Copyright: ©Author(s) 2026.
World J Gastroenterol. Apr 14, 2026; 32(14): 115790
Published online Apr 14, 2026. doi: 10.3748/wjg.v32.i14.115790
Published online Apr 14, 2026. doi: 10.3748/wjg.v32.i14.115790
Figure 1 Study population flowchart.
1Significant hemoglobin drop (SHD): The occurrence of SHD event is declared, if hemoglobin (Hb) decreases by > 2 g/dL compared with the baseline Hb. SHD: Significant hemoglobin drop; ICH: Intracerebral hemorrhage; IVH: Intraventricular hemorrhage; CKD: Chronic kidney disease; PPI: Proton pump inhibitor; H2RA: H2 receptor antagonist; GI: Gastrointestinal; DA: Damaging agent; PA: Protective agent.
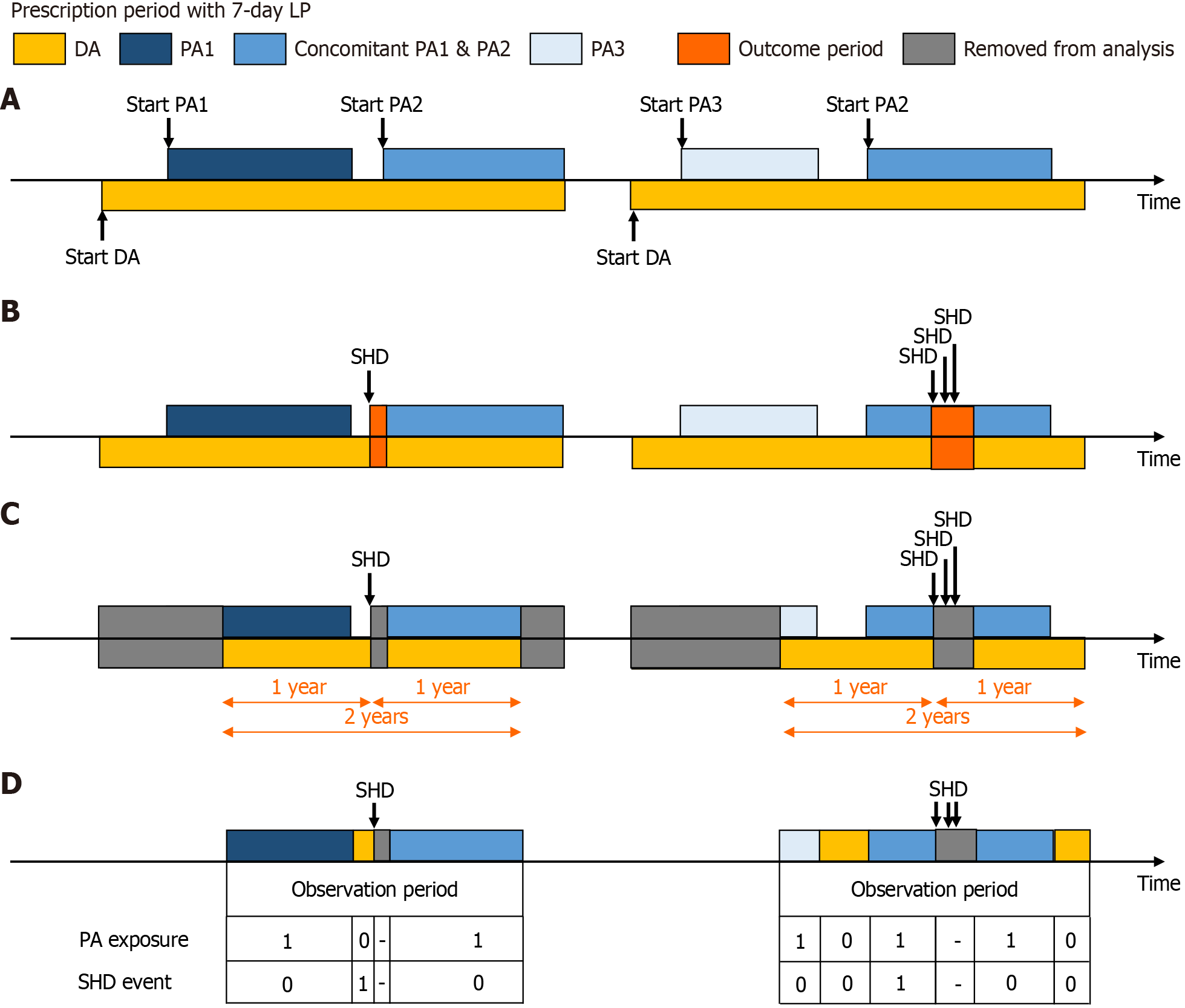
Figure 2 Schematic illustration of self-controlled case series design for a representative patient.
A: Observation period is defined as the entire duration of continuous mucosal damaging agent (DA) prescription periods plus the latent period. Within the observational period, protective agent (PA)-exposure period includes all continuous durations exposed to PAs (either a single agent or combination of agents) plus latent periods. The remaining observation period (i.e., non-PA periods or non-exposure periods) is the duration of DA alone; B: Outcome period is defined as the 30-day period following any significant hemoglobin drop (SHD) events. Overlapping outcome periods due to multiple SHDs were merged into a single outcome. The start of outcome period is considered the index date; C: For each SHD event, all observation periods are restricted to one year before and after the index date. The outcome periods are removed from the analysis; D: The incidence of SHD was assessed and compared between PA-exposure and non-PA periods. Using conditional Poisson regression models, incidence rate ratios for PA-exposure period were estimated compared to non-PA period. SDH: Significant hemoglobin drop; DA: Damaging agent; PA: Protective agent; LP: Latent period.
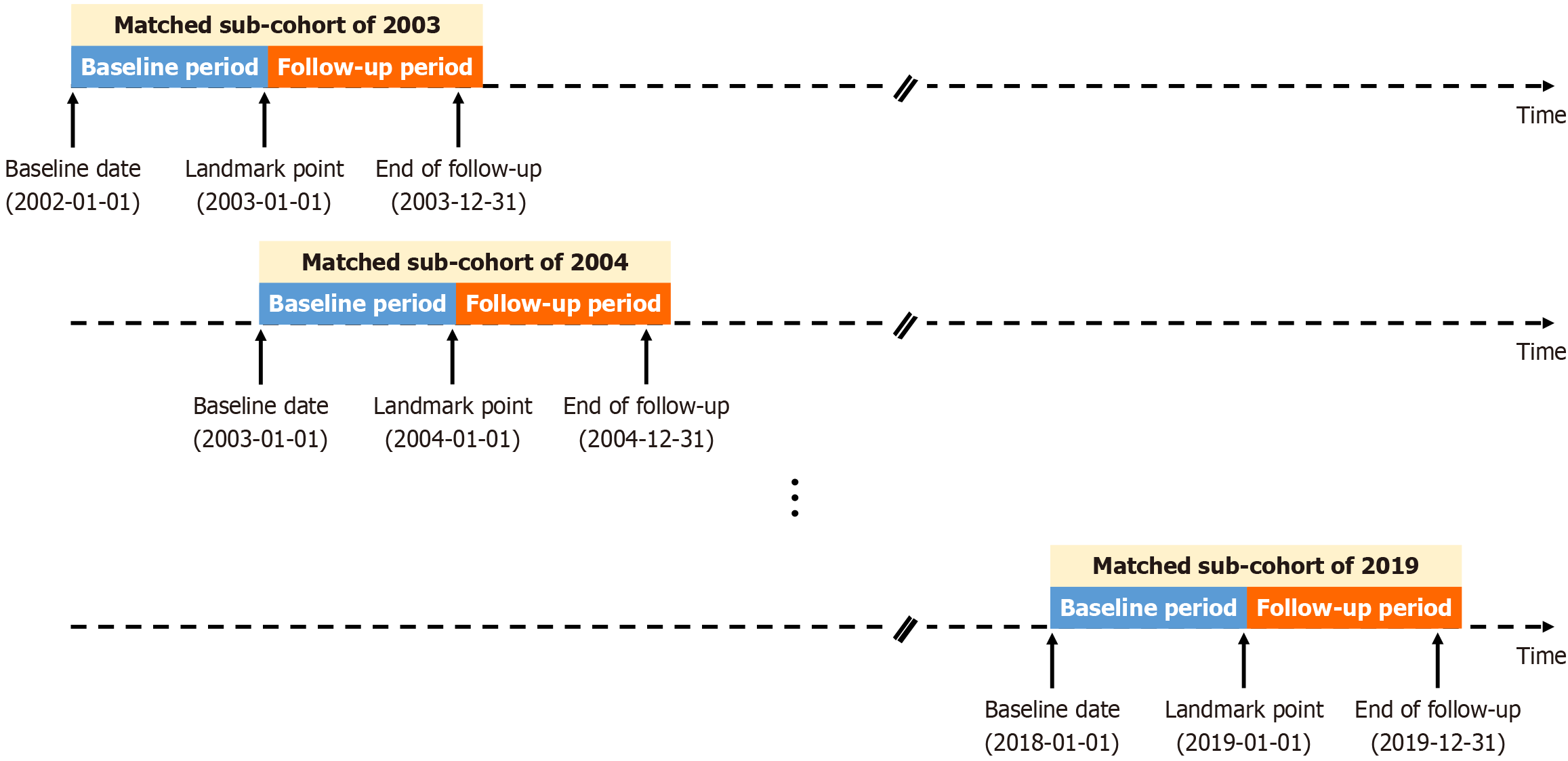
Figure 3
Schematic illustration of retrospective cohort design for the matched landmark analysis.
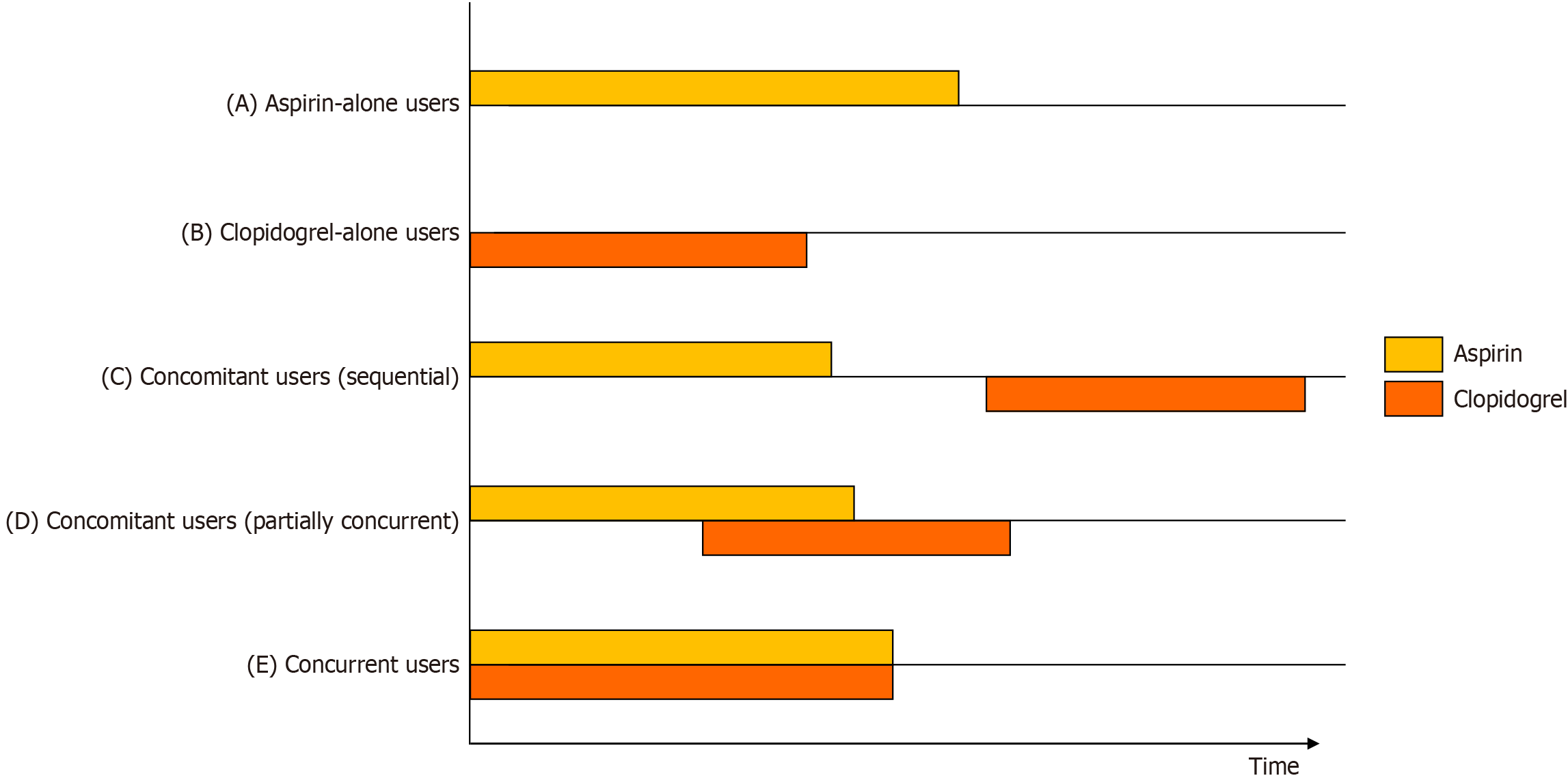
Figure 4 Conceptual diagram of antiplatelet user subgroups.
Subgroups of antiplatelet users based on aspirin and clopidogrel use are illustrated. Aspirin-alone users (A) and clopidogrel-alone users (B) are defined as patients receiving either agent without exposure to the other. Concomitant users are defined as patients exposed to both agents, regardless of taking two agents at the same time (overlapping). Concomitant users include sequential users (C) of two agents without overlapping, partially concurrent users (D) of two agents with partial overlap, and concurrent users (E), taking two agents at the same time without using single agent alone.
- Citation: Kim M, Chi SA, Kim JE, Kim ER, Hong SN, Kim YH, Kim K, Chang DK. Optimal strategies for mitigating gastrointestinal bleeding in patients receiving antiplatelet therapy: Real-world study. World J Gastroenterol 2026; 32(14): 115790
- URL: https://www.wjgnet.com/1007-9327/full/v32/i14/115790.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i14.115790