Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.115400
Revised: December 7, 2025
Accepted: January 15, 2026
Published online: April 19, 2026
Processing time: 165 Days and 20.8 Hours
Couples undergoing in vitro fertilization (IVF)-embryo transfer (ET) often face psychological and physiological stressors that may impact treatment outcomes. However, non-pharmacological adjunct therapies, which are intended to address these challenges, remain underexplored in infertility care settings.
To examine the efficacy of a culturally adapted psychological program involving affectionate touch for couples undergoing IVF-ET in Shanghai (China).
A controlled trial with 100 couples (51 intervention/49 control) was conducted. The intervention group received a 4-week affectionate touch program, alongside routine IVF-ET. Outcomes assessed included marital relationships, mental health status, sperm motility, and pregnancy rates. Longitudinal data were analyzed using generalized estimating equations.
Intervention group showed significant improvements in total scores (male: β = 5.24, P < 0.001; female: β = 5.82, P < 0.001) and communication subdimension (male: β = 1.85, P = 0.014; female: β = 2.11, P = 0.022). Female also improved in emotional bonding subdimension (β = 2.20, P = 0.011). Male intervention group exhibited reduced anxiety (β = -2.63, P < 0.001) and stress (β = -4.31, P < 0.001), with significant group difference (β = -1.73, β = -2.11; P = 0.030, P =0.025); females intervention group showed stress reduction (β = -4.98, P < 0.001) with significant group difference (β = -2.61, P = 0.029). Intervention male had improved sperm progressive motility (week 3: β = 7.81, P < 0.001; week 4: β = 7.91, P = 0.042), and intervention females showed higher clinical pregnancy rates (76.5% vs 40.8%).
These findings highlight the potential of affectionate touch, such as gentle massage or comforting physical contact, as a low-cost adjunct treatment to address psychological and physiological challenges in infertility care.
Core Tip: This study aimed to examine the efficacy of a culturally adapted psychological program incorporating affectionate touch for couples undergoing in vitro fertilization embryo transfer in Shanghai (China). The results underscore the potential of affectionate touch as a low-cost, accessible adjunctive therapy to routine in vitro fertilization embryo transfer care. This intervention addresses both the psychological challenges such as anxiety and depression and the physiological challenges, including hormonal imbalances and stress-related symptoms, experienced by infertile couples. It works through specific neurobiological mechanisms, including oxytocin release and modulation of the hypothalamic-pituitary-adrenal axis. These mechanisms contribute to stress-mitigating effects, suggesting the intervention’s role in reducing treatment-related stress and fostering relational resilience.
- Citation: Zeng WF, Zhang Q, Wang XR, Xu ZY, Huang CW, Zhang QQ, Yan HL, Wang YX. Efficacy evaluation of an affectionate touch-based psychological intervention program for in vitro fertilization-embryo transfer patients: A controlled trial. World J Psychiatry 2026; 16(4): 115400
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/115400.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.115400
Infertility has been recognized by the World Health Organization (WHO) as a global public health issue, defined as the failure to achieve pregnancy after 12 months or more of regular unprotected sexual intercourse[1]. In recent years, influenced by various factors, the number of people affected by infertility has been rising[2]. Consequently, the increasing infertility rates have accelerated the development and application of treatment options, particularly assisted reproductive technology (ART). According to WHO statistics, approximately one in six couples worldwide experience infertility during their lifetime. A WHO systematic review of 133 relevant studies revealed that the global lifetime prevalence of infertility reaches 17.6%, with a current prevalence of 12.6% - including primary infertility at 9%-9.6% and secondary infertility at 4.9%-6.5%[3]. The Lancet 2021 report on China’s reproductive, maternal, neonatal, child, and adolescent health over the past seven decades indicates that China’s current infertility prevalence is 15.5%, which is significantly higher than the 9% in 1990 and 11.9% in 2007[4]. Addressing the increasing prevalence of infertility is not only crucial for safeguarding individual and family rights but also is a vital issue concerning social development and national demographics.
The rising prevalence of infertility has driven the application and promotion of various treatments, with ART being the most significant. According to the report on the “Development of Maternal and Child Health” in China, there were 497 approved ART medical institutions in China, with over 300000 infants born annually through ART treatments by the end of 2018[5]. China’s first in vitro fertilization (IVF) baby was born in Beijing in 1988, marking a milestone in the development of ART in mainland China[6]. In 2021, the National Healthcare Security Administration encouraged the inclusion of ART services in medical insurance coverage. By 2022, Beijing had incorporated 16 ART procedures, including artificial insemination and embryo transfer (ET), into its medical insurance system[7]. With national promotion and support for ART, meeting patients’ multi-dimensional and personalized physical and psychological needs has become a critical research focus in this field[8].
Although not life-threatening, infertility profoundly impacts patients’ psychological well-being, interpersonal relationships, and quality of life. For most couples, infertility itself constitutes a stressor and psychological trauma. Patients frequently experience emotional distress, including anxiety, depression, self-blame, and loneliness, with anxiety and depression being the most common psychological stress responses. While ART offers hope, its prolonged treatment cycles, outcome uncertainty, and high costs impose significant psychological burdens on patients, leading to emotional distress and interpersonal conflicts. Anxiety and depression - closely associated with poor marital quality, reduced life satisfaction, low treatment compliance, and adverse IVF outcomes - are particularly prevalent[9,10]. Despite providing reproductive hope, ART’s invasive procedures, high costs, and unpredictable outcomes create additional stressors[11]. Moreover, infertility diagnosis and ART treatment may impact marital relationships depending on couples’ coping strategies. Negative coping mechanisms such as avoidance may disrupt marital harmony and lead to divorce, whereas positive communication and joint problem-solving can strengthen relationships[12]. Over 80% of infertile couples experience relationship issues, with 8.6% reporting family conflicts and 7%-8% facing marital crises[13]. Such negative emotional experiences and relational strain not only impair mental health but are also negatively correlated with treatment outcomes.
Multiple studies demonstrate that effective psychological interventions not only improve patients’ mental state but also enhance treatment compliance and ART success rates[14]. Appropriate interventions can alleviate emotional stress, improve interpersonal relationships, and potentially increase pregnancy rates[15]. However, current research on psychological interventions for infertility predominantly originates from Western countries[16], focusing on limited, time-intensive approaches such as support groups, cognitive behavioral therapy, and mindfulness-based interventions[17,18]. These methods require sustained patient participation. This requirement proves challenging because of frequent treatment discontinuation and high intervention dropout rates. Additionally, significant stigma and resistance toward psychological treatment persist within China’s socio-cultural context[19]. Therefore, developing novel, convenient, and evidence-based psychological interventions for infertility patients holds crucial significance for enhancing both mental health and treatment outcomes.
Affectionate touch, defined as warm physical contact conveying affection and support, serves as a vital behavior for establishing and maintaining social relationships. It frequently occurs in intimate relationships across the lifespan - from childhood to adulthood and into old age. Early research on affectionate touch primarily focused on mother-infant bonding. In this context, maternal touch functions as a critical mechanism for developing attachment[20]. Multiple studies have provided empirical evidence for its role in child-rearing and developmental outcomes[21]. Moreover, over the past decade, the psychological benefits of receiving affectionate touch in adult intimate relationships have emerged as a key research focus. For example, in Ditzen et al’s experiment[22], participants in the intervention group received standardized shoulder and neck massages from partners before completing stress tasks, while the control group received no massage or physical contact support. The intervention group exhibited lower cortisol levels and reduced heart rate response during tasks compared to the control group. Floyd et al[23] intervention study randomly assigned adults to either increase the frequency and duration of kissing with partners or to maintain current levels of affection over six weeks. Participants completing the kissing intervention showed significantly improved relationship satisfaction compared to the control group. The pleasurable effects of affectionate touch are mediated by neurophysiological mechanisms involving low-threshold C-tactile mechanoreceptive afferent fibers in superficial skin layers. These biological foundations, together with cultural influences and developmental experiences, shape unique patterns of tactile-emotional expression. Positive tactile interactions can sustainably enhance intimacy between individuals, fostering stronger relational bonds[24].
In adult intimate relationships, affectionate touch yields long-term benefits across three domains: Relational health, mental health, and physical health. Jakubiak and Feeney[24] theoretical framework proposes that affectionate touch in significant relationships mitigates adult stress, strengthens intimacy, and improves physical well-being. Relational health includes relationship satisfaction, commitment, low conflict, and durability. Mental health involves positive affect, subjective well-being, and reduced psychological distress. Physical health encompasses disease prevention and reduced morbidity. The mental health benefits of affectionate touch align with the stress-buffering hypothesis, where social support mitigates stress-related health impacts[25]. The study reveals that days involving physical touch from a partner are associated with increased positive emotions - such as relaxation and alertness - and decreased negative emotions, including irritability and fatigue[26]. Affectionate touch confers physical health advantages, such as lower resting blood pressure and heart rate. Post-touch physical symptom alleviation (e.g., pain reduction, improved sleep) has been documented[27]. In contrast, social isolation is associated with elevated mortality and increased health risks. Affectionate touch promotes social inclusion and encourages health-conscious behaviors (e.g., exercise, dietary management) while discouraging harmful habits (e.g., smoking). When partners share awareness of each other’s health needs and concerns, this mutual health consciousness further reinforces self-care and strengthens relational commitment, creating a positive feedback loop that benefits physical well-being. Despite these established benefits, research on affectionate touch interventions for infertility patients remains limited, highlighting the urgent need for empirical studies in this area.
In summary, affectionate touch holistically enhances relational, mental, and physical health, fostering overall well-being and healthy lifestyles. For infertility patients, it represents a novel and practical psychological intervention with significant therapeutic potential. The current study aimed to examine the efficacy of a culturally adapted, affectionate touch-based psychological program for couples undergoing IVF-ET in Shanghai (China).
Through a controlled trial, longitudinal tracking assessments were conducted during the affectionate touch practice intervention on both intervention and control groups. Table 1 presents the intervention protocol, which is designed to improve emotional well-being and enhance marital relationships in IVF-ET patients. This is achieved through interactive exercises based on affective touch. The protocol comprises three first-level indicators, eight second-level indicators, and eighteen third-level indicators, each representing different components of the intervention framework. The intervention protocol was culturally adapted to the local context and gender norms. For instance, within the “tactile actions” module, culturally familiar gestures such as “arm around the waist” and “hugging the shoulder” were explicitly included. This approach moved beyond the direct application of Western paradigms. In the “positive acceptance” module, emphasis on action-oriented support, such as “creating convenience for the partner”, and “sharing responsibilities”, aligned with relationship maintenance patterns in Eastern cultures that value practical actions alongside verbal affection. These adaptations were designed to enhance the cultural relevance and acceptability of the intervention.
| Level-1 indicator | Level-2 indicator | Level-3 indicator | Content |
| Tactile actions | Affectionate touch | Hugging, arm around waist or shoulder: Conveying security and intimacy | Explain the categories of affective touch actions to participants, emphasizing their positive effects and significance. Instruct participants to perform daily conscious tactile actions. Perform affectionate touch actions (e.g., hug, kiss) at least once daily upon waking up, before going out, after work, and before sleep. Perform at least one playful touch and/or deep touch action weekly. Log the completion of these actions daily before sleep |
| Playful touch | Playful or teasing touches: Light-hearted physical interactions such as patting the head, gentle pinching, or tickling | ||
| Deep touch | Massage: Relieving stress and fatigue through massage to enhance physical and emotional connection | ||
| Sitting/Lying next to the partner with bodies overlapping or touching | |||
| Body exploration: Exploring each other’s bodies with mutual consent to build trust and intimacy | |||
| Shared activities | Leisure activities | Engaging in casual conversation or deep, intimate dialogue with the partner | Explain the concept and significance of shared activities to participants. Instruct participants to complete at least one shared activity per week, each lasting no less than 30 minutes. Log the completion of the activity |
| Engaging in shared leisure activities or spending time together (e.g., taking a walk, watching a movie) | |||
| Shared responsibilities | Making plans or working (handling affairs) with the partner | ||
| Doing housework together with the partner | |||
| Positive acceptance | Verbal expression | Expressing understanding of the partner’s feelings | Explain the concept and significance of positive acceptance to participants. Instruct participants to reflect on and summarize positive interactions with their partner daily and complete a daily behavior log |
| Expressing confidence in the partner | |||
| Expressing acceptance or care for the partner | |||
| Complimenting the partner | |||
| Telling the partner positive or good things about him/her | |||
| Action support | Creating convenience for the partner | ||
| Sharing or lightening the partner’s responsibilities | |||
| Emotional support | Showing interest in learning about the partner’s day | ||
| Making the partner feel valued in multiple ways, including but not limited to eye contact, feeling seen/understood, and unconditional understanding |
Differences between the groups in stress levels, anxiety, and depression, marital relationships, male semen quality, and female pregnancy outcomes were compared to investigate the effects of a daily affectionate touch practice lasting 4 weeks on IVF-ET patients. The primary objective of this study was to evaluate the efficacy of an affectionate touch-based couples’ interaction intervention in improving marital relationships among IVF-ET patients. Secondary objectives included: (1) Evaluating the intervention’s effects on reducing stress and negative emotions, such as anxiety and depression, in IVF-ET patients; and (2) Exploring its potential impact on male semen quality and female clinical pregnancy rates. This study was approved by the Ethics Committee of Shanghai Changhai Hospital (Approval No. 2023-S-006).
Due to practical limitations in enrolling patients consecutively according to their treatment cycles, a non-randomized controlled design was employed. Although no statistically significant differences were found in baseline demographic variables and key clinical variables between groups, this non-randomized allocation may introduce unknown selection biases, which could potentially affect the comparability of the groups and the internal validity of the findings. The study was conducted at a tertiary hospital’s reproductive medicine center in Shanghai from June 2023 to November 2023. After obtaining voluntary consent from patients, we sequentially assigned couples to either the intervention group, which received affectionate touch therapy, or the control group, which received no additional intervention beyond standard care.
Clinical characteristics: Patients must be first-time IVF-ET patients with female patients aged ≤ 40 years and male patients aged ≤ 45 years. Infertility diagnosis involves either female or male factors: Female: Unilateral or bilateral fallopian tube obstruction (confirmed by laparoscopy or hysterosalpingography) or unexplained infertility after exclusion of other causes; male: Oligospermia or asthenospermia.
Eligibility and compliance: Patients must be proficient in Chinese reading and writing to independently complete assessment scales; must be cohabiting couples with stable relationships; and have no history of severe psychiatric disorders.
Psychosocial status: Patients must have experienced no major life events (e.g., bereavement, unemployment) within the past 6 months; must have signed informed consent and committed to complete the 28-day intervention and 3-month follow-up.
Reproductive abnormalities: Female: Primary ovarian insufficiency, endometriosis, or history of recurrent miscarriage; male: Azoospermia, severe teratozoospermia, chromosomal abnormalities (e.g., Klinefelter syndrome), or genetic disorders (e.g., Y-chromosome microdeletion).
Comorbidities and confounding factors: Uncontrolled chronic conditions such as hypertension, diabetes, or thyroid dysfunction; immune disorders (e.g., systemic lupus erythematosus, antiphospholipid antibody syndrome); recent psychological interventions or participation in other clinical trials within 6 months; intended use of other assisted reproductive technologies (e.g., intracytoplasmic sperm injection, preimplantation genetic testing) during the study.
Withdrawal criteria include: (1) Discontinuation of treatment cycle for medical reasons; (2) Occurrence of major life events during the intervention; and (3) Patients who fail to complete intervention tasks for 3 consecutive days and cannot be contacted.
Participants were recruited from the reproductive medicine center of the hospital based on inclusion criteria. After explaining the study’s purpose, procedures, and participant roles, eligible couples provided written informed consent to participate. Due to recruitment challenges and variability in the timing of IVF-ET treatment cycle initiation, a non-randomized controlled trial design was adopted. Participants voluntarily chose to enroll in either the daily practice (intervention) group or the non-intervention control group, with group assignment based on their preference.
Pre-intervention assessments were conducted upon enrollment and included collection of demographic data, the Depression, Anxiety, and Stress Scale-21, the Marital Relationship Questionnaire, measures of affectionate touch frequency, and male semen quality for both groups. Post-intervention assessments were completed after the 4-week (28-day) affectionate touch practice. Pregnancy outcomes were followed up within three months after the intervention.
The intervention group followed the affectionate touch interaction protocol developed based on literature review and expert consultation. The protocol contains three modules: Affectionate touch behavior, shared positive activities, and relational acceptance (Supplementary material). On day 1, researchers conducted face-to-face sessions to explain the intervention’s three modules, to demonstrate interaction techniques, and to address questions. Participants received a practice manual and were instructed to perform daily affectionate touch interactions and to complete a daily practice log before bedtime. To ensure adherence, researchers remotely monitored participants’ progress via a WeChat group for 4 weeks. The control group received routine IVF-ET care, which included standard medical treatment and support, and was added to a WeChat group for communication purposes only, with no additional interventions.
This design allowed for a comparison of outcomes between structured affectionate touch practice and standard care while accommodating the practical constraints posed by IVF-ET treatment schedules.
Sociodemographic characteristics: Sociodemographic data were collected using a researcher-designed basic information questionnaire, covering age, height, weight, exercise habits, smoking and alcohol consumption, educational level, years of marriage, and duration of infertility treatment.
Emotional state and stress level assessment: The Depression-Anxiety-Stress Scale-21 consists of three subscales (depression, anxiety, stress) with a total of 21 items. Items are rated on a 4-point scale ranging from 0 (never) to 3 (always). Subscale scores are calculated as the sum of item scores multiplied by 2, with a range of 0-42. Higher scores indicate greater severity of symptoms. Reliability and validity: Cronbach’s alpha - depression (0.861), anxiety (0.788), stress (0.837); Kaiser-Meyer-Olkin (KMO) values - depression (0.846), anxiety (0.852), stress (0.871); Bartlett’s sphericity tests were significant (P < 0.001).
Marital relationship assessment: The Marital Relationship Questionnaire[28] includes 14 items across three dimensions: Marital affection (5 items, e.g., “I trust my spouse will treat me well”); cognitive understanding (5 items, e.g., “I usually know my spouse’s thoughts”); and marital communication (4 reverse-scored items, e.g., “I don’t need to share my feelings directly”). Items are rated on a 5-point Likert scale. Higher total scores indicate better marital relationship quality. Reliability and validity: Cronbach’s alpha - marital affection (0.806), cognitive understanding (0.860), communication (0.722), total score (0.820); KMO values - marital affection (0.791), cognitive understanding (0.819), communication (0.699), total score (0.854); Bartlett’s tests were significant (P < 0.001).
Affectionate touch assessment: The Affectionate Touch Scale (adapted from Light et al[29]) assesses the frequency of partner touch behaviors during the past month. Frequency options range from 0 (never/almost never) to 4 (five or more times daily). Reliability and validity: Cronbach’s alpha: 0.901; KMO value: 0.858; Bartlett’s test of sphericity was sig
IVF-ET reproductive indicators: For females, clinical pregnancy status post-IVF-ET was recorded. For males, semen analysis parameters assessed included semen volume, sperm motility rate, progressive motility rate, sperm concentration, and total sperm count.
This comprehensive assessment framework ensures robust evaluation of sociodemographic, psychological, relational, and clinical factors in the study population.
Stringent quality control measures were implemented throughout all stages of this study. The research team developed the intervention protocol based on a literature review, which defined the daily exercise components - including duration, specific actions, and requirements. To ensure participants better understood and engaged with the intervention exercises, a face-to-face guidance session was subsequently introduced. Prior to the intervention and data collection, all research personnel received unified training. This training covered communication strategies with participants, questionnaire content, specific precautions to ensure participant safety and data integrity, as well as potential issues and corresponding solutions. During the actual intervention phase, establishing a good rapport and trust with patients was prioritized to ensure the smooth progression of the study. Researchers sent daily reminders via WeChat or phone calls at 20:00 to prompt participants to complete the exercises and log their exercise completion. Participants who either failed to com
The research personnel were blinded to group allocation. Semen samples were labeled only with identification numbers, and the group allocation data were maintained by an independent research team. Follow-up assessments for pregnancy outcomes were all conducted by nurses from the reproductive medical center who were not involved in the intervention (implementing blinding procedures). Key variables (semen parameters and pregnancy outcomes) were entered independently by two individuals. The data were locked only after achieving a data entry consistency rate of no less than 98%.
Statistical analyses were performed using SPSS version 26.0 and R (version 4.3.2).
Data comparisons: Categorical variables: Expressed as frequencies and analyzed using χ2 tests with cross-tabulation. Normally distributed continuous variables: Reported as mean ± SD and compared between groups via independent samples t-tests. Non-normally distributed continuous variables: Reported as median (interquartile range) and compared using the nonparametric Mann-Whitney U test.
Longitudinal data analysis: Generalized estimating equations (GEE) models (via the geepack package in R) were applied to assess group differences in repeated measures. Correlation structure: Exchangeable. Model covariates included time, group, time × group interaction, age, sex, education level, years of marriage, treatment duration, body mass index (BMI), smoking, alcohol use, daily exercise duration, and baseline levels of all indicators. Estimated marginal means across time points were visualized using bar plots.
Pregnancy outcome analysis: Pregnancy success rates were reported as n (%). Binary logistic regression was used to compare success rates between groups, with odds ratios and 95% confidence intervals (CIs) calculated.
Statistical threshold: All tests were two-tailed, with a statistical significance level α = 0.05. Analyses and visualizations were conducted in R (version 4.3.2). This approach ensures a rigorous evaluation of group differences, longitudinal trends, and clinical outcomes while controlling for covariates and baseline variability.
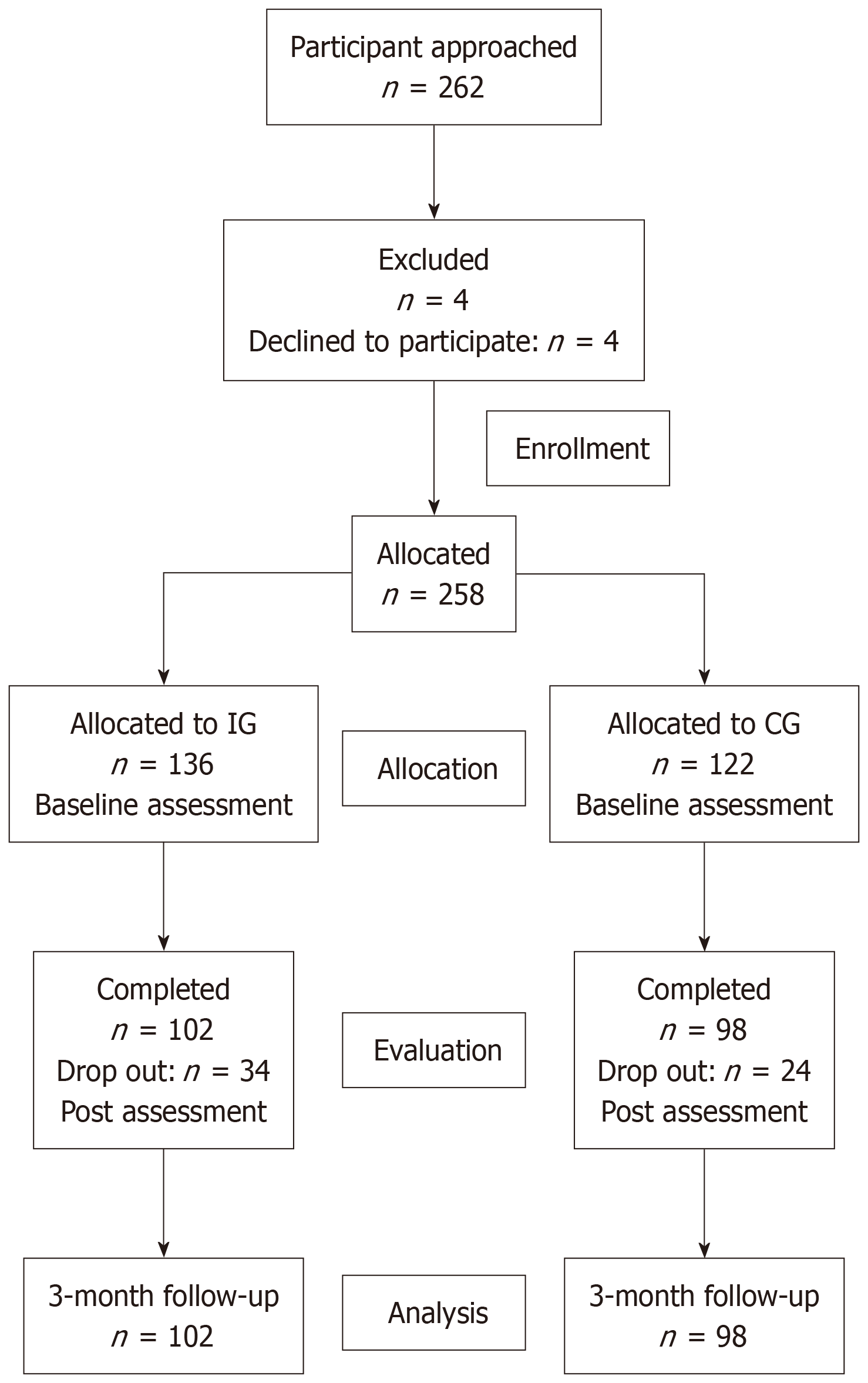
A total of 262 participants met the inclusion criteria for this study, but four individuals were unable to participate due to scheduling conflicts. Based on participants’ preferences, the remaining 258 were allocated to either the intervention group or the control group. Attrition rates were 25% in the intervention group, with 34 participants withdrawing due to failure to complete intervention tasks for three consecutive days or being lost to follow-up, and 19.7% in the control group, with 24 participants who were unable to complete pre-assessments and post-assessments due to personal or logistical reasons. Consequently, 200 participants completed the entire study: 102 in the intervention group (including 51 males and 51 females) and 98 in the control group (including 49 males and 49 females). The trial flowchart is shown in Figure 1.
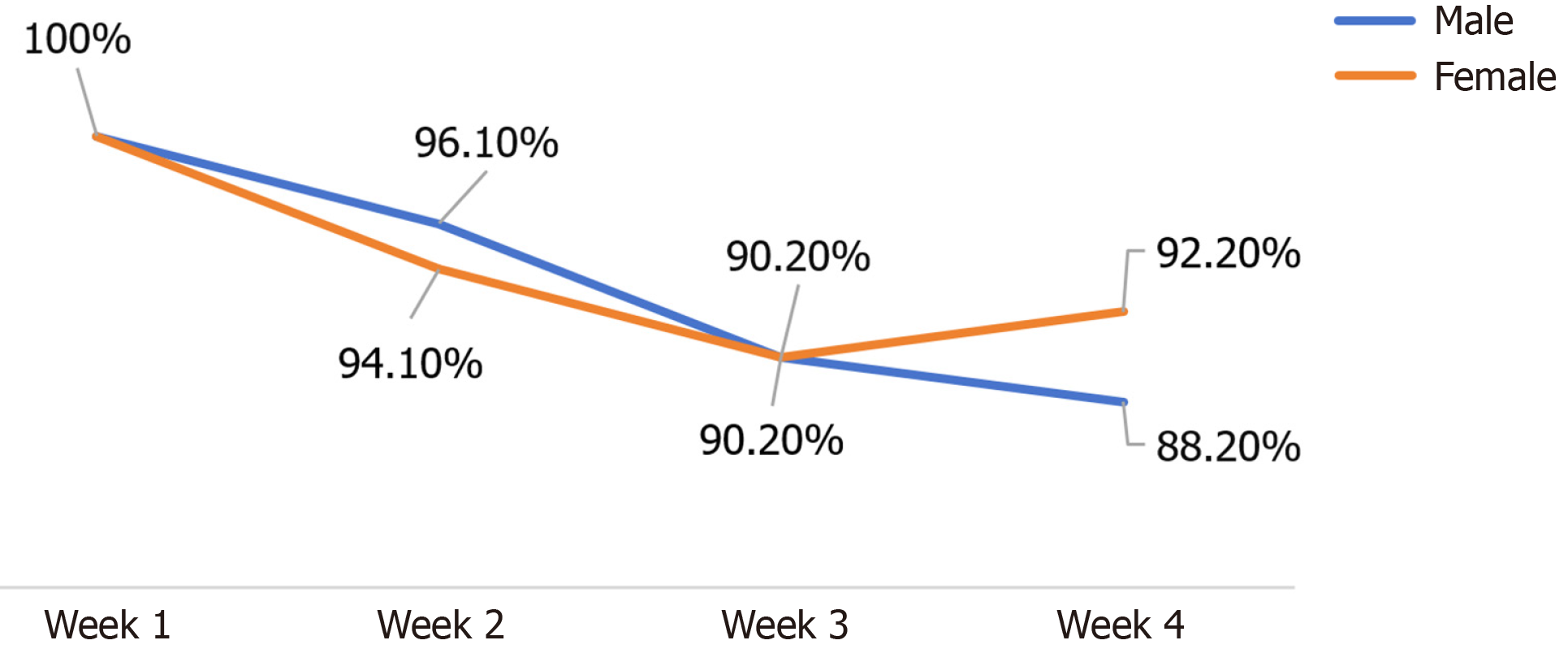
Regarding intervention adherence, all 102 participants in the intervention group completed and logged their daily practice tasks during week 1 (100%). In the following weeks, adherence rates were as follows: Week 2, 49 males (96.1%), and 48 females (94.1%); week 3, 46 males (90.2%), and 46 females (90.2%); week 4, 45 males (88.2%), and 47 females (92.2%). Detailed adherence trends are illustrated in Figure 2.
The randomization procedure established baseline equivalency between treatment conditions. Baseline demographic characteristics are shown in Table 2; no significant group differences between the intervention and control groups were identified in age, BMI, education level, smoking status, alcohol use, marriage duration, or treatment duration. Furthermore, as presented in Table 3, no significant baseline differences were observed between the intervention and control groups in marital relationship quality, stress, anxiety, depression, or levels of affectionate touch for either males or females (P > 0.05).
| Male (n = 100) | χ2/Z/t | P value | Female (n = 100) | χ2/Z/t | P value | |||
| Intervention group (n = 51) | Control group (n = 49) | Intervention group (n = 51) | Control group (n = 49) | |||||
| Age | 35 (31-37) | 33 (30.5-39.5) | Z = -0.304 | 0.761 | 33(30-35) | 32 (29-35) | Z = -0.948 | 0.343 |
| BMI | 24.25 ± 3.08 | 24.61 ± 3.66 | t = -0.53 | 0.597 | 22.22 (21.1-24.1) | 23.67(20.5-25.6) | Z = -1.048 | 0.295 |
| Education | χ2f = 0.260 | 1 | χ2f = 5.075 | 0.161 | ||||
| Middle school | 5 (9.8) | 5 (10.2) | 6 (11.8) | 9 (18.4) | ||||
| High school | 7 (13.7) | 7 (14.3) | 4 (7.8) | 10 (20.4) | ||||
| Undergraduate | 35 (68.6) | 34 (69.4) | 36 (70.6) | 25 (51) | ||||
| Graduate | 4 (7.8) | 3 (6.1) | 5 (9.8) | 5 (10.2) | ||||
| Smoking | χ2 = 3.780 | 0.286 | χ2f = 0.970 | 1 | ||||
| No | 33 (64.7) | 25 (51) | 50 (98) | 49 (100) | ||||
| Seldom | 7 (13.7) | 6 (12.2) | 1 (2) | 0 (0) | ||||
| Occasional | 4 (7.8) | 10 (20.4) | ||||||
| Often | 7 (13.7) | 8 (16.3) | ||||||
| Drinking | χ2f = 4.487 | 0.201 | χ2a = 0.223 | 0.637 | ||||
| No | 22 (43.1) | 29 (59.2) | 49 (96.1) | 45 (91.8) | ||||
| Seldom | 23 (45.1) | 13 (26.5) | 2 (3.9) | 4 (8.2) | ||||
| Occasional | 4 (7.8) | 6 (12.2) | ||||||
| Often | 2 (3.9) | 1 (2) | ||||||
| Marriage length | 5 (3-9) | 4 (3-8) | Z = -1.117 | 0.264 | 5(3-8) | 5 (3-6.5) | Z = -0.902 | 0.367 |
| Treatment length | 2 (2-3) | 2 (1-3) | Z = -1.786 | 0.074 | 2 (1-3) | 2 (1-3) | Z = -1.027 | 0.304 |
| Daily exercise time | 0.8 (0.5-1) | 1 (0.5-1) | Z = -1.143 | 0.253 | 0.5 (0.5-1) | 0.5 (0.5-1) | Z = -0.318 | 0.751 |
| Male (n = 100) | Z/t | P value | Female (n = 100) | Z/t | P value | |||
| IG (n = 51) | CG (n = 49) | IG (n = 51) | CG (n = 49) | |||||
| Total score | 55 (51-61) | 55 (49.5-60) | Z = -0.784 | 0.433 | 58 (53-62) | 55 (48.5-62.5) | Z = -1.478 | 0.139 |
| Affection | 21 (20-24) | 20 (19-23) | Z = -1.713 | 0.087 | 21 (20-22) | 20 (18.5-25) | Z = -0.423 | 0.672 |
| Cognitive | 20 (19-23) | 21 (20-24.5) | Z = -1.234 | 0.217 | 21 (20-23) | 20 (19.5-24.5) | Z = -0.472 | 0.637 |
| Communication | 14 (12-16) | 14 (8-16) | Z = -0.942 | 0.346 | 15 (13-16) | 15 (12-16.5) | Z = -1.073 | 0.283 |
| Depression | 4 (2-8) | 4 (0-9) | Z = -0.601 | 0.548 | 8 (4-10) | 6 (2-12) | Z = -0.017 | 0.986 |
| Anxiety | 6 (2-10) | 4 (2-10) | Z = -0.08 | 0.936 | 6 (4-10) | 6 (4-12) | Z = -1.041 | 0.298 |
| Stress | 10 (6-14) | 10 (6-14) | Z = -0.146 | 0.884 | 12.35 ± 5.56 | 13.18 ± 7.19 | t = -0.644 | 0.521 |
| Affectionate touch | 14 (10-19) | 17 (12-19) | Z = -1.381 | 0.167 | 15 (10-19) | 17 (11.5-20) | Z = -1.049 | 0.294 |
The GEE results for marital relationships are summarized in Table 4 (males) and Table 5 (females). For male participants, there was a significant group × time interaction for the total marital relationship score (δ = 3.79, 95%CI: 1.29-6.28, Wald = 8.85, P = 0.003), indicating that the intervention group experienced a significantly greater improvement over time compared to the control group. The effect size for this difference was medium (Cohen’s d = 0.60). In the subscale analysis, a significant interaction was observed for marital communication (δ = 1.85, 95%CI: 0.37-3.33, P = 0.014, Cohen’s d = 0.49). However, no significant group × time interactions were found for marital affection (P = 0.135) or cognitive understanding (P = 0.107), although the intervention group showed significant within-group improvements in these domains.
| Time | Control group | Intervention group | Within-group change (control) β (95%CI); P value | Within-group change (intervention) β (95%CI); P value | Group-time interaction difference Δ (95%CI) | Interaction P value | Effect size (Cohen’s d) |
| Total | |||||||
| Pre-intervention | 55.46 ± 0.60 | 55.97 ± 0.60 | |||||
| Post-intervention | 56.90 ± 0.91 | 61.20 ± 0.70 | 1.45 (-0.66 to 3.56); P = 0.180 | 5.24 (3.90-6.57); P < 0.001 | 3.79 (1.29-6.28) | 0.003 | 0.6 |
| Affection | |||||||
| Pre-intervention | 20.97 ± 0.24 | 21.35 ± 0.24 | |||||
| Post-intervention | 22.01 ± 0.38 | 23.23 ± 0.25 | 1.04 (0.05-2.04); P = 0.042 | 1.88 (1.40-2.36); P < 0.001 | 0.84 (-0.26 to 1.95) | 0.135 | 0.3 |
| Cognitive | |||||||
| Pre-intervention | 21.49 ± 0.34 | 21.31 ± 0.32 | |||||
| Post-intervention | 21.57 ± 0.54 | 22.49 ± 0.42 | 0.08 (-1.05 to 1.21); P = 0.888 | 1.18 (0.47-1.88); P = 0.001 | 1.09 (-0.24 to 2.42) | 0.107 | 0.32 |
| Communication | |||||||
| Pre-intervention | 13.10 ± 0.36 | 13.36 ± 0.34 | |||||
| Post-intervention | 13.43 ± 0.47 | 15.53 ± 0.40 | 0.33 (-0.89 to 1.54); P = 0.600 | 2.18 (1.33-3.02); P < 0.001 | 1.85 (0.37-3.33) | 0.014 | 0.49 |
| Time | Control group | Intervention group | Within-group change (control) β (95%CI); P value | Within-group change (intervention) β (95%CI); P value | Group-time interaction difference Δ (95%CI) | Interaction P value | Effect size (Cohen’s d) |
| Total | |||||||
| Pre-intervention | 54.93 ± 0.59 | 56.05 ± 0.43 | |||||
| Post-intervention | 56.46 ± 1.08 | 61.87 ± 0.58 | 1.53 (-1.44 to 4.50); P = 0.313 | 5.82 (4.31-7.34); P < 0.001 | 4.29 (0.96-7.62) | 0.012 | 0.51 |
| Affection | |||||||
| Pre-intervention | 20.28 ± 0.33 | 20.64 ± 0.22 | |||||
| Post-intervention | 20.65 ± 0.47 | 23.21 ± 0.35 | 0.37 (-1.05 to 1.79); P = 0.613 | 2.57 (1.65-3.48); P < 0.001 | 2.20 (0.51-3.89) | 0.011 | 0.51 |
| Cognitive | |||||||
| Pre-intervention | 20.45 ± 0.34 | 20.84 ± 0.24 | |||||
| Post-intervention | 21.35 ± 0.65 | 21.72 ± 0.36 | 0.90 (-0.74 to 2.54); P = 0.285 | 0.88 (0.06-1.70); P = 0.036 | -0.02 (-1.85 to 1.82) | 0.987 | 0 |
| Communication | |||||||
| Pre-intervention | 14.12 ± 0.32 | 14.60 ± 0.23 | |||||
| Post-intervention | 14.38 ± 0.64 | 16.97 ± 0.32 | 0.27 (-1.36 to 1.89); P = 0.750 | 2.37 (1.61-3.14); P < 0.001 | 2.11 (0.31-3.91) | 0.022 | 0.46 |
For female participants, a significant group × time interaction was observed for the total marital relationship score (δ = 4.29, 95%CI: 0.96-7.62, P = 0.012), with a medium effect size (Cohen’s d = 0.51). Regarding subscales, significant interactions were found for both marital affection (δ = 2.20, P = 0.011, Cohen’s d = 0.51) and marital communication (δ = 2.11, P = 0.022, Cohen’s d = 0.46). No significant interaction was observed for cognitive understanding (P = 0.987).
The changes in depression, anxiety, and stress are presented in Table 6 (males) and Table 7 (females). For male participants, the GEE analysis revealed a significant group × time interaction for anxiety (δ = -1.73, 95%CI: -3.29 to -0.17, P = 0.030), with a medium effect size (Cohen’s d = 0.44). Similarly, a significant interaction was observed for stress (δ = -2.11, 95%CI: -3.95 to -0.27, P = 0.025, Cohen’s d = 0.45). While the intervention group showed a significant within-group reduction in depression scores (P < 0.001), the group × time interaction for depression was not statistically significant (P = 0.319).
| Time | Control group | Intervention group | Within-group change (control) β (95%CI); P value | Within-group change (intervention) β (95%CI); P value | Group-time interaction difference Δ (95%CI) | Interaction P value | Effect size (Cohen’s d) |
| Depression | |||||||
| Pre-intervention | 6.27 ± 0.48 | 6.23 ± 0.48 | |||||
| Post-intervention | 5.04 ± 0.78 | 3.92 ± 0.59 | -1.22 (-3.00 to 0.55); P = 0.179 | -2.31 (-3.51 to -1.12); P < 0.001 | -1.09 (-3.23 to 1.05) | 0.319 | 0.2 |
| Anxiety | |||||||
| Pre-intervention | 6.07 ± 0.33 | 5.94 ± 0.36 | |||||
| Post-intervention | 5.17 ± 0.51 | 3.31 ± 0.40 | -0.90 (-2.14 to 0.34); P = 0.157 | -2.63 (-3.57 to -1.68); P < 0.001 | -1.73 (-3.29 to -0.17) | 0.03 | 0.44 |
| Stress | |||||||
| Pre-intervention | 10.34 ± 0.38 | 10.37 ± 0.41 | |||||
| Post-intervention | 8.13 ± 0.61 | 6.06 ± 0.54 | -2.20 (-3.67 to -0.74); P = 0.004 | -4.31 (-5.42 to -3.20); P < 0.001 | -2.11 (-3.95 to -0.27) | 0.025 | 0.45 |
| Time | Control group | Intervention group | Within-group change (control) β (95%CI); P value | Within-group change (intervention) β (95%CI); P value | Group-time interaction difference Δ (95%CI) | Interaction P value | Effect size (Cohen’s d) |
| Depression | |||||||
| Pre-intervention | 7.38 ± 0.36 | 7.43 ± 0.36 | |||||
| Post-intervention | 6.12 ± 0.85 | 4.60 ± 0.48 | -1.27 (-3.34 to 0.81); P = 0.234 | -2.82 (-3.95 to -1.69); P < 0.001 | -1.09 (-3.23 to 1.05) | 0.319 | 0.2 |
| Anxiety | |||||||
| Pre-intervention | 7.58 ± 0.28 | 7.26 ± 0.30 | |||||
| Post-intervention | 6.03 ± 0.60 | 5.46 ± 0.50 | -1.55 (-3.03 to -0.07); P = 0.041 | -1.80 (-2.96 to -0.65); P = 0.003 | -1.73 (-3.29 to -0.17) | 0.03 | 0.44 |
| Stress | |||||||
| Pre-intervention | 12.93 ± 0.39 | 12.66 ± 0.33 | |||||
| Post-intervention | 10.57 ± 0.74 | 7.68 ± 0.55 | -2.37 (-4.27 to -0.46); P = 0.016 | -4.98 (-6.35 to -3.61); P < 0.001 | -2.11 (-3.95 to -0.27) | 0.025 | 0.45 |
For female participants, a significant group × time interaction was observed only for stress (δ = -2.61, 95%CI: -4.96 to -0.26, P = 0.029), with a medium effect size (Cohen’s d = 0.44). Although female participants in the intervention group showed significant within-group reductions in depression and anxiety (both P < 0.01), the group × time interactions for these outcomes did not reach statistical significance (P = 0.196 for depression; P = 0.792 for anxiety).
For male participants, changes in semen parameters (semen volume, sperm motility, progressive motility, sperm concentration, and total sperm count) analyzed by GEE-adjusted models are presented in Table 8. Over the 4-week period, improvement in progressive motility was detected. Notably, in week 3, the intervention group showed a significant increase from baseline (β = 7.81, 95%CI: 3.55-12.08, P < 0.001) and a significant difference compared to controls in progressive motility. In week 4, further improvement in the intervention group was observed (β = 7.91, 95%CI: 0.30-15.51, P = 0.042) compared to controls. For female participants, pregnancy rate was assessed during a 3-month follow-up period after the intervention. Female participants in the intervention group (39/51, 76.5%) demonstrated a higher pregnancy rate compared with the control group (20/49, 40.8%).
| Time | CG | IG | CG difference β (95%CI); P value | IG difference β (95%CI); P value | Difference β (95%CI) | P value |
| Semen volume | ||||||
| T1 | 3.22 ± 0.05 | 3.15 ± 0.05 | -0.07 (-0.20 to 0.07) | 0.342 | ||
| T2 | 3.27 ± 0.15 | 3.22 ± 0.15 | 0.05 (-0.29 to 0.39); P = 0.767 | 0.07 (-0.27 to 0.41); P = 0.691 | 0.02 (-0.46 to 0.50) | 0.939 |
| T3 | 2.71 ± 0.26 | 2.85 ± 0.21 | -0.51 (-1.04 to 0.02); P = 0.062 | -0.31 (-0.74 to 0.13); P = 0.174 | 0.20 (-0.49 to 0.90) | 0.564 |
| Sperm motility rate | ||||||
| T1 | 46.56 ± 0.33 | 46.25 ± 0.32 | -0.31 (-1.21 to 0.59) | 0.500 | ||
| T2 | 46.84 ± 1.73 | 54.87 ± 1.45 | 0.27 (-3.31 to 3.86); P = 0.881 | 8.62 (5.58-11.66); P < 0.001 | 8.34 (3.64 to 13.05) | 0.001 |
| T3 | 46.39 ± 2.16 | 51.81 ± 2.62 | -0.17 (-4.42 to 4.08); P = 0.937 | 5.56 (0.30-10.81); P = 0.039 | 5.73 (-1.04 to 12.50) | 0.097 |
| Progressive motility rate | ||||||
| T1 | 37.85 ± 0.34 | 37.43 ± 0.34 | -0.42 (-1.35 to 0.51) | 0.372 | ||
| T2 | 38.50 ± 1.67 | 45.89 ± 1.20 | 0.64 (-2.84 to 4.12); P = 0.718 | 8.46 (5.99-10.92); P < 0.001 | 7.81 (3.55-12.08) | < 0.001 |
| T3 | 36.68 ± 2.39 | 44.16 ± 2.89 | -1.18 (-6.00 to 3.64); P = 0.633 | 6.73 (0.87-12.59); P = 0.025 | 7.91 (0.30-15.51) | 0.042 |
| Sperm concentration | ||||||
| T1 | 76.53 ± 1.92 | 80.06 ± 2.44 | 3.52 (-2.42 to 9.47) | 0.245 | ||
| T2 | 79.94 ± 7.14 | 87.21 ± 8.69 | 3.41 (-12.72 to 19.53); P = 0.679 | 7.15 (-11.13 to 25.42); P = 0.444 | 3.74 (-20.66 to 28.14) | 0.764 |
| T3 | 85.91 ± 5.74 | 86.63 ± 13.84 | 9.38 (-2.28 to 21.05); P = 0.116 | 6.57 (-21.82 to 34.96); P = 0.651 | -2.81 (-33.41 to 27.78) | 0.857 |
| Total sperm count | ||||||
| T1 | 231.98 ± 7.99 | 233.20 ± 8.02 | 1.23 (-21.01 to 23.46) | 0.914 | ||
| T2 | 257.58 ± 25.47 | 260.17 ± 32.48 | 25.61 (-29.41 to 80.62); P = 0.363 | 26.97 (-42.66 to 96.59); P = 0.449 | 1.36 (-87.29 to 90.01) | 0.976 |
| T3 | 205.24 ± 27.64 | 217.25 ± 32.16 | -26.74 (-85.54 to 32.06); P = 0.374 | -15.95 (-84.83 to 52.93); P = 0.650 | 10.79 (-79.09 to 100.67) | 0.814 |
A summary of all statistically significant primary and secondary outcomes, including effect sizes and interaction effects, is provided in Table 9.
| Outcome measure | Measurement | Gender | Key finding (intervention vs control) | Statistical significance (interaction) | Clinical/practical implication |
| Marital relationship | Communication | Male | Significant improvement in communication scores over time relative to control | β = 1.85, P = 0.014 | Improved verbal/non-verbal exchange |
| Communication | Female | Significant improvement in communication scores | β= 2.11, P = 0.022 | Enhanced emotional expression | |
| Affection | Female | Significant increase in affection scores | β = 2.20, P = 0.011 | Stronger emotional bonding | |
| Total score | Both | Significant improvement in overall relationship quality | Male: P = 0.003; female: P = 0.012 | Better dyadic adjustment during IVF | |
| Mental health | Anxiety | Male | Significant reduction in anxiety symptoms | β = -1.73, P = 0.030 | Reduced treatment-related arousal |
| Stress | Both | Significant reduction in stress levels | Male: P = 0.025; female: P = 0.029 | Effective stress buffering | |
| Reproductive | Sperm progressive motility | Male | Significant increase in progressive motility at week 3 and week 4 | Week 3: P < 0.001; week 4: P = 0.042 | Improved functional semen quality |
This study employed a non-randomized controlled trial to investigate the efficacy of a 4-week affectionate touch intervention for couples undergoing IVF-ET. The focus was on its impact on marital relationships, stress, negative emotions, and reproductive indicators. The completion rate for this program was quite high, with 93.6% for males and 94.1% for females. Compared to adherence rates for common behavioral health interventions (41%-90%)[30], and psychiatric pharmacotherapy (50%-63%)[31], the high adherence in this study may be attributed to the accessibility and remote monitoring of the program. Moreover, the intervention required minimal effort, as couples could perform daily touch exercises in familiar settings. Researchers used daily reminders via WeChat to reinforce habit formation and adherence to the intervention.
Our main results indicated that the affectionate touch-based program made a significant contribution to the impro
We also found gender differences in the results. Female participants exhibited greater improvements in the marital affection subscale. This result is potentially due to neurobiological sensitivity and cultural factors. Higher neural responsiveness to affectionate touch in females has been linked to increased body and relational satisfaction[33]. In Confucian societies, women often bear disproportionate stigma for infertility[36], making partner support via touch particularly impactful. Within many East Asian societies, traditional gender norms often socialize men to be stoic providers and women to be nurturing caregivers. The intervention, by offering a structured and sanctioned context for intimacy, may have uniquely empowered men to express vulnerability and engage in emotional communication. These behaviors are often discouraged by traditional masculinity norms. Conversely, for women, who are often socialized to prioritize emotional connection, the practice of affective touch may have been more readily integrated as a validation of their relational needs. This, in turn, deepened their experience of received affection.
Consistent with hypotheses, affectionate touch significantly alleviated stress in both genders, aligning with prior evidence of its role in mitigating anxiety, depressive behaviors, and physiological markers (e.g., blood pressure, heart rate). However, there are gender differences in the mental health benefits of the intervention based on affectionate touch. For the intervention group male participants, there is a significant reduction in anxiety compared to controls, but not in depression. Anxiety is often characterized by hyperarousal of the autonomic nervous system, and is highly responsive to the calming, regulatory effects of supportive touch. Anxiety, often linked to acute stress responses, is modulated by affectionate touch through parasympathetic activation and oxytocin release, which together rapidly suppress HPA axis activity and cortisol levels[37]. Depression, on the other hand, is frequently associated with anhedonia and psychomotor retardation, which are more complex and deeply entrenched states that may require longer or different therapeutic approaches to shift. The intervention’s immediate stress-buffering and physiological co-regulation effects may therefore have provided a more direct and potent antidote to the physiological underpinnings of anxiety. This aligns with the ‘tend-and-befriend’ model, which suggests that social bonding behaviors (like touch) are a primary stress regulation strategy for humans. Male participants might have previously lacked opportunities for such social bonding due to socio-cultural norms.
In contrast, depression involves chronic maladaptive cognitive patterns and dysregulation of serotonin and dopamine systems[38], which short-term behavioral interventions may not adequately address. Conversely, female participants showed no significant alleviation in either anxiety or depression, possibly due to multifaceted stressors experienced by women undergoing IVF-ET that require complementary strategies. The intervention’s effects on anxiety and depression were partial, with notable efficacy only for anxiety in male participants. A meta-analysis of 144 studies confirms touch’s broad benefits (e.g., pain relief, immune enhancement)[39]; however, our results suggest that the intervention duration (4 weeks) may be insufficient for sustained neurotransmitter modulation or cognitive changes.
Further, this study revealed that the affectionate touch intervention was significantly associated with improved progressive sperm motility in male participants over time, showing a marked difference compared to the control group. Existing research indicates that psychological interventions can positively influence semen quality; for instance, a 4-week mindfulness-based stress reduction program was shown to enhance sperm DNA integrity[40]. However, no significant changes were observed in total sperm count or sperm concentration in this study. Since sperm motility is a functional parameter, while count and concentration are quantitative parameters, these results suggest that the affectionate touch intervention may improve semen quality by enhancing functional aspects such as motility rather than by affecting spermatogenesis. The finding that the benefits of the intervention were primarily observed in sperm motility, rather than in concentration or count, significantly strengthens the biological plausibility of our results. Existing evidence suggests that psychological stress, primarily through the activation of the HPA axis and elevated cortisol levels, can impair sperm maturation and function in the epididymis more rapidly and reversibly, while suppressive effects on spermatogenesis require a longer duration to manifest[41,42]. Therefore, the observed improvement in motility following our short-term intervention aligns well with established neuroendocrine mechanisms. Prior studies confirm a negative correlation between anxiety and semen quality, with anxiety alleviation linked to improved sperm parameters[43]. Consequently, affectionate touch may mitigate oxidative damage by reducing cortisol levels, thereby improving sperm motility.
The intervention group demonstrated a significantly higher pregnancy rate than the control group, suggesting an association between psychological interventions and improved outcomes in assisted reproduction. A meta-analysis by Dube et al[44] highlighted the potential of psychological interventions to enhance pregnancy outcomes. Studies suggest that frequent affectionate touch between partners enhances physiological synchrony (e.g., heart rate, hormonal rhythms), serving as a proxy for empathy and triggering internal positive changes, such as activation of brain reward pathways and endogenous opioid release. These effects collectively promote well-being[34,45]. In participants receiving the intervention, the hormonal and HPA axis modulation induced by affectionate touch may indirectly contribute to improved clinical pregnancy rates. While the observed improvement in clinical pregnancy rates among the intervention group is encouraging, it is critical to interpret this finding with caution. These outcomes are observational and represent secondary endpoints. Although we controlled for key known confounders, such as age, BMI, and infertility duration, numerous other unmeasured factors could influence pregnancy success: For example, embryonic quality, specific laboratory protocols, and subtle immunological factors, including immune tolerance mechanisms. Nevertheless, the association between the intimacy intervention and pregnancy outcomes should not be interpreted as indicating causality. Instead, these results are hypothesis-generating and indicate a promising pathway for future research. Such research should employ designs that can more robustly test causality, including larger multi-center trials with stricter laboratory controls.
Admittedly, there are limitations of the current study, and further research is needed to confirm the efficacy of the current program. First, the primary limitation of this study is its non-randomized design. Despite attempts to enhance comparability by applying strict eligibility criteria and conducting baseline comparisons, residual selection bias due to patient self-selection or subtle preferences of clinicians during patient assignment cannot be ruled out. For instance, patients more willing to participate might have had higher baseline motivation or better initial relationship quality with their healthcare providers, potentially inflating the estimated treatment effects. Consequently, future research should employ randomized controlled trials to provide higher-quality evidence. Moreover, the study was conducted at a single center in Shanghai with a relatively small sample size (200 participants), limiting extrapolation to broader populations. Furthermore, due to the intervention’s cultural specificity, its efficacy in Confucian-influenced contexts (e.g., gender roles in infertility stigma) may not be generalizable to other cultural settings. Multicenter randomized trials with larger cohorts, extended follow-up periods, and biomarker assessments (e.g., cortisol, oxytocin) are needed to validate these findings. Integrating affectionate touch interventions with existing therapies, such as cognitive behavioral therapy, could optimize holistic outcomes for IVF-ET patients.
In addition to the limitations mentioned above, several others should be noted. First, there is potential for reporting bias in the self-logged records of daily tactile practices. Participants may have over-reported their adherence to the protocol due to social desirability bias or recall inaccuracies, which could inflate the perceived effectiveness of the intervention. To enhance data reliability, future studies could incorporate more objective monitoring tools or verification by a study partner involved in the intervention. Second, the follow-up duration for confirming pregnancy outcomes varied among participants because of differences in their individual treatment schedules within the IVF-ET process. Thirdly, while the GEE approach efficiently handles missing data, the attrition rate observed, primarily due to the logistical burden of IVF treatment and strict protocol adherence requirements. This heterogeneity in timing may have introduced variability in the assessment of study endpoints. Future research would benefit from implementing a standardized follow-up window timed from a fixed point in the treatment cycle (e.g., after ET) to ensure comparability across all participants.
The findings of this controlled trial demonstrate that a 4-week culturally adapted affectionate touch-based intervention - defined as structured, supportive physical contact aimed at enhancing emotional connection - significantly improved marital relationship quality, particularly communication, among couples undergoing IVF-ET in Shanghai. The intervention also alleviated psychological distress, with men showing reduced anxiety and stress, and women experiencing similarly reduced anxiety and stress. Moreover, the program was associated with enhanced sperm progressive motility and a higher clinical pregnancy rate in the intervention group (76.5% compared to 40.8% in the control group). These results underscore the potential of affectionate touch as a low-cost, accessible adjunct to routine IVF-ET care, addressing both psychological and physiological challenges faced by infertile couples. By engaging neurobiological pathways and stress-buffering mechanisms - such as modulation of the HPA axis and oxytocin release - the intervention helps mitigate treatment-related stress. This process fosters relational resilience and enhances couples’ ability to cope with infertility.
| 1. | Eisenberg ML, Esteves SC, Lamb DJ, Hotaling JM, Giwercman A, Hwang K, Cheng YS. Male infertility. Nat Rev Dis Primers. 2023;9:49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 329] [Article Influence: 109.7] [Reference Citation Analysis (0)] |
| 2. | Feng J, Wu Q, Liang Y, Liang Y, Bin Q. Epidemiological characteristics of infertility, 1990-2021, and 15-year forecasts: an analysis based on the global burden of disease study 2021. Reprod Health. 2025;22:26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 29] [Article Influence: 29.0] [Reference Citation Analysis (1)] |
| 3. | Infertility prevalence estimates, 1990-2021. Geneva: World Health Organization, 2023. |
| 4. | Qiao J, Wang Y, Li X, Jiang F, Zhang Y, Ma J, Song Y, Ma J, Fu W, Pang R, Zhu Z, Zhang J, Qian X, Wang L, Wu J, Chang HM, Leung PCK, Mao M, Ma D, Guo Y, Qiu J, Liu L, Wang H, Norman RJ, Lawn J, Black RE, Ronsmans C, Patton G, Zhu J, Song L, Hesketh T. A Lancet Commission on 70 years of women's reproductive, maternal, newborn, child, and adolescent health in China. Lancet. 2021;397:2497-2536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 471] [Cited by in RCA: 400] [Article Influence: 80.0] [Reference Citation Analysis (0)] |
| 5. | [Report on the Development of Maternal and Child Health in China (2019) (Part I)]. Zhongguo Fuyou Weisheng Zazhi. 2019;10:1-8. |
| 6. | Qiao J, Feng HL. Assisted reproductive technology in China: compliance and non-compliance. Transl Pediatr. 2014;3:91-97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 34] [Reference Citation Analysis (0)] |
| 7. | Wang L, Zhu Y, Wang T, Xu X, Tang Q, Li J, Wang Y, Hu W, Wu W. Feasibility analysis of incorporating infertility into medical insurance in China. Front Endocrinol (Lausanne). 2022;13:967739. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 8. | Marcellusi A, Scortichini M, Guarnotta G, Connolly M, Busnelli A. The economic benefit of implementing assisted reproductive technology within a national health system: insights from the Italian model. Health Econ Rev. 2025;15:76. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 9. | Li C, Fan Y, Chen D, Wu Y, Huang Y, Liu D. Relationship between marital satisfaction and defeat in IVF-ET: based on actor-partner interdependence model approach. Psychol Health. 2025;40:2146-2159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 10. | Zou F, Fang Y, Lin Y, Feng Z, Cai S, Huang J, Zheng S, Li J. Pathway analysis of the impact of family function and self-efficacy on depression and anxiety in patients undergoing in vitro fertilization-embryo transfer. BMC Psychol. 2024;12:749. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Park JC. Assessment of anxiety, depression, and sexual dysfunction in women undergoing fertility treatment. Clin Exp Reprod Med. 2025;52:38-43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 12. | Casu G, Zaia V, Fernandes Martins MDC, Parente Barbosa C, Gremigni P. A dyadic mediation study on social support, coping, and stress among couples starting fertility treatment. J Fam Psychol. 2019;33:315-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 13. | Gurunath S, Pandian Z, Anderson RA, Bhattacharya S. Defining infertility--a systematic review of prevalence studies. Hum Reprod Update. 2011;17:575-588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 206] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 14. | Purewal S, Chapman SCE, van den Akker OBA. Depression and state anxiety scores during assisted reproductive treatment are associated with outcome: a meta-analysis. Reprod Biomed Online. 2018;36:646-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 61] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 15. | Abdollahpour S, Taghipour A, Mousavi Vahed SH, Latifnejad Roudsari R. The efficacy of cognitive behavioural therapy on stress, anxiety and depression of infertile couples: a systematic review and meta-analysis. J Obstet Gynaecol. 2022;42:188-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 16. | Chow KM, Cheung MC, Cheung IK. Psychosocial interventions for infertile couples: a critical review. J Clin Nurs. 2016;25:2101-2113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 54] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 17. | Gaitzsch H, Benard J, Hugon-Rodin J, Benzakour L, Streuli I. The effect of mind-body interventions on psychological and pregnancy outcomes in infertile women: a systematic review. Arch Womens Ment Health. 2020;23:479-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 18. | Zhou R, Cao YM, Liu D, Xiao JS. Pregnancy or Psychological Outcomes of Psychotherapy Interventions for Infertility: A Meta-Analysis. Front Psychol. 2021;12:643395. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 19. | Zhang F, Lv Y, Wang Y, Cheng X, Yan Y, Zhang Y, Wang Y. The social stigma of infertile women in Zhejiang Province, China: a questionnaire-based study. BMC Womens Health. 2021;21:97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 20. | Fuertes M, Faria A, Gonçalves JL, Antunes S, Dionisio F. Antibiotic Consumption, Illness, and Maternal Sensitivity in Infants with a Disorganized Attachment. Children (Basel). 2023;10:1232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Shorey S, Asurlekar AR, Chua JS, Lim LHK. Influence of oxytocin on parenting behaviors and parent-child bonding: A systematic review. Dev Psychobiol. 2023;65:e22359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 22. | Ditzen B, Hoppmann C, Klumb P. Positive couple interactions and daily cortisol: on the stress-protecting role of intimacy. Psychosom Med. 2008;70:883-889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 97] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 23. | Floyd K, Boren JP, Hannawa AF, Hesse C, McEwan B, Veksler AE. Kissing in Marital and Cohabiting Relationships: Effects on Blood Lipids, Stress, and Relationship Satisfaction. West J Commun. 2009;73:113-133. [DOI] [Full Text] |
| 24. | Jakubiak BK, Feeney BC. Affectionate Touch to Promote Relational, Psychological, and Physical Well-Being in Adulthood: A Theoretical Model and Review of the Research. Pers Soc Psychol Rev. 2017;21:228-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 136] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 25. | Schneider E, Hopf D, Aguilar-Raab C, Scheele D, Neubauer AB, Sailer U, Hurlemann R, Eckstein M, Ditzen B. Affectionate touch and diurnal oxytocin levels: An ecological momentary assessment study. Elife. 2023;12:e81241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 26. | Wingenbach TSH, Ribeiro B, Nakao C, Gruber J, Boggio PS. Evaluations of affective stimuli modulated by another person's presence and affiliative touch. Emotion. 2021;21:360-375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Sorokowska A, Kowal M, Saluja S, Aavik T, Alm C, Anjum A, Asao K, Batres C, Bensafia A, Bizumic B, Boussena M, Buss DM, Butovskaya M, Can S, Carrier A, Cetinkaya H, Conroy-Beam D, Cueto RM, Czub M, Dural S, Espinosa A, Esteves CS, Frackowiak T, Contreras-Garduño J, Guemaz F, Hromatko I, Iskra H, Jiang F, Kafetsios K, Kavcic T, Kervyn N, Köbis NC, Kostić A, Láng A, Lindholm T, Manesi Z, Meskó N, Misra G, Monaghan C, Natividade JC, Nizharadze G, Oberzaucher E, Oleszkiewicz A, Pagani AF, Pakalniskiene V, Parise M, Pejičić M, Pisanski A, Pisanski K, Popa C, Prokop P, Sargautyte R, Sharad S, Simonetti F, Sorokowski P, Stefanczyk MM, Szagdaj A, Tadinac M, González KU, Uhryn O, Vauclair CM, Yoo G, Zupančič M, Croy I. Love and affectionate touch toward romantic partners all over the world. Sci Rep. 2023;13:5497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 28. | Chen SM, Zhang JF, Xiang YH. [Questionaire and Testing of Reliability and Validity about Marital Relationship]. Zhongguo Jiankang Xinlixue Zazhi. 2010;18:223-225. |
| 29. | Light KC, Grewen KM, Amico JA. More frequent partner hugs and higher oxytocin levels are linked to lower blood pressure and heart rate in premenopausal women. Biol Psychol. 2005;69:5-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 353] [Cited by in RCA: 231] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 30. | Davis MJ, Addis ME. Predictors of attrition from behavioral medicine treatments. Ann Behav Med. 1999;21:339-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 90] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 31. | Leon AC, Mallinckrodt CH, Chuang-Stein C, Archibald DG, Archer GE, Chartier K. Attrition in randomized controlled clinical trials: methodological issues in psychopharmacology. Biol Psychiatry. 2006;59:1001-1005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 205] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 32. | Jakubiak BK, Feeney BC. Hand-in-Hand Combat: Affectionate Touch Promotes Relational Well-Being and Buffers Stress During Conflict. Pers Soc Psychol Bull. 2019;45:431-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 33. | Campbell JT, Bennett-Brown M, Kaufman EM, Gesselman AN, Frederick DA, Garcia JR, Mark KP. Women Who Experience More Affectionate Touch Report Better Body Satisfaction and Relationship Outcomes. J Sex Res. 2025;62:776-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 34. | Jakubiak BK. Affectionate touch in satisfying and dissatisfying romantic relationships. J Soc Pers Relatsh. 2022;39:2287-2315. [DOI] [Full Text] |
| 35. | Olausson H, Wessberg J, Morrison I, McGlone F, Vallbo A. The neurophysiology of unmyelinated tactile afferents. Neurosci Biobehav Rev. 2010;34:185-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 477] [Cited by in RCA: 357] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 36. | Pu Y, Xu W. Parenting Desire Among Sexual Minority Women in China: From the Stigma Perspective. Arch Sex Behav. 2024;53:293-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 37. | Chang L, He Y, Tian T, Li B. Nucleus accumbens ghrelin signaling controls anxiety-like behavioral response to acute stress. Behav Brain Funct. 2024;20:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 38. | Aroke EN, Robinson AN, Wilbanks BA. Perioperative Considerations for Patients With Major Depressive Disorder Undergoing Surgery. J Perianesth Nurs. 2020;35:112-119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 39. | Moyer CA, Rounds J, Hannum JW. A meta-analysis of massage therapy research. Psychol Bull. 2004;130:3-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 455] [Cited by in RCA: 354] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 40. | Gautam S, Chawla B, Bisht S, Tolahunase M, Dada R. Impact of mindfulness based stress reduction on sperm DNA damage. J Anat Soc India. 2018;67:124-129. [RCA] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 41. | Wang X, Wang Y. The Effectiveness of Mindfulness-Based Intervention on Emotional States of Women Undergoing Fertility Treatment: A Meta-Analysis. J Sex Marital Ther. 2023;49:249-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 42. | Nargund VH. Effects of psychological stress on male fertility. Nat Rev Urol. 2015;12:373-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 146] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 43. | Pan Y, Wang S, Kang J, Cao T, Liu J, Zhang L, Niu S, Liu X. Association between generalized anxiety symptoms and semen quality in infertile men: A multicentre study in North China. Andrologia. 2022;54:e14449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 44. | Dube L, Bright K, Hayden KA, Gordon JL. Efficacy of psychological interventions for mental health and pregnancy rates among individuals with infertility: a systematic review and meta-analysis. Hum Reprod Update. 2023;29:71-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 41] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 45. | Wang X, Yu H, Zeng W, Hong Y, Huang F, Yang X, Wang Y. Psychological Well-being of Patients in Reproductive Medicine Center: Clinicians' Perspectives. J Reprod Infant Psychol. 2024;42:855-868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |