Copyright: ©Author(s) 2026.
World J Gastroenterol. Apr 7, 2026; 32(13): 115710
Published online Apr 7, 2026. doi: 10.3748/wjg.v32.i13.115710
Published online Apr 7, 2026. doi: 10.3748/wjg.v32.i13.115710
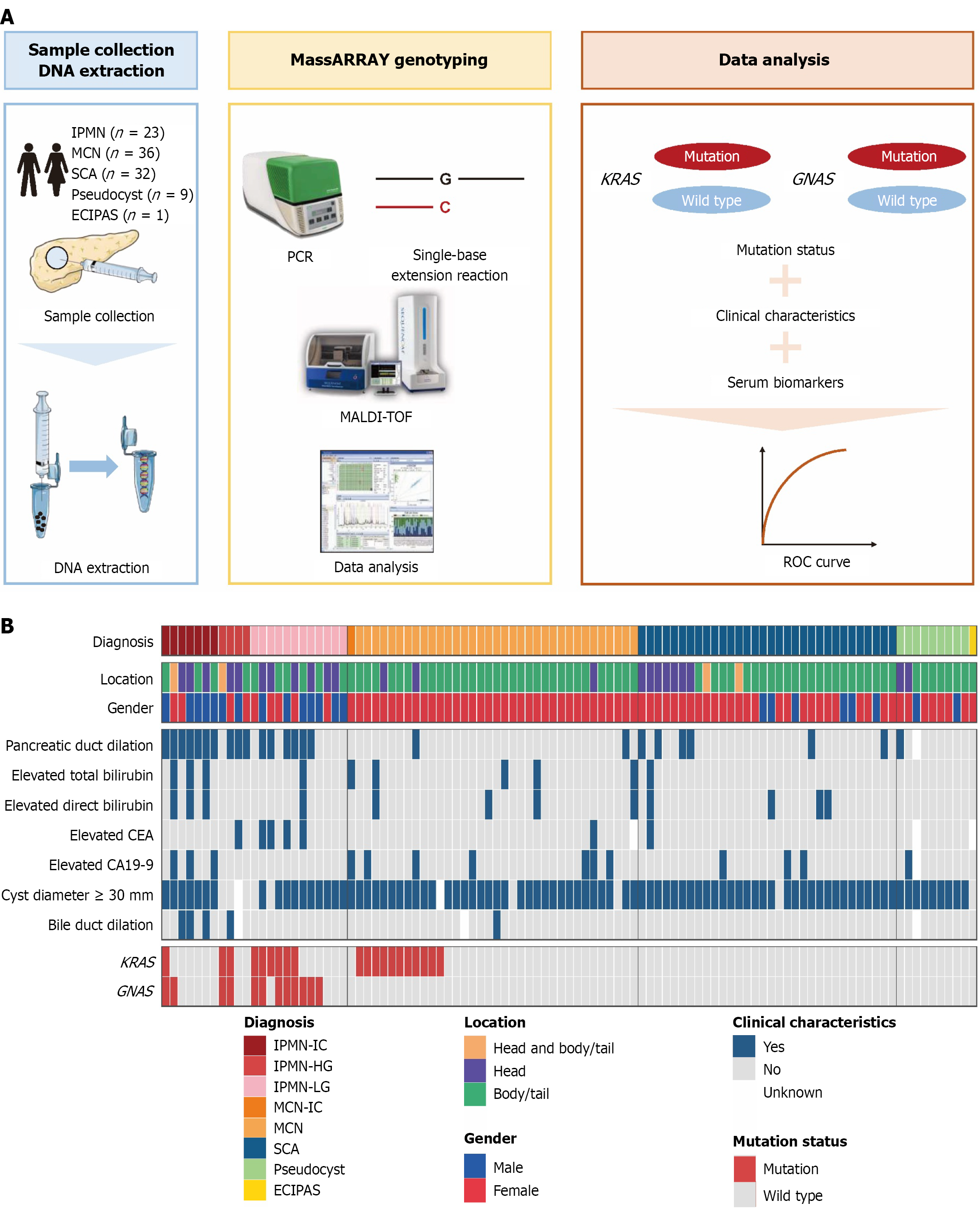
Figure 1 Workflow of this study and heatmap summarizing the clinicopathological, radiological characteristics and hotspot mutation analysis of pancreatic cystic lesions.
A: DNA samples were extracted from pancreatic cystic fluid samples obtained from 101 pancreatic cystic lesions (PCLs) patients for KRAS and GNAS hotspot mutation analysis by MassARRAY, and the results were utilized for PCLs subtype diagnosis; B: Clinicopathological and radiological characteristics, KRAS and GNAS mutation status of 101 PCLs. IPMN: Intraductal papillary mucinous neoplasm; MCN: Mucinous cystic neoplasm; SCA: Serous cystadenoma; ECIPAS: Epidermoid cyst within an intrapancreatic accessory spleen; MALDI-TOF: Matrix-assisted laser desorption ionization time-of-flight; ROC: Receiver operating characteristic; CEA: Carcinoembryonic antigen; CA19-9: Cancer antigen 19-9; IPMN-IC: Intraductal papillary mucinous neoplasm with invasive carcinoma; IPMN-HG: Intraductal papillary mucinous neoplasm with high grade dysplasia; IPMN-LG: Intraductal papillary mucinous neoplasm with low grade dysplasia; MCN-IC: Mucinous cystic neoplasm with invasive carcinoma.
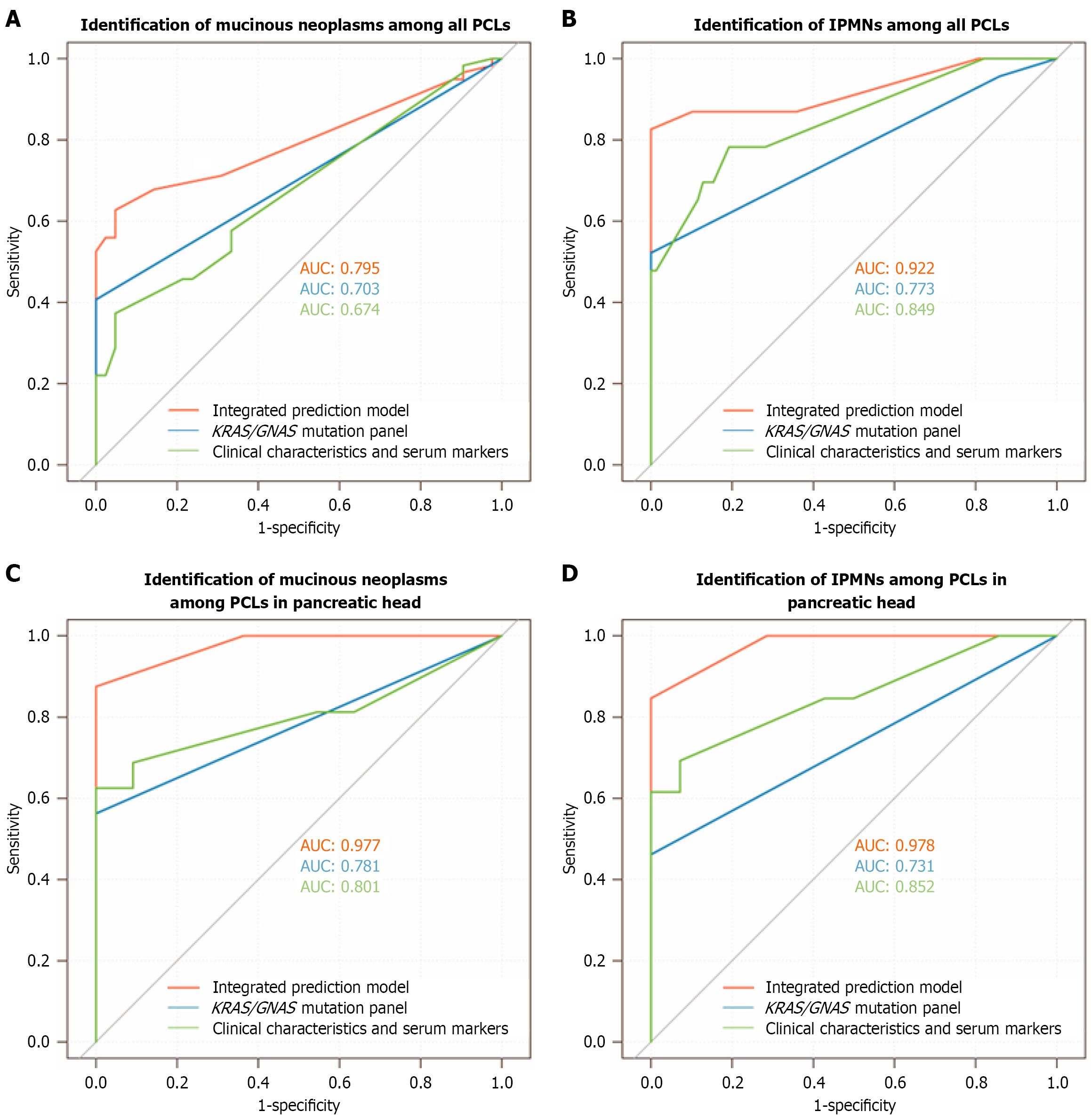
Figure 2 Receiver operating characteristic curves for identification of mucinous neoplasms and intraductal papillary mucinous neo
- Citation: Diao WF, Cui M, Chen TQ, Xiao JH, Yang S, Zheng QY, Xu RY, Han XL, Hu Y. MassARRAY-based KRAS and GNAS hotspot mutation analysis of cystic fluid enables accurate classification of pancreatic cystic lesions. World J Gastroenterol 2026; 32(13): 115710
- URL: https://www.wjgnet.com/1007-9327/full/v32/i13/115710.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i13.115710