Published online May 19, 2026. doi: 10.5498/wjp.v16.i5.117187
Revised: January 9, 2026
Accepted: February 12, 2026
Published online: May 19, 2026
Processing time: 141 Days and 0.6 Hours
Hepatocellular carcinoma (HCC), a prevalent primary liver cancer with high mortality, is often linked to chronic liver disease. Despite advances in treatment, prognosis remains poor due to high recurrence and metastasis. Alcohol dehydrogenase 4 (ADH4), involved in ethanol metabolism, is understudied in HCC regarding its expression, clinical relevance, and prognosis. Additionally, psychological distress in HCC patients may affect both quality of life and disease progr
To investigate the expression level of ADH4 in HCC and its relationship with tumor invasiveness, patient survival prognosis, and anxiety-depression status to provide evidence for the clinical diagnosis, treatment, and comprehensive mana
A retrospective cohort study design was adopted, including 148 patients with HCC who underwent surgical resection between January 2018 and January 2023 with complete clinicopathological data and follow-up inf
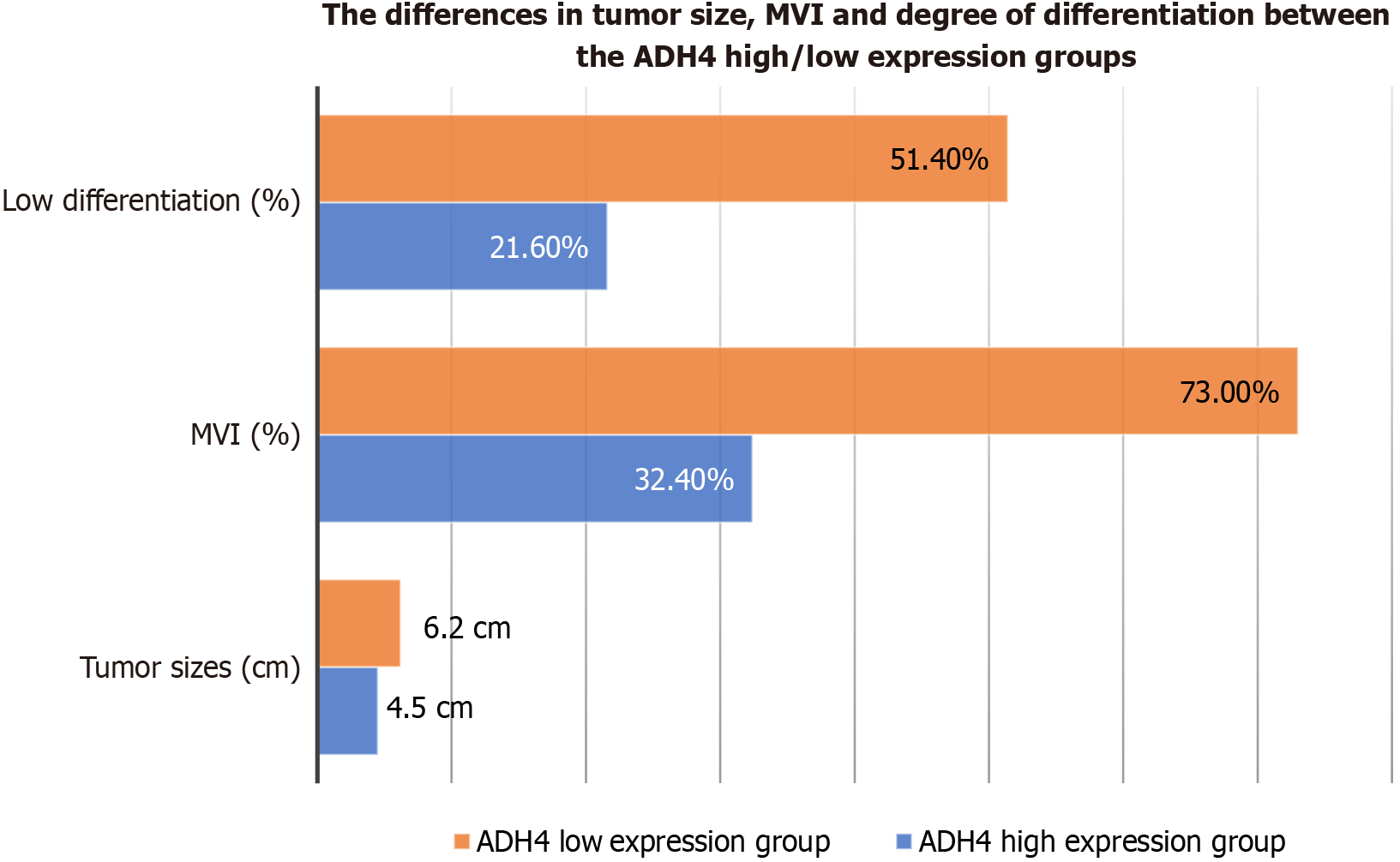
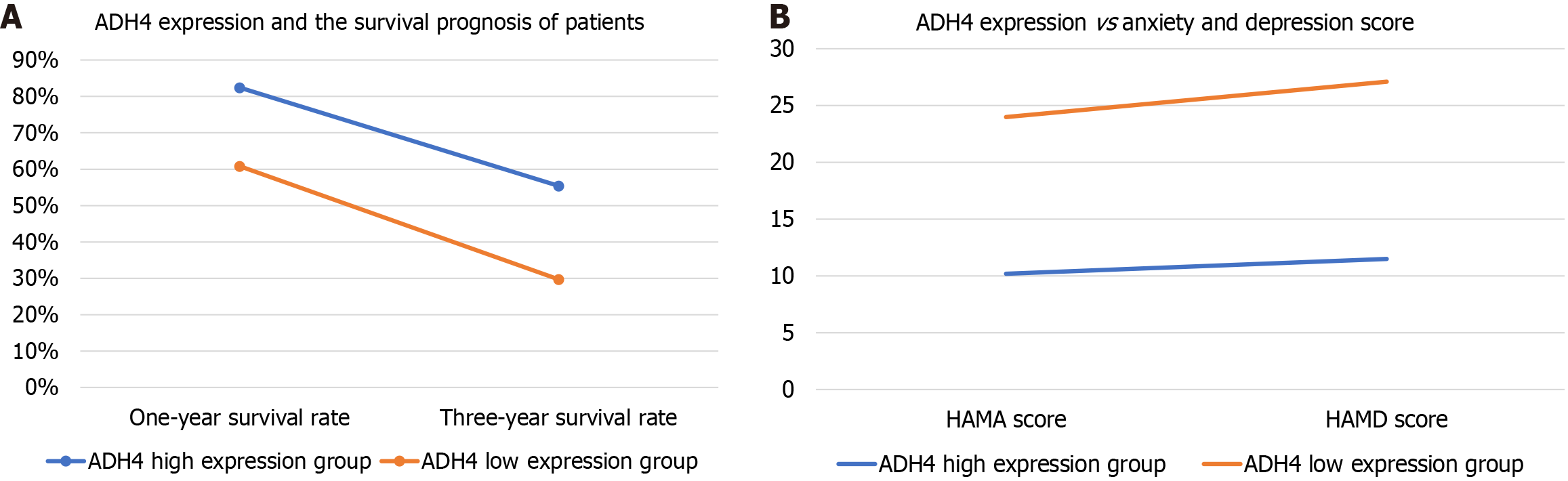
The mean maximum tumor diameter in the ADH4 low expression group (6.2 ± 2.5 cm) was significantly larger than that in the high expression group (4.5 ± 1.8 cm) (t = -4.218, P < 0.001). ADH4 expression was positively correlated with the degree of tumor differentiation (r = 0.432, P < 0.001), and the proportion of high ADH4 expression in well-differentiated tumors (76.9%) was significantly higher than that in poorly differentiated tumors (27.3%). The MVI positivity rate in the low expression group (73.0%) was significantly higher than that in the high expression group (32.4%) (χ² = 22.563, P < 0.001). Survival analysis showed that the 1-year and 3-year survival rates in the high expression group (82.4%, 55.4%) were significantly higher than those in the low expression group (60.8%, 29.7%) (χ² = 15.347, P < 0.001). Multivariate Cox regression analysis confirmed that low ADH4 expression [hazard ratio (HR) = 0.486, 95%CI: 0.305-0.775, P = 0.002], large tumor size (HR = 1.768, 95%CI: 1.135-2.750, P = 0.012), and MVI positivity (HR = 2.047, 95%CI: 1.315-3.186, P = 0.002) were independent risk factors for poor prognosis in patients with HCC. Psychological assessment revealed that HAMA and HAMD scores in the low expression group (13.8 ± 4.2 points, 15.6 ± 4.5 points) were significantly higher than those in the high expression group (10.2 ± 3.5 points, 11.5 ± 3.8 points) (t = -5.217, t = -5.528, respectively, both P < 0.001), and ADH4 expression was negatively correlated with both HAMA and HAMD scores (r = -0.456, r = -0.482, respectively, both P < 0.001).
