Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.117207
Revised: December 29, 2025
Accepted: January 22, 2026
Published online: April 19, 2026
Processing time: 119 Days and 7.6 Hours
Adolescents with type 1 diabetes face a disproportionately high risk of suicidal ideation, non-suicidal self-injury, and completed suicide, constituting a severe dual crisis of endocrinology and psychology. From a biopsychosocial perspective, this review critically analyzes the complex, bidirectional maladaptive feed-for
Core Tip: This review elucidates the lethal, bidirectional “neurotoxic cycle” connecting comorbid depression and glycemic dysregulation in adolescents with type 1 diabetes, identifying intentional insulin omission as a critical phenotype of covert self-harm. Beyond epidemiological associations, we synthesize evidence linking chronic hyperglycemia to neuroinflammatory changes in emotional regulation centers. Consequently, we advocate for a clinical paradigm shift from a singular “glucose-centric” approach to an integrated “psycho-metabolic” model, positioning routine suicide risk screening and means restriction as fundamental components of modern diabetes care.
- Citation: Wang DY, Yuan MY, Zhi H. Comorbid depression and glycemic instability in adolescent type 1 diabetes: Clinical insights into suicide risk. World J Psychiatry 2026; 16(4): 117207
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/117207.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.117207
Type 1 diabetes (T1D), one of the most common autoimmune chronic metabolic diseases in childhood and adolescence, has a peak incidence that coincides with a critical period of physical and psychological development. Adolescence is a tumultuous phase characterized by the transition from dependence to independence, the formation of identity, and the remodeling of emotional regulation networks[1]. For adolescents with T1D, this normal trajectory of biopsychosocial development is forcibly interrupted by lifelong and demanding disease management requirements. While exogenous insulin replacement therapy sustains life, it demands continuous, precise calculations balancing diet, exercise, emotions, and dosage[2]. The contradiction between this relentless cognitive burden and the delayed maturation of the adolescent prefrontal cortex renders this population highly susceptible to psychopathology. In recent years, while advances in the management of diabetic complications have reduced mortality from traditional causes such as renal failure or car
A more severe clinical challenge lies in the specificity and covert nature of suicidal behaviors in T1D patients. Unlike the general population, T1D patients possess ready access to a highly lethal means-insulin[5]. As a medication with a narrow therapeutic index, insulin is both a life-sustaining necessity and a potential biochemical weapon. In clinical practice, endocrinologists frequently encounter unexplained severe hypoglycemic events or recurrent episodes of diabetic ketoacidosis (DKA). Accumulating evidence suggests that these so-called “accidents” often conceal unrecognized suicide attempts or a paradoxical “gambling-like” self-harm behavior[6]. Patients may intentionally withhold insulin to induce ketosis, seeking pathological emotional release or self-punishment. Although the primary intent may not be immediate death, physiological consequences are often fatal[7].
However, despite the high clinical urgency of this issue, the current healthcare system still largely operates with a mind-body dichotomy. Endocrinologists often focus on achieving glycated hemoglobin (HbA1c) targets while neglecting the underlying psychological crisis, whereas psychiatrists may lack sufficient awareness of the neuropsychiatric symptoms triggered by acute glycemic fluctuations[8]. This interdisciplinary barrier has resulted in a significant number of T1D adolescents on the verge of suicide failing to receive timely identification and intervention. This review aims to systematically synthesize frontier literature from the past five years, deeply analyzing the complex interaction mechanisms between comorbid depression and glycemic dysregulation in elevating suicide risk from neurobiological, psychodynamic, and social environmental dimensions, and attempts to construct an integrated clinical diagnostic and treatment framework to provide a theoretical barrier for the safety of this vulnerable population.
The suicide risk in T1D adolescents does not exist in isolation but is the result of the interaction between demographic characteristics, disease-specific factors, and the psychosocial environment. Understanding the distribution of these epidemiological features is a prerequisite for clinicians to identify potential high-risk individuals and develop precise prevention strategies.
Suicidal ideation, as a precursor to suicidal behavior, is far more prevalent among T1D adolescents than the public expects. Multiple large-scale population-based cohort studies and meta-analyses indicate that the lifetime prevalence of suicidal ideation in T1D adolescents is as high as 26.4%, significantly higher than the 15.2% observed in nondiabetic peers[9]. This disparity not only reflects the psychological burden imposed by chronic illness itself but also reveals a generalized weakening of psychological resilience under specific disease states. Notably, mere prevalence data may mask a more core crisis: The low threshold for the transition from ideation to action. In the general adolescent population, the translation of suicidal ideation into actual attempts usually involves a psychological buffering period and is limited by the difficulty of accessing suicide means. However, in the T1D population, this translation pathway is significantly shortened.
This lowered threshold is primarily attributed to the accessibility of means. For patients with T1D, insulin is a drug they depend on for survival. The dual attribute of this “life-saving drug” as a “lethal poison” allows patients to imp
Through the in-depth analysis of numerous retrospective studies, it is evident that suicide risk in T1D adolescents is not uniformly distributed but presents a significant characteristic of risk clustering. This suggests that clinicians should focus on high-risk subgroups with specific clinical profiles.
First is the unique manifestation of gender differences. In suicide research within the general population, a “gender paradox” has long existed, where females have higher rates of suicide attempts but significantly lower mortality rates than males. However, in the T1D population, this protective effect appears to disappear or even reverse. Multiple studies have pointed out that female adolescents with T1D not only have higher rates of suicide attempts but also have a RR of suicide death far exceeding that of males (compared to healthy same-sex controls)[13]. This may be related to the fact that adolescent females are more susceptible to body image dissatisfaction, and the high-risk behavior of intentional insulin omission for weight control (i.e., dibulimia) is more prevalent among females, drastically increasing the risk of fatal metabolic complications[14].
Extreme abnormalities in metabolic control are another potent predictor of risk. Long-term HbA1c levels exceeding 9.0% (75 mmol/mol) are not only a sign of poor compliance but also a biological surrogate marker for chronic psychological distress[15]. Sustained hyperglycemia reflects the patient’s complete abandonment of disease management or a profound sense of helplessness. A more specific indicator is a history of frequent DKA admissions. Excluding objective factors such as insulin pump failure and acute infection, more than one episode of unprovoked DKA within a year should be regarded as a red alert for a psychological crisis[16]. Such recurrent admissions are often a powerful “cry for help” emitted through somatic collapse when adolescents are unable to verbally express their pain. Additionally, risk varies across different stages of the disease course. The first five years after diagnosis are a high-risk period, as patients undergo a grieving process from denial and anger to acceptance; late adolescence (15-19 years old) is another risk peak, where direct parental supervision is gradually withdrawn, and the conflict between the adolescent’s need for autonomy and their immature executive functions intensifies, easily leading to a collapse of the management system[17].
The toxic effect of the family environment cannot be ignored either. High-conflict, low-cohesion family environments are strong predictors of suicidal behavior in T1D adolescents[18]. Especially in families that moralize blood glucose values, where parents equate high blood glucose with the child’s moral defects or misconduct, this accusatory parenting style significantly intensifies the adolescent’s guilt and self-loathing. When adolescents feel that no matter how hard they try, they cannot meet parental or medical expectations, learned helplessness spontaneously arises, potentially leading to suicide as a means of escape from endless blame and a sense of failure. The multidimensional assessment indicators for identifying these high-risk profiles are summarized in Table 1.
| Dimension | High-risk indicators | Clinical pathological significance & mechanism |
| Metabolic | HbA1c persistently > 9.0% | Indicates chronic diabetes burnout or covert insulin omission; sustained hyperglycemic environment induces central neuroinflammation, impairing emotional regulation circuits |
| Recurrent DKA (> 1 episode in the past year) | After excluding device failure and infection, highly suspect behavioral expression of “emotional dysregulation” or implicit suicide attempts. Each DKA episode intensifies cerebral hypoxia and inflammatory damage | |
| Frequent severe iatrogenic hypoglycemia | Screen for self-harm via insulin overdose or confusion in diet/insulin matching due to severe depression and cognitive decline | |
| Psychobehavioral | Diagnosed with MDD or anxiety disorder | Depression increases suicide risk 3-6-fold; core symptoms of depression (e.g., avolition, hopelessness) directly undermine the willpower required to maintain complex self-management |
| Eating disorders (ED-DMT1/diabulimia) | Insulin omission is the core symptom, carrying an extremely high lethality rate. This is not just an eating disorder but a chronic, devastating form of self-destruction | |
| History of NSSI (cutting, burning, etc.) | The strongest single predictor of suicide attempts; indicates the patient is habituated to using somatic pain to relieve mental anguish and has increased pain tolerance (acquired capability for suicide) | |
| Social environment | High diabetes-specific family conflict | Parental overcontrol deprives adolescents of autonomy, whereas neglect leads to a lack of support. Both destroy the adolescent’s self-efficacy |
| Bullying and social exclusion | Peer rejection due to public device wearing or insulin injection intensifies “disease shame” and social isolation, reflecting “thwarted belongingness” in the interpersonal theory of suicide |
Applying a non-linear biopsychosocial lens, factors such as family conflict and social exclusion add to psychological despair and act as chronic stressors that recursively amplify neuroinflammatory signaling. This psycho-metabolic feedback loop means that a hostile social environment can biologically lower the threshold for suicide by further compromising the neurocircuitry of emotional regulation.
Rather than being discrete contributors, neuroinflammation, hypothalamic-pituitary-adrenal (HPA) axis dysfunction, and prefrontal impairment form a self-reinforcing pathophysiological network. Proinflammatory cytokines [e.g., interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α)] not only drive neurotoxicity but also impair glucocorticoid receptor sensitivity, disrupting the negative feedback loop of the HPA axis. This synergy leads to sustained HPA hyperactivity, further suppressing brain-derived neurotrophic factor (BDNF) expression, and accelerating structural degradation in the prefrontal-striatal-limbic circuits.
