Published online Jan 26, 2020. doi: 10.12998/wjcc.v8.i2.479
Peer-review started: October 30, 2019
First decision: November 13, 2019
Revised: December 13, 2019
Accepted: December 22, 2019
Article in press: December 22, 2019
Published online: January 26, 2020
Processing time: 78 Days and 19.3 Hours
Moonwort is a widely used Chinese herbal medicine. It has various pharmacological effects, such as relieving cough and preventing asthma. To date, multiple organ dysfunction and rhabdomyolysis caused by moonwort poisoning have not been reported.
Here we report four cases of moonwort poisoning that presented with multiple organ dysfunction and rhabdomyolysis accompanied by vomiting, fatigue, and muscle aches. One patient was an adult male, two were adult females, and one was a boy, with an age range of 7–64 years. The adults were treated with hemoperfusion and symptomatic therapies, while the child was treated with plasma exchange and symptomatic therapies. All four patients recovered.
Blood purification combined with symptomatic treatment may be an effective method for managing multiple organ dysfunction and rhabdomyolysis caused by acute moonwort poisoning.
Core tip: Moonwort is a widely used Chinese herbal medicine. To date, multiple organ dysfunction and rhabdomyolysis caused by moonwort poisoning have not been reported. In this case report, we describe the effects of blood purification combined with symptomatic treatment in patients with multiple organ dysfunction and rhabdomyolysis caused by moonwort poisoning. Use of this treatment was effective and safe in these patients.
- Citation: Li F, Chen AB, Duan YC, Liao R, Xu YW, Tao LL. Multiple organ dysfunction and rhabdomyolysis associated with moonwort poisoning: Report of four cases. World J Clin Cases 2020; 8(2): 479-486
- URL: https://www.wjgnet.com/2307-8960/full/v8/i2/479.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v8.i2.479
Core tip: Moonwort is a widely used Chinese herbal medicine. To date, multiple organ dysfunction and rhabdomyolysis caused by moonwort poisoning have not been reported. In this case report, we describe the effects of blood purification combined with symptomatic treatment in patients with multiple organ dysfunction and rhabdomyolysis caused by moonwort poisoning. Use of this treatment was effective and safe in these patients.
Moonwort is a plant of the genus Pteridaceae and the genus Pteridium. Moonwort is widely distributed in Yunnan, Hubei, Zhejiang, and other provinces in China. It is a widely used Chinese herbal medicine, which has numerous pharmacological effects, such as relieving cough and preventing asthma[1,2]. In some parts of China, residents have the habit of eating moonwort. However, the chemical composition of moonwort is complex and diverse. Its effective dose and toxic dose are unknown. To date, few studies have investigated moonwort poisoning[3-5]. The present report describes four cases of multiple organ dysfunction and rhabdomyolysis caused by moonwort poisoning.
Among the four patients, one was an adult male, two were adult females, and one was a boy. The age range of the patients was 7–64 years. On October 22, 2018, the four patients developed symptoms such as vomiting, fatigue, and muscle aches and pain after eating a mixture of “herbal medicine” and herbal liquid.
These patients are members of the same family (Table 1). Case 2 had a history of hypertension. The other cases had no particular medical history.
| Case | Age (yr) | Weight (kg) | Past medical history | Time since poisoning to symptoms, h | Dose of herbal liquid and medicine |
| Case 1 | 64 | 65 | No | 6 | 200 mL + 20 g |
| Case 2 | 60 | 51 | Hypertension | 6 | 150 mL + 10 g |
| Case 3 | 36 | 53 | No | 2 | 450 mL + 40 g |
| Case 4 | 7 | 28 | No | 4 | 100 mL + 10 g |
The patients visited the local hospital on the same day and were diagnosed with acute herbal poisoning. After receiving gastric lavage, laxative drugs, and other treatments, the symptoms were not relieved. The patients were subsequently transferred to our hospital on October 23. The four patients had typical vital signs. There were no abnormal signs on physical examination (Table 2).
