Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.116620
Revised: December 1, 2025
Accepted: December 23, 2025
Published online: March 25, 2026
Processing time: 118 Days and 15.9 Hours
Focal segmental glomerulosclerosis (FSGS) represents a major cause of nephrotic syndrome, with glucocorticoids constituting first-line therapy. When patients develop steroid dependence or resistance, calcineurin inhibitors (CNIs) serve as effective alternatives. However, CNI dependence or resistance may emerge, and prolonged use of either steroid or CNI therapy carries substantial toxicity risks. These limitations create an unmet need for safer treatments capable of main
The patient initially presented with acute anasarca, nephrotic-range proteinuria (5.6 g/24 hours), hypoalbuminemia (2.1 g/dL), and preserved renal function. She experienced repeated relapses after corticosteroid and cyclosporine tapering, ul
This case contributes to the growing evidence supporting rituximab’s therapeutic potential in adult FSGS, while highlighting the need for randomized controlled trials to establish its efficacy and optimal use in this challenging patient popula
Core Tip: This case report describes a patient with steroid- and calcineurin inhibitor-dependent primary focal segmental glomerulosclerosis (FSGS) who achieved complete and sustained remission for five years following a two-dose regimen of rituximab (RTX). The case highlights the potential of RTX as a steroid- and calcineurin inhibitor-sparing agent in adult FSGS, particularly in patients with significant toxicity or dependence on standard immunosuppressive therapy. The extended follow-up adds valuable insight into the long-term efficacy and safety of RTX in this challenging population.
- Citation: Moura AF, Rocha PN, Moura-Landim D, Moura Jr JA, Moura-Neto JA. Five-year follow-up sustained remission with rituximab in a patient with focal segmental glomerulosclerosis: A case report. World J Nephrol 2026; 15(1): 116620
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/116620.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.116620
Focal segmental glomerulosclerosis (FSGS) is a common cause of nephrotic syndrome, histologically characterized by segmental areas of sclerosis affecting at least one glomerulus in a kidney biopsy specimen[1]. Although its global pre
In patients with primary FSGS, an additional classification based on therapeutic response has been proposed[1]: (1) Steroid-resistant primary FSGS; (2) Steroid-dependent primary FSGS; (3) CNI-resistant primary FSGS; and (4) CNI-dependent primary FSGS. Steroid resistance is defined as persistent nephrotic-range proteinuria (> 3.5 g/day) with ≤ 50% reduction from baseline after 16 weeks of high-dose oral glucocorticoid therapy[1]. Steroid dependence refers to a relapse occurring during glucocorticoid treatment or within two weeks of its discontinuation[1]. CNI resistance is defined by per
A deeper understanding of FSGS pathophysiology is likely essential for the development of new therapeutic strategies. Rituximab (RTX) may also exert direct podocyte-protective effects by interacting with SMPDL-3b and stabilizing the actin cytoskeleton, as demonstrated in experimental models[7]. This mechanistic hypothesis provides additional biological plausibility for its use in FSGS beyond simple B-cell depletion. To date, the most extensively studied treatments remain glucocorticoid-based regimens and CyA[6,8,9]. However, options are limited for patients with contraindications to glu
RTX, an anti-CD20 monoclonal antibody, has been increasingly used as a steroid-sparing agent in nephrotic syndrome[10]. Although its efficacy is well established in pediatric steroid-dependent forms, data in adults with FSGS remain li
A 34-year-old woman presented with progressive lower limb edema evolving into anasarca and foamy urine, consistent with nephrotic syndrome.
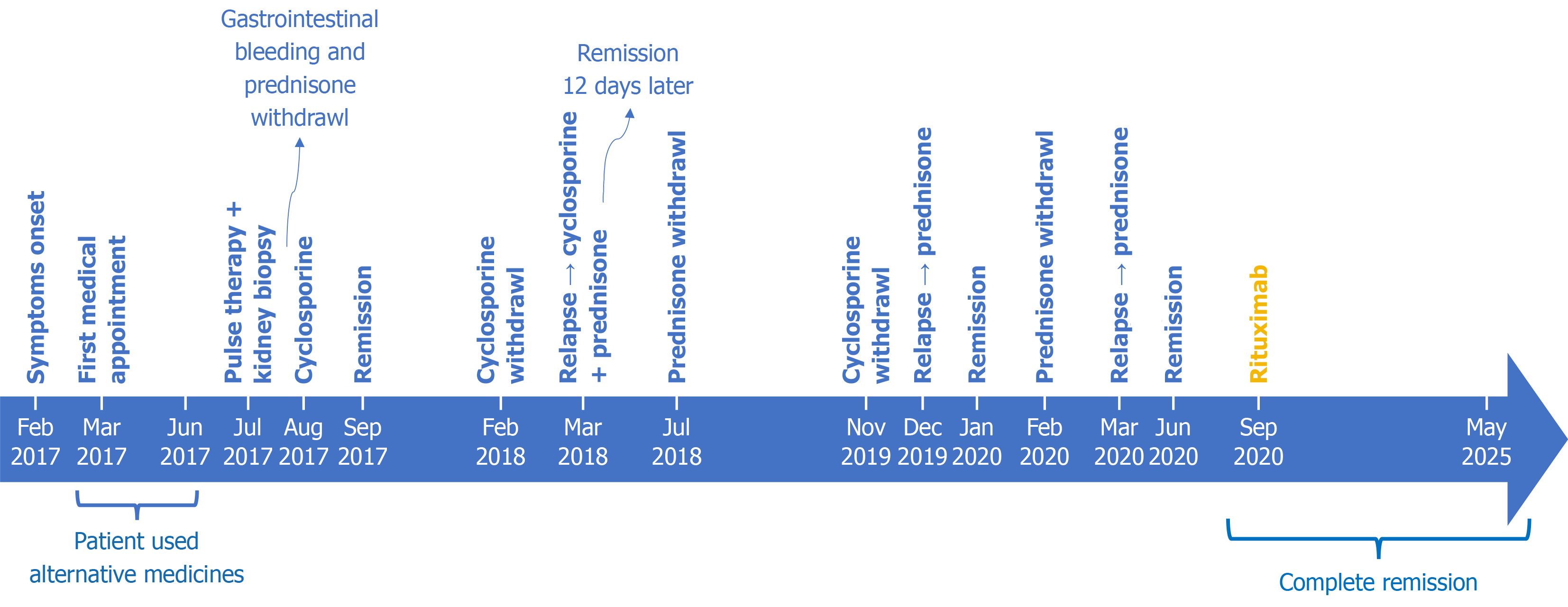
The symptoms began acutely in February 2017. Despite initial medical evaluation and a recommendation for kidney biopsy, the patient chose to postpone further investigations and follow-up. Over the subsequent three months, she self-treated with herbal remedies without clinical improvement.
In July 2017, due to persistent edema and worsening symptoms, she was admitted for empirical therapy with in
Following discontinuation of CyA, the patient relapsed two weeks later with nephrotic-range proteinuria (5587.7 mg/24 hours) and edema. Remission was again induced with CyA and low-dose prednisone (15 mg/day), which were conti
| March 2017 | July 2017 | August 2017 | September 2017 | January 2018 | March 2018 | April 2018 | June 2018 | July 2018 | May 2019 | November 2019 | December 2019 | January 2020 | May 2020 | June 2, 2020 | June 11, 2020 | September 2020 | July 2021 | May 2022 | May 2025 | |
| LDL-C (mg/dL) | 228 | 245 | 237.6 | 75.6 | 79.3 | 123.7 | 154.1 | 101 | 78 | 112 | 105 | 168 | 285 | 107 | 129.4 | 120.8 | 121.2 | 106 | 108.4 | 107 |
| Triglycerides (mg/dL) | 69 | 127 | 212.2 | 106.1 | 99.3 | 137.9 | 121 | 55 | 74 | 39 | 66 | 116 | 130 | 97 | 141.7 | 175.9 | 62.1 | 39 | 88 | 73 |
| Serum albumin (g/dL) | 2.6 | 2.1 | 3.3 | 4.7 | 3.9 | 2.4 | 4.5 | 4.6 | 4.7 | 4.4 | 2.6 | 4.2 | 4.1 | 3.9 | 4.4 | 4.7 | 4.4 | |||
| Urea (mg/dL) | 15 | 31 | 17.6 | 16.4 | 21.3 | 13.5 | 24 | 23 | 26 | 24 | 26 | 19 | 17 | 19.1 | 22.8 | 21.9 | 13 | 19.3 | 24 | |
| Creatinine (mg/dL) | 0.5 | 0.5 | 0.6 | 0.7 | 0.7 | 0.5 | 0.6 | 0.7 | 0.8 | 0.8 | 0.7 | 0.7 | 0.7 | 0.7 | 0.8 | 0.8 | 0.8 | 0.6 | 0.7 | 0.7 |
| UACR (mg/g) | 4100 | 4.8 | 2.9 | 15.1 | 60 | 2.0 | 6.2 | 1.4 | 5.0 | 1240 | 430 | 2 | 5 | 24 | 1.7 | |||||
| 24-hour proteinuria | 5757 mg/24 hours | 5581.7 mg/24 hours | 1524 mg/24 hours | 1272 mg/24 hours | 6645 mg/24 hours | 112.2 mg/24 hours | ||||||||||||||
| Urine analysis | Protein 1+; hemoglobin 3+ | Protein 1+; hemoglobin 3+ | Protein neg; hemoglobin neg | Protein neg; hemoglobin neg | Protein neg; hemoglobin neg | Protein 2+; hemoglobin neg | Protein neg; hemoglobin neg | Protein neg; hemoglobin neg | ||||||||||||
| Cyclosporine serum level (ng/mL) | 104 | 111 | 91 | 126 |
The patient had no known prior chronic illnesses, comorbidities, or relevant previous hospitalizations before the onset of nephrotic syndrome.
No personal history of smoking, alcohol or drug use. No known family history of kidney disease, autoimmune disorders, or hereditary conditions.
At the time of initial hospital admission in July 2017, the patient presented with significant anasarca, including periorbital and lower limb edema. Blood pressure and heart rate were within normal limits. No signs of systemic infection or or
Initial workup revealed nephrotic-range proteinuria, hypoalbuminemia, and hypercholesterolemia with preserved renal function (Table 1). Serologies for viral hepatitis and human immunodeficiency virus were negative. Autoimmune sc
Renal ultrasound showed normal-sized kidneys with preserved corticomedullary differentiation and no structural abnor
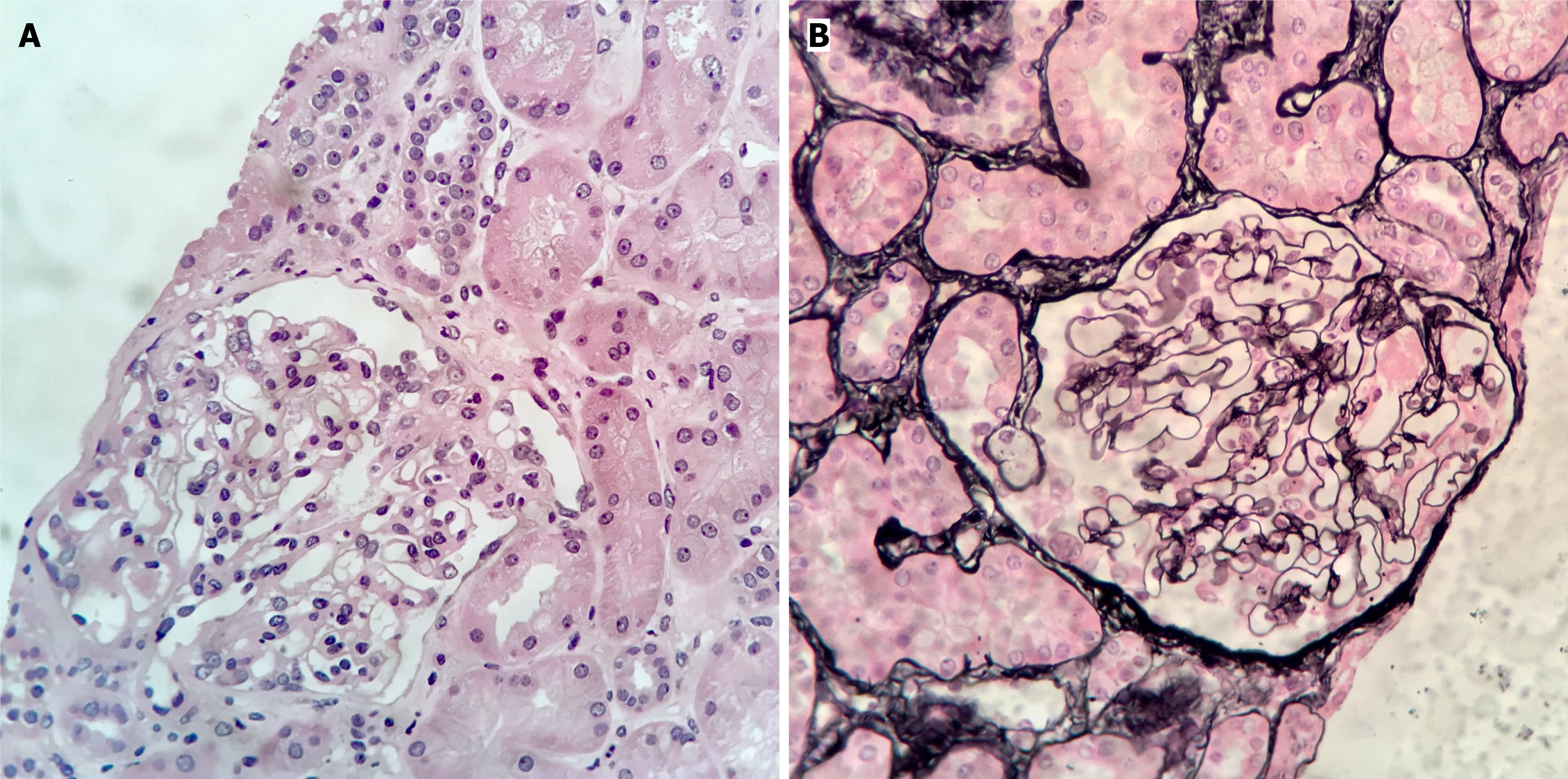
Kidney biopsy performed during hospitalization revealed segmental sclerosis and podocyte hypertrophy in a subset of glomeruli, consistent with a diagnosis of FSGS. Light microscopy demonstrated a glomerulus with flocculocapsular adhesion at the hilar pole and podocyte hypertrophy, suggestive of tip lesion variant, as seen with Grocott-Methenamine-Silver staining and hematoxylin-eosin staining at 400× magnification (Figure 2). No significant interstitial fibrosis or tubular atrophy was noted. While electron microscopy and immunofluorescence were not available due to logistical limi
Primary FSGS, likely tip lesion variant, steroid- and CNI-dependent.
