Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.110380
Revised: July 7, 2025
Accepted: October 21, 2025
Published online: June 20, 2026
Processing time: 322 Days and 13.3 Hours
Cancer increases the risk of developing dangerous and sometimes deadly blood clots, which are known as cancer-associated thrombosis (CAT). While direct oral anticoagulants (DOACs) can be considered for CAT, evidence is limited regarding their use in comparison to low molecular weight heparin (LMWH).
To compare the efficacy, bleeding risks, reoccurrence rate, and patient-reported outcomes of DOACs compared to LMWH in CAT of different cancer types.
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2009 checklist was followed during the search and screening of publications. Articles were searched systematically through biomedical databases such as PubMed, Scopus, and Cochrane covering studies published from January 1, 2003 to Decem
The 21371 cancer patients were included in 29 studies (15 randomized clinical trials and 14 observational studies). Rates of VTE recurrence between DOAC (5.6%-11%) and LMWH (7.9%-11%) in major trials were similar. There was a lot of difference to bleeding risks based on subtypes of cancers. In upper gastro
Doctors use both LMWH and DOACs for CAT, the decision of which patient to use DOACs depends on bleeding risk, with a specific focus on the risk by cancer subtypes. CAT management needs to be tailored to the specific cancer sub-type (especially upper vs lower gastrointestinal cancers), the risk of bleeding, and the patient's preferences or abilities.
Core Tip: Patients with cancer, direct oral anticoagulants (DOACs) are effective, as are low-molecular-weight heparin, in preventing new blood clots. DOACs raise the risk of bleeding, mainly in the digestive tract and urinary organs. Patients choose DOACs since they improve their quality of life and are convenient. Doctors have to consider the cancer type, possibility of bleeding and platelet levels before deciding on the treatment. Taking into account what the patient wants and what the medical team considers best is necessary. Anticoagulation treatment can be improved in multidisciplinary thrombosis programs, emphasizing the importance of providing ongoing information and support to patients.
- Citation: Asad D, Abid A, Hassan MB, Nenwani MK, Dev S, Kumar S, Sapna F, Bai P, Siddique S, Jawed I, Alam F, Razzaq FA, C Mekowulu F, Jabeen S, Mirza AM. Direct oral anticoagulants vs LMWH for cancer-associated thrombosis - updated evidence on bleeding risk, recurrence and patient-reported outcomes: A systematic review. World J Methodol 2026; 16(2): 110380
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/110380.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.110380
Cancer-associated thrombosis is a difficult condition for clinicians to handle in cancer patients as it affects about 20% of cases and comes in second after cancer progression as a cause of death[1]. Chemotherapy treatment, tumor-related changes to blood clotting and additional patient factors join to form a hypercoagulable state, increasing the risk of venous thromboembolism. Long ago, low-molecular-weight heparin was considered the tip of the anti-clotting strategy, as trials showed that it outperformed vitamin K antagonists in people with cancer[2].
Using direct oral anticoagulants has revolutionized anticoagulation treatment for various clinical needs, allowing for oral administration, maintaining stable blood levels, and requiring less monitoring compared to traditional methods[3]. However, due to cancer patients' special challenges (in drug metabolism, potential drug reactions, higher risk of bleeding and cancer treatment changes), careful study of DOACs was required to ensure they are suitable and effective for them.
There has been significant growth in recent clinical evidence on the use of DOACs in cancer-related blood clots, thanks to randomized controlled trials and observational studies providing information on how they compare to LMWH in terms of both effectiveness and safety[4-10]. The studies performed have found that the advantages and risks of treating blood clots with medication are not straightforward and depend on the type of cancer, the stage of treatment, and who is receiving the medication. Since the populations with cancer are not the same and their bleeding risks differ, as well as what patients say about their outcomes, thorough evidence reviews are needed to help doctors decide how to treat patients.
It is now recognized that the quality of life for patients and their perceptions of their treatment is crucial in cancer care, as they are considered equally important as the effectiveness of the therapy[11,12]. Dealing with daily shots, check-ups, and reduced freedom in lifestyle has led to studies on the experiences of patients using newer oral anticoagulants. Nowadays, these matters are especially significant for people with cancer because their treatment and illness can cause major problems with quality of life.
Sufficient evidence about the use of DOACs in cancer-related blood clots is emerging, indicating a need to review this evidence systematically and provide clinicians with useful guidance. There are ongoing efforts to use DOACs for certain cancer patients, especially thrombocytopenia or gastrointestinal (GI) cancer patients. However, many issues remain unclear, including who to treat, dosage, and other points.
ESMO, and ISTH, as well as highlighting preoccupying rates of bleeding in a subgroup of cancers, primarily GI and genitourinary malignancies. There are still critical areas of knowledge missing since the vast majority of trials excluded patients with a history of thrombocytopenia/active bleeding/elevated bleeding risk (which are populations regularly seen during clinical practice) and there is a lack of granular risk stratification by cancer subtype, which precludes the ability to make a specific clinical decision.
With remarkable increases in clinical evidence since 2018 and demonstrated practice variation among oncologists, hematologists, and primary care providers, it has become clear that the time has come to conduct an updated systematic synthesis. The systematic reviews published between 2019 and 2021 had smaller evidence bases. They were not sufficiently targeted at cancer subtype-specific outcomes, whereas randomized trials and real-world observational studies published between 2020 and 2024 have not been thoroughly incorporated into systematic reviews. This systematic review identifies knowledge gaps by synthesizing 29 studies involving 21371 cancer patients, with a particular focus on cancer subtype-specific bleeding risk stratification and pragmatic clinical decision-making frameworks. The aim is to provide clinicians with practical tools to optimize the use of DOACs in cancer-related thrombosis.
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2009 statement was followed in the preparation of this systematic review. This study did not have a review protocol that was in place before its initiation. A set of rules for reviewing the evidence was established beforehand to minimize mistakes and ensure thorough coverage of the evidence. A set of objectives, conditions for selecting studies, instructions for searching, methods for gathering data, and strategies for analysis were established before the review to maintain consistency in methodology throughout the study period.
Population (P): Adult cancer patients (18 years and older) who were diagnosed with venous thromboembolism (caner-associated blood clots), including those still undergoing cancer treatment, patients in remission, or those with stable disease post-treatment.
Intervention (I): Oral anticoagulants called DOACs (apixaban, rivaroxaban, edoxaban, and dabigatran) are given to patients for treating cancer-related venous thromboembolism. It is necessary to mention that dabigatran was included in the original Population Intervention Comparator Outcomes structure. However, it was not included in the final analysis because the necessary evidence was lacking in the chosen research.
Comparator (C): Low molecular weight heparin or LMWH (dalteparin, enoxaparin, and tinzaparin) are among the standard treatments for cancer-associated thrombosis.
Outcomes (O): Primary outcomes considered were major bleeding (as defined by the International Society on Thrombosis and Haemostasis and including the criteria of life-threatening bleeds, clinically relevant non- major bleeding, and minor bleeding), clinically significant but less severe bleeding, blood clots in veins [venous thromboembolism (VTE) re
Major bleeding: The life-threatening bleed or those that require intervention to avoid obvious harm.
Minor bleeding: A bleeding condition that is clinically insignificant and does not need intervention.
Clinically relevant non-major bleeding: Non-severe bleeding, which may or may not need clinical intervention, including a change in anticoagulant therapy.
