Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.113663
Revised: September 20, 2025
Accepted: November 26, 2025
Published online: June 20, 2026
Processing time: 234 Days and 21.2 Hours
Little information exists on the amount or type of training required for diaphragm ultrasound (DUS) competency.
To determine the number of directly-supervised DUS cases required for DUS-naive trainees to achieve competency for independent practice.
A prospective observational study was conducted in the intensive care unit, among trainees without prior DUS experience. Trainees completed a theoretical module and questionnaire, then performed a standardized DUS protocol on both right and left hemi-diaphragms under supervision. Trainees were tasked to determine: (1) Diaphragm thickening fraction (DTF), as a percentage difference of the diaphragm thickness at end-inspiration compared to thickness at end-expi
Thirteen trainees (mean age 29.5 years; 7 females) scanned 5 patients each, whereby each trainee would have acquired 150 hemi-diaphragm images for 20 sets of DTF measurements and 10 sets of DE measurements. Across 65 supervised assessments on 52 patients, measurement agreement improved with repeated attempts, particularly for DTF, reaching near 100% by the fifth attempt. Interpretation agreement was consistently high (> 80%) from the start and also approached 100% with training. However, some trainees still required assistance with left diaphragm assessments, especially excursion.
Our training approach allowed DUS-naive trainees to achieve adequate skills to perform unsupervised DUS evaluation of the right hemi-diaphragm after at least 5 patient encounters. For left DTF and DE, some trainees require more supervised scans to achieve competency for independent practice.
Core Tip: We performed a prospective observational study to determine the number of directly-supervised diaphragm ultrasound (DUS) cases required for DUS-naive trainees to achieve competency. Thirteen trainees completed a theoretical module and questionnaire, then performed a standardized DUS protocol on both right and left hemi-diaphragms under supervision. Our training approach allowed DUS-naive trainees to achieve adequate skills to perform unsupervised right DUS evaluation over a relatively short training period which included at least 20 sets of supervised measurements to assess diaphragm thickening fraction, and at least 10 sets of supervised measurements to assess diaphragm excursion. A higher number of supervised scans may be needed for left DUS training.
- Citation: Ng KWP, See KC. Basic diaphragm ultrasound training: Curriculum implementation and learning trajectory among diaphragm ultrasound naive trainees. World J Methodol 2026; 16(2): 113663
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/113663.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.113663
Diaphragm ultrasound (DUS) has clinical use in the evaluation of critically-ill patients due to its utility in serial radiation-free bedside evaluations of diaphragmatic thickness, thickening fraction and excursion[1]. It has been used in the diagnosis and prognostication of patients with respiratory failure from diaphragmatic dysfunction by providing useful additional information to fluoroscopy and electromyography[2], and has also been used in critically-ill, mechanically ventilated patients in predicting successful extubation[3-5]. However, ultrasound is an operator-dependent skill, and adequate training is a pre-requisite for effective clinical use. It has been shown that trainees who undergo practical training have more accurate DUS assessments compared to when they undergo a theoretical module alone[6]. While there have been some studies evaluating the training approach for cardiac and lung ultrasound assessments[7-10], there has been no standardized training approach for DUS. Little information exists on the efficacy of a training approach for independent DUS, and on how much practical training is required for independent practice by a trainee without sono
We therefore aimed to study the learning trajectory (image acquisition, measurements and interpretation), to determine a threshold number of directly-supervised cases above which competency for independent practice may be achieved. Our hypothesis is that a minimum threshold number of directly-supervised cases is required to be performed by a DUS-naive trainee before being competent for independent practice.
We conducted a prospective observational study of DUS training from July 2020 to July 2021 in our medical intensive care unit (ICU) within the National University Hospital. The study was approved by the local ethics committee (approval No. 2020/00177) and written informed consent was obtained from the trainee participants. DUS screening was to be done for patients admitted to the ICU who required monitoring with or without resultant invasive or non-invasive ventilation for their respiratory parameters. We aimed to recruit voluntary trainees with no experience of DUS among medical residents and respiratory therapists. These trainees would be recruited opportunistically when doing their training or job rotations through ICU. Two trainers were designated (both authors). The first author (Ng KWP) has 7 years of neuromuscular ultrasound experience with 5 years of DUS experience in an ICU and outpatient setting and is a trainer in her hospital in DUS. The other author (See KC) has 16 years of critical care ultrasound experience with a 5-year experience of DUS in an ICU setting and is a trainer in his hospital in thoracic and DUS.
The DUS curriculum was created by the authors. A set of training slides on the performance was given to the trainees. This deck of slides focused on key learning concepts of the DUS technique, including use of ultrasound equipment, probe positioning, acoustic windows, anatomical landmarks for DUS, and calculation and interpretation of normality. These concepts were directed to allow trainees to rapidly develop DUS skills within a busy clinical environment by providing practical theoretical knowledge.
The trainees were also asked to complete a questionnaire consisting of 15 multiple-choice questions before and after reading the slides. Results of the pre-test and post-test were recorded, but the results of the post-test were not used to determine progression to the practical training, as the tested concepts would be reiterated by the trainers during the practical training period. Course trainees were given up to 1 month for self-learning using the deck of training slides and they were commenced on practical training of DUS within 1 week of the post-test.
Trainees in the practical training were trained by either of the expert trainers (Ng KWP or See KC), where they received in-person explanations and an initial interactive demonstration on how to perform DUS. Trainees were then asked to independently perform DUS on both right and left diaphragms. All measurements were performed independently by the trainees after images’ acquisition and storage. The trainer would be present in the room during this acquisition and would record the trainees’ measurements. The trainer would perform his own DUS and acquire and record his own measurements if he judged the images or measurements were incorrectly acquired. The supervisor would render assistance for image acquisition whenever the trainee struggled for more than a minute in obtaining images. While blinding of the trainers and trainees were not possible, we also avoided looking at the clinical notes of the imaged patients till ultrasound image acquisition and interpretation was completed.
Either of the following ultrasound devices was used: SparqTM Ultrasound System (Philips Healthcare, Andover, MA, United States), and GE LOGIQ eTM Ultrasound System (GE HealthCare, Chicago, IL, United States). These were equipped with a low frequency (1-5 MHz) phased array probe, as well as with a high frequency (4-13 MHz) linear array probe.
Under direct supervision, trainees carried out sonographic evaluation on both right and left hemi-diaphragms on the patient lying in a minimally elevated position, as previously described[11-13]. Trainees were tasked to ascertain 2 main parameters of the diaphragm function: (1) Diaphragm thickening fraction (DTF) calculated from diaphragm thickness; and (2) Diaphragm displacement for diaphragm excursion (DE). Imaging for the diaphragm thickness and excursion would be done with the patient on their current ventilatory settings if ventilated.
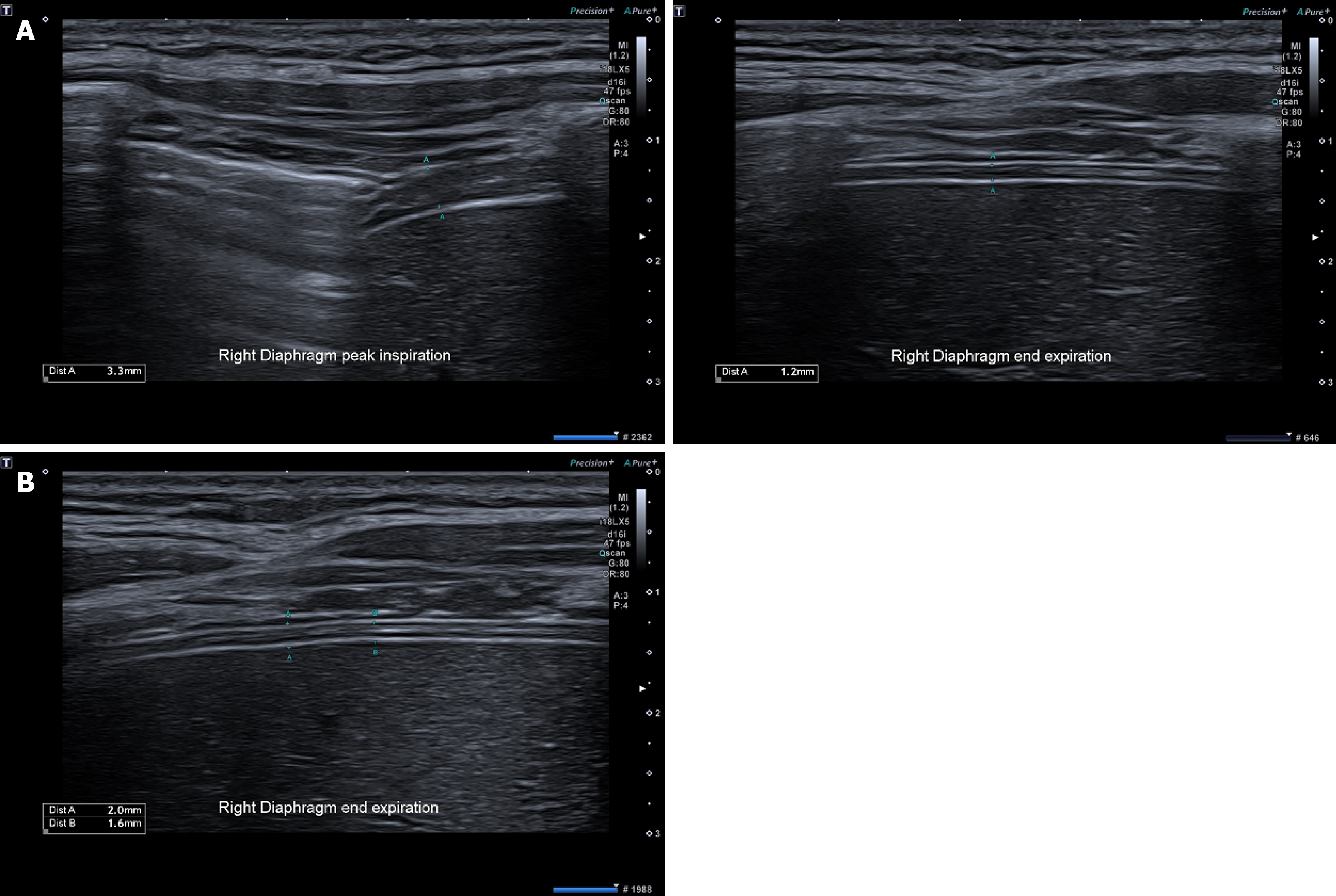
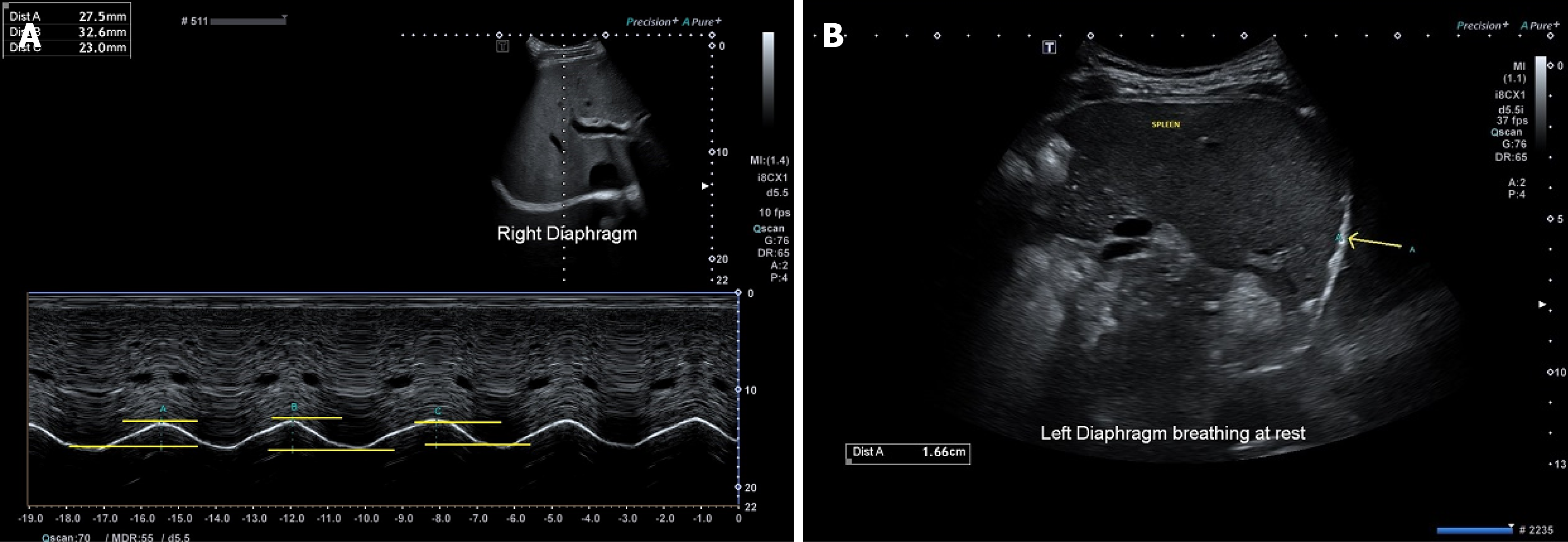
For diaphragm thickness, a high frequency linear array transducer was used to directly visualize the diaphragm’s thickness at its zone of apposition, and then again at the mid-axillary line, using B-mode ultrasound. The surface landmark of the zone of apposition is along the antero-axillary line at the level of the 9th or 10th intercostal space, and the diaphragm was identified as a 3-layered structure with the pleural and peritoneal membranes above and below respectively. Image and measurement acquisition was deemed satisfactory when the diaphragm was visible between the diaphragmatic pleural line and the peritoneal line, and measurement calipers placed within the two lines and not including the two lines into the total thickness (Figure 1). The trainee would need to determine the diaphragmatic thickness at end-inspiration and end-expiration on B-mode and acquire and store the relevant images. The 3 consecutive measurements of the diaphragmatic thickness at both end-inspiration and end-expiration would be obtained and averaged to calculate the DTF. The trainee would derive the DTF (%), calculated as (thickness at end-inspiration - thickness at end-expiration) divided by thickness at end-expiration × 100 (%), as previously described[11,12].
DE was evaluated by measuring the extent of diaphragm displacement. For DE, a low frequency phased array ultrasound transducer is placed below the right and left costal margins and directed towards the respective diaphragms to identify the diaphragms as a hyperechogenic curve. The M-mode (“time-motion” mode) is then used along the selected line to show the displacement of the hemi-diaphragms during the respiratory phases, and this displacement can be measured as the amount of diaphragmatic excursion, with the measurement calipers placed at the trough of the end-expiration phase, and at the peak of the inspiration slope[11,13]. For DE, the trainee would need to identify the hyper
The 3 consecutive measurements of diaphragm thickness at inspiration and expiration at both zone of apposition and mid-axillary line would result in a total of 12 images of diaphragm thickness for the right hemi-diaphragm and 12 images for the left hemi-diaphragm per patient. The 3 consecutive measurements of DE would result in 3 images for right hemi-diaphragm and 3 images for the left hemi-diaphragm per patient. Therefore, each trainee would acquire a total of 150 hemi-diaphragm images for 20 sets of DTF measurements and 10 sets of DE measurements, from 5 patient encounters.
Diaphragm dysfunction in the patient was determined to be present if: (1) DE < 10 mm[3,14]; or (2) If DTF < 20%[15-17]. Based on literature review[6], and agreement between the two expert trainers, trainee measurements of the diaphragm displacement in DE and DTF were considered to be in agreement with the trainers’ measurements if they satisfied the following criteria: (1) Diaphragm displacement on DE ± 2 mm from the value derived by the trainer; and (2) DTF ± 10% of the similar measurement derived by the trainer, respectively. We also compared the interpretation of the presence of diaphragm dysfunction by both trainer and trainee. The trainee was considered to have an overall pass of each parameter if he or she had both: (1) Measurements in agreement with the trainers; and (2) Similar interpretation of the presence of diaphragm dysfunction as the trainer.
Pre-test and post-test scores were compared with the Wilcoxon signed rank test. Data is presented as mean ± SD or as median (interquartile range). The Wilcoxon signed rank test was used to compare non-parametric matched data, while Fisher exact test was used to compare categorical data. The paired t test was used to compare normally-distributed paired measurements. Statistical significance was taken as P < 0.05.
Thirteen trainees completed the theoretical training (which included the self-reading of training slides, and multiple-choice questionnaire test before and after the self-reading), and the practical portion of training on patients admitted to the ICU. Of this, 6 trainees were male, 7 were female, mean age was 29.5 ± 2.0 years old. 5 trainees had no prior sonographic experience, while 8 had prior ultrasound experience mainly in a critical care setting (7 having 2 or less years of experience, while 1 had 6 years of experience). None of them had prior experience with DUS. 6 trainees were physiotherapists, 2 were internal medicine residents, 5 were fellows (4 were fellows in neurology, 1 was a fellow in respiratory and critical care medicine). They had a median of 6 years of work experience (range 2-10 years).
A Wilcoxon signed rank test indicated that the median post-reading multiple-choice question scores (median = 11) were significantly higher than the median pre-reading multiple-choice question scores (median = 6), P = 0.00148. In total, the trainees imaged 52 patients. Some patients had repeated imaging, and so the patient characteristics presented in Table 1 refer to the separate occasions upon which the trainees were assessed (n = 65), with trainees attempting on 5 patients each. Ng KWP supervised and reviewed 46 (70.8%) of the cases, while the remaining (n = 19, 29.2%) were supervised and reviewed by See KC. Analyses of the trainer and trainees’ assessment of right DE, left DTF at the mid-axillary line, and left DE were performed only for 12 trainees each due to missing data, when local factors resulted in an inability to visualize the diaphragm adequately.
| Characteristics | |
| Age (years) | 57.5 ± 14.0 |
| Gender | Male: 49 (75.4); female: 16 (24.6) |
| BMI (kg/m2) | 26.2 ± 9.3 |
| Primary diagnosis | Gastrointestinal bleed: 1 (1.5); IHD/cardiac failure: 6 (9.2); ischemic stroke: 23 (35.4); intracranial events other than ischemic stroke (CVT, ICH, abscess, status epilepticus): 12 (18.5); myasthenia gravis: 2 (3.1); metabolic abnormality (hypokalaemia, myxoedema coma): 2 (3.1); polytrauma: 1 (1.5); pneumonia: 17 (26.2); urosepsis: 1 (1.5) |
| Presence of neuromuscular condition | 4 (6.2)1 |
| Respiratory rate (median, range) | 20 (13-39) |
| Ventilation modes | Nil ventilation 26 (40); non-invasive ventilation 5 (7.7); invasive ventilation 34 (52.3) |
Table 2 shows the median (interquartile range) diaphragmatic parameters of DTF (%) and DE (cm) for both trainer and trainee. None of the median measurements had a significant difference between the trainer or the trainee. There was also no significant difference in the proportion of DUS that were interpreted as normal by both trainer and trainee (Table 3).
| Try 1 | Try 2 | Try 3 | Try 4 | Try 5 | ||
| Right DTF ZOA (%, n = 13) | Trainer | 40.9 (31.2-46.3) | 27.3 (11.3-32.1) | 26.7 (14.2-38.1) | 17.8 (15.1-38.7) | 30.4 (28.4-42.0) |
| Trainee | 29.9 (24.9-52.2) | 27.5 (6.7-36.7) | 25.0 (12.7-28.9) | 16.3 (5.8-38.7) | 29.2 (28.4-42.0) | |
| P value | 0.66 | 0.76 | 0.06 | 0.68 | 0.34 | |
| Right DTF mid-axillary (%, n = 13) | Trainer | 38.0 (22.4-77.5) | 19.3 (7.0-24.6) | 20.6 (14.2-26.1) | 31.6 (23.6-55.3) | 24.4 (21.6-41.2) |
| Trainee | 53.9 (13.1-79.9) | 19.6 (11.0-27.7) | 21.4 (12.8-26.1) | 31.6 (15.7-55.3) | 24.4 (21.6-41.2) | |
| P value | 0.13 | 0.43 | 0.87 | 0.22 | 1 | |
| Right diaphragm excursion (cm, n = 12) | Trainer | 1.27 (1.04-1.82) | 1.07 (0.66-1.45) | 0.96 (0.81-1.45) | 0.88 (0.83-1.38) | 1.18 (1.04-1.40) |
| Trainee | 1.16 (0.92-1.84) | 1.03 (0.66-1.36) | 1.02 (0.82-1.45) | 0.88 (0.70-1.45) | 1.18 (1.04-1.40) | |
| P value | 0.09 | 0.19 | 0.19 | 0.49 | 1 | |
| Left DTF ZOA (%, n = 13) | Trainer | 25.3 (19.4-72.4) | 34.6 (26.1-47.1) | 33.3 (23.1-35.3) | 31.0 (15.4-48.4) | 29.9 (15.3-37.8) |
| Trainee | 56.2 (14.8-72.4) | 34.6 (21.1-53.0) | 33.3 (27.7-38.9) | 28.6 (15.4-42.3) | 29.9 (15.3-37.8) | |
| P value | 0.31 | 0.78 | 0.55 | 0.54 | 0.34 | |
| Left DTF mid-axillary (%, n = 12) | Trainer | 32.5 (15.2-53.0) | 21.5 (14.6-40.2) | 22.9 (10.5-37.9) | 31.3 (24.0-39.1) | 24.0 (17.6-36.5) |
| Trainee | 20.5 (15.1-53.0) | 18.1 (7.8-41.3) | 18.9 (8.8-37.9) | 29.0 (22.8-39.1) | 24.0 (13.6-36.5) | |
| P value | 0.48 | 0.55 | 0.17 | 0.34 | 0.34 | |
| Left diaphragm excursion (cm, n = 12) | Trainer | 1.20 (1.11-1.58) | 1.16 (0.83-2.17) | 1.19 (0.94-1.34) | 1.01 (0.77-1.12) | 1.14 (1.05-1.31) |
| Trainee | 1.24 (1.11-1.60) | 1.15 (0.83-2.17) | 1.19 (0.89-1.34) | 1.01 (0.77-1.12) | 1.15 (1.07-1.42) | |
| P value | 0.71 | 0.25 | 0.34 | 0.34 | 0.34 |
| Try 1 | Try 2 | Try 3 | Try 4 | Try 5 | ||
| Right DTF at ZOA (n = 13) | Trainee with normal interpretation | 13 (100) | 8 (61.5) | 8 (61.5) | 5 (38.5) | 11 (84.6) |
| Trainer with normal interpretation | 12 (92.3) | 8 (61.5) | 7 (53.8) | 6 (46.2) | 11 (84.6) | |
| P value | 1 | 1 | 1 | 1 | 1 | |
| Right DTF at mid-axillary line (n = 13) | Trainee with normal interpretation | 8 (61.5) | 6 (46.2) | 7 (53.8) | 9 (69.2) | 10 (76.9) |
| Trainer with normal interpretation | 10 (76.9) | 6 (46.2) | 7 (53.8) | 10 (76.9) | 10 (76.9) | |
| P value | 0.6728 | 1 | 1 | 1 | 1 | |
| Right excursion (n = 12) | Trainee with normal interpretation | 7 (58.3) | 6 (50.0) | 6 (50.0) | 5 (41.7) | 9 (75.0) |
| Trainer with normal interpretation | 9 (75.0) | 8 (66.7) | 6 (50.0) | 5 (41.7) | 9 (75.0) | |
| P value | 0.6668 | 0.6802 | 1 | 1 | 1 | |
| Left DTF at ZOA (n = 13) | Trainee with normal interpretation | 9 (69.2) | 10 (76.9) | 10 (76.9) | 9 (69.2) | 9 (69.2) |
| Trainer with normal interpretation | 9 (69.2) | 11 (84.6) | 10 (76.9) | 9 (69.2) | 9 (69.2) | |
| P value | 1 | 1 | 1 | 1 | 1 | |
| Left DTF at mid-axillary line (n = 12) | Trainee with normal interpretation | 6 (50.0) | 6 (50.0) | 6 (50.0) | 10 (83.3) | 7 (58.3) |
| Trainer with normal interpretation | 7 (58.3) | 8 (66.7) | 7 (58.3) | 11 (91.7) | 8 (66.7) | |
| P value | 1 | 0.6802 | 1 | 1 | 1 | |
| Left excursion (n = 12) | Trainee with normal interpretation | 10 (83.3) | 6 (50.0) | 8 (66.7) | 6 (50.0) | 10 (83.3) |
| Trainer with normal interpretation | 10 (83.3) | 6 (50.0) | 8 (66.7) | 6 (50.0) | 10 (83.3) | |
| P value | 1 | 1 | 1 | 1 | 1 |
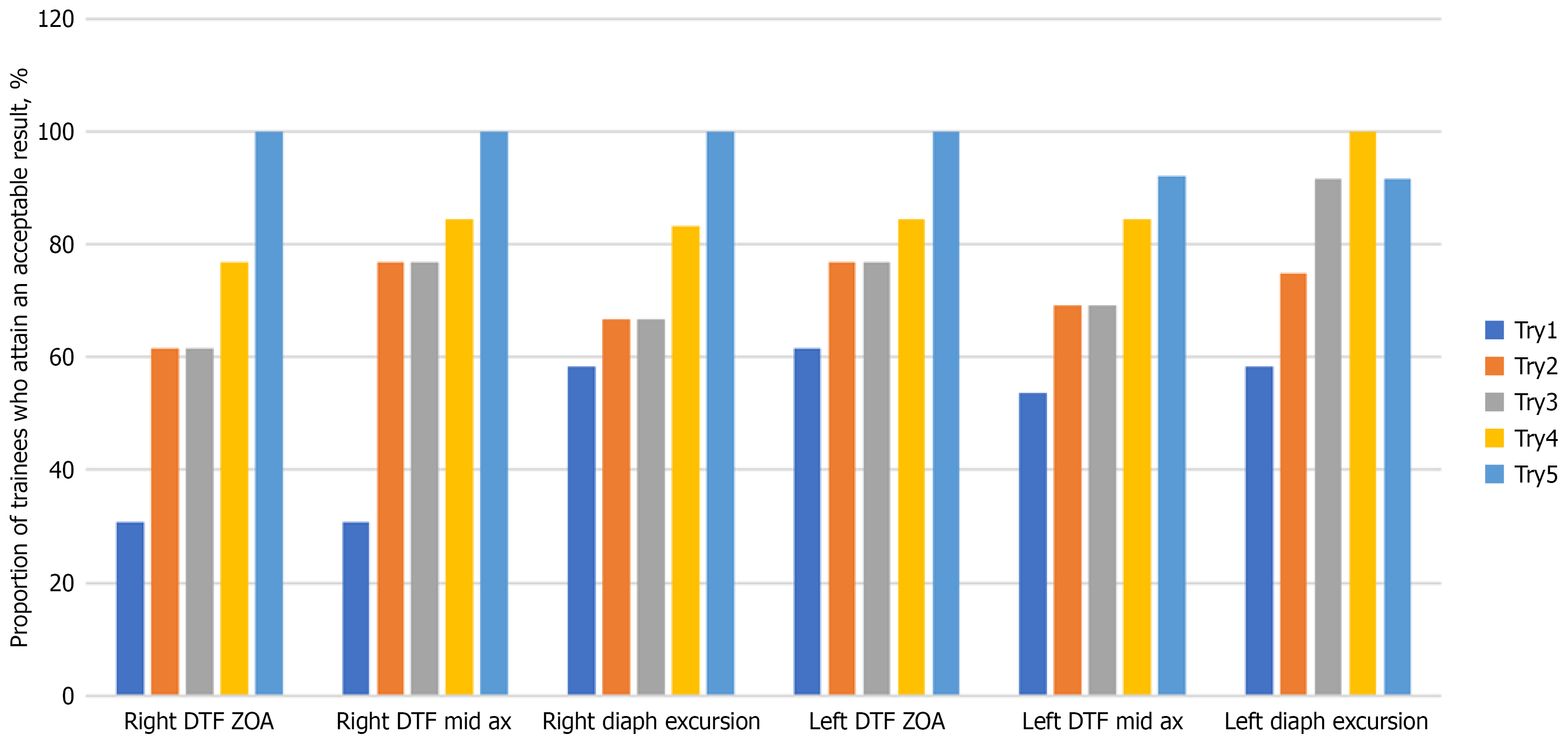
The proportions of trainees who had measurements in agreement with the trainers, similar interpretation of the presence of diaphragm dysfunction and who required assistance were also determined (Table 4). For measurement agreement, there appears to be a steeper learning curve for the right DTF at the zone of apposition and mid-axillary line between patient encounter 1 and 2, and again between patient encounter 3 and 4. A higher proportion of trainees had measurement agreement by the time they were attempting the left DTF (which was trained in sequence following training for the right diaphragm thickening and excursion). Measurement agreement for DTF approaches or equals to 100% at patient 5. For interpretation agreement, agreement was generally good, with nearly all attempts having more than 80% agreement, even from the first attempt. This approaches or equals 100% by the 5th patient encounter. Measurements and interpretation agreement is also close to or equal to 100% for evaluation of the right and left DE by the 5th patient encounter. A proportion of trainees still needed assistance for the left hemi-diaphragm assessment at the 5th patient encounter, with the highest proportion of trainees needing assistance for imaging the excursion of the left hemi-diaphragm (25%).
| Try 1 | Try 2 | Try 3 | Try 4 | Try 5 | |||
| Right DTF at ZOA (n = 13) | Measurement | Trainee/trainer measurement in agreement | 4 (30.8) | 8 (61.5) | 9 (69.2) | 10 (76.9) | 13 (100) |
| Interpretation | Trainee/trainer interpretation in agreement | 12 (92.3) | 11 (84.6) | 12 (92.3) | 12 (92.3) | 13 (100) | |
| Assistance | Needed help | 12 (92.3) | 8 (61.5) | 12 (92.3) | 5 (38.5) | 0 (0) | |
| Right DTF at mid-axillary line (n = 13) | Measurement | Trainee/trainer measurement in agreement | 4 (30.8) | 10 (76.9) | 10 (76.9) | 12 (92.3) | 13 (100) |
| Interpretation | Trainee/trainer interpretation in agreement | 11 (84.6) | 13 (100) | 13 (100) | 12 (92.3) | 13 (100) | |
| Assistance | Needed help | 11 (84.6) | 6 (46.2) | 6 (46.2) | 7 (53.8) | 0 (0) | |
| Right diaphragm excursion (n = 12) | Measurement | Trainee/trainer measurement in agreement | 7 (58.3) | 8 (66.7) | 8 (66.7) | 10 (83.3) | 12 (100) |
| Interpretation | Trainee/trainer interpretation in agreement | 10 (83.3) | 10 (83.3) | 12 (100) | 12 (100) | 12 (100) | |
| Assistance | Needed help | 12 (100) | 12 (100) | 8 (66.7) | 5 (41.7) | 0 (0) | |
| Left DTF at ZOA (n = 13) | Measurement | Trainee/trainer measurement in agreement | 8 (61.5) | 10 (76.9) | 10 (76.9) | 11 (84.6) | 13 (100) |
| Interpretation | Trainee/trainer interpretation in agreement | 13 (100) | 12 (92.3) | 13 (100) | 11 (84.6) | 13 (100) | |
| Assistance | Needed help | 12 (92.3) | 9 (69.2) | 10 (76.9) | 7 (53.8) | 2 (15.4) | |
| Left DTF at mid-axillary line (n = 12) | Measurement | Trainee/trainer measurement in agreement | 8 (66.7) | 9 (75.0) | 9 (75.0) | 11 (91.7) | 12 (100) |
| Interpretation | Trainee/trainer interpretation in agreement | 9 (75.0) | 10 (83.3) | 11 (91.7) | 11 (91.7) | 11 (91.7) | |
| Assistance | Needed help | 10 (83.3) | 7 (58.3) | 5 (41.7) | 2 (16.7) | 1 (8.3) | |
| Left excursion (n = 12) | Measurement | Trainee/trainer measurement in agreement | 7 (58.3) | 9 (75.0) | 11 (91.7) | 12 (100) | 11 (91.7) |
| Interpretation | Trainee/trainer interpretation in agreement | 12 (100) | 12 (100) | 12 (100) | 12 (100) | 12 (100) | |
| Assistance | Needed help | 12 (100) | 11 (91.7) | 6 (50.0) | 6 (50.0) | 3 (25.0) |
Figure 3 shows the proportion of trainees who were considered to have an overall pass of each parameter of the training session, where an overall pass was defined if he or she had both: (1) Measurements in agreement with the trainers; and (2) Similar interpretation of the presence of diaphragm dysfunction as the trainer. Trainees tended to do worse on the left DTF at the mid-axillary line and the left DE assessments.
DUS competency in this study was defined as the acquisition of the technical skills required for independent DUS practice. The trainers agreed this could be defined as achieved if > 90% trainees and trainers had measurements and interpretation in agreement, as well as the trainee not requiring further assistance. This study showed that among trainees with varied backgrounds and ultrasound experience, DUS skills and interpretation accuracy improved with practice, reaching near-complete agreement with trainers by the 5th patient for most parameters, particularly for the right hemi-diaphragm. However, left DTF and excursion required more supervised scans for some trainees to achieve independent competency.
The utility of DUS has been studied to look for evidence of diaphragmatic dysfunction, and in the critically-ill population[11,18]. Applications include diagnosing diaphragmatic dysfunction[3,14], serial assessments of atrophy in mechanically ventilated patients[19,20], predict weaning outcomes from mechanical ventilation[3-5], titration of ventilatory support through assessment of diaphragmatic contractile activity[20-22], and detection of patient-ventilator interactions by looking at contractile activity and excursion of the diaphragm[23,24].
However, little information exists on the amount or type of training required for DUS competency. Dugar et al[25] found that trainees who completed an online DUS training platform and independently completed 17 DUS examinations could obtain measurements of diaphragm thickness at end-expiration and diaphragmatic thickening fraction during inspiration with an acceptable agreement to expert measurements. Garofalo et al[6] found that using a combination of a theoretical module followed by a practical session improved trainees’ measurement accuracy compared to when they completed a theoretical module alone.
While Garofalo et al[6] showed the importance of a supervised practical session, it is not known what is the optimum number of supervised examinations that would be enough to achieve sufficient DUS competency for examining critically-ill patients. Garofalo et al[6] suggested that 25 supervised examinations in healthy subjects would be sufficient, based on the number required for lung ultrasound examinations[9]. Expert consensus on diaphragm ultrasonography in the critically ill suggested using a minimum of 40 (ideally bilateral) examinations, of which at least 20 should be under indirect supervision of an expert teacher, for training for independent use of DUS[18]. More recently, Puchongmart et al[26] reported that practicing on five patients was sufficient for emergency physicians to achieve an adequate level of competency in conducting right-sided diaphragmatic ultrasound examinations. However, unlike our study, the residents performing the ultrasound examinations had been receiving training in the application of point-of-care ultrasound (POCUS) from the start of their first year of residency with each having done at least 100 POCUS scans and so may have been further along the ultrasound trajectory than our trainee cohort. Their residents also obtained a total 20 images showing diaphragmatic excursion and diaphragmatic thickness from a block of five patients, with half for excursion and half for thickness evaluation.
In practice, the amount of training for DUS studies has varied. In pediatric patients, a 4-hour practical training course for trainees resulted in high inter-operator reliability when compared to the expert[27]. For adult patients, 3 to 5 training sessions for the trainee DUS operator, each lasting 10-15 minutes, was adequate to achieve an intraobserver variability of < 10% in measuring percentage change in diaphragm thickness[4]. Goligher et al[12] reported their observer training on 15 DUS examinations before beginning data collection, with sufficient feasibility and reproducibility in mechanically ventilated patients.
Despite the variation in training protocols, a fairly steep learning curve for DUS has been suggested. Pirompanich and Romsaiyut[28] showed that inter-observer correlation between trainee and expert was high for both right and left diaphragm assessment, with intra-observer correlation of the trainee increasing sharply to almost 0.9 for the first ten patients, with only slight increase after that. DUS may be most similar to lung ultrasound, where a steeper learning curve for lung ultrasound has been suggested with quick psychomotor skill acquisition for novices[29].
We showed that agreement between trainees without prior DUS experience and experts for evaluation of the left or right DTF could reach close to or equal to 100% after 10 sets of measurements using 60 images on each side (5 sets of measurements at the zone of apposition, and 5 sets of measurements at the mid-axillary line). For DE, agreement between trainees and experts for evaluation of the right or left DE could reach close to or equal to 100% after 5 sets of measure
We had chosen two sites to determine diaphragm thickening (zone of apposition and mid-axillary line) to simulate real-life experience where the most optimal site of visualization of the diaphragm may lie between the mid-axillary or antero-axillary line[11]. Using two sites as individual examination locations also contributes to the overall number of supervised examinations for determination of diaphragm thickness and thickening fraction. Therefore, for diaphragmatic thickening fraction, our trainees performed 30 individual examinations per side over 5 patient encounters. Similarly, in obtaining high intra-observer correlation after the first ten patients, Pirompanich and Romsaiyut[28] required the trainee to perform the diaphragmatic thickening fraction of three breaths on each side, which equates to 30 individual examinations per side. It is our postulation that the number of image acquisitions is a more accurate reflection of the amount of training required as opposed to the number of patients imaged. Separately, more supervised examinations may be required for the training of DTF compared to DE because of the increased difficulty in determining very small changes in thickness with the pre-existing small dimensions of the diaphragm for calculation of DTF.
Our practical training protocol followed a set sequence of measurements, where the right side of the diaphragm was routinely assessed before the left. The following parameters were assessed in this order: Right DTF at zone of apposition, right DTF at mid-axillary line, right diaphragmatic excursion, left DTF at zone of apposition, left DTF at mid-axillary line, left diaphragmatic excursion. It is possible that the experience of evaluating the contralateral side contributes to the overall learning experience of one side of the diaphragm. If training only focuses on one side, a higher number of dedi
Our study also highlighted the increased difficulty with assessing the left diaphragm compared to the right, in particular when assessing left DE, because of the smaller acoustic window of the spleen and the interposition of gas in the stomach. No assistance was required by the 5th attempt for the right diaphragm parameters, while assistance was required at the 5th attempt for the left DTF (8.3%-15.4%), while 25% still required assistance at the 5th attempt of determining the left DE. Interpretation and measurement accuracy was also less than 100%, at 91.7% for the left DTF at mid-axillary line and left DE respectively. This implies that more than 30 examinations for left DTF and 15 examinations for left DE may be required for the trainee to independently perform left DUS examinations. This suggests the need for a separate, higher competency threshold for left-sided DUS, especially for imaging of the left diaphragm at the mid-axillary line and left DE assessments. Our study also showed the increased difficulty with subcostal visualization of the diaphragm[3,11], where 1 out of the 13 patients in our study had poor ultrasound imaging quality of left and right DE, leading to exclusion from our data analysis.
Our study has several strengths. Firstly, our study was performed on critically-ill patients instead of healthy volun
Our study also had limitations. Firstly, we studied a small cohort of trainees recruited in a single center. Secondly, potential bias in measurements and interpretation could occur as our trainers were not blinded. To minimize bias, we followed a pre-determined protocol for measurements and interpretation, including sequence of parameter evaluation. Our protocol also stipulated that the trainer should completely not intervene (neither with verbal feedback nor image acquisition) for at least a minute while the trainee attempted to obtain images and measurements. This was followed in all patients to minimise bias. Trainers and trainees also avoided looking at the clinical notes of the imaged patients till ultrasound image acquisition and interpretation was completed. Our results are also in general agreement with prior expert suggestions and data[6,18,25,28], so major bias is less likely. While we had considered analyzing independent image acquisition and performing blinded measurements at a later time on saved clips, we ultimately decided on real time measurements for the following reasons: (1) We felt it was beneficial to the trainee for the trainer to be able to observe in real time any inaccuracies of the trainees’ acquisition of images or measurements, and so be able to give feedback via repeat demonstration of image acquisition and measurements. Being present in real time also allowed us to record the need for trainer assistance; and (2) We felt that thickness measurements done in the B mode for the diaphragm were best done in real time, in order to use anatomic landmarks (e.g., visualization of lung sliding) to accurately deter
Thirdly, determining the need for trainer assistance for image acquisition and measurement was subjective and could lead to either excess or limited assistance. However, we tried to minimize this by standardizing that the trainer would assist whenever the trainee struggled for more than a minute at scanning. Fourthly, about 5 (approximately 40%) of our trainees were completely ultrasound-naive, which may mean that our number of supervised scans for competency may be a conservative estimate for others who have prior ultrasound experience for other organs or procedures.
In conclusion, trainees can therefore achieve adequate DUS technical skills to perform unsupervised DUS measurements in the critically-ill after completing a theoretical self-study module, followed by at least 20 supervised sets of DTF measurements and 10 supervised sets DE measurements for the right hemi-diaphragm, while a separate, higher competency threshold with more supervised sets may be needed to achieve the technical skills required for independent imaging of the left hemi-diaphragm. This does not equate to competency of the trainee to independently integrate their ultrasound findings in actual patient management, which likely requires further workplace-based assessment. Rather, determining this minimum threshold of supervised cases helps suggest the ideal timing for further training through independent practice, given that higher numbers of POCUS examinations performed by trainees have been shown to be associated with improved learning outcomes[29]. The results of our study would therefore serve to inform a minimum threshold number of directly-supervised patient encounters, above which competency for independent practice may be obtained. This would help with the development of a standardized training method of DUS, and with future certification and credentialing processes for DUS.
| 1. | Zambon M, Greco M, Bocchino S, Cabrini L, Beccaria PF, Zangrillo A. Assessment of diaphragmatic dysfunction in the critically ill patient with ultrasound: a systematic review. Intensive Care Med. 2017;43:29-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 229] [Article Influence: 25.4] [Reference Citation Analysis (3)] |
| 2. | Boon AJ, Sekiguchi H, Harper CJ, Strommen JA, Ghahfarokhi LS, Watson JC, Sorenson EJ. Sensitivity and specificity of diagnostic ultrasound in the diagnosis of phrenic neuropathy. Neurology. 2014;83:1264-1270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 124] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 3. | Kim WY, Suh HJ, Hong SB, Koh Y, Lim CM. Diaphragm dysfunction assessed by ultrasonography: influence on weaning from mechanical ventilation. Crit Care Med. 2011;39:2627-2630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 335] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 4. | DiNino E, Gartman EJ, Sethi JM, McCool FD. Diaphragm ultrasound as a predictor of successful extubation from mechanical ventilation. Thorax. 2014;69:423-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 374] [Cited by in RCA: 286] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 5. | Ferrari G, De Filippi G, Elia F, Panero F, Volpicelli G, Aprà F. Diaphragm ultrasound as a new index of discontinuation from mechanical ventilation. Crit Ultrasound J. 2014;6:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 184] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 6. | Garofalo E, Bruni A, Pelaia C, Landoni G, Zangrillo A, Antonelli M, Conti G, Biasucci DG, Mercurio G, Cortegiani A, Giarratano A, Vetrugno L, Bove T, Forfori F, Corradi F, Vaschetto R, Cammarota G, Astuto M, Murabito P, Bellini V, Zambon M, Longhini F, Navalesi P, Bignami E. Comparisons of two diaphragm ultrasound-teaching programs: a multicenter randomized controlled educational study. Ultrasound J. 2019;11:21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 7. | Díaz-Gómez JL, Perez-Protto S, Hargrave J, Builes A, Capdeville M, Festic E, Shahul S. Impact of a focused transthoracic echocardiography training course for rescue applications among anesthesiology and critical care medicine practitioners: a prospective study. J Cardiothorac Vasc Anesth. 2015;29:576-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 8. | See KC, Ong V, Ng J, Tan RA, Phua J. Basic critical care echocardiography by pulmonary fellows: learning trajectory and prognostic impact using a minimally resourced training model*. Crit Care Med. 2014;42:2169-2177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 42] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 9. | Rouby JJ, Arbelot C, Gao Y, Zhang M, Lv J, An Y, Chunyao W, Bin D, Valente Barbas CS, Dexheimer Neto FL, Prior Caltabeloti F, Lima E, Cebey A, Perbet S, Constantin JM; APECHO Study Group. Training for Lung Ultrasound Score Measurement in Critically Ill Patients. Am J Respir Crit Care Med. 2018;198:398-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 154] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 10. | See KC, Ong V, Wong SH, Leanda R, Santos J, Taculod J, Phua J, Teoh CM. Lung ultrasound training: curriculum implementation and learning trajectory among respiratory therapists. Intensive Care Med. 2016;42:63-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 82] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 11. | Tuinman PR, Jonkman AH, Dres M, Shi ZH, Goligher EC, Goffi A, de Korte C, Demoule A, Heunks L. Respiratory muscle ultrasonography: methodology, basic and advanced principles and clinical applications in ICU and ED patients-a narrative review. Intensive Care Med. 2020;46:594-605. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 168] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 12. | Goligher EC, Laghi F, Detsky ME, Farias P, Murray A, Brace D, Brochard LJ, Bolz SS, Rubenfeld GD, Kavanagh BP, Ferguson ND. Measuring diaphragm thickness with ultrasound in mechanically ventilated patients: feasibility, reproducibility and validity. Intensive Care Med. 2015;41:642-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 357] [Cited by in RCA: 283] [Article Influence: 25.7] [Reference Citation Analysis (0)] |
| 13. | Boussuges A, Gole Y, Blanc P. Diaphragmatic motion studied by m-mode ultrasonography: methods, reproducibility, and normal values. Chest. 2009;135:391-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 611] [Cited by in RCA: 500] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 14. | Mariani LF, Bedel J, Gros A, Lerolle N, Milojevic K, Laurent V, Hilly J, Troché G, Bedos JP, Planquette B. Ultrasonography for Screening and Follow-Up of Diaphragmatic Dysfunction in the ICU: A Pilot Study. J Intensive Care Med. 2016;31:338-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 40] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 15. | Jung B, Moury PH, Mahul M, de Jong A, Galia F, Prades A, Albaladejo P, Chanques G, Molinari N, Jaber S. Diaphragmatic dysfunction in patients with ICU-acquired weakness and its impact on extubation failure. Intensive Care Med. 2016;42:853-861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 215] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 16. | Blumhof S, Wheeler D, Thomas K, McCool FD, Mora J. Change in Diaphragmatic Thickness During the Respiratory Cycle Predicts Extubation Success at Various Levels of Pressure Support Ventilation. Lung. 2016;194:519-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 66] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 17. | Boon AJ, Harper CJ, Ghahfarokhi LS, Strommen JA, Watson JC, Sorenson EJ. Two-dimensional ultrasound imaging of the diaphragm: quantitative values in normal subjects. Muscle Nerve. 2013;47:884-889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 221] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 18. | Haaksma ME, Smit JM, Boussuges A, Demoule A, Dres M, Ferrari G, Formenti P, Goligher EC, Heunks L, Lim EHT, Mokkink LB, Soilemezi E, Shi Z, Umbrello M, Vetrugno L, Vivier E, Xu L, Zambon M, Tuinman PR. EXpert consensus On Diaphragm UltraSonography in the critically ill (EXODUS): a Delphi consensus statement on the measurement of diaphragm ultrasound-derived parameters in a critical care setting. Crit Care. 2022;26:99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 124] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 19. | Zambon M, Beccaria P, Matsuno J, Gemma M, Frati E, Colombo S, Cabrini L, Landoni G, Zangrillo A. Mechanical Ventilation and Diaphragmatic Atrophy in Critically Ill Patients: An Ultrasound Study. Crit Care Med. 2016;44:1347-1352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 89] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 20. | Goligher EC, Fan E, Herridge MS, Murray A, Vorona S, Brace D, Rittayamai N, Lanys A, Tomlinson G, Singh JM, Bolz SS, Rubenfeld GD, Kavanagh BP, Brochard LJ, Ferguson ND. Evolution of Diaphragm Thickness during Mechanical Ventilation. Impact of Inspiratory Effort. Am J Respir Crit Care Med. 2015;192:1080-1088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 470] [Cited by in RCA: 387] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 21. | Goligher EC, Dres M, Fan E, Rubenfeld GD, Scales DC, Herridge MS, Vorona S, Sklar MC, Rittayamai N, Lanys A, Murray A, Brace D, Urrea C, Reid WD, Tomlinson G, Slutsky AS, Kavanagh BP, Brochard LJ, Ferguson ND. Mechanical Ventilation-induced Diaphragm Atrophy Strongly Impacts Clinical Outcomes. Am J Respir Crit Care Med. 2018;197:204-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 608] [Cited by in RCA: 499] [Article Influence: 62.4] [Reference Citation Analysis (1)] |
| 22. | Umbrello M, Formenti P, Longhi D, Galimberti A, Piva I, Pezzi A, Mistraletti G, Marini JJ, Iapichino G. Diaphragm ultrasound as indicator of respiratory effort in critically ill patients undergoing assisted mechanical ventilation: a pilot clinical study. Crit Care. 2015;19:161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 211] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 23. | Vivier E, Haudebourg AF, Le Corvoisier P, Mekontso Dessap A, Carteaux G. Diagnostic Accuracy of Diaphragm Ultrasound in Detecting and Characterizing Patient-Ventilator Asynchronies during Noninvasive Ventilation. Anesthesiology. 2020;132:1494-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 24. | Soilemezi E, Vasileiou M, Spyridonidou C, Tsagourias M, Matamis D. Understanding Patient-Ventilator Asynchrony Using Diaphragmatic Ultrasonography. Am J Respir Crit Care Med. 2019;200:e27-e28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 25. | Dugar S, Mehkri O, Li M, Hastings A, Siuba MT, Vashisht R, Thiruchelvam N, Wong J, Vorona S, Duggal A, Goligher EC. Validation of a Web-based Platform for Online Training in Point-of-Care Diaphragm Ultrasound. ATS Sch. 2022;3:13-19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 26. | Puchongmart C, Nakornchai T, Rittayamai N, Monsomboon A, Prapruetkit N, Limsuwat C, Ruangsomboon O, Chakorn T. Number of attempts required by emergency physicians to achieve competency in diaphragmatic ultrasound imaging. J Clin Ultrasound. 2022;50:256-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 27. | Sanchez de Toledo J, Munoz R, Landsittel D, Shiderly D, Yoshida M, Komarlu R, Wearden P, Morell VO, Chrysostomou C. Diagnosis of abnormal diaphragm motion after cardiothoracic surgery: ultrasound performed by a cardiac intensivist vs. fluoroscopy. Congenit Heart Dis. 2010;5:565-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 49] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 28. | Pirompanich P, Romsaiyut S. Use of diaphragm thickening fraction combined with rapid shallow breathing index for predicting success of weaning from mechanical ventilator in medical patients. J Intensive Care. 2018;6:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 60] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 29. | Breunig M, Hanson A, Huckabee M. Learning curves for point-of-care ultrasound image acquisition for novice learners in a longitudinal curriculum. Ultrasound J. 2023;15:31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 33] [Reference Citation Analysis (0)] |