The association between glycemic control, depression, and suicide risk is by no means solely a psychological reaction or caused by life stress; its roots are deeply embedded in the pathophysiological alterations of the central nervous system. This is a bidirectional, self-reinforcing “neurotoxic cycle”, where hyperglycemia is not only the result of metabolic dysregulation but also the initiating factor for impaired brain emotional regulation function.
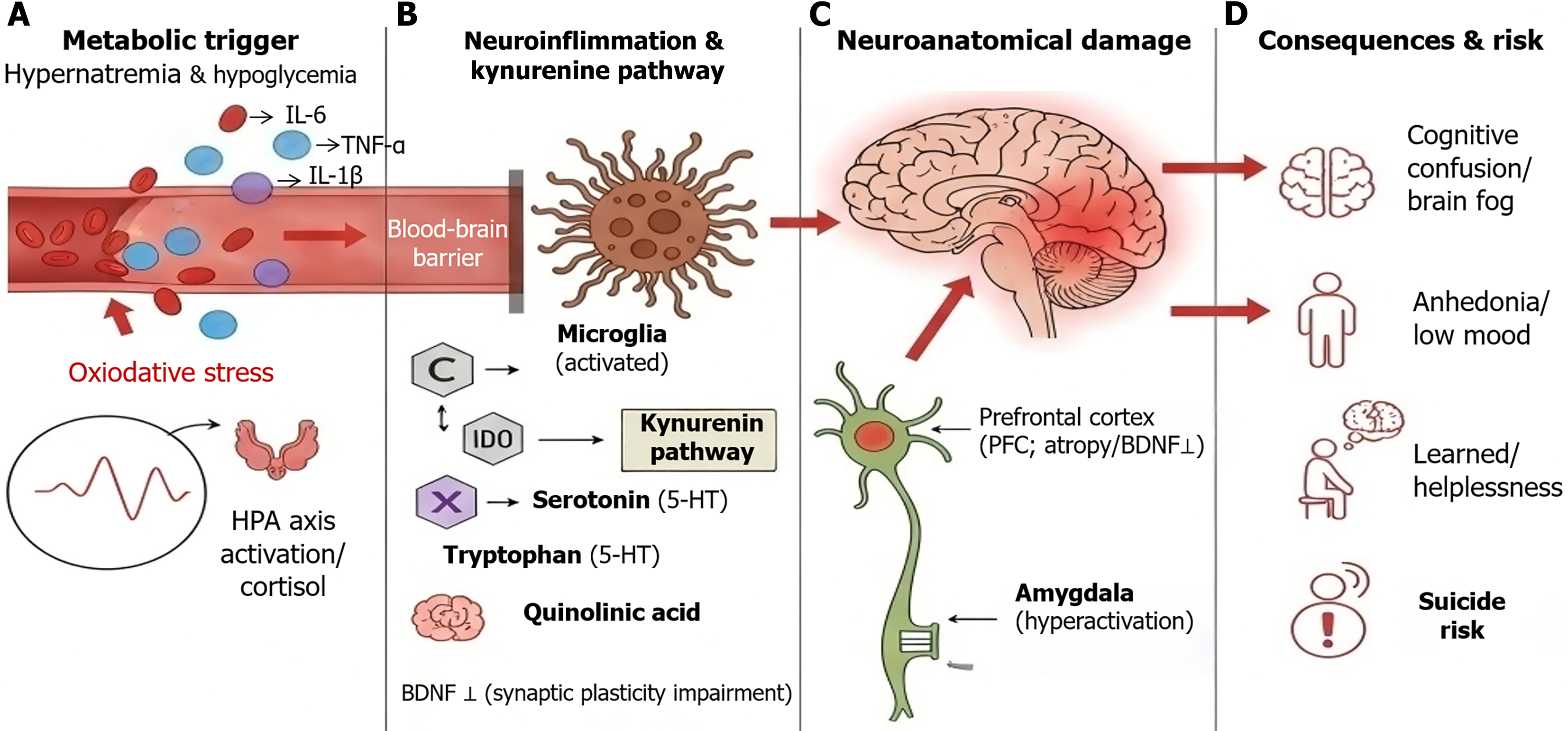
In recent years, the development of immunopsychiatry has provided a novel perspective for understanding the comorbid mechanisms of diabetes and depression. A chronic hyperglycemic state manifests as elevated glucose concentrations in the peripheral blood and essentially as a systemic state of chronic low-grade inflammation. The hyperglycemic environment induces oxidative stress through the activation of the polyol pathway, the hexosamine pathway, and the extensive deposition of advanced glycation end-products. This oxidative stress subsequently activates the peripheral immune system, leading to the systemic release of proinflammatory cytokines such as interleukin-6 (IL-6), TNF-α, and IL-1β. Notably, these peripheral inflammatory mediators can transmit inflammatory signals to the central nervous system by crossing the compromised blood-brain barrier or via vagal afferent pathways, activating microglia within the brain[19].
The activation of microglia triggers central neuroinflammation, which has profound effects on neurotransmitter metabolism. The most critical mechanism involves the alteration of the tryptophan metabolic pathway. Proinflammatory cytokines can upregulate the activity of indoleamine 2,3-dioxygenase (IDO), a key rate-limiting enzyme in tryptophan metabolism. Increased IDO activity accelerates the shunting of tryptophan toward the kynurenine pathway, thereby directly causing the depletion of tryptophan-the precursor for serotonin (5-HT) synthesis[20]. As a key neurotransmitter regulating mood, sleep, and impulse control, the reduction of serotonin concentration in the synaptic cleft is the classic biological basis for depression. Furthermore, downstream products of the kynurenine pathway, such as QA, possess direct neurotoxicity and can agonize NMDA receptors, leading to glutamate excitotoxicity and further neuronal damage. This neurobiochemical change driven by hyperglycemia explains why T1D adolescents with long-term poor glycemic control are more prone to anhedonia, low mood, and impulse control disorders.
At the neuroanatomical level, long-term metabolic dysregulation leads to structural remodeling of specific brain regions. High-resolution magnetic resonance imaging studies show that compared with healthy controls, T1D adolescents with long-term poor glycemic control exhibit significantly reduced gray matter volume in the hippocampus[21]. The hippocampus is not only a key brain region for memory formation but also an important center for negative feedback regulation of the HPA axis and participates in emotional regulation. Hippocampal atrophy weakens the brain’s ability to buffer stress. Concurrently, the amygdala-the brain region responsible for processing fear and anxiety-shows abnormally enhanced functional connectivity. This hyperactivation of the “emotional brain” (amygdala) and weakened inhibition by the “rational brain” (prefrontal cortex) constitute the neuroanatomical basis for susceptibility to suicidal behavior, making patients more prone to catastrophic cognition and extreme behavioral responses when facing negative emotions. The relevant mechanism is illustrated in Figure 1.
Recent advancements in transcriptomic profiling have provided deeper insights into the molecular signatures linking T1D and depression. Exploratory RNA-sequencing studies of peripheral blood mononuclear cells in adolescents with T1D and comorbid depression have identified significant upregulation in pathways related to Toll-like receptor signaling and proinflammatory cytokine production. Furthermore, integrated analysis of publicly available transcriptomic datasets suggests that chronic hyperglycemia-induced stress triggers a distinct gene expression pattern in neural-related tissues, characterized by the downregulation of neuroplasticity genes (e.g., BDNF, SYN1) and the overactivation of kynurenine pathway-related transcripts. These molecular alterations suggest that the neurotoxic cycle is encoded at the transcriptomic level, reinforcing the transition from metabolic stress to persistent suicidal ideation.
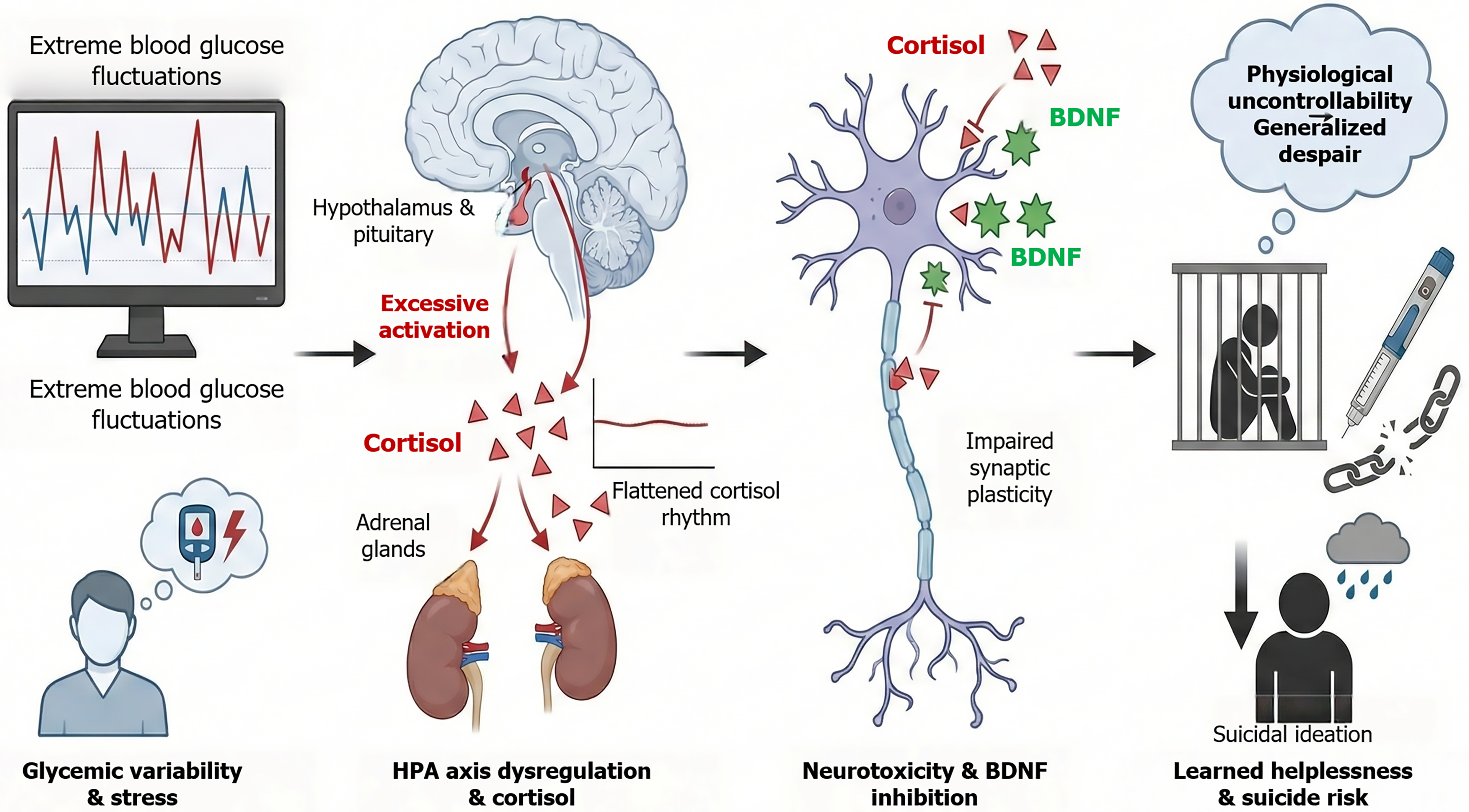
Besides chronic hyperglycemia, glycemic variability is a potent physiological stressor. Drastic fluctuations in blood glucose, especially frequent episodes of hypoglycemia followed by rebound hyperglycemia, are recognized by the body as threats to survival, leading to recurrent, excessive activation of the HPA axis. This sustained activation leads to dysregulation of cortisol secretion rhythms, manifesting as flattening of the circadian rhythm of cortisol or persistently elevated baseline levels[22].
Long-term exposure to high levels of glucocorticoids has significant toxic effects on the central nervous system. Glucocorticoids can inhibit the expression of BDNF[23]. BDNF is a key protein that maintains neuronal survival and promotes synaptic plasticity and neurogenesis. Decreased BDNF levels lead to impaired neural plasticity, rendering the brain unable to adapt to environmental stress or recover from negative emotions. Moreover, long-term dysfunction of the HPA axis leads to abnormal stress responses-blunted or exaggerated reactions when facing new psychosocial stressors (e.g., academic failure and interpersonal conflict). This physiological stress regulation disorder translates into Learned Helplessness at the psychological level. When adolescents feel that no matter how hard they try to control diet and inject insulin, blood glucose still fluctuates violently. This physiological uncontrollability generalizes to other areas of life, forming a diffuse sense of despair, which is the core psychological mechanism generating suicidal ideation. Figure 2 illustrates the relevant mechanism.
Severe hypoglycemic episodes are not only acute metabolic crises but also have non-negligible long-term impacts on brain function. The brain relies primarily on glucose as an energy substrate and lacks energy reserves, making it extremely sensitive to hypoglycemia. Recurrent severe hypoglycemia leads to neuronal energy failure and even irreversible neuronal death. This damage is brain region-specific, with the prefrontal cortex being one of the most severely affected areas.
The prefrontal cortex is responsible for higher cognitive functions, namely, executive function, including impulse control, decision-making, emotional regulation, and cognitive flexibility[24]. Impairment of executive function has direct predictive significance for suicide risk. For adolescents who have already developed suicidal ideation, the functional integrity of the prefrontal cortex acts as a “braking system”, inhibiting instantaneous suicidal impulses and guiding the individual to seek alternative coping strategies. However, under the battering of recurrent hypoglycemia, this braking system gradually fails. Clinical studies have found that patients with T1D with a history of severe hypoglycemia have a significantly increased risk of impulsive suicide attempts. In addition, acute cognitive confusion and emotional disinhibition accompanying hypoglycemia may trigger irrational self-harm behaviors in an instant[25]. Therefore, hypoglycemia is not only a side effect of treatment but also a pathophysiological accelerator that damages brain defense mechanisms and elevates suicide risk. Figure 1 depicts the relevant mechanism.
The transition from molecular insults to suicidal behavior is mediated by the disruption of integrated brain network dynamics. Specifically, the cellular damage described above compromises the prefrontal-striatal-limbic system, where diminished prefrontal inhibitory control fails to regulate a hyperactive amygdala. This functional imbalance at the circuit level provides a more precise biological explanation for the emergence of clinical impulsivity and profound hopelessness observed in T1D adolescents.
In clinical practice, the psychological crisis of T1D adolescents does not always present as typical depression but manifests as various specific clinical phenotypes closely intertwined with diabetes management. Accurately identifying these phenotypes and distinguishing their underlying psychodynamic mechanisms is key to developing effective intervention strategies.
Endocrinologists must possess the ability to distinguish between diabetes stress (DD) and major depressive disorder (MDD) in clinical work. Although there is overlap in symptom presentation and common comorbidity, fundamental differences exist in etiology, core experience, and treatment response. DD is not a psychopathological diagnosis but refers to the specific emotional reaction to the heavy burden of chronic disease management. Its core triggers stem directly from the pressure of diabetes management, such as persistent fear of hypoglycemic episodes, catastrophic imagination of long-term complications, and conflicts with healthcare providers or family members regarding disease management[26].
At the symptomatic level, the core experience of DD is typically described as “feeling overwhelmed”, “exhausted”, and anxiety and avoidance regarding blood glucose data. In contrast, the core symptom of MDD is Anhedonia, a loss of interest in all areas of life (including non-diabetes-related academics, social interactions, and hobbies), accompanied by pervasive sadness, low self-worth, and guilt[27]. Patients with DD usually maintain relatively intact functioning in non-diabetes-related social areas, and their emotional distress is context-specific. For example, an adolescent with severe DD may get along well with friends at school but exhibit extreme anxiety or anger upon mention of blood glucose testing or seeing a doctor.
This differential diagnosis has extremely important therapeutic implications. Studies indicate that antidepressant medication alone is very limited or ineffective in alleviating DD[28]. This is because medication cannot resolve the frustration and helplessness stemming from disease management tasks. For patients with high levels of DD, the most effective interventions are targeted diabetes education, behavioral interventions aimed at enhancing self-efficacy, and peer support. Blind use of antidepressants may not only increase the patient’s psychological burden due to side effects (e.g., weight gain, sexual dysfunction) but may also lead to deeper despair due to “treatment failure”. Therefore, clinical screening cannot rely solely on generic depression scales [e.g., Patient Health Questionnaire 9 (PHQ-9)] but must be combined with specific diabetes distress scales [e.g., Problem Areas in Diabetes-Teen (PAID-T) or T1 Diabetes Distress Scale] for comprehensive assessment[29]. A detailed comparison to aid in this differential diagnosis is provided in Table 2.
| Feature | DD | MDD |
| Core trigger | Directly related to diabetes management, fear of complications, doctor-patient relationship | No specific trigger or related to broad life stress events or genetic susceptibility |
| Symptom scope | Confined to diabetes-related contexts; patient may function well in non-diabetes areas | Pervasive; affects all areas of life (academics, social, hobbies, sleep, appetite) |
| Core experience | Feeling overwhelmed by disease, powerlessness, anxiety/avoidance of glucose data, and treatment burnout | Anhedonia, low self-worth, pervasive sadness, guilt, and thoughts of death |
| Diagnostic tools | PAID-T, T1-DDS | PHQ-9, Children’s Depression Inventory, and Beck Depression Inventory |
| Relation to HbA1c | Strong independent correlation; higher DD usually correlates with worse HbA1c | Weaker correlation; often affects HbA1c indirectly via DD as a mediator or appetite changes |
| First-line intervention | Diabetes education, self-efficacy-enhancing behavioral intervention, peer support, and resolving specific management barriers | Psychotherapy (CBT/IPT), psychopharmacology (SSRIs) |
In the spectrum of self-harm in T1D adolescents, intentional insulin omission occupies an extremely special and dangerous position, often referred to by media and clinicians as “diabulimia”. This is a complex pathological state where an eating disorder (usually bulimia or binge eating) intertwines with T1D, and it is one of the leading causes of suicide death and early disability in T1D adolescents[14].
From a pathophysiological mechanism perspective, insulin is an anabolic hormone that promotes glucose entry into cells for conversion into fat and glycogen. Lack of insulin causes the body to enter a catabolic state, where blood glucose rises extremely high and exceeds the renal threshold, excreting large amounts of glucose through urine (osmotic diuresis), thereby carrying away substantial calories[30,31]. Many weight-conscious T1D adolescents (especially young females) keenly discover this “shortcut”, using intentional underdosing or skipping of insulin to achieve rapid weight loss. This is essentially an extremely dangerous “metabolic purging” behavior[32].
However, in-depth psychodynamic analysis reveals that the motivation for insulin omission extends far beyond weight control. For many adolescents, it is a covert emotional regulation strategy and a means of self-harm. In a life rigorously controlled by blood glucose numbers, diet calculations, and medication dosages, adolescents often feel extreme constraint and loss of control. By “refusing injection”, they gain an illusory sense of control over their bodies and a sense of rebellion against the disease. Furthermore, some patients admit that the severe physical discomfort, confusion, and subsequent emergency hospitalization caused by inducing DKA allow them to temporarily escape unbearable reality pressures (e.g., academics, family conflict) and obtain attention and care from the outside world (pathological secondary gain). More severely, a significant proportion of adolescents use this method to express deep self-loathing or engage in a chronic, passive suicide attempt[11]. They are aware of the dangers of hyperglycemia but harbor a “devastating indifference”, allowing their bodies to corrode in ketoacidosis; this behavior constitutes so-called “chronic suicide”. The motivations for intentional insulin omission represent a clinical spectrum: From instrumental use for weight control (diabulimia), to emotional regulation through seeking DKA-related attention, and finally to passive suicidality characterized by fatalistic indifference to metabolic collapse.
The clinical consequences are devastating. Besides the acute mortality risk from recurrent DKA, this behavior leads to the explosive onset of microvascular complications at an extremely young age. Retinopathy leading to blindness, diabetic nephropathy leading to renal failure, and peripheral neuropathy leading to amputation-complications typically seen in middle-aged and elderly patients-may occur in patients in their early 20s. Rapid loss of physical function further intensifies depression and despair, forming an inescapable spiral of death[33].
Given the multidimensionality and complexity of suicide risk in T1D adolescents, the single biomedical treatment model has distinct limitations. To break the maladaptive feed-forward loop of hyperglycemia and depression, an integrated multidisciplinary team intervention system must be established, seamlessly embedding mental health management into routine diabetes care workflows[8].
Clinicians cannot passively rely on patients’ spontaneous reports to discover psychological problems because shame, fear of forced hospitalization, and worry about being labeled as mentally ill often cause adolescents to choose silence. Therefore, establishing an active, routine, multidimensional screening mechanism is crucial. International guidelines recommend baseline psychological assessment upon T1D diagnosis and routine implementation in subsequent quarterly follow-ups[34].
Screening should follow the principle of stratification. In level 1 universal screening, a combination of PHQ-9 for depressive symptom screening and PAID-T for diabetes distress screening should be used. This combination effectively distinguishes general depressive mood from disease-specific distress. Additionally, considering the high prevalence of eating disorders, it is recommended to routinely use the 5-item Sick, Control, One, Fat, Food questionnaire or Diabetes Eating Problem Survey-Revised Version scale to screen for insulin omission tendencies in adolescent female patients[29]. In level 2 targeted assessment, for patients with abnormal clinical indicators, even if questionnaire results are negative, they should be regarded as “psychological high-risk”. For instance, in patients with HbA1c persistently above 9.0% or recurrent unexplained DKA, their denial of psychological problems may be a psychological defense mechanism. For patients with a PHQ-9 score > 10 or a positive response to item 9 (suicidal ideation), a crisis assessment procedure must be immediately initiated using the Columbia-Suicide Severity Rating Scale to clarify the intensity, frequency, feasibility of the specific plan, and immediate impulse of suicidal ideation, and decide whether emergency referral to psychiatry or hospitalization is required[30].
For T1D adolescents assessed as having a suicide risk, the most direct and effective life-saving measure is means restriction. Since insulin is an essential medication carried by patients, completely cutting off access is impossible; therefore, the core strategy lies in “supervision”. For high-risk patients, clinicians should explicitly advise parents or guardians to temporarily retract the patient’s right to independently manage insulin. This means that insulin should be kept by parents, and every injection should be supervised or administered by parents. This not only prevents impulsive suicide using insulin overdose but also effectively curbs insulin omission behavior, breaking the “hyperglycemia-depression” physiological cycle[5].
Concurrently, developing a written safety plan is an important component of crisis intervention. Unlike merely establishing a “no-suicide contract”, safety planning focuses on empowering patients with specific skills to cope with crises. Doctors should work with patients to identify warning signs triggering suicidal impulses (e.g., specific emotions, physical sensations, or situations) and list hierarchical coping strategies: From internal coping alone (e.g., deep breathing, listening to music), to contacting specific friends or family members for distraction, to contacting professional medical personnel or calling crisis hotlines, and finally going to the nearest emergency room[10]. This plan should be written on paper or stored in a mobile phone to ensure that patients can access and execute it at any time during moments of crisis when cognitive function is impaired.
For adolescents with moderate to severe depression comorbid with T1D, pharmacotherapy is often necessary when psychotherapy is ineffective. However, the use of psychotropic drugs in patients with T1D requires high caution as the drugs themselves may interfere with glucose metabolism and produce complex interactions. Selective serotonin reuptake inhibitors (SSRIs) are typically used as first-line medications, such as fluoxetine and sertraline. Besides antidepressant effects, studies have found that certain SSRIs (especially fluoxetine) may have a direct hypoglycemic effect, increasing peripheral tissue insulin sensitivity[31]. While this sounds beneficial, in the initial stage of treatment, if insulin dosage is not adjusted synchronously, it may lead to an increased risk of severe hypoglycemia. Therefore, endocrinologists must work closely with psychiatrists to strengthen blood glucose monitoring during the first few weeks of initiating antidepressant treatment and proactively down-titrate total insulin dosage by 10%-20%.
On the other hand, the use of drugs causing significant weight gain or metabolic deterioration should be strictly avoided. Certain antipsychotics (e.g., olanzapine) or antidepressants (e.g., mirtazapine) have strong side effects of increased appetite and weight gain. For adolescents with T1D who are already sensitive to body shape and at risk of insulin omission, weight gain may exacerbate their body image anxiety, thereby triggering or aggravating insulin omission behavior, leading to catastrophic deterioration of glycemic control. Therefore, in drug selection, priority should be given to drugs with a neutral effect on weight or even those helpful for weight loss (e.g., bupropion, but use with caution in those with seizure risk or extremely high risk of eating disorders).
In recent years, the proliferation of continuous glucose monitoring (CGM) and automated insulin delivery (AID) systems (also known as artificial pancreas) has made revolutionary progress in improving metabolic control in patients with T1D. From a suicide prevention perspective, AID systems have significant advantages. They can automatically adjust basal rates during patient sleep, reducing hypoglycemia occurrence and improving sleep quality, thereby enhancing daytime emotional regulation ability. More importantly, AID systems alleviate the cognitive burden of constant dosage calculation to some extent, relieving diabetes burnout[35].
However, technology is also a double-edged sword. For some adolescents with obsessive tendencies, perfectionism, or high levels of anxiety, the real-time data stream provided by CGM can become a new source of stress. Continuous alarms and constantly changing trend arrows on the screen may keep the patient in a constant “fight or flight” stress state. To pursue a “flat” glucose curve, patients may engage in frequent corrective injections or excessive data checking, leading to “data burnout”. If data are not ideal, this immediate feedback reinforces the patient’s frustration and self-negation. Therefore, the introduction of technology should be based on an individualized psychological assessment. For certain extremely anxious patients, temporary “technological downgrading” (e.g., turning off nonurgent alarms, reducing checking frequency, or even briefly pausing CGM in favor of fingerstick glucose) may be a necessary psychological protective measure to rebuild a healthy relationship between the patient and disease management[34]. Consequently, we propose a stratified intervention framework tailored to the patient’s specific risk level (Table 3).
| Risk level | Clinical features | Recommended intervention strategy |
| Level 1 (low risk) | Good glycemic control (HbA1c < 7.5%), PHQ-9 < 5, occasional mild diabetes distress, and good family support | Basic support & prevention: Provide routine diabetes education emphasizing mental health importance; encourage participation in diabetes camps or peer support groups to enhance belonging; maintain quarterly routine psychological screening |
| Level 2 (moderate risk) | HbA1c 7.5%-9.0%, moderate diabetes distress, PHQ-9 indicates mild depression, no suicidal ideation, fluctuating compliance | Targeted intervention: Introduce a psychology nurse specialist or social worker for brief intervention; focus on resolving specific diabetes distress sources (e.g., needle phobia, social embarrassment); consider introducing CGM to reduce management burden but monitor anxiety levels; family sessions to resolve minor conflicts |
| Level 3 (high risk) | HbA1c > 9.0%, diagnosed with MDD or anxiety disorder, presence of NSSI behavior or eating disorder tendency, and distinct family conflict | Multidisciplinary team intervention: Establish joint rounds with endocrinology + psychiatry + nutrition; initiate CBT (for depression) or DBT (for self-harm) psychotherapy; cautiously initiate SSRI pharmacotherapy; recommend parents partially take over glycemic management responsibility to relieve adolescent burden |
| Level 4 (extremely high risk) | Recent suicide attempt, recurrent DKA admissions, severe insulin omission, specific suicide plan, and extreme hopelessness | Crisis intervention & hospitalization: Immediate hospitalization (endocrinology ward with enhanced monitoring or psychiatry closed ward); complete takeover of glycemic management responsibility, depriving the patient of independent access to insulin; strict means restriction; initiate family-based therapy to rebuild the family support system and resolve core conflicts |
The escalating risk of suicide and self-harm among adolescents with T1D, driven by the lethal intersection of neurobiological vulnerability and treatment-specific means like insulin omission, demands an urgent paradigm shift from a singular “glucose-centric” focus to an integrated “psycho-metabolic” model. Clinicians must recognize unexplained hyperglycemia as a potential physiological cry for help and implement routine, stratified mental health screening and means restriction within diabetes care. Ultimately, bridging the gap between endocrinology and psychiatry through multidisciplinary collaboration is the only sustainable path to breaking the maladaptive feed-forward loop of depression and dysregulation, safeguarding metabolic stability and the lives of this vulnerable population.
| 1. | Shah AS, Zeitler PS, Wong J, Pena AS, Wicklow B, Arslanian S, Chang N, Fu J, Dabadghao P, Pinhas-Hamiel O, Urakami T, Craig ME. ISPAD Clinical Practice Consensus Guidelines 2022: Type 2 diabetes in children and adolescents. Pediatr Diabetes. 2022;23:872-902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 108] [Article Influence: 27.0] [Reference Citation Analysis (3)] |
| 2. | Brazeau AS, Mircescu H, Desjardins K, Leroux C, Strychar I, Ekoé JM, Rabasa-Lhoret R. Carbohydrate counting accuracy and blood glucose variability in adults with type 1 diabetes. Diabetes Res Clin Pract. 2013;99:19-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 166] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 3. | AbdElmageed RM, Mohammed Hussein SM. Risk of Depression and Suicide in Diabetic Patients. Cureus. 2022;14:e20860. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 4. | Hill RM, Gallagher KAS, Eshtehardi SS, Uysal S, Hilliard ME. Suicide Risk in Youth and Young Adults with Type 1 Diabetes: a Review of the Literature and Clinical Recommendations for Prevention. Curr Diab Rep. 2021;21:51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 5. | Assad MA, Alawami FA, Al Khatem RS, Al Daif ZY, Alrumaih ZA, Menezes RG. Suicide and attempted suicide by insulin: A systematic review. F1000Res. 2023;12:59. [DOI] [Full Text] |
| 6. | Renaud-Charest O, Stoljar Gold A, Mok E, Kichler J, Nakhla M, Li P. Suicidal Ideation, Suicide Attempts, and Suicide Deaths in Adolescents and Young Adults With Type 1 Diabetes: A Systematic Review and Meta-analysis. Diabetes Care. 2024;47:1227-1237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 7. | Wojujutari Ajele K, Sunday Idemudia E. The role of depression and diabetes distress in glycemic control: A meta-analysis. Diabetes Res Clin Pract. 2025;221:112014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 8. | Young-Hyman D, de Groot M, Hill-Briggs F, Gonzalez JS, Hood K, Peyrot M. Psychosocial Care for People With Diabetes: A Position Statement of the American Diabetes Association. Diabetes Care. 2016;39:2126-2140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 861] [Cited by in RCA: 749] [Article Influence: 74.9] [Reference Citation Analysis (4)] |
| 9. | Butwicka A, Frisén L, Almqvist C, Zethelius B, Lichtenstein P. Risks of psychiatric disorders and suicide attempts in children and adolescents with type 1 diabetes: a population-based cohort study. Diabetes Care. 2015;38:453-459. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 143] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 10. | Chu C, Buchman-Schmitt JM, Stanley IH, Hom MA, Tucker RP, Hagan CR, Rogers ML, Podlogar MC, Chiurliza B, Ringer FB, Michaels MS, Patros CHG, Joiner TE. The interpersonal theory of suicide: A systematic review and meta-analysis of a decade of cross-national research. Psychol Bull. 2017;143:1313-1345. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 984] [Cited by in RCA: 773] [Article Influence: 85.9] [Reference Citation Analysis (0)] |
| 11. | Goddard G, Oxlad M. Caring for individuals with Type 1 Diabetes Mellitus who restrict and omit insulin for weight control: Evidence-based guidance for healthcare professionals. Diabetes Res Clin Pract. 2022;185:109783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 12. | Pompili M, Forte A, Lester D, Erbuto D, Rovedi F, Innamorati M, Amore M, Girardi P. Suicide risk in type 1 diabetes mellitus: A systematic review. J Psychosom Res. 2014;76:352-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 55] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 13. | Barnard‐Kelly K, Holt R, O’neill S. Suicide and type 1 diabetes: a complex issue. Pract Diabetes. 2022;39:10. [DOI] [Full Text] |
| 14. | Quiram N, Salam T, Sadjadpour F, Hosseinichimeh N, Jarvis L, Soghier L. A literature review of remote mental health screening: barriers, potential solutions, and tools. Front Digit Health. 2025;7:1670691. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 15. | Hagger V, Hendrieckx C, Sturt J, Skinner TC, Speight J. Diabetes Distress Among Adolescents with Type 1 Diabetes: a Systematic Review. Curr Diab Rep. 2016;16:9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 296] [Cited by in RCA: 249] [Article Influence: 24.9] [Reference Citation Analysis (0)] |
| 16. | Carreira M, Ruiz de Adana MS, Domínguez M, Valdés S, Almaraz MC, Olveira G, Anarte MT. Development and Validation of the Depression Inventory for Type 1 Diabetes (DID-1). Int J Environ Res Public Health. 2021;18:12529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 17. | Datye KA, Moore DJ, Russell WE, Jaser SS. A review of adolescent adherence in type 1 diabetes and the untapped potential of diabetes providers to improve outcomes. Curr Diab Rep. 2015;15:51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 107] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 18. | Savin KL, Hamburger ER, Monzon AD, Patel NJ, Perez KM, Lord JH, Jaser SS. Diabetes-specific family conflict: Informant discrepancies and the impact of parental factors. J Fam Psychol. 2018;32:157-163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 19. | Ducat L, Rubenstein A, Philipson LH, Anderson BJ. A review of the mental health issues of diabetes conference. Diabetes Care. 2015;38:333-338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 20. | Al-Jabi SW. Emerging global interest: Unraveling the link between diabetes mellitus and depression. World J Psychiatry. 2024;14:1127-1139. [PubMed] [DOI] [Full Text] |
| 21. | Wu A, Zhang J. Neuroinflammation, memory, and depression: new approaches to hippocampal neurogenesis. J Neuroinflammation. 2023;20:283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 230] [Cited by in RCA: 218] [Article Influence: 72.7] [Reference Citation Analysis (0)] |
| 22. | DeCosta P, Grabowski D, Skinner TC. The psychosocial experience and needs of children newly diagnosed with type 1 diabetes from their own perspective: a systematic and narrative review. Diabet Med. 2020;37:1640-1652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 23. | Adharshna TK, Gehlawat P, Nebhinani N, Singh K, Vyas V. Are children and adolescents with type 1 diabetes mellitus prone to psychiatric issues? A cross-sectional comparative study. Indian J Psychiatry. 2025;67:432-435. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Berg CA, Wiebe DJ, Suchy Y, Turner SL, Butner J, Munion A, Lansing AH, White PC, Murray M. Executive Function Predicting Longitudinal Change in Type 1 Diabetes Management During the Transition to Emerging Adulthood. Diabetes Care. 2018;41:2281-2288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 25. | Lawrence CM, Smart CE, Fatima A, King BR, Lopez P. Increased bolus overrides and lower time in range: Insights into disordered eating revealed by insulin pump metrics and continuous glucose monitor data in Australian adolescents with type 1 diabetes. J Diabetes Complications. 2024;38:108904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 26. | de Wit M, Gajewska KA, Goethals ER, McDarby V, Zhao X, Hapunda G, Delamater AM, DiMeglio LA. ISPAD Clinical Practice Consensus Guidelines 2022: Psychological care of children, adolescents and young adults with diabetes. Pediatr Diabetes. 2022;23:1373-1389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 132] [Cited by in RCA: 103] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 27. | Chou WC, Chou YY, Pan YW, Ou TY, Tsai MC. Correlates of disordered eating and insulin restriction behavior and its association with psychological health in Taiwanese youths with diabetes mellitus. J Eat Disord. 2023;11:158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 28. | Addala A, Howard KR, Hosseinipour Y, Ekhlaspour L. Discordance Between Clinician and Person-With-Diabetes Perceptions Regarding Technology Barriers and Benefits. J Diabetes Sci Technol. 2025;19:18-26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 29. | García-Ulloa AC, Miranda-Gil V, Díaz-Pineda M, Garnica-Carrillo MF, Serrano-Pérez NH, Tron-Gomez MS, Colorado Báez GP, Cruz Madrigal CA, Hernández-Jiménez S; Group of Study CAIPaDi. Comparison of Metabolic, Lifestyle and Mental Health Parameters in People with Diabetes and Relatives with and without Family Support. Diabetes Metab Syndr Obes. 2024;17:239-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | Fatima S, Prichett L, Campbell N, Snyder MCN, Bifano M, Wolf RM. Suicide Risk Screening in a Diverse Cohort of Youth With Type 1 and Type 2 Diabetes. Pediatr Diabetes. 2025;2025:6662248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 31. | Almeida AC, Leandro ME, Pereira MG. Adherence and Glycemic Control in Adolescents with Type 1 Diabetes: The Moderating Role of Age, Gender, and Family Support. J Clin Psychol Med Settings. 2020;27:247-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 32. | Pinhas-Hamiel O, Hamiel U, Levy-Shraga Y. Eating disorders in adolescents with type 1 diabetes: Challenges in diagnosis and treatment. World J Diabetes. 2015;6:517-526. [PubMed] [DOI] [Full Text] |
| 33. | Ghotane S, Hirve R, Forman J, Tan D, Achercouk Z, Wolfe I. Integrated care for children and young people with special health and care needs: a systematic review. Arch Dis Child. 2024;109:924-931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 34. | Bassi G, Mancinelli E, Dell’Arciprete G, Salcuni S. The Impact of the Covid-19 Pandemic on the Well-Being and Diabetes Management of Adolescents With Type 1 Diabetes and Their Caregivers: A Scoping Review. Front Clin Diabetes Healthc. 2022;3:835598. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 35. | Jeter K, Fisher R, Traino K, Dattilo T, Allen R, Anderson M, Cutler J, Sparling D, Beck J. The impact of continuous glucose monitoring on diabetes distress among high-risk adolescents and young adults: Secondary outcomes from a pilot randomized controlled trial. Psychol Health Med. 2025;1-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |