INTRODUCTION
Depression is a mental disorder characterized by persistently low mood and a steadily increasing incidence worldwide, constituting a major public health challenge. As a leading contributor to global disability, it affects approximately 350 million people, with its economic burden accounting for 4.3% of the world’s population and continuing to rise[1,2]. It is estimated that mental disorders will cause global economic losses of up to 16.3 United Stated dollars trillion between 2011 and 2030[3]. Owing to its complexity and heterogeneity-driven by interactions between genetic and environmental factors-the biological mechanisms underlying depression remain unclear. Current pathogenic theories primarily include the monoamine depletion hypothesis, the neuroplasticity hypothesis, and the hypothalamic-pituitary-adrenal (HPA) axis hypothesis[4]. Currently, depression is primarily managed with antidepressants. Although monoamine reuptake inhibitors have been used clinically for more than 50 years, their limitations-such as delayed onset, poor patient compliance, and high relapse rates after discontinuation-pose substantial risks, particularly for patients with suicidal tendencies[5]. In recent years, rapid-acting antidepressant therapy has attracted considerable attention. Ketamine, as a representative agent, can produce sustained antidepressant effects lasting approximately 1 week after a single administration within 2 hours[6]. However, its clinical application is limited by adverse effects, including addiction potential and neurotoxicity[7]. Therefore, the development of fast-acting and safe antidepressants remains an urgent priority.
The exploration of new antidepressants derived from traditional Chinese medicine (TCM) has become a research priority in recent years[8,9], with TCM demonstrating certain advantages in the management of depression[10]. Yueju pill is a classical TCM compound formulated more than 800 years ago and has long been used for syndromes associated with emotional disorders[11]. Studies have shown that the volatile oil of Yueju pill can inhibit inflammation via modulation of the AGE/PI3K/Akt signaling axis[12]. Furthermore, by improving intestinal flora and regulating systemic metabolism, Yueju pill alleviates depressive symptoms in animal models[13]. These findings suggest that Yueju pill exerts antidepressant effects through unique intracellular signaling mechanisms. Consistent with its traditional use in relieving “stagnation syndrome”, Yueju pill has been reported to modulate monoamine neurotransmitters (NTs), such as serotonin and norepinephrine (NE), and to attenuate hyperactivity of the HPA axis in chronic stress models[11,14]. Subsequent studies further identified the synergistic effects of its active components (e.g., geniposidic acid) in regulating neuroinflammation via NF-κB inhibition and oxidative stress via Nrf2 activation[15,16]. Autophagy is a conserved cellular self-degradation process that provides energy for cellular homeostasis and enables adaptation to nutritional and environmental stress. By eliminating misfolded or aggregated proteins, damaged organelles (including mitochondria, endoplasmic reticulum, and peroxisomes), and invading pathogens, autophagy maintains intracellular homeostasis and play a critical regulatory role in various diseases[17-19]. Increasing evidence indicates that dysregulated is involved in the onset and progression of depression[20]. The SIRT1/FOXO3 axis is an important pathway regulating autophagy and has been shown to mediate mitochondrial autophagy, thereby exerting neuroprotective effects in cerebral ischemia-reperfusion injury[21]. SIRT1 is a widely distributed deacetylase in the brain that regulates protein expression by deacetylating various substrates, including NF-κB, and participates in physiological processes such as cell survival and energy metabolism, contributing to central nervous system protection[22,23]. Notably, erythropoietin alleviates chronic stress-induced depression by regulating SIRT1-mediated mitochondrial function[24]. FoxO transcription factors play essential roles in several physiological and biological processes[25], and transcriptional regulation is closely implicated in depression pathogenesis. As intermediaries between intracellular signaling cascades and gene transcription, transcription factors may contribute to depression onset and progression and represent potential therapeutic targets[26]. However, the antidepressant role of the SIRT1/FoxO3a-mediated autophagy pathway has not been clearly elucidated, and it remains unknown whether Yueju pill exerts antidepressant effects through the regulation of this autophagy pathway. Accordingly, a chronic unpredictable mild stress (CUMS)-induced depression model was established in rats, with Yueju pill as an intervention. This study aimed to elucidate the underlying mechanism of Yueju pill and to provide experimental evidence supporting its potential clinical application.
MATERIALS AND METHODS
Experimental animals
A total of 70 specific pathogen free-grade adult male Sprague-Dawley rats (180-220 g) were obtained from Fujian Anburui Biotechnology Co., LTD. Animals were housed under a 12 hours/12 hours light-dark cycle at an ambient temperature of 20-25 °C and relative humidity of 40%-70%, with access to water and a standard maintenance diet. After a 2-week acclimation period, a sucrose preference test (SPT) was conducted. Once baseline sucrose preference stabilized, rats with sucrose preference values lower than baseline were excluded (n = 10). The remaining rats were assigned to a blank group (n = 15) and a model group (n = 45) according to a 1:3 allocation ratio. Rats in the model group underwent CUMS to induce depression, whereas blank-group rats were maintained under normal conditions without stress exposure.
Construction and intervention of the depression model
Before intervention, fluoxetine (10 mg/kg) was dissolved in normal saline (1 mL) for intraperitoneal administration. Yueju pill (composed of Rhizoma Cyperi 12 g, Rhizoma Atractylodis 12 g, Rhizoma Chuanxiong 9 g, Fructus Gardeniae 9 g, and Massa Medicata Fermentata 12 g) was administered at 800 mg/kg, dissolved in 1 mL of normal saline, via intraperitoneal injection. Model rats were further randomized into model, fluoxetine, and Yueju pill groups (n = 15 per group). Rats in the blank and model groups received intraperitoneal injections of normal saline (1 mL), whereas rats in the fluoxetine (10 mg/kg) and Yueju pill (800 mg/kg) groups received the corresponding treatments. All injections were administered once daily between 9:30 AM and 10:30 AM for 28 consecutive days.
For CUMS induction, rats were exposed to various stressors, including alterations in lighting and circadian rhythm (continuous lighting for 24-48 hours, overnight lighting, or lights off for 3 hours), changes in housing conditions (wet bedding, cage tilting at 45° for 24 hours, or cage shaking for 15 minutes), food and water deprivation (24 hours), tail clip stimulation (tail clamping for 1 minute, 1 cm from the base), behavioral restraint (2 hours), cold stress (swimming in 4 °C water for 5 minutes), and heat stress (swimming in 42 °C water for 5 minutes). One stressor was applied randomly each day for 28 days, with no consecutive repetition of the same stimulus. Blank-group rats were housed separately and maintained without exposure to stressors. Body weight was recorded weekly.
Behavioral detection
Following a 2-week acclimation period, SPT, forced swimming test (FST), open field test (OFT), and tail suspension test (TST) results obtained before stress exposure were used as behavioral baseline values.
SPT
The SPT was conducted on days 19-21. Rats were habituated to sucrose solution for 72 hours before testing. Each cage was provided with two bottles containing 1% sucrose solution and pure water, with bottle positions alternated every 12 hours, followed by 22 hours of food and water deprivation. During the formal test, rats were housed individually and given access to 1% sucrose solution and pure water ad libitum for 2 hours. Consumption of sucrose solution and pure water was recorded, and sucrose preference was calculated as sucrose solution intake/(sucrose solution intake + pure water intake).
FST
The FST was conducted on day 23 using a transparent cylindrical container (50 cm × 25 cm). On the first day, the container was filled with water to a depth of 30 cm (23 ± 3 °C), preventing the hind limbs from contacting the bottom, and rats were allowed to swim freely for 6 minutes for habituation. On the following day, rats were again placed in water under identical conditions for 6 minutes. Immobility time during the final 4 minutes was recorded by observers blinded to group allocation. Immobility was defined as cessation of active struggle, floating on the water surface, or minimal limb movements sufficient only to maintain flotation.
OFT
The OFT was conducted on day 24 in an open box (50 cm high), with the floor divided into 25 equal squares (70 cm × 70 cm) and all walls painted black. Testing was conducted in a quiet environment. Rats were placed in the center of the box, and activity was recorded for 5 minutes. Horizontal activity (number of crossings) and vertical activity (rearing frequency) were scored by observers blinded to the experimental design.
TST
The OFT was conducted on day 25. Rats were transferred to the testing room 1 hour in advance for acclimation. The tail was secured with medical adhesive tape 2-3 cm from the tip, rats were suspended from a fixed platform (75 cm height). Behavior was recorded for 6 minutes, and immobility time during the final 4 minutes was analyzed using a double-blind method.
Serum NT levels
Before modeling and after completion of behavioral testing, 1 mL of tail-vein blood was collected to determine serum 5-hydroxytryptamine (5-HT), NE, and dopamine (DA) levels using ELISA kits (Shanghai Yuanye Biotech).
Transmission electron microscope
After behavioral testing and blood collection, rats were anesthetized and brain tissue was collected. Hippocampal tissue was rapidly isolated on ice and stored at -80 °C. Samples (1 mm × 1 mm × 3 mm) were fixed in 3% glutaraldehyde, washed three times with PBST, and post-fixed in osmium tetroxide. Following dehydration, infiltration, embedding, and sectioning, samples were stained with 2% uranyl acetate and lead citrate for 15 minutes each. Autophagosome formation in hippocampal neurons was observed using transmission electron microscope (TEM).
Western blotting
Frozen (-80 °C) left hippocampal tissue (20 mg) was homogenized in lysis buffer and centrifuged for supernatant collection. Protein concentrations were determined using a BCA assay. Proteins were separated using SDS-PAGE, transferred to membranes (90 V, 50 minutes), and blocked with 5% skim milk at 4 °C overnight. Membranes were incubated with primary antibodies against brain-derived neurotrophic factor (BDNF; 1:1000), cAMP response element-binding protein (CREB; 1:1000), SIRT1 (1:1000), FOXO3a (1:1000), Beclin-1 (1:2000), and LC3B (1:2000; all from Abcam), followed by incubation with HRP-conjugated secondary antibodies for 2 hours at room temperature. Signals were visualized via ECL, and band intensities were quantified using Image J to calculate relative protein expression.
Statistical analysis
Data were analyzed using SPSS 25.0, and figures were generated using GraphPad Prism 8.0. Measurement data are expressed as mean ± SD. Comparisons between two groups were performed using the t-test, while one-way analysis of variance followed by the LSD-t test was used for multigroup comparisons. A value of P < 0.05 was considered statistically significant.
RESULTS
SPT and FST
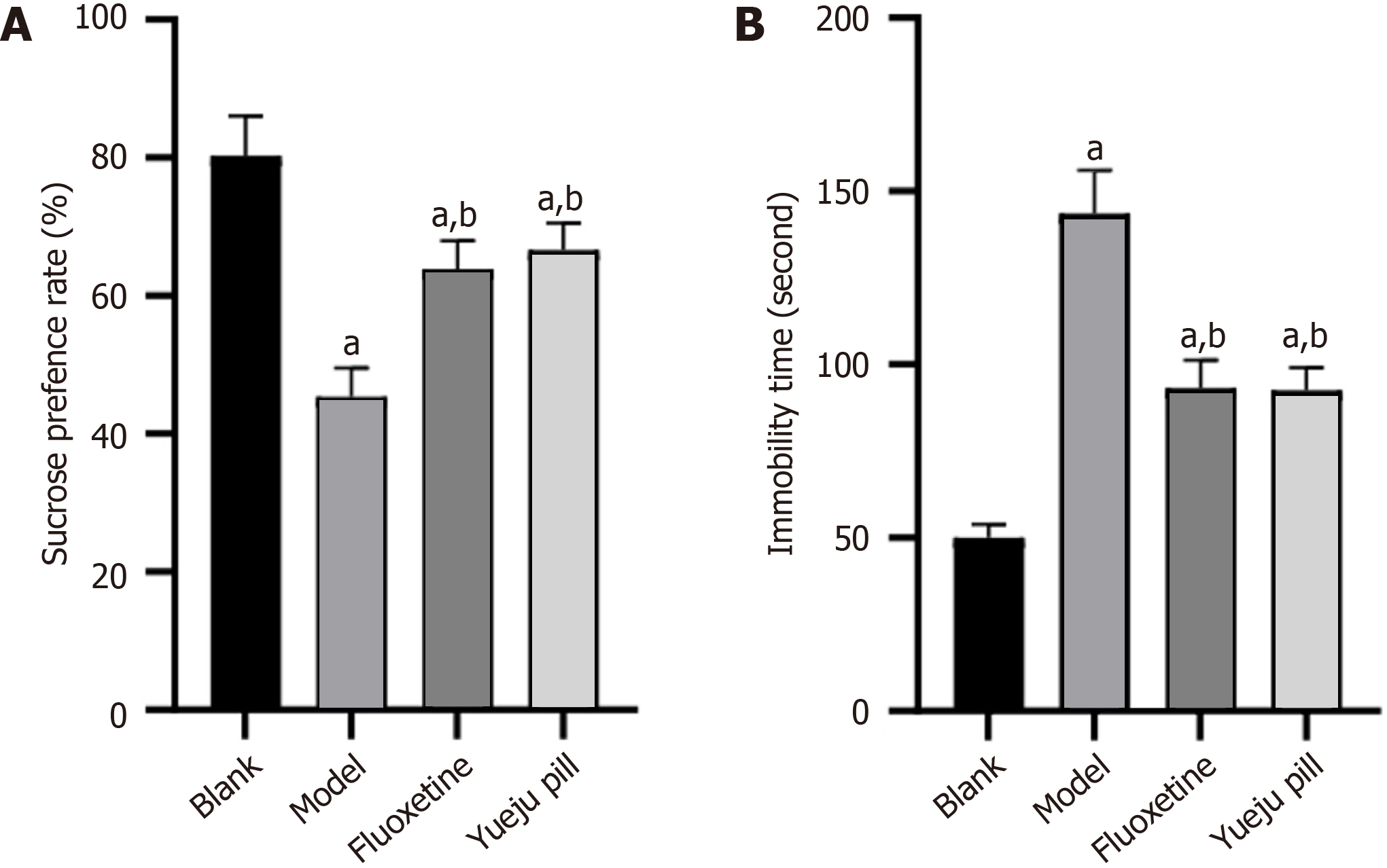
SPT showed reduced sucrose preference rate in the model group compared with the blank group (P < 0.05), while fluoxetine- or Yueju pill-treated rats exhibited higher sucrose preference than model animals (P < 0.05). No significant difference was observed between the fluoxetine and Yueju pill groups (P > 0.05; Figure 1A). In the FST, immobility time was prolonged in the model group compared with the blank group (P < 0.05) but was significantly shortened in the fluoxetine and Yueju pill groups compared with the model group (P < 0.05). Immobility time did not differ significantly between the fluoxetine and Yueju pill groups (P > 0.05; Figure 1B).
Figure 1 Sucrose preference and forced swimming test results.
A: Sucrose preference rates across groups; B: Immobility time in each group. aP < 0.05 vs blank group; bP < 0.05 vs model group.
OFT and TST
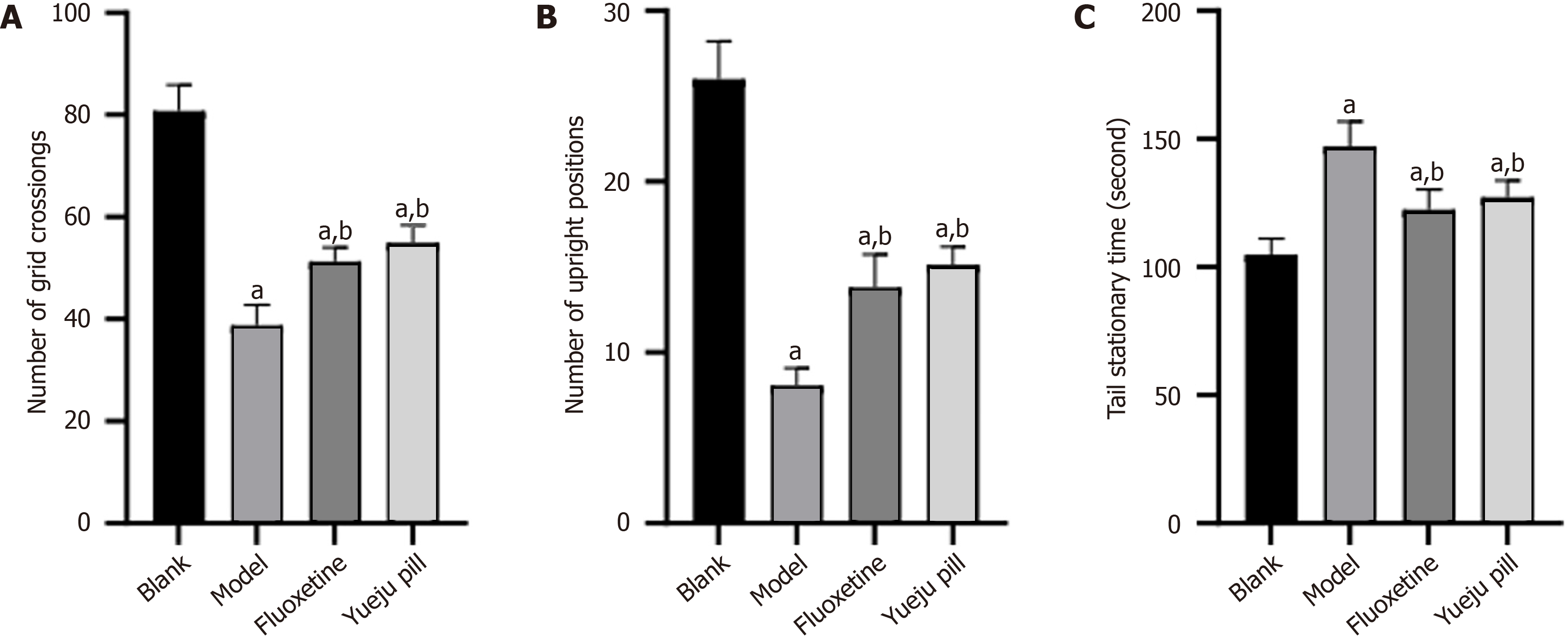
In the OFT (Figure 2A), the number of grid crossings and rearing frequency were lower in the model group than in the blank group (P < 0.05) but were increased in the fluoxetine and Yueju pill groups compared with the model group (P < 0.05). No significant differences were found between the fluoxetine and Yueju pill groups (P > 0.05). In the TST, tail immobility time was longer in the model group than in the blank group (P < 0.05) and was similarly reduced in the fluoxetine and Yueju pill groups compared with the model group (P < 0.05), with no difference between the two treatment groups (P > 0.05; Figure 2B and C).
Figure 2 Rat behavior test results.
A: Comparison of the number grid crossings; B: Rearing frequency comparison; C: Comparison of tail stationary time. aP < 0.05 vs blank group; bP < 0.05 vs model group.
Serum NTs
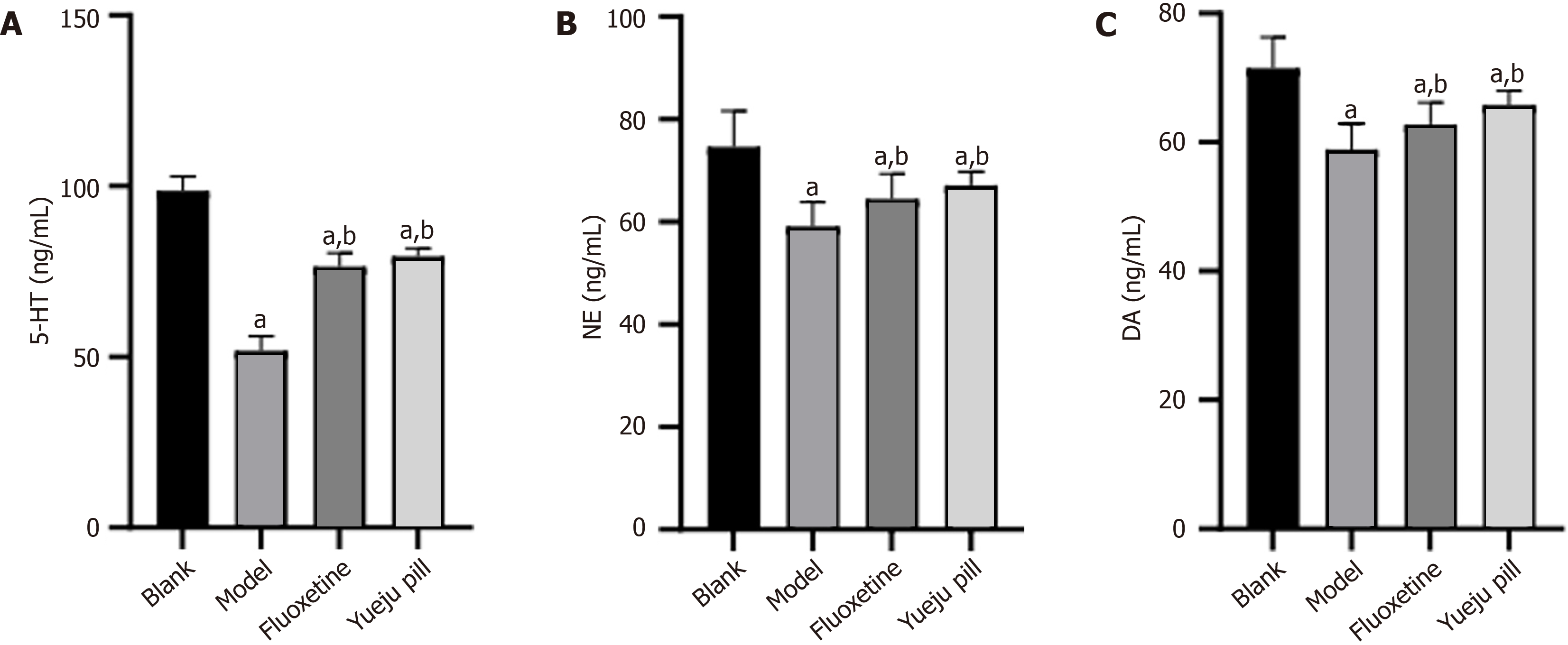
Serum 5-HT, NE, and DA levels were lower in the model group than in the blank group (P < 0.05). Fluoxetine and Yueju pill groups had increased 5-HT, NE, and DA levels compared with the model group (P < 0.05), with no significant differences between the two treatment groups (P > 0.05; Figure 3).
Figure 3 Serum neurotransmitter levels.
A: Serum 5-hydroxytryptamine comparison across groups; B: Serum norepinephrine comparison; C: Serum dopamine comparison. aP < 0.05 vs blank group; bP < 0.05 vs model group. 5-HT: 5-hydroxytryptamine; NE: Norepinephrine; DA: Dopamine.
TEM findings of hippocampal tissue
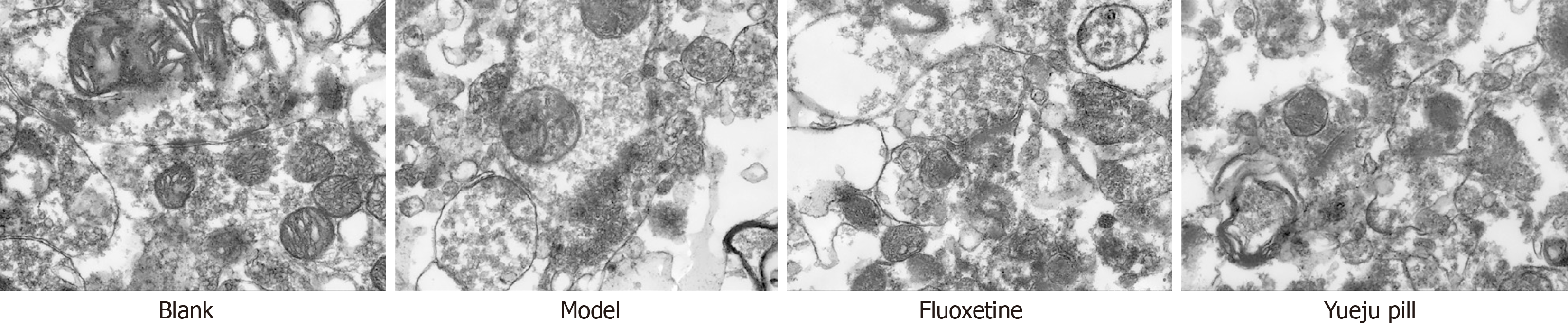
TEM examination revealed relatively intact mitochondrial morphology in blank group rats, with well-defined double membrane and cristae and no obvious autophagosomes. In the model group, mitochondria count was reduced, with obvious vacuolation, cristae breakdown or lysis, and membrane rupture, blurred boundaries, and the presence of autophagosomes. In fluoxetine and Yueju pill groups, neuronal ultrastructure was clear, mitochondria count was increased, and structural damage was reduced, with occasional cristae disorganization, increased free ribosomes, preserved endoplasmic reticulum structure, and no obvious autophagosomes (Figure 4).
Figure 4 Hippocampus observed under transmission electron microscopy.
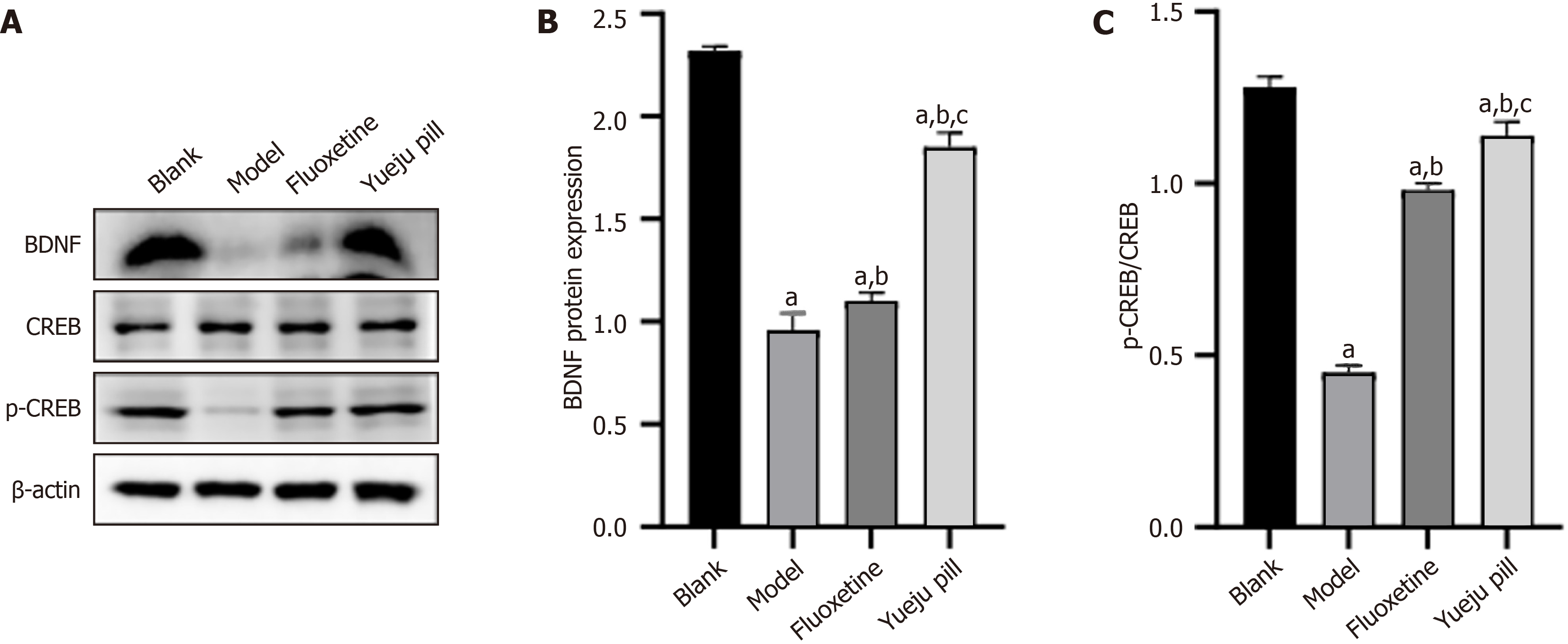
BDNF/CREB axis-related protein expression
Western blotting showed reduced BDNF expression and p-CREB/CREB ratios in the model group compared with the blank group (P < 0.05). Fluoxetine and Yueju pill treatment upregulated BDNF expression and p-CREB/CREB compared with the model group (P < 0.05), with higher levels observed in the Yueju pill group than in the Fluoxetine group (P < 0.05; Figure 5).
Figure 5 Brain-derived neurotrophic factor/cAMP response element-binding protein axis-related protein expression.
A: Brain-derived neurotrophic factor (BDNF)/cAMP response element-binding protein (CREB) axis-associated protein bands across groups; B: BDNF protein expression; C: CREB protein expression. aP < 0.05 vs blank group; bP < 0.05 vs model group; cP < 0.05 vs fluoxetine group. BDNF: Brain-derived neurotrophic factor; CREB: CAMP response element-binding protein.
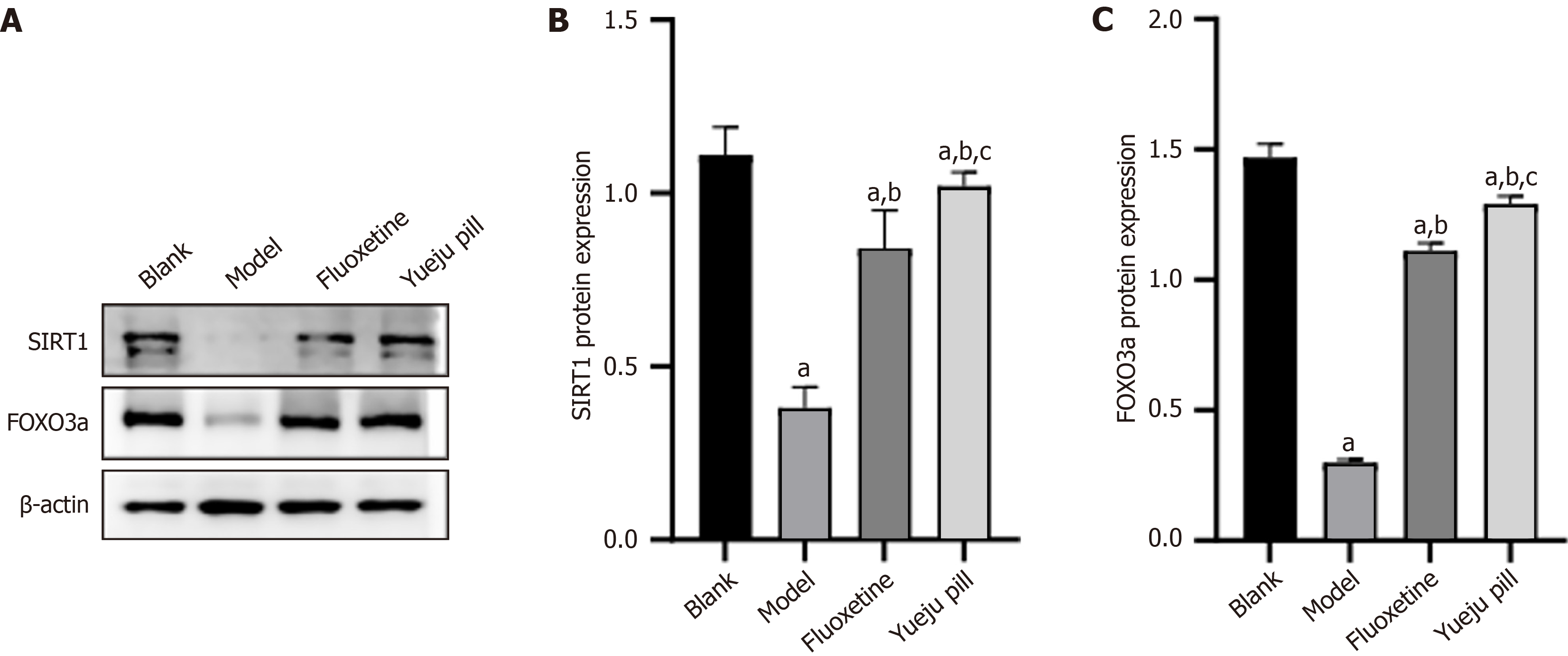
SIRT1/FOXO3a pathway protein expression
SIRT1 and FOXO3a protein levels were lower in the model group than in the blank group (P < 0.05). Fluoxetine and Yueju pill treatment significantly upregulated SIRT1 and FOXO3a expression level compared with the model group (P < 0.05), with higher levels found in the Yueju pill group than in the fluoxetine group (P < 0.05; Figure 6).
Figure 6 SIRT1/FOXO3a pathway protein expression.
A: SIRT1/FOXO3a axis-related protein bands across groups; B: SIRT1 protein expression; C: FOXO3a protein expression. aP < 0.05 vs blank group; bP < 0.05 vs model group; cP < 0.05 vs fluoxetine group.
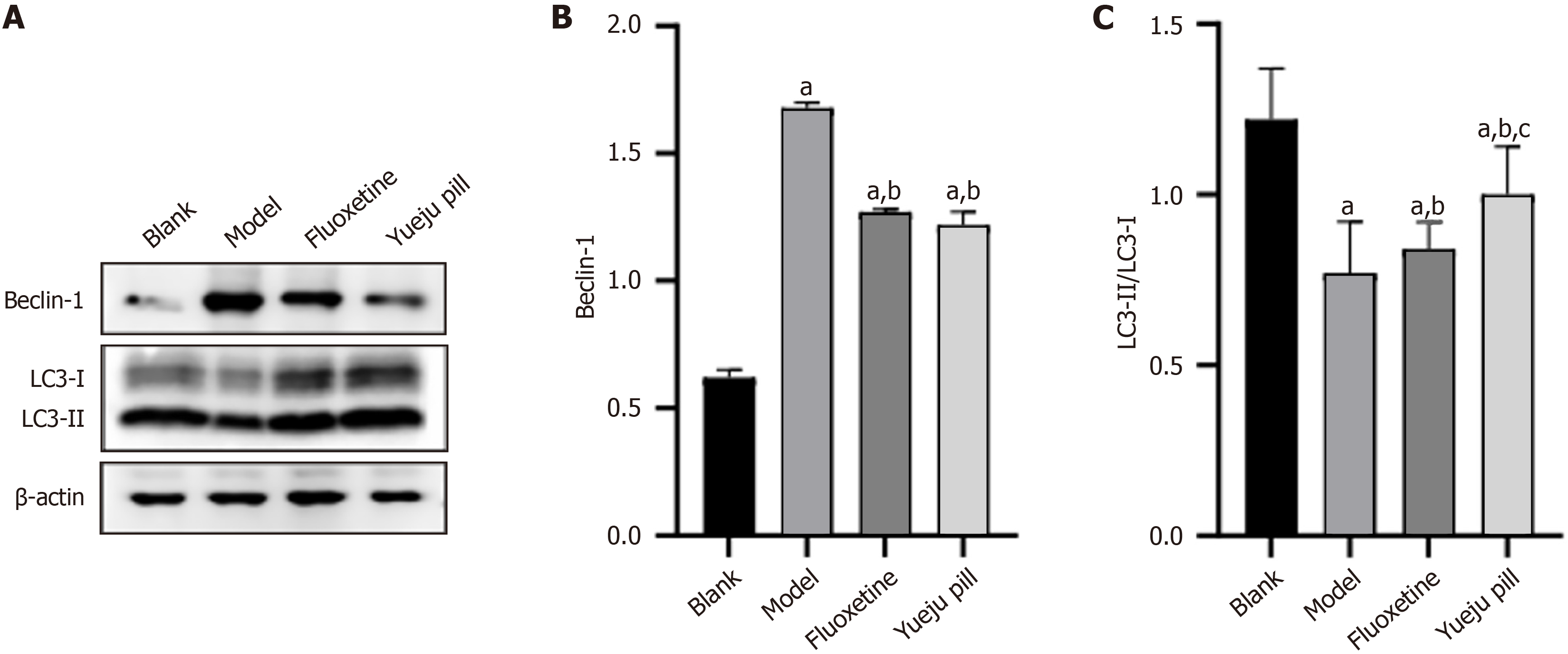
Autophagy-related protein expression
Compared with the blank group, the model group showed increased Beclin-1 protein expression and reduced LC3 protein levels (P < 0.05). Compared with the model group, Beclin-1 protein expression was decreased and LC3 protein levels were increased in the fluoxetine and Yueju pill groups, with statistically significant differences also observed between the two treatment groups (P < 0.05; Figure 7).
Figure 7 Autophagy-related protein expression.
A: Autophagy-related protein bands across group; B: Beclin-1 protein expression; C: LC3 protein expression. aP < 0.05 vs blank group; bP < 0.05 vs model group; cP < 0.05 vs fluoxetine group.
DISCUSSION
Yueju pill, formulated by Dan-Xi Zhu, a renowned theorist and physician of TCM, has long been used to treat disorders rooted in depression (stagnation syndrome). Dan-Xi Zhu proposed six forms of “depression”-qi, blood, fire, dampness, food, and phlegm stagnation-often triggered by stress responses and collectively contributing to emotional and somatic dysfunction[27]. These patterns manifest as low mood, chest pain or tightness, irritability, and a feeling of throat blockage. This study demonstrates that Yueju pill alleviates CUMS-induced depression, possibly through the modulation of SIRT1/FoxO3a axis-mediated autophagy.
Compared with blank rats, model animals showed reduced horizontal and vertical movements in the OFT, decreased sucrose preference in the SPT, and prolonged immobility time in the FST. In contrast, both fluoxetine and Yueju pill treatment improved performance across these behavioral tests, indicating that Yueju pill effectively ameliorates depressive-like behaviors, with efficacy comparable to fluoxetine as a positive control. Among the proposed mechanisms of depression, the monoamine NT hypothesis-mainly encompassing 5-HT, DA, and NE-remains central[28]. Monoamine NTs regulate multiple physiological, behavioral, and endocrine functions in the central nervous system[29], and their dysfunction is closely linked to depressive pathology[30]. Deficiencies in 5-HT, NE, and DA are diagnostically relevant and can aggravate depressive behaviors[31]. Consistently, our findings showed that both fluoxetine and Yueju pill increased serum DA, 5-HT, and NE levels compared with the model group. Given the complex interplay among NT systems, chronic stress-induced reductions in monoanimes and related metabolites across brain regions such as the cortex and hippocampus may underlie the observed behavioral deficits.
TEM analysis further revealed that fluoxetine and Yueju pill alleviated ultrastructural damage to hippocampal neurons, including dendrites, dendritic spines, and synapses. The synaptic microenvironment-compromising presynaptic and postsynaptic structures, synaptic clefts, energy supply, and NTs-is fundamental to neuronal communication[32]. NT imbalance can disrupt synaptic plasticity, and impaired hippocampal synaptic plasticity is a recognized contributor to[33]. These may explain Yueju pill’s antidepressant effects. Besides, Yueju pill significantly enhanced hippocampal CREB and BDNF protein expression compared with the model group. The BDNF/TrkB/CREB axis is critical for neuronal growth, synaptic remodeling, and emotional regulation. BDNF plays a key role in neuronal differentiation, survival, and neurogenesis, while CREB acts as an important upstream regulator of BDNF transcription[34,35].
At the molecular level, both fluoxetine and Yueju pill increased LC3-II/LC3-I ratios as well as SIRT1, and FOXO3a expression but lower the expression of Beclin-1, suggesting activation of autophagy pathways. These findings suggest that Yueju pill may exert antidepressant effects by regulating SIRT1/FoxO3a-mediated autophagy. Increasing evidence indicates that various antidepressants, including conventional agents, TCM formulations, and plant-derived small-molecule compounds, exert antidepressant effects through autophagy regulation[36]. Fluoxetine, for example, protects astrocytes in chronic mild stress-induced mice by promoting autophagosome formation and accelerating clearance of damaged mitochondria via a p53-dependent mechanism[37]. Similarly, multiple natural compounds (e.g., luteolin, apigenin, hypericin, resveratrol, salvianolic acid B, isoliquiritigenin, nobiletin, andrographolide, and rubescensine A) target autophagy and neuroinflammation by regulating AMPK, LC3-II/LC3-I, and Beclin-1 expression, suppressing NLRP3 inflammasome activation and pro-inflammatory cytokine release (e.g., Il-1β/18), and thereby exerting antidepressant effects in rodents[38,39]. In this context, the observed upregulation of SIRT1 and FOXO3a expression supports a neuroprotective role for the SIRT1/FOXO3a axis in central nervous system disorders via autophagy regulation.
This study still has several limitations. First, autophagy-related signaling pathways are complex, and this study focused primarily on the SIRT1/FOXOP3 pathway without examining upstream or downstream regulators. Second, although our data suggest that autophagy regulation contributes to the antidepressant effects of Yueju pill, further studies are required to explore the potential mechanisms of autophagy in depression. Although this study integrates findings from behavioral testing, NTs, ultrastructure, BDNF/CREB, and autophagy pathways, the results remain insufficient and require further comprehensive investigations to supplement them.