Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.111570
Revised: July 26, 2025
Accepted: October 22, 2025
Published online: June 20, 2026
Processing time: 294 Days and 12.3 Hours
Traumatic brain injury (TBI) remains a significant global health burden. Electrolyte imbalances contribute to secondary brain injury by exacerbating cerebral edema, neuronal dysfunction, and increasing mortality. Electrolyte-derived clinical indices, based on routine laboratory tests, provide rapid and cost-effective tools for clinical decision-making and prognostication. This narrative review summarizes the current evidence on key electrolyte indices relevant to TBI, including the glucose-to-potassium ratio (GPR), plasma and effective osmolality, ion shift index (ISI), anion gap, Combined Electrolyte Disturbance, and Corrected Electrolytes. Each index is discussed in terms of its physiological basis, clinical utility, limitations, and prognostic relevance. Among these, GPR and osmolality have demonstrated substantial predictive value for mortality and functional outcomes, while ISI and combined electrolyte abnormalities are promising but require further validation. Adjustments to sodium and calcium levels, respectively, for glucose and albumin, prevent misdiagnosis and support more accurate treatment. These indices help bridge the gap between bedside monitoring and early risk stratification in the management of TBI. Their utility lies in their accessibility and potential to individualize care in both resource-rich and resource-limited settings. Future research should prioritize multicenter prospec
Core Tip: Electrolyte tests are used for monitoring physiological stress in traumatic brain injury. Basic electrolyte calcu
- Citation: Mekkodathil A, El-Menyar A, Rizoli S, Al-Thani H. Electrolyte-derived clinical indices in traumatic brain injury: A narrative review. World J Methodol 2026; 16(2): 111570
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/111570.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.111570
Traumatic brain injury (TBI) is a significant global health burden, with an estimated 69 million cases annually, making it a leading cause of mortality and long-term disability, particularly among young adults and the elderly[1]. TBI results from external mechanical forces disrupting the skull, meninges, brain parenchyma, or intracranial vasculature, leading to a spectrum of neurological impairments ranging from mild concussions to severe, life-altering disabilities[2]. The pathophysiology of TBI is complex, involving primary mechanical damage followed by secondary injury cascades that exacerbate neuronal loss and functional deficits over hours to weeks post-injury[3]. Among these secondary mechanisms, electrolyte dysregulation is a critical, yet modifiable, contributor to adverse outcomes, driving cerebral edema, elevation of intracranial pressure (ICP), and cellular dysfunction[4].
Electrolyte imbalances in TBI arise from trauma-induced cellular disruption, neuroendocrine dysregulation, or iatrogenic factors such as fluid resuscitation, osmotic diuretics (e.g., mannitol), or massive blood loss[5-7]. These im
The high prevalence and profound impact of electrolyte disturbances in TBI underscore the need for early, reliable prognostic indicators to guide clinical management. Individual electrolyte abnormalities (sodium, potassium, chloride, calcium, magnesium, and phosphate) are well-documented in their correlation with TBI outcomes[6,8,9]. Electrolyte levels from routine blood tests at hospital admission should be monitored, including potassium, calcium, and mag
There are several electrolyte-related clinical indices in TBI, including the glucose-to-potassium ratio (GPR), plasma osmolality (Posm), effective osmolality, anion gap (AG), ion shift index (ISI), Combined Electrolyte Disturbances, and Corrected Electrolytes (Sodium and Calcium Adjustments) (Table 1).
| Index | Components | Clinical relevance | Prognostic use in TBI | Limitations |
| GPR[11-20] | Glucose/K+ | Reflects neuroendocrine stress | Predicts mortality, poor outcomes (OR: 4.08-5.26); GPR > 50 linked to poor prognosis | Cutoffs vary and are affected by glucose/K+ fluctuations; there are few multicenter validations |
| Plasma osmolality[28-32] | 2 × Na+ + glucose/18 + BUN/2.8 | Indicates total osmotic load | Levels > 320 mOsm/kg associated with mortality, BBB disruption | BUN may overestimate osmolality |
| Effective osmolality[29,30-34] | 2 × Na+ + Glucose/18 | Reflects true water shifts across the BBB | Critical in managing ICP, preventing edema | Requires the exclusion of urea |
| Ion shift index[35,36] | (K+ + Mg2+ + | Marker of cellular membrane failure | Promising in trauma and OHCA; not yet validated in TBI | Lack of prospective TBI studies |
| Anion gap[42-44] | Na+ – (Cl- + HCO3-) | Marker of metabolic acidosis | Suggests hypoperfusion, a potential prognostic tool | Not specific to brain injury; influenced by many variables |
| Corrected sodium | Na+ + 0.016 × (glucose-100) | Prevents pseudohyponatremia | Guides the osmotherapy decisions | Formula-specific interpretation |
| Corrected calcium | Ca + 0.8 × (4–albumin) | Accounts for hypoalbuminemia | Identifies true hypocalcemia | Ionized Ca is preferred, but not always available |
The GPR has emerged as a promising and accessible prognostic biomarker in patients with TBI and other acute brain insults due to its simplicity, cost-effectiveness, and rapid availability[11-13]. Its utility lies in capturing the combined impact of systemic metabolic stress through two commonly available serum markers: Glucose and potassium. GPR is calculated as the ratio of serum glucose (in mg/dL or mmol/L) to serum potassium (in mEq/L or mmol/L). Compared to either marker alone, this ratio provides a more comprehensive view of the physiological disturbance following acute neurological injury[11-20]. Several studies have established the association of elevated GPR with poor outcomes in TBI and hemorrhagic stroke. Zhou et al[14] reported that GPR was an independent predictor of 30-day mortality in severe TBI. Similarly, Liu et al[19] showed that higher GPR quartiles were linked with increased mortality at 30, 90, and 365 days in critically ill patients with intracerebral hemorrhage.
