Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.109986
Revised: July 3, 2025
Accepted: October 15, 2025
Published online: June 20, 2026
Processing time: 331 Days and 15.9 Hours
About 25%-40% of people with epidermal growth factor receptor (EGFR)-mutated non-small cell lung cancer (NSCLC) experience brain metastases, which cause major challenges in their treatment. There continues to be no agreement on the ideal method for using EGFR- tyrosine kinase inhibitors (TKIs) and radiotherapy in management.
To systematically assess how effective and safe EGFR-TKIs are when used together with or without radiotherapy in people with EGFR mutation and brain metastases.
According to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, a complete literature search was initiated. We selected studies of two designs: Randomized trials and observational studies, which focused on therapies like EGFR-TKIs, radiotherapy, or approaches that combine both. Results for primary outcomes focused on how long people lived without the cancer spreading, whether the brain tumor shrank, and how safe the treatment was.
Ten randomized controlled trials and 17 observational studies, totaling 14955 patients, met the inclusion criteria. Osimertinib and similar third-generation EGFR-TKIs performed better in the brain, helping patients with central nervous system (CNS) responses of 60% to 91%. Treatment involving combined EGFR-TKI medicine and radio
Third-generation EGFR-TKIs have good CNS effects in EGFR-mutated NSCLC. Treatments that include radiotherapy are sometimes used together, and choosing the right method depends on the amount of cancer, its effect on the person, and patient factors, which includes brain metastases (e.g., > 3), performance status (e.g., ECOG 0-2), and severity of the symptom (e.g., moderate to severe neurological symptoms).
Core Tip: The appearance of brain metastases in epidermal growth factor receptor (EGFR)-mutated non-small cell lung cancer makes therapy harder. A review of 27 studies involving 14955 patients shows that osimertinib and other third-generation EGFR tyrosine kinase inhibitors achieve higher activity inside the brain and produce better responses than earlier drugs. Those with multiple brain metastases who are treated with combined radiation therapy and osimertinib do better than those treated with osimertinib alone. Cognitive function is better preserved by stereotactic radiosurgery than whole-brain radiotherapy, all while giving the same good results in managing tumors, backing individualized methods linked to the disease and patient profile.
- Citation: Javed M, Fatima M, Hassan MB, Kumar S, Pawan R, Singh V, Kumar K, Amir R, Karim A, Jawed I, Alam F, Kumar D, Rai R, Tilokani H, Rizvi SAFA. Radiation therapy plus osimertinib vs osimertinib alone in epidermal growth factor receptor mutated non-small cell lung cancer: A systematic review. World J Methodol 2026; 16(2): 109986
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/109986.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.109986
Most (85 percent) of lung cancer cases around the globe are non-small cell lung cancer (NSCLC), which is a major cause of cancer-related deaths worldwide. The introduction and clinical use of molecular-targeted therapies have greatly changed how this difficult cancer is treated. One major advance is when a gene called the epidermal growth factor receptor (EGFR) is altered, which can be seen in about 10%-15% of people in the West and up to 50% in some Asian populations, particularly among those who do not smoke, are women and have the adenocarcinoma type[1].
Although EGFR mutations are relevant to the prognosis, they are mainly significant for the many treatments available for patients. This genomic subtype of NSCLC is distinguished by its special biology, how it responds to therapy, and the types of resistance it can experience. Almost half of EGFR mutations are exon 19 deletions, and nearly as many are L858R point mutations in exon 21; these both increase the response to EGFR-targeted treatments. There are extra difficulties with treating less common mutations, such as those in exon 20, and compared mutations, and proper treatment approaches are required[1,2].
About 25%-40% of patients with EGFR-mutated NSCLC experience brain metastases at some stage during their illness. It appears that EGFR-mutated cancers often affect the brain more frequently than EGFR wild-type disease. Perhaps EGFR-targeted drugs work for so long that the tumor has time to develop metastases in the brain, and brain factors may also predispose these tumors to spread into the brain. A patient can have brain metastases at the very beginning, and others may develop them as treatment for their cancer is ongoing[2,3].
For much of the twentieth century, managing brain metastases in NSCLC involved mainly radiotherapy, and most cases involved whole-brain radiotherapy (WBRT). WBRT delivered lots of coverage to the brain and relieve symptoms, yet it caused serious cognitive issues and did not always help much with life expectancy. Stereotactic radiosurgery (SRS) now provides a much more focused therapy for individuals with few brain metastases, helping to guard their memory and ensuring the tumor does not come back. Still, the main treatments for tumors at the time were chemotherapies with poor ability to enter the brain, along with only moderate results.
Precision oncology and molecularly targeted therapy in NSCLC began when EGFR-tyrosine kinase inhibitors (TKIs) were developed. In EGFR-mutated NSCLC patients who had never been treated, the first-generation EGFR-TKIs gefitinib and erlotinib yielded systemic efficacy with 60%-80% response rates and progression-free survival for 9-13 months. Yet, these drugs could not deeply act on the central nervous system (CNS) because very little got past the blood-brain barrier and were carried away by efflux pumps in the brain. Despite these problems, several investigations have found CNS effects in the first generation of EGFR-TKIs, suggesting systemic drugs may have a role in CNS treatment[4,5].
The arrival of afatinib and dacomitinib was meant to prevent EGFR from attaching to many cell structures and changing shape to fit several EGFR members. While they could overcome some resistance and worked well elsewhere in the body, their poor CNS penetration meant they were often not useful for brain metastases. Most research on these agents has concentrated mainly on helping with systemic disease and not their effects on the brain[6,7].
The arrival of osimertinib among third-generation EGFR-TKIs greatly revolutionized care for patients with EGFR-mutated NSCLC that had metastasized to the brain. Osimertinib was developed to address T790M-caused resistance, yet it still affects EGFR mutations that form the basis for prior very effective treatments. Key to its success, the molecule was built to pass more easily into the brain, requiring a weaker bond with efflux pumps and a better ability to cross the blood-brain barrier. According to lab research, this class of drugs reaches the brain better than previous types, explaining the strong CNS effects observed in clinical studies[8-11].
A major barrier in treating brain cancers is the blood-brain barrier, which stops many drugs from entering the brain from the rest of the body. The boundary material includes cells joined by tight junctions, efflux pumps, and enzymes that together slow the entry of drugs. Classic chemotherapies cannot effectively enter the brain, so new treatment methods are developed for brain metastases. Acknowledging these pharmacological issues has encouraged scientists to invent new methods of drug delivery and develop brain-targeted agents[8,12].
Currently, the techniques used in radiotherapy have advanced a great deal, and advanced planning, image guidance, and optimal dose calculation have made treatment much more precise. For patients with just a few brain tumors, stereotactic radiosurgery is now the preferred form of radiotherapy, as it provides strong local control and few side effects. Today, hippocampal-avoidance whole-brain radiotherapy can help keep people's minds sharp, even as they receive effective treatment. Advanced imaging, including magnetic resonance imaging and positron emission tomo
It is now common to involve different teams and consider personalized treatments for EGFR-mutated NSCLC with brain metastases. Currently, cancer care includes molecular diagnostics, advanced imaging, precise radiation, and CNS-targeted drugs. At the same time, various questions about how to offer care are still unknown, including deciding the best order of selected drugs, choosing patients who can benefit from combined therapy, coping with drug resistance, and managing treatment effects on cognitive and quality of life[16-18].
People given EGFR-targeting drugs usually become resistant to them after about nine to eighteen months, making treatment resistance a common problem. Close to half of all cases of acquired drug resistance are due to the T790M gatekeeper mutation; other mechanisms are MET amplification, HER2 amplification, PIK3CA mutations, and changing to small-cell lung cancer. As the CNS can develop immunity separately from disease elsewhere, it needs unique care and breakthrough methods of treatment[19-21].
With modern targeted therapy, there has been more emphasis on how patients live and think as survival rates continue to increase. Protecting the brain, thinking ability, and quality of life are very important goals that must be balanced with efforts to control the disease. Patient-reported measurements and thorough neuropsychological reviews are increasingly used in clinical trials and decisions about treatment[8,14,22].
In this review, we attempt to provide a clear assessment of the present evidence for EGFR-TKI treatment, along with or without radiotherapy, for EGFR-mutated NSCLC patients with brain cancer. The authors aim to support clinicians by reviewing studies, including randomized trials and observational studies, to find the best treatments, identify information gaps, and outline what should be prioritized in related future studies.
This systematic review was conducted using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The protocol contains the study objectives, patterns for selecting subjects, data gathering methods, and analysis rules, which were decided in advance.
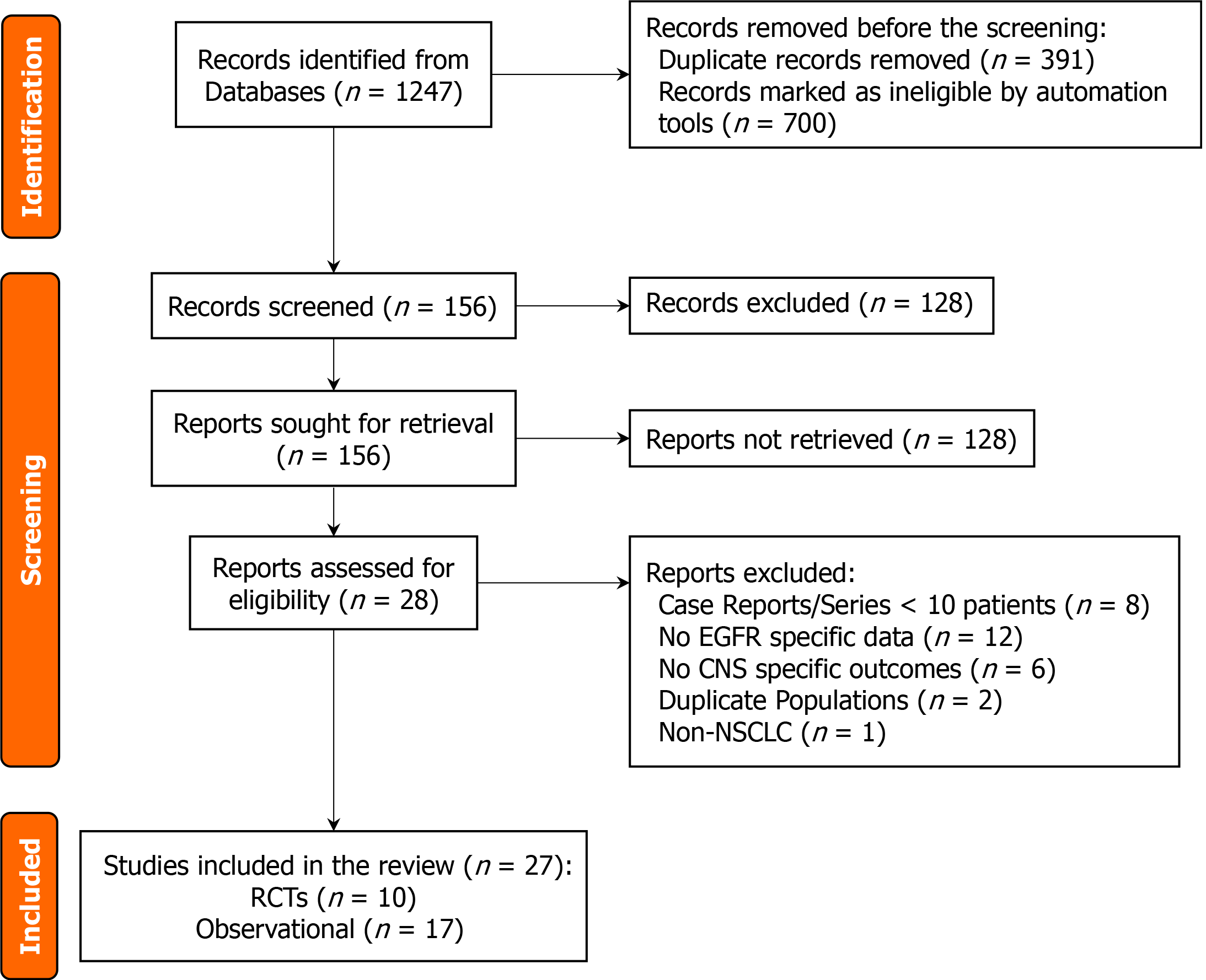
We followed the protocol outlined by PRISMA 2009[23] to select studies and extract their data. The study selection process is clearly shown in Figure 1.
The review used the population, intervention, comparison, and outcomes framework.
Population: Adult patients whose NSCLC was confirmed by biopsy to have EGFR mutations and whose disease has spread to the brain.
Intervention: First, second, or third-generation EGFR-TKIs can be used alone or in combination with radiotherapy as an intervention.
Comparison: EGFR-TKI generations, radiotherapy techniques, and whether to use combination or monotherapy.
Outcomes: Results include assessments of overall survival, time until a patient's cancer starts progressing, response rates for the CNS, time with no intracranial progression, treatment safety, and quality of life.
PubMed, EMBASE, Cochrane Central Register of Controlled Trials, and Web of Science were used to search the literature to search the literature. The search terms used were combinations of "EGFR", "epidermal growth factor receptor", "non-small cell lung cancer", "NSCLC", "brain metastases", "central nervous system", "tyrosine kinase inhibitor", "TKI", "osimertinib", "gefitinib", "erlotinib", "afatinib", "radiotherapy", "stereotactic radiosurgery" and "whole brain radio
The approach to search was discussed with a medical librarian and tried out to make sure all the relevant papers were included. The results were exported to EndNote 20, where duplicates were identified and grouped for screening.
Two experts independently reviewed all titles and abstracts, and they resolved disagreements together or by consulting with a third person. Articles were assessed for inclusion in the study by two individuals who were not collaborating.
The analysis of inter-rater agreement was performed at several phases of review. To measure agreement between the two independent reviewers, the Cohen's kappa coefficient was used to screen titles and abstracts. Both Cohen's kappa and percentage agreement were calculated to assess the accuracy of full-text screening and data extraction. According to the criteria of Landis and Koch, an excellent agreement was defined as 0.80 or above, substantial as 0.60-0.79, moderate as 0.40-0.59, and poor as less than 0.40. The statistical tests for inter-rater reliability were conducted using IBM SPSS version 27.0.
Inclusion criteria: Randomized controlled trials or/ and observational studies. Patients 18 years and older whose NSCLC is diagnosed by tissue examination. Verified mutations in the gene EGFR, for example, as in exon 19 deletions, can be categorized as L858R, T790M, or other sensitizing mutations. Confirmed Brain metastases by looking at neuro images. Treatment with EGFR-TKIs with or without radiotherapy. Included outcomes are survival, response rate, and safety information.
Exclusion criteria: Reports or series focused on a few patients (< 10 patients). Trials that do not provide data on patients with EGFR mutations. The absence of outcomes for brain metastases. Articles in languages other than English. Duplicate publications or patients included in more than one study.
We undertook the following steps to avoid double-counting of participants from papers reporting the same or overlapping cohorts. We screened studies for overlapping populations by comparing: (1) Study periods and timeframes; (2) Participating institutions and study groups; (3) Patient eligibility criteria and sample sizes; (4) Author affiliations and collaborative networks; and (5) Trial registration numbers.
Where more than one publication appeared to be a study of the same cohort of patients with different endpoints or duration of follow-up, we categorized them as follows:
Primary efficacy analysis: The primary efficacy assessment will be the earliest publication reporting progression-free survival (PFS) outcomes.
Overall survival update: The latter reports mature overall survival (OS) data with extended follow-up from earlier publications.
Subgroup analyses: Publications focusing on patients with specific characteristics (e.g., CNS metastases cohort).
There were separate data sources for each outcome, ensuring that no data was double-counted while providing full outcome data. For example, initial PFS data from FLAURA[10] and mature OS data from FLAURA-OS[9], which included the same patients with prolonged follow-up, were included for their respective outcomes (without double-counting). We reached out to the corresponding authors to confirm patient overlap in case of observational studies from the same institution with overlapping enrollment periods. In cases of confirmed overlap or inability to rule it out, we only included the most comprehensive or recent paper.
Two people independently extracted data using identical, already-developed forms. The data analyzed involved the design and setting of the original studies, patient statistics (age, sex, performance status), types of EGFR mutations, different treatment procedures, measures of effectiveness, results, and any safety-related findings. Resolving discrepancies often needed an open discussion and a consensus. We also had a third reviewer help if necessary.
To maintain reliability in the data extraction process, the two reviewers extracted data separately on a pilot sample of 10% of the included studies, after which data were extracted from all the studies. Cohen's kappa coefficient was used to evaluate the level of inter-rater agreement on key data (study design, patient characteristics, interventions and primary outcomes). Any data extraction forms that involved disagreement were revised collaboratively, and extraction rules were modified to enhance consistency.
We used the I² statistic and the χ2 test to assess the heterogeneity of the studies. Due to the high heterogeneity observed (I² > 75% for primary outcomes), which exceeded the threshold for meta-analysis, we performed a narrative synthesis instead of a quantitative analysis. Study design variations (10 randomized controlled trials vs 17 observational studies), EGFR-TKI heterogeneity generation, radiotherapy types, patient populations, and outcome measurement. The statistical strategy of meta-analysis was deemed inappropriate as pooling would violate the assumption of a common underlying effect and lead to misleading summary estimates. The results are explained clearly, paying attention to important outcomes for patients, how various treatments compare, and common patterns across subtypes of patients and their treatments.
For every study design, quality assessment was done using the appropriate methods. We applied the Cochrane Risk of Bias tool (known as RoB 2) to assess randomized controlled trials. This evaluation checked the processes of randomization, whether the intervention was implemented as intended, whether crucial data was missing, measurements of the outcomes, and the selection of study outcomes presented. Every study in the review was rated as having a low, some, or high risk of bias.
In the observational studies, we employed the propensity score matching strategy and covariate technique to balance the baseline characteristics of different treatment groups, thereby eliminating the selection bias that occurs with the retrospective design. We had also avoided confounding variables, such as age, comorbidities, and performance status, by controlling for them through multivariate studies. Other stratified analyses were also applied to ensure that the comparisons took into consideration important factors, including the number of brain metastases, which allowed us to strengthen our findings.
To evaluate observational studies, the Newcastle-Ottawa Scale was chosen and used to evaluate these main issues in groups: Representation, matching between groups, and measurements of outcomes. A study was given 7-9 stars if it was of high quality, 5-6 stars for moderate quality, and less than 5 stars for low quality.
Two viewers each performed the quality assessment, and disagreements were settled by discussion and agreement. Studies were arranged based on their overall degree of potential bias to interpret findings and combine the evidence.
The evidence quality of each outcome area was determined using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) procedure. The confidence in the evidence was assessed in five domains by two reviewers separately, namely, risk of bias, inconsistency, indirectness, imprecision, and publication bias.
In the case of randomized controlled trials, the evidence quality initially begins at a high level and can be downgraded due to severe limitations. In the case of observational studies, quality evidence was initially classified as "low" and could be improved if there were large effect sizes, a dose-response gradient, or a lack of confounding, which would dampen the observed effects.
The category of evidence quality was as follows: High: Very specific that the estimated effect is not far from the estimate. Moderate: Moderately confident in the estimate of an effect. Low: Low levels of confidence in the effect estimate. Very low: Slightly low confidence in the effect estimate. GRADE analysis was conducted using the GRADEpro GDT software package, and in cases of disagreement, a consensus was reached through discussion and agreement.
The process of searching and selecting literature is displayed in Figure 1 below. This first stage resulted in identifying 1247 possibly relevant articles from every database. No duplicate records or ineligible reports were left after using automation tools. As a result, 156 records were analyzed by screening their titles and abstracts. A full-text review of 28 articles selected 27 studies, including 10 randomized controlled trials and 17 observational studies that contained information on 14955 patients who had EGFR-mutated NSCLC and brain metastases. A summary of the characteristics of each study is shown in Table 1.
| Ref. | Design | Population | Patient characteristics | Intervention | Comparator | Primary outcome | Key findings |
| Soria et al[10], 2018 (FLAURA) | RCT (n = 556) | Treatment-naïve advanced NSCLC | Median age 64 years, 64% female, 62% Asian, PS 0-1 | Osimertinib 80 mg daily | Erlotinib 150 mg or gefitinib 250 mg daily | PFS | mPFS: 18.9 months vs 10.2 months (HR: 0.46, P < 0.001) |
| Ramalingam et al[9], 2020 (FLAURA-OS) | RCT (n = 556) | Treatment-naïve advanced NSCLC | Same cohort as FLAURA | Osimertinib 80 mg daily | Erlotinib or gefitinib | OS | mOS: 38.6 months vs 31.8 months (HR: 0.80, P = 0.046) |
| Reungwetwattana et al[24], 2018 | RCT (n = 144) | Treatment-naïve with CNS metastases | Median age 62 years, 69% female, 83% Asian, all with baseline BM | Osimertinib 80 mg daily | Erlotinib or gefitinib | CNS ORR | CNS ORR: 91% vs 68% (P = 0.0009) |
| Cho et al[25], 2019 (FLAURA Asian) | RCT (n = 357) | Asian treatment-naïve advanced NSCLC | 100% Asian, median age 62 years, 62% female | Osimertinib 80 mg daily | Erlotinib or gefitinib | PFS | mPFS: 16.5 months vs 11.0 months (HR: 0.46, P < 0.001) |
| Wu et al[11], 2018 (AURA3-CNS) | RCT (n = 144) | T790M+ post-EGFR-TKI progression | Median age 61 years, 61% female, 73% Asian, all with BM | Osimertinib 80 mg daily | Platinum-pemetrexed | CNS ORR | CNS ORR: 70% vs 31% (P = 0.015) |
| Brown et al[14], 2020 (RTOG 0933) | RCT (n = 518) | Brain metastases | Median age 61 years, 48% female, mixed primary tumors | HA-WBRT + memantine | Historical WBRT controls | Cognitive function | HVLT-R decline: 7.0% vs 30% historical (P < 0.001) |
| Brown et al[13], 2017 (NCCTG N107C) | RCT (n = 194) | Resected brain metastases | Median age 60 years, 48% female, 85% NSCLC | Post-op SRS | Post-op WBRT | Cognitive function | Better cognitive preservation with SRS |
| Brown et al[15], 2016 | RCT (n = 213) | 1-3 brain metastases | Median age 60 years, 46% female, 84% NSCLC | SRS alone | SRS + WBRT | Cognitive function | Cognitive decline: 63.5% vs 91.7% (P < 0.001) |
| Jänne et al[26], 2024 (FLAURA2-CNS) | RCT (n = 557) | Treatment-naïve advanced NSCLC | Median age 64 years, 61% female, 48% Asian | Osimertinib + chemotherapy | Osimertinib alone | PFS | Enhanced CNS activity with combination |
| Li et al[19], 2023 (FLOWERS) | RCT (n = 200) | MET aberrant, EGFR+ | Treatment-naïve with MET amplification/mutation | Osimertinib ± savolitinib | Osimertinib monotherapy | PFS | Ongoing trial design rationale |
| Shi et al[2], 2014 (PIONEER) | Cohort (n = 1482) | Asian adenocarcinoma | Median age 58 years, 58% female, 100% Asian | Observational | Not applicable | Mutation prevalence | EGFR mutations in 60.9% of adenocarcinomas |
| Gu et al[31], 2024 | Cohort (n = 186) | Advanced adenocarcinoma with BM | Median age 58 years, 55% female, TP53 co-mutations | EGFR-TKI therapy | TP53 wild-type comparison | iPFS | TP53 co-mutation: Worse iPFS (7.1 months vs 14.8 months) |
| Rangachari et al[3], 2015 | Cohort (n = 351) | EGFR+ vs ALK+ with BM | EGFR+: Median age 65 years, 76% female | Mixed therapy | ALK+ comparison | Survival patterns | BM in 25.4% EGFR+, mOS 19.9 months post-BM |
| Magnuson et al[27], 2016 | Cohort (n = 351) | Treatment-naïve with BM | Median age 65 years, 76% female, PS 0-2 | RT deferral analysis | Upfront RT comparison | OS | Comparable survival: 46 months vs 46 months (P = 0.95) |
| Ballard et al[8], 2016 | Cohort (n = 189) | Preclinical + clinical BM | Mixed characteristics | Osimertinib activity | Historical EGFR-TKI comparison | Response rates | Superior CNS penetration and activity |
| Magnuson et al[28], 2017 | Cohort (n = 351) | EGFR-TKI-naïve with BM | Median age 65 years, 76% female, multiple BM 68% | Combined vs EGFR-TKI alone | EGFR-TKI monotherapy | Intracranial control | 12 months IC progression: 21% vs 47% (P = 0.014) |
| Chen et al[29], 2019 | Cohort (n = 105) | Asymptomatic BM | Median age 59 years, 62% female, PS 0-1 | RT + TKI vs EGFR-TKI alone | EGFR-TKI monotherapy | iPFS, OS | iPFS: 19.1 months vs 10.2 months; OS: 41.2 months vs 23.1 months |
| Miyawaki et al[30], 2020 | Cohort (n = 180) | EGFR+ with BM | Median age 64 years, 59% female, 1-3 BM 48% | Sequencing analysis | Treatment sequence comparison | iPFS | 1-3 BM: Comparable; ≥ 4 BM: Benefit from upfront RT |
| Iuchi et al[5], 2013 | Cohort (n = 41) | Japanese with BM | Median age 65 years, 61% female, PS 0-2 | Gefitinib monotherapy | Historical controls | Response rate | Intracranial ORR: 87.8%, mOS: 18.8 months |
| Eichler et al[4], 2010 | Cohort (n = 82) | NSCLC with BM | Median age 58 years, 57% female | Survival comparison | EGFR wild-type | OS | EGFR+: 15 months vs 5 months wild-type (P = 0.003) |
| Yamamoto et al[33], 2017 | Cohort (n = 1194) | Multiple BM | Median age 64 years, 58% female, 2-10 BM | SRS outcomes | Single BM comparison | Toxicity, cognition | Low toxicity rates, preserved cognition |
| Tatineni et al[32], 2023 | Cohort (n = 267) | NSCLC with BM | Median age 66 years, 59% female, 2010-2019 era | EGFR-TKI + RT | Historical comparison | Survival outcomes | Improved outcomes with modern approaches |
| Yan et al[20], 2019 | Cohort (n = 89) | Leptomeningeal metastases | Median age 57 years, 62% female, poor PS | WBRT analysis | Non-WBRT comparison | OS | WBRT did not improve survival |
| Hsu et al[6], 2025 | Cohort (n = 127) | Baseline BM | Median age 64 years, 65% female, treatment-naïve | 1st vs 2nd gen EGFR-TKI | Generation comparison | PFS | 2nd gen: Modest improvement (11.2 months vs 8.7 months) |
| Zhong et al[21], 2025 | Cohort (n = 42) | Leptomeningeal metastases | Median age 58 years, 64% female, poor prognosis | Intrathecal pemetrexed | Historical controls | Response | Feasible but limited efficacy |
| Pan et al[34], 2025 | Cohort (n = 156) | EGFR+ with BM | Median age 61 years, 58% female, multiple BM | Brain RT analysis | Non-RT comparison | Efficacy | RT benefit in patients (> 3 brain metastases, ECOG 0-2, and moderate-severe symptoms) |
| Dohm et al[22], 2022 | Cohort (n = 389) | NSCLC BM | Median age 63 years, 56% female, mixed treatments | SRS + systemic therapy | Treatment comparison | OS | Variable outcomes by systemic therapy type |
It was found that, in general, the studies had solid methods. Across randomized controlled trials, most studies had limited bias in most areas, though issues with blinding occurred in open-label studies (as indicated in Table 2). According to observational studies, results were not very uniform; even so, the majority scored moderately to highly on the Newcastle-Ottawa Scale, but the selection bias was introduced using retrospective designs (Table 3).
| Ref. | Randomization process | Deviations from interventions | Missing outcome data | Measurement of outcomes | Selection of results | Overall risk |
| Soria et al[10], FLAURA | Low | Some concerns | Low | Low | Low | Some concerns |
| Ramalingam et al[9], FLAURA-OS | Low | Some concerns | Low | Low | Low | Some concerns |
| Reungwetwattana et al[24] | Low | Some concerns | Low | Low | Low | Some concerns |
| Cho et al[25], FLAURA Asian | Low | Some concerns | Low | Low | Low | Some concerns |
| Wu et al[11], AURA3-CNS | Low | Low | Low | Low | Low | Low |
| Brown et al[14], RTOG 0933 | Low | Low | Some concerns | Low | Low | Some concerns |
| Brown et al[13], NCCTG N107C | Low | Low | Low | Low | Low | Low |
| Brown et al[15], SRS study | Low | Low | Low | Low | Low | Low |
| Jänne et al[26], FLAURA2-CNS | Low | Some concerns | Low | Low | Low | Some concerns |
| Li et al[19], FLOWERS | Low | Some concerns | Some concerns | Low | Low | Some concerns |
| Ref. | Selection (0-4) | Comparability (0-2) | Outcome (0-3) | Total score | Quality rating |
| Shi et al[2], PIONEER | 4 | 2 | 3 | 9 | High |
| Gu et al[31] | 3 | 1 | 3 | 7 | High |
| Rangachari et al[3] | 4 | 2 | 2 | 8 | High |
| Magnuson et al[27], 2016 | 3 | 2 | 3 | 8 | High |
| Ballard et al[8] | 3 | 1 | 2 | 6 | Moderate |
| Magnuson et al[28], 2017 | 4 | 2 | 3 | 9 | High |
| Chen et al[29] | 3 | 2 | 3 | 8 | High |
| Miyawaki et al[30] | 3 | 2 | 3 | 8 | High |
| Iuchi et al[5] | 2 | 1 | 2 | 5 | Moderate |
| Eichler et al[4] | 3 | 2 | 2 | 7 | High |
| Yamamoto et al[33] | 4 | 1 | 3 | 8 | High |
| Tatineni et al[32] | 3 | 2 | 3 | 8 | High |
| Yan et al[20] | 2 | 1 | 2 | 5 | Moderate |
| Hsu et al[6] | 3 | 2 | 2 | 7 | High |
| Zhong et al[21] | 2 | 1 | 2 | 5 | Moderate |
| Pan et al[34] | 3 | 2 | 2 | 7 | High |
| Dohm et al[22] | 4 | 2 | 3 | 9 | High |
Third-generation EGFR-TKIs: The FLAURA study showed that osimertinib was better than other standard EGFR-TKIs in patients who had not yet been treated for advanced EGFR-mutated NSCLC[10]. The first analysis revealed that osimertinib was better than chemotherapy at keeping the disease from getting worse (HR: 0.46, P < 0.001) and resulted in slightly longer survival (HR: 0.80, P = 0.046)[9].
CNS-focused analyses demonstrated that osimertinib worked better in brain tumors than erlotinib[24]. Patients receiving osimertinib at the start of their treatment for CNS metastases registered an objective CNS response of 91%, whereas the reaction to comparator EGFR-TKIs was 68%. Treating patients with osimertinib led to a much longer progression-free survival in the CNS than with placebo (not reached vs 13.9 months, HR: 0.48, 95%CI: 0.26-0.86, P = 0.014).
The Asian analysis from FLAURA revealed that these results apply to the group with the highest rate of EGFR mutations[25]. Osimertinib was better at stopping disease growth, keeping the disease at bay for 16.5 months compared to 11.0 months for the other group, with HR: 0.46 (95%CI: 0.33-0.64).
Osimertinib was investigated in a group of T790M-positive NSCLC patients, and it worsened after previous treatment with EGFR-TKIs[11]. CNS efficacy analysis showed that osimertinib reached an intracranial response rate of 70% vs 31% with platinum-pemetrexed chemotherapy (this difference was significant: P = 0.015). Patients on osimertinib had better progression-free survival of the brain (11.7 months vs 5.6 months; HR: 0.32, 95%CI: 0.15-0.69, P = 0.004).
Osimertinib combined with chemotherapy was recently studied using results from the FLAURA2 trial[26]. A com
In the RTOG 0933 trial, researchers gave patients with brain metastases radiation to the brain and nerve medicine memantine instead of directly treating the hippocampus[14]. According to the results, cognitive function was similar to earlier studies, and dementia symptoms were better, with a decline in verbal learning test recall of just 7.0%, compared to 30% historically (P < 0.001). The analysis of EGFR-mutated NSCLC patients was not well represented in the subgroup.
The NCCTG N107C study tested postoperative stereotactic radiosurgery against whole-brain radiation after brain tumor resection[13]. SRS kept the mind sharp without lowering survival, although results for EGFR mutation were not separated in the main analysis.
When testing 1-3 brain metastase cases, SRS alone preserved mental functions just as well as SRS with WBRT[15]; the cognitive decline was much greater after SRS plus WBRT at 3 months, with 91.7% compared to just 63.5% after SRS alone (P < 0.001).
EGFR-TKI monotherapy: The PIONEER study in Asian patients with lung cancer[2] made a detailed molecular analysis of EGFR mutation possible. EGFR mutations were found in 60.9% of the adenocarcinomas studied among the 1482 patients. In 28.7% of EGFR-mutated patients, tumors spread to the brain more frequently in young adults and women.
Rangachari et al[3] compared how brain metastasis occurred in patients with EGFR-mutated and ALK-rearranged NSCLC. About 25.4% of EGFR-mutated patients had brain metastases, and these were likely to appear much later in the course of their disease than for ALK-positive patients. The median time patients lived after being diagnosed with brain metastases was 19.9 months for those with EGFR mutations.
The phase II experiment by Iuchi et al[5] looked at treatment with just gefitinib in Japanese NSCLC patients with brain metastases and no need for radiotherapy. Eighty-seven percent of patients responded in the brain. The median time before brain cancer progression was 14.5 months. All-cause survival was 18.8 months, meaning some brain activity was found in the first-generation EGFR-TKIs.
Eichler et al[4] compared survival in NSCLC patients with brain metastases based on whether they had EGFR mutations. Patients with tumors activating EGFR mutations lived longer than those with wild-type disease (15 months vs 5 months; P = 0.003).
Magnuson et al[27] studied the results of withholding radiotherapy from EGFR-mutated NSCLC patients diagnosed with brain metastases. Among patients who did not have EGFR-TKI upfront with radiotherapy, those who only received EGFR-TKI also had comparable survival (46 months) to those who received radiotherapy alone (46 months).
Magnuson et al[28] conducted a multi-institutional retrospective analysis to compare outcomes in EGFR-TKI mutated patients. Patients who received a combination of EGFR-TKIs and radiotherapy had less progression inside the brain than those on EGFR-TKIs alone (12-month rate of intracranial progression 21% vs 47%; P = 0.014).
Chen et al[29] reviewed data comparing brain radiotherapy plus anti-EGFR drugs with using anti-EGFR drugs alone for patients with asymptomatic brain metastases. Fewer new or growing cancers inside the brain were seen in combined therapy (19.1 months vs 10.2 months), and patients receiving that therapy survived longer (41.2 months vs 23.1 months).
Miyawaki et al[30] investigated the best ways to give local therapy and EGFR-TKIs depending on the number of brain metastases. Patients with only one to three brain metastases had similar results from starting with EGFR-TKI treatment as they did from combined approaches; when there are four or more brain metastases, radiotherapy and EGFR-TKIs used jointly provide the most benefit for patients.
Clinical practice surveys have recently observed the effects of third-generation EGFR-TKIs on patients. Gu et al[31] looked at how TP53 co-mutations influenced the outcomes of EGFR-mutated patients with brain metastases receiving EGFR-TKIs. An association was seen between TP53 co-mutation and poor intracranial progression-free survival (7.1 months vs 14.8 months; P = 0.041).
Tatineni et al[32] carried out a study in 2020 using data from the previous decade. The use of SRS and third-generation EGFR-TKIs rose in the series, which led to better patient survival over the years.
Hsu et al[6] examined how first-generation and second-generation EGFR-TKIs affected the treatment of untreated patients with brain metastases at the start of their illness. Second-generation EGFR-TKIs helped patients live significantly longer without intracranial relapse than first-generation drugs (11.2 months vs 8.7 months; P = 0.032), although all were inferior to the results seen with third-generation EGFR-TKIs.
Treatment approaches led to noticeably varied safety results. Most patients did not experience serious side effects from EGFR-TKI monotherapy, and common events involved diarrhea, a rash, and infections under the nails of the toes and hands. Doctors found that third-generation EGFR-TKIs caused less skin and gut toxicity than other EGFR-TKIs.
Although a third-generation EGFR-TKI agent, such as osimertinib, has been reported to be less dermatotoxic compared to second-generation TKIs, like afatinib, few direct comparisons have been conducted between afatinib and third-generation EGFR-TKI agents. Some studies have, however, indicated greater dermatologic toxicities associated with afatinib.
In most clinical series, both treatments showed an acceptable number of side effects together. The likelihood of lung inflammation with both drugs used simultaneously caused many centers to prefer to give the treatments one after another rather than together. People treated with stereotactic radiosurgery and EGRA-TKI regimens reported better toleration and improved their cognitive and quality of life.
Patients treating their cancers with several radiotherapy methods, especially those receiving a strong focal dose, were found to have radiation necrosis as a risk. Most current series use modern techniques and scheduling, keeping the rates below 5%.
Overcoming resistance to EGFR-TKIs in the brain is unique and presents several new challenges. When leptomeningeal disease occurs, the patient has few effective treatment options, and the outcome is usually poor. Yan et al[20] showed that using WBRT failed to increase survival in EGFR-mutated patients with leptomeningeal metastases, so it is important to find new therapies.
Some patients have benefited from using intrathecal chemotherapy in novel salvage approaches. According to the report by Zhong et al[21], treating NSCLC patients with leptomeningeal disease with pemetrexed given in the spine and standard therapy was feasible; however, it was not very successful.
Fourth-generation EGFR-TKIs and various new combinations are currently a lively area of research. The purpose of the FLOWERS trial is to study osimertinib with or without ruxolitinib in patients with MET changes to learn how resistance arises[19].
The data shows that osimertinib, among third-generation EGFR-TKIs, works much better to prevent brain metastases in EGFR-mutated NSCLC patients than earlier-generation EGFR-TKI drugs. The use of combined radiotherapy approaches brings further benefits to a group of patients based on disease severity, symptoms experienced, and personal health factors. For patients receiving stereotactic radiosurgery, it usually provides better preservation of brain function than whole-brain radiotherapy. Resisting treatment, especially for leptomeningeal disease, is still a problem, so scientists continue to seek new ways to treat it.
Table 4 presents an overview of the quality of evidence assessment, as applied using the GRADE methodology, and the level of certainty regarding significant treatment comparisons and outcomes.
| Outcome | Studies (design) | Participants | Effect estimate | Quality of evidence | Rationale for downgrading |
| Third-generation EGFR-TKIs vs first/second-generation | |||||
| Progression-free survival | 4 RCTs | 1201 | HR: 0.46 (0.38-0.56) | High | No serious limitations |
| CNS objective response rate | 3 RCTs | 644 | OR: 4.2 (2.8-6.3) | High | No serious limitations |
| Overall survival | 2 RCTs | 913 | HR: 0.80 (0.64-0.99) | Moderate | Wide confidence intervals |
| Combined EGFR-TKI + radiotherapy vs EGFR-TKI alone | |||||
| Intracranial control | 6 observational | 1156 | HR: 0.52 (0.38-0.71) | Low | Observational studies with inherent limitations and significant heterogeneity between studies (I² > 50%) |
| Overall survival | 4 observational | 892 | HR: 0.68 (0.51-0.91) | Low | Observational studies with inherent limitations and wide confidence intervals |
| Stereotactic radiosurgery vs whole brain radiotherapy | |||||
| Cognitive preservation | 3 RCTs | 925 | OR: 5.8 (3.2-10.5) | Moderate | Mixed population studies with limited EGFR-mutant specific data |
| Local control | 5 mixed studies | 1689 | OR: 1.8 (1.2-2.7) | Low | Observational studies with inherent limitations and significant heterogeneity between studies (I² > 50%) |
| Treatment sequencing | |||||
| Upfront EGFR-TKI vs Combined | 4 observational | 567 | Variable outcomes | Very low | Observational studies with inherent limitations, significant heterogeneity between studies (I² > 50%), and wide confidence intervals |
The treatment of brain metastases in EGFR-mutated NSCLC has completely transformed within the last 10 years. When histology was involved, doctors mainly gave radiotherapy and used drugs to help manage symptoms. Since targeted therapy arrived, EGFR-TKIs have become the preferred treatment and have been shown to impact cancer in the brain.
Osimertinib, in particular, has made a significant difference in therapy aimed at the brain[9,10,24]. As a result of better uptake in the brain, better accuracy at the site of disease, and less risk to patients, osimertinib has become the first therapy recommended for treating EGFR-mutated NSCLC, whether the cancer has spread to the brain or not. Because CNS performs reliably for various patients, including those from Asian populations where EGFR mutations are common[25], the drug can be used worldwide.
There are no direct, head-to-head, randomized studies comparing second-generation (afatinib, dacomitinib) EGFR-TKIs to third-generation EGFR-TKIs (osimertinib) in patients with brain metastases, which is a major limitation of the evidence base. Hsu et al’s study[6] (11.2 months vs 8.7 months for first-generation; P = 0.032) showed a modest im
This gap in evidence may be clinically significant, particularly in resource-limited settings where cost may favor second-generation agents, or for patients with resistance to Osimertinib, for whom second-generation TKIs may be considered. Future randomized trials specifically enrolling patients with incident or baseline brain metastases that compare contemporary second- and third-generation agents with CNS-directed endpoints could provide definitive evidence to inform treatment algorithms. Until this data is available, osimertinib’s overall efficacy trial superiority, CNS penetration, pharmacokinetic benefits, and better toxicity profile are driving treatment selection despite the absence of direct comparative evidence in the brain metastases population.
Using an EGFR-TKI alone is highly active in the brain, but adding radiotherapy can further increase the response in a small group of patients. The results indicate that combining several treatments provides better control over the tumor and a longer survival for patients with many brain tumors or disease symptoms[28-30]. Although observational studies indicate potential benefits of combining radiotherapy and TKIs, randomized controlled trials are necessary to confirm the definitive superiority of this approach over TKI monotherapy.
The selection of patients for combined tactics needs attention to how serious the disease is, what symptoms are present, how well patients can function, and what they want from treatment. For those with small, silent brain metastases, taking an EGFR-TKI at the start often makes sense, while radiotherapy is used after disease progression or when patients have symptoms[27,30]. Still, people who have several brain tumors or big lesions or are already showing symptoms may be best treated with combined therapy straight away.
Stereotactic radiotherapy is showing strong support over WBRT in some medical conditions[13,15]. Because patients can now survive longer with modern treatments, preserving mental function is crucial. Both approaches protect against cognitive decline following radiation for a brain tumor[14,15].
It is still being studied how to best organize EGFR-TKI and radiotherapy treatments[16,32]. There is concern that pairing drugs can cause side effects, especially pneumonitis, pushing many to administer each drug one after the other. Even so, researchers still need to examine how far apart treatments should be and which treatments last longer.
The relation between when local therapy is given and when the disease spreads matters. Some patients could first be treated with CNS-active EGFR-TKIs, while radiotherapy is introduced if the treatment stops working. This approach could be especially effective now that new EGFR-TKIs can better cross the blood-brain barrier.
Differences in molecules in EGFR-mutated NSCLC can change both treatment results and tumors' ability to resist drugs. Multiple alterations, such as changes in TP53, may alter the response and pattern of growth seen in the CNS[31]. Knowledge about these molecular interactions will support better decisions about which treatment should be given and when.
The blood-brain barrier and specific conditions near tumors in the brain can change how well brain cancer treatments operate. Looking at how CNS-specific resistance mutations and clonal evolution patterns develop is an important question for future research.
Using different types of medications together may help improve patient treatment. Investigators are studying how immunotherapy, targeted agents, and radiotherapy can be used together, though deciding on the right order and who should receive the treatment is still difficult[16,22].
The significant variation in outcomes, with heterogeneity higher than 50% and I2 across studies, implies that several factors contributed to the shift in treatment outcomes. Ontological differences appeared to be one of the primary sources with Asian populations contributing between 62-100 percent of study participants and leading to the occurrence of different frequencies of EGFR mutation (up to 60.9 percent compared to 10-15 percent in Western cohorts) and the change in baseline patient characteristics such as age (57-66 years) and gender (55-76 percent of women)[2]. The potential factor that had a significant influence on the outcome variability was radiotherapy protocol heterogeneity, as stereotactic radiosurgery protocols differed in terms of fractionation schemes (single-fraction vs hypo-fractionated), dose prescriptions (18-24 Gy in single lesions), and administration schedules (concomitant, sequential, or salvage).
Fourth-generation EGFR-TKIs, created to overcome known resistances, could rescue patients from disease progression. In addition, two different strategies administering medicine directly into the spinal fluid and disrupting the brain's protective barrier may lead to better drugs reaching the CNS[21].
Since today's therapy can extend lifespan, it is more important to consider how treatment affects a person's mental abilities and quality of life. Choosing a certain radiotherapy technique strongly impacts cognition, and group-classified SRS and hippocampal avoidance are best for preserving mental functions[13-15].
Treatment decisions should include results from patient surveys and tests of daily activity, particularly for those patients with no brain metastasis symptoms who can still enjoy good overall health.
The synergy observed between EGFR-TKIs and radiotherapy can be attributed to radio-sensitization resulting from the inhibition of major molecular pathways. The alteration of the process of tumor cell preservation and DNA repair occurs through EGFR-TKIs, especially with osimertinib, which disrupts essential signaling pathways, including the PI3K/Akt and MAPK pathways. Through the blocking of the same pathways, EGFR-TKIs increase the effectiveness of radiotherapy by suppressing the repair of radiation-induced DNA damage, which subsequently leads to increased tumor cell death. Preclinical investigations have demonstrated that the addition of EGFR-TKIs to radiotherapy leads to improved control of the tumor, especially in the brain metastases, where the blood-brain barrier restricts drug diffusion. These molecular mechanisms need further investigation to streamline treatment regimens.
Several limitations to the present systematic review should be carefully considered when interpreting the findings and their clinical implications. The quantitative assessment revealed significant statistical heterogeneity (I² > 75%) and did not support meta-analysis. There was considerable clinical heterogeneity across study design types (randomized clinical trials vs observational studies), intervention types (three generations of EGFR-TKIs with different brain penetrance profiles), radiotherapy treatment protocols (single-fraction vs multi-fraction SRS with different dose prescriptions), and patient groups (treatment-naive vs resistant disease; brain metastases 1-3 vs brain metastases ≥ 4). Although narrative synthesis is useful to incorporate wide-ranging evidence, it does not produce pooled effect estimates or allow formal testing for publication bias using a funnel plot analysis. It does not allow meta-regression to explore the source of heterogeneity or conduct a sensitivity analysis excluding high-risk-of-bias studies. As a result of this analytical constraint, limited treatment effect estimates in precision and definite ordering in effectiveness. In the 18 observational studies (64 percent of the included articles), significant selection bias is inrounded by the use of retrospective designs, possibly preferentially selecting the patients with better performance status, whereas the heterogeneity of the treated population spans geographic spectral (Asian-predominant vs. Western population with varying EGFR mutations frequency distribution), mutation subtypes (exon 19 deletions to L858R, T790M which may respond differently to treatment), disease burden heterogeneity single brain metastases to multiple brain metastases.
Most importantly, criteria variations over time will be substantial, as revolutionary changes in NSCLC management are expected to occur between 2010 and 2025 (a 15-year study period), which will present a significant source of bias in applicability to current practice. The first-generation EGFR-TKIs were primarily used in early research and were not deeply rooted in the CNS, unlike whole-brain radiotherapy, which could lead to cognitive toxicity. In modern practice, third-generation EGFR-TKIs (such as osimertinib), with high brain bioavailability, are used in conjunction with stereotactic radiosurgery procedures. It implies that there is a real risk that pooling of trials can significantly underestimate the actual effect size of existing standard-of-care interventions, because initial modest findings with original generation methods dilute the dramatic CNS activity shown with the current modern interventions. At the same time, advances in the accuracy of molecular diagnostic testing, imaging sensitivity in the detection of brain metastases, and the use of supportive care regimens have necessitated changes in the eligibility criteria for patients and the tolerability of treatments, making historical comparisons problematic. Their clinical applicability to modern-day practice may be limited by the size of the treatment impacts as measured in studies during this time of transformational change.
Based on GRADE assessment, moderate-to-low quality of evidence in combined modality approaches highlights the considerable difficulty in conducting randomized trials in this context. This is based mainly on retrospective studies of observational design, which have the potential for selection bias and confounding. The heterogeneity of patient populations, treatment protocols, and the definition of outcomes further limits the applicability of the pooled estimates.
Besides third-generation EGFR-TKIs, such as osimertinib, newer molecules, including BLU-945, a fourth-generation EGFR-TKI, and patritumab deruxtecan, an antibody-drug conjugate, are emerging as potential agents that may address the resistance problem associated with second-generation EGFR-TKIs. BLU-945 is active against an EGFR C797S mutation, which is a typical resistance mechanism to osimertinib. Patritumab deruxtecan targets EGFR and carries a cytotoxic payload to cancer cells, which would positively impact the results in cases of EGFR-mutated NSCLC, including those with brain metastases. Implementation of these novel therapies into the treatment algorithm for the EGFR-mutated NSCLC population may provide a more personalized approach to patients, particularly those at advanced or resistant stages.
Although third-generation EGFR-TKIs, such as osimertinib, are superior to second-generation EGFR-TKIs in treating EGFR-mutated brain metastases, radiotherapy may also be necessary for patients with more extensive CNS invasion or symptomatic brain metastases.
In the future, more research is required to compare modern EGFR-TKIs monotherapy to combined strategies, identify the best ways to use radiotherapy, decide on how to better categorize patients using biomarkers, and monitor and evaluate patient quality of life. Standard measures and responses for neurological disorders would make future comparisons in this field much easier.
Assessing cognitive function over a long period is vital, as survival for many chronic diseases is improving. Trying new medications and ways to get them into the brain may also improve CNS-directed treatments.
Osimertinib and similar third-generation EGFR-TKIs now play a major role in handling brain metastases in NSCLC that contain EGFR mutations. Our systematic review, supported by GRADE evidence evaluation, has demonstrated that these agents are highly active in the brain, and their benefits over previous-generation agents are backed by high-quality evidence to support their effectiveness, setting new standards of treatment for EGFR-mutated NSCLC regardless of whether the disease spreads to the brain.
Although stereotactic radiotherapy methods for cognitive protection have moderate-quality evidence, they tend to be superior to whole-brain therapy when applicable, yielding better maintenance of cognitive ability with no reduction in tumor control. In patients with more than three brain metastases, the combination of SRS and EGFR-TKIs should be preferred to preserve cognitive functions. WBRT should be used only in cases of significant disease or when SRS is not feasible. However, the low-quality evidence of some combined modality methods requires a specific decision-making process. Adding radiotherapy to other treatments benefits patients with less than three brain metastases, ECOG 0-2, and moderate to severe neurological symptoms, though patients with one to three brain metastases or those who are asymptomatic are likely to respond best to EGFR-TKI treatment alone as first-line therapy. The order and timing at which combined methods should be used are yet to be fully understood.
Multimodal therapies, including the use of third-generation EGFR-TKIs and radiotherapy, are expensive and in short supply in many regions of resource deficiency; therefore, a more sequential treatment strategy may need to be resorted to. Clinicians may consider implementing EGFR-TKI monotherapy and stereotactic radiosurgery as cost-effective alternatives to combinations of whole-brain radiotherapy treatment regimens in these settings. There have been no studies or trials comparing second-generation EGFR-TKIs with third-generation TKIs in patients with brain metastases. This presents a major evidence gap. However, considering the pharmacokinetic benefits of osimertinib, its CNS activity is superior to others in subgroup analyses, and data demonstrating overall benefit, we preferentially recommend this drug as the first-line agent in patients with EGFR-mutated NSCLC, with or without CNS involvement.
Individual patient-related factors, including the number of lesions, performance status (ECOG 0-2), neurological symptoms, and the type of disease, should guide doctors in selecting the most effective treatment to control the tumor while minimizing the impact on the patient's cognitive function and quality of life. When making a decision, clinicians need to consider a combination of therapies in light of the patient's cognitive function status, their tumor burden, and their priorities regarding whether it is more important to live longer or achieve a better quality of life. The difference in the quality of evidence among various interventions underscores the importance of collaborative decision-making among patients and multidisciplinary teams, taking into account both the quality of evidence as a criterion and individual patient factors. Close monitoring and follow-up are necessary to assess the response to medical intervention and potential treatment-related neurocognitive deficits.
Future studies are encouraged to focus on well-designed randomized trials to determine the optimal sequence of steps in terms of treatment and selection criteria, the application of biomarkers to associate drugs with patients, the combination of different drugs, and all patient responses. The latest research suggests that an integrated plan from oncologists, radiation specialists, and neurosurgeons can give patients with NSCLC EGFR mutations the best possible results. However, more work and study will be needed to improve how this challenging problem is treated.
| 1. | Midha A, Dearden S, McCormack R. EGFR mutation incidence in non-small-cell lung cancer of adenocarcinoma histology: a systematic review and global map by ethnicity (mutMapII). Am J Cancer Res. 2015;5:2892-2911. [PubMed] |
| 2. | Shi Y, Au JS, Thongprasert S, Srinivasan S, Tsai CM, Khoa MT, Heeroma K, Itoh Y, Cornelio G, Yang PC. A prospective, molecular epidemiology study of EGFR mutations in Asian patients with advanced non-small-cell lung cancer of adenocarcinoma histology (PIONEER). J Thorac Oncol. 2014;9:154-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1205] [Cited by in RCA: 1224] [Article Influence: 102.0] [Reference Citation Analysis (0)] |
| 3. | Rangachari D, Yamaguchi N, VanderLaan PA, Folch E, Mahadevan A, Floyd SR, Uhlmann EJ, Wong ET, Dahlberg SE, Huberman MS, Costa DB. Brain metastases in patients with EGFR-mutated or ALK-rearranged non-small-cell lung cancers. Lung Cancer. 2015;88:108-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 333] [Cited by in RCA: 436] [Article Influence: 39.6] [Reference Citation Analysis (0)] |
| 4. | Eichler AF, Kahle KT, Wang DL, Joshi VA, Willers H, Engelman JA, Lynch TJ, Sequist LV. EGFR mutation status and survival after diagnosis of brain metastasis in nonsmall cell lung cancer. Neuro Oncol. 2010;12:1193-1199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 217] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 5. | Iuchi T, Shingyoji M, Sakaida T, Hatano K, Nagano O, Itakura M, Kageyama H, Yokoi S, Hasegawa Y, Kawasaki K, Iizasa T. Phase II trial of gefitinib alone without radiation therapy for Japanese patients with brain metastases from EGFR-mutant lung adenocarcinoma. Lung Cancer. 2013;82:282-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 218] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 6. | Hsu CC, Chiu LC, Ko HW, Wu CE, Kuo SC, Ju JS, Huang AC, Wang CC, Yang CT, Hsu PC. Clinical outcome analysis of different first and secondgeneration EGFRtyrosine kinase inhibitors in untreated patients with EGFRmutated nonsmall cell lung cancer with baseline brain metastasis. Oncol Lett. 2025;29:201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 7. | Poels KE, Schoenfeld AJ, Makhnin A, Tobi Y, Wang Y, Frisco-Cabanos H, Chakrabarti S, Shi M, Napoli C, McDonald TO, Tan W, Hata A, Weinrich SL, Yu HA, Michor F. Identification of optimal dosing schedules of dacomitinib and osimertinib for a phase I/II trial in advanced EGFR-mutant non-small cell lung cancer. Nat Commun. 2021;12:3697. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 8. | Ballard P, Yates JW, Yang Z, Kim DW, Yang JC, Cantarini M, Pickup K, Jordan A, Hickey M, Grist M, Box M, Johnström P, Varnäs K, Malmquist J, Thress KS, Jänne PA, Cross D. Preclinical Comparison of Osimertinib with Other EGFR-TKIs in EGFR-Mutant NSCLC Brain Metastases Models, and Early Evidence of Clinical Brain Metastases Activity. Clin Cancer Res. 2016;22:5130-5140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 595] [Cited by in RCA: 587] [Article Influence: 58.7] [Reference Citation Analysis (0)] |
| 9. | Ramalingam SS, Vansteenkiste J, Planchard D, Cho BC, Gray JE, Ohe Y, Zhou C, Reungwetwattana T, Cheng Y, Chewaskulyong B, Shah R, Cobo M, Lee KH, Cheema P, Tiseo M, John T, Lin MC, Imamura F, Kurata T, Todd A, Hodge R, Saggese M, Rukazenkov Y, Soria JC; FLAURA Investigators. Overall Survival with Osimertinib in Untreated, EGFR-Mutated Advanced NSCLC. N Engl J Med. 2020;382:41-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2467] [Cited by in RCA: 2221] [Article Influence: 370.2] [Reference Citation Analysis (3)] |
| 10. | Soria JC, Ohe Y, Vansteenkiste J, Reungwetwattana T, Chewaskulyong B, Lee KH, Dechaphunkul A, Imamura F, Nogami N, Kurata T, Okamoto I, Zhou C, Cho BC, Cheng Y, Cho EK, Voon PJ, Planchard D, Su WC, Gray JE, Lee SM, Hodge R, Marotti M, Rukazenkov Y, Ramalingam SS; FLAURA Investigators. Osimertinib in Untreated EGFR-Mutated Advanced Non-Small-Cell Lung Cancer. N Engl J Med. 2018;378:113-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4505] [Cited by in RCA: 4104] [Article Influence: 513.0] [Reference Citation Analysis (3)] |
| 11. | Wu YL, Ahn MJ, Garassino MC, Han JY, Katakami N, Kim HR, Hodge R, Kaur P, Brown AP, Ghiorghiu D, Papadimitrakopoulou VA, Mok TSK. CNS Efficacy of Osimertinib in Patients With T790M-Positive Advanced Non-Small-Cell Lung Cancer: Data From a Randomized Phase III Trial (AURA3). J Clin Oncol. 2018;36:2702-2709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 404] [Article Influence: 50.5] [Reference Citation Analysis (1)] |
| 12. | Hendriks LE, Schoenmaekers J, Zindler JD, Eekers DB, Hoeben A, De Ruysscher DK, Dingemans AM. Safety of cranial radiotherapy concurrent with tyrosine kinase inhibitors in non-small cell lung cancer patients: A systematic review. Cancer Treat Rev. 2015;41:634-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 13. | Brown PD, Ballman KV, Cerhan JH, Anderson SK, Carrero XW, Whitton AC, Greenspoon J, Parney IF, Laack NNI, Ashman JB, Bahary JP, Hadjipanayis CG, Urbanic JJ, Barker FG 2nd, Farace E, Khuntia D, Giannini C, Buckner JC, Galanis E, Roberge D. Postoperative stereotactic radiosurgery compared with whole brain radiotherapy for resected metastatic brain disease (NCCTG N107C/CEC·3): a multicentre, randomised, controlled, phase 3 trial. Lancet Oncol. 2017;18:1049-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 571] [Cited by in RCA: 911] [Article Influence: 101.2] [Reference Citation Analysis (0)] |
| 14. | Brown PD, Gondi V, Pugh S, Tome WA, Wefel JS, Armstrong TS, Bovi JA, Robinson C, Konski A, Khuntia D, Grosshans D, Benzinger TLS, Bruner D, Gilbert MR, Roberge D, Kundapur V, Devisetty K, Shah S, Usuki K, Anderson BM, Stea B, Yoon H, Li J, Laack NN, Kruser TJ, Chmura SJ, Shi W, Deshmukh S, Mehta MP, Kachnic LA; for NRG Oncology. Hippocampal Avoidance During Whole-Brain Radiotherapy Plus Memantine for Patients With Brain Metastases: Phase III Trial NRG Oncology CC001. J Clin Oncol. 2020;38:1019-1029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 704] [Cited by in RCA: 638] [Article Influence: 106.3] [Reference Citation Analysis (0)] |
| 15. | Brown PD, Jaeckle K, Ballman KV, Farace E, Cerhan JH, Anderson SK, Carrero XW, Barker FG 2nd, Deming R, Burri SH, Ménard C, Chung C, Stieber VW, Pollock BE, Galanis E, Buckner JC, Asher AL. Effect of Radiosurgery Alone vs Radiosurgery With Whole Brain Radiation Therapy on Cognitive Function in Patients With 1 to 3 Brain Metastases: A Randomized Clinical Trial. JAMA. 2016;316:401-409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1434] [Cited by in RCA: 1306] [Article Influence: 130.6] [Reference Citation Analysis (0)] |
| 16. | Cao Z, Deng K, Jiang J, Tian K, Wang B. Combined treatment of small cell lung cancer using radiotherapy and immunotherapy: Challenges and updates. Biomed Pharmacother. 2025;182:117727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 17. | Bowen Jones S, Chan C, Filippi AR, Harada K, Louie AV, Lindsay CR, Nadal E, Munoz Schuffenegger P, Woolf D, Faivre-Finn C; IASLC Advanced Radiation Technologies (ART) Subcommittee. Emerging Role of Targeted Therapies Combined With Radiotherapy in Inoperable Stages I to III NSCLC: A Review From the IASLC ART Subcommittee. J Thorac Oncol. 2025;20:1018-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 18. | Zhu Z, Ni J, Cai X, Su S, Zhuang H, Yang Z, Chen M, Ma S, Xie C, Xu Y, Li J, Ge H, Liu A, Zhao L, Rao C, Xie C, Bi N, Hui Z, Zhu G, Yuan Z, Wang J, Zhao L, Zhou W, Rim CH, Navarro-Martin A, Vanneste BGL, Ruysscher D, Choi JI, Jassem J, Chang JY, Kepka L, Käsmann L, Milano MT, Van Houtte P, Suwinski R, Traverso A, Doi H, Suh YG, Noël G, Tomita N, Kowalchuk RO, Sio TT, Li B, Lu B, Fu X. International consensus on radiotherapy in metastatic non-small cell lung cancer. Transl Lung Cancer Res. 2022;11:1763-1795. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 19. | Li A, Chen HJ, Yang JJ. Design and Rationale for a Phase II, Randomized, Open-Label, Two-Cohort Multicenter Interventional Study of Osimertinib with or Without Savolitinib in De Novo MET Aberrant, EGFR-Mutant Patients with Advanced Non-Small-Cell Lung Cancer: The FLOWERS Trial. Clin Lung Cancer. 2023;24:82-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 20. | Yan W, Liu Y, Li J, Han A, Kong L, Yu J, Zhu H. Whole brain radiation therapy does not improve the overall survival of EGFR-mutant NSCLC patients with leptomeningeal metastasis. Radiat Oncol. 2019;14:168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 21. | Zhong W, Wu L, Qiu Z, Yu W, Liu L, Shi H, Wu S. Intrathecal pemetrexed chemotherapy combined with systemic therapy in patients with non-small cell lung cancer and leptomeningeal metastases: a retrospective study. Front Oncol. 2025;15:1545174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 22. | Dohm AE, Tang JD, Mills MN, Liveringhouse CL, Sandoval ML, Perez BA, Robinson TJ, Creelan BC, Gray JE, Etame AB, Vogelbaum MA, Forsyth P, Yu HM, Oliver DE, Ahmed KA. Clinical outcomes of non-small cell lung cancer brain metastases treated with stereotactic radiosurgery and immune checkpoint inhibitors, EGFR tyrosine kinase inhibitors, chemotherapy and immune checkpoint inhibitors, or chemotherapy alone. J Neurosurg. 2023;138:1600-1607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 23. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 24. | Reungwetwattana T, Nakagawa K, Cho BC, Cobo M, Cho EK, Bertolini A, Bohnet S, Zhou C, Lee KH, Nogami N, Okamoto I, Leighl N, Hodge R, McKeown A, Brown AP, Rukazenkov Y, Ramalingam SS, Vansteenkiste J. CNS Response to Osimertinib Versus Standard Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitors in Patients With Untreated EGFR-Mutated Advanced Non-Small-Cell Lung Cancer. J Clin Oncol. 2018;JCO2018783118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 630] [Article Influence: 78.8] [Reference Citation Analysis (0)] |
| 25. | Cho BC, Chewaskulyong B, Lee KH, Dechaphunkul A, Sriuranpong V, Imamura F, Nogami N, Kurata T, Okamoto I, Zhou C, Cheng Y, Cho EK, Voon PJ, Lee JS, Mann H, Saggese M, Reungwetwattana T, Ramalingam SS, Ohe Y. Osimertinib versus Standard of Care EGFR TKI as First-Line Treatment in Patients with EGFRm Advanced NSCLC: FLAURA Asian Subset. J Thorac Oncol. 2019;14:99-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 103] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 26. | Jänne PA, Planchard D, Kobayashi K, Cheng Y, Lee CK, Valdiviezo N, Laktionov K, Yang TY, Yu Y, Kato T, Jiang L, Chewaskulyong B, Lucien Geater S, Maurel JM, Rojas C, Takahashi T, Havel L, Shepherd FA, Tanaka K, Ghiorghiu D, Amin NP, Armenteros-Monterroso E, Huang X, Chaudhry AA, Yang JC. CNS Efficacy of Osimertinib With or Without Chemotherapy in Epidermal Growth Factor Receptor-Mutated Advanced Non-Small-Cell Lung Cancer. J Clin Oncol. 2024;42:808-820. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 112] [Article Influence: 56.0] [Reference Citation Analysis (0)] |
| 27. | Magnuson WJ, Yeung JT, Guillod PD, Gettinger SN, Yu JB, Chiang VL. Impact of Deferring Radiation Therapy in Patients With Epidermal Growth Factor Receptor-Mutant Non-Small Cell Lung Cancer Who Develop Brain Metastases. Int J Radiat Oncol Biol Phys. 2016;95:673-679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 78] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 28. | Magnuson WJ, Lester-Coll NH, Wu AJ, Yang TJ, Lockney NA, Gerber NK, Beal K, Amini A, Patil T, Kavanagh BD, Camidge DR, Braunstein SE, Boreta LC, Balasubramanian SK, Ahluwalia MS, Rana NG, Attia A, Gettinger SN, Contessa JN, Yu JB, Chiang VL. Management of Brain Metastases in Tyrosine Kinase Inhibitor-Naïve Epidermal Growth Factor Receptor-Mutant Non-Small-Cell Lung Cancer: A Retrospective Multi-Institutional Analysis. J Clin Oncol. 2017;35:1070-1077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 357] [Article Influence: 39.7] [Reference Citation Analysis (0)] |
| 29. | Chen Y, Wei J, Cai J, Liu A. Combination therapy of brain radiotherapy and EGFR-TKIs is more effective than TKIs alone for EGFR-mutant lung adenocarcinoma patients with asymptomatic brain metastasis. BMC Cancer. 2019;19:793. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 30. | Miyawaki E, Kenmotsu H, Mori K, Harada H, Mitsuya K, Mamesaya N, Kawamura T, Kobayashi H, Nakashima K, Omori S, Wakuda K, Ono A, Naito T, Murakami H, Endo M, Nakasu Y, Gon Y, Takahashi T. Optimal Sequence of Local and EGFR-TKI Therapy for EGFR-Mutant Non-Small Cell Lung Cancer With Brain Metastases Stratified by Number of Brain Metastases. Int J Radiat Oncol Biol Phys. 2019;104:604-613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 31. | Gu W, Liu P, Tang J, Lai J, Wang S, Zhang J, Xu J, Deng J, Yu F, Shi C, Qiu F. The prognosis of TP53 and EGFR co-mutation in patients with advanced lung adenocarcinoma and intracranial metastasis treated with EGFR-TKIs. Front Oncol. 2023;13:1288468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 32. | Tatineni V, O'Shea PJ, Saxena S, Khosla AA, Ozair A, Kotecha RR, Jia X, Rauf Y, Murphy ES, Chao ST, Suh JH, Peereboom DM, Ahluwalia MS. Combination of EGFR-Directed Tyrosine Kinase Inhibitors (EGFR-TKI) with Radiotherapy in Brain Metastases from Non-Small Cell Lung Cancer: A 2010-2019 Retrospective Cohort Study. Cancers (Basel). 2023;15:3015. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 33. | Yamamoto M, Serizawa T, Higuchi Y, Sato Y, Kawagishi J, Yamanaka K, Shuto T, Akabane A, Jokura H, Yomo S, Nagano O, Aoyama H. A Multi-institutional Prospective Observational Study of Stereotactic Radiosurgery for Patients With Multiple Brain Metastases (JLGK0901 Study Update): Irradiation-related Complications and Long-term Maintenance of Mini-Mental State Examination Scores. Int J Radiat Oncol Biol Phys. 2017;99:31-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 175] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 34. | Pan K, Wang B, Xu X, Tang Y, Liang J, Ma S, Xia B, Zhu L. Efficacy analysis of brain radiotherapy in EGFR mutation non-small cell lung cancer with brain metastasis: a retrospective study. Discov Oncol. 2025;16:488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |