Published online May 26, 2026. doi: 10.12998/wjcc.v14.i15.119781
Revised: March 13, 2026
Accepted: April 8, 2026
Published online: May 26, 2026
Processing time: 85 Days and 1.8 Hours
Systemic treatment options for hepatocellular carcinoma (HCC) following atezolizumab plus bevacizumab (Atez/Bev) failure are limited, particularly for patients with deteriorated liver function and poor tolerance to tyrosine kinase inhibitors (TKIs). This case highlights the potential role of ramucirumab (RAM) as later-line therapy in challenging clinical settings.
An 83-year-old man with a history of hepatitis C-related HCC developed disseminated recurrence involving the splenic hilum 12 years after curative resection. The lesion was surgically resected following four courses of Atez/Bev. After 17 months, intrahepatic and liver surface recurrences were detected. Atez/Bev was reintroduced as a first-line therapy but resulted in disease progression after 17 courses. Second-line lenvatinib and third-line sorafenib were discontinued because of severe fatigue, appetite loss, marked tumor progression, and wor
Following Atez/Bev failure, RAM may provide mechanistically rational and effective therapy in AFP-high HCC for patients intolerant to kinase inhibitor.
Core Tip: Hepatocellular carcinoma with elevated alpha-fetoprotein levels that progresses after treatment with atezolizumab plus bevacizumab may involve enhanced vascular endothelial growth factor receptor-2 signaling, making ramucirumab a potentially effective treatment option.
- Citation: Fukushima M, Nakao Y, Sasaki R, Haraguchi M, Miuma S, Miyaaki H. Dramatic response to fourth-line ramucirumab after atezolizumab plus bevacizumab failure in advanced hepatocellular carcinoma: A case report. World J Clin Cases 2026; 14(15): 119781
- URL: https://www.wjgnet.com/2307-8960/full/v14/i15/119781.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i15.119781
In Japan, there are currently six available regimens as first-line drug therapies for hepatocellular carcinoma (HCC), including immune checkpoint inhibitor regimens such as nivolumab plus ipilimumab, tremelimumab plus durvalumab, atezolizumab plus bevacizumab (Atez/Bev), and durvalumab monotherapy, as well as the multi-kinase inhibitors sorafenib and lenvatinib (LEN)[1]. Regorafenib, ramucirumab (RAM), and cabozantinib can also be used as second-line and subsequent drug therapies, with nine regimens available. Therefore, it is necessary to select the appropriate medication for each patient. RAM can extend overall survival compared with placebo in patients with HCC and alpha-fetoprotein (AFP) levels ≥ 400 ng/mL who had a history of sorafenib treatment. In the REACH-2 trial, RAM showed an overall response rate of 4.6% (combining complete and partial responses) and a disease control rate of 59.9% when patients with stable diseases were included[2]. Therefore, rather than expecting significant tumor shrinkage, RAM is often used in second- or later-line treatment, with the goal of disease control. Herein, we report a case in which RAM administered as a fourth-line treatment produced remarkable therapeutic effects.
An 83-year-old man experienced HCC-related recurrence in both the intrahepatic and liver surface areas and was referred to our department.
Upon referral to our hospital at 68 years old for HCC and chronic hepatitis C, a 10 mm HCC was identified in liver segment 3, and partial resection was performed. Pathologically, the tumor was a well-differentiated HCC. After HCC treatment, a sustained response to hepatitis C was achieved with pegylated interferon plus ribavirin therapy. Twelve years later, at age 80 years old, a single 20 mm mass was identified at the splenic hilum. The tumor markers for HCC, namely, serum AFP and des-gamma carboxyprothrombin (DCP), were elevated, and recurrence due to HCC-related dissemination was suspected. Because disseminated recurrence was considered, a total of four courses of combination therapy with Atez (1200 mg)/Bev (15 mg/kg) were administered every 3 weeks. After confirming that there were no recurrences at other sites, the tumor and spleen were surgically resected. Seventeen months later, the patient experienced recurrence in both the intrahepatic and liver surface areas and was referred to our department.
The patient achieved a sustained virologic response for hepatitis C with pegylated interferon plus ribavirin therapy at 68 years of age.
The patient had no significant family history of malignancy or known genetic predisposition, nor did he have any significant history of environmental exposure.
No notable findings were observed on physical examination.
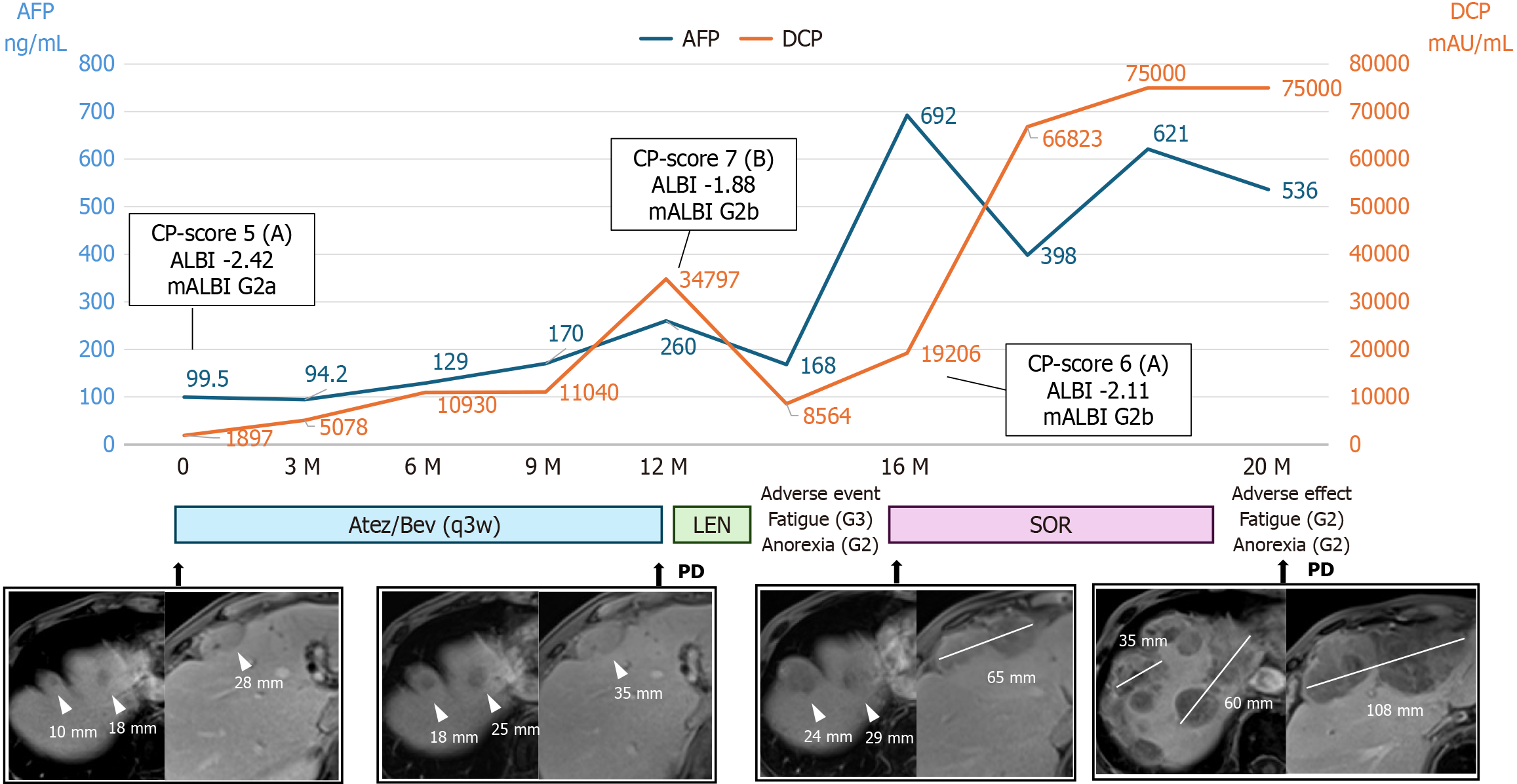
Blood test results at the time of initial referral were as follows: Platelet count, 17.6 × 104/μL; total bilirubin, 1.0 mg/dL (normal range: 0.4-1.5 mg/dL); albumin, 3.8 g/dL (normal range: 4.1-5.1 g/dL); prothrombin time (PT), 76% (PT-international normalized ratio: 1.14); Child-Pugh score, 5 (Class A), and albumin-bilirubin (ALBI) score, -2.42. His serum AFP and DCP levels were 99.5 ng/mL and 1897 mAU/mL, respectively.
Contrast-enhanced computed tomography (CT) revealed two intrahepatic tumors measuring 10 mm and 18 mm in maximum diameter, both showing early arterial enhancement and late-phase washout. In addition, an extrahepatic lesion with a maximum diameter of 28 mm was identified on the liver surface. Given the elevated AFP and DCP serum levels and the history of treatment for splenic hilum area seeding in HCC, HCC recurrence was suspected.
Recurrence of HCC disseminated nodules on the liver surface.
With a Child-Pugh score of 5 and a performance status of 1, Atez/Bev was initiated as first-line therapy. Tumor markers gradually increased during treatment, and after 17 courses, contrast-enhanced CT demonstrated growth of the intrahepatic target lesions from a total of 28 mm to 43 mm, along with growth of the extrahepatic lesion from 28 mm to 35 mm in maximum diameter, leading to a diagnosis of progressive disease according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. At this time, the Child-Pugh score was 7 points (Class B) and the ALBI score was
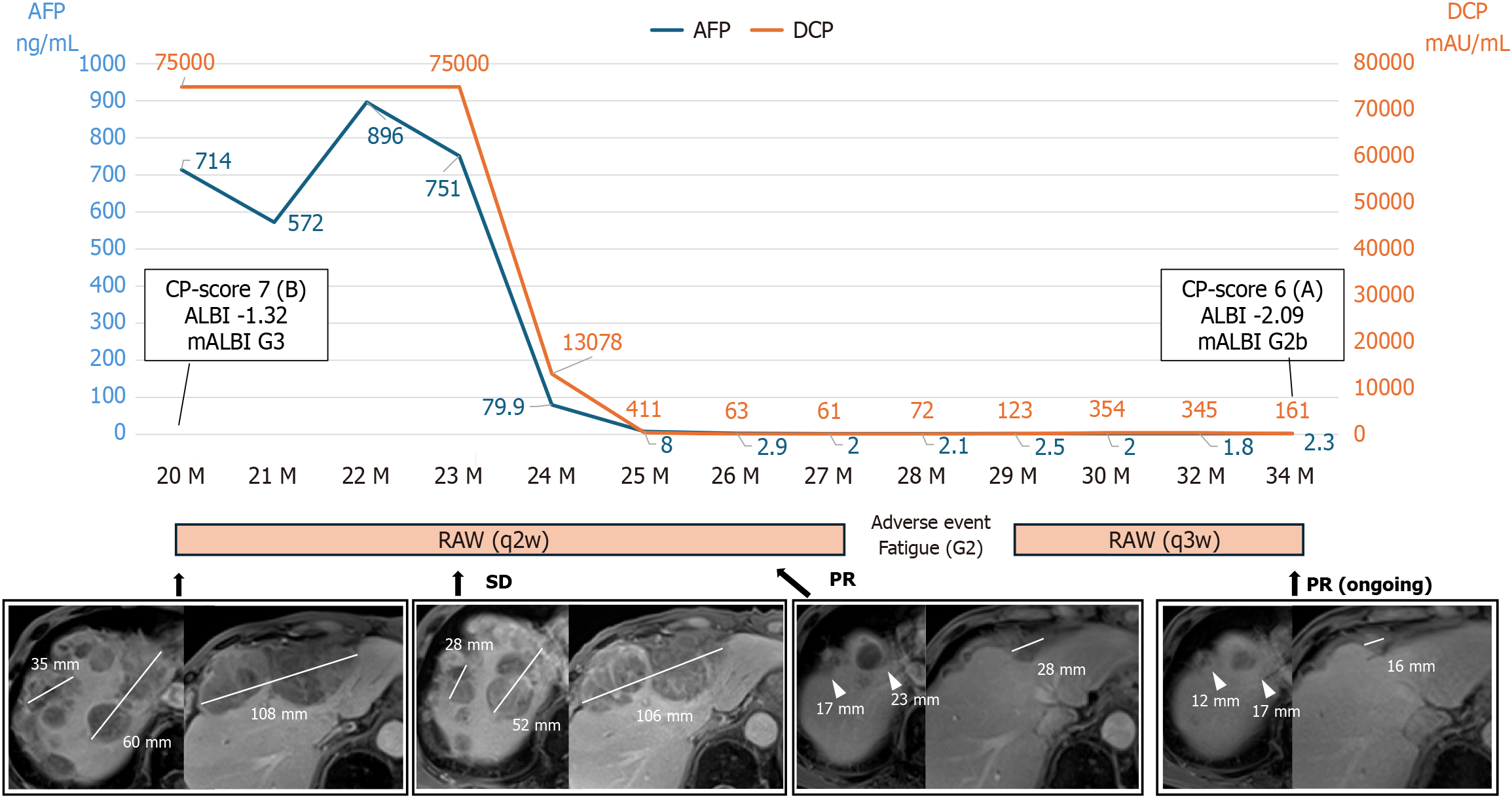
After 14 courses of RAM (7 months after initiation), the patient developed grade 2 fatigue, resulting in treatment interruption for approximately 2 months. RAM was subsequently resumed with a modified schedule of administration every 3 weeks. To date, treatment has been continued for more than 14 months, and a partial response has been maintained. Concomitantly, liver function improved to a Child-Pugh score of 6 (Class A), an ALBI score of -2.09 (mALBI grade 2b), and a performance status of 1 (Table 1).
| Parameter | Reference range | Atez/Bev | LEN | SOR | RAM | RAM (14 months) |
| WBC (μL) | 3300-8600 | 4900 | 3500 | 6100 | 5300 | 6000 |
| Hb (g/dL) | 13.7-16.8 | 14.5 | 13.7 | 13.9 | 13.4 | 11.8 |
| Plt (× 104/μL) | 15.8-34.8 | 19.2 | 22.7 | 22.3 | 29.2 | 14.4 |
| PT-INR | 1.00 | 1.14 | 1.23 | 1.07 | 1.19 | 0.98 |
| TBIL (mg/dL) | 0.4-1.5 | 1.0 | 1.1 | 0.9 | 1.3 | 0.7 |
| Alb (g/dL) | 4.1-5.1 | 3.8 | 3.2 | 3.4 | 2.6 | 3.3 |
| AST (U/L) | 13-30 | 33 | 37 | 47 | 65 | 42 |
| ALT (U/L) | 10-42 | 25 | 18 | 47 | 26 | 15 |
| ALP (U/L) | 38-113 | 145 | 96 | 153 | 194 | 137 |
| GGT (U/L) | 13-64 | 77 | 116 | 195 | 213 | 108 |
| Child-Pugh score | 5 | 7 | 6 | 7 | 6 | |
| ALBI score | -2.42 | -1.88 | -2.11 | -1.32 | -2.09 | |
| AFP (ng/mL) | < 7 | 99.5 | 260 | 692 | 714 | 2.3 |
| DCP (mAU/mL) | < 28 | 1897 | 34797 | 19206 | > 75000 | 161 |
We encountered a patient for whom RAM, administered as fourth-line therapy, yielded a remarkable effect. The patient showed slow progressive disease with Atez/Bev, intolerance to LEN, and progressive disease even with a low dose of sorafenib. However, fourth-line RAM was well tolerated, demonstrating efficacy and extended survival. This case involved an older adult patient with a Child-Pugh score of 7 (Class B), indicating decreased liver functional reserve. However, RAM was used safely.
The patient developed disseminated recurrence in the splenic hilum, leading to recurrence in the liver and on the liver surface. Recurrence in the splenic hilum was surgically treated after four courses of Atez/Bev; however, pathology findings showed that 90% of the HCC was viable, indicating that Atez/Bev was largely ineffective. Therefore, it was deemed highly likely that the subsequent recurrent lesions shared similar characteristics with HCC. The patient did not respond further to Atez/Bev. Therefore, we did not combine durvalumab and tremelimumab or ipilimumab and nivolumab as second-line therapy. Moreover, out of concern for immune-related adverse events associated with these agents, we chose to administer a TKI. Treatment with LEN markedly decreased tumor marker levels, suggesting that treatment may have been effective if tolerability had not been an issue. However, sorafenib was ineffective, possibly because of the low dose used.
Vascular endothelial growth factor (VEGF)-A, released by tumor cells and vascular endothelial cells at the tumor site, activates and recruits numerous types of inhibitory immune cells, including tumor-associated macrophages, regulatory T cells, and myeloid-derived suppressor cells, creating an immune-inhibitory microenvironment that favors tumor immune escape[3]. Bev inhibits VEGF-A, thereby improving the immunosuppressive microenvironment and enhancing the therapeutic effect when used in combination with immune checkpoint inhibitors. VEGF-A is also a central regulator of tumor angiogenesis, endothelial proliferation, permeability, and survival. VEGF-A binds with high affinity to two structurally similar tyrosine kinase receptors, VEGFR-1 and VEGFR-2, both of which are expressed in the tumor vasculature[4]. Thus, the inhibition of angiogenesis by Bev also has antitumor effects. Additionally, LEN and sorafenib exert antitumor effects by inhibiting multiple tyrosine kinases involved in angiogenesis in addition to VEGFR-1 and VEGFR-2, including PDGFRβ. LEN has a stronger affinity for receptors than sorafenib[5,6]. Yang et al[7] reported that Bev inhibition of VEGF-A leads to a compensatory increase in VEGF-C and VEGF-D, which act on VEGFR-2, resulting in reactivation of VEGFR-2-mediated signaling and potentially promoting tumor progression. RAM is a monoclonal antibody that acts specifically on VEGFR-2[4]; in this case, RAM was considered because enhanced VEGFR-2 signaling is the main driver of tumor growth. This was further supported by the observed decrease in tumor markers with LEN treatment, which strongly inhibited VEGFR-1 and VEGFR-2.
Approximately 10 months passed between the final administration of Atez/Bev and RAM administration. Atez’s half-life is reported to be 27 days[8]; therefore, we deemed it unlikely that an enhanced therapeutic effect owing to a pseudo-combination effect with Atez at the time of RAM administration applied. Regarding the therapeutic effect of RAM after Bev, Shimose et al[9] reported that RAM after Atez/Bev treatment had better progression-free survival than other combinations. These results support the model in which RAM is effective after Bev because of increased VEGFR-2 signaling[7].
AFP-high tumors exhibit a distinct phenotype characterized by poor differentiation, enrichment of progenitor features, and enhanced proliferation. Montal et al[10] reported that, VEGF-B is overexpressed and preferentially occupies VEGFR-1 in AFP-high HCC, resulting in a decreased decoy function of VEGFR-1 and a relative increase in VEGF-A binding and signaling through VEGFR-2.
Therefore, in addition to the compensatory increase in VEGF-C/D associated with resistance to Atez/Bev therapy, a phenotypic shift in the tumor to an AFP-high phenotype may occur. Against the background of increased VEGF-B, it is possible that VEGFR-2 signaling, re-induced by VEGF-A, is further enhanced. While VEGFR-1 has a high affinity for VEGF-A, its kinase activity is relatively weak and it primarily functions as a regulatory receptor that modulates VEGFR-2–mediated angiogenic signaling. Under such circumstances, where VEGFR-2–dependent angiogenesis becomes predominant, RAM acts effectively through direct inhibition of VEGFR-2.
In conclusion, in patients with high AFP levels following Atez/Bev treatment, the VEGFR-2 pathway may be activated. For cases in which tolerability to TKIs such as LEN, which also has strong VEGFR-2 inhibitory effects, cannot be ensured, there is value in proactively considering RAM as a therapeutic option.
| 1. | Ikeda M, Morizane C, Ueno M, Okusaka T, Ishii H, Furuse J. Systemic therapy for hepatocellular carcinoma, from the early to the advanced stage: a Japanese perspective. Jpn J Clin Oncol. 2025;55:465-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 2. | Zhu AX, Kang YK, Yen CJ, Finn RS, Galle PR, Llovet JM, Assenat E, Brandi G, Pracht M, Lim HY, Rau KM, Motomura K, Ohno I, Merle P, Daniele B, Shin DB, Gerken G, Borg C, Hiriart JB, Okusaka T, Morimoto M, Hsu Y, Abada PB, Kudo M; REACH-2 study investigators. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased α-fetoprotein concentrations (REACH-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20:282-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1404] [Cited by in RCA: 1317] [Article Influence: 188.1] [Reference Citation Analysis (6)] |
| 3. | Kudo M. Immuno-Oncology Therapy for Hepatocellular Carcinoma: Current Status and Ongoing Trials. Liver Cancer. 2019;8:221-238. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 54] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 4. | Spratlin JL, Cohen RB, Eadens M, Gore L, Camidge DR, Diab S, Leong S, O'Bryant C, Chow LQ, Serkova NJ, Meropol NJ, Lewis NL, Chiorean EG, Fox F, Youssoufian H, Rowinsky EK, Eckhardt SG. Phase I pharmacologic and biologic study of ramucirumab (IMC-1121B), a fully human immunoglobulin G1 monoclonal antibody targeting the vascular endothelial growth factor receptor-2. J Clin Oncol. 2010;28:780-787. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 468] [Cited by in RCA: 456] [Article Influence: 28.5] [Reference Citation Analysis (3)] |
| 5. | Yamamoto Y, Matsui J, Matsushima T, Obaishi H, Miyazaki K, Nakamura K, Tohyama O, Semba T, Yamaguchi A, Hoshi SS, Mimura F, Haneda T, Fukuda Y, Kamata JI, Takahashi K, Matsukura M, Wakabayashi T, Asada M, Nomoto KI, Watanabe T, Dezso Z, Yoshimatsu K, Funahashi Y, Tsuruoka A. Lenvatinib, an angiogenesis inhibitor targeting VEGFR/FGFR, shows broad antitumor activity in human tumor xenograft models associated with microvessel density and pericyte coverage. Vasc Cell. 2014;6:18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 435] [Cited by in RCA: 405] [Article Influence: 33.8] [Reference Citation Analysis (4)] |
| 6. | Okamoto K, Ikemori-Kawada M, Jestel A, von König K, Funahashi Y, Matsushima T, Tsuruoka A, Inoue A, Matsui J. Distinct binding mode of multikinase inhibitor lenvatinib revealed by biochemical characterization. ACS Med Chem Lett. 2015;6:89-94. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 246] [Cited by in RCA: 210] [Article Influence: 19.1] [Reference Citation Analysis (4)] |
| 7. | Yang Z, Suda G, Maehara O, Ohara M, Yoda T, Sasaki T, Kohya R, Yoshida S, Hosoda S, Tokuchi Y, Kitagataya T, Suzuki K, Kawagishi N, Nakai M, Sho T, Natsuizaka M, Ogawa K, Ohnishi S, Sakamoto N. Changes in Serum Growth Factors during Resistance to Atezolizumab Plus Bevacizumab Treatment in Patients with Unresectable Hepatocellular Carcinoma. Cancers (Basel). 2023;15:593. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 8. | Stroh M, Winter H, Marchand M, Claret L, Eppler S, Ruppel J, Abidoye O, Teng SL, Lin WT, Dayog S, Bruno R, Jin J, Girish S. Clinical Pharmacokinetics and Pharmacodynamics of Atezolizumab in Metastatic Urothelial Carcinoma. Clin Pharmacol Ther. 2017;102:305-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 129] [Article Influence: 14.3] [Reference Citation Analysis (6)] |
| 9. | Shimose S, Sugimoto R, Hiraoka A, Tanaka M, Iwamoto H, Tanaka Y, Tada F, Ohama H, Niizeki T, Shirono T, Moriyama E, Noda Y, Kamachi N, Nakano M, Kuromatsu R, Koga H, Kawaguchi T. Significance of ramucirumab following atezolizumab plus bevacizumab therapy for hepatocellular carcinoma using real-world data. Hepatol Res. 2023;53:116-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 10. | Montal R, Andreu-Oller C, Bassaganyas L, Esteban-Fabró R, Moran S, Montironi C, Moeini A, Pinyol R, Peix J, Cabellos L, Villanueva A, Sia D, Mazzaferro V, Esteller M, Llovet JM. Molecular portrait of high alpha-fetoprotein in hepatocellular carcinoma: implications for biomarker-driven clinical trials. Br J Cancer. 2019;121:340-343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 76] [Article Influence: 10.9] [Reference Citation Analysis (0)] |