Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.113265
Revised: September 21, 2025
Accepted: December 30, 2025
Published online: June 20, 2026
Processing time: 246 Days and 11 Hours
Heparin-induced thrombocytopenia (HIT) is a severe immune adverse drug reac
To systematically compare the efficacy and safety of fondaparinux with that of argatroban and bivalirudin in patients who have been suspected or confirmed to have had HIT.
A systematic review of the literature has been conducted according to the PRISMA 2020 guidelines. Electronic databases were searched until January 2025. Randomized controlled trials (RCTs) and observational studies comparing the parenteral anticoagulant in patients with HIT were included. Study quality was assessed by two independent reviewers, based on the Cochrane Risk of Bias tool for RCTs and the Newcastle-Ottawa Scale for observational studies. Evidence certainty was conducted using the GRADE method.
The 2867 patients with HIT were identified in 16 of the studies that comprised this review (1 RCT, 15 RCTs). Limited head-to-head evidence was available from the single RCT. Across the studies, thrombotic events occurred at rates of 5%-15% and major bleeding at rates of 5%-15%. Research has shown differences in anticoagulation efficiency, with methodological differences being significant. Fondaparinux's safety characteristics were found favorable in retrospective reviews, whereas argatroban and bivalirudin displayed similar efficacy characteristics. The evidence certainty was classified as low to very low due to study design limitations and inconsistencies among key outcomes.
This systematic review identified significant gap in the comparative evidence to manage HIT using parenteral anticoagulants. Based on one RCT study and 15 observational studies (n = 2867), no single anticoagulant agent was definitively superior, and the certainty level of all outcomes was low to very low. Observational evidence and its methodological heterogeneity do not allow for ranking the treatment in an evidence-based treatment. Well-designed RCT are needed to guide in selecting the best anticoagulant to use in patients with HIT.
Core Tip: Heparin-induced thrombocytopenia is life-threatening and needs alternative anticoagulation. The systematic review included sixteen studies on 2867 patients treated with fondaparinux, bivalirudin and argatroban. None of these agents proved superior, and evidence certainty was low to very low, mainly because most studies were observational and showed heterogeneity. All three agents demonstrated acceptable efficacy and safety; however, treatment choice should be patient specific and clinically informed. Randomized trials are urgently needed to support evidence-based treatment decisions.
- Citation: Rehman A, Abid M, Jamil H, Siddique S, Parkash S, Nasrullah F, Sapna F, Lohana B, Lohana K, Jawed I, Tufail M, Sharaf MS. Comparative effectiveness of parenteral anticoagulants (fondaparinux, argatroban, bivalirudin) in heparin-induced thrombocytopenia: A systematic review. World J Methodol 2026; 16(2): 113265
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/113265.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.113265
Heparin-induced thrombocytopenia (HIT) is one of the most clinically important immune-mediated adverse drug reactions in modern medicine because it occurs in an estimated 0.2%-5% of patients receiving heparin therapy[1,2]. The pathological state presents as a paradoxical prothrombotic disorder despite thrombocytopenia. The antibodies that form against platelet factor 4-heparin complexes cause platelet activation and an increase in thrombosis[3,4]. HIT has serious clinical implications, with 30-50 percent of afflicted patients developing thrombotic complications, which have high morbidity, such as amputation of limbs, stroke, heart attack, and death unless it is diagnosed early and treated appro
HIT management recommends the immediate discontinuation of any product associated with heparin and the initiation of alternative anticoagulation to prevent or treat obscure situations of thrombotic complications[1,2]. Over the last twenty years, the choice of effective alternative anticoagulants has undergone significant changes, with at least ten parenteral agents emerging, including direct thrombin inhibitors such as argatroban and bivalirudin, as well as the exemplar of a synthetic pentasaccharide, fondaparinux[5-7]. Each agent has unique pharmacological characteristics, dosing, monitoring, and safety profiles that could be relevant to their broader appropriateness in specific clinical settings and various patients[8-10].
Argatroban is an artificial direct thrombin inhibitor that is one of the first directly approved drugs for the treatment of HIT, with long-term clinical experience in its use. Its hepatic metabolism makes it quite desirable in a patient with renal impairment. However, this characteristic requires dose reductions in hepatic dysfunction and close monitoring of various coagulation parameters[10,11]. Bivalirudin is a more direct thrombin inhibitor, and although it has a shorter half-life and is primarily eliminated by the kidneys, in theory, it exhibits more consistent pharmacokinetics and fewer drug interactions as well[6,9].
Fondaparinux has a distinctly different mechanism of action, as it is an indirect factor Xa inhibitor, typical of antithrombin activation[7,12,13]. The drug possesses once-daily dosing, few drug interactions, and superior bioavailability, which makes it an attractive option. However, the drug has not been applied to patients with severe renal impairment, and a specific antagonist is absent, which has limited its application in certain medical practices[7,12]. Additionally, fondaparinux lacks official approval as a HIT treatment agent in most jurisdictions and is used off-label based on growing clinical evidence[7,13,14].
Despite all these therapeutic options, when it comes to using the anticoagulant of choice in HIT patients, an issue of debate and clinical insecurity still exists[1,2]. Clinical practice guidelines are currently based on expert opinion rather than comparative data, and most evidence comes from single-arm trials, case series, and retrospective studies, rather than head-to-head comparative trials[3,15]. Limited amount of comparative evidence has resulted in a significant variation in practice and uncertainty regarding the relative benefits and risks of the alternative anticoagulant strategies.
HIT patients are clinically heterogeneous, and the optimal anticoagulant choice may vary depending on renal, hepatic, bleeding risk, thrombotic burden, concurrent medications, or clinical circumstances[2,15]. Patients with isolated HIT without thrombosis might not need a certain therapy as much as the presence of thrombotic complications, and seriously ill patients might have another concern than the ones in general medical wards[1-3].
To date, there have been very few systematic reviews that have comprehensively compared fondaparinux to arga
The 2020 PRISMA statement has been adhered to in order to report on a systematic review[16]. The protocol for the review was designed beforehand, and the eligibility criteria, search strategy, and analysis plans were defined.
Electronic databases searched included MEDLINE (PubMed), EMBASE, and the Cochrane Central Register of Controlled Trials. The search was performed from the database inception to January 2025. A comprehensive literature search was conducted with the assistance of a medical librarian, who formulated a strategy using MeSH terms and free-text keywords related to HIT and parenteral anticoagulants. Web of Science tracked citations, and duplicates were removed using EndNote X9 (Clarivate Analytics, Philadelphia, PA, United States) as the reference manager.
The Population, Intervention, Comparison, and Outcomes framework was used to formulate the research question, ensuring it was extensive and comprehensive in identifying pertinent studies[17].
Population: Adult patients (≥ 18 years) who have either confirmed or suspected HIT with or without the thrombotic manifestations.
Intervention: Administration of parenteral anticoagulants, including fondaparinux, argatroban, or bivalirudin.
Comparison: Direct comparison between any two of the specified anticoagulants, or indirect comparison using a control such as a placebo or conventional medical practice.
Outcomes: Primary outcomes included thrombotic events (arterial and venous), major bleeding, and all-cause mortality. Secondary outcomes included minor bleeding, platelet recovery, hospital stay, and limb amputation rates.
Inclusion and exclusion criteria mentioned below were then predetermined to select high-quality evidence related to the research question, designed to limit heterogeneity[18].
Inclusion criteria: Randomized controlled trials and observational studies (cohort studies, case-control studies). Studies involving adult patients who had confirmed or suspected HIT. Studies evaluating fondaparinux, argatroban, or bivalirudin drugs as primary interventions. Studies providing data on comparative effectiveness between interventions. Studies published in the English language. Studies in which the outcome of interest is defined clearly.
Exclusion criteria: Case reports and case series with fewer than 10 patients. Studies restricted to pediatric populations. Studies that were limited to oral anticoagulants. Studies with unclear or inadequate HIT diagnostic criteria. Conference abstracts that do not provide full-text availability. Studies lacking a deficiency in outcome data.
Studies not written in English were excluded due to the resources required and translation issues. The introduction of more language biases is always a cause of concern. However, to favorably include the most relevant high-quality evidence, a comprehensive search of major international databases was conducted. Also, most pivotal studies in this field get published in English-language journals.
The screening of the retrieved records was conducted by two independent reviewers, who assessed the titles and abstracts of the records based on predetermined eligibility criteria using Rayyan (Qatar Computing Research Institute, Doha, Qatar), a web-based systematic review management system[19]. After that, the same reviewers used standardized screening forms to see full-text articles of potentially eligible studies. The decision on the disagreements was made through discussion, and in cases of need, a third reviewer was consulted. To establish the index of inter-reviewer agreement, the kappa coefficient (Cohen)[20] was applied, as computed using SPSS version 28.0 (IBM Corp., Armonk, NY, United States).
Two reviewers completed data extraction using an individually designed data extraction sheet, which was assembled according to Cochrane guidelines and created in Microsoft Excel (Microsoft Corporation, Redmond, WA, United States) in 2019. Extracted variables included the design, setting, and duration of the study, as well as the features of the participants (age, gender, comorbidities, and criteria used in diagnosing HIT). Additionally, the variables included the intervention (drug, dose, duration, and monitoring) and the outcome measure. Any deviation in the extraction of data would be addressed by documentation of the discussion of points and revisions of source documents.
The determination of risk of bias was made by two independent reviewers using validated instruments to assess the risk of bias in the study type. The revised Cochrane Risk of Bias tool version 2[21] was used to assess randomized controlled trials; it has five domains: Bias arising due to what has happened during the randomization process, bias occurring from the differences in what was supposed to be happening, bias developing because of missing results, bias either in the process of measuring the outcomes, and bias in the selection of the reported outcome. All the domains were assessed as having either a low risk of bias, some concerns, or a high risk of bias.
The Newcastle-Ottawa Scale (NOS)[22] was used to determine the quality of non-randomized studies, i.e., non-randomized (observational) research. The NOS examines three general opinions which is the groups selection (representativeness of exposed cohort, selection of non-exposed cohort, ascertainment of exposure, demonstration that outcome was not present at the start of the study), similarity of groups (comparability on the basis of design or analysis) and mea
Each study was evaluated independently by both reviewers, with differences resolved through discussion with the assistance of a third reviewer when necessary. The Cohen kappa coefficient as the rate of inter-reviewer agreement when it came to quality evaluation was read as follows in accordance with the cut-off values of 0.01-0.20 (slight agreement), 0.21-0.40 (fair agreement), 0.41-0.60 (moderate agreement), 0.61-0.80 (substantial agreement) and 0.81-1.00 (almost perfect agreement), and was calculated using SPSS version 28.0 (IBM Corp., Armonk, NY, United States). Other quality factors considered were the possibility of obscuring factors, the sufficiency of methods of statistical analysis and the comprehensiveness in reporting the outcome.
