Published online Jan 16, 2025. doi: 10.12998/wjcc.v13.i2.94530
Revised: October 1, 2024
Accepted: October 25, 2024
Published online: January 16, 2025
Processing time: 232 Days and 22.7 Hours
Intramuscular corticosteroid injection may cause adverse effects such as dermal and/or subcutaneous atrophy, alopecia, hypopigmentation, and hyperpigmen
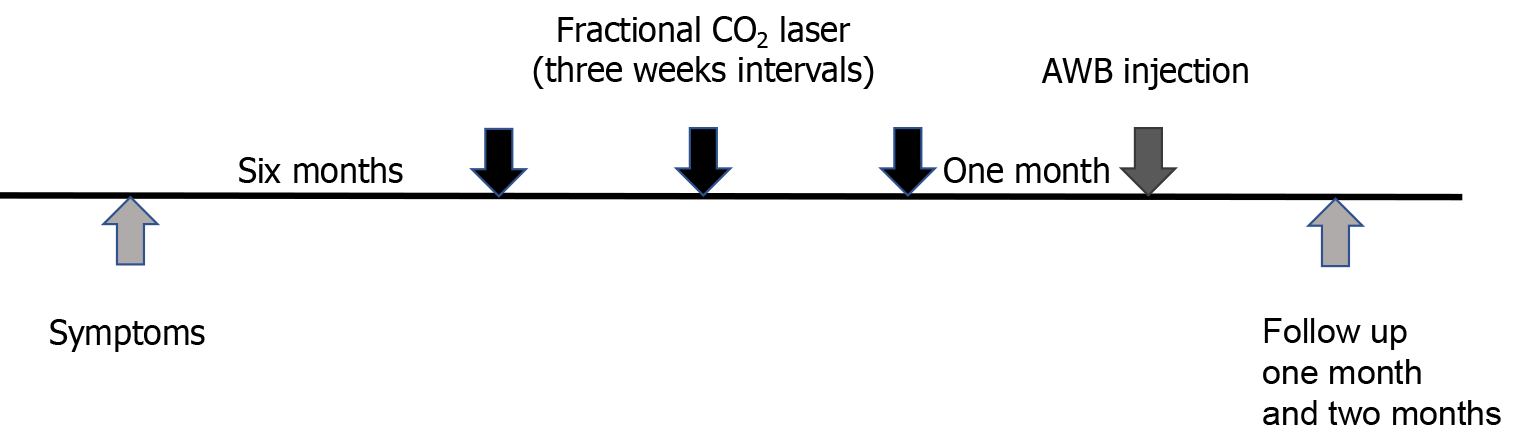
In this paper, we report a case of corticosteroid injection induced lipoatrophy treated with autologous whole blood (AWB) injection, as the condition had been unresponsive to fractional laser therapy. A 29-year-old female patient visited the dermatology clinic complaining of skin depression on her right buttock area, which had appeared six months earlier. There had been only subtle improvement at the margins after fractional CO2 laser treatment; therefore, after obtaining informed consent from the patient, AWB treatment was initiated. One month after the first AWB injection, the size and depth of the lesion had noticeably improved, and a slight improvement was also observed in discoloration.
Close observation is the initial treatment of choice for steroid induced skin atro
Core Tip: Intramuscular corticosteroid injection may cause adverse effects such as dermal and/or subcutaneous atrophy, alopecia, hypopigmentation, and hyperpigmentation. Although cutaneous atrophy can spontaneously resolve, several treatment options have been suggested for this condition. Here, we report a case of corticosteroid injections induced lipoatrophy treated with autologous whole blood (AWB) injection, which was unresponsive to fractional laser therapy. Close observation is the initial treatment of choice for steroid induced skin atrophy, however for patient in need of immediate cosmetic improvement, AWB injection may be a safe and cost-effective solution for treatment.
- Citation: Kim W, Park JS, Kim EH. Refractory lipoatrophy treated with autologous whole blood injection: A case report. World J Clin Cases 2025; 13(2): 94530
- URL: https://www.wjgnet.com/2307-8960/full/v13/i2/94530.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v13.i2.94530
In fields of medicine such as dermatology, plastic surgery and orthopedics, corticosteroid injections are frequently performed. Intramuscular corticosteroid injection may cause adverse effects such as dermal and/or subcutaneous atrophy, alopecia, hypopigmentation/depigmentation, and hyperpigmentation[1]. Previous reports have demonstrated that skin atrophy may resolve spontaneously, but no definite data are available, and the outcome may differ from person to person[2,3]. Several treatment options have been suggested for skin atrophy such as autologous fat grafting, intralesional saline injections, surgical excision, and poly-L-lactic acid injections, all with different success rates. Although the exact mechanism of action of autologous whole blood (AWB) injection remains unclear, in experimental and clinical models it seems to affect immune function. By injecting the patient’s AWB into the affected tissue, the body's own tissue-healing mechanisms may be stimulated possibly through activation of cellular and humoral mediators[4,5]. In this paper, we report a case of corticosteroid injection induced lipoatrophy treated with AWB injection.
A 29-year–old female patient visited the dermatology clinic complaining of skin depression on her right buttock area, which had appeared six months earlier.
The patient had previously received an intramuscular triamcinolone acetonide injection to alleviate a severely pruritic maculopapular rash that occurred after consuming some health products. She noticed the deformation approximately two months after the injection.
No known past history.
Not applicable.
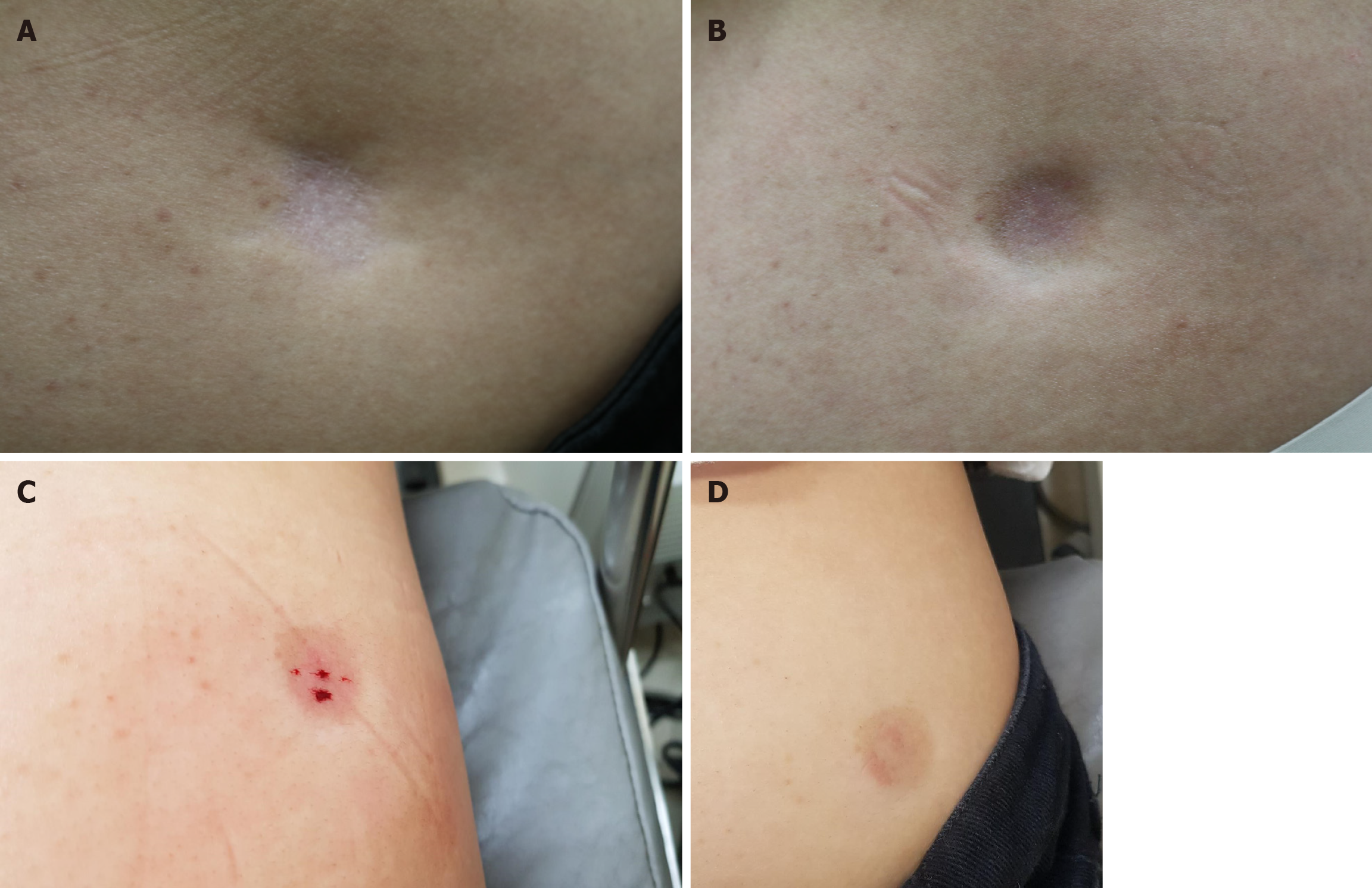
At the site of corticosteroid injection, depigmentation, epidermal/dermal atrophy, and subcutaneous fat atrophy were observed in the right gluteal region (Figure 1A).
Not applicable.
Not applicable.
Localized subcutaneous lipoatrophy was diagnosed based on the patient's past history and clinical characteristics.
Because the lesion was disfiguring and causing stress to the patient, several modes of treatment were explained and fractional CO2 laser (eCO2, Lutronic, Korea) treatment was initiated. Fluence ranging from 30 J/cm2 was used at densities of 100-150 MTZ/cm2, and single or double pass was used over the scar and also its margins. The patient received four laser treatments at three-week intervals (Figure 2). There was subtle improvement at the margins after the laser treatment, but the main depression was still quite noticeable (Figure 1B). In addition, the laser treatment induced post-inflammatory hyperpigmentation. Therefore, after obtaining informed consent, AWB injection was started. Using a 5 cc syringe with a 21-gauge 38 mm needle, 4 cc AWB was collected from peripheral antecubital veins. The AWB was injected into the subcutaneous fat tissue immediately after the blood was drawn. The needle was held at a 90° angle and 0.2-0.4 mL AWB was injected into the deep subcutaneous fat tissue and superficial subcutaneous fat tissue of the atrophic area (Figure 1C)[6].
Other than mild bruising and slight induration one week after the injection, there were no side effects. One month after the first AWB treatment, the lesion size and depth had markedly improved, and the discoloration had also mildly resolved. At the 2-month follow-up, induration and purpura had improved, and the clinical effect was maintained (Figure 1D). The patient was content with the results of the treatment.
The pathomechanism of subcutaneous lipoatrophy after steroid injection is unclear; however, an increase in macrophage cells accompanied by a decrease in both the number and size of adipocytes has been observed[7,8]. Moreover, in histologic evaluations of animal models, microscopic corticosteroid crystals were found after a localized steroid injection[9].
Hyaluronic acid filler injections, fat grafting, saline solution injections and observation are possible options of treatment for cutaneous atrophy[10]. Fat grafting can restore the volume loss and, through the stem cells in fat tissue, stimulate tissue regeneration, hence it may be an effective solution[10]. Regrettably, the process can be complex and invasive[11]. Hyaluronic acid fillers are another common treatment, but these may induce inflammatory reactions at the injection site or cause granuloma formation[12]. Saline solution injections can be administered locally to dilute and/or dissolve the steroid crystals found in the tissues of patients with subcutaneous lipoatrophy[13]. However, several treat
During AWB treatment, the patient’s blood is drawn from the peripheral veins and then promptly injected into the affected regions. Immediate improvement may have been achieved through the volumetric filling effect of AWB injected at the affected site. Moreover, the growth factors from platelets, such as transforming growth factor-beta 1, platelet-derived growth factor alpha and beta, epithelial growth factor, and insulin-like growth factor I, are key factors for skin regeneration[20]. AWB also contains hormonal and cellular mediators that induce collagen regeneration, cell proliferation, and stimulate the production of angiogenic factors resulting in tissue healing[4,22]. AWB injections are frequently used in the treatment of musculoskeletal diseases such as patellar tendinopathy, lateral epicondylitis, and Achilles tendinopathy[23,24]. They are also used for chronic urticaria[25]. A number of patients visit the dermatology clinic complaining of steroid induced lipoatrophy in the ankle, elbow, and buttock areas resulting from various treatments. Thus, AWB injections may constitute both a curative and preventive form of treatment for these patients.
Close observation is the initial treatment of choice for steroid induced skin atrophy; however, for patients in need of immediate cosmetic improvement, AWB injections may be considered a safe and cost-effective alternative. Future clinical trials with large sample sizes are needed to draw more reliable conclusions.
| 1. | Friedman SJ, Butler DF, Pittelkow MR. Perilesional linear atrophy and hypopigmentation after intralesional corticosteroid therapy. Report of two cases and review of the literature. J Am Acad Dermatol. 1988;19:537-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 70] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 2. | Brinks A, Koes BW, Volkers AC, Verhaar JA, Bierma-Zeinstra SM. Adverse effects of extra-articular corticosteroid injections: a systematic review. BMC Musculoskelet Disord. 2010;11:206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 160] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 3. | Pace CS, Blanchet NP, Isaacs JE. Soft Tissue Atrophy Related to Corticosteroid Injection: Review of the Literature and Implications for Hand Surgeons. J Hand Surg Am. 2018;43:558-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 4. | Kim EH. Pruritic Urticarial Papules and Plaques of Pregnancy Occurring Postpartum Treated with Intramuscular Injection of Autologous Whole Blood. Case Rep Dermatol. 2017;9:151-156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 5. | Kumar S, Mahajan BB, Kaur S, Singh A. Autologous therapies in dermatology. J Clin Aesthet Dermatol. 2014;7:38-45. [PubMed] |
| 6. | Frojo G, Halani SH, Pessa JE, Pazmiño P, Wall S, Kenkel JM, Del Vecchio DA. Deep Subcutaneous Gluteal Fat Compartments: Anatomy and Clinical Implications. Aesthet Surg J. 2023;43:76-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 7. | Ahmed I. Post-injection involutional lipoatrophy: Ultrastructural evidence for an activated macrophage phenotype and macrophage related involution of adipocytes. Am J Dermatopathol. 2006;28:334-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 8. | Dahl PR, Zalla MJ, Winkelmann RK. Localized involutional lipoatrophy: a clinicopathologic study of 16 patients. J Am Acad Dermatol. 1996;35:523-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 65] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 9. | Fülöp E. Mechanism of local skin atrophy caused by intradermally injected corticosteroids. Dermatologica. 1976;152 Suppl 1:139-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 10. | Imagawa K, Ohkuma S. A case of fat injection for treating subcutaneous atrophy caused by local administration of corticosteroid. Tokai J Exp Clin Med. 2010;35:66-69. [PubMed] |
| 11. | Coleman SR. Structural fat grafting: more than a permanent filler. Plast Reconstr Surg. 2006;118:108S-120S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 833] [Cited by in RCA: 837] [Article Influence: 41.9] [Reference Citation Analysis (0)] |
| 12. | Hausauer AK, Jones DH. Long-Term Correction of Iatrogenic Lipoatrophy With Volumizing Hyaluronic Acid Filler. Dermatol Surg. 2018;44 Suppl 1:S60-S62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 13. | Birnbaum A, Yoon MY, Struhl S. Serial saline solution injections for the treatment of lipoatrophy and depigmentation after corticosteroid injection for medial epicondylitis. JSES Int. 2020;4:1002-1005. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 14. | Leo MS, Kumar AS, Kirit R, Konathan R, Sivamani RK. Systematic review of the use of platelet-rich plasma in aesthetic dermatology. J Cosmet Dermatol. 2015;14:315-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 102] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 15. | Ogretmen Z. Efficacy of Platelet Rich Plasma in the Treatment of lipoatrophy. JOJCS. 2017;3:555622. [DOI] [Full Text] |
| 16. | Alster T, Zaulyanov L. Laser scar revision: a review. Dermatol Surg. 2007;33:131-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 43] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 17. | Orringer JS, Rittié L, Baker D, Voorhees JJ, Fisher G. Molecular mechanisms of nonablative fractionated laser resurfacing. Br J Dermatol. 2010;163:757-768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 60] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 18. | Xatzipsalti M, Alvertis H, Vazeou A. Laser treatment for lipoatrophy in children with diabetes type 1. Diabetol Int. 2022;13:452-455. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 19. | Barros Mourão CDA, Gheno E, Lourenço E, Barbosa RL, Kurtzman G, Javid K, Mavropoulos E, Benedicenti S, Calasans-maia M, de Mello Machado R, Alves G. Characterization of a new membrane from concentrated growth factors associated with denaturized Albumin (Alb-CGF) for clinical applications: A preliminary study. Int J Growth Factors Stem Cells Dent. 2018;1:64. [RCA] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 20. | Fujioka-Kobayashi M, Schaller B, Mourão CFAB, Zhang Y, Sculean A, Miron RJ. Biological characterization of an injectable platelet-rich fibrin mixture consisting of autologous albumin gel and liquid platelet-rich fibrin (Alb-PRF). Platelets. 2021;32:74-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 86] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 21. | Majewska L. Autologous Plasma Gel as an Effective Method of Facial Volume Restoration and Skin Rejuvenation. Dermatol Ther. 2023;2023:7. [DOI] [Full Text] |
| 22. | Anitua E, Andía I, Sanchez M, Azofra J, del Mar Zalduendo M, de la Fuente M, Nurden P, Nurden AT. Autologous preparations rich in growth factors promote proliferation and induce VEGF and HGF production by human tendon cells in culture. J Orthop Res. 2005;23:281-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 380] [Cited by in RCA: 311] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 23. | Nuhmani S. Injection therapies for patellar tendinopathy. Phys Sportsmed. 2020;48:125-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 24. | Kıvrak A, Ulusoy I. Comparison of the Clinical Results of Platelet-Rich Plasma, Steroid and Autologous Blood Injections in the Treatment of Chronic Lateral Epicondylitis. Healthcare (Basel). 2023;11:767. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 25. | Kr R, H B. Efficacy of autologous serum therapy in positive and negative autologous serum skin test patients in chronic urticaria in a tertiary care centre. Dermatol Reports. 2023;15:9525. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/