Published online Apr 6, 2026. doi: 10.12998/wjcc.v14.i10.118630
Revised: February 16, 2026
Accepted: March 10, 2026
Published online: April 6, 2026
Processing time: 84 Days and 11.5 Hours
Blau syndrome (BS) is a rare sporadic or autosomal dominant multi-system inf
We describe two sporadic cases with different clinical profiles. Case 1: A 5-year-old male presented with a history of erythematous skin lesions all over the body since the age of 6 months and recurrent fever and knee and wrist joint pains for the past year, associated with progressive diminution of vision. He had dry, scaly skin, malar erythema and bilaterally swollen wrist, ankle and knee joints, bilateral band-shaped keratopathy, and bilateral post-uveitis cataracts. Case 2: An 11-year-old male previously diagnosed with polyarticular juvenile idiopathic arthritis, tubulointerstitial nephritis, seizure disorder, restrictive lung disease, and multidrug-resistant pulmonary tuberculosis, presented with bilateral granulomatous uveitis. Fundus examination of the right eye showed fibrovascular proliferation over the disc secondary to retinal vasculitis and exudative shallow retinal detachment. Both patients were diagnosed with BS and treated with corticosteroids and ada
The diagnosis of BS is important to consider in very young children with arthritis and uveitis. Timely interdepartmental referral and collaboration is prudent. Prompt multidisciplinary management is required to prevent sig
Core Tip: Blau syndrome (BS) manifests as uveitis, arthritis, and skin rash, and although rare, is an important consideration in children with such manifestations. It may pose a diagnostic dilemma in the presence of systemic comorbidities. A multidisciplinary approach leads to management of this condition with satisfactory outcomes. The present series describes the presentation, diagnosis, and management of two sporadic cases of BS.
- Citation: Raj A, Nishant P, Kumar R, Sharma A, Singh P, Patra PK, Sinha S. Blau syndrome at a tertiary care center of Eastern India: Two case reports and review of literature. World J Clin Cases 2026; 14(10): 118630
- URL: https://www.wjgnet.com/2307-8960/full/v14/i10/118630.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i10.118630
Blau syndrome (BS), also known as juvenile systemic granulomatosis, is a monogenic disease resulting from gain-of-function mutations in the pattern recognition receptor nucleotide binding oligomerization domain containing 2 (NOD2) and is phenotypically characterized by the triad of granulomatous polyarthritis, dermatitis, and uveitis[1]. Skin rash is the first symptom to appear, usually in infants. Between the ages of 2 and 4 years, a boggy polyarthritis is observed. Finally, uveitis develops in 60%-80% of patients at about 48 months of age[2]. BS can mimic juvenile idiopathic arthritis (JIA) as
Previous reports have noted a paucity of literature on the ocular manifestations of BS from the Asian Indian population[3]. Most of the reports are accounts of individual cases or small case series[4-14] (Table 1). Here, we present two sporadic cases of BS with varied clinical profiles from a tertiary care center in East India with satisfactory outcomes obtained by a multidisciplinary approach, highlighting its varied presentation, clinical course, and management.
| Ref. | Region | Number of cases | Unique features | Disease progression pattern | Mainstay immunosuppressive treatment | Remark |
| Jain et al[4], 2018 | East | 1 | Vitreous hemorrhage without vascular abnormalities, hepato-splenomegaly | Therapeutic vitrectomy led to remission of ocular disease despite waxing and waning systemic disease | Oral steroids and methotrexate | Novel mutation in helical domain 2 of NOD2 in sporadic BS discovered |
| Naik et al[5], 2018 | South | 1 | Child born out of second-degree consanguineous marriage but no genetic mutations typical of BS found | Recurrent uveitis and arthritis, but maintaining normal visual acuity since remission | Adalimumab monotherapy (after failure of topical steroids, systemic steroids, methotrexate, and infliximab) | First case of sporadic BS in India with long follow-up on adalimumab monotherapy |
| Janarthanan et al[6], 2019 | South | 3 | First molecularly confirmed report of familial BS from India | Recurrent episodes with residual joint deformities | Systemic steroids, methotrexate, topical non-steroidal anti-inflammatory drugs | |
| Babu et al[7], 2021 | South | 7 | Keratoconjunctivitis sicca, conjunctival granulomas, subepithelial corneal opacities, subretinal granuloma | Recurrent episodes with but with good visual prognosis | Oral steroids, methotrexate, mycophenolate mofetil | Largest case series at that time |
| Babu et al[8], 2025 | South | 10 | Almost 4/5ths of the patients had no inflammation at last follow-up | Oral steroids, methotrexate, mycophenolate mofetil, adalimumab, tofacitinib, infliximab and combinations of biologic agents including canakinumab | ||
| Baisya et al[9], 2023 | South | 5 | Fever, lymphadenopathy, serositis, organomegaly, subcutaneous nodule | Patients responded well (details not mentioned) | Oral steroids, methotrexate | Familial BS in 3 families |
| Jindal et al[10], 2021 | North | 1 | Disseminated granulomas in liver and kidneys. Unique sequence and timing of organ system involvement: Symmetric polyarthritis at 3 years of age, granulomatous uveitis at 13 years of age, disseminated granulomas in liver and kidneys at 21 years of age | Persistent splenomegaly and thrombocytopenia, leucopenia, and anemia despite treatment and follow-up of 20 years | Adalimumab | |
| Banday et al[11], 2022 | North | 2 | No uveitis at presentation of child | Remained well at follow-up | Methotrexate | Grandmother, mother and child affected |
| Kumrah et al[12], 2022 | North | 11 | Chronic kidney disease, nephrotic range proteinuria | Progressive and refractory to treatment, leading to residual joint deformities | Methotrexate, adalimumab, azathioprine, mycophenolate mofetil, leflunomide | |
| Nagpal and Singh[13], 2024 | North | 1 | Good vision in operated and unoperated eye | oral corticosteroids and methotrexate | Biopsy refused. Genetic screening of family members not done due to constraints. | |
| Sharma et al[14], 2025 | North | 3 | Bilateral small vessel Retinal vasculitis in all cases | One of the cases had very late onset of uveitis at 28 years of age, without history of arthritis or dermatitis | Not reported | Retinal vasculitis reported as a rare phenotype of BS |
| Present report | East | 2 | One case had typical manifestations. The other had history of tubulointerstitial nephritis, seizure disorder, restrictive lung disease, and multi-drug-resistant tuberculosis, and developed uveitic sequelae post-cataract surgery, oral ulcers, retinal vasculitis and exudative shallow retinal detachment | Waxing and waning course of systemic and intraocular inflammation, however, with fair visual outcome but loss to follow up | Adalimumab with steroid therapy as initial bridge; intra-articular Triamcinolone acetate after withdrawal of methotrexate due to adverse effects | Detailed ocular examination and treatment course mentioned in detail. Ultrasonography of joints, biopsy and genetic testing refused due to constraints |
Case 1: A 5-year-old male child presented to the pediatric rheumatologist with fever and recurrent knee and wrist joint pains for the past year, which worsened in the past month. He also complained of gradually progressive diminution of vision with intermittent redness and pain in both eyes for the past week.
Case 2: An 11-year-old boy presented to the pediatric rheumatologist with fever on and off for the past 2 months and difficulty breathing for the past 10 days.
Case 1: The parents reported that the fever was low-grade and resolved on taking over-the-counter medicines, only to recur again without any specific interval. The joint pains were accompanied by mild swelling of the joints.
Case 2: The child’s mother informed that the child had had multiple similar episodes in the past but none in the pre
Case 1: The child’s father informed that the patient had had erythematous skin lesions all over the body since the age of 6 months.
Case 2: The parents reported that at 6 months of age, the child had bilateral nodular swelling of ankles and wrists, and at 1-year, bilateral swelling of knees and ankles. At 3 years of age, he developed high fever with cough associated with fast breathing. An acute episode of tubulointerstitial nephritis, seizure disorder, and restrictive lung disease was documented in the medical records. A synovial biopsy had been performed, and the child was diagnosed with polyarticular JIA. Oral prednisolone acetate (1 mg/kg body weight) was given intermittently for the same for 3 years. High-resolution computed tomography of the chest at follow-up had revealed consolidation with collapse and nodular infiltration and scarring suggestive of pulmonary TB. Culture of bronchoalveolar lavage grew multi-drug resistant TB. He received therapy for extrapulmonary TB for 2 years followed by oral methotrexate (10 mg/m²/dose) with folic acid 5 mg weekly and pre
Case 1 and Case 2: There was no significant family history of any other systemic illnesses, and no history of similar illness in grandparents, parents, or siblings. Both children had been delivered full term and had attained developmental mil
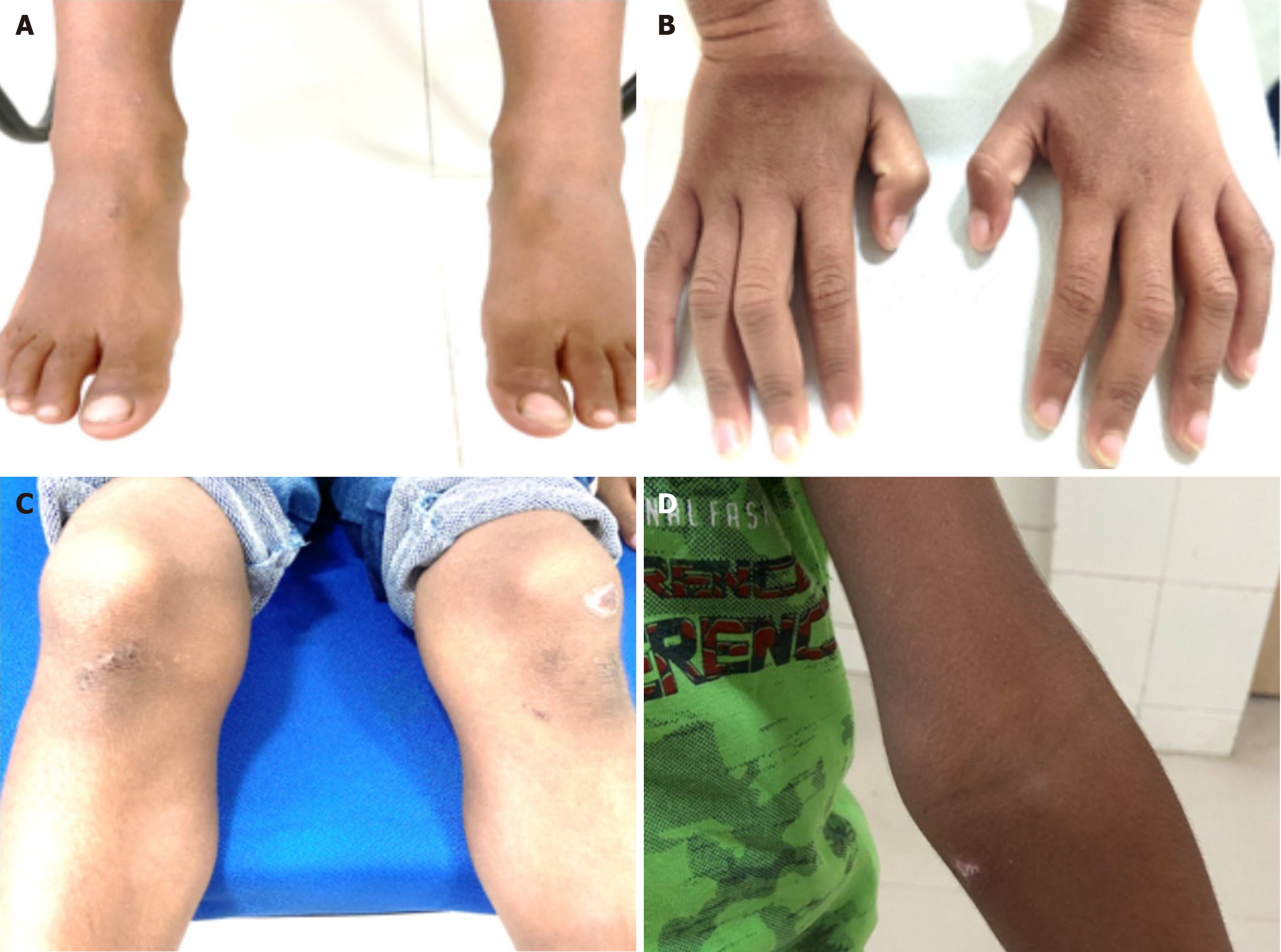
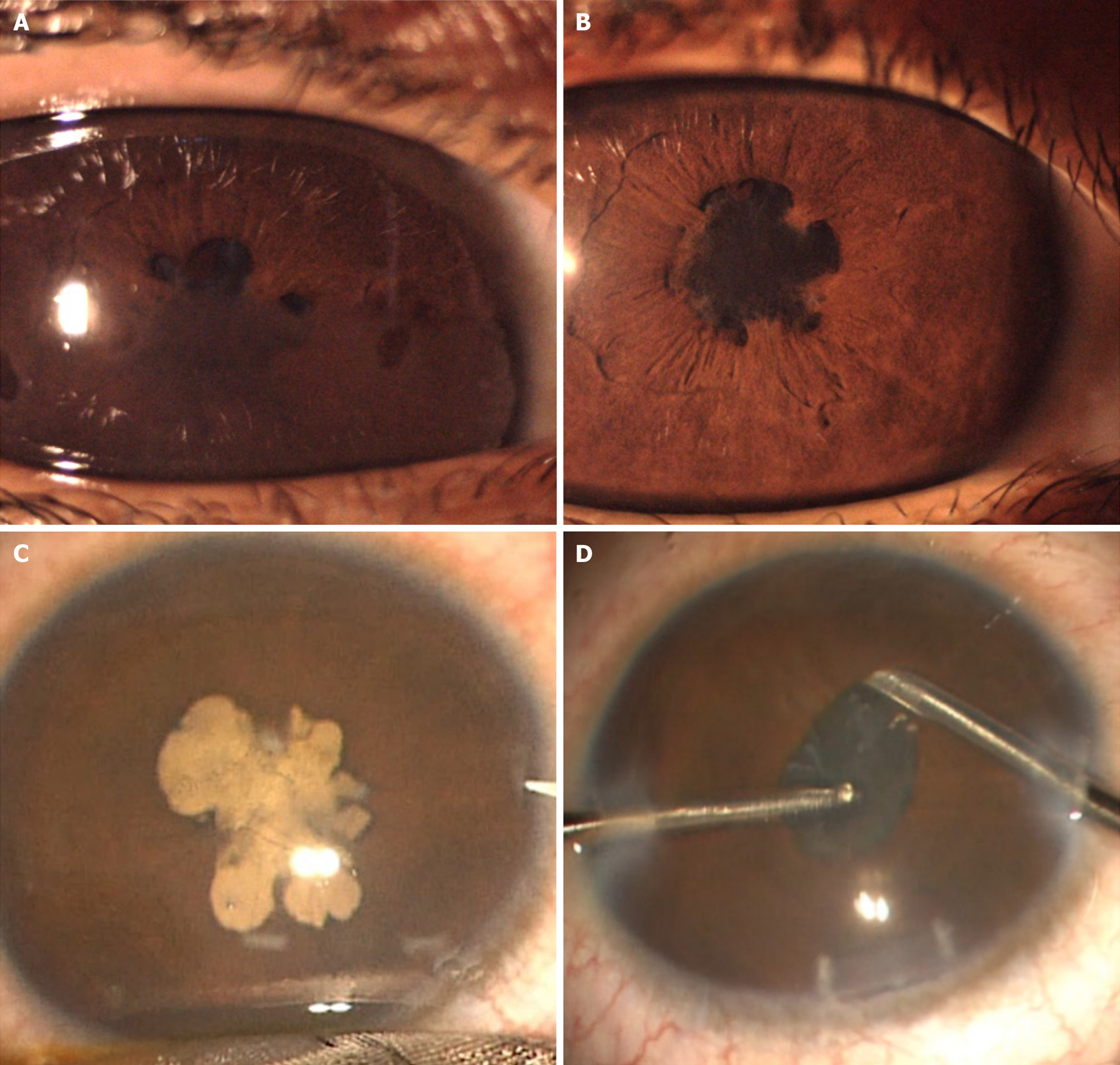
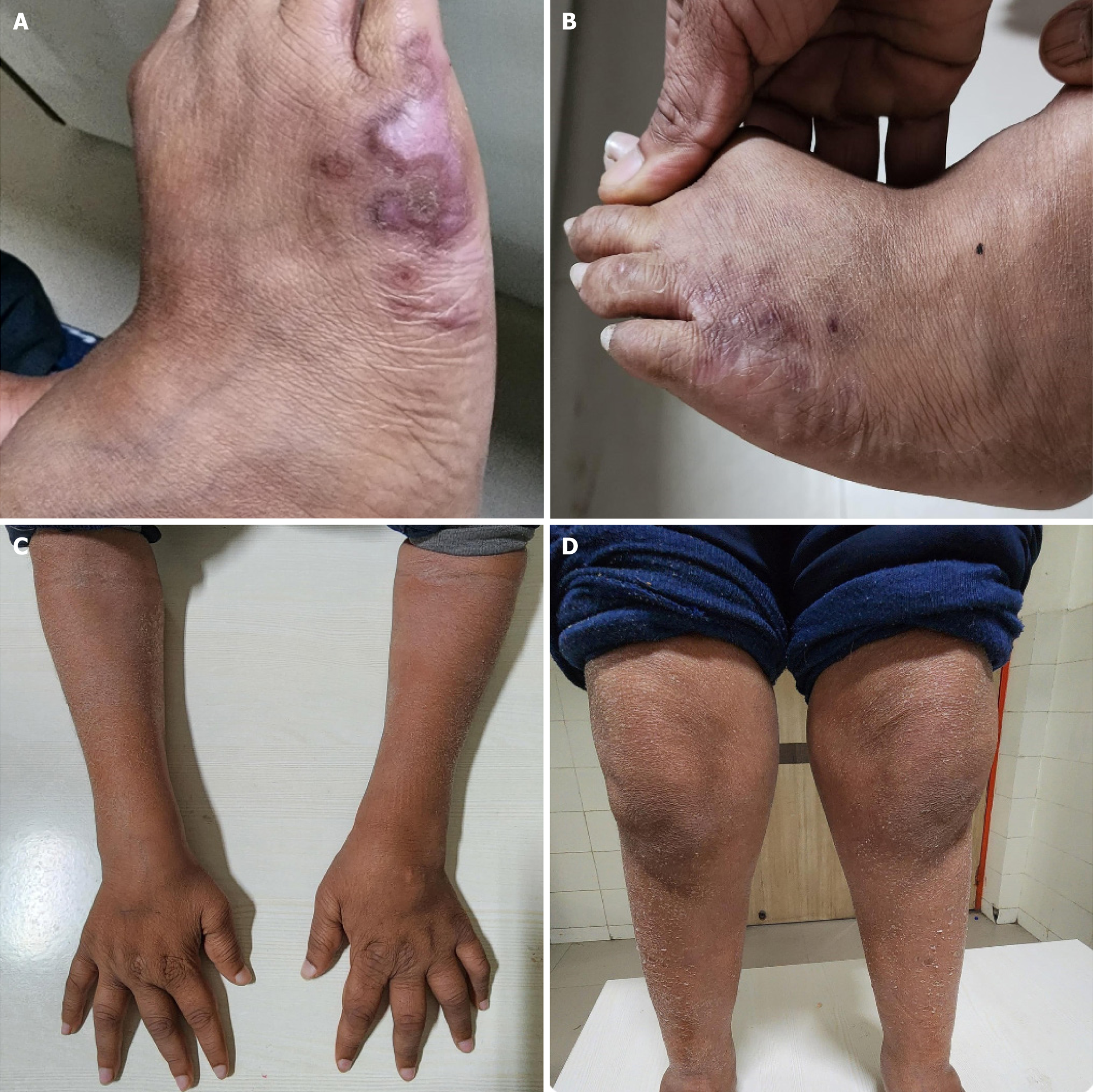
Case 1: Systemic examination showed dry, scaly skin, malar erythema. and bilaterally swollen but nontender wrist, ankle, and knee joints (Figure 1). Ophthalmic examination showed presenting uncorrected visual acuity (UCVA) of 6/30 OU using Cardiff’s cards at a working distance of 50 cm, not improving with refractive correction. Digitally, the intraocular pressures (IOP) seemed normal in both eyes. Ocular examination showed bilateral band-shaped keratopathy (BSK), 2+ anterior chamber cells, festooned pupils, posterior synechiae and complicated cataracts (Figure 2).
Case 2: The child looked ill with nasal flare, soft palpable bilateral axillary lymph nodes, and deformities of the hands and feet. On ophthalmic examination, UCVA in the right eye (oculus dexter [OD]) was 1/60, improving to 2/60 with pinhole, and OS 6/36, not improving with pinhole. The IOP by noncontact tonometry was 9 mmHg OD and 20 mmHg OS. OD had leucomatous corneal opacity with superficial vascularization, optic capture in the superior quadrant with one haptic in the anterior chamber, significant iris atrophy, posterior synechiae, and hyphema. Fundus examination OD showed hazy media, a vertical cup disc ratio of 0.4:1 with mild disc pallor and vitreous condensation. The peripheral fundus could not be visualized. OS showed occlusio pupillae, a patent peripheral iridectomy, and a flat bleb. The po
Case 1 and Case 2: Routine blood tests including total red blood cell and white blood cell count, erythrocyte sedi
Case 1: B-scan ultrasonography OU revealed bilateral vitritis with no evidence of retinal or choroidal detachment. Axial length was 22.14 mm in both eyes. The retina-choroid-sclera complex was within normal limits.
Case 2: B-scan ultrasonography OU revealed an anechoic vitreous cavity with no retinal or choroidal detachment. Axial length was 23.13 mm OD and 23.74 mm OS. The retina-choroid-sclera complex appeared thick, measuring 1.24 mm in OD and 1.16 mm in OS. Parents of both patients refused ultrasonography of the joints citing financial constraints.
Multidisciplinary expert consultation between the ophthalmologists and pediatric rheumatologist considered the dermatological, ophthalmic, and arthritic manifestations in both patients. A provisional diagnosis of BS was made. Differential diagnoses were Behçet’s Disease (systemic autoimmune vasculitis associated with HLA-B*51 allele)-considered less likely on the basis of younger age, absence of genital ulcers and lower grade of intraocular inflammation in our patients-and Yao syndrome (systemic autoinflammatory disease with fever, joint pain, and skin rashes mimicking BS except for a variant NOD2 gene mutation), excluded on the basis of the younger age of our patients, absence of gastrointestinal com
A clinical diagnosis of BS was made in both patients. Parents and siblings of both patients were examined and showed no evidence of any autoimmune disorder; hence, the inheritance was deemed to be sporadic in both cases.
The child was initiated on 1% prednisolone acetate eye drops 4 times/day and 1% atropine sulphate eye drops 3 times/day. The pediatric rheumatologist prescribed intravenous methylprednisolone 400 mg in 250 mL normal saline for 3 days, subcutaneous injection methotrexate 7.5 mg (10 mg/m²/dose) weekly, oral prednisolone acetate 25 mg once daily post meal, folic acid 5 mg once a week and naproxen 5 mg/kg of body weight twice daily. Bilateral ethylene-diamine-tetra-acetate (EDTA) chelation was carried out for BSK after control of intraocular inflammation, after which he was disc
The child was admitted to the pediatric rheumatology ward and administered subcutaneous injections of adalimumab 0.4 mL (40 mg/0.8 mL) weekly. He was followed up after 1 month and started on oral prednisolone 40 mg once daily (1 mg/kg/day) to be tapered weekly, syrup levetiracetam 4 mL twice daily (100 mg/mL, 40 mg/kg/day), and vitamin D3 0.25 mg once daily.
The patient was started on topical 1% prednisolone acetate and 2% homatropine eye drops OU. At 2 weeks, UCVA improved to 5/60 OD and 6/18 OS, and at 3 months, 6/24 OD and 6/12 OS. Injection methotrexate was also started but withheld at 3 months due to adverse effects of epistaxis and neutropenia which required hospitalization. Triamcinolone acetate 40 mg was injected in both knees under ultrasound guidance for the patient’s arthritic symptoms at this visit, following which he was discharged.
Both children were also advised to use masks in crowded public places in view of ongoing immunosuppressive treatment. Their parents were counselled about their condition with advice to follow up in the uvea clinic for visual ac
The patient later underwent phacoaspiration with single-piece hydrophobic foldable posterior chamber intraocular lenses and peripheral iridectomy under general anesthesia in the right eye (OD) followed by the left eye (OS) 1 month later. He recovered a best corrected visual acuity (BCVA) of 6/24 N18 OD and 6/19 N12 OS (by Cardiff’s visual acuity test). Executive bifocals were prescribed, immunosuppressive therapy continued and the child was asked to review after 1 month.
However, the patient was lost to follow-up and reviewed 2 years later with recurrence of systemic features and BCVA of 6/36 OD and 6/9 OS. Examination revealed bilateral anterior uveitis with patent peripheral iridotomy and early BSK OS. Digitally, IOP appeared raised. He was started on subcutaneous injection adalimumab 20 mg (0.4 mL) fortnightly with eye drops 1% prednisolone acetate hourly, 0.5% timolol maleate twice, 2% homatropine thrice and 0.5% carboxymethylcellulose four times daily in both eyes.
Within 2 weeks, the BCVA and systemic features had improved. However, the child developed recurrent anterior uveitis in OS at 1.5 months after the initial dose of adalimumab. Oral prednisolone acetate was added, 10 mg twice a day after meals, followed by fortnightly tapering. After 2 months, the patient’s vision had dropped to OD 6/30 and OS 6/24 by Cardiff’s visual acuity testing. He underwent membranectomy with EDTA chelation OS with guarded visual pro
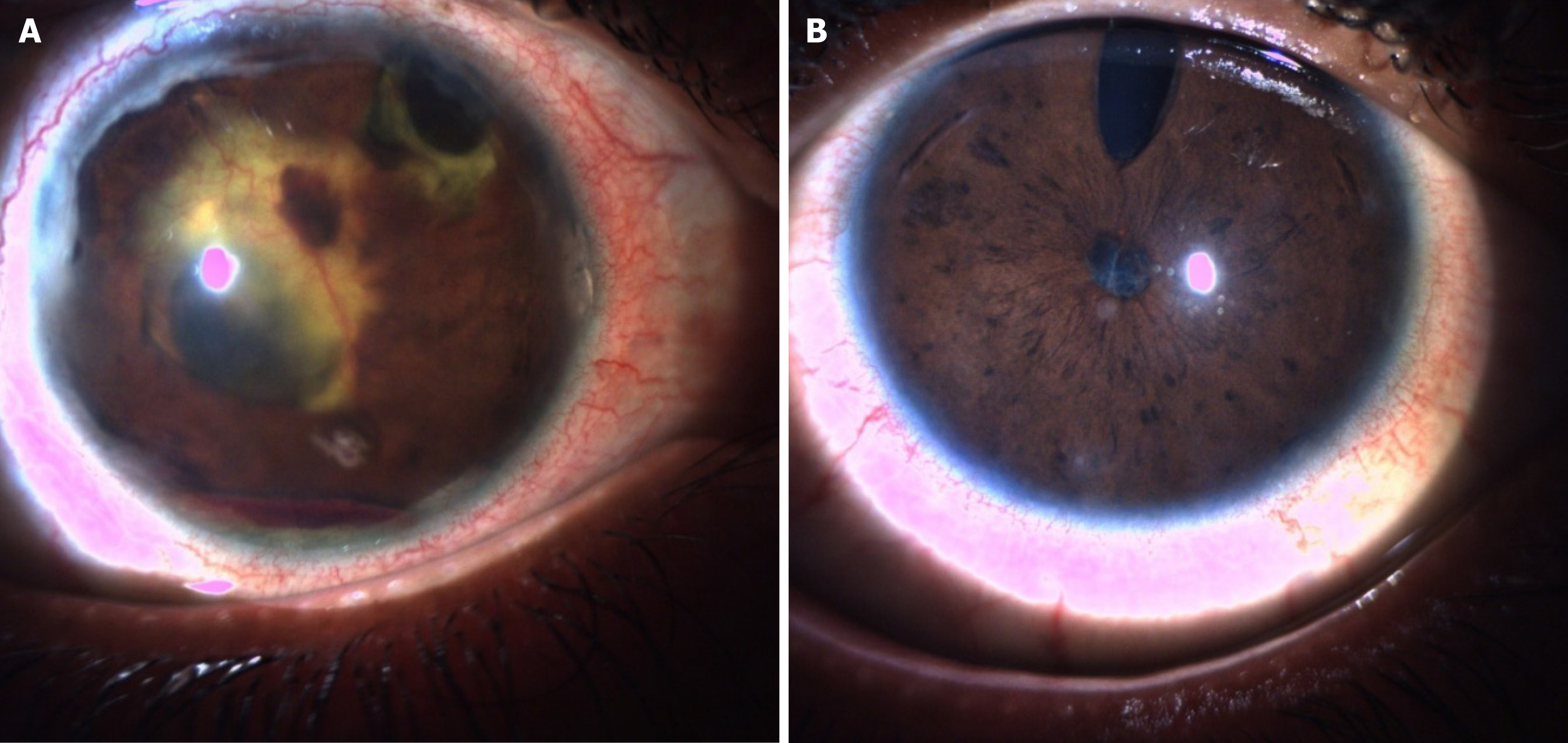
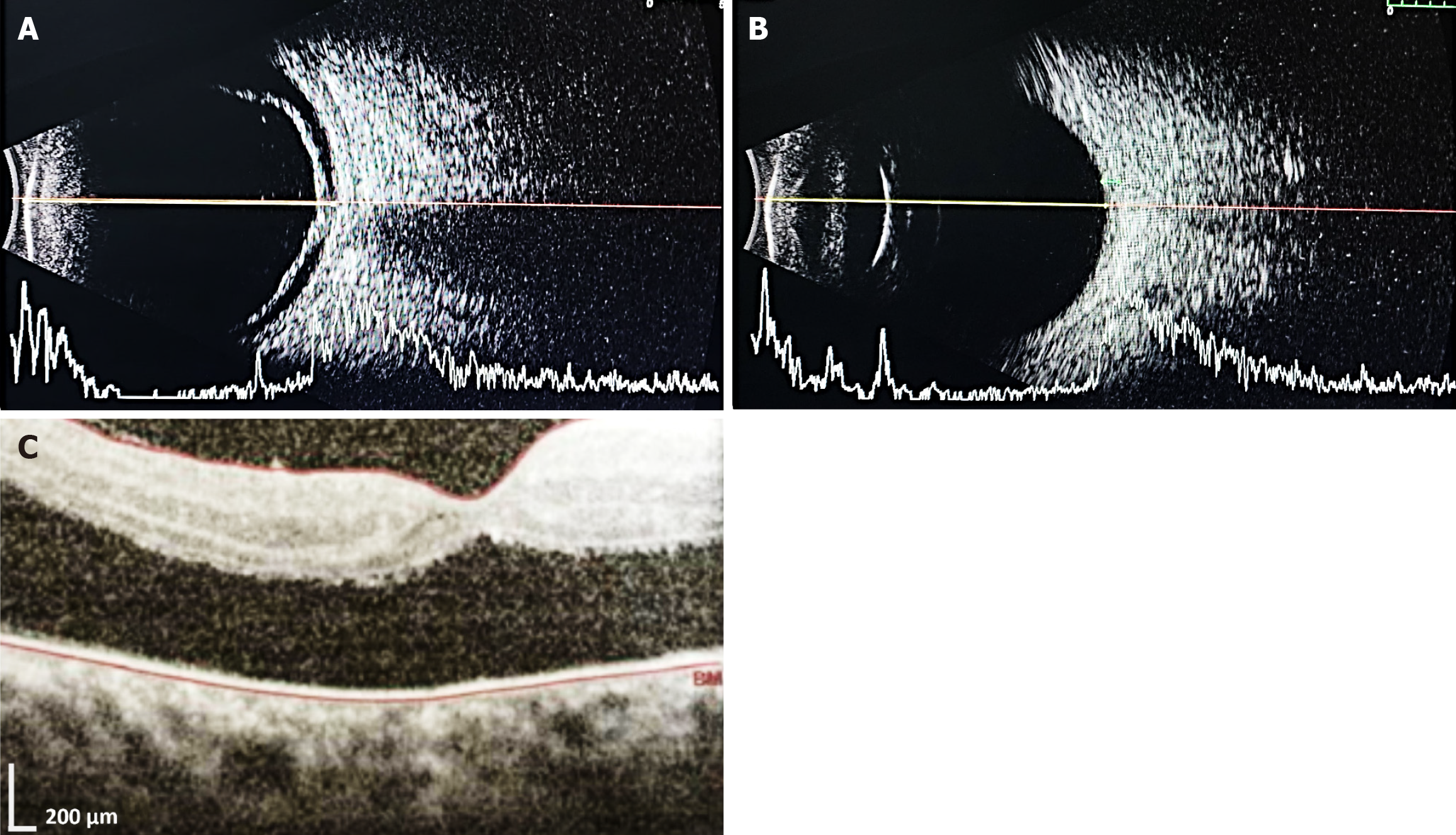
The patient was readmitted after 5 months for oral mucosal ulcers and received an injection of methotrexate 9 mg stat (10 mg/m²) with leucovorin rescue. Three doses of adalimumab (20 mg/0.4 mL) were administered subcutaneously at monthly intervals over the next 3 months. Fundus examination OD during this period revealed media haze with fibrovascular proliferation over the disc secondary to retinal vasculitis and exudative shallow retinal detachment (sparing the superior quadrant), which was confirmed by ultrasonography B-scan examination and optical coherence tomography (Figure 4). B-scan OS revealed an anechoic vitreous cavity, an intact retina-choroid-sclera complex, and a normal optic nerve head. Topical prednisolone and homatropine eye drops were restarted in OD, after which the clinical condition gradually improved. Two months later, the child suddenly developed painful bilateral foot ulcers on the lateral aspects of the soles, which were initially small (approximately 1 cm) but later became matted and enlarged (Figure 5) with ass
The earliest description of BS in 1985 was that of a familial granulomatous illness of the skin, joints, and eyes affecting 11 family members over four generations[17]. Early-onset sarcoidosis is a misnomer occasionally used to describe the illness when it manifests sporadically. The median age of onset of the illness is about 2 years, while ocular manifestations are seen at a median age of 4.4 years[3]. Initially, BS was reported majorly in Caucasians[18], but the condition is now increasingly being reported in other ethnicities as well-including the Indian and Chinese population. An account of cases of BS reported from India is provided in Table 1.
BS usually manifests with a classic triad. Skin rash is the most common manifestation in infancy[1]. While the majority of patients with BS experience uveitis by the age of four, arthritis follows between two and four years of life. The rash, usually thin, erythematous and maculopapular is a common early presentation, although it can also occasionally be dry and scaly. The arthritis is usually non-destructive, nontender and boggy, affecting the knees, wrists, and proximal interphalangeal joints of hands[19,20]. In the absence of prompt medical therapy, uveitis (which is initially posterior) eventually transforms into panuveitis, usually leading to considerable morbidity[21,22].
The spectrum of presenting features of BS varies, and most of these cases present to the rheumatologist when associated with systemic manifestations[7]. Fever and other systemic symptoms may be seen in almost half of all patients, as in our cases[19]. This condition is frequently linked to systemic diseases like nephrolithiasis, which causes severe renal damage from high calcium intake[23]. Non-caseating granulomas in the afflicted tissues are another characteristic of the disease, leading to diagnostic dilemmas. Prominent tenosynovitis on ultrasonography has been recently described as useful diagnostic sign for BS, especially in children with presumed JIA[11,20].
Associated ocular abnormalities due to chronic uveitis include BSK, a chronic degenerative disorder characterized by the development of grey opacification of the superficial cornea due to calcium deposition in confluent areas with scattered holes (Swiss-cheese appearance) as in our Case 1[24]. BS can also manifest as peripapillary nodules, disc edema, or disc pallor[25]. In a South Indian case series, there were six cases of keratoconjunctivitis sicca, three cases each of granulomatous panuveitis, anterior uveitis and conjunctival granulomas, one case of subepithelial corneal opacities and one case of subretinal granuloma, apart from cataract and BSK[7]. Retinal vasculitis and retinal detachment have also been reported as rare manifestations of BS, as in our Case 2[14,25].
Various mutations can alter the expression of the NOD2 gene and lead to different autoimmune disorders with varying presenting features[12]. Mutations associated with the ATP/Mg(2+)-binding site and helical domains of the central nucleotide-binding domain have been largely implicated in these disorders, affecting nuclear factor kappa-B signaling[26]. In BS, the p.Arg334Trp (p.R334W) missense mutation is the most common, while R334Q, R334W, E383G, E383K, R426H, and T605P mutations are the most destabilizing[27,28]. A case series from India reported NOD2 exon 4 mutations in 3 families-c.1000C>T (p.Arg334Trp), c.1147G>A (p.Glu383 Lys) and c.1324C>T (p.Lys442Phe), each with different predominant manifestations[9]. Other associated mutations include E600K, Y563S, and M513T[19]. Cases of BS with NOD2 variants may present with varied cutaneous changes, including lichenoid or granulomatous papules and nodules, dermatitis, chronic urticaria, ichthyosiform eruptions, vasospastic disorders, and oral ulcers, as in the second case[29]. Retinal vasculitis, as observed in our second case, has been reported as a rare manifestation in familial BS linked to the p.R334W variant[14]. Genetic testing is expensive and time-consuming, and may not be resorted to due to various constraints[13]. Although not yet widely available, a test for flow cytometric expression of NOD2 protein has been rec
The therapy consists of systemic and topical steroids along with disease-modifying anti-rheumatic drugs like methotrexate, but biologics are particularly beneficial in cases of severe intraocular inflammation[22]. Previous reports have highlighted successful control of intraocular inflammation with oral steroids and methotrexate, with the occasional addition of azathioprine, mycophenolate mofetil, leflunomide and hydroxychloroquine[32-35]. Cutaneous complications with methotrexate occur mostly in patients with risk factors for systemic toxicity and include skin-fold erosions, localized ulcers, epidermal necrosis or necrolysis, and erosions localized to psoriatic plaques, posing a diagnostic dilemma due to overlap with BS manifestations[33].
Biologic agents, typically tumor necrosis factor alpha (TNF-α) antagonists, are hence preferred in refractory non-infectious pediatric uveitis, as in BS[33,34,36]. In case the patient responds incompletely, a switch to another anti-TNF-α agent is warranted[37]. In our cases, adalimumab therapy was the cornerstone controlling systemic and ocular inflammation, with initial intensive steroids administered as a bridge. However, at present, there is no evidence to establish definite superiority of a particular agent for the management of uveitis in BS, and some reports have highlighted a role for therapeutic vitrectomy[4,29]. Interleukin 1 (IL-1) antagonists (anakinra, canakinumab) and IL-6 inhibitors (toci
In summary, the present report is the one of the few describing the clinical profile of sporadic cases of BS in East Indian patients[3]. Both cases improved on systemic steroids and adalimumab therapy. Timely detection and review assessments by the pediatric rheumatologist played a key role in the early management of the systemic disease. Prompt ophthalmic referral followed by meticulous diagnosis and management aided in effective response, reducing further morbidity. Limitations of the present case series include the absence of genetic analysis to look for NOD2 mutations due to the unavailability of this facility at our institute and financial limitations. In addition, ultrasonography of affected joints and histopathological examination of biopsy specimen could not be performed in our cases due to denial of consent.
BS remains an under-reported entity in India, and must be especially suspected when treating very young children with arthritis and uveitis, and when ophthalmic and rheumatological presentations suggest a diagnosis of JIA, or tubercular uveitis. Although it is important to elicit a proper family history and examine family members, BS may occur sporadically as well. Genetic counseling must be offered to the family if a child has been diagnosed with this entity. Timely interdepartmental referral and collaboration between pediatric rheumatologists and ophthalmologists is prudent. Prompt management with immunosuppressive pharmacotherapy and judicious surgical intervention are pertinent to avoid signi
| 1. | Wouters CH, Maes A, Foley KP, Bertin J, Rose CD. Blau syndrome, the prototypic auto-inflammatory granulomatous disease. Pediatr Rheumatol Online J. 2014;12:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 135] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 2. | Rosé CD, Wouters CH, Meiorin S, Doyle TM, Davey MP, Rosenbaum JT, Martin TM. Pediatric granulomatous arthritis: an international registry. Arthritis Rheum. 2006;54:3337-3344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 132] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 3. | Agarwal A, Karande S. Blau syndrome: An under-reported condition in India? J Postgrad Med. 2022;68:63-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 4. | Jain L, Gupta N, Reddy MM, Mittal R, Barik MR, Panigrahi B, Monie T, Basu S. A Novel Mutation in Helical Domain 2 of NOD2 in Sporadic Blau Syndrome. Ocul Immunol Inflamm. 2018;26:292-294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 5. | Naik AU, Annamalai R, Biswas J. Uveitis in sporadic Blau syndrome: Long-term follow-up of a refractory case treated successfully with adalimumab. Indian J Ophthalmol. 2018;66:1483-1485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 6. | Janarthanan M, Poddar C, Sudharshan S, Seabra L, Crow YJ. Familial Blau syndrome:First molecularly confirmed report from India. Indian J Ophthalmol. 2019;67:165-167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 7. | Babu K, Rao AP. Clinical Profile in Genetically Proven Blau Syndrome: A Case Series from South India. Ocul Immunol Inflamm. 2021;29:250-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 8. | Babu K, Padmapriya PG, Mooss V. Paediatric Ocular Sarcoidosis - Clinical Profiles from a Tertiary Eye Care Centre in South India. Ocul Immunol Inflamm. 2025;33:1995-1998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Baisya R, Manthri R, Tyagi M, Uppin SG, Rajasekhar L. Distinct NOD2 mutations reported in three families with Blau syndrome (BS) from a single center in India - Case series and review of literature. Clin Immunol. 2023;255:109743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 10. | Jindal AK, Pilania RK, Suri D, Gupta A, Gattorno M, Ceccherini I, Kumar N, Bansal R, Nada R, Singh S. A young female with early onset arthritis, uveitis, hepatic, and renal granulomas: a clinical tryst with Blau syndrome over 20 years and case-based review. Rheumatol Int. 2021;41:173-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 11. | Banday AZ, Mukherjee S, Kumrah R, Suri D, Jindal AK. Prominent tenosynovitis on ultrasonography: A useful sign for the diagnosis of Blau syndrome. Eur J Rheumatol. 2022;9:58-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 12. | Kumrah R, Pilania RK, Menia NK, Rawat A, Sharma J, Gupta A, Vignesh P, Jindal AK, Rikhi R, Agarwal A, Gupta V, Singh S, Suri D. Blau syndrome: Lessons learned in a tertiary care centre at Chandigarh, North India. Front Immunol. 2022;13:932919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 13. | Nagpal S, Singh V. An Interesting Case of Blau Syndrome – A Rare Autoinflammatory Disease. Indian J Med Spec. 2024;15:137-139. [DOI] [Full Text] |
| 14. | Sharma M, Arora A, Viswanath S, Jindal A, Rawat A, Suri D, Singh S, Gupta V. Retinal Vasculitis in Familial Blau Syndrome. Ocul Immunol Inflamm. 2025;1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 15. | Khalid A, Kaell A. Yao Syndrome: An Overview of Genotypic Associations, Clinical Manifestations, Diagnosis, and Treatment. Int Arch Allergy Immunol. 2025;186:189-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 16. | Batalli A, Liehr T, Temaj G. Behçet’s Disease: A Comprehensive Overview of Symptoms, Pathology, Genetics, and Treatment. J Mother Child. 2026;30:1-10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 17. | Blau EB. Familial granulomatous arthritis, iritis, and rash. J Pediatr. 1985;107:689-693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 303] [Cited by in RCA: 265] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 18. | Coto-Segura P, Mallo-Garcia S, Costa-Romero M, Arostegui JI, Yague J, Ramos-Polo E, Santos-Juanes J. A sporadic case of early-onset sarcoidosis resembling Blau syndrome due to the recurrent R334W missense mutation on the NOD2 gene. Br J Dermatol. 2007;157:1257-1259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 19. | Rosé CD, Pans S, Casteels I, Anton J, Bader-Meunier B, Brissaud P, Cimaz R, Espada G, Fernandez-Martin J, Hachulla E, Harjacek M, Khubchandani R, Mackensen F, Merino R, Naranjo A, Oliveira-Knupp S, Pajot C, Russo R, Thomée C, Vastert S, Wulffraat N, Arostegui JI, Foley KP, Bertin J, Wouters CH. Blau syndrome: cross-sectional data from a multicentre study of clinical, radiological and functional outcomes. Rheumatology (Oxford). 2015;54:1008-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 104] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 20. | Ikeda K, Kambe N, Satoh T, Matsue H, Nakajima H. Preferentially inflamed tendon sheaths in the swollen but not tender joints in a 5-year-old boy with Blau syndrome. J Pediatr. 2013;163:1525.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 21. | Sarens IL, Casteels I, Anton J, Bader-Meunier B, Brissaud P, Chédeville G, Cimaz R, Dick AD, Espada G, Fernandez-Martin J, Guly CM, Hachulla E, Harjacek M, Khubchandani R, Mackensen F, Merino R, Modesto C, Naranjo A, Oliveira-Knupp S, Özen S, Pajot C, Ramanan AV, Russo R, Susic G, Thatayatikom A, Thomée C, Vastert S, Bertin J, Arostegui JI, Rose CD, Wouters CH. Blau Syndrome-Associated Uveitis: Preliminary Results From an International Prospective Interventional Case Series. Am J Ophthalmol. 2018;187:158-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 56] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 22. | Achille M, Ilaria P, Teresa G, Roberto C, Ilir A, Piergiorgio N, Rolando C, Gabriele S. Successful treatment with adalimumab for severe multifocal choroiditis and panuveitis in presumed (early-onset) ocular sarcoidosis. Int Ophthalmol. 2016;36:129-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 23. | Jhanji V, Rapuano CJ, Vajpayee RB. Corneal calcific band keratopathy. Curr Opin Ophthalmol. 2011;22:283-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 24. | O’Brart DP, Gartry DS, Lohmann CP, Patmore AL, Kerr Muir MG, Marshall J. Treatment of band keratopathy by excimer laser phototherapeutic keratectomy: surgical techniques and long term follow up. Br J Ophthalmol. 1993;77:702-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 58] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 25. | Kreps EO, Al Julandani D, Guly CM, Arostegui JI, Dick AD, Ramanan AV. Long-Term Visual Outcome of Patients with Blau Syndrome. Ocul Immunol Inflamm. 2024;32:1728-1732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 26. | Parkhouse R, Boyle JP, Monie TP. Blau syndrome polymorphisms in NOD2 identify nucleotide hydrolysis and helical domain 1 as signalling regulators. FEBS Lett. 2014;588:3382-3389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 27. | P R, Ramireddy S, Chakraborty S, Mukherjee S, J S, C S. Structural localization of pathogenic mutations in the central nucleotide-binding domain (NBD) of nucleotide-binding oligomerization domain-2 (NOD2) protein and their inference in inflammatory disorders. Nucleosides Nucleotides Nucleic Acids. 2021;40:1198-1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 28. | Thirumal Kumar D, Udhaya Kumar S, Nishaat Laeeque AS, Apurva Abhay S, Bithia R, Magesh R, Kumar M, Zayed H, George Priya Doss C. Computational model to analyze and characterize the functional mutations of NOD2 protein causing inflammatory disorder - Blau syndrome. Adv Protein Chem Struct Biol. 2020;120:379-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 29. | Mohandesi NA, Mittal S, Orandi AB, Smith WM, Khan NS, Davis DM. Expanding the Cutaneous Presentation of Blau Syndrome, Response to Treatment, and Correlation With Genetics. Pediatr Dermatol. 2025;42:956-960. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | Kumrah R, Sharma J, Arora K, Rawat A, Gupta V, Singh S, Suri D. Flow Cytometric Expression of Nucleotide-Binding Oligomerization Domain Containing 2 (NOD2) Protein: An Effective Screening Tool for Blau Syndrome. Int J Rheum Dis. 2025;28:e70397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 31. | Okazaki F, Wakiguchi H, Korenaga Y, Nakamura T, Yasudo H, Uchi S, Yanai R, Asano N, Hoshii Y, Tanabe T, Izawa K, Honda Y, Nishikomori R, Uchida K, Eishi Y, Ohga S, Hasegawa S. A novel mutation in early-onset sarcoidosis/Blau syndrome: an association with Propionibacterium acnes. Pediatr Rheumatol Online J. 2021;19:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 32. | Maccora I, Wouters C, Rosè CD, Maniscalco V, de Masi S, Mastrolia MV, Marrani E, Pagnini I, Simonini G. Treatment of uveitis in Blau syndrome: A systematic review and meta-analysis. J Autoimmun. 2025;153:103401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 33. | Berna R, Rosenbach M, Margolis DJ, Mitra N, Baumrin E. Methotrexate Cutaneous Ulceration: A Systematic Review of Cases. Am J Clin Dermatol. 2022;23:449-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 34. | Chen J, Luo Y, Zhao M, Wu D, Yang Y, Zhang W, Shen M. Effective treatment of TNFα inhibitors in Chinese patients with Blau syndrome. Arthritis Res Ther. 2019;21:236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 35. | Jensen ME, Harrell K, McBride JD. Case Report: Methotrexate and hydroxychloroquine in combination for the treatment of NOD2-mutation-associated Blau syndrome. Front Immunol. 2023;14:1279329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 36. | Wang OJE, Jacob M, Crawford RI, Lam JM. Blau Syndrome (Juvenile Systemic Granulomatosis): State-Of-The-Art Review. Pediatr Dermatol. 2025;42:728-734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 37. | Lassoued Ferjani H, Kharrat L, Ben Nessib D, Kaffel D, Maatallah K, Hamdi W. Management of Blau syndrome: review and proposal of a treatment algorithm. Eur J Pediatr. 2024;183:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |