Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.112692
Revised: August 22, 2025
Accepted: January 9, 2026
Published online: June 20, 2026
Processing time: 263 Days and 18.4 Hours
Salt restriction is commonly recommended for patients with cirrhosis to manage ascites; however, its safety and efficacy in patients with hyponatremia remain un
To evaluate the effects of a salt-restricted diet (SRD; 5 g/day) vs a salt-unrestricted diet (SUD; 10 g/day) on renal, nutritional, and hemodynamic outcomes, as well as short-term mortality in patients with cirrhosis with moderate to severe hyponat
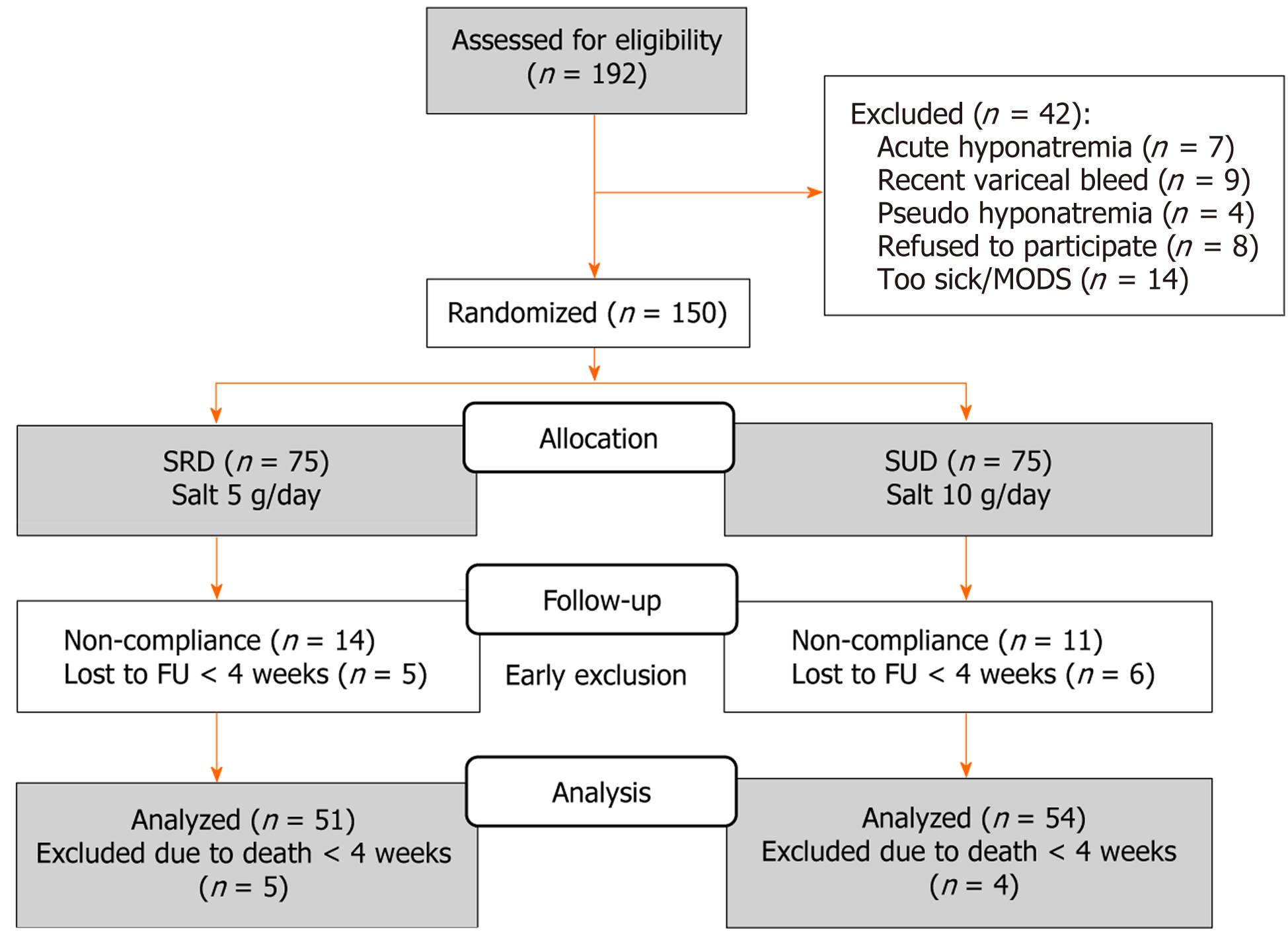
A total of 150 patients with serum sodium < 130 mEq/L were randomized to either SRD or SUD. After excluding patients who died, were non-adherent, or were lost to follow-up within 4 weeks, 105 participants (SRD: 51; SUD: 54) were included in the outcomes analysis. The primary outcome was the incidence of acute kidney injury (AKI) over 6 months; secondary outcomes included ascites control, nutritional markers, and mortality.
Baseline characteristics were largely comparable. AKI occurred in 45 patients (42.8%), which was significantly more in the SRD group (58.8% vs 27.7%; P = 0.005). SRD was an independent predictor of AKI (odds ratio: 4.85), and the risk of AKI was 66% lower in the SUD group (hazard ratio: 0.34; P < 0.001). While ascites control was numerically better in the SUD group, the difference was not statistically significant. The SUD group exhibited better hemo
Salt restriction in patients with cirrhosis with moderate to severe hyponatremia may predispose them to renal dysfunction, nutritional decline, and increased short-term mortality without providing a clear benefit in ascites control. These findings underscore the need for individualized sodium recommendations in patients with ad
Core Tip: This study highlights the potential harm of salt restriction in patients with cirrhosis with moderate to severe hyponatremia. Patients in the salt-restricted diet group (5 g/day) had significantly higher risk of acute kidney injury, poorer nutritional and hemodynamic profiles, and increased short-term mortality compared to those in the salt-unrestricted diet group (10 g/day). Importantly, no significant improvement in ascites control was observed with salt restriction. These results question the routine use of salt restriction in this subgroup and emphasize the need for a more tailored dietary approach to optimize renal function, nutrition, and survival outcomes in patients with hyponatremic cirrhosis.
- Citation: Marrapu S, Kumar R, Bhushan D, Kumar S, Kumar A, Priyadarshi RN, Anand U. Open-label randomized controlled study of salt-restricted vs salt-unrestricted diet in patients with cirrhosis with hyponatremia. World J Methodol 2026; 16(2): 112692
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/112692.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.112692
Hyponatremia is a frequent and clinically significant complication in patients with decompensated cirrhosis, affecting up to 50% of hospitalized individuals and often serving as a surrogate marker for disease severity and poor prognosis[1,2]. It typically results from impaired free water clearance driven by non-osmotic vasopressin release and a hyperdynamic circulatory state characterized by splanchnic vasodilation[3]. This pathophysiological environment predisposes these patients to effective arterial hypovolemia, activating neurohormonal pathways that ultimately lead to renal vasoconstriction and acute kidney injury (AKI)[4]. Traditionally, sodium restriction has been a cornerstone in managing ascites and fluid overload in patients with cirrhosis. Since salt is the primary source of sodium in our diet, guidelines from major liver societies recommend limiting salt intake to ≤ 5.2 g/day in patients with moderate to severe ascites[5]. However, there is a notable lack of consensus across existing clinical guidelines, with substantial inconsistencies in the recom
The physiological rationale for salt restriction may become counterproductive in the setting of impaired intravascular volume. Sodium depletion in such conditions can aggravate intravascular hypovolemia and impair renal perfusion, thereby increasing the risk of AKI[8,9,11,12]. AKI remains one of the strongest predictors of mortality in cirrhosis, with even small increases in serum creatinine associated with poor outcomes[13]. While many contributors to AKI are non-modifiable, dietary sodium intake represents a potentially modifiable factor. Observational data suggest that liberal sodium intake may enhance renal blood flow and reduce the incidence of AKI in patients with cirrhosis with ascites[8]. However, there is a lack of studies specifically evaluating the impact of sodium restriction on renal outcomes in patients with cirrhosis with hyponatremia - a subgroup that may be particularly vulnerable to volume shifts and renal dysf
Despite this evolving understanding, most dietary guidelines for patients with cirrhosis continue to endorse uniform sodium restriction, often without accounting for individual variability in disease physiology or clinical context. This randomized controlled trial (RCT) was designed to address this gap by evaluating whether salt restriction offers any advantage in terms of renal, nutritional, or hemodynamic outcomes in patients with cirrhosis with moderate to severe hyponatremia, defined as a serum sodium concentration of < 130 mEq/L. The primary outcome was the incidence of AKI over 6 months. Secondary outcomes included changes in ascites control, nutritional markers, mean arterial pressure (MAP), and mortality.
This was a single-center, prospective, open-label, parallel-arm RCT conducted in the Department of Gastroenterology at a tertiary care hospital in India, from August 1, 2023 to April 1, 2025. The study protocol was approved by the Institutional Ethics Committee (No. AIIMS/Pat/IEC/PGT/Jan23-06) and conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants prior to enrollment. The trial was exempted from man
All adult patients aged 18 years or older with a diagnosis of cirrhosis were eligible for inclusion if they had chronic mo
Randomization was performed using a computer-generated permuted block randomization technique with variable block sizes (4 and 6) to ensure balance and unpredictability. The allocation sequence was generated by a statistician not involved in patient recruitment or outcome assessment. Sequentially numbered, opaque, sealed envelopes were utilized to ensure allocation concealment. Participants were assigned in a 1:1 ratio to one of two dietary interventions: (1) SRD with a salt intake of 5 g/day, or (2) SUD with salt consumption similar to the general population (10 g/day). Due to the nature of the dietary intervention, blinding was not feasible. To ensure adherence, all participants were provided a pre
Demographic data, including age, sex, comorbidities, cirrhosis etiology, and anthropometric measurements, were collected at baseline. Dry weight estimates for corrected body mass index calculations were obtained by deducting 5%, 10%, or 15% of the actual weight in cases of mild, moderate, or severe ascites, respectively, with an additional 5% dedu
All patients received standardized medical care for cirrhosis in accordance with contemporary guidelines. Both groups received diuretics, beta-blockers, lactulose, rifaximin, or albumin as clinically indicated. Diuretic doses and the requirement for large-volume paracentesis (LVP) were documented. Patients were followed up at 4-week intervals over a 6-month period. At each visit, clinical assessments included vital signs, ascites status, CTP and MELD-Na scores, nutri
Based on a prior study reporting AKI in 13.86% of patients on an SRD vs 0% on an SUD[8], 49 patients per group were needed to achieve 80% power at a two-sided alpha of 0.05. With a 20% dropout adjustment, the target sample size was 124. However, 150 patients were ultimately enrolled in this study to accommodate a longer follow-up period.
