Copyright: ©Author(s) 2026.
World J Gastroenterol. Mar 28, 2026; 32(12): 114576
Published online Mar 28, 2026. doi: 10.3748/wjg.v32.i12.114576
Published online Mar 28, 2026. doi: 10.3748/wjg.v32.i12.114576
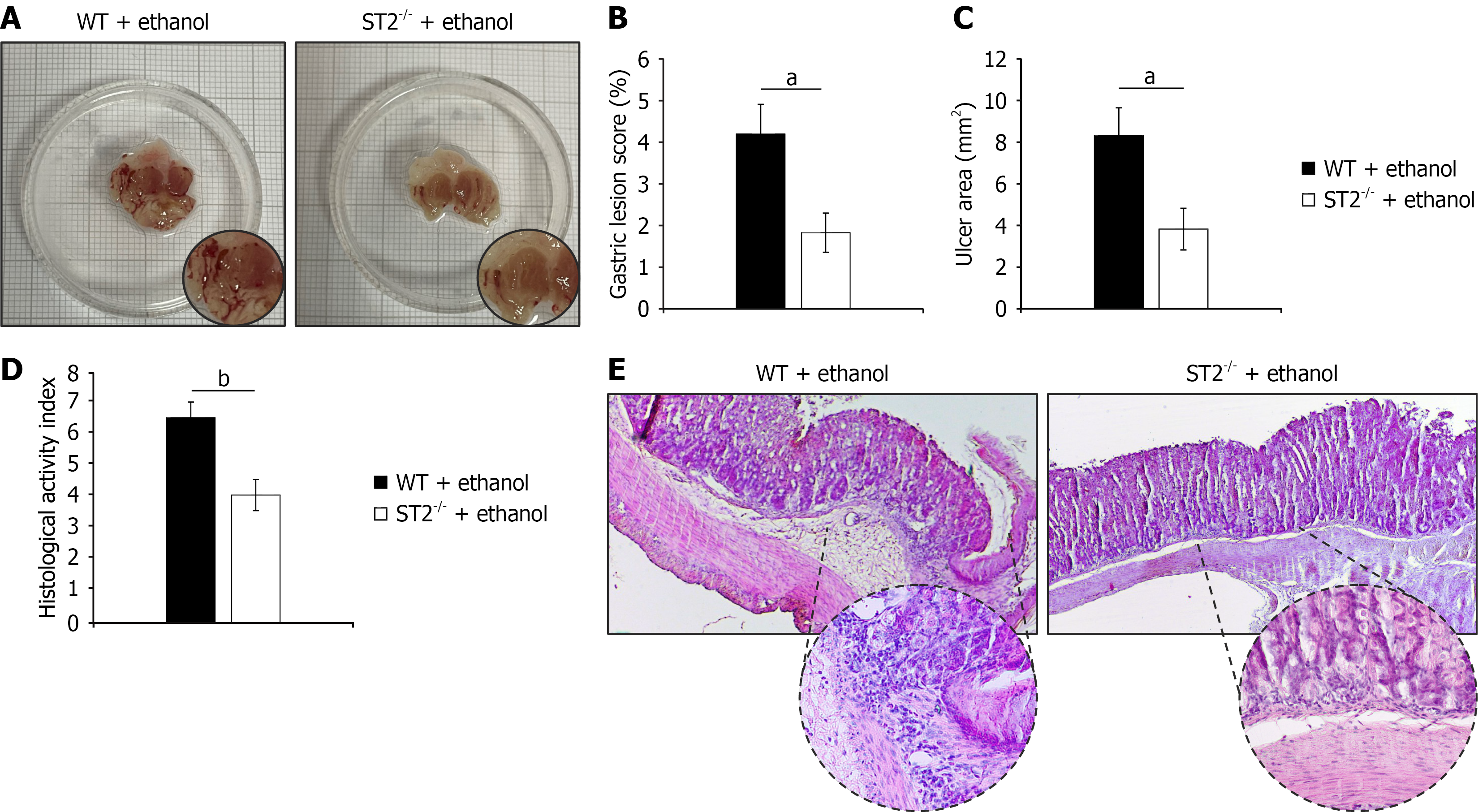
Figure 1 Suppression of tumorigenicity 2 gene deficiency mitigates acute gastric injury.
A: Representative macroscopic images of gastric tissue showing differences in the severity of mucosal damage following ethanol exposure; B and C: Quantitative evaluation of macroscopic gastric injury. Gastric lesion score (%) (B) and ulcer area (mm2) (C) were determined by capturing digital images of the inner gastric surface and analyzing them using ImageJ software; D: Microscopic assessment of tissue damage based on histological activity index; E: Representative hematoxylin and eosin staining images of gastric tissue sections (magnified × 10 and × 40) illustrating differences in epithelial integrity, inflammatory infiltration, and overall tissue architecture between experimental groups. Data are presented as mean ± SEM; n = 10 mice per group. aP < 0.05, bP < 0.01. WT: Wild type; ST2: Suppression of tumorigenicity 2.
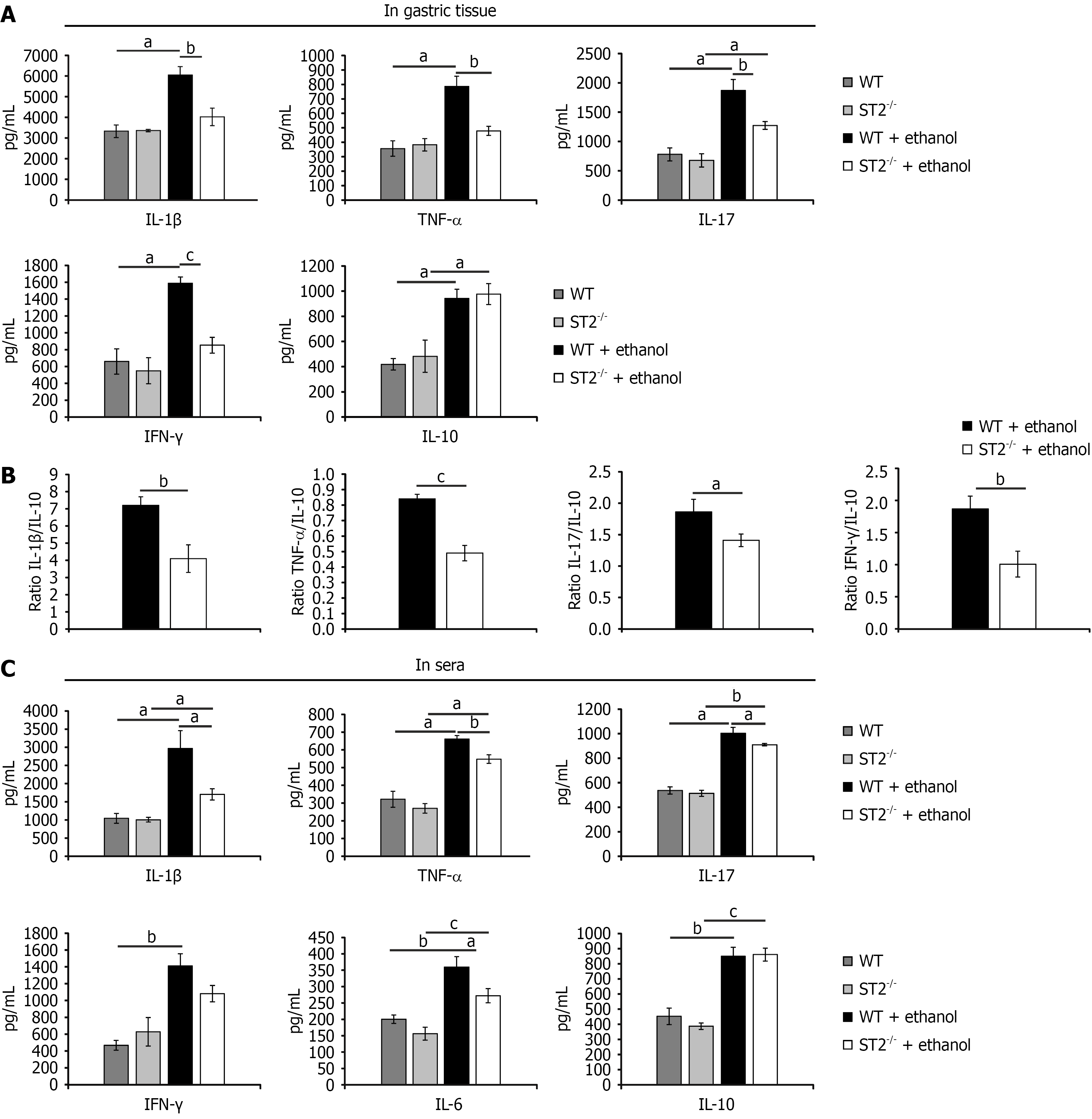
Figure 2 Attenuated systemic and local proinflammatory milieu in suppression of tumorigenicity 2 gene knockout mice during acute gastric injury.
A: Concentrations of interleukin (IL)-1β, tumor necrosis factor-α (TNF-α), IL-17, interferon (IFN)-γ, and IL-10 in gastric tissue homogenates; B: Ratios of proinflammatory to anti-inflammatory cytokines (IL-1β/IL-10, TNF-α/IL-10, IL-17/IL-10, and IFN-γ/IL-10) in gastric tissue to evaluate the inflammatory balance; C: Serum concentrations of IL-1β, TNF-α, IL-17, IFN-γ, IL-6, and IL-10. Cytokine levels were measured using enzyme-linked immunosorbent assay. Results are displayed as mean ± SEM; n = 10 mice per group. aP < 0.05, bP < 0.01, cP < 0.001. WT: Wild type; ST2: Suppression of tumorigenicity 2; IFN: Interferon; TNF-α: Tumor necrosis factor-α.
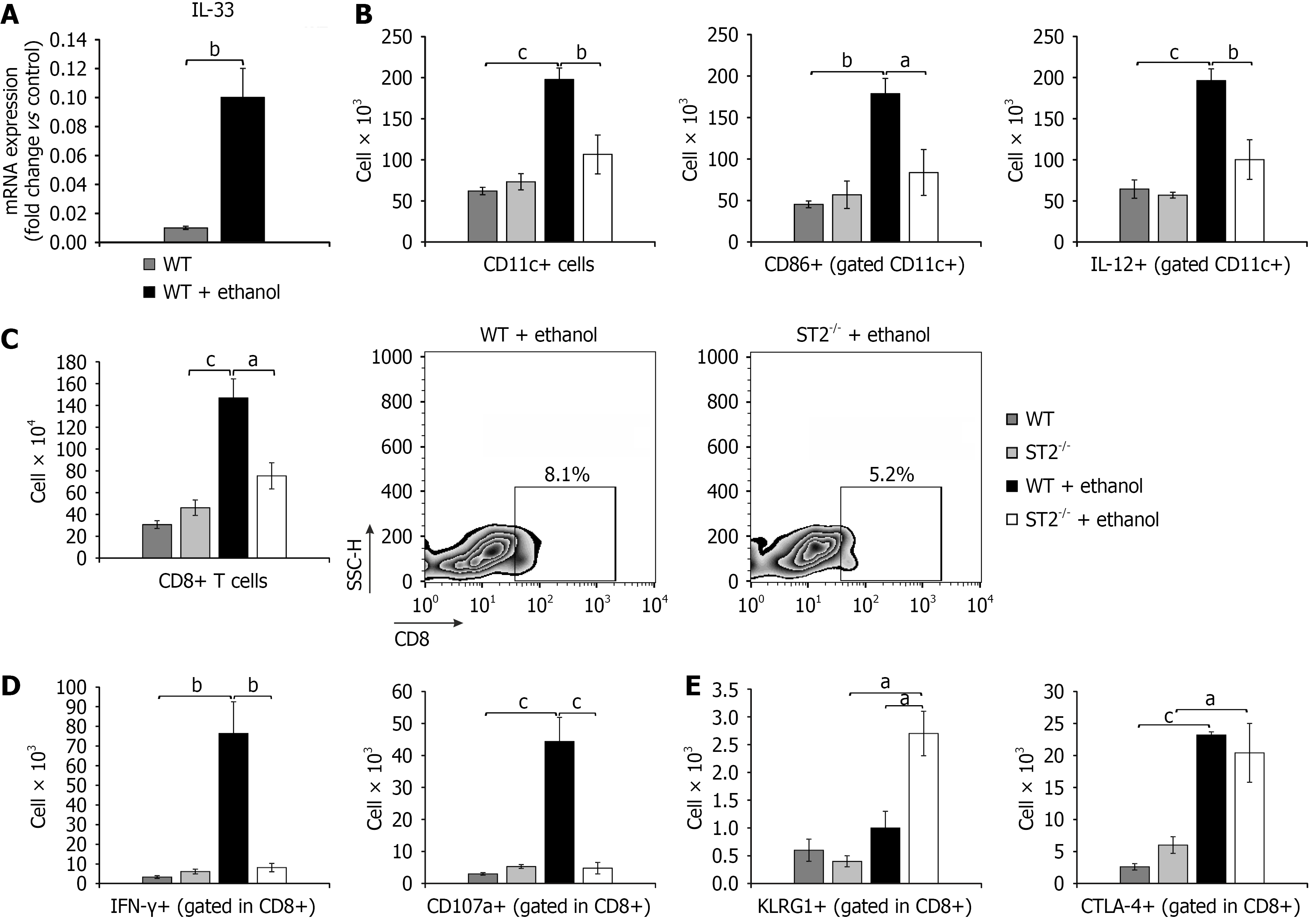
Figure 3 Interleukin-33 upregulation in acute gastric injury enhances type 1 cytotoxic activation and dendritic cells recruitment.
A: Interleukin-33 mRNA expression levels in gastric tissue following ethanol administration, measured by real-time quantitative reverse transcriptase polymerase chain reaction; B: Total count of dendritic cells (CD11c+), including the subsets expressing CD86 and producing interleukin-12, assessed by flow cytometry; C-E: Total count and representative flow cytometry dot plots of CD8+ cells in ethanol-treated mice; quantification of CD8+ cells subsets (C). Producing interferon-γ and CD107a (D), and expressing the inhibitory molecules killer cell lectin-like receptor subfamily G member 1 and cytotoxic T-lymphocyte-associated protein 4 (E). Results are displayed as mean ± SEM; n = 10 mice per group. aP < 0.05, bP < 0.01, cP < 0.001. WT: Wild type; ST2: Suppression of tumorigenicity 2; IFN: Interferon; KLRG1: Killer cell lectin-like receptor subfamily G member 1; CTLA-4: Cytotoxic T-lymphocyte-associated protein 4.
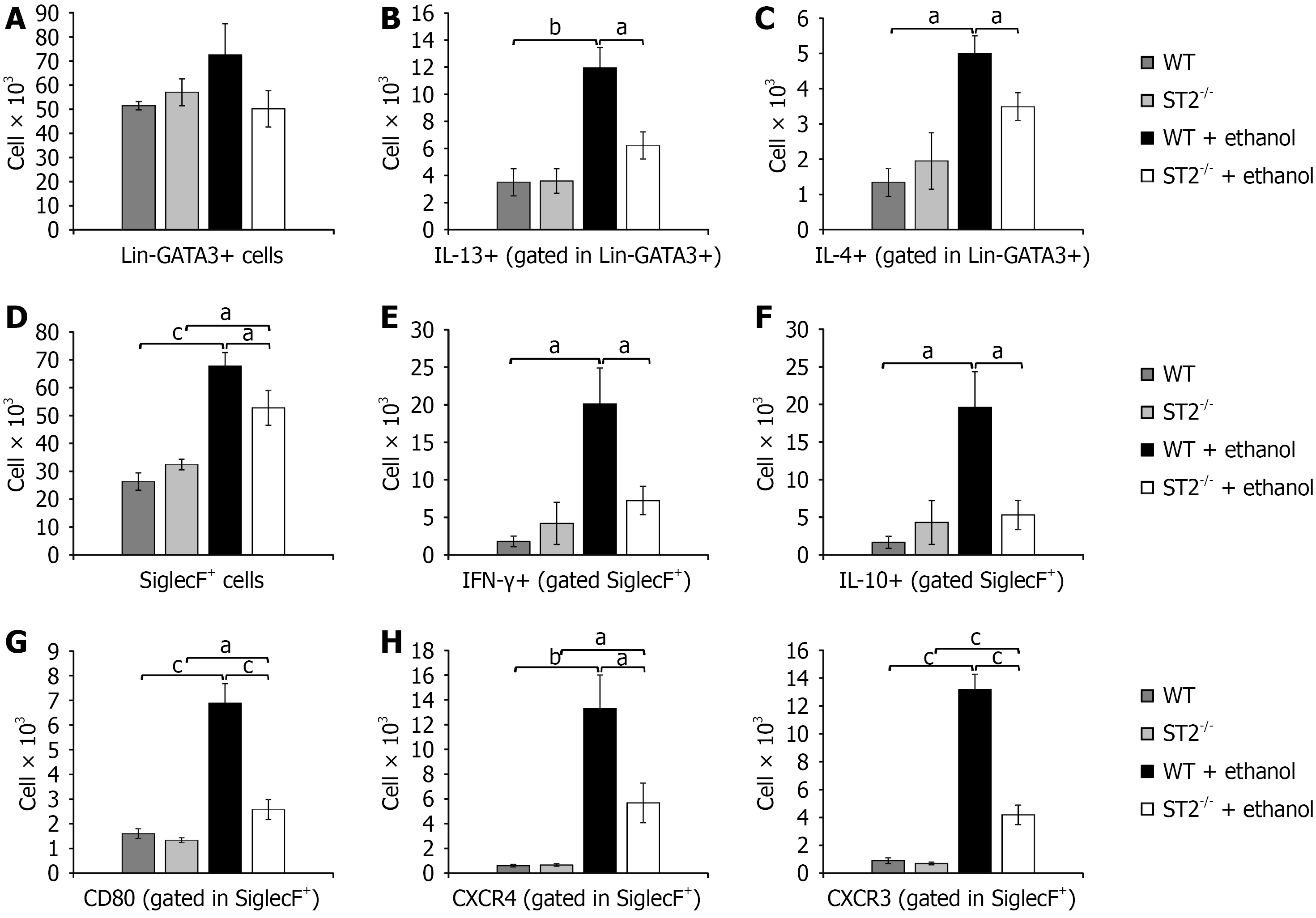
Figure 4 The interplay between interleukin-33, ILC2s, and eosinophils exacerbates acute gastric injury.
A-C: Total count of ILC2 (Lin-GATA3+) in gastric tissue (A), interleukin (IL)-13-producing (B), and IL-4-producing (C) ILC2 subsets following ethanol administration; D-H: Total count of eosinophils (SiglecF+) (D); interferon-γ-producing (E) and IL-10-producing eosinophils (F); CD80+ (G), CXCR4+ and CXCR3+ (H) eosinophils in gastric tissue following ethanol administration. All parameters were analyzed by flow cytometry. Data are expressed as mean ± SEM; n = 10 mice per group. aP < 0.05, bP < 0.01, cP < 0.001. GATA3: GATA binding protein 3; WT: Wild type; ST2: Suppression of tumorigenicity 2; IL: Interleukin; IFN: Interferon; CXCR: C-X-C motif chemokine receptor.
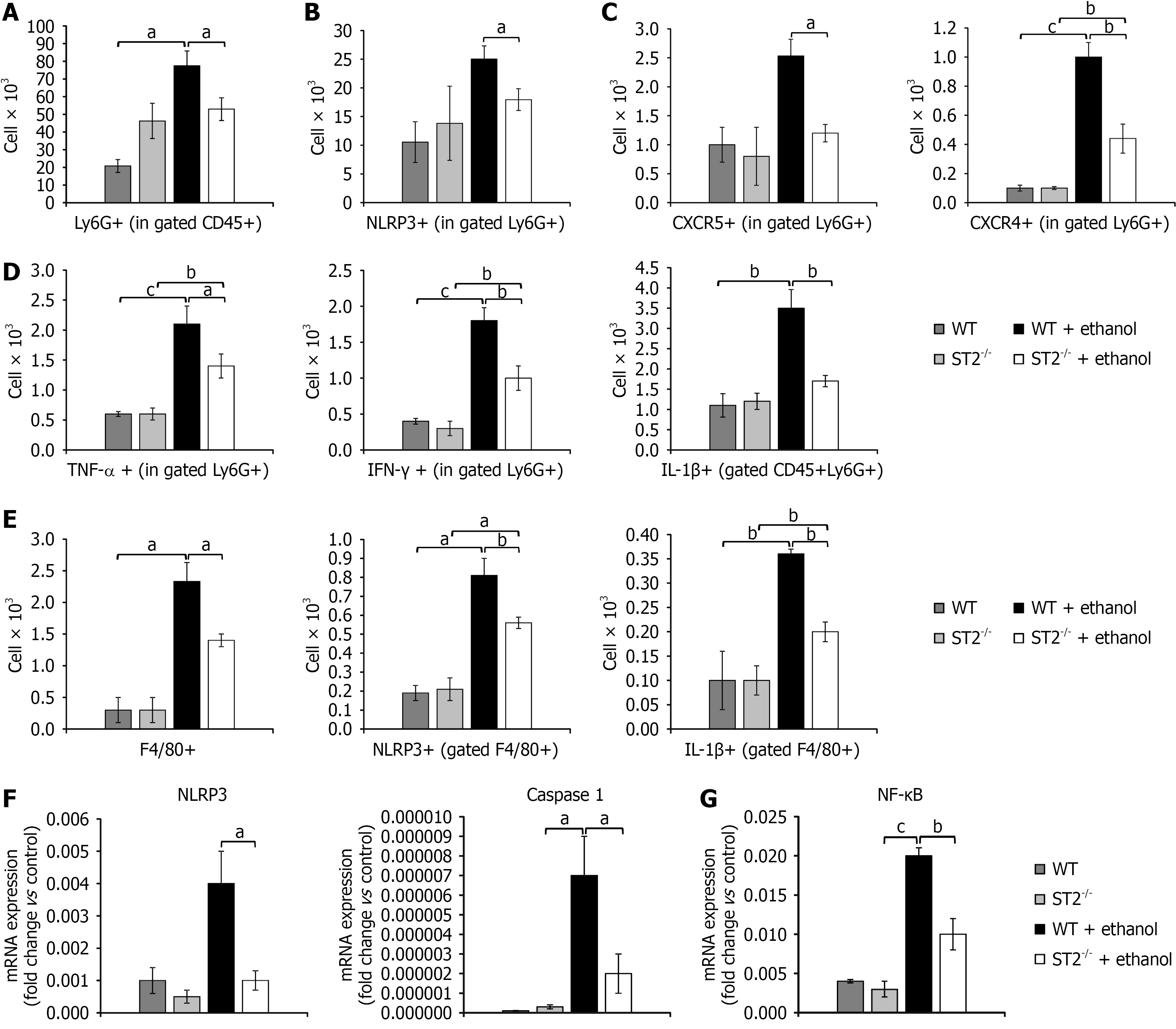
Figure 5 Suppression of tumorigenicity 2 gene deficiency attenuates neutrophil and macrophage activation and reduces gene expression of nuclear factor kappa B and NOD-like receptor family, pyrin domain containing 3.
A-D: Total count of neutrophils (Ly6G+) (A) in gastric tissue following ethanol administration, along with neutrophil subsets expressing NOD-like receptor family, pyrin domain containing 3 (NLRP3) (B), expressing C-X-C motif chemokine receptor 5 and C-X-C motif chemokine receptor 4 (C), and producing tumor necrosis factor-α, interferon-γ, and interleukin-1β (D); E: Total count of macrophages (F4/80+) in gastric tissue following ethanol exposure, including M1 macrophage subsets expressing NLRP3 and producing interleukin-1β. All parameters were analyzed by flow cytometry; F and G: The mRNA expression levels of NLRP3, caspase-1 (F), and nuclear factor kappa B (G) in gastric tissue detected by real-time quantitative reverse transcriptase polymerase chain reaction. Data are presented as mean ± SEM; n = 10 mice per group. aP < 0.05, bP < 0.01, cP < 0.001. WT: Wild type; ST2: Suppression of tumorigenicity 2; NLRP3: NOD-like receptor family, pyrin domain containing 3; CXCR: C-X-C motif chemokine receptor; TNF-α: Tumor necrosis factor-α; IL: Interleukin; NF-κB: Nuclear factor kappa B.
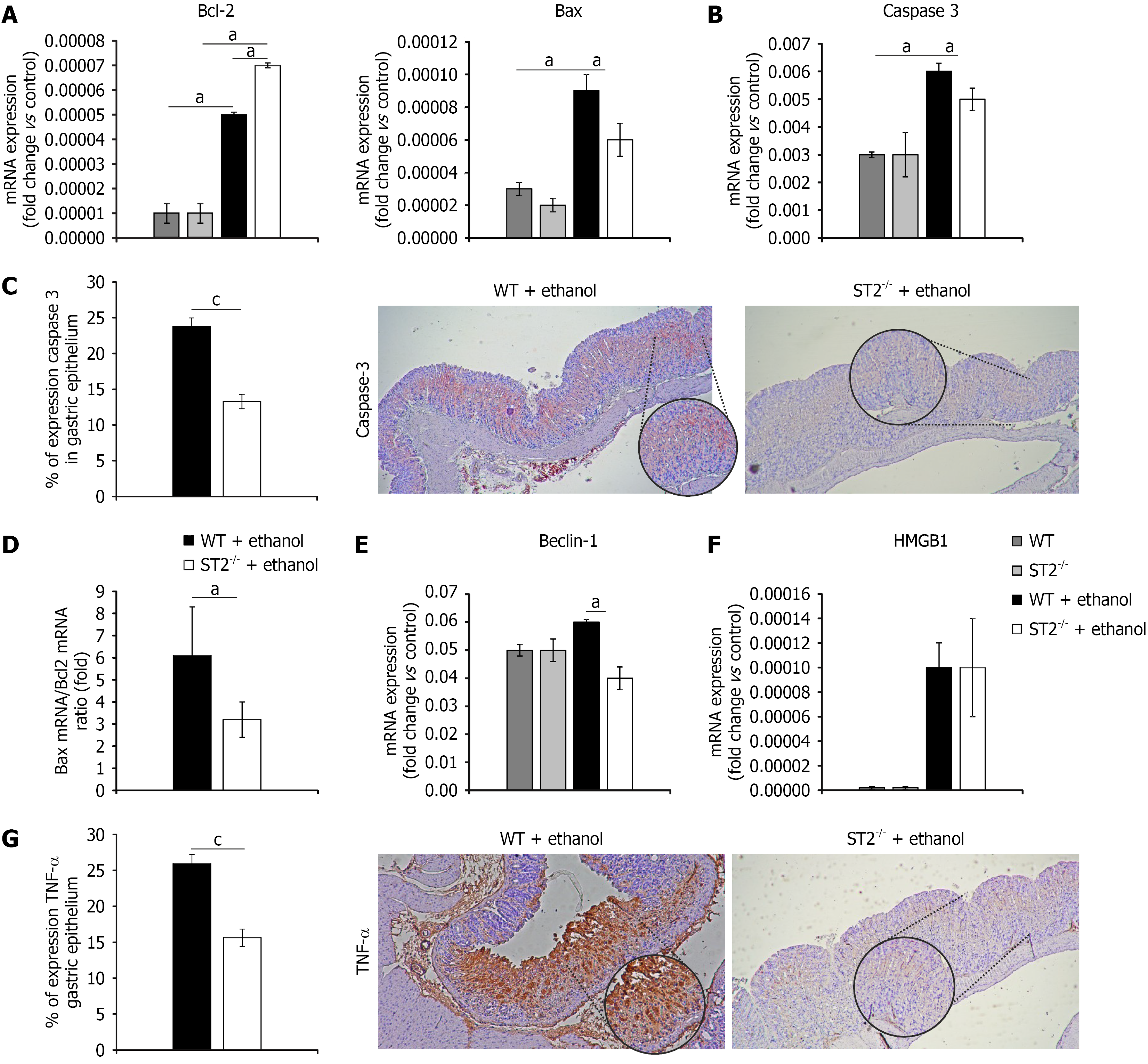
Figure 6 Suppression of tumorigenicity 2 deletion downregulates diverse cell death signaling in acute gastric damage.
A and B: Real-time quantitative reverse transcriptase polymerase chain reaction (RT-qPCR) analysis of mRNA expression levels of Bcl-2 and Bax (A), and caspase 3 (B) in gastric tissue; C: Percentage of caspase 3 positive cells in the gastric epithelium assessed by immunohistochemical, accompanied by representative images (magnified × 10 and × 40); D: RT-qPCR analysis of mRNA expression levels of Bax/Bcl-2 expression ratio; E and F: The mRNA expression levels of Beclin-1 (E) and high mobility group box-1 (F) gastric tissue detected by RT-qPCR; G: Percentage of tumor necrosis factor-α-positive cells in the gastric epithelium, evaluated by immunohistochemical and illustrated with representative images (magnified × 10 and × 40). Data are expressed as mean ± SEM; n = 10 mice per group. aP < 0.05, cP < 0.001. WT: Wild type; ST2: Suppression of tumorigenicity 2; HMGB1: High mobility group box-1; TNF-α: Tumor necrosis factor-α.
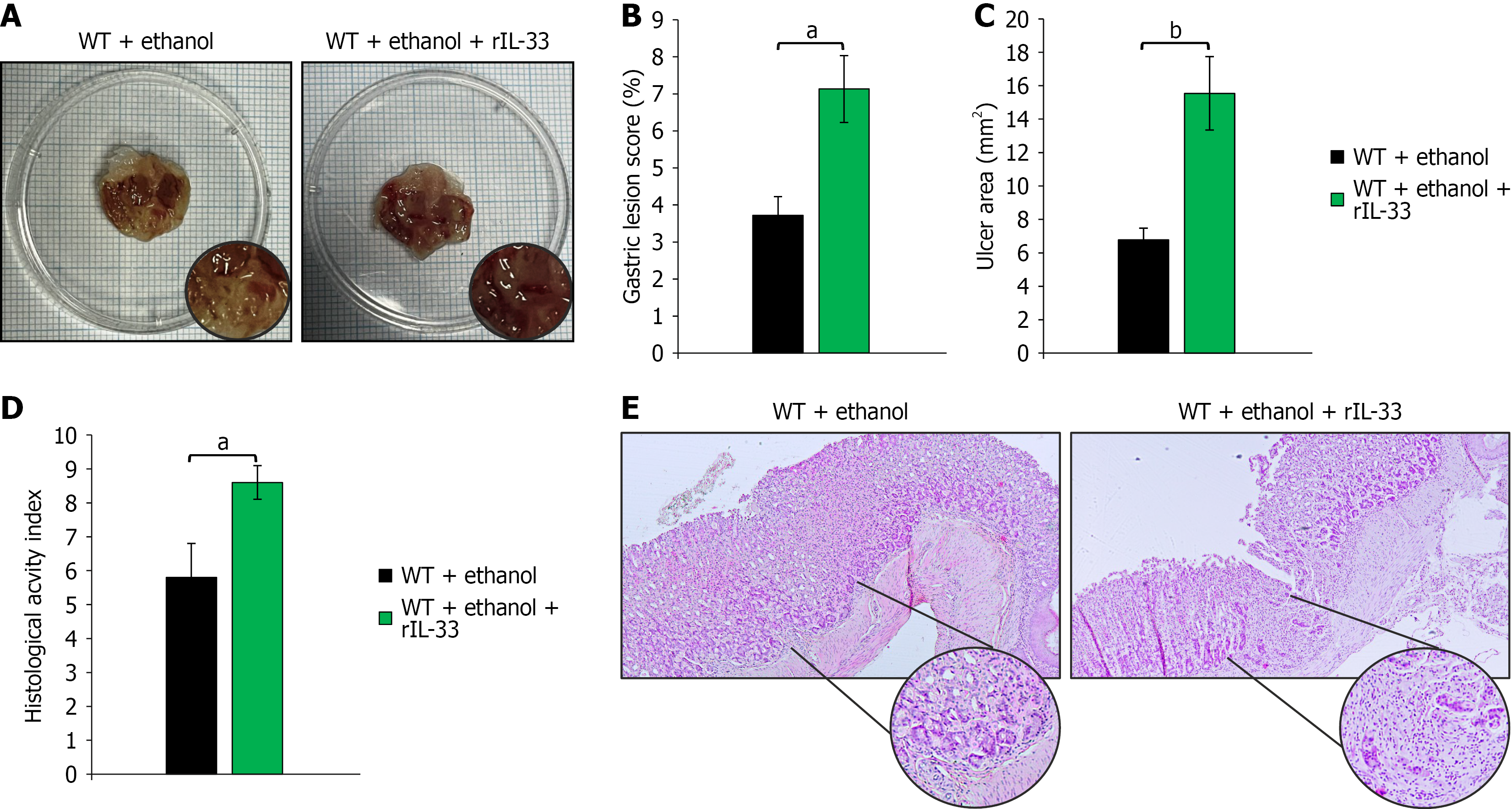
Figure 7 Recombinant interleukin-33 aggravates acute gastric tissue injury.
A: Representative macroscopic images illustrating the extent of gastric mucosal damage; B and C: Quantitative evaluation of gastric injury: Gastric lesion score (%) (B) and ulcer area (mm²) (C), analyzed using ImageJ software; D: Microscopic assessment of tissue damage based on histological activity index, accompanied by representative hematoxylin and eosin staining images (magnified × 10 and × 40). Data are presented as mean ± SEM; n = 10 mice per group. aP < 0.05, bP < 0.01. WT: Wild type; rIL-33: Recombinant interleukin-33.
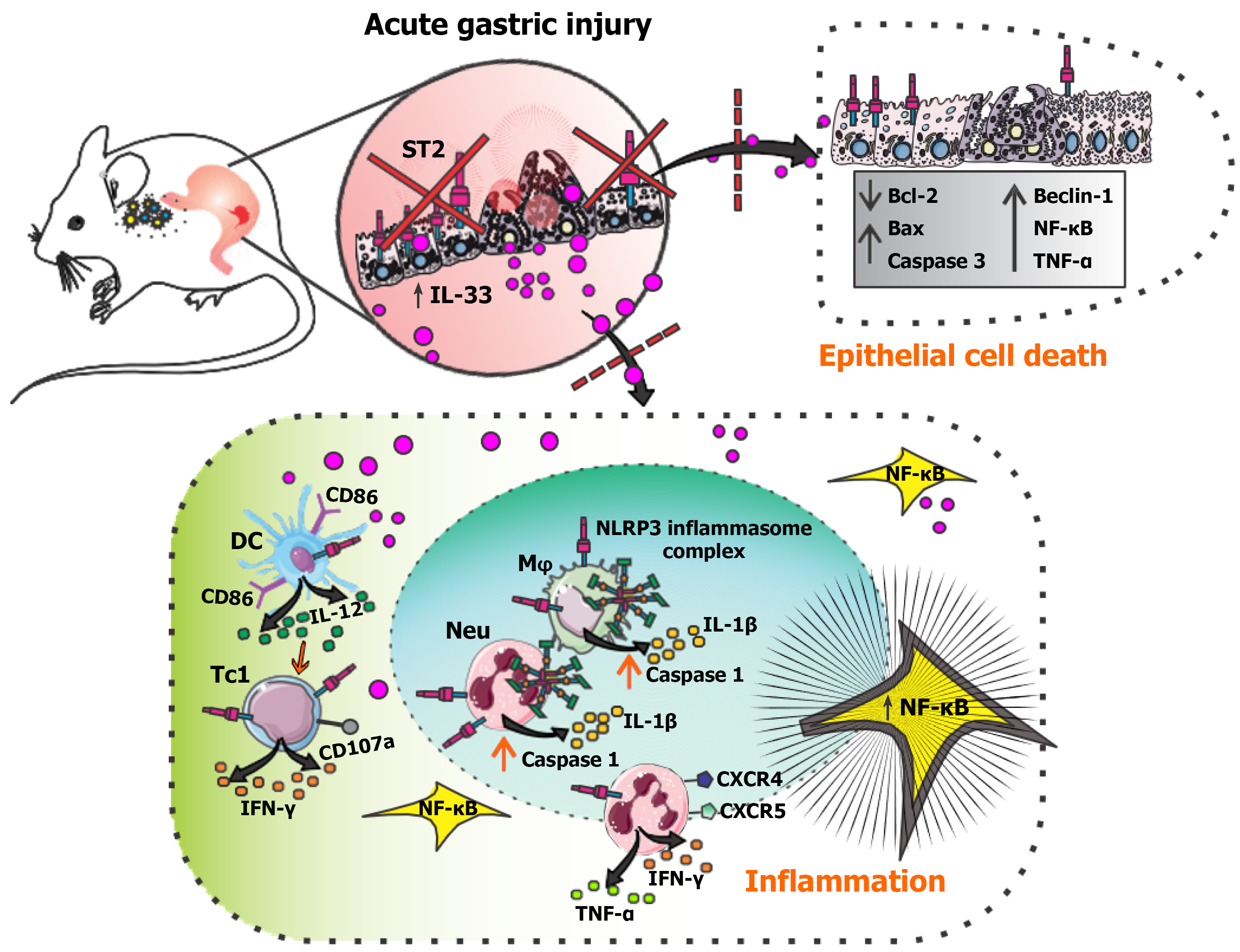
Figure 8 Schematic representation of the immunopathological mechanisms regulated by suppression of tumorigenicity 2 deletion in acute gastric injury.
Ethanol exerts both direct cytotoxic effects on the gastric epithelium and indirect effects through the induction of inflammation. Tissue injury triggers the release of the nuclear alarmin interleukin (IL)-33 from gastric mucosal cells, which, by binding to its receptor suppression of tumorigenicity 2 (ST2), orchestrates a broad range of immune responses. ST2 deletion attenuates the infiltration and activation of dendritic cells, reducing the number of IL-12-producing and CD86-expressing dendritic cells, and impairs type 1 cytotoxic polarization, resulting in decreased interferon-γ production. Furthermore, ST2 gene deficiency limits the infiltration of neutrophils and macrophages, with a decreased number of neutrophils expressing C-X-C motif chemokine receptor 4 and C-X-C motif chemokine receptor 5, as well as reduced tumor necrosis factor-α and interferon-γ production. Both cell types exhibit decreased activation of the NOD-like receptor family, pyrin domain containing 3 inflammasome and caspase-1, resulting in lower IL-1β production. This cascade is further supported by dampened activation of nuclear factor kappa B, a transcription factor driving the expression of multiple proinflammatory mediators. In gastric epithelial cells, ST2 deletion decreases apoptosis and pyroptosis, while reduced levels of tumor necrosis factor-α and Beclin-1 suggest potential involvement of necroptosis and autophagy-associated cell death. Altogether, the IL-33/ST2 axis blockade uncouples epithelial injury from downstream innate and adaptive immune activation and limits engagement of multiple regulated cell death pathways, thereby mitigating acute gastric mucosal damage. ST2: Suppression of tumorigenicity 2; IL-33: Interleukin-33; TNF-α: Tumor necrosis factor-α; NF-κB: Nuclear factor kappa B; Neu: Neutrophils; Mφ: Macrophages; DC: Dendritic cell; Tc1: Type 1 cytotoxic; NLRP3: NOD-like receptor family, pyrin domain containing 3; CXCR: C-X-C motif chemokine receptor; IFN: Interferon.
- Citation: Corovic IF, Pantic JM, Stanisavljevic IA, Pavlovic SM, Jovanovic IP, Radosavljevic GD, Simovic Markovic BJ. ST2 gene deficiency alleviates acute gastric injury in mice by modulating inflammation and epithelial cell death. World J Gastroenterol 2026; 32(12): 114576
- URL: https://www.wjgnet.com/1007-9327/full/v32/i12/114576.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i12.114576