Published online Jul 6, 2022. doi: 10.12998/wjcc.v10.i19.6360
Peer-review started: January 23, 2022
First decision: March 23, 2022
Revised: April 1, 2022
Accepted: May 12, 2022
Article in press: May 12, 2022
Published online: July 6, 2022
Processing time: 152 Days and 5.2 Hours
Non-small-cell lung cancer (NSCLC) causes significant mortality worldwide. Patients with chronic renal failure have an increased risk of developing lung cancer. NSCLC Patients with chronic renal failure undergoing hemodialysis (HD) often exhibit poor performance, and chemotherapy is generally contraindicated. Oral epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors (TKIs) are effective treatment agents for NSCLC patients. However, the benefits and adverse effects of EGFR-TKIs in NSCLC undergoing HD are known. There are no clinical studies on the effects of EGFR-TKIs on NSCLC patients undergoing HD. We reviewed all previous case reports about EGFR-TKIs in NSCLC patients undergoing HD. It is difficult to design studies about the effects of EGFR-TKIs in patients undergoing HD, and this review is quite important. EGFR-TKIs are well tolerated in patients undergoing HD. The main routes of elimination of EGFR-TKIs are metabolism via the liver, and renal elimination is minor. The recommended doses and pharmacokinetics of these EGFR-TKIs for patients undergoing HD are similar to those for patients with normal renal function. The plasma protein binding of EGFR-TKIs is very high, and it is not necessary to adjust the dose after HD. In conclusion, EGFR-TKIs are effective and well tolerated in patients undergoing HD.
Core Tip: Lung cancer causes significant mortality worldwide. Patients with chronic renal failure increase the difficulty of treatment. Oral epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors (TKIs) are effective treatment agents for non-small-cell lung cancer (NSCLC) patients. According to this review, EGFR-TKIs are suitable for treating NSCLC patients undergoing hemodialysis (HD) owing to their good response rates and tolerance. The main routes of elimination of EGFR-TKIs are via the liver and excretion via feces. The recommended doses and pharmacokinetics of EGFR-TKIs for patients undergoing HD are similar to those with normal renal function. The plasma protein binding of EGFR-TKIs is high, and it is not necessary to adjust the dose after HD.
- Citation: Lan CC, Hsieh PC, Huang CY, Yang MC, Su WL, Wu CW, Wu YK. Review of epidermal growth factor receptor-tyrosine kinase inhibitors administration to non-small-cell lung cancer patients undergoing hemodialysis. World J Clin Cases 2022; 10(19): 6360-6369
- URL: https://www.wjgnet.com/2307-8960/full/v10/i19/6360.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v10.i19.6360
Lung cancer causes significant mortality worldwide[1]. Lung cancer is classified into small-cell lung cancer (SCLC) and non-small-cell lung cancer (NSCLC), with NSCLC accounting for 80%–85% of lung cancer cases and adenocarcinoma accounting for 80% of NSCLC cases[1]. Despite the development of modern medicines, the outcomes of lung cancer remain poor[1]. The treatment of NSCLC depends strictly on the disease stage, and 80% of NSCLC patients are diagnosed at advanced (IIIB/IV) stages[1]. Since the lungs are often diagnosed at advanced stages and are unresectable, targeted therapy and chemotherapy are the major treatments for these patients[1].
In general, chronic renal failure (CRF) patients undergoing hemodialysis (HD) exhibit a higher incidence of cancers, including lung cancer[2]. The risk factors for developing cancers include smoking, air pollution, genetic susceptibility, and occupational exposure[1]. It is suspected that the reasons for the higher incidence of cancers in patients undergoing HD are further related to weakened immunity, chronic inflammation, malnutrition, and impaired DNA repair[2]. CRF increases the incidence of cancers as well as the difficulty in treating it. Therefore, the treatment of cancers in such patients is important.
For patients with cancer and CRF, who often exhibit poor performance, chemotherapy is generally contraindicated owing to its greater adverse effects (AEs). Moreover, chemotherapeutic drugs, such as cisplatin, are regarded as nephrotoxic agents and are not suitable for the treatment of CRF patients. Cisplatin is one of the most widely used chemotherapeutic agent for cancers. However, its clinical application is limited by its adverse effects, such as bone marrow suppression leading to hematopoietic abnormalities[3]. An increased incidence of adverse reactions to cisplatin has been reported in patients with renal insufficiency[4]. Therefore, the management of anti-cancer treatments for CRF patients undergoing HD is challenging.
Oral epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors (TKIs) are promising treatment agents for NSCLC patients. EGFR is a transmembrane receptor on the surface of epithelial cells and is considered an important molecular target in cancer treatment. EGFR gene mutation is a predictor of EGFR-TKI efficacy. EGFR-gene-activating mutations occur in 50%–60% of Asian and 10%–20% of Caucasian subjects with NSCLC[5]. Common mutations, including deletions in exon 19 and substitution of L858R in exon 21, comprise approximately 85% of EGFR mutations[5]. Rare mutations (about 15%) include point mutations, insertions, and deletions within exons 18–25 of the EGFR gene[5].
EGFR-TKIs exert low side effects and less impact on kidney function, and patients exhibit better tolerability for them[6]. EGFR-TKIs, including gefitinib and erlotinib (first-generation), afatinib (second-generation), and osimertinib (third-generation), have shown significant benefits, with improved overall response rates (ORRs), longer progression-free survival (PFS), and better overall survival rate (OS) in patients with NSCLC harboring EGFR mutations[6]. Lung cancer patients undergoing HD sometimes exhibit poor performance; EGFR-TKIs are often a treatment choice for such patients. Since physicians worry about AEs and nephrotoxicity of chemotherapy agents in patients undergoing HD, EGFR-TKIs are often an optional strategy for such patients. However, most clinical trials have excluded patients undergoing HD, and the applicability of EGFR-TKIs for such patients has not been studied. Therefore, we aimed to review the benefits, AEs, and pharmacokinetics of EGFR-TKIs in patients undergoing HD.
Gefitinib was approved for metastatic NSCLC patients with sensitive EGFR mutations in July 2015[7]. It has been demonstrated that using gefitinib as the first line of treatment for NSCLC with sensitive EGFR mutations results in an ORR of 62%–71%, PFS of 8–13 mo, and OS of 21–30 mo[7]. The common AEs are skin rash, acne, and diarrhea[7]. Most AEs resolve on their own or after medical treatment. Elevation in liver function test parameters has been observed, but these elevations are often not accompanied by any symptoms. The incidence of rare AEs, such as interstitial lung disease (ILD), is approximately 1%[7].
Bersanelli et al[8] reported a 60-year-old female Caucasian non-smoker undergoing HD with right lung adenocarcinoma with mediastinal lymph nodes and bone metastasis. Her initial ECOG PS score was 0, and she received radiation therapy on the spine and was administered gemcitabine and cisplatin. Owing to progression of bone metastasis after three cycles of gemcitabine and cisplatin, second-line treatment with gefitinib was initiated. Gefitinib induced a good response of the disease to treatment for 7 years without complications. Del Conte et al[9] reported a 70-year-old male non-smoker undergoing HD with right middle lobe lung adenocarcinoma. The patient underwent right middle lobe lobectomy for pT1N0M0 NSCLC (stage IA). After 3.5 years from surgery, recurrent lung adenocarcinoma with deletion of 15 nucleotides of exon 19 was found. He received gefitinib and exhibited only a grade 1 cutaneous rash. After 2 years of gefitinib treatment, he was still in complete remission without serious AEs[9]. Bersanelli et al[8] and Del Conte et al[9] showed that gefitinib was effective and well tolerated in patients undergoing HD.
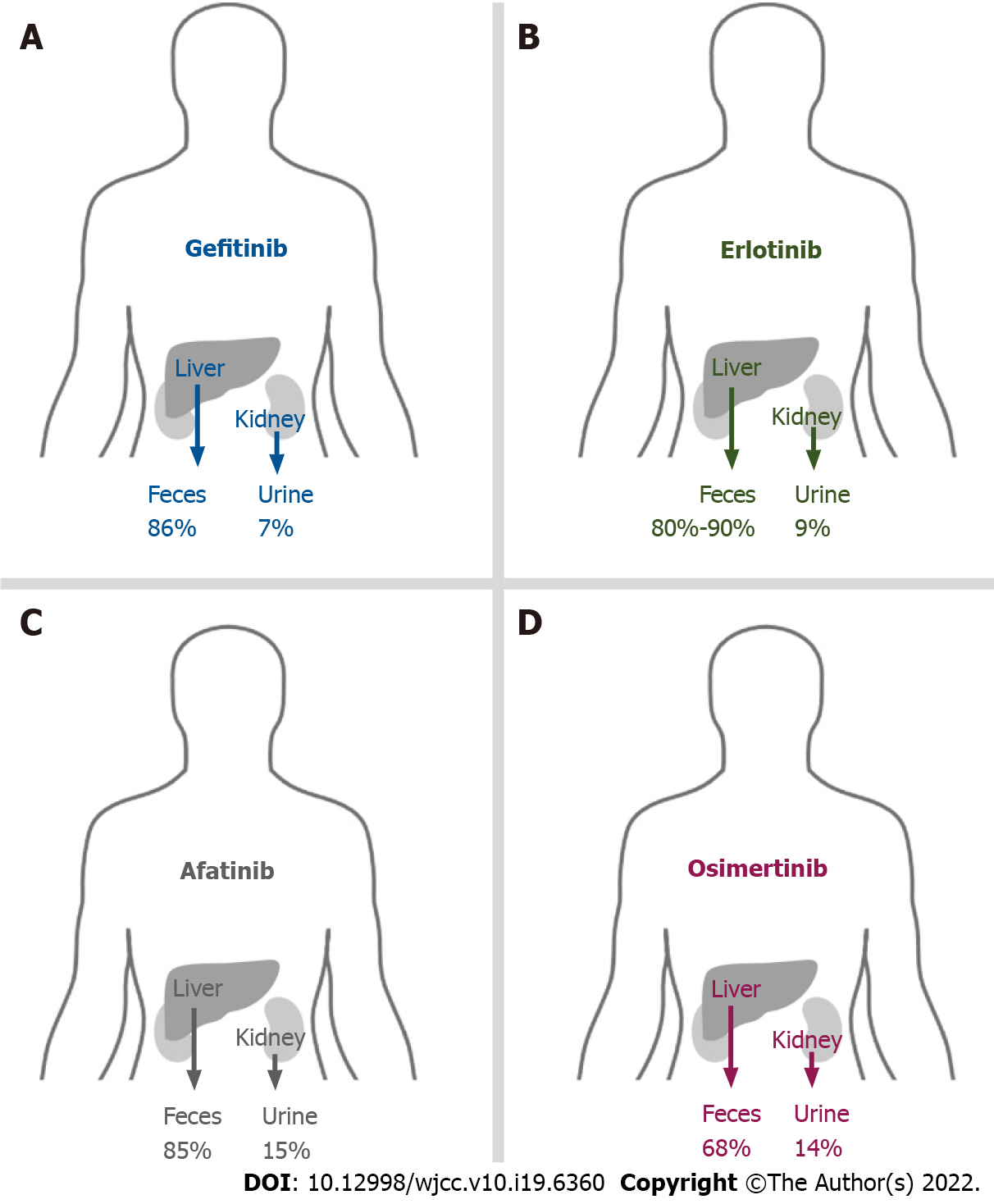
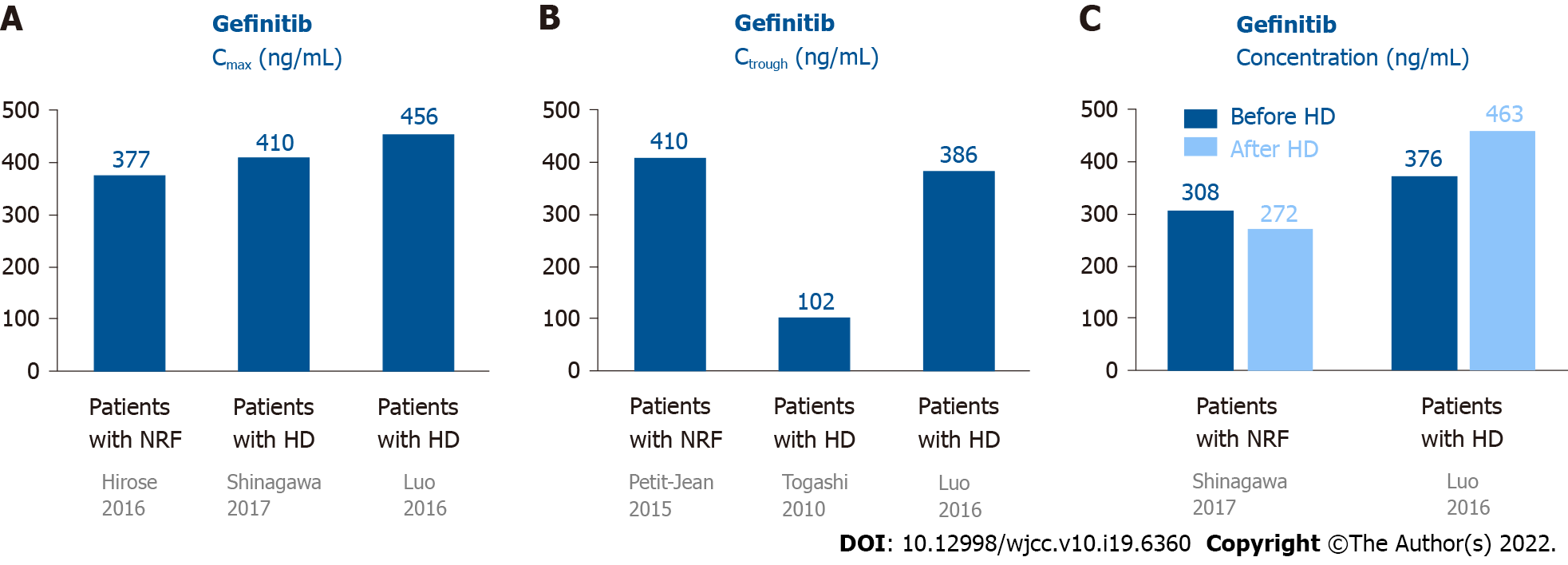
Most gefitinib is metabolized by the liver and excreted via feces (86%), and a small portion (7%) is excreted via urine (Figure 1A)[10]. Approximately 90% of gefitinib binds to plasma proteins in the blood[10]. The half-life of gefitinib is 41 h, and steady-state plasma concentrations are attained by day 7–10[11]. The maximum concentration (Cmax) is attained 5 h (3–7 h) after oral dosing, the median Cmax is 377 (168–781) ng/mL, the area under the curve of the plasma concentration from 0 to 24 h (AUC0-24) is 4893 (698–13991) ng· hr/mL, and the trough concentration (Ctrough) is 410 (115–1021) ng/mL[11].
There are only two case reports on the pharmacokinetics of gefitinib in patients undergoing HD. Shinagawa et al[12] performed the first pharmacokinetic analysis of gefitinib in a 58-year-old female non-smoker undergoing HD for 8 years. She developed lung adenocarcinoma (pT1N0M0, stage IA) and underwent left lower lobectomy (LLL)[12]. Two years later, she had recurrent lung cancer, which had metastasized to the left subclavian lymph node, and meningitis. The EGFR mutation showed a deletion of 15 nucleotides in exon 19 in tumor tissues. She was administered 250 mg gefitinib daily. The Cmax and Ctrough of gefitinib were 410.4 and 102.7 ng/mL, respectively. The plasma concentrations were 308.3 and 272.2 ng/mL before and after HD (88% maintained throughout HD), respectively[12]. There were no serious AEs following gefitinib administration[12]. Luo et al[13] reported a 75-year-old female non-smoker with lung adenocarcinoma (pT1N0M0, stage IA) who underwent right lower lobectomy. Nine years later, she underwent HD due to hypertension. However, the lung cancer relapsed with multiple nodules in both lungs. EGFR mutations in the tumor tissue revealed an L858R mutation in exon 21. She was then administered 250 mg gefitinib daily. The Cmax was 456 and 463 ng/mL on non-HD and HD days, respectively. The Ctrough was 386 ng/mL. The plasma concentrations were 376 and 463 ng/mL before and after HD, respectively. The initial response to gefitinib was a decrease in tumor size, but the tumor increased in size after 8 mo of treatment. No severe AEs were reported after gefitinib administration.
The data on the pharmacokinetics of gefitinib in patients undergoing HD and normal renal function (NRF) are shown in Figure 2. These data revealed that the pharmacokinetics of gefitinib in patients undergoing HD were similar to subjects with NRF[12,13]. Since 90% of gefitinib is bound to plasma proteins[10], it was rational that 90% of gefitinib was retained in the blood after HD. In addition, these patients showed a good response to gefitinib and did not have elevated AEs[12,13].
Erlotinib is a reversible EGFR-TKI that was approved in 2004 for patients harboring EGFR exon 21 L858R mutations and exon 19 deletion[6]. Previous studies using erlotinib as the first line of treatment for NSCLC with sensitive EGFR gene mutations showed an ORR of 58%–83%, PFS of 9.7–13 mo, and OS of 23–33 mo[14]. Common AEs included skin rash, diarrhea, nausea, anorexia, dermatitis acneiform, fatigue, anemia, and ILD[14]. However, grade 3 or 4 AEs rarely occurred in subjects upon administration of erlotinib[6].
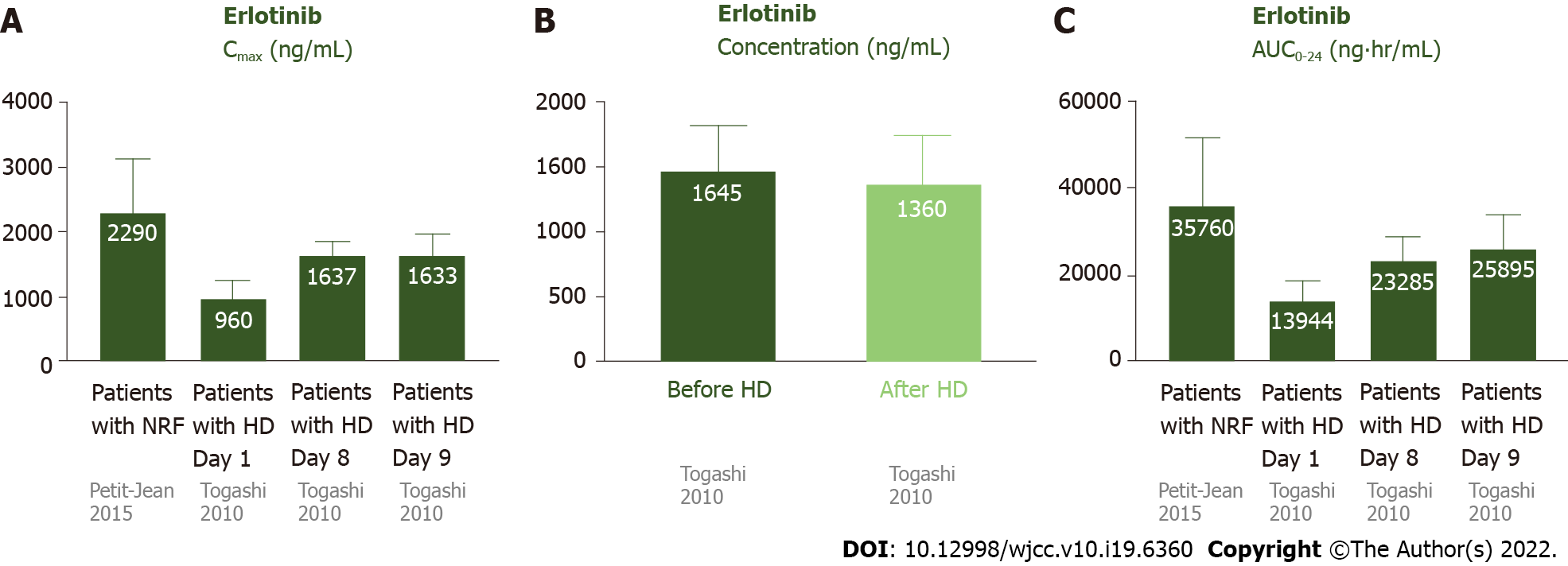
Most erlotinib is metabolized by the liver and excreted via feces (80%–90%), and a small portion (9%) is excreted via urine (Figure 1B)[15]. Approximately 95% of erlotinib is bound to plasma proteins[15]. The half-life of erlotinib is 21.86 ± 28.35 h, and steady-state concentrations in plasma are attained by day 8–10[16]. The Cmax is attained 3.69 ± 3.21 h after dosing, and the median Cmax is 2290 ± 840 ng/mL. The median AUC0-24 is 35760 ± 15720 ng· hr/mL[16].
Togashi et al[17] reported three NSCLC patients undergoing HD who were treated with erlotinib. Case 1 was a 74-year-old male ex-smoker undergoing HD for 5 years, who had lung adenocarcinoma (cT1N3M1, stage IV) without any EGFR mutations. Case 2 was a 74-year-old female non-smoker undergoing HD for 3 years, who had adenocarcinoma (cT2N0M0, stage IB) without any EGFR mutations. Initially, she received irradiation therapy with a dose of 54 Gy for the tumor. The tumor enlarged, with invasion to the chest wall, and metastasized to the hilar lymph node (cT3N1M0, IIIA). Case 3 was a 69-year-old male current smoker undergoing HD for 1 year, with lung squamous cell carcinoma (cT2N1M1, stage IV). All 3 patients were administered 150 mg erlotinib daily. Pharmacokinetic analyses of erlotinib were conducted on the first day (non-HD), eighth day (non-HD), and ninth day (HD) after administration[17]. For these 3 patients undergoing HD, the Cmax, Ctrough, and AUC0-24 on day 1 were 960 ± 287 ng/mL, 338 ± 116 ng/mL, and 13944 ± 4590 ng· hr/mL, respectively. On day 8 without HD, the Cmax, Ctrough, and AUC0-24 were 1638 ± 206 ng/mL, 494 ± 336 ng/mL, and 23285 ± 5338 ng· hr/mL, respectively. On day 9 (HD day), the Cmax was 1633 ± 338 ng/mL. The plasma concentrations of erlotinib before and after HD were 1465 ± 350 and 1360 ± 375 ng/mL, respectively. The pharmacokinetic parameters on the ninth day were similar to those on the eighth day, indicating that erlotinib was hardly eliminated by HD. In addition, the data of patients undergoing HD were similar to subjects with NRF. There were no serious AEs in these cases.
The data on the pharmacokinetics of erlotinib in patients undergoing HD and subjects with NRF are shown in Figure 3. These data suggested that the pharmacokinetics of erlotinib in patients undergoing HD were similar to subjects with NRF[16,17]. Since 95% of erlotinib is bound to plasma proteins[15], it was rational that most erlotinib was retained in the plasma after HD.
Afatinib is an irreversible covalent inhibitor of the Erb-B receptor tyrosine kinase family, including EGFR, Erb-B2 receptor tyrosine kinase 2/human epidermal growth factor receptor 2, and Erb-B4 receptor tyrosine kinase 4/human epidermal growth factor receptor 4[18]. It was approved by the Food and Drug Administration in 2013 for NSCLC with exon 21 L858R substitutions and exon 19 deletions[18]. Recent studies suggested that afatinib is effective against other uncommon mutations, such as L861Q in exon 21 and G719X in exon 18, and it was therefore approved by the Food and Drug Administration in 2018 for these uncommon EGFR mutations[18]. Previous studies using afatinib as the first line of treatment for NSCLC with sensitive EGFR gene mutations showed an ORR of 70%–81.8%, PFS of 13.4–15.2 mo, and OS of 27.9–49 mo[18]. The commonly reported AEs were diarrhea, skin rash, paronychia, mucosal inflammation, dry skin, stomatitis, skin fissures, nausea, dermatitis acneiform, and conjunctivitis[18]. Most AEs were of grade 1–2, and serious AEs were uncommon. However, serious AEs are more frequently reported for afatinib than for gefitinib and erlotinib[18].
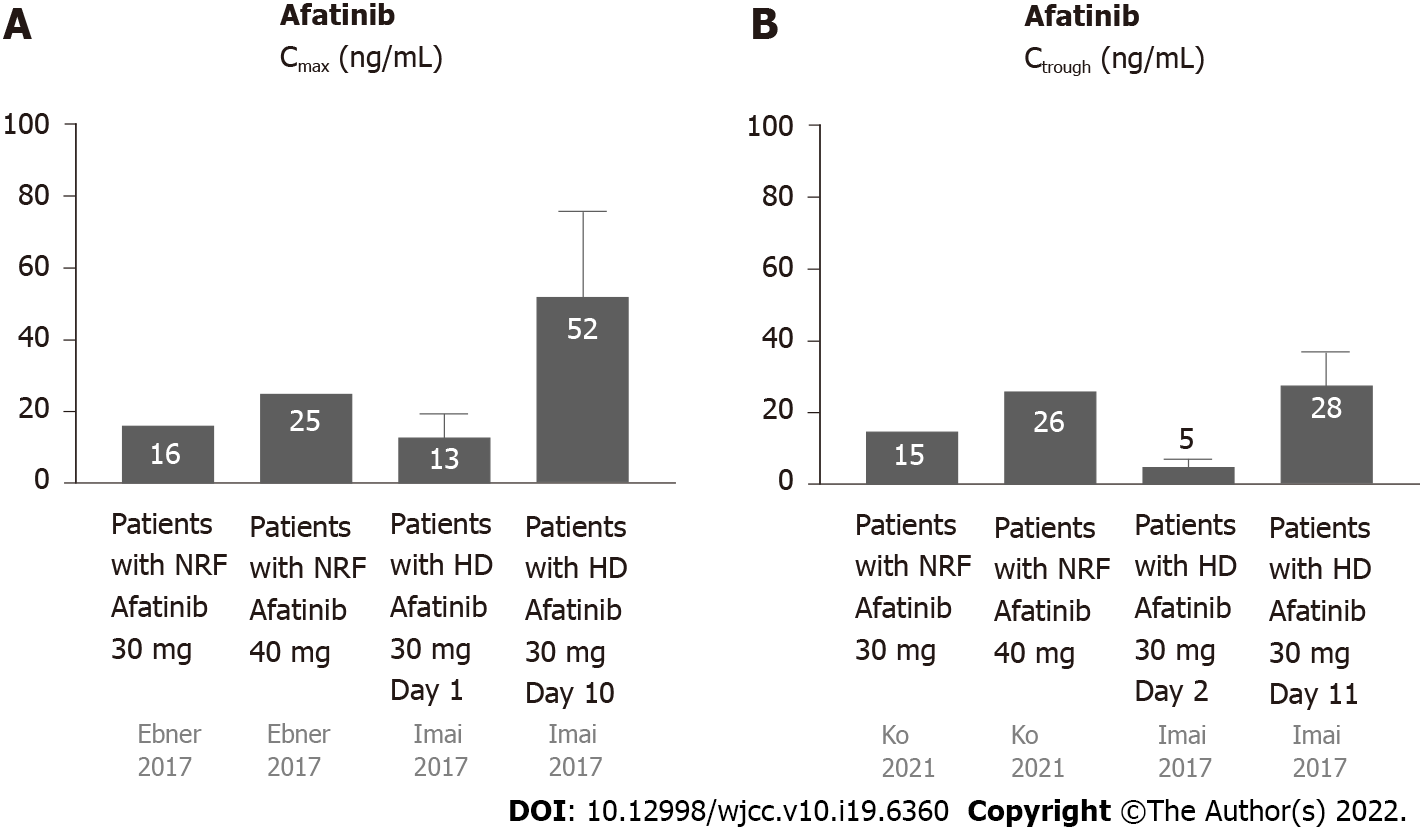
Most afatinib is excreted through feces (85%), and a small amount (15%) is excreted via urine (Figure 1C)[19]. Approximately 95% of afatinib is bound to plasma proteins[20]. The half-life periods for 30 and 40 mg afatinib are 21.3 and 26.9 h, respectively[19]. Steady-state plasma concentrations are attained in approximately 8 d[19]. The Cmax is attained approximately 2–5 h after administration[20]. For daily administration of 30 mg afatinib, the median Cmax, Ctrough, and AUC0-24 are 16.3 ng/mL, 15.1 ng/mL (8.1–38.1 ng/mL), and 189 ng· hr/mL, respectively[21,22]. For daily administration of 40 mg afatinib, the median Cmax, Ctrough, and AUC0-24 are 25.2 ng/mL, 18.2–34.1 ng/mL, and 324 ng· hr/mL, respectively[21,22].
Yamaguchi et al[23] reported a 59-year-old male ex-smoker undergoing HD for 17 years and diagnosed with stage IV lung adenocarcinoma (cT4N3M1b) with multiple pulmonary, hepatic, and bony metastases. Analysis of EGFR mutations revealed a G719A point mutation in exon 18. The patient received oral afatinib (30 mg) daily. The treatment was well tolerated, with only mild skin rash and no diarrhea. The tumors shrank after 2 mo of treatment. Bersanelli et al[8] reported afatinib as a third-line treatment for an NSCLC patient undergoing HD. Initially, the patient received cisplatin and gemcitabine (first-line) and gefitinib (second-line). After tumor progression, the patient was started on 30 mg afatinib daily. The treatment was well tolerated, with only mild asthenia. After 2 mo, the afatinib dosage was increased to 40 mg daily, which caused significant asthenia, vomiting, and nausea[8].
The United States prescribing information recommends that the dose of afatinib be decreased to 30 mg daily for subjects with renal impairment. However, this recommendation cannot be made for patients with severe renal impairment or those undergoing HD, as afatinib has not been studied in these populations[20]. Based on these two case reports, we suggest that 30 mg afatinib is safe and effective in patients undergoing HD[8,23].
Yamaguchi et al performed the first pharmacokinetic analysis of daily administration of 30 mg afatinib to patients undergoing HD[23]. The Ctrough values were 10.2 (day 2), 15.7 (day 3), 22.8 (day 10), 27.3 (day 11), and 23.8 ng/mL (day 12). Steady-state concentrations were attained by day 12. Yamaguchi et al[23] suggested that daily administration of 30 mg afatinib was safe for patients undergoing HD. Imai et al[24] also performed pharmacokinetic analysis of daily administration of 30 mg afatinib as first-line afatinib therapy for three lung cancer patients undergoing HD. Case 1 was a 78-year-old male with stage IV adenocarcinoma (cT1bN0M1b) with an EGFR mutation (exon 19 deletion). Case 2 was a 75-year-old male ex-smoker with lung adenocarcinoma (pT1aN0M0) with an EGFR mutation (exon 19 deletion) undergoing HD. However, postoperative mediastinal lymph nodes recurred. Case 3 was a 62-year-old male non-smoker with adenocarcinoma (pT2aN1M0) with an EGFR mutation (exon 21 L858R). Multiple pulmonary metastases with malignant pleural effusion occurred after the surgery. The pharmacokinetic analyses of the 3 cases revealed that the Cmax was 13.4 ± 6.2 and 51.7 ± 23.8 ng/mL on days 1 and 10, respectively, and the Ctrough was 4.5 ± 2.3 and 28.3 ± 9.0 ng/mL on days 2 and 11, respectively. The principal AEs were skin rash, dry skin, diarrhea, and malaise, and no severe AEs were reported except 1 case of grade 3 diarrhea. All the patients were maintained at partial response until the article was published (135–456 d).
The data on the pharmacokinetics of afatinib in patients undergoing HD and subjects with NRF are shown in Figure 4. Since approximately 95% of afatinib is bound to plasma proteins[25], afatinib is poorly eliminated during HD. The pharmacokinetics of afatinib in the aforementioned patients undergoing HD were comparable with subjects with NRF[25].
Osimertinib selectively and potently inhibits sensitive EGFR gene mutations[26]. Osimertinib also has a good response in NSCLC with acquired EGFR T790M resistance after treatment with first-or second-generation EGFR-TKIs.
Therefore, it is an effective first-line treatment for NSCLC patients with activating EGFR mutations, and an effective second-line treatment for NSCLC patients with T790 M resistance mutations[26]. Previous studies using osimertinib as the first line of treatment for NSCLC with sensitive EGFR gene mutations showed an ORR of approximately 80%, PFS of 18.9 mo, and OS of 38.6 mo[26]. After administration of osimertinib as the second-line treatment for NSCLC patients with T790 M resistance mutations, the ORR was approximately 71%, PFS was approximately 10.1 mo, and OS was 26.8 mo[27]. The common AEs of osimertinib were skin rash and diarrhea, and the others were paronychia, dry skin, stomatitis, nausea, anorexia, headache, constipation, anemia, pruritus, fatigue, and cough[27]. Osimertinib has fewer serious AEs than the first- and second-generation TKIs[27].
Iwafuchi et al[28] reported a 64-year-old female non-smoker undergoing HD with left lung adenocarcinoma (cT3N1M1a, stage IV, with malignant pleural effusion) with EGFR deletion in exon 19. She received gefitinib (first-line), erlotinib (second-line), taxotere (third-line), afatinib (fourth-line), and another six cycles of taxotere (fifth-line). After 5 years of treatment, the tumor was enlarged with liver and splenic metastases. For T790 M mutation, daily oral administration of 80 mg osimertinib was prescribed. The tumor improved to partial remission, and the liver and splenic metastases disappeared. The patient experienced no serious AEs and showed a good response. This case presented good effects and tolerability in patients undergoing HD even with sixth-line therapy.
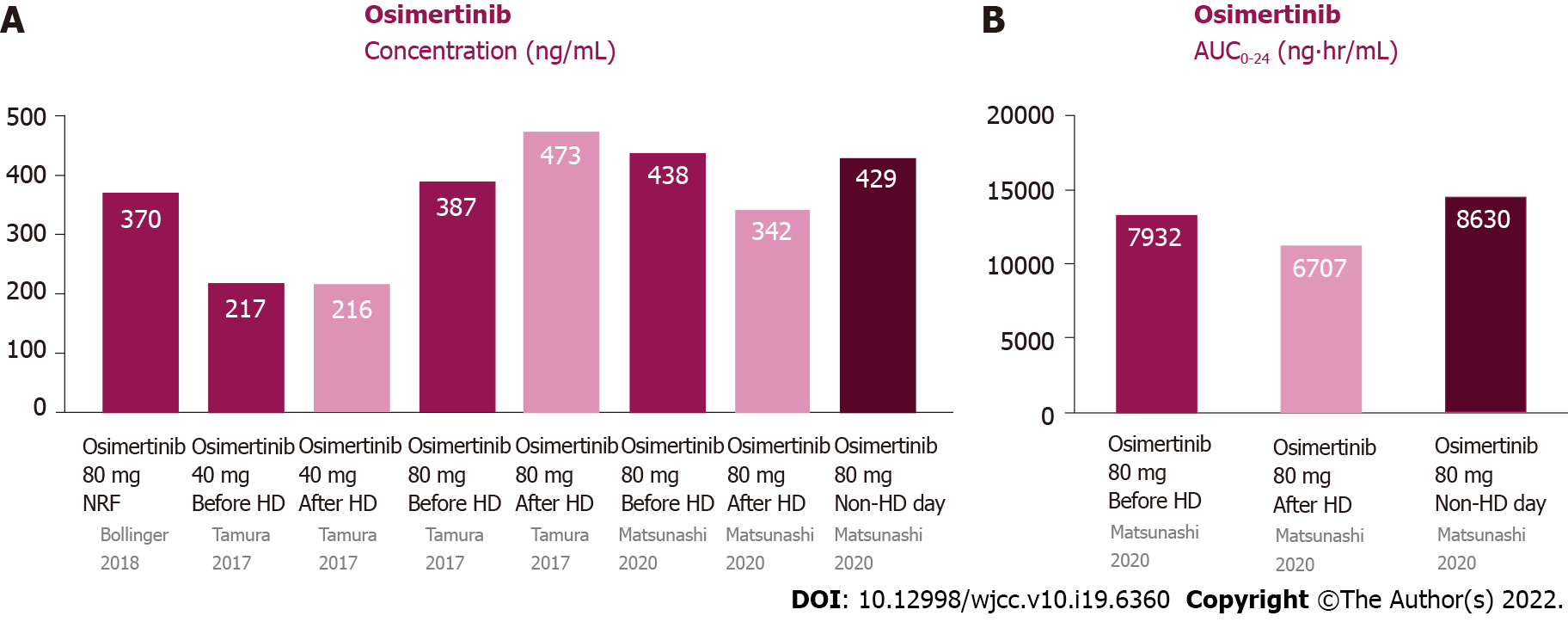
Most osimertinib is metabolized by the liver and excreted via feces (68%), and a small portion (14%) is excreted via urine (Figure 1D)[29]. The plasma protein binding ability of osimertinib is very high (98%)[30]. The half-life is 48.3 h, and steady state plasma concentrations are attained in approximately 10 d[30]. The time to attain Cmax is 6 h (3–24 h) after dosing; the Cmax and Ctrough are 371 and 230 ng/mL, respectively[31].
Tamura et al[32] reported the first pharmacokinetic analysis of osimertinib in patients undergoing HD. They reported a 72-year-old male ex-smoker with lung adenocarcinoma (T2aN3M1b, stage IV, with pleural and bone metastasis) with EGFR exon 19 deletion. He was initially receiving 250 mg gefitinib daily, but the treatment was discontinued, owing to liver toxicity, in 2 mo. After his recovery, he received 150 mg erlotinib daily and showed a good response. However, due to skin toxicity, the erlotinib dosage was gradually reduced to 50 mg daily. During the treatment, HD was initiated owing to worsening diabetic nephropathy. Four years after erlotinib treatment, the primary tumor was enlarged and had metastasized to the supraclavicular lymph nodes. The tumor tissue presented a T790M mutation in exon 20. He was initially administered 40 mg osimertinib daily for the previous liver and skin AEs of gefitinib and erlotinib. As treatment was well tolerated, pharmacokinetic analysis of osimertinib was performed 4 mo after administration[32]. Pharmacokinetic analysis of osimertinib (40 mg daily) was performed on HD and non-HD days. The Cmax was 218 and 217 ng/mL on HD and non-HD days, respectively. The osimertinib dosage was increased to 80 mg daily for only mild AEs. Pharmacokinetic analysis was performed after 6 d and revealed that the Cmax was 388 and 473 ng/mL on HD and non-HD days, respectively. The patient received 80 mg osimertinib daily, without any AEs[32]. Matsunashi et al[29] reported a 66-year-old male undergoing HD with relapsed stage IV NSCLC with an EGFR mutation in exon 21 (L858R) 2 years after body radiotherapy. He received 80 mg osimertinib daily as first-line treatment. The Cmax was 400-476 and 335–351 ng/mL before and after HD, respectively. When osimertinib was administered on HD days, the Cmax was 430 ng/mL[29].The AUC0–24 before HD, after HD and on non-HD day were 7022–8842, 6376–7039 and 8631 ng· hr/mL.
The data on the pharmacokinetics of gefitinib in patients undergoing HD and subjects with NRF are shown in Figure 5. Since the protein binding ability of osimertinib is very high, its dialyzability rate is relatively low and it is minimally affected by HD[30]. The pharmacokinetic parameters on non-HD days were almost the same as those on HD days.
This review has several limitations. Firstly, the literature cited in this review on EGFR-TKIs in patients undergoing HD are all case reports. There have been no cohorts or randomized controlled trials with EGFR-TKIs in patients undergoing HD. However, it is difficult to conduct research on HD patients owing to their rarity. Therefore, this review is very important for providing recommendations for EGFR-TKIs in such patients. Secondly, this review focuses on EGFR-TKIs in lung cancer patients undergoing HD. It is unknown whether the conclusions drawn are applicable to other cancers.
There have been no clinical studies on EGFR-TKIs in NSCLC patients undergoing HD. We suggest that EGFR-TKIs are suitable for such patients. The recommended doses and pharmacokinetics of these EGFR-TKIs for patients undergoing HD are similar to those for patients with NRF. EGFR-TKI treatment is effective and well tolerated in patients undergoing HD.
| 1. | Chang YS, Tu SJ, Chen YC, Liu TY, Lee YT, Yen JC, Fang HY, Chang JG. Mutation profile of non-small cell lung cancer revealed by next generation sequencing. Respir Res. 2021;22:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 2. | Mok Y, Ballew SH, Sang Y, Coresh J, Joshu CE, Platz EA, Matsushita K. Albuminuria, Kidney Function, and Cancer Risk in the Community. Am J Epidemiol. 2020;189:942-950. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 3. | Moon HH, Seo KW, Yoon KY, Shin YM, Choi KH, Lee SH. Prediction of nephrotoxicity induced by cisplatin combination chemotherapy in gastric cancer patients. World J Gastroenterol. 2011;17:3510-3517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 16] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 4. | Aldossary SA. Review on Pharmacology of Cisplatin: Clinical Use, Toxicity and Mechanism of Resistance of Cisplatin. BPJ. 2019;11:07-15. [RCA] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 212] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 5. | Harrison PT, Vyse S, Huang PH. Rare epidermal growth factor receptor (EGFR) mutations in non-small cell lung cancer. Semin Cancer Biol. 2020;61:167-179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 520] [Cited by in RCA: 459] [Article Influence: 76.5] [Reference Citation Analysis (0)] |
| 6. | You JHS, Cho WCS, Ming WK, Li YC, Kwan CK, Au KH, Au JS. EGFR mutation-guided use of afatinib, erlotinib and gefitinib for advanced non-small-cell lung cancer in Hong Kong - A cost-effectiveness analysis. PLoS One. 2021;16:e0247860. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 7. | Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I, Fujita Y, Okinaga S, Hirano H, Yoshimori K, Harada T, Ogura T, Ando M, Miyazawa H, Tanaka T, Saijo Y, Hagiwara K, Morita S, Nukiwa T; North-East Japan Study Group. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362:2380-2388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4530] [Cited by in RCA: 4368] [Article Influence: 273.0] [Reference Citation Analysis (0)] |
| 8. | Bersanelli M, Tiseo M, Artioli F, Lucchi L, Ardizzoni A. Gefitinib and afatinib treatment in an advanced non-small cell lung cancer (NSCLC) patient undergoing hemodialysis. Anticancer Res. 2014;34:3185-3188. [PubMed] |
| 9. | Del Conte A, Minatel E, Schinella D, Baresic T, Basso SM, Lumachi F. Complete metabolic remission with Gefitinib in a hemodialysis patient with bone metastases from non-small cell lung cancer. Anticancer Res. 2014;34:319-322. [PubMed] |
| 10. | Zhao C, Han SY, Li PP. Pharmacokinetics of Gefitinib: Roles of Drug Metabolizing Enzymes and Transporters. Curr Drug Deliv. 2017;14:282-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 11. | Hirose T, Fujita K, Kusumoto S, Oki Y, Murata Y, Sugiyama T, Ishida H, Shirai T, Nakashima M, Yamaoka T, Okuda K, Ohmori T, Sasaki Y. Association of pharmacokinetics and pharmacogenomics with safety and efficacy of gefitinib in patients with EGFR mutation positive advanced non-small cell lung cancer. Lung Cancer. 2016;93:69-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 12. | Shinagawa N, Yamazaki K, Asahina H, Agata J, Itoh T, Nishimura M. Gefitinib administration in a patient with lung cancer undergoing hemodialysis. Lung Cancer. 2007;58:422-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 13. | Luo J, Ni L, Wang M, Zhong W, Xiao Y, Zheng K, Hu P. Pharmacokinetic analysis of gefitinib in a patient with advanced non-small cell lung cancer undergoing hemodialysis. Thorac Cancer. 2016;7:251-253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 14. | Lin YT, Chen JS, Liao WY, Ho CC, Hsu CL, Yang CY, Chen KY, Lee JH, Lin ZZ, Shih JY, Yang JC, Yu CJ. Clinical outcomes and secondary epidermal growth factor receptor (EGFR) T790M mutation among first-line gefitinib, erlotinib and afatinib-treated non-small cell lung cancer patients with activating EGFR mutations. Int J Cancer. 2019;144:2887-2896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 15. | Evelina Cardoso, Guidi M, Khoudour N, Pascaline Boudou-Rouquette, Fabre E, Tlemsani C, Arrondeau J, François Goldwasser, Vidal M, Schneider MP, Wagner AD, Widmer N, Blanchet B, Csajka C. Population Pharmacokinetics of Erlotinib in Patients With Non-small Cell Lung Cancer: Its Application for Individualized Dosing Regimens in Older Patients. Clin Ther. 2020;42:1302-1316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 16. | Petit-Jean E, Buclin T, Guidi M, Quoix E, Gourieux B, Decosterd LA, Gairard-Dory AC, Ubeaud-Séquier G, Widmer N. Erlotinib: another candidate for the therapeutic drug monitoring of targeted therapy of cancer? Ther Drug Monit. 2015;37:2-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 17. | Togashi Y, Masago K, Fukudo M, Terada T, Ikemi Y, Kim YH, Fujita S, Irisa K, Sakamori Y, Mio T, Inui K, Mishima M. Pharmacokinetics of erlotinib and its active metabolite OSI-420 in patients with non-small cell lung cancer and chronic renal failure who are undergoing hemodialysis. J Thorac Oncol. 2010;5:601-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 18. | de Marinis F, Laktionov KK, Poltoratskiy A, Egorova I, Hochmair M, Passaro A, Migliorino MR, Metro G, Gottfried M, Tsoi D, Ostoros G, Rizzato S, Mukhametshina GZ, Schumacher M, Novello S, Dziadziuszko R, Tang W, Clementi L, Cseh A, Kowalski D. Afatinib in EGFR TKI-naïve patients with locally advanced or metastatic EGFR mutation-positive non-small cell lung cancer: Interim analysis of a Phase 3b study. Lung Cancer. 2021;152:127-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 19. | Stopfer P, Marzin K, Narjes H, Gansser D, Shahidi M, Uttereuther-Fischer M, Ebner T. Afatinib pharmacokinetics and metabolism after oral administration to healthy male volunteers. Cancer Chemother Pharmacol. 2012;69:1051-1061. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 111] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 20. | Keating GM. Afatinib: A Review in Advanced Non-Small Cell Lung Cancer. Target Oncol. 2016;11:825-835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 21. | Wind S, Schnell D, Ebner T, Freiwald M, Stopfer P. Clinical Pharmacokinetics and Pharmacodynamics of Afatinib. Clin Pharmacokinet. 2017;56:235-250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 147] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 22. | Ko R, Shukuya T, Imamura CK, Tokito T, Shimada N, Koyama R, Yamada K, Ishii H, Azuma K, Takahashi K. Phase I study of afatinib plus bevacizumab in patients with advanced non-squamous non-small cell lung cancer harboring EGFR mutations. Transl Lung Cancer Res. 2021;10:183-192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 23. | Yamaguchi T HH, Isogai S, Hayashi M, Uozu S, Goto Y, Nakanishi T, Sugiyama T, Itoh Y, Imaizumi K. Afatinib administration in a patient with non-small cell lung cancer harboring uncommon EGFR mutation G719A undergoing hemodialysis. Cancer Treat Commun. 2015;4:169-171. [DOI] [Full Text] |
| 24. | Imai H, Kaira K, Naruse I, Hayashi H, Iihara H, Kita Y, Mizusaki N, Asao T, Itoh Y, Sugiyama T, Minato K, Yamada M. Successful afatinib treatment of advanced non-small-cell lung cancer patients undergoing hemodialysis. Cancer Chemother Pharmacol. 2017;79:209-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 25. | Peters S, Zimmermann S, Adjei AA. Oral epidermal growth factor receptor tyrosine kinase inhibitors for the treatment of non-small cell lung cancer: comparative pharmacokinetics and drug-drug interactions. Cancer Treat Rev. 2014;40:917-926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 132] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 26. | Ramalingam SS, Vansteenkiste J, Planchard D, Cho BC, Gray JE, Ohe Y, Zhou C, Reungwetwattana T, Cheng Y, Chewaskulyong B, Shah R, Cobo M, Lee KH, Cheema P, Tiseo M, John T, Lin MC, Imamura F, Kurata T, Todd A, Hodge R, Saggese M, Rukazenkov Y, Soria JC; FLAURA Investigators. Overall Survival with Osimertinib in Untreated, EGFR-Mutated Advanced NSCLC. N Engl J Med. 2020;382:41-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2467] [Cited by in RCA: 2220] [Article Influence: 370.0] [Reference Citation Analysis (3)] |
| 27. | Liu J, Li X, Shao Y, Guo X, He J. The efficacy and safety of osimertinib in treating nonsmall cell lung cancer: A PRISMA-compliant systematic review and meta-analysis. Medicine (Baltimore). 2020;99:e21826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 28. | Iwafuchi Y, Saito I, Narita I. Efficacy and Safety of Osimertinib in a Hemodialysis Patient With Advanced Non-Small Cell Lung Cancer. Ther Apher Dial. 2017;21:416-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 29. | Matsunashi A, Fujimoto D, Hosoya K, Irie K, Fukushima S, Tomii K. Osimertinib in a patient with non-small cell lung cancer and renal failure undergoing hemodialysis: a case report. Invest New Drugs. 2020;38:1192-1195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 30. | Planchard D, Brown KH, Kim DW, Kim SW, Ohe Y, Felip E, Leese P, Cantarini M, Vishwanathan K, Jänne PA, Ranson M, Dickinson PA. Osimertinib Western and Asian clinical pharmacokinetics in patients and healthy volunteers: implications for formulation, dose, and dosing frequency in pivotal clinical studies. Cancer Chemother Pharmacol. 2016;77:767-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 113] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 31. | Bollinger MK, Agnew AS, Mascara GP. Osimertinib: A third-generation tyrosine kinase inhibitor for treatment of epidermal growth factor receptor-mutated non-small cell lung cancer with the acquired Thr790Met mutation. J Oncol Pharm Pract. 2018;24:379-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 32. | Tamura T, Takagi Y, Okubo H, Yamaguchi S, Kikkawa Y, Hashimoto I, Kaburagi T, Miura M, Satoh H, Hizawa N. Plasma concentration of osimertinib in a non-small cell lung cancer patient with chronic renal failure undergoing hemodialysis. Lung Cancer. 2017;112:225-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Critical care medicine
Country/Territory of origin: Taiwan
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Hu QD, China; Rostoker G, France S-Editor: Liu JH L-Editor: Filipodia P-Editor: Liu JH