Published online Mar 7, 2026. doi: 10.3748/wjg.v32.i9.111199
Revised: September 29, 2025
Accepted: January 19, 2026
Published online: March 7, 2026
Processing time: 247 Days and 17.2 Hours
Liver transplantation is the only treatment for acute and chronic liver failure, but the global organ shortage has increased reliance on extended criteria donor livers, which are more susceptible to ischemia-reperfusion injury. While static cold sto
To summarize the current state of small animal liver machine perfusion (MP), highlight variability in protocols, and emphasize the need for standardization to guide future research.
A comprehensive literature search of PubMed was conducted to identify studies on small animal (rat and mouse) ex vivo liver MP. Only English-language animal studies were included, with no restrictions on publication date. Relevant full-text articles were reviewed, and reference lists were screened to ensure completeness.
Small animal liver MP provides a cost-effective model to explore dynamic preservation strategies. Rat perfusion studies face challenges including dual-vessel perfusion, maintaining physiological perfusate volumes, and lack of standardized protocols. Open- and closed-circuit setups have distinct advantages and limitations, and experimental designs vary widely across studies.
This review illustrates the wide variability in small animal liver MP protocols and underscores the urgent need for standardization. Addressing these inconsistencies will enhance reproducibility, facilitate comparison across stu
Core Tip: Small animal liver machine perfusion models are essential for studying dynamic liver preservation in transplantation research. Rat liver perfusion provides a cost-effective and accessible platform, but currently no standardized protocols exist, limiting reproducibility and progress. This review highlights variations in existing studies, technical challenges, and limitations, emphasizing the urgent need for standardization. By summarizing key developments and system differences, it offers researchers practical insights to optimize perfusion strategies, improve reproducibility, and reduce animal use in future studies.
- Citation: Bickaite-Bausiene K, Kvietkauskas M, Leber B, Bausys B, Brislinger D, Strupas K, Stiegler P. Small animal ex vivo machine perfusion of the liver: A comprehensive literature review. World J Gastroenterol 2026; 32(9): 111199
- URL: https://www.wjgnet.com/1007-9327/full/v32/i9/111199.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i9.111199
Liver transplantation (LTx) is the only possible treatment option for acute and chronic liver failure. Recent advances in LTx outcomes, immunosuppressive techniques, and cancer therapies have expanded the eligibility of individuals for inclusion in transplant waiting lists[1]. The global organ shortage obligates centers to use grafts from extended criteria donors (ECDs) including grafts donated after circulatory death (DCD)[2]. ECD livers are susceptible to greater ischemia reperfusion injury, contributing to inferior graft function and outcomes[3]. While static cold storage (SCS) has long been considered the gold standard, ECD do not fare well under this traditional preservation method. Dynamic preservation methods such as normothermic (NMP), subnormothermic (SNMP), and hypothermic machine perfusion (HMP) have emerged to enhance the viability of donor livers and optimize transplantation outcomes[4,5].
In contrast to conventional SCS, ex situ machine perfusion (MP) enhances LTx outcomes, enabling extended preservation times and viability testing[6,7]. The prospective application of MP is anticipated to be even more extensive, with currently investigated therapeutic interventions like defatting cocktails, RNA interference, senolytics, and stem cell therapy showing promise in facilitating the repair and regeneration of injured livers before LTx[8].
Large animal studies on MP are expensive and face feasibility challenges[9]. Therefore, small animal experiments are necessary to clarify potential applications in the future. Adherence to the 3R rule (replacement, reduction, refinement) is crucial for ethical animal experimentation[10]. While the demand for animal experiments in MP is significant, a universally accepted standard protocol for its implementation is currently lacking.
This literature review outlines the steps and diverse possibilities involved in establishing MP for small animal livers, aiming to assist in considering essential elements and ultimately contributing to a reduction in the number of animals used for experiments.
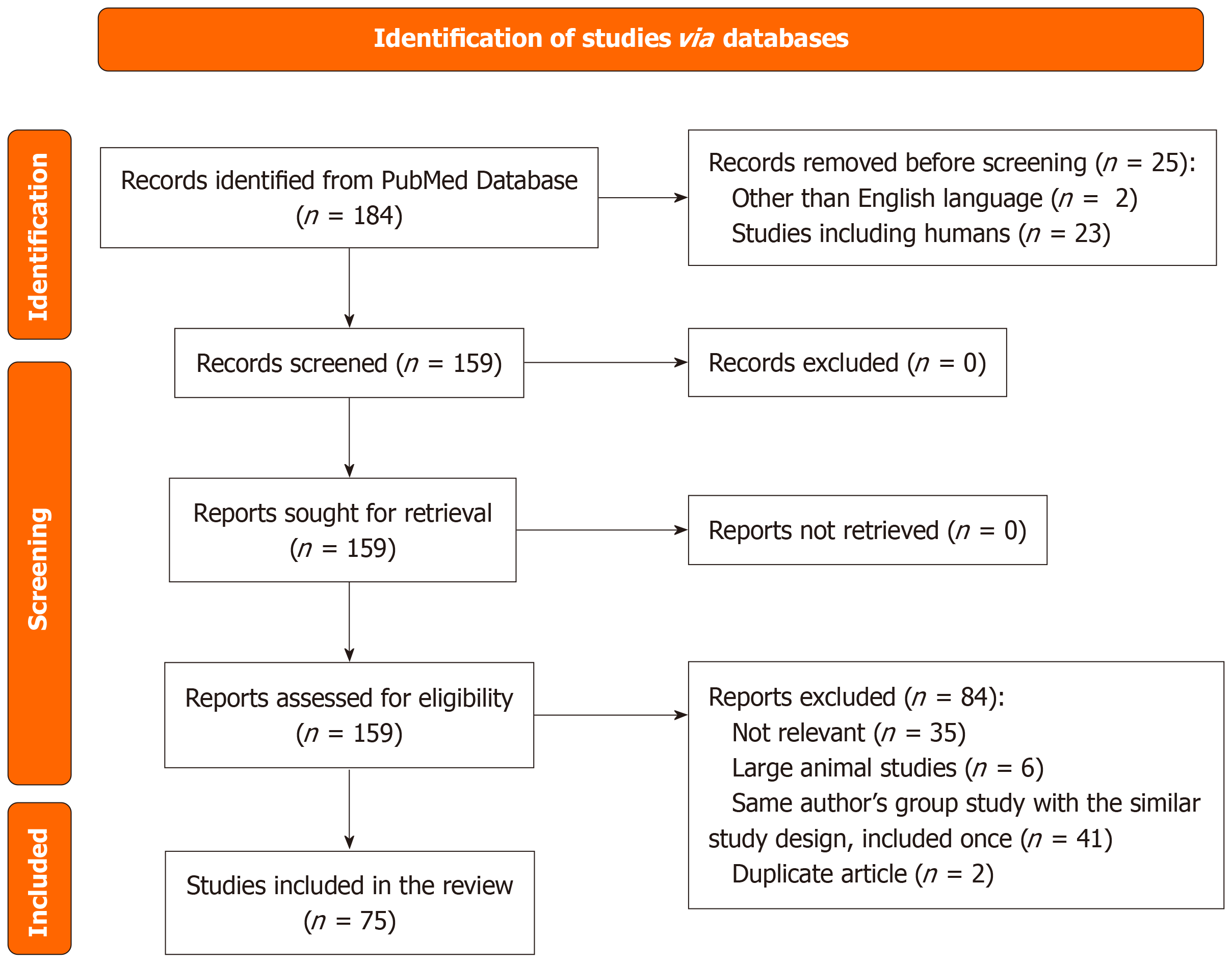
The comprehensive literature search was conducted using the PubMed database. The following combination of medical subject headings and keywords with the employment of “AND” or “OR” or “NOT” Boolean operators were used: (“HMP” OR “hope” OR “NMP” OR “SNMP” OR “machine perfusion”) AND “liver perfusion” AND (“rat” OR “mice” OR “small animal”) NOT “kidneys”. Only articles written about animals and not humans were included in the search. No restrictions on publication dates were applied, allowing for the inclusion of studies from all available years. Only abstracts written in the English language were reviewed. Full-text articles were retrieved if relevant abstracts were identified. An additional manual search of the reference lists was performed to ensure the comprehensive literature search procedure. The most recent search was performed on June 23rd, 2024. A preferred reporting items for systematic reviews and meta-analyses flow diagram of the literature search and study selection is presented in Figure 1.
Several different techniques are described in literature. Before connecting the liver to the ex vivo MP circuit, a standard cold flush in situ is performed to remove blood remnants and reduce warm ischemia (WI) time[11,12]. Clear descriptions of the flushing technique are crucial for achieving favorable outcomes in liver preservation[13]. Table 1 outlines various liver flushing solutions, routes, and volumes. The administration routes for the cold flush vary among authors. Most perform the flush through the portal vein (PV) only[14-66]. Others use a dual vessel approach, flushing through both the PV and the aorta (A)[67-74] or the PV and the hepatic artery (HA)[75-77]. Flushing via the A alone is another option[78-84]. The use of HA exclusively is a rare approach, employed by only a single author[85].
| Ref. | Via | Liver flush before MP |
| Kim et al[14] | PV | 10 mL of cold UW-G solution |
| Dutkowski et al[15] | PV | 2 mL + 3 mL + 1 mL of 4 °C UW solution (hydroxyethyl starch and glutathione free) |
| Compagnon et al[67] | A, PV | 25 mL of ice-cold Celsior-HES solution with 500 IU of heparin |
| Lauschke et al[16] | PV | 60 mL of HTK or Belzer MPS solutions |
| Lee et al[90] | NA | NA |
| Tan et al[91] | NA | NA |
| Xu et al[92] | NA | NA |
| Bessems et al[17] | PV | 50 mL of ringer lactate |
| Dutkowski et al[18] | PV | 2 mL + 3 mL + 1 mL of 4 °C UW solution (hydroxyethyl starch and glutathione free) |
| Tolboom et al[19] | PV | 5 mL + 5 mL of 4 °C UW solution |
| Vairetti et al[20] | PV | Oxygenated KH medium |
| Manekeller et al[21] | PV | HTK or Belzer MPS solutions |
| Stegemann et al[22] | PV | 20 mL of HTK or Custodiol-N solution at 4 °C |
| Ferrigno et al[23] | PV | Oxygenated KH medium |
| Lüer et al[24] | PV | 20 mL of HTK solution |
| Olschewski et al[25] | PV | 20 mL of Lifor organ preservation solution (at 4 °C, 12 °C and 21 °C) |
| Minor et al[26] | PV | 20 mL of HTK solution |
| Tolboom et al[27] | PV | 10 mL of saline |
| Giannone et al[28] | PV | 20 mL of cold Celsior solution (in situ) + 30 mL of cold Celsior solution |
| Perk et al[29] | PV | 10 mL of saline |
| Schlegel et al[30] | PV | 6 mL of 20 °C heparinized (1 IU/mL) saline |
| Bruinsma et al[88] | NA | Cold UW solution |
| Liu et al[31] | PV | 10 mL of 4 °C perfusate (control or defatting) |
| Carnevale et al[93] | NA | NA |
| Schlegel et al[32] | PV | 6 mL of 20 °C heparinized (1 IU/mL) saline |
| Schlegel et al[33] | PV | 6 mL of heparinized (1 IU/mL) saline at room temperature and UW solution at 4 °C |
| Bae et al[34] | PV | 200 mL of 0.9% saline |
| Niu et al[68] | A, PV | 50 mL of ice-cold ross perfusion fluid with 10 IU/mL heparin. 12 mL of cold supplemented UW solution. 20 mL of Hartmann’s solution with heparin (5 U/mL) |
| Tarantola et al[35] | PV | KH medium |
| Bruinsma et al[36] | PV | 10 mL of 21 °C 3-OMG loading solution + 10 mL of 21 °C 3-OMG loading solution |
| Ferrigno et al[37] | PV | 50 mL of modified KHB |
| Jia et al[38] | PV | Cooled saline containing 25 IU/mL heparin |
| Westerkamp et al[94] | NA | NA |
| Carbonell et al[95] | NA | NA |
| Op den Dries et al[75] | PV, HA | 5 mL of 37 °C 0.9% NaCl via PV + 5 mL of 4 °C 0.9% NaCl via SIVC + 20 mL of 4 °C 0.9% NaCl via PV + 5 mL of 4 °C 0.9% NaCl via HA |
| Okamura et al[39] | PV | 50 mL of preservation solution |
| Berardo et al[40] | PV | Ringer lactate |
| Chai et al[89] | NA | Cold UW solution until liver color changed to khaki |
| Zeng et al[41] | PV | 20 mL of 4 °C HTK solution |
| Beal et al[42] | PV | 60 mL of cold 0.9% saline with 1 mL heparin (100 U) |
| Tabka et al[69] | A, PV | 15 mL of 20 °C Ringer’s lactate or Celsior or with Celsior supplemented with 10-9 M of Ang IV |
| Xue et al[43] | PV | 20 mL of 4 °C HTK solution |
| He et al[78] | A | Approximately 10 mL of 0-4 °C saline |
| Gassner et al[70] | A, PV | 20 mL of 4 °C HTK solution (with/without 12 mmol/L glycine) |
| Zeng et al[79] | A | 2 mL of cold HTK solution with 10 IU/mL heparin |
| Jia et al[80] | A | Cold saline with 25 IU/mL of heparin |
| Oldani et al[44] | PV | 20 mL of cold IGL-1 solution with 100 IU heparin |
| Chin et al[45] | PV | 50 mL of 4 °C 0.9% NaCl |
| Gillooly et al[46] | PV | 10 CC of cold saline with 100 IU heparin |
| Martins et al[47] | PV | 4 °C Celsior solution |
| Scheuermann et al[48] | PV | 40 mL of cold UW solution |
| Claussen et al[71] | A, PV | 20 mL of 4 °C HTK solution supplemented with 12 mmol/L glycine |
| Haque et al[49] | PV | 50 mL of ice cold Ringer’s lactate with heparin |
| Schlegel et al[50] | PV | 10 mL of cold heparinized IGL-1 solution |
| Nösser et al[51] | PV | 20 mL of Ringer solution |
| Yamada et al[96] | NA | NA |
| Hu et al[52] | PV | 20 mL of 0-4 °C HTK solution |
| Von Horn and Minor[53] | PV | 60 mL of HTK solution |
| Raigani et al[54] | PV | 50 mL of ice-cold 0.9% saline |
| Yang et al[55] | PV | 2 mL of saline |
| Westerkamp et al[76] | PV, HA | 10 mL of 37 °C 0.9% NaCl via PV. 5 mL of 4 °C HTK solution via PV. 20 mL of 4 °C HTK solution via PV. 5 mL of 4 °C HTK solution via HA |
| De Vries et al[86] | NA | 60 mL of ice-cold saline |
| Liu et al[85] | HA | Heparin saline 40 mL (50 IU/L) |
| Lin et al[56] | PV | NA |
| Rigo et al[57] | PV | 10 mL of cold Celsior solution. 30 mL of cold Celsior solution. 10 mL of WEM |
| Xu et al[97] | NA | NA |
| Carlson et al[58] | PV | 20 mL of NaCl saline |
| Zhou et al[81] | A | 50 mL of 4 °C HTK solution |
| Cao et al[59] | PV | 10 mL of UW solution |
| De Stefano et al[60] | PV | 10 mL of saline or Celsior solution |
| Sun et al[61] | PV | NA |
| Jennings et al[62] | PV | 20 mL of cold 0.9% saline |
| Wang et al[72] | A, PV | 3 mL of cold HTK solution |
| Asong-Fontem et al[73] | A, PV | 50 mL of preservation solution. 20 mL of preservation solution at 5 °C ± 3 °C |
| Shi et al[77] | PV, HA | 0-4 °C heparinized saline |
| Von Horn et al[63] | PV | 60 mL of 4 °C HTK solution |
| Zhou et al[82] | A | 50 mL of 4 °C HTK solution |
| Luo et al[83] | A | 40 mL of heparinized saline (50 IU/mL) |
| Ohara et al[64] | PV | Ringer’s solution |
| Chen et al[65] | PV | 5 mL of 37 °C heparinized saline (2500 IU/mL) |
| Fukai et al[74] | A, PV | 60 mL of ice-chilled saline 25 mL of ice-chilled Belzer UW or Belzer MPS solutions |
| Hughes et al[84] | A | 80 mL of 4 °C HTK solution |
| Bai et al[98] | NA | NA |
| Von Horn et al[66] | PV | 60 mL of 4 °C HTK solution |
| Li et al[87] | NA | NaCl solution |
The solutions used for cold flushes vary significantly, with volumes ranging from 3 mL to 80 mL. Some researchers choose simple heparinized saline, which is a standard choice for removing blood remnants[80,83,85-87]. In contrast, others use more advanced preservation solutions such as University of Wisconsin (UW), Histidine Tryptophan Ketoglutarate (HTK), Krebs Henseleit, Belzer MP Solution (Belzer MPS), 3-O-methyl-D-glucose, Institute George Lopez (IGL)-1, and William’s E Medium (WEM)[66-73,79,81,82,84,88,89]. These more complex solutions are often chosen because they are also used during the ex vivo MP process, potentially offering better preservation properties.
Additionally, the temperature at which these solutions are administered can vary. Many protocols employ solutions cooled to 0-4 °C to minimize metabolic activity. Others use solutions at higher temperatures, such as 20 °C or even 37 °C[30,33,36,65,75]. Some authors perform the flush with a solution at the same temperature that will be used for the subsequent MP[25]. While cold flush has long been standard procedure before MP, controversially, some suggest that a cold flush before NMP subjects grafts to higher WI damage[12]. Some studies also incorporate oxygenated solutions, which might affect oxygen delivery and tissue viability during the flushing process[20,23].
While authors suggest different solutions and volumes for cold flush, it is important that the pressure during the cold flush for small animal livers is not too high and that adequate blood remnant washout is achieved. Some authors described using constant flow for the cold flush[67], others performed the cold flush until the liver color changed to khaki[89], while the majority applied different volumes and performed it multiple times, both in situ and ex vivo[90-98]. However, some studies do not detail any flushing procedure before applying MP, and future studies should not omit the description of this crucial step.
Heparin, an essential anticoagulant that prevents clotting, is crucial for use during rat liver explant surgery[99,100]. Administering heparin before liver perfusion in the donor and/or into the perfusion solution aids in optimizing liver harvesting[101]. Authors propose diverse routes and dosages for heparin administration, noting its non-liver-toxic effect in rats[102]. Several studies have delved into the administration of heparin via the vena cava (VC), with dosages spanning from 250 IU to 5000 IU[17,20,23,28,31,35,37,40,45,48,54,56]. While other suggested heparinization via iliac vein or abdominal A[41,43,52,78,80]. Alternative routes, including the tibial vein and dorsal penile vein, were explored in other investigations, employing heparin dosages ranging from 100 IU to 500 IU[59,68,75,76]. Moreover, some researchers chose to apply heparin via intraperitoneal administration, dosages ranged from 1000 IU to 1500 IU[60,79]. Various techniques have been employed for administering heparin during liver flushing via the PV, with some studies opting to incorporate heparin into the perfusate solutions[30,42,50,65,77]. These findings show the variability in heparin administration protocols and highlight the importance of further research to establish standardized guidelines for optimal anticoagulation during liver explant surgery and MP.
Anatomically, small animal livers are perfused by proportionally less arterial and more portal blood compared to large animals[103]. Thus, a significant number of authors prefer single-vessel MP via the PV, which necessitates simpler surgery and a more cost-effective MP circuit. Oxygenated MP through the PV provides oxygen and perfusate to the entire graft, including the extrahepatic biliary tree[104]. In contrast, other groups argue that although the PV supplies nutrients to hepatocytes and maintains a higher flow rate compared to the HA, it does not serve as the liver’s primary route for oxygen delivery and does not support the vascularization of the biliary tree as effectively as the HA does[105]. In addition, when selecting a perfusion circuit among the PV, VC, and HA, it’s noted that retrograde perfusion is comparable to PV perfusion, while perfusion via the HA is considered less advantageous[67]. Furthermore, concerns arise regarding the potential direct damage caused by arterial cannulation to the arterial intima, which could compromise vascular anastomosis[106].
However, some authors advocate for dual vessel MP, asserting that it results in superior outcomes compared to single-vessel MP[71]. Although HA cannulation is a complicated procedure, scientists often opt to cannulate via the celiac artery due to its larger diameter[39,84].
To establish a closed MP circuit, the VC must be cannulated, with the other VC outflow either ligated or sutured. Alternatively, the perfusate can flow freely via the infrahepatic and/or suprahepatic VC into the organ chamber, where it is immediately recirculated inside the system[20,23,35].
Bile duct cannulation is essential for measuring bile output. Moreover, it allows to test bile composition which is a great marker of biliary viability[107]. While some authors provide detailed descriptions of the bile duct cannulation process, others may omit it entirely. Nevertheless, it is crucial not to overlook this step. Bile flow is heavily influenced by perfusion temperature and oxygen delivery rate. At 37 °C and adequate oxygenation, bile flow should be at least 1 μL/minute/g liver[108]. Table 2 summarizes which vessels (PV, HA, VC) and common bile duct cannulations were performed by different authors in their MP settings.
| Ref. | Portal vein | Hepatic artery | Vena cava | Common bile duct |
| Kim et al[14]; Manekeller et al[21]; Stegemann et al[22]; Giannone et al[28]; Perk et al[29]; Bae et al[34]; Zeng et al[79]; Jia et al[80]; Gillooly et al[46]; Martins et al[47]; Haque et al[49]; Schlegel et al[50]; Cao et al[59]; Wang et al[72]; Asong-Fontem et al[73]; Li et al[87] | Yes | No or NA | No or NA | No or NA |
| Dutkowski et al[15]; Lee et al[90]; Bessems et al[17]; Olschewski et al[25]; Tolboom et al[27]; Bruinsma et al[88]; Carnevale et al[93]; Niu et al[68]; Bruinsma et al[36]; Zeng et al[41]; Beal et al[42]; Tabka et al[69]; Xue et al[43]; Gassner et al[70]; Oldani et al[44]; Nösser et al[51]; Hu et al[52]; Lin et al[56]; Xu et al[97]; Sun et al[61] | Yes | No or NA | Yes | Yes |
| Compagnon et al[67]; Westerkamp et al[94]; Op den Dries et al[75]; Okamura et al[39]; Claussen et al[71] | Yes | Yes | Yes | Yes |
| Lauschke et al[16]; Xu et al[92]; Dutkowski et al[18]; Tolboom et al[19]; Vairetti et al[20]; Ferrigno et al[23]; Lüer et al[24]; Minor et al[26]; Liu et al[31]; Tarantola et al[35]; Ferrigno et al[37]; Berardo et al[40]; He et al[78]; Chin et al[45]; Scheuermann et al[48]; Yamada et al[96]; Von Horn and Minor[53]; Raigani et al[54]; Yang et al[55]; De Vries et al[86]; Rigo et al[57]; Carlson et al[58]; De Stefano et al[60]; Jennings et al[62]; Von Horn et al[63]; Luo et al[83]; Chen et al[65]; Fukai et al[74]; Von Horn et al[66] | Yes | No or NA | No or NA | Yes |
| Tan et al[91]; Carbonell et al[95]; Chai et al[89] | No or NA | No or NA | No or NA | No or NA |
| Schlegel et al[30]; Schlegel et al[32]; Zhou et al[81]; Zhou et al[82] | Yes | No or NA | Yes | No or NA |
| Schlegel et al[33] | Yes | Yes | Yes | No or NA |
| Jia et al[38]; Shi et al[77] | Yes | Yes | No or NA | No or NA |
| Westerkamp et al[76]; Liu et al[85]; Ohara et al[64]; Hughes et al[84]; Bai et al[98] | Yes | Yes | No or NA | Yes |
Scientists have studied dynamic preservation methods under different durations and temperatures, with varying warm and cold ischemia times before applying MP. As there are still no standard times for the MP application, various studies demonstrate possible outcomes under different durations ranging from 30 minutes to 120 hours (Table 3). Some authors aim to minimize MP time which results in improved liver function[50,72,81,95,96], while others seek to extend MP time as much as possible to prolong preservation[24,26,36,49,88]. Additionally, some researchers focus on determining the optimal time for MP application[52].
| Ref. | Animal | WI time before MP | Length of SCS before MP | MP type and length of MP |
| Kim et al[14] | Rats | NA | 0 hour | HMP: 48 hours |
| Dutkowski et al[15] | Rats | 11.4 minutes 0.8 minutes or 4.2 minutes 0.4 minutes | NA | HMP: 10 hours |
| Compagnon et al[67] | Rats | < 20 seconds | NA | HMP: 24 hours or 48 hours; NMP: 2 hours |
| Lauschke et al[16] | Rats | 60 minutes | NA | HMP: 24 hours; NMP (reperfusion): 45 minutes |
| Lee et al[90] | Rats | 30 minutes | NA | HMP: 5 hours |
| Tan et al[91] | Rats | NA | 30 minutes | HMP: 36 hours |
| Xu et al[92] | Rats | NA | 0 hour (before HMP) or 24 hours (before NMP) | HMP: 24 hours; NMP: 1 hour |
| Bessems et al[17] | Rats | NA | NA | HMP: 24 hours; NMP (reperfusion): 1 hour |
| Dutkowski et al[18] | Rats | NA | 10 hours | HOPE: 3 hours; NMP (reperfusion): 40 minutes |
| Tolboom et al[19] | Rats | NA | 0 hour | NMP: 6 hours |
| Vairetti et al[20] | Rats | NA | NA | 4 °C, 10 °C, 20 °C, 25 °C, 30 °C, or 37 °C MP: 6 hours, NMP (reperfusion): 2 hours |
| Manekeller et al[21] | Rats | 30 minutes | NA | HMP: 18 hours; NMP (reperfusion): 2 hours |
| Stegemann et al[22] | Rats | 30 minutes | NA | HMP: 18 hours; NMP (reperfusion): 2 hours |
| Ferrigno et al[23] | Rats | NA | NA | HMP or SNMP: 6 hours; NMP (reperfusion): 2 hours |
| Lüer et al[24] | Rats | NA | NA | HMP: 18 hours; NMP (reperfusion): 2 hours |
| Olschewski et al[25] | Rats | 60 minutes | NA | HMP or SNMP: 6 hours; NMP (reperfusion): 6 hours |
| Minor et al[26] | Rats | 30 minutes | NA | HMP: 18 hours; NMP (reperfusion): 2 hours |
| Tolboom et al[27] | Rats | 1 hour | NA | SNMP: 5 hours; NMP: 5 hours |
| Giannone et al[28] | Rats | NA | NA | Normobaric or hyperbaric HMP: 24 hours |
| Perk et al[29] | Rats | 60 minutes or 90 minutes | NA | NMP: 6 hours |
| Schlegel et al[30] | Rats | 30 minutes | 4 hours | HMP: 1 hour |
| Bruinsma et al[88] | Rats | NA | 0 hour, 24 hours, 48 hours, 72 hours and 120 hours | SNMP: 3 hours |
| Liu et al[31] | Rats | NA | NA | SNMP: 6 hours |
| Carnevale et al[93] | Rats | 45 minutes | NA | HMP: 24 hours; NMP (reperfusion): 1.5 hours |
| Schlegel et al[32] | Rats | NA | 30 minutes | HMP or HNP: 1 hour |
| Schlegel et al[33] | Rats | 30 minutes or 60 minutes | NMP: 0 minute or 15 minutes; HOPE: 4 hours | NMP: 4 hours; HOPE: 1 hour |
| Bae et al[34] | Rats | 30-40 minutes | NA | HMP: 8 hours; NMP (reperfusion): 90 minutes |
| Niu et al[68] | Rats | 60 minutes | 5 hours | NMP: 2 hours |
| Tarantola et al[35] | Rats | NA | 6 hours | SNMP, NMP (reperfusion): 6 hours |
| Bruinsma et al[36] | Rats | NA | NA | Loading SNMP: 80 minutes; supercooling: 3-4 days; recovery SNMP: 5 hours |
| Ferrigno et al[37] | Rats | NA | NA | 10 °C, 20 °C, 30 °C or 37 °C MP: 6 hours |
| Jia et al[38] | Rats | NA | 0 hour or 6 hours | HMP: 6 hours or 0 hour |
| Westerkamp et al[94] | Rats | 30 minutes | 6 hours | HMP or SNMP or rewarming MP: 1 hour; NMP: 2 hours |
| Carbonell et al[95] | Rats | NA | NA | NMP: 15 minutes to stabilize (all groups); SNMP or NMP: 30 minutes |
| Op den Dries et al[75] | Rats | 0 minute or 30 minutes | 0 hour or 3 hours | NMP: 3 hours; NMP (reperfusion): 2 hours |
| Okamura et al[39] | Rats | NA | NA | SNMP: 4 hours; NMP: 2 hours |
| Berardo et al[40] | Rats | NA | 6 hours | SNMP: 6 hours; NMP: 2 hours |
| Chai et al[89] | Rats | NA | NA | HMP: 2 hours or 12 hours |
| Zeng et al[41] | Rats | 30 minutes | NA | HMP: 3 hours; NMP (reperfusion): 2 hours |
| Beal et al[42] | Rats | NA | NA | NMP: 4 hours |
| Tabka et al[69] | Rats | NA | NA | SNMP: 6 hours; NMP (reperfusion): 2 hours |
| Xue et al[43] | Rats | 30 minutes | NA | HMP: 3 hours; NMP: 1 hour |
| He et al[78] | Rats | NA | NA | HMP: 3 hours or 6 hours |
| Gassner et al[70] | Rats | 30 minutes | NA | NMP: 6 hours |
| Zeng et al[79] | Mice | 30 minutes | 4 hours | HOPE or HNPE: 1 hour; NMP (reperfusion): 2 hours |
| Jia et al[80] | Rats | NA | NA | HMP: 6 hours |
| Oldani et al[44] | Rats | 1 hour | 30 minutes | HOPE or NMP: 2 hours |
| Chin et al[45] | Rat | NA | NA | NMP: 6 hours |
| Gillooly et al[46] | Rats | 25 minutes | NA | HMP, NMP: 4 hours |
| Martins et al[47] | Rats | NA | 12 hours | NMP: 1 hour |
| Scheuermann et al[48] | Rats | NA | 0 hour | SNMP or NMP: 4 hours; NMP (reperfusion): 2 hours |
| Claussen et al[71] | Rats | < 15 minutes | < 60 minutes | NMP: 6 hours |
| Haque et al[49] | Rats | NA | NA | NMP: 24 hours |
| Schlegel et al[50] | Rats | 30 minutes | 4 hours | NMP or HOPE: 1 hour |
| Nösser et al[51] | Rats | NA | 81.71 minutes ± 28.44 minutes | SNMP or NMP: 6 hours or 12 hours |
| Yamada et al[96] | Rats | 30 minutes | 6 hours | SNMP or NMP: 30 minutes, 60 minutes or 90 minutes |
| Hu et al[52] | Rats | 30 minutes | NA | HMP: 1 hour, 3 hours, 4 hours, 12 hours, 24 hours; NMP: 2 hours |
| Von Horn and Minor[53] | Rats | 20 minutes | 18 hours | NMP: 2 hours |
| Raigani et al[54] | Rats | NA | < 10 minutes | NMP: 6 hours |
| Yang et al[55] | Rats | 30 minutes | NA | NMP: 8 hours |
| Westerkamp et al[76] | Rats | NA | 4 hours | NMP: 3 hours |
| De Vries et al[86] | Rats | 0 h | 0 hour or 24 hours or 72 hours | SNMP: 3 hours |
| Liu et al[85] | Rats | 0 minute, 10 minutes or 30 minutes | NA | HMP or NMP: 4 hours |
| Lin et al[56] | Rats | NA | 1 hour | HMP: 3 hours |
| Rigo et al[57] | Rats | 3.10 minutes (0.35) mean (SEM) | NA | NMP: 4 hours |
| Xu et al[97] | Rats | NA | NA | NMP: 4 hours |
| Carlson et al[58] | Rats | NA | < 5 minutes | NMP: 4 hours |
| Zhou et al[81] | Rats | 30 minutes | 23 hours | HOPE: 1 hour; NMP: 1 hour |
| Cao et al[59] | Rats | 30 minutes | NA | NMP: 4 hours |
| De Stefano et al[60] | Rats | 60 minutes | No or 34 minutes ± 7 minutes | NMP: 6 hours |
| Sun et al[61] | Rats | 30 minutes | NA | NMP: 6 hours |
| Jennings et al[62] | Rats | NA | < 5 minutes | NMP: 4 hours |
| Wang et al[72] | Mice | 10 minutes | 11 hours | HOPE: 1 hour; NMP (reperfusion): 2 hours |
| Asong-Fontem et al[73] | Rats | NA | 24 hours | HOPE: 2 hours; NMP (reperfusion): 2 hours |
| Shi et al[77] | Rats | 30 minutes | 8 hours | NMP: 2 hours |
| Von Horn et al[63] | Rats | 20 minutes | 17 hours or 18 hours | HMP: 1 hour; NMP (reperfusion): 2 hours |
| Zhou et al[82] | Rats | 30 minutes | 0 hour or 23 hours | HOPE: 1 hour; NMP: 1 hour |
| Luo et al[83] | Rats | 30 minutes | 3 hours or 4 hours | HMP or HOPE: 1 hour; NMP: 2 hours |
| Ohara et al[64] | Rats | 60 minutes | NA | NMP: 4 hours |
| Chen et al[65] | Mice | NA | NA | NMP: 12 hours |
| Fukai et al[74] | Rats | 30 minutes | NA | HMP: 3 hours; NMP (reperfusion): 90 minutes |
| Hughes et al[84] | Rats | 30 minutes | During MP priming | NMP: 4 hours |
| Bai et al[98] | Rats | 30 minutes | 8 hours | NMP: 2 hours |
| Von Horn et al[66] | Rats | 20 minutes | 18 hours | Rewarming MP: 2 hours; NMP: 1 hour |
| Li et al[87] | Rats | NA | NA | NMP: 3 hours |
Ex vivo rat liver MP studies are performed for a number of different reasons. They compare dynamic preservation techniques with SCS, especially concerning older or fatty livers[67,75,78,90,93,94]. Comparisons between dynamic preservation methods aim to identify the most effective approach[33,37,50,63,96]. Some authors study the role of oxygenation in liver preservation, providing evidence of its advantageous outcomes in improving liver function[22,24]. Researchers investigate various perfusates[17,21,92], including additives such as tacrolimus[32], α-tocopherol[34], pegylated-catalase[42], dopamine[26], angiotensin IV[69], metamizole[71], metformin[76], oxygen carriers[62], and IGL-2[73], to understand how they impact liver function. Some studies explore the introduction of stem cells to improve liver function[55,59-61], while others aim to treat fatty liver disease during MP[23,31,35,39,54,56,72,87,97]. Additionally, efforts are made to enhance donation after DCD liver function[81-85,94,96]. Moreover, there are promising initial attempts to apply gene therapy during MP[46].
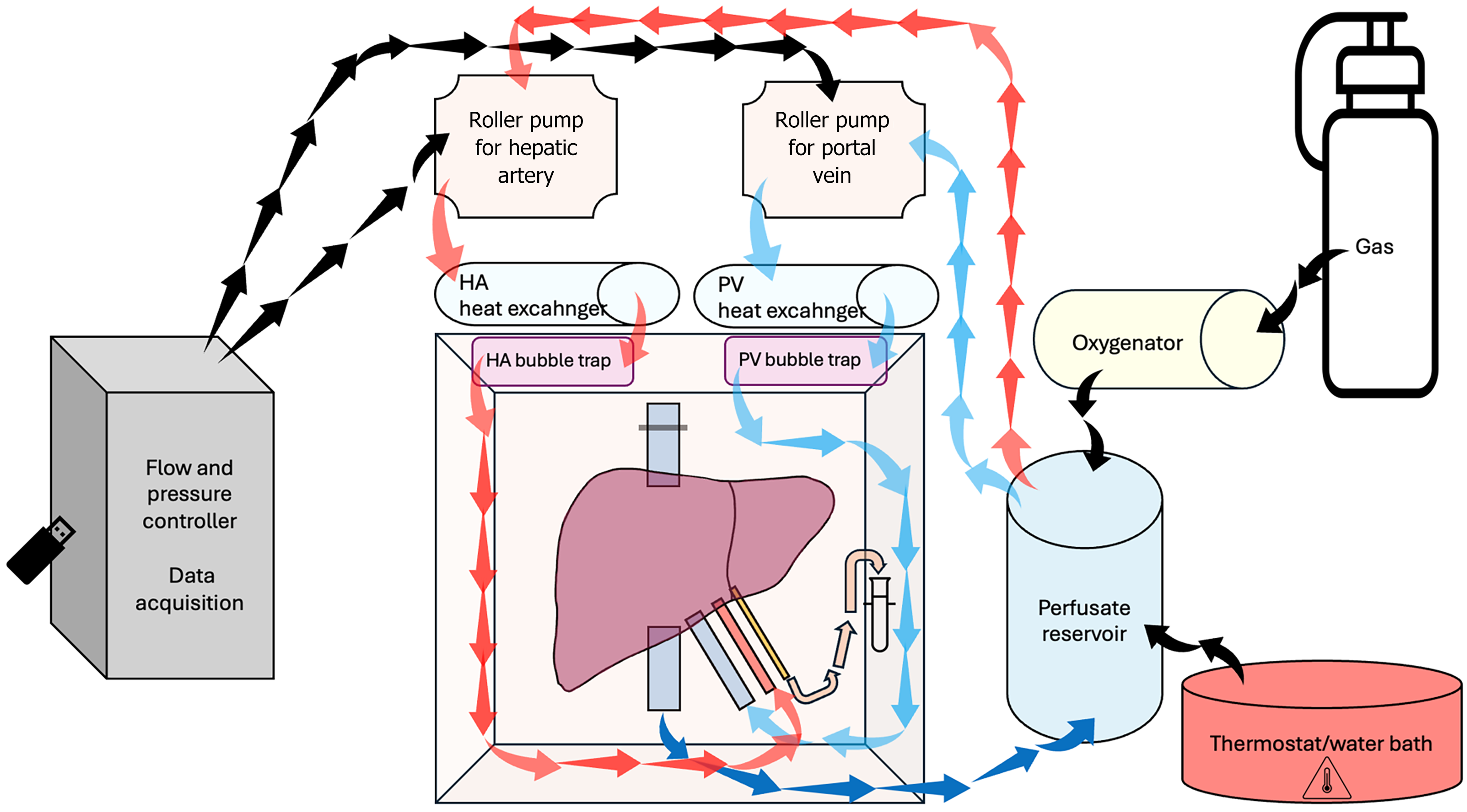
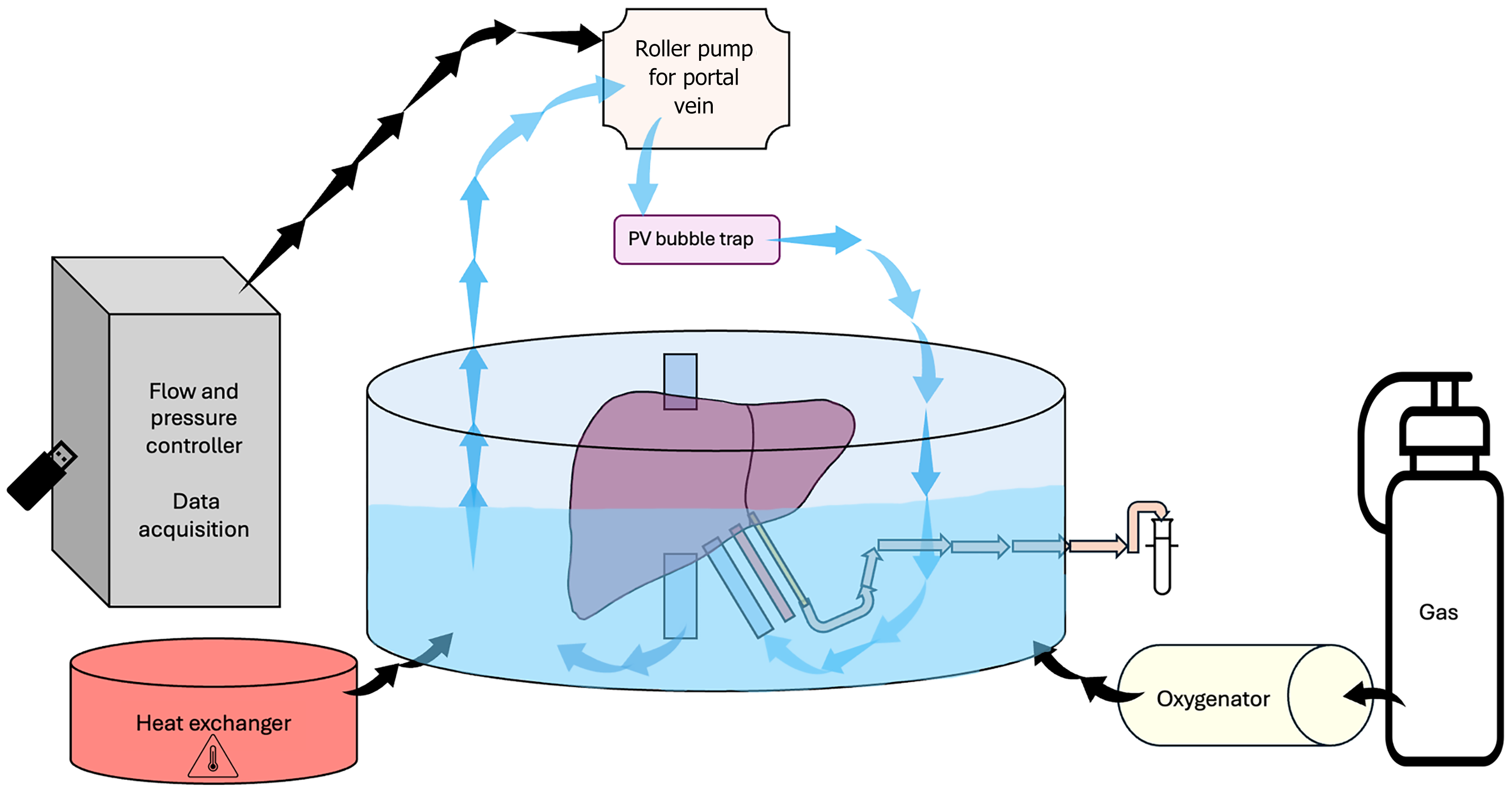
A standard perfusion system is made of organ chamber, perfusate reservoir, peristaltic pump, heat exchanger, bubble trap, oxygenator and tubing[65] (Figures 2 and 3). Various MP systems have been described, ranging from simpler self-made setups to more complex designs[70-110]. The organ chamber lies at the center of the MP system, where the liver is carefully positioned hilum facing upwards. Some authors suggest using an elastic pillow to aid in positioning the liver[51]. The perfusate reservoir, connected to the chamber, holds the perfusion solution to be pumped through the organ. Alternatively, the organ chamber could also serve as perfusate reservoir[19]. The peristaltic pump regulates the flow rate of the perfusate, ensuring precise delivery to the organ. When perfusion is performed via both PV and HA, two peristaltic pumps are necessary[11,75]. Additionally, controlling temperature is crucial, often achieved through the heat exchanger, thermostat[76], heating water bath with temperature sensor[41,52], combined heat exchanger-oxygenator[54], and other alternatives, maintaining the perfusate at the desired temperature throughout the process. The oxygenator enriches the solution with oxygen to sustain the metabolic needs of the organ. Various types of oxygenators, including membrane[29,47,55,59,61,75,76,84,88], hollow-fiber[32,52,57,77], bubble[93], tubing[15,53,58] and others, are discussed by the authors. The bubble trap removes air bubbles from the perfusate and prevents air embolism. Tubing connects these components, allowing for the seamless flow of the perfusate through the system. The implementation of sample ports is also crucial for obtaining perfusate samples during the MP process[93]. Monitoring MP parameters is also highly significant. Some researchers choose basic manometers to measure pressure[105], while others prefer sophisticated equipment equipped with diverse sensors and data acquisition devices, enabling real-time analysis and display of parameters[75]. Some researchers propose incorporating a dialysis unit into the MP circuit, which could potentially improve preservation outcomes during NMP[27,51].
According to the literature, the volume of perfusate used in small animal MP can range from 2 mL to 500 mL, as detailed in Table 4. Although there are no specific recommendations regarding the total perfusion volume, some authors propose reducing it to 50 mL. Notably, when the perfusate includes red blood cells, reducing the volume has been shown to increase hematocrit levels and decrease the release of transaminases[51].
| Ref. | Animal weight | Liver weight | Perfusate volume | Perfusate composition |
| Kim et al[14] | 200-400 g | NA | 200 mL | Cold UW-G solution |
| Dutkowski et al[15] | 250-300 g | 10.4 ± 0.3 g | 500 mL | Modified UW solution: Starch and glutathione was omitted, supplemented with 80 mg/L gentamycin, and 5000 IU/L heparin |
| Compagnon et al[67] | 300 ± 50 g | NA | 150 mL | HMP: Celsior-HES solution. NMP: KHB buffer with 5% bovine serum albumin |
| Lauschke et al[16] | 250-300 g | NA | 125 mL | HMP: HTK or Belzer MPS solution supplemented with 6000 IU superoxide dismutase. NMP: KH buffer |
| Lee et al[90] | 200-250 g | NA | 100 mL | KH solution, then switched to UW solution (starch omitted) |
| Tan et al[91] | 180-220 g | NA | 120 mL | Modified Hoffmann perfusate: Hydroxyethyl starch 50 g/L, calcium gluconate 80 mmol/L, raffinose 10 mmol/L, KH2PO4 25 mmol/L, hydroxyethyl piperazine 10 mmol/L, dexamethasone 12 mg/L, penicillin 2 × 105 units/L, insulin 100 units/L, and with/without 25 mmol/L MgCl and/or 5 mmol/L ATP |
| Xu et al[92] | 200-250 g | NA | NA | HMP: UW solution with/without starch. NMP: KH buffer solution containing 112 μmol/L taurocholic acid, and 150 μg/L hyaluronic acid |
| Bessems et al[17] | 350 ± 50 g | 16.5 ± 0.5 g | 250 mL | HMP: The UW-G solution or polysol. NMP: KHB without bovine serum albumin |
| Dutkowski et al[18] | 250-300 g | 10.8 ± 1.4 g | 450 mL | HOPE: Modified starch-free UW solution. NMP: KHB buffer |
| Tolboom et al[19] | 250-300 g | 9.74-0.81 g | 55-60 mL | NMP: Phenol red-free WEM supplemented with 2 IU/L insulin, 40000 IU/L penicillin, 40000 mg/L streptomycin, 0.292 g/L L-glutamine, 10 mg/L hydrocortisone, 1000 IU/L heparin with 25% (v/v) freshly isolated rat plasma and freshly isolated rat erythrocytes to a hematocrit of 16% to 18% |
| Vairetti et al[20] | 250-300 g | NA | 200 mL | KH solution with 1.25 mmol/L CaCl2 or with 0.25 mmol/L CaCl2 |
| Manekeller et al[21] | 250-300 g | NA | 125 mL | HMP: HTK or Blezer MPS solutions. NMP: KH buffer |
| Stegemann et al[22] | 250-300 g | NA | HMP: 125 mL; NMP: 250 mL | HMP: HTK or modified HTK solution (custodiol-N). NMP: KH buffer with 3 g/100 mL of bovine serum albumin |
| Ferrigno et al[23] | 375 ± 15 g and 300 ± | NA | 200 mL | KH medium |
| Lüer et al[24] | 250-300 g | NA | 125 mL | HMP: HTK solution. NMP: WEM solution, supplemented with 3 mg/100 mL of bovine serum albumin |
| Olschewski et al[25] | 250-280 g | NA | NA | HMP, SNMP: Lifor solution. NMP: KH solution |
| Minor et al[26] | 250-300 g | NA | HMP: 125 mL; NMP: 250 mL | HMP: HTK solution supplemented with 0 μmol/L, 10 μmol/L, 50 μmol/L or 100 μmol/L of dopamine. NMP: WEM solution with 3 mg/100 mL of bovine serum albumin |
| Tolboom et al[27] | 250-300 g | NA | 55-60 mL | WEM with autologous erythrocytes and plasma. To this were added: Insulin (2 IU/L), penicillin (40000 IU/L)/streptomycin (40000 μg/L), L-glutamine (0.292 g/L), hydrocortisone (10 mg/L), and heparin (1000 IU/L) |
| Schlegel et al[30] | 250-300 g | 9.83 ± 0.95 g | 50 mL | Modified starch free UW-solution |
| Giannone et al[28] | 250-300 g | NA | NA | Celsior solution |
| Perk et al[29] | 200-300 g | NA | 55-60 mL | Phenol red-free WEM supplemented with 2 IU/L insulin (28.85 units/mg), 100000 IU/L penicillin, 100 mg/L streptomycin sulfate, 0.292 g/L L-glutamine, 10 mg/L hydrocortisone, and 1000 IU/L heparin. Fresh rat plasma (25% v/v) and erythrocytes (18%-20% v/v) were collected and added to the perfusate |
| Bruinsma et al[88] | 180-250 g | NA | 500 mL | WE supplemented with insulin (2 IU/L), penicillin (40000 IU/L)/streptomycin (40000 μg/L), L-glutamine (0.292 g/L), hydrocortisone (10 mg/L) |
| Liu et al[31] | NA | 21.4 ± 3.6 g (control) 22.9 ± 5.8 g (defatting group) | 200 mL | The control perfusate: Minimum essential medium supplemented with 3% wt/vol bovine serum albumin, 1.07 mmol/L lactic acid, and 0.11 mmol/L pyruvic acid. The defatting perfusate: The control perfusate supplemented with the 6 defatting agents (forskolin, GW7647, scoparone, hypericin, visfatin, and GW501516) |
| Carnevale et al[93] | 250-300 g | NA | 250 mL | HMP: HTK solution: 100 mmol/L sodium gluconate, 7 mmol/L potassium gluconate, 20 mmol/L sucrose, 30 mmol/L BES, 2.5 mmol/L KH2PO4, and 0.03 mmol/L polyethylene glycol (35 kDa), 5 mmol/L MgSO4, 3 mmol/L glutathione, 5 mmol/L adenosine, and 15 mmol/L glycine, together with 0.25 mg/mL streptomycin and 10 IU/mL penicillin G. NMP: KH buffer with 4% dextran added |
| Schlegel et al[32] | 250-320 g | 9.7 ± 1.5 g | 50 mL | Modified starch free UW-solution |
| Schlegel et al[33] | 250-320 g | 10.14 ± 2.73 g | 50 mL | NMP: Diluted full blood or leukocyte and platelet depleted blood perfusate. HOPE: Modified starch-free UW solution |
| Bae et al[34] | 300 ± 25 g | NA | 100 mL | HMP: KPS-1 solution (Identical to Belzer’s UW MPS) or KPS-1, enhanced with α-ketoglutarate, L-arginine, N-acetylcysteine, nitroglycerin, and prostaglandin E1 or KPS-1, with 5.4 × 10-2 mmol/L of α-tocopherol diluted in acetone. NMP: KHB buffer |
| Niu et al[68] | 261 ± 4 g | NA | 353 ± 11 mL | KH buffer |
| Tarantola et al[35] | 375 ± 15 g and 300 ± | NA | 200 mL | KH medium |
| Bruinsma et al[36] | 250-300 g | Approximately 10 g | 100 mL | 3-OMG loading solution: 500 mL phenol-red free WEM, 5 mL of 200 mmol/L L-glutamine (0.292 mg/L), 4 mL of penicillin-streptomycin (5000 IU/mL), 5 mg of hydrocortisone, 5000 U of sodium heparin, 375 U of insulin, 19.42 g of 3-O-methyl glucose (0.2 M) |
| Ferrigno et al[37] | 250-300 g | NA | NA | A modified KH buffer |
| Jia et al[38] | 250-300 g | NA | 60 mL | UW or HTK solutions |
| Westerkamp et al[94] | 290-320 g | NA | 100 mL | HMP, SNMP, rewarming MP: Belzer MPS. NMP: 25 mL of human red blood cell concentrate (final hematocrit 25%), 53.9 mL of WEM solution, 20 mL of human albumin (200 g/L), 1 mL of insulin (100 IU/mL), and 0.1 mL of unfractionated heparin (5000 IU/mL) |
| Carbonell et al[95] | 225-250 g | NA | NA | KH buffer: 118 mmol/L NaCl, 4.7 mmol/L KCl, 1.2 mmol/L MgSO4, 1.2 mmol/L KH2PO4, 2.5 mmol/L CaCl2, 25 mmol/L NaHCO3, 20 mmol/L HEPES |
| Op den Dries et al[75] | 303 ± 4 g (mean ± SEM) | 100 mL | 20 mL of human red blood cell concentrate (final hematocrit 15%-20%), 59 mL of WEM solution, 20 mL of human albumin (200 g/L), 1 mL of insulin (100 IU/mL), and 0.1 mL of unfractionated heparin (5000 IU/mL) | |
| Okamura et al[39] | 250-300 g | NA | 300 mL | SNMP: Polysol solution. NMP: KH buffer |
| Berardo et al[40] | 250-300 g | NA | NA | KH buffer |
| Chai et al[89] | 250-300 g and 600-630 g | NA | 80 mL | UW solution with/without 0.165 mg/L of metformin |
| Zeng et al[41] | 250-300 g | NA | 150 mL | HTK solution |
| Beal et al[42] | 250-350 g | NA | 300 mL | 86 mL of 25% albumin, 184 mL of WEM, 30 mL of penicillin/streptomycin (10 IU/mL penicillin and 0.01 mg/mL streptomycin), insulin (50 IU/L), heparin (0.01 IU/mL), L-glutamine (0.292 g/L), and hydrocortisone (0.010 g/L). Addition of 625 IU/mL pegylated-catalase |
| Tabka et al[69] | 250-300 g | NA | SNMP: 150 mL; NMP: 140 mL | SNMP: Celsior with/without Ang IV. NMP: KBB enriched with 5% albumin |
| Xue et al[43] | 250 ± 10 g | NA | HMP: 100 mL; NMP: 250 mL | HMP: HTK solution. NMP: KH buffer with 4% dextran |
| He et al[78] | 250-300 g | NA | NA | HTK solution |
| Gassner et al[70] | 280-350 g | 13.6 g ± 2.15 g | 50 mL | NMP: Low-glucose DMEM supplemented with the rat erythrocyte concentrate and 12.5 mL strain specific rat plasma |
| Zeng et al[79] | 20-24 g | NA | 50 mL | HTK solution |
| Jia et al[80] | 250-300 g | NA | 60 mL | HTK solution |
| Oldani et al[44] | 176 (155-193) g | NA | 50 mL | HOPE: IGL-1 solution with 150 IU heparin. NMP: 25 mL Fischer rat blood with 25 mL normal saline, and with 150 IU heparin |
| Chin et al[45] | 200-250 g | NA | 150 mL | DMEM supplemented with 200 mmol/L L-glutamine, 10% v/v FBS, 5% with bovine serum albumin, 8 mg/L dexamethasone, 2000 IU/L heparin, 2 IU/L insulin, and 5 × 106 engineered rat fibroblasts |
| Gillooly et al[46] | Approximately 300 g | NA | 100 mL | 99 mL WEM with 10 IU insulin, and lipid nanoparticles (50 nM siRNA) |
| Martins et al[47] | 320-350 g | NA | NA | 50% plasma-Lyte and 50% KH solution |
| Scheuermann et al[48] | 296 g ± 8 g (mean ± SEM) | NA | 100 mL | SNMP, NMP: 80 mL of WEM supplemented with 5% bovine serum albumin, 20 mL of type O + human RBCs, 0.2 IU insulin, 29.2 mg L-glutamine, 1 mg hydrocortisone, and 500 IU heparin. NMP: KH buffer supplemented with 5% bovine serum albumin |
| Claussen et al[71] | 280-350 g | NA | 50 mL | 10 mL of the erythrocyte concentrate, 35 mL of DMEM, 5 mL of strain specific rat plasma supplemented with 1000 IU heparin and 12 mmol/L glycine |
| Haque et al[49] | 250-300 g | NA | 500 mL | NMP: 950 mL of William’s E media, 20 g of bovine serum albumin, 20 g of polyethylene glycol 35000, 20 mg of dexamethasone, 2 mL of heparin, 1 mL of regular insulin, 10 mL penicillin-streptomycin, 10 mL of antibiotic-antimycotic, and 2.2 g of sodium bicarbonate |
| Schlegel et al[50] | 250-320 g | 9.8 0.6 g | 100 mL | HOPE: Belzer MPS. NMP: Belzer MPS or diluted heparinized blood |
| Nösser et al[51] | 398.87 ± 133.12 g | 14.15 ± 2.66 g | 250 mL, 100 mL, 80 mL, 50 mL | SNMP: 500 Ml DMEM supplemented with 100 μg/mL penicillin and streptomycin, 4 mmol/L L-glutamine/L-alanine, 1 μM human insulin, 14 ng/mL glucagon, 1 μM dexamethasone. NMP: The isolated RBCs suspended in DMEM, isolated rat plasma (10% of the total volume), and 500 IU of heparin |
| Yamada et al[96] | 260-350 g | DCD 10.12 ± 0.93 g | NA | KH buffer |
| Hu et al[52] | 250-300 g | NA | 150 mL | HTK solution |
| Von Horn and Minor[53] | 250-300 g | NA | 200 mL | Aqix RS-I solution |
| Raigani et al[54] | NA | NA | 500 mL | High-glucose DMEM supplemented with 10% v/v FBS, 2% v/v penicillin-streptomycin, and 3% w/v bovine serum albumin. Defatting cocktail agents include 10 μM forskolin, 1 μM GW7647, 1 μM GW501516, 10 μM scoparone, 10 μM hypericin, 0.4 ng/mL visfatin, 0.8 mmol/L L-carnitine, and additional amino acids |
| Yang et al[55] | 200-220 g | NA | NA | 60 mL DMEM/F12 (1:1) containing 20% FBS and 1% penicillin-streptomycin solution (penicillin 10000 IU/mL, streptomycin 10000 μg/mL), 20 mL of fresh blood, 5 IU/mL of heparin, 2 IU/L of insulin, and 2.5 μg/mL of dexamethasone |
| Westerkamp et al[76] | 270-300 g | NA | 100 mL | 25 mL human red blood cell concentrate, 53.9 mL WEM solution, 20 mL human albumin (200 g/L), 1 mL insulin (100 IU/mL), and 0.1 mL unfractionated heparin (5000 IU/mL) |
| De Vries et al[86] | 200-250 g | NA | 500 mL | WEM supplemented with sodium bicarbonate (2.2 g/L), dexamethasone (24 mg/L), insulin (5 IU/L), heparin (2000 IU/L), and bovine serum albumin (10 mg/mL) |
| Liu et al[85] | NA | NA | 70 mL | HMP: UW solution with 250 IU heparin, 20000 IU penicillin and 2 mg hydrocortisone. Addition of CIRP competitive inhibitor (C23, 300 ng/mL). NMP: 20 mL rat blood, 50 mL WEM, 250 IU heparin, 20000 IU penicillin, 2 mg hydrocortisone, 0.4 IU insulin, and 0.0292 g glutamine |
| Lin et al[56] | 180 ± 20 g | NA | 50 mL | HTK solution with or without various defatting agents (10 mmol/L forskolin; 1 mmol/L GW7647; 10 mmol/L hypericin; 10 mmol/L scoparone; 0.4 ng/mL visfatin; 1 mmol/L GW501516) |
| Rigo et al[57] | 200-250 g | 10.35 g (0.41) mean (SEM) | 70 mL | NMP: 20 mL of fresh rat blood, 50 mL of complete phenol red-free WEM, supplemented with 11.6 mmol/L glucose, 50 IU/mL penicillin, 50 μg/mL streptomycin, 5 mmol/L L-glutamine, 1 IU/mL insulin, 1 IU/mL heparin, and 2 mEq of sodium bicarbonate |
| Xu et al[97] | Approximately 500 g | Approximately 15 g | 60-110 mL | Whole blood-based perfusate with different defatting components |
| Carlson et al[58] | 331 ± 16 g (mean ± SEM) | NA | 95 mL | WEM, 3250 IU each penicillin/streptomycin, 0.65 mmol/L sodium pyruvate, 1.30 mmol/L L-glutamine, 1% human albumin, 500 IU heparin, 15 mg papaverine, 1 mg insulin, 1.25 mg hydrocortisone, and 30 mL of leukoreduced, packed RBCs |
| Zhou et al[81] | 250-300 g | NA | NA | NA |
| Cao et al[59] | 200-220 g | NA | 80 mL | DMEM/F12, 20 mL rat blood, 100 IU/mL penicillin, 100 μg/mL streptomycin, and 5 IU/mL heparin |
| De Stefano et al[60] | 200-250 g | Control group: 17.10 (1.93). WI group: 15.17 (0.83) | 150 mL | 100 mL of phenol red-free WEM, supplemented with 100 IU/mL penicillin, 100 μg/mL streptomycin, 0.292 g/L L-glutamine, 1 IU/mL insulin, 1 IU/mL heparin, and 50 mL of recently expired (max 5 days) human red blood cell |
| Sun et al[61] | 200-220 g | NA | 2 mL | Normal saline or single cell suspension containing 1× 107-3 × 107 BMMSCs |
| Jennings et al[62] | 320 ± 11 g (mean ± SEM) | NA | 130 mL or 146 mL | WEM (65 mL for pRBC oxygen carriers and 50 mL for oxyglobin) with 3250 U each penicillin/streptomycin, 500 U heparin, 1 mg insulin, 1.25 mg hydrocortisone, and 15 mg papaverine. In addition, sodium pyruvate 0.65 mmol/L, L-glutamine 130 mmol/L, and human albumin 1%. The oxygen carriers added to the perfusate were human pRBC (30 mL), rat pRBC (30 mL), or oxyglobin (46 mL) |
| Wang et al[72] | NA | NA | HOPE: 50 mL; NMP: 30 mL | HOPE: HTK solution. NMP: 7.5 mL blood, 22.5 mL KHB solution containing 2% FBS, 60 mg glucose, 1 IU insulin, and 150 IU heparin |
| Asong-Fontem et al[73] | NA | NA | NA | HOPE: IGL-2 or PERF-GEN (Belzer MPS) solutions. NMP: Williams medium E supplemented with insulin 2 U/L, penicillin (40000 U/L)/streptomycin (40000 μg/L), L-glutamine, hydrocortisone 10 mg/L and heparin (1000 U/L) |
| Shi et al[77] | 320-350 g | NA | 36 mL | 24 mL whole heparinized blood supplemented with 10% sodium citrate, 1% penicillin and streptomycin, and 12 mL circuit priming solution with 45% lactated ringer, 5% sodium bicarbonate and 50% hydroxyethyl starch |
| Von Horn et al[63] | 250-300 g | NA | 150 mL | HMP: Aqix RS-I solution. NMP: WEM supplemented with 3 mg/100 mL of bovine serum albumin |
| Zhou et al[82] | 250-300 g | NA | NA | NA |
| Luo et al[83] | 250-300 g | NA | HMP: 150 mL; NMP: 180 mL | HMP: HTK solution. NMP: DMEM/F12, 20% FBS, 1% penicillin streptomycin solution (penicillin 10000 IU/mL, streptomycin 10000 mg/mL), 5 IU/mL of heparin, 2 IU/L of insulin, and 2.5 mg/mL of dexamethasone. Full rat blood (30-45 mL) was reconstituted up to a total volume of 180 mL perfusate |
| Ohara et al[64] | 250-350 g | NA | 100 mL | Rat whole blood |
| Chen et al[65] | 34 ± 4 g (mean ± SEM) | Approximately 1 g | 300 mL | WEM was supplemented with 20% FBS, 1% penicillin/streptomycin, 5000 IU/L heparin, 50 IU/L insulin, and 0.010 g/L hydrocortisone |
| Fukai et al[74] | NA | NA | 300 mL | HMP: UW-MPS solution. NMP: KHB solution |
| Hughes et al[84] | 250-300 g | 14 ± 0.2 g | 150 mL | Phenol red-free WEM with addition of 25% bovine albumin, rat RBCs, 2 IU/L insulin, 40000 IU/L penicillin, 40000 μg/L streptomycin, 0.292 g/L L-glutamine, 10 mg/L hydrocortisone, 1000 IU/L heparin, and bicarbonate 75% |
| Bai et al[98] | 360-380 g | NA | 36 mL | 24 mL whole heparinized blood supplemented with 10% sodium citrate, 1% penicillin, and streptomycin and 12 mL circuit priming solution with 45% lactated ringer, 5% sodium bicarbonate, and 50% hydroxyethyl starch |
| Von Horn et al[66] | 250-300 g | NA | NA | Rewarming MP: Diluted Steen solution or Belzer MPS. NMP: WEM supplemented with 3 mg/100 mL of bovine serum albumin |
| Li et al[87] | 250-350 g | NA | 100 mL | 90 mL WEM with 10 mL rat blood cells supplemented with 10 mg/L hydrocortisone, 5 IU/mL heparin, 1 IU/mL insulin, 5 mmol/L L-glutamine, 40 IU/mL penicillin, 40 μg/mL streptomycin, and with/without 10 mmol/L epigallocatechin gallate |
There is a wide range of suggested perfusate compositions for HMP and SNMP in the literature. Some of the most commonly used perfusates are Krebs Henseleit bicarbonate, UW solution, HTK solution, Belzer MPS, WEM and others. Some researchers use modified perfusates by omitting starch[15,18,30,32,33,90], while others argue that starch-containing solutions enhance endothelial cell function and reduce hepatocellular damage compared to starch-free solutions[92]. In all three types of MP authors usually supplement perfusate with antibiotics and heparin[73,83-85,87,98]. Supplementation with other various medications is less common in HMP and SNMP setting as liver function is suppressed in lower temperatures[109]. However, some authors suggest supplementation in order to improve MP effectiveness and improve liver preservation outcomes. For instance, adding dopamine has been shown to potentially improve HMP effectiveness[26]. Additionally, supplementation with low-dose tacrolimus has resulted in better graft function and survival[32]. Adding α-tocopherol to the perfusate has demonstrated a reduction in inflammatory cytokines[42], while the inclusion of metformin in the perfusion solution has decreased rat liver injury[89]. Furthermore, perfusate supplementation with IGL-2 has been reported to reduce transaminases and significantly lower levels of glycocalyx proteins, CASP3, and HMGB1, indicating its protective role in preserving fatty livers[73]. Supplementation with angiotensin IV has been found to decrease the median effect concentration value and improve endothelium-dependent relaxation of HA rings[69].
For NMP, authors typically use various perfusates mixed with whole fresh blood, artificial blood, red blood cells, or other oxygen carriers. During NMP perfusate supplementation with erythrocytes reduces cell damage and improves liver function[51]. However, some protocols have demonstrated that 12-hour erythrocyte-free NMP in mice has no significant impact on histological structure[65]. Supplementation with additional medications such as insulin, glucose, heparin, hydrocortisone, albumin, and amino acids is more frequent in NMP to better mimic the in vivo liver environment. Some researchers have added pegylated-catalase to the base perfusate, which has reduced liver preservation injury[42]. Glycine treatment has synergistically preserved the integrity of both normal and donation after DCD liver grafts[70]. Supplementation with metamizole has led to higher bile production, lower transaminase levels, and reduced necrosis in liver and bile duct tissue[71]. Several researchers have investigated perfusate supplementation with bone marrow mesenchymal stem cells (BM-MSCs), which have shown promise in enhancing liver quality in rat DCD livers by reducing oxidative stress, improving mitochondrial function, lowering reactive oxygen species and free ferrous ion levels, and repairing the morphology and function of donor livers[55,61]. Additionally, BM-MSCs modified with heme oxygenase 1 have inhibited natural killer cell and cluster of differentiation 8+ T cell activation, thus reducing acute graft rejection[59]. Other medications, such as defatting agents, are used when researchers aim not only to bridge the time until LTx but also to treat conditions like fatty liver disease[56,73,87,97].
Table 4 represents various perfusate compositions and volumes proposed by the authors. To highlight the importance of adjusting perfusate volume according to liver weight, we included the animal and liver weights. This provides a more accurate representation of the relationship between organ weight and perfusate volume used.
Ex vivo rat MP studies are published with a variety of temperature, flow, and pressure settings, although detailed descriptions are missing in several studies. Table 5 represents possible MP parameters used in small animal liver MP models.
| Ref. | HMP | SNMP | NMP | ||||||
| Temperature | Flow | Pressure | Temperature | Flow | Pressure | Temperature | Flow | Pressure | |
| Kim et al[14] | 4 °C | 0.5 mL/minute/g liver | 11.2 ± 0.4 mmHg | ||||||
| Dutkowski et al[15] | 3-6 °C | 0.43-0.44 mL/minute/g liver | 4.48-0.47 mmHg | ||||||
| Compagnon et al[67] | 4 °C | 0.1-0.4 mL/minute/g liver | PV, VC: ≤ 1 mmHg; HA: ≤ 11 mmHg | 37 °C | 3 mL/minute/g wet liver | NA | |||
| Lauschke et al[16] | 4 °C | 0.5 mL/minute/g | NA | 37 °C | 3 mL/g/minute | NA | |||
| Lee et al[90] | 4-5 °C | 0.4 mL/minute/g liver | < 3 mmHg | ||||||
| Tan et al[91] | 6-8 °C | 0.1 mL/minute/g | NA | ||||||
| Xu et al[92] | 4 °C | 0.4 mL/minute/g liver | NA | 37 °C | NA | NA | |||
| Bessems et al[17] | 4 °C | < 1 mL/minute/g liver | < 20 | 37.1 °C ± 0.4 °C | < 3 mL/minute/g liver | < 20 | |||
| Dutkowski et al[18] | 3-5 °C | 2.75-3.25 mL/minute | 4.4 ± 0.5 mmHg | 37 °C | 15 mL/minute | NA | |||
| Tolboom et al[19] | 37.5 °C | 1.8 mL/ minute/g ± 0.12 mL/ minute/g wet liver | 12-15 cmH2O | ||||||
| Vairetti et al[20] | 4 °C, 10 °C | NA | NA | 20 °C, 25 °C, 30 °C | NA | NA | 37 °C | Initial: 1 mL/minute/g, increased to 4 mL/minute/g | NA |
| Manekeller et al[21] | 4 °C | NA | NA | 37 °C | 3 mL/g/minute | NA | |||
| Stegemann et al[22] | 4 °C | 0.5 mL/minute/g | NA | 37 °C | 3 mL/g/minute | NA | |||
| Ferrigno et al[23] | 4 °C or 8 °C | 4 mL/minute/g | 7.4 ± 0.6 mmHg (lean, 4 °C), 8.7 ± 2.1 mmHg (fat, 4 °C), 6.9 ± 0.8 mmHg (lean, 8 °C), 7.1 ± 0.9 mmHg (fat, 8 °C) | 20 °C | 4 mL/minute/g | 7.3 ± 0.8 mmHg (lean) 7.5 ± 1.6 mmHg (fat) | 37 °C | 4 mL/minute/g | NA |
| Lüer et al[24] | 4 °C | 0.5 mL/minute/g | NA | 37 °C | 3 mL/g/minute | NA | |||
| Olschewski et al[25] | 4 °C or 12 °C | 1 mL/minute/g liver | NA | 21 °C | 1 mL/minute/g liver | NA | 37 °C | 3 mL/minute/g liver | NA |
| Minor et al[26] | 4 °C | 0.5 mL/minute/g | NA | 37 °C | 3 mL/g/minute | NA | |||
| Tolboom et al[27] | 20 °C or 30 °C or 37 °C | 2 mL/minute/g | 10-14 cmH2O | 37 °C | 2 mL/minute/g | 10-14 cmH2O | |||
| Giannone et al[28] | 4 °C | 1 mL/minute/g liver | NA | ||||||
| Perk et al[29] | 37 °C | Pressure dependent | 10-12 cmH2O | ||||||
| Schlegel et al[30] | 4 °C | Pressure dependent | ≤ 3 mmHg | ||||||
| Bruinsma et al[88] | Room temperature | NA | NA | ||||||
| Liu et al[31] | 20 °C | 1 mL/minute/g | NA | ||||||
| Carnevale et al[93] | 5.0 °C ± 0.5 °C | 0.23 mL/minute/g liver | 40 mmH2O (25% of the NMP PV pressure) | 37 °C | NA | NA | |||
| Schlegel et al[32] | 4 °C | Pressure dependent | ≤ 3 mmHg | ||||||
| Schlegel et al[33] | 4 °C | PV: 1-2 mL/minte | PV: 3 mmHg | 37 °C | HA: 6 mL/minute; PV: 15 mL/minute | PV: 8 mmHg | |||
| Bae et al[34] | 4 °C | 3.5 mL/minute/g of liver | NA | 37 °C | NA | NA | |||
| Niu et al[68] | 37 °C | 20 mL/minute | NA | ||||||
| Tarantola et al[35] | 20 °C | 4 mL/minute/g | NA | 37 °C | 4 mL/minute/g | NA | |||
| Bruinsma et al[36] | 21°C | 8-12 mL/minute | 10-15 cmH2O | ||||||
| Ferrigno et al[37] | 10 °C | 2.6 mL/minute/g | 5.8 ± 0.2 mmHg | 20 °C or 30 °C | 2.6 mL/minute/g | 4.9 ± 0.1 mmHg, 4.9 mmHg ± 0.2 mmHg | 37 °C | 2.6 mL/minute/g | 4.2 ± 0.1 mmHg |
| Jia et al[38] | 4 °C | 1.4 mL/minute | NA | ||||||
| Westerkamp et al[94] | 8 °C | Pressure dependent | PV: 3 mmHg; HA: 25 mmHg | 20 °C | Pressure dependent | PV: 4 mmHg; HA: 40 mmHg | 37 °C | Pressure dependent | PV: 11 mmHg. HA: 110 mmHg |
| Carbonell et al[95] | 22 °C or 26 °C | 3 mL/minute/g | NA | 37 °C | 3 mL/minute/g | NA | |||
| Op den Dries et al[75] | 37 °C | PV: 22.6 ± 0.8 mL/minute; HA: 5.3 ± 0.4 mL/minute | PV: < 11 mmHg. HA: < 110 mmHg | ||||||
| Okamura et al[39] | 20-24 °C | PV: 1 mL/g-liver/minute; HA: 0.1 mL/g-liver/minute | NA | 37 °C | PV: 3 mL/g-liver/minute | NA | |||
| Berardo et al[40] | 20 °C | NA | Starting: 6-7 mmHg | 37 °C | NA | Starting: 6-7 mmHg | |||
| Chai et al[89] | 4 °C | 4 mL/minute | NA | ||||||
| Zeng et al[41] | 0-4 °C | 0.5 mL/g/minute | NA | 36-37 °C | Pressure dependent | 10.3 mmHg | |||
| Beal et al[42] | 37 °C | 1-2 mL/minute | 10-16 cmH2O | ||||||
| Tabka et al[69] | 20 °C | 0.5 mL/g/minute | NA | 37 °C | NA | NA | |||
| Xue et al[43] | 0-4 °C | 0.23 mL/minute/g | NA | 36-37 °C | 3 mL/minute/g | NA | |||
| He et al[78] | 4 °C | 1.4 mL/minute | NA | ||||||
| Gassner et al[70] | 37 °C | 1 mL/minute/g liver weight | 4-9 mmHg | ||||||
| Zeng et al[79] | Approximately 4 °C | 0.2 mL/minute/g | < 2 mmHg | 36.5 °C ± 0.5 °C | 2.5 mL/minute/g | NA | |||
| Jia et al[80] | 4 °C | 1.4 mL/minute | 4.61 mmHg (mean) | ||||||
| Oldani et al[44] | 4 °C | Pressure dependent | 4 mmHg | 37 °C | Pressure dependent | 8 mmHg | |||
| Chin et al[45] | 37 °C | Initial: 5 mL/minute, then pressure dependent | 5 mmHg | ||||||
| Gillooly et al[46] | 4-7 °C | Pressure dependent | 10 mmHg | 37 °C | Pressure dependent | 10 mmHg | |||
| Martins et al[47] | 32 °C or 37 °C | NA | NA | ||||||
| Scheuermann et al[48] | 25 ± 0.1 °C or 30 °C | 1.80 mL/minute/g liver | NA | 37 °C | 1.80 mL/minute/g liver. Reperfusion: 2.84 ± 0.04 mL/minute/g liver | NA | |||
| Claussen et al[71] | 37 °C | PV: 1 mL/minute/g liver. HA: 0.1 mL/minute/g liver | PV: 5.65-9 mmHg. HA: 48.8-110 mmHg | ||||||
| Haque et al[49] | 37 °C | 25-30 mL/minute | < 12 mmHg | ||||||
| Schlegel et al[50] | 10 °C | 1-2 mL/minute | ≤ 3 mmHg | 37 °C | 15-18 mL/minute | 12 mmHg | |||
| Nösser et al[51] | 21 °C | 1 mL/g wet liver/minute | 5.0 mmHg | 37 °C | 1 mL/g wet liver/minute | 5.0 mmHg | |||
| Yamada et al[96] | 20-25 °C | NA | NA | 37 °C | NA | NA | |||
| Hu et al[52] | 0-4 °C | 0.5 mL/g/minute | NA | 36.5 °C ± 0.5 °C | NA | 10.3 mmHg | |||
| Von Horn and Minor[53] | 35-42 °C | NA | Initial: 3 mmHg. Later: 5 mmHg | ||||||
| Raigani et al[54] | 37 °C | NA | 10-12 mmHg | ||||||
| Yang et al[55] | 35-38 °C | 2 mL/g wet liver/minute | 10-12 mmH2O | ||||||
| Westerkamp et al[76] | 37 °C | NA | PV: 11 mmHg. HA: 110 mmHg | ||||||
| De Vries et al[86] | 21 ± 1 °C | ≤ 25 mL/minute | 5 mmHg | ||||||
| Liu et al[85] | 4 °C | PV: 8 mL/minute | NA | 37 °C | PV: 8 mL/minute; HA: 4 mL/minute | NA | |||
| Lin et al[56] | 4 °C | < 0.15 mL/minute/g | < 3 mmHg | ||||||
| Rigo et al[57] | 37 °C | 1.1-1.3 mL/minute/g | 8-10 mmHg | ||||||
| Xu et al[97] | 37 °C | NA | NA | ||||||
| Carlson et al[58] | 37 °C | 1.8 mL/minute/g | NA | ||||||
| Zhou et al[81] | NA | NA | NA | NA | NA | NA | |||
| Cao et al[59] | 36-38 °C | 1.5 mL/minute/g wet liver | 10-14 cmH2O | ||||||
| De Stefano et al[60] | 37 °C | NA | 12-16 mmHg | ||||||
| Sun et al[61] | NA | NA | NA | ||||||
| Jennings et al[62] | 37 °C | 1.8 mL/minute/g | NA | ||||||
| Wang et al[72] | 4 °C | NA | NA | 36.5 ± 0.5 °C | NA | NA | |||
| Asong-Fontem et al[73] | 5 °C ± 3 °C | NA | NA | 37 °C | NA | NA | |||
| Shi et al[77] | 38 °C | PV: 5-15 mL/minute. PV and HA flow ratio 3:1 | PV: 8-10 mmHg. HA: 90-100 mmHg | ||||||
| Von Horn et al[63] | 8 °C | NA | 5 mmHg | 37 °C | 3 mL/g minute | NA | |||
| Zhou et al[82] | NA | NA | NA | NA | NA | NA | |||
| Luo et al[83] | 4 °C | 1.2 mL/minute | NA | 35-37 °C | 2 mL/g/minute | NA | |||
| Ohara et al[64] | 37 °C | PV: 1 mL/minute/g liver. HA: 0.25 mL/minute/g liver | NA | ||||||
| Chen et al[65] | 37 °C | 1 mL/minute | 7-10 mmHg | ||||||
| Fukai et al[74] | 7-10 °C | 0.5 mL/minute/g liver | 4-6 cmH2O | 37 °C | NA | 8-12 cmH2O | |||
| Hughes et al[84] | 36-37.5 °C | PV: 1.2 mL/g-liver/minute; HA: 0.2 mL/g-liver/minute | NA | ||||||
| Bai et al[98] | NA | NA | NA | ||||||
| Von Horn et al[66] | Risen from 8 to 35 °C | 5.5 ± 1.4 mL/g/minute or 5.2 ± 1.1 mL/g/minute | Risen from 3 mmHg to 6 mmHg | 37 °C | 3 mL/g/minute | NA | |||
| Li et al[87] | 37 °C | Pressure dependent | < 4 mmHg | ||||||
Perfusion applied via PV exhibits varying flow settings depending on the type of MP employed: In HMP, the potential flow settings range from 0.1 mL/minute/g to 0.5 mL/minute/g liver; In SNMP, the range extends from 1 mL/minute/g to 12 mL/minute/g liver; And in NMP, the range spans from 1 mL/minute/g to 30 mL/minute/g liver. Another feasible approach is to tailor the flow based on targeted pressure levels[29,44,46,94]. However, HA flow and pressure descriptions are less common, as the majority of authors prefer single-vessel MP. In NMP, HA flow settings range from 4 mL/minute to 6 mL/minute or from 0.1 mL/minute/g to 0.25 mL/minute/g liver weight, while in SNMP, it is suggested to be 0.1 mL/g of liver/minute. Some authors propose setting PV and HA flow rates in a 3:1 ratio[77].
As there are three main types of MP, literature reveals significant inconsistency regarding the specific temperatures associated with these terms[13]. Most authors perform HMP at 4 °C, though some use lower temperatures between 0 °C and 4 °C, and a few employ temperatures as high as 12 °C. NMP is generally conducted at 37 °C, with slight variations of up to 2 degrees in some studies. SNMP exhibits the most diverse temperature settings, ranging from 20 °C to 35 °C. Table 5 presents a range of temperature settings employed in small animal HMP, SNMP, and NMP.
Over the past two decades, MP has been considered a significant advancement in the field of transplantation[110]. While it has shown superiority over standard SCS and the potential to enhance suboptimal livers, many of its possible applications remain unexplored. Utilizing small animal liver MP presents an excellent opportunity to investigate these potential roles. Rat liver MP is an ideal model for investigating possible MP applications due to its lower costs compared to large animal studies[49]. In rats, around 10% of the liver’s blood supply is arterial and 90% is venous, compared to larger animals. Most literature suggests that perfusion through the PV alone is sufficient and that LTx can be done without reconstructing the HA. Moreover, rat liver explant surgery and transplantation operating times can be under 1 hour, making the process faster, cheaper, and easier compared to large animals. Additionally, the wide availability of inbred rat strains allows researchers to avoid immunological compatibility issues during MP and transplantation[19].
For liver explant surgeries in experimental animals, ensuring proper anesthesia is crucial for maintaining the welfare and stability of the subjects throughout the procedure. Researchers employ various anesthesia protocols tailored to meet experimental requirements and ethical considerations. One common approach involves isoflurane inhalation for sedation, combined with subcutaneous administration of analgesics[21,44,71,76,90]. Alternatively, some researchers opt for intramuscular injection of ketamine hydrochloride (90 mg/kg) and xylazine (10 mg/kg) to induce anesthesia[16,22,24,26]. Conversely, other authors utilize intraperitoneal administration of anesthetics such as sodium pentobarbital at different dosages ranging from 20 mg/kg to 60 mg/kg[35,43,52,82,89,95], or chloral hydrate at varying concentrations (5% 10 mL/kg or 10% 3 mL/kg or 500 mg/kg)[41,55,93]. Each method aims to ensure adequate sedation and pain management for the animals undergoing liver explant surgery, thereby facilitating a successful experimental procedure with minimal discomfort or distress to the subjects.
The first description of rat orthotopic LTx dates back to 1979 and has since remained the gold standard, despite its complexity and long learning curve[111,112]. Although various alternative techniques were proposed to simplify the surgery and make it easier to learn, liver explant surgery remains complex and demands meticulous training[99,113,114]. In the study by Tolboom et al[19], a surgeon with prior experience of over 100 orthotopic liver transplants in rats, performed all surgeries. Specific expertise in liver surgery, prior training, and a deep understanding of the procedure are crucial for successful outcomes[115]. Unfortunately, many studies lack detailed descriptions of liver explant surgery and information about surgeons’ training backgrounds.
Currently, no studies have definitively analyzed which MP setup closed or open circuit is superior. Both configurations are shown to be effective in preserving liver function according to the articles reviewed in this study. A closed circuit, while requiring additional cannulation and precise surgery to prevent perfusate leakage and ensure continuous perfusion, may offer certain advantages. In contrast, an open circuit simplifies the procedure by not requiring VC cannulation. Dual vessel MP, involving both the PV and HA, is generally superior to PV-only perfusion, as it better supports the vascularization of the biliary tree and ensures adequate oxygen delivery[64]. However, it is also more expensive and necessitates advanced techniques to avoid damage to arterial intima[106]. Some authors suggest that single-vessel PV or retrograde perfusion is sufficient for liver preservation. Perfusion via the HA alone is less advantageous because this artery supplies roughly one-tenth of the liver[67]. Dual vessel MP offers superior outcomes but at a higher cost and complexity, whereas single-vessel PV or retrograde perfusion can be sufficient in many cases. Bile duct cannulation is essential and should always be performed, as it allows for the measurement of bile output and the assessment of bile composition, which are critical markers of biliary viability[107].
The heterogeneity of MP setups makes it difficult to select the best one. Key considerations in setting up the apparatus include: Ensuring the organ chamber accommodates the liver position without bending its edges and remains close to physiological conditions, positioning cannulas connected to the tubing close to the anatomical position of vessels, ensuring adequate oxygenation and temperature control, utilizing pumps and sensitive sensors for necessary flow and pressure adjustments, and incorporating bubble traps into the system to prevent air embolisms. We recommend avoiding excessively large tubing or perfusate reservoirs, as miniaturization is crucial for the successful establishment of small animal MP[70].
The choice of perfusate is as critical as the setup of the perfusion apparatus. Compositions and volumes of perfusates proposed by different authors vary significantly, yet few studies have examined the relative superiority of one perfusate over another[17,21]. Future research should focus on identifying the optimal perfusate composition. Regardless of the chosen perfusate composition, we recommend adjusting the perfusate potential of hydrogen (pH) to the normal range before connecting the liver to the MP circuit. Additionally, mild acidosis of the perfusate is believed to enhance cytoprotection during hypothermia, which can be considered an added advantage[17]. Therefore, it is advisable to maintain the perfusate within the normal pH range and avoid alkalosis.
HMP and SNMP do not require the addition of red blood cells or hourly supplementation of the perfusate because liver metabolic activity is suppressed and the risk of clotting and hemolysis is higher at lower temperatures[109]. During NMP, supplementation with red blood cells or other oxygen carriers is crucial for adequate oxygen delivery to the cells[116]. Additionally, during NMP, consideration should be given to hourly supplementation with heparin, insulin, amino acids, or other additives throughout the entire perfusion process as at 37 °C the liver is metabolically active and should be provided with substances similar to in vivo conditions[19].
This review highlights the current state of small animal liver MP systems, emphasizing their benefits and challenges in experimental research. Rat liver MP models offer a cost-effective and accessible alternative to larger animal studies, with simplified perfusion processes and shorter operative times, making them valuable for LTx and preservation research. However, significant challenges remain, particularly in dual-vessel perfusion, which enhances vascularization and oxygenation but requires advanced surgical skills and additional resources. Furthermore, the lack of a standardized protocol limits reproducibility across studies. Maintaining adequate perfusate volume is another challenge, as the small blood volume of rats often falls short of the fluid requirements of the perfusion apparatus. Closed-circuit systems may improve preservation through continuous perfusion but require meticulous handling, while open-circuit setups simplify procedures at the cost of reduced control over perfusion parameters. Likewise, perfusate composition remains an area of ongoing investigation, as factors such as red blood cell content, heparin use, and pH adjustments significantly impact liver viability, particularly in NMP perfusion. In conclusion, while dual-vessel closed-circuit MP offers superior physiological outcomes, single-vessel and open-circuit approaches may still be suitable for many studies, depending on research objectives. Future efforts should focus on optimizing perfusate composition and refining MP setups to improve reproducibility and minimize animal use. Establishing a standardized protocol will be crucial for advancing small animal MP research, facilitating faster implementation, and reducing the number of animals required for experimentation. We hope this review serves as a valuable resource for researchers seeking to streamline their MP protocols and enhance experimental efficiency.
| 1. | Chotai P, Matsuoka L. Reassessing the role of liver transplantation for patients with metastatic colorectal cancer to the liver. Curr Opin Organ Transplant. 2019;24:118-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 2. | Tingle SJ, Dobbins JJ, Thompson ER, Figueiredo RS, Mahendran B, Pandanaboyana S, Wilson C. Machine perfusion in liver transplantation. Cochrane Database Syst Rev. 2023;9:CD014685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 3. | Widmer J, Eden J, Carvalho MF, Dutkowski P, Schlegel A. Machine Perfusion for Extended Criteria Donor Livers: What Challenges Remain? J Clin Med. 2022;11:5218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 4. | Goutard M, de Vries RJ, Tawa P, Pendexter CA, Rosales IA, Tessier SN, Burlage LC, Lantieri L, Randolph MA, Lellouch AG, Cetrulo CL Jr, Uygun K. Exceeding the Limits of Static Cold Storage in Limb Transplantation Using Subnormothermic Machine Perfusion. J Reconstr Microsurg. 2023;39:350-360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 5. | Nasralla D, Coussios CC, Mergental H, Akhtar MZ, Butler AJ, Ceresa CDL, Chiocchia V, Dutton SJ, García-Valdecasas JC, Heaton N, Imber C, Jassem W, Jochmans I, Karani J, Knight SR, Kocabayoglu P, Malagò M, Mirza D, Morris PJ, Pallan A, Paul A, Pavel M, Perera MTPR, Pirenne J, Ravikumar R, Russell L, Upponi S, Watson CJE, Weissenbacher A, Ploeg RJ, Friend PJ; Consortium for Organ Preservation in Europe. A randomized trial of normothermic preservation in liver transplantation. Nature. 2018;557:50-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1064] [Cited by in RCA: 945] [Article Influence: 118.1] [Reference Citation Analysis (2)] |
| 6. | de Meijer VE, Fujiyoshi M, Porte RJ. Ex situ machine perfusion strategies in liver transplantation. J Hepatol. 2019;70:203-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 7. | Czigany Z, Pratschke J, Froněk J, Guba M, Schöning W, Raptis DA, Andrassy J, Kramer M, Strnad P, Tolba RH, Liu W, Keller T, Miller H, Pavicevic S, Uluk D, Kocik M, Lurje I, Trautwein C, Mehrabi A, Popescu I, Vondran FWR, Ju C, Tacke F, Neumann UP, Lurje G. Hypothermic Oxygenated Machine Perfusion Reduces Early Allograft Injury and Improves Post-transplant Outcomes in Extended Criteria Donation Liver Transplantation From Donation After Brain Death: Results From a Multicenter Randomized Controlled Trial (HOPE ECD-DBD). Ann Surg. 2021;274:705-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 194] [Article Influence: 38.8] [Reference Citation Analysis (1)] |
| 8. | Lascaris B, de Meijer VE, Porte RJ. Normothermic liver machine perfusion as a dynamic platform for regenerative purposes: What does the future have in store for us? J Hepatol. 2022;77:825-836. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 50] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 9. | Boteon YL, Hessheimer AJ, Brüggenwirth IMA, Boteon APCS, Padilla M, de Meijer VE, Domínguez-Gil B, Porte RJ, Perera MTPR, Martins PN. The economic impact of machine perfusion technology in liver transplantation. Artif Organs. 2022;46:191-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 10. | Muth V, Gassner JMGV, Moosburner S, Lurje G, Michelotto J, Strobl F, Knaub K, Engelmann C, Tacke F, Selzner M, Pratschke J, Sauer IM, Raschzok N. Ex Vivo Liver Machine Perfusion: Comprehensive Review of Common Animal Models. Tissue Eng Part B Rev. 2023;29:10-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 11. | von Horn C, Hannaert P, Hauet T, Leuvenink H, Paul A, Minor T; COPE consortium partners. Cold flush after dynamic liver preservation protects against ischemic changes upon reperfusion - an experimental study. Transpl Int. 2019;32:218-224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 12. | Fabry G, Doorschodt BM, Grzanna T, Boor P, Elliott A, Stollenwerk A, Tolba RH, Rossaint R, Bleilevens C. Cold Preflush of Porcine Kidney Grafts Prior to Normothermic Machine Perfusion Aggravates Ischemia Reperfusion Injury. Sci Rep. 2019;9:13897. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 13. | Karangwa SA, Dutkowski P, Fontes P, Friend PJ, Guarrera JV, Markmann JF, Mergental H, Minor T, Quintini C, Selzner M, Uygun K, Watson CJ, Porte RJ. Machine Perfusion of Donor Livers for Transplantation: A Proposal for Standardized Nomenclature and Reporting Guidelines. Am J Transplant. 2016;16:2932-2942. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 128] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 14. | Kim JS, Boudjema K, D'Alessandro A, Southard JH. Machine perfusion of the liver: maintenance of mitochondrial function after 48-hour preservation. Transplant Proc. 1997;29:3452-3454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 15. | Dutkowski P, Odermatt B, Heinrich T, Schönfeld S, Watzka M, Winkelbach V, Krysiak M, Junginger T. Hypothermic oscillating liver perfusion stimulates ATP synthesis prior to transplantation. J Surg Res. 1998;80:365-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 72] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 16. | Lauschke H, Olschewski P, Tolba R, Schulz S, Minor T. Oxygenated machine perfusion mitigates surface antigen expression and improves preservation of predamaged donor livers. Cryobiology. 2003;46:53-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | Bessems M, Doorschodt BM, van Vliet AK, van Gulik TM. Improved rat liver preservation by hypothermic continuous machine perfusion using polysol, a new, enriched preservation solution. Liver Transpl. 2005;11:539-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 52] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 18. | Dutkowski P, Graf R, Clavien PA. Rescue of the cold preserved rat liver by hypothermic oxygenated machine perfusion. Am J Transplant. 2006;6:903-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 93] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 19. | Tolboom H, Pouw R, Uygun K, Tanimura Y, Izamis ML, Berthiaume F, Yarmush ML. A model for normothermic preservation of the rat liver. Tissue Eng. 2007;13:2143-2151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 43] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 20. | Vairetti M, Ferrigno A, Rizzo V, Boncompagni E, Carraro A, Gringeri E, Milanesi G, Barni S, Freitas I, Cillo U. Correlation between the liver temperature employed during machine perfusion and reperfusion damage: role of Ca2+. Liver Transpl. 2008;14:494-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 21. | Manekeller S, Schuppius A, Stegemann J, Hirner A, Minor T. Role of perfusion medium, oxygen and rheology for endoplasmic reticulum stress-induced cell death after hypothermic machine preservation of the liver. Transpl Int. 2008;21:169-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Stegemann J, Hirner A, Rauen U, Minor T. Gaseous oxygen persufflation or oxygenated machine perfusion with Custodiol-N for long-term preservation of ischemic rat livers? Cryobiology. 2009;58:45-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 23. | Ferrigno A, Carlucci F, Tabucchi A, Tommassini V, Rizzo V, Richelmi P, Gringeri E, Neri D, Boncompagni E, Freitas I, Cillo U, Vairetti M. Different susceptibility of liver grafts from lean and obese Zucker rats to preservation injury. Cryobiology. 2009;59:327-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 24. | Lüer B, Koetting M, Efferz P, Minor T. Role of oxygen during hypothermic machine perfusion preservation of the liver. Transpl Int. 2010;23:944-950. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 25. | Olschewski P, Gass P, Ariyakhagorn V, Jasse K, Hunold G, Menzel M, Schöning W, Schmitz V, Neuhaus P, Puhl G. The influence of storage temperature during machine perfusion on preservation quality of marginal donor livers. Cryobiology. 2010;60:337-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 71] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 26. | Minor T, Lüer B, Efferz P. Dopamine improves hypothermic machine preservation of the liver. Cryobiology. 2011;63:84-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 27. | Tolboom H, Izamis ML, Sharma N, Milwid JM, Uygun B, Berthiaume F, Uygun K, Yarmush ML. Subnormothermic machine perfusion at both 20°C and 30°C recovers ischemic rat livers for successful transplantation. J Surg Res. 2012;175:149-156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 28. | Giannone FA, Treré D, Domenicali M, Grattagliano I, Baracca A, Sgarbi G, Maggioli C, Longobardi P, Solaini G, Derenzini M, Bernardi M, Caraceni P. An innovative hyperbaric hypothermic machine perfusion protects the liver from experimental preservation injury. ScientificWorldJournal. 2012;2012:573410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 29. | Perk S, Izamis ML, Tolboom H, Uygun B, Yarmush ML, Uygun K. A fitness index for transplantation of machine-perfused cadaveric rat livers. BMC Res Notes. 2012;5:325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 30. | Schlegel A, Graf R, Clavien PA, Dutkowski P. Hypothermic oxygenated perfusion (HOPE) protects from biliary injury in a rodent model of DCD liver transplantation. J Hepatol. 2013;59:984-991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 146] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 31. | Liu Q, Berendsen T, Izamis ML, Uygun B, Yarmush ML, Uygun K. Perfusion defatting at subnormothermic temperatures in steatotic rat livers. Transplant Proc. 2013;45:3209-3213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 32. | Schlegel A, Kron P, Graf R, Clavien PA, Dutkowski P. Hypothermic Oxygenated Perfusion (HOPE) downregulates the immune response in a rat model of liver transplantation. Ann Surg. 2014;260:931-7; discussion 937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 110] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 33. | Schlegel A, Kron P, Graf R, Dutkowski P, Clavien PA. Warm vs. cold perfusion techniques to rescue rodent liver grafts. J Hepatol. 2014;61:1267-1275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 140] [Article Influence: 11.7] [Reference Citation Analysis (3)] |
| 34. | Bae C, Pichardo EM, Huang H, Henry SD, Guarrera JV. The benefits of hypothermic machine perfusion are enhanced with Vasosol and α-tocopherol in rodent donation after cardiac death livers. Transplant Proc. 2014;46:1560-1566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 35. | Tarantola E, Bertone V, Milanesi G, Gruppi C, Ferrigno A, Vairetti M, Barni S, Freitas I. Dipeptidylpeptidase-IV activity and expression reveal decreased damage to the intrahepatic biliary tree in fatty livers submitted to subnormothermic machine-perfusion respect to conventional cold storage. Eur J Histochem. 2014;58:2414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 36. | Bruinsma BG, Berendsen TA, Izamis ML, Yeh H, Yarmush ML, Uygun K. Supercooling preservation and transplantation of the rat liver. Nat Protoc. 2015;10:484-494. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 76] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 37. | Ferrigno A, Di Pasqua LG, Bianchi A, Richelmi P, Vairetti M. Metabolic shift in liver: correlation between perfusion temperature and hypoxia inducible factor-1α. World J Gastroenterol. 2015;21:1108-1116. [PubMed] [DOI] [Full Text] |
| 38. | Jia JJ, Zhang J, Li JH, Chen XD, Jiang L, Zhou YF, He N, Xie HY, Zhou L, Zheng SS. Influence of perfusate on liver viability during hypothermic machine perfusion. World J Gastroenterol. 2015;21:8848-8857. [PubMed] [DOI] [Full Text] |
| 39. | Okamura Y, Hata K, Tanaka H, Hirao H, Kubota T, Inamoto O, Kageyama S, Tamaki I, Yermek N, Yoshikawa J, Uemoto S. Impact of Subnormothermic Machine Perfusion Preservation in Severely Steatotic Rat Livers: A Detailed Assessment in an Isolated Setting. Am J Transplant. 2017;17:1204-1215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 40. | Berardo C, Di Pasqua LG, Siciliano V, Rizzo V, Richelmi P, Ferrigno A, Vairetti M. Machine Perfusion at 20°C Prevents Ischemic Injury and Reduces Hypoxia-Inducible Factor-1α Expression During Rat Liver Preservation. Ann Transplant. 2017;22:581-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 41. | Zeng C, Hu X, He W, Wang Y, Li L, Xiong Y, Ye Q. Hypothermic machine perfusion ameliorates inflammation during ischemia-reperfusion injury via sirtuin-1-mediated deacetylation of nuclear factor-κB p65 in rat livers donated after circulatory death. Mol Med Rep. 2017;16:8649-8656. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 42. | Beal EW, Dumond C, Kim J, Akateh C, Eren E, Maynard K, Sen CK, Zweier JL, Washburn K, Whitson BA, Black SM. A Small Animal Model of Ex Vivo Normothermic Liver Perfusion. J Vis Exp. 2018;136:57541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 43. | Xue S, He W, Zeng X, Tang Z, Feng S, Zhong Z, Xiong Y, Wang Y, Ye Q. Hypothermic machine perfusion attenuates ischemia/reperfusion injury against rat livers donated after cardiac death by activating the Keap1/Nrf2-ARE signaling pathway. Mol Med Rep. 2018;18:815-826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 44. | Oldani G, Peloso A, Slits F, Gex Q, Delaune V, Orci LA, van de Looij Y, Colin DJ, Germain S, de Vito C, Rubbia-Brandt L, Lacotte S, Toso C. The impact of short-term machine perfusion on the risk of cancer recurrence after rat liver transplantation with donors after circulatory death. PLoS One. 2019;14:e0224890. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 45. | Chin LY, Carroll C, Raigani S, Detelich DM, Tessier SN, Wojtkiewicz GR, Schmidt SP, Weissleder R, Yeh H, Uygun K, Parekkadan B. Ex vivo perfusion-based engraftment of genetically engineered cell sensors into transplantable organs. PLoS One. 2019;14:e0225222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 46. | Gillooly AR, Perry J, Martins PN. First Report of siRNA Uptake (for RNA Interference) During Ex Vivo Hypothermic and Normothermic Liver Machine Perfusion. Transplantation. 2019;103:e56-e57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 47. | Martins RM, Teodoro JS, Furtado E, Oliveira RC, Tralhão JG, Rolo AP, Palmeira CM. Mild hypothermia during the reperfusion phase protects mitochondrial bioenergetics against ischemia-reperfusion injury in an animal model of ex-vivo liver transplantation-an experimental study. Int J Med Sci. 2019;16:1304-1312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 48. | Scheuermann U, Zhu M, Song M, Yerxa J, Gao Q, Davis RP, Zhang M, Parker W, Hartwig MG, Kwun J, Brennan TV, Lee J, Barbas AS. Damage-Associated Molecular Patterns Induce Inflammatory Injury During Machine Preservation of the Liver: Potential Targets to Enhance a Promising Technology. Liver Transpl. 2019;25:610-626. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 49. | Haque O, Pendexter CA, Cronin SEJ, Raigani S, de Vries RJ, Yeh H, Markmann JF, Uygun K. Twenty-four hour ex-vivo normothermic machine perfusion in rat livers. Technology (Singap World Sci). 2020;8:27-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 50. | Schlegel A, Muller X, Mueller M, Stepanova A, Kron P, de Rougemont O, Muiesan P, Clavien PA, Galkin A, Meierhofer D, Dutkowski P. Hypothermic oxygenated perfusion protects from mitochondrial injury before liver transplantation. EBioMedicine. 2020;60:103014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 213] [Cited by in RCA: 191] [Article Influence: 31.8] [Reference Citation Analysis (4)] |
| 51. | Nösser M, Gassner JMGV, Moosburner S, Wyrwal D, Claussen F, Hillebrandt KH, Horner R, Tang P, Reutzel-Selke A, Polenz D, Arsenic R, Pratschke J, Sauer IM, Raschzok N. Development of a Rat Liver Machine Perfusion System for Normothermic and Subnormothermic Conditions. Tissue Eng Part A. 2020;26:57-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 52. | Hu X, Wang W, Zeng C, He W, Zhong Z, Liu Z, Wang Y, Ye Q. Appropriate timing for hypothermic machine perfusion to preserve livers donated after circulatory death. Mol Med Rep. 2020;22:2003-2011. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 53. | von Horn C, Minor T. Transient hyperthermia during oxygenated rewarming of isolated rat livers. Transpl Int. 2020;33:272-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 54. | Raigani S, Carroll C, Griffith S, Pendexter C, Rosales I, Deirawan H, Beydoun R, Yarmush M, Uygun K, Yeh H. Improvement of steatotic rat liver function with a defatting cocktail during ex situ normothermic machine perfusion is not directly related to liver fat content. PLoS One. 2020;15:e0232886. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 55. | Yang L, Cao H, Sun D, Lin L, Zheng WP, Shen ZY, Song HL. Normothermic Machine Perfusion Combined with Bone Marrow Mesenchymal Stem Cells Improves the Oxidative Stress Response and Mitochondrial Function in Rat Donation After Circulatory Death Livers. Stem Cells Dev. 2020;29:835-852. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 56. | Lin F, Zhen F, Yan X, Shaojun Y, Guizhu P, Yanfeng W, Qifa Y. Hypothermic oxygenated perfusion with defatting cocktail further improves steatotic liver grafts in a transplantation rat model. Artif Organs. 2021;45:E304-E316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 57. | Rigo F, Navarro-Tableros V, De Stefano N, Calleri A, Romagnoli R. Ex Vivo Normothermic Hypoxic Rat Liver Perfusion Model: An Experimental Setting for Organ Recondition and Pharmacological Intervention. Methods Mol Biol. 2021;2269:139-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 58. | Carlson KN, Pavan-Guimaraes J, Verhagen JC, Chlebeck P, Verhoven B, Jennings H, Najmabadi F, Liu Y, Burlingham W, Capitini CM, Al-Adra DP. Interleukin-10 and Transforming Growth Factor-β Cytokines Decrease Immune Activation During Normothermic Ex Vivo Machine Perfusion of the Rat Liver. Liver Transpl. 2021;27:1577-1591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 59. | Cao H, Wu L, Tian X, Zheng W, Yuan M, Li X, Tian X, Wang Y, Song H, Shen Z. HO-1/BMMSC perfusion using a normothermic machine perfusion system reduces the acute rejection of DCD liver transplantation by regulating NKT cell co-inhibitory receptors in rats. Stem Cell Res Ther. 2021;12:587. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 60. | De Stefano N, Navarro-Tableros V, Roggio D, Calleri A, Rigo F, David E, Gambella A, Bassino D, Amoroso A, Patrono D, Camussi G, Romagnoli R. Human liver stem cell-derived extracellular vesicles reduce injury in a model of normothermic machine perfusion of rat livers previously exposed to a prolonged warm ischemia. Transpl Int. 2021;34:1607-1617. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 61. | Sun D, Yang L, Zheng W, Cao H, Wu L, Song H. Protective Effects of Bone Marrow Mesenchymal Stem Cells (BMMSCS) Combined with Normothermic Machine Perfusion on Liver Grafts Donated After Circulatory Death via Reducing the Ferroptosis of Hepatocytes. Med Sci Monit. 2021;27:e930258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 62. | Jennings H, Carlson KN, Little C, Verhagen JC, Nagendran J, Liu Y, Verhoven B, Zeng W, McMorrow S, Chlebeck P, Al-Adra DP. The Immunological Effect of Oxygen Carriers on Normothermic Ex Vivo Liver Perfusion. Front Immunol. 2022;13:833243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 63. | von Horn C, Zlatev H, Pletz J, Lüer B, Minor T. Comparison of thermal variations in post-retrieval graft conditioning on rat livers. Artif Organs. 2022;46:239-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 64. | Ohara M, Ishikawa J, Yoshimoto S, Hakamata Y, Kobayashi E. A rat model of dual-flow liver machine perfusion system. Acta Cir Bras. 2023;38:e387723. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 65. | Chen H, Dirsch O, Albadry M, Ana PH, Dahmen U. Normothermic Ex Vivo Liver Machine Perfusion in Mouse. J Vis Exp. 2023;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 66. | von Horn C, Lüer B, Malkus L, Minor T. Role of perfusion medium in rewarming machine perfusion from hypo- to normothermia. Artif Organs. 2024;48:150-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 67. | Compagnon P, Clément B, Campion JP, Boudjema K. Effects of hypothermic machine perfusion on rat liver function depending on the route of perfusion. Transplantation. 2001;72:606-614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 68. | Niu X, Huang WH, De Boer B, Delriviere L, Mou LJ, Jeffrey GP. Iron-induced oxidative rat liver injury after non-heart-beating warm ischemia is mediated by tumor necrosis factor α and prevented by deferoxamine. Liver Transpl. 2014;20:904-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 69. | Tabka D, Bejaoui M, Javellaud J, Achard JM, Ben Abdennebi H. Angiotensin IV improves subnormothermic machine perfusion preservation of rat liver graft. Biomed Pharmacother. 2018;104:841-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 70. | Gassner JMGV, Nösser M, Moosburner S, Horner R, Tang P, Wegener L, Wyrwal D, Claussen F, Arsenic R, Pratschke J, Sauer IM, Raschzok N. Improvement of Normothermic Ex Vivo Machine Perfusion of Rat Liver Grafts by Dialysis and Kupffer Cell Inhibition With Glycine. Liver Transpl. 2019;25:275-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 71. | Claussen F, Gassner JMGV, Moosburner S, Wyrwal D, Nösser M, Tang P, Wegener L, Pohl J, Reutzel-Selke A, Arsenic R, Pratschke J, Sauer IM, Raschzok N. Dual versus single vessel normothermic ex vivo perfusion of rat liver grafts using metamizole for vasodilatation. PLoS One. 2020;15:e0235635. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 72. | Wang S, Zeng X, Yang Y, Li S, Wang Y, Ye Q, Fan X. Hypothermic oxygenated perfusion ameliorates ischemia-reperfusion injury of fatty liver in mice via Brg1/Nrf2/HO-1 axis. Artif Organs. 2022;46:229-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 73. | Asong-Fontem N, Panisello-Rosello A, Sebagh M, Gonin M, Rosello-Catafau J, Adam R. The Role of IGL-2 Preservation Solution on Rat Livers during SCS and HOPE. Int J Mol Sci. 2022;23:12615. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 74. | Fukai M, Sakamoto S, Bochimoto H, Zin NKM, Shibata K, Ishikawa T, Shimada S, Kawamura N, Fujiyoshi M, Fujiyoshi S, Nakamura K, Shimamura T, Taketomi A. Hypothermic Machine Perfusion with Hydrogen Gas Reduces Focal Injury in Rat Livers but Fails to Restore Organ Function. Transplant Proc. 2023;55:1016-1020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 75. | Op den Dries S, Karimian N, Westerkamp AC, Sutton ME, Kuipers M, Wiersema-Buist J, Ottens PJ, Kuipers J, Giepmans BN, Leuvenink HG, Lisman T, Porte RJ. Normothermic machine perfusion reduces bile duct injury and improves biliary epithelial function in rat donor livers. Liver Transpl. 2016;22:994-1005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 57] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 76. | Westerkamp AC, Fujiyoshi M, Ottens PJ, Nijsten MWN, Touw DJ, de Meijer VE, Lisman T, Leuvenink HGD, Moshage H, Berendsen TA, Porte RJ. Metformin Preconditioning Improves Hepatobiliary Function and Reduces Injury in a Rat Model of Normothermic Machine Perfusion and Orthotopic Transplantation. Transplantation. 2020;104:e271-e280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 77. | Shi JH, Yang DJ, Jin Q, Cheng N, Shi YB, Bai Y, Yu DS, Guo WZ, Ge GB, Zhang SJ. Cytochrome P450 2E1 predicts liver functional recovery from donation after circulatory death using air-ventilated normothermic machine perfusion. Sci Rep. 2022;12:7446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 78. | He N, Jia JJ, Xie HY, Li JH, He Y, Yin SY, Liang RP, Jiang L, Liu JF, Xu KD, Zhang ZH, Zhou L, Zheng SS. Partial Inhibition of HO-1 Attenuates HMP-Induced Hepatic Regeneration against Liver Injury in Rats. Oxid Med Cell Longev. 2018;2018:9108483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 79. | Zeng X, Wang S, Li S, Yang Y, Fang Z, Huang H, Wang Y, Fan X, Ye Q. Hypothermic oxygenated machine perfusion alleviates liver injury in donation after circulatory death through activating autophagy in mice. Artif Organs. 2019;43:E320-E332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 80. | Jia JJ, Xie HY, Li JH, He Y, Jiang L, He N, Zhou L, Wang W, Zheng SS. Graft protection of the liver by hypothermic machine perfusion involves recovery of graft regeneration in rats. J Int Med Res. 2019;47:427-437. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 81. | Zhou W, Zhong Z, Lin D, Liu Z, Zhang Q, Xia H, Peng S, Liu A, Lu Z, Wang Y, Ye S, Ye Q. Hypothermic oxygenated perfusion inhibits HECTD3-mediated TRAF3 polyubiquitination to alleviate DCD liver ischemia-reperfusion injury. Cell Death Dis. 2021;12:211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 82. | Zhou W, Peng S, Du P, Zhou P, Xue C, Ye Q. Hypothermic oxygenated perfusion combined with TJ-M2010-5 alleviates hepatic ischemia-reperfusion injury in donation after circulatory death. Int Immunopharmacol. 2022;105:108541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 83. | Luo J, Hu Y, Qiao Y, Li H, Huang J, Xu K, Jiang L, Wu H, Hu X, Jia J, Zhou L, Xie H, Li J, Zheng S. Hypothermic Oxygenated Machine Perfusion Promotes Mitophagy Flux against Hypoxia-Ischemic Injury in Rat DCD Liver. Int J Mol Sci. 2023;24:5403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 84. | Hughes CB, Nigmet Y, Villanueva FS, Chen X, Demetris AJ, Stolz DB, Pacella JJ, Humar A. Ultrasound-Targeted Microbubble Cavitation During Machine Perfusion Reduces Microvascular Thrombi and Graft Injury in a Rat Liver Model of Donation After Circulatory Death. Transplant Proc. 2023;55:485-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 85. | Liu W, Fan Y, Ding H, Han D, Yan Y, Wu R, Lv Y, Zheng X. Normothermic machine perfusion attenuates hepatic ischaemia-reperfusion injury by inhibiting CIRP-mediated oxidative stress and mitochondrial fission. J Cell Mol Med. 2021;25:11310-11321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 86. | de Vries RJ, Pendexter CA, Cronin SEJ, Marques B, Hafiz EOA, Muzikansky A, van Gulik TM, Markmann JF, Stott SL, Yeh H, Toner M, Uygun K, Tessier SN. Cell release during perfusion reflects cold ischemic injury in rat livers. Sci Rep. 2020;10:1102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 87. | Li S, Zhi Y, Mu W, Li M, Lv G. Exploring the effects of epigallocatechin gallate on lipid metabolism in the rat steatotic liver during normothermic machine perfusion: Insights from lipidomics and RNA sequencing. Eur J Pharmacol. 2024;964:176300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 88. | Bruinsma BG, Berendsen TA, Izamis ML, Yarmush ML, Uygun K. Determination and extension of the limits to static cold storage using subnormothermic machine perfusion. Int J Artif Organs. 2013;36:775-780. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 89. | Chai YC, Dang GX, He HQ, Shi JH, Zhang HK, Zhang RT, Wang B, Hu LS, Lv Y. Hypothermic machine perfusion with metformin-University of Wisconsin solution for ex vivo preservation of standard and marginal liver grafts in a rat model. World J Gastroenterol. 2017;23:7221-7231. [PubMed] [DOI] [Full Text] |
| 90. | Lee CY, Jain S, Duncan HM, Zhang JX, Jones JW Jr, Southard JH, Clemens MG. Survival transplantation of preserved non-heart-beating donor rat livers: preservation by hypothermic machine perfusion. Transplantation. 2003;76:1432-1436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 63] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 91. | Tan XD, Egami H, Wang FS, Ogawa M. Protective effect of exogenous adenosine triphosphate on hypothermically preserved rat liver. World J Gastroenterol. 2004;10:871-874. [PubMed] [DOI] [Full Text] |
| 92. | Xu H, Zhang JX, Jones JW, Southard JH, Clemens MG, Lee CY. Hypothermic machine perfusion of rat livers preserves endothelial cell function. Transplant Proc. 2005;37:335-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 93. | Carnevale ME, Balaban CL, Guibert EE, Bottai H, Rodriguez JV. Hypothermic machine perfusion versus cold storage in the rescuing of livers from non-heart-beating donor rats. Artif Organs. 2013;37:985-991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 94. | Westerkamp AC, Mahboub P, Meyer SL, Hottenrott M, Ottens PJ, Wiersema-Buist J, Gouw AS, Lisman T, Leuvenink HG, Porte RJ. End-ischemic machine perfusion reduces bile duct injury in donation after circulatory death rat donor livers independent of the machine perfusion temperature. Liver Transpl. 2015;21:1300-1311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 95. | Carbonell T, Alva N, Sanchez-Nuño S, Dewey S, Gomes AV. Subnormothermic Perfusion in the Isolated Rat Liver Preserves the Antioxidant Glutathione and Enhances the Function of the Ubiquitin Proteasome System. Oxid Med Cell Longev. 2016;2016:9324692. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 96. | Yamada S, Miyagi S, Hara Y, Kakizaki Y, Sasajima H, Mitsui K, Fujimori K, Unno M, Kamei T, Goto M. Effects of Short-Term Normothermic and Subnormothermic Perfusion After Cold Preservation on Liver Transplantation From Donors After Cardiac Death. Transplant Proc. 2020;52:1639-1642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 97. | Xu M, Zhou F, Ahmed O, Upadhya GA, Jia J, Lee C, Xing J, Ye L, Shim SH, Zhang Z, Byrnes K, Wong B, Kim JS, Lin Y, Chapman WC. A Novel Multidrug Combination Mitigates Rat Liver Steatosis Through Activating AMPK Pathway During Normothermic Machine Perfusion. Transplantation. 2021;105:e215-e225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 98. | Bai Y, Shi JH, Liu Q, Yang DJ, Yan ZP, Zhang JK, Tang HW, Guo WZ, Jin Y, Zhang SJ. Charged multivesicular body protein 2B ameliorates biliary injury in the liver from donation after cardiac death rats via autophagy with air-oxygenated normothermic machine perfusion. Biochim Biophys Acta Mol Basis Dis. 2023;1869:166686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 99. | Kashfi A, Mehrabi A, Pahlavan PS, Schemmer P, Gutt CN, Friess H, Gebhard MM, Schmidt J, Büchler MW, Kraus TW. A review of various techniques of orthotopic liver transplantation in the rat. Transplant Proc. 2005;37:185-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 100. | Yener AÜ, Çiçek MC, Genç SB, Özkan T, Doğan E, Bilgin BC, Akın T, Erdem H, Ankarali H. Protective role of heparin in the injury of the liver and kidney on the experimental model of ischemia/reperfusion. J Cardiothorac Surg. 2014;9:35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 101. | Kamada N, Calne RY. A surgical experience with five hundred thirty liver transplants in the rat. Surgery. 1983;93:64-69. [PubMed] |
| 102. | Capuano G, Rodinò S, D'Agostino L, Budillon G. Evaluation of heparin toxicity in the isolated and perfused rat liver. Enzyme. 1986;35:77-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 103. | Kruepunga N, Hakvoort TBM, Hikspoors JPJM, Köhler SE, Lamers WH. Anatomy of rodent and human livers: What are the differences? Biochim Biophys Acta Mol Basis Dis. 2019;1865:869-878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 104. | Schlegel A, Kron P, De Oliveira ML, Clavien PA, Dutkowski P. Is single portal vein approach sufficient for hypothermic machine perfusion of DCD liver grafts? J Hepatol. 2016;64:239-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 105. | Reichen J. Role of the hepatic artery in canalicular bile formation by the perfused rat liver. A multiple indicator dilution study. J Clin Invest. 1988;81:1462-1469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 106. | Monbaliu D, Brassil J. Machine perfusion of the liver: past, present and future. Curr Opin Organ Transplant. 2010;15:160-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 69] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 107. | de Jong IEM, Bodewes SB, van Leeuwen OB, Oosterhuis D, Lantinga VA, Thorne AM, Lascaris B, van den Heuvel MC, Wells RG, Olinga P, de Meijer VE, Porte RJ. Restoration of Bile Duct Injury of Donor Livers During Ex Situ Normothermic Machine Perfusion. Transplantation. 2023;107:e161-e172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 108. | Ferrigno A, Richelmi P, Vairetti M. Troubleshooting and improving the mouse and rat isolated perfused liver preparation. J Pharmacol Toxicol Methods. 2013;67:107-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 109. | van Beekum CJ, Vilz TO, Glowka TR, von Websky MW, Kalff JC, Manekeller S. Normothermic Machine Perfusion (NMP) of the Liver - Current Status and Future Perspectives. Ann Transplant. 2021;26:e931664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 110. | Martins PN, Buchwald JE, Mergental H, Vargas L, Quintini C. The role of normothermic machine perfusion in liver transplantation. Int J Surg. 2020;82S:52-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 111. | Kamada N, Calne RY. Orthotopic Liver Transplantation in the Rat. Transplantation. 1979;28:47-50. [RCA] [DOI] [Full Text] [Cited by in Crossref: 569] [Cited by in RCA: 527] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 112. | Hori T, Nguyen JH, Zhao X, Ogura Y, Hata T, Yagi S, Chen F, Baine AM, Ohashi N, Eckman CB, Herdt AR, Egawa H, Takada Y, Oike F, Sakamoto S, Kasahara M, Ogawa K, Hata K, Iida T, Yonekawa Y, Sibulesky L, Kuribayashi K, Kato T, Saito K, Wang L, Torii M, Sahara N, Kamo N, Sahara T, Yasutomi M, Uemoto S. Comprehensive and innovative techniques for liver transplantation in rats: a surgical guide. World J Gastroenterol. 2010;16:3120-3132. [PubMed] [DOI] [Full Text] |
| 113. | Oldani G, Lacotte S, Morel P, Mentha G, Toso C. Orthotopic liver transplantation in rats. J Vis Exp. 2012;4143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 114. | Delrivière L, Gibbs P, Kobayashi E, Goto S, Kamada N, Gianello P. Detailed modified technique for safer harvesting and preparation of liver graft in the rat. Microsurgery. 1996;17:690-696. [PubMed] [DOI] [Full Text] |
| 115. | Czigány Z, Iwasaki J, Yagi S, Nagai K, Szijártó A, Uemoto S, Tolba RH. Improving Research Practice in Rat Orthotopic and Partial Orthotopic Liver Transplantation: A Review, Recommendation, and Publication Guide. Eur Surg Res. 2015;55:119-138. [PubMed] [DOI] [Full Text] |
| 116. | Muth V, Strobl F, Michelotto J, Gilles L, Kirwan JA, Eisenberger A, Marchand J, Roschke NN, Moosburner S, Pratschke J, Sauer IM, Raschzok N, Gassner JMGV. Quality Assessment by Bile Composition in Normothermic Machine Perfusion of Rat Livers. Tissue Eng Part A. 2025;31:244-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/