Published online Mar 28, 2026. doi: 10.3748/wjg.v32.i12.113939
Revised: October 24, 2025
Accepted: January 28, 2026
Published online: March 28, 2026
Processing time: 192 Days and 22.6 Hours
Ballooned hepatocytes are a histological hallmark in the diagnosis of metabolic dysfunction-associated steatohepatitis (MASH). Identifying ballooned hepa
To investigate the utility of immunostaining for positive SHH protein expression in ballooned hepatocytes in MASH.
Clinicopathological data from hospitalized patients with metabolic dysfunction-associated steatotic liver (MASL) disease at the Second Hospital of Nanjing from January 2020 to November 2022 were analyzed. The Nonalcoholic Steatohepatitis Clinical Research Network scoring system was used. Post-staining, digitized images were acquired, and area quantification algorithms were used to quantify SHH expression.
A total of 190 MASL disease patients who underwent liver biopsy were enrolled in this study; 58.9% (112/190) had definite MASH, and 41.1% (78/190) had MASL. There were significant differences in body mass index (P < 0.001), diabetes (P < 0.01), metabolic syndrome (P < 0.02), and circulating M65 and M30 (P < 0.001), as well as aspartate aminotransferase (AST), alanine aminotransferase, glucose, uric acid, controlled attenuation parameter (CAP), liver stiffness measurement, and nonalcoholic fatty liver disease fibrosis score (P < 0.05). Serum M30 and M65 levels were almost three times greater in MASH than in MASL patients. Hepatic SHH expression correlated with cir
Hepatic SHH protein expression assisted in the diagnosis of MASH. SHH immunostaining may be useful for classifying and quantifying ballooned hepatocytes by artificial intelligence algorithms.
Core Tip: This is a retrospective cohort study of the characteristics of metabolic dysfunction-associated steatohepatitis, in which we found the suggestive role of sonic hedgehog immunostaining in the diagnosis of metabolic dysfunction-associated steatohepatitis. We also found that histological ballooning hepatocytes and circulating M30 as independent predictors of advanced fibrosis.
- Citation: Han X, Chen MY, Xiong QF, Zhong YD, Liu DX, Li J, Yang YF. Role of hepatic sonic hedgehog protein expression in the diagnosis of metabolic dysfunction-associated steatohepatitis. World J Gastroenterol 2026; 32(12): 113939
- URL: https://www.wjgnet.com/1007-9327/full/v32/i12/113939.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i12.113939
Metabolic dysfunction-associated steatotic liver disease (MASLD), previously termed nonalcoholic fatty liver disease (NAFLD), is defined as steatotic liver disease in the presence of one or more cardiometabolic risk factors and the absence of harmful alcohol intake. The spectrum of MASLD includes liver steatosis (MASL), metabolic dysfunction-associated steatohepatitis (MASH), previously termed nonalcoholic steatohepatitis (NASH), fibrosis, cirrhosis, and MASH-related hepatocellular carcinoma (HCC)[1]. The estimated global prevalence of MASLD in the general population has increased from 25% in 2016[2] to > 30% at present, and the incidence is continually increasing[2,3]. Therefore, the substantial socioe
MASH is characterized by histological features of hepatocellular ballooning and lobular inflammation. For the de
Immunohistochemistry (IHC) for cytokeratin 8/18 (CK8/18) has been shown by several groups to enhance the detection of ballooned hepatocytes[7,8]. CK8/18 shows diffuse cytoplasmic expression in normal but not in ballooned hepatocytes, highlighting that Mallory-Denk bodies are present in degenerated hepatocytes[7]. Hepatocyte sonic hed
In the present study, immunostained hepatic SHH was quantified using computer-assisted morphometry[10]. This study aimed to determine whether SHH expression indicates disease severity, as determined by circulating CK18 (M65) and its caspase degradation product (M30), which have been shown to correlate with histologic severity in NAFLD[11], along with pathologic evaluations. Hepatic expression of SHH is associated with ballooned hepatocytes. This study investigated the utility of immunostaining for positive SHH protein expression in ballooned hepatocytes in MASH.
We retrospectively selected hospitalized patients who were diagnosed with MASLD through liver biopsy at the Second Hospital of Nanjing between January 2020 and November 2022. Patients were not required to give informed consent to the study because the analysis used anonymous clinical data that were obtained after each patient agreed to treatment by written consent. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki and was ap
Patients who underwent liver biopsy and staining with hematoxylin and eosin (H&E) and Masson’s trichrome were included. Patients who met the criteria for MASLD, with histopathologically proven liver steatosis and at least one cardiometabolic risk factor[12]: (1) Body mass index (BMI) ≥ 23 kg/m2 or waist circumference ≥ 90/80 cm for Asian men and women; (2) Triglycerides ≥ 1.70 mmol/L or receiving specific drug treatment; (3) Blood pressure ≥ 130/85 mmHg or specific drug treatment; (4) High-density lipoprotein cholesterol ≤ 1.0 mmol/L for men and ≤ 1.3 mmol/L for women; and (5) Type 2 diabetes or prediabetes (prediabetes glycated hemoglobin: 5.7%-6.4%, fasting glucose levels of 5.6 mmol/L to 6.9 mmol/L, or 2 hours glucose levels of 7.8 mmol/L to 11.0 mmol/L). A MAXCORE disposable automatic biopsy needle (16 G, Bard, Tempe, AZ, United States) was used to obtain liver tissue samples through percutaneous liver biopsy. Each liver tissue sample was > 2 cm in length and contained > 10 portal areas.
The exclusion criteria were age < 18 years, inadequate biopsy, excessive alcohol use, and a history of viral hepatitis or other liver diseases.
