Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.115150
Revised: October 20, 2025
Accepted: December 26, 2025
Published online: June 20, 2026
Processing time: 196 Days and 16.2 Hours
The 72-hour supervised fasting test (72-HSFT) remains the gold standard for investigating fasting hypoglycemia in patients without diabetes, which is a rare but potentially fatal clinical condition. However, testing inaccuracies often lead to diagnostic ambiguities and unnecessary healthcare expenditures. Baseline data collection over 1 year revealed that 66% of 72-HSFT tests at our institution were performed incorrectly, with dynamic hormones such as C-peptide being ordered four times per patient. The goal of this quality improvement project (QIP) was to reduce the frequency of unnecessary or incorrect 72-HSFT for hypoglycemia from 66% to 25% within 24 months. To achieve the goals of the QIP, educational inter
Core Tip: The 72-hour supervised fasting test is the cornerstone in evaluating fasting hypoglycemia, but remains vulnerable to procedural inconsistencies. This quality improvement project improved the diagnostic accuracy of the 72-hour supervised fasting test from 33.3% to 100% within two plan-do-study-act cycles. Key interventions included protocol standardization, an electronic medical records-integrated order bundle, targeted clinician and nursing education, and real-time clinical decision support prompts. These improvements were achieved without extending the length of stay, thus preserving patient flow. This project demonstrated promising sustainability and reproducibility in improving hypoglycemia evaluation, with multimodal effects on healthcare, including enhanced clinical outcomes, improved patient experiences, and potential cost reductions.
- Citation: Ata F, Khan AA, Mohammad Nofal MZ, Abdelmahmuod EA, Fawad R, Al Mohanadi D, Dabbous Z. Improving diagnostic accuracy of 72-hour supervised fasting test for hypoglycemia evaluation: A quality improvement project. World J Methodol 2026; 16(2): 115150
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/115150.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.115150
Hypoglycemia in patients without diabetes is a rare but bothersome and potentially fatal clinical condition encountered in endocrinology[1,2]. Hypoglycemia in patients without diabetes is very rare, with studies reporting a frequency of 36 per 10000 admissions[3]. However, severe hypoglycemia carries a significantly high 90-day mortality of 20.3% in patients without diabetes, compared to 1.6% in those with diabetes[4].
Hypoglycemia may result from a wide range of etiologies, including, but not limited to, insulinoma, exogenous insulin use, or ingestion of sulfonylureas. The etiologies can be broadly categorized into insulin-mediated and non-insulin-mediated[5]. It is essential to identify the exact cause of hypoglycemia, as management strategies vary significantly depending on the etiology. This requires inducing hypoglycemia under clinical supervision and then administering a set of dynamic hormones, including insulin, C-peptide, pro-insulin, and insulin-like growth factor, along with laboratory tests such as serum beta-hydroxybutyrate (BHB) and a hypoglycemic drug screen[5]. Historically, hypoglycemia could be subdivided based on its relation to meals, i.e., fasting or postprandial, to narrow down the underlying possibilities. However, more recent data show that this approach might be misleading. For instance, insulinoma, which typically causes fasting hypoglycemia, can also cause post-prandial hypoglycemia[5]. Hypoglycemia evaluation requires a precise diagnostic workup to identify the underlying cause accurately. The 72-hour supervised fasting test (72-HSFT) is the gold standard for the evaluation of mainly fasting hypoglycemia[6]. However, it has its intricacies and can be nondiagnostic, bothersome to patients, and very costly if not done properly[6]. Hypoglycemia is induced to a certain evidence-based threshold (< 3 mmol/L or 55 mg/dL) under supervised conditions, with the collection of dynamic hormones and blood tests. This process can only be helpful and diagnostic if conducted correctly and with the correct hypoglycemia threshold. However, when performed incorrectly, the test becomes not only clinically futile but also costly[6]. Despite the high sensitivity of 72-HSFT reaching > 90%, performance irregularities can render the test nondiagnostic[6,7].