Electronic databases, including PubMed, Scopus, and the Cochrane Library were employed to identify all studies that compared the differences between DOACs and LMWH in cancer patients with venous thromboembolism. A literature search was conducted from January 1, 2003, to December 31, 2024, and only studies published before 2012 that compared LMWH with VKAs were included. Any study carried out during this time that did not involve DOACs could not be included in this review, as it would not fall under the category to be analyzed. The plan was to search for both ran
Inclusion criteria: Studies based on randomization (randomized controlled trials) and studies following a group over time (cohort studies, case-control studies, and cross-sectional studies).
Patients who have cancer and develop cancer-related venous thromboembolism.
Studies conducted to compare the use of DOACs and LMWH for the treatment of cancer-associated VTE. Studies that report at least one of the principal outcomes: Bleeding, recurrence of VTE, or experiences reported by the patients. Studies published in the English language. Study that lasted for at least 30 days.
Exclusion criteria: Case reports, case series, editorials, letters and conference abstracts.
Research that is concerned only with preventing blood clots (VTE prevention or prophylaxis) rather than treatment.
Studies focused only on children. Trials that did not specifically look at the differences between DOACs and LMWHs.
Studies fail to record important information. More than one journal publication from the same group of patients.
Systematic data extraction was carried out independently by two reviewers by using a form created just for this review. Data management and organization of extracted data, as well as calculation of descriptive statistics for the study characteristics, were carried out using IBM SPSS Statistics Version 27.0. Among the data recorded for evaluation were descriptions of the studies (design, setting, and duration), patient information (such as age, sex, cancer type, and cancer stage), the method and length of treatment used, comparator data, descriptions of outcomes, the time of follow-up, and the reported outcomes. Discrepancies among reviewers were resolved through discussion. In the event of any disputes, the final decision was made by a third-party reviewer. The results were subsequently qualitatively synthesized, with special attention paid to main themes and the effects of interventions identified by the studies.
The systematic analysis of the clinical, methodological, and statistical heterogeneity was conducted. The assessment of clinical heterogeneity was based on the variation in study populations (types of cancer, disease severity, treatment environment), intervention protocols (type of DOAC, dose schedule), and outcome definitions. Comparison of studies in terms of study design, follow-up, measurement of outcomes, and evaluation of risk of bias was used to assess methodological homogeneity. A qualitative assessment of statistical heterogeneity was made by examining the ranges and distributions of effect estimates across studies that shared the same outcome measure.
A qualitative synthesis, in the form of a narrative review, was conducted to explore the diverse approaches, participant characteristics, and outcome measures, as well as the varying follow-up lengths. The synthesis was organized by the study design [randomized controlled trials (RCTs) compared to observational studies], the primary outcome (bleeding events and VTE recurrence), the secondary outcome (patient-reported outcomes and adherence), and by type of cancer as subgroup analyses to ensure a consistent view of the evidence available.
Predefined rules followed the narrative synthesis, and the descriptive analysis was presented systematically, encompassing elements of the study, participant demographics, and the frequency of outcomes. Thematic analysis was performed to identify common themes and patterns among the studies, and comparative analysis was used to investigate similarities and differences between the research studies in terms of their types and populations. To define specific patterns and trends in various patient groups, subgroup synthesis provided an analysis of the findings by type of cancer, type of DOAC, and study design.
A quantitative meta-analysis could not be performed as there was significant heterogeneity in the definition and measures of outcomes with varying follow-up duration (6-24 months), different populations of patients with cancer and different level of staging of these patients, lack of similar reports regarding the classification of bleeding criteria and different healthcare setting and treatment regimens. Such characteristics excluded the statistical pooling of the results, which would have otherwise been significant; hence, the most suitable method of synthesizing any available data was the qualitative one.
The study design was taken into account when conducting a risk-of-bias assessment. In the case of randomized controlled trials, we used the Cochrane Risk of Bias tool to assess whether there was bias in the randomization, intention-to-treat analysis, handling of missing data, methods of measuring outcomes, and the reporting of results. The bias risk of incorporating RCTs is overviewed in the following textual synthesis: Low risk of bias: 6 RCTs (40%); A few concerns: 8 RCTs (53%); High risk of bias: 1 RCT (7%).
The Cochrane Risk of Bias was generally moderate or low in the RCTs, although specific issues were noted, including those related to open-label design and randomization. Such issues may create bias in matters related to patient and investigator awareness.
The Newcastle-Ottawa Scale was used in observational studies to assess study quality in the domains of selecting groups, comparing these groups, and understanding the exposure and outcome. The research was scored on a scale of 0 to 9 stars, with the higher the number of stars it received, the higher its methodological quality. The observational studies with bias can be summarized according to the following textual synthesis: High quality (Score 7-9): 8 studies (57%); Moderate quality (Score 4-6): 5 (36%); Low quality (Score 0-3): 1 study (7%).
Most observational studies have been classified as high in quality, indicating good research methodology. Some of them, however, were found to be of moderate or low quality, as they had issues with patient selection, and outcome data were incomplete.
Additional elements in assessing quality included having a good sample size, using the right statistical methods, full reporting of outcomes, and the possibility of publication bias. Articles with significant flaws in their study methods were identified, and their potential impact on the final results was considered during interpretation.
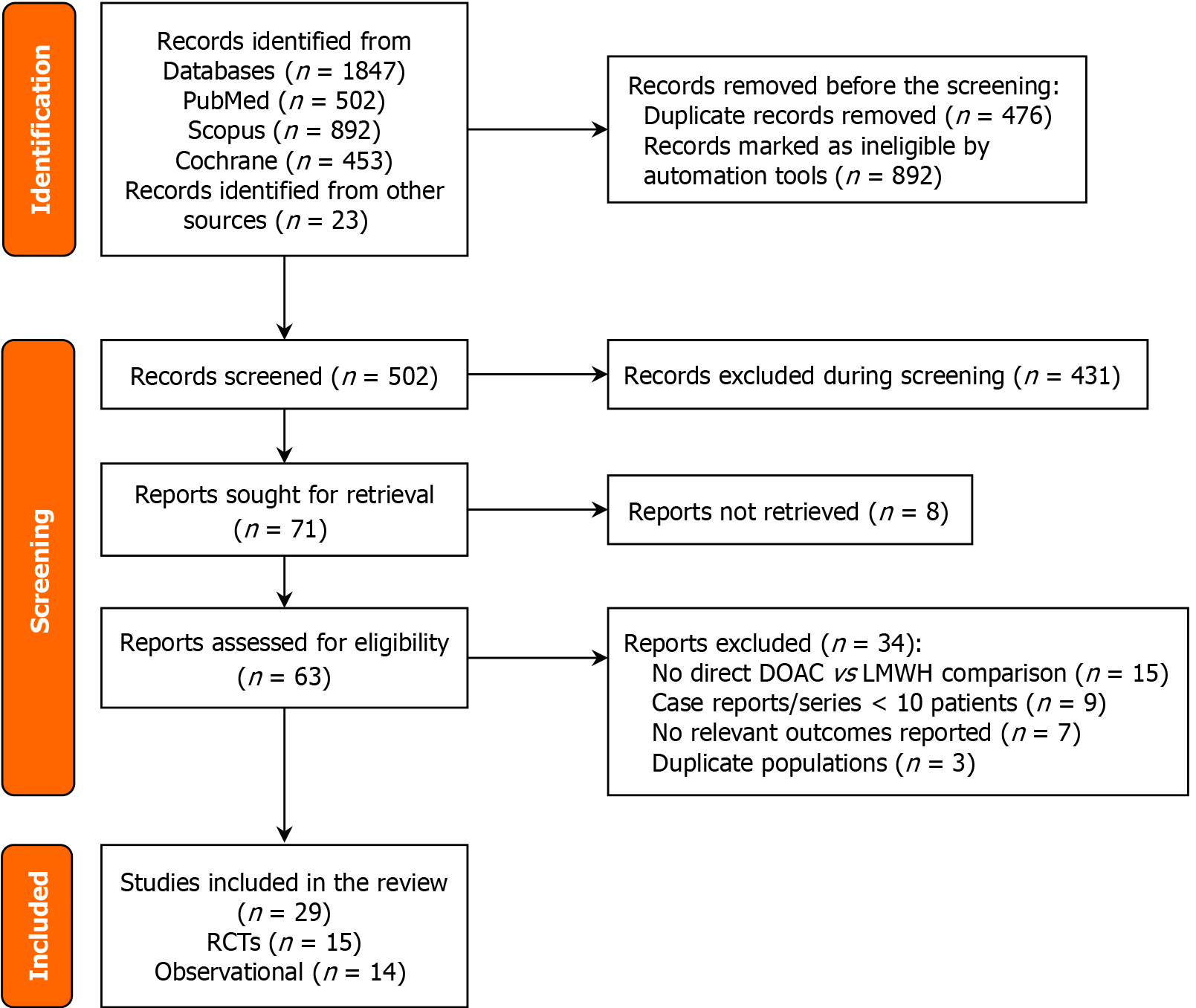
The initial database search yielded 1847 records retrieved from PubMed (502 records), Scopus (892 records), and the Cochrane Library (453 records). Reference list screening of included studies, citation searching of key review articles and consultations with scientific experts and their recommendations produced 23 records. 1368 records were excluded before screening due to duplicate records (476 records) and those deemed ineligible by automated tools (892 records). 502 unique records remained to be reviewed for title and abstract screening. After the title and the abstract screening, 431 records were excluded according to the following criteria: 15 records: Not related to cancer-associated thrombosis; 9 records: Pediatric populations only; 7 records: Focused on VTE prophylaxis instead of treatment; 3 records: Case reports, editorials, or conference abstracts; 12 records: Not comparing DOACs with LMWH; 6 records: Non-English language publications; 379 records: Other reasons.