Age, sex, diagnosis, BMI, systolic blood pressure, liver and kidney function tests, M30, M65, controlled attenuation pa
Each liver biopsy sample was stained with H&E and Masson’s trichrome. The histological features reflecting the degree of steatosis, ballooned hepatocytes, necroinflammatory lesions, and fibrosis resulting from MASH were evaluated ac
Immunohistochemical staining for SHH was performed via a standard procedure with an anti-SHH antibody (ab53281; Abcam, Cambridge, MA, United States) at a dilution of 1:4000. CK8-18 (clone EP17/EP30, dilution 1:50) staining was performed using the Ventana Benchmark. ImmPACT DAB (Vector Laboratories, Newark, CA, United States) was used for detection, and the slides were counterstained with hematoxylin before dehydration and mounting. SHH staining was semi-quantified using 10 × objective low-power fields (100 × magnification) as a percentage of the total surface area and graded into five categories: 1 (< 20%), 2 (20%-39%), 3 (40%-59%), 4 (60%-79%), and 5 (80%). The assessment was com
Slides were scanned at 40 × by use of an Aperio ScanScope system (Aperio, Vista, CA, United States) and digitized images were made available to the hepatopathologist for scoring according to the NASH-CRN scoring system. Two pathologists independently reviewed the images without accessing the clinical information. In cases where two pathologists reached different diagnoses, they rereviewed them for agreement.
Statistical analyses were performed with SPSS 20.0 and GraphPad Prism 8. Variables with skewed distributions are expressed as the range and median, whereas variables with normal distributions are expressed as the mean ± SD. The clinical data and some pathological features were analyzed via parametric tests, such as the t test, and nonparametric tests, such as the χ2 test, Kruskal-Wallis test, and Spearman rank correlation (according to the data type). Binary logistic regression analysis was subsequently performed to identify the risk factors for MASH and advanced fibrosis in patients with MASLD. Interobserver agreement was analyzed using Fleiss’ kappa. P ≤ 0.05 was considered statistically significant.
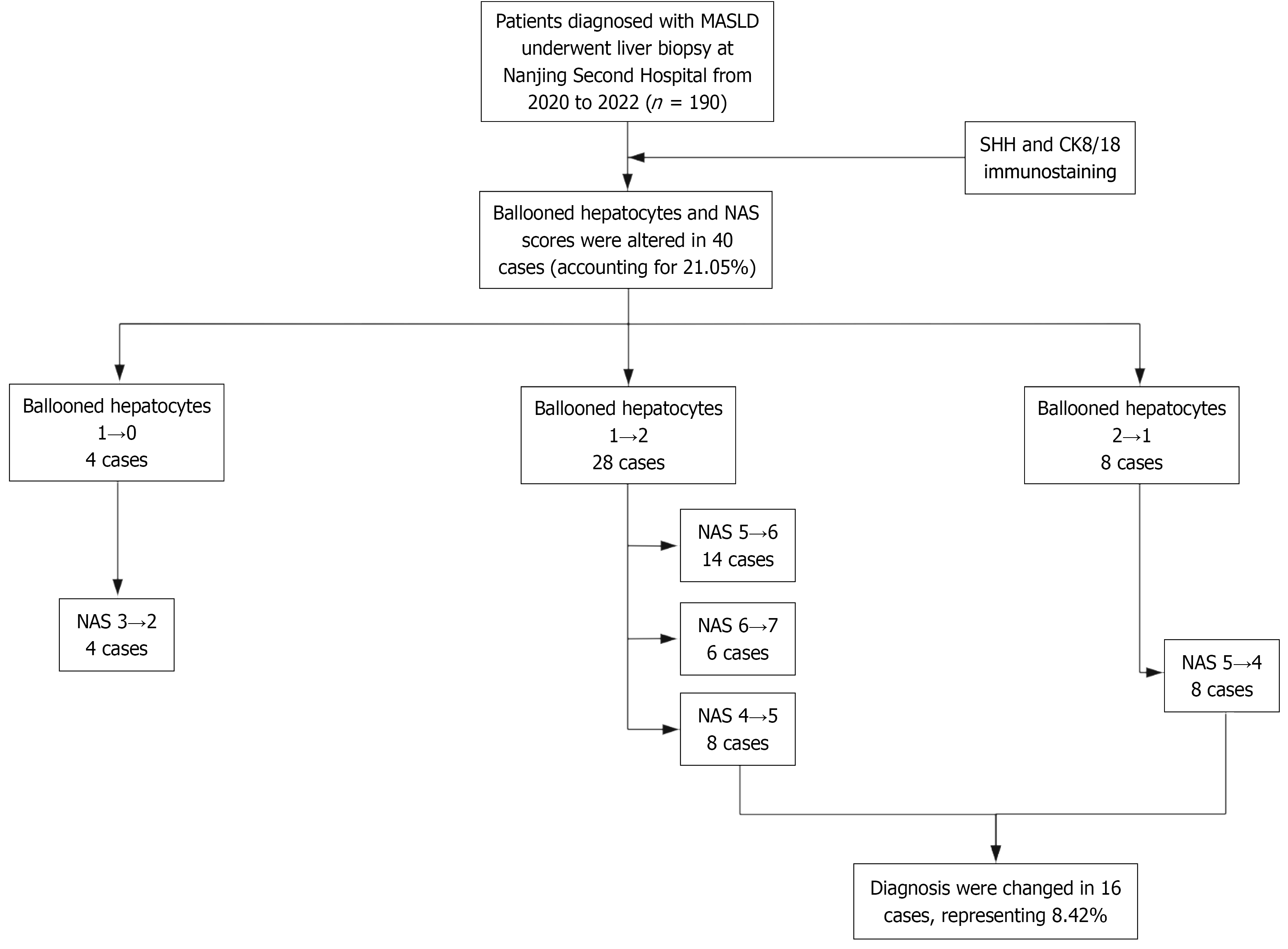
A total of 190 MASLD patients who underwent liver biopsy were enrolled. The histological features reflected the degree of steatosis, ballooned hepatocytes, inflammatory lesions, and fibrosis according to the NAS system. An NAS score ≥ 5 correlated with a diagnosis of MASH[13]. These evaluations were performed using routine H&E staining, followed by immunostaining for SHH and CK8/18. Compared with H&E staining, SHH and CK8/18 immunostaining resulted in an increase in ballooned hepatocyte scores in 28 patients and a decrease in 12. NAS scores were altered in 40 patients, accounting for 21.05% of the cohort, whereas diagnoses were changed in 16 patients, representing 8.42% of the cohort (Figure 1).
The clinical information is summarized in Table 1. Overall, 58.9% (112/190) of patients had definite MASH, whereas 41.1% (78/190) were diagnosed with MASL. The mean BMI of the MASH patients was 25.75 kg/m2, and 21.43%-38.74% of patients suffered from diabetes or metabolic syndrome. Significant differences were observed in BMI (P < 0.001), diabetes (P < 0.01), metabolic syndrome (P < 0.02), circulating M65 and M30 (P < 0.001), and aspartate aminotransferase (AST), alanine aminotransferase, glucose, uric acid, CAP, liver stiffness measurement, and NAFLD fibrosis score (P < 0.05). Serum M30 and M65 levels were almost three times greater in MASH than in non-MASH patients.
| MASL (n = 78) | MASH (n = 112) | P value | |
| Sex (male) | 48 (61.54) | 57 (50.89) | 0.147 |
| Age (year) | 45.54 ± 12.93 | 43.77 ± 15.34 | 0.391 |
| BMI (kg/m2, median) | 23.31 (22.2, 25.9) | 25.75 (23.9, 27.9) | 0.000 |
| Hypertension | 16 (20.51) | 34 (30.36) | 0.130 |
| Diabetes | 5 (6.14) | 24 (21.43) | 0.005 |
| Metabolic syndrome | 17 (21.52) | 43 (38.74) | 0.011 |
| ALT (U/L, median) | 49.60 (29.6, 87.1) | 98.60 (69.2, 163.1) | 0.000 |
| AST (U/L, median) | 28.750 (20.5, 40.1) | 56.550 (39.8, 81.0) | 0.000 |
| GGT (U/L, median) | 86.950 (28.9, 207.1) | 80.75 (45.2, 127.0) | 0.520 |
| ALP (U/L, median) | 91.70 (73.0, 117.0) | 86.30 (71.6, 105.0) | 0.261 |
| ALB | 43.90 (41.5, 47.1) | 45.050 (42.5, 47.0) | 0.341 |
| TBIL | 13.450 (9.6, 18.3) | 13.300 (9.6, 17.1) | 0.610 |
| BUN | 4.700 (4.1, 5.8) | 4.460 (3.8, 5.5) | 0.143 |
| Cr | 68.0 (58.0, 81.5) | 65.50 (52.8, 77.0) | 0.132 |
| UA | 348.0 (272.8, 402.0) | 364.50 (299.5, 449.0) | 0.028 |
| TG | 1.710 (1.1, 2.3) | 1.570 (1.3, 2.2) | 0.823 |
| TC | 4.50 (3.9, 5.2) | 4.760 (4.3, 5.5) | 0.078 |
| LDL | 2.89 ± 0.78 | 3.10 ± 0.82 | 0.092 |
| HDL | 1.11 ± 0.26 | 1.09 ± 0.21 | 0.602 |
| GLU | 5.240 (4.7, 5.6) | 5.425 (4.9, 6.1) | 0.011 |
| M30 | 153.0 (153.0, 228.6) | 476.0 (391.1, 549.0) | 0.000 |
| M65 | 168.0 (153.3, 282.0) | 579.0 (537.1, 621.3) | 0.000 |
| CAP | 250.0 (228.5, 261.0) | 291.0 (276.5, 313.0) | 0.000 |
| LSM | 5.050 (4.1, 6.7) | 7.40 (4.7, 10.2) | 0.003 |
| NFS | 2.283 (1.8, 2.6) | 2.626 (2.0, 3.2) | 0.006 |
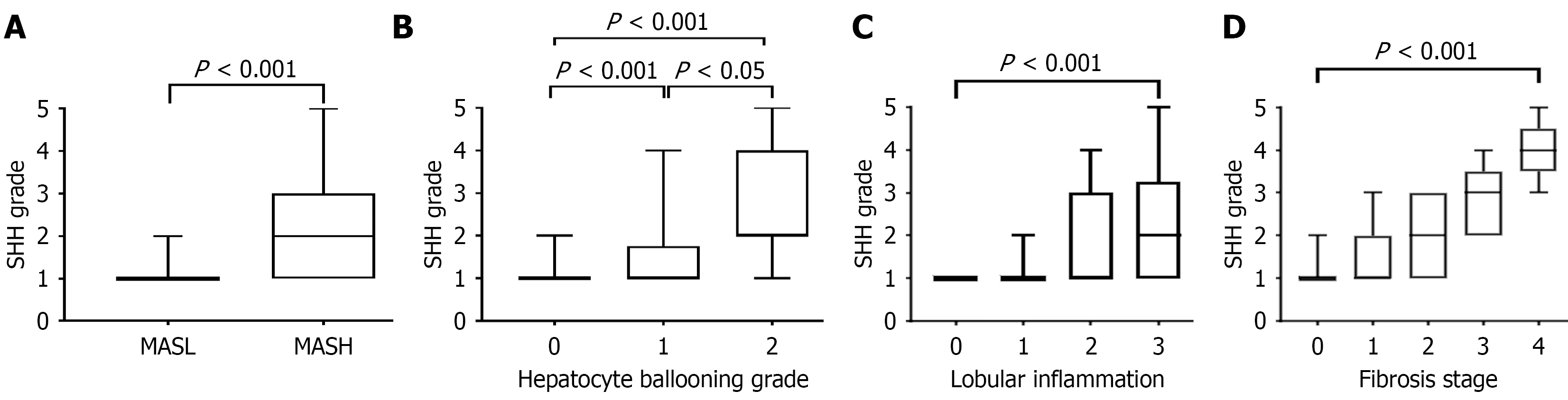
The pathological characteristics of the study population are summarized in Table 2. In the MASH group, 56.25% of patients had grade 2 hepatocyte ballooning, and 62.50% had advanced fibrosis (F2-F4). Hepatic SHH expression corre
| MASL (n = 78) | MASH (n = 112) | P value | |
| Steatosis | 0.000 | ||
| Score 1 | 71 (91.03) | 21 (18.75) | |
| Score 2 | 6 (7.69) | 50 (44.64) | |
| Score 3 | 1 (1.28) | 41 (36.61) | |
| Ballooned hepatocytes with H&E staining and SHH + CK8/18 immunostaining | 0.000 | ||
| Score 0 | 37 (47.44) | 0 | |
| Score 1 | 41 (52.56) | 49 (43.75) | |
| Score 2 | 0 | 63 (56.25) | |
| Lobular inflammation | 0.000 | ||
| Score 0 | 2 (2.56) | 0 | |
| Score 1 | 56 (71.79) | 4 (3.57) | |
| Score 2 | 19 (24.36) | 48 (42.86) | |
| Score 3 | 1 (1.28) | 60 (53.57) | |
| Fibrosis | 0.000 | ||
| Score 0 | 39 (50.0) | 11 (9.82) | |
| Score 1 | 30 (38.46) | 31 (27.68) | |
| Score 2 | 8 (10.26) | 32 (28.57) | |
| Score 3 | 1 (1.28) | 30 (26.79) | |
| Score 4 | 0 | 8 (7.14) |
The Froward Wald test was used in binary logistic regression analysis, and the final data included in the equation are shown in Tables 3 and 4. Logistic modeling indicated the following. Diabetes, AST, CAP, and hepatic SHH expression were independent predictors of MASH [defined as NAS score ≥ 5: Odds ratio (OR) = 20.95, P = 0.043, OR = 1.044, P = 0.023, OR = 1.034, P = 0.008, and OR = 7.151, P = 0.017, respectively]. Histological ballooning hepatocytes and circulating M30 were independent predictors of advanced fibrosis (defined as portal and pericellular fibrosis ≥ 2: OR = 6.440, P = 0.023, and OR = 1.012, P = 0.005, respectively).
| B | SE | Wald | df | Sig | Exp(B) | 95%CI for Exp(B) | |
| Diabetes | 3.042 | 1.504 | 4.094 | 1 | 0.043 | 20.95 | 1.100-399.132 |
| AST | 0.043 | 0.019 | 5.176 | 1 | 0.023 | 1.044 | 1.006-1.083 |
| CAP | 0.033 | 0.013 | 6.991 | 1 | 0.008 | 1.034 | 1.009-1.059 |
| SHH | 1.967 | 0.822 | 5.722 | 1 | 0.017 | 7.151 | 1.427-35.848 |
| Constant | -13.551 | 3.737 | 13.149 | 1 | 0.000 | 0.001 | - |
| B | SE | Wald | df | Sig | Exp(B) | 95%CI for Exp(B) | |
| Ballooned hepatocytes | 1.863 | 0.820 | 5.161 | 1 | 0.023 | 6.440 | 1.291-32.114 |
| M30 | 0.012 | 0.004 | 7.871 | 1 | 0.005 | 1.012 | 1.004-1.021 |
| Constant | 2.657 | 3.721 | 0.510 | 1 | 0.000 | 14.247 | - |
Fleiss’ kappa confirmed an increased level of agreement in interobserver ballooning assessment by use of SHH immunostaining, from 0.65 to 0.85.
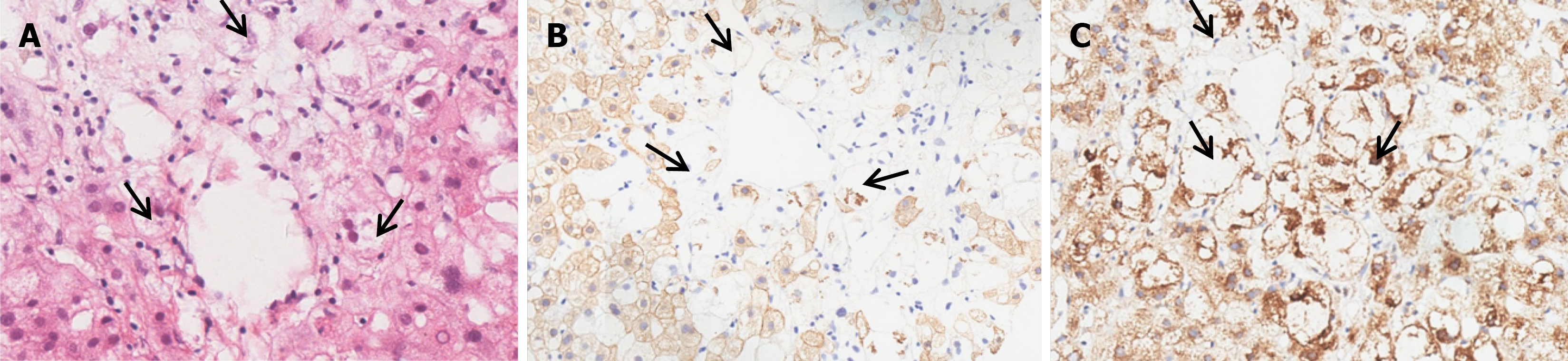
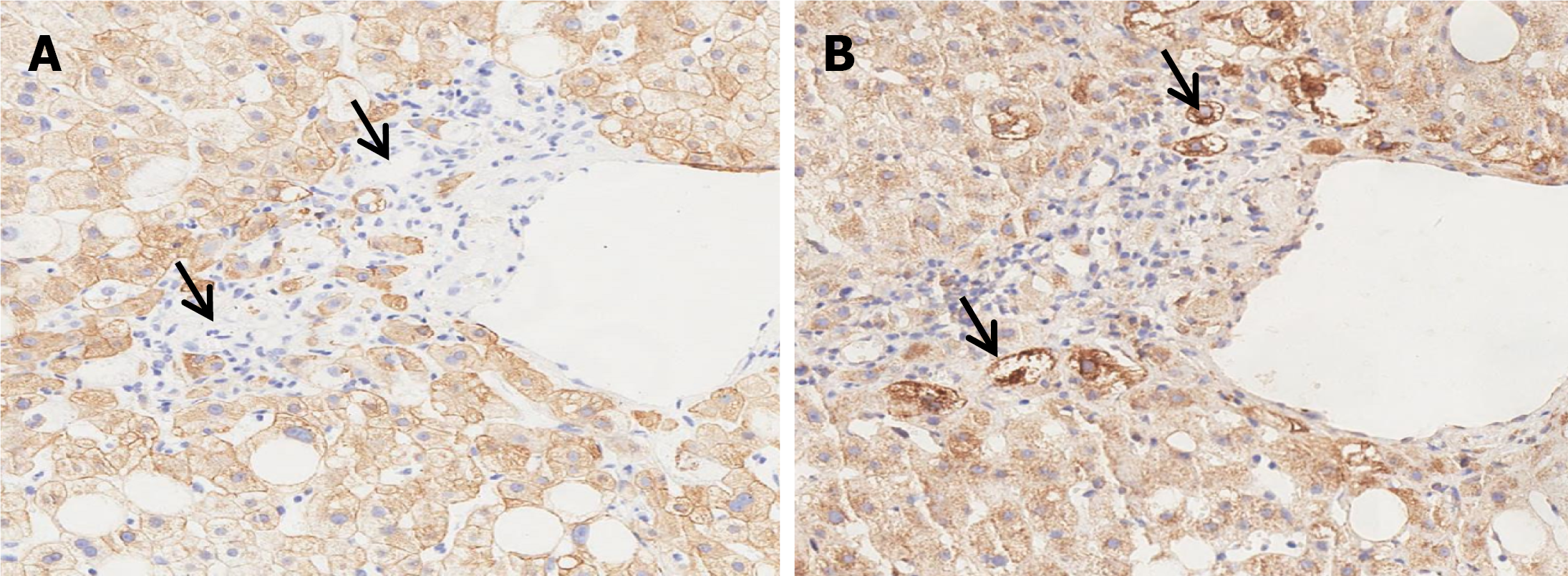
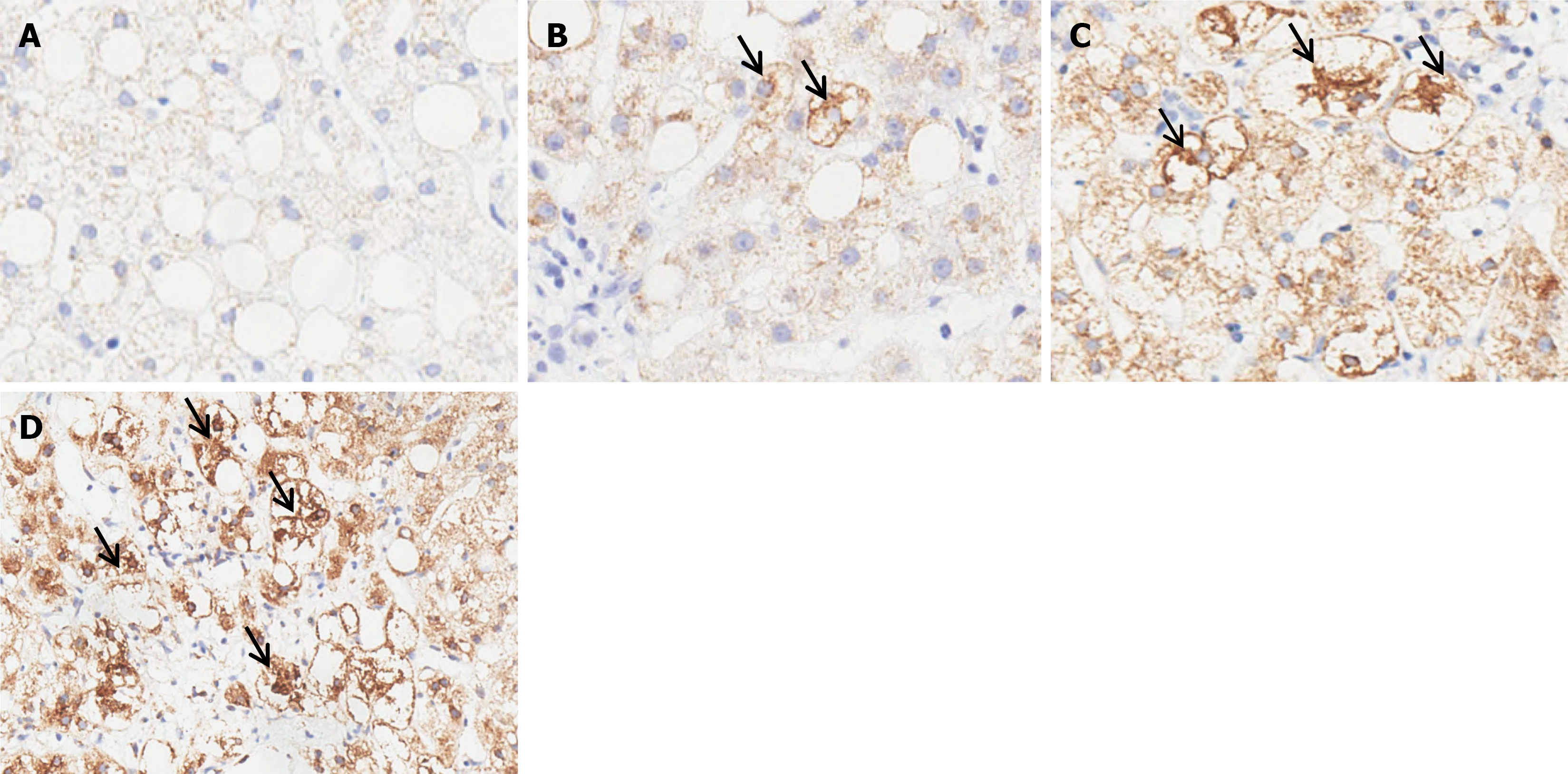
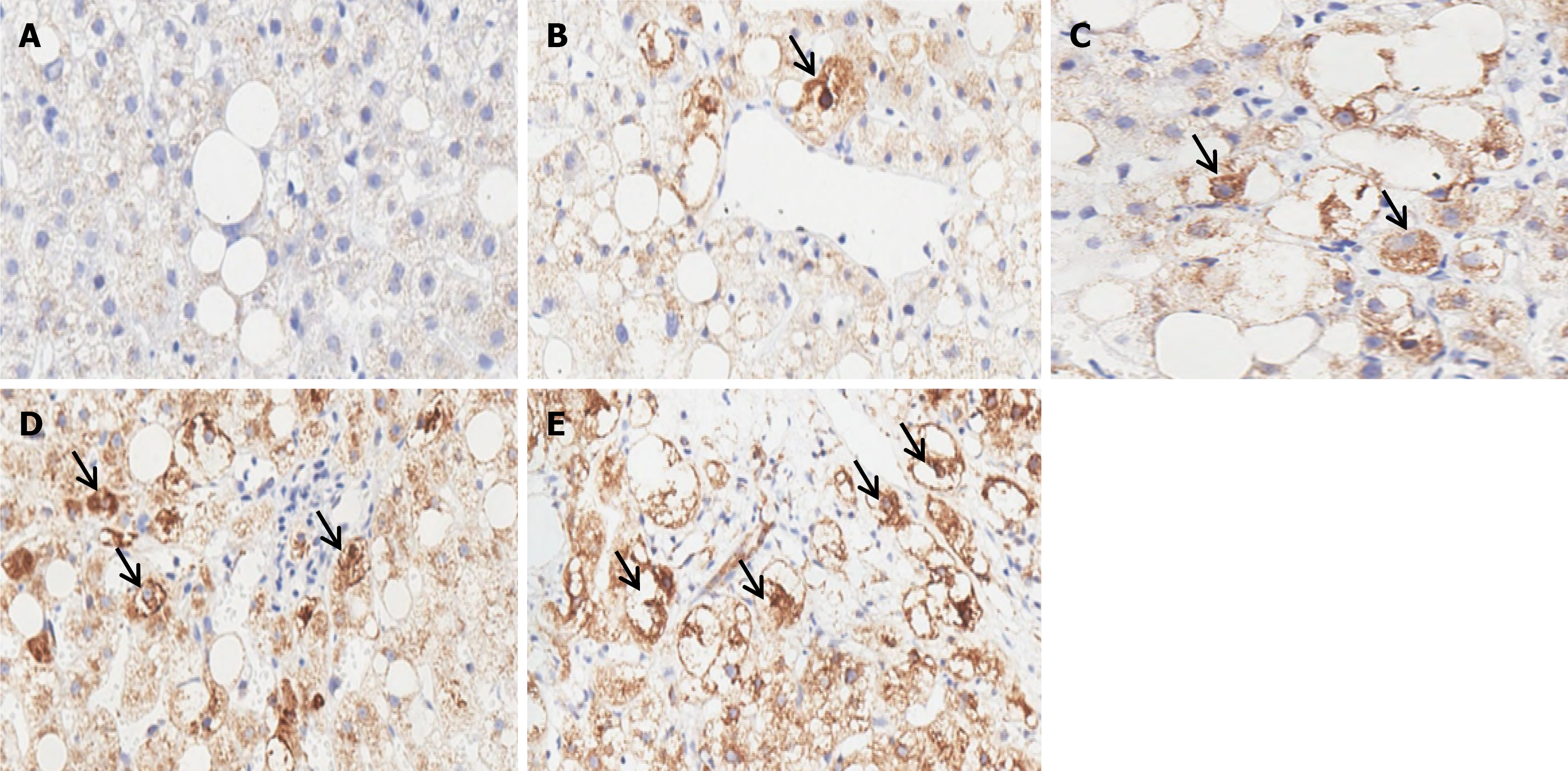
MASH is histologically characterized by lipid deposition in hepatocytes, ballooning hepatocytes, inflammatory injury and fibrotic changes in biopsy tissues. In the inflammatory foci, ballooned hepatocytes were typically interwoven with inflammatory cells (Figure 2A). Ballooned hepatocytes were identified as enlarged hepatocytes that lost cell polarity and frequently contained Mallory-Denk bodies (Figure 2A). Ballooned hepatocytes presented a loss of CK8/18 expression, whereas other nonballooned hepatocytes presented diffuse positivity for CK8/18 (Figures 2B and 3A). SHH immunostaining highlighted ballooned hepatocytes, whereas other nonballooned hepatocytes were negative for SHH (Figures 2C and 3B). SHH was strongly expressed in the cytoplasm of injured ballooned hepatocytes. Additionally, SHH expression was plotted relative to the degree of hepatocyte ballooning (Figure 4), as scored by the NASH-CRN Pathology Com
This study demonstrated that immunostaining for SHH and CK8/18 aids in the detection of ballooned hepatocytes, which are histological hallmarks of MASH. The scores for ballooned hepatocytes were increased in 28 (14.74%) patients and decreased in six (3.16%) with SHH and CK8/18 immunostaining, respectively. CK8/18 and SHH exhibit distinct expression profiles in normal vs ballooned hepatocytes. CK8/18 is diffusely expressed in the cytoplasm of normal hepatocytes but absent in ballooned cells. In contrast, SHH expression is undetectable in normal hepatocytes but present in ballooned hepatocytes. Pathologists must look for a small number of CK8/18-negative hepatocytes from among the CK8/18-positive hepatocyte population. Fleiss’ kappa also confirmed increased agreement in interobserver assessment of ballooning using SHH immunostaining.
This study revealed that increased SHH protein expression was significantly correlated with severity of hepatocyte ballooning, lobular inflammation, and fibrotic progression in MASH tissues. Questions have been raised regarding the applicability of evidence derived based on the NAFLD definition to patients with MASLD. Re-examinations from several existing cohort studies confirm that NAFLD-related findings can be fully extrapolated to individuals with MASLD[14]. In another investigation, a transgenic mouse model was employed, where a transposon carrying the SHH gene was hydro
Patients with NASH and advanced fibrosis (F2-F4 fibrosis) are at increased risk for liver-related events and mortality[16,17]. This study identified diabetes, AST, CAP, and hepatic SHH expression as independent predictors of MASH, whereas histological ballooning hepatocytes and circulating M30 were identified as independent predictors of advanced fibrosis. Additionally, circulating M30 and M65 levels were significantly higher in MASH than in MASL patients, suggesting their potential as markers for histological ballooning. In this study, circulating M30 and M65 were signi
The SHH signaling pathway is involved in endoplasmic reticulum stress, oxidative stress, and lipotoxic injury in the liver. These cell stress types induce hepatocyte damage in NASH[18]. Mechanistically, ballooned hepatocytes indicate sustained, strong endoplasmic reticulum stress and oxidative stress in NASH patients and are a major SHH production source. In addition, a reduction in SHH pathway activity by inhibitors may improve NASH in human and mouse models[19]. Therefore, in the current study, injured hepatocytes may have secreted SHH ligands, which recruited and activated inflammatory cells while inducing the proliferation of ductular/progenitor cells. Conversely, activated stromal cells enhanced the expression of chemokines and cytokines, leading to the recruitment of additional inflammatory cells and the accumulation of myofibroblasts, thereby accelerating the fibrogenic process. A recent study revealed that SHH secreted through damaged hepatocytes may result in the activation of transforming growth factor-β1 and the subsequent transformation of hepatic stellate cells, which together modulate progression of human NASH[20].
Our study had several limitations. First, it was a single-center retrospective study, where selection bias could occur, and incorporating prospective validation data from a multi-center cohort studies is needed to validate the results. Second, we could not obtain data on the amount of circulating SHH. In a related study[10], no correlations were found between circulating serum and hepatic SHH signals. This finding suggests the possibility of derangements in SHH post-translational modifications in ballooning hepatocytes, perhaps in the cholesterol addition/cleavage process of full-length SHH into N and C fragments or in the proteasome degradation of SHH C-terminal fragment. If so, accumulation of SHH or SHH C-terminal fragment within ballooning hepatocytes could theoretically be a marker for MASLD patients in danger of progressive fibrosis. The mechanistic insights into how SHH contributes to MASH progression require further elucidation. Third, we did not include follow-up data to analyze whether SHH expression is associated with disease pro
Artificial intelligence algorithms for assessing steatosis and fibrosis are the most advanced methods as the physicochemical characteristics of lipid droplets and collagen facilitate their automated identification[21,22]. In contrast, lobular and portal inflammation, along with various forms of hepatocyte injury, such as ballooning, is difficult in correct classification and quantification by artificial intelligence algorithms but is under development. SHH immunostaining may be useful for classifying and quantifying ballooned hepatocytes by artificial intelligence algorithms. Furthermore, dedicated mechanistic experiments investigating the precise role of the SHH signaling pathway in hepatocyte injury and fibro
The findings of this study highlight the utility of SHH immunostaining in detecting ballooned hepatocytes and predicting disease severity in MASH. SHH and CK8/18 immunostaining can be feasibly incorporated into routine clinical practice. Our findings are a step toward the goal of diminishing interobserver variability as it pertains to ballooning assessment and MASH diagnosis. SHH immunostaining may be useful for classifying and quantifying ballooned hepatocytes by artificial intelligence algorithms. Further research is warranted to validate these findings and explore the potential the
We thank all participants from the Second Hospital of Nanjing for their help in this study.
| 1. | European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). J Hepatol. 2024;81:492-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1461] [Cited by in RCA: 1230] [Article Influence: 615.0] [Reference Citation Analysis (2)] |
| 2. | Younossi ZM, Golabi P, Paik JM, Henry A, Van Dongen C, Henry L. The global epidemiology of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH): a systematic review. Hepatology. 2023;77:1335-1347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2563] [Cited by in RCA: 2258] [Article Influence: 752.7] [Reference Citation Analysis (6)] |
| 3. | Quek J, Chan KE, Wong ZY, Tan C, Tan B, Lim WH, Tan DJH, Tang ASP, Tay P, Xiao J, Yong JN, Zeng RW, Chew NWS, Nah B, Kulkarni A, Siddiqui MS, Dan YY, Wong VW, Sanyal AJ, Noureddin M, Muthiah M, Ng CH. Global prevalence of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in the overweight and obese population: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2023;8:20-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 563] [Cited by in RCA: 485] [Article Influence: 161.7] [Reference Citation Analysis (1)] |
| 4. | Karlsen TH, Sheron N, Zelber-Sagi S, Carrieri P, Dusheiko G, Bugianesi E, Pryke R, Hutchinson SJ, Sangro B, Martin NK, Cecchini M, Dirac MA, Belloni A, Serra-Burriel M, Ponsioen CY, Sheena B, Lerouge A, Devaux M, Scott N, Hellard M, Verkade HJ, Sturm E, Marchesini G, Yki-Järvinen H, Byrne CD, Targher G, Tur-Sinai A, Barrett D, Ninburg M, Reic T, Taylor A, Rhodes T, Treloar C, Petersen C, Schramm C, Flisiak R, Simonova MY, Pares A, Johnson P, Cucchetti A, Graupera I, Lionis C, Pose E, Fabrellas N, Ma AT, Mendive JM, Mazzaferro V, Rutter H, Cortez-Pinto H, Kelly D, Burton R, Lazarus JV, Ginès P, Buti M, Newsome PN, Burra P, Manns MP. The EASL-Lancet Liver Commission: protecting the next generation of Europeans against liver disease complications and premature mortality. Lancet. 2022;399:61-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 542] [Cited by in RCA: 484] [Article Influence: 121.0] [Reference Citation Analysis (2)] |
| 5. | Loomba R, Li S, Peng Y, Wang X, Harrison S. Health care costs are double for non-alcoholic fatty liver disease non-alcoholic steatohepatitis patients with compensated cirrhosis who progress to end-stage liver disease. J Hepatol. 2018;68:S719-S720. [DOI] [Full Text] |
| 6. | Davison BA, Harrison SA, Cotter G, Alkhouri N, Sanyal A, Edwards C, Colca JR, Iwashita J, Koch GG, Dittrich HC. Suboptimal reliability of liver biopsy evaluation has implications for randomized clinical trials. J Hepatol. 2020;73:1322-1332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 384] [Cited by in RCA: 342] [Article Influence: 57.0] [Reference Citation Analysis (2)] |
| 7. | Guy CD, Suzuki A, Burchette JL, Brunt EM, Abdelmalek MF, Cardona D, McCall SJ, Ünalp A, Belt P, Ferrell LD, Diehl AM; Nonalcoholic Steatohepatitis Clinical Research Network. Costaining for keratins 8/18 plus ubiquitin improves detection of hepatocyte injury in nonalcoholic fatty liver disease. Hum Pathol. 2012;43:790-800. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 8. | Lackner C, Gogg-Kamerer M, Zatloukal K, Stumptner C, Brunt EM, Denk H. Ballooned hepatocytes in steatohepatitis: the value of keratin immunohistochemistry for diagnosis. J Hepatol. 2008;48:821-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 158] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 9. | Guy CD, Suzuki A, Zdanowicz M, Abdelmalek MF, Burchette J, Unalp A, Diehl AM; NASH CRN. Hedgehog pathway activation parallels histologic severity of injury and fibrosis in human nonalcoholic fatty liver disease. Hepatology. 2012;55:1711-1721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 170] [Article Influence: 12.1] [Reference Citation Analysis (2)] |
| 10. | Estep M, Mehta R, Bratthauer G, Alaparthi L, Monge F, Ali S, Abdelatif D, Younoszai Z, Stepanova M, Goodman ZD, Younossi ZM. Hepatic sonic hedgehog protein expression measured by computer assisted morphometry significantly correlates with features of non-alcoholic steatohepatitis. BMC Gastroenterol. 2019;19:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 11. | Shen J, Chan HL, Wong GL, Chan AW, Choi PC, Chan HY, Chim AM, Yeung DK, Yu J, Chu WC, Wong VW. Assessment of non-alcoholic fatty liver disease using serum total cell death and apoptosis markers. Aliment Pharmacol Ther. 2012;36:1057-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 56] [Article Influence: 4.0] [Reference Citation Analysis (2)] |
| 12. | Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, Romero D, Abdelmalek MF, Anstee QM, Arab JP, Arrese M, Bataller R, Beuers U, Boursier J, Bugianesi E, Byrne CD, Castro Narro GE, Chowdhury A, Cortez-Pinto H, Cryer DR, Cusi K, El-Kassas M, Klein S, Eskridge W, Fan J, Gawrieh S, Guy CD, Harrison SA, Kim SU, Koot BG, Korenjak M, Kowdley KV, Lacaille F, Loomba R, Mitchell-Thain R, Morgan TR, Powell EE, Roden M, Romero-Gómez M, Silva M, Singh SP, Sookoian SC, Spearman CW, Tiniakos D, Valenti L, Vos MB, Wong VW, Xanthakos S, Yilmaz Y, Younossi Z, Hobbs A, Villota-Rivas M, Newsome PN; NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. J Hepatol. 2023;79:1542-1556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2209] [Cited by in RCA: 1961] [Article Influence: 653.7] [Reference Citation Analysis (0)] |
| 13. | Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, Ferrell LD, Liu YC, Torbenson MS, Unalp-Arida A, Yeh M, McCullough AJ, Sanyal AJ; Nonalcoholic Steatohepatitis Clinical Research Network. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9231] [Cited by in RCA: 8592] [Article Influence: 409.1] [Reference Citation Analysis (4)] |
| 14. | Younossi ZM, Paik JM, Stepanova M, Ong J, Alqahtani S, Henry L. Clinical profiles and mortality rates are similar for metabolic dysfunction-associated steatotic liver disease and non-alcoholic fatty liver disease. J Hepatol. 2024;80:694-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 224] [Article Influence: 112.0] [Reference Citation Analysis (1)] |
| 15. | Chung SI, Moon H, Ju HL, Cho KJ, Kim DY, Han KH, Eun JW, Nam SW, Ribback S, Dombrowski F, Calvisi DF, Ro SW. Hepatic expression of Sonic Hedgehog induces liver fibrosis and promotes hepatocarcinogenesis in a transgenic mouse model. J Hepatol. 2016;64:618-627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 92] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 16. | Dulai PS, Singh S, Patel J, Soni M, Prokop LJ, Younossi Z, Sebastiani G, Ekstedt M, Hagstrom H, Nasr P, Stal P, Wong VW, Kechagias S, Hultcrantz R, Loomba R. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: Systematic review and meta-analysis. Hepatology. 2017;65:1557-1565. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1666] [Cited by in RCA: 1545] [Article Influence: 171.7] [Reference Citation Analysis (1)] |
| 17. | Rinella ME, Neuschwander-Tetri BA, Siddiqui MS, Abdelmalek MF, Caldwell S, Barb D, Kleiner DE, Loomba R. AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology. 2023;77:1797-1835. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1980] [Cited by in RCA: 1760] [Article Influence: 586.7] [Reference Citation Analysis (2)] |
| 18. | Kakisaka K, Cazanave SC, Werneburg NW, Razumilava N, Mertens JC, Bronk SF, Gores GJ. A hedgehog survival pathway in 'undead' lipotoxic hepatocytes. J Hepatol. 2012;57:844-851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 19. | Chen Y, Choi SS, Michelotti GA, Chan IS, Swiderska-Syn M, Karaca GF, Xie G, Moylan CA, Garibaldi F, Premont R, Suliman HB, Piantadosi CA, Diehl AM. Hedgehog controls hepatic stellate cell fate by regulating metabolism. Gastroenterology. 2012;143:1319-1329.e11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 231] [Cited by in RCA: 236] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 20. | Zhou X, Wang P, Ma Z, Li M, Teng X, Sun L, Wan G, Li Y, Guo L, Liu H. Novel Interplay Between Sonic Hedgehog and Transforming Growth Factor-β1 in Human Nonalcoholic Steatohepatitis. Appl Immunohistochem Mol Morphol. 2020;28:154-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 21. | Liu F, Goh GB, Tiniakos D, Wee A, Leow WQ, Zhao JM, Rao HY, Wang XX, Wang Q, Wan WK, Lim KH, Romero-Gomez M, Petta S, Bugianesi E, Tan CK, Harrison SA, Anstee QM, Chang PJ, Wei L. qFIBS: An Automated Technique for Quantitative Evaluation of Fibrosis, Inflammation, Ballooning, and Steatosis in Patients With Nonalcoholic Steatohepatitis. Hepatology. 2020;71:1953-1966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 97] [Article Influence: 16.2] [Reference Citation Analysis (1)] |
| 22. | Taylor-Weiner A, Pokkalla H, Han L, Jia C, Huss R, Chung C, Elliott H, Glass B, Pethia K, Carrasco-Zevallos O, Shukla C, Khettry U, Najarian R, Taliano R, Subramanian GM, Myers RP, Wapinski I, Khosla A, Resnick M, Montalto MC, Anstee QM, Wong VW, Trauner M, Lawitz EJ, Harrison SA, Okanoue T, Romero-Gomez M, Goodman Z, Loomba R, Beck AH, Younossi ZM. A Machine Learning Approach Enables Quantitative Measurement of Liver Histology and Disease Monitoring in NASH. Hepatology. 2021;74:133-147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 174] [Cited by in RCA: 138] [Article Influence: 27.6] [Reference Citation Analysis (0)] |