Published online May 16, 2026. doi: 10.12998/wjcc.v14.i14.117795
Revised: February 16, 2026
Accepted: April 13, 2026
Published online: May 16, 2026
Processing time: 132 Days and 17.5 Hours
Sleeve gastrectomy (SG) is a widely performed bariatric procedure known for its technical simplicity, safety profile, and effectiveness in long-term weight re
We present a 47-year-old female with body mass index 52 kg/m2, qualified for metabolic bariatric surgery and selected for SG as a first-stage restrictive pro
Three-port robotic SG represents a safe and viable modification of conventional port configurations. Although early results are encouraging, further studies involving larger patient groups are necessary to confirm the reproducibility and broader clinical benefits of this technique.
Core Tip: This case report describes a three-port robotic sleeve gastrectomy in an obese patient (body mass index > 50 kg/m2), demonstrating technical feasibility and early postoperative safety. Using the da Vinci robotic system, the procedure was completed efficiently without a liver retractor, and the patient resumed oral intake within hours and was discharged the next day. The report highlights the potential of reduced-port robotic approaches to minimize invasiveness, improve cosmesis, and maintain operative efficiency, providing a foundation for future studies to evaluate reproducibility and broader clinical benefits in larger patient populations.
- Citation: Walędziak M, Różańska-Walędziak A. Three-port robotic sleeve gastrectomy-another step forward: A case report. World J Clin Cases 2026; 14(14): 117795
- URL: https://www.wjgnet.com/2307-8960/full/v14/i14/117795.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i14.117795
Sleeve gastrectomy (SG), aimed at treating obesity, is one of the most popular bariatric procedures worldwide[1]. It is considered one of the least complex bariatric operations, offering high reproducibility and a relatively low risk of complications as its major advantages[1-3]. Studies indicate that SG results in substantial long-term weight loss, im
Long-term outcomes demonstrate that patients maintain significant weight loss for several years after surgery, which translates into a reduced risk of complications associated with obesity, such as type 2 diabetes, hypertension, and cardiovascular diseases[6,7]. Additionally, many individuals report improvement in their mental well-being and daily life functioning[8].
Originally performed as an open procedure, SG was soon adapted to a laparoscopic approach, which remained the standard technique for several decades. In recent years, SG has increasingly been performed using the da Vinci robotic system, which provides greater precision and minimizes tissue trauma compared to conventional laparoscopy[9]. The robotic system enables surgeons to achieve an enhanced visualization of the surgical field, facilitating more accurate and controlled resection[10]. Initially, SG was performed using a five-port configuration; however, it was later observed that a high number of ports may not be necessary. The three-port laparoscopic SG has gained popularity in bariatric surgery due to its advantages, including reduced invasiveness, faster recovery, and improved cosmetic outcomes. This technique minimizes invasiveness, resulting in shorter recovery times and smaller scars[11,12]. This technique minimizes invasiveness and may contribute to shorter recovery times and smaller scars, which are particularly relevant from both functional and aesthetic perspectives. The first report of the use of only three trocars during robotic SG was published in 2024[13]. Recent studies indicate that this approach is beneficial for optimizing surgical tool usage and reducing overall operation time[14].
Three-port laparoscopic SG has been successfully implemented and routinely performed at the main author’s bariatric center for several years, and the experience gained from over 500 surgeries has allowed for the adaptation of the technique to the robotic platform.
A 47-year-old female patient with body mass index (BMI) 52 kg/m2 was qualified for metabolic bariatric surgery.
The patient denies symptoms associated with gastroesophageal reflux. Preoperative gastroscopy did not reveal any inflammation of the esophageal or gastric mucosa. No symptoms of hiatal hernia were detected during endoscopy. At the time of qualification, the patient did not express her consent to undergo surgery limiting the absorption of nutritional substances. However, she did consent to two-stage treatment, if necessary.
The patient had a history of two cesarean sections with no associated medical problems. Status after two cesarean sections in 2013 and 2015, respectively. The patient was not treated for chronic diseases, denies taking medications on a per
Unrelated to the current medical condition.
The patient is well-groomed, alert, and oriented to person, place, and time, in no acute distress. Posture and mobility are limited secondary to marked adiposity. Vital signs are stable; blood pressure and heart rate within acceptable range for age, respiratory rate regular, afebrile, oxygen saturation normal on room air.
Skin warm, intact, without active lesions; striae and intertriginous areas without signs of acute inflammation. Head normocephalic and atraumatic. Pupils equal, round, reactive to light; extraocular movements intact; sclerae anicteric. Oropharynx clear, mucous membranes moist. Neck supple, no palpable lymphadenopathy; thyroid not enlarged; ass
Chest expansion symmetrical; breath sounds vesicular bilaterally without wheezes or crackles, though auscultation partially limited by body habitus. Heart sounds regular, no audible murmurs; examination limited by thick chest wall. Peripheral pulses palpable; no significant peripheral edema.
Abdomen markedly enlarged, soft, non-tender; no palpable masses or organomegaly, though palpation limited due to abdominal adiposity. Bowel sounds present. Musculoskeletal examination reveals preserved joint range of motion with mild functional limitation related to body mass; muscle strength grossly preserved. Gait slow but stable. Neurologically intact; cranial nerves grossly normal, no focal deficits. Mood and affect appropriate.
No significant deviations. Within reference ranges.
Within reference ranges.
Overall findings consistent class III obesity (BMI > 52 kg/m2) without acute abnormalities on physical examination.
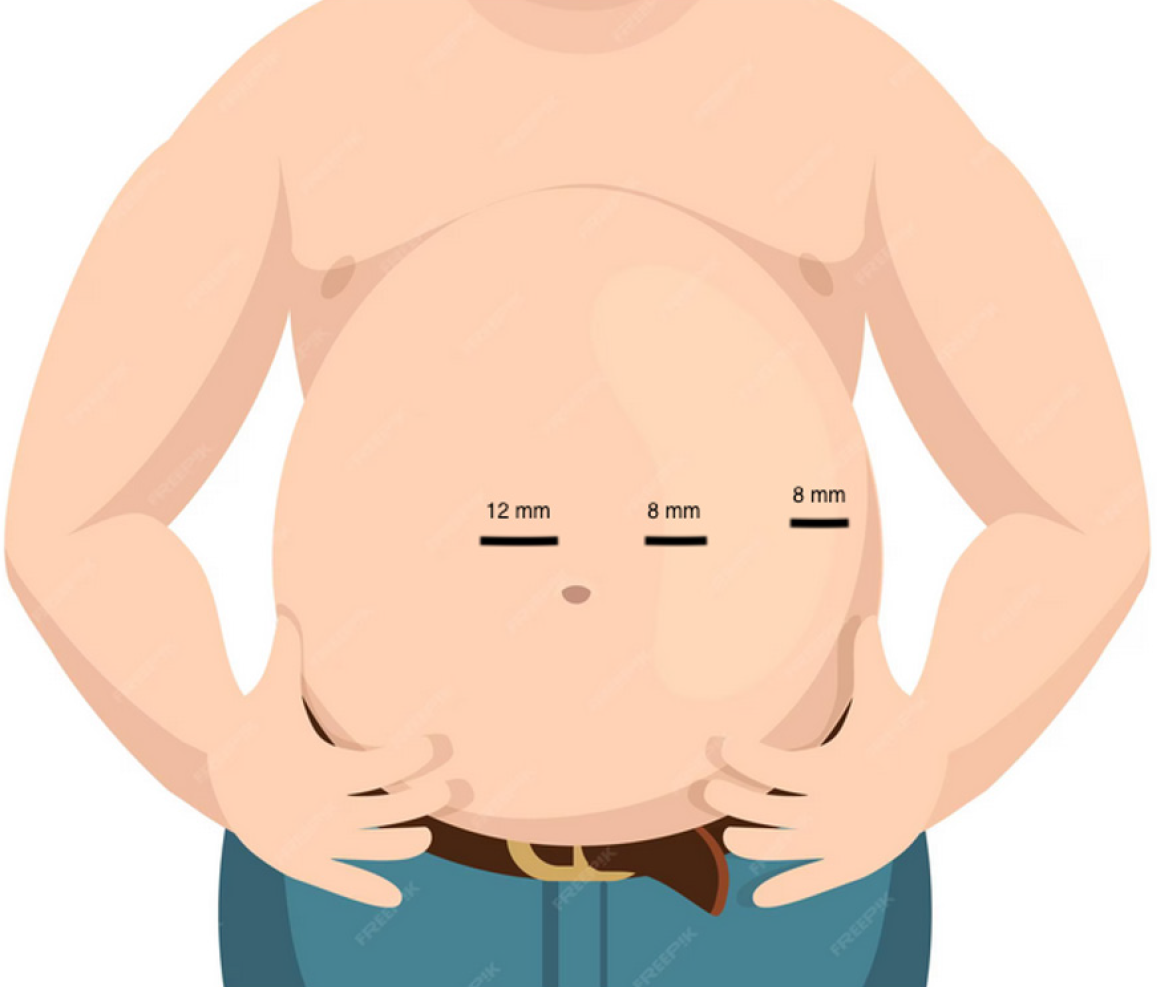
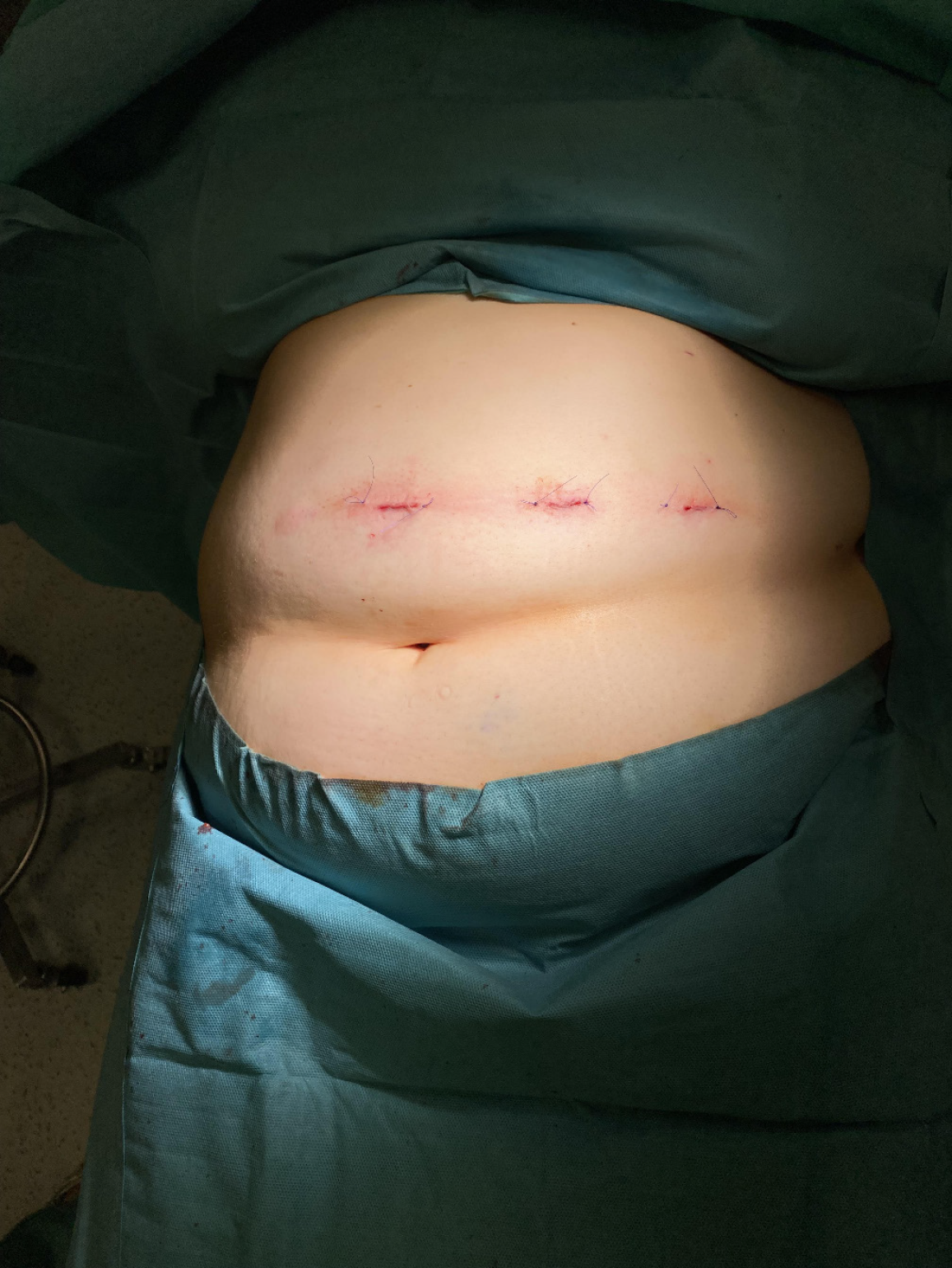
Three-port robotic SG (Video). The patient was selected for SG as the first-stage procedure based on BMI > 50 kg/m2, absence of reflux symptoms, and preference for a restrictive technique. The three-port robotic SG was performed. The patient was placed in a supine position. Both arms were secured along the body. The table was positioned in a 12°-15° reverse Trendelenburg and tilted 2°-3° to the right. The arrangement of the operating room is shown in the Figure 1. Pneumoperitoneum was established at 15 mmHg using a Veress needle at Palmer’s point. Three trocars (1 mm × 12 mm and 2 mm × 8 mm) were inserted in the typical location-at transverse plane 15 cm inferior from xiphoid process. First 8 mm port was placed 3 cm left of the midline, 12 mm port right lateral and second 8mm left lateral from first one as presented in Figure 2. The operation was started with Cadiere forceps in the left hand and Vessel Sealer in right hand during the dissection time of the surgery. For the stapling time, the robotic stapler was put in the right arm. No liver retractor was used.
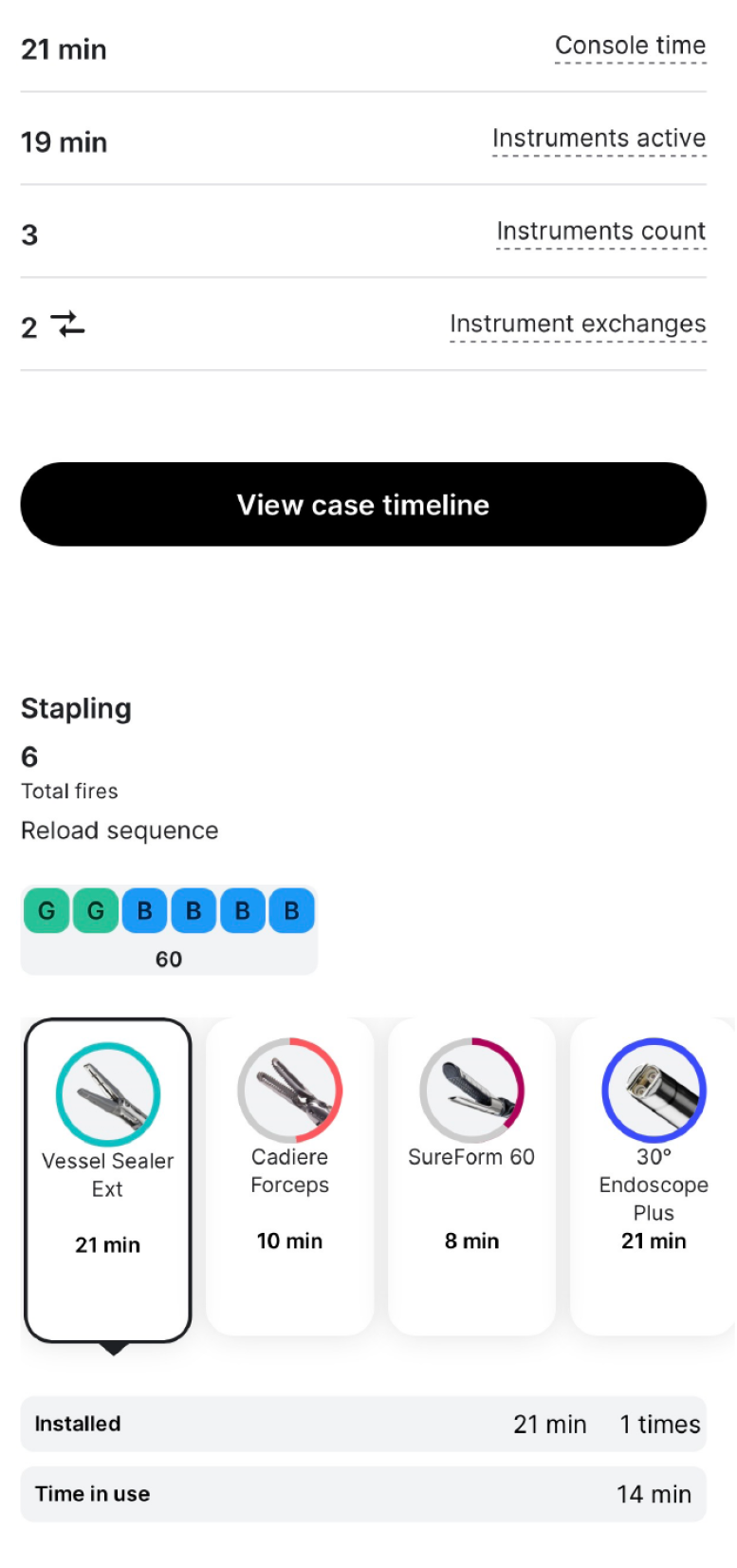
The greater curvature of the stomach was mobilized up to 5-7 cm from the pylorus by dividing the gastrocolic ligament and entering the lesser sac. A 36 F bougie was placed, secured at the lesser curvature, and while calibrating the future stomach, the excess was resected from the side of the greater curvature with staplers (SureForm 60 with two green reloads for the gastric body and four blue reloads till the level of the fundus). The staple line was checked with a methylene blue test to ensure integrity, and hemostasis was confirmed. The resected gastric specimen was extracted through the 12 mm incision. No drain was left during the surgery. The console time was 21 minutes, instruments were active for 19 minutes and there were 2 instrument exchanges needed during the surgery as presented in Figure 3.
The postoperative course was uneventful. The patient resumed oral liquids 2 hours after surgery and was discharged home on postoperative day one, approximately 24 hours after the operation. During the 6-month follow-up, no disturbing symptoms related to the surgery were observed. No readmission. The patient continued to take no medications due to her comorbidities. She reported a weight loss of approximately 17%.
Robotic SG is widely used bariatric procedures due to its high precision and minimal tissue trauma compared to traditional laparoscopic methods. The robotic system provides a three-dimensional view of the surgical field, resulting in improved accuracy and control. By utilizing smaller incisions, patients experience less postoperative pain and faster recovery, often allowing early discharge. Despite increased costs, robotic approach may represent an added value in more complex cases such as BMI > 50 kg/m2 patients[15].
This single-case report demonstrates the technical feasibility of performing robotic SG using a three-port configuration in a super-obese patient. However, given that this experience is limited to one case, no definitive conclusions can be drawn regarding safety, efficiency, superiority over conventional approaches, or comparative complication rates. Any potential benefits, such as reduced postoperative discomfort or improved cosmetic outcomes, remain hypothetical and would require validation in larger comparative studies.
| 1. | Schauer PR, Bhatt DL, Kirwan JP, Wolski K, Aminian A, Brethauer SA, Navaneethan SD, Singh RP, Pothier CE, Nissen SE, Kashyap SR; STAMPEDE Investigators. Bariatric Surgery versus Intensive Medical Therapy for Diabetes - 5-Year Outcomes. N Engl J Med. 2017;376:641-651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2384] [Cited by in RCA: 2069] [Article Influence: 229.9] [Reference Citation Analysis (5)] |
| 2. | Debel N, Snijkers E, van de Velde M, Joshi GP, Sauter AR, Freys S, Pogatzki-Zahn E, Dewinter G; on behalf of the PROSPECT working Group of the European Society of Regional Anaesthesia and Pain Therapy (ESRA). Pain management for laparoscopic sleeve gastrectomy: An update of the systematic review and procedure-specific postoperative pain management (PROSPECT) recommendations. Eur J Anaesthesiol. 2026;43:19-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 3. | Kowalewski PK, Olszewski R, Walędziak MS, Janik MR, Kwiatkowski A, Gałązka-Świderek N, Cichoń K, Brągoszewski J, Paśnik K. Long-Term Outcomes of Laparoscopic Sleeve Gastrectomy-a Single-Center, Retrospective Study. Obes Surg. 2018;28:130-134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 76] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 4. | Qi L, Guo Y, Liu CQ, Huang ZP, Sheng Y, Zou DJ. Effects of bariatric surgery on glycemic and lipid metabolism, surgical complication and quality of life in adolescents with obesity: a systematic review and meta-analysis. Surg Obes Relat Dis. 2017;13:2037-2055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 5. | El-Masry H, Mahmoud MH, Mohamed BS, Elraggal D, Elkholy MH, Abokhozima A. The Nutritional Challenges Following Revisional Bariatric Surgery After Sleeve Gastrectomy: A Systematic Review and Meta Analysis. Obes Surg. 2025;35:5515-5530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 6. | Tripathi S, Ray AK, Sinha Y, Reid A, Noormohamed S. Shaping the Future of Restrictive Bariatric Surgery: Clinical, Economic, and Long-Term Perspectives on Endoscopic and Laparoscopic Sleeve. Obes Surg. 2025;35:5550-5555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 7. | Adams TD, Gress RE, Smith SC, Halverson RC, Simper SC, Rosamond WD, Lamonte MJ, Stroup AM, Hunt SC. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;357:753-761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2072] [Cited by in RCA: 1698] [Article Influence: 89.4] [Reference Citation Analysis (0)] |
| 8. | GBD 2021 Adolescent BMI Collaborators. Global, regional, and national prevalence of child and adolescent overweight and obesity, 1990-2021, with forecasts to 2050: a forecasting study for the Global Burden of Disease Study 2021. Lancet. 2025;405:785-812. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 252] [Cited by in RCA: 227] [Article Influence: 227.0] [Reference Citation Analysis (0)] |
| 9. | Zhao S, Fu Y, Zhou J, Sun L, Li R, Tian Z, Cheng Y, Wang J, Wang W, Wang D. Comparing the Efficacy of Robotic Versus Laparoscopic Sleeve Gastrectomy: A Systematic Review and Meta-Analysis. Obes Surg. 2024;34:3493-3505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 10. | Shen D, Ye H, Wang Y, Ji Y, Zhan X, Zhu J, Li W. Comparison of short-term outcomes between laparoscopic greater curvature plication and laparoscopic sleeve gastrectomy. Surg Endosc. 2013;27:2768-2774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 11. | Dunford G, Philip S, Kole K. Three-Port Laparoscopic Sleeve Gastrectomy: A Novel Technical Modification. Surg Laparosc Endosc Percutan Tech. 2016;26:e174-e177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Gutiérrez-Ramírez L, Morandeira-Rivas A, Medina-Benítez E, Arias-Arias Á, Moreno-Sanz C. Reduced Port Laparoscopic Sleeve Gastrectomy: A Systematic Review and Meta-analysis. Obes Surg. 2024;34:4519-4530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Pascotto B, Azagra JS, González González L, Goergen M. Real Robotic 3-Arm Sleeve Gastrectomy. Obes Surg. 2024;34:295-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Arru L, Azagra JS, Goergen M, de Blasi V, de Magistris L, Facy O. [Three-port laparoscopic sleeve gastrectomy: feasibility and short outcomes in 25 consecutives super-obese patients]. Cir Esp. 2013;91:294-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 15. | Marincola G, Procopio PF, Pennestrì F, Gallucci P, Voloudakis N, Ciccoritti L, Greco F, Salvi G, Prioli F, De Crea C, Raffaelli M. Robot-assisted vs laparoscopic bariatric procedures in super-obese patients: clinical and economic outcomes. J Robot Surg. 2024;18:34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |