Published online Mar 18, 2026. doi: 10.13105/wjma.v14.i1.113789
Revised: October 9, 2025
Accepted: December 11, 2025
Published online: March 18, 2026
Processing time: 187 Days and 3.5 Hours
Metabolic dysfunction-associated fatty liver disease (MAFLD) may actively contribute to the development, progression, and outcomes of colorectal cancer (CRC) in addition to coexisting with it, according to mounting epidemiological and mechanistic evidence. Obesity, type 2 diabetes, dietary patterns, and systemic inflammation are among the risk factors that overlap between the two conditions, indicating a convergent pathophysiological axis. Insulin resistance, altered adi
Core Tip: Metabolic dysfunction-associated fatty liver disease may actively contribute to the development, progression, and outcomes of colorectal cancer in addition to coexisting with it, according to mounting epidemiological and mechanistic evidence. Obesity, type 2 diabetes, dietary patterns, and systemic inflammation are among the risk factors that overlap between the two conditions, indicating a convergent pathophysiological axis. Changing one’s lifestyle, controlling metabolic risk factors, and using new therapeutic targets like anti-inflammatory and antifibrotic drugs may help slow the progression of metabolic dysfunction-associated fatty liver disease and lower the burden of colorectal cancer. This perspective emphasized the necessity of multidisciplinary strategies aimed at prevention, early detection, and therapeutic optimization.
- Citation: Fouad Y, Pan Z, Mostafa AM, Eslam M. Metabolic dysfunction-associated fatty liver disease: A key player in colorectal cancer development and progression. World J Meta-Anal 2026; 14(1): 113789
- URL: https://www.wjgnet.com/2308-3840/full/v14/i1/113789.htm
- DOI: https://dx.doi.org/10.13105/wjma.v14.i1.113789
Metabolic dysfunction-associated fatty liver disease (MAFLD) affects over 25% of the global population and is recognized as the most prevalent chronic liver condition, significantly contributing to both hepatic and extrahepatic morbidity[1]. The updated terminology, MAFLD, emphasizes the strong link between the condition, metabolic dysfunction, and cardiometabolic risk[2].
Globally, approximately 1.9 million new cases of colorectal cancer (CRC) are reported annually, making it the second leading cause of cancer-related deaths and the third most common cancer overall[3]. While age and heredity are established risk factors, lifestyle and metabolic factors are increasingly implicated in the rising incidence of early-onset CRC, particularly in high-income countries[4].
Emerging evidence from epidemiological and Mendelian randomization studies identifies MAFLD as an independent risk factor for CRC alongside obesity and diabetes[5]. A bidirectional relationship is suggested, impacting diagnosis, disease progression, treatment outcomes, and recurrence through shared pathways, including insulin resistance, bile acid dysregulation, gut microbiome alterations, and chronic low-grade inflammation.
This article aimed to explore the intricate relationship between MAFLD and CRC by integrating historical context, molecular insights, and clinical data. It highlighted common etiological pathways, diagnostic challenges, and op
In 1836 Addison became the first to characterize fatty liver[6]. Subsequently, pathologists observed a link between liver histology changes in individuals with diabetes, severe obesity, and alcoholism. A potential link between obesity and fatty liver was observed by Pepper in 1885[7]. Later, in 1938, Connor[8] linked fatty liver infiltration to the development of cirrhosis in individuals with diabetes. The perilobular fibrosis observed was attributed to tissue anoxia and mechanical factors. The first histological description of fatty liver infiltration in individuals with obesity appeared in 1958, and the syndrome was further characterized pathologically and clinically by 1962[9]. Numerous studies between the 1950s and 1970s pathologically confirmed fatty liver disease in individuals with diabetes or obesity[10].
Since the initial description of nonalcoholic fatty liver disease (NAFLD) in 1980[11], there have been multiple attempts to rename the condition by various researchers and organizations. New definitions and diagnostic criteria have emerged in recent years due to the need to emphasize the clear link between liver steatosis and metabolic dysfunction (i.e. insulin resistance, overweight/obesity, type 2 diabetes, and metabolic syndrome) and the desire to define a condition in a positive rather than negative way. Bayoumi et al[12] proposed MAFLD in 2020. More recently, a Delphi consensus that was supported by a number of international hepatologic societies suggested a new set of diagnostic criteria as well as a new term: Metabolic dysfunction-associated steatotic liver disease (MASLD). There is a high degree of agreement between the definitions of MAFLD and MASLD. Their primary differences are in the quantity of metabolic abnormalities required to characterize “metabolic dysfunction” in people of normal weight and in alcohol intake. In fact, the recent Delphi consensus included the metabolic dysfunction and alcohol-related liver disease entity, a condition in which steatosis, metabolic dysfunction, and moderate alcohol intake coexist even though MAFLD does not exclude patients with significant alcohol consumption[13].
These straightforward criteria simplify the diagnosis of fatty liver disease. In addition to diabetes mellitus, obesity/overweight, or two of seven metabolic dysfunction criteria, the consensus supports diagnosing MAFLD based on steatosis, detected through imaging or histopathology. This new nomenclature and methodology enhance the un
CRC has been recognized since ancient times with the earliest documented cases dating back to 3000-1500 before common era (BCE) in Egyptian mummies. Around 1550 BCE, the Ebers Papyrus described rectal tumors and suggested early cauterization as a treatment. Hippocrates (460-370 BCE) categorized intestinal tumors as “karkinos” (cancer). The first successful surgical removal of a colon tumor occurred in 1739, but high death rates persisted until the 19th century when anesthesia and antisepsis improved surgical outcomes. The invention of colonoscopy in the 1960s transformed CRC diagnosis and prevention by enabling early detection and polyp removal[14].
We searched the PubMed, Web of Science, and Google Scholar databases for relevant literature for this review. The following keywords were used: Colorectal carcinoma; MAFLD; fatty liver; MASLD; NAFLD; and colon cancer. In
The highest prevalence of MAFLD is reported in the Middle East and South American regions. The increase in MAFLD prevalence parallels the global rise in diabetes, obesity, and lack of physical exercise rates.
On the other hand, the incidence rates of CRC vary significantly by region due to Westernized diets (high in red and processed meats, low in fiber), obesity, and sedentary lifestyles with the highest rates in developed countries like Australia, New Zealand, Europe, and North America. Despite rising incidence due to dietary changes and increased urbanization, lower-income countries in Africa and South-Central Asia have lower CRC rates[3].
Ninety percent of CRC cases occur in individuals aged 50 or older, primarily affecting elderly adults. However, recent trends indicate a concerning rise in early-onset CRC (diagnosed before age 50), especially in high-income countries. This increase may be related to lifestyle factors and metabolic syndrome. Males are 25%-30% more likely than females to be at risk, potentially due to hormonal differences and higher rates of alcohol and smoking. Socioeconomic disparities, genetic factors (such as a higher prevalence of KRAS mutations), and delayed screenings contribute to higher incidence and mortality rates among African Americans in the United States, highlighting racial disparities[4].
Modifiable risk factors for CRC include obesity, smoking, excessive alcohol use, and diets high in processed meats (classified as group 1 carcinogens by the International Agency of Research on Cancer)[15]. Non-modifiable risk factors include hereditary conditions like Lynch syndrome and familial adenomatous polyposis and inflammatory bowel disease. Early identification and screening programs (fecal immunochemical testing, colonoscopy) have significantly reduced CRC mortality rates[16]. Recent advances in immunotherapy (checkpoint inhibitors for microsatellite instability-high tumors) and targeted therapies [anti-epidermal growth factor receptor (EGFR) medications] have improved survival rates for patients with metastasis[17].
In conclusion, changing epidemiologic trends driven by lifestyle, screening availability, and hereditary factors continue to make both MAFLD and CRC a significant global health concern. Public health initiatives focused on early detection, lifestyle modifications, and equitable access to healthcare are essential to reduce the burden of CRC.
The link between MAFLD and CRC was initially suggested when metabolic syndrome was identified as a common risk factor for CRC in the early 2000s. Subsequently, multiple epidemiological studies have shown a direct link between MAFLD and CRC with a recent meta-analysis confirming that MAFLD is an independent predictor of colorectal adenomas and cancer even after adjusting for diabetes and obesity[18]. Notably, emerging research suggests that MAFLD criteria are superior to the former NAFLD criteria in identifying those at increased risk of both hepatocellular cancer and extrahepatic malignancy including CRC[19,20].
Another line of evidence indicating a higher prevalence of colorectal adenomas and CRC in individuals with MAFLD and suggesting a causal link between these two conditions came from a Mendelian randomization study[5]. For instance, a study included 770180 controls and 8434 patients with genetically predicted MAFLD. Forward analysis using the inverse variance weighted approach revealed a statistically significant positive correlation between a genetic predisposition to MAFLD and the risk of CRC [odds ratio = 1.270; 95% confidence interval (CI): 1.154-1.398; P = 1.092 × 10-6]. Interestingly, the reverse Mendelian randomization study did not support the hypothesis that CRC causes MAFLD, indicating that the relationship may be only unidirectional (from MAFLD to CRC)[5] and suggesting that MAFLD is more likely to act as a risk amplifier or precursor for colorectal neoplasia and cancer rather than a consequence of it.
Despite the value of the knowledge gained by the Mendelian randomization studies, challenges remain. While reducing confounding, they are heavily reliant on the quality and specificity of genetic instruments[21]. As the genetic architecture of MAFLD continues to evolve, future Mendelian randomization studies incorporating newer MAFLD criteria or hepatic fibrosis stages may further refine risk estimates.
According to recent studies, MAFLD may promote the liver metastases of CRC. After an average follow-up of 65.6 months, 21.4% of 388 patients who had CRC surgery between 2008 and 2015 experienced recurrence, according to a retrospective study. Twenty-five patients developed liver metastases: 17.8% in the MAFLD group (8/45) and 5.0% in the non-MAFLD group (17/343; P = 0.004). MAFLD and cancer stage were independently associated with recurrence, and the group with MAFLD had a significantly lower liver metastasis-free survival (P < 0.001)[22]. Hepatic steatosis was identified as a significant risk factor for metachronous colorectal liver metastasis in a thorough analysis of 1815 individuals (odds ratio = 1.90, 95%CI: 1.35-2.66, P < 0.001). According to the results, patients with MAFLD and a high fibrosis score at the time of CRC diagnosis are at a higher risk of developing metachronous liver metastases[23].
Although there is growing recognition of the connection between MAFLD and an increased risk of CRC, the reverse relationship is less recognized and how the management of CRC affects the progression and treatment of MAFLD.
Systemic chemotherapy, a key component in CRC treatment, can significantly impact liver health. For instance, irinotecan has been associated with damage resembling steatohepatitis, similar to metabolic steatohepatitis. On the other hand, oxaliplatin-based regimens, such as folinic acid, fluorouracil, and oxaliplatin, are linked to sinusoidal obstruction syndrome[24].
Liver resection is a common surgical approach, particularly for treating CRC liver metastases. However, patients with MAFLD undergoing liver resection are at a greater risk of liver failure and postoperative complications, especially if fibrosis is not adequately identified beforehand. Steatotic livers are more vulnerable to ischemia-reperfusion injury, which can affect their ability to regenerate and recover after a major hepatectomy. Therefore, managing liver health is crucial during perioperative planning for patients with CRC[25].
Sarcopenia, metabolic changes, and nutritional deficiencies are frequent side effects of CRC treatments. These changes can exacerbate insulin resistance, increase hepatic fat accumulation, and disrupt mitochondrial function in the liver, all of which contribute to the development of fatty liver disease. Conversely, intentional dietary modifications and exercise, often included in CRC survivorship programs, can offer opportunities to prevent or reverse the accumulation of hepatic fat. This underscores the importance of integrated metabolic care during oncology follow-ups[26,27].
New CRC treatments, particularly immune checkpoint inhibitors for microsatellite instability-high tumors, may significantly influence the pathophysiology of MAFLD. Cancer and its treatment alter the gut microbiota, which is known to affect both CRC and MAFLD. Additionally, immune modulation resulting from these treatments may impact liver inflammatory pathways. Patients with pre-existing liver conditions are also at risk for immune-related hepatotoxicity[28].
Several potential mechanisms have been postulated underlying this connection including insulin resistance, chronic systemic inflammation, altered gut microbiota, and pro-oncogenic lipid metabolites[29].
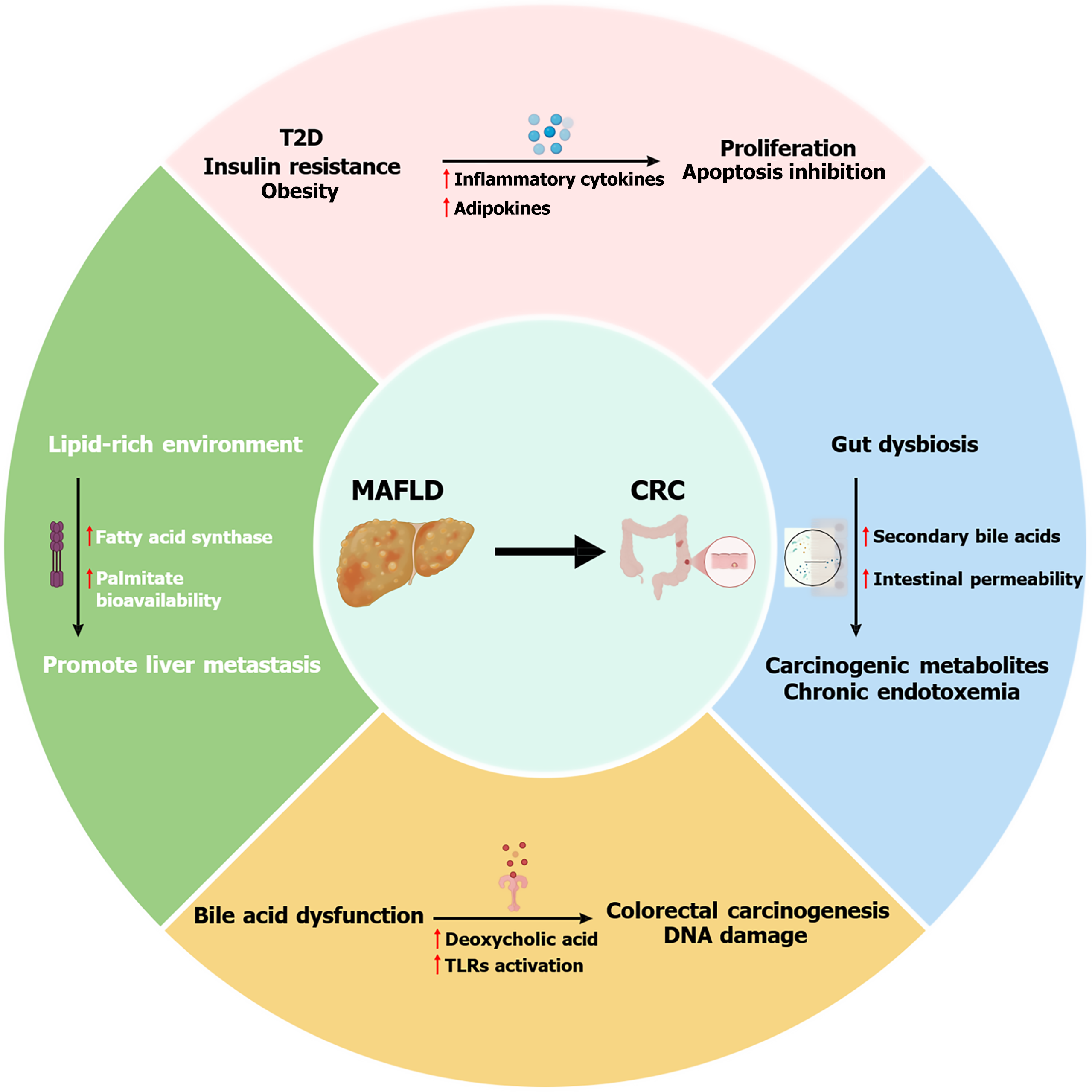
From a pathophysiological perspective, metabolic dysfunction is a key etiological factor in both CRC and MAFLD pathogenesis and likely is the main pathological link between the two conditions. Type 2 diabetes mellitus and insulin resistance in MAFLD can lead to hyperinsulinemia and increased insulin-like growth factor-1 activity, promoting epithelial cell proliferation and inhibiting apoptosis in the colonic mucosa - key processes in cancer development[30] fostering an environment that is both proinflammatory and procarcinogenic cytokines [e.g., tumor necrosis factor-α and interleukin (IL)-6][31].
Gut dysbiosis is increasingly recognized as another link between NAFLD and CRC. Patients with MAFLD often exhibit reduced microbial diversity and an overabundance of proinflammatory bacteria, leading to the production of carcinogenic metabolites (e.g., secondary bile acids) or increased intestinal permeability, which results in chronic endotoxemia (the presence of bacterial endotoxins in the bloodstream) and inflammation that can promote colon tumor development[32]. Changes in microbial composition (e.g., increased Fusobacterium nucleatum and decreased Faecalibacterium prausnitzii) disrupt intestinal barrier integrity and activate oncogenic pathways, contributing to the development of CRC and the progression of MAFLD[33,34].
Additionally, abnormalities in bile acid metabolism, which are prevalent in NAFLD, raise secondary bile acids such deoxycholic acid as well as activation of toll like receptor signaling, which has been linked to colorectal carcinogenesis and DNA damage[35,36]. These common mechanisms are made worse by lifestyle choices including a high-fat, low-fiber diet and inactivity.
Chronic low-grade inflammation caused by adipose tissue malfunction in obesity results in increased levels of inflammatory cytokines (e.g., tumor necrosis factor-α, interleukin 6) and adipokines (e.g., adiponectin and leptin), which in turn promote colorectal carcinogenesis and hepatic steatosis (a condition characterized by the abnormal accumulation of fat (specifically triglycerides) inside liver cells comprising at least 5% of the weight of the liver)[37].
Additionally, MAFLD facilitated the metabolic condition of CRC liver metastasis by promoting the stemness of metastatic CRC cells for their colonization and growth in hepatic niches. Metabolically, the lipid-rich environment in MAFLD promoted the formation of de novo palmitate in metastatic CRC cells by upregulating fatty acid synthase. Additionally, increased intracellular palmitate bioavailability promoted EGFR palmitoylation, enhancing the stability and plasma membrane localization of the protein. AN overview of the shared pathophysiological mechanisms between MAFLD and CRC is shown in Figure 1.
When two of the most common diseases in the world, MAFLD and CRC, coexist, diagnosing each can become particularly challenging. Their similar metabolic underpinnings, overlapping risk factors, and systemic symptoms can obscure clinical presentations and affect diagnostic accuracy. Prompt and accurate diagnosis is crucial as delayed recognition can impact staging, treatment planning, and patient outcomes.
Liver imaging plays a critical role in staging and monitoring CRC. However, hepatic steatosis and fibrosis associated with MAFLD can mask or resemble metastatic lesions on CT and ultrasound. Diffuse steatosis reduces liver parenchymal attenuation, making it difficult to identify small hypodense metastases, especially on non-contrast CT images[38]. In cases of cirrhosis or fibrosis linked to MAFLD, regenerative nodules can be mistaken for metastatic deposits.
The best way to identify and characterize focal liver lesions in fatty or fibrotic livers is through contrast-enhanced magnetic resonance imaging using liver-specific agents, such as gadoxetate disodium[39]. While positron emission tomography-CT has decreased sensitivity in steatotic livers, it may still help differentiate between inflammatory or fibrotic changes and actual metastases.
Liver function tests may be slightly elevated in both early-stage MAFLD and CRC with liver involvement. While even small-volume metastases in patients with CRC can raise alkaline phosphatase or gamma-glutamyl transferase levels, MAFLD alone can produce similar elevations without metastases. Additionally, elevated carcinoembryonic antigen levels in patients with MAFLD due to inflammation may incorrectly suggest cancer recurrence[40]. It is important to exercise caution when interpreting tumor markers like carcinoembryonic antigen in patients with active liver disease. Pairing biochemical alterations with fibrosis staging tools (such as fibrosis-4 index or transient elastography) and dynamic imaging results is advisable.
Nonspecific gastrointestinal symptoms such as bloating, fatigue, or discomfort in the right upper quadrant are common in patients with MAFLD. These symptoms, particularly in elderly individuals with multiple comorbidities, may obscure early signs of CRC, such as occult bleeding or changes in bowel habits. Physicians may erroneously attribute mild anemia to chronic liver disease rather than consider a colonic cause. Hence, in patients with MAFLD aged 50 or older, it is crucial to maintain a high index of suspicion for CRC, especially if they show symptoms like anemia, unexplained weight loss, or changes in bowel habits. Active colonoscopy-based CRC screening should be pursued according to risk-adjusted guidelines.
Chronic inflammation and dysbiosis are associated with both MAFLD and CRC. This systemic inflammation can impair mucosal healing and immune responses, potentially reducing the specificity of histopathological or endoscopic results. For instance, finding flat or subtle neoplastic lesions in patients with non-alcoholic steatohepatitis-related colitis may be more challenging[41]. For patients with MAFLD at a higher risk of CRC, high-definition colonoscopy combined with chromoendoscopy is recommended. Additionally, for greater sensitivity, stool-based molecular testing (such as fecal immunochemical test-DNA or methylated SEPT9 testing) may be used alongside colonoscopy.
The interaction between CRC and MAFLD can lead to delays in diagnosis for both conditions. For example, CRC-related cachexia or weight loss due to treatment may temporarily alleviate hepatic steatosis, misguiding clinicians about the severity of MAFLD. Conversely, a focus on managing fatty liver could postpone investigations for mild CRC symptoms, particularly in individuals with diabetes or obesity experiencing complex gastrointestinal issues. We recommend creating dual-assessment protocols and integrated diagnostic pathways for high-risk patients (e.g., colonoscopy combined with liver elastography in diabetics or obese adults). Furthermore, shared care models that promote collaboration among hepatologists, gastroenterologists, and oncologists should be encouraged.
In the future, AI-based imaging technologies that can differentiate between steatotic nodules and metastases show promise although they require further validation. Noninvasive, tumor-specific diagnostics unaffected by liver-related confounders may be available through liquid biopsy techniques. Advancing guidelines for CRC screening in high-risk MAFLD populations and vice versa is essential[42].
A common cause of the global increase in CRC and MAFLD is metabolic dysregulation brought on by contemporary lifestyle choices. Obesity, insulin resistance, chronic inflammation, and sedentary lifestyles are the main causes of the common coexistence of both illnesses. Remarkably, new research indicates that a number of management techniques not only lessen the impact of MAFLD but also lower risk and enhance CRC results. This makes it possible for clinical practice to adopt an integrated, dual-benefit strategy.
Lifestyle modification remains the first-line treatment in both MAFLD and CRC prevention. Caloric restriction, increased physical activity, and weight loss of at least 7%-10% have been shown to significantly reduce liver fat, improve hepatic inflammation, and reverse early fibrosis in MAFLD[1]. Both diseases respond to improved insulin sensitivity, lowered systemic inflammation, and modulation of gut microbiota diversity[34].
Exercise: Exercise can slow the progression of cancer, especially colon cancer, according to preclinical research. Furthermore, observational studies have demonstrated that individuals with CRC, especially those with stage III colon cancer, who engage in more recreational physical activity following treatment are less likely to die or see a recurrence of their disease. The effects of exercise on immunological function, metabolic growth factors, and inflammation may account for these connections. Due to the methodological constraints of observational designs, the results are uncertain even though these studies suggest a survival benefit associated with exercise[43].
A phase 3 randomized trial was conducted at 55 centers in which patients with resected colon cancer who had completed adjuvant chemotherapy were randomly assigned to either receive health education materials alone (health-education group) or participate in a structured exercise program (exercise group) for 3 years. The primary result was survival without disease. Between 2009 and 2024 a total of 889 patients were randomized to either the health-education group (444 patients) or the exercise group (445 patients). Compared with the health education group, the exercise group had a significantly longer disease-free survival at a median follow-up of 7.9 years. The disease-free survival rate for the exercise group was 80.3% during a 5-year period compared with 73.9% for the health education group. Starting a 3-year structured exercise program soon after adjuvant treatment for colon cancer resulted in significantly longer disease-free survival and outcomes consistent with prolonged overall life[44].
Dietary patterns of Mediterranean and plant-based diets: A diet high in whole grains, fruits, vegetables, legumes, olive oil, and seafood, known as the Mediterranean diet, has been associated with a lower incidence of both MAFLD and CRC. Plant-based diets that are high in dietary fiber and polyphenols decrease colon carcinogenesis and liver steatosis by promoting gut microbiota diversity and reducing hepatic lipogenesis. A shared mechanism of short-chain fatty acids, such as butyrate, which have anti-inflammatory and anti-tumor properties in the liver and colon, are elevated by dietary fiber[45].
Coffee consumption: It is becoming more widely acknowledged that advanced fibrosis in MAFLD represents a systemic carcinogenic state. Early CRC screening may be beneficial for patients with metabolic syndrome and MAFLD, especially if additional risk factors (such as obesity and family history) are present. Cost-effective cancer prevention may be possible with risk-adapted colonoscopy techniques. Coffee consumption has been associated with a decreased risk of CRC. The association between coffee drinking and all-cause mortality and CRC recurrence was evaluated using data from a prospective cohort analysis of 1719 stage I-III CRC patients in the Netherlands. Overall, 309 deaths were reported at a median follow-up of 6.6 years while 257 recurrences were reported at a normal follow-up of 6.2 years. Drinking more than four cups of coffee daily compared with fewer than two cups per day was associated with a 32% lower risk of CRC recurrence (95%CI: 0.49-0.94,). Three to five cups of coffee per day seems to be the ideal amount with four cups per day showing the lowest risk (hazard ratio = 0.68, 95%CI: 0.53-0.88). Coffee consumption and all-cause mortality had a U-shaped relationship. These results suggest that coffee consumption may be associated with a lower risk of CRC recurrence and all-cause death[46].
Coffee suppresses the kynurenine pathway, which dramatically reduces tumor development, prolongs survival, and boosts CD8+ T cell activity in CRC, according to a recent study that examined the potential for improving the efficacy of programmed cell death protein 1 (PD-1) immunotherapy in CRC. By reducing kynurenine via the Krüppel-like factor 4, collagen type XII alpha 1, mitogen-activated protein kinase, and indoleamine 2,3-dioxygenase 1 axis, caffeine lessens CD8+ T cell exhaustion. Together, caffeine and PD-1 therapy further improve survival, highlighting the necessity of including dietary advice in cancer treatment to improve outcomes and broaden the range of treatments accessible. The ability of caffeine to enhance PD-1 immunotherapy for CRC by blocking the kynurenine pathway raises the possibility that it could be taken as a dietary supplement[47].
Coffee has been shown to protect against liver and prostate cancer in both observational and magnetic resonance imaging investigations. The anti-inflammatory and antioxidant properties of the bioactive ingredients of coffee, such as caffeine, diterpenes, and chlorogenic acids, are believed to offer potential anticancer effects. These compounds can cause apoptosis, reduce oxidative stress, inhibit the growth of cancer cells, and change hormone levels. The review emphasized the need for more research to clarify dose-response correlations, causal relationships, and the molecular mechanisms underlying these linkages. Coffee consumption raises the risk of certain cancers, highlighting the complexity of its health consequences even if it appears to help prevent cancer and encourage healthy aging[48].
Glucagon-like peptide 1 (GLP-1) receptor agonists, such as semaglutide and liraglutide, were originally developed to treat diabetes but may also help treat MAFLD. By reducing fibrosis, inflammation, and hepatic steatosis, these medications can lead to significant histological remission of steatohepatitis[49]. Furthermore, new observational data and preclinical studies suggest that GLP-1 receptor agonists may have anti-tumorigenic effects in CRC through mechanisms like reduced insulin/insulin-like growth factor-1 signaling and modulation of the Wnt/β-catenin pathway[50]. Sodium-glucose co-transporter 2 inhibitors, such as empagliflozin, have shown preclinical protective effects against CRC by altering the adenosine monophosphate-activated protein kinase/mechanistic target of rapamycin pathway and decreasing liver fat and fibrosis[51].
Low-dose aspirin and other nonsteroidal anti-inflammatory drugs can protect against CRC by preventing cyclooxygenase-2-mediated prostaglandin synthesis. Additionally, aspirin may reduce hepatic inflammation and fibrosis in MAFLD by preventing platelet activation and Kupffer cell stimulation[52].
The Food and Drug Administration-approved fatty acid synthase inhibitor orlistat reduces the production of endogenous palmitate activated by MAFLD and blocks the palmitoylation of EGFR. When combined with conventional chemotherapy, orlistat may decrease CRC cell stemness and limit liver metastases. This suggests that the metabolic environment of MAFLD promotes the production of endogenous palmitate in metastatic CRC cells, enhancing cell stemness through palmitoylation of EGFR. Thus, orlistat could serve as a useful adjunct treatment for individuals with CRC who also have MAFLD to prevent liver metastases[53]. Another study demonstrated that in several mouse models MAFLD promotes the release of the chemokine C-X-C motif chemokine ligand 5 from F4/80+ Kupffer cells. This in turn attracts C-X-C motif chemokine receptor 2 + myeloid-derived suppressor cells, contributing to the development of colorectal liver metastases. A dual treatment approach using a C-X-C motif chemokine receptor 1/2 inhibitor, reparixin, has been shown to treat established colorectal liver metastases in mice with MAFLD[54].
As MAFLD is increasingly recognized as a multisystem disease with oncologic relevance, future guidelines must incorporate oncological history, particularly CRC, into risk stratification and management algorithms. Key implications include: (1) Pretherapeutic liver assessment. Routine hepatic imaging and noninvasive fibrosis scoring (e.g., fibrosis-4 index, FibroScan) should be integrated into baseline work-up for patients with CRC, especially those receiving hepatotoxic agents; (2) Surveillance post-therapy. CRC survivors with chemotherapy-induced liver injury or known MAFLD should undergo regular liver health monitoring to detect progression toward advanced fibrosis or cirrhosis; (3) Multidisciplinary care models. Incorporating hepatologists into CRC treatment teams may improve outcomes by optimizing liver health before, during, and after oncological therapies; (4) Personalized metabolic intervention. Tailored nutritional and physical activity plans should be developed in patients with CRC to reduce hepatic steatosis and counteract treatment-related metabolic derangements; and (5) Liver-sparing strategies in CRC treatment. When feasible, choosing less hepatotoxic agents or using protective strategies (e.g., bevacizumab to reduce sinusoidal obstruction syndrome) should be considered in patients with MAFLD undergoing chemotherapy.
This review emphasized the growing understanding of the complex relationship between MAFLD and CRC, highlighting the shared inflammatory and metabolic foundations of these two extremely common conditions. Even though there has been a lot of progress in understanding the reciprocal influence between MAFLD and CRC, there are still a number of important questions. The development of targeted therapies that target the shared pathways underlying both diseases, the improvement of risk prediction models, and the discovery of new biomarkers for early detection should be the main goals of future research. Additionally, to assess the effectiveness of dual-benefit interventions, like GLP-1 receptor agonists and microbiome-targeted strategies, in enhancing outcomes for patients with MAFLD and CRC, carefully planned clinical trials are required. In order to convert these research findings into better patient care, a multidisciplinary strategy combining expertise in hepatology, gastroenterology, and oncology will be crucial.
| 1. | Zhou XD, Targher G, Byrne CD, Somers V, Kim SU, Chahal CAA, Wong VW, Cai J, Shapiro MD, Eslam M, Steg PG, Sung KC, Misra A, Li JJ, Brotons C, Huang Y, Papatheodoridis GV, Sun A, Yilmaz Y, Chan WK, Huang H, Méndez-Sánchez N, Alqahtani SA, Cortez-Pinto H, Lip GYH, de Knegt RJ, Ocama P, Romero-Gomez M, Fudim M, Sebastiani G, Son JW, Ryan JD, Ikonomidis I, Treeprasertsuk S, Pastori D, Lupsor-Platon M, Tilg H, Ghazinyan H, Boursier J, Hamaguchi M, Nguyen MH, Fan JG, Goh GB, Al Mahtab M, Hamid S, Perera N, George J, Zheng MH. An international multidisciplinary consensus statement on MAFLD and the risk of CVD. Hepatol Int. 2023;17:773-791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 158] [Cited by in RCA: 141] [Article Influence: 47.0] [Reference Citation Analysis (1)] |
| 2. | Eslam M, Newsome PN, Sarin SK, Anstee QM, Targher G, Romero-Gomez M, Zelber-Sagi S, Wai-Sun Wong V, Dufour JF, Schattenberg JM, Kawaguchi T, Arrese M, Valenti L, Shiha G, Tiribelli C, Yki-Järvinen H, Fan JG, Grønbæk H, Yilmaz Y, Cortez-Pinto H, Oliveira CP, Bedossa P, Adams LA, Zheng MH, Fouad Y, Chan WK, Mendez-Sanchez N, Ahn SH, Castera L, Bugianesi E, Ratziu V, George J. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J Hepatol. 2020;73:202-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3640] [Cited by in RCA: 3345] [Article Influence: 557.5] [Reference Citation Analysis (6)] |
| 3. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69784] [Article Influence: 13956.8] [Reference Citation Analysis (49)] |
| 4. | Siegel RL, Jakubowski CD, Fedewa SA, Davis A, Azad NS. Colorectal Cancer in the Young: Epidemiology, Prevention, Management. Am Soc Clin Oncol Educ Book. 2020;40:1-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 117] [Article Influence: 19.5] [Reference Citation Analysis (1)] |
| 5. | Wang FC, Dong Y, Xu ZQ, Gao YJ, Yan JG, Cao LL, Feng DN, Liu C, Zhong YW, Zhang M, Xin SJ. Characteristics analysis of hepatitis B core-related antigen in children with hepatitis B e antigen-positive chronic viral hepatitis B infection. Eur J Gastroenterol Hepatol. 2024;36:1238-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 6. | Ayonrinde OT. Historical narrative from fatty liver in the nineteenth century to contemporary NAFLD - Reconciling the present with the past. JHEP Rep. 2021;3:100261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (2)] |
| 7. | Pepper W, editor. A System of Practical Medicine (In Five Volumes). Philadelphia: Lea Brothers and Company, 1885. |
| 8. | Connor CL. Fatty infiltration of the liver and the development of cirrhosis in diabetes and chronic alcoholism. Am J Pathol. 1938;14:347-364.9. [PubMed] |
| 9. | Burt AD, Ferrell LD, Hübscher SG. MacSween’s pathology of the liver. 8th ed. Edinburgh: Elsevier, 2022. |
| 10. | Brunt EM, Neuschwander-Tetri BA, Burt AD. Fatty liver disease: Alcoholic and nonalcoholic. In: Burt AD, Portmann BC, Ferrell LD, editors. In: MacSween's Pathology of the Liver: Expert Consult: Online and Print. Edinburgh: Elsevier, 2011: 293-359. |
| 11. | Ludwig J, Viggiano TR, McGill DB, Oh BJ. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin Proc. 1980;55:434-438. [PubMed] |
| 12. | Bayoumi A, Grønbæk H, George J, Eslam M. The Epigenetic Drug Discovery Landscape for Metabolic-associated Fatty Liver Disease. Trends Genet. 2020;36:429-441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 73] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 13. | Ciardullo S, Perseghin G. From NAFLD to MAFLD and MASLD: a tale of alcohol, stigma and metabolic dysfunction. Metab Target Organ Damage. 2024;4:30. [RCA] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 14. | David AR, Zimmerman MR. Cancer: an old disease, a new disease or something in between? Nat Rev Cancer. 2010;10:728-733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 109] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 15. | Bouvard V, Loomis D, Guyton KZ, Grosse Y, Ghissassi FE, Benbrahim-Tallaa L, Guha N, Mattock H, Straif K; International Agency for Research on Cancer Monograph Working Group. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 2015;16:1599-1600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1539] [Cited by in RCA: 1153] [Article Influence: 104.8] [Reference Citation Analysis (4)] |
| 16. | Zauber AG, Winawer SJ, O'Brien MJ, Lansdorp-Vogelaar I, van Ballegooijen M, Hankey BF, Shi W, Bond JH, Schapiro M, Panish JF, Stewart ET, Waye JD. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012;366:687-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2583] [Cited by in RCA: 2403] [Article Influence: 171.6] [Reference Citation Analysis (6)] |
| 17. | Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, Aulakh LK, Lu S, Kemberling H, Wilt C, Luber BS, Wong F, Azad NS, Rucki AA, Laheru D, Donehower R, Zaheer A, Fisher GA, Crocenzi TS, Lee JJ, Greten TF, Duffy AG, Ciombor KK, Eyring AD, Lam BH, Joe A, Kang SP, Holdhoff M, Danilova L, Cope L, Meyer C, Zhou S, Goldberg RM, Armstrong DK, Bever KM, Fader AN, Taube J, Housseau F, Spetzler D, Xiao N, Pardoll DM, Papadopoulos N, Kinzler KW, Eshleman JR, Vogelstein B, Anders RA, Diaz LA Jr. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357:409-413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5580] [Cited by in RCA: 5228] [Article Influence: 580.9] [Reference Citation Analysis (10)] |
| 18. | Zhao JF, Zhou BG, Lv Y, Teng QP, Wang XM, Li XY, Ding Y. Association between metabolic dysfunction-associated steatotic liver disease and risk of colorectal cancer or colorectal adenoma: an updated meta-analysis of cohort studies. Front Oncol. 2024;14:1368965. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 19. | Tandon P, Montano-Loza AJ, Lai JC, Dasarathy S, Merli M. Sarcopenia and frailty in decompensated cirrhosis. J Hepatol. 2021;75 Suppl 1:S147-S162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 333] [Cited by in RCA: 304] [Article Influence: 60.8] [Reference Citation Analysis (1)] |
| 20. | Lin YP, Wang PM, Chuang CH, Yong CC, Liu YW, Huang PY, Yao CC, Tsai MC. Metabolic Risks Are Increasing in Non-B Non-C Early-Stage Hepatocellular Carcinoma: A 10-Year Follow-Up Study. Front Oncol. 2022;12:816472. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 21. | Sutti S, Albano E. Adaptive immunity: an emerging player in the progression of NAFLD. Nat Rev Gastroenterol Hepatol. 2020;17:81-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 322] [Cited by in RCA: 310] [Article Influence: 51.7] [Reference Citation Analysis (2)] |
| 22. | Bath NM, Palettas M, Stevens L, Sarna A, Ejaz A, Kim A, Pawlik TM, Cloyd JM. Patient Perceptions of Care Coordination during Neoadjuvant Therapy for Gastrointestinal Cancers: A Mixed Methods Analysis. J Gastrointest Cancer. 2024;55:862-868. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 23. | Dai S, Liu C, Chen L, Jiang K, Kong X, Li X, Chen H, Ding K. Hepatic steatosis predicts metachronous liver metastasis in colorectal cancer patients: a nested case-control study and systematic review. Am J Cancer Res. 2024;14:1292-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 24. | Vauthey JN, Pawlik TM, Ribero D, Wu TT, Zorzi D, Hoff PM, Xiong HQ, Eng C, Lauwers GY, Mino-Kenudson M, Risio M, Muratore A, Capussotti L, Curley SA, Abdalla EK. Chemotherapy regimen predicts steatohepatitis and an increase in 90-day mortality after surgery for hepatic colorectal metastases. J Clin Oncol. 2006;24:2065-2072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1080] [Cited by in RCA: 922] [Article Influence: 46.1] [Reference Citation Analysis (2)] |
| 25. | Yura M, Takeuchi H, Fukuda K, Nakamura R, Suda K, Wada N, Kawakubo H, Kitagawa Y. High-risk group of upper and middle mediastinal lymph node metastasis in patients with esophagogastric junction carcinoma. Ann Gastroenterol Surg. 2018;2:419-427. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 26. | Kumar A, Gautam V, Sandhu A, Rawat K, Sharma A, Saha L. Current and emerging therapeutic approaches for colorectal cancer: A comprehensive review. World J Gastrointest Surg. 2023;15:495-519. [PubMed] [DOI] [Full Text] |
| 27. | Eslam M, Ahmed A, Després JP, Jha V, Halford JCG, Wei Chieh JT, Harris DCH, Nangaku M, Colagiuri S, Targher G, Joshi S, Byrne CD, Khunti K, Nguyen MH, Gish RG, George J. Incorporating fatty liver disease in multidisciplinary care and novel clinical trial designs for patients with metabolic diseases. Lancet Gastroenterol Hepatol. 2021;6:743-753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 75] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 28. | Zhan ZQ, Cheng ZJ. Impact of preoperative virtual reality education on surgical patients: additional considerations. Int J Surg. 2024;110:3988-3989. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 29. | Tilg H, Diehl AM. NAFLD and extrahepatic cancers: have a look at the colon. Gut. 2011;60:745-746. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 30. | Kasprzak A. Insulin-Like Growth Factor 1 (IGF-1) Signaling in Glucose Metabolism in Colorectal Cancer. Int J Mol Sci. 2021;22:6434. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 169] [Article Influence: 33.8] [Reference Citation Analysis (1)] |
| 31. | Younossi ZM, Corey KE, Lim JK. AGA Clinical Practice Update on Lifestyle Modification Using Diet and Exercise to Achieve Weight Loss in the Management of Nonalcoholic Fatty Liver Disease: Expert Review. Gastroenterology. 2021;160:912-918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 431] [Cited by in RCA: 381] [Article Influence: 76.2] [Reference Citation Analysis (1)] |
| 32. | Aron-Wisnewsky J, Vigliotti C, Witjes J, Le P, Holleboom AG, Verheij J, Nieuwdorp M, Clément K. Gut microbiota and human NAFLD: disentangling microbial signatures from metabolic disorders. Nat Rev Gastroenterol Hepatol. 2020;17:279-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 937] [Cited by in RCA: 815] [Article Influence: 135.8] [Reference Citation Analysis (5)] |
| 33. | Schott C, Lebedeva V, Taylor C, Abumelha S, Roshanov PS, Connaughton DM. Utility of Genetic Testing in Adults with CKD: A Systematic Review and Meta-Analysis. Clin J Am Soc Nephrol. 2025;20:101-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 34. | Chen F, Esmaili S, Rogers GB, Bugianesi E, Petta S, Marchesini G, Bayoumi A, Metwally M, Azardaryany MK, Coulter S, Choo JM, Younes R, Rosso C, Liddle C, Adams LA, Craxì A, George J, Eslam M. Lean NAFLD: A Distinct Entity Shaped by Differential Metabolic Adaptation. Hepatology. 2020;71:1213-1227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 270] [Article Influence: 45.0] [Reference Citation Analysis (9)] |
| 35. | Chen L, Zhang L, Hua H, Liu L, Mao Y, Wang R. Interactions between toll-like receptors signaling pathway and gut microbiota in host homeostasis. Immun Inflamm Dis. 2024;12:e1356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 44] [Article Influence: 22.0] [Reference Citation Analysis (1)] |
| 36. | Kubitschke M, Müller M, Wallhorn L, Pulin M, Mittag M, Pollok S, Ziebarth T, Bremshey S, Gerdey J, Claussen KC, Renken K, Groß J, Gneiße P, Meyer N, Wiegert JS, Reiner A, Fuhrmann M, Masseck OA. Next generation genetically encoded fluorescent sensors for serotonin. Nat Commun. 2022;13:7525. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 37. | Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ. Mechanisms of NAFLD development and therapeutic strategies. Nat Med. 2018;24:908-922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3652] [Cited by in RCA: 3330] [Article Influence: 416.3] [Reference Citation Analysis (6)] |
| 38. | Crișan G, Moldovean-Cioroianu NS, Timaru DG, Andrieș G, Căinap C, Chiș V. Radiopharmaceuticals for PET and SPECT Imaging: A Literature Review over the Last Decade. Int J Mol Sci. 2022;23:5023. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 281] [Cited by in RCA: 188] [Article Influence: 47.0] [Reference Citation Analysis (1)] |
| 39. | Yoon JH, Kim YK, Kim JW, Chang W, Choi JI, Park BJ, Choi JY, Kim SS, Park HS, Lee ES, Yu JS, Park SJ, You MW, Lee CH, Lee JM. Comparison of Four Diagnostic Guidelines for Hepatocellular Carcinoma Using Gadoxetic Acid-enhanced Liver MRI. Radiology. 2024;311:e233114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 40. | Lee JY, Lee DC, Lee JW. Serum carcinoembryonic antigen is associated with non-alcoholic fatty liver disease in healthy Korean non-smokers. Clin Chem Lab Med. 2013;51:1499-1504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 41. | Azimi A, Jolfayi AG, Rezayifar V, Ateen F, Fard HH, Mohammed SKM, Hosseinizadeh H, Faizabadi S, Keshavarzian A, Mansournia MA, Vosough M, Rahmanian M. The association between metabolic-associated fatty liver diseases and risk of colorectal polyps, neoplasia, and cancer: A systematic review and meta-analysis of over 56 million individuals. Clin Res Hepatol Gastroenterol. 2025;49:102652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 42. | Bui TT, Park E, Kang HY, Oh JK. Combined effects of smoking and alcohol consumption on the risk of liver cancer according to metabolic syndrome: A nested case-control study in South Korea. Int J Cancer. 2024;155:654-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 43. | Courneya KS, Vardy JL, O'Callaghan CJ, Gill S, Friedenreich CM, Wong RKS, Dhillon HM, Coyle V, Chua NS, Jonker DJ, Beale PJ, Haider K, Tang PA, Bonaventura T, Wong R, Lim HJ, Burge ME, Hubay S, Sanatani M, Campbell KL, Arthuso FZ, Turner J, Meyer RM, Brundage M, O'Brien P, Tu D, Booth CM; CHALLENGE Investigators. Structured Exercise after Adjuvant Chemotherapy for Colon Cancer. N Engl J Med. 2025;393:13-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 177] [Article Influence: 177.0] [Reference Citation Analysis (1)] |
| 44. | Kawaguchi T, Charlton M, Kawaguchi A, Yamamura S, Nakano D, Tsutsumi T, Zafer M, Torimura T. Effects of Mediterranean Diet in Patients with Nonalcoholic Fatty Liver Disease: A Systematic Review, Meta-Analysis, and Meta-Regression Analysis of Randomized Controlled Trials. Semin Liver Dis. 2021;41:225-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 80] [Article Influence: 16.0] [Reference Citation Analysis (2)] |
| 45. | Horesh N, Emile SH, Garoufalia Z, Gefen R, Zhou P, Wexner SD. Trends in management and outcomes of colon cancer in the United States over 15 years: Analysis of the National Cancer Database. Int J Cancer. 2024;155:139-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 46. | Liu Y, Liu Z, Hu Y, Ling Y, Qing S, Liu Y, Zhan Y, Shen Z, Fang Y, Deng H. Caffeine enhances antitumor T-cell activity by suppressing kynurenine pathway in colorectal cancer. Nat Commun. 2025;16:5906. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 47. | Kunutsor SK, Lehoczki A, Laukkanen JA. Coffee consumption, cancer, and healthy aging: epidemiological evidence and underlying mechanisms. Geroscience. 2025;47:1517-1555. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 48. | Armstrong MJ, Gaunt P, Aithal GP, Barton D, Hull D, Parker R, Hazlehurst JM, Guo K; LEAN trial team, Abouda G, Aldersley MA, Stocken D, Gough SC, Tomlinson JW, Brown RM, Hübscher SG, Newsome PN. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet. 2016;387:679-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1837] [Cited by in RCA: 1653] [Article Influence: 165.3] [Reference Citation Analysis (6)] |
| 49. | Janeeh AS, Bajbouj K, Rah B, Abu-Gharbieh E, Hamad M. Interplay between tumor cells and immune cells of the colorectal cancer tumor microenvironment: Wnt/β-catenin pathway. Front Immunol. 2025;16:1587950. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 50. | Rykova EY, Klimontov VV, Shmakova E, Korbut AI, Merkulova TI, Kzhyshkowska J. Anti-Inflammatory Effects of SGLT2 Inhibitors: Focus on Macrophages. Int J Mol Sci. 2025;26:1670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 46.0] [Reference Citation Analysis (1)] |
| 51. | Simon TG, Wilechansky RM, Stoyanova S, Grossman A, Dichtel LE, Lauer GM, Miller KK, Hoshida Y, Corey KE, Loomba R, Chung RT, Chan AT. Aspirin for Metabolic Dysfunction-Associated Steatotic Liver Disease Without Cirrhosis: A Randomized Clinical Trial. JAMA. 2024;331:920-929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 64] [Article Influence: 32.0] [Reference Citation Analysis (1)] |
| 52. | Zhang Q, Zhou Y, Feng X, Gao Y, Huang C, Yao X. Low-dose orlistat promotes the therapeutic effect of oxaliplatin in colorectal cancer. Biomed Pharmacother. 2022;153:113426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 53. | Yang Y, Chen Y, Liu Z, Chang Z, Sun Z, Zhao L. Concomitant NAFLD Facilitates Liver Metastases and PD-1-Refractory by Recruiting MDSCs via CXCL5/CXCR2 in Colorectal Cancer. Cell Mol Gastroenterol Hepatol. 2024;18:101351. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 54. | Bulut SD, Döndaş HA, Celebioglu HU, Sansano JM, Döndaş NY. Recent Insights About Probiotics Related Pharmabiotics in Pharmacology: Prevention and Management of Diseases. Probiotics Antimicrob Proteins. 2025;17:2262-2292. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |