©Author(s) (or their employer(s)) 2026.
World J Gastroenterol. Mar 7, 2026; 32(9): 115131
Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.115131
Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.115131
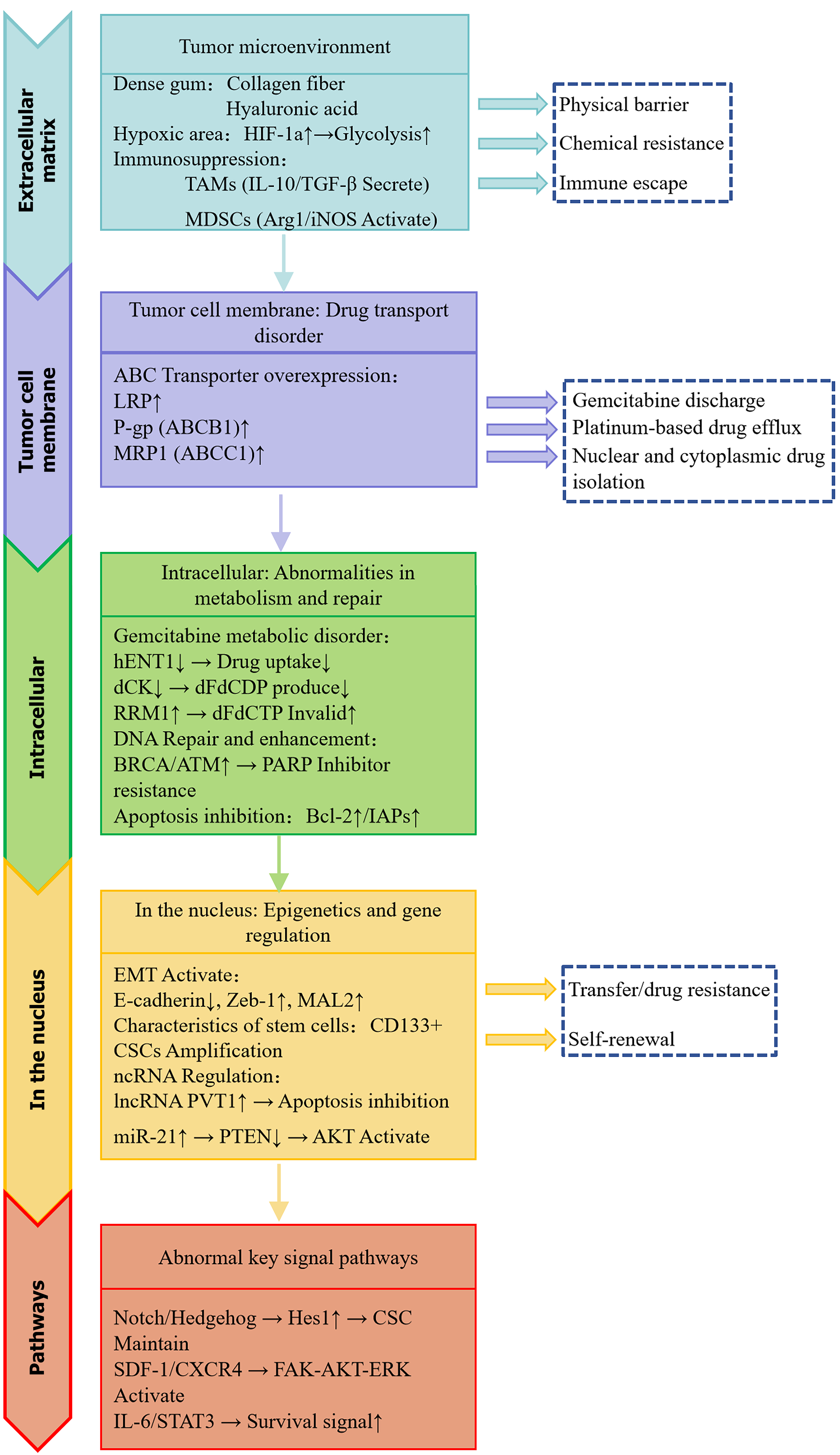
Figure 1 Flowchart of drug resistance mechanisms in pancreatic cancer.