We performed a qualitative synthesis of the evidence due to substantial heterogeneity regarding study design, popu
The level of certainty of the evidence was determined using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology[24], aided by GRADEpro GDT software (McMaster University and Evidence Prime Inc., Hamilton, ON, Canada). Evidence levels were categorized as high, moderate, low, or very low, based on the study design and the risk of bias, discrepancy, indirectness, and imprecision, among other factors, including publication bias and dose-response relationships.
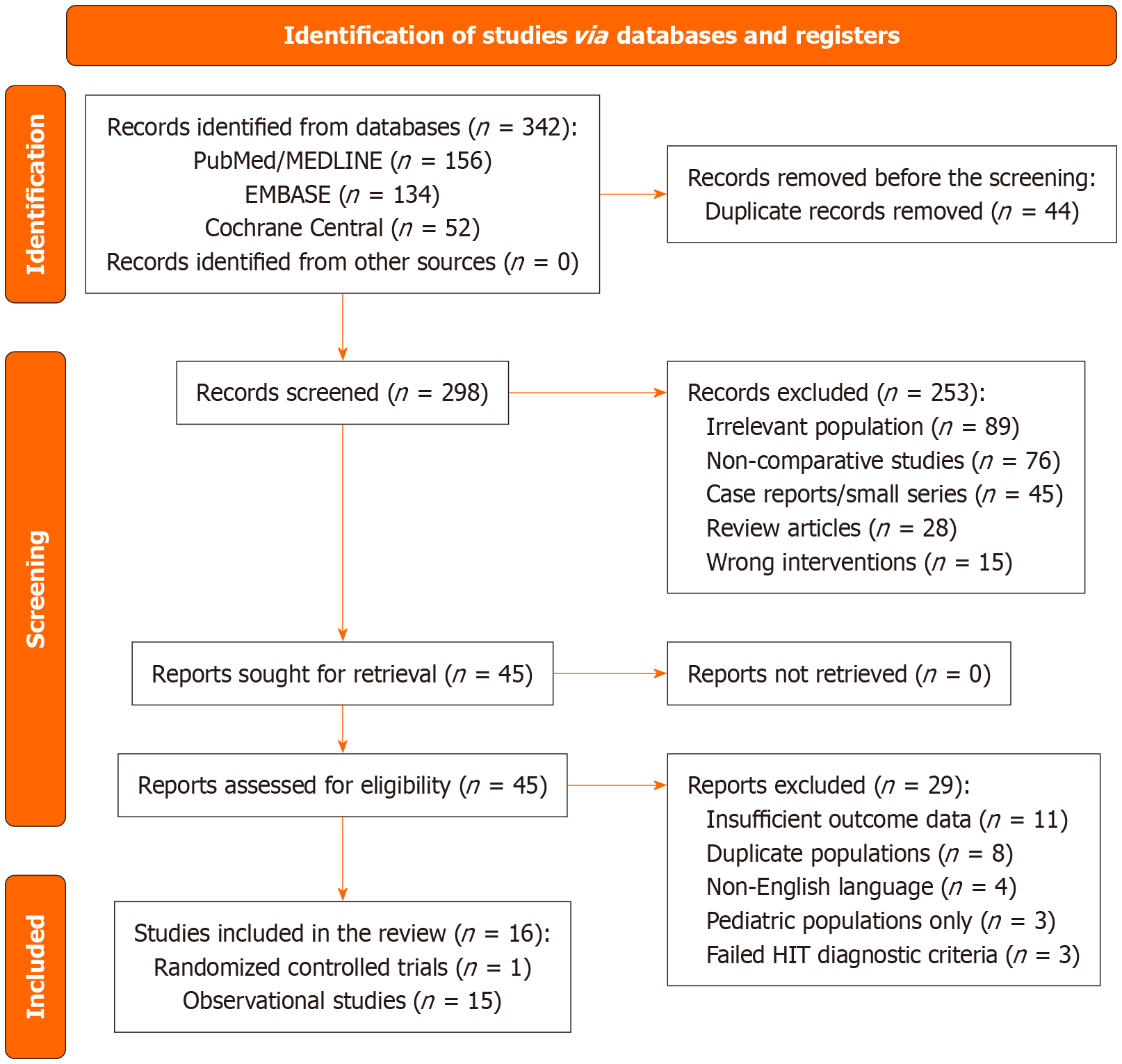
A systematic literature search yielded 342 records from three databases. A total of 298 records were screened for title and abstract after duplicates were removed (n = 44). After a complete evaluation of the text from 29 articles, we included 16 studies that fulfilled our inclusion criteria. These studies consisted of one randomized controlled trial and 15 observational studies based on their objectives and design. The detailed process of the article section process is depicted in the PRISMA flow diagram below (Figure 1).
The one randomized controlled trial and 15 observational studies included a total of 2867 patients with confirmed or suspected HIT[25-28]. Table 1 below presents characteristics of studies. Across all studies, thrombotic event rates ranged from 0% to 30% (median 10.5%), major bleeding rates from 5% to 15% (median 9.2%), and mortality rates from 0% to 22% (median 12.4%) (Table 1).
| Ref. | Design | Setting | Patients | HIT diagnosis | Interventions | Follow-up |
| Boyce et al[5] (2011) | RCT | Multi-center | 50/62.5 | Suspected HIT | Desirudin vs argatroban | 35 days |
| Joseph et al[6] (2014) | Retrospective cohort | Single center | 133/61.0 | Confirmed/suspected | Bivalirudin | Hospital stay |
| Bain and Meyer[9] (2015) | Retrospective cohort | Single center | 89/66.2 | Confirmed/suspected | Bivalirudin vs lepirudin vs Argatroban | Hospital stay |
| Duewell et al[10] (2021) | Retrospective cohort | Multi-center | 167/64.8 | Suspected/confirmed | Argatroban vs bivalirudin | 30 days |
| Al-Eidan et al[8] (2018) | Retrospective cohort | Single center | 34/65.4 | Isolated HIT | Argatroban vs fondaparinux | Hospital stay |
| Marchetti et al[11] (2022) | Retrospective cohort | Single center | 32/68.5 | Confirmed HIT | Argatroban | Treatment period |
| Davis et al[25] (2022) | Retrospective cohort | Multi-center | 135/63.2 | Confirmed/Suspected | DOACs | 90 days |
| Albuloushi et al[26] (2022) | Retrospective cohort | Single center | 48/66.8 | HIT | DOACs | 30 days |
| Snodgrass et al[13] (2016) | Retrospective cohort | Single center | 43/61.7 | Suspected HIT | Fondaparinux | Hospital stay |
| Pishko et al[15] (2019) | Retrospective cohort | Multi-center | 1291/63.1 | Suspected HIT | Various | 30 days |
| Carré et al[27] (2021) | Retrospective case series | Single center | 22/69.0 | HIT | DOACs | 6 months |
| Schindewolf et al[7] (2017) | Retrospective cohort | Multi-center | 616/67.2 | HIT | Fondaparinux vs others | Hospital stay |
| Kang et al[12] (2015) | Retrospective cohort | Single center | 78/64.5 | Suspected HIT | Fondaparinux | 30 days |
| Tardy-Poncet et al[28] (2015) | Prospective cohort | Multi-center | 62/66.8 | Confirmed HIT | Argatroban | Treatment period |
| Nand et al[3] (1997) | Retrospective cohort | Single center | 108/59.4 | HIT with thrombosis | Various | Hospital stay |
| Dulicek et al[14] (2019) | Retrospective cohort | Single center | 37/68.9 | HIT | Fondaparinux | Treatment period |
In the quality assessment, it was found that the included studies exhibited significant variability in methodological rigor. The single randomized controlled trial by Boyce et al[5] showed a low risk of bias in all domains. This assessment was based on proper outcome assessment, concealment of allocation, and adequate randomization. In contrast, the quality of observational studies varied widely. Common weaknesses included poor control over confounding factors, design-related risk of bias in treatment allocation, and inconsistent measurement of outcomes.
Most of the observational studies (n = 10, 67%) were ranked as having fair overall quality, whereas three studies received good ratings in quality, and five were rated as poor. The main areas of focus were comparability of treatment groups, with 9 of the studies (60%) having a high risk of bias due to inadequate research to correct for baseline patient characteristics, comorbidities, and degree of disease severity. The issue of selection bias was also a problem, and 5 studies (33%) were at high risk, either because their samples lacked representative patients or the inclusive criteria were poorly outlined. The scale of risk of bias in the included studies is presented in Table 2.
| Ref. | Study design | Selection | Comparability | Outcome assessment | Overall quality |
| Boyce et al[5] (2011) | RCT | Low risk | Low risk | Low risk | Good |
| Joseph et al[6] (2014) | Observational | Moderate risk | High risk | Moderate risk | Fair |
| Bain and Meyer[9] (2015) | Observational | Moderate risk | Moderate risk | Moderate risk | Fair |
| Duewell et al[10] (2021) | Observational | Moderate risk | Moderate risk | Low risk | Fair |
| Al-Eidan et al[8] (2018) | Observational | High risk | High risk | Moderate risk | Poor |
| Marchetti et al[11] (2022) | Observational | Moderate risk | High risk | Low risk | Fair |
| Davis et al[25] (2022) | Observational | Moderate risk | High risk | Moderate risk | Fair |
| Albuloushi et al[26] (2022) | Observational | Moderate risk | High risk | Moderate risk | Fair |
| Snodgrass et al[13] (2016) | Observational | High risk | High risk | Moderate risk | Poor |
| Pishko et al[15] (2019) | Observational | Low risk | Moderate risk | Low risk | Good |
| Carré et al[27] (2021) | Observational | High risk | High risk | Moderate risk | Poor |
| Schindewolf et al[7] (2017) | Observational | Low risk | Moderate risk | Low risk | Good |
| Kang et al[12] (2015) | Observational | Moderate risk | Moderate risk | Moderate risk | Fair |
| Tardy-Poncet et al[28] (2015) | Observational | Moderate risk | High risk | Low risk | Fair |
| Nand et al[3] (1997) | Observational | High risk | High risk | High risk | Poor |
| Dulicek et al[14] (2019) | Observational | High risk | High risk | Moderate risk | Poor |
Randomized controlled trial evidence: In one of the studies by Boyce et al[5], desirudin and argatroban were compared in 50 suspected HIT patients. This trial showed no difference in the compassionate treatment outcome of passing away, amputation, or the development of a new thrombotic event between the treatments (desirudin 16 percent vs argatroban 20 percent, P = 0.99). In the management of serious cases of bleeding, major bleeding occurred in 8 percent of desirudin-treated patients, whereas the argatroban-treated patients recorded 4 percent (P = 0.99). Although this study provides randomized evidence, it is not directly compared with the three major anticoagulants of interest (fondaparinux, argatroban, and bivalirudin), and it lacks a large sample size.
Observational study evidence: Observational studies provided most of the comparative effectiveness data, but they had their own limitations in establishing causal links between interventions and outcomes. Such studies encompassed a wide variety of clinical environments and populations, as well as various periods of follow-up, which represent reality in the form of clinical practice patterns and also contribute to providing significant heterogeneity in data interpretation. It is also found that the studies differed significantly in the way the diagnosis of HIT was carried out, with half requiring laboratory confirmation of the heparin-PF4 complex, whereas the other half used clinical criteria and a suspicion of HIT diagnosis.
Key findings presented in the observational evidence showed mixed efficacy among various anticoagulants, with thrombotic events ranging from 0% to 30%, and major bleeding ranging from 5% to 15% in the studies. It was not possible to make direct comparisons, as the available data was limited, but it is possible to state some of the trends that have been identified because of the existing data, e.g., the use of fondaparinux has a potentially positive effect in certain groups of patients, and argatroban has consistent effects on numerous clinical scenarios. The great variability in the definition of outcomes, follow-up, and patient characteristics reduces the ability to draw conclusive findings on comparative effectiveness. Table 3 provides detailed results and significant findings from the observational studies.
| Agent | Studies (n) | Thrombotic events (%) | Major bleeding (%) | Mortality (%) |
| Fondaparinux | 4 | 0-15.4 | 5.1-11.1 | 5.9-9.3 |
| Argatroban | 6 | 5.9-21.7 | 5.9-14.5 | 0-21.7 |
| Bivalirudin | 3 | 7.5-16.9 | 7.8-15.8 | 12.4-20.0 |
| Overall range | 16 | 0-30 | 5-15 | 0-22 |
Fondaparinux: Favorable outcomes in the area of fondaparinux therapy for HIT patients have been reported in various studies. In a multicenter study, Schindewolf et al[7] found that thrombotic events occurred in 6.8% vs 11.2% of patients (fondaparinux vs other anticoagulants). Kang et al[12] conducted a propensity-matched study and found the same effectiveness when compared to a historical control in argatroban. Evidence of consistent safety was evident[7,12-14], although almost all the studies had relatively small sample sizes compared to argatroban studies.
Argatroban: Argatroban was the most studied agent, demonstrating great efficiency in successive observational studies[8,10,11]. The percentages of thrombotic events varied between studies, ranging from 5.9% to 21.7%, most likely due to differences in patient populations and the severity of HIT. The incidence of serious bleeding was generally the same as that in other agents, at 5.9-14.5 percent.
Bivalirudin: It was also reported that bivalirudin was as effective as, or more effective than, several other agents in preventing thrombotic events in studies that compared this medication with others. The highest rates of thrombotic events were reported by Joseph et al[6] of 7.5% at a single center, and lower event rates were reported by Bain and Meyer[9] and compared favorably to lepirudin and argatroban.
The certainty of evidence on key outcomes of different comparisons has been assessed using the GRADE methodology, a systematic approach to rating the quality of evidence in research. The evaluation considered five areas that may reduce confidence in the evidence, which includes risk of bias (flaws in study design), inconsistency (differences among study results), indirectness (relevance of the evidence to the specific question), imprecision (lack of certainty in the results), and publication bias (selective publication of results). The evaluation also considered three factors that may increase confidence, including the magnitude of effect (how strong the results are), the dose-response gradient (whether greater exposure increases the effect), and confounding factors that reduce the observed effect (when outside factors make the observed effect smaller)[24].
The evaluation of evidence has revealed significant limitations for most comparisons and outcomes. The main reason for such a low rating was the observational character of most of the included studies, which makes it impossible to be confident about the causal relationship, along with the issue of risk of bias, where there is a risk of confounding and selection bias. Other reasons contributing to the low level of certainty ratings include severe imprecision due to the small sample sizes in most studies and a lack of uniformity in findings across study populations and contexts. The tabulation of the in-depth GRADE criterion is given in Table 4[25-27].
| Comparison | Outcome | Studies | Design | Risk of bias | Inconsistency | Indirectness | Imprecision | Certainty |
| Any anticoagulant vs control | Thrombotic events | 1 RCT, 8 Obs | Mixed | Serious | Not serious | Not serious | Serious | Low |
| Fondaparinux vs others | Thrombotic events | 5 Obs | Observational | Serious | Serious | Not serious | Serious | Very low |
| Argatroban vs bivalirudin | Thrombotic events | 3 Obs | Observational | Serious | Not serious | Not serious | Serious | Low |
| Any anticoagulant vs control | Major bleeding | 1 RCT, 10 Obs | Mixed | Serious | Serious | Not serious | Serious | Very low |
| Any anticoagulant vs control | Mortality | 1 RCT, 8 Obs | Mixed | Serious | Serious | Not serious | Serious | Very low |
The proposed systematic review will be the first of its kind to be conducted, reviewing comparative effectiveness evidence on parenteral anticoagulants in the management of HIT. The available evidence base revealed limitations associated with the strength of the available body of evidence, whereby one small randomized controlled trial provides direct comparative information, whereas the rest consists of more observational data, indicating a low degree of methodological strength. Irrespective of these limitations, several notable trends are evident in the available body of evidence that could inform the clinical decision-making approach, while also highlighting the most critical gaps in knowledge that should be addressed in future research.
The fact that high-quality evidence, in the form of randomization, is low is perhaps the most prominent result of this review. The RCT by Boyce et al[5] had an insufficient population (50 patients) and did not study both fondaparinux and bivalirudin, which were the major therapeutic options of interest at that point. This limitation is indicative of the wider challenge of performing randomized trials in the HIT setting, given the small incidence of the condition and its generally prothrombotic nature, as well as the practicality of such a trial not being able to tolerate substantial numbers of patients in the various arms. The outcome of this, however, is that the utilization of observational data creates a risk of selection bias, confounding, and heterogeneity, which makes it quite difficult to draw a definitive conclusion regarding comparative effectiveness.
The implication of the observational data, although imperfect in its own right, is that all three major parenteral agents appear to be equally effective in preventing thrombotic complications in patients with HIT. Thrombotic event rates in the studies varied widely, ranging from 0 to 30 percent, with most studies indicating rates between 5 and 15 percent[6-10,12,13,28]. The variation was perhaps due to differences in patient populations, the severity of HIT, diagnostic criteria, outcome definitions, and the length of follow-up, rather than variations in drug effect[3,15]. The relatively uniform outcomes among the various anticoagulants suggest that perhaps the type of anticoagulant used is not of paramount importance, as long as an effective alternative form of anticoagulation is quickly implemented[1,2].
In our study, fondaparinux showed promising results that should be considered hypothesis-generating rather than definitive, as some studies indicated positive thrombotic and bleeding results, even though its use is off-label in most jurisdictions[7,12-14]. The daily dosing of the agent is convenient, the pharmacokinetic profile is predictable, and interactions are minimal; these factors may be of special interest when dealing with complex hospitalized patients. Nevertheless, fewer patients and possible selection biases (where clinicians choose fondaparinux over other agents in lower-risk patients or when there may be specific indications to use the agent, e.g., specific contraindications to low-molecular-weight agents) do limit the evidence base for fondaparinux. In addition, it is not advisable to use it in severe kidney failure, and also, no specific reversal agent is available, which could restrict its utilization in certain clinical contexts.
Argatroban has the most clinical experience (argatroban has a consistent efficacy across a number of studies and encompasses a wide range of patients)[8,10,11,28]. Its liver metabolism means it will be more suitable for patients with renal dysfunction, which is another comorbidity in HIT patients. However, it can be challenging to apply in a clinical setup due to the need for frequent monitoring, an advanced dose-adjusting system, and the potential for drug interac
Bivalirudin has been shown to yield favorable outcomes in the limited number of comparative studies available and has the potential to achieve better thrombotic event outcomes than other direct thrombin inhibitors[6,9,10]. It potentially has an advantage when there is a high risk of bleeding or a frequent invasive procedure is contemplated, especially since it has a less toxic profile and a reliable dose-response relationship, as well as a shorter half-life. The improved cost-effectiveness and the limited availability of bivalirudin in most healthcare systems have the potential to limit its applicability, and there is a lack of sufficient evidence to demonstrate that bivalirudin is superior to alternative options.
All three agents exhibited an acceptable safety profile. However, meaningful comparisons are hindered by variously defined bleeding patterns, patient populations, and follow-up strategies[6,9,10,15]. All studies indicated a between-study rate of major bleeding that was approximately 5-15 percent, as one would anticipate with the inherent risk of therapeutic anticoagulation meted out to mostly critically ill patients[10,15,28]. Inconsistency in defining and reporting minor bleeding events limits our ability to draw definitive conclusions regarding relative safety profiles, as non-standard definitions often compromise the ability to establish a clear picture of the relative safety profiles of disparate comparisons[15].
Several clinical considerations should be taken into account regarding these results. A significant bias in patient selection may have occurred in the results of observational studies, as clinicians might have had a preferred choice of any particular agent for patients with specific risk factors or presentations. The additional complexity in interpreting comparative effectiveness is due to heterogeneity in the range of HIT diagnostic criteria, which include the presence of suspicion that can be followed by confirmation of HIT and may be accompanied by different approaches to antibody testing[3,15]. Moreover, the evolution of HIT management practices during the study period, such as diagnostic testing, monitoring procedures, and supportive care, between older studies and modern practice may affect the generalizability of older studies and their applicability to current practice.
The economic implications of the choice of anticoagulant are potentially another factor that was not considered sufficiently in the presented studies. These agents differ in costs, with bivalirudin being much more expensive than alternatives in most healthcare systems. A comprehensive pharmacoeconomic assessment should consider acquisition costs, monitoring, duration of therapy, hospital length of stay, and clinical outcomes, including thrombotic complications and major bleeding. The consequences that each agent has on the economy varies greatly. Coagulation monitoring with argatroban requires extensive laboratory and nursing costs, while fondaparinux requires little monitoring and has no readily available agents[7,12]. Bivalirudin’s relatively shorter half-life may facilitate perioperative management, but it requires continuous infusion, which increases pharmacy costs[6,9]. Decisions regarding drug formularies within the healthcare system often focus on acquisition costs, with less emphasis on the total cost of care, leading to inefficient economic outcomes. There is not many cost-effectiveness analyses related to HIT Management. This represents a significant gap that requires a health technology assessment to inform future formulary decision-making. There was a low to very low level of certainty in the evidence for all key outcomes in our GRADE assessment, with a high degree of study and bias-related imprecision, and most studies being observational in nature. The established finding highlights a dire need for high-quality randomized trials to further establish evidence-based hierarchies of treatment. The difficulties in carrying out such trials in HIT should not prevent attempts to innovate and develop new study designs, pragmatic trials, registry-based randomized studies, or adaptive trial designs that would allow for generating more viable comparative effectiveness data[23,29].
The implications of our findings are pertinent to individual-level treatment decisions, extending beyond the scope of healthcare policy and clinical practice guideline development[1,2]. The current guidelines are largely based on expert opinion and indirect evidence, resulting in inconsistent guidelines among professional societies and geographical areas. The development of more conclusive evidence on comparative effectiveness may encourage greater homogeneity in the treatments provided. The substantial heterogeneity in diagnostic criteria, patients, and outcomes across various studies has important consequences for guidelines, preventing the establishment of evidence-based hierarchies and contributing to differences in practice among institutions and regions. Current clinical guidelines, which often lack comparative data and rely primarily on expert opinion, lead to substantial discordance among professional societies. This difference in methods presents a major hurdle to the standardization of clinical practices and the delivery of optimal care.
The review also has several limitations, which are worth noting. The English-language culture of the publications might have overlooked relevant research; however, the thorough search strategy may have identified most high-quality comparative research[30]. It was impossible to conduct a quantitative meta-analysis due to heterogeneity in study designs, populations, and outcome measures; consequently, we were unable to make specific estimates of comparative effectiveness[23]. Additionally, the emphasis on parenteral anticoagulants did not consider direct oral anticoagulants, which are gaining usage in the management of HIT[25-27]. These are also used in practice, but their role remains under research.
The clinical implications of our findings suggest that, overall, the three major Parenteral anticoagulant agents appear to be acceptable options in therapy for HIT management. The choice may be influenced by patient-related factors, institutional bias, and logistical factors, rather than sound evidence-based comparative effectiveness evidence[1,2]. The provision of alternative anticoagulation should be the top priority for clinicians, and the specific monitoring needs and issues associated with each agent must be carefully considered, although clinicians cannot afford to spend too much time deliberating the benefits, risks, and suitability of each agent[5,6,10,28].
Future research priorities should entail conducting well-powered randomized controlled trials that evaluate primary parenteral anticoagulants in well-defined populations with a history of HIT[31]. They would need to apply standardized diagnostic criteria, endpoints, and safety metrics to facilitate comparisons. Additionally, the empirical design of predictive models or clinical decision tools to assist in the use of individualized anticoagulant selection based on patients' characteristics and risk factors may serve to maximize treatment outcomes until more conclusive comparative data become available[32]. The issue of the emerging impact of direct oral anticoagulants in the treatment of HIT should also be evaluated systematically with the dedicated comparative effectiveness studies[25-27].
This systematic review of 16 studies involving 2867 patients with proven or suspected HIT showed that fondaparinux, argatroban, and bivalirudin have similar efficacy and safety outcomes. The quality of evidence was classified as ‘limited’ due to the mainly observational design of the studies, with heterogeneous methodology and a risk of bias in the studies included in the review. In the absence of formal data on comparative effectiveness, the choice of anticoagulant should be individualized based on patient clinical markers, comorbid illnesses, institutional protocols, and pharmacological considerations. Future research priorities should consist of conducting well-powered multicenter RCTs with standardized outcome measures to inform evidence-based clinical practice guidelines.
| 1. | Fathi M. Heparin-induced thrombocytopenia (HIT): Identification and treatment pathways. Glob Cardiol Sci Pract. 2018;2018:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 2. | Páramo JA, Lozano ML, González-Porras JR, Mateo J. Current status of diagnosis and treatment of heparin-induced thrombocytopenia (HIT). Med Clín (Engl Ed). 2022;158:82-89. [DOI] [Full Text] |
| 3. | Nand S, Wong W, Yuen B, Yetter A, Schmulbach E, Gross Fisher S. Heparin-induced thrombocytopenia with thrombosis: Incidence, analysis of risk factors, and clinical outcomes in 108 consecutive patients treated at a single institution. Am J Hematol. 1997;56:12-16. [DOI] [Full Text] |
| 4. | Warkentin TE, Cook RJ, Marder VJ, Sheppard JA, Moore JC, Eriksson BI, Greinacher A, Kelton JG. Anti-platelet factor 4/heparin antibodies in orthopedic surgery patients receiving antithrombotic prophylaxis with fondaparinux or enoxaparin. Blood. 2005;106:3791-3796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 176] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 5. | Boyce SW, Bandyk DF, Bartholomew JR, Frame JN, Rice L. A randomized, open-label pilot study comparing desirudin and argatroban in patients with suspected heparin-induced thrombocytopenia with or without thrombosis: PREVENT-HIT Study. Am J Ther. 2011;18:14-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 6. | Joseph L, Casanegra AI, Dhariwal M, Smith MA, Raju MG, Militello MA, Gomes MP, Gornik HL, Bartholomew JR. Bivalirudin for the treatment of patients with confirmed or suspected heparin-induced thrombocytopenia. J Thromb Haemost. 2014;12:1044-1053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 7. | Schindewolf M, Steindl J, Beyer-Westendorf J, Schellong S, Dohmen PM, Brachmann J, Madlener K, Pötzsch B, Klamroth R, Hankowitz J, Banik N, Eberle S, Müller MM, Kropff S, Lindhoff-Last E. Use of Fondaparinux Off-Label or Approved Anticoagulants for Management of Heparin-Induced Thrombocytopenia. J Am Coll Cardiol. 2017;70:2636-2648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 49] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 8. | Al-Eidan FAS, Alrawkan S, Alshammary H, Crowther MA. Comparison of argatroban and fondaparinux for the management of patients with isolated heparin-induced thrombocytopenia. Ann Hematol. 2018;97:2055-2059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 9. | Bain J, Meyer A. Comparison of bivalirudin to lepirudin and argatroban in patients with heparin-induced thrombocytopenia. Am J Health Syst Pharm. 2015;72:S104-S109. [PubMed] [DOI] [Full Text] |
| 10. | Duewell BE, Briski MJ, Feih JT, Rinka JRG, Tawil JN. Argatroban Versus Bivalirudin in the Treatment of Suspected or Confirmed Heparin-Induced Thrombocytopenia. J Pharm Pract. 2021;34:529-534. [PubMed] [DOI] [Full Text] |
| 11. | Marchetti M, Barelli S, Gleich T, Gomez FJ, Goodyer M, Grandoni F, Alberio L. Managing argatroban in heparin-induced thrombocytopenia: A retrospective analysis of 729 treatment days in 32 patients with confirmed heparin-induced thrombocytopenia. Br J Haematol. 2022;197:766-790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 12. | Kang M, Alahmadi M, Sawh S, Kovacs MJ, Lazo-Langner A. Fondaparinux for the treatment of suspected heparin-induced thrombocytopenia: a propensity score-matched study. Blood. 2015;125:924-929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 87] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 13. | Snodgrass MN, Shields J, Rai H. Efficacy and Safety of Fondaparinux in Patients With Suspected Heparin-Induced Thrombocytopenia. Clin Appl Thromb Hemost. 2016;22:712-717. [PubMed] [DOI] [Full Text] |
| 14. | Dulicek P, Ivanova E, Kostal M, Fiedlerova Z, Sadilek P, Hirmerova J. Heparin-induced thrombocytopenia treated with fondaparinux: single center experience. Int Angiol. 2020;39:76-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 15. | Pishko AM, Lefler DS, Gimotty P, Paydary K, Fardin S, Arepally GM, Crowther M, Rice L, Vega R, Cines DB, Guevara JP, Cuker A. The risk of major bleeding in patients with suspected heparin-induced thrombocytopenia. J Thromb Haemost. 2019;17:1956-1965. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 16. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 17. | Schardt C, Adams MB, Owens T, Keitz S, Fontelo P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med Inform Decis Mak. 2007;7:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2284] [Cited by in RCA: 1933] [Article Influence: 101.7] [Reference Citation Analysis (0)] |
| 18. | Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 19. | Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan-a web and mobile app for systematic reviews. Syst Rev. 2016;5:210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18843] [Cited by in RCA: 15381] [Article Influence: 1538.1] [Reference Citation Analysis (6)] |
| 20. | Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33:159-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60504] [Cited by in RCA: 42870] [Article Influence: 874.9] [Reference Citation Analysis (4)] |
| 21. | Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22257] [Cited by in RCA: 20369] [Article Influence: 2909.9] [Reference Citation Analysis (7)] |
| 22. | Wells GA, Wells G, Shea B, Shea B, O'Connell D, Peterson J, Welch Losos M, Tugwell P, Ga SW, Zello GA, Petersen JA. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. [cited 20 November 2025]. Available from: https://www.semanticscholar.org/paper/The-Newcastle-Ottawa-Scale-(NOS)-for-Assessing-the-Wells-Wells/c293fb316b6176154c3fdbb8340a107d9c8c82bf. |
| 23. | Popay J, Roberts H, Sowden A, Petticrew M, Arai L, Rodgers M, Britten N, Roen K, Duffy S. Guidance on the conduct of narrative synthesis in systematic reviews. A product from the ESRC methods programme Version. Lancaster University, 2006; 1: b92. [DOI] [Full Text] |
| 24. | Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, Schünemann HJ; GRADE Working Group. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924-926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17839] [Cited by in RCA: 16615] [Article Influence: 923.1] [Reference Citation Analysis (4)] |
| 25. | Davis K, Sebaaly J, Wooten L, Khouli C, Mihm A, Nisly SA. A Multicenter Retrospective Evaluation of Direct Oral Anticoagulants for the Treatment of Heparin-Induced Thrombocytopenia. Am J Cardiovasc Drugs. 2022;22:417-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 26. | Albuloushi A, Rhoten M, Kelly J, Sylvester KW, Grandoni J, Connors JM. Evaluation of the use of direct oral anticoagulants for the management of heparin-induced thrombocytopenia. J Thromb Thrombolysis. 2022;54:597-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 27. | Carré J, Guérineau H, Le Beller C, Mauge L, Huynh B, Nili R, Planquette B, Clauser S, Smadja DM, Helley D, Lillo-Le Louet A, Gendron N, Calmette L. Direct Oral Anticoagulants as Successful Treatment of Heparin-Induced Thrombocytopenia: A Parisian Retrospective Case Series. Front Med (Lausanne). 2021;8:713649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 28. | Tardy-Poncet B, Nguyen P, Thiranos JC, Morange PE, Biron-Andréani C, Gruel Y, Morel J, Wynckel A, Grunebaum L, Villacorta-Torres J, Grosjean S, de Maistre E. Argatroban in the management of heparin-induced thrombocytopenia: a multicenter clinical trial. Crit Care. 2015;19:396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 29. | Ford I, Norrie J. Pragmatic Trials. N Engl J Med. 2016;375:454-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1240] [Cited by in RCA: 1094] [Article Influence: 109.4] [Reference Citation Analysis (0)] |
| 30. | Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20160] [Cited by in RCA: 17906] [Article Influence: 1053.3] [Reference Citation Analysis (4)] |
| 31. | Mehta CR, Pocock SJ. Adaptive increase in sample size when interim results are promising: a practical guide with examples. Stat Med. 2011;30:3267-3284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 264] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 32. | Steyerberg EW, Moons KG, van der Windt DA, Hayden JA, Perel P, Schroter S, Riley RD, Hemingway H, Altman DG; PROGRESS Group. Prognosis Research Strategy (PROGRESS) 3: prognostic model research. PLoS Med. 2013;10:e1001381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1210] [Cited by in RCA: 1119] [Article Influence: 86.1] [Reference Citation Analysis (0)] |