Copyright: ©Author(s) 2026.
World J Gastroenterol. Apr 21, 2026; 32(15): 115226
Published online Apr 21, 2026. doi: 10.3748/wjg.v32.i15.115226
Published online Apr 21, 2026. doi: 10.3748/wjg.v32.i15.115226
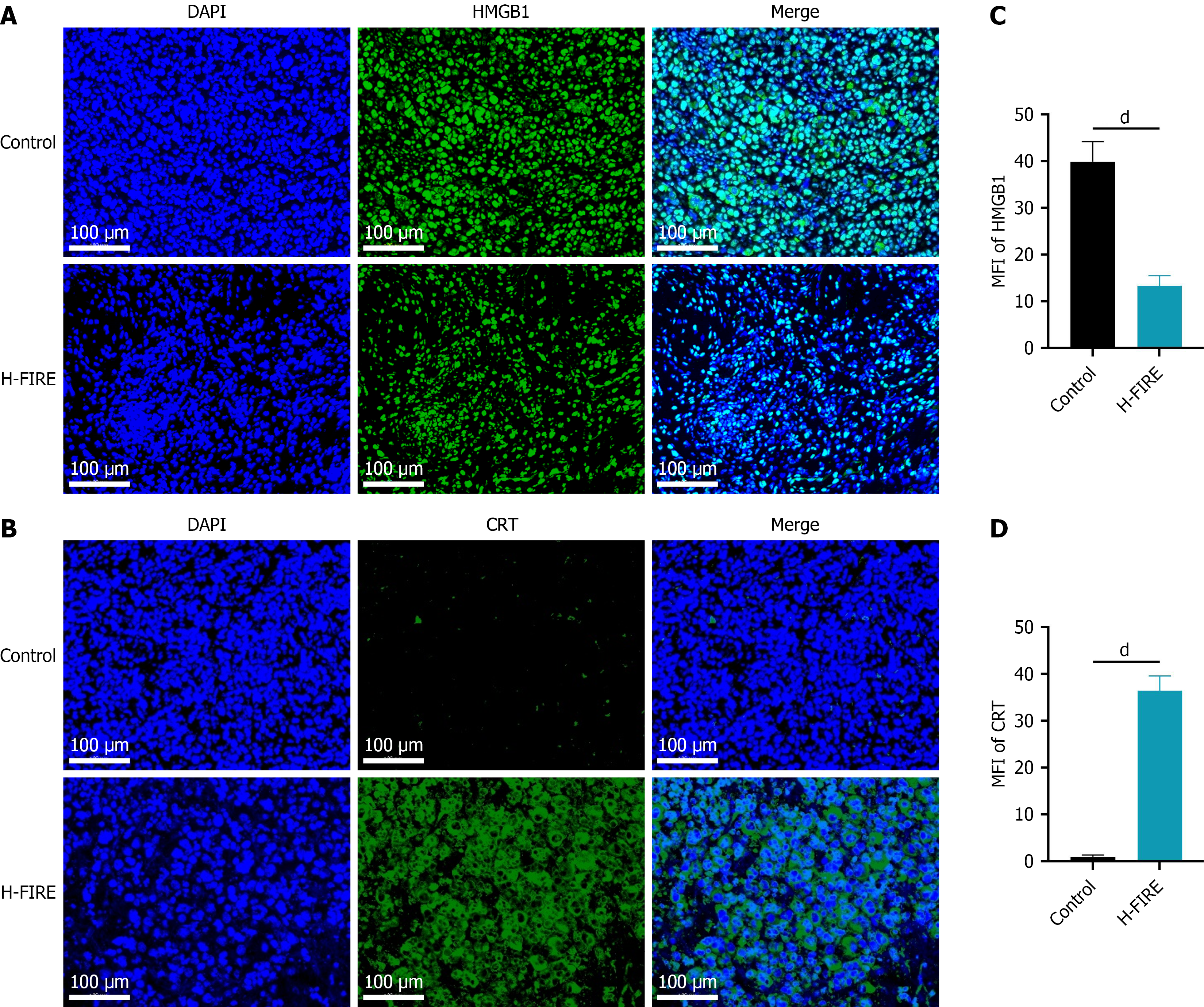
Figure 1 High-frequency irreversible electroporation induces immunogenic cell death in subcutaneous tumor cells.
A and B: Immu
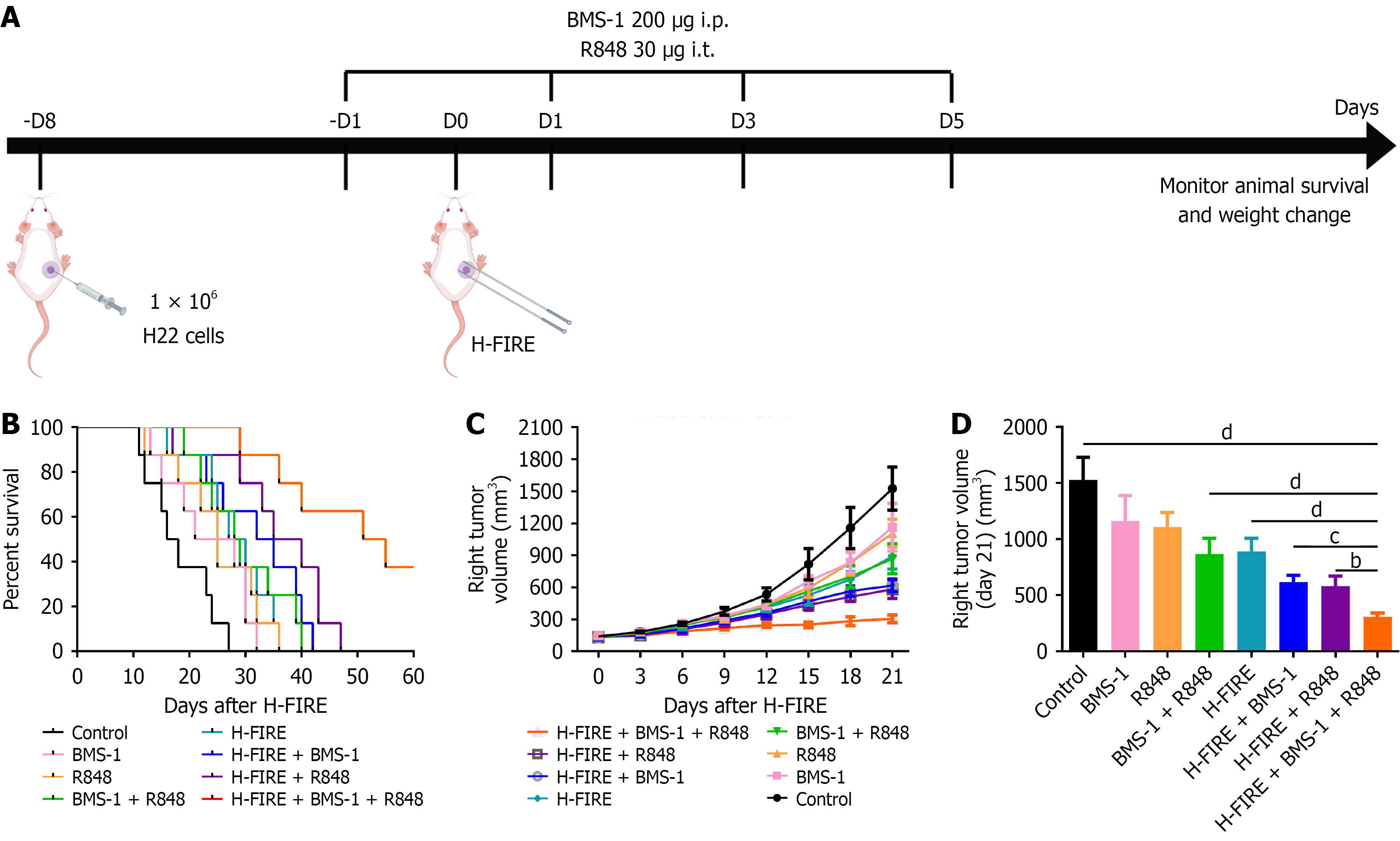
Figure 2 Combinatorial therapy with high-frequency irreversible electroporation, BMS-1, and R848 prolongs survival and inhibits tumor growth in hepatoma 22 subcutaneous tumor-bearing mice.
A: Schematic of the hepatoma 22 subcutaneous tumor model and experimental treatment timeline; B: Kaplan-Meier survival curves across the different treatment groups (n = 8); C: Tumor growth kinetics presented as dynamic volume curves over the observation period (n = 8); D: Comparison of subcutaneous tumor volumes measured on day 21 post-treatment (n = 8). bP < 0.01, cP < 0.001, and dP < 0.0001. i.p.: Intraperitoneal; i.t.: Intratumoral; H-FIRE: High-frequency irreversible electroporation; H22: Hepatoma 22.
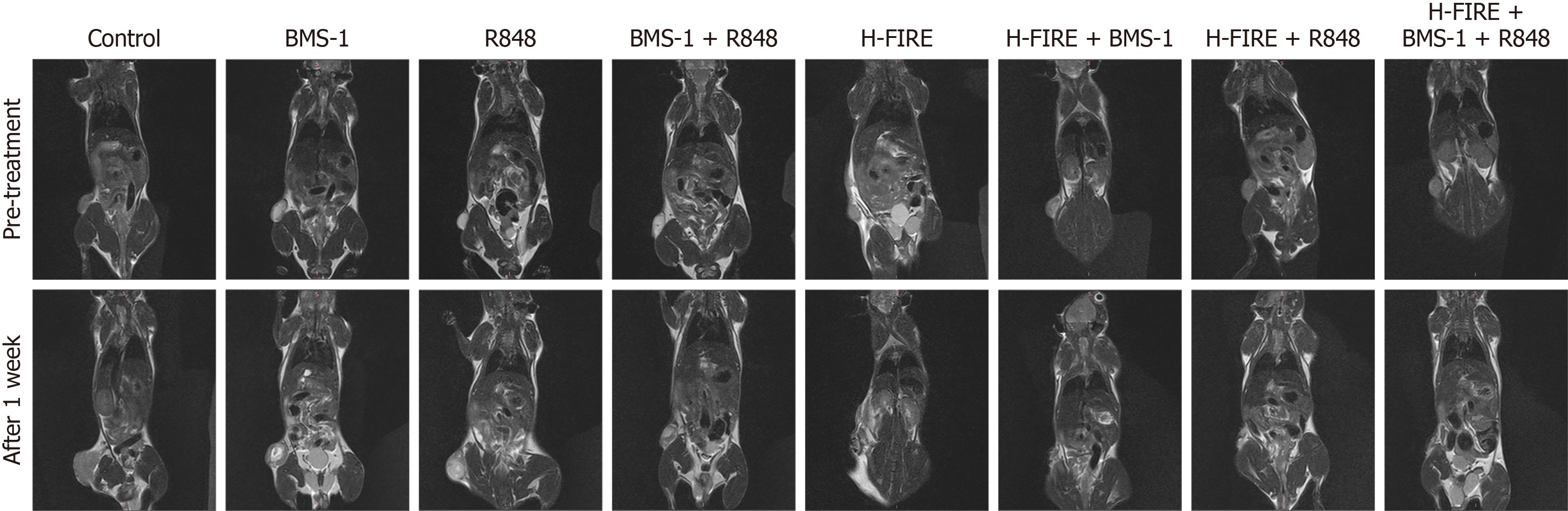
Figure 3 Representative coronal T2-weighted magnetic resonance scans of mouse tumors before treatment and 1 week post-treatment across all experimental groups.
H-FIRE: High-frequency irreversible electroporation.
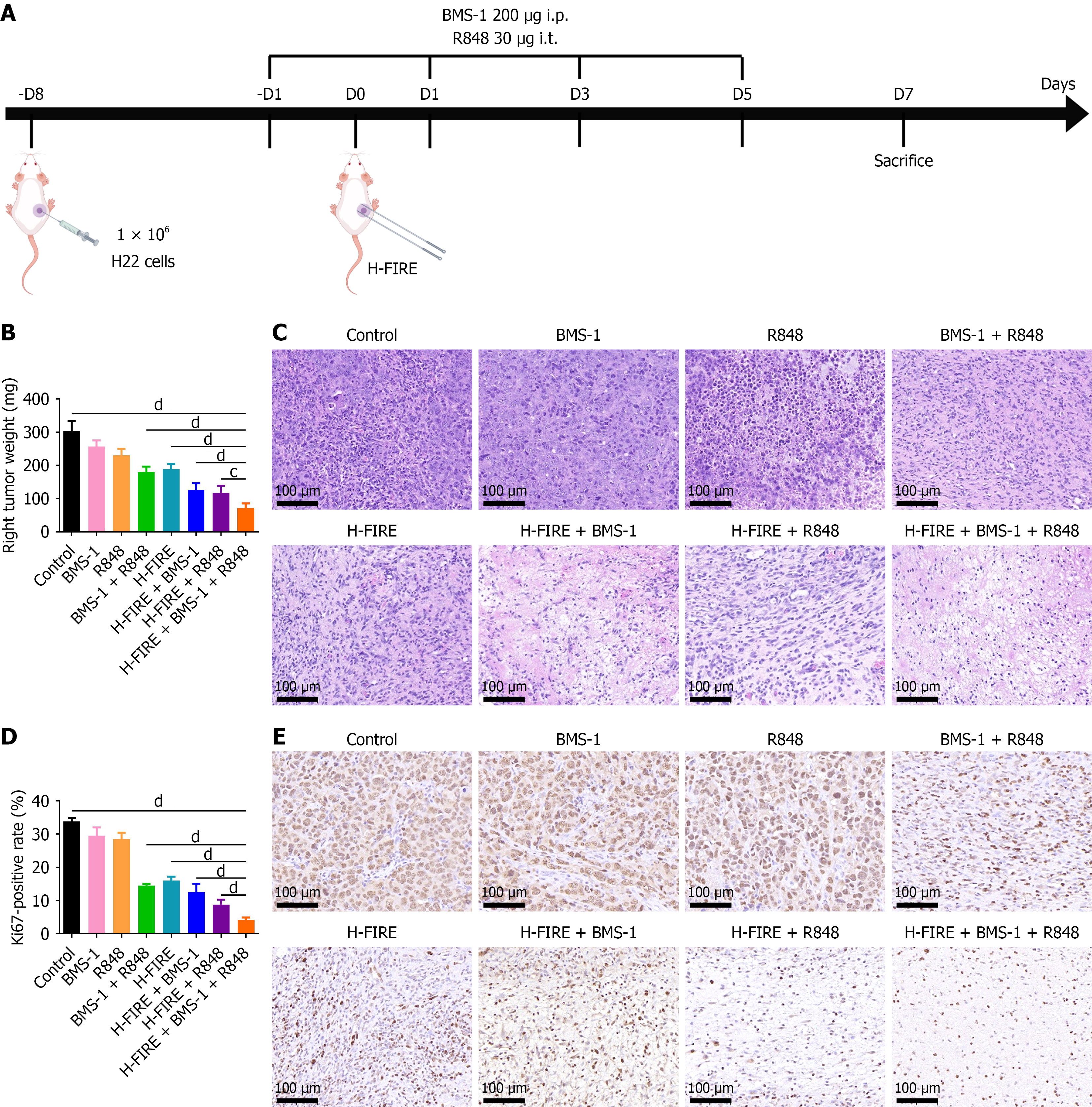
Figure 4 High-frequency irreversible electroporation combined with BMS-1 and R848 impairs tumor cell viability and suppresses proliferation in hepatoma 22 subcutaneous tumors.
A: Schematic of the hepatoma 22 subcutaneous tumor-bearing mouse model and treatment regimen; B: Weight of excised subcutaneous tumors from the right flank at 1 week post-treatment; C: Hematoxylin-eosin staining of subcutaneous tumor sections across treatment groups (n = 8); D: Quantitative analysis of Ki67-positive rate (%) in right-sided subcutaneous tumors 1 week post-treatment (n = 8); E: Representative images of Ki67 immunohistochemical staining in right-flank subcutaneous tumors 1 week post-treatment. cP < 0.001, and dP < 0.0001. i.p.: Intraperitoneal; i.t.: Intratumoral; H-FIRE: High-frequency irreversible electroporation; H22: Hepatoma 22.
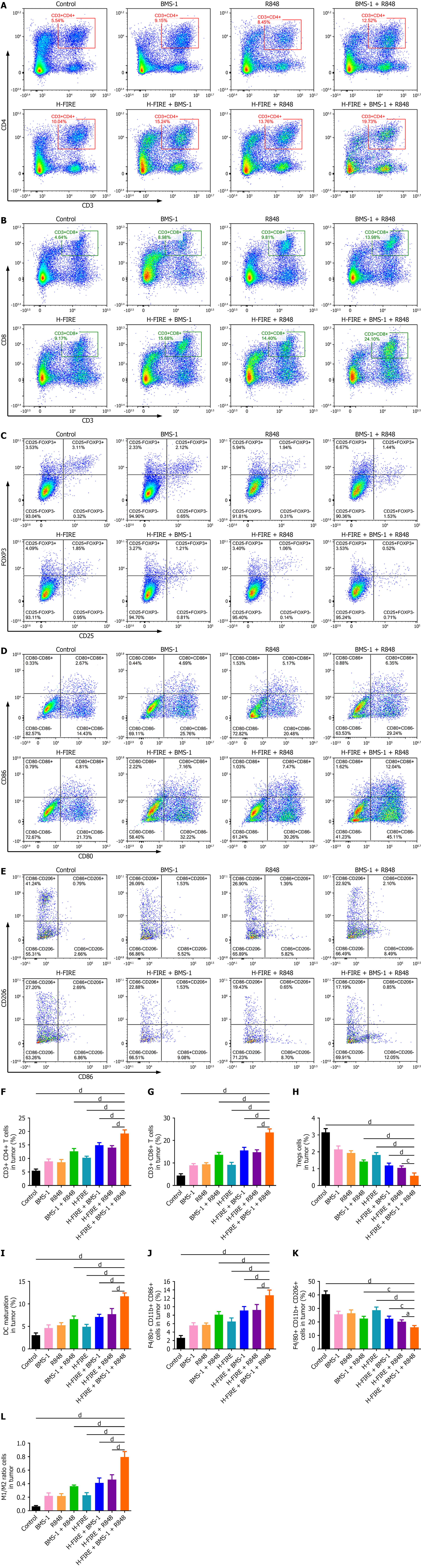
Figure 5 High-frequency irreversible electroporation combined with BMS-1 and R848 reprograms the tumor immune microenvironment in hepatoma 22 subcutaneous tumors.
A-E: Representative flow cytometry dot plots of intratumoral immune cell subsets post-treatment across all groups: CD3+ CD4+ T cells, CD3+ CD8+ T cells, CD4+ CD25+ FOXP3+ Tregs, CD11c+ CD80+ CD86+ dendritic cells, F4/80+ CD11b+ CD86+ M1 macrophages, and F4/80+ CD11b+ CD206+ M2 macrophages; F-K: Quantitative analysis of the proportions of the above-listed intratumoral immune cell subsets in each group post-treatment (n = 5); L: Quantitative analysis of the intratumoral M1/M2 macrophage ratio across groups post-treatment (n = 5). aP < 0.05, cP < 0.001, and dP < 0.0001. H-FIRE: High-frequency irreversible electroporation.
Figure 6 Safety assessment of high-frequency irreversible electroporation combined with BMS-1 and R848 in hepatoma 22 tumor-bearing mice.
A: Dynamic body weight changes of mice across all groups post-treatment (n = 8); B: Quantification of mouse body weights in each group at 21 days post-treatment (n = 8); C: Hematoxylin-eosin staining of major organs (heart, liver, spleen, lung, kidney) in mice from each group post-treatment, showing no obvious histopathological damage; D-H: Serum biochemical analysis shows no significant intergroup differences in liver function markers (aspartate aminotransferase, alanine aminotransferase), a cardiac injury marker (creatine kinase MB isoenzyme), and renal function parameters (creatinine, blood urea nitrogen) post-treatment (n = 8). H-FIRE: High-frequency irreversible electroporation; AST: Aspartate aminotransferase; ALT: Alanine aminotransferase; CK-MB: Creatine kinase MB isoenzyme; CREA: Creatinine; BUN: Blood urea nitrogen.
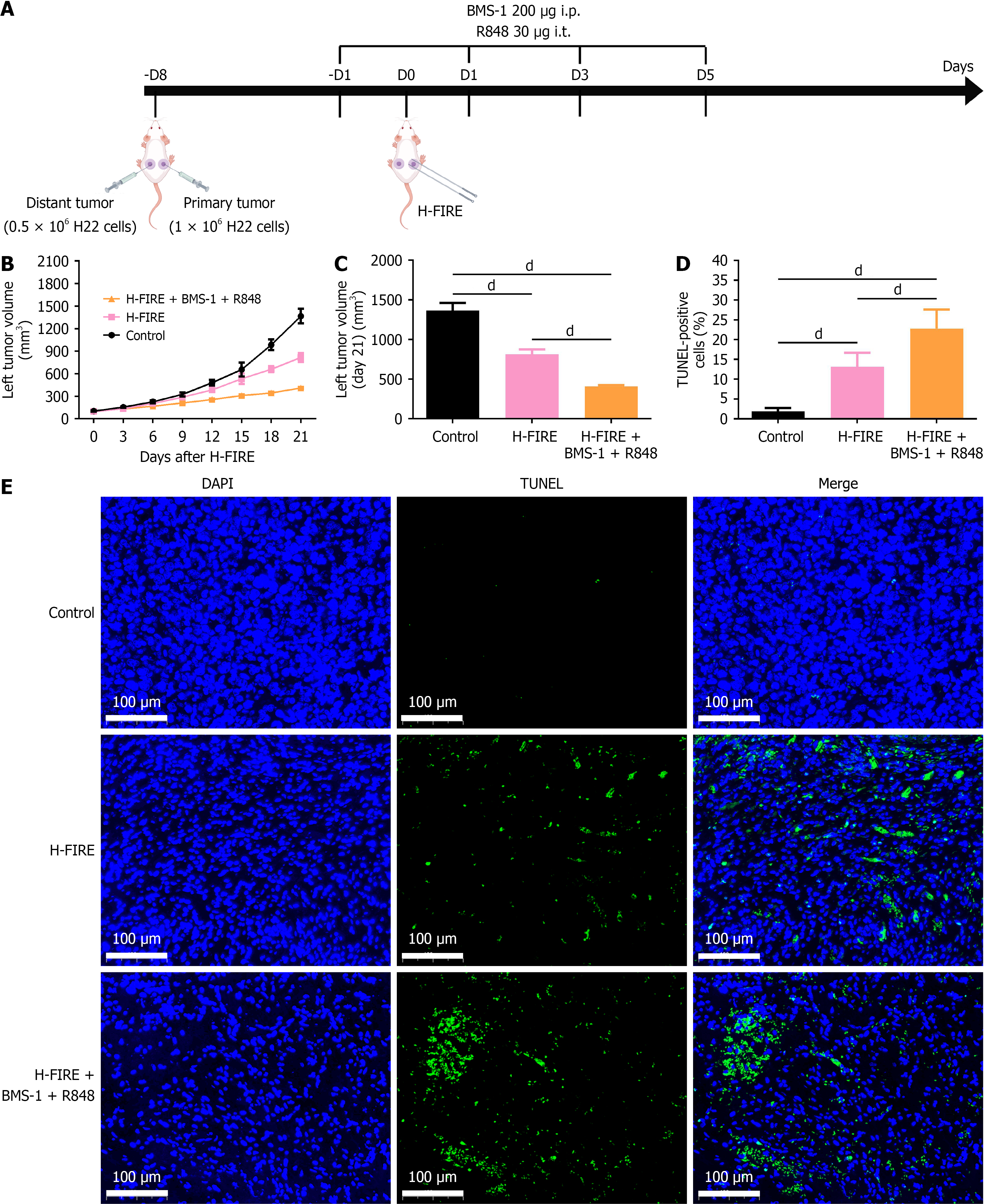
Figure 7 Therapeutic efficacy of high-frequency irreversible electroporation combined with BMS-1 and R848 in the hepatoma 22 bilateral tumor-bearing mice.
A: Schematic of the hepatoma 22 bilateral tumor-bearing mice and treatment regimen; B: Dynamic changes in left tumor volume of hepatoma 22 bilateral tumor-bearing mice post-treatment (n = 8); C: Comparison of left tumor volumes on day 21 post-treatment (n = 8); D: Quantitative analysis of TUNEL-positive cell proportion in left tumors 1 week post-treatment (n = 8); E: Representative TUNEL staining images of left-sided tumors from each group 1 week post-treatment. dP < 0.0001. i.p.: Intraperitoneal; i.t.: Intratumoral; H-FIRE: High-frequency irreversible electroporation; H22: Hepatoma 22.
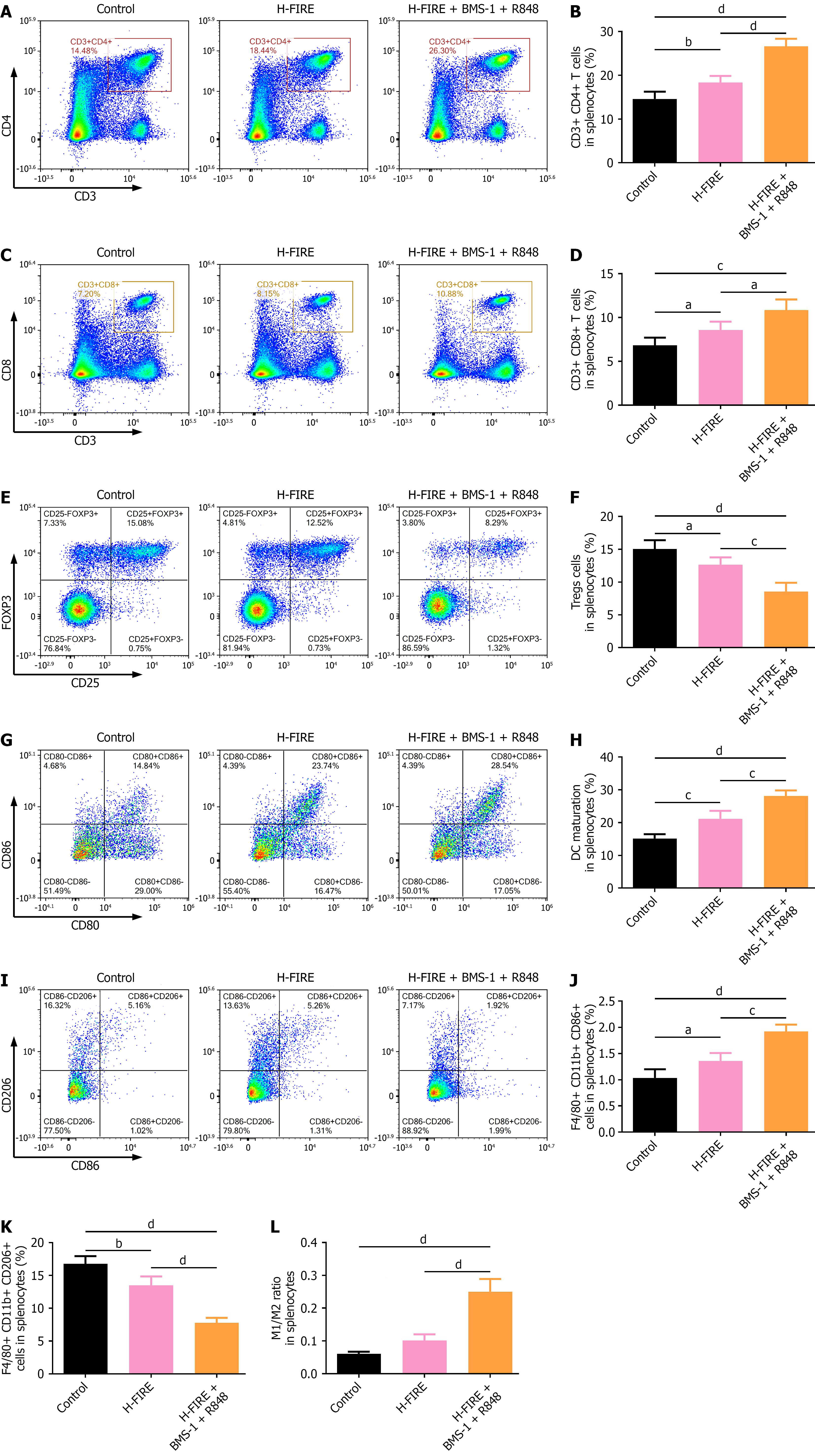
Figure 8 High-frequency irreversible electroporation combined with BMS-1 and R848 elicits systemic antitumor immunity by modulating splenic immune cell subsets.
A and B: Proportion of splenic CD3+ CD4+ T cells; C and D: Proportion of splenic CD3+ CD8+ T cells; E and F: Proportion of splenic CD4+ CD25+ FOXP3+ Tregs; G and H: Proportion of splenic CD11c+ CD80+ CD86+ dendritic cells; I-L: Polarization of splenic macrophages: Proportion of F4/80+ CD11b+ CD86+ M1 and F4/80+ CD11b+ CD206+ M2 macrophages and the M1/M2 macrophage ratio. Each panel shows representative flow cytometry plots alongside quantitative data (n = 5). aP < 0.05, bP < 0.01, cP < 0.001, and dP < 0.0001. H-FIRE: High-frequency irreversible electroporation.
- Citation: Huang SM, Zhang XB, Li J, Zhang GH, Zhang X, Wei YT, Sun H, Ma L, Wang ZJ, Yao DX, Shi HJ, Wang T, Xiao YY. High-frequency irreversible electroporation synergizes with PD-1/PD-L1 inhibitor BMS-1 and TLR7/8 agonist R848 to potentiate liver cancer antitumor immunity. World J Gastroenterol 2026; 32(15): 115226
- URL: https://www.wjgnet.com/1007-9327/full/v32/i15/115226.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i15.115226