This figure summarizes the primary mechanisms of drug resistance in pancreatic cancer. Resistance arises from extracellular matrix remodeling, overexpression of ATP-binding cassette transporters at the tumor cell membrane, intracellular defects in metabolism and DNA repair, nuclear epigenetic and gene regulatory changes, and activation of abnormal signaling pathways. These interconnected processes collectively reduce drug efficacy and facilitate tumor survival. HIF-1α: Hypoxia-inducible factor-1α; TAMs: Tumor-associated macrophages; IL: Interleukin; TGF: Transforming growth factor; MDSCs: Myeloid-derived suppressor cells; Arg1: Arginase 1; iNOS: Inducible nitric oxide synthase; ABC: ATP-binding cassette; LRP: Lung resistance-related protein; P-gp: P-glycoprotein; MRP1: Multidrug resistance-associated protein 1; hENT1: Human equilibrative nucleoside transporter 1; dCK: Deoxycytidine kinase; RRM1: Ribonucleotide reductase subunit 1; PARP: Polyadenosine-diphosphate-ribose polymerase; EMT: Epithelial-mesenchymal transition; MAL2: Myelin and lymphocyte protein 2; CSCs: Cancer stem cells; ncRNA: Non-coding RNA; lncRNA: Long non-coding RNAs; PVT1: Plasmacytoma variant translocation 1; PTEN: Phosphatase and tensin homolog; AKT: Protein kinase B; SDF-1: Stromal cell-derived factor 1; CXCR4: C-X-C receptor 4; FAK: Focal adhesion kinase; ERK: Extracellular signal-regulated kinase; STAT3: Signal transducer and activator of transcription 3.
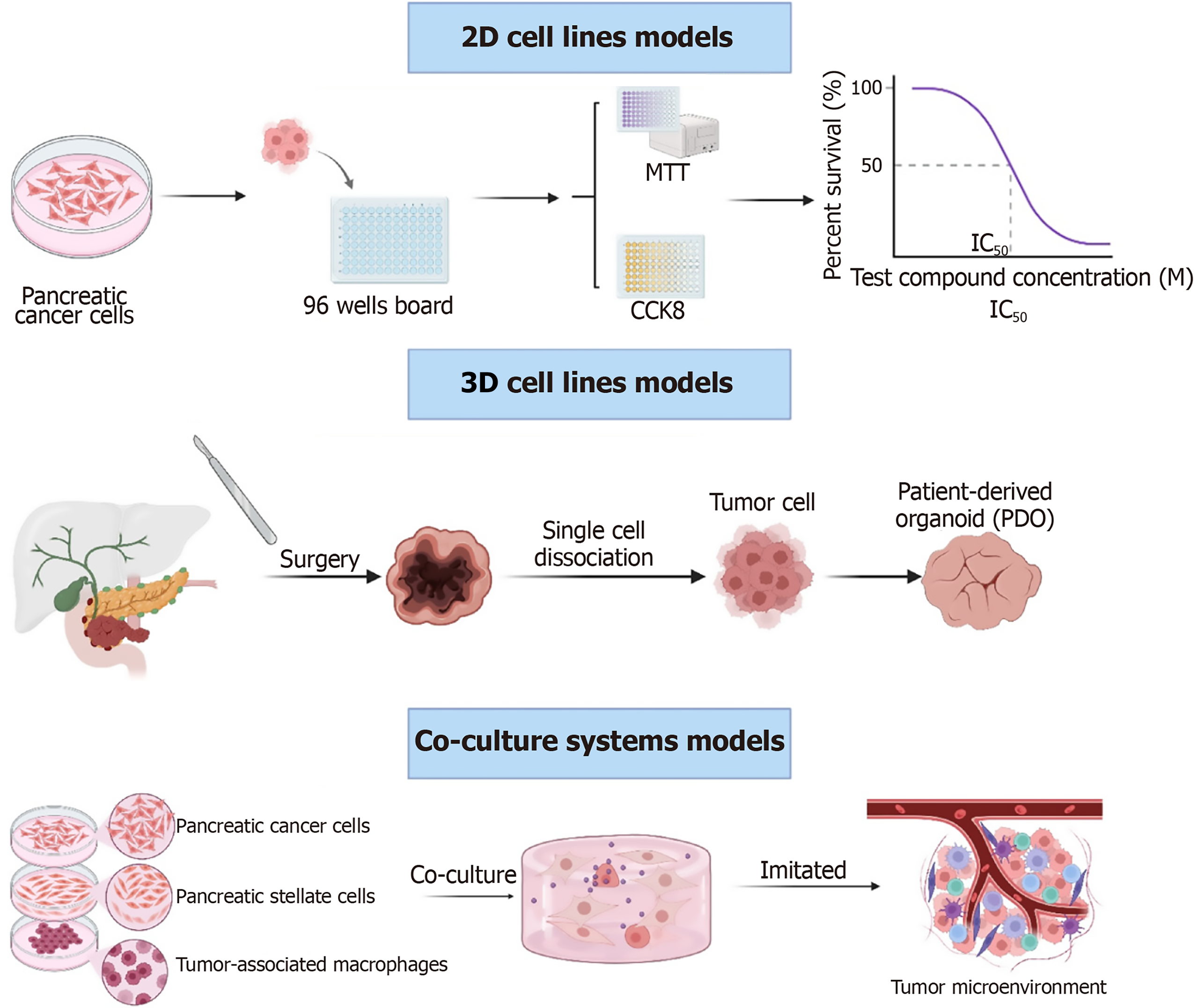
Figure 2 In vitro models for drug resistance research in pancreatic cancer.
Schematic overview of experimental models used in pancreatic cancer research. These include two-dimensional cell line models, three-dimensional models such as patient-derived organoids, and co-culture systems combining cancer cells with stromal or immune components. Each model provides distinct advantages for studying tumor biology, drug response, and tumor-microenvironment interactions.
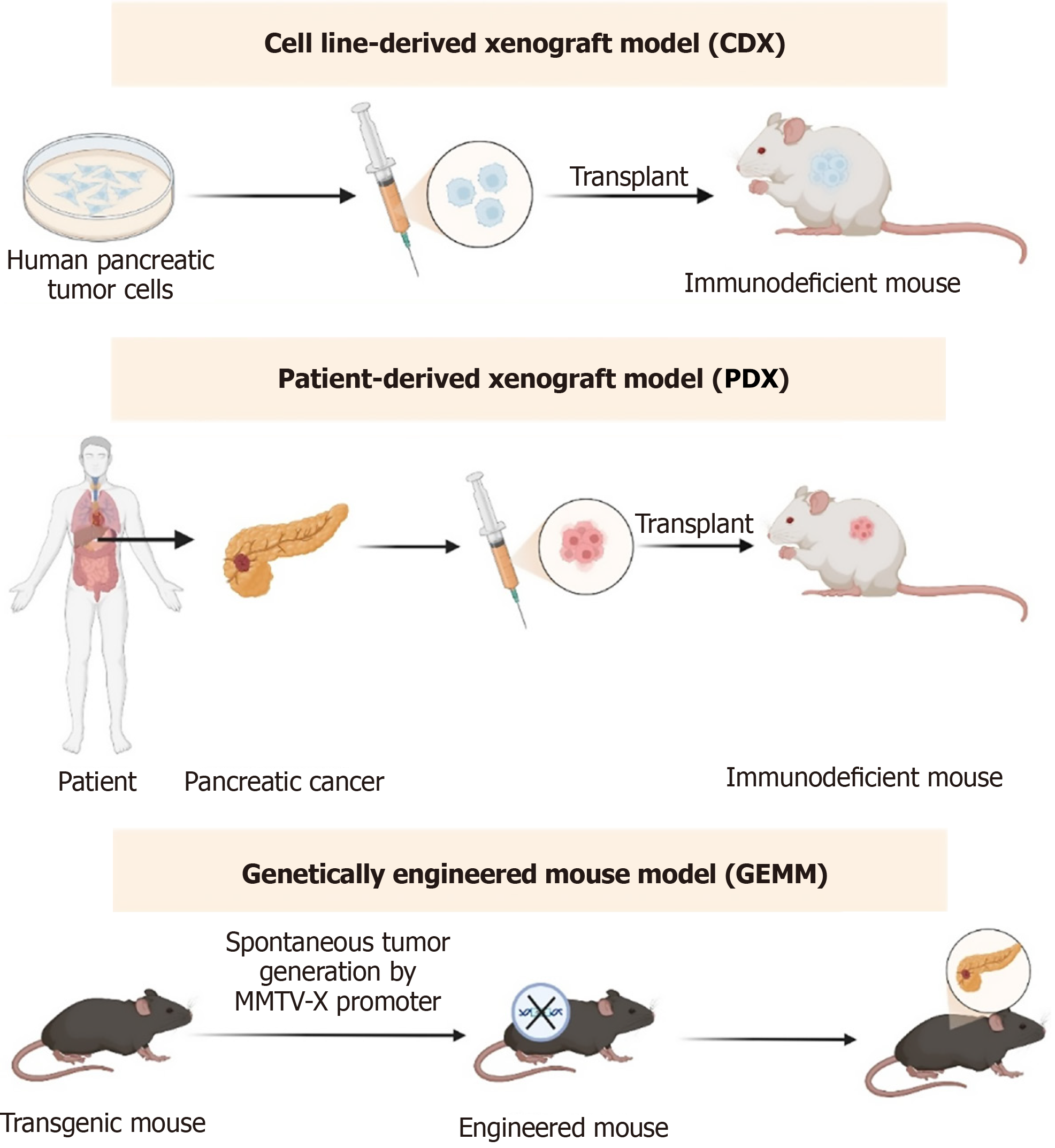
Figure 3 In vivo models for drug resistance research in pancreatic cancer.
These include cell line-derived xenografts, patient-derived xenografts, and genetically engineered mouse models. Each model offers distinct advantages for investigating tumor biology, therapeutic response, and mechanisms of drug resistance.
Figure 4 Summary of plant-derived chemical compounds.
Representative classes of natural compounds with anticancer potential, including polyphenols (e.g., resveratrol, curcumin), flavonoids (e.g., quercetin, apigenin), terpenoids (e.g., paclitaxel, artemisinin), alkaloids (e.g., berberine, camptothecin), organosulfur compounds (e.g., allicin, sulforaphane), and polysaccharides (e.g., lentinan). These bioactive molecules, originating from plants, fungi, and other natural sources, have been extensively investigated for their potential roles in tumor suppression and the modulation of drug resistance.
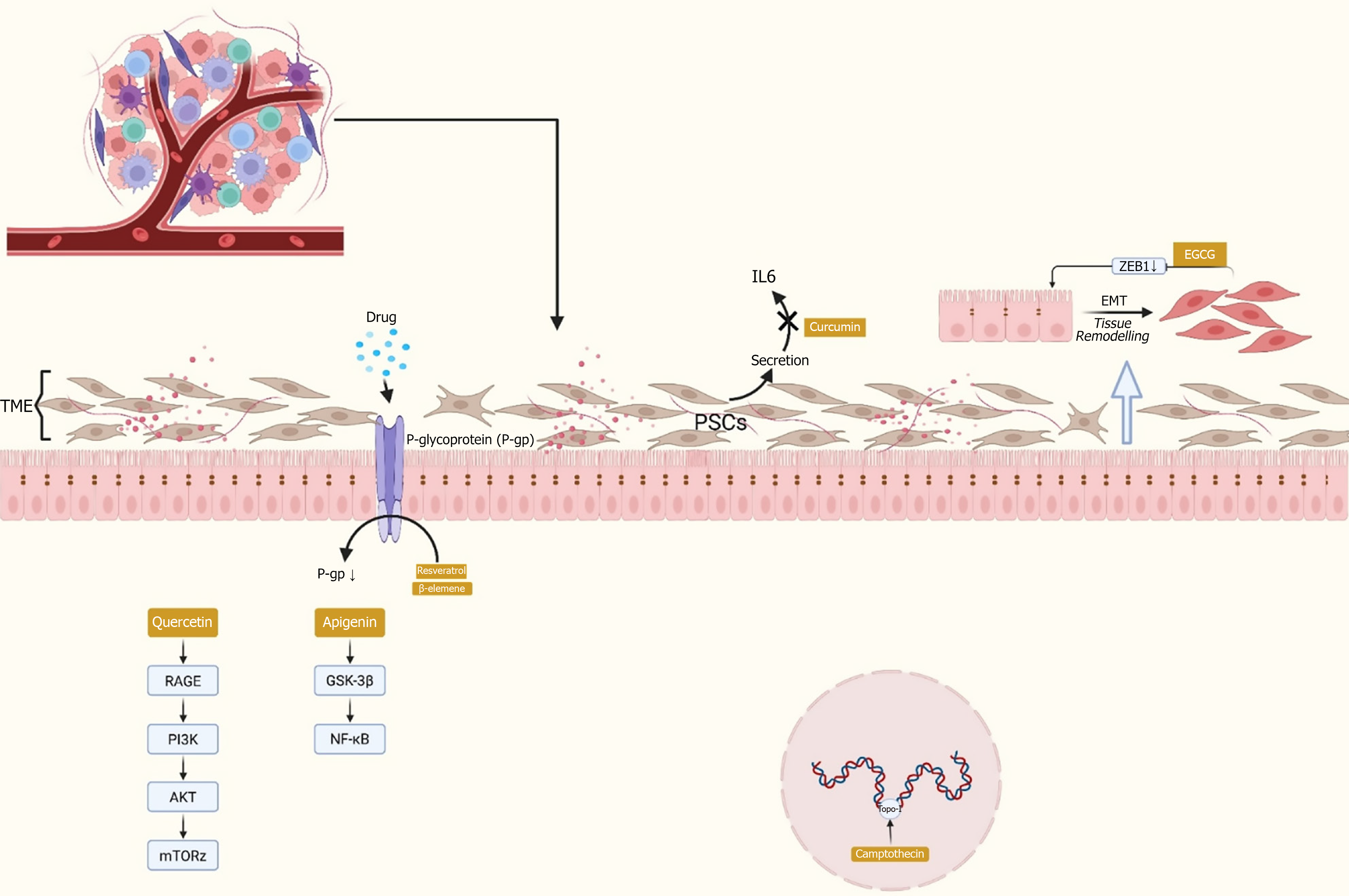
Figure 5 Plant-derived compounds influence drug resistance in pancreatic cancer through multiple mechanisms.
They regulate efflux transporters (e.g., P-glycoprotein), inhibit epithelial-mesenchymal transition, and modulate tumor-stroma interactions mediated by pancreatic stellate cells. The key signaling pathways include phosphatidylinositol 3-kinase/protein kinase B/mechanistic target of rapamycin, nuclear factor kappa B, Janus kinase/signal transducer and activator of transcription, and receptor for advanced glycation end-product pathways. Natural products such as quercetin and apigenin have been shown to target these pathways, thereby sensitizing tumor cells to chemotherapy. TME: Tumor microenvironment; IL: Interleukin; PSCs: Pancreatic stellate cells; ZEB1: Zinc-finger E-box-binding homeobox 1; EMT: Epithelial-mesenchymal transition; EGCG: Epigallocatechin gallate; P-gp: P-glycoprotein; RAGE: Receptor for advanced glycation end-product; PI3K: Phosphatidylinositol 3-kinase; AKT: Protein kinase B; mTOR: Mechanistic target of rapamycin; GSK-3β: Glycogen synthase kinase 3 beta; NF-кB: Nuclear factor kappa B.
- Citation: Yu JQ, Yu SJ, He YH, Xue YZ, Yu XF, Chen W, Hu LY, Fan XF, Gao ZF, Zhou HK, Liu XR, He XS, Wang XG. Research progress of plant-derived chemical compounds for overcoming pancreatic cancer drug resistance. World J Gastroenterol 2026; 32(9): 115131
- URL: https://www.wjgnet.com/1007-9327/full/v32/i9/115131.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i9.115131