Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.116148
Revised: November 29, 2025
Accepted: January 6, 2026
Published online: March 25, 2026
Processing time: 130 Days and 20.8 Hours
Glomerulonephritis (GN) is a major cause of chronic kidney disease and end-stage renal disease. Renal biopsy remains the gold standard for assessing interstitial fibrosis and tubular atrophy (IF/TA), but it is invasive and unsuitable for re
To evaluate renal elasticity using SWE in patients with GN and correlate SWE values with histopathology and serum transforming growth factor-β1 (TGF-β1) levels.
This prospective study included 201 consecutive adults with suspected GN undergoing renal biopsy. SWE was performed using a standardized protocol (6 cortical measurements per kidney). Histopathological IF/TA grade served as the reference standard. Serum TGF-β1 was measured using ELISA. Receiver operating characteristic analysis, analysis of variance, and Pearson correlation were applied. Additionally, 53 patients underwent follow-up SWE after 3-6 months.
Patients were classified as: No fibrosis (n = 22), mild (n = 96), moderate (n = 74), and marked fibrosis (n = 9). Mean SWE values increased progressively with IF/TA severity: 13.38 ± 1.36 kPa, 21.66 ± 4.49 kPa, 28.57 ± 4.91 kPa, and 40.31 ± 5.06 kPa, respectively (P < 0.001). SWE accurately detected any fibrosis (area under the curve = 0.986); an optimal cutoff of 13.98 kPa achieved 98% sensitivity and 73% specificity. Serum TGF-β1 levels showed an increasing trend across groups but were not statistically significant (P = 1.0). Etiology-wise, diabetic nephropathy exhibited the highest stiffness, while minimal change disease showed values comparable to the no-fibrosis group. Follow-up SWE revealed a small but statistically significant increase (23.48 ± 6.83 vs 24.04 ± 6.58 kPa, P < 0.001).
SWE correlates strongly with histological fibrosis in GN, outperforming conventional ultrasound and serum TGF-β1. It may serve as a non-invasive surrogate marker for moderate-to-marked fibrosis and assist in minimizing unnecessary biopsies. SWE also demonstrates potential for monitoring disease progression.
Core Tip: This research explored a new, painless ultrasound method that measures kidney stiffness. In patients with kidney inflammation (glomerulonephritis), we found that increased kidney stiffness directly corresponds to more scarring inside the organ. This technique proved to be highly accurate in detecting this scarring, which is a key factor in predicting kidney disease progression. Our findings suggest that this simple, non-invasive scan can be a valuable tool for doctors to identify scarring early, monitor the disease over time, and make more informed decisions about which patients truly need a biopsy.
- Citation: Prasad R, Verma R, Singh A, Behera MR, Yachha M, Kushwaha RS, Jain M, Agrawal V, Yadav P, Lal H. Renal elasticity assessment in patients of glomerulonephritis by shear-wave elastography and its correlation with histopathology and renal biomarkers. World J Nephrol 2026; 15(1): 116148
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/116148.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.116148
Glomerulonephritis (GN) remains a leading cause of chronic kidney disease (CKD) and end-stage renal disease (ESRD) worldwide[1]. The progression of GN is primarily driven by irreversible damage to the renal parenchyma, culminating in interstitial fibrosis and tubular atrophy (IF/TA). The extent of IF/TA is one of the strongest histological predictors of renal survival and long-term clinical outcomes[2]. Although renal biopsy is the gold standard for quantifying fibrosis, it is invasive, associated with bleeding complications[3], and limited by sampling error and inter-observer variability. Additionally, repeated biopsies for monitoring are impractical, highlighting the need for reliable, non-invasive markers of renal fibrosis.
Advances in ultrasound elastography have created opportunities for real-time assessment of tissue stiffness. Shear-wave elastography (SWE) uses acoustic radiation force to generate shear waves whose propagation provides a quan
Despite these advances, few studies have examined SWE exclusively in GN populations, where dynamic monitoring of fibrosis progression and treatment response is clinically important. Furthermore, the relationship between SWE, histopathological IF/TA, and fibrosis-related biomarkers such as transforming growth factor-β1 (TGF-β1)-a key mediator of renal fibrogenesis-remains incompletely understood[11].
This prospective study evaluates SWE in a large cohort of GN patients and correlates stiffness values with histopathological IF/TA grades and serum TGF-β1 levels. It also examines SWE differences across GN etiologies and provides follow-up data to explore SWE as a candidate tool for longitudinal fibrosis monitoring.
This prospective observational study was conducted at a tertiary academic medical center after approval from the Institutional Ethics Committee (IEC 2021-52-MD-EXP-36). From November 2020 to July 2022, 201 consecutive adults with clinically suspected GN undergoing renal biopsy were enrolled. Written informed consent was obtained.
Inclusion criteria: Age ≥ 18 years. Clinical suspicion of GN based on hematuria, proteinuria, or renal dysfunction. Planned renal biopsy.
Exclusion criteria: Body mass index (BMI) > 35 kg/m²; skin-to-kidney distance > 4 cm; prior renal interventions causing focal fibrosis; refusal to consent. Patients represented both acute and chronic GN presentations, including acute GN flares, acute kidney injury on GN, crescentic GN/rapidly progressive glomerulonephritis, lupus nephritis, anti-neutrophil cytoplasmic antibodies (ANCA)-associated vasculitis, and chronic parenchymal GN. Hypertension and diabetes, where present, were recorded.
All ultrasound examinations were performed using an Aixplorer® ultrasound system (SuperSonic Imagine, France) with a convex XC6-1 probe. The standardized protocol included: B-mode ultrasonography: Kidney length, cortical thickness, echotexture. Color Doppler: Resistive index and peak systolic velocity. SWE: Stiffness measurements were obtained from the renal cortex in upper, mid, and lower poles. Two measurements per pole were acquired, totaling six per kidney. A 6-mm Q-Box was placed in homogenous cortical regions during breath-hold. The mean of six values per patient was used for analysis. Elasticity was recorded in kPa.
Ultrasound-guided renal biopsy was performed using automated firing needles. Specimens were evaluated by experienced renal pathologists. IF/TA was graded as: Grade 0 (no fibrosis): < 5%; grade I (mild): 5%-25%; grade II (moderate): 26%-50%; grade III (marked): > 50%. GN etiologies [immunoglobulin A (IgA), focal and segmental glomerulosclerosis (FSGS), membranous, lupus, diabetic nephropathy, membranoproliferative glomerulonephritis (MPGN), ANCA vasculitis, anti-glomerular basement membrane (anti-GBM) disease, monoclonal gammopathies of renal significance (MGRS), multiple carboxylase deficiency] were recorded.
Serum TGF-β1 was quantified using a human TGF-β1 DuoSet® ELISA (R&D systems).
A subset of 53 patients underwent repeat SWE after 3-6 months to evaluate elasticity changes over time.
Continuous variables were expressed as mean ± SD. Differences across IF/TA groups were compared using one-way analysis of variance. Receiver operating characteristic (ROC) curve analysis assessed SWE performance in detecting any fibrosis (grade I-III vs grade 0). Pearson correlation evaluated relationships between SWE and continuous variables. Paired t-tests compared baseline and follow-up SWE. A P value < 0.05 was considered significant.
The study population comprised 201 patients with a mean age of 39.70 ± 15.58 years, including 129 (64.2%) males and 72 (35.8%) females. The patients were stratified into four groups based on histopathological assessment of IF/TA: No fibrosis (group A, n = 22), mild fibrosis (group B, n = 96), moderate fibrosis (group C, n = 74), and marked fibrosis (group D, n = 9). The groups were well-matched at baseline, with no statistically significant differences in age, gender, BMI, or serum creatinine levels (P > 0.05 for all), minimizing potential confounding effects on elastography measurements.
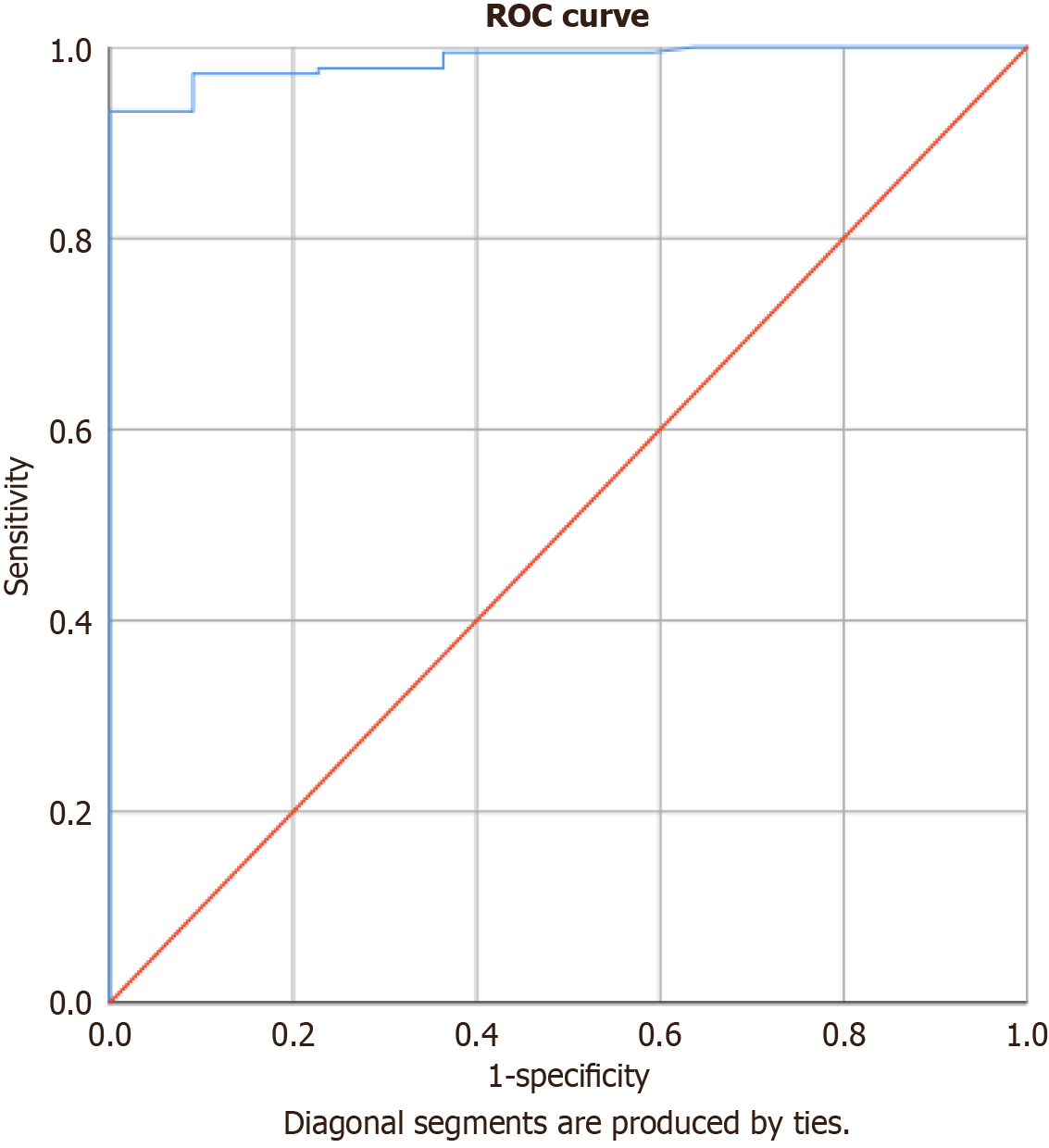
The central finding of this study was a strong, stepwise increase in renal cortical stiffness with advancing IF/TA grade. The mean SWE values demonstrated a highly significant progressive increase across the groups (P < 0.001) (Table 1, Figure 1). ROC curve analysis was performed to evaluate the diagnostic efficacy of SWE. For distinguishing any fibrosis (groups B-D) from no fibrosis (group A), the area under the curve (AUC) was 0.986 (Figure 2). A cutoff value of 13.98 kPa yielded a sensitivity of 98% and a specificity of 73%. These findings indicate that SWE reliably differentiates normal from fibrotic renal parenchyma and can quantify fibrosis severity. Representative examples of SWE findings and their direct histopathological correlation are illustrated in Figure 3.
| IF/TA grade | Mean SWE (kPa) | SD | Mean SWE (m/s) | SD |
| No fibrosis (A) | 13.35 | 1.37 | 2.09 | 0.11 |
| Mild fibrosis (B) | 21.66 | 4.50 | 2.66 | 0.28 |
| Moderate fibrosis (C) | 28.57 | 4.91 | 3.06 | 0.28 |
| Marked fibrosis (D) | 40.31 | 5.06 | 3.63 | 0.24 |
| P value | < 0.001 | < 0.001 |
A wide spectrum of glomerular diseases was represented in the cohort. IgA nephropathy was the most common etiology, accounting for 21.4% (43/201) of cases, followed by FSGS 13.9% (28/201), membranous nephropathy 12.9% (26/201), and lupus nephritis 10.9% (22/201). Diabetic nephropathy comprised 9.0% (18/201) of the cohort, while MPGN accounted for 5.5% (11/201), ANCA-associated vasculitis 4.0% (8/201), and anti-GBM disease 1.5% (3/201). MGRS/myeloma kidney was identified in 2.0% (4/201), and minimal change disease in 7.0% (14/201). The remaining 11.9% (24/201) were categorized as “other” or mixed GN patterns. Diabetic nephropathy, MPGN, and advanced lupus nephritis had the highest stiffness values, reflecting their greater fibrotic burden. Intermediate stiffness values were noted in IgA nephropathy, FSGS, and membranous nephropathy. Minimal change disease, being structurally preserved, showed values close to the no-fibrosis group. These patterns support the physiological link between pathological remodeling and SWE stiffness.
Serum TGF-β1 levels showed an upward trend from no fibrosis to marked fibrosis; however, the differences did not reach statistical significance (P = 1.0) (Table 2). Subsequently, ROC analysis for TGF-β1 as a diagnostic tool for fibrosis showed poor performance (AUC = 0.521, P = 0.817).
| IF/TA grade | Mean TGF-β1 (pg/mL) | SD |
| No fibrosis (A) | 11918.18 | 7636.16 |
| Mild fibrosis (B) | 11100.73 | 6089.92 |
| Moderate fibrosis (C) | 12433.00 | 6800.01 |
| Marked fibrosis (D) | 15438.10 | 7647.07 |
| P value | 1.0 |
A subset of 53 patients was available for follow-up examination between 3 months to 6 months. In this cohort, a small but statistically significant increase in mean SWE values was observed, from a baseline of 23.48 ± 6.83 kPa to 24.04 ± 6.58 kPa at follow-up (P < 0.001). In contrast, conventional B-mode ultrasound parameters (kidney dimensions, cortical thickness) and Doppler indices (Resistive Index, Peak Systolic Velocity) showed no significant changes over the same period.
Interstitial fibrosis is the final common pathway of most forms of chronic kidney injury and is a key determinant of progression to ESRD. In GN, assessing fibrosis accurately and non-invasively is particularly important due to the heterogeneous disease mechanisms, dynamic disease activity, and need for longitudinal monitoring. This study demonstrates that SWE provides a robust and reproducible measure of renal cortical stiffness that correlates strongly with histological IF/TA grades.
The most compelling observation is the clear stepwise increase in SWE values across the four IF/TA categories, from 13.38 kPa in non-fibrotic kidneys to 40.31 kPa in those with marked fibrosis. This progression aligns closely with the biological expectation that extracellular matrix deposition, including cross-linked collagen, increases tissue stiffness. These findings are consistent with earlier elastography literature, including the pioneering work of Samir et al[12] and subsequent studies demonstrating increased stiffness in fibrotic renal tissue[8-10]. However, most previous studies examined mixed CKD etiologies. Our work provides the largest GN-exclusive dataset to date, offering more disease-specific insight.
A key strength of this study is the detailed comparison of SWE values across GN etiologies. Diabetic nephropathy, MPGN, and advanced lupus nephritis demonstrated the highest stiffness values, reflecting the aggressive fibrotic remodeling common in these conditions. In contrast, minimal change disease, which often retains preserved parenchymal architecture, showed SWE values similar to the no-fibrosis group. These findings underscore SWE’s pathophysiological relevance and its ability to discriminate GN subtypes based on their fibrotic burden.
Unlike earlier studies, this work provides several novel contributions, including the largest prospective GN-specific cohort to date (n = 201), a comprehensive four-grade IF/TA comparison rather than a simple binary grouping, and the integration of serum TGF-β1 measurements to add a molecular dimension to fibrosis assessment. It also offers detailed etiology-wise SWE characterization-an aspect not previously explored-and incorporates follow-up SWE measurements that demonstrate the technique’s sensitivity to temporal changes in renal stiffness. The ROC analysis yielded an AUC of 0.986 with an optimal cutoff of 13.98 kPa, indicating excellent discriminatory performance. These findings suggest that SWE could complement or, in selected cases, reduce the need for renal biopsy when fibrosis assessment is the primary clinical concern, particularly in patients who are elderly, have significant comorbidities, possess a solitary kidney, or are at elevated bleeding risk.
An important discussion point is the conflicting literature regarding elastography in advanced CKD. Some ARFI-based studies reported lower stiffness in advanced disease due to reduced perfusion and loss of renal turgor[6,7]. We addressed this by emphasizing that our cohort predominantly represents GN, where early fibrosis, inflammation, and tissue remodeling dominate. These processes increase stiffness, and the perfusion-driven “softening effect” described in late CKD may not significantly influence this population. The consistency of rising stiffness values even in marked fibrosis supports this interpretation.
The lack of statistical significance in TGF-β1 values (P = 1.0) also warranted clarification. Although mean levels trended upward with increasing IF/TA, the large intra-group variance, combined with the systemic nature of TGF-β1 regulation, limited its discriminatory capacity. This reinforces the advantage of SWE as an organ-specific measurement, directly reflecting tissue-level mechanical changes. Biomarkers like TGF-β1 may be more useful when combined with imaging tools rather than as standalone fibrosis markers.
The follow-up SWE findings, while showing only a small absolute change, demonstrated statistically significant progression. This suggests that SWE is sensitive enough to detect early directional changes in parenchymal stiffness over short intervals. Although the clinical relevance of a 0.56-kPa increase requires larger longitudinal studies, it highlights SWE’s potential utility for monitoring disease activity or response to therapy. Conventional ultrasound and Doppler parameters remained unchanged, emphasizing SWE’s superior sensitivity to subtle structural alterations.
There are limitations to acknowledge. The group D sample size was small, reflecting the natural distribution of marked fibrosis in biopsy cohorts. SWE was performed by a single radiologist, preventing assessment of inter-observer variability-though this also reduced acquisition variability. Additionally, renal SWE lacks global standardization in terms of acquisition protocols, measurement depths, and machine-specific variability. Larger multicenter studies are needed to address these issues. Despite these limitations, the study provides strong evidence supporting the clinical value of renal SWE in GN.
Overall, this work demonstrates that SWE is a powerful, non-invasive tool for assessing renal fibrosis in GN. It shows strong correlation with histology, differentiates GN etiologies based on their fibrotic burden, and exhibits potential for longitudinal monitoring. With further validation and standardization, SWE could become an integral component of the nephrologist’s diagnostic armamentarium-reducing reliance on biopsy, improving risk stratification, and enhancing personalized GN management.
In conclusion, this study demonstrates that SWE is a reliable, non-invasive tool that correlates strongly with histological fibrosis in GN. SWE accurately differentiates IF/TA grades, reflects etiological differences, and detects subtle longitudinal changes better than conventional ultrasonography or serum TGF-β1. Its excellent diagnostic performance suggests an important complementary role to renal biopsy, particularly in patients where biopsy poses higher risk or limited benefit. With further validation and standardization, SWE has the potential to become an integral component of routine GN evaluation, guiding clinical decision-making and reducing dependence on invasive procedures.
| 1. | Anders HJ, Kitching AR, Leung N, Romagnani P. Glomerulonephritis: immunopathogenesis and immunotherapy. Nat Rev Immunol. 2023;23:453-471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 123] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 2. | Nath KA. Tubulointerstitial changes as a major determinant in the progression of renal damage. Am J Kidney Dis. 1992;20:1-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 790] [Cited by in RCA: 772] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 3. | Trajceska L, Severova-Andreevska G, Dzekova-Vidimliski P, Nikolov I, Selim G, Spasovski G, Rambabova-Busletik I, Ristovska V, Grcevska L, Sikole A. Complications and Risks of Percutaneous Renal Biopsy. Open Access Maced J Med Sci. 2019;7:992-995. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 4. | Sigrist RMS, Liau J, Kaffas AE, Chammas MC, Willmann JK. Ultrasound Elastography: Review of Techniques and Clinical Applications. Theranostics. 2017;7:1303-1329. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1574] [Cited by in RCA: 1261] [Article Influence: 140.1] [Reference Citation Analysis (1)] |
| 5. | Gennisson JL, Deffieux T, Fink M, Tanter M. Ultrasound elastography: principles and techniques. Diagn Interv Imaging. 2013;94:487-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 865] [Cited by in RCA: 654] [Article Influence: 50.3] [Reference Citation Analysis (0)] |
| 6. | Asano K, Ogata A, Tanaka K, Ide Y, Sankoda A, Kawakita C, Nishikawa M, Ohmori K, Kinomura M, Shimada N, Fukushima M. Acoustic radiation force impulse elastography of the kidneys: is shear wave velocity affected by tissue fibrosis or renal blood flow? J Ultrasound Med. 2014;33:793-801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 113] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 7. | Bob F, Bota S, Sporea I, Sirli R, Popescu A, Schiller A. Relationship between the estimated glomerular filtration rate and kidney shear wave speed values assessed by acoustic radiation force impulse elastography: a pilot study. J Ultrasound Med. 2015;34:649-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 8. | Leong SS, Wong JHD, Md Shah MN, Vijayananthan A, Jalalonmuhali M, Chow TK, Sharif NHM, Ng KH. Shear wave elastography accurately detects chronic changes in renal histopathology. Nephrology (Carlton). 2021;26:38-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 9. | Ng KH, Wong JHD, Leong SS. Shear wave elastography in chronic kidney disease - the physics and clinical application. Phys Eng Sci Med. 2024;47:17-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 10. | Lakshmana SK, Koteshwar P, Kamath T. Value of Shear Wave Elastography in the Evaluation of Chronic Kidney Disease. Int J Nephrol Renovasc Dis. 2024;17:307-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 11. | Meng XM, Tang PM, Li J, Lan HY. TGF-β/Smad signaling in renal fibrosis. Front Physiol. 2015;6:82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 562] [Cited by in RCA: 578] [Article Influence: 52.5] [Reference Citation Analysis (0)] |
| 12. | Samir AE, Allegretti AS, Zhu Q, Dhyani M, Anvari A, Sullivan DA, Trottier CA, Dougherty S, Williams WW, Babitt JL, Wenger J, Thadhani RI, Lin HY. Shear wave elastography in chronic kidney disease: a pilot experience in native kidneys. BMC Nephrol. 2015;16:119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 98] [Article Influence: 8.9] [Reference Citation Analysis (0)] |