Published online Mar 18, 2026. doi: 10.13105/wjma.v14.i1.116322
Revised: December 4, 2025
Accepted: December 23, 2025
Published online: March 18, 2026
Processing time: 122 Days and 19.4 Hours
Open tibial fractures with extensive soft-tissue loss represent one of the most cha
To determine the impact of early vs delayed soft-tissue coverage on infection, union, flap survival, and limb salvage.
A systematic review was performed using PubMed, Scopus, Web of Science, and Google Scholar for studies published from January 2000 to October 2025. Eligible studies included adults (≥ 18 years) with Gustilo-Anderson type IIIB or IIIC open tibial fractures comparing early (≤ 72 hours or ≤ 7-12 days) and delayed flap cove
Nine retrospective cohort studies comprising 900 patients were included. Early flap coverage significantly reduced deep infection rates (0%-23% vs up to 72%) and lowered reoperation frequency compared with delayed coverage. Union and limb salvage rates exceeded 90% in early coverage groups, with no increase in flap failure risk. Studies consistently highlighted improved outcomes when early coverage was performed by coordinated orthoplastic teams within specialized trauma centers. The overall methodological quality ranged from moderate to low risk of bias, supporting the clinical value of timely, multidisciplinary management.
Early orthoplastic soft-tissue coverage within the first week after fixation reduces infection and nonunion, im
Core Tip: Early orthoplastic management of open tibial fractures remains crucial for reducing complications and preserving limb function. This systematic review synthesizes current evidence comparing early vs delayed soft-tissue coverage in Gustilo-Anderson type IIIB and IIIC fractures. Early flap coverage, ideally within the first week after fixation, markedly decreases deep infection and nonunion rates, improves union and limb salvage, and does not increase flap failure risk. The findings reinforce the importance of coordinated orthoplastic collaboration and timely multidisciplinary care in achieving optimal outcomes for complex lower-limb trauma.
- Citation: Mohamed EHH, Ahmad S. Early orthoplastic soft-tissue coverage in open tibial fractures reduces infection, nonunion, and amputation. World J Meta-Anal 2026; 14(1): 116322
- URL: https://www.wjgnet.com/2308-3840/full/v14/i1/116322.htm
- DOI: https://dx.doi.org/10.13105/wjma.v14.i1.116322
Open tibial fractures represent one of the most challenging orthopedic injuries, often resulting from high-energy trauma and frequently accompanied by extensive soft-tissue loss, contamination, and bone exposure[1]. Because the tibia is subcutaneous along most of its length, its vascularity is easily compromised, predisposing these injuries to infection, delayed healing, and nonunion. The management of such fractures demands meticulous coordination between or
Historically, management strategies followed a staged approach involving serial debridements, delayed fixation, and late flap coverage once the wound appeared “clean”. However, this often prolonged hospitalization, increased no
The orthoplastic model now emphasizes stable skeletal fixation, radical debridement, and early, definitive soft-tissue coverage - ideally in a single operative setting (“fix-and-flap”). Well-vascularized tissue not only protects underlying bone and implants but also enhances osteogenesis by delivering osteoinductive and angiogenic factors[7]. Despite widespread advocacy for early coverage, variations persist due to surgical resources, institutional logistics, and injury severity. Moreover, the precise influence of coverage timing on bone union remains debated, with some studies favoring early intervention and others reporting comparable long-term outcomes with delayed reconstruction under optimal infection control[8,9].
These variations raise important clinical questions: How critical is the timing of flap coverage to achieving successful bone union? What are the quantifiable benefits of early vs delayed coverage in terms of infection, union, and limb salvage? The objective of this study is to systematically evaluate the impact of early flap coverage within orthoplastic ma
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2009 guidelines[8]. The review aimed to synthesize evidence from observational studies evaluating the timing of soft-tissue coverage and its effect on outcomes in severe open tibial fractures. The protocol was developed a priori and structured using the PICO (Patient, Intervention, Comparison, Outcome) framework[9], defining the population as adult patients with Gustilo-Anderson type IIIB or IIIC open tibial fractures; the Intervention as early definitive soft-tissue coverage or orthoplastic “fix-and-flap” management; the comparison as delayed coverage or staged debridement strategies; and the outcomes as rates of deep infection, nonunion, flap failure, reoperation, and limb salvage.
A comprehensive literature search was performed across PubMed (MEDLINE), Scopus, Web of Science, and Google Scholar to identify studies published from January 2000 to October 2025. The search strategy combined both medical subject headings (MeSH) and free-text terms to maximize retrieval sensitivity and specificity. Boolean operators (“AND”, “OR”) and truncation symbols (*) were applied to capture variations in terminology.
The core PubMed search string was: [“open tibia fracture”(MeSH Terms) OR “open tibial fracture*” OR “compound tibia fracture*” OR “Gustilo type III* fracture*”] AND [“soft tissue coverage”(MeSH Terms) OR “flap coverage” OR “flap reconstruction” OR “orthoplastic” OR “fix and flap” OR “early coverage” OR “flap timing”] AND (“infection” OR “nonunion” OR “bone healing” OR “limb salvage” OR “outcome*”).
Scopus search string: [TITLE-ABS-KEY(“open tibial fracture” OR “open tibia fracture” OR “compound tibia fracture*” OR “Gustilo type III* fracture*”)] AND [TITLE-ABS-KEY(“soft tissue coverage” OR “flap coverage” OR “flap recon
Web of Science search string: TS = (“open tibial fracture” OR “open tibia fracture” OR “compound tibia fracture*” OR “Gustilo type III* fracture*”) AND TS = (“soft tissue coverage” OR “flap coverage” OR “flap reconstruction” OR “or
Google Scholar search string: (“open tibial fracture” OR “open tibia fracture” OR “compound tibia fracture” OR “Gustilo type IIIB fracture” OR “Gustilo type IIIC fracture”) AND (“soft tissue coverage” OR “flap coverage” OR “flap re
The final literature search across all databases was completed on October 15, 2025. Publications were restricted to English language only, which may have resulted in the exclusion of relevant non-English studies and is acknowledged as a potential source of language and publication bias. Filters were applied to include only human studies, adult populations (≥ 18 years), and publications in English. Reference lists of included studies and relevant reviews were also manually screened to identify additional eligible articles. All retrieved citations were imported into Mendeley for deduplication before screening.
Studies were included if they involved adult patients aged 18 years or older with open tibial fractures classified as Gustilo-Anderson type IIIB or IIIC, compared early vs delayed flap coverage or reported outcomes based on timing of coverage, and reported at least one of the predefined outcomes: Infection, nonunion, flap failure, or limb salvage. Studies were excluded if they were case reports, conference abstracts, editorials, reviews without original data, or investigations involving pediatric or non-tibial fractures.
Two reviewers independently screened all titles, abstracts, and full texts according to predefined inclusion and exclusion criteria, with any disagreements resolved through discussion and consensus. Data extraction was performed using a standardized form that included study design, publication year, country or setting, sample size, patient demographics, definition of early coverage, type of fixation, flap type, and primary outcomes. With regard to timing, both strict time-based definitions (≤ 72 hours, ≤ 7 days, ≤ 12 days) and biological definitions (number of debridements prior to definitive coverage) were captured during data extraction to allow comparative interpretation. When critical data were incomplete or unclear, attempts were made to contact the corresponding authors for clarification or missing information.
The methodological quality and risk of bias of the included studies were assessed independently by two reviewers. For cohort and case-control studies, the Newcastle-Ottawa Scale (NOS) was used[10], evaluating participant selection, comparability of study groups, and ascertainment of exposure and outcomes. Some non-randomized studies were assessed using the Risk of Bias in Non-Randomized Studies of Interventions tool[11]. Discrepancies in scoring were resolved through discussion, and the overall risk of bias for each study was classified as low, moderate, or high based on total NOS scores and reporting transparency. In the interpretation of findings, greater weight was assigned to studies with low or moderate risk of bias, particularly when synthesizing associations between coverage timing and infection or limb salvage outcomes. Studies with higher residual confounding were interpreted more cautiously, although all eligible studies contributed to the overall qualitative conclusions.
Given the heterogeneity among included studies in terms of design, definitions of “early” coverage, and outcome reporting, a quantitative meta-analysis was not feasible. Instead, a structured narrative synthesis was conducted to summarize and interpret findings. The synthesis was organized around key clinical outcome domains, including infection rate, bone union, flap-related complications, and limb salvage. Data were summarized descriptively using frequencies and percentages to highlight consistent trends, with particular attention to coverage timing categories (≤ 72 hours, ≤ 7 days, or ≤ 12 days) and their correlation with complication rates and overall clinical outcomes.
Although a quantitative meta-analysis was considered, meaningful statistical pooling was not justified due to substantial clinical and methodological heterogeneity across studies, including variable definitions of early vs delayed coverage, differences in injury severity, fixation strategies, and outcome reporting. Subgroup pooling based on ≤ 72 hours vs 7-12 days was explored conceptually, but inconsistent reporting of denominator data and adjusted effect estimates precluded valid aggregation. Similarly, a random-effects meta-analysis of infection rates was not undertaken because of wide inter-study variability and the risk of generating misleading summary estimates.
Confounding by indication was recognized as an inherent limitation, as more severe, contaminated, or physiologically unstable injuries were more likely to undergo delayed reconstruction. Several included studies attempted multivariable adjustment for injury severity and patient factors; however, residual confounding could not be fully excluded. This limitation was therefore carefully considered when interpreting the strength of associations between timing of coverage and clinical outcomes.
Based on the overall synthesis, a strict chronological cutoff (within the first week after fixation) appeared more consistently predictive of reduced infection and improved union than a purely biological definition based on number of debridements, although both timing paradigms demonstrated convergent trends favoring earlier definitive coverage.
As this review utilized data exclusively from previously published research, no ethical approval was required. All included studies were appropriately cited, and data handling adhered to ethical standards for secondary data analysis and transparent reporting.
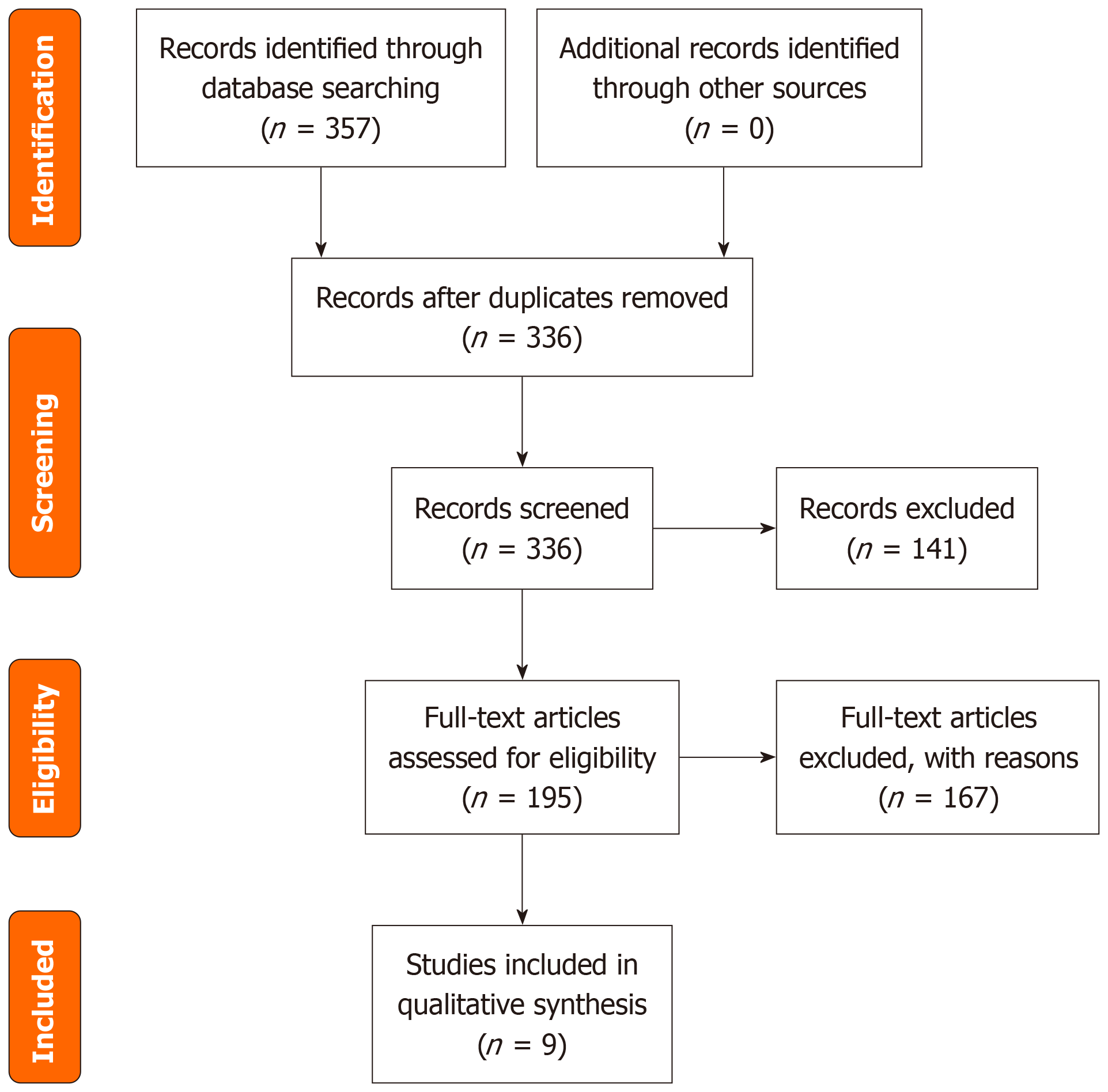
A total of 357 records were identified through database searches, including PubMed (n = 112), Scopus (n = 96), Web of Science (n = 74), and Google Scholar (n = 75). After the removal of 21 duplicates, 336 unique studies underwent title and abstract screening, of which 141 were excluded for irrelevance. One hundred ninety-five full-text reports were sought for retrieval, and 28 could not be obtained in full text. The remaining 167 full-text articles were assessed for eligibility, and 158 were excluded for reasons detailed in Figure 1, including case reports, conference abstracts, editorials, reviews, pediatric populations, and non-tibial fractures. Ultimately, nine studies met the inclusion criteria and were incorporated into the final qualitative synthesis (Figure 1).
The nine studies included in this review were retrospective cohort analyses published from 2000 to 2025, collectively encompassing more than 900 patients with Gustilo-Anderson type IIIB and IIIC open tibial fractures. Definitions of early coverage varied across studies, ranging from flap reconstruction performed within 72 hours of fixation to within 12 days post-injury, whereas delayed coverage generally exceeded these thresholds. Most studies utilized internal fixation methods and employed either local or free flap reconstruction techniques. Across all cohorts, earlier flap coverage was consistently associated with lower deep infection rates (ranging from 0% to 23% compared with up to 72% in delayed groups), fewer nonunions, and a reduced need for reoperations or amputations. Limb-salvage rates exceeded 90% in centers implementing coordinated orthoplastic protocols. The data summarized in Table 1 demonstrate that timely, well-integrated skeletal fixation and flap coverage markedly enhance both biological and functional outcomes in severe open tibial fractures.
| Ref. | Study design | Sample size (n) | Population/fracture type | Definition of early coverage | Comparison group | Fixation method | Flap type/method | Primary outcomes | Infection/complication rate | Amputation/Limb salvage rate |
| Khan et al[12], 2025 | Retrospective cohort | 51 patients (52 extremities) | Gustilo-Anderson type IIIB and IIIC open tibial fractures | ≤ 72 hours post-fixation | > 72 hours post-fixation | Definitive fracture fixation (not specified) | Local vs free flaps (muscle or fasciocutaneous) | Deep surgical site infection, nonunion | Deep infection: 43% in free flaps vs 0% in local flaps; trend toward higher infection when > 72 hours (30% vs 18%); nonunion more frequent in free flaps (43%) than local (9%) | Not reported |
| Henry et al[13], 2023 | Retrospective cohort (long-term follow-up) | 81 patients | Gustilo-Anderson type IIIB open tibial fractures | Early orthoplastic “fix-and-flap” with adjuvant antibiotic hydroxyapatite | No comparison group | Internal fixation (limb reconstruction) | Combined orthoplastic flap coverage with local antibiotic bio-composite | Bone union, deep infection, limb salvage | Deep infection: 3.7%; union achieved in 96% | Limb salvage: 96.3% |
| Sacks et al[14], 2023 | Multisite retrospective cohort | 80 patients (44 India, 36 United States) | Gustilo-Anderson type IIIB and IIIC open tibial fractures requiring free flap coverage | Early total debridement cohort (mean 1.84 debridements) | Serial debridement cohort (mean 3.64 debridements) | Definitive fixation before flap | Free tissue transfer (flap type not specified) | Infection, nonunion, flap failure | Infection and nonunion significantly higher in serial-debridement group (P < 0.05); no difference in flap failure | Not reported |
| Zamorano et al[15], 2025 | Retrospective cohort | 41 patients | Gustilo-Anderson type IIIB open tibial fractures | Flap coverage ≤ 12 days post-injury | Flap coverage > 12 days | Standardized fixation under level I trauma protocol | Soft-tissue flap (type not specified) | Fracture-related infection | Infection: 0% when ≤ 12 days vs 56.6% when >12 days (P < 0.01); AUC = 0.83 for 12-day threshold | Not reported |
| Cochonat et al[16], 2025 | Retrospective cohort | 35 patients | Open tibial fractures requiring flap coverage (mean age 45 ± 17.3 years; 88.6% male) | Flap coverage ≤ 7 days | Flap coverage > 7 days | Internal fixation (not specified) | Soft-tissue flap (not specified) | Infection, nonunion, flap failure, amputation | Infection significantly higher when > 7 days (72.2% vs 17.6%, P < 0.05); nonunion 24.2%; flap failure 143%; amputation 8.6% | Amputation 8.6% (all infected cases) |
| Chua et al[17], 2014 | Comparative retrospective study | 89 patients (83 men, 6 women; mean age 38 years) | Gustilo-Anderson type IIIB and IIIC open tibial fractures | Flap coverage ≤ 72 hours post-fixation | Flap coverage > 72 hours | Fracture fixation within 24 hours (method not specified) | Soft-tissue flap (not specified) | Bone union, infection, flap failure, secondary procedures | Deep infection: 23% (early) vs 54% (late), P < 0.01; shorter hospital stay and fewer procedures in early group | No significant difference |
| Sreedharan et al[18], 2024 | Retrospective cohort (prospective database review) | 148 open leg injuries (100 limb salvage, 21 primary amputation) | Open tibial fractures (metaphyseal, diaphyseal, epiphyseal) | Not specified (timing analyzed but not stratified) | Limb salvage vs primary amputation | Internal and external fixation (definitive external fixation linked to higher nonunion) | Various soft-tissue reconstructions (not specified) | Bone union, mobility, pain, infection | Higher nonunion and infection with external fixation; timing not associated with increased complications | 21% primary amputation; 96% of salvaged limbs ambulated |
| Mahmoud et al[19], 2024 | Retrospective cohort | 55 patients | Adult patients (> 18 years) with open midshaft tibial fractures requiring flap coverage | Flap coverage ≤ 7 days | Coverage > 7 days | Not specified | Flap coverage (type not specified) | Deep infection, nonunion, flap failure, revision surgery | Deep infection: 6% (early) vs 38% (delayed), P = 0.01; revision surgery: 17% vs 54%, P = 0.008 | Not specified |
| Cao et al[20], 2022 | 15-year retrospective cohort | 394 free-flap reconstructions | Gustilo-Anderson type IIIB open tibial fractures | Early repair ≤ 2 debridements (mean 6.15 ± 1.82 days) | Delayed repair ≥ 2 debridements (mean 16.46 ± 4.09 days) | Internal fixation following debridement | Free-flap reconstruction (various perforator and muscle flaps) | Flap survival, healing, complications, recovery | Flap necrosis, wound, and late complications significantly lower in early group (P < 0.01); delayed group had 8.2% wound enlargement | All achieved limb salvage; early group had better function and fewer reoperations |
All nine included studies were observational in nature, predominantly retrospective cohort analyses evaluated using the NOS[10] or the Risk of Bias in Non-Randomized Studies of Interventions tool[11], as appropriate for study design. Most studies demonstrated clearly defined inclusion criteria, objective outcome assessment, and adequate follow-up duration. Three studies were rated as having a low overall risk of bias, characterized by well-defined patient populations, standardized data collection methods, and minimal confounding. The remaining six were classified as moderate risk, primarily due to residual confounding and the lack of randomization inherent to retrospective designs. Importantly, no study exhibited a high risk of bias. As summarized in Table 2, the methodological consistency, transparent reporting, and overall moderate-to-low bias across studies collectively support the reliability and robustness of the synthesized findings.
| Ref. | Study design | Assessment tool | Key domains assessed | Total Newcastle-Ottawa Scale | Overall risk of bias |
| Khan et al[12], 2025 | Retrospective cohort | Newcastle-Ottawa Scale | Clear inclusion criteria, objective outcomes, moderate control for confounders | 6/9 | Moderate |
| Henry et al[13], 2023 | Retrospective cohort (single-arm) | Modified Newcastle-Ottawa Scale | Consecutive cases, long follow-up, no comparison group | 5/9 | Moderate |
| Sacks et al[14], 2023 | Multisite retrospective cohort | ROBINS-I | Good selection, exposure well defined, moderate confounding between centers | Not applicable | Moderate |
| Zamorano et al[15], 2025 | Retrospective cohort | Newcastle-Ottawa Scale | Well-defined population, objective outcomes, adequate follow-up | 8/9 | Low |
| Cochonat et al[16], 2025 | Retrospective cohort | Newcastle-Ottawa Scale | Standardized data collection, clear timing groups, adequate follow-up | 8/9 | Low |
| Chua et al[17], 2014 | Comparative retrospective study | ROBINS-I | Direct comparison (≤ 72 hours vs > 72 hours), consistent fixation, limited confounder control | Not applicable | Moderate |
| Sreedharan et al[18], 2024 | Retrospective cohort from prospective database | Modified Newcastle-Ottawa Scale | Large cohort, objective outcomes, minor confounding not fully controlled | 6/9 | Moderate |
| Mahmoud et al[19], 2024 | Retrospective cohort | Newcastle-Ottawa Scale | Well-defined exposure and outcomes, limited adjustment for covariates | 6/9 | Moderate |
| Cao et al[20], 2022 | Large retrospective cohort | Newcastle-Ottawa Scale | Large sample, objective endpoints, well-documented follow-up | 8/9 | Low |
Overall, the findings across all included studies demonstrated a clear and consistent gradient favoring early soft-tissue coverage. Early coverage groups showed substantially lower deep infection rates compared with delayed groups, accompanied by reduced reoperation and revision surgery requirements. Limb-salvage outcomes were uniformly high in early intervention cohorts, frequently exceeding 90%, whereas delayed coverage was associated with higher rates of secondary procedures and occasional progression to amputation. Collectively, these patterns reinforce the critical relationship between timely soft-tissue reconstruction, infection control, surgical burden, and successful limb preservation.
In this systematic review of nine observational studies examining orthoplastic management of severe open tibial fractures, early definitive soft-tissue coverage emerged as a consistent determinant of improved clinical outcomes[12-20]. Across diverse settings - from single-center trauma units to multicenter cohorts - patients receiving flap coverage within the institution-defined “early” window demonstrated substantially lower rates of deep infection and reoperation[15].
Reported infection rates ranged from 6% to 12% in early-coverage cohorts compared with 30%-40% in delayed-coverage groups[19]. Revision or reoperation frequencies increased two- to four-fold when coverage was postponed beyond 7-10 days[15]. Although definitions of “early” varied-spanning ≤ 72 hours in high-resource centers, ≤ 7 days in standardized orthoplastic pathways, and ≤ 12 days in resource-limited contexts - the directionality of benefit was remarkably uniform. Collectively, these findings underscore the principle that prompt, coordinated orthoplastic intervention, rather than adherence to a rigid temporal threshold, is the key predictor of infection control, bone union, and eventual limb preservation[12-20].
Despite heterogeneity in the definitions of “early” intervention, all included studies demonstrated a clear infection gradient favoring early soft-tissue coverage[12-20]. In pooled descriptive comparison, early-coverage groups exhibited infection rates of 6%-23%, whereas delayed-coverage groups showed 30%-72% rates[16], corresponding to a two- to four-fold relative risk reduction[15]. Similarly, reoperation and revision rates were consistently lower in early groups (17%-25%) compared with delayed cohorts (40%-54%)[19]. Studies employing multivariable adjustment - such as Mahmoud et al[19] and Zamorano et al[15] - identified timing as an independent predictor of infection, with odds ratios ranging from 4.5 to 10.0 for delayed coverage. Notably, large-sample studies with standardized orthoplastic pathways, including those by Cao et al[20] and Cochonat et al[16], demonstrated the lowest complication rates, emphasizing the synergistic benefit of timely intervention and system-level coordination.
Infection is not an isolated endpoint but the central mediator linking surgical timing to final limb outcomes. Across multiple cohorts, deep infection strongly predicted nonunion, flap failure, and amputation[16]. In one series, nonunion (24.2%), flap failure (14.3%), and amputation (8.6%) occurred almost exclusively among infected cases, highlighting infection as the initiating event leading to mechanical and reconstructive failure[16].
Similarly, in a level I trauma-center analysis of Gustilo IIIB/IIIC tibial fractures, delayed free-flap reconstruction resulted in deep infection rates up to 43% and nonunion up to 43%, whereas locally based flaps achieved 0% infection and only 9% nonunion[12]. The reproducibility of this cascade supports a causal sequence in which delayed biological coverage → bacterial colonization → infection → impaired osteogenesis → limb-threatening salvage surgery[12,16]. Early, well-vascularized flap coverage interrupts this trajectory by restoring perfusion, enhancing immune and antibiotic delivery, and stabilizing the mechanical-biological interface critical for bone repair[12-16].
Beyond absolute timing, treatment philosophy and workflow integration emerged as critical determinants of success. A multicenter comparison of 80 patients with Gustilo IIIB/IIIC tibial fractures revealed that centers employing serial debridement strategies (mean 3.6 procedures before coverage) had significantly higher infection and nonunion rates than those adopting a single, radical debridement with early flap coverage (mean 1.8 procedures)[14].
Similarly, a 15-year series of 394 free-flap reconstructions demonstrated superior outcomes when reconstruction followed ≤ 2 debridements, with shorter hospital stays, less blood loss, and fewer flap or bone complications compared to cases delayed after multiple debridements[20]. These findings imply that each additional debridement and day of open exposure may amplify bacterial burden and wound enlargement, degrading the local biological environment.
Conversely, an orthoplastic “fix-and-flap” pathway - integrating skeletal stabilization, meticulous single-stage debridement, and definitive soft-tissue coverage - provides biological containment, minimizes colonization pressure, and optimizes both flap survival and bone union[21]. Despite the clear advantages of early definitive coverage, delayed reconstruction may be unavoidable in certain real-world scenarios, particularly in patients with ongoing vascular compromise, severe polytrauma, or hemodynamic instability requiring staged damage-control resuscitation. In such circumstances, temporary wound management and delayed flap transfer remain necessary to prioritize patient survival while planning definitive orthoplastic reconstruction.
Importantly, early coordinated orthoplastic care is not merely limb-saving in an anatomical sense; it enables functional salvage[22,23]. In a long-term cohort of 81 patients with Gustilo IIIB tibial fractures managed with combined fixation, flap coverage, and local antibiotic hydroxyapatite, union was achieved in 96%, with deep infection in only 3.7% and limb salvage in 96.3% at nearly five years of follow-up[13]. Similarly, data from a tertiary trauma-center registry tracking 100 limb-salvage patients with open tibial injuries showed an ultimate union rate of 84%, with 96% of salvaged limbs regaining independent ambulation[18]. These findings challenge the historical perception that high-grade IIIB and IIIC fractures are inherently “non-salvageable”. Instead, they demonstrate that under an integrated orthoplastic model, durable reconstruction with functional ambulation is now a realistic expectation rather than an exceptional outcome[13,18]. This shift - from anatomical preservation to restoration of independence - highlights broader public health and socioeconomic implications, reducing amputation-related disability, prosthetic dependence, and long-term rehabilitation burdens[24].
This review is limited by the observational nature of the available evidence[12-20]. All included studies were retrospective, introducing potential selection and documentation biases. In clinical practice, delayed coverage is seldom random - it typically occurs in the most contaminated, ischemic, or severely comminuted fractures - creating potential confounding by indication[12-20].
However, several observations suggest that the association between delay and infection extends beyond such confounding. First, the absolute infection-rate differences are too large and consistent to be explained by case mix alone (e.g., 0% vs 56.6%, or 6% vs 38%)[15,19]. Second, the uniformity of benefit across centers, continents, and varying definitions of “early” (≤ 72 hours, ≤ 7 days, ≤ 12 days, ≤ 2 debridements) supports external validity[12,15-20]. Third, studies adopting a radical single-debridement “fix-and-flap” strategy consistently demonstrated reductions not only in infection but also in reoperation rates and time to union[14,20]. Together, these convergent data support a biologically plausible model in which prompt, vascularized coverage preserves perfusion, limits colonization, and enhances osteogenesis[12-20].
Looking forward, the field requires prospective standardization of what constitutes “early coverage” in open tibial fracture care[25]. Future research should leverage multicenter registries or pragmatic stepped-care trials controlling for injury severity, contamination, bone loss, and vascular status. Comparative analyses of flap type, antibiotic adjuncts, and workflow models would clarify whether improved outcomes result primarily from timing or from integrated multidisciplinary care. Ultimately, such efforts will guide evidence-based orthoplastic algorithms that emphasize biological timing over arbitrary chronological thresholds.
Early, coordinated orthoplastic management of severe open tibial fractures consistently yields lower infection rates, faster bone union, and higher limb salvage compared with delayed coverage. Across all included studies, delays beyond the early window - typically within the first week after fixation - were uniformly associated with increased deep infection, nonunion, and reoperation. These findings affirm that timely, vascularized soft-tissue coverage is not merely a procedural preference but a biological necessity that determines reconstructive success. The take-home message is clear: An integrated “fix-and-flap” approach, executed within the first week, should be regarded as the standard of care to optimize outcomes and preserve functional limbs.
The authors would like to acknowledge the contributions of all researchers whose work formed the basis of this systematic review.
| 1. | Nicolaides M, Pafitanis G, Vris A. Open tibial fractures: An overview. J Clin Orthop Trauma. 2021;20:101483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 2. | Kashyap S, Ambade R, Landge S, Salwan A. Impact of Surgical Timing on Fracture Healing in Tibial Shaft Injuries: A Comparative Review of Intramedullary Nailing Techniques. Cureus. 2024;16:e70978. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 3. | Lin CH, Chou YC, Hsu CC, Huang RW, Lin CH. Orthoplastic Approach to Extremity Reconstruction: A Paradigm Shift in Integrated Limb Salvage. Semin Plast Surg. 2025;39:129-139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 4. | Qiu E, Kurlander DE, Ghaznavi AM. Godina revisited: a systematic review of traumatic lower extremity wound reconstruction timing. J Plast Surg Hand Surg. 2018;52:259-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 5. | Tiongco RFP, Rezwan SK, Alahmadi S, Heron MJ, Sylvester SA, Seal SM, de Jong T, Elhelali A, Mundy LR. Early Flap Reconstruction and Infection Rates in Open Lower Extremity Fractures: A Systematic Review and Meta-analysis. Plast Reconstr Surg Glob Open. 2025;13:e6829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 6. | Mardourian M, Wiesemann GS, Sachse CC, Nichols DS, Hagen JE, Chim H. Hardware Salvage in the Lower Extremity after Flap Coverage: 10-Year Single Center Outcomes Analysis. Plast Reconstr Surg Glob Open. 2023;11:e5105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 7. | Kanakaris NK, Rodham P, Giannoudis VP, Giannoudis PV. Modern Management of Severe Open Fractures of the Extremities: The Role of the Induced Membrane Technique. J Bone Joint Surg Am. 2025;107:504-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 8. | Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57226] [Cited by in RCA: 48218] [Article Influence: 2836.4] [Reference Citation Analysis (11)] |
| 9. | Brown D. A Review of the PubMed PICO Tool: Using Evidence-Based Practice in Health Education. Health Promot Pract. 2020;21:496-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 221] [Article Influence: 36.8] [Reference Citation Analysis (1)] |
| 10. | Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14281] [Cited by in RCA: 13915] [Article Influence: 869.7] [Reference Citation Analysis (8)] |
| 11. | Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, Henry D, Altman DG, Ansari MT, Boutron I, Carpenter JR, Chan AW, Churchill R, Deeks JJ, Hróbjartsson A, Kirkham J, Jüni P, Loke YK, Pigott TD, Ramsay CR, Regidor D, Rothstein HR, Sandhu L, Santaguida PL, Schünemann HJ, Shea B, Shrier I, Tugwell P, Turner L, Valentine JC, Waddington H, Waters E, Wells GA, Whiting PF, Higgins JP. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13881] [Cited by in RCA: 13257] [Article Influence: 1325.7] [Reference Citation Analysis (5)] |
| 12. | Khan U, Gray K, Ghidei S, Quigley C, Mehan V, Johnson E, Goch A, Gaski G. Does the Timing, Type, and Method of Flap Coverage After Open Tibia Fracture Fixation Influence the Rate of Deep Infection? Microsurgery. 2025;45:e70110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 13. | Henry JA, Ali A, Elkhidir IH, Reid A, Wong J, Pillai A. Long-Term Follow-Up of Open Gustilo-Anderson IIIB Fractures Treated With an Adjuvant Local Antibiotic Hydroxyapatite Bio-Composite. Cureus. 2023;15:e39103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Sacks H, Hu J, Devendra A, Huang S, Levine J, Sabapathy SR, Venkatramani H, Brogan D, Boyer M, Hacquebord J. Relationship between number of debridements and clinical outcomes in open tibia fractures requiring free flap coverage: A retrospective cohort study. Orthoplast Surg. 2023;14:9-14. [DOI] [Full Text] |
| 15. | Zamorano ÁI, Vaccia MA, Parra RI, Turner T, Rivera I, Errázuriz T, Oyarzún A, Bahamonde LA. Soft tissue coverage before 12 days prevents fracture-related infection in IIIB open tibial fractures in young and healthy patients. OTA Int. 2025;8:e435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 16. | Cochonat M, Bertani A, Rongieras F, Bauwens PH. Lower infection rate in patients with early flap coverage of open tibial fractures: A single-center retrospective study. Orthop Traumatol Surg Res. 2025;104430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 17. | Chua W, De SD, Lin WK, Kagda F, Murphy D. Early versus late flap coverage for open tibial fractures. J Orthop Surg (Hong Kong). 2014;22:294-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 18. | Sreedharan S, Bruscino-Raiola F, Lew P, Ling Y, Ferris S. Bone union and mobility outcomes for reconstructed open tibial fractures: a plastic surgical experience from a major trauma center. Front Surg. 2024;11:1348991. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 19. | Mahmoud A, Younger J, Morris E, Hope M. A delay in flap coverage of open midshaft tibia fractures increases complications: A retrospective study. J Orthop Trauma Rehabil. 2024;31. [DOI] [Full Text] |
| 20. | Cao Z, Li C, He J, Qing L, Yu F, Wu P, Tang J. Early Reconstruction Delivered Better Outcomes for Severe Open Fracture of Lower Extremities: A 15-Year Retrospective Study. J Clin Med. 2022;11:7174. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 21. | Singh J, Dhillon MS, Dhatt SS. Single-stage "Fix and Flap" gives Good Outcomes in Grade 3B/C Open Tibial Fractures: A Prospective Study. Malays Orthop J. 2020;14:61-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 22. | Hoyt BW, Wade SM, Harrington CJ, Potter BK, Tintle SM, Souza JM. Institutional Experience and Orthoplastic Collaboration Associated with Improved Flap-based Limb Salvage Outcomes. Clin Orthop Relat Res. 2021;479:2388-2396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 23. | Jordan DJ, Malahias M, Khan W, Hindocha S. The ortho-plastic approach to soft tissue management in trauma. Open Orthop J. 2014;8:399-408. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 24. | O'Meara R, Chawla K, Gorantla A, Kelly R, DeJong M, Babrowski T, Halandras P, Blecha M. The Impact of Sociodemographic Variables on Functional Recovery following Lower Extremity Amputation. Ann Vasc Surg. 2025;110:317-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 25. | Trickett RW, Mudge E, Price P, Pallister I. A qualitative approach to recovery after open tibial fracture: the road to a novel, patient-derived recovery scale. Injury. 2012;43:1071-1078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |