Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.115933
Revised: December 17, 2025
Accepted: January 28, 2026
Published online: March 25, 2026
Processing time: 136 Days and 15 Hours
Patients with type 2 diabetes mellitus (T2DM) and chronic kidney disease (CKD) face high renal and car
To evaluate the real-world safety and effectiveness of finerenone in patients with T2DM and CKD across seven Latin American countries.
We conducted an observational, multicenter, retrospective cohort study based on real-world data in 347 patients with T2DM and CKD [urinary albumin-creatinine ratio (UACR) > 30 mg/g]. Patients received finerenone (10 mg or 20 mg daily), and clinical and laboratory parameters were evaluated at baseline and after six months of treatment.
At baseline, median values (interquartile range) were: Glycated hemoglobin A1c 7.6% (6.8%-8.1%); estimated glomerular filtration rate 39.0 mL/minute/1.73 m2 (30.0-50.0); UACR 345 mg/g (189-760); systolic blood pressure 143 mmHg (130-160); diastolic blood pressure 79 mmHg (70-82); and serum potassium 4.4 mmol/L (4.1-4.7). After six months, significant reductions were observed: Glycated hemoglobin A1c to 7.0% (6.5%-7.9%); UACR to 81 mg/g (28-167); systolic blood pressure to 130 mmHg (120-140); and diastolic blood pressure to 73 mmHg (70-80). Serum potassium increased to 4.7 mmol/L (4.3-5.0), while estimated glomerular filtration rate remained stable at 41.6 mL/minute/1.73 m2 (27.0-52.0).
In our cohort of patients with CKD associated with T2DM, finerenone proved to be an effective short-term therapy for reducing albuminuria, demonstrating very good tolerance and a low risk of hyperkalemia.
Core Tip: In a real-world observational study of 347 Latin American patients with type 2 diabetes mellitus and chronic kidney disease finerenone demonstrated significant short-term benefits. After six months, treatment led to major reductions in the urinary albumin-creatinine ratio and blood pressure. This efficacy was achieved with very good tolerance; the estimated glomerular filtration rate remained stable, and serum potassium increases were minimal, highlighting a low risk of hyperkalemia. This study provides crucial real-world evidence supporting finerenone’s safety and effectiveness.
- Citation: Arnedo RD, Polo VS, Garcia JB, Gutiérrez JF, Clason ER, Domínguez D, Rebutti GM, Mancia CM, López RT, Meza MR, López DT, Muñoz Zambrano JJ, Herrera EL, Dina-Batlle E, Terrones MC, de Moraes TP, Yánez TR, Osorio W, Cabeza AA, Rico-Fontalvo J. Efficacy and safety of finerenone in diabetic kidney disease: Latin American experience from FINDKDLATAM trial. World J Nephrol 2026; 15(1): 115933
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/115933.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.115933
Chronic kidney disease (CKD) occurs in approximately 40% of people with type 2 diabetes mellitus (T2DM)[1]. This is defined by the presence of an estimated glomerular filtration rate of less than 60 mL/minute/1.73 m2 and/or an albu
Several interrelated factors such as hyperglycemia, dyslipidemia, chronic arterial hypertension, activation of the renin angiotensin system, and inflammation have been implicated in the development of microvascular complications in DKD[8-11]. The active form of the mineralocorticoid receptor is associated with systemic inflammation and CKD progression[12-14]. Finerenone, a selective non-steroidal mineralocorticoid receptor antagonist has emerged as a therapeutic option in the management of DKD[15]. Its development represents a breakthrough in the fight against renal complications associated with T2DM. It has been evaluated in clinical studies such as FIDELIO-DKD and FIGARO-DKD, demonstrating significant reduction in the risk of renal disease progression and cardiovascular events in patients with T2DM and CKD with albuminuria[6,10]. These findings generated an important insight in its role not only in renal protection, but also as a prevention of cardiovascular complications, which is a significant benefit in patients with T2DM[16-18].
In the present study, we analyzed the effects of finerenone on blood pressure, glomerular filtration rate and changes in albuminuria/creatinuria ratio in a population of patients from Latin American countries with a diagnosis of CKD associated with T2DM.
An observational, multicenter study based on real-world data. Patients with a mean age between 55 years and 71 years. The objective of this study is to evaluate the efficacy and safety of the mineralocorticoid receptor antagonist finerenone in seven Latin American countries. All participants were informed in writing about the nature of this study and signed the informed consent form. The study was conducted in accordance with the standards of the Declaration of Helsinki. The Ethics Review Committee was the graduate school of the Faculty of Medical Sciences at the University of San Carlos in Guatemala. During this committee meeting, the ethical components, possible assessments in the use of patient infor
Inclusion criteria are adult patients older than 18 years, with a diagnosis of T2DM and CKD with a urinary albumin-creatinine ratio (UACR) > 30 mg/g, relevant history of cardiovascular disease and pharmacological interventions for diabetes-associated CKD.
Patients received finerenone in daily doses of 10 mg or 20 mg. Finerenone was prescribed according to standard clinical practice guidelines. The medication costs were covered by the patients’ health insurance providers or national health systems, reflecting a real-world setting rather than a sponsored clinical trial environment. This research received no external funding. Medical care, laboratory tests, and medication were covered by the standard of care/health insurance of the patients. The data collection and analysis were performed by the researchers as part of their academic and clinical duties without additional compensation. No comparison was made with other types of interventions, even though most patients were treated with other pharmacological groups [glucagon-like peptide (GLP)-1, sodium-glucose cotransporter 2, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers].
Several CKD-related variables were evaluated, including glycated hemoglobin A1c (HbA1c), estimated glomerular filtration rate (eGFR), albuminuria/creatinuria ratio, systolic blood pressure (SBP), diastolic blood pressure (DBP) and serum potassium, at baseline and at six months of treatment. The descriptive analysis of qualitative variables was per
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki, and it was approved by the local Ethics Committee and teaching reference of the Faculty of Medical Sciences of the University of San Carlos of Guatemala. The consent of the participants was not required for publication.
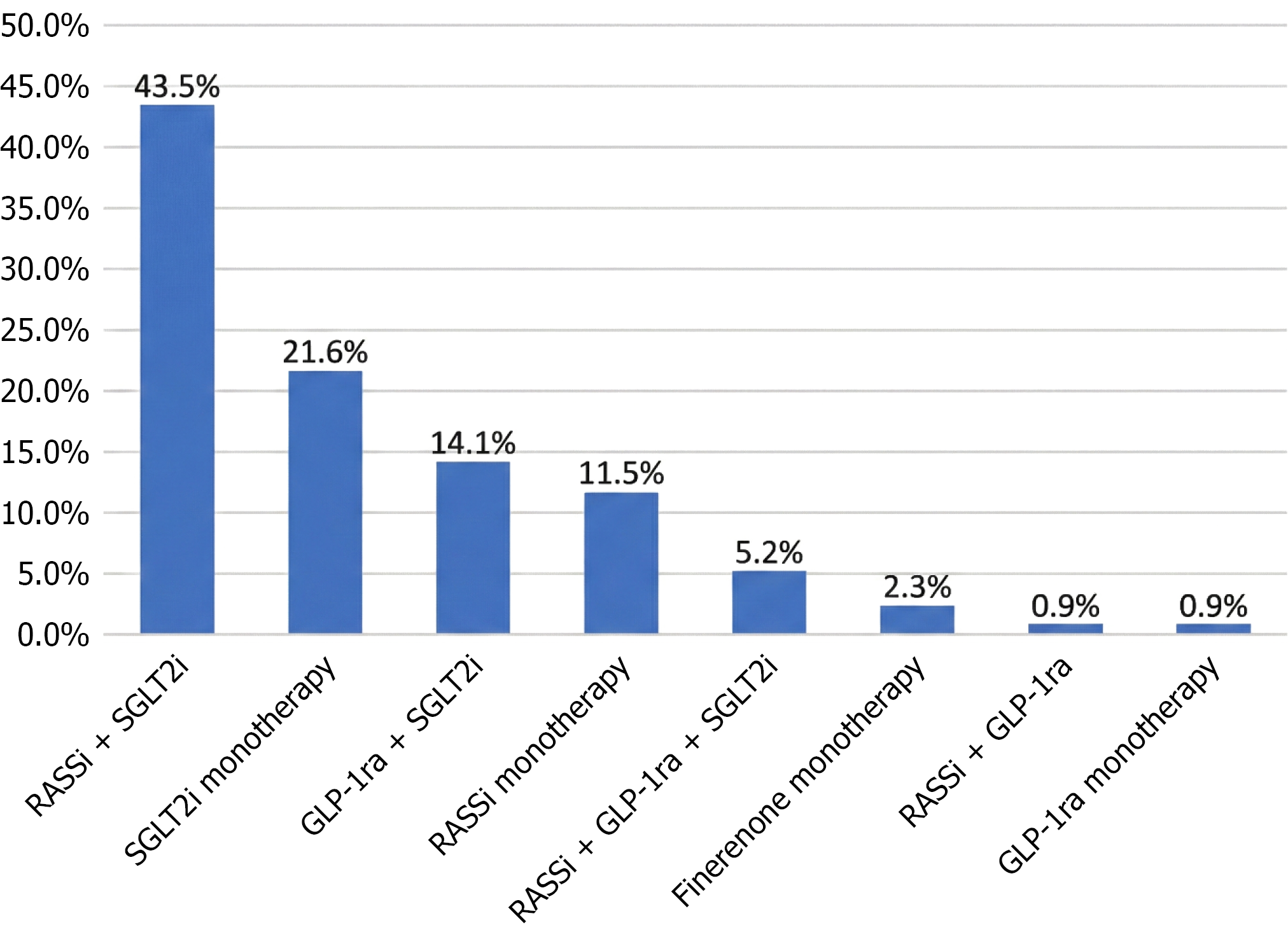
A total of 347 patients from seven countries in Central and South America were included, all with T2DM, individualized management based on international guidelines and therapy for CKD secondary to T2DM. 55.9% were male, the median age was 65 years (IQR 55-71) and the most frequent age group was under 65 years with 49.9%, followed by those between 65 years and 79 years with 45.2%. Among the relevant personal history, arterial hypertension stood out in 84.7% and cardiovascular diseases in 42.7%; obesity, acute myocardial infarction and smoking were found in less than 50% of the population. Pharmacological treatment included sodium-glucose cotransporter inhibitor (SGLT2i) in 84.4%, renin-angiotensin-system inhibitors (RASSI) 61.1% and GLP-1 21%, while finerenone was used at doses of 10 mg 55.9% and 20 mg 44.1%, Table 1. The combination of drugs firstly showed the use of RASSI + SGLT2i in 43.5%, followed by SGLT2i alone in 21.6%, GLP-1 + SGLT2i in 14.1%, RASSI alone in 11.5% and in proportions lower than 10% the combinations of RASSI + GLP-1 + SGLT2i, finerenone alone, RASSI + GLP-1 and GLP alone were used (Figure 1).
| n | % | |
| Gender | ||
| Female | 153 | 44.1 |
| Male | 194 | 55.9 |
| Age | 65 (55-71) | |
| < 65 years | 173 | 49.9 |
| 65-79 years | 157 | 45.2 |
| ≥ 80 years | 17 | 4.9 |
| Relevant background | ||
| AHT | 294 | 84.7 |
| Cardiovascular disease1 | 148 | 42.7 |
| Obesity | 113 | 32 6 |
| AMI | 73 | 21.0 |
| Smoking | 56 | 16.1 |
| Treatment | ||
| SGLT2i | 293 | 84.4 |
| RASSI | 212 | 61.1 |
| GLP-1 | 73 | 21.0 |
| Finerenone dosage | ||
| 10 | 194 | 55.9 |
| 20 | 153 | 44.1 |
When comparing the paraclinical parameters of HbA1c, eGFR, albuminuria/creatinuria ratio, SBP, DBP and potassium at baseline and six months after starting finerenone, we found medians IQRs for HbA1c 7.6 (6.8-8.1), eGFR 39.0 (30.0-50.0), albuminuria/creatinuria ratio 345 (189-760), SBP 143 (130-160), DBP 79 (70-82) and potassium 4.4 (4.1-4.7). The final assessment showed a statistically significant decrease in HbA1c, albuminuria/creatinuria ratio, SBP and DBP with res
| Baseline | Final | P value | |
| General | |||
| HbA1c | 7.6 (6.8-8.1) | 7.0 (6.5-7.9) | 0.0000 |
| eGFR | 39.0 (30.0-50.0) | 41.6 (27.0-52.0) | 0.7209 |
| ACR | 345 (189-760) | 81 (28-167) | 0.0000 |
| SBP | 143 (130-160) | 130 (120-140) | 0.0000 |
| DBP | 79 (70-82) | 73 (70-80) | 0.0000 |
| Potassium | 4.4 (4.1-4.7) | 4.7 (4.3-5.0) | 0.0000 |
| Dosage 10 | |||
| HbA1c | 7.8 (7.0-8.1) | 7.2 (6.5-8.0) | 0.0002 |
| eGFR | 33 (28-43) | 30 (26-43) | 0.0104 |
| ACR | 430 (257-898) | 90 (24-186) | 0.0000 |
| SBP | 145 (135-160) | 130 (120-133) | 0.0000 |
| DBP | 80 (70-87) | 70 (65-80) | 0.0000 |
| Potassium | 4.5 (4.2-4.8) | 5.0 (4.6-5.2) | 0.0000 |
| Dosage 20 | |||
| HbA1c | 7.3 (6.7-8.2) | 6.9 (6.5-7.5) | 0.0002 |
| eGFR | 46 (39-56) | 49 (42-58) | 0.3321 |
| ACR | 267 (153-383) | 78 (34-153) | 0.0000 |
| SBP | 140 (130-156) | 134 (122-144) | 0.0017 |
| DBP | 78 (70-80) | 75 (70-80) | 0.0048 |
| Potassium | 4.4 (4.0-4.6) | 4.4 (4.1-4.8) | 0.0098 |
Table 3 shows the comparative analysis of the baseline and final paraclinical parameters stratified by age group, which showed similar behavior to that of the general sample in the age groups under 65 years and between 65 years and 79 years, with statistically significant decreases in HbA1c, albuminuria/creatinuria ratio, SBP, DBP, a significant increase in potassium levels and no differences in eGFR values. However, in the age group of 80 years or older, statistical diffe
| Baseline | Final | P value | |
| < 65 years | |||
| HbA1c | 7.8 (6.9-8.3) | 7.1 (6.5-7.9) | 0.0000 |
| eGFR | 42 (30-50) | 44 (27-51) | 0.3656 |
| ACR | 352 (189-750) | 90 (29-194) | 0.0000 |
| SBP | 142 (130-160) | 130 (120-140) | 0.0000 |
| DBP | 78 (70-84) | 74 (70-80) | 0.0000 |
| Potassium | 4.4 (4.0-4.6) | 4.6 (4.3-5.0) | 0.0000 |
| 65-79 years | |||
| HbA1c | 7.5 (6.7-8.1) | 7.0 (6.5-7.9) | 0.0027 |
| eGFR | 38 (29-49) | 40 (29-55) | 0.7439 |
| ACR | 327 (197-850) | 86 (33-150) | 0.0000 |
| SBP | 145 (134-160) | 130 (120-140) | 0.0000 |
| DBP | 80 (70-82) | 72 (70-80) | 0.0000 |
| Potassium | 4.4 (4.1-4.7) | 4.8 (4.4-5.0) | 0.0000 |
| ≥ 80 years | |||
| HbA1c | 7.0 (6.4-7.7) | 6.9 (6.4-7.0) | 0.3292 |
| eGFR | 33 (30-50) | 32 (27-55) | 0.9365 |
| ACR | 297 (85-430) | 27 (23-125) | 0.0003 |
| SBP | 130 (124-150) | 120 (118-130) | 0.0928 |
| DBP | 77 (69-80) | 70 (64-80) | 0.4048 |
| Potassium | 4.5 (4.1-4.8) | 4.8 (4.5-5.1) | 0.0512 |
| Baseline | |
| RASSI + SGLT2i (n = 151) | |
| HbA1c | 7.5 (6.8-8.1) |
| eGFR | 44 (35-55) |
| ACR | 288 (156-454) |
| SBP | 142 (130-157) |
| DBP | 79 (70-81) |
| Potassium | 4.4 (4.1-4.6) |
| SGLT2i alone (n = 75) | |
| HbA1c | 7.8 (6.8-8.2) |
| eGFR | 32 (28-40) |
| ACR | 458 (270-900) |
| SBP | 145 (130-150) |
| DBP | 80 (70-86) |
| Potassium | 4.4 (4.1-4.6) |
| GLP-1 + SGLT2i (n = 49) | |
| HbA1c | 7.7 (7.0-8.1) |
| eGFR | 32 (28-44) |
| ACR | 644 (321-950) |
| SBP | 145 (135-160) |
| DBP | 75 (70-86) |
| Potassium | 4.4 (4.0-4.7) |
| RASSI alone (n = 40) | |
| HbA1c | 7.2 (6.6-8.0) |
| eGFR | 46 (34-57) |
| ACR | 266 (161-351) |
| SBP | 145 (131-160) |
| DBP | 79 (72-81) |
| Potassium | 4.4 (4.0-4.8) |
| RASSI + GLP-1 + SGLT2i (n = 18) | |
| HbA1c | 7.1 (6.7-9.0) |
| eGFR | 45 (39-59) |
| ACR | 289 (173-523) |
| SBP | 144 (120-156) |
| DBP | 77 (70-86) |
| Potassium | 4.3 (4.0-4.6) |
| Finerenone alone (n = 8) | |
| HbA1c | 7.5 (5.4-7.7) |
| eGFR | 30 (25-55) |
| ACR | 714 (300-1387) |
| SBP | 133 (127-170) |
| DBP | 78 (73-90) |
| Potassium | 4.8 (4.3-5.0) |
The clinical evolution of the patients showed the need for renal replacement therapy in 2.9% of the total sample, while 12.7% had hyperkalemia, 3.8% had to suspend treatment and 1.2% had to do so due to hyperkalemia. When comparing these events between the finerenone dose groups used, the 10 mg group had a higher frequency of hyperkalemia and the need for treatment interruption with 19.6% and 5.7%, respectively, which were higher than those observed in the 20 mg finerenone group, with hyperkalemia frequencies of 3.9%, P = 0.0000, and treatment interruption of 1.3%, P = 0.0335. No differences were observed in the need for renal replacement therapy or in the interruption of treatment due to hyper
| All (n = 347) | Dosage 10 (n = 194) | Dosage 20 (n = 153) | P value | |
| Required RRT | 10 (2.9) | 8 (4.1) | 2 (1.3) | 0.0800 |
| Hyperkalemia | 44 (12.7) | 38 (19.6) | 6 (3.9) | 0.0000 |
| Suspended treatment | 13 (3.8) | 11 (5.7) | 2 (1.3) | 0.0335 |
| Suspended due to hyperkalemia | 4 (1.2) | 4 (2.1) | 0 (0.0) | 0.1333 |
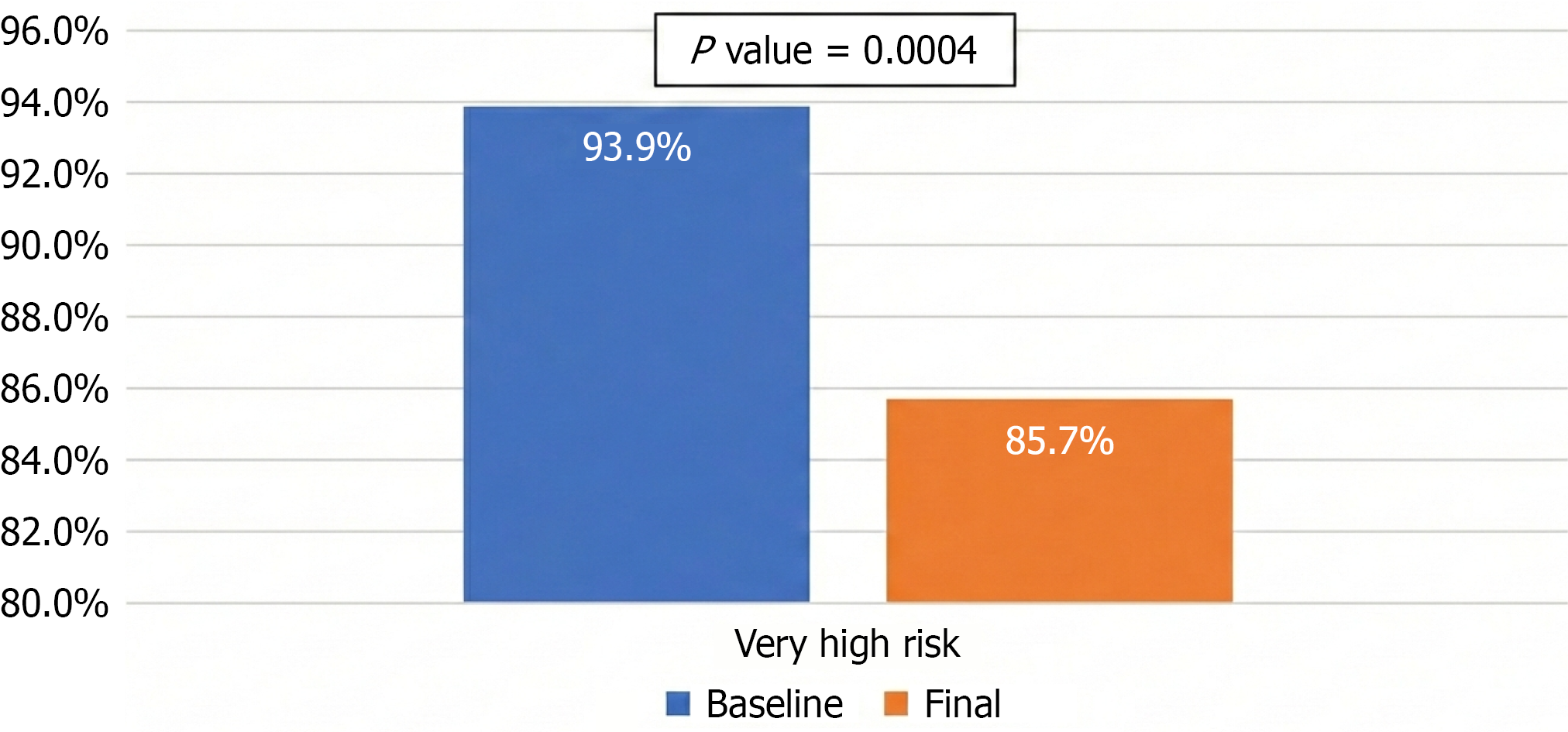
Tables 6 and 7 show the heat maps of patients’ cardiovascular risk according to eGFR and albuminuria/creatinuria ratio levels at baseline measurement (Table 6) and at six months of treatment (Table 7). Low risk was observed in 0.3% at baseline and 4.1% at six months. High risk was 14.0% in the baseline measurement and 31.2% in the final measurement. Finally, the very high-risk category had a baseline frequency of 93.3% and at six months it was 85.7%, which was statistically significant (P = 0.0004; Figure 2).
| CKD classification | ACR | |||||
| A1 (normal or average increase, < 30 mg/g) | A2 (moderate increase, 30-299 mg/g) | A3 (severe increase, ≥ 300 mg/g) | ||||
| GFR categories (mL/minute/1.73 m2) | G1 | Normal or high | ≥ 90 | 0 (0.0)1 | 3 (0.9)2 | 5 (1.5)3 |
| G2 | Average decrease | 60-89 | 1 (0.3)1 | 15 (4.4)2 | 18 (5.3)3 | |
| G3 | Average to moderate decrease | 45-59 | 2 (0.6)2 | 43 (12.6)3 | 44 (12.9)3 | |
| G4 | Moderate to severe decrease | 30-44 | 0 (0.0)3 | 52 (15.2)3 | 84 (24.6)3 | |
| G5 | Severe decrease | 15-29 | 0 (0.0)3 | 26 (7.6)3 | 49 (14.3)3 | |
| G6 | Kidney failure | < 15 | 0 (0.0)3 | 0 (0.0)3 | 0 (0.0)3 | |
| CKD classification | ACR | |||||
| A1 (normal or average increase, < 30 mg/g) | A2 (moderate increase, 30-299 mg/g) | A3 (severe increase, ≥ 300 mg/g) | ||||
| GFR categories (mL/minute/1.73 m2) | G1 | Normal or high | ≥ 90 | 2 (0.6)1 | 4 (1.3)2 | 1 (0.3)3 |
| G2 | Average decrease | 60-89 | 11 (3.5)1 | 19 (6.1)2 | 5 (1.6)3 | |
| G3 | Average to moderate decrease | 45-59 | 9 (2.9)2 | 74 (23.6)3 | 10 (3.2)3 | |
| G4 | Moderate to severe decrease | 30-44 | 18 (5.7)3 | 62 (19.7)3 | 5 (1.6)3 | |
| G5 | Severe decrease | 15-29 | 40 (12.7)3 | 38 (12.1)3 | 16 (5.1)3 | |
| G6 | Kidney failure | < 15 | 0 (0.0)3 | 0 (0.0)3 | 0 (0.0)3 | |
CKD and cardio-metabolic conditions often coexist in the same individual, sharing common pathophysiological path
The present study highlights the sample of 347 patients with CKD associated with T2DM, older adults on average, relevant cardiovascular comorbidities where the significant impact of the medicine on the variables studied is evident. Efficacy was initially evaluated at six months, identifying a 76.5% decrease in albuminuria/creatinuria ratio regardless of patient age and with no significant differences in medicine dosage.
This result was twice the magnitude of the decrease in albuminuria reported in other studies[10]. In the study by Bakris et al[10], the average decrease was 31% at 4 months, while in FIGARO-DKD, the average was 32%[6]. Other real-life studies such as that of Hanouneh et al[25], in which a 50% decrease over 8 months in albuminuria was documented in a cohort of 402 patients with CKD.
The mechanisms that could explain the positive effect of finerenone in reducing albuminuria are not fully understood. Blocking of the overactivity of the mineralocorticoid receptor that is expressed in the podocyte, endothelial cells and mesangial cells are reported to have a relevant role. However, the fact that albuminuria decreases in the first months of treatment could imply a possible additional hemodynamic effect that could be contributing. We have proposed two possible hemodynamic mechanisms that could explain this rapid decrease in albuminuria. The first is related to the acute effect observed in the first weeks of treatment with finerenone where an initial drop in glomerular filtration rate was observed, as evidenced in the FIDELIO-DKD and FIGARO-DKD studies, where an initial drop between 2-3 mL/minute was observed, which could attenuate the hyperfiltration state that characterizes patients with DKD, which could limit the amount of albumin that is filtered into the urinary space[23]. The other mechanism that could explain at least in part the early decrease of albuminuria is the effect of finerenone on blood pressure. In the FIDELIO-DKD and FIGARO-DKD studies, only a moderate decrease of 2-3 mmHg in SBP was observed vs placebo, and this could not fully explain the cardiorenal benefits; however, it should be considered that the blood pressure recordings in these studies were only made during medical consultation, which does not allow us to reliably infer the effect of finerenone on blood pressure over a 24-hour period.
The results are comparable with studies such as ARTS DN, a phase 2b study including patients with T2DM and CKD, where continuous ambulatory blood pressure monitoring recording of blood pressure was obtained, and results showed a significant decrease of 8.3-9.9 mmHg at 90 days for doses of 10-20 mg of finerenone, which are related to the decrease in intraglomerular pressure, which explains the filtration of proteins[26,27].
Despite the short half-life of finerenone, there appears to be a sustained effect on blood pressure which does not appear to be explained by pharmacokinetic effects. Therefore, the hypothesis arises that this effect could be related to genomic effects. In our cohort, we found no difference in glomerular filtration rate at baseline and at the end of follow-up, which speaks for the safety of the molecule in the short term, not increasing the risk of acute renal failure or impairment of renal function. Our findings are in line with the results of Agarwal et al[28], who reported an impairment of renal function vs placebo. In comparison to the first- and second-generation mineralocorticoid receptor antagonists, which in their studies (TOPCAT and EMPHASIS-HF) not only did not show benefits in glomerular filtration rate but also remained lower than that of patients assigned to placebo throughout follow-up, in addition to the fact that TOPCAT increased the risk of renal failure by 9% in patients receiving spironolactone[29,30]. Our results show significant decreases in SBP and DBP at six months, confirming these benefits for patients with CKD and AHT, and this would in part be a mediator of the renal effi
In the analysis of different subgroups, the trend on efficacy and safety results is consistent, with the exception that the group of patients receiving the 10 mg dose had more episodes of hyperkalemia and suspension of medication due to adverse effects vs those receiving 20 mg. The reason for this is that the patients with lower doses were sicker with lower glomerular filtration rate at baseline, 33 mL/minute vs 46 mL/minute, which were the patients who were receiving 20 mg doses.
Regarding hyperkalemia, in our cohort, the result was 12.7%, with no deaths related to hyperkalemia. The rate of treatment suspension due to episodes of hyperkalemia was also low (only 1.2%), a frequency very similar to that reported in other studies. FIGARO-DKD reported 1.2% vs 0.4% placebo, while FIDELIO-DKD reported 2.3% vs 0.9% placebo.
We also observed a significant improvement in patients’ metabolic control at six months, which could be related to the concomitant use of diabetes medicines, such as SGLT2i (84.4%), GLP-1 receptor agonists (21%), much more significantly than that reported in pivotal studies such as FIDELIO-DKD, where only 4.6% of patients reported using SGLT2i. The higher use of these medicines in our sample has to do with the current trend of prescribing SGLT2i, which are first-line therapy in the management of DKD[2,31].
Similarly, when we compared our study with FINEARTS-HF, which evaluated a sample of 6001 patients with mildly reduced or preserved heart failure, based on their glomerular filtration rate results and the need to start renal repla
We cannot fail to mention that in our cohort, cardiovascular risk was also modified at the end of follow-up. At baseline, patients with a very high-risk account for 93.9% of the total sample. At six months that proportion decreased to 85.7% with a statistically significant P-value, a similar finding described in the study by Agarwal et al[28], where a higher odds of improvement in the renal risk category in the Kidney Disease: Improving Global Outcomes heat map of 39% was reported, which allows us to understand that cardiovascular and renal risk is dynamic and bidirectional where a patient can migrate from a lower to a higher risk category and vice versa[33].
Our study has strengths and weaknesses. As a strength we should note that it is the first study to date of publication that has a representative sample of the region that demonstrates the efficacy and safety of finerenone in patients with CKD and diabetes mellitus. Information was obtained from 347 patients, and to date it is one of the real-life studies with the most patients.
The authors recognize several limitations inherent to the retrospective observational design of this study. First, the absence of a randomized control group and the non-randomized selection of participants introduce the potential for selection and information bias. Consequently, causality cannot be established, and we cannot rule out that unmeasured residual confounding influenced the outcomes, although the results are consistent with pivotal studies. Second, the follow-up period was limited to six months; while this duration was sufficient to observe meaningful hemodynamic and short-term metabolic changes (such as albuminuria reduction), it precludes the assessment of long-term hard outcomes, including progression to end-stage renal disease or cardiovascular mortality. Third, reliance on medical records from multiple centers across seven countries may introduce heterogeneity in data collection and laboratory measurement techniques. Finally, while the sample size represents a significant cohort for real-world evidence in Latin America, it is smaller than those of global pivotal trials; therefore, the analysis by subgroup should be considered hypothesis-gene
In our cohort of patients with T2DM and CKD in Latin America, finerenone was shown in a short-term follow-up to be a medicine with low risk of hyperkalemia, demonstrating improvement in blood pressure, decrease in urinary albumin-creatinine ratio and good tolerance. We consider expanding the fields of research in this intervention for the prediction of mortality and improvement of CKD in trials with a larger sample size, studies with a lower risk of bias and comparison of interventions with a good response to the pathology described.
| 1. | Tuttle KR, Wong L, St Peter W, Roberts G, Rangaswami J, Mottl A, Kliger AS, Harris RC, Gee PO, Fowler K, Cherney D, Brosius FC 3rd, Argyropoulos C, Quaggin SE; Diabetic Kidney Disease Collaborative Task Force. Moving from Evidence to Implementation of Breakthrough Therapies for Diabetic Kidney Disease. Clin J Am Soc Nephrol. 2022;17:1092-1103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 65] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 2. | Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024;105:S117-S314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3017] [Cited by in RCA: 2691] [Article Influence: 1345.5] [Reference Citation Analysis (9)] |
| 3. | American Diabetes Association Professional Practice Committee. 11. Chronic Kidney Disease and Risk Management: Standards of Care in Diabetes-2024. Diabetes Care. 2024;47:S219-S230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 183] [Article Influence: 91.5] [Reference Citation Analysis (1)] |
| 4. | Rico Fontalvo J, Vázquez Jiménez LC, Rodríguez Yánez T, Daza Arnedo R, Raad Sarabia M, Montejo Hernández JD, Lopera Vargas M, Jiménez Quintero J. Enfermedad renal diabética: puesta al día. An Fac Cienc Méd (Asunción). 2022;55:86-98. [RCA] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 5. | Duru OK, Middleton T, Tewari MK, Norris K. The Landscape of Diabetic Kidney Disease in the United States. Curr Diab Rep. 2018;18:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (4)] |
| 6. | Pitt B, Filippatos G, Agarwal R, Anker SD, Bakris GL, Rossing P, Joseph A, Kolkhof P, Nowack C, Schloemer P, Ruilope LM; FIGARO-DKD Investigators. Cardiovascular Events with Finerenone in Kidney Disease and Type 2 Diabetes. N Engl J Med. 2021;385:2252-2263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1436] [Cited by in RCA: 1151] [Article Influence: 230.2] [Reference Citation Analysis (8)] |
| 7. | Filippatos G, Anker SD, Pitt B, McGuire DK, Rossing P, Ruilope LM, Butler J, Jankowska EA, Michos ED, Farmakis D, Farjat AE, Kolkhof P, Scalise A, Joseph A, Bakris GL, Agarwal R. Finerenone efficacy in patients with chronic kidney disease, type 2 diabetes and atherosclerotic cardiovascular disease. Eur Heart J Cardiovasc Pharmacother. 2022;9:85-93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 8. | Inker LA, Eneanya ND, Coresh J, Tighiouart H, Wang D, Sang Y, Crews DC, Doria A, Estrella MM, Froissart M, Grams ME, Greene T, Grubb A, Gudnason V, Gutiérrez OM, Kalil R, Karger AB, Mauer M, Navis G, Nelson RG, Poggio ED, Rodby R, Rossing P, Rule AD, Selvin E, Seegmiller JC, Shlipak MG, Torres VE, Yang W, Ballew SH, Couture SJ, Powe NR, Levey AS; Chronic Kidney Disease Epidemiology Collaboration. New Creatinine- and Cystatin C-Based Equations to Estimate GFR without Race. N Engl J Med. 2021;385:1737-1749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3522] [Cited by in RCA: 3252] [Article Influence: 650.4] [Reference Citation Analysis (4)] |
| 9. | Baran W, Krzemińska J, Szlagor M, Wronka M, Młynarska E, Franczyk B, Rysz J. Mineralocorticoid Receptor Antagonists-Use in Chronic Kidney Disease. Int J Mol Sci. 2021;22:9995. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 10. | Bakris GL, Agarwal R, Anker SD, Pitt B, Ruilope LM, Rossing P, Kolkhof P, Nowack C, Schloemer P, Joseph A, Filippatos G; FIDELIO-DKD Investigators. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes. N Engl J Med. 2020;383:2219-2229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2319] [Cited by in RCA: 1935] [Article Influence: 322.5] [Reference Citation Analysis (10)] |
| 11. | Castillo GA, Aroca G, Buelvas J, Buitrago AF, Carballo V, Cárdenas JM, Gómez EA, Fériz K, Lopera JM, Melgarejo E, Restrepo K, Montejo JD, Pinzón JB, Quintero A, Rico JE, Rosero R. Recomendaciones para el manejo del riesgo cardiorrenal en el paciente con diabetes mellitus tipo 2. Rev Colomb Cardiol. 2020;27:3-22. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 12. | Chaudhuri A, Ghanim H, Arora P. Improving the residual risk of renal and cardiovascular outcomes in diabetic kidney disease: A review of pathophysiology, mechanisms, and evidence from recent trials. Diabetes Obes Metab. 2022;24:365-376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 75] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 13. | Kolkhof P, Bärfacker L. 30 years of the mineralocorticoid receptor: Mineralocorticoid receptor antagonists: 60 years of research and development. J Endocrinol. 2017;234:T125-T140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 195] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 14. | Rico Fontalvo J, Montejo Hernández J, Vázquez Jiménez L, Rodríguez Yánez T, Daza Arnedo R, Cardona Blanco MX, López Lozano J, Plaza Rivero M. Bloqueo del eje renina angiontensina aldosterona (RAAS) en la enfermedad renal diabética. Más allá del control de la hipertensión arterial. An Fac Cienc Méd (Asunción). 2023;56:46-57. [DOI] [Full Text] |
| 15. | Naaman SC, Bakris GL. Diabetic Nephropathy: Update on Pillars of Therapy Slowing Progression. Diabetes Care. 2023;46:1574-1586. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 232] [Cited by in RCA: 200] [Article Influence: 66.7] [Reference Citation Analysis (7)] |
| 16. | Rico-Fontalvo J, Daza-Arnedo R, Aguilar-Salcedo N, Alfaro M, Navas-Torrejano D, Cardona-Blanco M, Uparella-Gulfo I, Raad-Sarabia M, Abuabara-Franco E, Cabrales J. Nueva evidencia en el tratamiento de la enfermedad renal diabética: ¿qué aporta la finerenona? Rev Colomb Nefrol. 2022;9:e603. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Yama-Mosquera E, Montejo Hernández JD, Chacón Acevedo KR, Daza R, De la Espriella-Badel V, Machacon Miranda E, Henao CM, Guevara JG, Parra Serrano PA, Lopera Vargas JM, Pertuz A, López Garcés JG, Rozo Agudelo N, Cárdenas JG, López López E, Rico-Fontalvo J. Actualización de la guía colombiana de práctica clínica sobre enfermedad renal diabética. Rev Colomb Nefrol. 2024;11:e912. [DOI] [Full Text] |
| 18. | Rico Fontalvo JE. Enfermedad renal diabética: de cara a la prevención, diagnóstico e intervención temprana. Rev Colomb Nefrol. 2020;7:15-16. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 19. | Arellano AA, Castilla SM, Ortiz E, Lozano T. Perspectiva actual en manejo de Arritmias en Cardiomiopatía Chagásica. Rev Cienc Biomed. 2022;11:211-222. [DOI] [Full Text] |
| 20. | Currie G, Taylor AH, Fujita T, Ohtsu H, Lindhardt M, Rossing P, Boesby L, Edwards NC, Ferro CJ, Townend JN, van den Meiracker AH, Saklayen MG, Oveisi S, Jardine AG, Delles C, Preiss DJ, Mark PB. Effect of mineralocorticoid receptor antagonists on proteinuria and progression of chronic kidney disease: a systematic review and meta-analysis. BMC Nephrol. 2016;17:127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 146] [Article Influence: 14.6] [Reference Citation Analysis (4)] |
| 21. | Barrera-Chimal J, Girerd S, Jaisser F. Mineralocorticoid receptor antagonists and kidney diseases: pathophysiological basis. Kidney Int. 2019;96:302-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 188] [Article Influence: 26.9] [Reference Citation Analysis (3)] |
| 22. | Martinez Vargas V, Menon T, Castro M, Vijayaraghavan K. Evidence of clinical trials of cardiac outcomes on renal disease. In: Rao GHR, Das UN, editors. Cardiometabolic Diseases. Academic Press, 2025: 117-128. [DOI] [Full Text] |
| 23. | Fujii W, Shibata S. Mineralocorticoid Receptor Antagonists for Preventing Chronic Kidney Disease Progression: Current Evidence and Future Challenges. Int J Mol Sci. 2023;24:7719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 24. | Singh AK, Singh A, Singh R, Misra A. Finerenone in diabetic kidney disease: A systematic review and critical appraisal. Diabetes Metab Syndr. 2022;16:102638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 25. | Hanouneh M, Le D, Jaar BG, Tamargo C, Cervantes CE. Real-Life Experience on the Effect of SGLT2 Inhibitors vs. Finerenone vs. Combination on Albuminuria in Chronic Kidney Disease. Diagnostics (Basel). 2024;14:1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 26. | Bakris GL, Agarwal R, Chan JC, Cooper ME, Gansevoort RT, Haller H, Remuzzi G, Rossing P, Schmieder RE, Nowack C, Kolkhof P, Joseph A, Pieper A, Kimmeskamp-Kirschbaum N, Ruilope LM; Mineralocorticoid Receptor Antagonist Tolerability Study-Diabetic Nephropathy (ARTS-DN) Study Group. Effect of Finerenone on Albuminuria in Patients With Diabetic Nephropathy: A Randomized Clinical Trial. JAMA. 2015;314:884-894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 645] [Cited by in RCA: 539] [Article Influence: 49.0] [Reference Citation Analysis (7)] |
| 27. | Di Lullo L, Lavalle C, Scatena A, Mariani MV, Ronco C, Bellasi A. Finerenone: Questions and Answers-The Four Fundamental Arguments on the New-Born Promising Non-Steroidal Mineralocorticoid Receptor Antagonist. J Clin Med. 2023;12:3992. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 36] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 28. | Agarwal R, Filippatos G, Pitt B, Anker SD, Rossing P, Joseph A, Kolkhof P, Nowack C, Gebel M, Ruilope LM, Bakris GL; FIDELIO-DKD and FIGARO-DKD investigators. Cardiovascular and kidney outcomes with finerenone in patients with type 2 diabetes and chronic kidney disease: the FIDELITY pooled analysis. Eur Heart J. 2022;43:474-484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 809] [Article Influence: 161.8] [Reference Citation Analysis (4)] |
| 29. | Pitt B, Pfeffer MA, Assmann SF, Boineau R, Anand IS, Claggett B, Clausell N, Desai AS, Diaz R, Fleg JL, Gordeev I, Harty B, Heitner JF, Kenwood CT, Lewis EF, O'Meara E, Probstfield JL, Shaburishvili T, Shah SJ, Solomon SD, Sweitzer NK, Yang S, McKinlay SM; TOPCAT Investigators. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370:1383-1392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2427] [Cited by in RCA: 2084] [Article Influence: 173.7] [Reference Citation Analysis (4)] |
| 30. | Zannad F, McMurray JJ, Krum H, van Veldhuisen DJ, Swedberg K, Shi H, Vincent J, Pocock SJ, Pitt B; EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. 2011;364:11-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2600] [Cited by in RCA: 2159] [Article Influence: 143.9] [Reference Citation Analysis (6)] |
| 31. | Folkerts K, Millier A, Smela B, Olewinska E, Schmedt N, Mernagh P, Kovesdy CP. Real-world evidence for steroidal mineralocorticoid receptor antagonists in patients with chronic kidney disease. J Nephrol. 2023;36:1135-1167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 32. | Vaduganathan M, Filippatos G, Claggett BL, Desai AS, Jhund PS, Henderson A, Brinker M, Kolkhof P, Schloemer P, Lay-Flurrie J, Viswanathan P, Lam CSP, Senni M, Shah SJ, Voors AA, Zannad F, Rossing P, Ruilope LM, Anker SD, Pitt B, Agarwal R, McMurray JJV, Solomon SD. Finerenone in heart failure and chronic kidney disease with type 2 diabetes: FINE-HEART pooled analysis of cardiovascular, kidney and mortality outcomes. Nat Med. 2024;30:3758-3764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 87] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 33. | Bello AK, Okpechi IG, Levin A, Johnson DW. Variations in kidney care management and access: regional assessments of the 2023 International Society of Nephrology Global Kidney Health Atlas (ISN-GKHA). Kidney Int Suppl (2011). 2024;13:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |