Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.114321
Revised: November 22, 2025
Accepted: December 25, 2025
Published online: March 25, 2026
Processing time: 176 Days and 14.6 Hours
Liver transplantation (LT) remains the only curative therapy in patients with end stage liver disease. With improvement in surgical techniques and immunosuppression protocols, both 1-year and 5-year survival have improved significantly. Similarly, recurrent hepatitis B virus and hepatitis C virus infection and con
Core Tip: Effective management of viral hepatitis after liver transplantation hinges on early diagnosis, vigilant monitoring, and timely antiviral therapy. For hepatitis B virus, nucleos(t)ide analogues with high genetic barriers to resistance (e.g., entecavir, tenofovir alefnamide/tenofovir disoproxyl fumarate) are preferred, often with short-term hepatitis B immunoglobulin in high-risk patients. Directly acting antivirals have revolutionized treatment of patients with hepatitis C virus infection. Though timing of treatment must balance the baseline model for end stage liver disease score and graft outcomes. Hepatitis E virus infection should be suspected in unexplained graft dysfunction; ribavirin is effective when reduction of immunosupression fails. Recurrence with hepatitis D virus infection is rare if hepatitis B virus is well controlled. Infection with cytomegalovirus and Epstein-Barr virus require prophylaxis, pre-emptive monitoring, and individualized immunosuppression adjustment to reduce risk of post-transplant lymphoproliferative disorder (related to Epstein-Barr virus infection) and graft loss.
- Citation: Praharaj DL, Giri S, Anand AC, Mallick B, Nath P, Sahu SK, Pattnaik B, Acharya SK, Chawla YK. Viral hepatitis after liver transplantation: A brief overview. World J Virol 2026; 15(1): 114321
- URL: https://www.wjgnet.com/2220-3249/full/v15/i1/114321.htm
- DOI: https://dx.doi.org/10.5501/wjv.v15.i1.114321
Liver transplantation (LT) is a surgical procedure that replaces a diseased or cirrhotic liver with a healthy liver or liver portion from a donor (live donor or cadaveric donor). It is primarily performed to treat end-stage liver diseases, acute liver failure (ALF), and certain liver cancers. The procedure has become a well-established treatment, significantly improving survival and quality of life for patients with end stage liver diseases. Advances in surgical techniques, the advent of safe and highly effective immunosuppressive drugs, with improved donor management have contributed to increased transplant success rates. Despite these improvements, LT remains complex, requiring careful patient selection, thorough preoperative assessment, and lifelong post-LT care to monitor for complications such as rejection, infections, and recurrence of underlying diseases like viral hepatitis[1]. LT typically involves either deceased donor organs or living donor segments (especially in countries where donor organs are scarce). The procedure has revolutionized the mana
Viral hepatitis plays a crucial role in liver transplant outcomes due to its impact on graft health and patient survival. Infections by the hepatitis B virus (HBV) and the hepatitis C virus (HCV) can recur after transplantation, leading to ongoing liver damage and deterioration of graft function. Post-LT viral hepatitis, especially HCV, can cause rapid fibrosis and cirrhosis in the transplanted liver, increasing the risk of graft failure. Other viral infections that may complicate LT include hepatitis D virus (HDV), cytomegalovirus (CMV) and Epstein-Barr virus (EBV). Immunosuppression therapy (e.g., corticosteroids) required to prevent rejection can facilitate viral reactivation or increased replication, worsening liver injury. Persistent viral activity increases morbidity and mortality, affecting long-term graft viability and patient quality of life. The need for lifelong antiviral therapy, potential drug resistance, and side effects may further complicate post-transplant care. Thus, effective management of viral hepatitis in LT recipients is essential to optimize transplant success, reducing complications along with improving both long-term and short-term outcomes[1].
The scope of the present article is to provide a brief overview of common viral infections causing hepatitis in patients following LT. Clinical features, diagnosis and management of these infections will also be provided in detail.
Infection by HBV in LT recipients often occurs due to recurrence of infection which is associated with liver damage post-transplant. As estimated by the World Health Organization, about 296 million people are infected with HBV worldwide, and the virus results in about 8 Lakh deaths due to cirrhosis and hepatocellular carcinoma (HCC)[2]. Even after transplantation, the patient remains at risk of developing recurrent HBV infection of the graft liver. Prevention of recurrent HBV infection in the graft remains crucial to ensure graft and patient survival. At present, interferon (IFN) and nucleotide analogues (NAs) are used in the treatment of HBV infection. But use of IFN is often not possible in patients with decompensated cirrhosis (DC) or in post-LT settings due to fear of developing adverse events. Among the NAs, the agents with a high barrier to resistance [entecavir/tenofovir disoproxyl fumarate (TDF)/tenofovir alafenamide (TAF)] are being used at present. Though costly, the development of resistance even after using these agents for a long duration is rare[3]. Use of TAF in contrast to TDF is associated with a lower risk of osteopenia and renal side effects in view of low plasma concentration and high intrahepatic concentration of the drugs[4]. In terms of suppression of HBV-DNA levels, both TAF and TDF have been shown to be equally effective, though high-quality studies regarding the use of the former agent in the LT setting are lacking at present[5]. Thus, in current scenario, both TAF and entecavir are commonly used in patients with cirrhosis and LT recipients (to prevent recurrence of disease)[6,7].
Treatment of HBV infection prior to transplantation (in DC/ALF) has been shown to improve liver function and prevent re-infection in the transplanted liver. Use of NAs in patients with DC has also been shown to improve LT-free survival[8]. An important strategy to prevent recurrence of HBV infection after LT is to ensure almost undetectable HBV-DNA levels before LT[8,9]. Thus, once diagnosed[10], all patients with HBV-related DC must be started on NAs. With the use of NAs, the rate of HBV recurrence after LT has significantly reduced[11].
In patients with acute on chronic liver failure (ACLF), dual therapy has been shown to be better [especially in patients with high viral loads (> 108)] as compared to single NA therapy[10]. In contrast, patients with low HBV-DNA levels, either enticavir or TAF/TDF may be equally effective in achieving viral suppression[12]. Thus, a combination of newer NAs may be useful in selected ACLF patients with very high viral loads, as it may rapidly make the DNA levels undetectable.
In patients with ALF related to HBV infection, management remains the same as all other etiologies. All patients should be started on NAs. However, whenever a poor prognosis with conservative treatment is anticipated, the patients should be taken up for LT. In such a scenario, graft reinfection can be prevented by using hepatitis B immunoglobulin (HBIG) or NAs or both[12].
Hepatitis B infection in LT recipients can recur (in HBV-positive recipients) or de novo [from hepatitis B core antibody (anti-HBc)-positive donors][13]. The clinical presentation of recurrent hepatitis B may be acute hepatitis or chronic hepatitis. Rarely, these patients may also have fulminant hepatitis or fibrosing cholestatic hepatitis. These factors associated with high rate of virus reactivation include high viral load before transplantation, hepatitis B e antigen (HBeAg) reactivity, co-infection with human immunodeficiency virus, non-compliance to therapy, HCC at LT and antiviral drug resistance. In contrast, low viral load, anti-HBeAg positivity and anti-hepatitis B surface antigen (HBsAg) positivity is associated with low risk of recurrence[14,15]. De novo hepatitis occurs when a liver is received from a donor with a past history of HBV infection. In these donors, the hepatocytes of the grafted liver may contain covalant closed circular DNA (cccDNA), which gets reactivated with the use of immunosuppression. Despite being a DNA virus HBV replicates with a reverse transcription as intermediate. Establishment of cccDNA is crucial for the life cycle of HBV. This mini-chromosomal material acts as a power house of HBV transcription and plays important role in viral reactivation after LT. Despite use of the antivirals with high genotype (GT) barrier to resistance it is not possible to remove cccDNA from the mononuclear cells and hepatocytes altogether. Thus, donors with past history of infection with HBV (total anti-HBc antigen positivity) continue to harbour cccDNA in the hepatocytes despite being negative for HBsAg and HBV-DNA. In absence of prophylaxis, the recipients from these donors experience HBV recurrence following LT. Thus, all these recipients require lifelong prophylaxis with NAs[16,17]. The other mechanism leading to recurrence of HBV includes saturation of HBIG by very high viral load along with development of vaccine escape mutants (HBsAg mutation at codon 145 leading to glycine to arginine substitution)[18]. Co-infection with HDV may also increase risk of HBV recurrence following LT[19].
Recurrence of HBV infection after LT is defined as reappearance of HBsAg with or without detectable HBV-DNA. In contrast to immunocompetent individuals, DNA may be undetectable in LT recipients. Conversely, in view of mutation, HBsAg may also be undetectable in these patients[18]. Thus, in all patients with suspected HBV reactivation, both tests must be done. Without appropriate prophylaxis and treatment, recurrent HBV infection and consequent graft loss may be very high[20]. Risk factors of HBV recurrence in graft liver include very high viral load at transplantation, HBeAg positivity status and presence of anti-viral resistant strain[11]. An undetectable viral load prior to transplantation makes the risk of recurrent HBV infection almost nil[9]. HCC as an indication of LT and recurrence of HCC is also another predictor of recurrence of HBV[21].
Management of HBV recurrence after LT involves risk stratification of patients, followed by using HBIG, NAs, or both. Prophylactic use of HBIG ± NAs have revolutionized LT in HBV-related DC, before which recurrence of infection was almost universal[22]. Now, with the availability of newer NAs, treatment of HBV recurrence can also be done effectively.
HBIG is a polyclonal antibody against HBsAg, and it acts by neutralizing the circulating HBsAg and blocking the HBsAg receptors in extra-hepatic sites. Use of HBIG soon after LT led to a significant reduction in the rate of recurrence[23,24]. However, blocking the wild variant of HBsAg leads to the production of about 30 mutation-associated phenotypes of the surface antigen, thus gradually decreasing the efficacy of HBIG over time. More importantly, HBIG is expensive, has to be given by parenteral route for an indefinite period, thus increasing the cost of treatment. With the advent of NAs, use of HBIG alone has fallen out of favor and is being used only for a shorter period as combination therapy to prevent HBV recurrence[25,26].
Lamivudine and adefovir were the initial NAs that were used to prevent HBV recurrence post-LT. With the use of lamivudine mono-therapy, the rate of HBV reinfection was brought down to 3%-40%[27,28]. However, the use of these agents with a low genetic barrier to resistance led to the rapid emergence of resistance and HBV recurrence. Fung et al[29], for the first time, used entecavir monotherapy in LT recipients, 74% of whom had detectable HBV-DNA at transplantation. Using this regimen, 86% patients achieved sero-clearance at 1 year and 91% at 2 years. However, 10 patients had reappearance of HBsAg. This finding was later confirmed in another study where a cohort of 362 patients (HBV-related cirrhosis) who underwent LT. These patients received either lamivudine monotherapy, entecavir monotherapy, or a combination of the two. At 8 years of follow-up, 88% patients had undetectable HBsAg and 98% had undetectable HBV-DNA. Recurrence (mainly in the lamivudine group) occurred in about 16% patients[30]. In the Indian study by Wadhawan et al[31], using NA monotherapy, the rate of recurrence was 10% and 8% at 1 year and 2 years, respectively.
With the use of highly effective NAs, the role of HBIG in the prevention of HBV recurrence has significantly reduced. Nonetheless, HBIG may be used for a short period of time in a specific group of patients who are at high risk of having recurrent HBV infection; patients with high HBV-DNA levels prior to LT, patients undergoing transplant for HCC, patients infected with resistant strains of HBV and patients co-infected with HBV/HDV. In these high-risk patients, HBIG may be used with gradually reducing doses over 1 year. At present, there is insufficient evidence to consider total withdrawal of NAs in LT recipients[32]. Management of HBV infection in the setting of LT is summarized in Table 1.
| Donor status | Recipient status | Management |
| HBsAg positive or anti-HBc positive | HBsAg positive | Lifelong antiviral treatment ± hepatitis B immunoglobulin1 |
| Anti-HBc positive | Lifelong antiviral treatment | |
| Anti-HBs titre ≥ 10 mIU/mL | No therapy required | |
| Anti-HBs titre < 10 mIU/mL | Booster dose and repeat titre after 1 month; Lifelong antiviral treatment if transplanted before achieving protective antibody titre |
Recurrence of HBV infection can occur in recipients who receive grafts from anti-HBc-positive donors. The risk of de novo recurrence without NA prophylaxis ranges from 1% (recipients with anti-HBc and anti-hepatitis B surface positivity) to 47% (in HBV naive recipients). The evidence regarding the effectiveness of newer NAs in such settings is very limited[13]. Nonetheless, the American Society for Transplantation recommends NA prophylaxis to all recipients who receive a liver from an anti-HBc-positive graft[33]. In addition, Wang et al[34]. have described prevention of de novo recurrence of HBV in these recipients by using HBV vaccine (maintaining anti-hepatitis B surface titre > 100 U/L).
The other method to prevent the development of de novo HBV recurrence is the use of active immunization in LT recipients who are HBsAg negative and receiving liver from an anti-HBc positive donor. Ideally, vaccination should be done in all HBsAg-negative patients with cirrhosis at an early stage so as to elicit a robust immune response. In patients, DC vaccination may elicit sub-optimal immune response and may fail to prevent de novo HBV infection[35]. Vaccination following LT has also been shown to reduce the requirement of HBIG in patients at risk of acquiring de novo HBV infection. However, more future studies are required before the routine use of the HBV vaccine can be advocated following LT[36].
Infection with the HCV virus is a common cause of DC and HCC in both eastern and western countries. It is an important indication of LT all over the world. With the advent of directly acting antivirals (DAAs), the treatment of HCV has been simplified. The rate of sustained viral response (SVR) has improved. More importantly, treatment is now possible even in patients with DC and post-LT[37]. With improved SVR, most of the patients have undetectable HCV-RNA before LT. Thus, HCV recurrence is quite uncommon in the post-LT setting at present. However, a few concerns still remain[38].
During the IFN era, treatment of patients with DC was difficult due to poor efficacy, especially with GT-1, and tolerance. Overall, SVR with IFN therapy was about 19%[39]. Similarly, treatment after LT was also difficult and associated with low SVR (median 30%) and poor tolerance. Some of the patients needed dose reduction or discontinuation of therapy[40]. In addition, some patients also developed immunological complications including acute rejection, chronic rejection and autoimmune-like hepatitis[38,41].
In the DAA era, the treatment has been simplified and tolerance to treatment is better in DC. However, efficacy is lower in patients with poor liver function (low albumin and high bilirubin levels). In fact, patients with high Child-Turcotte-Pugh (CTP) and model for end stage liver disease (MELD) scores may actually worsen following DAA therapy[42,43]. More importantly, most of the currently available literature in DC is with GT-1 (most easily treatable with DAAs). Results with regard to the treatment of GT-3 patients with DC are limited to only a few studies[44,45]. In the study by Premkumar et al[44] about 1152 patients with HCV-related DC were included. The DAA regimen used was either sofosbuvir + velpatasvir + ribavirin or sofosbuvir + daclatasvir + ribavirin. The mean MELD score in this cohort was 16 ± 4. The SVR with this treatment was 82%-90%. In this study, about 25% patients achieved recompensation. Despite this, 145 patients died, and 6 patients underwent LT. High serum bilirubin, large varices and presence of gastric varices at baseline were negative predictors of recompensation. The summary of landmark studies looking into treatment of HCV-related DC is provided in Table 2[44-51]. In addition, patients can be easily treated with DAAs after LT even if there is a recurrence of HCV infection[52].
| Ref. | Patient population | SVR rates | Additional comments |
| Premkumar et al[44], 2024 | 1152 patients with DC (87% GT-3) | 82%-90% with sofosbuvir + velpatasvir + ribavirin or sofosbuvir + daclatasvir + ribavirin | Probably the largest study showing effect of directly acting antivirals in DC and GT-3 |
| Belperio et al[45], 2019 | 243 patients with GT-3 related DC | 87.5%-100% in various arms with sofosbuvir + daclatasvir/velpatasvir with or without ribavirin | Higher SVR (up to 92%) in patients with compensated cirrhosis |
| Lim et al[46], 2018 | 667 patients with GT-1, 231 non-transplanted GT | 87.5%-100% with ledipasvir + sofosbuvir with or without ribavirin | Higher SVR with compensated cirrhosis and in patients having normal bilirubin with high albumin |
| Manns et al[47], 2016 | 333 patients with DC, with GT-1 and 4 | CTP-B: 87%-96%, CTP-C: 78%-85% | 17 patients died during the study period |
| Poordad et al[48], 2016 | 60 patients with advanced cirrhosis | CTP-A: 93%, CTP-B: 93%, CTP-C: 56%, regimen used-sofosbuvir + daclatasvir + ribavirin | Presence of DC was a negative predictor of SVR |
| Reddy et al[49], 2017 | 240 patients with model for end stage liver disease score more than 10 | 35%-84% with sofosbuvir + simeprevir + ribavirin or sosfosbuvir + ribavirin | Low SVR rates with GT-1a; SVR only 35% with GT-3 |
| Curry et al[50], 2015 | 267 patients with DC, GT-3: 13%, GT-1: 75% | Sofosbuvir + velpatasvir-24 weeks (86%), sofosbuvir + velpatasvir-12 weeks (83%), sofosbuvir + velpatasvir + ribavirin (94%) | Serious adverse events in 16%-19% |
| Charlton et al[51], 2015 | 337 DC patients with GT-1 | 86%-89% with sofosbuvir + ledipasvir + ribavirin for 12-24 weeks | All but 1 patient with GT-1 |
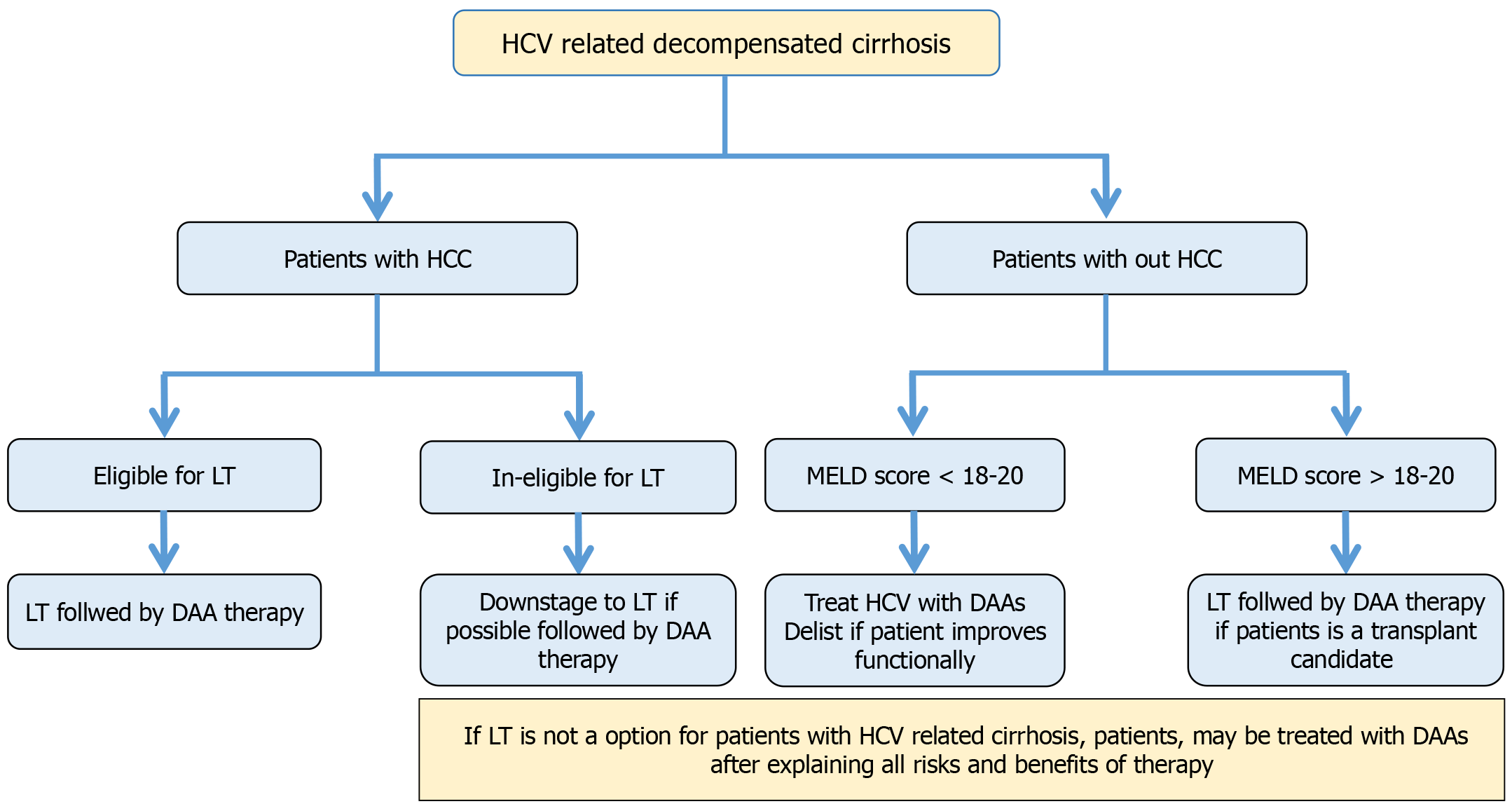
Treatment of DC patients with MELD scores more than 27 may actually reduce life expectancy. The European Association for the Study of the Liver recommends against the treatment of HCV-positive DC patients with MELD scores of more than 18-20 prior to LT[53].
Improvement in overall liver function, as reflected by a reduction in MELD score, is another concern. Two European studies have specifically looked into this. The study by Belli et al[54] included 103 patients from 11 European centers. The median MELD score was 16, and 14 patients had a MELD score of more than 20. In the latter group, only 2 patients could be taken out of the LT list. Moreover, improvement in both these patients was probably due to control of the infection that was present prior to initiation of DAAs. The extension of this study was further reported by Perricone et al[55], who included another 39 patients in addition to 103 patients as reported by Belli et al[54]. The median MELD score in this cohort was 16. The cumulative incidence of delisting was 7% at 48 weeks, 18% at 72 weeks and 27% at 96 weeks. The median MELD score of patients who were delisted was 14, in contrast to 16 in patients who could not be taken off the LT list. Fifteen patients had a MELD score of more than 20, out of which only 3 could be delisted. From the above two studies, it can be concluded that patients with high MELD scores should be treated following LT. Moreover, the incidence of adverse events was also higher in patients with high MELD scores and patients with CTP B/C (as compared to CTP-A)[56,57].
The pros and cons of treatment of HCV-positive DC patients prior to LT are given in Table 3.
| Pros of treatment | Cons of treatment with directly acting antivirals |
| Improvement of liver function with survival till LT | Slight improvement in MELD score may not be sufficient enough overall functional improvement |
| Some patients may get delisted from LT waiting list | Due to lower MELD score patients may not get a liver timely |
| Only option of treatment for patients not opting for LT | Sustained viral response rate may be lower in patients with decompensated cirrhosis |
| Reduced rate of recurrence after LT | Some patients may die during therapy |
| Less risk of recurrent cirrhosis due to hepatitis C virus |
Patients with HCV-related cirrhosis with HCC pose another challenge with regard to the use of DAAs. In this scenario, patients have to be treated for the HCV infection, underlying liver disease and the HCC itself. Though some initial studies showed possible increased recurrence of HCC with DAAs as compared to IFN-based therapy, it has now been proven beyond doubt that these agents reduce the rate of HCC recurrence[58]. In addition, some studies have shown reduced response to DAAs in patients with HCC due to an unfavorable tumor microenvironment[59,60]. In patients with successfully treated early HCC, the primary driver of mortality is usually the underlying diseased liver and decompensation rather than HCV infection[61]. Thus, all patients who are affordable or eligible for LT should be transplanted first. Patients who are not eligible for transplantation may be down-staged if possible using loco-regional therapy and immunotherapy. Once successfully down-staged, they may be transplanted. In all such scenarios, patients should be transplanted first, followed by DAA therapy after LT[62].
In contrast to patients with DC, treatment of patients after LT is easier. Serious adverse events are fewer, and the rate of SVR is also higher. A meta-analysis of 22 studies that included about 1700 patients showed an SVR rate of about 91% with the use of DAA following LT. Adverse events occurred in about 8% with 3% patients discontinuing therapy[52]. Two Indian studies have also shown excellent results with regard to attainment of SVR with the use of DAAs after LT[63,64]. The timing of initiation of treatment is not clear, though early initiation of therapy prior to graft fibrosis may improve overall outcome.
Similarly, data regarding the treatment of LT recipients with past DAA failure is also less clear. Successful use of sofosbuvir + velpatasvir + voxilaprevir with or without ribavirin has been documented in small case series and case reports. A mild reduction in the dose of calcinurin inhibitors may be needed[65].
Approach to patients with HCV-related cirrhosis in the setting of LT is provided in Figure 1.
Hepatitis E virus (HEV) infection is an emerging hepatotropic viral infection in developing countries. The infection is quite uncommon in developed countries. However, with population migration, the incidence of HEV infection is also increasing in Western countries. Transmission of HEV occurs by the feco-oral route. Zoonotic transmission has been documented after consumption of undercooked meat of pig, boar, deer, rabbit and camel[66,67]. In the setting of LT, HEV infection occurs by blood product transfusion and autochthonous route, though zoonotic transmission may also occur. In the setting of underlying immunosuppression in LT recipients, the disease may run a chronic course, leading to early fibrosis and cirrhosis. The infection in eastern countries is mostly due to GT-1 and GT-2, while in western parts of the world, it is due to GT-3[68-70]. Comparison of various GTs of HEV in the setting of LT is provided in Table 4.
| Characteristics | GT-1 | GT-2 | GT-3 | GT-4 |
| No of patients with LT | 6 patients | None reported | Most commonly reported GT in post-LT setting | 2 case reports |
| Mean age | 42 years | NA | Mostly in 40 years; rarely in 20-30 years of age | 68 |
| Country of origin | India | NA | Europe/South America/United States/Japan | United States |
| Chronicity of illness | None | NA | Yes | Patients developed fibrosis/cirrhosis |
The increased incidence and severity of HEV infection in the LT setting have been attributed to the use of tacrolimus instead of cyclosporine as the baseline immunosuppression and the use of DAAs for the treatment of HCV infection. IFN and ribavirin were the backbone of HCV therapy previously, which was also effective in the eradication of HEV infection. However, with the use of DAAs, this benefit has now been lost. Though some reports have documented successful use of sofosbuvir-based therapies for HEV infection, high-quality studies are needed before DAAs can be routinely prescribed for HEV infection[71,72].
Clinical features of patients in post-LT settings vary from asymptomatic, elevated liver enzymes or fulminant liver failure. In patients on induction chemotherapy or immunosuppression, the infection may run a chronic course leading to fibrosis or cirrhosis of the transplanted liver[70,73].
Due to extremely low prevalence and varied clinical presentation, pre-LT screening of HEV infection is usually not recommended[73]. A study from the UK specifically looked into the importance of pre-LT screening of HEV infection. They found out only 3 positive cases (detectable HEV-RNA) for HEV infection out of 262 LT recipients. One of these patients received treatment for infection both before and after transplant[71]. Similarly, routine screening of HEV infection in blood donors has not been proven cost-effective due to the very low prevalence of viral infection[74].
Hepatitis related to HEV infection should be suspected in any patient who presents with icteric hepatitis or ALF. Patients with underlying cirrhosis may present as ACLF, which is characterized by a sudden deterioration of the underlying liver function leading to worsening coagulopathy and encephalopathy[75]. In addition, pregnant women are also increasingly susceptible to developing HEV-related ALF, with case fatality going up to 20%[76].
However, the clinical course of HEV infection in the setting of LT with underlying immunosuppression is usually chronic with prolonged viremia[77]. Chronic HEV infection usually manifests as elevated transaminases without overt hepatitis. Rarely, a rapid progression of cirrhosis with graft failure in 1-2 years has also been reported[78,79]. The prospective study by Kamar et al[80] evaluated the evolution of liver fibrosis in 16 solid organ transplant (SOT) patients with chronic HEV infection. Three out of these patients progressed to cirrhosis, with two patients dying of decompensation. Some of the patients may also present with extrahepatic complications of HEV infection like neurological symptoms, renal injury and hematological disorders[81].
Testing for HEV infection is usually done by detection of virus-specific antibodies, positive HEV-RNA levels and liver biopsy. The first antibody to be detectable after HEV infection is immunoglobulin M anti-HEV, which is soon replaced by immunoglobulin G antibodies. However, in immunosuppressed individuals, the development of antibodies is often delayed. In addition, the antibody levels may remain unchanged even after successful treatment with IFN and ribavirin. In such a scenario, detection of HEV-RNA by PCR is the diagnostic modality of choice. Similarly, HEV-RNA PCR from liver biopsy tissue is also useful in making the diagnosis[82].
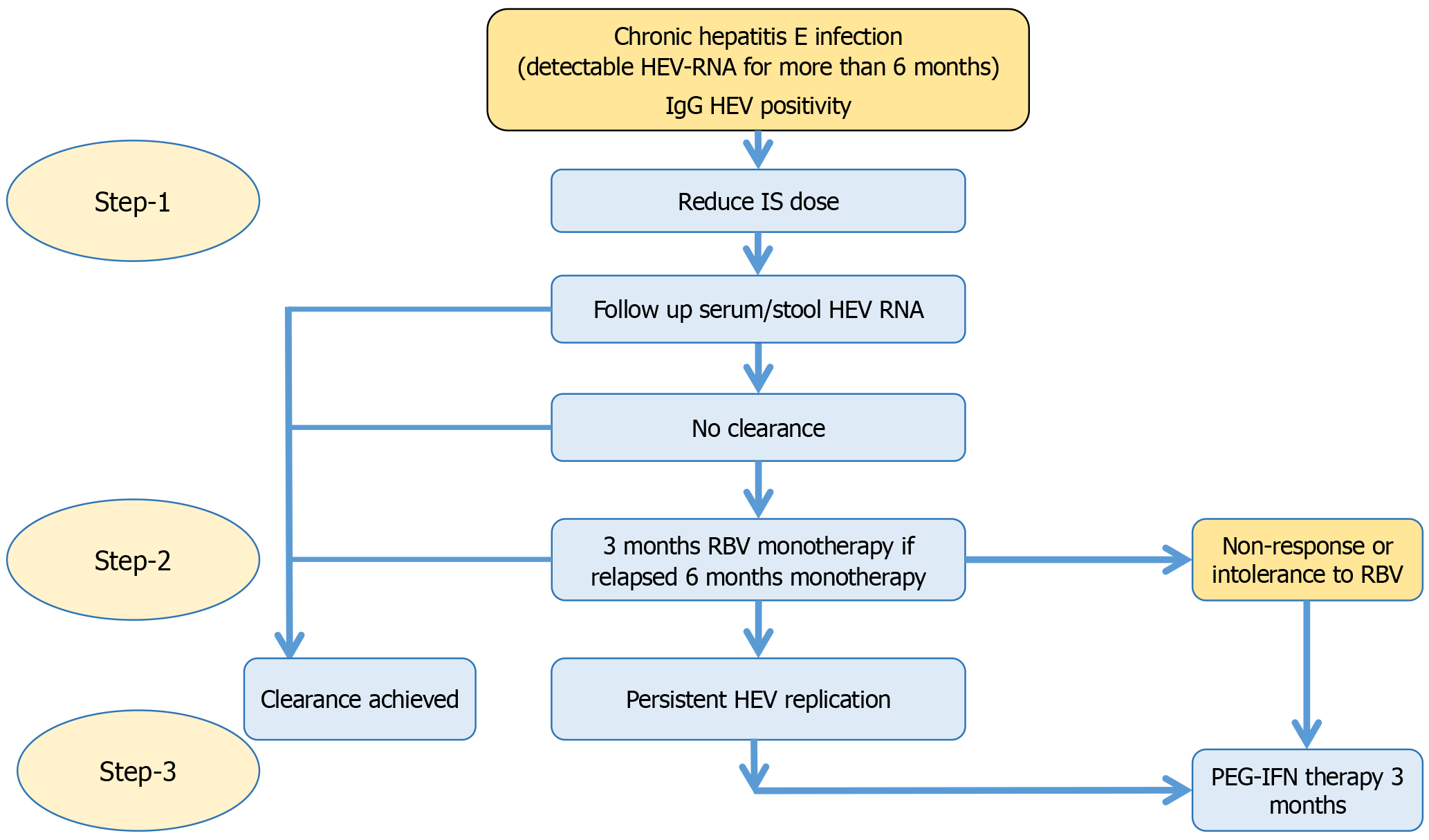
While in the general population, HEV infection resolves spontaneously, in SOT patients, chronic infection may lead to graft fibrosis, cirrhosis and liver failure[70]. Thus, these patients need to be treated. The first step in the treatment of HEV infection in SOT recipients is the reduction of immunosuppression, with which about 30% recipients will clear the virus[83]. Three patients with SOT (2 of 2 LT recipients and 1 out of 3 kidney transplants) achieved SVR at 3 months, 6 months, and 30 months after stopping immunosuppression[84]. Patients not responding to a reduction of immunosuppression or who continue to have deterioration of liver function should be started on ribavirin. Data regarding the use of ribavirin as HEV prophylaxis is currently lacking. The duration of treatment was 6-9 months with SVR between 67%-87%[84-86].
In the retrospective study by Kamar et al[87], 59 patients who had chronic HEV infection following SOT were included. Treatment with ribavirin was initiated after a median of 9 months of transplantation with a median dose of 600 mg/day (8 mg/kg/day). Ribavirin was given for a median of 3 months. All patients had HEV infection with GT-3. At the end of therapy, HEV clearance was noted in about 95% at the end of therapy. About 78% patients achieved SVR (HEV-RNA negative for 6 months after therapy). A high lymphocyte count at baseline was associated with a higher chance of achieving SVR. The other treatment option is the use of pegylated IFN, which should be used only in the setting of LT. Two case reports describe undetectable HCV-RNA for 20 weeks and 8 weeks of stopping treatment with IFN[88].
The current recommendation to treat HEV infection in the LT setting is to reduce immunosuppression and look for HEV seroclearance. If HEV-RNA remains positive, then treatment with ribavirin may be given for 3 months. If still HEV-RNA remains detectable, the duration of ribavirin therapy may be prolonged for 6 months. The next line of therapy is the use of peg-IFN for 3 months. Approach to HEV infection in LT recipients is given in Figure 2.
HDV, previously called the delta virus, is the third major cause of chronic viral hepatitis after HBV and HCV. Per se, HDV infection is not autonomous and occurs in individuals who are HBsAg positive. Exposure to the HDV infection is determined by the presence of anti-HDV antibody in serum and the detection of HDV-RNA by PCR[89,90].
Infection with HDV is widespread, with worldwide prevalence being 12-72 million. Injectable drug abusers are at high risk of acquiring the infection. However, with the availability of a highly effective vaccine and NAs with a high barrier to resistance, the prevalence of this infection has reduced significantly. Conversely, immigration of infected patients, especially from underdeveloped countries, is also increasing the prevalence of infection[91-93].
In general, the clinical course of HDV infection is more ominous than that of both HBV and HCV infection alone. The rapidity and rate of progression to cirrhosis are higher compared to infection with HBV alone. Super-infection with HDV may lead to the development of cirrhosis in 5-10 years in 60%-70% patients. Rapid decompensation rather than development of HCC is more often the indication for LT listing in the United States[94]. In the absence of an effective therapy, the rapid onset of decompensation makes LT an effective therapy for HDV infection. In fact, all patients with HBsAg-positive status should also be screened for HDV infection. However, diagnostic tests for HDV are not widely available and often not asked for due to the perception of decreasing prevalence of HDV infection with HBV vaccination. Moreover, access to LT is often low in the countries (Asia and Africa) with high HDV prevalence[95,96].
Initial studies with LT in HDV infection showed frequent reinfection (70%-80%) with a benign clinical course. Graft loss was minimal, isolated HDV reinfection. From these studies, it was concluded that recurrent HDV infection may not lead to graft loss without concomitant recurrence of HBV infection also[97]. In the landmark multicentric study by Caccamo[98], it was conclusively shown that HDV infection is not of much clinical significance in the absence of co-infection with HBV. With the use of HBIG and NAs with high barrier to resistance (enticavir/TDF), the level of HBV DNA is already low, thus reducing the risk of recurrence further. At present, the use of HBIG in the context of prevention of HBV reinfection in the transplanted liver is confined only to very high-risk cases. Withdrawal of HBIG while continuing NAs has been shown to be very effective in preventing HDV and HBV recurrence in various studies[99,100].
The Indian prospective study by Wadhawan et al[31] included 89 patients with HBV-related cirrhosis who underwent live-related LT. Out of these patients, 75 patients received only NAs without HBIG. At median follow-up of 21 months, all patients were HBV-DNA negative, 66 patients had cleared HBsAg, and 19 patients had developed antibody to HBsAg. The recurrence of HBV infection was noted only in 8 patients (due to NA withdrawal and resistance). No mortality occurred due to HBV recurrence.
From all these studies, it can be concluded that NAs alone without HBIG in most of the patients are sufficient to prevent HBV and consequent clinically significant HDV infection in transplanted liver. Buleviritide (entry inhibitor) was granted a conditional marketing authorization by the European Medicines Agency for the treatment of compensated cirrhosis related to HDV[101]. However, its use in the LT setting needs to be established in future studies.
The human CMV or human herpes virus 5 is the most common opportunistic infection in LT recipients. It belongs to the herpesviridae family. In the infected cells, it gives the appearance of intranuclear inclusion bodies in the infected cells. The primary infection tends to occur early in life, in the first two decades. In immunocompetent individuals, the infection is often self-limited and is cleared spontaneously. Afterwards, the body acts as the virus reservoir. Reactivation of infection occurs when the patient becomes immunodeficient (e.g., secondary to the use of immunosuppression in LT recipients). The seroprevalence in LT recipients in developing countries may be up to 97%[102-104]. Despite identification of risk factors and advances in early diagnosis and treatment, CMV infection continues to complicate about 30% LT recipients, increasing morbidity and mortality.
Infection in adults occurs by exposure to body fluids, tissues, including saliva, blood, tears, and transplanted grafts. After recovery from the primary infection, the virus remains latent in lymphoid and myeloid cells present in the liver. The latent virus gets reactivated after exposure to immunosuppressive following transplantation. The reactivated virus classically infects hepatocytes and macrophages, causing hepatitis. The proliferating virus ultimately disseminates to the bloodstream to other organs[105,106].
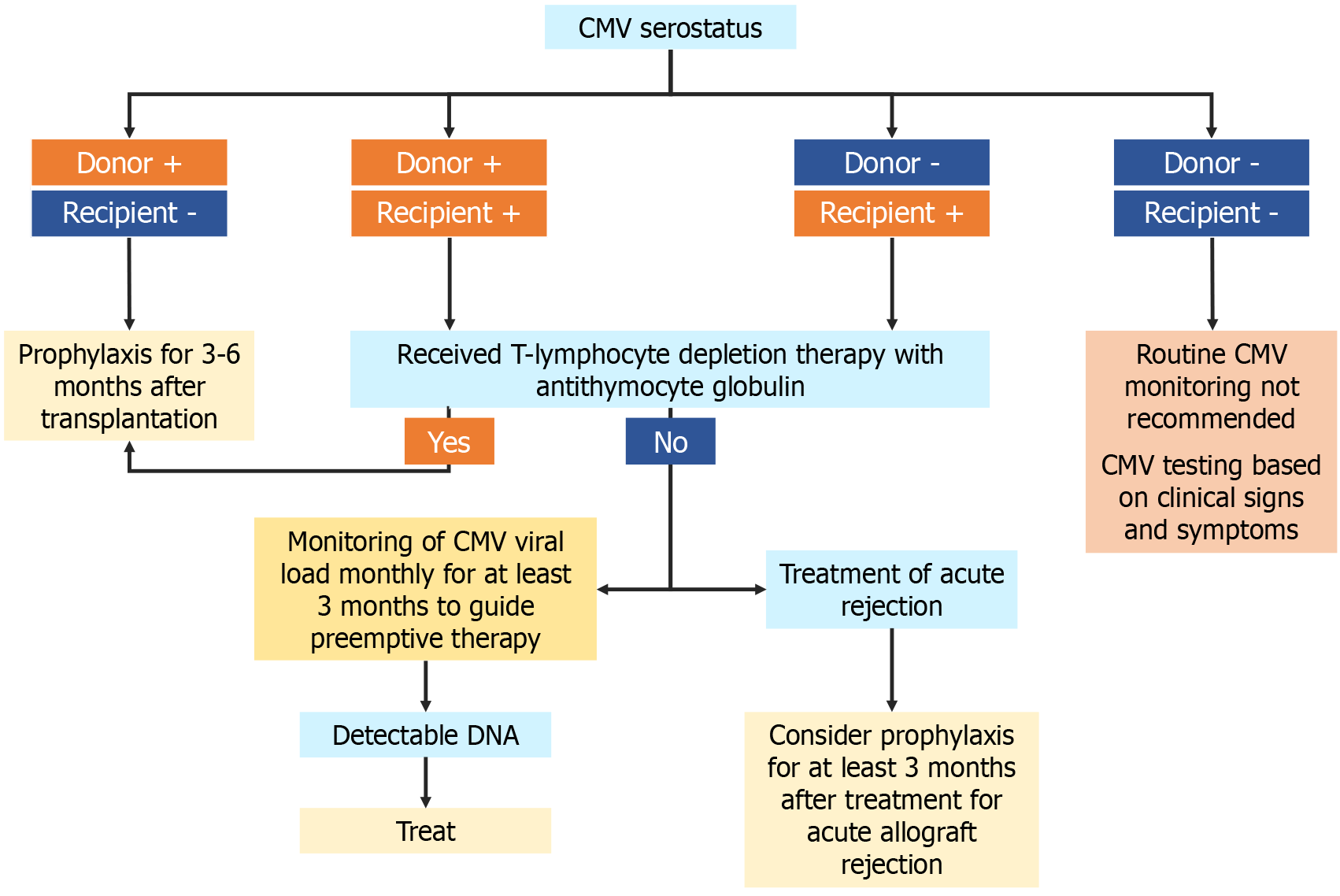
The biggest risk for the development of CMV infection post-LT is the CMV-seronegative status of the recipient. In LT with D + R- (Donor CMV+ with recipient CMV) CMV status, the risk of infection ranges from 78%-88% without prophylaxis. This risk decreases to about 13% when both are negative[107]. Pediatric LT recipients is associated with a higher risk of CMV recurrence as compared to adult recipients. More importantly, the latent period to develop recurrence is also lower in pediatric recipients[108]. Immunosuppressive medications cause lymphopenia and increase the risk of CMV infection. Higher doses of maintenance immunosuppression further increase the risk. In addition, use of lymphocyte depleting agents including muromonab, alemtuzumab and anti-thymocyte globulin[109,110]. In contrast, mammalian target of rapamycin inhibitors reduces the risk of CMV infection[111]. The risk of CMV disease is higher in LT recipients compared to renal transplant recipients. In contrast, the risk is lower compared to lungs, heart and intestine transplants[112-114]. Other risk factors for CMV infection post-LT include advanced donor/recipient age, human leukocyte antigen mismatch, immediate graft rejection by itself, impaired humoral immunity and co-infection with other herpes viruses[115,116].
A summary of the risks involved in CMV infection post-LT is provided in the Table 5. Pathophysiology of CMV infection after LT. Transmission of CMV infection occurs through body fluids and tissues like saliva, tears, blood and the transplanted graft. The primary infection occurs in the mucosal epithelial cells from which the virus disseminated through the CD14+ monocytes. After the primary infection, the virus remains latent in lymphoid and myeloid cells. As described above various risk factors are involved in development of recurrent CMV infection after LT. Among other organs, the virus preferentially attacks the transplanted liver (especially hepatocytes and macrophages) to cause CMV hepatitis. In the setting of intense immunosuppression following LT or during treatment of acute rejection the reactivation of the latent CMV virus may occur causing hepatitis. Conversely, CMV infection can potentiate allograft rejection by potentiating of immune response against the transplanted graft[117].
| Risk factors of CMV infection in LT patients |
| CMV sero-negative status |
| High dose maintenance immunosuppression |
| Use of lymphocyte depleting agents like anti-thymocyte globulin, alemtuzumab and muromonab |
| LT recipients more than kidney transplant recipients |
| Donor/recipient advanced age |
| Human leukocyte antigen mismatch |
| Immediate graft rejection |
| Impaired humoral immunity |
| Co-infection with other herpes virus |
The recently published American Society of Transplantation guidelines have standardized the definitions of various CMV-related illnesses: CMV infection is documented CMV replication (by nucleic acid testing) irrespective of symptomatology. The presence of CMV infection, along with the presence of symptoms, defines CMV disease. Asymptomatic CMV illness is defined as CMV replication without any obvious symptoms. Finally, CMV syndrome is characterized by the presence of CMV in blood along with at least 2 of the following features: (1) Fever > 38 degree for at least 48 hours; (2) New onset malaise or fatigue; (3) Leukopenia or neutropenia on two separate measurements; and (4) 5% atypical lym
In a post-LT setting, CMV infection usually presents with a mononucleosis syndrome characterized by fever, malaise and hematologic manifestations[119]. CMV hepatitis is characterized by jaundice (mixed hepatocellular/cholestatic) and elevated transaminases. In addition, the presence of the virus must be confirmed in liver tissue[120]. Rarely, patients may present with granulomatous hepatitis and fulminant hepatic failure[121,122]. Gastrointestinal (GI) CMV disease, as characterized by symptoms like odynophagia, dysphagia, hematochaezia, melena, vomiting, and diarrhoea, is also common in LT recipients. The most common site of GI involvement is the colon, which may be present in about 94% of cases[123]. In addition to the serological presence of CMV, histological demonstration of the virus is essential to confirm diagnosis[124].
While the clinical feature of CMV infection mimics those of acute cellular rejection or chronic rejection, the infection may lead to rejection by potentiation of allo-antigens[113]. Conversely, like other viral infections, CMV infection may worsen the immunosuppressive state of the LT recipient by inhibitory action on T-cells and B-cells. This leads to invasive fungal infection, bacteremia and EBV induced post-transplant lymphoproliferative disorders (PTLD)[118]. In addition, CMV infection may lead to graft vessel thrombosis due to the underlying pro-inflammatory state. It may also facilitate reactivation and replication of HCV[125].
Diagnosis of CMV infection in LT recipients is based on the clinical features with serological evidence of CMV replication. Whole blood sample rather than plasma, peripheral blood leucocyte, or peripheral blood mononuclear cells should be ideally used to demonstrate CMV replication[126]. Liver biopsy may be necessary to confirm the diagnosis and rule out concomitant illnesses like graft rejection. In the presence of GI symptoms, endoscopic/colonoscopy biopsies may be done to demonstrate viral cytopathic effect[127].
With the use of appropriate CMV prophylaxis, the morbidity and mortality related to infection in the post-LT setting have been reduced by about 80%[128]. The duration and nature of prophylaxis depend on the CMV status of the donor and recipients. As the risk of reactivation of infection is highest with D + R- state the prophylaxis should be given for longer periods (up to 6 months). In these patients, prophylactic therapy has been shown to be superior to pre-emptive therapy. In contrast, patients with D - R-status need not receive any prophylaxis. Prophylactic therapy should be started within 10 days of LT. Either intravenous (IV) gancyclovir (5 mg/kg per day) or oral valgancyclovir (900 mg/day) can be used. While studies have shown comparable efficacy with the use of either of the drugs, the United States Food and Drug Administration doesn’t approve the use of valgancyclovir as prophylaxis post-LT. However, due to oral usage, valgancyclovir is the popular prophylactic agent in the post-LT setting[129]. However, the dose of valgancyclovir has to be modified in the presence of renal insufficiency. After completion of CMV prophylaxis, patients should be monitored for post-prophylaxis delayed onset CMV disease weekly.
Pre-emptive therapy should be initiated once CMV viremia (Defined as CMV DNA level more than 3 times limit of normal) is confirmed. Preferred treatments include either IV gancyclovir (5 mg/kg/dose twice a day) or oral valgancyclovir (900 mg twice a day). The treatment should be continued till CMV-DNA PCR becomes negative.
Finally, for patients with CMV disease, IV gancyclovir should be used in severe cases at a dose of 5 mg/kg twice a day. In contrast, for mild to moderate cases, oral valgancyclovir (900 mg twice a day) can also be used with similar efficacy. The treatment should be given for at least 15 days. Treatment is discontinued once the patient is asymptomatic and DNA-PCR is negative. Persistence of CMV viremia after therapy and extensive GI involvement predicts relapse of infection[129].
Management of CMV infection in the LT setting is provided in Figure 3. Some of the patients may develop resistance to traditional therapy due to mutation of CMV-DNA polymerase or thymidate kinase. The prevalence of resistance is about 7% in SOT recipients. In case of resistance to valgancyclovir/gancyclovir, a combination of both therapies may be used. Alternatively, patients may receive cidofovir or foscarnet. A viral terminase inhibitor (Letermovir), approved to be used for CMV prophylaxis in hematopoietic stem cell transplantation, is not approved in LT recipients[117].
EBV is a double-stranded DNA virus belonging to the herpesviridae family that commonly affects people worldwide. The viral infection commonly occurs in childhood. Patients are often asymptomatic or present as infectious mononucleosis. In addition, the virus is also implicated in the pathogenesis of Burkitt’s lymphoma, nasopharyngeal carcinoma, Hodgkin’s lymphoma and multiple sclerosis. In addition, in post-LT settings, it is also associated with PTLD[130-133].
In the post-transplant setting, EBV infection plays a critical role in the development of PTLD by unchecked proliferation of B cells associated with inadequate T-cell-mediated immune surveillance[134,135]. While PTLD may or may not be related to EBV infection, most of the pediatric PTLD cases are related to the viral infection[136]. Moreover, primary EBV infection is more likely to evolve to PTLD in children as compared to adults[135].
The life cycle of EBV involves primary infection, the latent period and reactivation. Following infection of the pharyngeal epithelium, the virus migrates to B-cells. Following initial infection, the virus persists in memory B cells throughout life. During this latent phase, EBV expresses latency-associated proteins and EBV nuclear antigen, which facilitate immune evasion, viral replication and survival of the host cell[136]. Use of immunosuppressive therapy following LT allows the virus to enter the lytic phase, producing new virions and finally leading to PTLD[137,138].
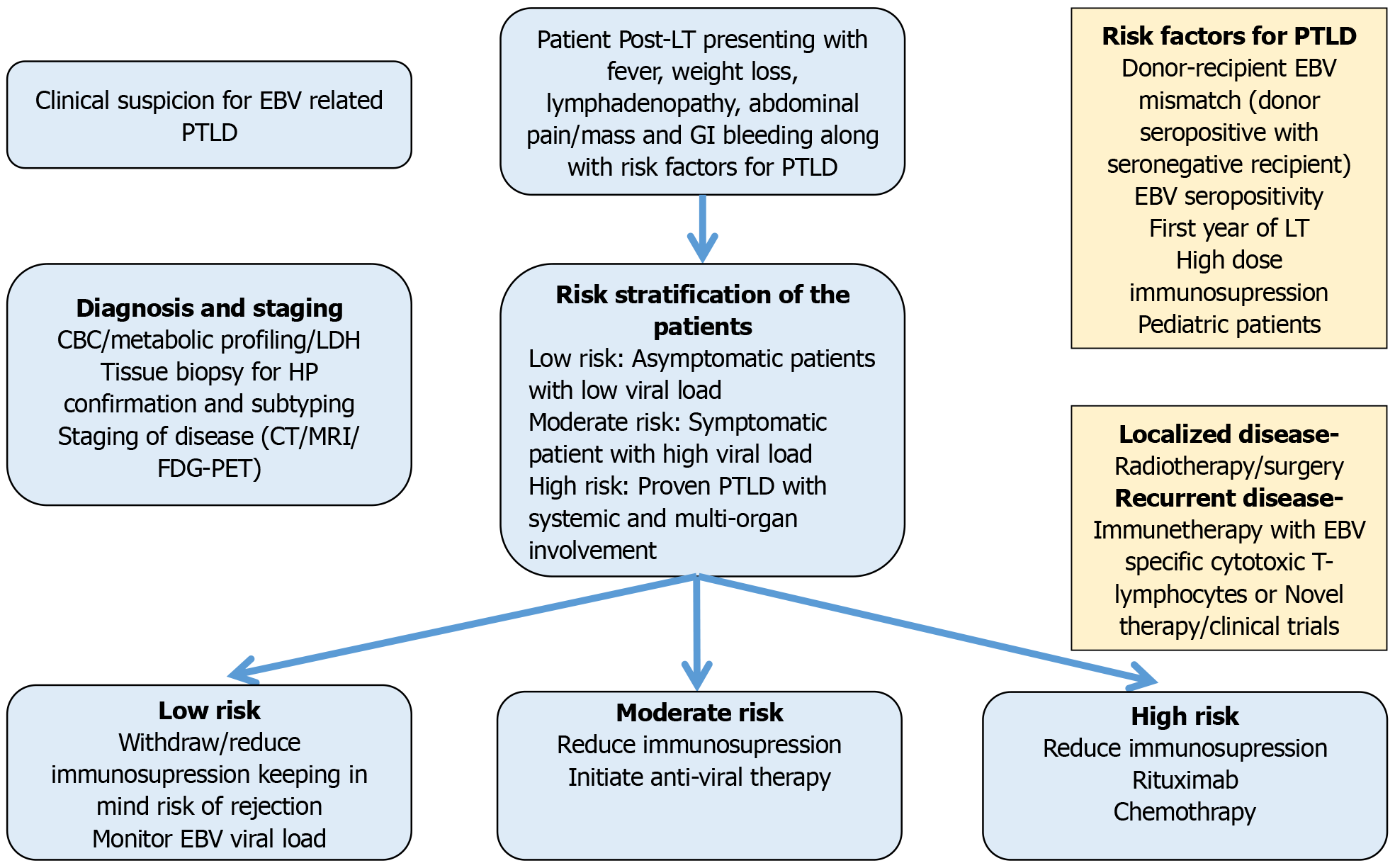
Immunosuppression is the most important risk factor for the development of PTLD, with a higher risk associated with high doses of maintenance immunosuppression. The EBV status of the donor and the recipient also plays an important role. Seropositive donor status with a seronegative recipient is also associated with a higher risk. Finally, the risk of developing PTLD is higher in children as compared to adults. In contrast to adults, children are often serologically negative for EBV due to the absence of primary infection, making them susceptible to PTLD[139].
Clinical features of PTLD is extremely variable and often non-specific. The time of presentation of PTLD is bimodal, with most of the cases presenting within 1 year of transplantation, while the rest present between 5-10 years. Patients may be asymptomatic or present with non-specific clinical features like fever, malaise, diarrhoea and anorexia. Some patients have an infectious mononucleosis-like presentation with lymphadenopathy. In addition, the presentation of PTLD may be focal (symptoms related to a single organ) or diffuse (systemic presentation like sepsis). Isolated tonsilar involvement may cause snoring, breathing difficulty, voice changes or cough[140,141]. GI involvement includes hepatosplenomegaly, GI bleed and abdominal pain. Central nervous system involvement may cause focal neurological deficit and seizures[142]. Finally, PTLD of the allograft may also occur and may mimic rejection[143]. While nodal involvement (mesenteric adenopathy/intussusception) is common in children, PTLD in adults is usually extranodal[144].
The definite diagnosis of PTLD is made by tissue biopsy, which is needed for histologic evaluation and classification. Initially, PTLT was classified as per the World Health Organization in 2017 as non-destructive, polymorphic, monomorphic and classic Hodgkin's disease[145]. This classification was later updated in 2022 to include immune dysregulation, aligning with clinical scenarios and patient management (Table 6)[146]. In addition to a biopsy, comprehensive laboratory and imaging studies should be done. Laboratory investigations include complete blood count, liver functions, renal functions, lactate dehydrogenase level, uric acid level, along with EBV serology and PCR. For detailed staging, a computed tomography scan/magnetic resonance imaging scan, along with a whole-body PET-computed tomography, may be done[142,147]. Presence of GI manifestations may warrant gastroduodenoscopy and biopsy of the affected areas[147].
| 2017 World Health Organization Classification | 2022 International consensus classification |
| Non-destructive PTLD | Hyperplasia arising at the time of immune deficiency/immune dysregulation |
| Polymorphic PTLD | Polymorphic lymphoproliferative disorder arising at the time of immune deficiency/dysregulation |
| Monomorphic PTLD | Lymphomas arising during immune-deficiency or dysregulation |
| Classic Hodgkin’s lymphoma PTLD |
The goal of treatment of PTLD is to achieve remission in addition to maintenance of graft status. Due to the rarity of the illness, there is currently no standard treatment. Treatment has to be individualized for every patient. A multidisciplinary approach with early involvement of an oncologist is essential to obtain an optimal outcome. The first step of treatment is the reduction of immunosuppression, followed by rituximab and chemotherapy. Surgery and radiotherapy may be needed for localized cases[146]. Novel therapeutic approaches include immunotherapy/cytokine-based therapy and anti-EBV therapy[148]. Management of PTLD has been provided in Figure 4.
Viral infection in the setting of LT is common and a major cause of morbidity and mortality. Use of NAs with high barrier to resistance and oral DAAs has significantly reduced the recurrence of HBV and HCV infections. However, data regarding timing and efficacy of DAAs post-LT is sparse. Hepatitis B vaccination and use of NAs have significantly reduced graft infection by HDV. However, routine screening of HDV infection in LT recipients is not done in most countries with high prevalence of HDV, which may underestimate the actual burden of HDV infection in this population. Prospective studies regarding use of HBV vaccination in post-LT setting is needed. Role of entry inhibitor (buleviritide) in the setting of LT is uncertain at present and needs to be clarified in future studies. Donor and recipient serological status for CMV infection must be ascertained prior to LT so as to identify and start prophylactic treatment of the high-risk patients. Similarly, use of letermovir to prevent CMV infection in post-LT settings needs to be clarified in future prospective studies. Treatment of PTLD related to EBV infection is not standardized and often underdiagnosed (due to the rarity of the illness). Personalized therapy with early involvement of an oncologist in cases of suspected PTLD is essential to obtain optimal outcomes. Finally, several novel therapies for PTLD, including cytokine inhibitors and T-cell-based therapy, are in the pipeline of development. More high-quality studies are needed to use these agents on a routine basis.
| 1. | Jothimani D, Venugopal R, Vij M, Rela M. Post liver transplant recurrent and de novo viral infections. Best Pract Res Clin Gastroenterol. 2020;46-47:101689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 2. | Jeng WJ, Papatheodoridis GV, Lok ASF. Hepatitis B. Lancet. 2023;401:1039-1052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 468] [Cited by in RCA: 393] [Article Influence: 131.0] [Reference Citation Analysis (1)] |
| 3. | Tacke F, Kroy DC. Treatment for hepatitis B in patients with drug resistance. Ann Transl Med. 2016;4:334. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 4. | Agarwal K, Fung SK, Nguyen TT, Cheng W, Sicard E, Ryder SD, Flaherty JF, Lawson E, Zhao S, Subramanian GM, McHutchison JG, Gane EJ, Foster GR. Twenty-eight day safety, antiviral activity, and pharmacokinetics of tenofovir alafenamide for treatment of chronic hepatitis B infection. J Hepatol. 2015;62:533-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 161] [Article Influence: 14.6] [Reference Citation Analysis (4)] |
| 5. | Agarwal K, Brunetto M, Seto WK, Lim YS, Fung S, Marcellin P, Ahn SH, Izumi N, Chuang WL, Bae H, Sharma M, Janssen HLA, Pan CQ, Çelen MK, Furusyo N, Shalimar D, Yoon KT, Trinh H, Flaherty JF, Gaggar A, Lau AH, Cathcart AL, Lin L, Bhardwaj N, Suri V, Mani Subramanian G, Gane EJ, Buti M, Chan HLY; GS-US-320-0110; GS-US-320-0108 Investigators. 96 weeks treatment of tenofovir alafenamide vs. tenofovir disoproxil fumarate for hepatitis B virus infection. J Hepatol. 2018;68:672-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 365] [Cited by in RCA: 327] [Article Influence: 40.9] [Reference Citation Analysis (3)] |
| 6. | Marcellin P, Heathcote EJ, Buti M, Gane E, de Man RA, Krastev Z, Germanidis G, Lee SS, Flisiak R, Kaita K, Manns M, Kotzev I, Tchernev K, Buggisch P, Weilert F, Kurdas OO, Shiffman ML, Trinh H, Washington MK, Sorbel J, Anderson J, Snow-Lampart A, Mondou E, Quinn J, Rousseau F. Tenofovir disoproxil fumarate versus adefovir dipivoxil for chronic hepatitis B. N Engl J Med. 2008;359:2442-2455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 963] [Cited by in RCA: 910] [Article Influence: 50.6] [Reference Citation Analysis (2)] |
| 7. | Fung J, Lai CL, Yuen MF. New paradigms for the treatment of chronic hepatitis B. J Gastroenterol Hepatol. 2008;23:1182-1192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (2)] |
| 8. | Jang JW, Choi JY, Kim YS, Woo HY, Choi SK, Lee CH, Kim TY, Sohn JH, Tak WY, Han KH. Long-term effect of antiviral therapy on disease course after decompensation in patients with hepatitis B virus-related cirrhosis. Hepatology. 2015;61:1809-1820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 126] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 9. | Marzano A, Gaia S, Ghisetti V, Carenzi S, Premoli A, Debernardi-Venon W, Alessandria C, Franchello A, Salizzoni M, Rizzetto M. Viral load at the time of liver transplantation and risk of hepatitis B virus recurrence. Liver Transpl. 2005;11:402-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 133] [Article Influence: 6.3] [Reference Citation Analysis (2)] |
| 10. | Lok AS, Trinh H, Carosi G, Akarca US, Gadano A, Habersetzer F, Sievert W, Wong D, Lovegren M, Cohen D, Llamoso C. Efficacy of entecavir with or without tenofovir disoproxil fumarate for nucleos(t)ide-naïve patients with chronic hepatitis B. Gastroenterology. 2012;143:619-628.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 117] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 11. | Manne V, Allen RM, Saab S. Strategies for the prevention of recurrent hepatitis B virus infection after liver transplantation. Gastroenterol Hepatol (N Y). 2014;10:175-179. [PubMed] |
| 12. | Li J, Hu C, Chen Y, Zhang R, Fu S, Zhou M, Gao Z, Fu M, Yan T, Yang Y, Li J, Liu J, Chen T, Zhao Y, He Y. Short-term and long-term safety and efficacy of tenofovir alafenamide, tenofovir disoproxil fumarate and entecavir treatment of acute-on-chronic liver failure associated with hepatitis B. BMC Infect Dis. 2021;21:567. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 13. | Cholongitas E, Papatheodoridis GV, Burroughs AK. Liver grafts from anti-hepatitis B core positive donors: a systematic review. J Hepatol. 2010;52:272-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 202] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 14. | Faria LC, Gigou M, Roque-Afonso AM, Sebagh M, Roche B, Fallot G, Ferrari TC, Guettier C, Dussaix E, Castaing D, Brechot C, Samuel D. Hepatocellular carcinoma is associated with an increased risk of hepatitis B virus recurrence after liver transplantation. Gastroenterology. 2008;134:1890-9; quiz 2155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 131] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 15. | Chauhan R, Lingala S, Gadiparthi C, Lahiri N, Mohanty SR, Wu J, Michalak TI, Satapathy SK. Reactivation of hepatitis B after liver transplantation: Current knowledge, molecular mechanisms and implications in management. World J Hepatol. 2018;10:352-370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 16. | Perrillo RP. Acute flares in chronic hepatitis B: the natural and unnatural history of an immunologically mediated liver disease. Gastroenterology. 2001;120:1009-1022. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 325] [Cited by in RCA: 290] [Article Influence: 11.6] [Reference Citation Analysis (1)] |
| 17. | Cheng AL, Hsiung CA, Su IJ, Chen PJ, Chang MC, Tsao CJ, Kao WY, Uen WC, Hsu CH, Tien HF, Chao TY, Chen LT, Whang-Peng J; Lymphoma Committee of Taiwan Cooperative Oncology Group. Steroid-free chemotherapy decreases risk of hepatitis B virus (HBV) reactivation in HBV-carriers with lymphoma. Hepatology. 2003;37:1320-1328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 227] [Article Influence: 9.9] [Reference Citation Analysis (1)] |
| 18. | Ghany MG, Ayola B, Villamil FG, Gish RG, Rojter S, Vierling JM, Lok AS. Hepatitis B virus S mutants in liver transplant recipients who were reinfected despite hepatitis B immune globulin prophylaxis. Hepatology. 1998;27:213-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 245] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 19. | Roche B, Samuel D. Liver transplantation in delta virus infection. Semin Liver Dis. 2012;32:245-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 20. | O'Grady JG, Smith HM, Davies SE, Daniels HM, Donaldson PT, Tan KC, Portmann B, Alexander GJ, Williams R. Hepatitis B virus reinfection after orthotopic liver transplantation. Serological and clinical implications. J Hepatol. 1992;14:104-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 268] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 21. | Saab S, Yeganeh M, Nguyen K, Durazo F, Han S, Yersiz H, Farmer DG, Goldstein LI, Tong MJ, Busuttil RW. Recurrence of hepatocellular carcinoma and hepatitis B reinfection in hepatitis B surface antigen-positive patients after liver transplantation. Liver Transpl. 2009;15:1525-1534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 68] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 22. | Saab S, Ham MY, Stone MA, Holt C, Tong M. Decision analysis model for hepatitis B prophylaxis one year after liver transplantation. Liver Transpl. 2009;15:413-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 23. | Sawyer RG, McGory RW, Gaffey MJ, McCullough CC, Shephard BL, Houlgrave CW, Ryan TS, Kuhns M, McNamara A, Caldwell SH, Abdulkareem A, Pruett TL. Improved clinical outcomes with liver transplantation for hepatitis B-induced chronic liver failure using passive immunization. Ann Surg. 1998;227:841-850. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 112] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 24. | Terrault NA, Zhou S, Combs C, Hahn JA, Lake JR, Roberts JP, Ascher NL, Wright TL. Prophylaxis in liver transplant recipients using a fixed dosing schedule of hepatitis B immunoglobulin. Hepatology. 1996;24:1327-1333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 208] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 25. | Lazarevic I. Clinical implications of hepatitis B virus mutations: recent advances. World J Gastroenterol. 2014;20:7653-7664. [PubMed] [DOI] [Full Text] |
| 26. | Loomba R, Rowley AK, Wesley R, Smith KG, Liang TJ, Pucino F, Csako G. Hepatitis B immunoglobulin and Lamivudine improve hepatitis B-related outcomes after liver transplantation: meta-analysis. Clin Gastroenterol Hepatol. 2008;6:696-700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 81] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 27. | Grellier L, Mutimer D, Ahmed M, Brown D, Burroughs AK, Rolles K, McMaster P, Beranek P, Kennedy F, Kibbler H, McPhillips P, Elias E, Dusheiko G. Lamivudine prophylaxis against reinfection in liver transplantation for hepatitis B cirrhosis. Lancet. 1996;348:1212-1215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 330] [Cited by in RCA: 285] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 28. | Mutimer D, Pillay D, Dragon E, Tang H, Ahmed M, O'Donnell K, Shaw J, Burroughs N, Rand D, Cane P, Martin B, Buchan S, Boxall E, Barmat S, Gutekunst K, McMaster P, Elias E. High pre-treatment serum hepatitis B virus titre predicts failure of lamivudine prophylaxis and graft re-infection after liver transplantation. J Hepatol. 1999;30:715-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 104] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 29. | Fung J, Cheung C, Chan SC, Yuen MF, Chok KS, Sharr W, Dai WC, Chan AC, Cheung TT, Tsang S, Lam B, Lai CL, Lo CM. Entecavir monotherapy is effective in suppressing hepatitis B virus after liver transplantation. Gastroenterology. 2011;141:1212-1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 155] [Article Influence: 10.3] [Reference Citation Analysis (3)] |
| 30. | Fung J, Chan SC, Cheung C, Yuen MF, Chok KS, Sharr W, Chan AC, Cheung TT, Seto WK, Fan ST, Lai CL, Lo CM. Oral nucleoside/nucleotide analogs without hepatitis B immune globulin after liver transplantation for hepatitis B. Am J Gastroenterol. 2013;108:942-948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 94] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 31. | Wadhawan M, Gupta S, Goyal N, Taneja S, Kumar A. Living related liver transplantation for hepatitis B-related liver disease without hepatitis B immune globulin prophylaxis. Liver Transpl. 2013;19:1030-1035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (3)] |
| 32. | Kumar N, Choudhary NS. Managing HBV and HCV Infection Pre- and Post-liver Transplant. J Clin Exp Hepatol. 2024;14:101287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (3)] |
| 33. | Te H, Doucette K. Viral hepatitis: Guidelines by the American Society of Transplantation Infectious Disease Community of Practice. Clin Transplant. 2019;33:e13514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 67] [Article Influence: 9.6] [Reference Citation Analysis (1)] |
| 34. | Wang SH, Loh PY, Lin TL, Lin LM, Li WF, Lin YH, Lin CC, Chen CL. Active immunization for prevention of De novo hepatitis B virus infection after adult living donor liver transplantation with a hepatitis B core antigen-positive graft. Liver Transpl. 2017;23:1266-1272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (2)] |
| 35. | Sahu M, Praharaj D, Bhadoria AS. Vaccination Strategies for a Liver Transplant Recipient. J Clin Exp Hepatol. 2025;15:102421. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 36. | Adler E, Krucker Y, Kolev M, Semmo N. Active hepatitis B virus vaccination in the prevention of viral reactivation in liver transplantation recipients with previous hepatitis B infection: a cohort study. Swiss Med Wkly. 2025;155:4116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 37. | Choudhary NS, Saraf N, Saigal S, Rastogi A, Bhangui P, Thiagrajan S, Soin AS. Outcome of hepatitis C-related liver transplantation in direct-acting antiviral era. Indian J Gastroenterol. 2020;39:539-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 38. | Anand AC. Potential Liver Transplant Recipients with Hepatitis C: Should They Be Treated Before or After Transplantation? J Clin Exp Hepatol. 2017;7:42-54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 39. | Xirouchakis E, Triantos C, Manousou P, Sigalas A, Calvaruso V, Corbani A, Leandro G, Patch D, Burroughs A. Pegylated-interferon and ribavirin in liver transplant candidates and recipients with HCV cirrhosis: systematic review and meta-analysis of prospective controlled studies. J Viral Hepat. 2008;15:699-709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 76] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 40. | Berenguer M. Systematic review of the treatment of established recurrent hepatitis C with pegylated interferon in combination with ribavirin. J Hepatol. 2008;49:274-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 298] [Cited by in RCA: 262] [Article Influence: 14.6] [Reference Citation Analysis (1)] |
| 41. | Choudhary NS, Saraf N, Saigal S, Gautam D, Rastogi A, Goja S, Bhangui P, Srinivasan T, Yadav SK, Soin A. Revisiting chronic rejection following living donor liver transplantation in the tacrolimus era: A single center experience. Clin Transplant. 2018;32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 42. | Cortesi PA, Belli LS, Facchetti R, Mazzarelli C, Perricone G, De Nicola S, Cesana G, Duvoux C, Mantovani LG, Strazzabosco M; European Liver and Intestine Transplant Association (ELITA). The optimal timing of hepatitis C therapy in liver transplant-eligible patients: Cost-effectiveness analysis of new opportunities. J Viral Hepat. 2018;25:791-801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 43. | Chhatwal J, Samur S, Kues B, Ayer T, Roberts MS, Kanwal F, Hur C, Donnell DM, Chung RT. Optimal timing of hepatitis C treatment for patients on the liver transplant waiting list. Hepatology. 2017;65:777-788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 79] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 44. | Premkumar M, Dhiman RK, Duseja A, Mehtani R, Taneja S, Gupta E, Gupta P, Sandhu A, Sharma P, Rathi S, Verma N, Kulkarni AV, Bhujade H, Chaluvashetty SB, Kalra N, Grover GS, Nain J, Reddy KR. Recompensation of Chronic Hepatitis C-Related Decompensated Cirrhosis Following Direct-Acting Antiviral Therapy: Prospective Cohort Study From a Hepatitis C Virus Elimination Program. Gastroenterology. 2024;167:1429-1445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 38] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 45. | Belperio PS, Shahoumian TA, Loomis TP, Mole LA, Backus LI. Real-world effectiveness of daclatasvir plus sofosbuvir and velpatasvir/sofosbuvir in hepatitis C genotype 2 and 3. J Hepatol. 2019;70:15-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 72] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 46. | Lim JK, Liapakis AM, Shiffman ML, Lok AS, Zeuzem S, Terrault NA, Park JS, Landis CS, Hassan M, Gallant J, Kuo A, Pockros PJ, Vainorius M, Akushevich L, Michael L, Fried MW, Nelson DR, Ben-Ari Z; HCV-TARGET Study Group. Safety and Effectiveness of Ledipasvir and Sofosbuvir, With or Without Ribavirin, in Treatment-Experienced Patients With Genotype 1 Hepatitis C Virus Infection and Cirrhosis. Clin Gastroenterol Hepatol. 2018;16:1811-1819.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 47. | Manns M, Samuel D, Gane EJ, Mutimer D, McCaughan G, Buti M, Prieto M, Calleja JL, Peck-Radosavljevic M, Müllhaupt B, Agarwal K, Angus P, Yoshida EM, Colombo M, Rizzetto M, Dvory-Sobol H, Denning J, Arterburn S, Pang PS, Brainard D, McHutchison JG, Dufour JF, Van Vlierberghe H, van Hoek B, Forns X; SOLAR-2 investigators. Ledipasvir and sofosbuvir plus ribavirin in patients with genotype 1 or 4 hepatitis C virus infection and advanced liver disease: a multicentre, open-label, randomised, phase 2 trial. Lancet Infect Dis. 2016;16:685-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 391] [Cited by in RCA: 357] [Article Influence: 35.7] [Reference Citation Analysis (2)] |
| 48. | Poordad F, Schiff ER, Vierling JM, Landis C, Fontana RJ, Yang R, McPhee F, Hughes EA, Noviello S, Swenson ES. Daclatasvir with sofosbuvir and ribavirin for hepatitis C virus infection with advanced cirrhosis or post-liver transplantation recurrence. Hepatology. 2016;63:1493-1505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 369] [Cited by in RCA: 347] [Article Influence: 34.7] [Reference Citation Analysis (1)] |
| 49. | Reddy KR, Lim JK, Kuo A, Di Bisceglie AM, Galati JS, Morelli G, Everson GT, Kwo PY, Brown RS Jr, Sulkowski MS, Akuschevich L, Lok AS, Pockros PJ, Vainorius M, Terrault NA, Nelson DR, Fried MW, Manns MP; HCV-TARGET Study Group. All-oral direct-acting antiviral therapy in HCV-advanced liver disease is effective in real-world practice: observations through HCV-TARGET database. Aliment Pharmacol Ther. 2017;45:115-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 54] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 50. | Curry MP, O'Leary JG, Bzowej N, Muir AJ, Korenblat KM, Fenkel JM, Reddy KR, Lawitz E, Flamm SL, Schiano T, Teperman L, Fontana R, Schiff E, Fried M, Doehle B, An D, McNally J, Osinusi A, Brainard DM, McHutchison JG, Brown RS Jr, Charlton M; ASTRAL-4 Investigators. Sofosbuvir and Velpatasvir for HCV in Patients with Decompensated Cirrhosis. N Engl J Med. 2015;373:2618-2628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 692] [Cited by in RCA: 627] [Article Influence: 57.0] [Reference Citation Analysis (4)] |
| 51. | Charlton M, Everson GT, Flamm SL, Kumar P, Landis C, Brown RS Jr, Fried MW, Terrault NA, O'Leary JG, Vargas HE, Kuo A, Schiff E, Sulkowski MS, Gilroy R, Watt KD, Brown K, Kwo P, Pungpapong S, Korenblat KM, Muir AJ, Teperman L, Fontana RJ, Denning J, Arterburn S, Dvory-Sobol H, Brandt-Sarif T, Pang PS, McHutchison JG, Reddy KR, Afdhal N; SOLAR-1 Investigators. Ledipasvir and Sofosbuvir Plus Ribavirin for Treatment of HCV Infection in Patients With Advanced Liver Disease. Gastroenterology. 2015;149:649-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 688] [Cited by in RCA: 633] [Article Influence: 57.5] [Reference Citation Analysis (7)] |
| 52. | Qu Y, Guo Y, Li T, Ye Q, Sun C, Wang L, Yang B. Efficacy and safety of sofosbuvir-based interferon-free therapies for hepatitis C in liver transplant recipients. J Gastroenterol Hepatol. 2017;32:740-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 53. | European Association for the Study of the Liver; Clinical Practice Guidelines Panel: Chair:; EASL Governing Board representative:; Panel members:. EASL recommendations on treatment of hepatitis C: Final update of the series(☆). J Hepatol. 2020;73:1170-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 999] [Cited by in RCA: 893] [Article Influence: 148.8] [Reference Citation Analysis (2)] |
| 54. | Belli LS, Berenguer M, Cortesi PA, Strazzabosco M, Rockenschaub SR, Martini S, Morelli C, Donato F, Volpes R, Pageaux GP, Coilly A, Fagiuoli S, Amaddeo G, Perricone G, Vinaixa C, Berlakovich G, Facchetti R, Polak W, Muiesan P, Duvoux C; European Liver and Intestine Association (ELITA). Delisting of liver transplant candidates with chronic hepatitis C after viral eradication: A European study. J Hepatol. 2016;65:524-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 255] [Article Influence: 25.5] [Reference Citation Analysis (3)] |
| 55. | Perricone G, Duvoux C, Berenguer M, Cortesi PA, Vinaixa C, Facchetti R, Mazzarelli C, Rockenschaub SR, Martini S, Morelli C, Monico S, Volpes R, Pageaux GP, Fagiuoli S, Belli LS; European Liver and Intestine Transplant Association (ELITA). Delisting HCV-infected liver transplant candidates who improved after viral eradication: Outcome 2 years after delisting. Liver Int. 2018;38:2170-2177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (1)] |
| 56. | Gray E, O'Leary A, Stewart S, Bergin C, Cannon M, Courtney G, Crosbie O, De Gascun CF, Fanning LJ, Feeney E, Houlihan DD, Kelleher B, Lambert JS, Lee J, Mallon P, McConkey S, McCormick A, McKiernan S, McNally C, Murray F, Sheehan G, Norris S; Irish Hepatitis C Outcomes and Research Network (ICORN). High mortality during direct acting antiviral therapy for hepatitis C patients with Child's C cirrhosis: Results of the Irish Early Access Programme. J Hepatol. 2016;65:446-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 57. | Dyson JK, Hutchinson J, Harrison L, Rotimi O, Tiniakos D, Foster GR, Aldersley MA, McPherson S. Liver toxicity associated with sofosbuvir, an NS5A inhibitor and ribavirin use. J Hepatol. 2016;64:234-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 71] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 58. | Mocan T, Nenu I, Crăciun R, Spârchez Z. Treatment of hepatitis C virus infection in patients with hepatocellular carcinoma: Truth or dare? J Gastroenterol Hepatol. 2021;36:1518-1528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 59. | Schmitz KJ, Wohlschlaeger J, Lang H, Sotiropoulos GC, Malago M, Steveling K, Reis H, Cicinnati VR, Schmid KW, Baba HA. Activation of the ERK and AKT signalling pathway predicts poor prognosis in hepatocellular carcinoma and ERK activation in cancer tissue is associated with hepatitis C virus infection. J Hepatol. 2008;48:83-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 244] [Article Influence: 13.6] [Reference Citation Analysis (2)] |
| 60. | Tahmasebi Birgani M, Carloni V. Tumor Microenvironment, a Paradigm in Hepatocellular Carcinoma Progression and Therapy. Int J Mol Sci. 2017;18:405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 145] [Article Influence: 16.1] [Reference Citation Analysis (1)] |
| 61. | Cabibbo G, Petta S, Barbara M, Attardo S, Bucci L, Farinati F, Giannini EG, Negrini G, Ciccarese F, Rapaccini GL, Di Marco M, Caturelli E, Zoli M, Borzio F, Sacco R, Virdone R, Marra F, Mega A, Morisco F, Benvegnù L, Gasbarrini A, Svegliati-Baroni G, Foschi FG, Olivani A, Masotto A, Nardone G, Colecchia A, Persico M, Craxì A, Trevisani F, Cammà C; Italian Liver Cancer (ITA. LI.CA) group. Hepatic decompensation is the major driver of death in HCV-infected cirrhotic patients with successfully treated early hepatocellular carcinoma. J Hepatol. 2017;67:65-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 88] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 62. | Huang AC, Mehta N, Dodge JL, Yao FY, Terrault NA. Direct-acting antivirals do not increase the risk of hepatocellular carcinoma recurrence after local-regional therapy or liver transplant waitlist dropout. Hepatology. 2018;68:449-461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 70] [Article Influence: 8.8] [Reference Citation Analysis (5)] |
| 63. | Choudhary NS, Saigal S, Gautam D, Saraf N, Rastogi A, Goja S, Bhangui P, Soin AS. Efficacy And Safety of Sofosbuvir Based Regimens For Treatment of Hepatitis C Recurrence After Living Donor Liver Transplantation: An Experience From India. J Clin Exp Hepatol. 2018;8:121-124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 64. | Anand AC, Agarwal SK, Garg HK, Khanna S, Gupta S. Sofosbuvir and Ribavirin for 24 Weeks Is An Effective Treatment Option for Recurrent Hepatitis C Infection After Living Donor Liver Transplantation. J Clin Exp Hepatol. 2017;7:165-171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 65. | Higley C, Hsu CC, Smith C, Nadella S, Lalos AT. Safety and efficacy of sofosbuvir/velpatasvir/voxilaprevir in post-liver transplant patients with previous direct-acting antiviral failure: Six case reports. World J Hepatol. 2020;12:1341-1348. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (2)] |
| 66. | Nimgaonkar I, Ding Q, Schwartz RE, Ploss A. Hepatitis E virus: advances and challenges. Nat Rev Gastroenterol Hepatol. 2018;15:96-110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 307] [Cited by in RCA: 264] [Article Influence: 33.0] [Reference Citation Analysis (2)] |
| 67. | van der Eijk AA, Pas SD, de Man RA. Hepatitis E virus: A potential threat for patients with liver disease and liver transplantation. Best Pract Res Clin Gastroenterol. 2017;31:143-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 68. | Donnelly MC, Scobie L, Crossan CL, Dalton H, Hayes PC, Simpson KJ. Review article: hepatitis E-a concise review of virology, epidemiology, clinical presentation and therapy. Aliment Pharmacol Ther. 2017;46:126-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 68] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 69. | Aggarwal A, Perumpail RB, Tummala S, Ahmed A. Hepatitis E virus infection in the liver transplant recipients: Clinical presentation and management. World J Hepatol. 2016;8:117-122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 70. | Hoofnagle JH, Nelson KE, Purcell RH. Hepatitis E. N Engl J Med. 2012;367:1237-1244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 380] [Article Influence: 27.1] [Reference Citation Analysis (2)] |
| 71. | Liang Z, Shu J, He Q, Zhang F, Dai L, Wang L, Lu F, Wang L. High dose sofosbuvir and sofosbuvir-plus-ribavirin therapy inhibit Hepatitis E Virus (HEV) replication in a rabbit model for acute HEV infection. Antiviral Res. 2022;199:105274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 72. | Kamar N, Pan Q. No Clear Evidence for an Effect of Sofosbuvir Against Hepatitis E Virus in Organ Transplant Patients. Hepatology. 2019;69:1846-1847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 73. | Webb GW, Dalton HR. Hepatitis E: an underestimated emerging threat. Ther Adv Infect Dis. 2019;6:2049936119837162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 69] [Article Influence: 9.9] [Reference Citation Analysis (1)] |
| 74. | Reekie I, Irish D, Ijaz S, Fox T, Bharucha T, Griffiths P, Thorburn D, Harber M, MacKinnon S, Sekhar M. Hepatitis E infection in stem cell and solid organ transplantpatients: A cross-sectional study: The importance of HEV RNA screening in peri-transplant period. J Clin Virol. 2018;107:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 75. | de Vos AS, Janssen MP, Zaaijer HL, Hogema BM. Cost-effectiveness of the screening of blood donations for hepatitis E virus in the Netherlands. Transfusion. 2017;57:258-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (2)] |
| 76. | Kumar A, Saraswat VA. Hepatitis E and Acute-on-Chronic Liver Failure. J Clin Exp Hepatol. 2013;3:225-230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 65] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 77. | Navaneethan U, Al Mohajer M, Shata MT. Hepatitis E and pregnancy: understanding the pathogenesis. Liver Int. 2008;28:1190-1199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 325] [Cited by in RCA: 275] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 78. | Wedemeyer H, Pischke S, Manns MP. Pathogenesis and treatment of hepatitis e virus infection. Gastroenterology. 2012;142:1388-1397.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 219] [Article Influence: 15.6] [Reference Citation Analysis (2)] |
| 79. | Kamar N, Garrouste C, Haagsma EB, Garrigue V, Pischke S, Chauvet C, Dumortier J, Cannesson A, Cassuto-Viguier E, Thervet E, Conti F, Lebray P, Dalton HR, Santella R, Kanaan N, Essig M, Mousson C, Radenne S, Roque-Afonso AM, Izopet J, Rostaing L. Factors associated with chronic hepatitis in patients with hepatitis E virus infection who have received solid organ transplants. Gastroenterology. 2011;140:1481-1489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 577] [Cited by in RCA: 502] [Article Influence: 33.5] [Reference Citation Analysis (2)] |
| 80. | Kamar N, Abravanel F, Selves J, Garrouste C, Esposito L, Lavayssière L, Cointault O, Ribes D, Cardeau I, Nogier MB, Mansuy JM, Muscari F, Peron JM, Izopet J, Rostaing L. Influence of immunosuppressive therapy on the natural history of genotype 3 hepatitis-E virus infection after organ transplantation. Transplantation. 2010;89:353-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 173] [Article Influence: 10.8] [Reference Citation Analysis (4)] |
| 81. | Kamar N, Rostaing L, Izopet J. Hepatitis E virus infection in immunosuppressed patients: natural history and therapy. Semin Liver Dis. 2013;33:62-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 79] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 82. | Prost S, Crossan CL, Dalton HR, De Man RA, Kamar N, Selves J, Dhaliwal C, Scobie L, Bellamy COC. Detection of viral hepatitis E in clinical liver biopsies. Histopathology. 2017;71:580-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 83. | Verna EC. Hepatitis viruses and liver transplantation: evolving trends in antiviral management. Clin Liver Dis. 2014;18:575-601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 84. | Debing Y, Gisa A, Dallmeier K, Pischke S, Bremer B, Manns M, Wedemeyer H, Suneetha PV, Neyts J. A mutation in the hepatitis E virus RNA polymerase promotes its replication and associates with ribavirin treatment failure in organ transplant recipients. Gastroenterology. 2014;147:1008-11.e7; quiz e15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 174] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 85. | Pischke S, Hardtke S, Bode U, Birkner S, Chatzikyrkou C, Kauffmann W, Bara CL, Gottlieb J, Wenzel J, Manns MP, Wedemeyer H. Ribavirin treatment of acute and chronic hepatitis E: a single-centre experience. Liver Int. 2013;33:722-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 141] [Article Influence: 10.8] [Reference Citation Analysis (1)] |
| 86. | Tavitian S, Peron JM, Huguet F, Kamar N, Abravanel F, Beyne-Rauzy O, Oberic L, Faguer S, Alric L, Roussel M, Gaudin C, Ysebaert L, Huynh A, Recher C. Ribavirin for Chronic Hepatitis Prevention among Patients with Hematologic Malignancies. Emerg Infect Dis. 2015;21:1466-1469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 87. | Kamar N, Izopet J, Tripon S, Bismuth M, Hillaire S, Dumortier J, Radenne S, Coilly A, Garrigue V, D'Alteroche L, Buchler M, Couzi L, Lebray P, Dharancy S, Minello A, Hourmant M, Roque-Afonso AM, Abravanel F, Pol S, Rostaing L, Mallet V. Ribavirin for chronic hepatitis E virus infection in transplant recipients. N Engl J Med. 2014;370:1111-1120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 459] [Cited by in RCA: 386] [Article Influence: 32.2] [Reference Citation Analysis (2)] |
| 88. | Haagsma EB, Riezebos-Brilman A, van den Berg AP, Porte RJ, Niesters HG. Treatment of chronic hepatitis E in liver transplant recipients with pegylated interferon alpha-2b. Liver Transpl. 2010;16:474-477. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 80] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 89. | Rizzetto M. Hepatitis D Virus: Introduction and Epidemiology. Cold Spring Harb Perspect Med. 2015;5:a021576. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 141] [Cited by in RCA: 107] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 90. | Taylor JM. Infection by Hepatitis Delta Virus. Viruses. 2020;12:648. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 91. | Stockdale AJ, Kreuels B, Henrion MYR, Giorgi E, Kyomuhangi I, de Martel C, Hutin Y, Geretti AM. The global prevalence of hepatitis D virus infection: Systematic review and meta-analysis. J Hepatol. 2020;73:523-532. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 583] [Cited by in RCA: 512] [Article Influence: 85.3] [Reference Citation Analysis (1)] |
| 92. | Miao Z, Zhang S, Ou X, Li S, Ma Z, Wang W, Peppelenbosch MP, Liu J, Pan Q. Estimating the Global Prevalence, Disease Progression, and Clinical Outcome of Hepatitis Delta Virus Infection. J Infect Dis. 2020;221:1677-1687. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 301] [Cited by in RCA: 274] [Article Influence: 45.7] [Reference Citation Analysis (1)] |
| 93. | Rizzetto M, Hamid S, Negro F. The changing context of hepatitis D. J Hepatol. 2021;74:1200-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 134] [Article Influence: 26.8] [Reference Citation Analysis (2)] |
| 94. | Kushner T, Da BL, Chan A, Dieterich D, Sigel K, Saberi B. Liver Transplantation for Hepatitis D Virus in the United States: A UNOS Study on Outcomes in the MELD Era. Transplant Direct. 2022;8:e1253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 95. | Kushner T, Serper M, Kaplan DE. Delta hepatitis within the Veterans Affairs medical system in the United States: Prevalence, risk factors, and outcomes. J Hepatol. 2015;63:586-592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 137] [Article Influence: 12.5] [Reference Citation Analysis (1)] |
| 96. | Safaie P, Razeghi S, Rouster SD, Privitera I, Sherman KE. Hepatitis D diagnostics:Utilization and testing in the United States. Virus Res. 2018;250:114-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 97. | Ottobrelli A, Marzano A, Smedile A, Recchia S, Salizzoni M, Cornu C, Lamy ME, Otte JB, De Hemptinne B, Geubel A. Patterns of hepatitis delta virus reinfection and disease in liver transplantation. Gastroenterology. 1991;101:1649-1655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 101] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 98. | Caccamo L. Long-term nucleos(t)ide analog(s) monoprophylaxis in Delta coinfected liver transplant recipients. Transpl Infect Dis. 2017;19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 99. | Cholongitas E, Goulis I, Antoniadis N, Fouzas I, Imvrios G, Giakoustidis D, Giouleme O, Papanikolaou V, Akriviadis E, Vasiliadis T. Nucleos(t)ide analog(s) prophylaxis after hepatitis B immunoglobulin withdrawal against hepatitis B and D recurrence after liver transplantation. Transpl Infect Dis. 2016;18:667-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 100. | Fernández I, Loinaz C, Hernández O, Abradelo M, Manrique A, Calvo J, Manzano M, García A, Cambra F, Castellano G, Jiménez C. Tenofovir/entecavir monotherapy after hepatitis B immunoglobulin withdrawal is safe and effective in the prevention of hepatitis B in liver transplant recipients. Transpl Infect Dis. 2015;17:695-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 101. | Wedemeyer H, Aleman S, Brunetto MR, Blank A, Andreone P, Bogomolov P, Chulanov V, Mamonova N, Geyvandova N, Morozov V, Sagalova O, Stepanova T, Berger A, Manuilov D, Suri V, An Q, Da B, Flaherty J, Osinusi A, Liu Y, Merle U, Schulze Zur Wiesch J, Zeuzem S, Ciesek S, Cornberg M, Lampertico P; MYR 301 Study Group. A Phase 3, Randomized Trial of Bulevirtide in Chronic Hepatitis D. N Engl J Med. 2023;389:22-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 169] [Article Influence: 56.3] [Reference Citation Analysis (1)] |
| 102. | Beam E, Razonable RR. Cytomegalovirus in solid organ transplantation: epidemiology, prevention, and treatment. Curr Infect Dis Rep. 2012;14:633-641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 94] [Article Influence: 6.7] [Reference Citation Analysis (2)] |
| 103. | Cannon MJ, Schmid DS, Hyde TB. Review of cytomegalovirus seroprevalence and demographic characteristics associated with infection. Rev Med Virol. 2010;20:202-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1216] [Cited by in RCA: 1056] [Article Influence: 66.0] [Reference Citation Analysis (5)] |
| 104. | Zuhair M, Smit GSA, Wallis G, Jabbar F, Smith C, Devleesschauwer B, Griffiths P. Estimation of the worldwide seroprevalence of cytomegalovirus: A systematic review and meta-analysis. Rev Med Virol. 2019;29:e2034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 779] [Cited by in RCA: 658] [Article Influence: 94.0] [Reference Citation Analysis (1)] |
| 105. | Razonable R. Direct and indirect effects of cytomegalovirus: can we prevent them? Enferm Infecc Microbiol Clin. 2010;28:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 106. | Razonable RR, Paya CV, Smith TF. Role of the laboratory in diagnosis and management of cytomegalovirus infection in hematopoietic stem cell and solid-organ transplant recipients. J Clin Microbiol. 2002;40:746-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 108] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 107. | Lautenschlager I, Halme L, Höckerstedt K, Krogerus L, Taskinen E. Cytomegalovirus infection of the liver transplant: virological, histological, immunological, and clinical observations. Transpl Infect Dis. 2006;8:21-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 50] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 108. | Verma Y, Gupta E, Kumar N, Hasnain N, Bhadoria AS, Pamecha V, Khanna R. Earlier and higher rates of cytomegalovirus infection in pediatric liver transplant recipients as compared to adults: An observational study. J Lab Physicians. 2018;10:221-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 109. | Gala-Lopez BL, Senior PA, Koh A, Kashkoush SM, Kawahara T, Kin T, Humar A, Shapiro AM. Late cytomegalovirus transmission and impact of T-depletion in clinical islet transplantation. Am J Transplant. 2011;11:2708-2714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 1.8] [Reference Citation Analysis (2)] |
| 110. | Portela D, Patel R, Larson-Keller JJ, Ilstrup DM, Wiesner RH, Steers JL, Krom RA, Paya CV. OKT3 treatment for allograft rejection is a risk factor for cytomegalovirus disease in liver transplantation. J Infect Dis. 1995;171:1014-1018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 90] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 111. | Brennan DC, Legendre C, Patel D, Mange K, Wiland A, McCague K, Shihab FS. Cytomegalovirus incidence between everolimus versus mycophenolate in de novo renal transplants: pooled analysis of three clinical trials. Am J Transplant. 2011;11:2453-2462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 124] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 112. | Ambrose T, Sharkey LM, Louis-Auguste J, Rutter CS, Duncan S, English S, Gkrania-Klotsas E, Carmichael A, Woodward JM, Russell N, Massey D, Butler A, Middleton S. Cytomegalovirus Infection and Rates of Antiviral Resistance Following Intestinal and Multivisceral Transplantation. Transplant Proc. 2016;48:492-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 113. | Beam E, Lesnick T, Kremers W, Kennedy CC, Razonable RR. Cytomegalovirus disease is associated with higher all-cause mortality after lung transplantation despite extended antiviral prophylaxis. Clin Transplant. 2016;30:270-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 56] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 114. | Nagai S, Mangus RS, Anderson E, Ekser B, Kubal CA, Fridell JA, Tector AJ. Cytomegalovirus Infection After Intestinal/Multivisceral Transplantation: A Single-Center Experience With 210 Cases. Transplantation. 2016;100:451-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 115. | Singh N, Wagener MM. Strategies to prevent organ disease by cytomegalovirus in solid organ transplant recipients. Ann Intern Med. 2006;144:456-7; author reply 457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 116. | Razonable RR. Cytomegalovirus infection after liver transplantation: current concepts and challenges. World J Gastroenterol. 2008;14:4849-4860. [PubMed] [DOI] [Full Text] |
| 117. | Lizaola-Mayo BC, Rodriguez EA. Cytomegalovirus infection after liver transplantation. World J Transplant. 2020;10:183-190. [PubMed] [DOI] [Full Text] |
| 118. | Razonable RR, Humar A. Cytomegalovirus in solid organ transplant recipients-Guidelines of the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant. 2019;33:e13512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 664] [Cited by in RCA: 550] [Article Influence: 78.6] [Reference Citation Analysis (2)] |
| 119. | Razonable RR, Emery VC; 11th Annual Meeting of the IHMF (International Herpes Management Forum). Management of CMV infection and disease in transplant patients. 27-29 February 2004. Herpes. 2004;11:77-86. [PubMed] |
| 120. | Paya CV, Hermans PE, Wiesner RH, Ludwig J, Smith TF, Rakela J, Krom RA. Cytomegalovirus hepatitis in liver transplantation: prospective analysis of 93 consecutive orthotopic liver transplantations. J Infect Dis. 1989;160:752-758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 95] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 121. | Shusterman NH, Frauenhoffer C, Kinsey MD. Fatal massive hepatic necrosis in cytomegalovirus mononucleosis. Ann Intern Med. 1978;88:810-812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 31] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 122. | Tjwa M, De Hertogh G, Neuville B, Roskams T, Nevens F, Van Steenbergen W. Hepatic fibrin-ring granulomas in granulomatous hepatitis: report of four cases and review of the literature. Acta Clin Belg. 2001;56:341-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 20] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 123. | You DM, Johnson MD. Cytomegalovirus infection and the gastrointestinal tract. Curr Gastroenterol Rep. 2012;14:334-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 72] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 124. | Patra S, Samal SC, Chacko A, Mathan VI, Mathan MM. Cytomegalovirus infection of the human gastrointestinal tract. J Gastroenterol Hepatol. 1999;14:973-976. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 125. | Aguilera V, Di Maira T, Conde I, Fornés-Ferrer V, Vinaixa C, Pallarés C, Carvalho-Gomes A, Cubells A, García M, Rubín Á, Benlloch S, Gonzalez-Dieguez L, Molina JM, Puchades L, López-Labrador FX, Prieto M, Berenguer M. Cytomegalovirus reactivation in liver transplant recipients due to hepatitis C cirrhosis is associated with higher cardiovascular risk - an observational, retrospective study. Transpl Int. 2018;31:649-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 126. | Razonable RR, Brown RA, Wilson J, Groettum C, Kremers W, Espy M, Smith TF, Paya CV. The clinical use of various blood compartments for cytomegalovirus (CMV) DNA quantitation in transplant recipients with CMV disease. Transplantation. 2002;73:968-973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 115] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 127. | Dioverti MV, Razonable RR. Cytomegalovirus. Microbiol Spectr. 2016;4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 98] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 128. | Lowance D, Neumayer HH, Legendre CM, Squifflet JP, Kovarik J, Brennan PJ, Norman D, Mendez R, Keating MR, Coggon GL, Crisp A, Lee IC. Valacyclovir for the prevention of cytomegalovirus disease after renal transplantation. International Valacyclovir Cytomegalovirus Prophylaxis Transplantation Study Group. N Engl J Med. 1999;340:1462-1470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 599] [Cited by in RCA: 505] [Article Influence: 18.7] [Reference Citation Analysis (1)] |
| 129. | Asberg A, Humar A, Rollag H, Jardine AG, Mouas H, Pescovitz MD, Sgarabotto D, Tuncer M, Noronha IL, Hartmann A; VICTOR Study Group. Oral valganciclovir is noninferior to intravenous ganciclovir for the treatment of cytomegalovirus disease in solid organ transplant recipients. Am J Transplant. 2007;7:2106-2113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 371] [Cited by in RCA: 295] [Article Influence: 15.5] [Reference Citation Analysis (2)] |
| 130. | Dunmire SK, Verghese PS, Balfour HH Jr. Primary Epstein-Barr virus infection. J Clin Virol. 2018;102:84-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 324] [Article Influence: 40.5] [Reference Citation Analysis (1)] |
| 131. | Houen G, Trier NH. Epstein-Barr Virus and Systemic Autoimmune Diseases. Front Immunol. 2020;11:587380. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 305] [Cited by in RCA: 254] [Article Influence: 50.8] [Reference Citation Analysis (2)] |
| 132. | Ascherio A, Munger KL, Lünemann JD. The initiation and prevention of multiple sclerosis. Nat Rev Neurol. 2012;8:602-612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 244] [Cited by in RCA: 202] [Article Influence: 14.4] [Reference Citation Analysis (1)] |
| 133. | Hjalgrim H, Askling J, Rostgaard K, Hamilton-Dutoit S, Frisch M, Zhang JS, Madsen M, Rosdahl N, Konradsen HB, Storm HH, Melbye M. Characteristics of Hodgkin's lymphoma after infectious mononucleosis. N Engl J Med. 2003;349:1324-1332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 344] [Cited by in RCA: 276] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 134. | Okamoto T, Okajima H, Uebayashi EY, Ogawa E, Yamada Y, Umeda K, Hiramatsu H, Hatano E. Management of Epstein-Barr Virus Infection and Post-Transplant Lymphoproliferative Disorder in Pediatric Liver Transplantation. J Clin Med. 2022;11:2166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 135. | Quintero Bernabeu J, Juamperez J, Mercadal-Hally M, Larrarte King M, Gallego Melcon S, Gros Subias L, Sábado Álvarez C, Soler-Palacin P, Melendo Pérez S, Esperalba J, Navarro Jiménez A, Garrido Pontnou M, Camacho Soriano J, Hidalgo Llompart E, Bilbao Aguirre I, Charco Torra R. Epstein-Barr virus-associated risk factors for post-transplant lymphoproliferative disease in pediatric liver transplant recipients. Pediatr Transplant. 2022;26:e14292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 136. | Jain A, Nalesnik M, Reyes J, Pokharna R, Mazariegos G, Green M, Eghtesad B, Marsh W, Cacciarelli T, Fontes P, Abu-Elmagd K, Sindhi R, Demetris J, Fung J. Posttransplant lymphoproliferative disorders in liver transplantation: a 20-year experience. Ann Surg. 2002;236:429-36; discussion 436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 203] [Cited by in RCA: 172] [Article Influence: 7.2] [Reference Citation Analysis (2)] |
| 137. | Yu H, Robertson ES. Epstein-Barr Virus History and Pathogenesis. Viruses. 2023;15:714. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 93] [Article Influence: 31.0] [Reference Citation Analysis (1)] |
| 138. | Cen H, Williams PA, McWilliams HP, Breinig MC, Ho M, McKnight JL. Evidence for restricted Epstein-Barr virus latent gene expression and anti-EBNA antibody response in solid organ transplant recipients with posttransplant lymphoproliferative disorders. Blood. 1993;81:1393-1403. [PubMed] |
| 139. | Allen UD, Preiksaitis JK; AST Infectious Diseases Community of Practice. Post-transplant lymphoproliferative disorders, Epstein-Barr virus infection, and disease in solid organ transplantation: Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant. 2019;33:e13652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 255] [Article Influence: 36.4] [Reference Citation Analysis (4)] |
| 140. | Roberts J, Powell J, Mather MW, Powell S, Brodlie M. A review of adenotonsillar hypertrophy and adenotonsillectomy in children after solid organ transplantation. Int J Pediatr Otorhinolaryngol. 2018;114:29-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 141. | Akbas A, Tiede C, Lemound J, Maecker-Kolhoff B, Kreipe H, Hussein K. Post-transplant lymphoproliferative disorders with naso- and oropharyngeal manifestation. Transpl Int. 2015;28:1299-1307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 142. | Rubinstein J, Toner K, Gross T, Wistinghausen B. Diagnosis and management of post-transplant lymphoproliferative disease following solid organ transplantation in children, adolescents, and young adults. Best Pract Res Clin Haematol. 2023;36:101446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 143. | Damania B, Kenney SC, Raab-Traub N. Epstein-Barr virus: Biology and clinical disease. Cell. 2022;185:3652-3670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 484] [Cited by in RCA: 407] [Article Influence: 101.8] [Reference Citation Analysis (0)] |
| 144. | Montanari F, Orjuela-Grimm M. Joining Efforts for PTLD: Lessons Learned from Comparing the Approach and Treatment Strategies Across the Pediatric and Adult Age Spectra. Curr Hematol Malig Rep. 2021;16:52-60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 145. | Alaggio R, Amador C, Anagnostopoulos I, Attygalle AD, Araujo IBO, Berti E, Bhagat G, Borges AM, Boyer D, Calaminici M, Chadburn A, Chan JKC, Cheuk W, Chng WJ, Choi JK, Chuang SS, Coupland SE, Czader M, Dave SS, de Jong D, Du MQ, Elenitoba-Johnson KS, Ferry J, Geyer J, Gratzinger D, Guitart J, Gujral S, Harris M, Harrison CJ, Hartmann S, Hochhaus A, Jansen PM, Karube K, Kempf W, Khoury J, Kimura H, Klapper W, Kovach AE, Kumar S, Lazar AJ, Lazzi S, Leoncini L, Leung N, Leventaki V, Li XQ, Lim MS, Liu WP, Louissaint A Jr, Marcogliese A, Medeiros LJ, Michal M, Miranda RN, Mitteldorf C, Montes-Moreno S, Morice W, Nardi V, Naresh KN, Natkunam Y, Ng SB, Oschlies I, Ott G, Parrens M, Pulitzer M, Rajkumar SV, Rawstron AC, Rech K, Rosenwald A, Said J, Sarkozy C, Sayed S, Saygin C, Schuh A, Sewell W, Siebert R, Sohani AR, Tooze R, Traverse-Glehen A, Vega F, Vergier B, Wechalekar AD, Wood B, Xerri L, Xiao W. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia. 2022;36:1720-1748. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3118] [Cited by in RCA: 2688] [Article Influence: 672.0] [Reference Citation Analysis (2)] |
| 146. | Campo E, Jaffe ES, Cook JR, Quintanilla-Martinez L, Swerdlow SH, Anderson KC, Brousset P, Cerroni L, de Leval L, Dirnhofer S, Dogan A, Feldman AL, Fend F, Friedberg JW, Gaulard P, Ghia P, Horwitz SM, King RL, Salles G, San-Miguel J, Seymour JF, Treon SP, Vose JM, Zucca E, Advani R, Ansell S, Au WY, Barrionuevo C, Bergsagel L, Chan WC, Cohen JI, d'Amore F, Davies A, Falini B, Ghobrial IM, Goodlad JR, Gribben JG, Hsi ED, Kahl BS, Kim WS, Kumar S, LaCasce AS, Laurent C, Lenz G, Leonard JP, Link MP, Lopez-Guillermo A, Mateos MV, Macintyre E, Melnick AM, Morschhauser F, Nakamura S, Narbaitz M, Pavlovsky A, Pileri SA, Piris M, Pro B, Rajkumar V, Rosen ST, Sander B, Sehn L, Shipp MA, Smith SM, Staudt LM, Thieblemont C, Tousseyn T, Wilson WH, Yoshino T, Zinzani PL, Dreyling M, Scott DW, Winter JN, Zelenetz AD. The International Consensus Classification of Mature Lymphoid Neoplasms: a report from the Clinical Advisory Committee. Blood. 2022;140:1229-1253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1308] [Cited by in RCA: 1164] [Article Influence: 291.0] [Reference Citation Analysis (2)] |
| 147. | DeStefano CB, Desai SH, Shenoy AG, Catlett JP. Management of post-transplant lymphoproliferative disorders. Br J Haematol. 2018;182:330-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 67] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 148. | Chen EY, Dilwali N, Mysore KR, Hassan S, Smith SK, Karnsakul W. Navigating Epstein-Barr Virus (EBV) and Post-Transplant Lymphoproliferative Disorder (PTLD) in Pediatric Liver Transplantation: Current Knowledge and Strategies for Treatment and Surveillance. Viruses. 2025;17:254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (1)] |