Data were analyzed using SPSS version 25. Continuous variables are expressed as the mean ± SD or median with range, depending on data distribution. Categorical variables were summarized as n (%). Normality was assessed using the Kolmogorov-Smirnov test and Q-Q plots. Intergroup comparisons were made using the Student’s t-test or Mann-Whitney U test for continuous variables and χ2 test or Fisher’s exact test for categorical variables, as applicable. Multivariable logistic regression was conducted to identify independent predictors of AKI. All clinically relevant variables dem
A total of 192 patients with cirrhosis and hyponatremia without AKI were screened for study inclusion. Forty-two patients were excluded for not meeting the inclusion criteria (Figure 1). Subsequently, 150 patients with serum sodium levels below 130 mEq/L were randomized into either the SRD or SUD group. Of these, 45 patients were further excluded from the outcome analysis due to early death, dietary non-compliance, or loss to follow-up within 4 weeks after randomization. The final cohort consisted of 105 patients: 51 in the SRD group and 54 in the SUD group (Figure 1). The two gr
| Variable (unit) | SRD (n = 51) | SUD (n = 54) | P value |
| Age, (years) | 49.33 ± 14.71 | 46.83 ± 11.94 | 0.340 |
| Sex, male:female | 36:15 (70.6:29.4) | 50:4 (92.6:7.4) | 0.003 |
| Etiology of cirrhosis | 0.150 | ||
| Ethanol | 12 (23.5) | 26 (48.1) | |
| Hepatitis B virus | 8 (15.7) | 5 (9.3) | |
| Hepatitis C virus | 2 (3.9) | 0 (0) | |
| MASLD | 9 (17.6) | 9 (16.7) | |
| Mixed/others | 20 (39.2) | 14 (25.9) | |
| ACLF at presentation | 5 (9.8) | 4 (7.4) | 0.608 |
| Diabetes mellitus | 13 (25.5) | 15 (27.8) | 0.791 |
| Mean arterial pressure (mmHg) | 82.77 ± 12.84 | 83.57 ± 11.72 | 0.736 |
| Body mass index (kg/m2) | 21.04 ± 4.86 | 20.44 ± 3.62 | 0.476 |
| Abdominal circumference (cm) | 92.30 ± 10.73 | 90.48 ± 10.67 | 0.387 |
| Severity of hyponatremia | 0.973 | ||
| Moderate (125-129 mEq/L) | 31 (60.8) | 33 (61.1) | |
| Severe (< 125 mEq/L) | 20 (39.2) | 21 (38.9) | |
| Grade of ascites | 0.214 | ||
| Controlled | 7 (13.7) | 9 (16.7) | |
| Grade I | 4 (7.8) | 6 (11.1) | |
| Grade II | 17 (33.3) | 13 (24.1) | |
| Grade III | 26 (51.0) | 23 (42.6) | |
| Prior LVP | 12 (23.5) | 11 (20.4) | 0.696 |
| Prior AKI | 10 (19.6) | 11 (20.4) | 0.922 |
| Thyroid-stimulating hormone (μIU/mL) | 5.82 ± 7.22 | 4.20 ± 2.68 | 0.127 |
| Cortisol (μg/dL) | 14.81 ± 6.17 | 14.35 ± 4.28 | 0.655 |
| TLC (cells/mm3) | 7115.69 ± 3652.82 | 8379.07 ± 5607.03 | 0.177 |
| Bilirubin (mg/dL) | 5.74 ± 7.23 | 4.66 ± 5.39 | 0.388 |
| Aspartate transaminase (U/L) | 109.24 ± 141.95 | 113.49 ± 87.19 | 0.853 |
| Alanine transaminase (U/L) | 60.05 ± 94.75 | 43.84 ± 20.68 | 0.223 |
| Potassium (mEq/L) | 4.18 ± 0.68 | 4.35 ± 0.63 | 0.198 |
| Hemoglobin (g/dL) | 8.65 ± 2.28 | 8.76 ± 2.31 | 0.803 |
| Urea (mg/dL) | 35.10 ± 14.09 | 31.83 ± 15.69 | 0.265 |
| Creatinine (mg/dL) | 0.83 ± 0.32 | 0.81 ± 0.26 | 0.779 |
| Sodium (mEq/L) | 125.60 ± 3.48 | 124.99 ± 4.30 | 0.429 |
| Chloride (mEq/L) | 97.13 ± 5.69 | 95.96 ± 6.80 | 0.342 |
| eGFR by MDRD6 (mL/minute/1.73 m2) | 97.68 ± 66.00 | 104.85 ± 57.54 | 0.558 |
| Albumin (g/dL) | 2.59 ± 0.46 | 2.74 ± 0.57 | 0.130 |
| Mid arm circumference (cm) | 21.09 ± 4.18 | 22.11 ± 3.12 | 0.161 |
| Handgrip strength (kg) female | 10.7 ± 4.67 | 12.2 ± 4.60 | 0.565 |
| Handgrip strength (kg) male | 18.8 ± 6.51 | 20.5 ± 6.51 | 0.151 |
| Child-Turcotte-Pugh score | 9.65 ± 2.17 | 9.13 ± 2.24 | 0.233 |
| MELD-Na score | 23.28 ± 7.80 | 22.41 ± 8.55 | 0.589 |
| Baseline calorie intake (kcal/day) | 1356.45 ± 512.33 | 1450.48 ± 517.93 | 0.352 |
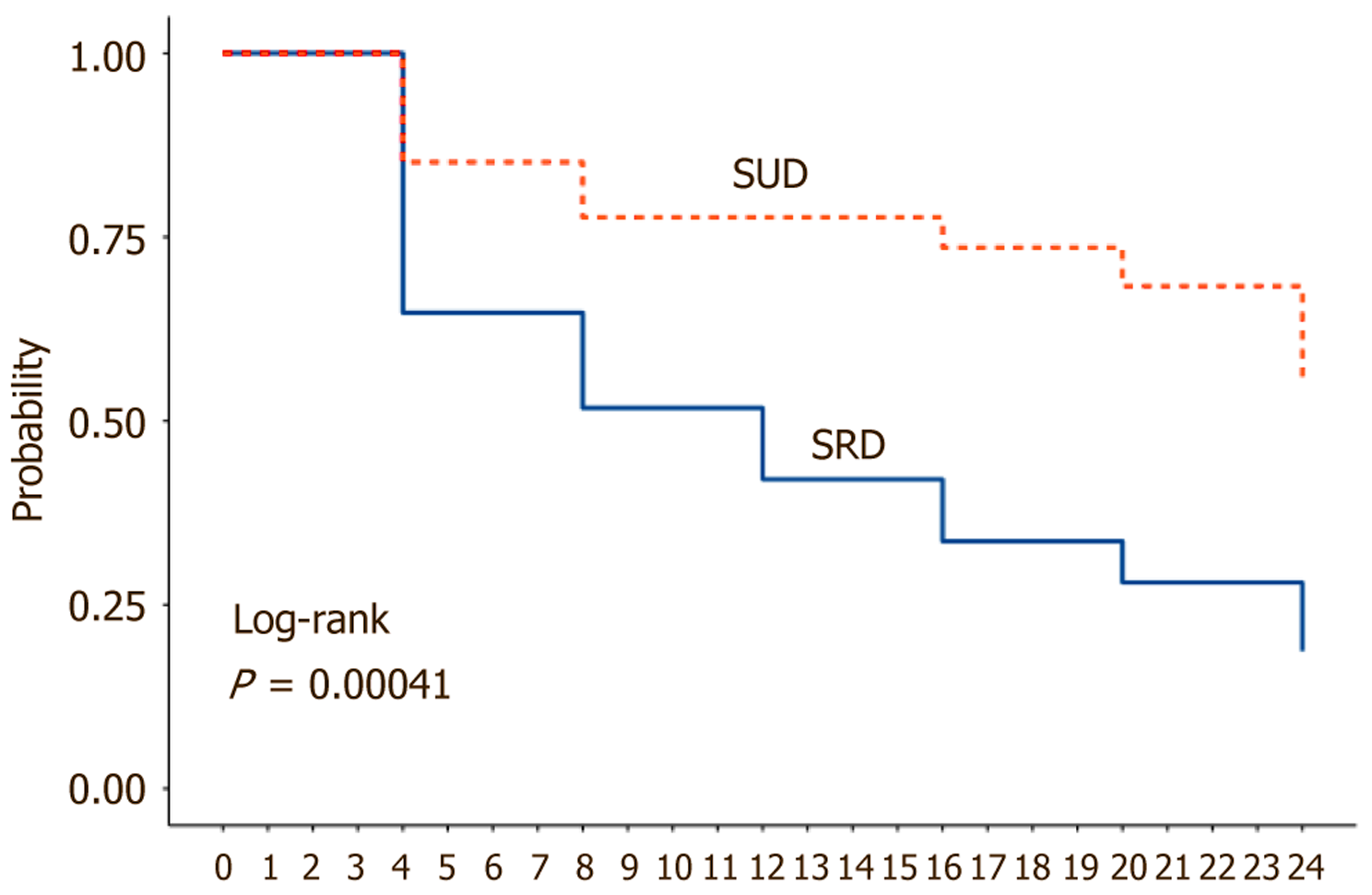
During follow-up, 45 patients developed AKI, corresponding to an overall incidence of 42.8% (95%CI: 33%-52%). The incidence was significantly higher in the SRD group (30 patients, 58.8% [95%CI: 44%-72%]) compared with the SUD group (15 patients, 27.7% [95%CI: 15%-39%]), with P = 0.005. Even on intention-to-treat analysis, AKI incidence remained higher in the SRD group (40.0% [30/75]) compared with the SUD group (20.0% [15/75]; P = 0.003). The severity of AKI, based on Kidney Disease: Improving Global Outcomes criteria, showed no significant intergroup difference. Stage 2 or 3 AKI occurred in 36% of the SRD group and 40% of the SUD group, indicating similar severity when AKI occurred. In the subgroup analysis, patients who developed AKI exhibited a significantly higher prevalence of dietary salt restriction (67% vs 35%; P = 0.002, crude OR: 3.71) and a prior history of AKI (35.6% vs 8.3%; P < 0.001, crude OR: 6.06), as well as a significantly lower MAP (79.91 ± 11.42 mmHg vs 85.63 ± 12.31 mmHg; P = 0.017, crude OR: 0.95) compared to those without AKI. However, there were no significant differences in sex distribution (P = 0.56), prior diuretic use (P = 0.95), LVP (P = 0.58), serum sodium (P = 0.78), serum albumin (P = 0.13), CTP score (P = 0.21), or MELD-Na score (P = 0.56) (Table 2). In logistic regression analysis, SRD had 4.85 times higher odds of developing AKI than those on SUD (95%CI: 1.67-14.07; P = 0.005) (Supplementary Table 1). Kaplan-Meier analysis showed that patients in the SUD group had a significantly higher cumulative probability of remaining AKI-free compared with those in the SRD group (Figure 2). The median time to AKI in the SRD group was 12 weeks (interquartile range: 8-18 weeks). At 24 weeks, 55.9% of participants in the SUD group remained free from AKI, in contrast to only 18.7% in the SRD group. Cox proportional hazards regr
| Variable | No AKI (n = 60) | AKI (n = 45) | P value |
| Age (years) | 47.17 ± 14.14 | 49.22 ± 12.26 | 0.43 |
| Sex | 0.56 | ||
| Male | 51 (85.0) | 35 (77.8) | |
| Female | 9 (15.0) | 10 (22.2) | |
| Dietary group | 0.002 | ||
| Salt restricted | 21 (35.0) | 30 (66.7) | |
| Salt unrestricted | 39 (65.0) | 15 (33.3) | |
| Prior history of AKI | 5 (8.3) | 16 (35.6) | < 0.001 |
| Prior diuretic use | 37 (61.7) | 28 (62.2) | 0.954 |
| Prior LVP | 12 (20.0) | 11 (24.4) | 0.58 |
| Urine protein present | 13 (21.7) | 11 (24.4) | 0.10 |
| TLC (cells/mm3) | 7010 ± 3986 | 8772 ± 5556 | 0.06 |
| MAP (mmHg) | 85.63 ± 12.31 | 79.91 ± 11.42 | 0.017 |
| MAC (cm) | 21.38 ± 4.04 | 21.92 ± 3.16 | 0.45 |
| BMI (kg/m2) | 20.45 ± 4.50 | 21.10 ± 3.93 | 0.44 |
| Sodium (mEq/L) | 125.19 ± 4.00 | 125.41 ± 3.83 | 0.78 |
| Chloride (mEq/L) | 95.86 ± 5.61 | 97.41 ± 7.05 | 0.21 |
| Albumin (g/dL) | 2.73 ± 0.48 | 2.57 ± 0.56 | 0.13 |
| Handgrip strength (kg) | 18.71 ± 6.74 | 18.13 ± 7.19 | 0.67 |
| CTP score | 9.15 ± 2.19 | 9.69 ± 2.22 | 0.21 |
| MELD-Na score | 22.43 ± 8.18 | 23.36 ± 8.19 | 0.56 |
| Baseline calorie intake (kcal/day) | 1458 ± 546.5 | 1333 ± 465.72 | 0.21 |
Improvement in ascites was numerically higher in the SUD group, although not statistically significant (48% vs 33%; P = 0.375). Similarly, non-improvement or worsening of ascites was non-significantly higher in the SRD group (67% vs 52%; P = 0.375). Diuretic requirements decreased in 19.6% and 35% of patients in the SRD and SUD groups, respectively (P = 0.197). Requirements for LVP were comparable across the groups. However, in the subgroup analysis, patients who rem
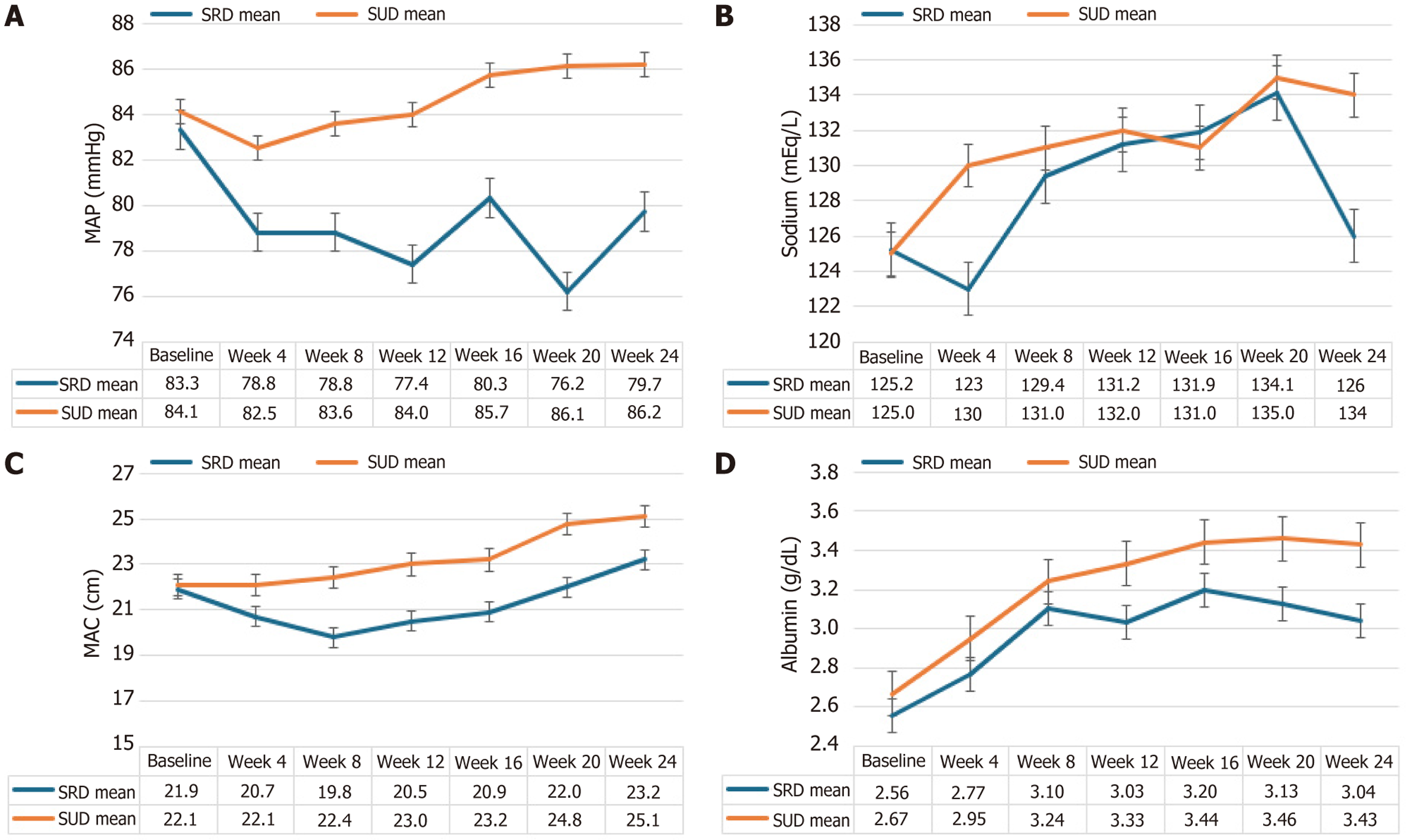
MAP remained consistently higher in the SUD group across follow-up visits (Figure 3A). At multiple time points, differences approached statistical significance. In the SRD group, MAP consistently remained below 82 mmHg, making patients more susceptible to AKI development. At 4 weeks, sodium levels were significantly higher in the SUD group (130.1 mEq/L vs 123.1 mEq/L; P = 0.04). This early divergence favored the SUD group; however, subsequent visits did not maintain statistical significance, potentially due to a decreasing number of participants (Figure 3B).
Patients in the SUD arm consumed more calories across all follow-up visits. MAC was significantly higher in the SUD group during early follow-up (weeks 8 to 16), indicating better preservation of muscle mass and nutritional status. Later visits showed a non-significant trend in favor of SUD (Figure 3C). Although the SUD group had a slightly higher mean albumin at baseline, the difference was not statistically significant (2.59 ± 0.46 g/dL vs 2.74 ± 0.57 g/dL; P = 0.130). Throughout follow-up, serum albumin levels remained consistently higher in the SUD group, but these differences did not reach statistical significance (Figure 3D). A significant and consistently higher HGS was noted in the SUD group across all follow-up points (Supplementary Figure 1).
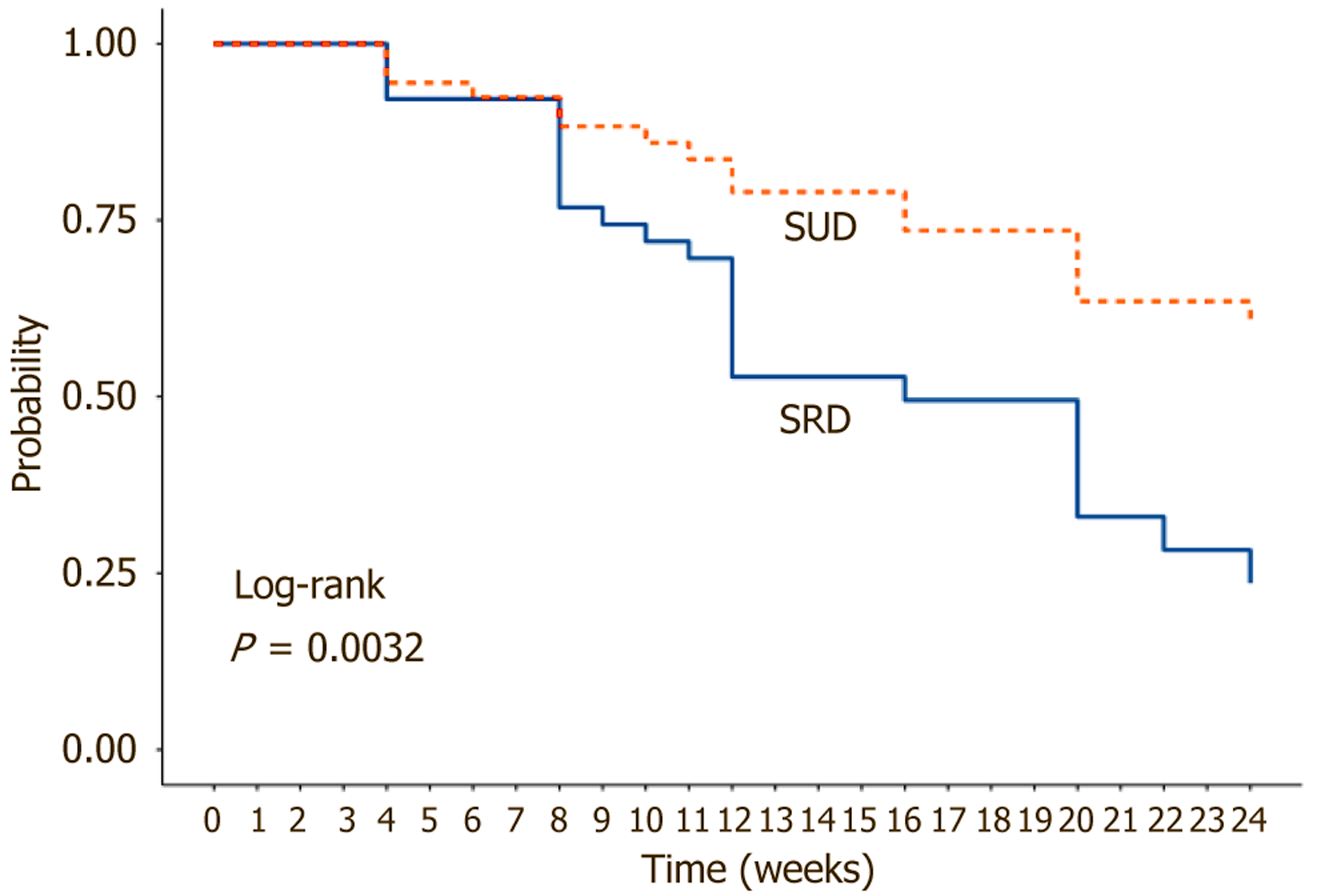
The SUD group consistently exhibited better CTP and MELD-Na scores across all time points, although these differences were not statistically significant (Supplementary Table 2). The overall mortality rate was 41% (44/105) across the cohort. None of the patients underwent liver transplantation due to a lack of available facilities. Mortality was significantly higher in the SRD group, with 28 deaths (54.9%) compared to 16 deaths (29.6%) in the SUD group (P = 0.009). The odds of death were 2.89 times higher in the SRD group than in the SUD group (95%CI: 1.29-6.46). Mortality was also higher among patients who developed AKI (24/45, 53.3%) compared to those who did not (20/60, 33.3%) (P = 0.04), indicating a more than two-fold increase in mortality associated with AKI. Patients who died had higher CTP scores (10.47 ± 1.8 vs 9.2 ± 2.2; P = 0.01), lower calorie intake (1201 ± 513 kcal/day vs 1399 ± 478 kcal/day; P = 0.02), and a trend toward lower albumin levels (2.49 ± 0.42 g/dL vs 2.67 ± 0.5 g/dL; P = 0.008). There was no difference in mortality rates by sex (41% in men vs 42% in women; P = 0.984). Kaplan-Meier analysis demonstrated better survival in the SUD group (Figure 4). At 24 weeks, survival was 59.8% (95%CI: 45.6%-78.4%) in the SUD group and 23.6% (95%CI: 11.8%-47.0%) in the SRD group (P = 0.003). Survival curves began to diverge at week 12 (SUD: 79.0% vs SRD: 52.8%) and consistently favored the SUD group throughout the follow-up.
The RCT demonstrated that salt restriction was associated with a significantly higher incidence of AKI, poorer nutritional parameters, and higher mortality, without significant benefits in ascites control among patients with cirrhosis with moderate to severe hyponatremia. These findings challenge conventional sodium restriction strategies in decompensated cirrhosis with hyponatremia and suggest the need for individualized dietary recommendations. A central finding was the significantly higher incidence of AKI in the SRD group (58.8%) compared to the salt-unrestricted group (27.7%), despite largely comparable baseline characteristics. This aligns with the pathophysiological understanding that, in cirrhosis, effe
Traditionally, sodium restriction has been recommended to reduce ascitic fluid accumulation. Several studies, including RCTs, have evaluated salt restriction in cirrhotic patients with ascites[8,9,11,12,18,19]. However, most of these studies are older and have methodological limitations, making it difficult to draw definitive conclusions. While some reported improved ascites control with strict salt restriction[18,19], others found no benefit[11,12]. Notably, two recent RCTs showed that an SUD led to better ascites resolution and reduced need for LVP compared to an SRD[8,9]. The authors suggested that salt restriction may worsen hyponatremia, impair diuretic response, and reduce renal perfusion, thereby exacerbating ascites. Our study also did not demonstrate any significant advantage of an SRD in controlling ascites; in fact, ascites control was non-significantly better in the SUD group. However, it should be noted that ascites assessment relied on clinical and imaging criteria without quantitative measurements, and standardized or blinded grading by independent evaluators was not possible due to limitations in available resources. Sodium depletion can blunt the diuretic response, often necessitating higher doses that may further impair renal function - a paradox observed in cirrhosis. Supporting this, prior studies have shown that hypertonic saline combined with diuretics enhances fluid mobilization in refractory ascites by improving intravascular volume and renal perfusion[20]. While the overall need for LVP did not significantly differ between groups in our study, subgroup analysis revealed a lower LVP requirement in patients without AKI, suggesting that renal stability may be more critical to ascites management than sodium restriction alone. Hemodynamic analysis revealed consistently higher MAP in the SUD group, with trends approaching significance at multiple visits. Cullaro et al[21] and Maiwall et al[22] demonstrated that maintaining a higher MAP improves renal out
Salt restriction can decrease the palatability of food, potentially resulting in reduced dietary protein and calorie intake[5]. This may have further adverse effects on patients with cirrhosis, who often exhibit poor appetite and varying degrees of protein-calorie malnutrition and sarcopenia[14]. In our study, nutritional outcomes were significantly more favorable in the SUD group. HGS, a validated marker of sarcopenia, was consistently superior in the SUD group, a finding with prognostic significance, as sarcopenia is linked to increased hospitalizations and mortality in cirrhosis[14,15,23]. Mechanistically, salt is essential for the intestinal absorption of glucose and amino acids; thus, salt restriction may impair their uptake and worsen hypoalbuminemia. Serum albumin levels consistently trended higher in the SUD group across all follow-up visits. Although these differences were not statistically significant, likely due to limited power for subgroup an
In our study, mortality was significantly higher in the SRD group (54.9%) compared to the SUD group (29.6%), with an OR of 2.89. These findings align with a recent study showing similarly increased mortality in the SRD group (67.4% vs 35.7%; P = 0.001), where SRD emerged as an independent predictor of mortality in patients with cirrhosis with ascites (OR = 1.65; P < 0.001)[10]. In our study, there was poor correlation between the severity of hyponatremia and the risk of AKI. The variable response to sodium intake on hemodynamic parameters may be attributed to differences in “salt sensitivity” and “third-space sodium storage”. It is known that salt sensitivity, which varies among individuals, leads to exaggerated hemodynamic responses to sodium fluctuations[24]. Furthermore, sodium can be stored in non-osmotic compartments, such as the skin interstitium and endothelium, without concurrent water retention. Dysregulation of this buffer in cirrhosis may impair fluid handling[25,26]. Sodium in the skin interstitium is also involved in fluid mobilization by inducing lymphatic flow. Severe salt restriction may suppress compensatory lymphatic drainage pathways[5,27].
The randomized design, homogeneous study population, and longitudinal follow-up are strengths of our study. However, some limitations must be acknowledged. Blinding was not feasible due to the nature of the intervention, which might have introduced bias. Assessing dietary compliance was challenging due to the ubiquitous presence of sodium in food and the inherent limitations of dietary recall methods, which lack sufficient objectivity and accuracy. Moreover, there is no reliable objective tool for measuring salt intake in cirrhosis. Even 24-hour urinary sodium estimation is limited by factors such as third-space sodium storage, frequent use of diuretics, and variable renal handling of sodium. The sex distribution was not balanced at baseline; however, gender did not affect the primary outcome of the study. There were no differences in the incidence of AKI or mortality rates by gender. As our sample size calculation was partly based on extrapolation from a related study, the unavailability of strong prior effect-size estimates was a limitation. There was a high patient attrition rate in our study, reflecting the advanced stage and frailty of the study population. This may have reduced the power to detect significant differences in secondary outcomes, contributing to several follow-up parameters remaining statistically insignificant despite observable trends. A substantial number of patients were excluded from analysis due to early death, dietary non-compliance, or loss to follow-up; however, comparable outcomes in intention-to-treat analysis made the possibility of bias and overestimation of treatment effects less likely. Our multivariable logistic regression was based on baseline predictors. A time-dependent covariate model could better capture changes in clinical variables over the 6-month follow-up. However, significant attrition over time limited our ability to robustly apply dynamic modeling without introducing additional bias. Furthermore, AKI and mortality in cirrhosis are multifactorial outcomes influenced by various dynamic variables. Although we took several steps to minimize confounding, the possibility of residual confounding, particularly due to sepsis and medications, cannot be ruled out. Confounders such as concomitant use of albumin, lactulose, and rifaximin were not stratified. However, only a few patients received these treatments transiently, and their distribution between groups was similar according to protocol. Lastly, although our findings indicate a significant association between salt restriction and mortality, causality cannot be definitively inferred, primarily due to the potential for residual confounding.
In conclusion, salt restriction may increase the risk of AKI and mortality in patients with cirrhosis with moderate to severe hyponatremia. Moreover, an SUD appears to be associated with better nutritional parameters without worsening ascites control. These results underscore the need for a more individualized approach to sodium management in patients with advanced cirrhosis. Nevertheless, our findings should be interpreted with caution, particularly regarding secondary analyses, given the modest sample size and the potential for residual confounding inherent to an open-label trial. Larger, multicenter studies employing propensity score adjustment and time-dependent statistical methods are warranted to refine clinical guidelines and optimize patient-specific dietary recommendations in cirrhosis with hyponatremia.
We acknowledge the support and cooperation of all of the study participants.
| 1. | Flores J, Maldonado AO, Pena C, Nugent K. Cirrhosis and hyponatremia: A review of pathogenesis, clinical relevance, and management. Am J Med Sci. 2025;370:209-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 2. | Alukal JJ, John S, Thuluvath PJ. Hyponatremia in Cirrhosis: An Update. Am J Gastroenterol. 2020;115:1775-1785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 66] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 3. | Bernardi M, Moreau R, Angeli P, Schnabl B, Arroyo V. Mechanisms of decompensation and organ failure in cirrhosis: From peripheral arterial vasodilation to systemic inflammation hypothesis. J Hepatol. 2015;63:1272-1284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 534] [Cited by in RCA: 463] [Article Influence: 42.1] [Reference Citation Analysis (1)] |
| 4. | Arroyo V, Ginès P, Gerbes AL, Dudley FJ, Gentilini P, Laffi G, Reynolds TB, Ring-Larsen H, Schölmerich J. Definition and diagnostic criteria of refractory ascites and hepatorenal syndrome in cirrhosis. International Ascites Club. Hepatology. 1996;23:164-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1037] [Cited by in RCA: 954] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 5. | Kumar R, Marrapu S. Dietary salt in liver cirrhosis: With a pinch of salt! World J Hepatol. 2023;15:1084-1090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 6. | Aithal GP, Palaniyappan N, China L, Härmälä S, Macken L, Ryan JM, Wilkes EA, Moore K, Leithead JA, Hayes PC, O'Brien AJ, Verma S. Guidelines on the management of ascites in cirrhosis. Gut. 2021;70:9-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 328] [Cited by in RCA: 288] [Article Influence: 57.6] [Reference Citation Analysis (1)] |
| 7. | Moore KP, Wong F, Gines P, Bernardi M, Ochs A, Salerno F, Angeli P, Porayko M, Moreau R, Garcia-Tsao G, Jimenez W, Planas R, Arroyo V. The management of ascites in cirrhosis: report on the consensus conference of the International Ascites Club. Hepatology. 2003;38:258-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 744] [Cited by in RCA: 629] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 8. | Gu XB, Yang XJ, Zhu HY, Xu BY. Effect of a diet with unrestricted sodium on ascites in patients with hepatic cirrhosis. Gut Liver. 2012;6:355-361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 9. | Sorrentino P, Castaldo G, Tarantino L, Bracigliano A, Perrella A, Perrella O, Fiorentino F, Vecchione R, D' Angelo S. Preservation of nutritional-status in patients with refractory ascites due to hepatic cirrhosis who are undergoing repeated paracentesis. J Gastroenterol Hepatol. 2012;27:813-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 44] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 10. | Elsabaawy M, Ragab M, Naguib M, Kamal E, Al-Khalifa M, Gamil K, Elfayoumy M. Impacts of salt restriction on nutritional status, sarcopenia, and mortality of cirrhotic patients with ascites. BMC Gastroenterol. 2025;25:521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 11. | Bernardi M, Laffi G, Salvagnini M, Azzena G, Bonato S, Marra F, Trevisani F, Gasbarrini G, Naccarato R, Gentilini P. Efficacy and safety of the stepped care medical treatment of ascites in liver cirrhosis: a randomized controlled clinical trial comparing two diets with different sodium content. Liver. 1993;13:156-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 64] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 12. | Descos L, Gauthier A, Levy VG, Michel H, Quinton A, Rueff B, Fermanian J, Fombonne E, Durbec JP. Comparison of six treatments of ascites in patients with liver cirrhosis. A clinical trial. Hepatogastroenterology. 1983;30:15-20. [PubMed] |
| 13. | Wong F, Nadim MK, Kellum JA, Salerno F, Bellomo R, Gerbes A, Angeli P, Moreau R, Davenport A, Jalan R, Ronco C, Genyk Y, Arroyo V. Working Party proposal for a revised classification system of renal dysfunction in patients with cirrhosis. Gut. 2011;60:702-709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 280] [Article Influence: 18.7] [Reference Citation Analysis (2)] |
| 14. | Prakash SS, Priyadarshi RN, Surya H, Kumar S, Anand U, Kumar R. Bedside assessment of sarcopenia in hospitalized patients with liver cirrhosis: Magnitude and clinical implications. Indian J Gastroenterol. 2024;43:821-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 15. | Montano-Loza AJ. Clinical relevance of sarcopenia in patients with cirrhosis. World J Gastroenterol. 2014;20:8061-8071. [PubMed] [DOI] [Full Text] |
| 16. | Johnson C, Praveen D, Pope A, Raj TS, Pillai RN, Land MA, Neal B. Mean population salt consumption in India: a systematic review. J Hypertens. 2017;35:3-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 17. | Fordtran JS, Rector FC Jr, Carter NW. The mechanisms of sodium absorption in the human small intestine. J Clin Invest. 1968;47:884-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 226] [Article Influence: 3.9] [Reference Citation Analysis (2)] |
| 18. | Reynolds TB, Lieberman FL, Goodman AR. Advantages of treatment of ascites without sodium restriction and without complete removal of excess fluid. Gut. 1978;19:549-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 19. | Gauthier A, Levy VG, Quinton A, Michel H, Rueff B, Descos L, Durbec JP, Fermanian J, Lancrenon S. Salt or no salt in the treatment of cirrhotic ascites: a randomised study. Gut. 1986;27:705-709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 48] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 20. | Licata G, Tuttolomondo A, Licata A, Parrinello G, Di Raimondo D, Di Sciacca R, Cammà C, Craxì A, Paterna S, Pinto A. Clinical Trial: High-dose furosemide plus small-volume hypertonic saline solutions vs. repeated paracentesis as treatment of refractory ascites. Aliment Pharmacol Ther. 2009;30:227-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 21. | Cullaro G, Chiou SH, Fenton C, Ge J, McCulloch CE, Rubin J, Shui AM, Yao F, Lai JC. Outpatient mean arterial pressure: A potentially modifiable risk for acute kidney injury and death among patients with cirrhosis. Liver Transpl. 2024;30:679-688. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 22. | Maiwall R, Rao Pasupuleti SS, Hidam AK, Kumar A, Tevethia HV, Vijayaraghavan R, Majumdar A, Prasher A, Thomas S, Mathur RP, Kumar G, Sarin SK. A randomised-controlled trial (TARGET-C) of high vs. low target mean arterial pressure in patients with cirrhosis and septic shock. J Hepatol. 2023;79:349-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (1)] |
| 23. | Kumar R, Prakash SS, Priyadarshi RN, Anand U. Sarcopenia in Chronic Liver Disease: A Metabolic Perspective. J Clin Transl Hepatol. 2022;10:1213-1222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 24. | Mishra S, Ingole S, Jain R. Salt sensitivity and its implication in clinical practice. Indian Heart J. 2018;70:556-564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 25. | Olde Engberink RHG, Selvarajah V, Vogt L. Clinical impact of tissue sodium storage. Pediatr Nephrol. 2020;35:1373-1380. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 26. | Titze J, Shakibaei M, Schafflhuber M, Schulze-Tanzil G, Porst M, Schwind KH, Dietsch P, Hilgers KF. Glycosaminoglycan polymerization may enable osmotically inactive Na+ storage in the skin. Am J Physiol Heart Circ Physiol. 2004;287:H203-H208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 245] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 27. | Kumar R, Anand U, Priyadarshi RN. Lymphatic dysfunction in advanced cirrhosis: Contextual perspective and clinical implications. World J Hepatol. 2021;13:300-314. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |