Published online Mar 18, 2026. doi: 10.13105/wjma.v14.i1.118496
Revised: January 30, 2026
Accepted: February 24, 2026
Published online: March 18, 2026
Processing time: 65 Days and 21.3 Hours
Diuretic resistance is a frequent and clinically important problem in acute decom
To determine the effectiveness and safety of adjunctive pharmacologic therapies for diuretic resistance in ADHF.
Randomized controlled trials enrolling adults hospitalized with ADHF receiving loop diuretics were identified through PubMed, EMBASE, Scopus, and the Cochrane Central Register of Controlled Trials to February 2025. Ad
Eight randomized controlled trials including 1758 participants were analyzed. Acetazolamide increased successful decongestion compared with placebo (42.2% vs 30.5%; risk ratios 1.46, 95% confidence interval: 1.17-1.82, study-level estimate). Thiazide augmentation produced greater weight loss at 72 hours (2.3 vs 1.5 kg) but increased renal dysfunction (46.5% vs 17.2%, study-level data). Sodium-glucose cotransporter 2 (SGLT2) inhibitors increased urine output and natriuresis and reduced worsening heart failure events in selected studies (10% vs 33%, based on individual trial findings). High-dose spironolactone, low-dose dopamine, and low-dose nesiritide showed no meaningful clinical benefit. Thiazide and thiazide-like diuretics were appropriately classified as distal convoluted tubule agents, whereas acetazolamide and SGLT2 inhibitors act primarily at the proximal nephron.
Adjunctive therapies targeting proximal sodium handling (acetazolamide, SGLT2 inhibitors) or distal sodium reabsorption (thiazide diuretics) improve decongestion and may serve as effective options when loop diuretics alone are insufficient. These findings reflect study-level evidence from individual randomized trials and support a mechanistically guided approach to treating diuretic resistance in ADHF.
Core Tip: Diuretic resistance remains a major obstacle to effective decongestion in acute decompensated heart failure (HF). This systematic review synthesizes randomized controlled trial evidence evaluating adjunctive pharmacologic therapies added to loop diuretics. Therapies targeting proximal tubular sodium handling, particularly acetazolamide, consistently improve decongestion and natriuresis without excess renal risk. Thiazide diuretics, which act at the distal convoluted tubule, enhance diuretic response but increase the risk of renal dysfunction and therefore require cautious use. Sodium-glucose cotransporter 2 inhibitors offer modest diuretic effects, reduce worsening HF events, and provide benefits that are not limited by baseline kidney function. In contrast, older adjuncts such as dopamine, nesiritide, and high-dose spironolactone show no clinical benefit in overcoming acute diuretic resistance.
- Citation: Patel V, Zameer R, Kumar B, Das M. Adjunctive pharmacologic therapies for diuretic resistance in acute decompensated heart failure: Systematic review of randomized trials. World J Meta-Anal 2026; 14(1): 118496
- URL: https://www.wjgnet.com/2308-3840/full/v14/i1/118496.htm
- DOI: https://dx.doi.org/10.13105/wjma.v14.i1.118496
Acute decompensated heart failure (ADHF) remains one of the leading causes of hospital admissions worldwide and is associated with high morbidity, frequent readmissions, and significant healthcare burden[1]. A central feature of ADHF is fluid overload, which contributes to dyspnea, peripheral edema, elevated filling pressures, and end-organ dysfunction. Intravenous loop diuretics are the cornerstone of therapy and are routinely used to achieve rapid decongestion. However, a substantial proportion of patients exhibit an inadequate response to loop diuretics, a phenomenon known as diuretic resistance[2,3].
Diuretic resistance is generally defined as a diminished natriuretic or diuretic effect despite escalating loop diuretic doses. This condition is clinically important because persistent congestion is strongly associated with poorer outcomes, including prolonged hospitalization, worsening renal function, and increased mortality[4,5]. Multiple pathophysiological mechanisms contribute to diuretic resistance, such as neurohormonal activation, impaired renal perfusion, reduced tubular responsiveness, and distal nephron sodium compensation. These mechanisms complicate management and create the need for therapeutic strategies that can enhance or restore diuretic efficacy[6].
Over the past decade, several adjunctive pharmacologic therapies have emerged as potential options to overcome diuretic resistance. These include agents that target different segments of the nephron-such as thiazide-type diuretics, carbonic anhydrase inhibitors, and mineralocorticoid receptor antagonists (MRA)-as well as newer drug classes like sodium-glucose cotransporter 2 (SGLT2) inhibitors and vasopressin antagonists[2,7]. Randomized controlled trials have evaluated these therapies as add-on strategies to loop diuretics, with the aim of improving urine output, enhancing natriuresis, accelerating decongestion, and stabilizing renal function. Despite growing interest, the evidence remains scattered, with varying clinical endpoints, patient characteristics, and therapeutic responses across studies[8].
Given the clinical relevance of diuretic resistance and the expanding range of adjunctive pharmacologic interventions, a comprehensive synthesis of randomized controlled trial data is essential. Such a review can help clinicians identify which therapies provide the greatest benefit, under what circumstances they are effective, and how they may be integrated into current ADHF management pathways.
The objective of this systematic review is to evaluate the effectiveness and safety of adjunctive pharmacologic therapies used alongside loop diuretics to overcome diuretic resistance in patients hospitalized with ADHF. Specifically, the review will synthesize evidence from randomized controlled trials assessing their impact on diuretic response, congestion relief, renal outcomes, length of stay, and key clinical endpoints.
This study was conducted as a systematic review of randomized controlled trials evaluating adjunctive pharmacologic therapies used to overcome diuretic resistance in adults hospitalized with ADHF. The review followed the PRISMA 2009 guidelines. A predefined protocol outlining the research question, eligibility criteria, search strategy, and data extraction methods was followed throughout the review process[9]. Given substantial methodological and clinical heterogeneity across trials, no statistical pooling or meta-analysis was performed.
The research question was guided by the PICO framework[10]. The population consisted of adult patients hospitalized with ADHF who demonstrated insufficient response to loop diuretic therapy or required enhanced decongestion. Interventions included adjunctive pharmacologic therapies administered in addition to loop diuretics, such as carbonic anhydrase inhibitors, thiazide or thiazide-like diuretics, vasopressin antagonists, SGLT2 inhibitors, and MRAs. To ensure mechanistic clarity, adjunctive therapies were classified according to their primary nephron site of action: Proximal tubular agents (e.g., acetazolamide, SGLT2 inhibitors) and distal convoluted tubule agents (e.g., thiazide and thiazide-like diuretics). Comparators included placebo, usual care, or other active adjunctive agents. Outcomes of interest included diuretic response parameters such as natriuresis, urine output, diuretic efficiency, weight change, improvement in congestion, renal function trajectories, adverse events, readmissions, and mortality. The primary aim was to determine the effectiveness and safety of adjunctive pharmacologic therapies in improving diuretic response in acute heart failure (HF).
A comprehensive electronic search was conducted in PubMed, EMBASE, Scopus, and the Cochrane Central Register of Controlled Trials from database inception to February 2025. Search terms included combinations of “acute heart failure”, “decompensated heart failure”, “diuretic resistance”, “diuretic response”, “adjunctive therapy”, “acetazolamide”, “thiazide”, “SGLT2 inhibitor”, “dapagliflozin”, “empagliflozin”, “tolvaptan”, “spironolactone”, and “randomized controlled trial”. Medical Subject Headings (MeSH) were incorporated to improve specificity. No language restrictions were applied. Reference lists of included articles and relevant reviews were screened for additional eligible studies.
Studies were eligible if they met the following criteria: Randomized controlled trial design, adult population hospitalized with ADHF, evaluation of an adjunctive pharmacologic therapy administered in conjunction with loop diuretics, and reporting of at least one clinically relevant outcome related to diuretic response, congestion, renal function, or safety. Studies were excluded if they were observational, non-randomized, conducted exclusively in chronic stable HF, or focused on non-pharmacologic adjuncts such as ultrafiltration.
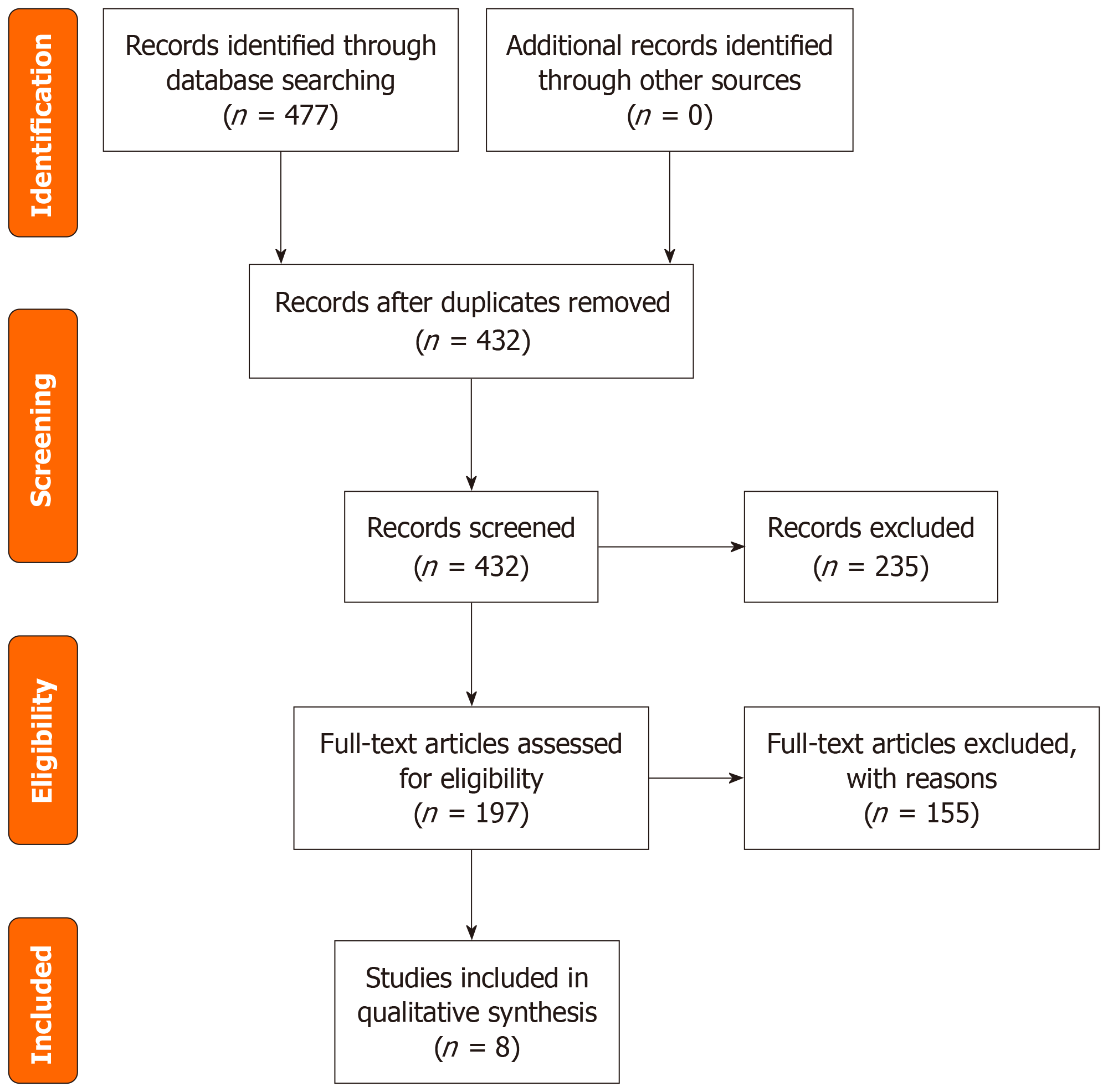
Two reviewers independently screened titles and abstracts for relevance, followed by full-text evaluation of potentially eligible studies. Discrepancies were resolved through discussion and consensus. The selection process was documented using the PRISMA 2009 flow diagram, detailing the number of records identified, screened, excluded, and finally included in the review.
Data were extracted independently by two reviewers using a standardized form. Extracted information included study characteristics, patient demographics, intervention details, comparator arms, dosing regimens, outcome definitions, follow-up durations, and reported results for all primary and secondary outcomes. For consistency, diuretic response metrics were recorded as reported in the original trials, including weight change, cumulative urine output, natriuresis, and diuretic efficiency. All effect estimates (e.g., risk ratios, confidence intervals, mean differences) were extracted directly from individual randomized trials without transformation, recalculation, or pooling.
The methodological quality of the included studies was evaluated using the Cochrane Risk of Bias tool for randomized trials (RoB 2.0)[11]. Each study was assessed across domains including randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result. Each domain was judged as having low risk, some concerns, or high risk of bias, and an overall judgment was assigned accordingly. Disagreements were resolved through consensus. A detailed risk-of-bias table was created to summarize domain-level assessments across studies and provide a clear overview of methodological quality.
A structured narrative synthesis was performed due to heterogeneity in interventions, diuretic dosing strategies, and outcome definitions across the included trials. Findings were summarized with attention to patterns in diuretic response, clinical decongestion, renal effects, and adverse event profiles. Direction-of-effect vote counting, consistent with Cochrane guidance, was used to qualitatively compare study findings without conducting quantitative pooling. Effect estimates and confidence intervals were extracted directly from published results and presented as study-level values. Where applicable, comparisons between adjunctive agents and placebo or standard care were described in a consistent format to enhance interpretability.
Figure 1 illustrates the study identification, screening, eligibility assessment, and inclusion process for this systematic review. A total of 477 records were identified through database searching, including PubMed, EMBASE, Scopus, and the Cochrane Central Register of Controlled Trials. After removal of duplicate records, 432 records were screened by title and abstract, and 235 records were excluded. Full-text articles were sought for 197 records; 34 reports could not be retrieved. The remaining 163 full-text articles were assessed for eligibility, of which 155 were excluded due to observational design, non-randomized methodology, chronic stable HF populations, or non-pharmacologic interventions. Ultimately, eight randomized controlled trials met all inclusion criteria and were included in the qualitative synthesis. No studies were included in quantitative synthesis.
The eight randomized controlled trials summarized in Table 1 evaluated a range of adjunctive pharmacologic therapies designed to enhance diuretic response in adults hospitalized with ADHF. Sample sizes ranged from small pilot cohorts of 40 participants to large multinational trials enrolling more than 500 participants. These studies included therapies acting at distinct nephron sites, such as proximal tubular agents (acetazolamide, SGLT2 inhibitors), distal convoluted tubule agents (thiazide and thiazide-like diuretics), and other adjuncts including MRAs, vasodilatory agents, and dopaminergic therapies. Although all trials targeted an inadequate response to loop diuretics, they differed in their definitions of diuretic resistance, baseline congestion severity, renal function profiles, and timing of adjunctive therapy initiation. Primary outcomes varied widely and included changes in weight, cumulative urine output, natriuresis, diuretic efficiency, biomarker trajectories, and hierarchical composite clinical measures. Follow-up durations were generally short, consistent with the acute inpatient setting, with most primary outcomes assessed during hospitalization or within the first 90 days. Collectively, these studies contributed heterogeneous but clinically meaningful study-level evidence, allowing comparative interpretation of mechanistically distinct adjunctive strategies for managing diuretic resistance in acute HF. Eight randomized controlled trials were included in the qualitative synthesis, comprising the ADVOR trial[12], the 3T trial[13], CLOROTIC[14], DICTATE-AHF[15], EMPA-RESPONSE-AHF[16], EMPULSE[17], ATHENA-HF[18], and ROSE-AHF[19].
| Ref. | Population characteristics | Intervention (adjunct therapy) | Comparator | Sample size (intervention/ | Primary outcome | Key findings | Statistical information1 |
| Mullens et al[12] | Acute decompensated heart failure; volume overload; elevated natriuretic peptides | Acetazolamide 500 mg IV daily (proximal tubule) + standardized loop diuretics | Placebo + standardized loop diuretics | 256/259 | Successful decongestion at 72 hours | Higher decongestion rates; increased natriuresis and urine output | RR 1.46; 95%CI: 1.17-1.82 |
| Cox et al[13] | Acute HF with documented loop resistance | Metolazone, IV chlorothiazide, or tolvaptan (distal tubule or V2 receptor) + loop diuretics | Active comparators | 20/20/20 | 48-hour weight loss | All adjuncts improved diuretic response; no between-group superiority | Weight loss 4.1-5.8 kg (study-level range) |
| Trullàs et al[14] | Hospitalized acute HF; volume overload | Hydrochlorothiazide (distal tubule) + IV furosemide | Placebo + IV furosemide | 115/115 | 72-hour body-weight change | Greater weight loss; improved diuresis; more renal impairment | Mean difference 114 kg; renal impairment 46.5% vs 17.2% |
| Cox et al[15] | Hypervolemic acute HF; hospitalized within 24 hours | Dapagliflozin 10 mg daily (proximal tubule via SGLT2) + protocolized diuretics | Structured usual care | 120/120 | Diuretic efficiency | No improvement in diuretic efficiency; reduced loop-diuretic needs; increased natriuresis | Odds ratio 0.65 for diuretic efficiency |
| Damman et al[16] | Acute decompensated HF with or without diabetes | Empagliflozin 10 mg daily (proximal tubule via SGLT2) | Placebo | 40/40 | Dyspnea and diuretic response | Higher urine output; fewer worsening-HF events | Composite events 10% vs 33% |
| Voors et al[17] | Stabilized acute HF; broad EF spectrum | Empagliflozin 10 mg daily | Placebo | 265/265 | Hierarchical composite | Significant clinical benefit; improved composite outcomes | Win ratio 1.36; 95%CI: 1.09-1.68 |
| Butler et al[18] | Acute HF; elevated natriuretic peptides | High-dose spironolactone 100 mg daily | Placebo or usual-care spironolactone | 180/180 | Change in natriuretic peptide levels | No improvement in congestion or outcomes | Reported as non-significant in original trial |
| Chen et al[19] | Acute HF with renal dysfunction | Low-dose dopamine or low-dose nesiritide + loop diuretics | Placebo | 122/119/119 | Urine volume and renal biomarkers | No benefit in decongestion or renal outcomes | Reported as non-significant in original trial |
The methodological quality of the included trials, summarized in Table 2, was generally robust, with most studies demonstrating low risk of bias across key domains. Randomization procedures were well described and appropriately implemented, minimizing concerns regarding allocation bias. Blinding was maintained in the majority of trials; however, a few open-label designs introduced some concerns related to deviations from intended interventions and outcome assessment. Missing outcome data were minimal across studies, reflecting short follow-up intervals and tightly supervised inpatient environments. Outcome measurements relied primarily on objective metrics-such as urine output, natriuresis, weight change, and biomarker levels-which reduced the likelihood of detection bias. While selective reporting was uncommon, smaller pilot studies and trials with multiple primary endpoints introduced areas of uncertainty. Overall, the risk of bias was judged to be low in five trials and of some concern in three, supporting the reliability of the findings while acknowledging methodological limitations that should be considered when interpreting study-specific results.
| Ref. | Randomization | Deviations from intended interventions | Missing data | Outcome measurement | Selective reporting | Overall risk |
| Mullens et al[12] | Low | Low | Low | Low | Low | Low |
| Cox et al[13] | Low | Low | Low | Low | Some concerns | Some concerns |
| Trullàs et al[14] | Low | Low | Low | Low | Low | Low |
| Cox et al[15] | Low | Some concerns | Low | Some concerns | Low | Some concerns |
| Damman et al[16] | Low | Low | Low | Low | Some concerns | Some concerns |
| Voors et al[17] | Low | Low | Low | Low | Low | Low |
| Butler et al[18] | Low | Low | Low | Low | Low | Low |
| Chen et al[19] | Low | Low | Low | Low | Low | Low |
This systematic review examined randomized controlled trials evaluating adjunctive pharmacologic therapies used to improve diuretic response in adults hospitalized with ADHF. Across the eight included studies, several important themes emerged. Acetazolamide, hydrochlorothiazide, and SGLT2 inhibitors demonstrated consistent signals of enhanced decongestion or improved natriuresis when added to loop diuretics, although the magnitude and clinical relevance varied between agents and trial designs. Notably, SGLT2 inhibitors provided decongestive and prognostic benefits across a broad range of kidney function, and their effects were not restricted by baseline estimated glomerular filtration rate (eGFR), supporting their use even in patients with moderate renal impairment. Other strategies, including high-dose spironolactone, low-dose dopamine, and low-dose nesiritide, did not improve congestion, renal function, or short-term clinical outcomes despite plausible physiologic mechanisms. However, the neutral findings with high-dose spironolactone should be interpreted in the context of widespread background renin-angiotensin-aldosterone system (RAAS) inhibition, potential ceiling effects from chronic MRA use, and the relatively delayed onset of MRA-mediated natriuresis in the acute setting. Collectively, the findings suggest that modern adjunctive therapies targeting proximal tubular sodium handling or glucose–sodium cotransport may offer more reliable benefits than older neurohormonal or vasodilatory strategies. In contrast, thiazide and thiazide-like diuretics augment loop diuretics through sequential nephron blockade at the distal convoluted tubule rather than the proximal nephron, highlighting the importance of correctly aligning mechanistic classification with site of action. The synthesis highlights the evolving landscape of diuretic resistance management and underscores the need to integrate mechanistic understanding with pragmatic clinical evidence when selecting adjunctive agents in the acute setting[20].
Interpreting the included trials through a mechanistic lens highlights why some adjunctive therapies offer meaningful benefits in ADHF while others do not. Acetazolamide, evaluated in the ADVOR trial[12], consistently enhanced decongestion and natriuresis when added to standardized loop diuretics, with approximately a one and a half fold higher likelihood of achieving complete decongestion compared with placebo. This benefit aligns well with its mechanism of proximal tubular sodium blockade, which increases distal sodium delivery and supports the pharmacologic action of loop diuretics. Thiazide-based strategies produced similarly encouraging effects[21]. In the 3T Trial, both metolazone and intravenous chlorothiazide improved diuretic response through sequential nephron blockade, and even the V2 receptor antagonist tolvaptan enhanced decongestion despite a different mechanism[13]. The CLOROTIC trial further demonstrated that hydrochlorothiazide combined with furosemide produced greater weight loss and higher diuretic efficiency, although this benefit came at the cost of more frequent renal impairment[14]. Importantly, thiazide and thiazide-like diuretics exert their action at the distal convoluted tubule rather than the proximal nephron, and their role in sequential nephron blockade should be interpreted in this mechanistic context. This pattern suggests that thiazide augmentation is effective but may be best applied as a rescue strategy in patients without significant renal vulnerability.
SGLT2 inhibitors showed a more nuanced profile across trials, reflecting their multifaceted renal and metabolic effects. In the pilot EMPA-RESPONSE-AHF study, empagliflozin did not significantly improve primary diuretic endpoints but increased total urine output and reduced a composite outcome of worsening HF, rehospitalization, or death[16]. The larger EMPULSE trial demonstrated clear clinical benefit at ninety days across diverse HF phenotypes, reinforcing that these agents contribute meaningfully even if their immediate diuretic effects are modest[17]. DICTATE-AHF further supported this interpretation by showing similar decongestion to structured usual care but with a lower cumulative loop diuretic dose and better natriuresis and urine output[15]. A key observation across these trials is that the decongestive and clinical benefits of SGLT2 inhibitors were not limited by baseline renal function, with meaningful effects observed even in patients with reduced eGFR. Together, these findings indicate that SGLT2 inhibitors enhance decongestion not by producing aggressive diuresis but by improving renal sodium handling, reducing diuretic requirements, and conferring early prognostic benefit.
In contrast, MRAs, dopamine, and nesiritide did not demonstrate meaningful benefits in the acute setting. ATHENA-HF showed that high-dose spironolactone was well tolerated yet did not improve NT-proBNP reduction, congestion, or short-term outcomes[18]. However, the neutral findings with spironolactone likely reflect important contextual factors, including background RAAS inhibition, which may blunt additional natriuretic benefit; potential ceiling effects from chronic MRA use; and the delayed onset of mineralocorticoid receptor blockade, which limits its utility in the acute phase. ROSE-AHF similarly found that low-dose dopamine and low-dose nesiritide failed to improve decongestion or preserve renal function, despite long-standing interest in these strategies[19]. These negative findings underscore that several older adjuncts lack clinical utility for managing diuretic resistance in the acute phase of HF. Overall, the mechanistic patterns observed across trials suggest that therapies targeting proximal sodium reabsorption or glucose-sodium cotransport provide more consistent and clinically meaningful augmentation of loop diuretic therapy than agents acting through neurohormonal or vasodilatory pathways[18].
A formal meta-analysis was not performed in this review because the included trials differed substantially in their primary endpoints, definitions of diuretic response, durations of follow-up, and comparator regimens, and because some studies were double-blind while others were open-label. These factors limit the validity of statistical pooling and increase the risk of drawing misleading conclusions from heterogeneous designs. Instead, the quantitative interpretation focuses on study-level effect estimates as reported by the original investigators. Accordingly, no pooled P values or combined statistical estimates were generated, and all results are presented as individual trial findings to align with narrative synthesis methodology. This approach allows meaningful comparisons by highlighting directional trends and approximate magnitudes rather than aggregated values. For example, acetazolamide improved decongestion rates by roughly 10 to 15 percentage points over placebo in ADVOR[12], while thiazide-based combinations in CLOROTIC produced greater weight loss at the expense of more renal dysfunction[14]. SGLT2 inhibitor trials consistently demonstrated neutral effects on formal diuretic efficiency but showed favorable patterns in urine output, natriuresis, and short-term clinical benefit. Importantly, these SGLT2-related benefits occurred independently of baseline kidney function, reinforcing their utility even at lower eGFRs. These findings are presented descriptively, without combining outcomes across studies, to maintain analytical rigor while still giving readers a clear understanding of the strengths and limitations of each intervention.
The evidence synthesized in this review supports a pragmatic, stepwise approach to adjunctive therapy in ADHF with insufficient diuretic response. Acetazolamide emerges as a particularly robust option for rapid decongestion, supported by improvements in natriuresis and clinically meaningful decongestion rates when added early to optimized loop therapy. Thiazide augmentation remains an effective strategy for intensifying diuresis but requires careful patient selection due to its association with renal dysfunction, suggesting a more reserved role in frail or renally vulnerable individuals. Thiazide and thiazide-like agents act at the distal convoluted tubule rather than the proximal nephron, underscoring their role as distal sequential blockade rather than proximal augmentation. SGLT2 inhibitors, including empagliflozin and dapagliflozin, demonstrate advantages that extend beyond immediate decongestion by improving natriuretic profiles, reducing loop requirements, and offering short-term prognostic benefit, making early inpatient initiation a reasonable consideration[22]. Moreover, their clinical utility is preserved across a wide spectrum of kidney function, further supporting their early use in acute HF regardless of baseline eGFR. Conversely, therapies such as high-dose spironolactone, low-dose dopamine, and nesiritide did not provide meaningful acute benefit and should not be considered primary adjuncts for overcoming diuretic resistance. The apparent lack of spironolactone benefit likely reflects confounding from background RAAS inhibitor use, potential ceiling effects from chronic MRA exposure, and the slow onset of mineralocorticoid receptor blockade, which limits its effectiveness in the acute setting. In clinical practice, diuretic resistance may be addressed by optimizing loop dosing, adding acetazolamide or an SGLT2 inhibitor as initial adjunctive therapies, and introducing a thiazide-type diuretic when additional intensification is needed and renal function permits[23].
Current HF guidelines emphasize optimization of loop diuretic therapy and support sequential nephron blockade with thiazides when clinical congestion persists, while also endorsing the early initiation of SGLT2 inhibitors as part of guideline-directed medical therapy. However, they offer limited guidance on the use of several other adjunctive agents historically considered for diuretic resistance, and they do not clearly define a preferred sequence for adjunctive pharmacologic escalation[24,25]. This review contributes to the existing literature by consolidating evidence from randomized controlled trials specifically evaluating adjunct therapies in the acute setting, an area where recommendations remain sparse. By distinguishing modern evidence-based strategies such as acetazolamide, hydrochlorothiazide, and SGLT2 inhibitors from older approaches like dopamine, nesiritide, and high-dose spironolactone that have not demonstrated meaningful benefit, the review provides a clearer mechanistic and clinical framework for decision-making. Rather than reiterating guideline statements, this synthesis clarifies where the evidence supports intervention, where it does not, and how the relative strengths and weaknesses of each strategy can inform a rational, stepwise approach to managing diuretic resistance in hospitalized patients[26].
This review has several notable strengths that enhance the reliability and clinical relevance of its findings. By including only randomized controlled trials, the synthesis draws from the highest level of primary evidence available for adjunctive pharmacologic therapy in ADHF. The included studies span multiple mechanistic classes, allowing a broad and integrated evaluation of strategies targeting different segments of renal sodium handling and neurohormonal pathways. The review followed a structured PICO framework and employed the RoB 2.0 tool to systematically assess study quality, further strengthening the rigor of the methodology. Importantly, the focus on acute HF addresses an area often overshadowed by research conducted in chronic stable HF, despite the high morbidity associated with acute presentations.
Several limitations should also be acknowledged. The included trials used heterogeneous endpoints ranging from weight loss and natriuresis to patient-reported dyspnea and composite clinical outcomes, limiting direct comparability. Some trials were modest in sample size and may be underpowered, while others, such as DICTATE-AHF[15], were open-label, raising the possibility that clinician behavior influenced diuretic titration and outcome assessment. Most studies evaluated short-term outcomes, usually within the index hospitalization or up to ninety days, leaving uncertainty regarding the long-term implications of initiating adjunctive therapy during the acute phase. Additionally, background treatment with RAAS inhibitors and chronic MRA exposure in several trials may have attenuated the incremental effects of certain adjunctive therapies-particularly high-dose spironolactone-introducing potential confounding rather than simple heterogeneity. For these reasons, a pooled quantitative synthesis was not performed; avoiding formal meta-analysis in the presence of substantial clinical and methodological heterogeneity represents a methodologically appropriate and responsible choice. Consistent with this approach, no pooled statistical estimates or P values were generated, and all quantitative findings are presented strictly as study-level results reported by the original investigators. Finally, larger, adequately powered head-to-head randomized trials are needed to directly compare mechanistically distinct adjunctive strategies and establish an evidence-based sequence for managing diuretic resistance in ADHF.
Future research should prioritize targeted, comparative trials that address key unanswered questions in the management of diuretic resistance in acute HF. High-quality head-to-head studies comparing acetazolamide, thiazide augmentation, and early SGLT2 inhibitor initiation in well-defined loop-resistant populations would provide essential insights into the optimal sequence and relative effectiveness of these widely used strategies. Incorporating biomarkers and dynamic measures such as urine sodium-guided algorithms could help personalize adjunct selection and dosing, moving beyond fixed-dose approaches. Trials specifically designed to evaluate therapeutic sequencing, such as progression from loop optimization to acetazolamide, followed by SGLT2 inhibition and finally thiazide intensification, may clarify which escalation strategy offers the best balance between efficacy and renal safety. Longer-term follow-up of patients initiated on SGLT2 inhibitors or acetazolamide during hospitalization would help determine whether early use confers sustained clinical benefit beyond the acute phase. In addition, dedicated studies in high-risk subgroups, including individuals with advanced chronic kidney disease, the very elderly, and those with preserved ejection fraction, are urgently needed to refine patient-centered treatment algorithms. Together, such research could meaningfully update clinical practice and establish a more evidence-based, individualized approach to managing diuretic resistance in ADHF.
This systematic review highlights that adjunctive pharmacologic therapies can meaningfully augment decongestion in ADHF, but their effectiveness varies considerably by mechanism and clinical context. Among the available options, acetazolamide and thiazide-based strategies demonstrated the most consistent improvements in diuretic response, while early initiation of SGLT2 inhibitors showed additional benefits in natriuresis, loop diuretic sparing, and short-term clinical outcomes. In contrast, older adjuncts such as high-dose spironolactone, low-dose dopamine, and nesiritide did not confer measurable advantages for decongestion or renal protection. By synthesizing randomized evidence across mechanistically distinct drug classes and applying structured methodological evaluation, this review provides an updated, clinically relevant framework for selecting adjunctive therapies when loop diuretics alone are insufficient. The findings underscore the importance of individualized, mechanism-guided decision-making and clarify where the current evidence can meaningfully guide practice, while also identifying key gaps that future trials must address. Together, these insights contribute to a more nuanced and evidence-driven approach to managing diuretic resistance in the acute care setting.
| 1. | Marcus G, Najjar M, Monayer A, Orbach A, Maymon SL, Kalmanovich E, Moravsky G, Grupper A, Fuchs S, Minha S. Temporal trends in acute decompensated heart failure outcomes: A single-center 11-year retrospective analysis. Int J Cardiol Cardiovasc Risk Prev. 2024;22:200306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 2. | Wu L, Rodriguez M, El Hachem K, Krittanawong C. Diuretic Treatment in Heart Failure: A Practical Guide for Clinicians. J Clin Med. 2024;13:4470. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 3. | Milhem F, Almur O, Hajjeh O, Bdair M, Dahmas AM, Haddad KB, Shubietah A, Al-Said OS, Al-Braik R, Abukhalil MM, Ayaseh QZ, Jallad H, Karaki L, Hamshari H, AbuBaha M. Advances and controversies in acute decompensated heart failure treatment: beta-blocker roles, emerging devices, and future directions. Ann Med Surg (Lond). 2025;87:5696-5719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Wilcox CS, Testani JM, Pitt B. Pathophysiology of Diuretic Resistance and Its Implications for the Management of Chronic Heart Failure. Hypertension. 2020;76:1045-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 128] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 5. | Horiuchi Y, Wettersten N. Treatment strategies for diuretic resistance in patients with heart failure. J Cardiol. 2025;85:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 6. | Guo L, Fu B, Liu Y, Hao N, Ji Y, Yang H. Diuretic resistance in patients with kidney disease: Challenges and opportunities. Biomed Pharmacother. 2023;157:114058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 7. | Blebea N, Pușcașu C, Ștefănescu E, Stăniguț AM. Diuretic Therapy: Mechanisms, Clinical Applications, and Management. J Mind Med Sci. 2025;12:26. [DOI] [Full Text] |
| 8. | Prata AA, Prata AA, Coan ACC, Scardini PG, Fernandes JM, Neto WF, Gioli-Pereira L. Protocolized natriuresis-guided diuretic therapy in acute heart failure: a systematic review and meta-analysis. Crit Care. 2025;29:474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 9. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 10. | Brown D. A Review of the PubMed PICO Tool: Using Evidence-Based Practice in Health Education. Health Promot Pract. 2020;21:496-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 220] [Article Influence: 36.7] [Reference Citation Analysis (1)] |
| 11. | Nejadghaderi SA, Balibegloo M, Rezaei N. The Cochrane risk of bias assessment tool 2 (RoB 2) versus the original RoB: A perspective on the pros and cons. Health Sci Rep. 2024;7:e2165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 113] [Article Influence: 56.5] [Reference Citation Analysis (0)] |
| 12. | Mullens W, Dauw J, Martens P, Verbrugge FH, Nijst P, Meekers E, Tartaglia K, Chenot F, Moubayed S, Dierckx R, Blouard P, Troisfontaines P, Derthoo D, Smolders W, Bruckers L, Droogne W, Ter Maaten JM, Damman K, Lassus J, Mebazaa A, Filippatos G, Ruschitzka F, Dupont M; ADVOR Study Group. Acetazolamide in Acute Decompensated Heart Failure with Volume Overload. N Engl J Med. 2022;387:1185-1195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 478] [Cited by in RCA: 400] [Article Influence: 100.0] [Reference Citation Analysis (0)] |
| 13. | Cox ZL, Hung R, Lenihan DJ, Testani JM. Diuretic Strategies for Loop Diuretic Resistance in Acute Heart Failure: The 3T Trial. JACC Heart Fail. 2020;8:157-168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 127] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 14. | Trullàs JC, Morales-Rull JL, Casado J, Carrera-Izquierdo M, Sánchez-Marteles M, Conde-Martel A, Dávila-Ramos MF, Llácer P, Salamanca-Bautista P, Pérez-Silvestre J, Plasín MÁ, Cerqueiro JM, Gil P, Formiga F, Manzano L; CLOROTIC trial investigators. Combining loop with thiazide diuretics for decompensated heart failure: the CLOROTIC trial. Eur Heart J. 2023;44:411-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 216] [Article Influence: 72.0] [Reference Citation Analysis (0)] |
| 15. | Cox ZL, Collins SP, Hernandez GA, McRae AT 3rd, Davidson BT, Adams K, Aaron M, Cunningham L, Jenkins CA, Lindsell CJ, Harrell FE Jr, Kampe C, Miller KF, Stubblefield WB, Lindenfeld J. Efficacy and Safety of Dapagliflozin in Patients With Acute Heart Failure. J Am Coll Cardiol. 2024;83:1295-1306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 113] [Article Influence: 56.5] [Reference Citation Analysis (0)] |
| 16. | Damman K, Beusekamp JC, Boorsma EM, Swart HP, Smilde TDJ, Elvan A, van Eck JWM, Heerspink HJL, Voors AA. Randomized, double-blind, placebo-controlled, multicentre pilot study on the effects of empagliflozin on clinical outcomes in patients with acute decompensated heart failure (EMPA-RESPONSE-AHF). Eur J Heart Fail. 2020;22:713-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 394] [Cited by in RCA: 345] [Article Influence: 57.5] [Reference Citation Analysis (3)] |
| 17. | Voors AA, Angermann CE, Teerlink JR, Collins SP, Kosiborod M, Biegus J, Ferreira JP, Nassif ME, Psotka MA, Tromp J, Borleffs CJW, Ma C, Comin-Colet J, Fu M, Janssens SP, Kiss RG, Mentz RJ, Sakata Y, Schirmer H, Schou M, Schulze PC, Spinarova L, Volterrani M, Wranicz JK, Zeymer U, Zieroth S, Brueckmann M, Blatchford JP, Salsali A, Ponikowski P. The SGLT2 inhibitor empagliflozin in patients hospitalized for acute heart failure: a multinational randomized trial. Nat Med. 2022;28:568-574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 760] [Cited by in RCA: 648] [Article Influence: 162.0] [Reference Citation Analysis (7)] |
| 18. | Butler J, Anstrom KJ, Felker GM, Givertz MM, Kalogeropoulos AP, Konstam MA, Mann DL, Margulies KB, McNulty SE, Mentz RJ, Redfield MM, Tang WHW, Whellan DJ, Shah M, Desvigne-Nickens P, Hernandez AF, Braunwald E; National Heart Lung and Blood Institute Heart Failure Clinical Research Network. Efficacy and Safety of Spironolactone in Acute Heart Failure: The ATHENA-HF Randomized Clinical Trial. JAMA Cardiol. 2017;2:950-958. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 273] [Cited by in RCA: 240] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 19. | Chen HH, Anstrom KJ, Givertz MM, Stevenson LW, Semigran MJ, Goldsmith SR, Bart BA, Bull DA, Stehlik J, LeWinter MM, Konstam MA, Huggins GS, Rouleau JL, O'Meara E, Tang WH, Starling RC, Butler J, Deswal A, Felker GM, O'Connor CM, Bonita RE, Margulies KB, Cappola TP, Ofili EO, Mann DL, Dávila-Román VG, McNulty SE, Borlaug BA, Velazquez EJ, Lee KL, Shah MR, Hernandez AF, Braunwald E, Redfield MM; NHLBI Heart Failure Clinical Research Network. Low-dose dopamine or low-dose nesiritide in acute heart failure with renal dysfunction: the ROSE acute heart failure randomized trial. JAMA. 2013;310:2533-2543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 437] [Cited by in RCA: 382] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 20. | Stachteas P, Nasoufidou A, Patoulias D, Karakasis P, Karagiannidis E, Mourtzos MA, Samaras A, Apostolidou X, Fragakis N. The Role of Sodium-Glucose Co-Transporter-2 Inhibitors on Diuretic Resistance in Heart Failure. Int J Mol Sci. 2024;25:3122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 21. | George J, Wilson Tang WH. Evolving Decongestion Strategies in the Management of Acute Heart Failure. Curr Treat Options Cardiovasc Med. 2025;27:71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 22. | Siddiqi AK, Maniya MT, Alam MT, Ambrosy AP, Fudim M, Greene SJ, Khan MS. Acetazolamide as an Adjunctive Diuretic Therapy for Patients with Acute Decompensated Heart Failure: A Systematic Review and Meta-Analysis. Am J Cardiovasc Drugs. 2024;24:273-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 23. | Ostrominski JW, Vaduganathan M. Evolving therapeutic strategies for patients hospitalized with new or worsening heart failure across the spectrum of left ventricular ejection fraction. Clin Cardiol. 2022;45 Suppl 1:S40-S51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 24. | Authors/Task Force Members:; McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, Burri H, Butler J, Čelutkienė J, Chioncel O, Cleland JGF, Coats AJS, Crespo-Leiro MG, Farmakis D, Gilard M, Heymans S, Hoes AW, Jaarsma T, Jankowska EA, Lainscak M, Lam CSP, Lyon AR, McMurray JJV, Mebazaa A, Mindham R, Muneretto C, Francesco Piepoli M, Price S, Rosano GMC, Ruschitzka F, Kathrine Skibelund A; ESC Scientific Document Group. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2022;24:4-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1836] [Cited by in RCA: 1599] [Article Influence: 399.8] [Reference Citation Analysis (4)] |
| 25. | Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, Deswal A, Drazner MH, Dunlay SM, Evers LR, Fang JC, Fedson SE, Fonarow GC, Hayek SS, Hernandez AF, Khazanie P, Kittleson MM, Lee CS, Link MS, Milano CA, Nnacheta LC, Sandhu AT, Stevenson LW, Vardeny O, Vest AR, Yancy CW. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79:e263-e421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2233] [Cited by in RCA: 1898] [Article Influence: 474.5] [Reference Citation Analysis (4)] |
| 26. | Milbradt TL, Sudo RYU, Gobbo MODS, Akinfenwa S, Moura B. Acetazolamide therapy in patients with acute heart failure: a systematic review and meta-analysis of randomized controlled trials. Heart Fail Rev. 2024;29:1039-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |