©Author(s) (or their employer(s)) 2026.
World J Gastroenterol. Mar 7, 2026; 32(9): 114580
Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.114580
Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.114580
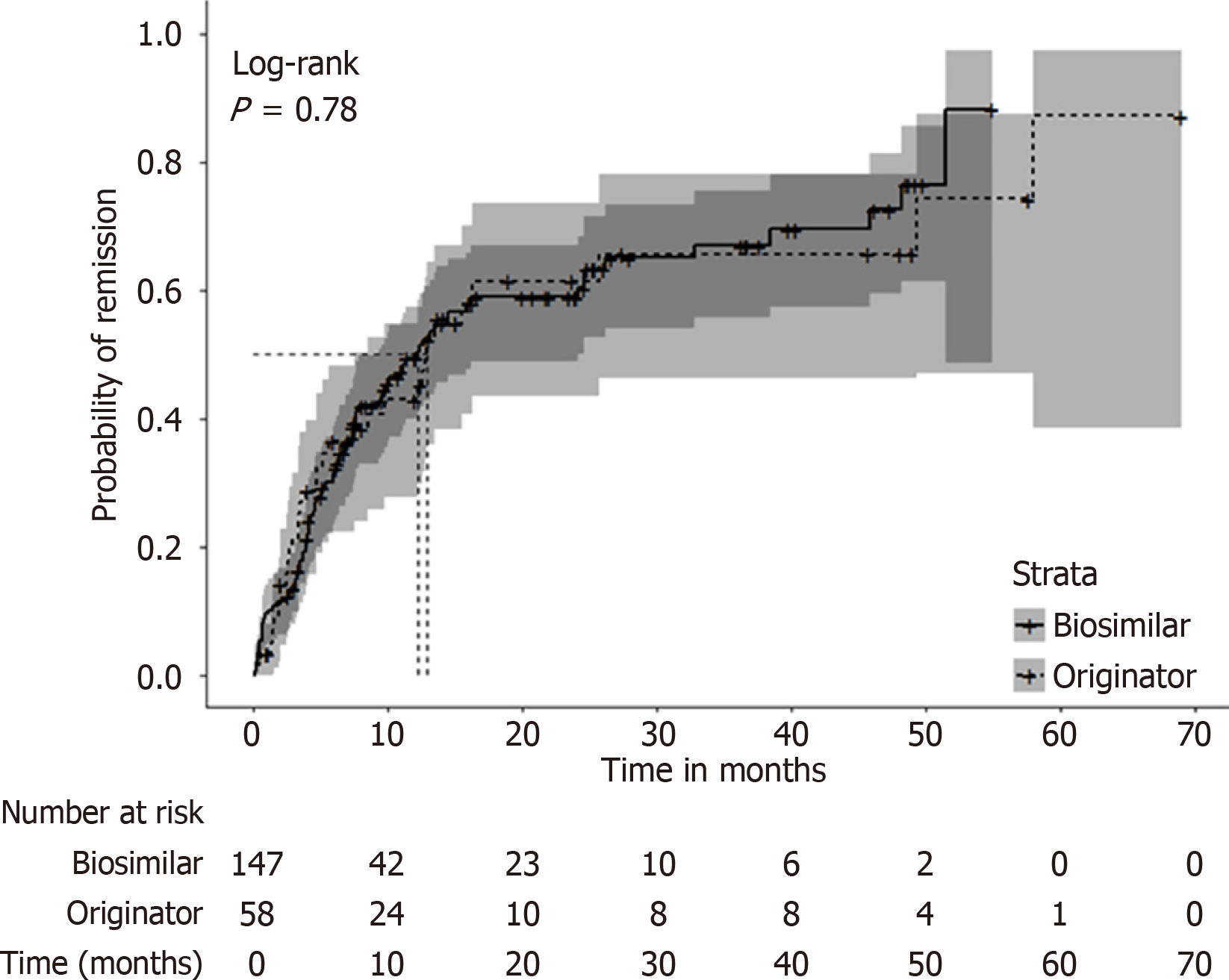
Figure 1 Kaplan-Meier curve for the time to first remission (among those without remission at baseline), stratified by originator biologic and biosimilar, Canadian IBD Research Consortium cohort, Canada, 2018-2024.
The median time to clinical remission was 12.8 months [95% con
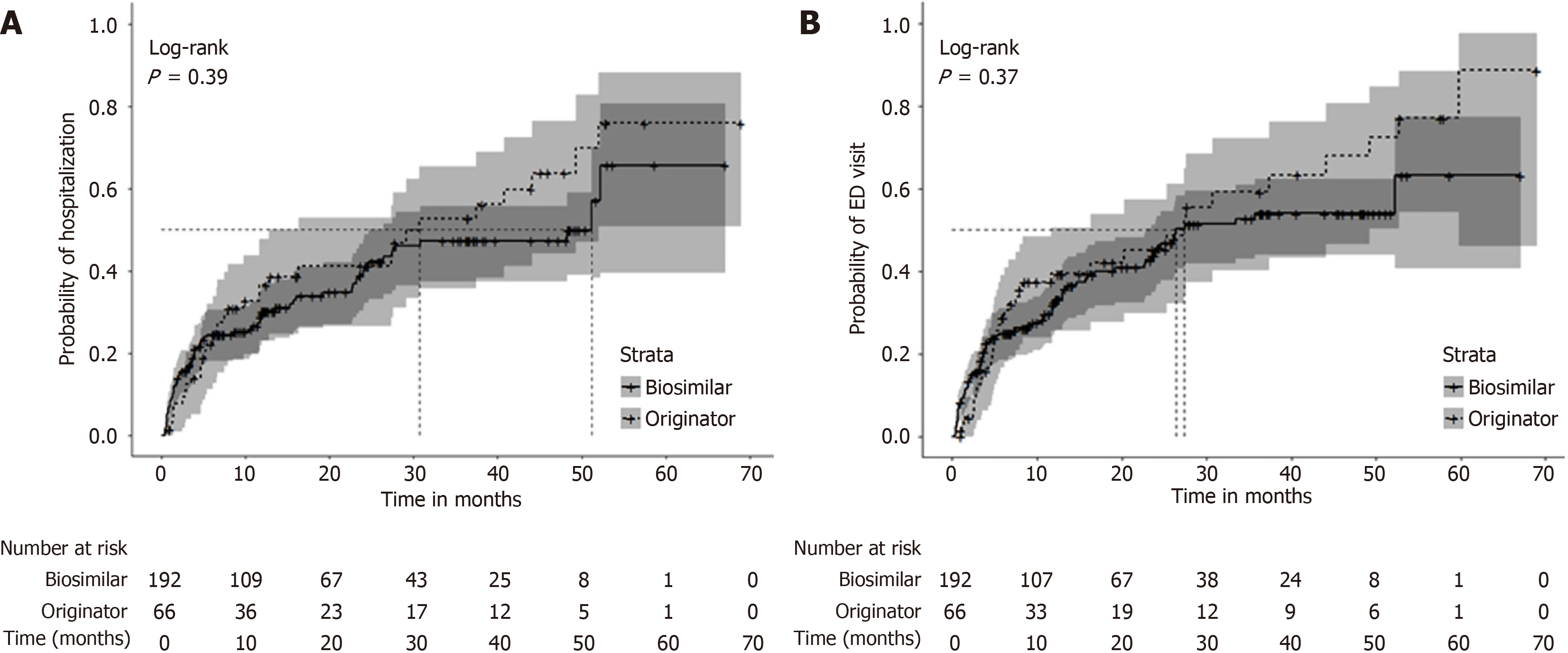
Figure 2 Kaplan-Meier curve for the time to: First hospitalization and first emergency department visit, stratified by originator biologic and biosimilar, Canadian IBD Research Consortium cohort, Canada, 2018-2024.
A: First hospitalization; B: First emergency department (ED) visit. The median time to first hospitalization estimated by Kaplan-Meier analysis was 30.7 months [95% confidence interval (95%CI): 12.8-52.1 months] for originator users and 51.2 months (95%CI: 24.6-72.3 months) for biosimilar users with no statistically significant difference between groups (A). The median time to first ED visit was 27.4 months (95%CI: 11.6-49.3 months) for originator users and 26.4 months (95%CI: 22.6-53.4 months) for biosimilar users (B). ED: Emergency department.
- Citation: Moura CS, Etingin A, Lukusa L, Singh H, Narula N, Targownik LE, Leung Y, Zezos P, Polewiczowska-Nowak B, Afif W, Bernatsky S. Comparable remission and health care use in real-world inflammatory bowel disease patients initiating originator biologics vs biosimilars. World J Gastroenterol 2026; 32(9): 114580
- URL: https://www.wjgnet.com/1007-9327/full/v32/i9/114580.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i9.114580