Low ADH4 expression in HCC tissues is closely associated with enhanced tumor invasiveness, larger tumor volume, lower degree of differentiation, and higher MVI incidence. Patients with high ADH4 expression have better anxiety and depression states but poor survival prognosis. ADH4 may be a potential biomarker for assessing the severity, prognosis, and psychological status of patients with HCC, providing an important reference for per
Core Tip: Low alcohol dehydrogenase 4 (ADH4) expression in hepatocellular carcinoma correlates with larger tumors, poor differentiation, high microvascular invasion, reduced 1-year and 3-year survival, and elevated anxiety-depression scores. Multivariate analysis identifies low ADH4 as an independent poor prognostic factor. These findings suggest ADH4 is a valuable biomarker for assessing tumor aggressiveness, predicting outcomes, and identifying patients requiring intensive therapy and psychosocial support, thus enabling personalized oncological and mental-health interventions to improve overall prognosis and quality of life.
- Citation: Xia T, Liu CL, Zhang JC, Meng P, Ma YB, Huang QK, Li HM, Zhao XY, Wang C, Zhao G, Kong LH. Alcohol dehydrogenase 4 expression in hepatocellular carcinoma and its association with tumor invasion, survival, and psychological status. World J Psychiatry 2026; 16(5): 117187
- URL: https://www.wjgnet.com/2220-3206/full/v16/i5/117187.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i5.117187
Hepatocellular carcinoma (HCC) is the most common histological type of primary liver cancer and poses a serious threat to human health. According to data released by the World Health Organization’s International Agency for Research on Cancer[1], approximately 906000 new cases of liver cancer will be reported globally by 2022, with HCC accounting for over 80% of all cases and 830000 deaths. Approximately 410000 new HCC cases will be reported in China in 2022, with HCC ranking fifth in malignancy incidence and second in mortality in China[2]. The high incidence and mortality of HCC impose heavy economic and mental burdens on the patients’ families and society.
Alcohol dehydrogenase 4 (ADH4), an important member of the alcohol dehydrogenase family, plays a key role in physiological processes, such as ethanol and retinol metabolism[3]. ADH4 plays a significant role in the occurrence and development of various tumors[3]. Research on breast cancer has reported that ADH4 expression levels are related to tumor cell proliferation and invasion capabilities. In gastric cancer tissues, low ADH4 expression is closely associated with poor patient prognosis[4]. However, research on ADH4 in HCC remains relatively scarce and the relationship between its expression levels, HCC invasiveness, and patient survival rates is not fully understood[5].
An in-depth investigation of ADH4 expression in HCC is of great significance for revealing the pathogenesis of HCC, identifying effective prognostic markers, and developing new therapeutic targets. Simultaneously, focusing on the anxiety and depression statuses of patients with HCC and analyzing their association with ADH4 expression helps to comprehensively understand the physical and mental health of patients, providing a basis for developing more comprehensive integrated treatment plans, thereby improving patients’ quality of life and survival rates[6,7].
This study aimed to analyze the clinicopathological data and follow-up information of patients with HCC to clarify the relationship between ADH4 expression and HCC invasiveness, survival rate, and anxiety-depression status. The specific research objectives were to analyze the correlation between ADH4 expression and clinicopathological parameters [such as tumor size, degree of differentiation, and microvascular invasion (MVI)][8] in patients with HCC, evaluate the impact of ADH4 expression on the survival prognosis of patients with HCC, and explore the association between ADH4 expression and the anxiety-depression status of patients with HCC, focusing on their psychological health status.
We proposed the following hypotheses: (1) ADH4 expression levels are correlated with HCC invasiveness, and low ADH4 expression may promote tumor invasion and metastasis; (2) ADH4 expression level is closely related to patient survival rate; (3) Patients expressing low levels of ADH4 have lower survival rates; (4) ADH4 expression is associated with patient anxiety-depression status; and (5) Patients with low ADH4 expression may be more prone to adverse psy
This study used a retrospective cohort design, including 148 patients with HCC with complete clinicopathological data and follow-up information who underwent surgical resection at the Air Force Medical Center between January 2018 and January 2023. The patients did not receive radiotherapy, chemotherapy, or other antitumor treatments before surgery.
Immunohistochemical staining results for ADH4 were used as the primary grouping criteria. Based on the immunohistochemistry score (H-score or Allred score)[9], patients with scores above the median or optimal cut-off value were included in the high ADH4 expression group (n = 74); otherwise, they were included in the low/negative ADH4 expr
The specific scoring criteria were as follows. Determination was mainly based on two dimensions: (1) Staining intensity; and (2) Proportion of positive cells. The staining intensity was divided into no staining (0 point), light yellow (1 point), brownish-yellow (2 points), and brownish-red (3 points). The proportion of positive cells was classified as follows: (1) < 5% (0 point); (2) 5%-25% (1 point); (3) 26%-50% (2 points); (4) 51%-75% (3 points); and (5) > 75% (4 points). The final ADH4 expression score was determined as the product of these two scores. Generally, a score ≥ 3 is defined as high expression, and a score < 3 is defined as low/negative expression. However, differences in antibody concentration, antigen remediation methods, and scoring thresholds between studies may lead to deviations in the determination of ADH4 expression levels.
The inclusion criteria were as follows: (1) HCC diagnosed by surgical resection and postoperative pathology; (2) Age between 18 years and 75 years; (3) Complete clinicopathological data, including preoperative laboratory results such as liver function, hepatitis B virus (HBV) serology, and alpha-fetoprotein levels; (4) Imaging data such as liver ultrasound, computed tomography, magnetic resonance imaging, intraoperative findings, and postoperative pathology reports; and (5) Complete follow-up information with a follow-up time of no less than 6 months.
The exclusion criteria were as follows: (1) Received preoperative radiotherapy, chemotherapy, targeted therapy, or immunotherapy; (2) Presence of other malignant tumors; (3) Presence of severe underlying diseases such as hepatic or renal insufficiency, cardiopulmonary dysfunction affecting survival, and psychological status assessment; and (4) Inco
Patient clinicopathological data, including age, sex, tumor size, tumor number, tumor location, degree of differentiation, MVI status, HBV infection status, cirrhosis status, and alpha-fetoprotein level, were collected using the hospital’s electronic medical record system and pathology department information system. Follow-up information was obtained through outpatient review, telephone follow-up, and recording of patient survival status, recurrence time, and time of death.
The main observation indicators were as follows: (1) Correlation between ADH4 expression and clinicopathological parameters, including age, sex, tumor size, tumor number, differentiation degree, MVI, HBV infection status, and cirrhosis status; (2) Relationship between ADH4 expression and patient survival prognosis using the Kaplan-Meier method to draw survival curves, the log-rank test to compare survival rate differences between the two groups[10], and Cox regression analysis to analyze independent risk factors affecting patient survival prognosis; and (3) Association analysis between ADH4 expression and patient anxiety-depression status using the Hamilton Anxiety Scale (HAMA) and the Hamilton Depression Scale (HAMD) to assess patients’ psychological status one month postoperatively.
The HAMA is used to evaluate the severity of anxiety symptoms in patients with anxiety and depressive disorders. All items were scored on a five-level scale ranging from 0 to 4 points. The criteria for each level were as follows: (1) 0 point: Asymptomatic; (2) 1 point: Light; (3) 2 points: Medium; (4) 3 points: Heavy; and (5) 4 points: Extremely heavy. A total score of 29 or higher indicates severe anxiety. A score of 21 or higher indicated obvious anxiety. A score of 14 or higher indicated anxiety. A score greater than 7 indicates anxiety. If the score was less than 7, there were no anxiety symptoms.
The HAMD is mainly used to assess the severity of neurotic and depressive symptoms (0: None; 1: Mild; 2: Moderate; 3: Severe; 4: Very serious). A few items were scored on a three-point scale ranging from 0 to 2 (0: None; 1: Suspicious or mild; 2: Obvious symptoms). A diagnosis of less than 8 points was considered normal. A score of 8-20 indicates possible depression. Depression was diagnosed at 21 points and 35 points. A score of > 35 points indicates severe depression.
SPSS (version 25.0; IBM, Armonk, NY, United States) was used for data processing. Measurement data are expressed as mean ± SD. Comparisons between two groups were performed using the independent-samples t-test, and comparisons among multiple groups were performed using analysis of variance with pairwise comparisons using the least-significant-differences t-test. Count data are expressed as numbers and percentages, and intergroup comparisons were performed using the χ2 test. Correlation analyses between ADH4 expression and clinicopathological parameters were performed by Spearman’s rank correlation analysis. Survival analysis was performed using the Kaplan-Meier method and log-rank test, and multivariate analysis was performed using Cox regression analysis. Statistical significance was set at P < 0.05.
The low ADH4 group exhibited remarkable results. larger tumors, poorer differentiation, higher incidence of MVI, and less favorable survival outcomes, including lower survival rates and higher recurrence rates than the high ADH4 group. In contrast, parameters such as age, sex, tumor number, HBV infection status, and presence of cirrhosis showed no significant differences between the two groups (Table 1).
| Clinicopathological parameter | High expression (n = 74) | Low expression/negative (n = 74) | Statistical value (t or χ² or Z) | P value |
| Age (years) | 52.3 ± 8.5 | 53.1 ± 9.2 | -0.582 | 0.561 |
| Male | 52 (70.3) | 54 (73.0) | 0.257 | 0.612 |
| Tumor size (cm) | 4.5 ± 1.8 | 6.2 ± 2.5 | -4.218 | < 0.001 |
| Tumor size | ||||
| < 5 cm | 47 (62.8) | 28 (37.8) | 10.324 | 0.001 |
| ≥ 5 cm | 27 (37.2) | 46 (62.2) | ||
| Tumor number | 2.836 | 0.092 | ||
| Single | 58 (78.4) | 49 (66.2) | ||
| Multiple | 16 (21.6) | 25 (33.8) | ||
| Differentiation degree | 0.432 | < 0.001 | ||
| Well | 20 (27.0) | 6 (8.1) | ||
| Moderate | 38 (51.4) | 30 (40.5) | ||
| Poor | 16 (21.6) | 38 (51.4) | ||
| Microvascular invasion | 22.563 | < 0.001 | ||
| Yes | 24 (32.4) | 54 (73.0) | ||
| No | 50 (67.6) | 20 (27.0) | ||
| Hepatitis B virus infection | 0.085 | 0.770 | ||
| Positive | 61 (82.4) | 62 (83.8) | ||
| Negative | 13 (17.6) | 12 (16.2) | ||
| Cirrhosis | 0.365 | 0.546 | ||
| Yes | 45 (60.8) | 48 (64.9) | ||
| No | 29 (39.2) | 26 (35.1) | ||
| 1-year survival | 61 (82.4) | 45 (60.8) | 15.347 | < 0.001 |
| 3-year survival | 41 (55.4) | 22 (29.7) | - | - |
| Recurrence status | 10.072 | 0.001 | ||
| Recurred | 26 (35.1) | 45 (60.8) | ||
| Not recurred | 48 (64.9) | 29 (39.2) | ||
| Median survival time (months) | 38.2 ± 4.5 | 25.6 ± 3.8 | 4.893 | < 0.001 |
Relationship between ADH4 expression and tumor size: Among the 148 patients with HCC in this study, the mean maximum tumor diameter was 4.5 ± 1.8 cm in the high ADH4 group and 6.2 ± 2.5 cm in the low/negative ADH4 group. The independent-samples t-test showed a significant difference in tumor size between the two groups (t = -4.218, P < 0.001). This indicates that ADH4 expression is negatively correlated with tumor size and that patients with low ADH4 expression have larger tumor volumes. Further analysis of ADH4 expression across different tumor size groups, dividing tumor size at the 5-cm cut-off, showed that in the < 5 cm group, 62.8% (47/75) of patients had high ADH4 expression; in the ≥ 5 cm group, 37.2% (27/73) of patients had high ADH4 expression, with a significant difference (χ² = 10.324, P = 0.001) (Figure 1).
Relationship between ADH4 expression and tumor differentiation degree: The proportions of patients with well-differentiated, moderately differentiated, and poorly differentiated HCC with high ADH4 expression were 76.9% (20/26), 55.9% (38/68), and 27.3% (16/58), respectively. Spearman’s rank correlation analysis showed that ADH4 expression positively correlated with the degree of tumor differentiation (r = 0.432, P < 0.001), indicating that higher ADH4 exp
Relationship between ADH4 Expression and MVI: The proportions of patients with and without MVI but with high ADH4 expression were 30.8% (24/78) and 69.2% (50/72), respectively, with a significant difference (χ² = 22.563, P < 0.001), indicating that ADH4 expression is negatively correlated with MVI, and patients with low ADH4 expression are more prone to MVI. MVI is an important risk factor for postoperative recurrence and metastasis of HCC, suggesting that ADH4 may influence the invasion and metastatic capability of HCC by affecting the occurrence of MVI (Figure 1).
Survival curve analysis: Kaplan-Meier survival curves showed that the overall survival rate of patients in the ADH4 high expression group was significantly higher than that of patients in the ADH4 low expression group. The 1-year and 3-year survival rates in the high ADH4 group were 82.4% (61/74) and 55.4% (41/74), respectively, and 60.8% (45/74) and 29.7% (22/74) in the low/negative ADH4 group, respectively (Figure 1). Log-rank test results showed a significant difference in survival rates between the two groups (χ² = 15.347, P < 0.001), indicating that ADH4 expression level is closely related to the survival prognosis of HCC (Figure 2A), and high ADH4 expression has a protective effect on patient survival.
Univariate and multivariate analysis: Univariate Cox regression analysis showed that tumor size, degree of tumor differentiation, MVI, and ADH4 expression were closely related to overall patient survival. Statistically significant factors in univariate analysis were included in the multivariate Cox regression model. The results showed that ADH4 expression, tumor size, and MVI were independent risk factors affecting patient survival, indicating that after controlling for other factors, low ADH4 expression remained an independent factor leading to reduced patient survival (Table 2).
| Factor | Univariate HR (95%CI) | Multivariate HR (95%CI) |
| Alcohol dehydrogenase 4 low expression | 0.402 (0.268-0.603) | 0.486 (0.305-0.775) |
| Large tumor size | 2.135 (1.426-3.192) | 1.768 (1.135-2.750) |
| Microvascular invasion positive | 2.568 (1.734-3.797) | 2.047 (1.315-3.186) |
Comparison of anxiety and depression scores: Psychological assessment using HAMA and HAMD one month postoperatively showed that the mean HAMA score was 10.2 ± 3.5 points in the high ADH4 group and 13.8 ± 4.2 points in the low/negative ADH4 group, with a significant difference between groups (t = -5.217, P < 0.001). The mean HAMD score was 11.5 ± 3.8 points in the high ADH4 group and 15.6 ± 4.5 points in the low/negative ADH4 group, with a significant difference between groups (t = -5.528, P < 0.001). This indicates that anxiety and depression levels in the low/negative ADH4 group were significantly higher than those in the high ADH4 group.
Correlation analysis: Spearman’s correlation coefficients between ADH4 expression and anxiety-depression scores were calculated. ADH4 expression was negatively correlated with HAMA (r = -0.456, P < 0.001) and HAMD scores (r = -0.482, P < 0.001) (Figure 2B), indicating a close association between ADH4 expression and the anxiety-depression status of patients with HCC and suggesting that ADH4 may play a role in regulating the patient’s psychological state.
MVI is crucial for tumor cells to enter the bloodstream and cause distant metastasis[11]. ADH4 may affect the occurrence of MVI by regulating angiogenesis-related factors in the tumor microenvironment. Tumor cell proliferation and invasion require an adequate blood supply, and angiogenic factors such as vascular endothelial growth factor play a key role in promoting tumor angiogenesis[12]. ADH4 may inhibit the expression or activity of angiogenic factors, such as vascular endothelial growth factor, reducing tumor angiogenesis, thereby decreasing the chance of tumor cells invading microvessels and inhibiting tumor metastasis[13]. When ADH4 is expressed at low levels, its inhibitory effect on angio
The correlation between ADH4 expression and tumor invasiveness indicates its potential value as a diagnostic marker and therapeutic target in HCC. In clinical diagnoses, detecting ADH4 expression can assist physicians in accurately assessing tumor invasiveness[17]. For patients with low ADH4 expression, which indicates potentially stronger inva
From the perspective of therapeutic targets, interventions targeting ADH4 and its related signaling pathways may provide new strategies for HCC treatment. If ADH4 expression can be upregulated through drugs or other means or if the biological function of ADH4 can be mimicked, it may help inhibit tumor cell proliferation and invasion, thereby reducing tumor malignancy[18]. Drugs that promote ADH4 expression could be developed, or inhibitors targeting key nodes in the downstream signaling pathway of ADH4 could be designed to block tumor cell proliferation and metastasis. Furthermore, exploring the combination of ADH4 with existing HCC treatments such as surgery, chemotherapy, targeted therapy, and immunotherapy could improve treatment efficacy.
In this study, the overall survival of patients in the high ADH4 group was significantly higher than that in the low/negative ADH4 group, and multivariate analysis showed that ADH4 expression was an independent risk factor affecting patient survival prognosis. From a tumor biology perspective, ADH4 may affect patient survival through various pathways[19]. ADH4 participates in retinol metabolism and affects intracellular ATRA levels, thereby regulating tumor cell proliferation and apoptosis. High ADH4 expression can maintain relatively high levels of ATRA, promoting tumor cell apoptosis and inhibiting proliferation, thus reducing tumor recurrence and metastasis and prolonging patient survival. In contrast, ADH4 may affect the tumor microenvironment by regulating the function of immune cells and expression of immune-related factors, enhancing the body’s immune surveillance, and killing tumor cells[20]. Studies have shown that immune cells in the tumor microenvironment, such as cytotoxic T-lymphocytes and tumor-associated macrophages, play important roles in tumor development and prognosis[21]. ADH4 may influence the tumor immune escape ability by regulating the activity and degree of infiltration of these immune cells, thereby affecting patient sur
The relationship between ADH4 expression and the survival prognosis of patients with HCC in this study was consistent with that described in other studies. Some studies based on large cohorts of patients with liver cancer have found that low ADH4 expression is significantly associated with poor prognosis[22], with significantly shorter recurrence-free and overall survival in the low expression group than in the high expression group, which is consistent with our results. However, the mechanisms and factors influencing ADH4 in tumors may differ[23]. In addition to affecting tumor cell biology through the retinol metabolism pathway, ADH4 may interact with other metabolic pathways, such as fatty acid metabolism, thereby affecting tumor cell energy metabolism and proliferation capacity. These differences may be related to factors such as the race of study participants, sample size, detection methods, and study design[24]. Future prospective research needs to be conducted with larger sample sizes across multiple centers and with more advanced detection technologies to explore the mechanism of ADH4 in HCC and its influencing factors to evaluate the impact of ADH4 on patient survival prognosis more comprehensively.
From an endocrinological perspective, ADH4 is the key enzyme in the conversion of retinol to retinoic acid, and retinoic acid signaling influences the hypothalamic-pituitary-adrenal (HPA) axis and monoamine system, thereby participating in emotional regulation[25]. The HPA axis is an important stress regulatory system in the human body. Under stress, the HPA axis is activated, leading to increased secretion of stress hormones such as cortisol. Chronic stress and high cortisol levels negatively affect the neurobiological functions in the brain, thereby increasing the risk of anxiety and depression. ADH4 may influence the HPA axis activity by regulating related signaling pathways, thereby modulating the body’s response to stress. Low expression of ADH4 may lead to HPA axis dysfunction, reducing stress tolerance in the body and making it more prone to emotional disorders such as anxiety and depression[26]. This study observed a close association between ADH4 expression and the anxiety-depression status in patients with HCC, with the low/negative ADH4 group having significantly higher anxiety and depression levels than the high ADH4 group. From a neurobiological perspective, ADH4 may regulate patients’ emotional states by affecting the metabolism and signaling of neurotransmitters. Neurotransmitters, such as serotonin and dopamine, play key roles in mood regulation, and their abnormal levels are closely related to mental disorders such as anxiety and depression[27]. ADH4 participates in retinol metabolism, and retinol and its metabolites may affect the function of the neurotransmitter system by regulating the synthesis, release, and uptake of neurotransmitters[28]. Studies have shown that retinoic acid can regulates the activity of tryptophan hyd
The association between ADH4 expression and patient anxiety-depression status suggests that both tumor treatment and psychological health should be considered in the comprehensive management of HCC[31]. For patients with low ADH4 expression, medical staff should strengthen the assessment and monitoring of their psychological state and promptly identify and intervene for anxiety and depression. Comprehensive intervention measures such as psychotherapy and medication, including cognitive behavioral therapy and supportive psychotherapy, can be adopted to help patients adjust their cognition and coping styles and alleviate anxiety and depression, whereas anti-anxiety and antidepressant drugs can be reasonably used under physician guidance for patients with severe anxiety and depression symptoms[32]. Simultaneously, when formulating tumor treatment plans, the impact of the patient’s psychological state on treatment efficacy should be considered by adopting personalized treatment strategies to improve patient treatment compliance and quality of life. For patients with a heavy psychological burden, the pace and intensity of treatment can be appro
This study had certain limitations. Although 148 patients with HCC were included, the sample size was relatively small compared with that of some large-scale studies, which may have led to insufficient representativeness of the results and certain biases. This study had a retrospective cohort design. Although the relationship between ADH4 expression and various observation indicators can be analyzed to a certain extent, retrospective studies carry the risk of information and selection bias, which may affect the accuracy of the results. Immunohistochemical staining for ADH4 expression is a common method; however, it has a certain degree of subjectivity and different observers may judge the staining results differently, thus affecting the reliability of the results.
This retrospective cohort analysis of 148 patients with HCC clarified the relationship between ADH4 expression and HCC invasiveness, survival rate, and anxiety-depression status. ADH4 expression was significantly correlated with tumor invasiveness indicators such as tumor size, differentiation degree, and MVI. Patients with low ADH4 expression have larger tumor volumes, poorer differentiation, and are more prone to MVI[33]. ADH4 expression was an independent risk factor affecting patient survival, with the overall survival rate of the high ADH4 group being significantly higher than that of the low/negative ADH4 groups. ADH4 expression was associated with the anxiety-depression status, with the low/negative ADH4 group having significantly higher anxiety and depression levels than the high ADH4 group.
This study had several important clinical implications. ADH4 expression may serve as a potential biomarker for assessing the invasiveness and prognosis of HCC. By detecting ADH4 expression, physicians can more accurately judge the severity of a patient’s condition and prognosis, thereby providing a basis for formulating personalized treatment plans. For patients with low ADH4 expression, indicating stronger tumor invasiveness and poorer prognosis, physicians can consider more aggressive treatment strategies, such as expanding the scope of surgical resection, postoperative adjuvant chemotherapy, or targeted therapy, and strengthening the follow-up monitoring of patients to detect tumor recurrence and metastasis early, thereby improving patient survival rates[34]. This study revealed the role of ADH4 in the path
Future research should expand the sample size and conduct multicenter prospective studies to improve the reliability and generalizability of the results. An in-depth exploration of the mechanism of ADH4 in HCC should clarify its upstream and downstream signaling pathways, providing theoretical support for the development of ADH4-based targeted drugs. In clinical practice, ADH4 testing should be incorporated into routine diagnosis and prognostic assessment systems for HCC, while paying attention to the psychological health status of the patient. For patients with low ADH4 expression and anxiety/depression, timely psychological intervention and support should be provided to improve their quality of life and treatment compliance, thereby improving their overall prognosis.
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69784] [Article Influence: 13956.8] [Reference Citation Analysis (48)] |
| 2. | Zheng R, Zhang S, Zeng H, Wang S, Sun K, Chen R, Li L, Wei W, He J. Cancer incidence and mortality in China, 2016. J Natl Cancer Cent. 2022;2:1-9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1126] [Cited by in RCA: 1071] [Article Influence: 267.8] [Reference Citation Analysis (4)] |
| 3. | Edenberg HJ. The genetics of alcohol metabolism: role of alcohol dehydrogenase and aldehyde dehydrogenase variants. Alcohol Res Health. 2007;30:5-13. [PubMed] |
| 4. | Aly R, Emmady PD. Paraneoplastic Cerebellar Degeneration. 2023 Jul 4. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 5. | Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR. A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nat Rev Gastroenterol Hepatol. 2019;16:589-604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3422] [Cited by in RCA: 3212] [Article Influence: 458.9] [Reference Citation Analysis (9)] |
| 6. | Steel JL, Kim KH, Dew MA, Unruh ML, Antoni MH, Olek MC, Geller DA, Carr BI, Butterfield LH, Gamblin TC. Cancer-related symptom clusters, eosinophils, and survival in hepatobiliary cancer: an exploratory study. J Pain Symptom Manage. 2010;39:859-871. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 7. | Wade R, South E, Anwer S, Sharif-Hurst S, Harden M, Fulbright H, Hodgson R, Dias S, Simmonds M, Rowe I, Thornton P, Eastwood A. Ablative and non-surgical therapies for early and very early hepatocellular carcinoma: a systematic review and network meta-analysis. Health Technol Assess. 2023;27:1-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 8. | Rodríguez-Perálvarez M, Luong TV, Andreana L, Meyer T, Dhillon AP, Burroughs AK. A systematic review of microvascular invasion in hepatocellular carcinoma: diagnostic and prognostic variability. Ann Surg Oncol. 2013;20:325-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 552] [Cited by in RCA: 541] [Article Influence: 41.6] [Reference Citation Analysis (5)] |
| 9. | Allred DC, Harvey JM, Berardo M, Clark GM. Prognostic and predictive factors in breast cancer by immunohistochemical analysis. Mod Pathol. 1998;11:155-168. [PubMed] |
| 10. | Dinse GE, Lagakos SW. Nonparametric estimation of lifetime and disease onset distributions from incomplete observations. Biometrics. 1982;38:921-932. [PubMed] |
| 11. | Roayaie S, Blume IN, Thung SN, Guido M, Fiel MI, Hiotis S, Labow DM, Llovet JM, Schwartz ME. A system of classifying microvascular invasion to predict outcome after resection in patients with hepatocellular carcinoma. Gastroenterology. 2009;137:850-855. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 555] [Cited by in RCA: 563] [Article Influence: 33.1] [Reference Citation Analysis (4)] |
| 12. | Zhang Y, Jiang HH, Wang ZY, Zhai B, Lin MB. Alcohol dehydrogenase 4 is a TP53-associated gene signature for the prediction of prognosis in hepatocellular carcinoma. Oncol Lett. 2023;25:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 13. | Li L, Huang YT, Wang LT, Wang XL, Chen ZY, Jiang SL, Zeng QL, Huang HP, Li XL. ADH4-a potential prognostic marker for hepatocellular carcinoma with possible immune-related implications. BMC Cancer. 2024;24:927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 14. | Xie YJ. Screening and Verifying the Value of ADH4 in the Prognosis and Diagnosis of Hepatocellular Carcinoma Based on Bioinformatics Analysis. PhD Thesis. Fujian Medical University. |
| 15. | Tang XH, Gudas LJ. Retinoids, retinoic acid receptors, and cancer. Annu Rev Pathol. 2011;6:345-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 515] [Cited by in RCA: 463] [Article Influence: 30.9] [Reference Citation Analysis (0)] |
| 16. | Kropotova ES, Zinov'eva OL, Zyrianova AF, Choĭnzonov EL, Afanas'ev SG, Cherdyntseva NV, Beresten' SF, Oparina NIu, Mashkova TD. [Expression of genes involved in retinoic acid biosynthesis in human gastric cancer]. Mol Biol (Mosk). 2013;47:317-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 17. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018;69:182-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6763] [Cited by in RCA: 6579] [Article Influence: 822.4] [Reference Citation Analysis (5)] |
| 18. | Mahipal A, Kommalapati A, Mehta R, Kim RD. Molecular-Targeted Therapies in Hepatocellular Carcinoma. 2019 Aug 6. In: Hepatocellular Carcinoma: Translational Precision Medicine Approaches [Internet]. Cham (CH): Humana Press; 2019–. [PubMed] |
| 19. | Hoshida Y, Villanueva A, Kobayashi M, Peix J, Chiang DY, Camargo A, Gupta S, Moore J, Wrobel MJ, Lerner J, Reich M, Chan JA, Glickman JN, Ikeda K, Hashimoto M, Watanabe G, Daidone MG, Roayaie S, Schwartz M, Thung S, Salvesen HB, Gabriel S, Mazzaferro V, Bruix J, Friedman SL, Kumada H, Llovet JM, Golub TR. Gene expression in fixed tissues and outcome in hepatocellular carcinoma. N Engl J Med. 2008;359:1995-2004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1071] [Cited by in RCA: 1016] [Article Influence: 56.4] [Reference Citation Analysis (4)] |
| 20. | Tateishi R, Fujiwara N. Precision Locoregional Therapies for Hepatocellular Carcinoma: Percutaneous Ablation and Radiotherapy. 2019 Aug 6. In: Hepatocellular Carcinoma: Translational Precision Medicine Approaches [Internet]. Cham (CH): Humana Press; 2019–. [PubMed] |
| 21. | Fu J, Xu D, Liu Z, Shi M, Zhao P, Fu B, Zhang Z, Yang H, Zhang H, Zhou C, Yao J, Jin L, Wang H, Yang Y, Fu YX, Wang FS. Increased regulatory T cells correlate with CD8 T-cell impairment and poor survival in hepatocellular carcinoma patients. Gastroenterology. 2007;132:2328-2339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 743] [Cited by in RCA: 712] [Article Influence: 37.5] [Reference Citation Analysis (3)] |
| 22. | Gao Q, Wang XY, Qiu SJ, Yamato I, Sho M, Nakajima Y, Zhou J, Li BZ, Shi YH, Xiao YS, Xu Y, Fan J. Overexpression of PD-L1 significantly associates with tumor aggressiveness and postoperative recurrence in human hepatocellular carcinoma. Clin Cancer Res. 2009;15:971-979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 672] [Cited by in RCA: 655] [Article Influence: 38.5] [Reference Citation Analysis (3)] |
| 23. | Nault JC, Martin Y, Caruso S, Hirsch TZ, Bayard Q, Calderaro J, Charpy C, Copie-Bergman C, Ziol M, Bioulac-Sage P, Couchy G, Blanc JF, Nahon P, Amaddeo G, Ganne-Carrie N, Morcrette G, Chiche L, Duvoux C, Faivre S, Laurent A, Imbeaud S, Rebouissou S, Llovet JM, Seror O, Letouzé E, Zucman-Rossi J. Clinical Impact of Genomic Diversity From Early to Advanced Hepatocellular Carcinoma. Hepatology. 2020;71:164-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 154] [Article Influence: 25.7] [Reference Citation Analysis (1)] |
| 24. | Wang SM, Ooi LL, Hui KM. Identification and validation of a novel gene signature associated with the recurrence of human hepatocellular carcinoma. Clin Cancer Res. 2007;13:6275-6283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 141] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 25. | Lin YL, Wei CW, Lerdall TA, Nhieu J, Wei LN. Crabp1 Modulates HPA Axis Homeostasis and Anxiety-like Behaviors by Altering FKBP5 Expression. Int J Mol Sci. 2021;22:12240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 26. | Miller AH, Maletic V, Raison CL. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry. 2009;65:732-741. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3206] [Cited by in RCA: 2888] [Article Influence: 169.9] [Reference Citation Analysis (4)] |
| 27. | Ressler KJ, Nemeroff CB. Role of serotonergic and noradrenergic systems in the pathophysiology of depression and anxiety disorders. Depress Anxiety. 2000;12 Suppl 1:2-19. [PubMed] [DOI] [Full Text] |
| 28. | Bremner JD, McCaffery P. The neurobiology of retinoic acid in affective disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32:315-331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 135] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 29. | Shearer KD, Goodman TH, Ross AW, Reilly L, Morgan PJ, McCaffery PJ. Photoperiodic regulation of retinoic acid signaling in the hypothalamus. J Neurochem. 2010;112:246-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 58] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 30. | Otto LR, Clemens V, Üsekes B, Cosma NC, Regen F, Hellmann-Regen J. Retinoid homeostasis in major depressive disorder. Transl Psychiatry. 2023;13:67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 31. | Davidson B, Gurusamy K, Corrigan N, Croft J, Ruddock S, Pullan A, Brown J, Twiddy M, Birtwistle J, Morris S, Woodward N, Bandula S, Hochhauser D, Prasad R, Olde Damink S, Coolson M, Laarhoven KV, de Wilt JH. Liver resection surgery compared with thermal ablation in high surgical risk patients with colorectal liver metastases: the LAVA international RCT. Health Technol Assess. 2020;24:1-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 32. | Caruso R, GiuliaNanni M, Riba MB, Sabato S, Grassi L. Depressive Spectrum Disorders in Cancer: Diagnostic Issues and Intervention. A Critical Review. Curr Psychiatry Rep. 2017;19:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 33. | Reig M, Forner A, Rimola J, Ferrer-Fàbrega J, Burrel M, Garcia-Criado Á, Kelley RK, Galle PR, Mazzaferro V, Salem R, Sangro B, Singal AG, Vogel A, Fuster J, Ayuso C, Bruix J. BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. J Hepatol. 2022;76:681-693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3613] [Cited by in RCA: 3335] [Article Influence: 833.8] [Reference Citation Analysis (6)] |
| 34. | Craig AJ, von Felden J, Garcia-Lezana T, Sarcognato S, Villanueva A. Tumour evolution in hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2020;17:139-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 676] [Cited by in RCA: 607] [Article Influence: 101.2] [Reference Citation Analysis (0)] |