©Author(s) (or their employer(s)) 2026.
World J Gastroenterol. Mar 7, 2026; 32(9): 114200
Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.114200
Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.114200
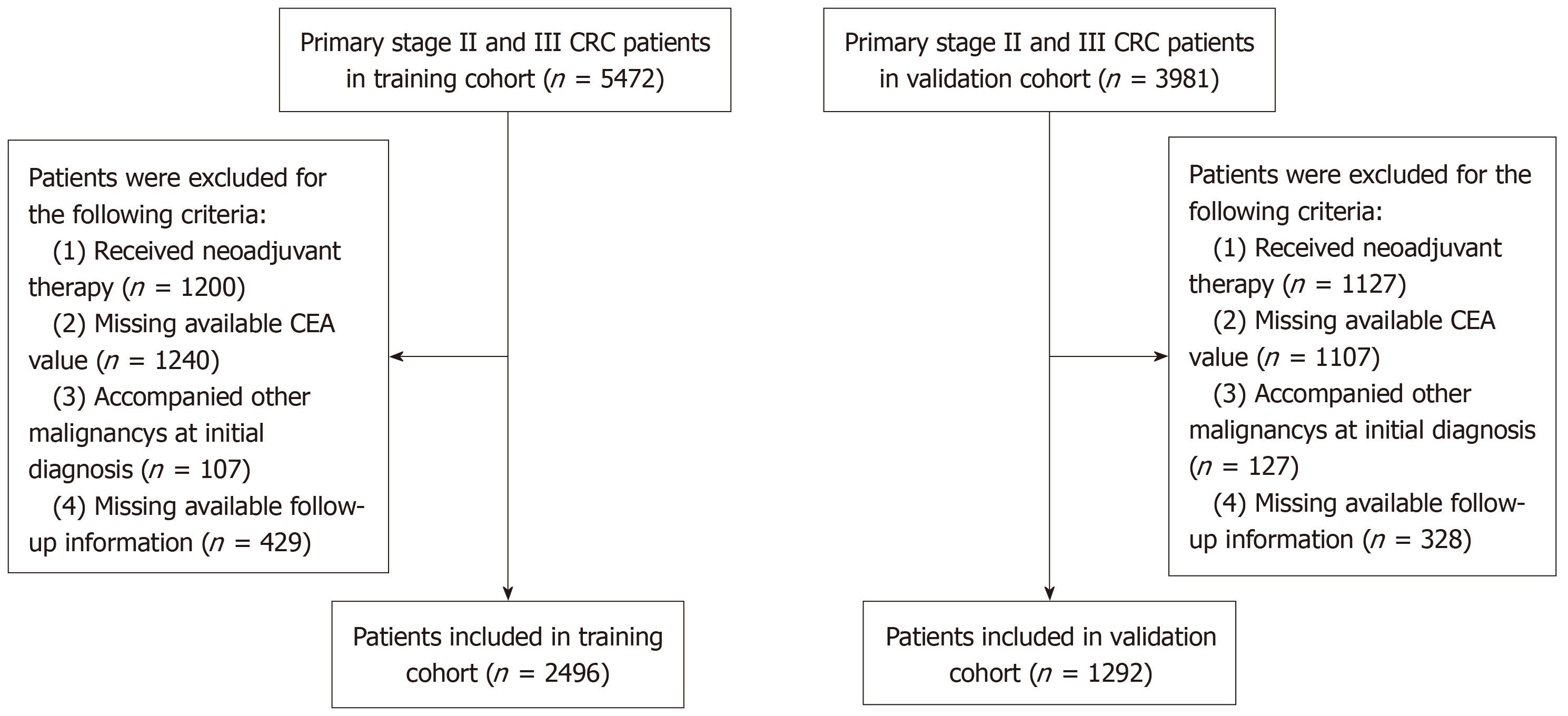
Figure 1 Flowchart of patient selection.
CRC: Colorectal cancer; CEA: Carcinoembryonic antigen.
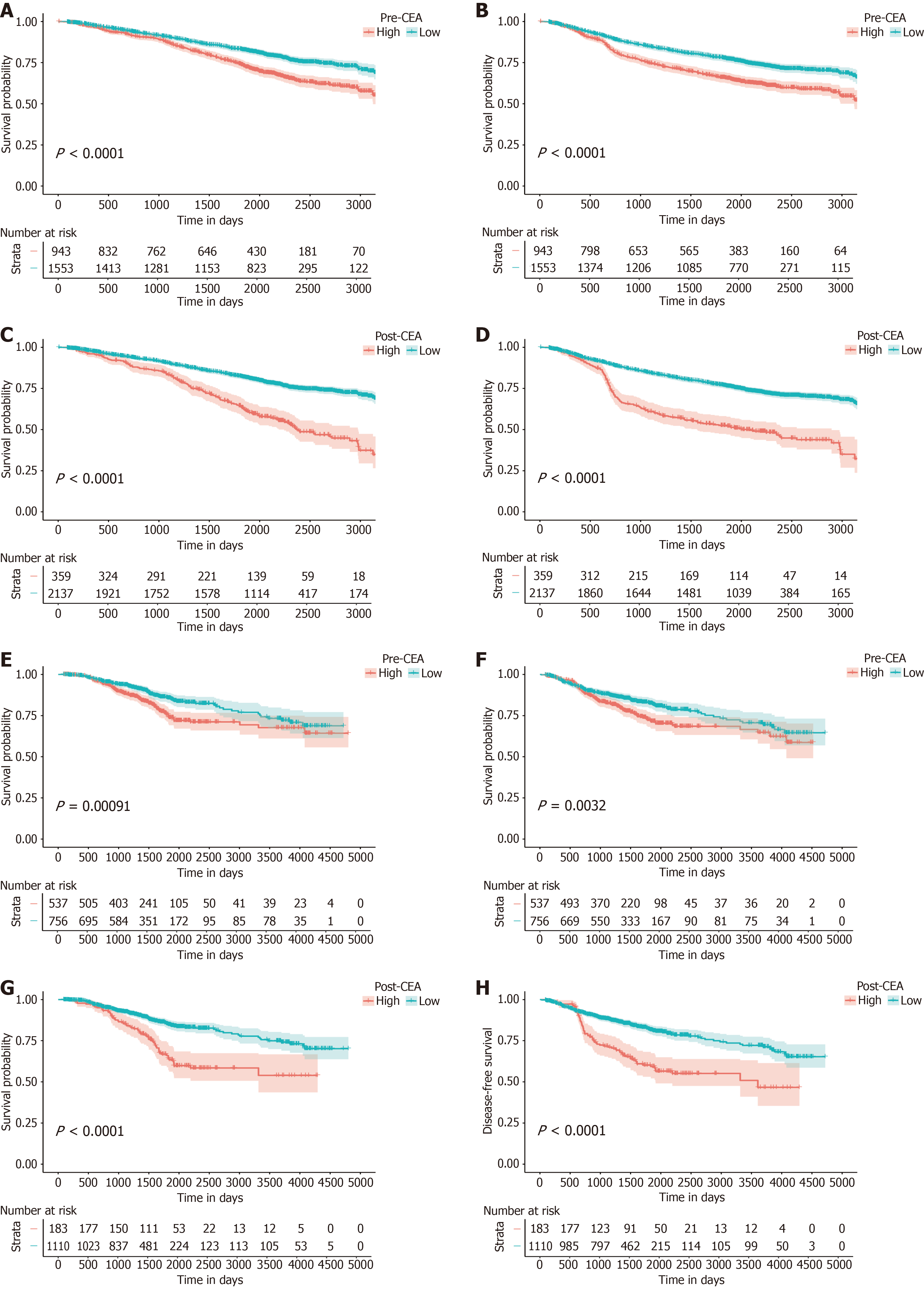
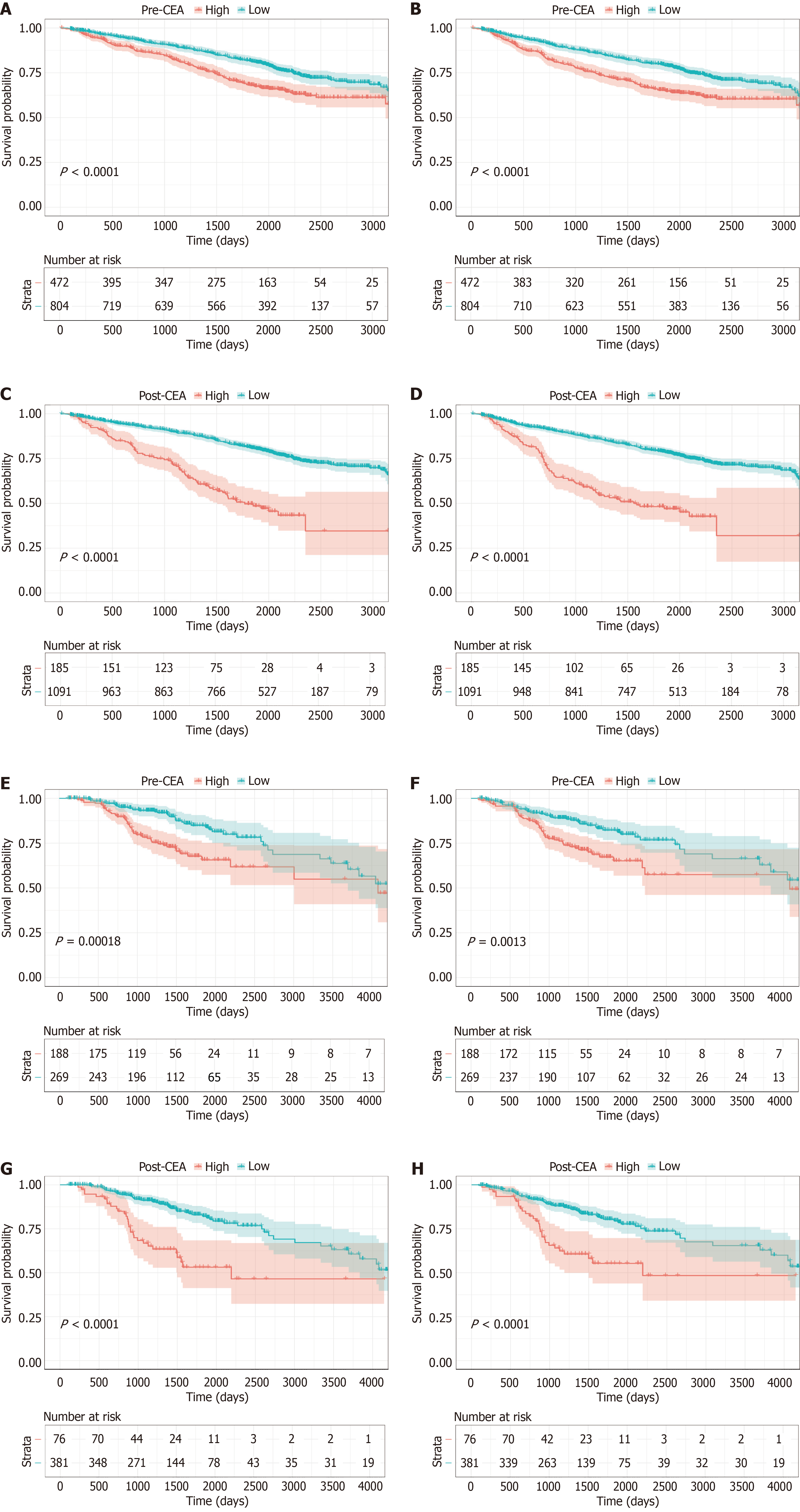
Figure 2 Kaplan-Meier survival analysis according to perioperative carcinoembryonic antigen.
A-D: Kaplan-Meier curves for overall survival (OS) and disease-free survival (DFS) for patients with different pre-carcinoembryonic antigen (CEA) (A and B) and post-CEA (C and D) in the training cohort; E-H: Kaplan-Meier curves of OS and DFS for patients with different pre-CEA (E and F) and post-CEA (G and H) in the validation cohort. pre-CEA: Preoperative carcinoembryonic antigen; post-CEA: Postoperative carcinoembryonic antigen.
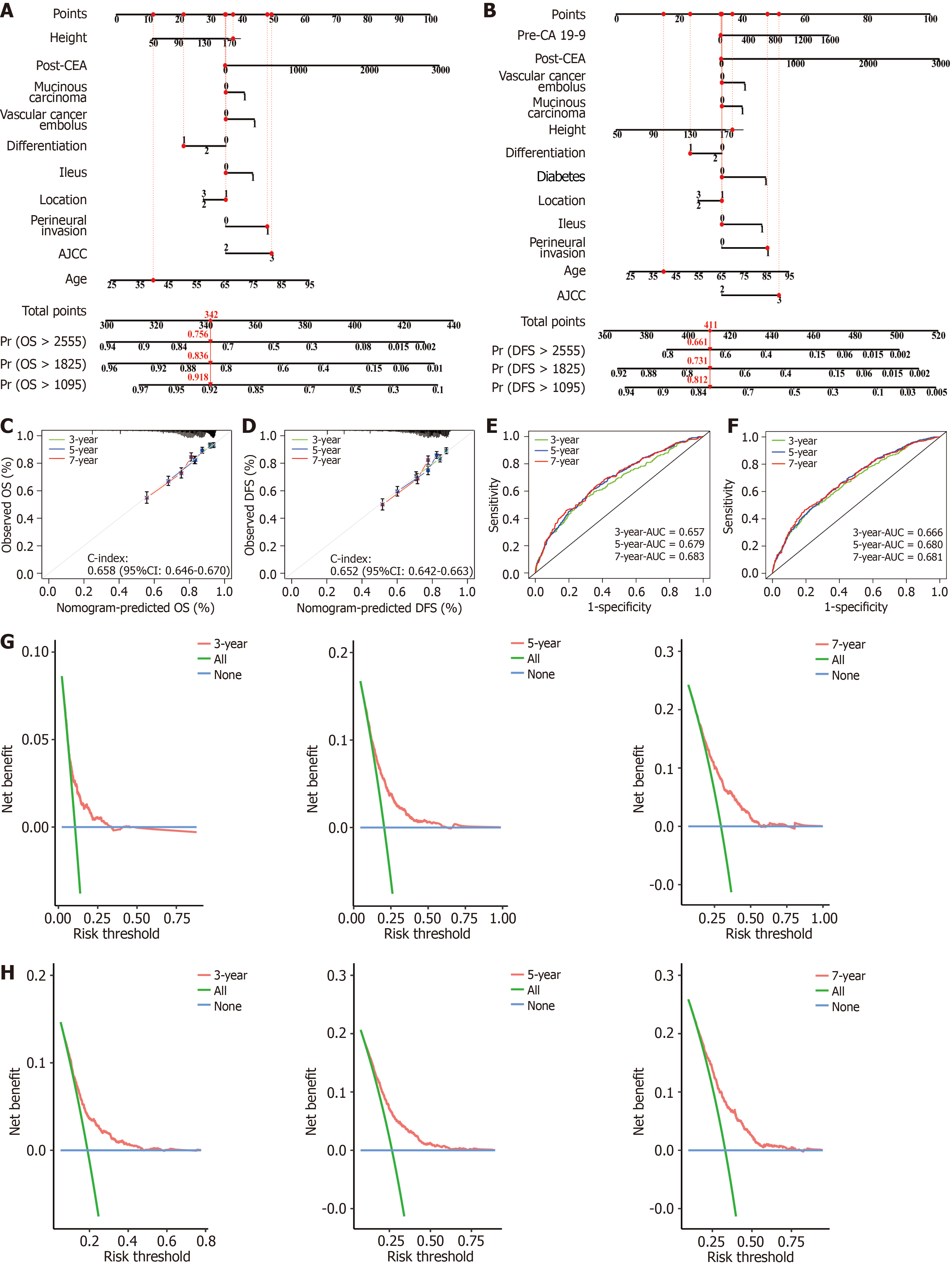
Figure 3 Nomograms for prognosis in the training cohort.
A and B: Nomograms of overall survival (OS) and disease-free survival (DFS) based on the training cohort; C: Calibration curves for the nomogram of OS at 3, 5, 7 years in the training cohort; D: Calibration curves for the nomogram of DFS at 3, 5, 7 years in the training cohort; E: Receiver operating characteristic (ROC) curves for the nomogram of OS at 3, 5, 7 years in the training cohort; F: ROC curves for the nomogram of DFS at 3, 5, 7 years in the training cohort; G and H: Decision curve analyses for the nomograms of OS (G) and DFS (H) at 3, 5, 7 years in the training cohort. OS: Overall survival; DFS: Disease-free survival; AUC: Area under the curve; post-CEA: Postoperative carcinoembryonic antigen; AJCC: American Joint Committee on Cancer; CI: Confidence interval.
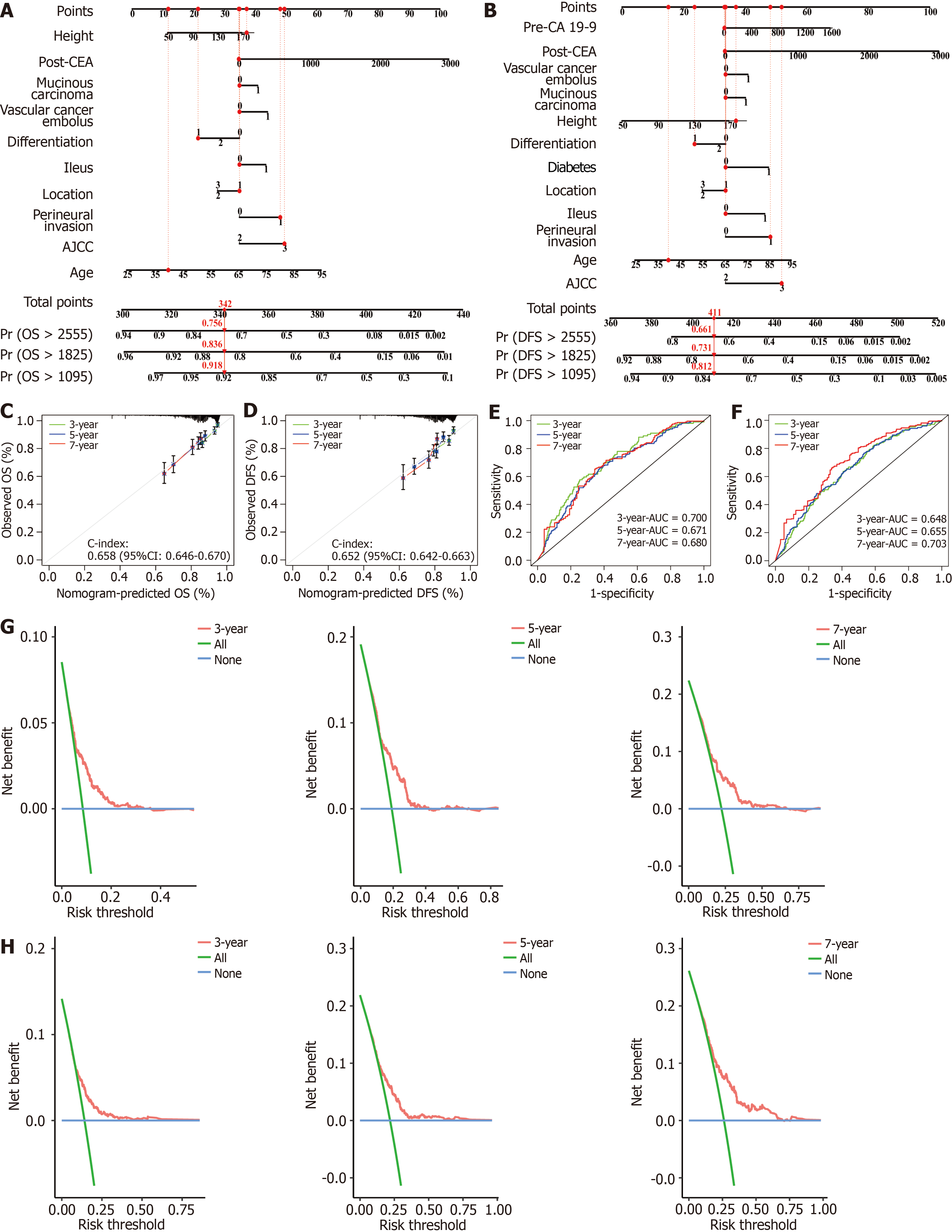
Figure 4 The performance of the nomograms in the validation cohort.
A and B: Nomograms of overall survival (OS) and disease-free survival (DFS) based on the training cohort; C: Calibration curves for the nomogram of OS at 3, 5, 7 years in the validation cohort; D: Calibration curves for the nomogram of DFS at 3, 5, 7 years in the validation cohort; E: Receiver operating characteristic (ROC) curves for the nomogram of OS at 3, 5, 7 years in the validation cohort; F: ROC curves for the nomogram of DFS at 3, 5, 7 years in the validation cohort; G and H: Decision curve analyses (DCA) for the nomograms of OS (G) and DFS (H) at 3, 5, 7 years in the validation cohort. OS: Overall survival; DFS: Disease-free survival; AUC: Area under the curve; post-CEA: Postoperative carcinoembryonic antigen; AJCC: American Joint Committee on Cancer; CI: Confidence interval.
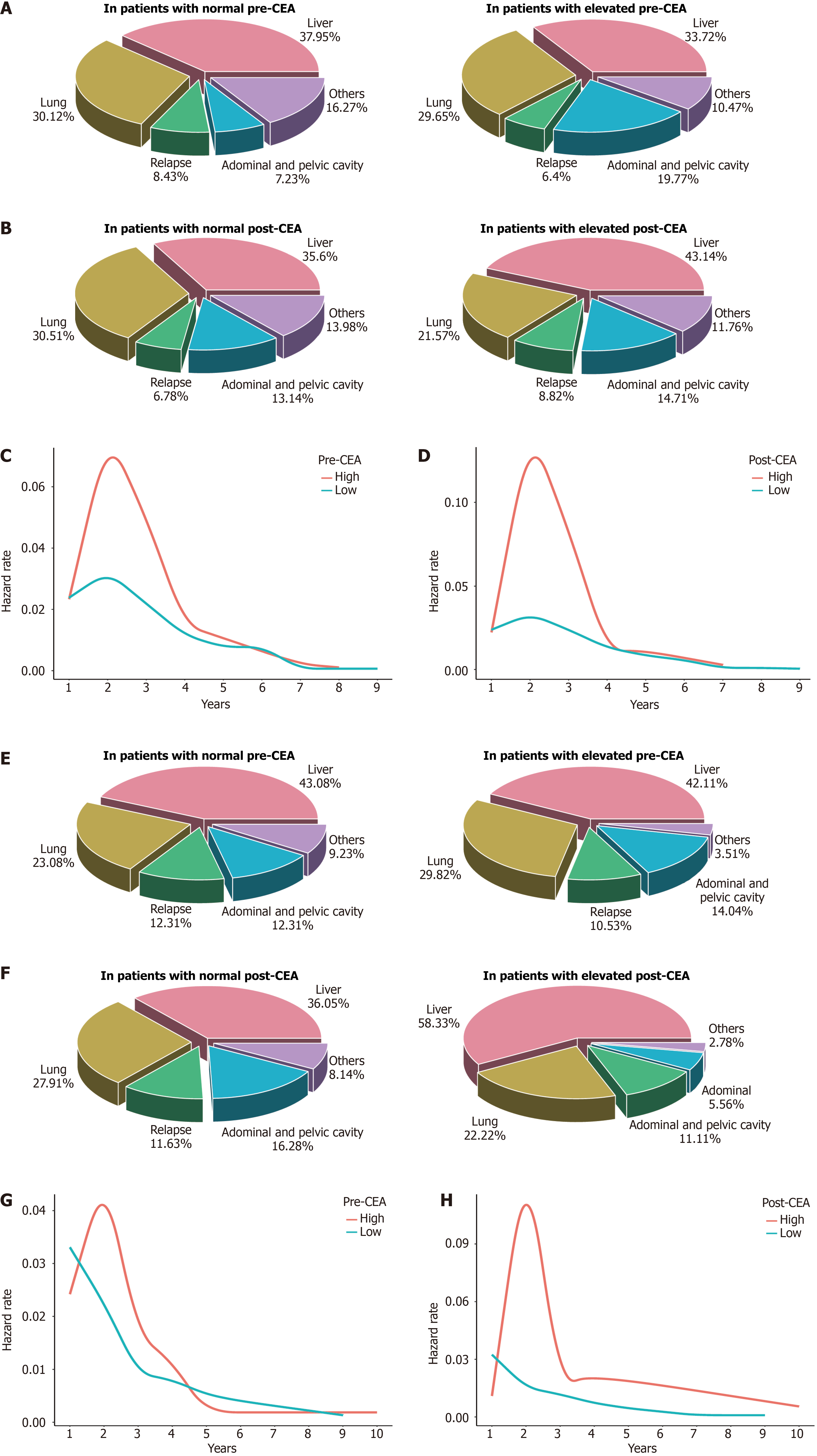
Figure 5 Recurrence and metastasis patterns based on perioperative carcinoembryonic antigen.
A: Relative frequencies of recurrence or metastasis sites for patients in the training cohort based on preoperative carcinoembryonic antigen (pre-CEA); B: Relative frequencies of recurrence or metastasis sites for patients in the training cohort based on postoperative carcinoembryonic antigen (post-CEA); C: Annual recurrence or metastasis hazard curves for patients in the training cohort based on pre-CEA; D: Annual recurrence or metastasis hazard curves for patients in the training cohort based on post-CEA; E: Relative frequencies of recurrence or metastasis sites for patients in the validation cohort based on pre-CEA; F: Relative frequencies of recurrence or metastasis sites for patients in the validation cohort based on post-CEA; G: Annual recurrence or metastasis hazard rates for patients in the validation cohort based on pre-CEA; H: Annual recurrence or metastasis hazard rates for patients in the validation cohort based on post-CEA. pre-CEA: Preoperative carcinoembryonic antigen; post-CEA: Postoperative carcinoembryonic antigen.
Figure 6 Potential of perioperative carcinoembryonic antigen in guiding treatment decision making.
A-D: Kaplan-Meier curves of overall survival (OS) and disease-free survival (DFS) for patients without adjuvant chemotherapy in the training cohort, grouping based on pre-CEA (A and B), grouping based on post-CEA (C and D); E-H: Kaplan-Meier curves of OS and DFS for patients without adjuvant chemotherapy in the validation cohort, grouping based on pre-CEA (E and F),grouping based on post-CEA (G and H).
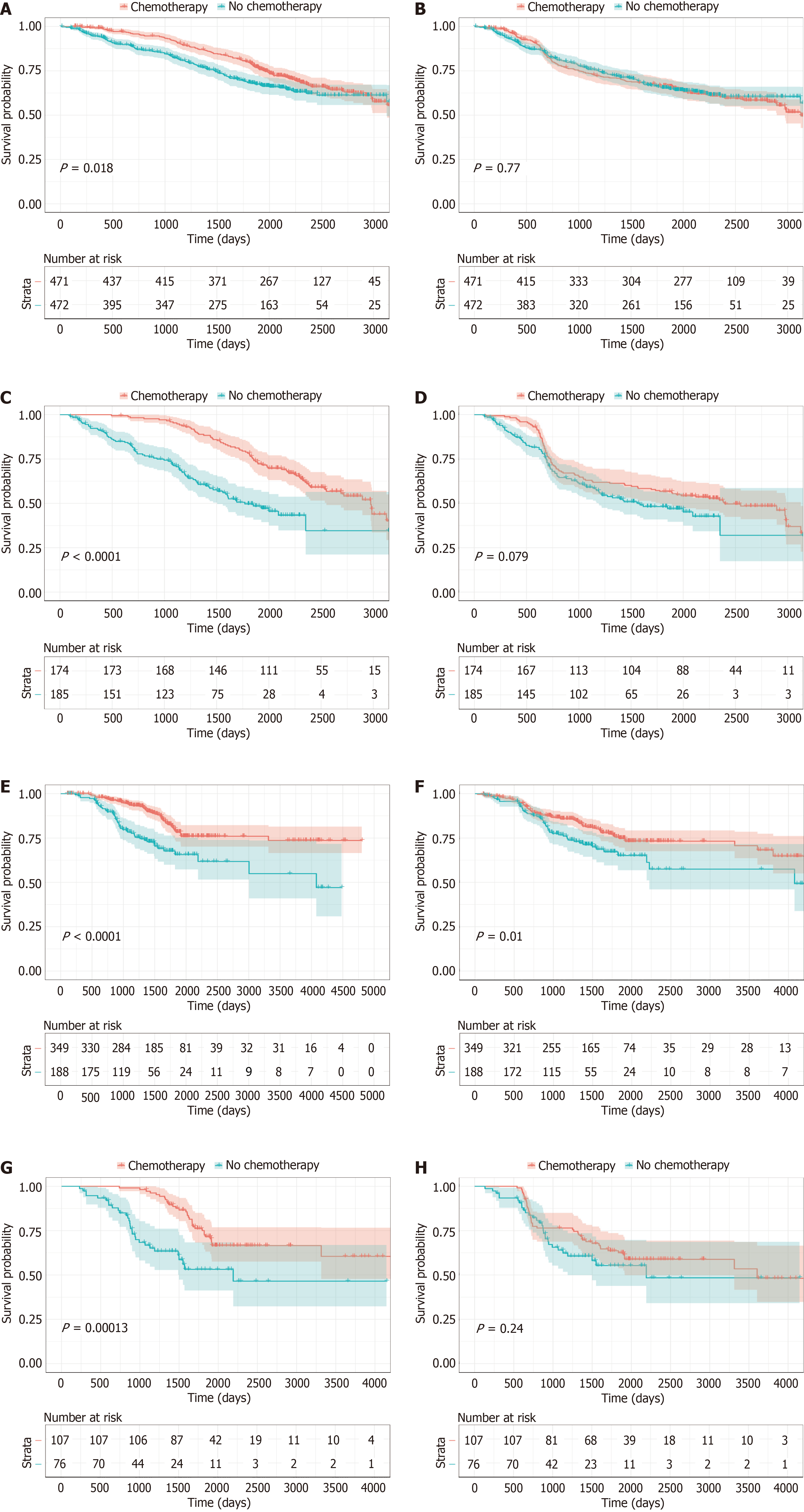
Figure 7 Impact of adjuvant chemotherapy on survival in patients with elevated perioperative carcinoembryonic antigen.
A and B: Kaplan-Meier curves of overall survival (OS) and disease-free survival (DFS) for patients with elevated pre-CEA in the training cohort who did or did not receive adjuvant chemotherapy; C and D: Kaplan-Meier curves of OS and DFS for patients with elevated post-CEA in the training cohort who did or did not receive adjuvant chemotherapy; E and F: Kaplan-Meier curves of OS and DFS for patients with elevated pre-CEA in the validation cohort who did or did not receive adjuvant chemotherapy; G: And H: Kaplan-Meier curves of OS and DFS for patients with elevated post-CEA in the validation cohort who did or did not receive adjuvant chemotherapy.
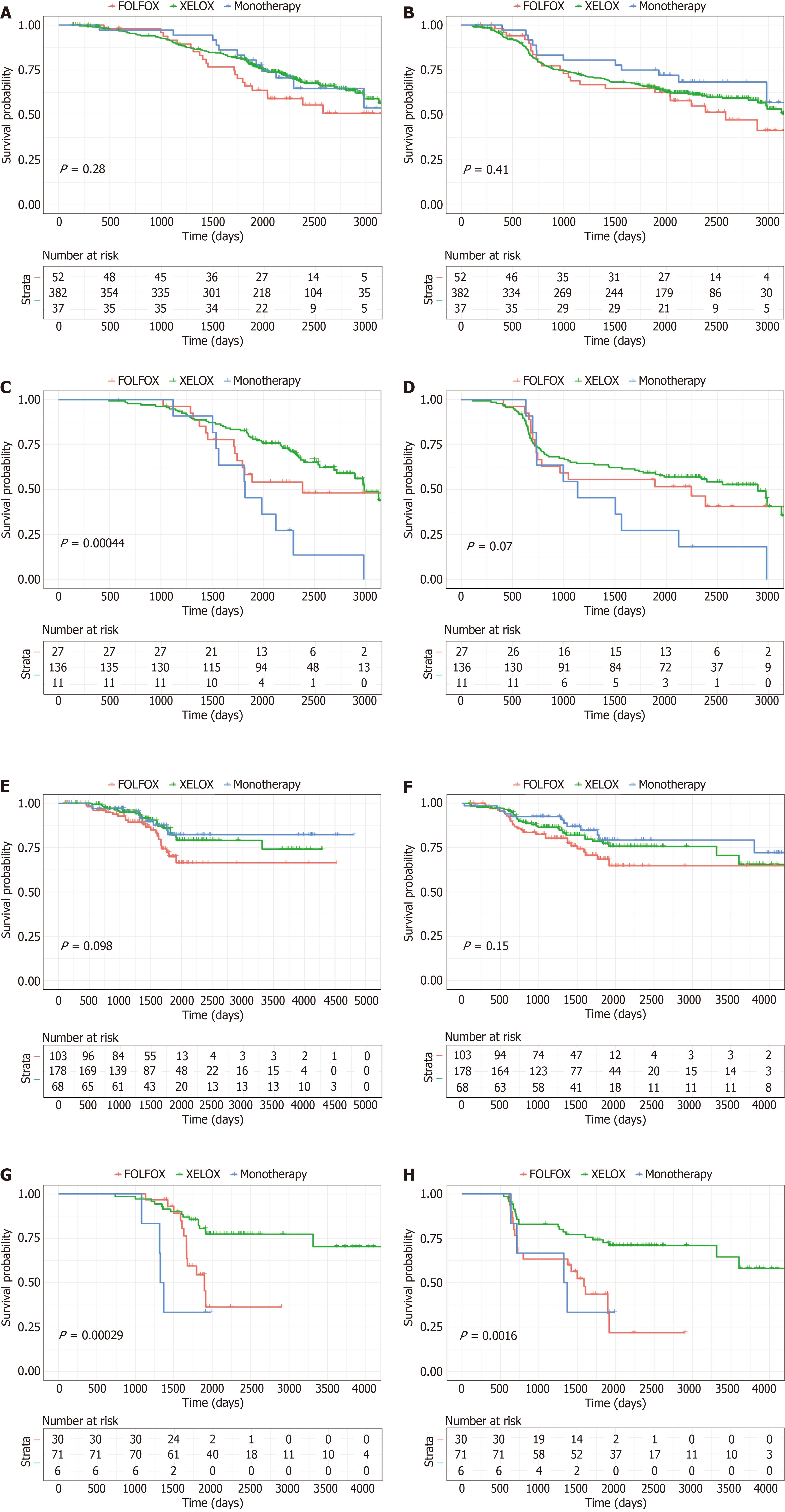
Figure 8 Potential of perioperative carcinoembryonic antigen in guiding the selection of adjuvant chemotherapy strategies.
A and B: Kaplan-Meier curves of overall survival (OS) and disease-free survival (DFS) for patients with elevated pre-CEA in the training cohort after receiving different adjuvant chemotherapy strategies; C and D: Kaplan-Meier curves of OS and DFS for patients with elevated post-CEA in the training cohort after receiving different adjuvant chemotherapy strategies; E and F: Kaplan-Meier curves of OS and DFS for patients with elevated pre-CEA in the validation cohort after receiving different adjuvant chemotherapy strategies; G and H: Kaplan-Meier curves of OS and DFS for patients with elevated post-CEA in the validation cohort after receiving different adjuvant chemotherapy strategies.
- Citation: Du FQ, Liu JL, Mai LD, Han XH, Song WJ, Yang D, Zhang QJ, Zhang R, Liu YL, Tong JX. Perioperative serum carcinoembryonic antigen: Powerful marker for prognostic prediction and adjuvant chemotherapy decision-making in patients with stage II and III colorectal cancer. World J Gastroenterol 2026; 32(9): 114200
- URL: https://www.wjgnet.com/1007-9327/full/v32/i9/114200.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i9.114200