Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.109312
Revised: June 12, 2025
Accepted: November 17, 2025
Published online: June 20, 2026
Processing time: 351 Days and 17.3 Hours
Informed consent is a cornerstone of ethical endoscopic practice, ensuring that patients understand and agree to the procedures that they undergo. Despite its importance, studies suggest that variations in consent practices per
To investigate current practices around consent taking among endoscopists in Nigeria.
This cross-sectional survey was conducted from August to September 2024 among endoscopists practicing in Nigeria. The participants completed a 32-part self-administered online questionnaire that documented the experiences of endoscopists concerning consent taking in endoscopy practice. These included the location of practice, awareness of legislation concerning informed consent in Nigeria, patient autonomy, various procedural risks discussed with patients, and challenges faced. At the end of the survey, the responses were analyzed using tables, pie charts, and figures.
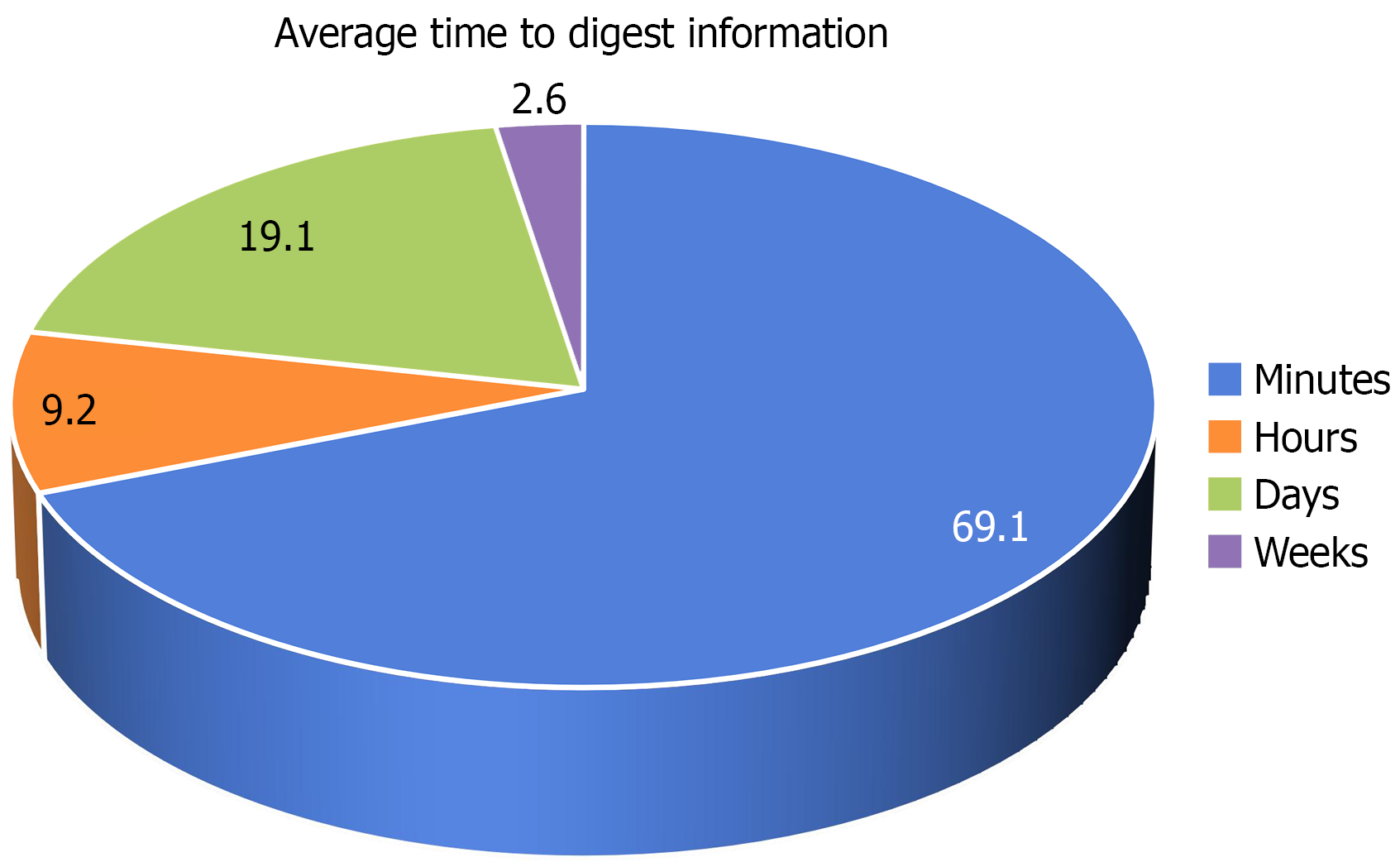
A total of 68 participants responded to the questionnaire. The mean age of the participants was 45.2 ± 6.6 years. There were 26 (38.2%) females and 42 (61.8%) males. Participants practicing in the northern part of the country comprised 41.1% (30) of the participants, while those practicing in southern Nigeria were 58.9% (38). Only 49.3% were aware of legislation about informed consent in Nigeria. The majority of the study participants (69.1%) gave the patients a few minutes to digest information before the procedure. A variety of risks were reported to be dis
Consent taking for endoscopy in Nigeria is not ideal; many healthcare workers only gave patients a few minutes to understand information about the procedure. To address this, training workshops and online webinars should be organized to train medical staff on how to do it properly. This will help improve patient care, ensure safety, and lead to better health results for patients.
Core Tip: Consent taking is important in endoscopic procedures so as to optimize procedural safety and patient outcomes. This is ethically important as part of good endoscopic practice. The process of consent taking is suboptimal in Nigeria. Specific measures such as training workshops and online webinars for endoscopists should be regularly held to improve the process in Nigeria.
- Citation: Okonkwo KC, Davwar PM, Musa Y, Bojuwoye MO, Onyia CP, Egboh SMC, Chukwurah SN, Obasi E, Asaolu OI, Adiri WN, Okeke UF, Dankiri NA, Udigwe CF. From understanding to agreement: The current state of informed consent in Nigerian endoscopic practice. World J Methodol 2026; 16(2): 109312
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/109312.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.109312
The demand for endoscopy procedures continues to increase and is likely to rise for the foreseeable future[1]. It is therefore imperative that the consent process is comprehensive and standardized to match the increasing complexity of both diagnostic and therapeutic procedures currently being performed by endoscopists[2].
Consent is a legal process whereby the patient is informed of the reason why the practicing clinician deems the procedure to be of benefit; it is an activity that involves adequate explanation of the potential risks or side effects of the procedure, and provision of alternatives[1].
Informed consent is also the mechanism by which a patient can choose the medical treatment that they receive, as it is they who will live with the consequences[3].
This is written in the simplest possible language that a patient can comprehend, often via oral communication, but supplemental written or video materials can be used[4].
This is in keeping with global best practices that promote a patient-centered model where risks, benefits, and alternatives, including not undergoing procedures, are discussed between the patient and clinician[5].
Few studies that have studied pre-surgery consent taking in Nigeria have described a paternalistic approach; there is, however, a paucity of data on the endoscopy consent-taking process in Nigeria. This creates a vacuum in ascertaining whether patients undergoing endoscopy are benefiting from global best practices[6-10].
This nationwide study examined the process of obtaining consent before endoscopy in Nigeria. It also highlights the varied methods and contexts of consent seeking in Nigeria.
The study was a cross-sectional survey conducted among clinicians performing gastrointestinal endoscopy across the six zones of Nigeria over two months, from August to September 2024. It was a descriptive study that examined the individual experiences of endoscopists on consent taking in Nigeria. Clinicians performing gastrointestinal endoscopy in all the Nigerian geo-political regions were encouraged to participate in the study.
All Nigerian clinicians performing endoscopy and who volunteered to participate in the study were included. The endoscopist who did not respond was excluded.
The study was conducted online using Google Forms. It involved a 32-part self-administered questionnaire that inquired about individual experiences concerning consent in endoscopy practice. It was shared on the social media handle of Gastroenterologists/Endoscopists in Nigeria. The Form had an opt-in preference in which respondents showed consent by filling out the questionnaire. The social media page had about 150 endoscopists, of whom 68 responded. The form captured demographic information, study center details, location of practice, and awareness of informed consent legislation in Nigeria. Additionally, the survey explored patient autonomy, risks discussed with patients, and challenges in the consent process. The form primarily employed dropdowns and multiple-choice questions, along with open-ended prompts for age and the name of the endoscopy center. At the end of the survey, responses were analyzed using SPSS version 26, and the data were presented in tables, pie charts, and figures. Inferential statistics were done using the χ2 test.
Ethical clearance for the study was obtained from the University of Medical Sciences, Ondo State Health Research and Ethics Committee, reference number NHREC/TR/UNIMED-HREC-Ondo St/22/06/21.
At the end of the 2-month survey, a total of 68 participants responded to the questionnaire. The mean age ± SD of the respondents was 45.2 ± 6.6 years, with a range of 37-69 years. There were 42 (61.8%) males with an M:F ratio of 1.6:1, with predominant response from North-Central Nigeria and least response from North-East. Detailed demography of the respondents is shown in Table 1.
| Variable | Frequency (n) | Percentage (%) |
| Age group (in years) | ||
| 37-44 | 37 | 54.4 |
| 45-54 | 26 | 38.2 |
| > 55 | 5 | 7.4 |
| Gender | ||
| Female | 26 | 38.2 |
| Male | 42 | 61.8 |
| Location of practice | ||
| North Central | 20 | 29.4 |
| North East | 3 | 4.4 |
| North West | 7 | 10.3 |
| South East | 14 | 20.6 |
| South South | 7 | 10.3 |
| South West | 17 | 25.0 |
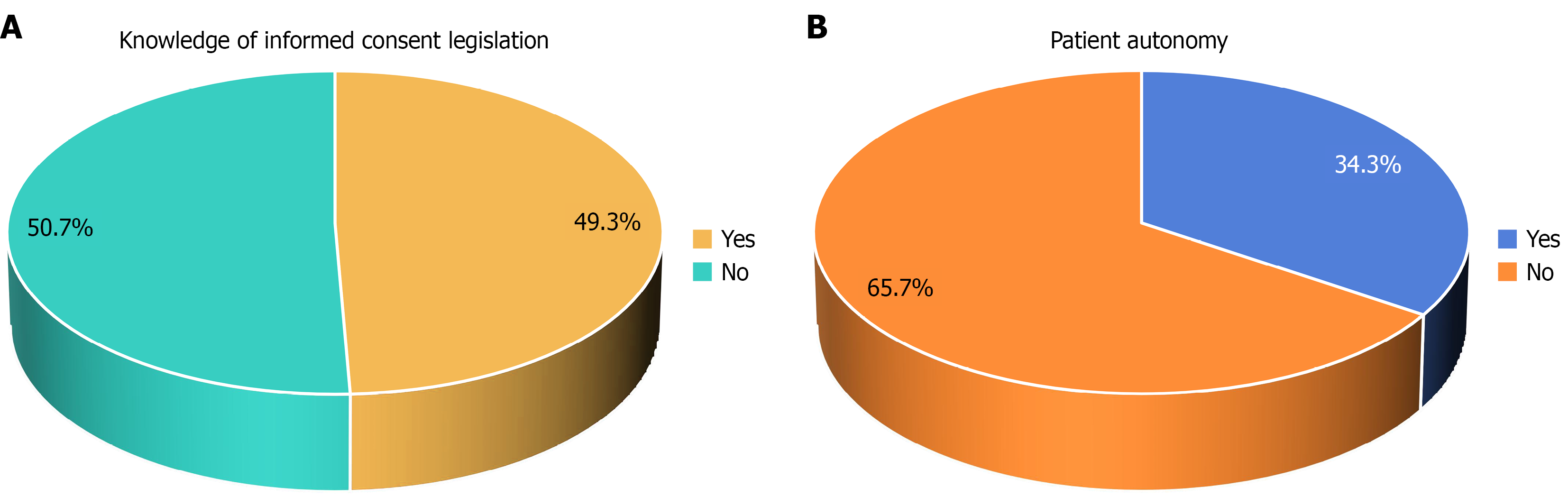
The survey revealed that at least 50.7% of respondents were aware of the legislation concerning informed consent in the country. However, there was no statistically significant regional difference in knowledge on consent legislation among the respondents as shown on Table 2 and Figure 1A. Additionally, 65.7% of endoscopists expressed a willingness to allow patients to decide whether their endoscopy can be used as a learning activity as Figure 1.
| Variable | Know about consent legislation | F test | P value | |
| Yes | No | |||
| North central | 9 (45.0) | 11 (55.0) | ||
| North East | 2 (67.7) | 1 (33.3) | 6.936 | 0.214 |
| North West | 1 (14.3) | 6 (85.7) | ||
| South East | 8 (57.1) | 6 (42.9) | ||
| South South | 2 (28.6) | 5 (71.4) | ||
| South West | 11 (64.7) | 6 (35.3) | ||
There was no statistically significance between the geographical regions in terms of knowledge of consent taking legislation in Nigeria (P > 0.05).
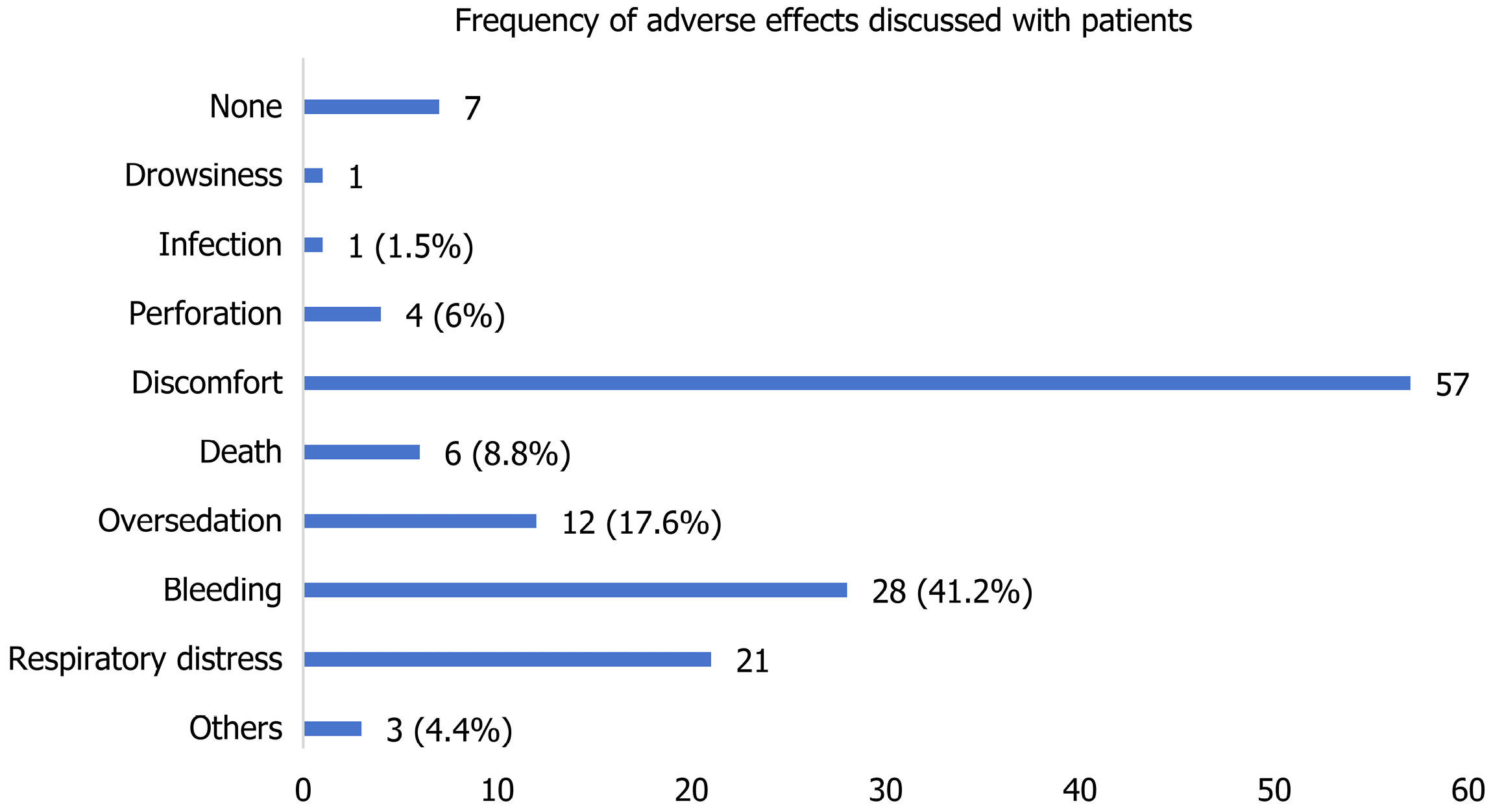
Most endoscopists focus primarily on informing patients about anticipated discomfort associated with the procedure. Notably, up to 10% do not provide any information regarding potential side effects of endoscopy (Figure 2).
Most endoscopists conduct counseling on the day of the procedure, allowing patients a few minutes to assimilate the information and make a decision regarding the procedure (Figure 3).
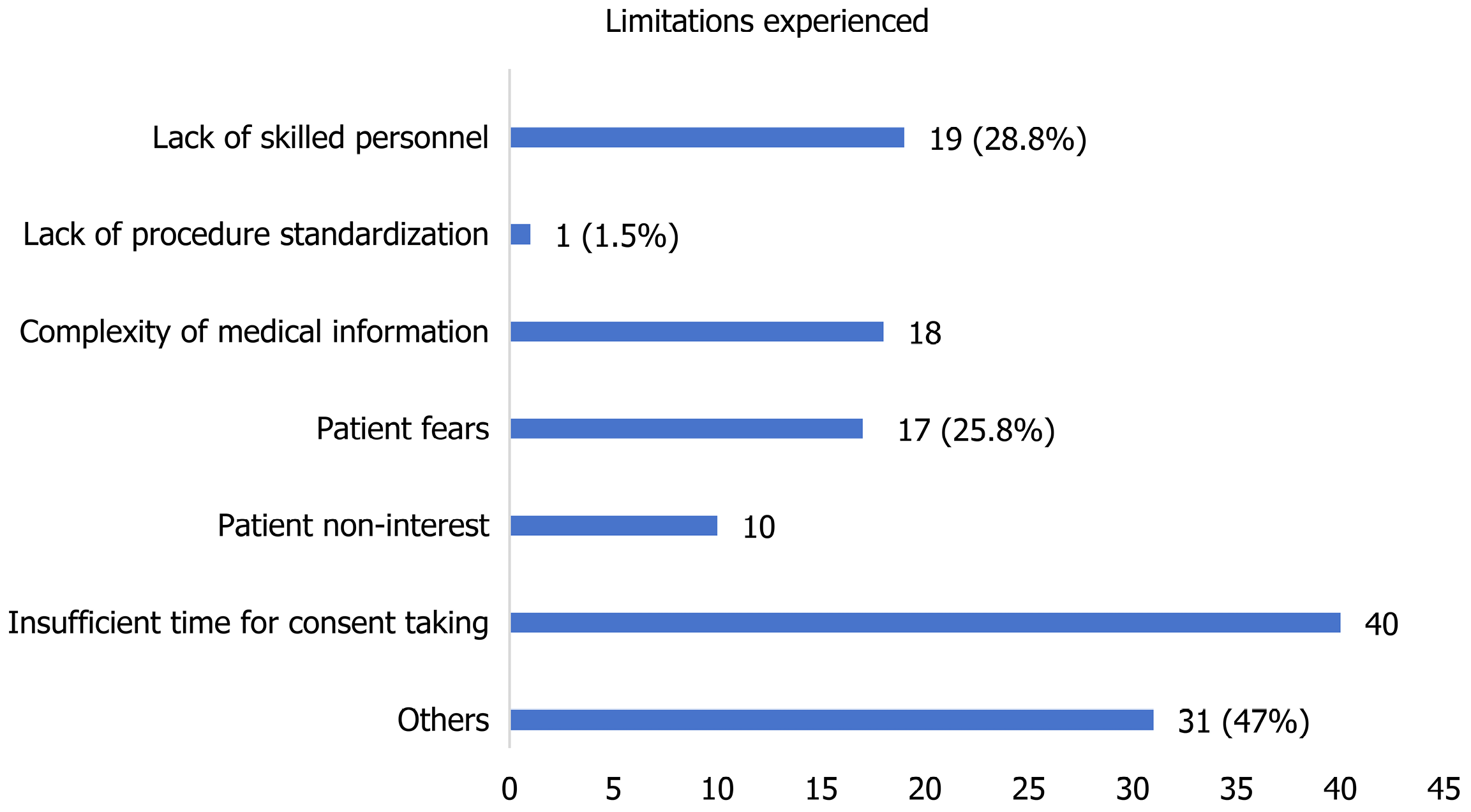
Many respondents reported that time constraints significantly hinder their ability to thoroughly explain the details of the endoscopy procedure before obtaining consent. This lack of adequate explanation can lead to misunderstandings and insufficiently informed consent. Other factors reported are illustrated in Figure 4.
This study investigated the factors influencing the process of informed consent for endoscopy among healthcare providers in Nigeria. A total of 68 participants completed the survey, revealing essential insights into their demographic characteristics, the information provided to patients regarding the procedure, and the time allocated for patient decision-making. This discussion will explore the implications of these findings concerning current practices and their potential impact on patient care and informed consent.
This study is the first attempt to examine the process of consent seeking among endoscopists in Nigeria, and it has a national outlook as respondents were from all the geopolitical regions. There have been a few studies on consent-taking before surgery in Nigeria, which have provided insight into what the process looks like, but not specifically for endo
In terms of patients’ autonomy, 34.3% of the respondents said that they discussed with the patient if they wanted students to observe the procedure as a learning activity. This low percentage is not in agreement with the recom
Furthermore, a large percentage (50.7%) of the respondents were not aware of any legislation in Nigeria regarding informed consent. This differs from the European Society of Gastrointestinal Endoscopy (ESGE) guideline, which states that practitioners must know and follow the laws of their own country[3]. A similar high level of unawareness has been previously documented among Nigerian healthcare practitioners in general. This is similar to a study in the Democratic Republic of Congo, which shows a poor consent-seeking process due to a lack of knowledge of legislation regarding consent seeking. Less than 50% of the respondents attested that they always obtained consent prior to an examination or to any procedure[11].
Respondents' lack of knowledge about consent laws in this study may be because Nigeria’s laws on consent are not clear enough[8]. Legislation on informed consent in Nigeria should be further developed and expanded to cater to the peculiarities of clinical practice, including endoscopy services.
Many of the respondents took their time to explain to patients a myriad of possible adverse effects of endoscopy. This is in tandem with the updated 2016 BSG guidelines, which state that clinical factors related to the patient’s symptoms, past medical history, and specific patient-related risk factors, including possible adverse outcomes related to the proposed procedure, should be individualized and discussed with each patient. This is a very positive finding as it shows that Nigerian endoscopists are comprehensive in discussing potential side-effects with patients so as to optimize their overall endoscopy experience. The fact that medical ethics is entrenched in the curriculum of medical students in Nigeria may explain this positive finding[12].
Most of the respondents (69.1%) gave the patients minutes to digest the information about endoscopy. This is suboptimal as this may not permit the time and space for questions and a comprehensive understanding of the procedure. The ESGE guideline, as revised in 2023, recommends that days to weeks be allowed for adequate interaction, depending on the complexity of the procedure to be done[5]. The most common limitation, as adduced by the respondents to comprehensive consent seeking, is inadequate time for the process. This may be due to the fact that the current doctor-to-patient ratio is low and is worsening as a result of the current tide of migration of health professionals[13].
One of the key patient factors as underlined by about a third of the respondents (27.3%) is the fact that patients do not often understand much information that should go into informed consent and this may retard the consent process. A study has confirmed this fact by showing that a considerable portion of the Nigerian population lacks basic reading and writing skills and may therefore find it difficult to understand the information provided by medical personnel[14]. Another study shows that less educated people are less likely to be satisfied with the explanations that come with consent taking. A plausible solution, as identified in some studies, is the need for consultation with loved ones of the patient, which will help understanding and eliminate anxiety[15,16].
Across the geo-political regions, there were no significant differences in knowledge about legislation in Nigeria (P = 0.214), though the North East, South East, and South West appeared to have more endoscopists with more information. This informs the fact that education about legislation on consent should be carried out nationwide.
In conclusion, consent taking in endoscopy is suboptimal in Nigeria. Steps must be taken to educate clinical personnel on consent taking in endoscopy with respect to implementation of standardized consent forms and, development of audio-visual aids, as this would improve patient care and safety as well as improve patient outcomes. A reform of the legislation involving the consent process in Nigeria, as well as subsequent enforcement of legal frameworks, is necessary to cater to peculiarities like culture, literacy rate, and paucity of medical personnel.
We acknowledge all members of the Gastro Consortium of Nigeria for their input toward the actualization of this manuscript.
| 1. | Mohanaruban A, Flanders L, Dor R. Consent in the endoscopy department. Frontline Gastroenterol. 2014;5:291-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 2. | Fitzgerald DW, Marotte C, Verdier RI, Johnson WD Jr, Pape JW. Comprehension during informed consent in a less-developed country. Lancet. 2002;360:1301-1302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 109] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 3. | Everett SM, Triantafyllou K, Hassan C, Mergener K, Tham TC, Almeida N, Antonelli G, Axon A, Bisschops R, Bretthauer M, Costil V, Foroutan F, Gauci J, Hritz I, Messmann H, Pellisé M, Roelandt P, Seicean A, Tziatzios G, Voiosu A, Gralnek IM. Informed consent for endoscopic procedures: European Society of Gastrointestinal Endoscopy (ESGE) Position Statement. Endoscopy. 2023;55:952-966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 4. | ASGE Standards of Practice Committee; Storm AC, Fishman DS, Buxbaum JL, Coelho-Prabhu N, Al-Haddad MA, Amateau SK, Calderwood AH, DiMaio CJ, Elhanafi SE, Forbes N, Fujii-Lau LL, Jue TL, Kohli DR, Kwon RS, Law JK, Pawa S, Thosani NC, Wani S, Qumseya BJ. American Society for Gastrointestinal Endoscopy guideline on informed consent for GI endoscopic procedures. Gastrointest Endosc. 2022;95:207-215.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 5. | Burr NE, Penman ID, Griffiths H, Axon A, Everett SM. Individualised consent for endoscopy: update on the 2016 BSG guidelines. Frontline Gastroenterol. 2023;14:273-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 6. | Lawal YZ, Garba ES, Ogirima MO, Dahiru IL, Maitama MI, Abubakar K. The doctrine of informed consent in surgical practice. Ann Afr Med. 2011;10:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 7. | Ezeome ER, Marshall PA. Informed consent practices in Nigeria. Dev World Bioeth. 2009;9:138-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 8. | Ezeuko MI. Nigerian laws on informed consent before a surgical procedure. Med Leg J. 2019;87:185-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Ogundiran TO, Adebamowo CA. Surgeons' opinions and practice of informed consent in Nigeria. J Med Ethics. 2010;36:741-745. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 10. | Ezeome ER, Chuke PI, Ezeome IV. Contents and readability of currently used surgical/procedure informed consent forms in Nigerian tertiary health institutions. Niger J Clin Pract. 2011;14:311-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 11. | Nzaumvila D, Ntotolo P, Govender I, Lukanu P, Niati JL, Sanduku D, Bongongo T. Knowledge and practices of seeking informed consent for medical examinations and procedures by health workers in the Democratic Republic of Congo. Afr Health Sci. 2021;21:478-488. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | Fadare JO, Desalu OO, Jemilohun AC, Babatunde OA. Knowledge of medical ethics among Nigerian medical doctors. Niger Med J. 2012;53:226-230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | Balogun SA, Ubom AE, Adesunkanmi AO, Ugowe OJ, Idowu AO, Mogaji IK, Nwigwe NC, Kolawole OJ, Nwebo EE, Sanusi AA, Odedeyi AA, Ogunrinde OV, Adedayo OO, Ndegbu CU, Ojo AS, Anele CO, Ogunjide OE, Olasehinde O, Awowole IO, Ijarotimi OA, Komolafe EO. Nigerian resident doctors' work schedule: A national study. Niger J Clin Pract. 2022;25:548-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 14. | Agu KA, Obi EI, Eze BI, Okenwa WO. Attitude towards informed consent practice in a developing country: a community-based assessment of the role of educational status. BMC Med Ethics. 2014;15:77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 15. | Osime OC, Okojie O, Osadolor F, Mohammed S. Current practices and medico-legal aspects of pre-operative consent. East Afr Med J. 2004;81:331-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 16. | Marshall PA. Informed consent in international health research. J Empir Res Hum Res Ethics. 2006;1:25-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 4.9] [Reference Citation Analysis (0)] |