Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.115981
Revised: December 16, 2025
Accepted: January 15, 2026
Published online: April 19, 2026
Processing time: 151 Days and 1.4 Hours
Depression represents a frequent comorbidity in breast cancer patients. However, many antidepressants have a certain degree of adverse reactions and decline pa
To systematically evaluate the efficacy of CHF interventions for depression in breast cancer patients and explore the potential mechanisms.
We systematically searched four English databases (PubMed, EMBASE, the Coch
Pooled data from 20 randomized trials (n = 1607) demonstrated CHF’s superiority over antidepressants in reducing Hamilton Depression Rating Scale scores (mean difference = -5.33, 95% confidence interval: -6.93 to -3.73, P < 0.05) and enhancing QOL (mean difference = 9.13, 95% confidence interval: 6.69-11.57, P < 0.05). CHF significantly ele
This systematic review establishes CHF as an effective and safe intervention for depression in breast cancer pa
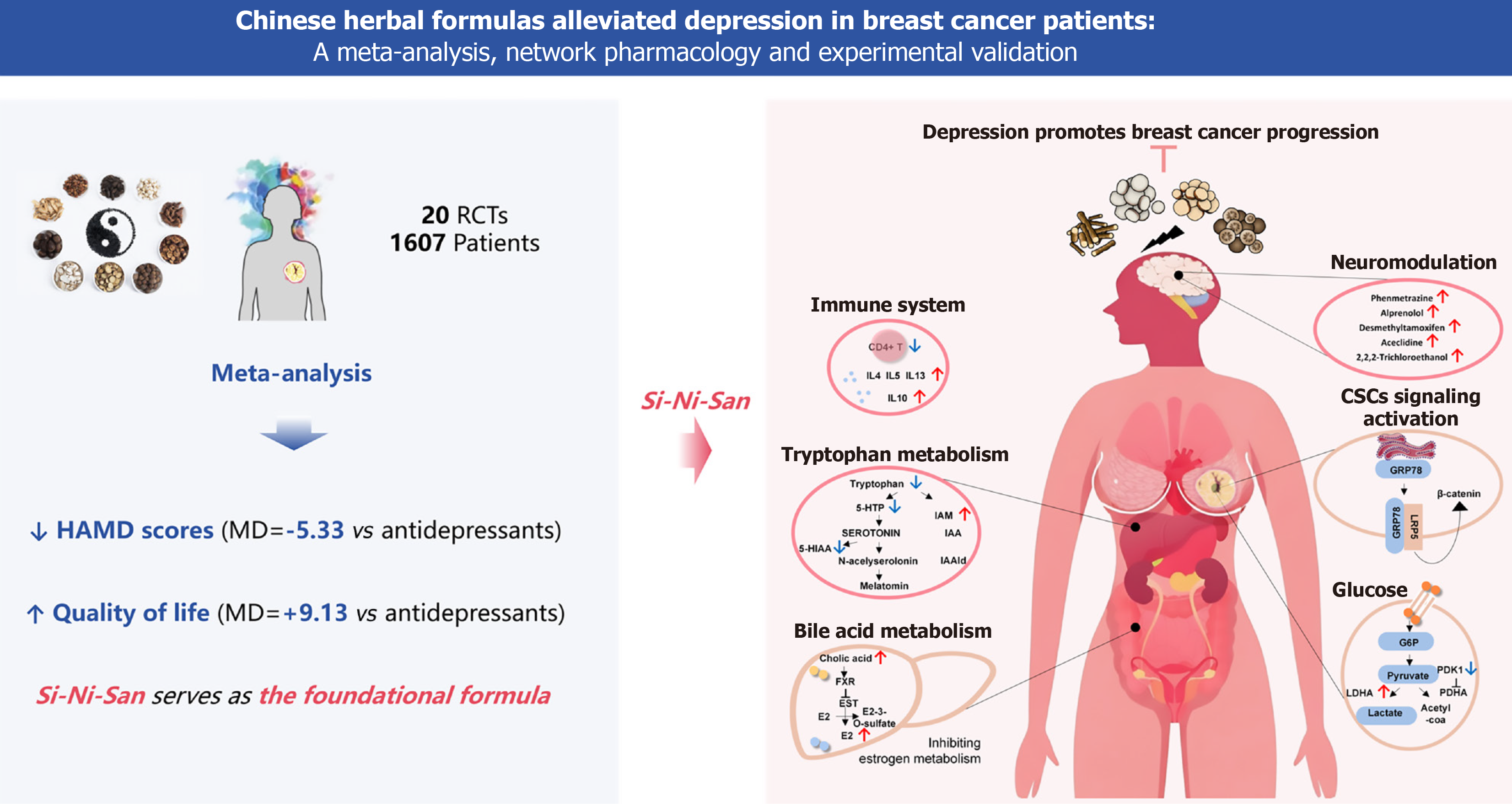
Core Tip: In present study, we found that Chinese herbal formulas outperformed antidepressants in reducing depression and improving quality of life, significantly elevated monoamine neurotransmitter levels, and selectively increased CD4+ T-cell counts. Network pharmacology revealed that Si-Ni-San may modulate immune responses and serotonergic synapses, while experimental validation indicated involvement of CD4+ T cells and rebalancing of neurotransmitter, tryptophan metabolism. These results advocate for the integration of Chinese herbal formulas especially Si-Ni-San into integrative care protocols for breast cancer-related depression and underscore the need for further mechanistic and clinical investigation.
- Citation: Hong SC, Du YY, Wang GB, Wang SQ, Zheng YF, Wang N, Situ HL, Wang ZY. Chinese herbal formulas alleviated depression in breast cancer patients: A meta-analysis, network pharmacology and experimental validation. World J Psychiatry 2026; 16(4): 115981
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/115981.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.115981
Breast cancer constitutes a significant public health concern globally, representing the most prevalent cancer diagnosis and a primary contributor to cancer-associated deaths among women. Current epidemiological reports indicate approximately 2.33 million new cases and 664000 fatalities annually[1]. Although developments in screening technologies and treatment modalities have enhanced survival outcomes[2], marked inequalities in mortality rates persist across different healthcare settings[3]. The condition’s impact transcends clinical parameters, generating considerable socioeconomic consequences as reflected by its prominent position among leading causes of disability-adjusted life years in multiple nations[4]. In response, the World Health Organization’s Global Breast Cancer Initiative has established ambitious targets to reduce mortality through 2040[5].
The psychological burden accompanying breast cancer diagnosis and treatment is substantial. Compared to the general population, depression occurs 3-5 times more frequently in cancer population than in the general community[6,7]. Clinical investigations report that nearly 50% of patients develop clinically relevant depressive symptoms within the initial treatment phase[8]. Meta-analytic evidence confirms a pooled depression prevalence of 32.2% among breast cancer patients[6], with mood disturbances independently predicting worse oncological outcomes. These affective disturbances demonstrate independent prognostic significance, correlating with elevated risks of cancer recurrence (24% increase) and disease-specific mortality (29% increase)[9]. This association was attributed to a variety of biological and behavioral mechanisms. It was reported that depression could affect immune and endocrine systems directly[10]. The underlying pathophysiology also involves complex interactions between neuroendocrine dysfunction, particularly hypothalamic-pituitary-adrenal axis dysregulation[11]. Additionally, depression even in subthreshold manifestations, is associated with maladaptive lifestyle habits and poor adherence to medical appointments and therapies[12,13], which may indirectly influence cancer progression. Therefore, to enhance the prognosis and overall survival of breast cancer patients, it is imperative to explore strategies for alleviating depression and optimizing long-term clinical outcomes.
At present, both non-pharmacological treatments and antidepressants are recommended as therapeutic approaches. However, psychological specialists may lack professional training in working with cancer patients, and the antidepressants may worsen existing symptoms and interact with chemotherapy agents[14]. In addition, previous studies indicated that antidepressants were found with low-certainty evidence for their application in cancer patients[15]. Traditional Chinese medicine (TCM) has long been appreciated to treat depression, particularly in Asia. Notably, its holistic, multi-ingredients and multi-targets nature fits well with modern systemic medicine. Chinese herbal formulas (CHF) usually combine two or more medicines to create synergistic effects and minimize toxicity, much like modern therapeutic strategies that require multiple drugs[16,17]. Xiao-Yao-San, a commonly employed anti-depression formula, has been shown to meliorate depressive-like behaviors induced by gut microbiota dysbiosis by regulating the gut microbiota and counteracting abnormal synaptic pruning[18]. Another anti-depression formula Si-Ni-San (SNS) has demonstrated antidepressant effects in various clinical contexts[19-23], while emerging studies have further underscored its potential in suppressing the growth and metastasis of breast cancer triggered by chronic psychological stress, potentially through interactions with the central nervous system, cancer stem cells and bile acids metabolism[24-26]. As the field of CHF continues to be explored, a variety of active ingredients and components with notable antidepressant properties have been identified. Saikosaponin A, a pivotal bioactive constituent derived from Radix Bupleuri, has been proposed to exert antidepressant efficacy by augmenting the expression level of proline-rich transmembrane protein 2 and dopamine (DA) levels content within the hippocampus[27]. Those CHF and ingredients appear to exert their effects by modulating various systems[28], which offer novel insights into the therapeutic mechanism underpinning the use of CHF for the management of depression. Therefore, the leading organizations in the field, such as the American Cancer Society, recommended integrative therapies for symptoms management during breast cancer treatment[29]. However, a systematic review is necessary to explore the therapeutic efficacy and safety of CHF for depression treatment in breast cancer patients.
To address this gap, we conducted a systematic review and meta-analysis to determine whether CHF therapy is beneficial in relieving depression among breast cancer patients. In present study, we found that CHF outperformed antidepressants in reducing depression and improving quality of life (QOL), significantly elevated monoamine neurotransmitter levels, and selectively increased CD4+ T-cell counts. Network pharmacology revealed that SNS may modulate immune responses and serotonergic synapses, while experimental validation indicated involvement of CD4+ T cells and rebalancing of neurotransmitter, tryptophan metabolism. These results advocate for the integration of CHF especially SNS into integrative care protocols for breast cancer-related depression and underscore the need for further mechanistic and clinical investigation.
This systematic review and meta-analysis adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The study protocol was prospectively registered in the PROSPERO international prospective register of systematic reviews (Registration No. CRD42024555279).
An extensive literature search was conducted across eight databases from inception through June 1, 2024, including both international databases (PubMed, EMBASE, Cochrane Library, and PsycINFO) and Chinese databases (CBM, CNKI, VIP, and Wanfang). Supplementary searches included: Manual screening of reference lists from eligible studies and relevant reviews, conference proceedings for unpublished data, existing systematic reviews for additional trials.
The search strategy incorporated three key domains: (1) Population: Breast cancer patients with depression (breast cancer, breast neoplasms, depression, and mental health); (2) Intervention: CHF therapies (TCM, Chinese herbal medicine, decoctions, formulas); and (3) Study design: Randomized controlled trials (RCTs). Two of us (Hong SC and Du YY) independently reviewed the abstracts and titles of preliminary literature, or the full texts as necessary, to establish the eligibility of the studies. Two independent reviewers (Hong SC and Du YY) screened titles/abstracts and full texts when necessary. Complete search terms are provided in Supplementary Tables 1 and 2.
Studies were included if they met the following criteria: (1) Participants: Individuals histopathologically diagnosed with breast cancer and meeting depression criteria as defined by the Diagnostic and Statistical Manual of Mental Disorders Fourth Edition, Research Diagnostic Criteria, International Statistical Classification of Diseases and Related Health Problems, or Chinese Classification of Mental Disorders Third Edition Revised; (2) Interventions and comparisons: The treatment group received CHF, and the control groups were categorized as either “antidepressant” or “no intervention”, the latter denoting the absence of any specific antidepressant intervention; (3) Outcome measures: Primary outcomes of depression included Hamilton Depression Rating Scale (HAMD) or Self-rating Depression Scale (SDS) values, and secondary outcomes encompassed QOL, serum levels of 5-hydroxytryptamine (5-HT), DA, and norepinephrine (NE), as well as immune function indices (CD4+, CD8+ T lymphocytes); and (4) Study design: Published RCTs. Exclusion criteria include: (1) Duplicate publications; (2) Studies lacking relevant outcome data; (3) Unavailable primary data after author contact; and (4) Non-English/Chinese publications.
The PRISMA criteria were adhered to[30]. EndNote 20 was utilized for literature management. Two researchers (Hong SC and Du YY) independently screened article titles and abstracts. Any disagreements during screening were resolved in consultation with another researcher (Wang GB). Relevant information was independently extracted and cross-checked by two researchers (Hong SC and Du YY), including: (1) Basic information on the article: Author’s name, year of pu
The data extraction process was conducted systematically by two independent reviewers who collected comprehensive information from each included study according to a predefined protocol. This encompassed four main categories of data: (1) Bibliographic details including authorship and publication year; (2) Methodological characteristics such as study design, sample size, intervention protocols for both treatment and control groups, specified outcome measures, and duration of intervention; (3) Demographic information about participants including age range and sex distribution; and (4) Contextual details regarding study location and healthcare setting. Any discrepancies between reviewers during this process were resolved through discussion and, when necessary, arbitration by a third senior investigator.
For quality assessment, we employed the Cochrane Risk of Bias tool (implemented in RevMan version 5.4) to rigo
The Grading of Recommendations Assessment, Development and Evaluation system and software (GRADE Pro GDT) were employed to rate the quality of all included RCTs. The overall certainty of evidence was categorized into four levels: High, moderate, low, or very low, based on the five GRADE domains. Any disagreements were resolved through consul
Review Manager software (version 5.4.1) was used for the meta-analysis. For continuous variables, effect estimates were calculated as mean differences (MD) with 95% confidence intervals (CI) presented in forest plots. If meta-analysis was not feasible, descriptive analysis was performed. A random-effects model was used to synthesize evidence. Sensitivity or subgroup analysis was conducted to identify the sources of heterogeneity. Sensitivity analysis was performed by sequentially deleting studies to ensure result stability. Subgroup analysis was based on the type of intervention (antidepressants or no intervention), antidepressant type, and CHF intervention duration (≤ 4 weeks or > 4 weeks). Publication bias was assessed using funnel plots when at least 10 trials were included. Statistical significance was set at P < 0.05.
The potential active ingredients from the top five herbs were identified using the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (CHFSP, https://CHFsp-e.com/CHFsp.php), applying cut-off criteria of oral bioavailability ≥ 30% and drug likeness ≥ 0.18. These ingredients were standardized using the UniProt protein database (https://www.uniprot.org). Breast cancer-related and depression-related targets were collected from the GeneCards database (https://www.genecards.org) (cut-off: Degree ≥ median). Interactions between top drug-related targets, breast cancer-related, and depression-related targets were analyzed using the STRING database (https://string-db.org), with a cut-off of a minimum interaction score of “highest confidence” (> 0.900). The results were visualized using Cytoscape 3.7.1. The intersection targets were imported into the Metascape platform for Gene Ontology and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis. Visualization analysis was performed using the Bioladder tool (https://www.bioladder.cn/).
Samples were collected from participants after 4-week SNS or placebo treatment in the previous study (ChiCTR2200065009). The original clinical trial was approved by the Ethics Committee of Guangdong Provincial Hospital of Chinese Medicine (Approval No. 2022-177-01). Written informed consent was obtained from all participants, which included consent for the secondary analysis of biospecimens for mechanistic research.
The enrolled breast cancer patients were in the rehabilitation phase (≥ 3 months post-completion of surgery, chemo
The clinical validation cohort comprised a total of n = 20 patients from the previous trial. They were allocated equally to the SNS treatment group (n = 10) and the matching placebo group (n = 10). Blood samples were collected between 7-9 AM after a 12-hour overnight fast. Samples were processed within 30 minutes of collection: Centrifuged at 1500 × g for 10 minutes at 4 °C, aliquoted into cryovials, and stored at -80 °C.
Peripheral blood samples of patients were collected and T lymphocyte subsets were detected by flow cytometry (FACS CANTO II, BD, San Jose, CA, United States). BD Multitest CD3/CD8/CD45/CD4 reagent (No. 340499, BD, San Jose, CA, United States) was used for the T lymphocyte percentage determinations. Laboratory tests were completed by the clinical laboratory department of Guangdong Provincial Hospital of Chinese Medicine.
Blood sample cytokines were examined using a QAH-CYT-1 antibody array (RayBiotech, United States), following the manufacturer’s guidelines. To measure cytokine secretion levels, 90 μL of concentrated conditioned medium was added to the array, incubated overnight at 4 °C, and then washed with a wash buffer. Biotinylated primary antibodies were added and incubated for 2 hours at room temperature, followed by additional washing. Subsequently, Cy3 equivalent dye-conjugated streptavidin was added and incubated for 1 hour at room temperature before scanning the array with an InnoScan 300 Microarray Scanner (Innopsys, France). An 8-point standard curve for each target protein was created using array-specific protein standards, and cytokine quantification was based on a standard curve generated from the same array. Partial least squares-discriminant analysis was conducted online through BioLadder (bioladder.cn).
The analysis of blood sample metabolites was conducted using an Ultimate U3000 HPLC system (Thermo Fisher Scientific, MA, United States) coupled with a Q Exactive Plus mass spectrometer (Thermo Fisher Scientific, MA, United States). Pooled quality control samples were analyzed every 10 injections. Pearson correlation analysis of quality control results demonstrated that R-values approaching 1 indicate superior analytical process stability and higher data quality. Chromatography was carried out on an Acquity UPLC BEH C18 column (1.7 μm, 100 mm × 2.1 mm; Waters), with solvent A composed of ultrapure water and solvent B of acetonitrile (CAS 75-05-08; Merck, Germany). The gradient conditions included: 5% solvent B for 0-1 minute, a transition to 5%-100% solvent B over 19 minutes, a hold at 100% solvent B for 1 minute, and a final return to 5% solvent B for 4 minutes. The flow rate was set at 200 μL/minute, with a column temperature of 37 °C and an injection volume of 5 μL. Mass spectrometer settings were as follows: Spray voltage of 2.5 kV in negative ion mode and 3.0 kV in positive ion mode; sheath gas flow of 40; auxiliary gas flow of 10; and sweep cone gas flow of 0. The capillary temperature was maintained at 320 °C. Data acquisition and analysis were performed using Xcalibur 4.0 software (Thermo Fisher Scientific, MA, United States). Subsequent to raw data acquisition, Compound Discover 3.0 software (Thermo Fisher Scientific, MA, United States) was employed for processing. This involved normalizing the original metabolomics data across batches for comparison. Metabolite identification was based on m/z ratios (± 3 ppm error), retention times, and comparison with a mass spectrometry database. Principal coordinates analysis was conducted online through BioLadder (bioladder.cn). All metabolites reported in this study adhered to the standards set by the Metabolomics Standards Initiative.
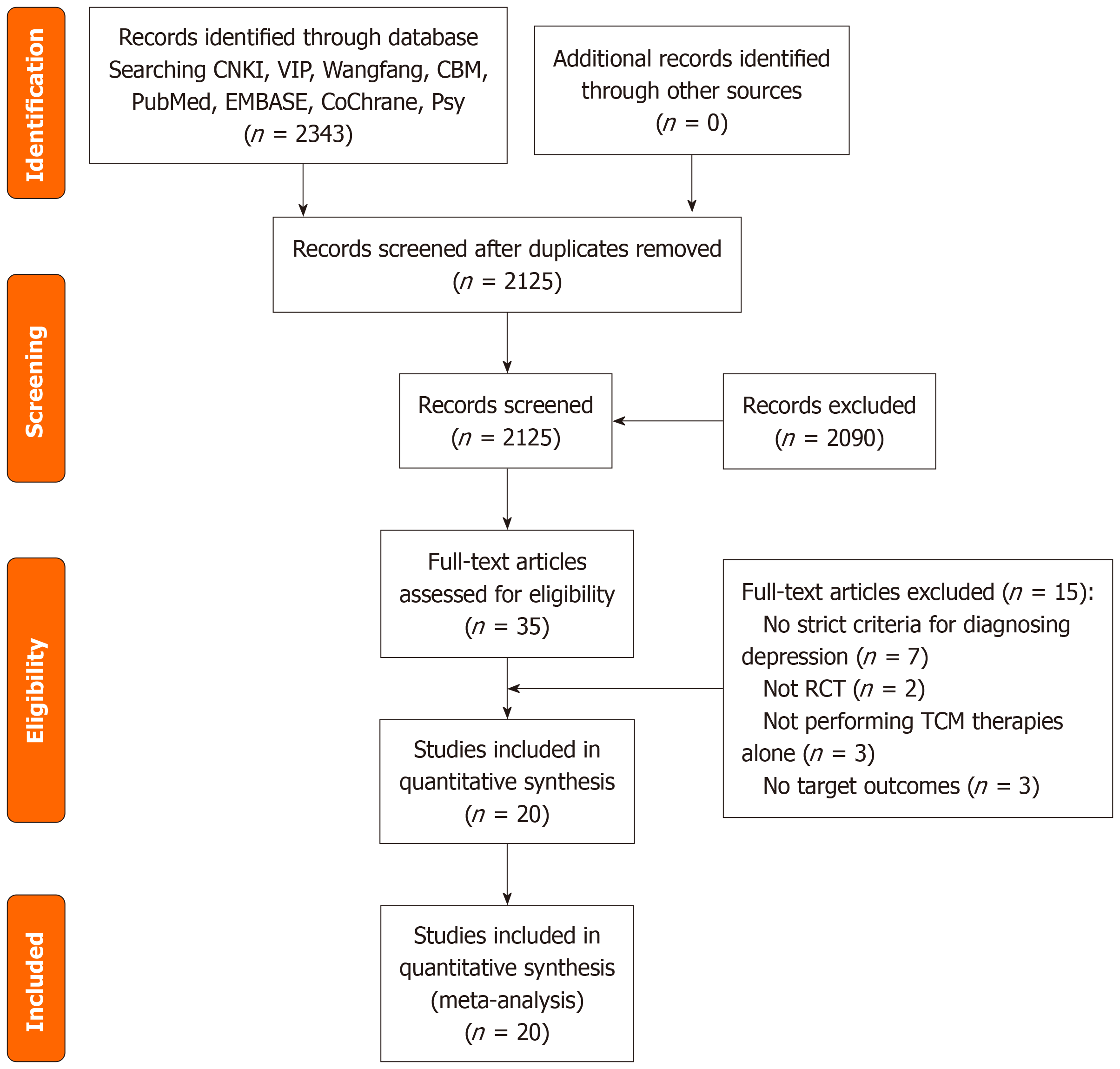
Study selection and characteristics: The systematic search identified 2343 records from eight databases. After removing 218 duplicates, 2125 abstracts were screened against eligibility criteria (Figure 1). Full-text review of 35 articles yielded 20 qualifying RCTs (n = 1607 patients), including: 18 trials (n = 1409) comparing CHF with antidepressants, 2 trials (n = 198) evaluating CHF vs no intervention. All studies were conducted in China with treatment durations ranging from 4 weeks to 6 months. Outcome reporting included: 18 trials evaluating depression scales (HAMD/SDS), four trials evaluating QOL, five trials evaluating neurotransmitter levels (5-HT/DA/NE) and two trials evaluating immune markers (CD4+/CD8+ T cell subsets).
Methodological quality of included studies: The risk of bias for the included studies is depicted in Supplementary Figure 1. Eleven RCTs employed the random number method and were assessed as having a low risk of random sequence generation[31-41]. The remaining nine RCTs did not specify their randomization methods, resulting in unclear risk[42-50]. None of the studies explicitly mentioned allocation concealment or blinding, leading to high or unclear risk of bias in these domains. All 20 studies completely reported outcome data, with similar reasons for missing data, thus rated as low risk of attrition bias. The characteristics of included trials were shown in Table 1. The GRADE assessment indicated moderate-quality evidence for primary outcomes (HAMD, SDS, QOL), as detailed in Supplementary Table 3.
| Ref. | Samples | Age (mean ± SD) | Intervention/experimental group | Control group | Intervention time/period | Outcomes | |
| Liu and Xiao[44], 2011 | 60 | E: 51.40 ± 8.7 | C: 51.30 ± 6.6 | Modified Xiaoyaosan (n = 30) | Flupentixol and melitracen tablets (n = 30) | 6 weeks | (1) |
| Zhang[49], 2011 | 96 | E: 35-70 | C: 36-69 | Modified Ganmaidazao decoction plus Xiaoyaosan (n = 48) | Alprazolam (n = 48) | 6 weeks | (1)(2)(3) |
| Sun et al[46], 2015 | 58 | E: 45.9 ± 11.7 | C: 58.6 ± 35 | Modified Xiaoyaosan (n = 29) | Flupentixol and melitracen tablets (n = 29) | 6 weeks | (2) |
| Yan et al[47], 2015 | 68 | 38-69 | Modified Xiaoyaosan (n = 34) | Paroxetine hydrochloride tablets (n = 34) | 4 weeks | (1) | |
| Zhang et al[48], 2015 | 60 | E: 40-65 | C: 35-67 | Modified Xiaoyaosan (n = 30) | Fluoxetine hydrochloride tablets (n = 30) | 3 weeks | (1) |
| Jin et al[43], 2016 | 82 | E: 23-65 | C: 24-67 | Fuzhengjieyu decoction (n = 41) | Alprazolam (n = 41) | 10 weeks | (1)(5) |
| Sun et al[36], 2016 | 64 | 24-73 | Modified Xiaoyaosan (n = 32) | No intervention (n = 32) | 6 weeks | (2)(4) | |
| Chang et al[32], 2017 | 84 | E: 39-70 | C: 38-68 | Shuganxiaobi decoction (n = 42) | Fluoxetine hydrochloride tablets (n = 42) | 4 weeks | (1) |
| Shi et al[45], 2017 | 66 | E: 35-68 | C: 32-71 | Modified Chaihuguizhi decoction (n = 36) | Paroxetine hydrochloride tablets (n = 30) | 8 weeks | (1) |
| Hu et al[34], 2018 | 80 | E: 47.37 ± 8.60 | C: 48.23 ± 8.10 | Jiawei Xiaoyao pill (n = 40) | Venlafaxine hydrochloride sustained-release tablets (n = 40) | 1 month | (4) |
| Yang[50], 2018 | 92 | E: 49.12 ± 7.96 | C: 48.86 ± 8.13 | Chaihulonggumuli decoction (n = 46) | Fluoxetine hydrochloride tablets (n = 46) | 1 month | (1) |
| He[33], 2019 | 70 | E: 51.83 ± 8.15 | C: 50.77 ± 9.20 | Danzhixiaoyao powder (n = 35) | Flupentixol and melitracen tablets (n = 35) | 4 weeks | (1) |
| Huang et al[42], 2019 | 77 | E: 46.26 ± 2.32 | C: 45.72 ± 2.05 | Modified Ganmaidazao decoction (n = 39) | Amitriptyline hydrochloride tablets (n = 38) | 4 weeks | (1)(3) |
| Jiang[37], 2019 | 60 | E: 42.08 ± 5.10 | C: 41.93 ± 5.37 | Chaihu plus Longgumuli decotion (n = 30) | Fluoxetine hydrochloride tablets (n = 30) | 4 weeks | (1) |
| Wang et al[38], 2019 | 134 | E: 42.3 ± 12.34 | C: 40.57 ± 13.91 | Xiaoyao powder (n = 67) | No intervention (n = 67) | 6 months | (1)(3)(5) |
| Lin[35], 2022 | 70 | E: 47.33 ± 5.96 | C: 46.82 ± 6.14 | Jieyusanjie decoction (n = 35) | Sertraline hydrochloride tablets (n = 35) | 4 weeks | (1) |
| Xiao[39], 2021 | 100 | E: 45.81 ± 11.56 | C: 46.37 ± 12.21 | Modified Xiaoyao powder (n = 50) | Paroxetine Hydrochloride tablets (n = 50) | 3 months | (1)(4) |
| An et al[31], 2022 | 120 | E: 55.76 4 ± 8.81 | C: 56.05 ± 8. 73 | Chaihu plus Longgumuli decoction (n = 60) | Escitalopram tablets (n = 60) | 60 days | (1)(4) |
| Zhang[40], 2022 | 60 | E: 54.67 ± 6.87 | C: 54.20 ± 7.56 | Chaihushugan powder (n = 30) | Flupentixol and melitracen tablets (n = 30) | 4 weeks | (2)(3) |
| Zhong et al[41], 2023 | 106 | E: 52.09 ± 9.07 | C: 51.23 ± 8.91 | Modified Fangjidihuang (n = 53) | Sertraline hydrochloride tablets (n = 53) | 8 weeks | (1)(4) |
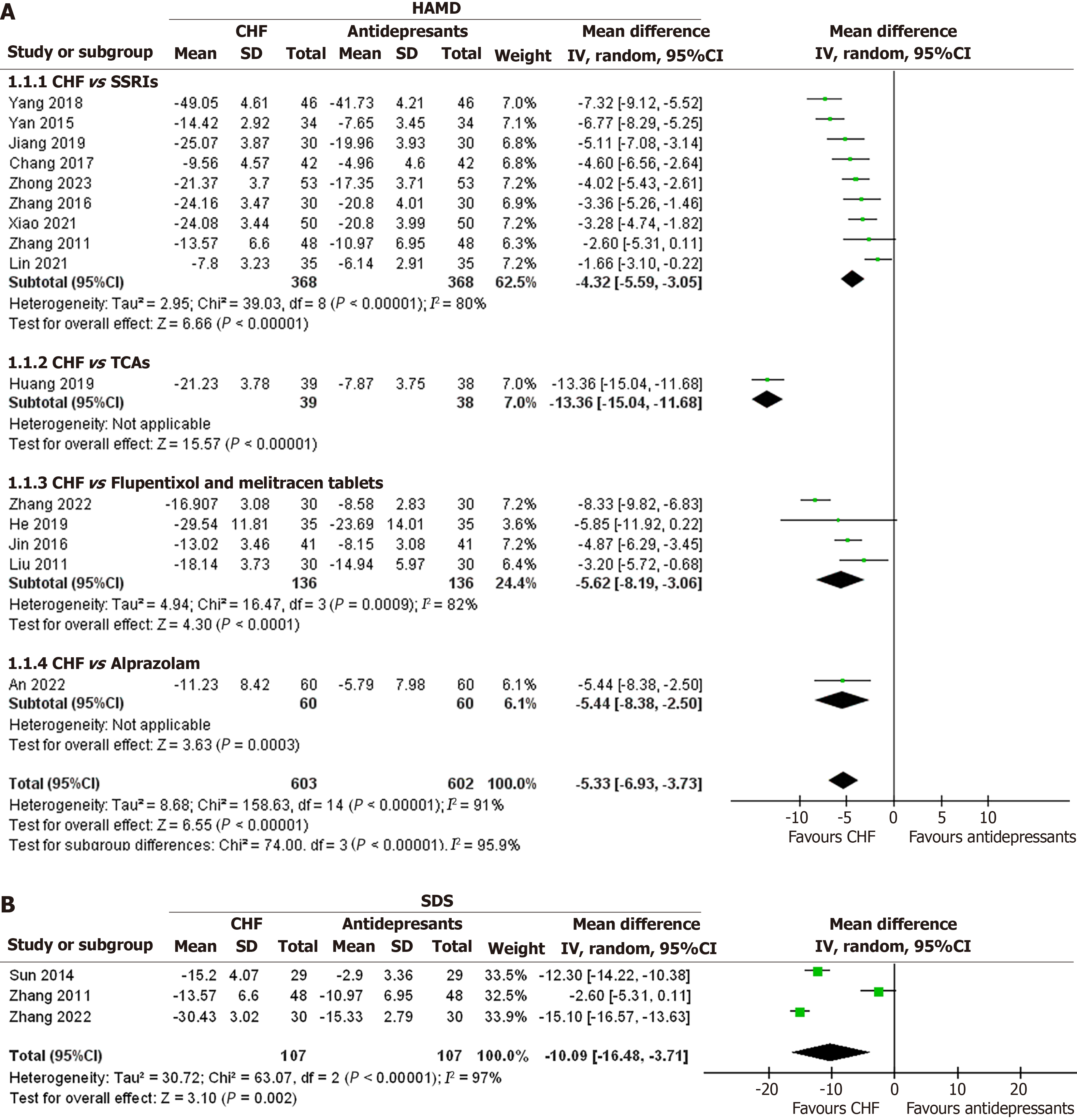
Therapeutic efficacy of CHF on relieving depression in breast cancer patients: A total of 18 RCTs involving 1461 patients reported depression outcomes. The meta-analysis of direct comparisons included 16 RCTs (n = 1263) that compared CHF with antidepressants, using either HAMD or SDS. Two additional RCTs (n = 198) comparing CHF with no intervention were described narratively due to differences in measurement tools. For the comparison between CHF and antidepressants assessed by HAMD, 15 studies involving 1205 breast cancer patients were included (Figure 2A). CHF therapies significantly reduced HAMD scores compared to selective serotonin reuptake inhibitors (MD = -4.32, 95%CI: -5.59 to -3.05, P < 0.00001). Besides, CHF also showed superiority over flupentixol and melitracen in improving depression scores (MD = -5.62, 95%CI: -8.19 to -3.06, P < 0.0001). CHF presented a significant advantage in reducing depression score compared with antidepressants (MD = -5.33, 95%CI: -6.93 to -3.73, P < 0.00001). For the comparison between CHF and antidepressants assessed by SDS, three studies involving 214 breast cancer patients were included (Figure 2B). CHF significantly reduced depression scores compared to antidepressants (MD = -10.09, 95%CI: -16.48 to 3.71, P = 0.002). In addition, compared to no intervention, Sun et al[36] reported CHF reduced SDS scores (MD = -15.43, 95%CI: -21.33 to -9.52), while Wang et al[38] reported CHF reduced HAMD scores (MD = -9.41, 95%CI: -14.14 to -4.68).
The influence of CHF on improving QOL in breast cancer patients: Four RCTs involving 367 patients reported on QOL as assessed by the Karnofsky Performance Scale and were included in the meta-analysis (Supplementary Figure 2). Results from three studies involving 233 breast cancer patients showed that CHF treatment significantly improved QOL compared to antidepressants (MD = 9.13, 95%CI: 6.69-11.57, P < 0.00001). One study also reported that CHF was significantly better than no intervention.
The influence of CHF treatment duration on depression and frequency statistics of herbal medicine: In order to evaluate the influence of treatment duration on the therapeutic efficacy of CHF, a subgroup analysis was performed according to different intervention duration (≤ 4 weeks or > 4 weeks). As shown in Supplementary Figure 3, CHF treatment could lead to improvements in depression either for using shorter or longer than four weeks when compared to antidepressants. Notably, patients with treatment duration exceeding four weeks seemed to obtain more advantage in relieving depression (MD = -4.99, 95%CI: -6.34 to -3.64, P < 0.00001) compared to the antidepressant group. These findings revealed that the intervention duration may be a crucial factor influencing the therapeutic efficacy of CHF.
Safety outcomes: In this study, ten RCTs reported that the incidence of adverse events (AEs). AEs were actively collected through structured interviews at each study visit and/or patient diaries in the original trials. The reported AEs included dry mouth, constipation, dizziness and headache, poor appetite and indisgestion, insomnia, abdominal pain, somnolence, lack of strength, nausea and vomiting, liver and kidney dysfunction, alopecia, blurred vision, as well as fever. The incidence of these AEs may be attributed to breast cancer patients undergoing concurrent radiotherapy, chemotherapy, and endocrine therapy often experience varying degrees of breast cancer treatment-related adverse effects. As shown in Supplementary Figure 4, the CHF treatment group was associated with a significantly lower incidence of adverse reactions compared to the control group (risk ratio = 0.36, 95%CI: 0.25-0.51, P < 0.00001).
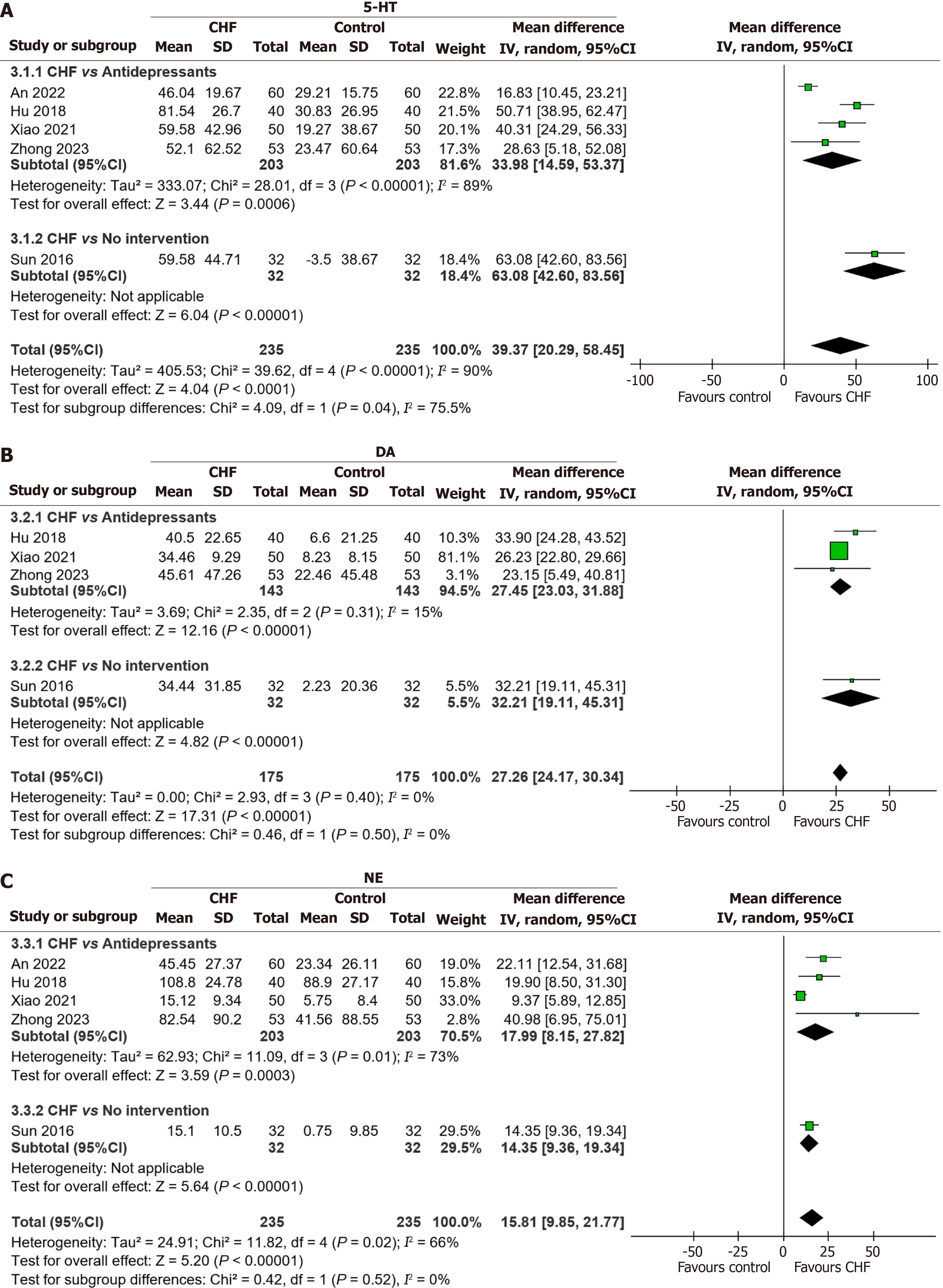
The influence of CHF on 5-HT, DA, and NE in breast cancer patients: Data on 5-HT and NE levels were reported in five RCTs (n = 470), while DA levels (pre- and post-treatment) were reported in four RCTs (n = 350); all were included in the meta-analysis. Compared to antidepressants, CHF significantly increased 5-HT (MD = 33.98, 95%CI: 14.59-53.37, P = 0.0006), DA (MD = 27.45, 95%CI: 23.03-31.88, P < 0.00001), and NE (MD = 17.99, 95%CI: 8.15-27.82, P = 0.0003) (Figure 3). One study also favored CHF over no intervention in regulating 5-HT, DA, and NE[34].
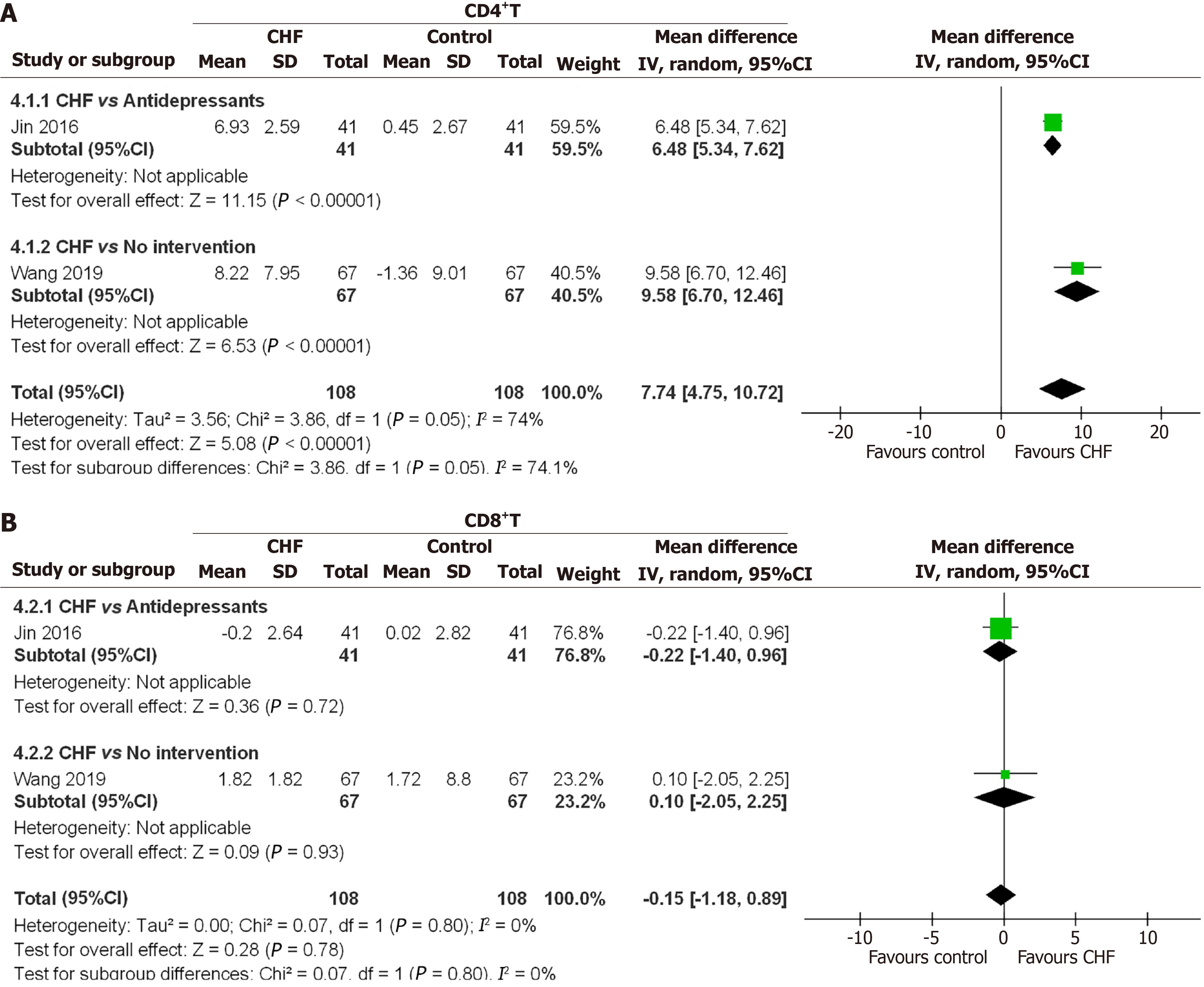
The influence of CHF on CD4+ and CD8+ T lymphocytes in breast cancer patients: Two RCTs involving 216 cases reported the changes of CD4+ and CD8+ T lymphocytes, one vs antidepressants while another vs no intervention, respectively. All were included in the meta-analysis. It was found that CHF treatment greatly improved CD4+ T population either comparing with antidepressants or placebo group (MD = 7.74, 95%CI: 4.75-10.72, P < 0.00001). However, little statistical difference was found in the clinical efficacy of CHF elevating CD8+ T population (MD = -0.15, 95%CI: -1.18 to 0.89, P = 0.78) (Figure 4).
Publication bias and sensitivity analysis: Evaluation of publication bias through funnel plot analysis revealed no complete symmetry and indicated the existence of publication bias (Supplementary Figure 5). The publication bias may be associated with negative results not being published. To assess result stability, we performed sensitivity testing by systematically removing each study and recalculating effect sizes, which demonstrated consistent treatment effects across all iterations, supporting the reliability of our primary conclusions.
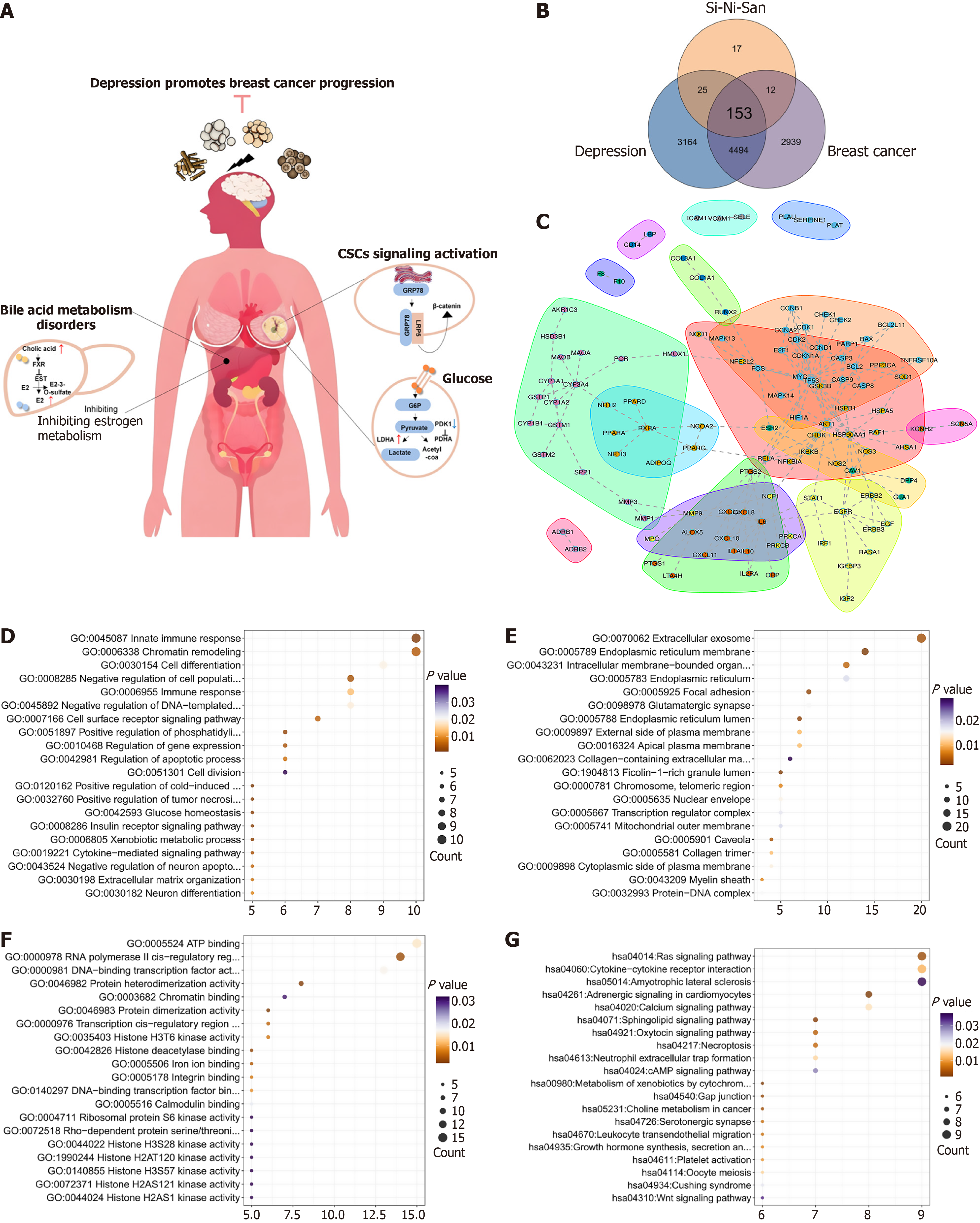
The meta-analysis indicated that the majority of the formulations included were derived from the classical antidepressant formula SNS. Preclinical evidence supports the potential of SNS in suppressing breast cancer growth and metastasis induced by chronic psychological stress (Figure 5A). Specifically, SNS disrupts the 78kDa glucose regulated protein-lipoprotein receptor-related protein 5 interaction on the cell membrane, thereby inhibiting Wnt/β-catenin signaling in breast cancer stem cells[25]. Furthermore, naringenin, a bioactive compound present in SNS, has demonstrated the ability to impede breast cancer progression through modulation of the farnesoid X receptor/hepatocyte nuclear factor 4α/estrogen sulfotransferase pathway[26]. SNS also downregulates the glycolysis-related proteins that are upregulated by chronic psychological stress[51]. Nevertheless, a thorough understanding of the pharmacological mechanisms underlying the use of SNS for the treatment of depression in patients with breast cancer necessitates additional research.
Using network pharmacology, we identified 57 active ingredients within SNS. Meanwhile, 193 unique potential targets of these components were retained after removing duplicates. Furthermore, we obtained 9151 targets related to breast cancer and 9121 targets associated with depression from GeneCards. Intersection of these target sets yielded 153 common genes (Figure 5B). Protein-protein interaction analysis further highlighted a total of 93 significant genes, including interleukin-10 (IL10), IL1A, CXC motif chemokine ligand 11 (CXCL11), CXCL10, IL6, CXCL2, CXCL8, etc. (Figure 5C). Gene Ontology enrichment analysis indicated that these genes are primarily involved in immune-related biological processes such as positive regulation of transcription driven by immune response and tumor necrosis factor production (Figure 5D). Within the cellular component category, extracellular exosome was the most significantly enriched term (Figure 5E). For molecular function, ATP binding was the most prominent (Figure 5F). KEGG pathway analysis revealed significant enrichment in pathways including choline metabolism in cancer and the serotonergic synapse pathway (Figure 5G), both of which are implicated in cancer progression and depression.
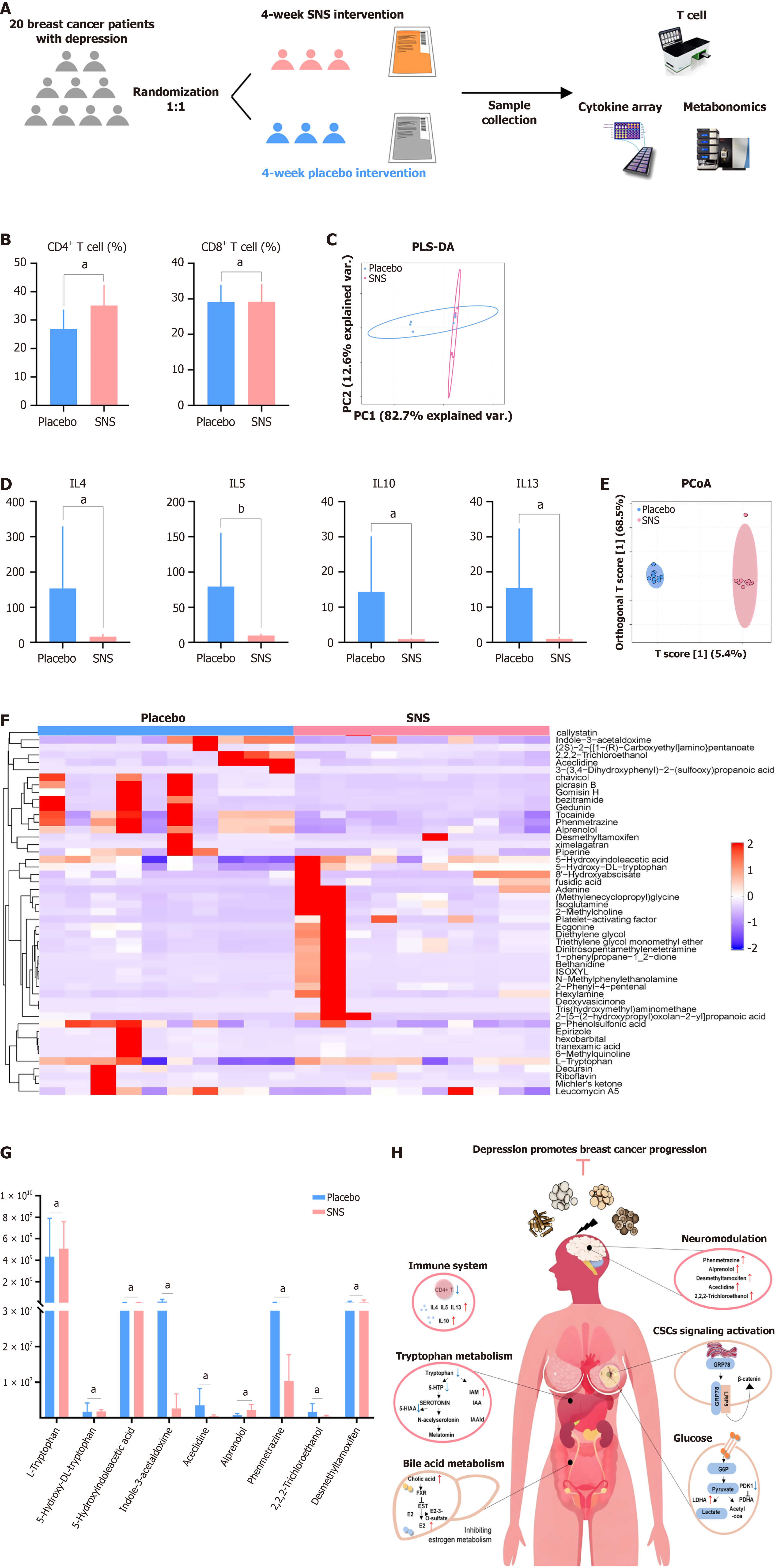
Despite the encouraging preclinical data that suggest the effectiveness of SNS in ameliorating depressive symptoms and enhancing the prognosis of breast cancer, there is a paucity of well-controlled clinical trials among breast cancer patients with depression. To validate our findings in meta-analysis and network pharmacology, we utilized clinical specimens derived from a trial intended to assess the effectiveness of SNS in treating breast cancer patients experiencing depression. The experimental flow chart was summarized in Figure 6A. Notably, SNS administration significantly increased CD4+ T cell levels in two independent comparisons (P < 0.05; Figure 6B). Meanwhile, partial least squares-discriminant analysis of cytokine profiles revealed clear separation between the SNS and placebo groups (Figure 6C), indicating that SNS markedly altered the cytokine landscape between groups. In particular, the key cytokines derived from CD4+ T cells, including IL-4, IL-5, IL-10, and IL-13, were significantly downregulated following SNS treatment (Figure 6D), further underscoring the pivotal role of CD4+ T cells in SNS-mediated antidepressant effects.
Metabolomic profiling via principal coordinates analysis also showed distinct clustering between groups (Figure 6E), demonstrating substantial shifts in the metabolome. Upon the application of a stringent threshold (|fold change| > 5), we identified 49 metabolites exhibiting differential expression (Figure 6F). Among these, the levels of L-tryptophan, 5-hydroxy-DL-tryptophan, and 5-hydroxyindoleacetic acid were increased significantly following SNS treatment (P < 0.05). Meanwhile, the levels of metabolites such as indole-3-acetaldoxime, aceclidine, alprenolol, phenmetrazine, 2,2,2-trichloroethanol and desmethyltamoxifen were markedly inhibited after SNS treatment (P < 0.05; Figure 6G). Therefore, integrating these metabolic alterations with the established pathophysiology of depression and previous findings[24-26] (Figure 6H), we propose that SNS alleviates depression through a multi-system mechanism encompassing immunomodulation, systemic neuromodulation, metabolic reprogramming, and pathways related to cancer stem cells and glycolysis.
In this study, we revealed that CHF intervention showed great efficacy in relieving depression, and demonstrated a stronger alleviating effect on symptoms when compared with antidepressants. Additionally, CHF therapies significantly enhance QOL and increase serum levels of 5-HT, DA and NE, while improving the immune index of CD4+ T lymphocytes. It is noteworthy that the classical formula SNS has emerged as a foundational element supporting these therapeutic effects. Further experimental analyses indicated that SNS may exert its antidepressant actions primarily through modulating CD4+ T cell function and multi-organ regulation (Figure 7).
Unlike previous meta-analysis of CHF applied in mental disorders[52-54] or mixed cancers[55,56], our study focused on breast cancer patients with depression. Compared with depression in general population, breast cancer related depression is persistent, more disseminated and poorly controlled[57]. Meanwhile, the impact of antidepressants on breast cancer progression is also a serious concern. Long-term use of antipsychotic medications has been linked to an increased risk of breast cancer[58]. Notably, CYP2D6 inhibitors like paroxetine, when co-administered with tamoxifen, have been reported to increase breast cancer-related mortality[59]. These factors significantly affect breast cancer patients’ compliance with antidepressants due to potential drug interactions with chemotherapeutic agents or biological therapies. In contrast, CHF, which have been used for millennia to treat mental disorders, are characterized by multiple targets, low toxicity, and systematic efficacy. Consistent with previous reports[60-62], we also validated that CHF can significantly improve depressive symptoms of breast cancer patients as an adjunctive therapy. Notably, our results showed that the formulas present better efficacy than antidepressants when the administration duration was longer than four weeks. Considering the mild pharmacological activity of CHF, we speculate that CHF may require a longer duration to reach consistent efficacy. As expected, our results showed that the formulas present better efficacy than antidepressants when the administration duration was longer than four weeks. This temporal pattern likely reflects CHF’s gradual system-regulating effects rather than immediate pharmacological action. In addition, the significantly reduced AE incidence further supports CHF’s clinical utility, as recognized by leading institutions like MD Anderson Cancer Center through their incorporation of Chinese medicine into integrative oncology programs[63]. These collective findings position CHF as a valuable complementary approach for managing depression in breast cancer patients, though additional controlled trials are needed to establish optimal treatment protocols and duration. Research should particularly focus on standardizing formulations and evaluating long-term outcomes to facilitate clinical implementation. In addition, CHF has demonstrated distinctive therapeutic advantages in managing numerous complex conditions, such as rheumatoid arthritis, irritable bowel syndrome, and mammary hyperplasia[64-67]. These clinical benefits are fundamentally attributable to their multi-target, multi-component, and multi-level regulatory mechanisms, which enable a holistic modulation of disease networks. For example, beyond its efficacy in breast cancer-related depression, SNS has been shown to alleviate a spectrum of disorders including adolescent depression, liver injury, acute colitis, and anxious insomnia[68-71]. Our study also revealed that the pharmacological activities of SNS were closely correlated with the modulation of immune system, tryptophan metabolism, bile acid metabolism, neurotransmitters, glucose metabolism, as well as cancer stem cell signaling activation[26,51,72]. Collectively, these phenomena underscore the core TCM principle of “treating different diseases with the same strategy”, wherein a single formula exerts therapeutic effects across multiple diseases through its system-regulating action.
The pathophysiology of depression is complex and involves multiple pathways, including the hypothalamic-pituitary-adrenal axis, the gut-brain-microbiome axis, brain-derived neurotrophic factor, monoamine neurotransmitters, and cytokines[73]. The monoamine hypothesis of depression posits that the dysregulation of 5-HT, NE and DA contributes to depression occurrence and establishes the cornerstone of current pharmacological treatments for depression[74]. In the present study, we observed that CHF interventions were more effective than antidepressants in increasing levels of 5-HT, DA, and NE. KEGG pathway analysis highlighted the neuroactive ligand-receptor interaction pathway as significantly enriched, suggesting that CHF may modulate the key neurotransmitter system to improve the prognosis of depression. This is consistent with previous meta-analyses that have reported an elevation of 5-HT, DA, and NE levels following treatment with CHF[75]. With advancements in multidisciplinary techniques, the antidepressant mechanisms of CHF have been increasingly elucidated.
To elucidate the specific mechanisms underlying this modulation, we conducted an untargeted metabolomic analysis of the foundational formula SNS. The findings uncovered a synchronized reorganization of metabolic processes, characterized by a deliberate improvement of the serotonergic pathway, as indicated by marked increases in L-tryptophan, 5-hydroxy-DL-tryptophan, and 5-hydroxyindoleacetic acid, coupled with a reduction in Indole-3-acetaldoxime. This signature suggests a shift in tryptophan metabolism away from competing catabolic routes and toward serotonin synthesis, a process potentially facilitated via the gut-brain axis[76,77]. Concurrently, we observed a marked decline in various neuroactive substances, including phenmetrazine, alprenolol, desmethyltamoxifen, aceclidine and 2,2,2-trichloroethanol. This coordinated decrease signals a reduction in the overall neuroactive load, reflecting a systemic neuromodulatory reset induced by SNS treatment. Integrating these metabolic findings with the established pathophysiology of depression and prior findings[24-26], we propose that SNS exerts its antidepressant effect via a multi-system mechanism. This mechanism entails immunomodulation, a reduction in neuroactive burden, a reprogramming of tryptophan meta
Immune suppression microenvironment plays a crucial role in cancer development, particularly given the pivotal role of T lymphocytes in the anti-cancer battle[78]. The interplay between immune indices and depression has also emerged as a significant topic in oncology research[79]. In this study, CHF treatment was significantly more effective in increasing CD4+ T lymphocytes compared to antidepressant intervention. This finding aligns with studies reporting a negative correlation between CD4+ T levels and depression, suggesting that an increased CD4+ population may ameliorate depressive symptoms[80]. Single-cell RNA sequencing analysis demonstrated a reduced presence of naive T cells, especially CD4+ naive T cells, in depression patients relative to controls, and highlighted the ETS-mitogen-activated protein kinase pathway in T cell activation during antidepressant treatment in major depressive disorder[81]. Meanwhile, higher levels of CD3+, CD4+, and CD8+ T cells have been associated with improved survival rates among breast cancer patients[82]. In present study, CD4+ T cell and its key cytokines including IL-4, IL-5, IL-10, IL-13 were significantly modulated following SNS treatment. The observed increase in CD4+ T cell may reflect the activation of beneficial immune responses induced by SNS treatment. Simultaneously, the reduction in type 1 helper T (Th2)-associated cytokines, including IL-4, IL-5, and IL-13[83], likely indicates a favaroble shift, suggesting that SNS attenuates Th2-polarized immunity, which is typically deemed detrimental to anti-cancer defense mechanisms. As a key immunosuppressive cytokine predominantly produced by regulatory T cells, the decline in IL-10 likely reflects a reduction in regulatory T cell activity or abundance. Those points to a shift away from an immunosuppressive state towards a more immunopermissive microenvironment, which may not only restore anti-tumor immune competence but also contribute to the alleviation of inflammation-associated depressive symptoms. This immunomodulatory pattern is consistent with previously reported effects of various bioactive components in Chinese Medicine, including Ganoderma lucidum polysaccharide and Ginseng polysaccharides, which have been shown to inhibit Th2 cytokines and promote Th1 responses[84,85]. Collectively, these findings collectively underscore the unique advantage of immunomodulation effect offered by CHF.
QOL is a vital consideration for cancer survivors, as the five-year survival ratio has been greatly prolonged with the development of multidisciplinary strategies. For breast cancer, the poor QOL exited in most patients, with domains including physical, emotional and social domains[86]. Notably, QOL was also closely associated with mortality risk in breast cancer patients[87]. Our analysis confirms CHF’s positive impact across multiple QOL parameters, consistent with previous reports of improved functioning and symptom control[60,61]. American Cancer Society also suggest acupuncture and Qigong should be considered for improving QOL in cancer patients[29]. According to a dual-center trial in America, acupuncture improved QOL for patients with breast cancer over time[88]. Regarding CHF, breast cancer patients receiving formulas during cancer treatment reported higher QOL scores, compared to those without formula interventions[89]. Additionally, a significant improvement in QOL has been demonstrated for breast cancer patients receiving the CHF PRM1201 during chemotherapy, with multiple indexes including overall life quality, physical capacity, role performance, emotional well-being, and mitigating fatigue and appetite loss showing improvement after PRM1201 administration[90]. However, the beneficial effects of CHF are still needed to be validated by large-scale multi-center RCTs. These positive findings encourage the potential practice of CHF in enhancing QOL of breast cancer patients.
This study demonstrates the superior efficacy and safety of CHF for breast cancer-related depression, evidenced by improving depressive symptoms, QOL, monoamine neurotransmitter levels and immune function. Several limitations should be considered. First, the majority of included RCTs were conducted in China, and language restrictions may introduce selection bias and limit the sample size and generalizability of the findings. Second, the impact of CHF on varying severity levels of depression could not be assessed due to insufficient reporting in the original studies. Third, the potential benefits of CHF on long-term oncological outcomes such as overall survival or recurrence rate, remain unclear, as included trials had short follow-up periods. Furthermore, while mechanistic insights from SNS provide valuable clues for understanding the anti-depression mechanism of CHF, these findings require further experimental validation. To address these gaps, future research should prioritize large-scale, multi-regional RCTs that adhere to standardized protocols and reporting guidelines, such as the CONSORT statement. Emphasis should be placed on rigorous randomization, blinding, and allocation concealment. Additionally, the utilization of standardized assessment instruments to gauge the severity of depression and the integration of sustained longitudinal follow-up are imperative for confirming the enduring efficacy of CHF and its impact on survival metrics. Such efforts would enhance the reproducibility, translational value, and global applicability of CHF in treating breast cancer-related depression.
In conclusion, this study suggests that CHF, particularly SNS, is associated with greater improvement in depression outcomes compared to conventional antidepressants among breast cancer patients. This is supported by observed positive effects on monoamine neurotransmitter levels and CD4+ T lymphocyte counts. However, these conclusions must be interpreted with caution given the methodological limitations of the included trials, such as inadequate blinding and allocation concealment, as well as the observed funnel plot asymmetry. While the findings support the potential integration of CHF into management strategies for breast cancer-related depression, further rigorous, large-scale RCTs with robust blinding and longer follow-up are needed to confirm these benefits, elucidate the mechanisms, and assess the long-term impact on both depression and oncological outcomes.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14995] [Article Influence: 7497.5] [Reference Citation Analysis (23)] |
| 2. | Plevritis SK, Munoz D, Kurian AW, Stout NK, Alagoz O, Near AM, Lee SJ, van den Broek JJ, Huang X, Schechter CB, Sprague BL, Song J, de Koning HJ, Trentham-Dietz A, van Ravesteyn NT, Gangnon R, Chandler Y, Li Y, Xu C, Ergun MA, Huang H, Berry DA, Mandelblatt JS. Association of Screening and Treatment With Breast Cancer Mortality by Molecular Subtype in US Women, 2000-2012. JAMA. 2018;319:154-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 266] [Cited by in RCA: 234] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 3. | DeSantis CE, Bray F, Ferlay J, Lortet-Tieulent J, Anderson BO, Jemal A. International Variation in Female Breast Cancer Incidence and Mortality Rates. Cancer Epidemiol Biomarkers Prev. 2015;24:1495-1506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 613] [Cited by in RCA: 508] [Article Influence: 46.2] [Reference Citation Analysis (4)] |
| 4. | Qiu H, Cao S, Xu R. Cancer incidence, mortality, and burden in China: a time-trend analysis and comparison with the United States and United Kingdom based on the global epidemiological data released in 2020. Cancer Commun (Lond). 2021;41:1037-1048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 852] [Cited by in RCA: 731] [Article Influence: 146.2] [Reference Citation Analysis (4)] |
| 5. | Trapani D, Ginsburg O, Fadelu T, Lin NU, Hassett M, Ilbawi AM, Anderson BO, Curigliano G. Global challenges and policy solutions in breast cancer control. Cancer Treat Rev. 2022;104:102339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 209] [Article Influence: 52.3] [Reference Citation Analysis (0)] |
| 6. | Pilevarzadeh M, Amirshahi M, Afsargharehbagh R, Rafiemanesh H, Hashemi SM, Balouchi A. Global prevalence of depression among breast cancer patients: a systematic review and meta-analysis. Breast Cancer Res Treat. 2019;176:519-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 228] [Article Influence: 32.6] [Reference Citation Analysis (3)] |
| 7. | Hashemi SM, Rafiemanesh H, Aghamohammadi T, Badakhsh M, Amirshahi M, Sari M, Behnamfar N, Roudini K. Prevalence of anxiety among breast cancer patients: a systematic review and meta-analysis. Breast Cancer. 2020;27:166-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 144] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 8. | Elimimian EB, Elson L, Stone E, Butler RS, Doll M, Roshon S, Kondaki C, Padgett A, Nahleh ZA. A pilot study of improved psychological distress with art therapy in patients with cancer undergoing chemotherapy. BMC Cancer. 2020;20:899. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 9. | Wang X, Wang N, Zhong L, Wang S, Zheng Y, Yang B, Zhang J, Lin Y, Wang Z. Prognostic value of depression and anxiety on breast cancer recurrence and mortality: a systematic review and meta-analysis of 282,203 patients. Mol Psychiatry. 2020;25:3186-3197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 387] [Cited by in RCA: 340] [Article Influence: 56.7] [Reference Citation Analysis (0)] |
| 10. | Maddock C, Pariante CM. How does stress affect you? An overview of stress, immunity, depression and disease. Epidemiol Psichiatr Soc. 2001;10:153-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 75] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 11. | Argentieri MA, Nagarajan S, Seddighzadeh B, Baccarelli AA, Shields AE. Epigenetic Pathways in Human Disease: The Impact of DNA Methylation on Stress-Related Pathogenesis and Current Challenges in Biomarker Development. EBioMedicine. 2017;18:327-350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 127] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 12. | Strine TW, Mokdad AH, Dube SR, Balluz LS, Gonzalez O, Berry JT, Manderscheid R, Kroenke K. The association of depression and anxiety with obesity and unhealthy behaviors among community-dwelling US adults. Gen Hosp Psychiatry. 2008;30:127-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 444] [Cited by in RCA: 383] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 13. | Kisely S, Crowe E, Lawrence D. Cancer-related mortality in people with mental illness. JAMA Psychiatry. 2013;70:209-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 274] [Article Influence: 21.1] [Reference Citation Analysis (1)] |
| 14. | Pitman A, Suleman S, Hyde N, Hodgkiss A. Depression and anxiety in patients with cancer. BMJ. 2018;361:k1415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 615] [Cited by in RCA: 472] [Article Influence: 59.0] [Reference Citation Analysis (0)] |
| 15. | Ostuzzi G, Matcham F, Dauchy S, Barbui C, Hotopf M. Antidepressants for the treatment of depression in people with cancer. Cochrane Database Syst Rev. 2018;4:CD011006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 78] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 16. | Zha LH, He LS, Lian FM, Zhen Z, Ji HY, Xu LP, Tong XL. Clinical Strategy for Optimal Traditional Chinese Medicine (TCM) Herbal Dose Selection in Disease Therapeutics: Expert Consensus on Classic TCM Herbal Formula Dose Conversion. Am J Chin Med. 2015;43:1515-1524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 17. | Wang Y, Li JW, Qin YN, Sun CP, Chen JJ, Ruan YY, Chen LX, Liu S, Liu GY. Clinical observation on the effect of Chinese medicine-"TCM formula" intervention on recurrence and metastasis of triple negative breast cancer. Complement Ther Med. 2020;52:102456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 18. | Hao W, Ma Q, Wang L, Yuan N, Gan H, He L, Li X, Huang J, Chen J. Gut dysbiosis induces the development of depression-like behavior through abnormal synapse pruning in microglia-mediated by complement C3. Microbiome. 2024;12:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 82] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 19. | Han Y, Wang H. [Curative effect observation of Sini San Jiawei for treatment of 30 cases functional indigestion accompanied by depression]. Beijing Zhongyiyao. 2011;30:457-458. |
| 20. | Chu C. [Clinical Effect of Sini San Combined with Xiaoyao Wan in Treatmenting Postpartum Depression]. Xinzhongyi. 2015;47:120-122. [DOI] [Full Text] |
| 21. | He X, Zhang R, Li Z, Yao Z, Xie X, Bai R, Li L, Zhang X, Zhang S, Shen Y, Li S, Hui Z, Liu R, Chen J. Sini powder with paroxetine ameliorates major depressive disorder by modulating circadian rhythm: A randomized, double-blind, placebo-controlled trial. J Pineal Res. 2022;73:e12832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 22. | Xie J, Geng X, Fan F, Fu X, He S, Li T. The efficacy of therapies for post-stroke depression in aging: An umbrella review. Front Aging Neurosci. 2022;14:993250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 23. | Cai L, Jiejie L, Hu Y, Chen W, Wei L, He Q, Hu Y. Efficacy and safety of modified Sini San for treating poststroke depression: A meta-analysis of randomized controlled trials. Explore (NY). 2021;17:162-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 24. | Zheng Y, Zhang J, Huang W, Zhong LLD, Wang N, Wang S, Yang B, Wang X, Pan B, Situ H, Lin Y, Liu X, Shi Y, Wang Z. Sini San Inhibits Chronic Psychological Stress-Induced Breast Cancer Stemness by Suppressing Cortisol-Mediated GRP78 Activation. Front Pharmacol. 2021;12:714163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 25. | Shen Z, Yu M, Dong Z. Research Progress on the Pharmacodynamic Mechanisms of Sini Powder against Depression from the Perspective of the Central Nervous System. Medicina (Kaunas). 2023;59:741. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Zhang J, Wang N, Zheng Y, Yang B, Wang S, Wang X, Pan B, Wang Z. Naringenin in Si-Ni-San formula inhibits chronic psychological stress-induced breast cancer growth and metastasis by modulating estrogen metabolism through FXR/EST pathway. J Adv Res. 2023;47:189-207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (4)] |
| 27. | Guo J, Zhang F, Gao J, Guan X, Liu B, Wang X, Qin Z, Tang K, Liu S. Proteomics-based screening of the target proteins associated with antidepressant-like effect and mechanism of Saikosaponin A. J Cell Mol Med. 2020;24:174-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 28. | Ding W, Wang L, Li L, Li H, Wu J, Zhang J, Wang J. Pathogenesis of depression and the potential for traditional Chinese medicine treatment. Front Pharmacol. 2024;15:1407869. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 29. | Greenlee H, DuPont-Reyes MJ, Balneaves LG, Carlson LE, Cohen MR, Deng G, Johnson JA, Mumber M, Seely D, Zick SM, Boyce LM, Tripathy D. Clinical practice guidelines on the evidence-based use of integrative therapies during and after breast cancer treatment. CA Cancer J Clin. 2017;67:194-232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 534] [Cited by in RCA: 493] [Article Influence: 54.8] [Reference Citation Analysis (4)] |
| 30. | Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57226] [Cited by in RCA: 48213] [Article Influence: 2836.1] [Reference Citation Analysis (11)] |
| 31. | An Y, Liu Z, Wang S, Wang Q, Zhang C, Zhang L, Xu Y, Ge Y, Zheng M, Zhang S, Zheng H, Yu Q. Effect of Chaihu plus Longgu Muli decoction plus five-element music therapy in the treatment of cancer-related depression. Support Care Cancer. 2022;30:7955-7962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 32. | Chang J, Wang T, Zhang W, Wang F. [Research on treatment of fluoxetine combined with Shuganxiaobi prescription in the treatment of breast cancer patients with depression]. Guoji Jingshenbingxue Zazhi. 2017;44:128-131. |
| 33. | He H. Clinical observation of Danzhi xiaoyao powder in the treatment of anxiety and depression in patients with fire derived from stagnation of liver-qi type breast cancer. M.Sc. Thesis, Chengdu University of Traditional Chinese Medicine. 2019. Available from: https://www.doc88.com/p-54759402439574.html. |
| 34. | Hu X, Li F, Shang Y, Wang N, Han T, Li J. [Effect of Jiawei Xiaoyao pill on plasma 5-HT, DA and NE in depression after breast cancer surgery]. Changchun Zhongyiyao Daxue Xuebao. 2018;34:725-727. |
| 35. | Lin J. A clinical study about Jieyu Sanjie Decoction treating the syndrome of liver depression and spleen deficency of Breast cancer-related depression. M.Sc. Thesis, Guangzhou University of Chinese Medicine. 2022. Available from: https://www.zhangqiaokeyan.com/academic-journal-foreign_detail_thesis/0204142907512.html. |
| 36. | Sun S, Yang L, Zhang H, Wu Y. [Effects of Modified Xiaoyao Powder on breast cancer patients with depressive disorder treated with chemotherapy after operation]. Zhonghua Zhongyiyao Zazhi. 2016;31:1499-1502. |
| 37. | Jiang A. [Clinical study of Chaihu plus Longgumuli Decotion in the treatment of depression in patients with breast cancer]. Zhongxiyi Jiehe. 2019;19:204-206. [DOI] [Full Text] |
| 38. | Wang H, Sun H, Chen G. [Randomized Parallel Control Study on the Effect of Xiaoyao Powder on Depression in Postoperative Patients with Breast Cancer]. Zhongyiyao Daobao. 2019;25:67-70. |
| 39. | Xiao F. [Clinical efficacy of modified Xiaoyao powder combined with Paroxetine in the treatment of postoperative depression of breast cancer]. Linchuang Heli Yongyao Zazhi. 2021;14:78-80. [DOI] [Full Text] |
| 40. | Zhang H. Observation on Curative Effect of Chaihu Shugan Powder Combined with Deanxit in the Treatment of Breast Cancer Related Depression. M.Sc. Thesis, Chongqing Medical University. 2022. Available from: https://d.wanfangdata.com.cn/thesis/D02769890. |
| 41. | Zhong M, Zhang H, Jiang D, Zhou J, Zhu L. [Modified Fangjidihuang decoction combined with sertraline in the treatment of breast cancer patients with depression]. Jianyanyixue Yu Linchuang. 2023;20:3560-3563. [DOI] [Full Text] |
| 42. | Huang J, Shen H, Wu Y, Zhou X, Li M. [Effect of Jiawei Ganmai Dazao Decoction combined with amitriptyline in patients with depression and anxiety after breast tumor resection and its effect on sleep and quality of life]. Xiandai Zhongxiyi Jiehe Zazhi. 2019;28:1496-1499. [DOI] [Full Text] |
| 43. | Jin J, Yu Z, Xu B. [Clinical observation on Fuzheng jieyu decoction in treatment of hepatic stagnation and spleen deficiency type depression after breast cancer operation]. Zhongguo Xiandai Yisheng. 2016;54:123-126. |
| 44. | Liu Z, Xiao B. [Chinese medicine diagnosis and treatment for breast cancer clinical observation of 30 cases of depression]. Zhongyiyao Daobao. 2011;17:13-15. [DOI] [Full Text] |
| 45. | Shi G, Shi H, Ye X, Lu N, Li M, Xu Y. [Effects of Modified Chaihuguizhi Decotion on 36 breast cancer patients with depression]. Zhejiang Zhongyi Zazhi. 2017;52:36-37. [DOI] [Full Text] |
| 46. | Sun S, Zhang H, Yang L, Zhang Y. [Clinical Research on Modified Xiaoyaosan in Treating Breast Cancer Postoperative Depression]. Zhongyi Xuebao. 2014;29:1708-1709. |
| 47. | Yan Y, Fu W, Zhou J, Hu L, Fan W. [Modified Xiaoyao Powder in treatment of postoperative breast cancer accompanied with depression]. Changchun Zhongyiyao Daxue Xuebao. 2015;31:781-782. |
| 48. | Zhang L, Zhuang Z, Wang J. [Clinical Research on the Treatment of Postoperative Breast Cancer with Depression by Modified Xiaoyao Powder Combined with Fluoxertine Hydrochloride Capsules]. Zhongyi Xuebao. 2015;30:785-787. |
| 49. | Zhang A. [Clinical Research on Ganmai Dazao Decoction Combined with Xiaoyaosan in Treating Breast Cancer with Depression]. Zhongyi Xuebao. 2011;26:910-911. |
| 50. | Yang Y. [The Application of Chaihu Longgu Muli Decoction in the Treatment of Breast Cancer Complicated with Depression]. Zhongguo Zhongyiyao Xiandai Yuancheng Jiaoyu. 2018;16:113-114. |
| 51. | Zheng Y, Zhan D, Hong S, Wang N, Wang Z. [Si Ni San Suppresses Chronic Psychological Stress-induced Breast Cancer Progression via Glycolysis Inhibition]. Zhongyi Zhongliuxue Zazhi. 2022;4:50-59. |
| 52. | Qi H, Liu R, Zheng W, Zhang L, Ungvari GS, Ng CH, Wang G, Xiang YT. Efficacy and safety of traditional Chinese medicine for Tourette's syndrome: A meta-analysis of randomized controlled trials. Asian J Psychiatr. 2020;47:101853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 53. | Sun YJ, Yuan JM, Yang ZM. Effectiveness and safety of moxibustion for primary insomnia: a systematic review and meta-analysis. BMC Complement Altern Med. 2016;16:217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 54. | Xue D, Zhang Y, Song Z, Jie X, Jia R, Zhu A. Integrated meta-analysis, data mining, and animal experiments to investigate the efficacy and potential pharmacological mechanism of a TCM tonic prescription, Jianpi Tongmai formula, in depression. Phytomedicine. 2022;105:154344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 55. | Tao WW, Jiang H, Tao XM, Jiang P, Sha LY, Sun XC. Effects of Acupuncture, Tuina, Tai Chi, Qigong, and Traditional Chinese Medicine Five-Element Music Therapy on Symptom Management and Quality of Life for Cancer Patients: A Meta-Analysis. J Pain Symptom Manage. 2016;51:728-747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 155] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 56. | Ge L, Wang Q, He Y, Wu D, Zhou Q, Xu N, Yang K, Chen Y, Zhang AL, Hua H, Huang J, Hui KK, Liang F, Wang L, Xu B, Yang Y, Zhang W, Zhao B, Zhu B, Guo X, Xue CC, Zhang H; International Trustworthy traditional Chinese Medicine Recommendations (TCM Recs) Working Group. Acupuncture for cancer pain: an evidence-based clinical practice guideline. Chin Med. 2022;17:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 57. | Yi JC, Syrjala KL. Anxiety and Depression in Cancer Survivors. Med Clin North Am. 2017;101:1099-1113. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 268] [Cited by in RCA: 221] [Article Influence: 24.6] [Reference Citation Analysis (4)] |
| 58. | Taipale H, Solmi M, Lähteenvuo M, Tanskanen A, Correll CU, Tiihonen J. Antipsychotic use and risk of breast cancer in women with schizophrenia: a nationwide nested case-control study in Finland. Lancet Psychiatry. 2021;8:883-891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 103] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 59. | Kelly CM, Juurlink DN, Gomes T, Duong-Hua M, Pritchard KI, Austin PC, Paszat LF. Selective serotonin reuptake inhibitors and breast cancer mortality in women receiving tamoxifen: a population based cohort study. BMJ. 2010;340:c693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 350] [Cited by in RCA: 275] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 60. | Ge A, Yang K, Deng X, Zhao D, Ge J, Liu L. The efficacy and safety of Xihuang Pill/capsule in adjuvant treatment of breast cancer: A systematic review and meta-analysis of 26 randomized controlled trials. J Ethnopharmacol. 2022;295:115357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 61. | Kim W, Lee WB, Lee JW, Min BI, Baek SK, Lee HS, Cho SH. Traditional herbal medicine as adjunctive therapy for breast cancer: A systematic review. Complement Ther Med. 2015;23:626-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 62. | Zhang H, Chen T, Shan L. ShenQi FuZheng injection as an adjunctive treatment to chemotherapy in breast cancer patients: a meta-analysis. Pharm Biol. 2019;57:612-624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 63. | Lu W, Rosenthal DS. Oncology Acupuncture for Chronic Pain in Cancer Survivors: A Reflection on the American Society of Clinical Oncology Chronic Pain Guideline. Hematol Oncol Clin North Am. 2018;32:519-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (3)] |
| 64. | Gong X, Kang J, Chang T, Zhao Y, Liu W, Tang X, Yang X, Liu W, Zong R, Wu C, Liu X, Guo Y, Zhu Q, Fan B, Yue Y, Wang Y, Yang H, Xie J, Li Y, Zhang Y, Peng Q, Liu Y, Zhang C, Xia C, Jiao J, Su Y, Zeng X, Huang Q, Jiang Q; PRE-ERODE group. Efficacy of Qingrehuoxue granule for bone protection in patients with active rheumatoid arthritis in China: The PRE-ERODE randomized controlled trial. J Ethnopharmacol. 2025;353:120427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 65. | Bensoussan A, Kellow JE, Bourchier SJ, Fahey P, Shim L, Malcolm A, Boyce P. Efficacy of a Chinese Herbal Medicine in Providing Adequate Relief of Constipation-predominant Irritable Bowel Syndrome: A Randomized Controlled Trial. Clin Gastroenterol Hepatol. 2015;13:1946-54.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 66. | Liang SB, Cheng HJ, Zhang QY, Han M, Li YF, Cao HJ, Yu ZY, Kong LY, Cai YM, An LB, Zhao BT, Xu SS, Yan L, Zhang NW, Jia BY, Liu WF, Niu F, Wu BT, Song JM, Jia SX, Shi MM, Zhang XN, Chung VCH, Robinson N, Liu JP. Chinese herbal formula Tongxie Yaofang granules for diarrhoea-predominant irritable bowel syndrome: a randomised, double-blind, placebo-controlled, phase II trial. BMJ Open. 2025;15:e088410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 67. | Li X, Xin P, Wang C, Wang Z, Wang Q, Kuang H. Mechanisms of Traditional Chinese Medicine in the Treatment of Mammary Gland Hyperplasia. Am J Chin Med. 2017;45:443-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 68. | Zhou L, Zhang C, Xie Z, Yu Q, Wang J, Gong Y, Zhao J, Bai S, Yang L, Deng D, Zhang R, Shi Y. Neural Circuit Mechanisms of Sinisan formula for the Treatment of adolescent Depression: prefrontal cortex to dorsal raphe nucleus. J Ethnopharmacol. 2024;334:118529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 69. | Xu W, Du X, Li J, Zhang Z, Ma X, Luo D, Xiao M, Sun Q. SiNiSan alleviates liver injury by promoting hepatic stem cell differentiation via Wnt/β-catenin signaling pathway. Phytomedicine. 2022;99:153969. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 70. | Cai YJ, Lan JH, Li S, Feng YN, Li FH, Guo MY, Liu RP. Sinisan, a compound Chinese herbal medicine, alleviates acute colitis by facilitating colonic secretory cell lineage commitment and mucin production. J Integr Med. 2025;23:429-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 71. | Lin CT, Lin HY, Peng WH, Wu LY. Multi-target Mechanisms of Si-Ni-San on Anxious Insomnia: An Example of Network-pharmacology and Molecular Docking Analysis. Curr Med Chem. 2025;32:2640-2663. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 72. | Zhan D, She Y, Zhang X, Zhang J, Wang S, Hong S, Zheng Y, Wang N, Wang Z. Chronic Psychological Stress Activates TRP/TAM/CXCL1 Signaling to Promote Breast Cancer Adipocyte Lipolysis via KEAP1 m(6)A Demethylation. Research (Wash D C). 2025;8:0980. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 73. | Fries GR, Saldana VA, Finnstein J, Rein T. Molecular pathways of major depressive disorder converge on the synapse. Mol Psychiatry. 2023;28:284-297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 408] [Cited by in RCA: 350] [Article Influence: 116.7] [Reference Citation Analysis (0)] |
| 74. | Mulinari S. Monoamine theories of depression: historical impact on biomedical research. J Hist Neurosci. 2012;21:366-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 74] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 75. | Jia X, Chen J, Huang R, Wang D, Wang X. Effect-enhancing and toxicity-reducing effects of Chaihu Jia Longgu Muli decoction in the treatment of multimorbidity with depression: a systematic review and meta-analysis. Pharm Biol. 2023;61:1094-1106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 76. | Zhou M, Fan Y, Xu L, Yu Z, Wang S, Xu H, Zhang J, Zhang L, Liu W, Wu L, Yu J, Yao H, Wang J, Gao R. Microbiome and tryptophan metabolomics analysis in adolescent depression: roles of the gut microbiota in the regulation of tryptophan-derived neurotransmitters and behaviors in human and mice. Microbiome. 2023;11:145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 196] [Cited by in RCA: 173] [Article Influence: 57.7] [Reference Citation Analysis (0)] |
| 77. | Xue C, Li G, Zheng Q, Gu X, Shi Q, Su Y, Chu Q, Yuan X, Bao Z, Lu J, Li L. Tryptophan metabolism in health and disease. Cell Metab. 2023;35:1304-1326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 789] [Cited by in RCA: 646] [Article Influence: 215.3] [Reference Citation Analysis (1)] |
| 78. | Xie Q, Ding J, Chen Y. Role of CD8(+) T lymphocyte cells: Interplay with stromal cells in tumor microenvironment. Acta Pharm Sin B. 2021;11:1365-1378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 97] [Article Influence: 19.4] [Reference Citation Analysis (4)] |
| 79. | Xue H, Chen J, Fan W. Assessing the causal relationship between immune cell traits and depression by Mendelian randomization analysis. J Affect Disord. 2024;356:48-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 80. | Laumet G, Edralin JD, Dantzer R, Heijnen CJ, Kavelaars A. CD3(+) T cells are critical for the resolution of comorbid inflammatory pain and depression-like behavior. Neurobiol Pain. 2020;7:100043. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 81. | Sun Z, Zhang B, Zhou J, Luo Y, Zhu X, Wang Y, He Y, Zheng P, Zhang L, Yang J, Wang G. Integrated Single-Cell RNA-seq and ATAC-seq Reveals Heterogeneous Differentiation of CD4(+) Naive T Cell Subsets is Associated with Response to Antidepressant Treatment in Major Depressive Disorder. Adv Sci (Weinh). 2024;11:e2308393. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 82. | Castaneda CA, Mittendorf E, Casavilca S, Wu Y, Castillo M, Arboleda P, Nunez T, Guerra H, Barrionuevo C, Dolores-Cerna K, Belmar-Lopez C, Abugattas J, Calderon G, De La Cruz M, Cotrina M, Dunstan J, Gomez HL, Vidaurre T. Tumor infiltrating lymphocytes in triple negative breast cancer receiving neoadjuvant chemotherapy. World J Clin Oncol. 2016;7:387-394. [PubMed] [DOI] [Full Text] |
| 83. | Sun L, Su Y, Jiao A, Wang X, Zhang B. T cells in health and disease. Signal Transduct Target Ther. 2023;8:235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 839] [Cited by in RCA: 700] [Article Influence: 233.3] [Reference Citation Analysis (2)] |
| 84. | Li W, Zhou Q, Lv B, Li N, Bian X, Chen L, Kong M, Shen Y, Zheng W, Zhang J, Luo F, Luo Z, Liu J, Wu JL. Ganoderma lucidum Polysaccharide Supplementation Significantly Activates T-Cell-Mediated Antitumor Immunity and Enhances Anti-PD-1 Immunotherapy Efficacy in Colorectal Cancer. J Agric Food Chem. 2024;72:12072-12082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 36] [Article Influence: 18.0] [Reference Citation Analysis (6)] |
| 85. | Ma J, Liu H, Wang X. Effect of ginseng polysaccharides and dendritic cells on the balance of Th1/Th2 T helper cells in patients with non-small cell lung cancer. J Tradit Chin Med. 2014;34:641-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 86. | Doege D, Thong MS, Koch-Gallenkamp L, Bertram H, Eberle A, Holleczek B, Pritzkuleit R, Waldeyer-Sauerland M, Waldmann A, Zeissig SR, Jansen L, Brenner H, Arndt V. Health-related quality of life in long-term disease-free breast cancer survivors versus female population controls in Germany. Breast Cancer Res Treat. 2019;175:499-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 87. | Suzuki K, Morishita S, Nakano J, Okayama T, Inoue J, Tanaka T, Fukushima T. Association between quality of life and mortality risk in patients with breast cancer: a systematic review and meta-analysis. Breast Cancer. 2024;31:552-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 88. | Bao T, Cai L, Snyder C, Betts K, Tarpinian K, Gould J, Jeter S, Medeiros M, Chumsri S, Bardia A, Tan M, Singh H, Tkaczuk KH, Stearns V. Patient-reported outcomes in women with breast cancer enrolled in a dual-center, double-blind, randomized controlled trial assessing the effect of acupuncture in reducing aromatase inhibitor-induced musculoskeletal symptoms. Cancer. 2014;120:381-389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 89. | Chu YR, Kung PT, Liu LC, Lin CY, Ou-Yang F, Yue CH, Su SY, Chen YY, Wang WC, Kao HF, Chou WY, Tsai WC. Comparison of Quality of Life Between Breast Cancer Patients Treated With and Without Adjunctive Traditional Chinese Medicine in Taiwan. Integr Cancer Ther. 2023;22:15347354221150907. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 90. | Jia R, Liu N, Cai G, Zhang Y, Xiao H, Zhou L, Ji Q, Zhao L, Zeng P, Liu H, Huo J, Yue X, Zhang Y, Wu C, Sun X, Feng Y, Liu H, Liu H, Han Z, Lai Y, Zhang Y, Han G, Gong H, Wang Y, Li Q. Effect of PRM1201 Combined With Adjuvant Chemotherapy on Preventing Recurrence and Metastasis of Stage III Colon Cancer: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Front Oncol. 2021;11:618793. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (4)] |