The outcome of this screening process was 71 full-text articles, 8 of which were not retrieved, and 68 records were evaluated as eligible. In full-text assessment, 34 further articles were disregarded for the following reasons: 15 articles: No direct DOAC vs LMWH comparison; 9 articles: Case reports/series less than ten patients; 7 articles: No relevant outcomes reported; 3 articles: Duplicate patient populations from multi-publication studies.
Using all inclusion/exclusion criteria, 29 studies were finally included in the qualitative synthesis, which consisted of: 15 RCTs; 14 observational studies (including cohort studies, case-control studies, and registry analyses).
Studies included in this review were conducted between 2003 and 2024, with participant numbers ranging from 84 to 18223 patients. Patients were studied in several countries and various healthcare systems, including academic medical centers, community, and population-based registries. Based on cancer-specific research, the total sample amounted to 21371 patients in all the studies provided.
Studies conducted before 2012, when DOACs began to be widely used when treating cancer-associated thrombosis (CAT), were included in the study, provided they compared low-molecular-weight heparin (LMWH) to vitamin K antagonists that were standard treatment methods at the time. Before the advent of DOACs, the literature mainly consisted of LMWH comparisons with VKAs and lies outside the present review, which is aimed at comparing DOACs with LMWH.
The percentage agreement concerning study selection among inter-reviewers was high, with Cohen's kappa coefficients of 0.85 and 0.91 obtained in title/abstract and full-text assessments, respectively. The agreement was reached through discussion, and three disputes required consultation with a third reviewer.
All the studies had some consistent outcome measures, such as major bleeds, relevant non-major bleeds, VTE recurrence, and well-being, including satisfaction with the treatment, quality of life survey scores, and adherence to medication rules. Common secondary goals involved tracking how many people died in the trial, cancer progression and how frequently healthcare was delivered. The selection of studies is illustrated in the PRISMA flow diagram (Figure 1), and complete descriptions of all included studies are presented in Table 1.
| Ref. | Design | N | Population | Patient characteristics | Intervention | Comparator | Primary outcome | Key findings |
| Agnelli et al[4], 2020 | RCT | 1155 | Cancer patients with VTE | Active cancer, mixed tumor types | Apixaban 10 mg BID × 7 days, then 5 mg BID | Dalteparin 200 IU/kg daily | Recurrent VTE or major bleeding | Non-inferiority demonstrated; higher bleeding in GI/GU cancers |
| McBane et al[6], 2020 | RCT | 300 | Active malignancy with VTE | Predominantly solid tumors | Apixaban 10 mg BID × 7 days, then 5 mg BID | Dalteparin 200 IU/kg daily | Major bleeding | Similar efficacy; increased bleeding risk with apixaban |
| Raskob et al[8], 2018 | RCT | 1050 | Cancer-associated VTE | Mixed cancer types, active disease | Edoxaban 60 mg daily | Dalteparin 200 IU/kg daily | Composite of VTE recurrence or major bleeding | Non-inferiority shown; higher bleeding with edoxaban |
| Young et al[10], 2018 | RCT | 406 | Cancer patients with VTE | Mixed solid tumors | Rivaroxaban 15 mg BID × 21 days, then 20 mg daily | Dalteparin 200 IU/kg daily | VTE recurrence | Reduced VTE recurrence; increased bleeding risk |
| Marshall et al[5], 2020 | RCT | 406 | Cancer patients with VTE | 12-month follow-up of SELECT-D | Rivaroxaban vs placebo | Dalteparin | VTE recurrence | Sustained VTE reduction; persistent bleeding concern |
| Planquette et al[7], 2022 | RCT | 370 | Cancer-associated thrombosis | Mixed cancer types | Rivaroxaban 15 mg BID × 21 days, then 20 mg daily | Dalteparin 200 IU/kg daily | Net clinical benefit | Effective VTE prevention; bleeding concerns in GI cancers |
| Lee et al[29], 2003 | RCT | 672 | Cancer patients with VTE | Mixed cancer types | Dalteparin | Warfarin | VTE recurrence | LMWH superior to warfarin in cancer patients |
| Lee et al[2], 2015 | RCT | 900 | Active cancer with VTE | Mixed tumor types | Tinzaparin | Warfarin | VTE recurrence | Tinzaparin effective vs warfarin |
| Schrag et al[9], 2023 | RCT | 671 | Cancer patients with VTE | Real-world population | DOACs (mixed) | LMWH | VTE recurrence | Comparable effectiveness in broader population |
| Hull et al[27], 2006 | RCT | 200 | Proximal VTE with cancer | Mixed cancer types | Long-term LMWH | Usual care | VTE recurrence | Long-term LMWH beneficial |
| Deitcher et al[30], 2006 | RCT | 102 | Active cancer with VTE | Mixed solid tumors | Enoxaparin alone | Enoxaparin→Warfarin | VTE recurrence | LMWH alone superior to transition |
| Riess et al[23], 2015 | RCT | 200 | Cancer patients with VTE | German population | Rivaroxaban | LMWH | Patient satisfaction | Improved satisfaction with rivaroxaban |
| Di Nisio et al[21], 2019 | Post-hoc RCT | 1050 | Cancer patients with VTE | Extended treatment analysis | Edoxaban | Dalteparin | Extended treatment outcomes | Benefits maintained with extended treatment |
| Mulder et al[22], 2020 | Post-hoc RCT | 1050 | Different cancer types | Cancer type-specific analysis | Edoxaban | Dalteparin | Cancer type-specific outcomes | Variable outcomes by cancer type |
| Kraaijpoel et al[24], 2018 | Post-hoc RCT | 1050 | Cancer patients with bleeding | Bleeding impact analysis | Edoxaban | Dalteparin | Clinical impact of bleeding | Bleeding events clinically significant |
| Coleman et al[13], 2023 | Observational | 7441 | Cancer-associated VTE | Real-world cohort | Rivaroxaban | LMWH | Effectiveness and safety | Comparable real-world effectiveness |
| Kang et al[14], 2024 | Observational | 15656 | Cancer-associated VTE | Switching patterns analysis | Continuous LMWH vs switch to DOAC | LMWH | Safety of switching | Continuous LMWH safer than switching |
| Carney et al[16], 2021 | Observational | 121 | Cancer with thrombocytopenia | Thrombocytopenic patients | DOACs | LMWH | Bleeding in thrombocytopenia | Increased bleeding risk in both groups |
| Weitz et al[15], 2020 | Registry | 1162 | Cancer-associated thrombosis | Global registry data | Variable DOACs | LMWH | Real-world treatment patterns | Geographic variations in treatment |
| Guo et al[26], 2020 | Observational | 18223 | Cancer-associated thrombosis | US treatment patterns | DOACs | LMWH | Treatment patterns and costs | Economic implications of treatment choice |
| Farge et al[11], 2018 | Observational | 365 | Cancer patients on LMWH | Quality of life study | LMWH | N/A | Quality of life | Significant QoL burden with LMWH |
| Schaefer et al[12], 2021 | Observational | 3139 | Cancer-associated thrombosis | Adherence comparison | DOACs | LMWH | Medication adherence | Improved adherence with DOACs |
| Caroti et al[25], 2023 | Observational | 2071 | Lower bleeding risk CAT | DOAC comparison | Rivaroxaban vs Apixaban | N/A | DOAC safety comparison | Similar safety between DOACs |
| Lee et al[17], 2019 | Observational | 84 | Lung cancer with VTE | Lung cancer-specific | Rivaroxaban | Dalteparin | Efficacy and safety | Effective in lung cancer patients |
| Benzidia et al[19], 2022 | Observational | 95 | Cancer patients with VTE | Multidisciplinary care | Various anticoagulants | Standard care | Care program effectiveness | Improved outcomes with coordinated care |
| Pelzer et al[28], 2014 | Pilot | 312 | Pancreatic cancer | Pancreatic cancer-specific | Heparin with chemotherapy | Standard care | Safety and feasibility | Feasible in pancreatic cancer |
| How et al[18], 2024 | Observational | 486 | Myeloproliferative neoplasms | Splanchnic vein thrombosis | Various anticoagulants | Variable | Management in MPN | Specialized considerations for MPN |
| Font et al[20], 2023 | Observational | 287 | Cancer-associated VTE | Multinational study | Various anticoagulants | Variable | Adherence and QoL | International perspective on outcomes |
| Zabeida et al[31], 2023 | Observational | 146 | Pediatric COVID-19 | Children with coagulopathy | Anticoagulation | Variable | COVID-associated coagulopathy | Pediatric coagulation considerations |
Patient stratification by cancer status revealed significant differences in treatment outcomes. In patients receiving active cancer treatment, upper gastrointestinal (particularly gastroesophageal and pancreatic cancers) and genitourinary cancers were at the highest risk of a significant bleeding event, with the highest risks in patients receiving DOAC therapy (6.2%) compared to LMWH therapy (4.5%). In lower GI cancers, the risk was of an intermediate level, but colorectal cancers indicated a moderate risk of bleeding compared to upper GI cancers. The percentages of recurrence of VTE were comparable in DOACs (9.3%) and LMWH (10.1%). Conversely, such reduced major bleeding rates (2.1% with DOACs and 3.0% with LMWH) were observed in patients who had remission or stable disease, with similar rates of VTE recurrence (5.8% with DOACs and 6.3% with LMWH). The quality of life and patient satisfaction were also high in the remission/stable disease group, particularly in the DOAC cohort (80% satisfied), compared to the active treatment group (60%). The results of statistical analyses indicated a moderately high risk of bleeding in the sample with active treatment (P = 0.032). In contrast, the level of VTE recurrence was not significantly higher (P = 0.225), suggesting the need to balance approaches towards anticoagulant treatment based on cancer status.
For every included study, the risk of bias was assessed using appropriate, validated tools. The levels of evidence from randomized controlled trials were mainly moderate to high, as most studies featured appropriate random allocation and results reporting. Most investigations found that failing to blind patients and not including certain outcomes could be issues with open-label designs caused by the various administration methods used for oral or subcutaneous anticoagulants.
The examined studies differed in quality, and generally, larger registry-based studies were better reported than single-center investigations. Most of these studies adequately accounted for major confounding issues; however, some con
| Ref. | Randomization process | Deviations from intended interventions | Missing outcome data | Measurement of outcomes | Selection of reported results | Overall risk of bias |
| Agnelli et al[4] | Low | Some concerns | Low | Low | Low | Some concerns |
| McBane et al[6] | Low | Some concerns | Low | Low | Low | Some concerns |
| Raskob et al[8] | Low | Some concerns | Some concerns | Low | Low | Some concerns |
| Young et al[10] | Low | Some concerns | Low | Low | Low | Some concerns |
| Marshall et al[5] | Low | Some concerns | Low | Low | Low | Some concerns |
| Planquette et al[7] | Low | Some concerns | Low | Low | Low | Some concerns |
| Lee et al[29] | Low | Low | Low | Low | Low | Low |
| Lee et al[2] | Low | Low | Some concerns | Low | Low | Some concerns |
| Schrag et al[9] | Low | Some concerns | Low | Low | Low | Some concerns |
| Hull et al[27] | Some concerns | Some concerns | Some concerns | Low | Low | Some concerns |
| Deitcher et al[30] | Some concerns | High | Some concerns | Low | Low | High |
| Riess et al[23] | Some concerns | Some concerns | Low | Some concerns | Low | Some concerns |
| Di Nisio et al[21] | Low | Some concerns | Low | Low | Low | Some concerns |
| Mulder et al[22] | Low | Some concerns | Low | Low | Low | Some concerns |
| Kraaijpoel et al[24] | Low | Some concerns | Low | Low | Low | Some concerns |
| Ref. | Selection (4 points) | Comparability (2 points) | Outcome (3 points) | Total score | Quality rating |
| Coleman et al[13] | 4 | 2 | 3 | 9/9 | High |
| Kang et al[14] | 4 | 2 | 3 | 9/9 | High |
| Carney et al[16] | 3 | 1 | 2 | 6/9 | Moderate |
| Weitz et al[15] | 4 | 2 | 3 | 9/9 | High |
| Guo et al[26] | 4 | 1 | 3 | 8/9 | High |
| Farge et al[11] | 3 | 1 | 2 | 6/9 | Moderate |
| Schaefer et al[12] | 4 | 2 | 3 | 9/9 | High |
| Caroti et al[25] | 3 | 2 | 3 | 8/9 | High |
| Lee et al[17] | 2 | 1 | 2 | 5/9 | Moderate |
| Benzidia et al[19] | 2 | 1 | 2 | 5/9 | Moderate |
| Pelzer et al[28] | 2 | 1 | 2 | 5/9 | Moderate |
| How et al[18] | 3 | 1 | 3 | 7/9 | High |
| Font et al[20] | 3 | 2 | 2 | 7/9 | High |
| Zabeida et al[31] | 2 | 1 | 2 | 5/9 | Moderate |
Fifteen randomized controlled trials were found that compared the use of direct oral anticoagulants (DOACs) to LMWH in patients with VTE who had cancer. Major trial examples are the Caravaggio trial with apixaban vs dalteparin (n = 1155), the ADAM VTE study with apixaban vs dalteparin (n = 300), the Hokusai VTE Cancer trial with edoxaban vs dalteparin (n = 1050) and the SELECT-D trial with rivaroxaban vs dalteparin (n = 406). In every primary study, it was found that DOACs did not increase the rate of VTE recurrence compared to LMWH, with recurrence rates ranging from 5.6% to 11% for DOACs and from 7.9% to 11% for LMWH.
However, DOAC users experienced a greater risk of bleeding, especially significant bleeding, in patients having upper gastrointestinal (particularly gastroesophageal malignancies) or genitourinary cancer. The risk was highest among gastroesophageal cancer, and the risk stratification varied among the various GI-type cancers. The results from the Caravaggio trial gave 3.8% (apixaban) bleeding rates against 4.0% (dalteparin), whereas higher rates were found in the Hokusai VTE Cancer study of 6.9% (edoxaban) vs 4.0% (dalteparin). Patient self-reported results showed that using DOACs led to a better quality of life, higher treatment satisfaction (approximately 75% vs 45%-65% for LMWH), and better adherence to medications when the risk of bleeding was under control.
Fourteen studies examined the use of DOACs and LMWH in cancer-associated thrombosis across various healthcare settings. According to registry studies by Coleman et al[13] and Kang et al[14] (n = 7441 and n = 15656), the recurrence of VTE was the same for both DOACs and LMWH, and Kang et al[14] found that keeping patients on LMWH was safer than switching them to DOACs in those with high VTE risk. Researchers from the GARFIELD-VTE registry (Weitz et al[15], sample size n = 1162) discovered major differences in treatment choices between regions and institutions. Examining special populations, anticoagulation therapy in patients with low platelet counts was associated with a higher risk of bleeding (Carney et al[16], n = 121). However, for individuals with cancer, the outcomes varied depending on the cancer type (Lee et al[17], lung cancer, n = 84; How et al[18], myeloproliferative neoplasms, n = 486).
Schaefer et al[12] (n = 3139) and Farge et al[11] (n = 365) both reported that patients using DOACs are more likely to follow their prescription than those using LMWH, and Farge et al[11] also found that receiving daily LMWH injections hurt patients' quality of life. According to economic analyses (including 18223 people), using DOACs may lead to cost savings since these drugs do not require constant monitoring. In both multidisciplinary programs studied by Benzidia et al[19] (n = 95) and international studies by Font et al[20] (n = 287), the results showed that care programs were helpful, but treatment options varied by the healthcare system.
All the studies showed similar patterns of bleeding risks and factors based on cancer type, with more related with cancer sub-types. Across several studies[4,7,8,10], upper gastrointestinal cancers were a common reason for major bleeding when patients received DOACs, especially for patients with gastroesophageal cancers[4]. Based on differences in tumor vascularity, mucosa involvement, and location, the ranking of bleeding risk was gastroesophageal, pancreatic, colorectal, and other lower GI cancers.
Genitourinary cancers are associated with an increased risk of bleeding when treated with DOACs[4], especially in people with active bleeding in the urine or recent treatment for genitourinary diseases. Most patient populations had very low rates of severe bleeding, and these bleeds could be well-managed.
Thrombocytopenia turned out to be a significant cause of bleeding for patients using any anticoagulant. Still, studies have shown that DOACs may have increased the risk of bleeding compared to LMWH in individuals with very low platelet counts (below 100000/μL). Still, many studies failed to include patients with thrombocytopenia, which limited the evidence for this finding.
Most cancers showed the same or nearly similar risk of VTE with DOACs and LMWH[4-10], and studies also suggested that DOACs might be more effective in preventing VTE[5,10]. Additionally, when comparing these differences in recurrence rates, doctors must weigh them against concerns about bleeding and determine what is best for each patient.
Both the growth of cancer and the variation in its treatment have been reported to affect the likelihood of VTE recurrence, and studies suggest that cancer progression and patient response to treatment may be more critical predictors for these patients than the type of blood thinner used[21,22].
Research has shown that patients on DOACs tend to have a better quality of life, report greater satisfaction with their treatment, and are more likely to continue their medication than patients on other types of anticoagulation, especially when bleeding risks are similar[11,12,20,23]. The primary reasons for improved patient experiences were the removal of daily needles and fewer required checks.
Personal situations, experiences with treatment, and the healthcare system played a significant role in what patients wanted. Several patients chose LMWH out of familiarity or because they believed that not taking oral medications could protect them from harmful reactions to their cancer treatment.
Data studies have shown that treatment adherence is influenced by the ease with which patients can take medication, their level of education, and the type of care they receive[12]. Studies have shown that proper patient education and regular support should be provided to patients, regardless of the anticoagulant used.
Nearly all studies have shown that DOACs are as effective as or better than LMWH in preventing recurrent VTE. The key randomised controlled trials showed that VTE might recur in 5.6% to 11% of DOAC users and 7.9% to 11% of LMWH users, with most cases reported within the trials' non-inferiority limit[4-10].
The risk of significant bleeding was seen to vary more among the treatment groups, and several studies found that bleeding rates were slightly higher when patients took DOACs. According to the Caravaggio trial[4], the use of apixaban resulted in major bleeding in 3.8% of cases, compared to 4.0% for dalteparin. In contrast, the Hokusai VTE Cancer trial[8] revealed that using edoxaban increased the major bleeding rate to 6.9% when compared to using dalteparin. In the SELECT-D trial[10], 6% of patients who received rivaroxaban experienced major bleeding, compared to 4% of those on dalteparin.
The studies showed that DOACs led to higher rates of clinically important non-major bleeding, between 9% and 16%, when compared to LMWH, which caused non-major bleeding in about 4% to 6% of patients. Still, the significance of these bleeding events in clinical practice varied, but most could be managed by reducing the dose or taking a short break from therapy[24].
The analysis by cancer type found that bleeding risk patterns vary strongly, with upper gastrointestinal cancers had the biggest differences. Many studies found that patients with upper gastrointestinal cancers (particularly gastroesophageal and pancreatic cancers) had twice or three times as many major bleedings if they received DOAC treatment instead of LMWH[4,7,8,10]. Other low GI cancers (colorectal cancers in particular) demonstrated even more modest increases in the risk of bleeding, implying a gradient of risk along the GI tract.
DOAC treatment was preferred by patients over warfarin therapy as long as bleeding dangers were tolerable. The quality of life for patients was higher with oral anticoagulation[11,23], and over 75% of users expressed satisfaction, whereas LMWH treatment received satisfaction ratings of only 45%-65%. Data from various observational studies have revealed an improvement in adherence to DOAC therapy, ranging from 15% to 25%, compared to LMWH[12,25].
Using the same healthcare utilization metrics, the point is that the financial benefits of DOAC treatment are evident, as patients require fewer visits, fewer adjustments, and interact with fewer healthcare providers. On the other hand, the fact that these medications cost more and that patients may end up needing additional care following a bleed needs to be considered[26].
Research results demonstrated that treatment methods did not have a noticeable effect on mortality in most patients since cancer progression was generally the primary cause of death and not blood clots or bleeding[9,21,22,27]. None
It is apparent from the extensive study of DOACs vs LMWH that because both treatments have potential risks and benefits, treating cancer-associated thrombosis in an individual needs to be carefully thought out. The evidence drawn from both experimental and everyday studies helps us learn about how these anticoagulation methods measure up in terms of safety and effectiveness.
The results showing VTE recurrence prevention with all types of DOACs[4-10] is as good as warfarin treatment, leading to new oral anticoagulation options for cancer patients and clinicians. The clear signal that patients with geni
The different bleeding risks seen in cancer patients, depending on their type, imply that the location and nature of the tumour play important roles in choosing treatments[3]. Upper gastrointestinal cancers (particularly gastroesophageal and pancreatic cancers) tend to bleed more when patients take DOACs, which may be caused by the drugs irritating the stomach or intestine, the tumor’s blood vessels or by being in direct contact with lesions in the digestive tract[4,7,8,10]. This risk has an anatomical gradient with the upper GI cancers where the risk is highest, then pancreatic cancers, and then colorectal cancers and the least risk in other lower GI malignancies. The research data shows that LMWH should be the main choice for treatment in cancer patients with active upper gastrointestinal cancers (particularly gastroesophageal and pancreatic cancers). In contrast, DOACs may be used in patients with lower-risk GI cancers or others who are already stabilized in care. The decision to be made must include cancer subtype-specific bleeding risk stratifications instead of considering all the GI cancers as a whole.
Considering the common occurrence of treatment-related cytopenias in cancer patients, the presence of thrombocytopenia should be noted as important in clinical care[16]. Even though studies including thrombocytopenic patients are often not done, what we have seen so far suggests that low platelet counts increase a patient's risk of bleeding with any of these blood-thinning agents. However, due to the limited evidence, the increased risk of thrombocytopenia in patients using DOACs must be considered, and a risk-benefit assessment must be made individually. In cases where more substantial evidence is lacking, LMWH may be chosen for patients with thrombocytopenia.
Patient opinions about treatment through these measures reveal important details about the acceptability and results of therapy in everyday life[11,12,20,23]. Because dialysis patients can experience bleeding, oral anticoagulation is a natural choice, as daily LMWH injections and seeing a doctor for checks every 2 weeks are demanding. Based on these findings, shared decision-making should consider the patient's wishes, well-being and psychological wellness in addition to clinical effectiveness and safety.
Adherence shows that taking medication easily, informing patients well and achieving good clinical outcomes are all closely related and cannot be reduced to medicine form only[12]. Although oral medications often encourage patients to adhere to their treatment, studies indicate that providing patients with thorough support and education helps achieve better results for all types of anticoagulants. This information highlights the need for increased investments in programs designed to teach and help patients manage cancer-related blood clots.
Evidence from observational studies and randomized trials helps explain and confirm the outcomes found in clinical trials while also describing what happens in typical clinical care settings. Researchers can generalize trial results because the findings typically remain consistent across various healthcare systems and patient populations; however, some variations in treatment may exist between locations and institutions, which could be attributed to differences in guidelines or resources[15,26].
Although economic factors are not the primary focus of this review, they are significant in influencing the way treatments are chosen and decisions are made in healthcare systems[26]. Though taking DOACs may be more expensive than other drugs, the ease of monitoring could save money when it comes to visiting the doctor, but economic studies on cancer patients are not numerous.
Changes in cancer treatment, such as the inclusion of immunotherapy and targeted agents, introduce new aspects to anticoagulation management that aren't discussed in current guidelines[1]. Because of possible drug interactions, altered bleeding risks and changing survival rates in cancer patients, the best strategy for anticoagulation may need to be studied, and guidelines can be updated as required.
When choosing treatment for elderly patients, those with renal problems or those on intensive chemotherapy, extra attention is needed, but findings on these specific groups are limited[18,28]. Because there are many types of cancers and various treatment methods, personalized care through thorough risk assessment will continue to be essential.
A common finding in studies[19] is that bringing together experts from different fields is more important than selecting a particular anticoagulant for achieving good results. Such results encourage healthcare systems to support more advanced thrombosis programs, helping to inform difficult treatment choices.
Important areas that still need more exploration in future research have been identified[1,3]. Long-term information on how well DOACs work and are safe in cancer patients is scarce, especially for periods beyond 6-12 months. Cancer guidelines must be updated with new information, especially regarding advancements in treatments and changes in patient care.
Developing bleeding risk prediction models for cancer patients on anticoagulation could enhance both the selection of the right treatment and help patients understand their medications. Likewise, developing specialized quality-of-life tools for cancer patients undergoing anticoagulation may help us determine if the treatment is suitable for them and beneficial.
Studies on transitioning from LMWH to DOAC therapy when clinical circumstances change are an area that requires further examination. Regular monitoring while moving from active cancer treatment, having surgery or at the end of life should be studied further.
If treatment and outcome measures are common across nations, it will help researchers in their work and make clinical practice more consistent internationally[1]. Routines for shared decision-making and how patients are taught could improve healthcare outcomes and offer greater patient satisfaction.
Several major issues impact the current body of evidence. Since there is a lot of variation among patient groups, the types of cancers they have, the disease's progression, and the simultaneous management, it is challenging to recommend specific treatments for each group. Additionally, the short follow-up periods in most studies (6-12 months) do not provide insight into long-term safety and effectiveness.
Results from randomized trials do not often apply to patients with thrombocytopenia, bleeding, or a high likelihood of bleeding, which are frequently encountered in clinical practice. Additionally, since studies frequently employ their types of outcomes and measurement methods, it is challenging to compare and combine the findings.
Current evidence gaps include limited data on rare cancer types and novel treatment modalities such as immunotherapy and targeted therapy, which may alter anticoagulation management considerations. The predominance of studies from high-resource healthcare settings may limit applicability to diverse healthcare environments with varying treatment protocols and monitoring capabilities.
Since there are no standardized approaches to handling shared decision-making and patient education, it is chal
The current systematic review summarized the evidence from 29 studies (15 randomized controlled trials and 14 observational studies), including 21371 cancer patients with venous thromboembolism, comparing DOACs with LMWH for managing cancer-associated thrombosis. As the evidence shows similar effectiveness in preventing VTE recurrence in both DOACs and LMWH, the rates are 5.6%-11% for DOACs and 7.9%-11% for LMWH in major trials. Nevertheless, the bleeding risks of DOACs indicate discordant findings; gastrointestinal and genitourinary malignancies have higher rates of major bleeding events with DOACs (risk ratios of 1.5-3.0 compared with LMWH), whereas patient-reported outcomes continuously favored DOACs, which show a better quality of life and treatment satisfaction of 75%-83% compared to 45%-65% of LMWH.
When bleeding with DOACs is not a major concern, patients usually prefer them, making it helpful to include such preferences in treatment decisions. All the evidence indicates that, depending on the specific cancer sub-type, the presence of increased risk of bleeding, and patient or caregiver preferences, DOACs are a valid alternative to LMWH in treating cancer-related thrombosis, especially in low-risk bleeding cancer patients (lung, breast, hematologic mali
The quality of evidence was overall of moderate to poor quality, with the majority of RCTs, except for those related to outcomes, having inadequate randomization and open-label design, and observational studies exhibiting heterogeneous reporting of patient and outcome measures, which prohibited quantitative synthesis. Most trials exclude high-risk patients, making them less applicable to real-world clinical practice.
The review was limited by a high degree of clinical and methodological heterogeneity in the included studies, which prevented conducting a quantitative meta-analysis. Additionally, the majority of studies were conducted in high-resource settings, which limits the applicability of the findings to diverse healthcare settings. Most studies involve short follow-up and recall, which makes it impossible to measure long-term results. Additionally, many studies have excluded high-risk patients, resulting in limited external validity.
Future research gaps encompass long-term safety and efficacy data that are exceeding 12 months, standard methods of measuring outcomes and measuring the risk of hemorrhage, how DOAC-based regimens perform in populations at higher risk of developing cancer, specific studies in patients with thrombocytopenia to form clear anticoagulation recommendations in this group of patients, and, finally, studies of how new cancer therapies, such as immunotherapy and targeted agents, are managed with anticoagulation. Overall, the evidence favours the incorporation of DOACs into treatment algorithms for cancer-associated thrombosis, highlighting the exceptional role of individualized risk-benefit evaluation and shared decision-making in selecting specific treatments.
| 1. | Hoang SV, Ho THQ, Dang THQ, Nguyen HT, Pham PC, Do TA, Bui HH, Nguyen HT, Vuong HDT, Dang DQ, Than THN, Nguyen HH, Phan TT, Dinh HTT. Cancer-associated Thrombosis in Vietnam: Literature Review and Expert Perspectives. J Appl Hematol. 2024;15:9-21. [DOI] [Full Text] |
| 2. | Lee AYY, Kamphuisen PW, Meyer G, Bauersachs R, Janas MS, Jarner MF, Khorana AA; CATCH Investigators. Tinzaparin vs Warfarin for Treatment of Acute Venous Thromboembolism in Patients With Active Cancer: A Randomized Clinical Trial. JAMA. 2015;314:677-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 450] [Cited by in RCA: 480] [Article Influence: 43.6] [Reference Citation Analysis (3)] |
| 3. | Pernod G, Joly M, Sonnet B. Direct oral anticoagulant (DOAC) versus low-molecular-weight heparin (LMWH) for the treatment of cancer-associated thrombosis (which agent for which patient). J Med Vasc. 2020;45:6S17-6S23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 4. | Agnelli G, Becattini C, Meyer G, Muñoz A, Huisman MV, Connors JM, Cohen A, Bauersachs R, Brenner B, Torbicki A, Sueiro MR, Lambert C, Gussoni G, Campanini M, Fontanella A, Vescovo G, Verso M; Caravaggio Investigators. Apixaban for the Treatment of Venous Thromboembolism Associated with Cancer. N Engl J Med. 2020;382:1599-1607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 473] [Cited by in RCA: 757] [Article Influence: 126.2] [Reference Citation Analysis (0)] |
| 5. | Marshall A, Levine M, Hill C, Hale D, Thirlwall J, Wilkie V, French K, Kakkar A, Lokare A, Maraveyas A, Chapman O, Arif A, Petrou S, Maredza M, Hobbs R, Dunn JA, Young AM. Treatment of cancer-associated venous thromboembolism: 12-month outcomes of the placebo versus rivaroxaban randomization of the SELECT-D Trial (SELECT-D: 12m). J Thromb Haemost. 2020;18:905-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 62] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 6. | McBane RD 2nd, Wysokinski WE, Le-Rademacher JG, Zemla T, Ashrani A, Tafur A, Perepu U, Anderson D, Gundabolu K, Kuzma C, Perez Botero J, Leon Ferre RA, Henkin S, Lenz CJ, Houghton DE, Vishnu P, Loprinzi CL. Apixaban and dalteparin in active malignancy-associated venous thromboembolism: The ADAM VTE trial. J Thromb Haemost. 2020;18:411-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 424] [Article Influence: 70.7] [Reference Citation Analysis (0)] |
| 7. | Planquette B, Bertoletti L, Charles-Nelson A, Laporte S, Grange C, Mahé I, Pernod G, Elias A, Couturaud F, Falvo N, Sevestre MA, Ray V, Burnod A, Brebion N, Roy PM, Timar-David M, Aquilanti S, Constans J, Bura-Rivière A, Brisot D, Chatellier G, Sanchez O, Meyer G, Girard P, Mismetti P; CASTA DIVA Trial Investigators. Rivaroxaban vs Dalteparin in Cancer-Associated Thromboembolism: A Randomized Trial. Chest. 2022;161:781-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 125] [Article Influence: 31.3] [Reference Citation Analysis (0)] |
| 8. | Raskob GE, van Es N, Verhamme P, Carrier M, Di Nisio M, Garcia D, Grosso MA, Kakkar AK, Kovacs MJ, Mercuri MF, Meyer G, Segers A, Shi M, Wang TF, Yeo E, Zhang G, Zwicker JI, Weitz JI, Büller HR; Hokusai VTE Cancer Investigators. Edoxaban for the Treatment of Cancer-Associated Venous Thromboembolism. N Engl J Med. 2018;378:615-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1368] [Cited by in RCA: 1166] [Article Influence: 145.8] [Reference Citation Analysis (3)] |
| 9. | Schrag D, Uno H, Rosovsky R, Rutherford C, Sanfilippo K, Villano JL, Drescher M, Jayaram N, Holmes C, Feldman L, Zattra O, Farrar-Muir H, Cronin C, Basch E, Weiss A, Connors JM; CANVAS Investigators. Direct Oral Anticoagulants vs Low-Molecular-Weight Heparin and Recurrent VTE in Patients With Cancer: A Randomized Clinical Trial. JAMA. 2023;329:1924-1933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 114] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 10. | Young AM, Marshall A, Thirlwall J, Chapman O, Lokare A, Hill C, Hale D, Dunn JA, Lyman GH, Hutchinson C, MacCallum P, Kakkar A, Hobbs FDR, Petrou S, Dale J, Poole CJ, Maraveyas A, Levine M. Comparison of an Oral Factor Xa Inhibitor With Low Molecular Weight Heparin in Patients With Cancer With Venous Thromboembolism: Results of a Randomized Trial (SELECT-D). J Clin Oncol. 2018;36:2017-2023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 670] [Cited by in RCA: 955] [Article Influence: 119.4] [Reference Citation Analysis (4)] |
| 11. | Farge D, Cajfinger F, Falvo N, Berremili T, Couturaud F, Bensaoula O, Védrine L, Bensalha H, Bonnet I, Péré-Vergé D, Coudurier M, Li V, Rafii H, Benzidia I, Connors JM, Resche-Rigon M. Quality of life in cancer patients undergoing anticoagulant treatment with LMWH for venous thromboembolism: the QUAVITEC study on behalf of the Groupe Francophone Thrombose et Cancer (GFTC). Oncotarget. 2018;9:26990-26999. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 12. | Schaefer JK, Li M, Wu Z, Basu T, Dorsch MP, Barnes GD, Carrier M, Griggs JJ, Sood SL. Anticoagulant medication adherence for cancer-associated thrombosis: A comparison of LMWH to DOACs. J Thromb Haemost. 2021;19:212-220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 13. | Coleman CI, Caroti KS, Abdelgawwad K, Psaroudakis G, Fatoba S, Rivera M, Schaefer B, Brobert G, Khorana AA, Becattini C, Lee AYY, Ekbom A, Carrier M, Brescia C, Cohen AT. Effectiveness and Safety of Rivaroxaban and Low Molecular Weight Heparin in Cancer-Associated Venous Thromboembolism. JACC CardioOncol. 2023;5:189-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 14. | Kang W, Huang C, Yan VKC, Wei Y, Shami JJP, Li STH, Yang Y, Ye X, Tang J, Lee SF, Lee VHF, Chan SL, El Helali A, Lam KO, Ngan RKC, Wong ICK, Chan EW. Effectiveness and safety of continuous low-molecular-weight heparin versus switching to direct oral anticoagulants in cancer-associated venous thrombosis. Nat Commun. 2024;15:5657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 15. | Weitz JI, Haas S, Ageno W, Goldhaber SZ, Turpie AGG, Goto S, Angchaisuksiri P, Nielsen JD, Kayani G, Farjat AE, Schellong S, Bounameaux H, Mantovani LG, Prandoni P, Kakkar AK; GARFIELD-VTE investigators. Cancer associated thrombosis in everyday practice: perspectives from GARFIELD-VTE. J Thromb Thrombolysis. 2020;50:267-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 80] [Article Influence: 13.3] [Reference Citation Analysis (3)] |
| 16. | Carney BJ, Wang TF, Ren S, George G, Al Homssi A, Gaddh M, Connolly GC, Shah VI, Bogue T, Bartosic A, Neuberg D, Baumann Kreuziger L, Zwicker JI; VENUS Network Investigators. Anticoagulation in cancer-associated thromboembolism with thrombocytopenia: a prospective, multicenter cohort study. Blood Adv. 2021;5:5546-5553. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 17. | Lee JH, Hyun DG, Choi CM, Lee JC, Kim WS, Oh YM, Lee SD, Lee JS. A Retrospective Study on Efficacy and Safety of Rivaroxaban and Dalteparin for Long-Term Treatment of Venous Thromboembolism in Patients with Lung Cancer. Respiration. 2019;98:203-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 18. | How C, Chrysafi P, Ko A, Zon R, Ghorbanzadeh A, Sastow DL, Georgen M, Prabhu A, Pachpande V, Valisekka SS, Agarwal M, Nayak AR, Cheong MA, Iurlo A, Cattaneo D, Chiasakul T, Dhawan R, Sanfilippo KM, Hobbs GS, Houghton DE, Beckman J, Baumann Kreuziger L, Tremblay D, Patell R, Lauw MN. Anticoagulation Management of Splanchnic Vein Thrombosis in Myeloproliferative Neoplasms: A Global Abdominal/Splanchnic Thrombosis Retrospective Observational Study in 486 MPN Patients (GASTRO-MPN). Blood. 2024;144:16-16. [DOI] [Full Text] |
| 19. | Benzidia I, Crichi B, Montlahuc C, Rafii H, N'Dour A, Sebuhyan M, Gauthier H, Ait Abdallah N, Benillouche P, Villiers S, Le Maignan C, Farge D. Effectiveness of a multidisciplinary care program for the management of venous thromboembolism in cancer patients: a pilot study. J Thromb Thrombolysis. 2022;53:417-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 20. | Font C, Gomez-Mesa JE, López-Núñez JJ, Calderón C, Galindo-Coral S, Wu CC, Ma J, Kroll MH, Rojas-Hernandez CM. Measurement of adherence and health-related quality of life during anticoagulation therapy in cancer-associated venous thromboembolism (VTE): a multicenter quantitative study. Support Care Cancer. 2023;31:615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 21. | Di Nisio M, van Es N, Carrier M, Wang TF, Garcia D, Segers A, Weitz J, Buller H, Raskob G. Extended treatment with edoxaban in cancer patients with venous thromboembolism: A post-hoc analysis of the Hokusai-VTE Cancer study. J Thromb Haemost. 2019;17:1866-1874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 22. | Mulder FI, van Es N, Kraaijpoel N, Di Nisio M, Carrier M, Duggal A, Gaddh M, Garcia D, Grosso MA, Kakkar AK, Mercuri MF, Middeldorp S, Royle G, Segers A, Shivakumar S, Verhamme P, Wang T, Weitz JI, Zhang G, Büller HR, Raskob G. Edoxaban for treatment of venous thromboembolism in patient groups with different types of cancer: Results from the Hokusai VTE Cancer study. Thromb Res. 2020;185:13-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 52] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 23. | Riess H, Sinn M, Kreher S; für den Arbeitskreis Hämostaseologie der Deutschen Gesellschaft für Hämatologie und Medizinische Onkologie (DGHO). [CONKO-011: Evaluation of patient satisfaction with the treatment of acute venous thromboembolism with rivaroxaban or low molecular weight heparin in cancer patients. A randomized phase III study]. Dtsch Med Wochenschr. 2015;140 Suppl 1:S22-S23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 24. | Kraaijpoel N, Di Nisio M, Mulder FI, van Es N, Beyer-Westendorf J, Carrier M, Garcia D, Grosso M, Kakkar AK, Mercuri MF, Middeldorp S, Hernandez CR, Santamaria A, Schwocho L, Segers A, Verhamme P, Wang TF, Weitz JI, Zhang G, Zwicker JI, Büller HR, Raskob GE. Clinical Impact of Bleeding in Cancer-Associated Venous Thromboembolism: Results from the Hokusai VTE Cancer Study. Thromb Haemost. 2018;118:1439-1449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 158] [Article Influence: 19.8] [Reference Citation Analysis (3)] |
| 25. | Caroti KS, Becattini C, Carrier M, Cohen AT, Ekbom A, Khorana AA, Lee AYY, Brescia C, Abdelgawwad K, Psaroudakis G, Rivera M, Schaefer B, Brobert G, Coleman CI. Rivaroxaban versus Apixaban for Treatment of Cancer-Associated Venous Thromboembolism in Patients at Lower Risk of Bleeding. TH Open. 2023;7:e206-e216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 26. | Guo JD, Hlavacek P, Poretta T, Wygant G, Lane D, Gorritz M, Wang X, Chen CC, Wade RL, Pan X, Rajpura J, Stwalley B, Rosenblatt L. Inpatient and outpatient treatment patterns of cancer-associated thrombosis in the United States. J Thromb Thrombolysis. 2020;50:386-394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 27. | Hull RD, Pineo GF, Brant RF, Mah AF, Burke N, Dear R, Wong T, Cook R, Solymoss S, Poon MC, Raskob G; LITE Trial Investigators. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med. 2006;119:1062-1072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 455] [Cited by in RCA: 421] [Article Influence: 21.1] [Reference Citation Analysis (0)] |
| 28. | Pelzer U, Hilbig A, Stieler JM, Bahra M, Sinn M, Gebauer B, Dörken B, Riess H. Intensified chemotherapy and simultaneous treatment with heparin in outpatients with pancreatic cancer - the CONKO 004 pilot trial. BMC Cancer. 2014;14:204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 29. | Lee AY, Levine MN, Baker RI, Bowden C, Kakkar AK, Prins M, Rickles FR, Julian JA, Haley S, Kovacs MJ, Gent M; Randomized Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer (CLOT) Investigators. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med. 2003;349:146-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1831] [Cited by in RCA: 1735] [Article Influence: 75.4] [Reference Citation Analysis (4)] |
| 30. | Deitcher SR, Kessler CM, Merli G, Rigas JR, Lyons RM, Fareed J; ONCENOX Investigators. Secondary prevention of venous thromboembolic events in patients with active cancer: enoxaparin alone versus initial enoxaparin followed by warfarin for a 180-day period. Clin Appl Thromb Hemost. 2006;12:389-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 276] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 31. | Zabeida A, Winikoff R, Pelland-Marcotte MC, Charlebois J, Sabapathy C. COVID-19-associated coagulopathy in children: A multicenter observational cohort study. Pediatr Blood Cancer. 2023;70:e30079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |