Published online Oct 16, 2021. doi: 10.12998/wjcc.v9.i29.8749
Peer-review started: January 29, 2021
First decision: May 2, 2021
Revised: May 20, 2021
Accepted: September 8, 2021
Article in press: September 8, 2021
Published online: October 16, 2021
Processing time: 259 Days and 5.1 Hours
There is little evidence about the association of pre-existing hepatitis C infection (HCV) with outcomes in patients with coronavirus disease 2019 (COVID-19).
To assess the prevalence of history of HCV among patients with COVID-19 and to study the relationship of in-hospital mortality in relation with other predictors of poor outcomes in the presence or absence of COVID-19 induced acute liver injury.
In a retrospective single-center study design, 1193 patients with COVID-19 infection were studied. Patients were then classified into those with and without a history of HCV, 50 (4.1%) and 1157 (95.9%) respectively.
Multivariate cox-regression models showed that age, HCV, D-Dimer, and ferritin were the only predictors of in-hospital mortality. Acute liver injury and fibrosis score (Fib-4 score) were not different between both groups. Multivariate cox-regression model for liver profile revealed that aspartate aminotransferase/ alanine aminotransferase ratio, Fib-4 score, and HCV were predictors of in-hospital mortality. After propensity score matching HCV was the only predictor of mortality in the multivariate cox-regression model. A model including HCV was found to add predictive value to clinical and laboratory parameters.
In patients with COVID-19, history of HCV infection leads to an accentuated severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virulence, irres
Core Tip: In patients with coronavirus disease 2019 (COVID-19), history of hepatitis C infection accentuated severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) viral virulence and is a strong predictor of in-hospital mortality irrespective of baseline comorbidities, laboratory parameters, or COVID-19-induced liver injury. History of hepatitis C infection (HCV) in these patients seems to add a cumulative mortality risk to any clinical or laboratory profile. The mechanisms involved may be related to extra hepatic effects of HCV leading to enhanced ACE-2/TMPRSS mechanisms of SARS-CoV-2 virus and subsequent endothelial dysfunction. The realization is important for better characterization of the disease and triage this sub-group as high risk for therapeutic or prophylactic measures like vaccines.
- Citation: Ronderos D, Omar AMS, Abbas H, Makker J, Baiomi A, Sun H, Mantri N, Choi Y, Fortuzi K, Shin D, Patel H, Chilimuri S. Chronic hepatitis-C infection in COVID-19 patients is associated with in-hospital mortality. World J Clin Cases 2021; 9(29): 8749-8762
- URL: https://www.wjgnet.com/2307-8960/full/v9/i29/8749.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i29.8749
Coronavirus disease 2019 (COVID-19) is caused by the recently identified novel severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). Since December 2019, COVID-19 has caused a global outbreak and continuously spreading in more than 200 countries. World Health Organization estimates 3.4% mortality for the disease. Epidemiological reports show that mortality is higher in the elderly and in patients with underlying comorbidities like diabetes, hypertension, and heart disease. Identifying the predictors of mortality in patients with COVID-19 gains significant importance given the efforts to develop a new vaccine, as this could be an initial step towards the selection of the population at risk which can benefit the most from the developing vaccines at an earlier stage of the disease.
Besides the initial pulmonary manifestations of COVID-19, multi-organ involvement such as cardiovascular, neurologic, renal, and gastrointestinal manifestations have been reported. Hepatic manifestations, in particular, occur in about 50% of patients with COVID-19 which can range from asymptomatic transaminitis to, less frequent acute liver failure[1]. The etiology behind the hepatic manifestations of COVID-19 remains unclear and postulated mechanisms include ischemic liver injury, immune-mediated liver injury, drug-induced liver injury, and a direct viral cytopathy[1,2].The correlation of the worse outcomes of COVID-19 and pre-existing viral hepatitis is unclear, as there is little evidence to support this association.
The effect of preexisting chronic hepatitis C virus (HCV) infection, which is known to cause several extrahepatic manifestations independent of liver injury[3], in COVID-19 patients has not been studied. In one meta-analysis of the COVID-19 patients have reported an estimate of 3% prevalence of pre-existing chronic liver disease[4]. To the best of our knowledge, this data does not provide specific information regarding the prevalence and impact of viral hepatitis, specifically HCV in COVID-19 patients. Initial reports have suggested that COVID-19 patients with HCV are at increased risk for poor outcomes especially if accompanied by other risk factors such as advanced age and hypertension. The aim of our work was to assess the prevalence of the history of HCV among patients with COVID-19 and to study the relationship between in-hospital mortality with other predictors of poor outcomes in the presence or absence of baseline liver dysfunction or COVID-19 induced acute liver injury (ALI).
Our study is a retrospective review and included all hospitalized patients at our institution with COVID-19 between March 15, 2020 and May 30, 2020. All patients had nasal swabs positive for SARS-CoV-2. Patients below the age of 18 years were excluded from study.
We reviewed medical charts and the laboratory parameter (HCV antibody) to obtain the HCV infection status. The patient who did not have HCV antibody testing was not included in the analysis. HCV viral load levels were abstracted from the electronic medical records. Patients were then divided into two groups based on the presence or absence of a history of HCV and their demographic, clinical, laboratory variables, and outcomes were compared. The primary endpoint of our study was in-hospital mortality.
Continuous variables were expressed as mean ± SD, and nominal and categorical variables were expressed as numbers (%). The independent sample t-test and one-way ANOVA were used to compare the mean values of different groups, and chi-square test was used for comparison of nominal and categorical variables. Predictors of mortality were checked using univariate and multivariate Cox regression models. Propensity score matching was used between patients with and without HCV to create subgroups matched for age, sex, body mass index (BMI), and all co-morbidities and all models were repeated for subgroups. Kaplan-Myer survival curves were used to test the difference in cumulative in-hospital outcomes. A 2-log likelihood ratio was initiated to compare 4 different models for the prediction of in-hospital mortality. The first model (clinical model) consisted of clinical parameters reported in the literature to be associated with mortality in COVID-19 patients, namely, age, sex, hypertension (HTN), diabetes mellitus (DM), history of respiratory disease, and history of cardiac disease. The second model (laboratory model) consisted of laboratory values reported in the literature to be associated with worse in-hospital mortality, namely D-dimer, lactate dehydrogenase (LDH), high sensitivity C-reactive protein (hsCRP), and serum creatinine. The third model was a combined clinical and laboratory model, and the fourth model was the combined model with the addition of the history of HCV. For all statistical tests, a P value < 0.05 was considered statistically significant. All analyses were performed with commercially available software (SPSS version 23.0; SPSS, Inc).
During the study period, 1207 patients with confirmed COVID-19 were identified. Seven patients were excluded as their age was ≤ 18 years and another 7 patients were excluded as their HCV status was not clear and accordingly 1193 patients were included in the analysis. The baseline demographic, clinical, laboratory and imaging criteria for all patients are summarized in Supplementary Table 1. Of the study group, 50 (5%) had HCV infection, and 1157 (95%) did not have HCV infection. Baseline demographic clinical and laboratory data compared for patients with and without HCV are summarized in Table 1. Briefly, HCV patients had lower BMI, higher incidence of HIV, asthma, chronic obstructive pulmonary disease (COPD), congestive heart failure (CHF), and chronic kidney disease (CKD) at baseline. In addition, patients with HCV presented to the hospital earlier than patients without HCV, and, on hospital admission, had higher lymphocytic count and lower CRP, however, had worse serum creatinine, aspartate aminotransferase (AST), conjugated bilirubin, higher pro-calcitonin, lower hemoglobin, and lower platelet count. All other demographic, clinical, and laboratory values were not different between both groups.
| All patients (n = 1193) | Propensity score matched1 (n = 98) | |||||
| No HCV (n = 1143) | HCV (n = 50) | P value | No HCV (n = 48) | HCV (n = 50) | P value | |
| Age (yr) | 62 ± 16 | 64 ± 9 | 0.402 | 64 ± 15 | 64 ± 9 | 0.803 |
| Gender (female) | 456 (39.90) | 14 (28.00) | 0.092 | 20 (41.67) | 14 (28.00) | 0.155 |
| Body mass index (kg/m2) | 30 ± 7 | 27.7 ± 8.5 | 0.026 | 27.9 ± 6.8 | 28 ± 9 | 0.917 |
| Hypertension | 703 (61.50) | 31 (62.00) | 0.944 | 33 (68.75) | 31 (62.00) | 0.483 |
| Diabetes mellitus) | 530 (46.37) | 17 (34.00) | 0.086 | 17 (35.42) | 17 (34.00) | 0.883 |
| Human immune-deficiency virus | 58 (05.07) | 21 (42.00) | < 0.001 | 17 (35.42) | 21 (42.00) | 0.504 |
| Asthma | 144 (12.60) | 10 (20.00) | < 0.001 | 9 (18.75) | 10 (20.00) | 0.612 |
| Chronic obstructive pulmonary disease | 105 (09.19) | 14 (28.00) | < 0.001 | 9 (18.75) | 14 (28.00) | 0.280 |
| Hepatitis B infection | 1 (00.09) | 3 (06.00) | < 0.001 | 0 (00.00) | 3 (06.00) | 0.085 |
| Congestive heart failure | 105 (09.19) | 12 (24.00) | 0.001 | 10 (20.83) | 12 (24.00) | 0.707 |
| Coronary artery disease | 119 (10.41) | 6 (12.00) | 0.720 | 6 (12.50) | 6 (12.00) | 0.940 |
| Chronic kidney disease | 89 (07.79) | 9 (18.00) | 0.01 | 5 (10.42) | 9 (18.00) | 0.284 |
| End stage renal disease | 77 (06.74) | 5 (10.00) | 0.372 | 2 (04.17) | 5 (10.00) | 0.262 |
| Fever | 753 (65.88) | 31 (62.00) | 0.525 | 26 (54.17) | 31 (62.00) | 0.585 |
| Cough | 771 (67.45) | 24 (48.00) | 0.003 | 27 (56.25) | 24 (48.00) | 0.294 |
| Shortness of breath | 787 (68.85) | 40 (80.00) | 0.110 | 34 (70.83) | 40 (80.00) | 0.478 |
| Myalgia | 352 (30.80) | 11 (22.00) | 0.175 | 19 (39.58) | 11 (22.00) | 0.04 |
| Chest discomfort | 144 (12.60) | 5 (10.00) | 0.572 | 3 (06.25) | 5 (10.00) | 0.538 |
| Atypical symptoms | 392 (34.30) | 19 (38.00) | 0.611 | 20 (41.67) | 19 (38.00) | 0.585 |
| Symptom onset to hospitalization (d) | 5.85 ± 5.56 | 3.79 ± 4.04 | 0.034 | 6.2 ± 5.7 | 3.8 ± 4.04 | 0.053 |
| Length of hospital stay (d) | 8.05 ± 7.05 | 6.2 ± 4.3 | 0.072 | 7 ± 5.79802 | 6.2 ± 4.3 | 0.454 |
| Hemoglobin | 13.0 ± 2.2 | 12.2 ± 2.7 | 0.021 | 12.5 ± 2.1 | 12.2 ± 2.7 | 0.598 |
| Total white count | 8.6 ± 6.6 | 9.8 ± 13.1 | 0.244 | 7.78 ± 3.5 | 9.8 ± 13.1 | 0.300 |
| Platelets | 221.7 ± 103.3 | 183 ± 73.5 | 0.015 | 234.4 ± 100 | 183 ± 73 | 0.007 |
| Neutrophilic count | 6.8 ± 4.05 | 6.1 ± 3.8 | 0.224 | 6.3 ± 3.4 | 6.1 ± 3.8 | 0.780 |
| Lymphocyte count | 1.18 ± 4.86 | 2.86 ± 12.3 | 0.037 | 0.99 ± 0.52 | 2.86 ± 12.3 | 0.302 |
| D-dimer | 3115 ± 9997 | 2270 ± 5383 | 0.582 | 6883 ± 28017 | 2270 ± 5383 | 0.293 |
| Lactate dehydrogenase | 581 ± 393 | 623 ± 391 | 0.477 | 523 ± 419 | 623 ± 391 | 0.248 |
| High sensitivity C-reactive protein | 143 ± 113 | 98 ± 76 | 0.013 | 129 ± 89 | 98 ± 75 | 0.095 |
| Ferritin | 1148 ± 1574 | 1265 ± 2098 | 0.647 | 1053 ± 1267 | 1265 ± 2098 | 0.576 |
| Lactate | 2.3 ± 1.9 | 2.7 ± 2.6 | 0.265 | 2.07 ± 1.3 | 2.7 ± 2.6 | 0.189 |
| Prothrombin time (s) | 14 ± 10.7 | 13.4 ± 2.5 | 0.716 | 13.6 ± 2.3 | 13.4 ± 2.4 | 0.700 |
| Activated partial thromboplastin time (s) | 32.2 ± 7.9 | 32.7 ± 6.3 | 0.67 | 31.4 ± 5.4 | 32.7 ± 6.3 | 0.317 |
| Creatinine | 1.9 ± 2.6 | 3.3 ± 3.5 | < 0.001 | 2.01 ± 2.4 | 3.3 ± 3.5 | 0.039 |
| Alanine transaminase | 43.7 ± 66.4 | 53.2 ± 121.7 | 0.386 | 40.8 ± 37.5 | 53.2 ± 121.7 | 0.538 |
| Aspartate transaminase | 68.6 ± 90.8 | 128.1 ± 367.2 | 0.001 | 59.8 ± 43.5 | 128.2 ± 367 | 0.246 |
| Alkalaine phosphatase | 94.2 ± 86.4 | 111.5 ± 143 | 0.224 | 82.3 ± 40.7 | 111.5 ± 143 | 0.217 |
| Total bilirubin | 0.6 ± 0.7 | 0.8 ± 0.86 | 0.111 | 0.54 ± 0.41 | 0.8 ± 0.9 | 0.087 |
| Combined bilirubin | 0.27 ± 0.45 | 0.45 ± 0.68 | 0.02 | 0.26 ± 0.28 | 0.45 ± 0.68 | 0.115 |
| Total protein | 6.98 ± 0.83 | 7.2 ± 0.98 | 0.163 | 6.91 ± 0.72 | 7.2 ± 0.98 | 0.175 |
| Albumin | 3.59 ± 0.55 | 3.44 ± 0.56 | 0.089 | 3.6 ± 0.5 | 3.4 ± 0.6 | 0.209 |
| Procalcitonin | 2.34 ± 9.4 | 7.27 ± 21.4 | 0.02 | 3.08 ± 8.8 | 7.3 ± 21.4 | 0.453 |
| Hydroxychloroquine | 878 (76.82) | 36 (72.00) | 0.711 | 33 (68.75) | 36 (72.00) | 0.725 |
| Azithromycin | 839 (73.40) | 32 (64.00) | 0.303 | 35 (72.92) | 32 (64.00) | 0.343 |
| Tocilizumab | 78 (06.82) | 0 (00.00) | 0.149 | 4 (08.33) | 0 (00.00) | 0.054 |
| Mechanical ventilation | 449 (39.28) | 29 (58.00) | 0.003 | 4 (08.33) | 29 (58.00) | < 0.001 |
| Death | 386 (33.77) | 28 (56.00) | 0.004 | 7 (14.58) | 28 (56.00) | < 0.001 |
The use of hydroxychloroquine, azithromycin, and tocilizumab was not different between both groups, however, it’s to be noted that none of the HCV patients received tocilizumab. Importantly, both mortality and in-hospital mechanical ventilation were significantly higher in patients with HCV.
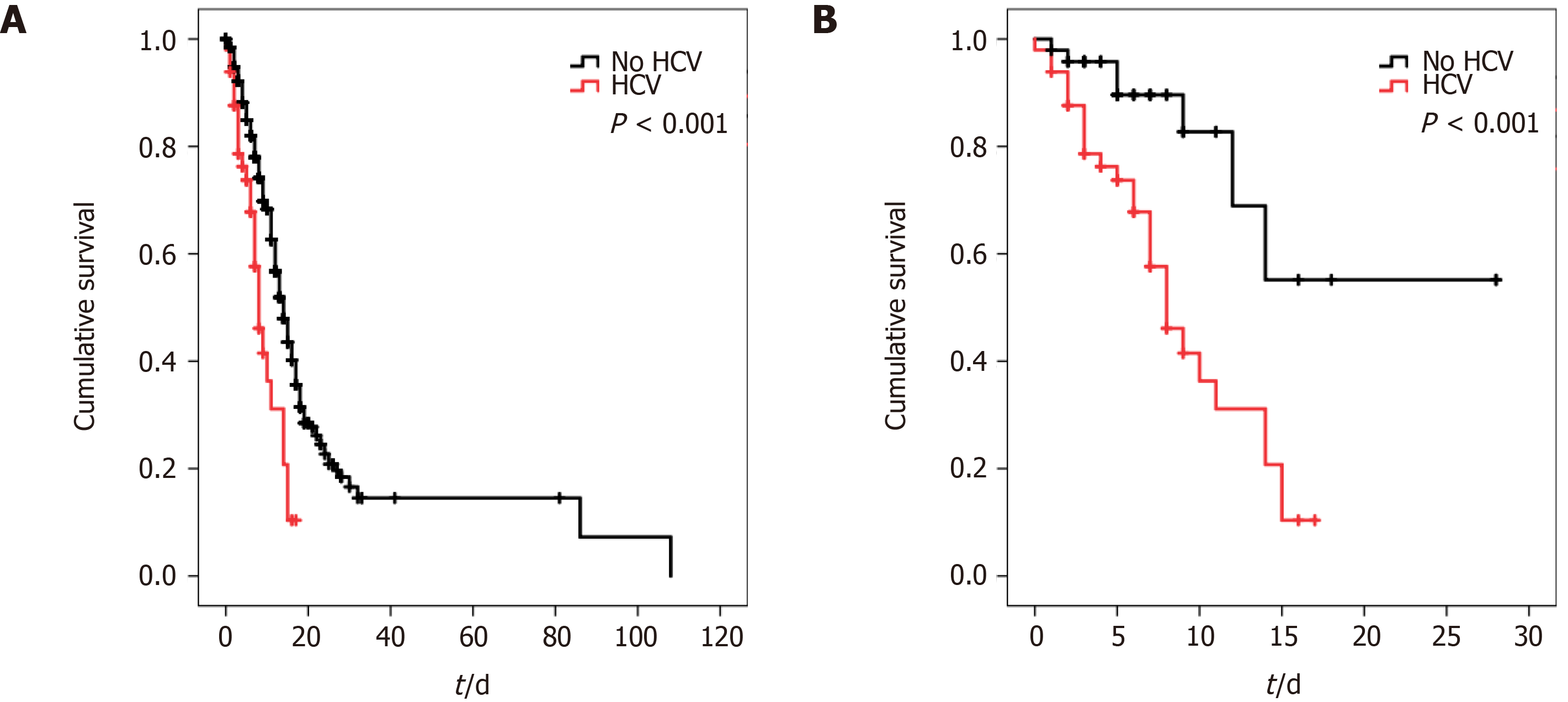
The overall predictors of in-hospital mortality are summarized in Table 2. Univariate Cox-regression model showed that the independent predictors of in-hospital mortality were age, sex, HTN, DM, HIV, COPD, HCV, CHF, CKD, neutrophilic count, D-dimer, LDH, hsCRP, ferritin, total and conjugated bilirubin, albumin, pro-calcitonin levels, and platelet count. Multivariate cox-regression models however showed that age, HCV, D-Dimer, and ferritin were the only predictor of in-hospital mortality, while all other variables lost statistical significance. Kaplan-Meier curves were initiated for patients with and without HCV and revealed that patients with HCV have worse cumulative in-hospital mortality compared to patients without HCV (P < 0.001, Figure 1).
| All patients (n = 1193) | Propensity score matched (n = 98) | |||||||
| Univariate | Multivariate | Univariate | Multivariate | |||||
| Exp (B) | Sig. | Exp (B) | Sig. | Exp (B) | Sig. | Exp (B) | Sig. | |
| Age (yr) | 1.028 | < 0.001 | 1.029 | < 0.001 | 1.022 | 0.183 | - | - |
| Gender (female) | 0.675 | < 0.001 | 0.865 | 0.42 | 0.488 | 0.05 | 0.714 | 0.110 |
| Body mass index (kg/m2) | 0.991 | 0.184 | - | - | 1.014 | 0.514 | - | - |
| Hypertension | 1.611 | < 0.001 | 0.843 | 0.386 | 2.87 | 0.004 | 2 | 0.051 |
| Diabetes mellitus | 1.394 | 0.001 | 1.208 | 0.277 | 1.06 | 0.864 | - | - |
| Human immune-deficiency virus | 1.797 | < 0.001 | 1.251 | 0.492 | 1.92 | 0.064 | - | - |
| Asthma | 0.864 | 0.341 | - | - | 1.39 | 0.357 | - | - |
| Chronic obstructive pulmonary disease | 1.435 | 0.013 | 1.043 | 0.879 | 1.222 | 0.591 | - | - |
| Hepatitis B infection | 1.984 | 0.237 | - | - | 1.04 | 0.962 | - | - |
| HCV | 2.224 | < 0.001 | 2.435 | 0.041 | 3.88 | 0.001 | 3.46 | 0.004 |
| Congestive heart failure | 1.4 | 0.02 | 1.249 | 0.356 | 1.253 | 0.545 | - | - |
| Coronary artery disease | 1.387 | 0.023 | 1.092 | 0.734 | 1.05 | 0.923 | - | - |
| End stage renal disease | 1.208 | 0.305 | - | - | 1.013 | 0.983 | - | - |
| Chronic kidney disease | 1.437 | 0.016 | 1.188 | 0.535 | 1.024 | 0.957 | - | - |
| Hemoglobin | 0.966 | 0.123 | - | - | 0.925 | 0.337 | - | - |
| Total white count | 1.006 | 0.376 | - | - | 1.004 | 0.736 | - | - |
| Platelets | 0.998 | < 0.001 | 0.999 | 0.438 | 0.999 | 0.525 | - | - |
| Neutrophilic count | 1.023 | 0.028 | 0.991 | 0.658 | 1.072 | 0.145 | - | - |
| Lymphocyte count | 0.993 | 0.641 | 1 | 0.163 | 1.000 | 0.976 | - | - |
| D-dimer | 1 | < 0.001 | 1.001 | < 0.001 | 1.000 | 0.09 | - | - |
| Lactate dehydrogenase | 1.001 | < 0.001 | 1.001 | 0.374 | 1.000 | 0.714 | - | - |
| High sensitivity C-reactive protein | 1.002 | < 0.001 | 1 | 0.553 | 0.999 | 0.788 | - | - |
| Ferritin | 1 | 0.002 | 1.053 | 0.028 | 1.000 | 0.902 | - | - |
| Prothrombin time (s) | 1.001 | 0.89 | - | - | 1.1 | 0.281 | - | - |
| Activated partial thromboplastin time (s) | 0.998 | 0.804 | - | - | 0.966 | 0.281 | - | - |
| Creatinine | 1.072 | < 0.001 | - | - | 1.08 | 0.91 | - | - |
| Alanine transaminase | 0.999 | 0.325 | - | - | 0.998 | 0.371 | - | - |
| Aspartate transaminase | 1 | 0.391 | - | - | 0.999 | 0.402 | - | - |
| Alkalaine phosphatase | 0.999 | 0.222 | - | - | 0.275 | 0.995 | - | - |
| Total bilirubin | 1.131 | 0.002 | 1.026 | 0.925 | 1.16 | 0.475 | - | - |
| Combined bilirubin | 1.327 | < 0.001 | 1.091 | 0.733 | 1.17 | 0.510 | - | - |
| Total protein | 0.932 | 0.235 | - | - | 0.892 | 0.560 | - | - |
| Albumin | 0.681 | < 0.001 | 0.933 | 0.678 | 0.613 | 0.114 | - | - |
| Procalcitonin | 1.014 | < 0.001 | 1.001 | 0.793 | 1.008 | 0.544 | - | - |
Liver function tests were available in only 1065 patients. Liver enzymes [AST and alanine aminotransferase (ALT)] were normal in 416 patients. Only ALT was elevated in 6 patients (all of whom did not have HCV), only AST was elevated in 423 patients, and both AST and ALT were elevated in 201 patients. Overall, AST was higher than ALT in 633 patients with a mean absolute AST/ALT of 1.88 ± 1.5 in all patients. It was noted that, with the exception of the absolute AST/ALT ratio which was higher in patients with HCV (P = 0.023), none of the previous parameters was different between the two groups. Moreover, history of fatty liver disease, history of cirrhosis, and the fibrosis score (Fib-4 score) was not different between both groups.
ALI, was defined in our study as an increase in both admission AST and ALT 2 times above the upper normal limit or elevation of either admission AST or ALT 2 times above the upper normal limit in addition to at least one of the following: Elevated admission combined bilirubin, elevated admission total bilirubin or elevated admission PT more than 2 times the normal limit. Based on our definition, ALI occurred in 107 (10%) of all patients and was not different between patients without or with HCV (100 vs 7, respectively, P = 0.127).
Univariate Cox-regression models revealed that ALI, AST/ALT ratio, the presence of any elevation of liver enzyme even if it was less than 2 times the normal value, and overall AST more than ALT levels, and Fib-4 score were all predictive of in-hospital mortality in addition to HCV. The multivariate Cox-regression model, however, revealed that only AST/ALT ratio, Fib-4 score, and HCV were predictors of in-hospital mortality.
Next, propensity score matching was initiated between patients with and without HCV for age, sex, BMI, HTN, DM, HIV, COPD, CHF, and CKD. As seen in Table 1, almost all differences between the groups were eliminated after matching with the exception of lower platelet count in patients with HCV. It is important to note that the worse in-hospital mortality and need for mechanical ventilation in patients with HCV persisted despite matching. Moreover, univariate and multivariate models for in-hospital mortality prediction were re-initiated and it was found that sex, HTN, and HCV were the only univariate predictors of mortality, while HCV was the only predictor of mortality in the multivariate model as sex and HTN lost statistical significance (Table 2). Kaplan Meier curves were also reinitiated for matched patients with and without HCV and revealed that patients with HCV continued to have worse cumulative in-hospital mortality compared to patients without HCV (P < 0.001, Figure 2)
Comparison for parameters of liver functions, chronic liver disease, and ALI was similar in the matched and unmatched groups as AST/ALT ratio and history of chronic liver disease remained significantly higher in the HCV group (Table 3). Importantly, univariate Cox-regression models revealed that only HCV and Fib-4 score were predictive of mortality, while the multivariate cox-regression model showed that HCV was the only predictor of mortality among all liver function parameters (Table 4). HCV: 65%, no HCV: 34%, P < 0.001 ALI: 53% vs 34% and 34% in patients without HCV and without ALI, respectively.
| All (n = 1065) | ALI patients (n = 1065) | Propensity score matched (n = 81) | |||||
| No HCV (n = 1014) | HCV (n = 41) | P value | No HCV (n = 40) | HCV (n = 41) | P value | ||
| AST/ALT | 1.88 ± 1.5 | 1.86 ± 1.5 | 2.4 ± 1.3 | 0.023 | 1.8 ± 0.88 | 2.4 ± 1.27 | 0.015 |
| Any liver enzyme elevated | 639 (60) | 610 (60) | 29 (71) | 0.174 | 25 (63) | 29 (71) | 0.423 |
| Normal liver enzymes | 416 (39) | 311 (31) | 10 (24) | 0.242 | 15 (38) | 10 (24) | 0.223 |
| Only ALT elevated | 6 (1) | 6 (1) | 0 (0) | 0 (0) | 0 (0) | ||
| Only AST elevated | 432 (41) | 409 (40) | 23 (56) | 15 (38) | 2356 | ||
| Both elevated | 201 (19) | 195 (19) | 6 (15) | 10 (25) | 6 (15) | ||
| AST > ALT | 423 (40) | 399 (39) | 20 (49) | 0.394 | 0.394 | ||
| All AST > ALT | 633 (59) | 604 (60) | 29 (71) | 0.153 | 25 (63) | 29 (71) | 0.423 |
| Fatty liver | 50 (5) | 48 (5) | 2 (5) | 0.931 | 1 (3) | 2 (5) | 0.582 |
| History of cirrhosis | 6 (1) | 5 (1) | 1 (2) | 0.228 | 2 (5) | 1 () | 0.534 |
| Fib-4 score | 3.43 ± 5.5 | 3.47 ± 5.6 | 3.1 ± 3.6 | 0.642 | 3.4 ± 2.25 | 3.1 ± 3.6 | 0.249 |
| ALI1 | 107 (10) | 100 (10) | 7 (17) | 0.128 | 3 (8) | 7 (17) | 0.166 |
| ALl patients (n = 1065) | Propensity score matched (n = 81) | |||||||
| Univariate | Multivariate | Univariate | Multivariate | |||||
| Exp (B) | Sig. | Exp (B) | Exp (B) | Sig. | Exp (B) | Sig. | Sig. | |
| AST/ALT | 1.1 | < 0.001 | 1.06 | < 0.001 | 1.204 | 0.221 | - | - |
| Any liver enzyme elevated | 1.427 | 0.002 | 0.001 | 0.823 | 1.204 | 0.624 | - | - |
| All AST > ALT | 1.455 | 0.001 | 0.001 | 0.852 | 1.204 | 0.624 | - | - |
| History of cirrhosis | 1.368 | 0.659 | - | - | 1.95 | 0.513 | - | - |
| ALI1 | 1.418 | 0.015 | 1.045 | 0.792 | 1.132 | 0.817 | - | - |
| Fib-4 score | 1.02 | < 0.001 | 1.022 | < 0.001 | 1.1 | 0.042 | 1.1 | 0.08 |
| HCV | 2.224 | < 0.001 | 2.15 | < 0.001 | 3.88 | 0.001 | 3.75 | 0.002 |
It is to be noted that, all HCV patients in our study received anti-viral treatment. Twenty-six of these patients had sustained viral response. Viral load was still detectable in the remaining 24 patients (chronic HCV, viral loads: 1688184 ± 1439771, median: 1380000, SE: 434107, minimum: 269, maximum: 3610259 IU/mL). Comparisons between this the subgroup of chronic HCV patients and matched controls continued to show no differences in baseline demographic, clinical, and laboratory variables, however, death and need for mechanical ventilation continued to be observed more frequently in chronic HCV patients (Table 5). A subgroup Cox-regression analysis for predictors of in-hospital mortality (matched controls and chronic HCV, n = 72) showed that hypertension and HCV were the only predictors of mortality in the univariate analysis. While both HCV and hypertension retained their predictive ability in the multivariate model, HCV was significantly stronger as an independent predictor of mortality (Table 5).
| Propensity score matched (n = 72) | |||||||
| Comparison between groups | Cox-regression analysis | ||||||
| Univariate | Multivariate | ||||||
| NO HCV (n = 48) | HCV with viral load (n = 24) | P value | Exp (B) | Sig. | Exp (B) | Sig. | |
| Age (yr) | 64.4 ± 15.1 | 64.4 ± 9.8 | 0.995 | 1.021 | 0.293 | - | - |
| Gender (female) | 28 (58) | 17 (71) | 0.302 | 0.481 | 0.088 | - | - |
| Body mass index (kg/m2) | 27.9 ± 6.8 | 26.0 ± 6.2 | 0.252 | 1.014 | 0.625 | - | - |
| Hypertension | 33 (69) | 16 (67) | 0.858 | 3.03 | 0.014 | 2.55 | 0.035 |
| Diabetes mellitus | 17 (35) | 7 (29) | 0.596 | 1.05 | 0.902 | - | - |
| Human immune-deficiency virus | 17 (35) | 9 (38) | 0.862 | 1.79 | 0.185 | - | - |
| Asthma | 9 (19) | 4 (17) | 0.828 | 1.81 | 0.251 | - | - |
| Chronic obstructive pulmonary disease | 9 (19) | 5 (21) | 0.833 | 1.386 | 0.483 | - | - |
| Hepatitis B infection | 1 (0.2) | 0 (0) | 0.476 | 1.04 | 0.976 | - | - |
| HCV | 0 (0) | 24 (100) | - | 3.47 | 0.007 | 3.2 | 0.014 |
| Congestive heart failure | 10 (21) | 5 (21) | 1.000 | 1.41 | 0.448 | - | - |
| Coronary artery disease | 6 (13) | 2 (8) | 0.596 | 1.12 | 0.845 | - | - |
| End stage renal disease | 2 (0.4) | 1 (4) | 1.000 | 1.65 | 0.628 | - | - |
| Chronic kidney disease | 5 (10) | 6 (25) | 0.105 | 1.1 | 0.875 | - | - |
| Hemoglobin | 12.5 ± 2.1 | 11.7 ± 2.6 | 0.166 | 0.921 | 0.424 | - | - |
| Total white count | 7.8 ± 3.5 | 12 ± 17.9 | 0.122 | 1.006 | 0.618 | - | - |
| Platelets | 234.4 ± 100.3 | 185 ± 86.2 | 0.051 | 1.000 | 0.978 | - | - |
| Neutrophilic count | 6.27 ± 3.4 | 6.24 ± 4.3 | 0.981 | 1.11 | 0.056 | - | - |
| Lymphocyte count | 0.99 ± 0.5 | 4.62 ± 17.0 | 0.145 | 1.002 | 0.897 | - | - |
| D-dimer | 6883 ± 28017 | 1154 ± 1807 | 0.333 | 1.000 | 0.054 | - | - |
| Lactate dehydrogenase | 523 ± 419 | 649 ± 484 | 0.273 | 1.000 | 0.898 | - | - |
| High sensitivity C-reactive protein | 129 ± 90 | 113 ± 89 | 0.518 | 0.999 | 0.751 | - | - |
| Ferritin | 1053 ± 1267 | 1373 ± 2775 | 0.533 | 1.000 | 0.661 | - | - |
| Prothrombin time (s) | 13.6 ± 2.3 | 13.5 ± 2.5 | 0.820 | 1.05 | 0.636 | - | - |
| Activated partial thromboplastin time (s) | 31.4 ± 5.4 | 32.9 ± 7.28 | 0.365 | 0.935 | 0.138 | - | - |
| Creatinine | 2.01 ± 2.4 | 3.2 ± 4. | 0.132 | 1.06 | 0.315 | - | - |
| Alanine transaminase | 40.8 ± 37.5 | 62.1 ± 169.8 | 0.448 | 0.998 | 0.427 | - | - |
| Aspartate transaminase | 59.8 ± 43.5 | 169.8 ± 518.6 | 0.185 | 0.999 | 0.433 | - | - |
| Alkaline phosphatase | 82.3 ± 40.7 | 114.5 ± 165.7 | 0.246 | 0.996 | 0.376 | - | - |
| Total bilirubin | 0.54 ± 0.4 | 0.93 ± 1.1 | 0.05 | 1.236 | 0.336 | - | - |
| Combined bilirubin | 0.26 ± 0.28 | 0.58 ± 0.9 | 0.05 | 1.223 | 0.428 | - | - |
| Total protein | 6.91 ± 0.72 | 7.0 ± 0.95 | 0.618 | 0.813 | 0.380 | - | - |
| Albumin | 3.6 ± 0.51 | 3.3 ± 0.52 | 0.073 | 0.509 | 0.079 | - | - |
| Procalcitonin | 3.1 ± 8.8 | 4.6 ± 11.7 | 0.695 | 0.984 | 0.538 | - | - |
| Hydroxychloroquine | 33 (69) | 18 (75) | 0.582 | - | - | - | - |
| Azithromycin | 35 (73) | 13 (54) | 0.112 | - | - | - | - |
| Tocilizumab | 4 (8) | 0 (0) | 0.146 | - | - | - | - |
| Mechanical ventilation | 4 (8) | 15 (63) | < 0.001 | - | - | - | - |
| Death | 7 (15) | 15 (63) | < 0.001 | - | - | - | - |
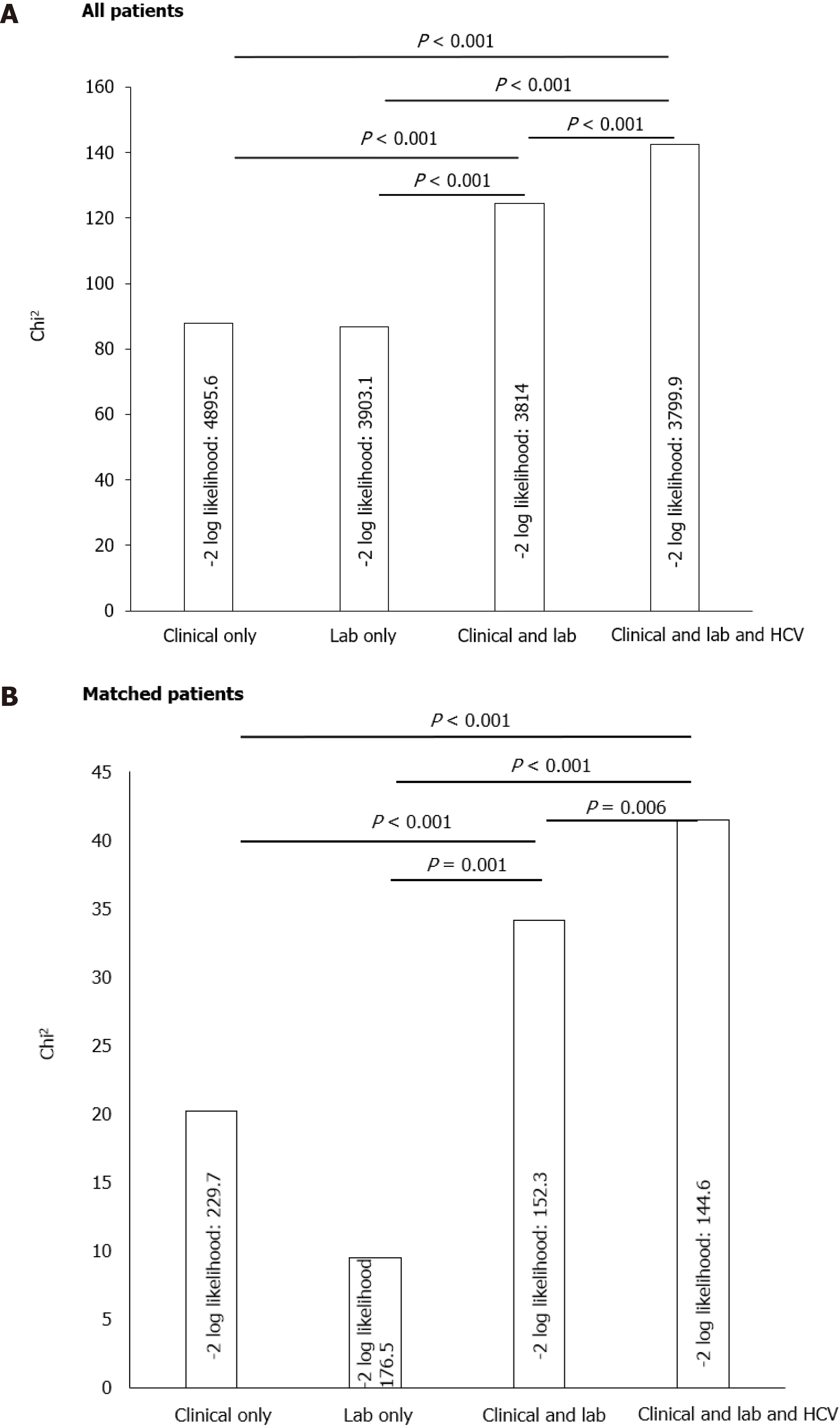
Model comparisons were done using the –2-log likelihood test which was initiated to assess the cumulative predictive abilities of 4 different models for in-hospital mortality. The first model was composed of the most reported clinical predictors of mortality (clinical model), the second model was composed of the most important reported laboratory variables associated with in-hospital mortality (laboratory model), the third model was a combination of clinical and laboratory model, and the fourth model represented the third model with the addition of history of HCV. It was found that, while the clinical and laboratory models seem to be additive for the prediction of mortality, the addition of HCV to any of these models including the combined model increased the predictive accuracy for in-hospital mortality (Figure 3).
The findings of our study can be summarized as follows: First, in a cohort of COVID-19 patients in a single-center, the frequency of history of chronic HCV infection is 4.1%, the frequency of COVID-19 induced ALI is 10%, both of which were associated with significant in-hospital mortality compared to patients without HCV or ALI. Second, in those patients, HCV seems to be an independent predictor of in-hospital mortality and seems to be stronger than the traditionally reported predictors of mortality in COVID-19 patients. The predictive ability of history of HCV infection for mortality in COVID-19 persists independent of baseline liver functions, fibrosis, and the acute COVID-19 induced ALI. Finally, the history of HCV infection seems to add cumulative predictive ability to other traditional clinical and laboratory risk factors for in-hospital mortality.
In this single-center study, traditional predictors of mortality in patients with COVID-19 were strongly associated with in-hospital mortality as reported in several studies which was in agreement with previous reports. Interestingly, however, HCV was overall strongly associated with in-hospital mortality. The incidence of history of HCV infection in our study was on the higher side of that reported in the literature (50 patients, 4.1%). In a large series of 5700 hospitalized patients with COVID-19, HCV infection was encountered in < 0.1% of patients[5]. There is a local higher prevalence of injection drug abuse in the community wherefrom the study cohort was included (Bronx, New York), which is reportedly significantly associated with increased incidence and prevalence of HCV and may explain the higher HCV prevalence in our cohort[6]. Initial reports have suggested that COVID-19 patients with HCV are at increased risk for poor outcomes especially if accompanied by other risk factors of poor outcomes such as advanced age and hypertension[5]. In our study, despite that HCV adds a cumulative increased risk of mortality to both clinical and laboratory predictors of mortality risk, HCV stood out as the only strong predictor of mortality after matching patients with and without HCV for all other baseline confounding clinical and laboratory predictors of mortality which may suggest that the pathological effect of the COVID-19 virus is accentuated in HCV patients compared to patients without HCV irrespective of their age, baseline clinical status, or admission laboratory parameters.
Moreover, in our study, the incidence of COVID-associated ALI was comparable to previous studies[7] (107 patients, 10%) and was associated with 53% mortality, which was significantly higher than patients without ALI (34%, P < 0.001). However, almost all parameters of ALI, chronic liver disease, as well as liver fibrosis, despite associated with mortality, were not different between patients with and without HCV, and, moreover, HCV outperformed ALI as a predictor of mortality, both overall and after matching, which brings the question whether ALI in patients with COVID-19 is a result of direct SARS-CoV-2 viral effect on the liver or just a result of cytokine storm, multi-organ failure or even medication-induced. The lack of differences of ALI between patients with and without HCV may also indicate that liver injury in HCV patients was not the primary cause of the accentuated mortality effect of the SARS-CoV-2 virus.
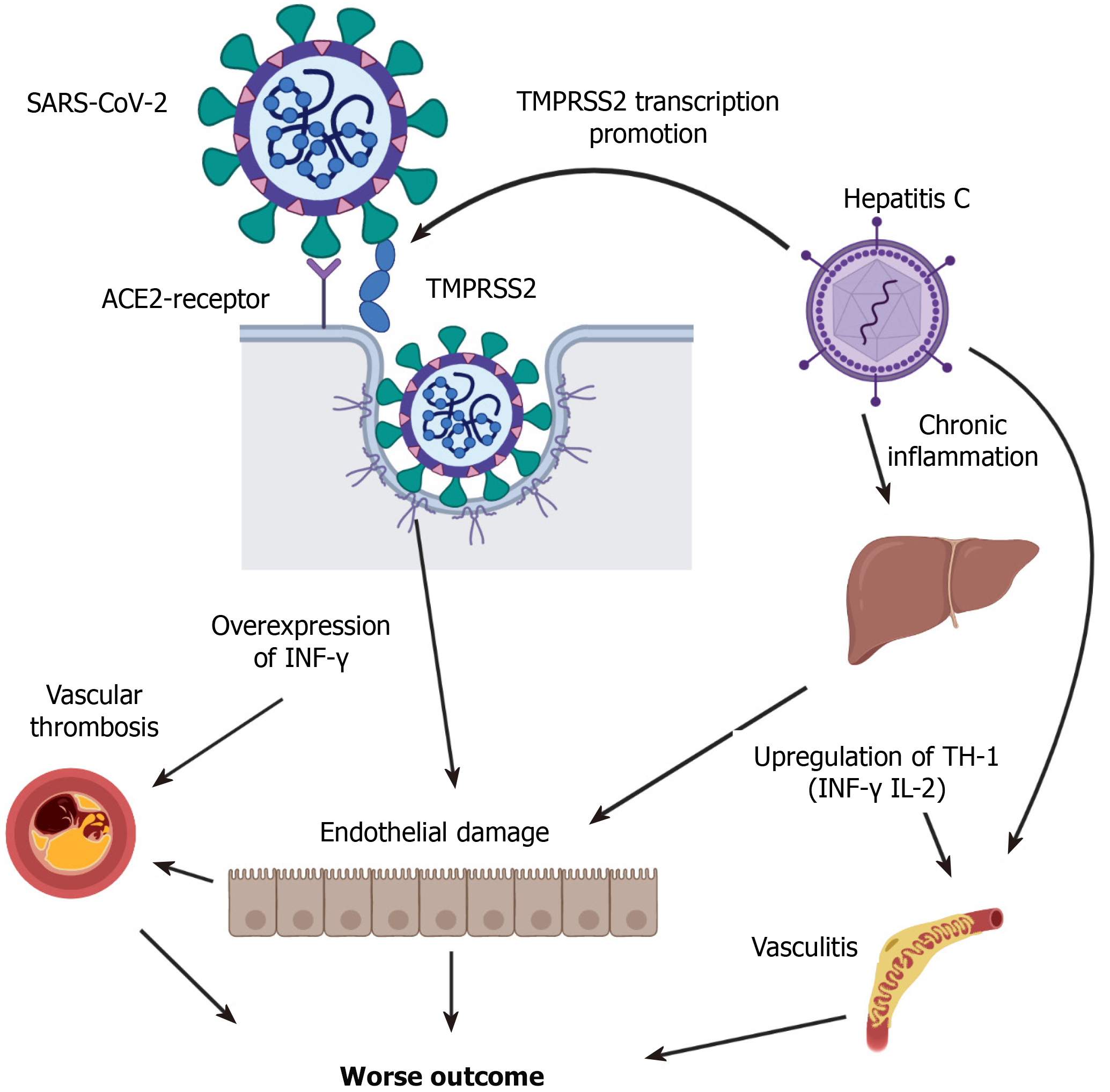
A review of the pathophysiology of the infection of HCV and SARS-CoV-2 elucidates several overlapping mechanisms (Figure 3). SARS-CoV-2 uses the ACE-2 receptor as a main point of entry to the target cell[8]. ACE2 receptors exist on the surface of the cholangiocytes indicating that the liver could be a potential target for SARS-CoV-2, however[8], data so far shows that SARS-CoV-2 seldom affects the liver severely. One molecule that has been identified to potentiate SARS-CoV-2 viral entry is the transmembrane protease serine 2 (TMPRSS2), which is suggested to affect the S protein at the cell surface and induces SARS-CoV-2-cellular membrane fusion[9]. Importantly, TMPRSS2 is over-expressed in patients with HCV which may lead the exaggerated SARS-CoV-2 infection in these patients[10]. Second, HCV is a systemic disease, associated with significant extrahepatic morbidity despite that can occur independently of liver injury[11]. Reportedly, HCV antibodies seropositivity was associated with 1.5-folds increased non-liver-related mortality[12]. The mechanisms may involve the induction of circulating immune complexes and immune-mediated cellular toxicity, which is reported to occur in SARS-CoV-2 and may be related to the potentiated effects on the virus in HCV patients. Third, there seems to be an immune response overlap between chronic HCV and SARS-CoV-2 infection[13]. Studies have shown that in chronic HCV infection there is a correlation between the production of pro-inflammatory cytokines, such as INF-γ and TNF-α, and progressive liver injury, while the regulatory cytokines such as IL-4 and IL-10 may modulate the pro-inflammatory immune response induced by the virus[14,15]. Importantly, several cytokines are elevated in SARS-CoV-2 and are significantly associated with mortality[16-18]. A recent study showed that higher levels of INF-γ, elevated in both conditions, are associated with elevated mortality in patients with COVID-19[16]. Moreover, HCV triggers a systemic cytokine response controlled by TNF and IL-10. This may indicate that a baseline inflammatory status in patients with HCV could be related to the higher mortality in those patients when contracting COVID-19. Reportedly, the cytokine response to HCV infection mirrors the liver fibrosis score[17], which sheds the light on the fact that despite the FIB-4 score in our study was not different between HCV and non-HCV patients, it was the only other predictor of mortality in the matched group.
Finally, Vascular endothelial dysfunction is another overlapping mechanism between HCV and SARS-CoV-2 viruses. Reportedly, chronic HCV accelerates atherosclerotic process by inducing endothelial dysfunction and is significantly influenced by the progression of liver fibrosis[18,19]. Recent pathological reports have shown that viral elements exist within endothelial cells of patients with COVID-19 associated with the accumulation of inflammatory cells, causing endotheliitis[15]. Reportedly, the integrity of the endothelial function and immune system is crucial for a regulated immune response good enough to control the virus spread[20,21]. As such, direct endothelial infection in COVID-19 patients can be aggravated by the baseline endothelial dysfunction reported to occur in patients with chronic HCV infection.
The following limitations existed in our study and should be investigated in future studies: First, the sample size particularly that of HCV patients is small, however, this is a limitation that arises from the prevalence of HCV in COVID-19 patients. Future studies and meta-analyses should be done to check if the findings of the current study can be reproduced. Second, viral loads were not obtained for all the patients with a history of HCV, and as such, the status of their HCV infection, viral clearance and its effects on mortality could not be assessed precisely. Third, despite the fact that the postulated mechanisms are all dependent on pathological, functional, or microbiological evidence, none of these have been measured in our study. Information regarding differences in cytokines, endothelial function, and ACE-2 activity should be evaluated in future studies between COVID-19 patients with and without HCV.
In patients with COVID-19, a history of HCV and seropositive HCV infection leads to accentuated SARS-CoV-2 viral virulence and is a strong predictor of in-hospital mortality irrespective of baseline comorbidities, admission laboratory variables, or COVID-19-induced liver injury. The history of HCV in these patients seems to add a cumulative mortality risk to any clinical or laboratory profile. The mechanisms involved may be related to extrahepatic effects of HCV leading to enhanced ACE-2/TMPRSS mechanisms of SARS-CoV-2 viral entry and may also be related to baseline cytokine-mediated pro-inflammation and endothelial dysfunction. The realization and understanding of these mechanisms may help in better characterization of the disease and investigating possible therapeutic options in this subgroup of patients, which is of significant importance as an initial step towards the selection of at-risk groups that can benefit the most of the developing vaccines at an earlier stage of the disease.
The effect of preexisting chronic hepatitis C infection in coronavirus disease 2019 (COVID-19) patients has not been studied. Initial reports have suggested that COVID-19 patients with hepatitis C virus (HCV) are at increased risk for poor outcomes especially if accompanied by other risk factors such as advanced age and hypertension.
We have found that hepatitis C is associated with increased in-hospital mortality in patients hospitalized with COVID-19.
We aimed at assessing the relationship between in-hospital mortality with other predictors of poor outcomes in the presence or absence of baseline liver dysfunction or COVID-19 induced acute liver injury.
We reviewed retrospectively the medical charts of patients with COVID-19 admission and the laboratory parameter in patients with and without history of HCV infection. Patients were then divided into two groups based on the presence or absence of a history of HCV and their demographic, clinical, laboratory variables, and outcomes were compared. The primary endpoint of our study was in-hospital mortality.
Multivariate cox-regression model for liver profile revealed that aspartate aminotransferase/ alanine aminotransferase ratio, Fib-4 score, and HCV were predictors of in-hospital mortality. After propensity score matching HCV was the only predictor of mortality in the multivariate cox-regression model. 2-log likelihood ratio was done to compare different diagnostic models and it was found that a model including HCV add a predictive value to clinical and laboratory parameters.
HCV infection accentuated severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) viral virulence in our study and had a strong association with in-hospital mortality irrespective of baseline co-morbidities, admission laboratory variables, or COVID-19-induced liver injury. The mechanisms involved may be related to extra-hepatic effects of HCV, baseline cytokine-mediated pro-inflammation, and endothelial dysfunction. Understanding of these mechanisms may help in better characterization of the disease and investigating possible therapeutic options in this subgroup of patients.
COVID-19 is significantly heterogeneous. Predictors of Mortality are numerous and association of baseline risk and co-morbidities are still developing. Based on an observation of HCV association with increased risk in our institution we sought to investigate the relationship between HCV and COVID-19 mortality
| 1. | Portincasa P, Krawczyk M, Machill A, Lammert F, Di Ciaula A. Hepatic consequences of COVID-19 infection. Lapping or biting? Eur J Intern Med. 2020;77:18-24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 77] [Article Influence: 12.8] [Reference Citation Analysis (1)] |
| 2. | Napodano C, Pocino K, Stefanile A, Marino M, Miele L, Gulli F, Basile V, Pandolfi F, Gasbarrini A, Rapaccini GL, Basile U. COVID-19 and hepatic involvement: The liver as a main actor of the pandemic novel. Scand J Immunol. 2021;93:e12977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 3. | Cacoub P, Comarmond C, Domont F, Savey L, Desbois AC, Saadoun D. Extrahepatic manifestations of chronic hepatitis C virus infection. Ther Adv Infect Dis. 2016;3:3-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 128] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 4. | Kovalic AJ, Satapathy SK, Thuluvath PJ. Prevalence of chronic liver disease in patients with COVID-19 and their clinical outcomes: a systematic review and meta-analysis. Hepatol Int. 2020;14:612-620. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 97] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 5. | Reddy KR. SARS-CoV-2 and the Liver: Considerations in Hepatitis B and Hepatitis C Infections. Clin Liver Dis (Hoboken). 2020;15:191-194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 6. | Des Jarlais DC, Cooper HLF, Arasteh K, Feelemyer J, McKnight C, Ross Z. Potential geographic "hotspots" for drug-injection related transmission of HIV and HCV and for initiation into injecting drug use in New York City, 2011-2015, with implications for the current opioid epidemic in the US. PLoS One. 2018;13:e0194799. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 7. | Phipps MM, Barraza LH, LaSota ED, Sobieszczyk ME, Pereira MR, Zheng EX, Fox AN, Zucker J, Verna EC. Acute Liver Injury in COVID-19: Prevalence and Association with Clinical Outcomes in a Large U.S. Cohort. Hepatology. 2020;72:807-817. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 300] [Cited by in RCA: 278] [Article Influence: 46.3] [Reference Citation Analysis (0)] |
| 8. | Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631-637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4648] [Cited by in RCA: 4074] [Article Influence: 185.2] [Reference Citation Analysis (7)] |
| 9. | Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020; 181: 271-280. e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17180] [Cited by in RCA: 14386] [Article Influence: 2397.7] [Reference Citation Analysis (7)] |
| 10. | Esumi M, Ishibashi M, Yamaguchi H, Nakajima S, Tai Y, Kikuta S, Sugitani M, Takayama T, Tahara M, Takeda M, Wakita T. Transmembrane serine protease TMPRSS2 activates hepatitis C virus infection. Hepatology. 2015;61:437-446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 11. | Gill K, Ghazinian H, Manch R, Gish R. Hepatitis C virus as a systemic disease: reaching beyond the liver. Hepatol Int. 2016;10:415-423. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 109] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 12. | Hisada M, Chatterjee N, Kalaylioglu Z, Battjes RJ, Goedert JJ. Hepatitis C virus load and survival among injection drug users in the United States. Hepatology. 2005;42:1446-1452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 13. | Thurairajah P, Khanna A, Mutimer D. Add-on combination therapy with adefovir dipivoxil induces renal impairment in patients with lamivudine-refractory hepatitis B virus (J Viral Hepat 2010 Feb 1;17 (2):123-9). J Viral Hepat. 2011;18:820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 14. | Hofmann SR, Ettinger R, Zhou YJ, Gadina M, Lipsky P, Siegel R, Candotti F, O'Shea JJ. Cytokines and their role in lymphoid development, differentiation and homeostasis. Curr Opin Allergy Clin Immunol. 2002;2:495-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 78] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 15. | Rehermann B. Hepatitis C virus versus innate and adaptive immune responses: a tale of coevolution and coexistence. J Clin Invest. 2009;119:1745-1754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 425] [Cited by in RCA: 420] [Article Influence: 24.7] [Reference Citation Analysis (1)] |
| 16. | Gadotti AC, de Castro Deus M, Telles JP, Wind R, Goes M, Garcia Charello Ossoski R, de Padua AM, de Noronha L, Moreno-Amaral A, Baena CP, Tuon FF. IFN-γ is an independent risk factor associated with mortality in patients with moderate and severe COVID-19 infection. Virus Res. 2020;289:198171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 141] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 17. | de Souza-Cruz S, Victória MB, Tarragô AM, da Costa AG, Pimentel JP, Pires EF, Araújo Lde P, Coelho-dos-Reis JG, Gomes Mde S, Amaral LR, Teixeira-Carvalho A, Martins-Filho OA, Victória Fda S, Malheiro A. Liver and blood cytokine microenvironment in HCV patients is associated to liver fibrosis score: a proinflammatory cytokine ensemble orchestrated by TNF and tuned by IL-10. BMC Microbiol. 2016;16:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 18. | Oliveira CP, Kappel CR, Siqueira ER, Lima VM, Stefano JT, Michalczuk MT, Marini SS, Barbeiro HV, Soriano FG, Carrilho FJ, Pereira LM, Alvares-da-Silva MR. Effects of hepatitis C virus on cardiovascular risk in infected patients: a comparative study. Int J Cardiol. 2013;164:221-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 70] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 19. | Barone M, Viggiani MT, Amoruso A, Schiraldi S, Zito A, Devito F, Cortese F, Gesualdo M, Brunetti N, Di Leo A, Scicchitano P, Ciccone MM. Endothelial dysfunction correlates with liver fibrosis in chronic HCV infection. Gastroenterol Res Pract. 2015;2015:682174. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 20. | Perico L, Benigni A, Casiraghi F, Ng LFP, Renia L, Remuzzi G. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat Rev Nephrol. 2021;17:46-64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 501] [Cited by in RCA: 399] [Article Influence: 79.8] [Reference Citation Analysis (0)] |
| 21. | Ronderos Botero DM, Omar AMS, Sun HK, Mantri N, Fortuzi K, Choi Y, Adrish M, Nicu M, Bella JN, Chilimuri S. COVID-19 in the Healthy Patient Population: Demographic and Clinical Phenotypic Characterization and Predictors of In-Hospital Outcomes. Arterioscler Thromb Vasc Biol. 2020;40:2764-2775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/Licenses/by-nc/4.0/
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Kumar R, Shahini E S-Editor: Liu M L-Editor: A P-Editor: Li JH