| Case | Blood pressure (mmHg) | Pulse (beats/min) | Respiratory rate (breaths/min) | Temperature (ºC) | SpO2 (%) |
| Case 1 | 114/69 | 93 | 19 | 36 | 96 |
| Case 2 | 167/77 | 92 | 26 | 37 | 99 |
| Case 3 | 138/97 | 110 | 28 | 36.2 | 95 |
| Case 4 | 124/80 | 109 | 24 | 37.2 | 98 |
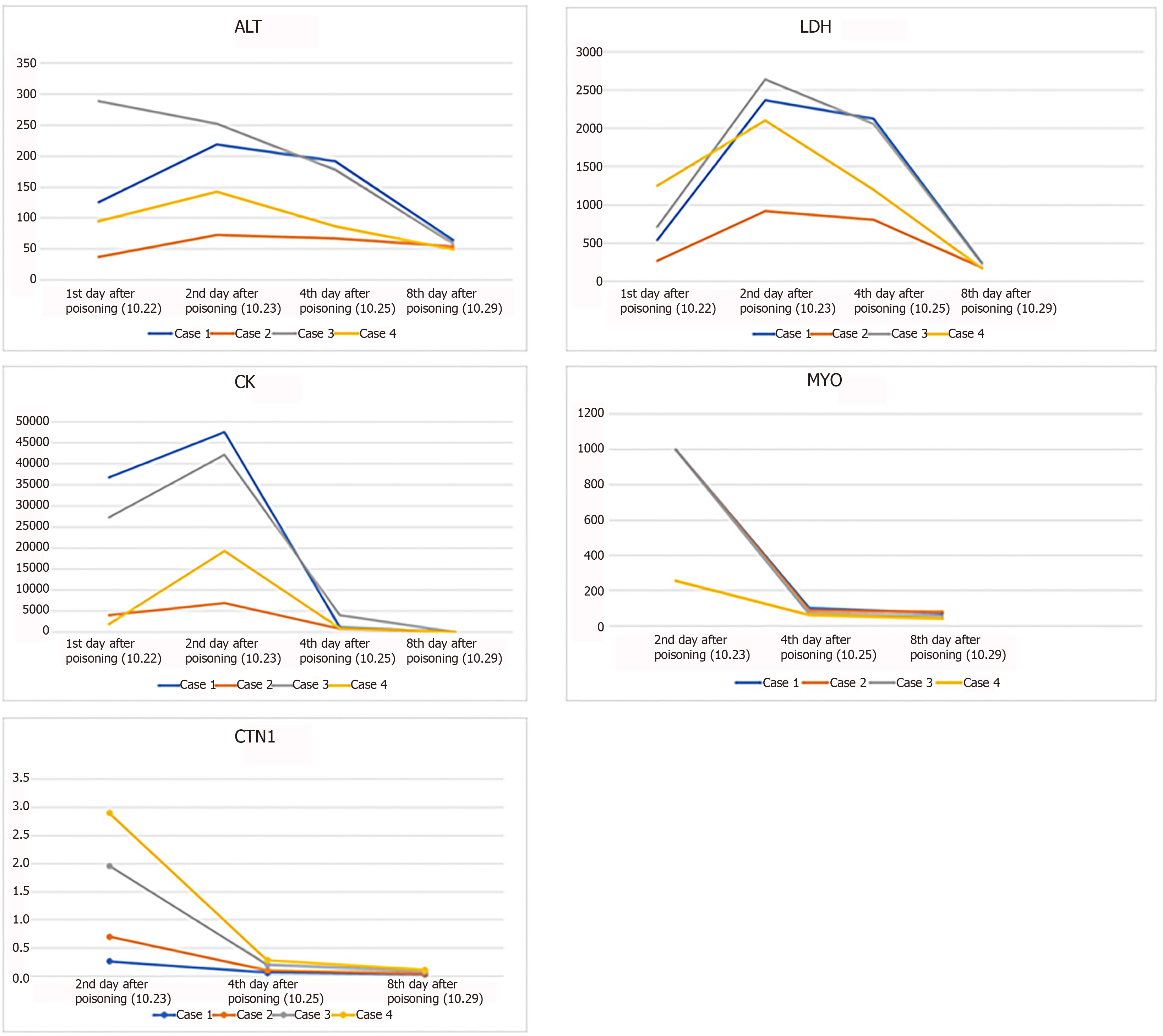
Admission-related investigations revealed that all four patients had liver dysfunction and elevated cardiac enzymes. However, all four patients showed marked elevations in serum levels of cardiac troponin I, myoglobin, and creatine kinase (Table 3). There were also mild increases in white blood cell count and neutrophil count (Table 3). However, electrolyte analysis did not demonstrate hyperkalemia in any of the four patients (Table 3).
| Case | Day1 1 | Day1 1 after treatment2 | Day1 2 after treatment2 | Day1 7 | |
| K+, mmol/L | Case 1 | 3.95 | 4.29 | 3.39 | 4.23 |
| Case 2 | 2.93 | 3.02 | 2.82 | 3.59 | |
| Case 3 | 3.33 | 3.72 | 3.33 | 4.33 | |
| Case 4 | 3.57 | 3.77 | 3.22 | 4.09 | |
| Ca2+, mmol/L | Case 1 | 2.06 | 2.01 | 1.97 | 2.2 |
| Case 2 | 2.11 | 1.96 | 1.98 | 2.19 | |
| Case 3 | 2.08 | 2.02 | 2.05 | 2.27 | |
| Case 4 | 2.27 | 2.17 | 2.19 | 2.39 | |
| Na+, mmol/L | Case 1 | 140.5 | 138.1 | 139.3 | 138.4 |
| Case 2 | 136.1 | 141 | 144.8 | 146.6 | |
| Case 3 | 140.3 | 138.3 | 143.9 | 144.8 | |
| Case 4 | 141.6 | 140.8 | 138.9 | 140.9 | |
| Cl-, mmol/L | Case 1 | 106 | 103.2 | 109.6 | 102.3 |
| Case 2 | 103.4 | 108.5 | 113 | 105 | |
| Case 3 | 106 | 103.2 | 111.7 | 104.2 | |
| Case 4 | 100 | 104.6 | 100.3 | 99.1 | |
| Cardiac troponin I, ng/mL | Case 1 | 0.26 | 0.14 | 0.06 | 0.03 |
| Case 2 | 0.44 | 0.32 | 0.04 | 0.01 | |
| Case 3 | 1.26 | 0.62 | 0.1 | 0.04 | |
| Case 4 | 0.94 | 0.48 | 0.08 | 0.03 | |
| Myoglobin, ng/mL | Case 1 | > 1000 | 316.3 | 102 | 68 |
| Case 2 | > 1000 | 229 | 85 | 57 | |
| Case 3 | > 1000 | 468.7 | 70 | 55 | |
| Case 4 | 225.5 | 225.5 | 40 | 29 | |
| Creatine kinase isoenzyme, ng/mL | Case 1 | > 500 | 318.8 | 40.8 | 2 |
| Case 2 | 155.8 | 84.1 | 11.2 | 3.4 | |
| Case 3 | > 500 | 171.2 | 41.4 | 2.6 | |
| Case 4 | 489.5 | 489.5 | 116.6 | 3.07 | |
| Creatine kinase, U/L | Case 1 | 47643 | 30233 | 30028 | 69 |
| Case 2 | 7039 | 5101 | 2796 | 86 | |
| Case 3 | 42251 | 28228 | 13446 | 112 | |
| Case 4 | 19350 | 5814 | 1458 | 58 | |
| White blood cell count, 109/L | Case 1 | 11.17 | 12.41 | 15.23 | 9.05 |
| Case 2 | 13.14 | 13.03 | 12.93 | 9.38 | |
| Case 3 | 15.14 | 13.68 | 14 | 9.11 | |
| Case 4 | 11.22 | 10.67 | 11.74 | 6.92 | |
| Neutrophil count, 109/L | Case 1 | 9.39 | 11.82 | 13.71 | 6.2 |
| Case 2 | 10.56 | 12.14 | 11.51 | 6.26 | |
| Case 3 | 13.11 | 12.91 | 13.16 | 6.07 | |
| Case 4 | 8.54 | 9.43 | 8.85 | 4.69 | |
| Lymphocyte count, 109/L | Case 1 | 1.37 | 0.58 | 1.13 | 1.27 |
| Case 2 | 1.71 | 0.63 | 1.06 | 2.66 | |
| Case 3 | 1.59 | 0.7 | 0.69 | 1.68 | |
| Case 4 | 1.99 | 1 | 1.84 | 1.61 | |
| Monocyte count, 109/L | Case 1 | 0.37 | 0.01 | 0.38 | 0.39 |
| Case 2 | 0.86 | 0.26 | 0.35 | 0.52 | |
| Case 3 | 0.4 | 0.07 | 0.14 | 0.58 | |
| Case 4 | 0.62 | 0.21 | 1.02 | 0.57 | |
| Eosinophilic granulocyte count, 109/L | Case 1 | 0.03 | 0 | 0 | 0 |
| Case 2 | 0.01 | 0 | 0 | 0 | |
| Case 3 | 0.04 | 0 | 0.01 | 0.02 | |
| Case 4 | 0.04 | 0.01 | 0.01 | 0.03 | |
| Basophilic granulocyte count, 109/L | Case 1 | 0.01 | 0 | 0.01 | 0.01 |
| Case 2 | 0 | 0 | 0.01 | 0.01 | |
| Case 3 | 0 | 0 | 0 | 0.01 | |
| Case 4 | 0.03 | 0.02 | 0.02 | 0.02 | |
| Red blood cell count, 109/L | Case 1 | 5.3 | 4.97 | 4.71 | 4.66 |
| Case 2 | 5.06 | 4.85 | 4.58 | 4.04 | |
| Case 3 | 4.88 | 4.75 | 4.66 | 4 | |
| Case 4 | 4.94 | 4.99 | 4.57 | 4.7 | |
| Blood platelet count, 109/L | Case 1 | 264 | 185 | 111 | 127 |
| Case 2 | 178 | 135 | 108 | 132 | |
| Case 3 | 404 | 320 | 258 | 273 | |
| Case 4 | 364 | 335 | 313 | 266 | |
| Hemoglobin, g/L | Case 1 | 163 | 154 | 145 | 141 |
| Case 2 | 148 | 145 | 136 | 121 | |
| Case 3 | 116 | 113 | 111 | 117 | |
| Case 4 | 135 | 135 | 125 | 137 |
Pulmonary computed tomography for the four patients showed different degrees of exudation. There were no abnormalities in renal function tests, routine urine tests, or the electrocardiogram in any of the four patients.
Based on the clinical history, symptom pattern, and elevated levels of creatine kinase and myoglobin, the admission diagnosis was acute herbal poisoning, multiple organ dysfunction, and rhabdomyolysis[6,7]. During treatment at our department, the family provided the fresh and complete plants of the “herbal medicine” consumed by the patients (Figure 1). After consulting the literature and relevant Chinese herbal medicine experts, the poison was identified as moonwort. Hence, the patients were diagnosed with acute moonwort poisoning, multiple organ dysfunction (heart, liver, and lung), and rhabdomyolysis.
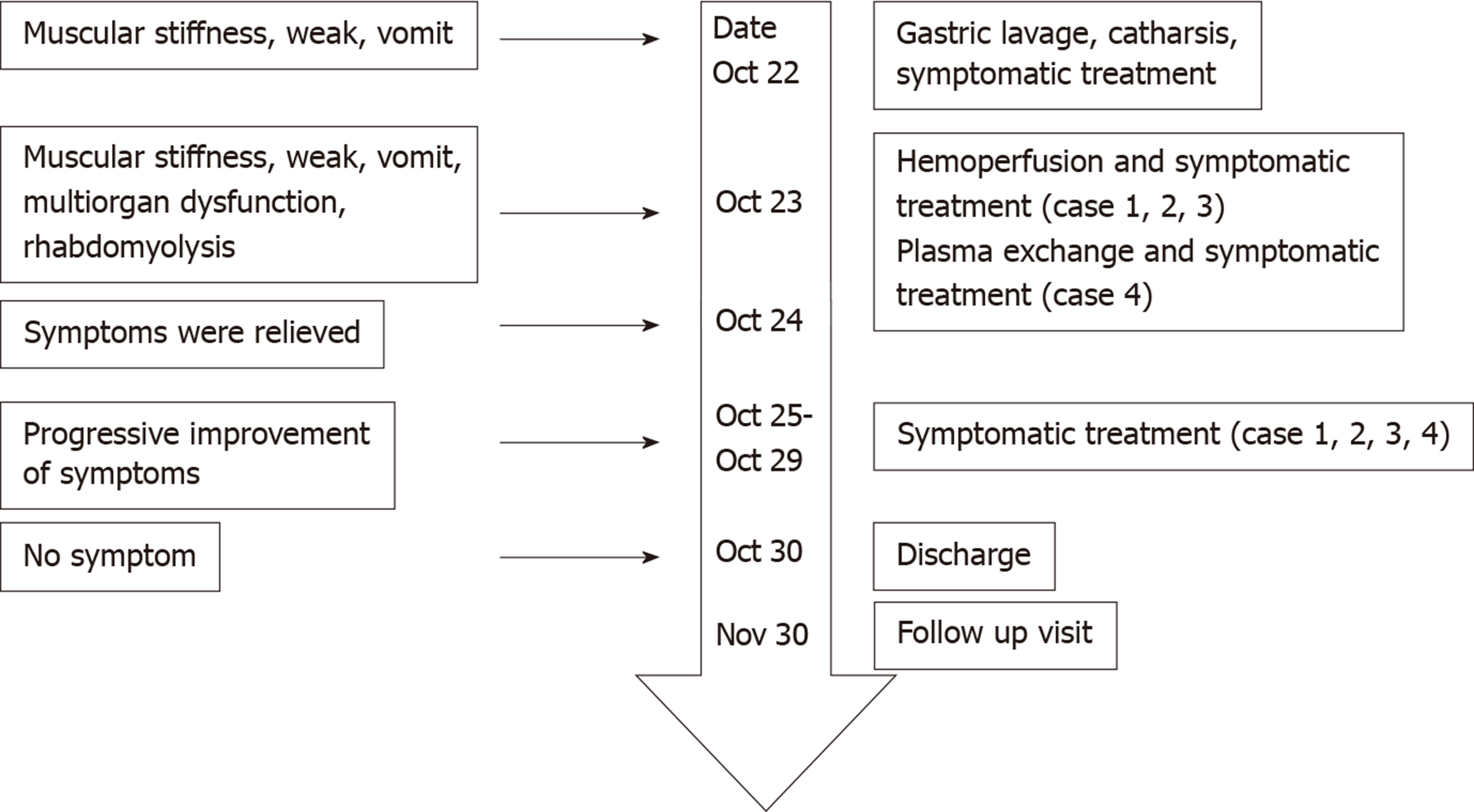
From the day of admission, all patients were treated with oxygen therapy, liver protecting agents, cardiac nutritional treatments, anti-oxidants, anti-inflammatory drugs, and methylprednisolone. The therapeutic dose of methylprednisolone was calculated based on the patient’s body weight. After consent of the patient and their family members, cases 1, 2, and 3 underwent two sessions of hemoperfusion for 2 hours on the day of admission and the second day after admission. Due to the absence of a pediatric hemoperfusion device, case 4 received two sessions of plasma exchanges on the day of admission and the second day after admission (Table 4). The methylprednisolone dose was gradually reduced on October 29. During the blood purification treatment, the patients did not develop complications such as hemorrhage or shock. After treatment, the patients’ symptoms were gradually relieved, and there was no other discomfort. Blood test indicators slowly returned to normal (Figure 2, Table 3). On October 30, all patients had recovered and were discharged.
The four patients were followed at 30 d after discharge, and no further symptoms were reported. The treatment process and timeline are shown in Figure 3.
Here, we have reported four cases of acute moonwort poisoning with multiple organ dysfunction (heart, liver, and lung) and rhabdomyolysis. The mechanism of toxic fern poisoning remains unclear. A previous study[8] found that moonwort contains flavonoids, plant proteins, cardiac glycosides, saponins, and phenols, but not alkaloids or oils. However, since no previous studies have identified which components of moonwort have the ability to induce rhabdomyolysis, it is not possible to draw any conclusions regarding the causative compound(s) in the four cases described here. At present, the identification of moonwort is mainly based on trait and microscopy. Zhou et al[9] reported a detailed analysis and description of the morphology of the moonwort plant. After a comparative review, the four patients were diagnosed with acute moonwort poisoning. The time to symptom onset was 2–6 h, and the disease course was 8 d. The clinical condition of the patients was worse than that described previously in the literature[3-5]. In these reports, acute moonwort poisoning caused damage to the liver, kidneys, and some other organs, but no lung injury or rhabdomyolysis occurred. The time to symptom onset was 2–8 h, and the patients recovered after symptomatic treatment within 72 h. The severity of our cases in comparison to those in previous reports may be related to factors such as a higher dose of moonwort, consumption during a different season, and different origin of the moonwort. The fertility of moonwort in different habitats is not the same[8], and the efficacy of moonwort has noticeable seasonal differences[10].
Rhabdomyolysis is an acquired or inherited clinical syndrome characterized by the destruction of skeletal muscle and release of intracellular constituents (such as myoglobin, enzymes, and electrolytes) that lead to a variety of systemic complications[6]. The causes of acquired rhabdomyolysis are varied and include trauma, intense exertion, ischemia, thermal injury, drugs, and toxins[6,11]. Regardless of the underlying cause, direct injury to the skeletal muscle membrane or energy depletion results in an increase in intracellular calcium that activates proteases and apoptotic pathways, leading to the generation of oxygen free radicals, mitochondrial dysfunction, and cell death[12,13]. Rhabdomyolysis typically presents with myalgia, weakness, and/or myoglobinuria, but an accurate diagnosis is facilitated by the detection of an elevated creatine kinase level[6,7,11-13]. Acute kidney injury, the most frequent systemic complication of rhabdomyolysis, occurs with an incidence of 10%–40% and is associated with a poor prognosis, especially if multiple organ failure is also present[6,13]. Other complications include electrolyte disturbances (such as hyperkalemia), hypovolemia, compartment syndrome, and disseminated intravascular coagulation[13]. The management of rhabdomyolysis includes treatment of the underlying cause, infusion of fluids to correct hypovolemia and electrolyte disturbances, alkalinization of the urine with sodium bicarbonate, and decompression of muscle compartments[13,14].
Rhabdomyolysis may be associated with hyperkalemia due to the loss of large quantities of intracellular K+ from the damaged skeletal muscle and the development of acute kidney injury[15]. It is essential that hyperkalemia is rapidly corrected because it can result in potentially life-threatening arrhythmia[6]. However, it was notable that none of the four patients in our study exhibited an elevated level of plasma K+ before treatment. This may have been due to the rapid initiation of treatment and the absence of renal injury in these patients, which allowed the kidneys to compensate for the loss of K+ from skeletal muscle.
There is no specific antidote for acute moonwort poisoning. Gastric lavage, purgative agents, protection of essential organ functions, scavenging of oxygen free radicals, and prevention of complications are recommended. The four patients in this report developed liver, heart, and lung dysfunction as well as rhabdomyolysis after acute moonwort poisoning. Their condition was more serious than that described in previous reports. Since moonwort contains various chemicals, we used repeated hemoperfusion or plasma exchange along with symptomatic treatment. Hemoperfusion can eliminate medium- and large-sized toxic molecules and is widely used in the treatment of acute poisoning in Asia. It has a good effect on various poisons, such as paraquat and organic phosphorus[16]. In previous studies, some patients with severe poisoning also underwent repeated hemoperfusion, and good therapeutic results were achieved[17]. Plasma exchange is less applicable than hemoperfusion in acute poisoning. However, the daily replacement of up to 1–2 times the patient's plasma volume until symptom relief is achieved can effectively reduce the damage caused by the poison[18]. Acetylcysteine may also have an important role in the management of poisoning. Acetylcysteine is used as an anti-oxidant to prevent and treat cell damage, reduce apoptosis, and improve mitochondrial function[19-21]. Acetylcysteine can also improve the patient’s renal metabolism and microcirculation[22]. These effects may be beneficial in alleviating multiple organ dysfunction and improving renal function after rhabdomyolysis.
We have reported four cases of multiple organ dysfunction and rhabdomyolysis caused by moonwort poisoning. The patients were managed using blood purification techniques and symptomatic treatments. Their symptoms completely disappeared on the ninth day after poisoning. Blood purification combined with symptomatic treatment may be an effective method for acute moonwort poisoning. However, further research is required to confirm these findings.
| 1. | Yuan Y, Yang B, Ye Z, Zhang M, Yang X, Xin C, Lin M, Huang P. Sceptridium ternatum extract exerts antiasthmatic effects by regulating Th1/Th2 balance and the expression levels of leukotriene receptors in a mouse asthma model. J Ethnopharmacol. 2013;149:701-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 2. | Chuang SY, Lee SC, Hsieh YT, Pan WH. Trends in hyperuricemia and gout prevalence: Nutrition and Health Survey in Taiwan from 1993-1996 to 2005-2008. Asia Pac J Clin Nutr. 2011;20:301-308. [PubMed] |
| 3. | Liu ML, Yuan WT. Analysis of 15 cases of acute poisoning of moonwort fern. Shijie Linchuang Yixue Zazhi. 2016;10:227. |
| 4. | Gao Y, Yang LY, Yin F, Liu RC. One case of multiple organ dysfunction caused by overdose of moonwort. Shiyong Yixue Zazhi. 2017;71. [DOI] [Full Text] |
| 5. | Dai EF. Two cases of fern poisoning in the genus. Zhongcaoyao Zazhi. 1999;1. [DOI] [Full Text] |
| 6. | Chavez LO, Leon M, Einav S, Varon J. Beyond muscle destruction: a systematic review of rhabdomyolysis for clinical practice. Crit Care. 2016;20:135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 327] [Cited by in RCA: 278] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 7. | Zimmerman JL, Shen MC. Rhabdomyolysis. Chest. 2013;144:1058-1065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 243] [Article Influence: 20.3] [Reference Citation Analysis (1)] |
| 8. | Farrar DR, Stensvold MC. Observations on bipolar disjunctions of moonwort ferns (Botrychium, Ophioglossaceae). Am J Bot. 2017;104:1675-1679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 9. | Zhou C, Lu XW, Wei JL, Zhan XP, Zhou HH. Plant morphology and microscopic identification of a cloud of Miao medicine (F. fuliginea). Zhongguo Minzu Minjian Yiyao Zazhi. 2011;20:8-9. [DOI] [Full Text] |
| 10. | Wu S, Li J, Wang Q, Cao H, Cao J, Xiao J. Seasonal dynamics of the phytochemical constituents and bioactivities of extracts from Stenoloma chusanum (L.) Ching. Food Chem Toxicol. 2017;108:458-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 11. | Coban YK. Rhabdomyolysis, compartment syndrome and thermal injury. World J Crit Care Med. 2014;3:1-7. [PubMed] [DOI] [Full Text] |
| 12. | Giannoglou GD, Chatzizisis YS, Misirli G. The syndrome of rhabdomyolysis: Pathophysiology and diagnosis. Eur J Intern Med. 2007;18:90-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 214] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 13. | Torres PA, Helmstetter JA, Kaye AM, Kaye AD. Rhabdomyolysis: pathogenesis, diagnosis, and treatment. Ochsner J. 2015;15:58-69. [PubMed] |
| 14. | Cervellin G, Comelli I, Benatti M, Sanchis-Gomar F, Bassi A, Lippi G. Non-traumatic rhabdomyolysis: Background, laboratory features, and acute clinical management. Clin Biochem. 2017;50:656-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 83] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 15. | Chatzizisis YS, Misirli G, Hatzitolios AI, Giannoglou GD. The syndrome of rhabdomyolysis: complications and treatment. Eur J Intern Med. 2008;19:568-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 171] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 16. | Hsu CW, Lin JL, Lin-Tan DT, Chen KH, Yen TH, Wu MS, Lin SC. Early hemoperfusion may improve survival of severely paraquat-poisoned patients. PLoS One. 2012;7:e48397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 62] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 17. | Li C, Hu D, Xue W, Li X, Wang Z, Ai Z, Song Y, Liu X, Cheng J, Fan S, Zhao L, Wang L, Mohan C, Peng A. Treatment Outcome of Combined Continuous Venovenous Hemofiltration and Hemoperfusion in Acute Paraquat Poisoning: A Prospective Controlled Trial. Crit Care Med. 2018;46:100-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 18. | Szczepiorkowski ZM, Winters JL, Bandarenko N, Kim HC, Linenberger ML, Marques MB, Sarode R, Schwartz J, Weinstein R, Shaz BH; Apheresis Applications Committee of the American Society for Apheresis. Guidelines on the use of therapeutic apheresis in clinical practice--evidence-based approach from the Apheresis Applications Committee of the American Society for Apheresis. J Clin Apher. 2010;25:83-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 385] [Cited by in RCA: 338] [Article Influence: 21.1] [Reference Citation Analysis (4)] |
| 19. | de Jesus Pires de Moraes A, Andreato LV, Branco BHM, da Silva EL, Gonçalves MA, Dos Santos RZ, Becker AM, da Silveira Cavalcante L, da Silva Casagrande F, Benetti M. Effects of N-acetylcysteine supplementation on cellular damage and oxidative stress indicators in volleyball athletes. J Exerc Rehabil. 2018;14:802-809. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 20. | Al-Nahdi AMT, John A, Raza H. Cytoprotective Effects of N-Acetylcysteine on Streptozotocin- Induced Oxidative Stress and Apoptosis in RIN-5F Pancreatic β-Cells. Cell Physiol Biochem. 2018;51:201-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 21. | Zhang H, Li Y, Chen Y, Zhang L, Wang T. N-Acetylcysteine protects against intrauterine growth retardation-induced intestinal injury via restoring redox status and mitochondrial function in neonatal piglets. Eur J Nutr. 2019;58:3335-3347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 22. | Ergin B, Guerci P, Zafrani L, Nocken F, Kandil A, Gurel-Gurevin E, Demirci-Tansel C, Ince C. Effects of N-acetylcysteine (NAC) supplementation in resuscitation fluids on renal microcirculatory oxygenation, inflammation, and function in a rat model of endotoxemia. Intensive Care Med Exp. 2016;4:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See:
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Coban YK S-Editor: Zhang L L-Editor: Wang TQ E-Editor: Xing YX