In TBI and other acute neurological conditions, the neuroendocrine stress response triggers a cascade involving hy
Prognostic utility of GPR: Elevated GPR has consistently been associated with poor outcomes in TBI. Studies show that a GPR ≥ 50 is linked with increased mortality, poor GCS scores, and unfavorable Glasgow Outcome Scale scores. Zhou et al[14] demonstrated its predictive value for 30-day mortality (OR: 5.256), while Liu et al[19] confirmed associations with 90-day and 1-year mortality. Additionally, high GPR is correlated with functional disability (e.g., mRS ≥ 4)[13,16] and even rebleeding risk in aneurysmal subarachnoid hemorrhage[12].
GPR is a practical, rapid, and cost-effective tool in neurocritical care. It relies on routine laboratory values, making it accessible in both high- and low-resource settings. Clinicians can obtain GPR results quickly, within minutes, supporting early decision-making during triage and intensive care. However, several limitations should be considered. Most GPR studies are retrospective and conducted at a single center, which limits their generalizability. For example, Shibata et al[11] and Zhou et al[14] conducted retrospective analyses with relatively small cohorts. Liu et al[19] excluded patients with diabetes, renal failure, and malignancies to minimize confounding, but this limitation reduces the real-world applicability. The time of measurement was inconsistent. Wang et al[12] and Fujiki et al[17] assessed GPR only at admission, missing potential prognostic value in later phases of care. Furthermore, the impact of therapeutic interventions, such as insulin use or potassium correction, was not addressed in studies like Alışkan et al[16]. These methodological constraints underscore the need for prospective, multicenter research to validate GPR thresholds, assess temporal dynamics, and determine its impact on clinical decisions.
Posm and effective osmolality (tonicity) are crucial parameters in the management of TBI, as they have a profound impact on cerebral edema, ICP, and ultimately, neurological outcomes. Disruptions in osmotic homeostasis, whether hy
Plasma osmolality quantifies the total concentration of osmotically active particles in plasma, mainly sodium, glucose, and urea. It is commonly calculated using the following formula:
Plasma osmolality (mOsm/kg H2O) = 2 × serum Na+ (mEq/L) + glucose (mg/dL)/18 + BUN (mg/dL)/2.8[4,29].
Normal plasma osmolality ranges between 280 and 295 mOsm/kg H2O, and even slight deviations from this range can significantly impact cellular and cerebral function[4]. In the context of TBI, hyperosmolality frequently occurs due to osmotherapy, such as the use of mannitol or hypertonic saline, or as a result of underlying metabolic derangements. While hyperosmolar therapies are essential for lowering ICP and controlling cerebral edema, plasma osmolality should be carefully managed to avoid exceeding 320 mOsm/kg H2O, as higher levels can impair renal, cardiac, and immune function, and compromise the blood–brain barrier[28,30-32]. Hypo-osmolality, although less common, may arise due to conditions such as the syndrome of inappropriate SIADH and can rapidly worsen cerebral edema. This is particularly concerning in acute TBI, where even transient hyponatremia can exacerbate secondary brain injury[4,31]. Measurement of Posm, especially via direct osmometry, is considered more accurate than calculation and is strongly recommended for critically ill neurologic patients.
Effective osmolality, or tonicity, specifically refers to the concentration of impermeable solutes that drive osmotic water movement across cellular membranes, primarily sodium and its accompanying anions. Unlike total Posm, it excludes urea, which readily equilibrates across membranes and thus does not contribute to transcellular water shifts. Effective osmolality is calculated as:
Effective osmolality (mOsm/kg H2O) ≈ 2 × serum Na+ (mEq/L) + glucose (mg/dL)/18[29,33].
This measure is crucial because these effective solutes drive water movement into or out of brain cells. Rapid fluctuations in effective osmolality can lead to significant complications, including cerebral swelling due to acute hypotonicity or cellular dehydration, and osmotic demyelination resulting from sudden hypertonicity[4].
Effective regulation of osmolality is central to the management of TBI. Osmotherapy, utilizing agents such as mannitol or hypertonic saline, is commonly employed to increase plasma osmolality and facilitate osmotic diuresis, which effectively reduces cerebral edema and ICP. The therapeutic target for plasma osmolality is typically maintained between 300 mOsm/kg H2O and 320 mOsm/kg H2O[30,34]. However, excessive hyperosmolality, defined as levels ≥ 320 mOsm/kg H2O, has been linked to adverse outcomes, including disruption of the blood-brain barrier, organ dysfunction, and increased mortality in TBI patients[31,32]. Furthermore, disrupted osmoregulation may indicate underlying hypothalamic injury, which is frequently associated with poor clinical outcomes. Specifically, sustained elevations in plasma osmolality beyond recommended thresholds have been correlated with higher mortality rates and worse functional recovery[28].
Notably, effective osmolality is a more accurate representation of the osmotic gradient affecting brain tissue volume. This has implications not only for acute ICP management but also for the prevention of delayed cerebral ischemia, electrolyte shifts, and osmotic demyelination.
In short, both plasma osmolality and effective osmolality are pivotal parameters in the management of TBI. Accurate assessment and cautious regulation of these indices are crucial to minimizing cerebral edema, preventing secondary injury, and enhancing patient outcomes. Clinical management should aim to maintain plasma osmolality within the range of 300–320 mOsm/kg H2O, ensuring that therapeutic benefits are balanced against the risks of osmotic stress. Monitoring both total and effective osmolality offers a physiologically grounded approach to optimizing care for neurocritically ill patients.
The ISI is an emerging biochemical marker designed to quantify the degree of acute ionic imbalance following severe physiological insults such as TBI, major trauma, or cardiac arrest. This composite index reflects the dysregulation of key electrolytes resulting from cellular energy failure and membrane pump dysfunction, hallmarks of early secondary injury across critical illnesses[35,36].
The ISI is calculated using the following formula:
ISI = [Potassium (mmol/L) + Magnesium (mmol/L) + Phosphate (mmol/L)]/Calcium (mmol/L)[35].
This formula integrates key intracellular ions (potassium, phosphate, magnesium), which leak into the extracellular space during acute injury, and contrasts them against extracellular calcium, which typically floods into cells during excitotoxic or ischemic events. As such, ISI reflects the net ion shift across the cell membrane, serving as a surrogate for membrane integrity, energy failure, and cell death; higher values indicate more severe cellular injury[35,36].
Although not yet widely applied in TBI-specific prognostic models, the theoretical foundation for ISI in TBI is robust. Following a concussive or diffuse axonal brain injury, rapid fluxes of potassium, calcium, and glutamate occur, which overwhelm neuronal homeostasis and contribute to secondary injury cascades. Intracellular ATP depletion impairs Na+/K+ pumps, leading to ionic shifts and cytotoxic edema[35]. Potassium efflux and calcium influx are well-documented in the early phases of TBI, reflecting energy-dependent pump failure and excitotoxic neurotransmission[37-39]. Therefore, ISI holds theoretical validity in TBI as a real-time marker of neuronal injury severity, although large-scale validation studies are still lacking.
In polytrauma patients (excluding isolated TBI), ISI has demonstrated strong prognostic utility. Lee et al[35] found that initial ISI values, measured upon arrival at the emergency department, were independently associated with in-hospital mortality in patients with severe trauma. Higher ISI reflected greater physiological derangement and worse outcomes. Similarly, Kim et al[36] demonstrated that the ISI was a significant predictor of 30-day mortality in patients undergoing damage control laparotomy. The ISI outperformed several conventional scoring systems, underscoring its predictive precision and ease of calculation. These findings position ISI as a valuable early tool for risk stratification in trauma resuscitation protocols.
In survivors of out-of-hospital cardiac arrest (OHCA), the ISI offers a rapid biochemical snapshot of cellular viability and systemic stress. Kim et al[36] demonstrated that an elevated ISI within one hour of return of spontaneous circulation was strongly associated with poor neurological outcomes at 6 months. Notably, ISI showed comparable prognostic performance to established cardiac arrest scores, suggesting its potential as a simpler, physiology-based early warning tool.
The ISI provides a quantitative biochemical signature of cellular injury severity, capturing the systemic impact of trauma, hypoxia, or ischemia. Its relevance in TBI is supported by the central role of ionic imbalance in secondary brain injury[35-41], its ability to integrate multiple electrolytes into a single dynamic index[35,36], and its rapid measurability using routine lab parameters.
In trauma and cardiac arrest patients, ISI has proven helpful for early prognosis and clinical decision-making, outperforming traditional scoring systems in some settings. For TBI, its potential lies in its incorporation into multimodal prognostic models, real-time monitoring of electrolyte-based injury patterns, and possibly guiding electrolyte repletion or neuroprotective strategies.
While further validation is needed in TBI-specific cohorts, the ISI holds significant promise as a next-generation electrolyte-based marker for assessing systemic stress and neuronal injury. Its integration into TBI care pathways could enhance early prognosis, support individualized treatment plans, and enrich our understanding of cellular-level dysfunction in critical neurologic illness.
The AG is a derived biochemical index used to evaluate acid–base disturbances and underlying metabolic imbalances, particularly metabolic acidosis. It is calculated as the difference between the major measured cations and anions in the serum:
AG = [Na+] − ([Cl-] + [HCO3-])[42].
A normal AG typically ranges from 10 to 16 mEq/L, depending on laboratory parameters. An elevated AG signals the presence of unmeasured anions, such as lactate, ketones, or organic acids, and is frequently seen in shock states, sepsis, or prolonged ischemia.
While AG is widely used in critical care and trauma resuscitation, its direct prognostic application in TBI has not been fully established. However, several lines of evidence suggest its potential clinical utility in this setting. TBI patients often develop metabolic acidosis due to secondary systemic insults like hypoxia, hypotension, or seizures, which may lead to AG elevation[42]. An elevated AG reflects anaerobic metabolism and tissue hypoperfusion, conditions known to exacerbate secondary brain injury and worsen outcomes in polytrauma[43]. Although most existing AG research focuses on general trauma or subarachnoid hemorrhage, its pathophysiological relevance in TBI is compelling, especially in cases of prolonged hypotension, delayed resuscitation, or ongoing seizures[43,44]. Routine monitoring of AG in TBI is standard in many intensive care units (ICUs). However, its use as a standalone prognostic marker remains to be validated in TBI-specific cohorts.
In summary, although the AG is not yet a validated prognostic index in TBI, it holds promise as an indirect marker of metabolic crisis, particularly in the presence of systemic derangements commonly associated with moderate-to-severe TBI. Future research should investigate AG dynamics in relation to lactate, base deficit, and electrolyte-based indices (e.g., GPR, ISI) to determine whether it can serve as part of a multi-marker panel for outcome prediction in TBI populations.
While individual electrolyte abnormalities, such as hypernatremia, hypokalemia, hypocalcemia, and hypomagnesemia, have been independently linked to poor outcomes in TBI, recent research is shifting toward examining combinations of these disturbances as potential prognostic markers[8] (Table 2). The emerging concept of “electrolyte signatures”, or clinically relevant clusters of concurrent electrolyte derangements, is gaining recognition for its potential to indicate increased clinical severity in critical illness[45-47].
| Electrolyte pattern | Prognostic signal | Evidence strength | Remarks |
| Hypernatremia + hypocalcemia | ↑ Mortality (univariate) | Limited | Not confirmed in multivariate analysis[45] |
| Hypernatremia + hypokalemia + metabolic acidosis | Hypoperfusion marker | Limited | Triad not validated in TBI studies[5,8,9] |
| Hypernatremia + hypokalemia + hypophosphatemia | Cellular derangement | Theoretical | Not studied; potential "danger triad"[8] |
| Hypocalcemia alone | Strong adverse prognostic marker | High | Independently predicts mortality[9,46] |
| Hypernatremia alone | Linked with mortality and DI | High | Especially with high ICP or osmotherapy[6,46] |
| Hypokalemia alone | Associated with poor outcomes | High | Early TBI marker[8,9] |
Critically ill patients, including those with TBI, often exhibit multiple electrolyte abnormalities simultaneously. These patterns are associated with increased morbidity, mortality, and a more complex clinical trajectory[45-47]. Although the prognostic utility of individual electrolyte disturbances is well supported, the additive or synergistic value of specific combinations remains under investigation.
Hypernatremia and hypocalcemia: Wang et al[46] observed that both hypernatremia and hypocalcemia were indivi
Hypernatremia, hypokalemia, and metabolic acidosis: Each of these derangements has been independently associated with increased mortality and tissue hypoperfusion in TBI and critical illness[5,8,9]. Nonetheless, no prospective studies have validated this triad as a distinct or superior predictor of mortality in the postoperative or acute TBI setting.
Hypernatremia + hypokalemia + hypophosphatemia: This combination may reflect profound metabolic disruption. Although each abnormality is concerning[8], the co-occurrence of all three has not been established as a validated predictor of adverse outcomes in TBI.
Recent cohort studies report high prevalence rates of individual electrolyte abnormalities in moderate-to-severe TBI: Hypocalcemia (up to 67.3%), hypokalemia (up to 48.1%), and hypernatremia (up to 36.5%)[9,46,48]. Hypomagnesemia is also observed, but it is reported less consistently.
These abnormalities follow distinct temporal patterns. Hypocalcemia and hypokalemia often occur early post-injury[9,46,48]. Hypernatremia becomes prominent in patients with rising ICP, particularly those requiring hyperosmolar therapy or developing central diabetes insipidus[6]. In later stages, hyponatremia may develop due to SIADH or cerebral salt wasting[6,46].
Underlying metabolic changes often complicate the interpretation of electrolytes in critically ill patients. In TBI, where precise osmotic balance and ionic stability are crucial for cerebral homeostasis, adjusting key electrolyte values to account for confounding factors such as hyperglycemia or hypoalbuminemia is necessary for accurate diagnosis and safe intervention. Two such corrections, correcting sodium for hyperglycemia and correcting calcium for albumin levels, are particularly relevant in TBI management[49-51] (Table 3).
| Correction | Formula | Use case | Importance |
| Corrected sodium (mg/dL glucose)[50,51] | Na+ + 0.016 × (glucose − 100) | Hyperglycemia | Prevents misdiagnosis of hyponatremia |
| Corrected sodium (mmol/L glucose)[50,51] | Na+ + 0.024 × (glucose − 5.6) | Hyperglycemia (SI units) | Same as above |
| Corrected calcium (g/dL albumin)[51] | Ca + 0.8 × (4 − albumin) | Hypoalbuminemia | Prevents under-treatment of hypocalcemia |
| Corrected calcium (g/L albumin)[51] | Ca + 0.02 × (40 − albumin) | Hypoalbuminemia (SI units) | Same as above |
Stress-induced hyperglycemia is common following TBI and is independently associated with increased mortality, worsened neurological outcomes, and prolonged ICU stays[52]. High blood glucose levels increase serum osmolality, creating an osmotic force that draws water out of cells and dilutes extracellular sodium, resulting in pseudohypo
Failure to correct sodium for hyperglycemia may lead to inappropriate fluid restriction or excessive osmotherapy, both of which can exacerbate cerebral injury[50,53,54]. Therefore, corrected sodium is essential for guiding the management of ICP and cerebral edema in hyperglycemic TBI patients. To account for hyperglycemia-induced pseudohyponatremia, sodium should be corrected using the formula:
Corrected sodium (mmol/L) = Measured Na+ + 0.016 × [Glucose (mg/dL) − 100].
An alternate formula can be used when glucose is measured in mmol/L:
Corrected Na+ = Measured Na+ + 0.024 × [Glucose (mmol/L) − 5.6][50,51].
In critically ill patients, total serum calcium levels may not accurately reflect ionized (active) calcium, especially in the presence of hypoalbuminemia. Albumin binds a significant portion of calcium in the plasma. When albumin is low, as commonly seen in TBI due to inflammation, catabolic stress, or fluid shifts, total calcium can appear falsely low[55,56].
Ionized calcium is essential for neuronal excitability, blood coagulation, and membrane stability. Inaccurate assessment of calcium status may result in missed hypocalcemia, which can contribute to seizures, cardiac arrhythmia, or coagulopathy in TBI patients. Corrected calcium is often used to guide calcium replacement decisions when ionized calcium measurement is unavailable, aiming to provide a more physiologically relevant value[55,56]. When albumin levels are low, total calcium must be corrected using:
Corrected calcium (mg/dL) = Measured Ca + 0.8 × [4.0 − Albumin (g/dL)] or corrected calcium (mmol/L) = Measured Ca + 0.02 × [40 − Albumin (g/L)] for SI units[9,51].
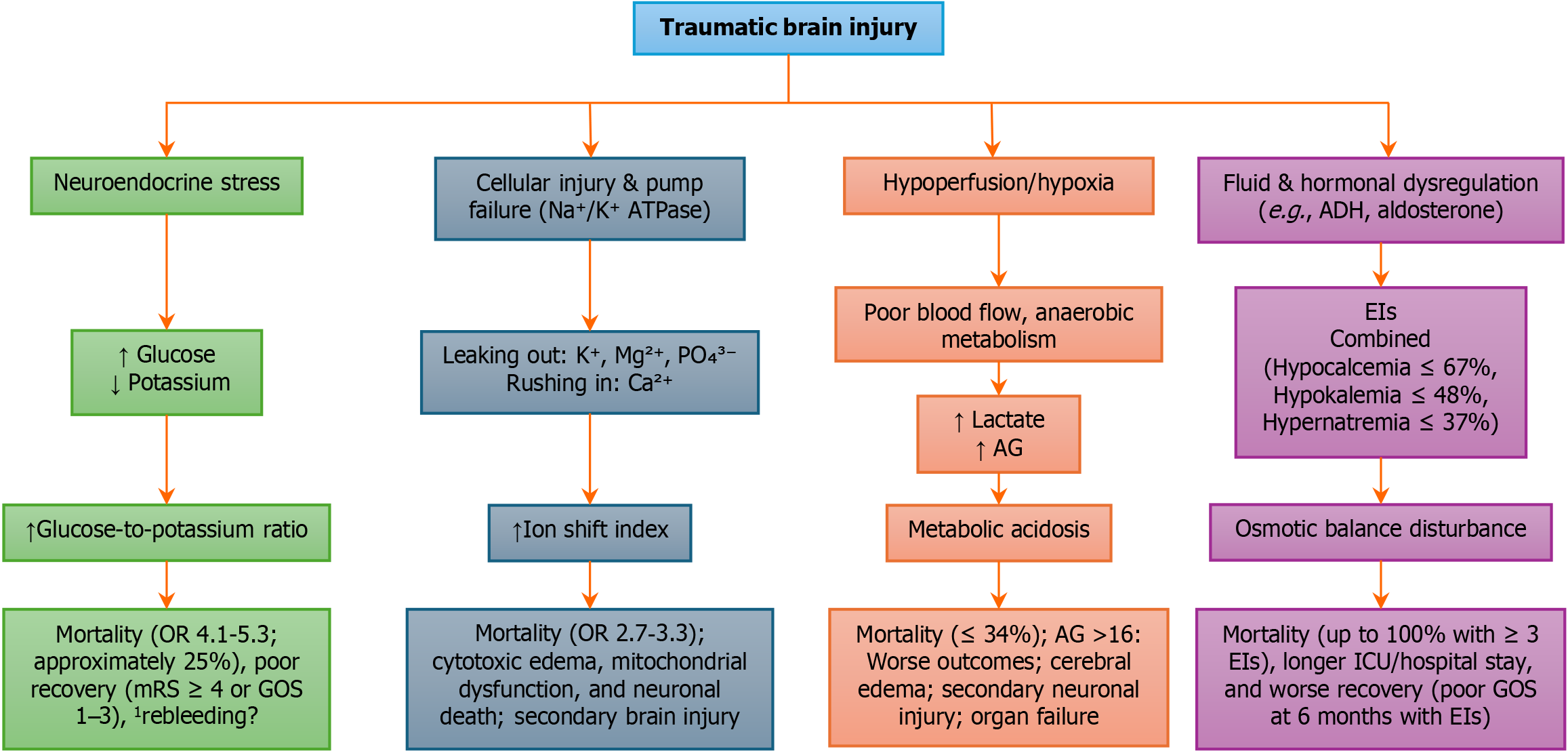
In essence, routine use of corrected sodium and corrected calcium is recommended in the neurocritical management of TBI. These adjustments help clinicians avoid diagnostic pitfalls and tailor electrolyte therapies more precisely, particularly in the presence of hyperglycemia or hypoalbuminemia, which are common yet under-recognized disruptors of accurate biochemical interpretation in TBI patients. Figure 1 summarizes the pathophysiological pathways linking clinical indices to secondary brain injury in TBI.
Electrolyte-derived clinical indices represent a promising and underutilized domain in the management of TBI. Tools such as the GPR, ISI, AG, and osmolality-based calculations provide rapid, bedside-accessible insights into physiological stress and injury severity. By utilizing standard laboratory data, these indices support real-time risk stratification, early triage decisions, and targeted interventions, particularly in settings where access to neuroimaging or biomarkers is limited. From a clinical perspective, integrating these indices into decision-making could reduce the time to intervention, optimize use of hyperosmolar therapy, and identify patients at risk for deterioration. For instance, routine calculation of GPR or ISI in the emergency or ICU setting could trigger early alerts for intensive monitoring or neuroprotective strategies. Future research should validate these markers in large, prospective, multicenter studies and standardize cutoff values based on TBI severity and clinical stage. It should explore their temporal dynamics, treatment responses, and integration into composite scoring systems for better prognostic accuracy.
Electrolyte disturbances are common and clinically significant in patients with TBI. Individual abnormalities, such as hypernatremia, hypokalemia, hypocalcemia, and altered osmolality, have established associations with poor outcomes. This review highlights the unique value of simple, widely available electrolyte-derived indices, such as the GPR, ISI, and AG, as rapid and cost-effective prognostic tools. These indices not only reflect physiological stress and injury severity but also offer opportunities for real-time decision-making, especially in settings with limited access to neuroimaging or advanced biomarkers. Importantly, this is one of the first narrative reviews to collectively evaluate these indices and propose their integration into individualized care models for TBI. The discussion of combined electrolyte disturbances and correction formulas also highlights how routine tests can be utilized more effectively to prevent misinterpretation and treatment delays. As TBI management evolves toward precision medicine, incorporating these accessible and biologically meaningful indices could improve early risk stratification, guide interventions, and enhance long-term outcomes. Future multicenter prospective studies are essential to standardize thresholds, validate composite scores, and embed these tools into clinical workflows.
| 1. | Dewan MC, Rattani A, Gupta S, Baticulon RE, Hung YC, Punchak M, Agrawal A, Adeleye AO, Shrime MG, Rubiano AM, Rosenfeld JV, Park KB. Estimating the global incidence of traumatic brain injury. J Neurosurg. 2019;130:1080-1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2083] [Cited by in RCA: 1821] [Article Influence: 260.1] [Reference Citation Analysis (0)] |
| 2. | Maas AIR, Menon DK, Adelson PD, Andelic N, Bell MJ, Belli A, Bragge P, Brazinova A, Büki A, Chesnut RM, Citerio G, Coburn M, Cooper DJ, Crowder AT, Czeiter E, Czosnyka M, Diaz-Arrastia R, Dreier JP, Duhaime AC, Ercole A, van Essen TA, Feigin VL, Gao G, Giacino J, Gonzalez-Lara LE, Gruen RL, Gupta D, Hartings JA, Hill S, Jiang JY, Ketharanathan N, Kompanje EJO, Lanyon L, Laureys S, Lecky F, Levin H, Lingsma HF, Maegele M, Majdan M, Manley G, Marsteller J, Mascia L, McFadyen C, Mondello S, Newcombe V, Palotie A, Parizel PM, Peul W, Piercy J, Polinder S, Puybasset L, Rasmussen TE, Rossaint R, Smielewski P, Söderberg J, Stanworth SJ, Stein MB, von Steinbüchel N, Stewart W, Steyerberg EW, Stocchetti N, Synnot A, Te Ao B, Tenovuo O, Theadom A, Tibboel D, Videtta W, Wang KKW, Williams WH, Wilson L, Yaffe K; InTBIR Participants and Investigators. Traumatic brain injury: integrated approaches to improve prevention, clinical care, and research. Lancet Neurol. 2017;16:987-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 983] [Cited by in RCA: 1794] [Article Influence: 199.3] [Reference Citation Analysis (0)] |
| 3. | Ng SY, Lee AYW. Traumatic Brain Injuries: Pathophysiology and Potential Therapeutic Targets. Front Cell Neurosci. 2019;13:528. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 170] [Cited by in RCA: 576] [Article Influence: 82.3] [Reference Citation Analysis (0)] |
| 4. | Tran V, Flores J, Sheldon M, Pena C, Nugent K. Fluid and Electrolyte Disorders in Traumatic Brain Injury: Clinical Implications and Management Strategies. J Clin Med. 2025;14:756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 5. | Kocik VI, April MD, Rizzo JA, Dengler BA, Schauer SG. A Review of Electrolyte, Mineral, and Vitamin Changes After Traumatic Brain Injury. Mil Med. 2024;189:e101-e109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 6. | Tomkins M, Green D, O'Reilly MW, Sherlock M. Fluid and electrolyte disorders following traumatic brain injury. Best Pract Res Clin Endocrinol Metab. 2025;39:102014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 7. | Pin-On P, Saringkarinkul A, Punjasawadwong Y, Kacha S, Wilairat D. Serum electrolyte imbalance and prognostic factors of postoperative death in adult traumatic brain injury patients: A prospective cohort study. Medicine (Baltimore). 2018;97:e13081. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 8. | Ngatuvai M, Martinez B, Sauder M, Beeton G, Andrade R, Maka P, Smith CP, Kornblith L, Elkbuli A. Traumatic Brain Injury, Electrolyte Levels, and Associated Outcomes: A Systematic Review. J Surg Res. 2023;289:106-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 9. | Dey S, Kumar R, Tarat A. Evaluation of Electrolyte Imbalance in Patients With Traumatic Brain Injury Admitted in the Central ICU of a Tertiary Care Centre: A Prospective Observational Study. Cureus. 2021;13:e17517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 10. | Chen X, Xu J, Li Y, Xu X, Shen B, Zou Z, Ding X, Teng J, Jiang W. Risk Scoring Systems Including Electrolyte Disorders for Predicting the Incidence of Acute Kidney Injury in Hospitalized Patients. Clin Epidemiol. 2021;13:383-396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 11. | Shibata A, Matano F, Saito N, Fujiki Y, Matsumoto H, Mizunari T, Morita A. Serum Glucose-To-Potassium Ratio as a Prognostic Predictor for Severe Traumatic Brain Injury. J Nippon Med Sch. 2021;88:342-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 12. | Wang J, Hong C, Feng Q, Wu B, Li S, Yan C, Gao H. Glucose-potassium ratio: a prognostic biomarker enhancing outcome prediction in mild-to-moderate traumatic brain injury. Front Neurol. 2025;16:1577390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 13. | Wu XY, Zhuang YK, Cai Y, Dong XQ, Wang KY, Du Q, Yu WH. Serum glucose and potassium ratio as a predictive factor for prognosis of acute intracerebral hemorrhage. J Int Med Res. 2021;49:3000605211009689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (4)] |
| 14. | Zhou J, Yang CS, Shen LJ, Lv QW, Xu QC. Usefulness of serum glucose and potassium ratio as a predictor for 30-day death among patients with severe traumatic brain injury. Clin Chim Acta. 2020;506:166-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 15. | Marini JI, Sein ME. The Role of the Glucose Potassium Ratio in the Management of Traumatic Brain Injury. Korean J Neurotrauma. 2023;19:82-89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 16. | Alışkan H, Kılıç M, Ak R. Usefulness of plasma glucose to potassium ratio in predicting the short-term mortality of patients with aneurysmal subarachnoid hemorrhage. Heliyon. 2024;10:e38199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 17. | Fujiki Y, Matano F, Mizunari T, Murai Y, Tateyama K, Koketsu K, Kubota A, Kobayashi S, Yokota H, Morita A. Serum glucose/potassium ratio as a clinical risk factor for aneurysmal subarachnoid hemorrhage. J Neurosurg. 2018;129:870-875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 18. | Jung HM, Paik JH, Kim SY, Hong DY. Association of Plasma Glucose to Potassium Ratio and Mortality After Aneurysmal Subarachnoid Hemorrhage. Front Neurol. 2021;12:661689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 19. | Liu J, Luo F, Guo Y, Li Y, Jiang C, Pi Z, Luo J, Long Z, Wen J, Huang Z, Zhu J. Association between serum glucose potassium ratio and mortality in critically ill patients with intracerebral hemorrhage. Sci Rep. 2024;14:27391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 20. | Liu Y, Qiu T, Fu Z, Wang K, Zheng H, Li M, Yu G. Systemic immune-inflammation index and serum glucose-potassium ratio predict poor prognosis in patients with spontaneous cerebral hemorrhage: An observational study. Medicine (Baltimore). 2024;103:e39041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 21. | Lundqvist MH, Pereira MJ, Almby K, Hetty S, Eriksson JW. Regulation of the Cortisol Axis, Glucagon, and Growth Hormone by Glucose Is Altered in Prediabetes and Type 2 Diabetes. J Clin Endocrinol Metab. 2024;109:e675-e688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 22. | Jones BJ, Tan T, Bloom SR. Minireview: Glucagon in stress and energy homeostasis. Endocrinology. 2012;153:1049-1054. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 91] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 23. | Reid JL, Whyte KF, Struthers AD. Epinephrine-induced hypokalemia: the role of beta adrenoceptors. Am J Cardiol. 1986;57:23F-27F. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 80] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 24. | Darbar D, Smith M, Mörike K, Roden DM. Epinephrine-induced changes in serum potassium and cardiac repolarization and effects of pretreatment with propranolol and diltiazem. Am J Cardiol. 1996;77:1351-1355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 25. | Krogager ML, Mortensen RN, Lund PE, Bøggild H, Hansen SM, Kragholm K, Aasbjerg K, Søgaard P, Torp-Pedersen C. Risk of Developing Hypokalemia in Patients With Hypertension Treated With Combination Antihypertensive Therapy. Hypertension. 2020;75:966-972. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 26. | Alamri FF, Almarghalani DA, Alraddadi EA, Alharbi A, Algarni HS, Mulla OM, Alhazmi AM, Alotaibi TA, Beheiry DH, Alsubaie AS, Alkhiri A, Alatawi Y, Alzahrani MS, Hakami AY, Alamri A, Al Sulaiman K. The utility of serum glucose potassium ratio as a predictive factor for haemorrhagic transformation, stroke recurrence, and mortality among ischemic stroke patients. Saudi Pharm J. 2024;32:102082. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 27. | Schubert KM, Schmick A, Stattmann M, Galovic M. Prognostic models for seizures and epilepsy after stroke, tumors and traumatic brain injury. Clin Neurophysiol Pract. 2025;10:116-128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 28. | Trost HA, Gaab MR. Plasma osmolality, osmoregulation and prognosis after head injury. Acta Neurochir (Wien). 1992;116:33-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 29. | Rasouli M. Basic concepts and practical equations on osmolality: Biochemical approach. Clin Biochem. 2016;49:936-941. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 128] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 30. | Lal S, Moore M. Osmotherapy and the management of traumatic brain injury: still a dilemma. Br J Hosp Med (Lond). 2023;84:1-2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 31. | Dabrowski W, Siwicka-Gieroba D, Robba C, Bielacz M, Sołek-Pastuszka J, Kotfis K, Bohatyrewicz R, Jaroszyński A, Malbrain MLNG, Badenes R. Potentially Detrimental Effects of Hyperosmolality in Patients Treated for Traumatic Brain Injury. J Clin Med. 2021;10:4141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 32. | Ziaei M, Galavi M, Bahmani A, Abdolrazaghnejad A. The Level of Serum Osmolarity at Admission in Prognosis of Nosocomial Mortality in Patients with Severe Brain Trauma. Adv Biomed Res. 2023;12:40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 33. | Gennari FJ. Current concepts. Serum osmolality. Uses and limitations. N Engl J Med. 1984;310:102-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 174] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 34. | Williams GV. Prehospital osmotherapy in isolated traumatic brain injury: a systematic review. J Paramed Pract. 2021;13:114-124. [DOI] [Full Text] |
| 35. | Lee HY, Lee SM, Jeung KW, Lee BK, Han JH, Heo T, Min YI. Association between ion shift index and prognosis in severe trauma patients without isolated head injury. Injury. 2021;52:1151-1157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 36. | Kim B, Kwon H, Kim SM, Kim JS, Ryoo SM, Kim YJ, Kim WY. Ion Shift Index at the Immediate Post-Cardiac Arrest Period as an Early Prognostic Marker in Out-of-Hospital Cardiac Arrest Survivors. J Clin Med. 2022;11:6187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 37. | Prins M, Greco T, Alexander D, Giza CC. The pathophysiology of traumatic brain injury at a glance. Dis Model Mech. 2013;6:1307-1315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 220] [Cited by in RCA: 265] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 38. | Giza CC, Hovda DA. The Neurometabolic Cascade of Concussion. J Athl Train. 2001;36:228-235. [PubMed] |
| 39. | Blennow K, Hardy J, Zetterberg H. The neuropathology and neurobiology of traumatic brain injury. Neuron. 2012;76:886-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 554] [Cited by in RCA: 483] [Article Influence: 34.5] [Reference Citation Analysis (0)] |
| 40. | Jha RM, Kochanek PM, Simard JM. Pathophysiology and treatment of cerebral edema in traumatic brain injury. Neuropharmacology. 2019;145:230-246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 408] [Cited by in RCA: 351] [Article Influence: 50.1] [Reference Citation Analysis (0)] |
| 41. | Katayama Y, Mori T, Maeda T, Kawamata T. Pathogenesis of the mass effect of cerebral contusions: rapid increase in osmolality within the contusion necrosis. Acta Neurochir Suppl. 1998;71:289-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 42. | Kraut JA, Madias NE. Serum anion gap: its uses and limitations in clinical medicine. Clin J Am Soc Nephrol. 2007;2:162-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 249] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 43. | Caputo ND, Kanter M, Fraser R, Simon R. Comparing biomarkers of traumatic shock: the utility of anion gap, base excess, and serum lactate in the ED. Am J Emerg Med. 2015;33:1134-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 44. | Leskovan JJ, Justiniano CF, Bach JA, Cook CH, Lindsey DE, Eiferman DS, Papadimos TJ, Steinberg SM, Bergese SD, Stawicki SP, Evans DC. Anion gap as a predictor of trauma outcomes in the older trauma population: correlations with injury severity and mortality. Am Surg. 2013;79:1203-1206. [PubMed] [DOI] [Full Text] |
| 45. | Bulloch MN, Cardinale-king M, Cogle S, Radparvar S, Effendi M, Jagpal S, Dixit D. Correction of Electrolyte Abnormalities in Critically Ill Patients. Intensive Care Res. 2024;4:19-37. [DOI] [Full Text] |
| 46. | Wang GH, Yan Y, Shen HP, Chu Z. The Clinical Characteristics of Electrolyte Disturbance in Patients with Moderate and Severe Traumatic Brain Injury Who Underwent Craniotomy and Its Influence on Prognosis. J Korean Neurosurg Soc. 2023;66:332-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 47. | Ghannam M, Malihi P, Laudanski K. Examination of Electrolyte Replacements in the ICU Utilizing MIMIC-III Dataset Demonstrates Redundant Replacement Patterns. Healthcare (Basel). 2021;9:1373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 48. | Gupta H, Saxena T, Rathi P, Pandey V. Evaluation of Electrolyte Imbalance in Patients with Traumatic Brain Injury Admitted to the Central ICU of Chirayu Medical College & Hospital: A Prospective Observational Study. Afr J Biol Sci. 2024;6:4037-4041. |
| 49. | Adrogué HJ, Madias NE. Hypernatremia. N Engl J Med. 2000;342:1493-1499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 787] [Cited by in RCA: 523] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 50. | Hillier TA, Abbott RD, Barrett EJ. Hyponatremia: evaluating the correction factor for hyperglycemia. Am J Med. 1999;106:399-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 364] [Cited by in RCA: 320] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 51. | Liamis G, Liberopoulos E, Barkas F, Elisaf M. Spurious electrolyte disorders: a diagnostic challenge for clinicians. Am J Nephrol. 2013;38:50-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 52. | Cui S, Xu D, Xiong H, Zhuang Y, He Z. Stress-induced hyperglycemia and mortality in patients with traumatic brain injury without preexisting diabetes: A meta-analysis. Biomol Biomed. 2025;25:291-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 53. | Shi J, Dong B, Mao Y, Guan W, Cao J, Zhu R, Wang S. Review: Traumatic brain injury and hyperglycemia, a potentially modifiable risk factor. Oncotarget. 2016;7:71052-71061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 103] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 54. | Braun MM, Barstow CH, Pyzocha NJ. Diagnosis and management of sodium disorders: hyponatremia and hypernatremia. Am Fam Physician. 2015;91:299-307. [PubMed] |
| 55. | Kenny CM, Murphy CE, Boyce DS, Ashley DM, Jahanmir J. Things We Do for No Reason™: Calculating a "Corrected Calcium" Level. J Hosp Med. 2021;16:499-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 56. | Desgagnés N, King JA, Kline GA, Seiden-Long I, Leung AA. Use of Albumin-Adjusted Calcium Measurements in Clinical Practice. JAMA Netw Open. 2025;8:e2455251. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 23] [Article Influence: 23.0] [Reference Citation Analysis (0)] |