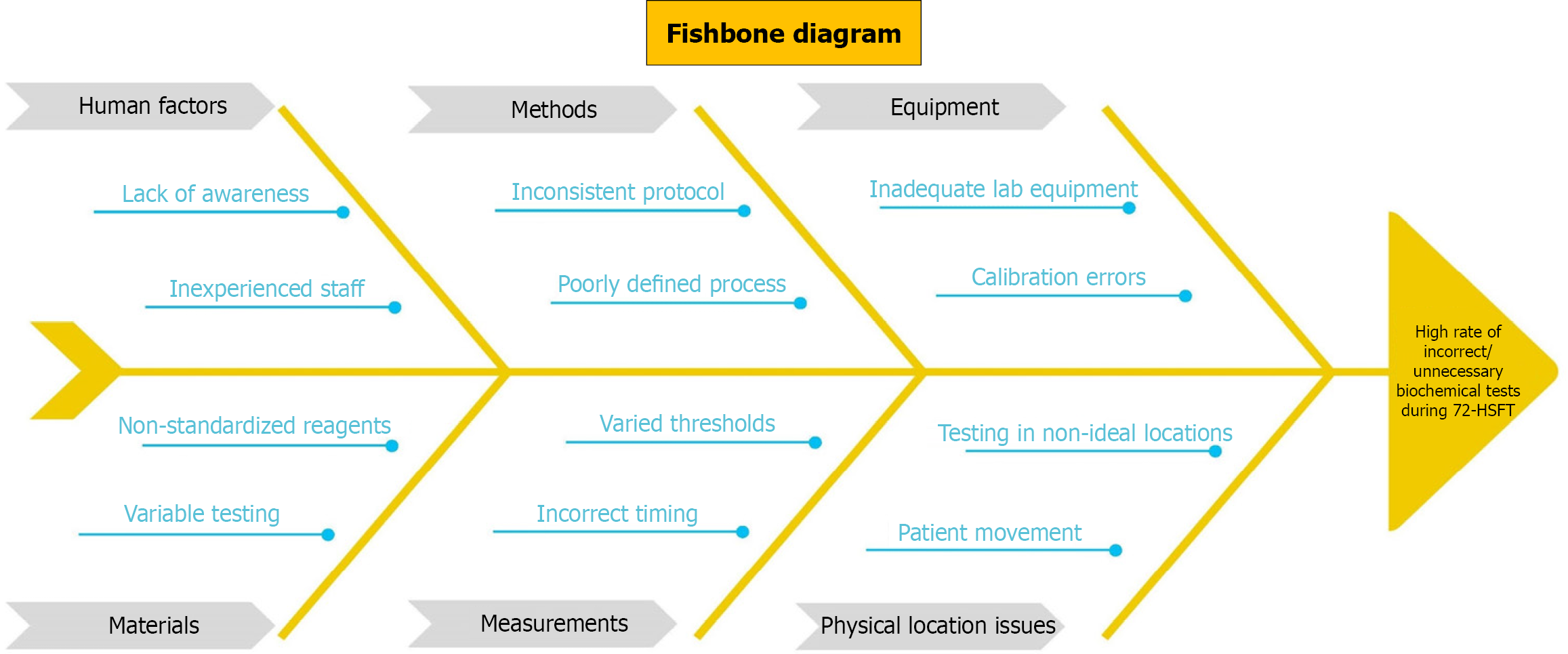
We identified various irregularities in the 72-HSFT at our institution. Despite the existence of a clinical protocol on the hospital website at our institution, an audit of 12 months’ data revealed a high error rate in protocol adherence, resulting in irregularities in the result reporting with repetitions, overuse of dynamic hormonal panels, leading to diagnostic confusion, patient discomfort, and unnecessary healthcare expenditure.
Before the implementation of specific interventions through this quality improvement project (QIP), the 72-HSFT was performed inconsistently across the emergency and medical wards of our institution. We evaluated the diagnostic performance of the 72-HSFT using baseline patient data (June 2022 to June 2023). Baseline data showed only 33.3% of the tests executed accurately. The low diagnostic accuracy risked both overdiagnosis and missed diagnoses, while inflating hospital resources. The primary aim of this QIP was to enhance the diagnostic accuracy of the 72-HSFT by reducing the percentage of unnecessary or incorrect biochemical tests from 66% to 25% over 24 months, through multiple practical system-level interventions.
This QIP was conducted in the medical wards of Hamad General Hospital (Hamad Medical Corporation), Qatar, a 600-bed academic public sector general hospital and part of a larger medical corporation that manages 16 public hospitals, all of which adhere to the same clinical guidelines and protocols. The institution is Joint Commission International certified, and the medical and endocrine training are accredited by the Accreditation Council for Graduate Medical Education International.
This QIP followed the methodology outlined in the Standards for Quality Improvement (QI) Reporting Excellence guidelines for reporting QI studies[8]. The design included baseline data collection to quantify the problem, followed by the use of a fishbone analysis to identify the key root causes of the problem. This was then followed by the imple
Adult patients admitted with hypoglycemia without diabetes and who underwent a 72-HSFT were eligible for inclusion in this QIP for baseline data collection and PDSA cycles. Patients who underwent mixed meal tests for hypoglycemia evaluation and those who had diabetes were excluded from this study.
As this was a QIP, an IRB approval was not required. The Department of Internal Medicine approved the project charter before initiating the interventions.
Using a fishbone (Ishikawa) analysis (Figure 1), we systematically categorized root causes into six categories: Human factors, methods, equipment, measurements, materials, and physical location-related issues. Among these, the most critical drivers of incorrect testing were thought to be measurement inconsistencies, such as sending hormonal panels at lower glucose thresholds and sending the tests multiple times. Additionally, human factors, particularly the lack of protocol awareness among staff and patients, were also significant factors leading to diagnostic inaccuracy. Therefore, we decided to work around these factors and design specific interventions to streamline the 72-HSFT process with a focus on minimizing human error.
Using a specific, measurable, applicable, realistic, timely statement, QIP was initiated with the introduction of a set of interventions, including patient, physician, and nursing staff education on 72-HSFT, incorporation of a hospital-wide Cerner® auto text for the 72-HSFT protocol, and addition of a hospital-wide Cerner® order set mandating to order all hormonal labs together with a single click with clinical decision support alerts guiding to send the labs only once venous blood glucose is less than 3 mmol/L.
The QIP team involved physicians from the departments of endocrinology and medicine, as well as members from the nursing department, laboratory department, and the health information technology team. The project was led by Ata F, a clinical fellow from the department of endocrinology.
To assess the diagnostic accuracy of the 72-HSFT, we collected baseline data from patients’ electronic medical records (EMR) Cerner®, who were admitted for testing in the emergency and medical wards of our institution from June 2022 to June 2023. We collected patients’ demographic details, indications for testing, initial diagnosis, the number of times C-peptide was tested per patient, frequency of other related tests (insulin, BHB, proinsulin, and sulfonylurea screen), and median length of stay (LOS) among other variables. A total of 33 patients were admitted for hypoglycemia evaluation during the defined baseline data review period, of whom nine underwent the 72-HSFT. The mean age of the participants was 34.5 years, with the majority (66.6%) being females. Only 33.3% of the tests were completed accurately, whereas the remaining 66.6% encountered various procedural challenges. These included problems such as the inappropriate timing of laboratory testing related to the glucose threshold, missing laboratory tests from the panel, and premature termination of the fasting period (Table 1).
| Variable | Results |
| Number of patients | 9 |
| Mean age (years), mean ± SD | 34.5 ± 11.7 |
| Gender | |
| Females | 6 (66.6) |
| Males | 3 (33.3) |
| Nationality | |
| Qatari | 5 (55.5) |
| Egyptian | 1 (11.1) |
| Sudanese | 1 (11.1) |
| Saudi | 1 (11.1) |
| Filipino | 1 (11.1) |
| Presumed diagnosis | |
| Possible drug induced hypoglycemia | 1 (11.1) |
| Non-specific hypoglycemia for workup | 2 (22.2) |
| Dumping syndrome with recurrent hypoglycemia | 2 (22.2) |
| Recurrent hypoglycemia for workup | 1 (11.1) |
| Possible insulinoma | 3 (33.3) |
| 72-HSFT done correctly | |
| Yes | 3 (33.3) |
| No | 6 (66.6) |
| Issues in the 72-HSFT (n = 6) | |
| Labs sent 1 hour after vein glucose reached 3 mmol/L threshold | 1 (16.6) |
| Fasting broken before finishing the test | 1 (16.6) |
| Labs sent with glucose level of 3.7 mmol/L | 1 (16.6) |
| Not done at time of hypoglycemia | 1 (16.6) |
| Done when vein glucose 39 mmol/L | 1 (16.6) |
| Not all labs sent with hypoglycemic event | 1 (16.6) |
| C-peptide (number of times ordered) | 36 (4 times per person) |
| Insulin level (number of times ordered) | 35 (3.8 times per person) |
| BHB (number of times ordered) | 35 (3.8 times per person) |
The primary outcome measure was the frequency of inaccurate testing. A test was defined as accurate if a venous glucose threshold of at least 3 mmol/L was reached, allowing for the measurement of at least serum insulin, C-peptide, and BHB. A secondary outcome measure was the number of times C-peptide was sent per patient, which would give an idea of resource utilization.
All analyses were conducted using STATA 19.5 BE.
The QI team consisted of a lead endocrinology fellow, two supporting endocrinology fellows, two internal medicine residents, and one supervising consultant from the department of endocrinology. Using the PDSA model of QI, the following interventions for change were introduced.
Interactive teaching sessions tailored to nurses and physicians in the emergency and internal medicine wards: Physi
Introduction of an EMR Cerner® auto-text protocol detailing stepwise 72-HSFT instructions available in the patients’ clinical notes: Once a patient is admitted with hypoglycemia, the 72-HSFT can start at any time point, in the emergency department, even before consulting the medical team for admission, and at other times after admission on medical floors. Hence, the involvement of different teams led to inconsistent approaches to following the 72-HSFT protocol. The purpose of incorporating a hospital-wide auto-text for the protocol was to address this issue, as the protocol should ideally be available in every progress note or, at the very least, in the admission note, allowing any team member caring for the patient to review and follow it.
Protocol added in the auto-text: Start the 72-hour fasting protocol as follows: (1) Note the date and time of the last ingestion of calories; (2) Document the baseline vital signs; (3) Stop all foods and drinks except calorie-free and caffeine-free beverages and water; (4) Ensure that the patient is active during waking hours; (5) Check finger stick blood sugar every 2 hours until blood gas is 60 mg/dL (3.3 mmol/L) or less; (6) Check finger stick blood sugar every 1 hour after blood gas is 60 mg/dL (3.3 mmol/L) or less; (7) Check blood for venous blood glucose (on a VBG machine) once the point of care glucose is 3 mmol/L or less. At the same time, collect blood samples for the following: C-peptide, insulin level, BHB, sulfonylurea level, insulin antibody, and proinsulin level; (8) Once the VBG confirms that plasma glucose is 3 mmol/L or less, the remaining samples must be sent to the lab for processing. After confirmation from the lab that the samples have been received and processed, the fast can be broken with a glucagon stimulation test; (9) Do not break the fast if venous glucose does not reach 3 or if the lab does not confirm the above, in which case the other samples should be discarded, and the above must be repeated until venous glucose is 3 or less or the above criteria are met; (10) Before breaking the fast, give 1 mg intravenous glucagon and measure glucose at 0 minute, 20 minutes, and 30 minutes: Glucagon response test. After this, the patient can resume feeding; and (11) If 72 hours have passed and the patient has not reached the target venous glucose level of 3 mmol/L, or if the patient is experiencing severe symptoms, then send all labs. The fast can be broken after the glucagon stimulation test, as described in point 10.
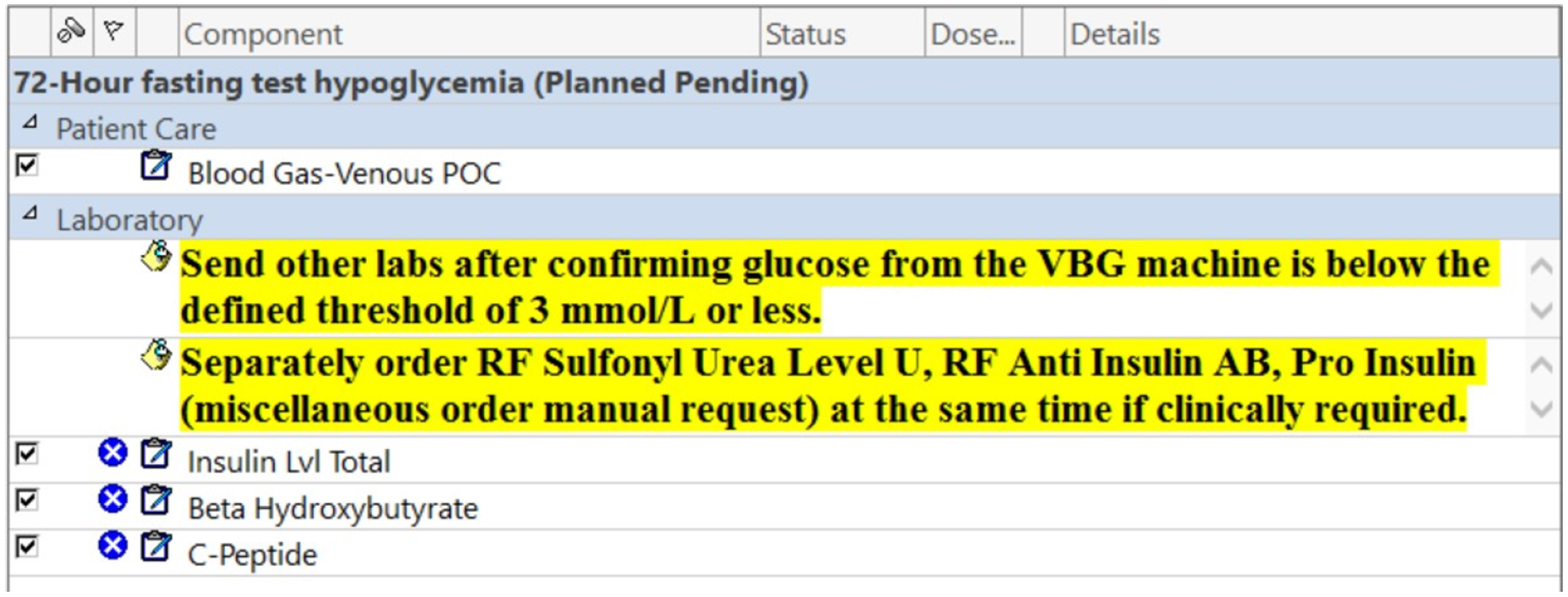
A standardized biochemical order set was developed to streamline test ordering during the 72-hour fasting test: This bundled order included serum insulin, BHB, C-peptide, and venous blood glucose measured via VBG. All tests were integrated into a single click within the Cerner®. To enhance adherence to protocol, two electronic alert prompts were embedded: A warning to only send the labs when the bedside VBG confirms a venous glucose level of ≤ 3.0 mmol/L. A secondary prompt reminds clinicians to order sulfonylurea screening, proinsulin, and insulin antibodies separately only if clinically indicated, as these tests are sent externally and are not part of the routine default testing (Figure 2).
As a reinforcement strategy, a mandatory endocrinology consult was implemented for all patients admitted for the 72-HSFT. This allowed the QI team to conduct retrospective, post-discharge feedback reviews of the cases after intervention without influencing real-time clinical decision-making.
To address the inconsistencies in the performance of the 72-HSFT at our institution, we used a structured approach using two PDSA cycles following the Standards for QI Reporting Excellence reporting guidelines for QI studies[8]. Before initiating interventions, a 12-month retrospective review of baseline data (June 2022 to June 2023) revealed the infrequent nature of the testing. Hence, we decided to run each PDSA cycle for six months to capture at least three cases, allowing sufficient time for intervention implementation and evaluation.
Plan: The first PDSA cycle focused on reducing unnecessary C-peptide and related hormonal panel testing and enhancing protocol adherence during the 72-HSFT. Interventions that were successfully implemented included patient and staff education, the introduction of a Cerner auto-text protocol detailing step-by-step instructions to follow, as well as the implementation of a bundle order set for insulin, C-peptide, BHB, and venous glucose.
Do: These interventions were implemented hospital-wide. The QI team retrospectively reviewed the patients to evaluate the implementation.
Study: Five patients were evaluated during this cycle. The median age was 29 years (range 20-35), and the LOS was 4 days (interquartile range 4-5). Test adherence improved to 80% (4 out of 5 tests executed correctly). C-peptide orders dropped to 1.2 per patient (from 4.0 at baseline). An issue that still persisted was the early termination of fasting and the sending of laboratory workups at a glucose level above the set threshold in the protocol, underscoring the need for a stronger emphasis on glucose thresholds and more education for patients and staff.
Act: The QI team reinforced the glucose threshold for sending laboratory workups through more education. A lecture on correct 72-HSFT was delivered during medical grand rounds, attended by corporation-wide faculty and trainees in the Department of Medicine. Concerns that physicians had regarding glucose thresholds and other nuances of the 72-HSFT were addressed in a detailed question and answer session following the lecture.
Plan: Cycle 2 focused on sustaining the improvement rates achieved and further reducing testing inaccuracies. It continued all previous interventions with an emphasis on protocol adherence and use of EMR prompts and order sets.
Do: The same QI structure remained in place. Education was refreshed mid-cycle with a PowerPoint presentation in morning reports, and the QI team continued to monitor the testing retrospectively.
Study: Three patients underwent 72-HSFT in the pre-defined period of the second PDSA cycle. The median age was 26 years (range, 25-42), and the LOS remained stable at 4 days. All three tests (100%) were conducted correctly, and C-peptide testing was reduced to one test per patient. No premature fast terminations or sampling errors were documented.
Act: Given the remarkable improvement, the interventions were deemed adequate and the QIP successful. The QI team submitted recommendations to the health information technology team for the formal adoption of auto-text, standardized bundle order sets, and Cerner® prompts corporation-wide to enhance effective test performance.
The LOS was selected as a balancing measure to ensure that efforts to standardize and streamline the 72-HSFT protocol did not inadvertently prolong hospitalization. An increase in the LOS could suggest that more stringent test performance might delay discharge or create hurdles in patient flow. On the contrary, a stable or decreased LOS would support the idea that our interventions can improve the diagnostic efficiency of the 72-HSFT without compromising patient flow. Throughout the PDSA cycles, the median LOS remained stable at 4 days (one day less than the baseline LOS), indicating that the implementation of interventions to improve the diagnostic accuracy of the 72-HSFT did not adversely affect patients’ hospitalization and flow.
The 72-HSFT is the gold standard diagnostic test for evaluating fasting hypoglycemia. However, in many centers, protocol deviations due to multifactorial reasons significantly impact the diagnostic yield of the test. This QIP is a pioneer effort from the region to demonstrate significant measurable improvements in the performance of the 72-HSFT through multiple targeted interventions (patient and staff education, a standardized bundle order set for laboratory workup, electronic prompts, and protocol embedded in the patient notes).
The frequency of correct testing improved markedly from baseline through the two PDSA cycles. The primary aim of reducing the testing inaccuracy to 25% was achieved successfully in both cycles, with inaccuracy reaching 0% by the end of cycle 2 (Table 2). C-peptide testing frequency dropped by 75%, demonstrating enhanced efficiency without com
| Cycle | n | Median age (years) | Median LOS | Correct test (%) with 95%CI1 | C-peptide (total) | Average C-peptide per patient |
| Baseline | 9 | 34.5 (mean) | 5 (4-6) | 33.3 (12.1-64.6) | 36 | 4.0 |
| PDSA 1 | 5 | 29 (20-35) | 4 (4-5) | 80 (37.6-96.4) | 6 | 1.2 |
| PDSA 2 | 3 | 26 (25-42) | 4 (4-4) | 100 (43.9-100) | 3 | 1.0 |
This QIP highlights the importance of system-level interventions in improving the diagnostic accuracy of dynamic testing in endocrine medicine. This QIP also highlighted the blend of traditional and advanced interventions in QIPs. The initial overuse of C-peptide and related tests was a consequence of multiple-level errors in the testing, which required more than one-dimensional interventions. By embedding order sets, decision-support tools, and patient and staff education into practice, we not only improved test precision but also reduced unnecessary investigations. Wider adoption of similar interventions in other dynamic endocrine testing can enhance diagnostic quality and optimize institutional policies and best-practice guidelines.
Limitations include small sample sizes and a limited number of PDSA cycles due to the rarity of the condition under study, as well as a single-center design. Seasonal staff turnover and patient mix may have introduced confounders. A small number of patients in this QIP is largely due to the intrinsic rarity of fasting hypoglycemia in patients without diabetes, limiting the number of eligible cases during the pre-defined study periods. Despite this, our QIP methodology prioritizes measurability, achievability, and sustainability of a system change over statistical power. A sustained improvement across two cycles, as demonstrated in this study, suggests that our interventions were not only potentially impactful but also reproducible. Furthermore, given that the 72-hour fasting protocol is standard in most centers, the findings are potentially generalizable to other institutions using similar testing methodologies. This QIP represents an initial phase, with additional longer cycles planned for even broader and more sustained evaluation, with the addition of data relevant to healthcare resource utilization, such as cost analysis.
A 72-HSFT remains of high diagnostic value in the investigation of fasting hypoglycemia in patients without diabetes. However, inconsistencies in the testing methodologies render it nondiagnostic in many circumstances due to various reasons. Through structured interventions involving patient and staff education, protocol incorporation, and the addition of bundle order sets in the EMR, the performance of the 72-HSFT for fasting hypoglycemia evaluation was significantly enhanced. The changes demonstrated in this QIP are sustainable, easily implementable, and expandable. However, it is worth noting that the sample size in this QIP was small due to the rarity of the condition. Future work will include cost-reduction analysis, staff and patient satisfaction surveys, and adaptation to other dynamic endocrine tests.
| 1. | Elghobashy M, Gama R, Sulaiman RA. Investigation and Causes of Spontaneous (Non-Diabetic) Hypoglycaemia in Adults: Pitfalls to Avoid. Diagnostics (Basel). 2023;13:3275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 2. | Davis SN, Duckworth W, Emanuele N, Hayward RA, Wiitala WL, Thottapurathu L, Reda DJ, Reaven PD; Investigators of the Veterans Affairs Diabetes Trial. Effects of Severe Hypoglycemia on Cardiovascular Outcomes and Death in the Veterans Affairs Diabetes Trial. Diabetes Care. 2019;42:157-163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 93] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 3. | Nirantharakumar K, Marshall T, Hodson J, Narendran P, Deeks J, Coleman JJ, Ferner RE. Hypoglycemia in non-diabetic in-patients: clinical or criminal? PLoS One. 2012;7:e40384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 4. | Tsujimoto T, Yamamoto-Honda R, Kajio H, Kishimoto M, Noto H, Hachiya R, Kimura A, Kakei M, Noda M. Prediction of 90-day mortality in patients without diabetes by severe hypoglycemia: blood glucose level as a novel marker of severity of underlying disease. Acta Diabetol. 2015;52:307-314. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 5. | Palani G, Stortz E, Moheet A. Clinical Presentation and Diagnostic Approach to Hypoglycemia in Adults Without Diabetes Mellitus. Endocr Pract. 2023;29:286-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 6. | Farahmand A, Kerr JM, Lawler HM. Maximizing The Utility Of The 72-Hour Fast In Evaluating Hypoglycemia. Endocr Pract. 2021;27:80-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 7. | Kelz RR, Schwartz TA, Haut ER. SQUIRE Reporting Guidelines for Quality Improvement Studies. JAMA Surg. 2021;156:579-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 8. | Ogrinc G, Davies L, Goodman D, Batalden P, Davidoff F, Stevens D: SQUIRE 2. 0 (Standards for QUality Improvement Reporting Excellence): revised publication guidelines from a detailed consensus process. BMJ Qual Saf. 2016;25:986-992. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2333] [Cited by in RCA: 2074] [Article Influence: 207.4] [Reference Citation Analysis (0)] |