The patient was initially treated with high-dose steroids, then switched to CyA due to gastrointestinal complications. After several relapses under steroid and CNI therapy, the patient received two 1 g infusions of RTX 14 days apart in 2019. No maintenance immunosuppression was used after that. Prednisone was tapered over 8 weeks following RTX infusion.
The patient achieved complete remission after RTX administration and has maintained sustained remission without relapse or additional immunosuppressive therapy for five years.
This case provides an uncommon and informative contribution due to several distinguishing features. The patient is a young adult woman with no comorbidities, who presented with severe nephrotic syndrome, experienced life-threatening corticosteroid-associated gastrointestinal bleeding, and demonstrated both steroid- and CNI-dependence. Notably, complete remission was achieved and sustained for five years after a single course of RTX without additional immu
Glucocorticoids are the cornerstone of initial FSGS therapy, supported by observational studies demonstrating their efficacy in inducing proteinuria remission and a relatively low incidence of severe adverse effects, even at high doses[1,6,8]. However, high-dose steroids may cause serious complications, precluding their use in some patients. Similarly, pro
Most studies on CNIs in FSGS are observational, primarily focusing on glucocorticoid-dependent or glucocorticoid-resistant primary FSGS[9,12-14]. The evidence supporting CyA as an alternative therapy remains of low quality. Alth
Prolonged use of immunosuppressive agents, even with therapeutic drug monitoring, carries significant risks, in
For patients with previously steroid-sensitive primary FSGS, such as the case reported here, KDIGO recommends management similar to minimal change disease (MCD)[1]. RTX is a therapeutic option, showing acceptable efficacy in pe
The efficacy of RTX in adults with nephrotic syndrome remains uncertain. A meta-analysis of 14 observational studies involving patients ≥ 18 years with FSGS or MCD reported a 53.6% remission rate among those with FSGS receiving RTX[10].
An Italian study of 31 patients with steroid-resistant or steroid-dependent primary FSGS treated with RTX and followed for a mean of 17 months reported remission rates of 39%, 52%, and 42% at 3 months, 6 months, and 12 months, respectively, with higher remission rates at 6 months in steroid-dependent patients[11]. Another study of 14 FSGS pa
The mechanism by which RTX induces remission in FSGS and MCD remains unclear. RTX, a monoclonal antibody targeting CD20-positive B lymphocytes, is approved for B cell-mediated malignancies and connective tissue diseases but lacks formal approval for nephrotic syndrome. Its high cost further limits global access. While the mechanism of RTX-induced remission in our patient remains unclear, several hypotheses may explain the sustained response. RTX’s deple
Emerging data suggest that the therapeutic effects of RTX in FSGS may extend beyond B-cell depletion. In experimental models, RTX has been shown to interact directly with podocytes through SMPDL-3b and acid sphingomyelinase signaling pathways, stabilizing the actin cytoskeleton and promoting podocyte survival[7]. Moreover, a subset of patients may harbor circulating permeability factors or autoantibodies that are neutralized or modulated by RTX, indirectly preventing podocyte injury. Recent immunophenotyping studies have proposed that certain B-cell subtypes, such as memory B cells and regulatory B cells, may correlate with treatment response in nephrotic syndrome[19]. Ongoing clinical trials-such as NCT04716231-are exploring the broader immunologic effects and clinical efficacy of RTX in glo
Emerging reports support the safe use of RTX[10,14,16]; however, robust studies specifically evaluating RTX in FSGS are lacking, with most data derived from MCD. Randomized controlled trials are needed to confirm RTX’s efficacy in adult FSGS patients, potentially supporting its inclusion in standard treatment protocols. Case reports with extended follow-up, like this study, provide valuable evidence of RTX’s therapeutic success and may advocate for its approval in FSGS treatment.
Despite its strengths, this report is subject to the inherent limitations of a single-case observation, including limited generalizability and the inability to infer causality. The absence of genetic testing and electron microscopy further res
Primary FSGS is a leading cause of nephrotic syndrome, typically managed with glucocorticoids due to their efficacy in achieving proteinuria remission and favorable safety profile. However, steroid- and CNI-dependent cases, like the one reported here, pose significant therapeutic challenges. This case demonstrates the successful use of two 1 g doses of RTX in a patient with steroid- and CNI-dependent primary FSGS, achieving complete remission sustained for five years. While RTX shows promise in reducing relapses in pediatric nephrotic syndrome, its efficacy in adults remains uncertain. Well-designed clinical trials are needed to validate RTX’s role in adult FSGS and expand therapeutic options for managing complex cases.
| 1. | Kidney Disease: Improving Global Outcomes (KDIGO) Glomerular Diseases Work Group. KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases. Kidney Int. 2021;100:S1-S276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1772] [Cited by in RCA: 1547] [Article Influence: 309.4] [Reference Citation Analysis (1)] |
| 2. | Haas M, Meehan SM, Karrison TG, Spargo BH. Changing etiologies of unexplained adult nephrotic syndrome: a comparison of renal biopsy findings from 1976-1979 and 1995-1997. Am J Kidney Dis. 1997;30:621-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 271] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 3. | Bahiense-Oliveira M, Saldanha LB, Mota EL, Penna DO, Barros RT, Romão-Junior JE. Primary glomerular diseases in Brazil (1979-1999): is the frequency of focal and segmental glomerulosclerosis increasing? Clin Nephrol. 2004;61:90-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 4. | Braden GL, Mulhern JG, O'Shea MH, Nash SV, Ucci AA Jr, Germain MJ. Changing incidence of glomerular diseases in adults. Am J Kidney Dis. 2000;35:878-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 153] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 5. | Goldszer RC, Sweet J, Cotran RS. Focal segmental glomerulosclerosis. Annu Rev Med. 1984;35:429-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 6. | Korbet SM. Treatment of primary FSGS in adults. J Am Soc Nephrol. 2012;23:1769-1776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 124] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 7. | Fornoni A, Sageshima J, Wei C, Merscher-Gomez S, Aguillon-Prada R, Jauregui AN, Li J, Mattiazzi A, Ciancio G, Chen L, Zilleruelo G, Abitbol C, Chandar J, Seeherunvong W, Ricordi C, Ikehata M, Rastaldi MP, Reiser J, Burke GW 3rd. Rituximab targets podocytes in recurrent focal segmental glomerulosclerosis. Sci Transl Med. 2011;3:85ra46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 466] [Cited by in RCA: 410] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 8. | Fernandez-Juarez G, Villacorta J, Ruiz-Roso G, Panizo N, Martinez-Marín I, Marco H, Arrizabalaga P, Díaz M, Perez-Gómez V, Vaca M, Rodríguez E, Cobelo C, Fernandez L, Avila A, Praga M, Quereda C, Ortiz A. Therapeutic variability in adult minimal change disease and focal segmental glomerulosclerosis. Clin Kidney J. 2016;9:381-386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 9. | Gorsane I, Helal I, Yacoub I, Hamida FB, Abderrahim E, Abdallah TB. Cyclosporine therapy in steroid-dependent or steroid-resistant idiopathic focal and segmental glomerulosclerosis. Saudi J Kidney Dis Transpl. 2016;27:958-965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 10. | Hansrivijit P, Cheungpasitporn W, Thongprayoon C, Ghahramani N. Rituximab therapy for focal segmental glomerulosclerosis and minimal change disease in adults: a systematic review and meta-analysis. BMC Nephrol. 2020;21:134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 49] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 11. | Tedesco M, Mescia F, Pisani I, Allinovi M, Casazza G, Del Vecchio L, Santostefano M, Cirillo L, Ferrario F, Esposito C, Esposito P, Santoro D, Lazzarin R, Rossi GM, Fiaccadori E, Ferrantelli A, Sinico RA, Cozzolino M, Gallieni M; Italian Society of Nephrology Immunopathology Working Group, Cirami L, Scolari F, Vaglio A, Alberici F. The Role of Rituximab in Primary Focal Segmental Glomerular Sclerosis of the Adult. Kidney Int Rep. 2022;7:1878-1886. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 12. | D'Agati VD, Fogo AB, Bruijn JA, Jennette JC. Pathologic classification of focal segmental glomerulosclerosis: a working proposal. Am J Kidney Dis. 2004;43:368-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 623] [Cited by in RCA: 499] [Article Influence: 22.7] [Reference Citation Analysis (3)] |
| 13. | Banfi G, Moriggi M, Sabadini E, Fellin G, D'Amico G, Ponticelli C. The impact of prolonged immunosuppression on the outcome of idiopathic focal-segmental glomerulosclerosis with nephrotic syndrome in adults. A collaborative retrospective study. Clin Nephrol. 1991;36:53-59. [PubMed] |
| 14. | Ponticelli C, Villa M, Banfi G, Cesana B, Pozzi C, Pani A, Passerini P, Farina M, Grassi C, Baroli A. Can prolonged treatment improve the prognosis in adults with focal segmental glomerulosclerosis? Am J Kidney Dis. 1999;34:618-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 83] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 15. | Ito S, Kamei K, Ogura M, Udagawa T, Fujinaga S, Saito M, Sako M, Iijima K. Survey of rituximab treatment for childhood-onset refractory nephrotic syndrome. Pediatr Nephrol. 2013;28:257-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 56] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 16. | Aslam A, Koirala A. Review of the Role of Rituximab in the Management of Adult Minimal Change Disease and Immune-Mediated Focal and Segmental Glomerulosclerosis. Glomerular Dis. 2023;3:211-219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 17. | Wang L, Yu L, Wang Y, Guo Y, Zhai Z, Tang L. Rituximab treatment of adults with primary focal segmental glomerulosclerosis. Sci Rep. 2023;13:6740. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 18. | Lan L, Lin Y, Yu B, Wang Y, Pan H, Wang H, Lou X, Lang X, Zhang Q, Jin L, Yang Y, Xiao L, Chen J, Han F. Efficacy of Rituximab for Minimal Change Disease and Focal Segmental Glomerulosclerosis with Frequently Relapsing or Steroid-Dependent Nephrotic Syndrome in Adults: A Chinese Multicenter Retrospective Study. Am J Nephrol. 2024;55:25-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 19. | Colucci M, Carsetti R, Cascioli S, Casiraghi F, Perna A, Ravà L, Ruggiero B, Emma F, Vivarelli M. B Cell Reconstitution after Rituximab Treatment in Idiopathic Nephrotic Syndrome. J Am Soc Nephrol. 2016;27:1811-1822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 179] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 20. |
|