Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.114462
Revised: November 23, 2025
Accepted: January 4, 2026
Published online: April 19, 2026
Processing time: 164 Days and 0.5 Hours
Two severely disabling complications - levodopa-induced dyskinesia and de
To examines how serum 5-hydroxytryptamine (5-HT) concentrations relate to motor impairment (dyskinesia) and depressive symptoms in PD patients. Our goal is to assess whether serum 5-HT can function as a unified biomarker for these prevalent complications.
Between January 2019 and August 2019, we enrolled 70 PD patients from our hospital’s inpatient and outpatient services in an observational investigation. Participants were divided into two groups: Those with dyskinesia (n = 33) and those without (n = 37). Using standardized assessment tools, we evaluated depression and subsequently classified patients according to whether depressive symptoms were present. High-performance liquid chromatography enabled measurement of serum 5-HT concentrations. We gathered clinical information encompassing Hoehn-Yahr (H-Y) staging, levodopa equivalent daily dose, dyskinesia severity scores, and depression scale ratings. SPSS version 22.0 software facilitated our statistical analyses.
The dyskinesia group exhibited significantly reduced serum 5-HT concentrations relative to the non-dyskinesia group. Analysis of 5-HT distribution revealed that 78.8% (26/33) of dyskinesia patients had concentrations falling below the median value, contrasting sharply with just 29.7% (11/37) in the non-dyskinesia group (χ2 = 16.84, P < 0.001). Further stratification within the dyskinesia group utilized Abnormal Involuntary Movement Scale (AIMS) scores to assess dyskinesia severity. Patients with mild dyskinesia (AIMS 1-6, n = 14) demonstrated elevated 5-HT levels compared to those experiencing moderate-to-severe dyskinesia (AIMS ≥ 7, n = 19). While no significant association emerged between serum 5-HT concentrations and H-Y stages among dyskinesia patients (H-Y 2-2.5 vs H-Y 3-4: P = 0.073), the near-significant P value warrants cautious interpretation considering the constrained sample size. Similarly, maximum daily levodopa dosage showed no significant correlation.
PD patients presenting with dyskinesia and depression display markedly reduced serum 5-HT concentrations, indicating that serum 5-HT may represent a valuable marker for tracking both motor and non-motor complications. These results reinforce the contribution of serotonergic dysfunction to the underlying pathophysiology of dyskinesia and depression in PD, which may guide therapeutic approaches targeting serotonin-related pathways.
Core Tip: This study investigated the role of serum serotonin [5-hydroxytryptamine (5-HT)] as a biomarker for complications in Parkinson’s disease (PD). We found that reduced serum 5-HT levels were independently associated with both levodopa-induced dyskinesia and depression, two common motor and non-motor complications of PD. Serum 5-HT showed good discriminative ability for identifying at-risk patients and may provide a simple peripheral marker for early risk stratification and personalized management in PD.
- Citation: Chen YS, Yang SY, Lu KY, Li M, Liu T, Zhu SG, Lu HL, Hu K. Serum 5-hydroxytryptamine levels as biomarkers for motor dysfunction and depression in Parkinson’s disease patients. World J Psychiatry 2026; 16(4): 114462
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/114462.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.114462
For over half a century, levodopa has maintained its status as the gold standard therapeutic agent for Parkinson’s disease (PD), demonstrating superior efficacy compared to alternative treatments[1]. Despite its remarkable effectiveness in ameliorating PD motor manifestations, prolonged levodopa therapy frequently precipitates or intensifies motor complications[2]. These motor complications encompass three principal manifestations: Diminished therapeutic response, unpre
Non-motor symptoms represent an additional burden for PD patients beyond the motor complications, with de
Among PD motor complications, dyskinesia represents a particularly severe manifestation, distinguished by paroxys
Current limitations in understanding the pathogenic mechanisms underlying both dyskinesia and depression in PD, combined with restricted effective diagnostic and therapeutic options, render the management of these complications particularly challenging, seriously compromising patients’ quality of life[18]. Attempting to ameliorate dyskinesia through levodopa dose reduction proves counterproductive for motor symptom control, while depression management necessitates meticulous consideration of potential drug interactions with anti-parkinsonian medications[19]. Early identification of risk factors associated with dyskinesia and depression development in PD patients would enable proactive therapeutic strategy adjustments, potentially preventing or postponing the onset of these complications[20].
The diagnostic and prognostic utility of potential cerebrospinal fluid and blood biomarkers that closely mirror the pathophysiological foundations of both dyskinesia and depression has been demonstrated in several studies. However, the invasive nature of lumbar puncture procedures limits their widespread clinical implementation. Consequently, the identification of specific peripheral biomarkers would facilitate prediction of both motor and non-motor complication occurrence in PD patients.
Progressive striatal dopaminergic neuron degeneration in PD patients leads to a compensatory mechanism whereby serotonergic neurons expressing aromatic L-amino acid decarboxylase acquire the capacity to convert levodopa into dopamine. Serotonergic neurons located within the dorsal raphe nucleus functionally substitute for dopaminergic neurons in dopamine synthesis and release. These dorsal raphe nucleus serotonergic neurons, however, lack the feedback regulatory mechanisms necessary for controlled dopamine synthesis and synaptic release. This absence of regulation results in uncontrolled dopamine release into the hypersensitized striatum. Beyond contributing to dyskinesia, this dysregulation may also disrupt mood regulation circuits dependent upon proper serotonergic function.
Following dopamine denervation, striatal 5-hydroxytryptaminergic innervation undergoes significant augmentation, enabling the majority of levodopa to undergo metabolism via 5-hydroxytryptaminergic axon terminals, with subsequent release as dopamine. Furthermore, progressive dopaminergic neuron loss results in gradual replacement of stored 5-hydroxytryptamine (5-HT) within serotonergic neurons by dopamine, consequently diminishing serotonin release from axonal terminals and reducing its binding to autoreceptors. This reduction in serotonin availability serves dual functions: It amplifies feedback activity in serotonergic neurons, promoting enhanced dopamine release, while simultaneously contributing to depressive symptom development by disrupting normal serotonergic neurotransmission within limbic and cortical regions. Through vesicular monoamine transporter 2, dopamine demonstrates faster vesicular transport and filling rates compared to 5-HT, competitively inhibiting 5-HT production and release - a mechanism that may explain the concurrent manifestation of dyskinesia and depression in PD patients.
Recent evidence from both animal models and human investigations indicates strong correlations between serum 5-HT concentrations and those measured in brain tissue and cerebrospinal fluid samples, suggesting that peripheral serum 5-HT markers may partially reflect central 5-HT activity. Additional research has demonstrated that chronic levodopa administration stimulates angiogenesis. Elevated vascular endothelial growth factor levels and increased striatal capillary density compromise blood-brain barrier integrity, resulting in enhanced permeability to medium and large amino acids including 5-HT. These findings collectively suggest that monitoring peripheral serum 5-HT levels in PD patients may facilitate identification of individuals at elevated risk for developing both dyskinesia and depression.
A clear clinical need exists for biomarkers capable of predicting both motor and non-motor complication occurrence in PD while elucidating their underlying mechanisms. The present investigation examines the relationship between serum 5-HT concentrations and both dyskinesia and depression in PD patients. Our research establishes a foundation for early diagnosis of these complications and enhanced patient quality of life through more comprehensive and personalized therapeutic approaches.
Seventy patients with PD who were admitted to our hospital from January 2019 to August 2019 were enrolled in this observational study. Inclusion criteria: (1) The patients are able to complete the physical examination and questionnaire survey independently; (2) The patients have been taking levodopa for at least one month; and (3) The diagnosis of PD is according to the Movement Disorders Society diagnostic criteria of PD. Exclusion criteria: (1) The patients are suspected of having secondary Parkinson’s syndrome; (2) There is a co-morbidity with serious diseases, such as heart, liver, and kidney failure; (3) The patients have serious intellectual and communication impairments which could affect the eva
To ensure the validity of serum 5-HT measurements, we confirmed that none of the enrolled patients were taking medications known to significantly alter serotonin levels, including selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, tricyclic antidepressants, monoamine oxidase inhibitors, triptans, or St. John’s wort (Hypericum perforatum). Patients who had taken any such medications were required to complete a minimum 4-week washout period before enrollment. At the time of blood sample collection, all participants were confirmed to be free from these medications through careful medication history review and verification. Additionally, patients taking other medi
For the enrolled patients with PD, medical history collection, physical examination of the nervous system, and ques
Dyskinesia classification: Dyskinesia was clinically defined as a series of dystonia or dance-like involuntary movements that occur in patients with PD after taking dopaminergic drugs. According to dyskinesia’s clinical definition, patients with PD were classified into the dyskinesia group and non-dyskinesia group.
Depression classification: Depression was assessed using the Hamilton Depression Rating Scale (HAM-D) and the Beck Depression Inventory-II (BDI-II). Patients with HAM-D scores ≥ 14 or BDI-II scores ≥ 14 were classified as having clinically significant depressive symptoms.
Patients categorized: (1) PD with dyskinesia and depression; (2) PD with dyskinesia without depression; (3) PD without dyskinesia but with depression; and (4) PD without dyskinesia and without depression.
The 4 mL of fasting venous blood was drawn from all subjects between 7:00 to 8:00. Four vessels were filled with blood and then rotated 4 times gently to prevent coagulation. Within 30 minutes, the four blood vessels were centrifuged at room temperature at 2000 × g for 15 minutes. The supernatant was then added into a microcentrifuge tube and stored in a refrigerator at -80 °C. The levels of 5-HT in human serum were determined by high-performance liquid chromatography-tandem mass spectrometry.
Triple Quadrupole Mass Spectrometer by Waters (United States), equipped with electrospray ion source, Masslynx data processing software by Waters (United States), Waters ACQUITY UPLC I-Class high-performance liquid chromatography by Waters (United States), multi-tube vortex mixer by Hangzhou Youning Company (China), high-speed refrigerated centrifuge by Zhongke Zhongjia Company (China), and electronic balance by Shanghai Haosheng Company (China), were used. In this study, we used 5-HT by Beijing Solarbio (China), adriamycin by Shanghai Aladdin (China), formic acid by Fisher (United States), acetonitrile by Fisher (United States), methanol by Fisher (United States), and distilled water by Watson’s Water (China).
Involuntary Movements Rating Scale: The specialists from the PD Center of our hospital surveyed and evaluated patients with dyskinesia, and used Abnormal Involuntary Movement Scale (AIMS) to assess the clinical severity of dyskinesia. The AIMS is a scale designed by the National Institute of Mental Health to assess abnormal involuntary movements.
Unified PD Rating Scale: Unified PD Rating Scale (UPDRS) is a scale commonly used to assess PD’s severity worldwide. Patients with PD were evaluated with the third part of the UPDRS and the Hoehn-Yahr (H-Y) classification scale in the “off-period” to evaluate their motor symptoms and disease status.
General mental state: Patients with PD were assessed with UPDRS part 1 in the “off-period” to evaluate the general mental state of the patients.
Cognitive function: Mini-Mental State Examination scale was used to evaluate the cognitive function of the patients.
Multiple validated instruments facilitated clinical assessment of depressive symptoms. The HAM-D 17, a clinician-admi
For elderly participants, the Geriatric Depression Scale 15 served as a specialized screening instrument validated specifically for geriatric populations, with ≥ 5 representing the cut-off score indicating potential depressive symptoms. The PD Questionnaire 39 (PDQ-39) provided health-related quality of life evaluation, with particular emphasis on emotional well-being and social support network domains. Assessment reliability and bias minimization were ensured through evaluation conduct by experienced neurologists who remained blinded to participants’ serum 5-HT concentrations. Standardized examination protocols and assessment guidelines received consistent application across all subjects. Critically, all clinical assessments occurred during the medication-off state, which required participants to discontinue anti-parkinsonian medications for a minimum 12-hour period prior to evaluation.
SPSS version 22.0 facilitated all statistical analyses. Normally distributed continuous variables received presentation as mean ± SD for descriptive statistics, whereas non-normally distributed data utilized median (interquartile ranges).
The Kruskal-Wallis H test was employed for comparisons involving three or more independent groups, with sub
The study included 70 PD patients, with 37 in the non-dyskinesia group and 33 in the dyskinesia group. Gender distribution was similar between groups (P = 0.578), with no significant differences in height, weight, or cognitive function as measured by Mini-Mental State Examination scores. However, patients in the dyskinesia group were significantly younger at the time of evaluation (63.03 ± 10.34 years vs 69.00 ± 8.86 years, P = 0.013) and had an earlier age of onset (55.12 ± 10.72 years vs 63.43 ± 8.63 years, P = 0.001), suggesting that younger-onset PD patients may be at higher risk for developing dyskinesia. Patients with dyskinesia demonstrated more advanced disease characteristics. They had a longer disease duration (7.97 ± 3.97 years vs 5.16 ± 3.61 years, P = 0.004) and higher H-Y staging scores (2.97 ± 0.98 vs 2.41 ± 0.90, P = 0.017), indicating more severe motor disability. Non-motor symptoms were also more pronounced in the dyskinesia group, as evidenced by significantly higher UPDRS I scores (4.39 ± 3.94 vs 2.70 ± 2.83, P = 0.050), which assess mentation, behavior, and mood disturbances. Although UPDRS III motor scores showed a trend toward being higher in the dyskinesia group (52.06 ± 20.87 vs 42.62 ± 19.85), this difference did not reach statistical significance (P = 0.056, Table 1).
| Variable | Non-dyskinesia (n = 37) | Dyskinesia (n = 33) | t/χ2 | P value |
| Gender (male/female) | 19/18 | 15/18 | χ2 = 0.31 | 0.578 |
| Age (years) | 69.00 ± 8.86 | 63.03 ± 10.34 | t = 2.56 | 0.013a |
| Height (cm) | 163.64 ± 7.55 | 162.46 ± 7.66 | t = 0.65 | 0.520 |
| Weight (kg) | 63.11 ± 10.32 | 60.76 ± 11.47 | t = 0.90 | 0.373 |
| MMSE | 22.97 ± 6.29 | 21.79 ± 6.98 | t = 0.74 | 0.462 |
| Age of onset (years) | 63.43 ± 8.63 | 55.12 ± 10.72 | t = 3.50 | 0.001b |
| Course of disease (years) | 5.16 ± 3.61 | 7.97 ± 3.97 | t = -2.96 | 0.004b |
| UPDRS I (off phase) | 2.70 ± 2.83 | 4.39 ± 3.94 | t = -2.00 | 0.050a |
| UPDRS III (off phase) | 42.62 ± 19.85 | 52.06 ± 20.87 | t = -1.95 | 0.056 |
| H-Y stage (off phase) | 2.41 ± 0.90 | 2.97 ± 0.98 | t = -2.45 | 0.017a |
| Duration of dopaminergic treatment (years) | 3.12 ± 3.22 | 7.33 ± 4.14 | t = -4.74 | < 0.001c |
| Onset-to-treatment interval (years) | 1.37 ± 2.79 | 0.53 ± 1.22 | t = 1.59 | 0.117 |
| Duration of wearing-off (months) | 1.78 ± 6.46 | 19.70 ± 21.21 | t = -4.45 | < 0.001c |
| Duration of on-off (months) | 1.46 ± 4.78 | 21.49 ± 29.21 | t = -3.69 | < 0.001c |
| Equivalent dose for wearing-off (mg) | 40.54 ± 132.73 | 428.03 ± 326.96 | t = -6.23 | < 0.001c |
| Equivalent dose for on-off (mg) | 47.30 ± 149.98 | 393.94 ± 306.12 | t = -5.92 | < 0.001c |
| Maximum daily levodopa (mg) | 409.67 ± 294.09 | 735.39 ± 277.80 | t = -4.78 | < 0.001c |
Depression was identified in 31 (44.3%) of the 70 PD patients based on HAM-D scores ≥ 14 or BDI-II scores ≥ 14. The prevalence of depression was significantly higher in the dyskinesia group compared to the non-dyskinesia group (21/33, 63.6% vs 10/37, 27.0%; χ2 = 9.42, P = 0.002). Among patients with depression, the mean HAM-D score was 18.5 ± 4.8, indicating moderate to severe depressive symptoms. The BDI-II scores correlated strongly with HAM-D scores (r = 0.847, P < 0.001), confirming the reliability of depression assessment. Patients with depression had significantly higher PDQ-39 total scores (52.3 ± 15.7 vs 28.4 ± 12.1, P < 0.001), indicating poorer quality of life. The emotional well-being domain of PDQ-39 showed the most pronounced difference between depressed and non-depressed patients (68.2 ± 18.3 vs 22.1 ± 14.6, P < 0.001). Depression severity was positively correlated with disease duration (r = 0.412, P = 0.001) and UPDRS I score (r = 0.689, P < 0.001), but showed no significant correlation with UPDRS III motor scores (r = 0.186, P = 0.124) or H-Y staging (r = 0.203, P = 0.089, Table 2).
| Indicator | Depression (n = 31) | Non-depression (n = 39) | t/χ2/r | P value |
| Depression cases | 31 | 39 | 9.42 | 0.002a |
| HAM-D | 18.5 ± 4.8 | 8.2 ± 3.1 | 6.85 | < 0.001b |
| BDI-II | 19.1 ± 5.0 | 7.9 ± 2.8 | 7.10 | < 0.001b |
| PDQ-39 total | 52.3 ± 15.7 | 28.4 ± 12.1 | 7.37 | < 0.001b |
| PDQ-39 emotional well-being | 68.2 ± 18.3 | 22.1 ± 14.6 | 11.82 | < 0.001b |
| Correlation with disease duration | 0.412 | 0.180 | 0.412 | 0.001a |
| Correlation with UPDRS I | 0.689 | 0.250 | 0.689 | < 0.001b |
| Correlation with UPDRS III | 0.186 | 0.120 | 0.186 | 0.124 |
| Correlation with H-Y stage | 0.203 | 0.095 | 0.203 | 0.089 |
Serum 5-HT levels were significantly lower in the dyskinesia group compared to the non-dyskinesia group [median (interquartile rage): 145.2 (118.7-178.9) ng/mL vs 198.4 (165.3-245.7) ng/mL; Z = -3.047, P = 0.002]. The distribution of 5-HT levels showed that 78.8% (26/33) of patients in the dyskinesia group had 5-HT levels below the median value of the entire study, compared to only 29.7% (11/37) in the non-dyskinesia group (χ2 = 16.84, P < 0.001). Within the dyskinesia group, patients were further stratified by dyskinesia severity using AIMS scores. Mild dyskinesia (AIMS 1-6, n = 14) was associated with higher 5-HT levels compared to moderate-to-severe dyskinesia [AIMS ≥ 7, n = 19; 162.3 (138.2-189.5) ng/mL vs 128.7 (98.4-158.2) ng/mL; Z = -2.38, P = 0.017]. There was no significant difference in serum 5-HT levels between patients with different H-Y stages within the dyskinesia group (H-Y 2-2.5 vs H-Y 3-4: P = 0.073), nor was there a correlation with maximum daily levodopa dosage (r = -0.041, P = 0.815).
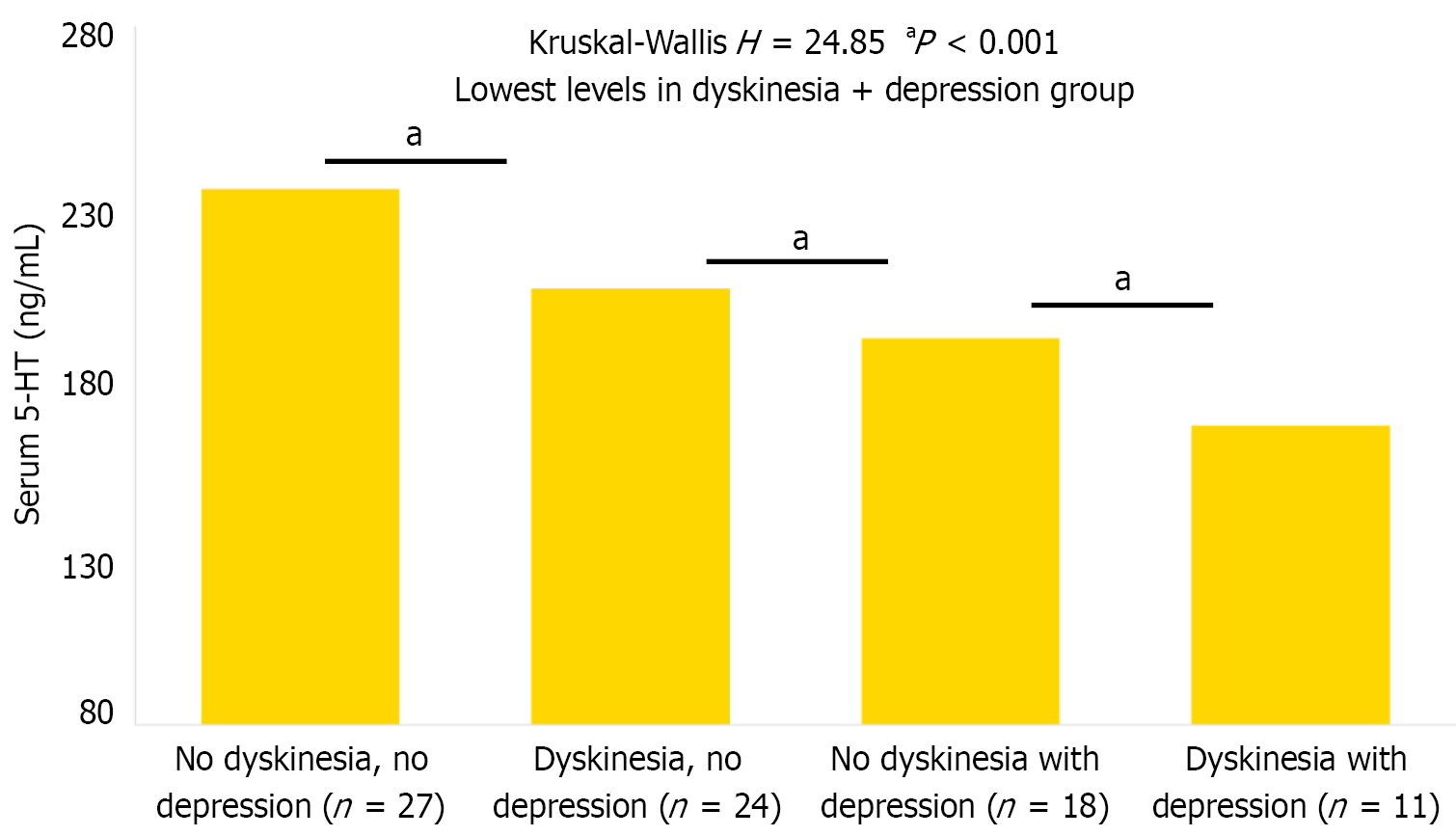
PD patients with depression demonstrated significantly reduced serum 5-HT levels compared to non-depressed patients [median (interquartile rage): 138.6 (105.2-172.8) ng/mL vs 201.5 (168.9-248.3) ng/mL; Z = -4.12, P < 0.001]. The relationship between 5-HT levels and depression severity was examined using correlation analysis, revealing a significant negative correlation between serum 5-HT concentrations and both HAM-D scores (r = -0.524, P < 0.001) and BDI-II scores (r = -0.487, P < 0.001). When patients were categorized into four subgroups based on the presence of dyskinesia and depression: (1) Dyskinesia with depression (n = 21): 122.4 (95.7-151.3) ng/mL; (2) Dyskinesia without depression (n = 12): 178.9 (152.6-205.4) ng/mL; (3) No dyskinesia but with depression (n = 10): 156.8 (128.5-189.2) ng/mL; and (4) No dyskinesia and no depression (n = 27): 215.7 (182.4-251.8) ng/mL. The differences between all four groups were statistically significant (Kruskal-Wallis H = 24.85, P < 0.001), with the lowest 5-HT levels observed in patients with both dyskinesia and depression (Figure 1).
Spearman correlation analysis revealed several significant associations between serum 5-HT levels and clinical variables. Serum 5-HT levels showed negative correlations with disease duration (r = -0.358, P = 0.002), H-Y staging (r = -0.312, P = 0.008), and UPDRS I score (r = -0.445, P < 0.001). There was a weak negative correlation with UPDRS III scores (r = -0.267, P = 0.025) and duration of dopaminergic treatment (r = -0.389, P = 0.001). Interestingly, 5-HT levels demonstrated a strong negative correlation with motor fluctuation parameters, including duration of wearing-off episodes (r = -0.512, P < 0.001) and on-off phenomena (r = -0.478, P < 0.001). The correlation with maximum daily levodopa dose was moderate but significant (r = -0.334, P = 0.005). Age at disease onset showed a positive correlation with 5-HT levels (r = 0.298, P = 0.012), while current age showed no significant correlation (r = 0.156, P = 0.196). Quality of life measures showed strong negative correlations with 5-HT levels, particularly the PDQ-39 total score (r = -0.567, P < 0.001) and the emotional well-being domain (r = -0.612, P < 0.001). These findings suggest that lower 5-HT levels are associated with more severe disease progression, greater motor complications, more pronounced depression, and poorer quality of life (Table 3).
| Variable | r | P value |
| Disease duration | -0.358 | 0.002b |
| H-Y staging | -0.312 | 0.008b |
| UPDRS I | -0.445 | < 0.001c |
| UPDRS III | -0.267 | 0.025a |
| Duration of dopaminergic treatment | -0.389 | 0.001b |
| Duration of wearing-off episodes | -0.512 | < 0.001c |
| Duration of on-off phenomena | -0.478 | < 0.001c |
| Maximum daily levodopa dose | -0.334 | 0.005b |
| Age at disease onset | 0.298 | 0.012a |
| Current age | 0.156 | 0.196 |
| PDQ-39 total score | -0.567 | < 0.001c |
| PDQ-39 emotional well-being | -0.612 | < 0.001c |
Multivariate logistic regression analysis was performed to identify independent predictors of dyskinesia and depression occurrence. For dyskinesia prediction, the final model included age at onset [odds ratio (OR) = 0.921, 95% confidence interval (CI): 0.867-0.979, P = 0.009], duration of dopaminergic treatment (OR = 1.298, 95%CI: 1.112-1.515, P = 0.001), maximum daily levodopa dose (OR = 1.003, 95%CI: 1.001-1.005, P = 0.008), and serum 5-HT levels (OR = 0.984, 95%CI: 0.975-0.993, P = 0.001). The model demonstrated good discriminative ability with an area under the curve (AUC) of 0.867 (95%CI: 0.785-0.949). For depression prediction, independent risk factors included H-Y staging (OR = 2.156, 95%CI: 1.234-3.767, P = 0.007), UPDRS I scores (OR = 1.387, 95%CI: 1.178-1.632, P < 0.001), disease duration (OR = 1.142, 95%CI: 1.012-1.289, P = 0.031), and serum 5-HT levels (OR = 0.981, 95%CI: 0.971-0.991, P < 0.001). This model achieved an AUC of 0.823 (95%CI: 0.731-0.915). When both outcomes were considered simultaneously in a combined model, serum 5-HT levels remained a significant independent predictor for both dyskinesia (OR = 0.986, 95%CI: 0.976-0.996, P = 0.006) and depression (OR = 0.983, 95%CI: 0.972-0.994, P = 0.003), suggesting its potential utility as a comprehensive biomarker (Table 4).
| Endpoint | Variable | OR | 95%CI | P value | AUC 95%CI |
| Dyskinesia | Age at onset | 0.921 | 0.867-0.979 | 0.009 | 0.867 (0.785-0.949) |
| Duration of dopaminergic treatment | 1.298 | 1.112-1.515 | 0.001 | ||
| Maximum daily levodopa dose | 1.003 | 1.001-1.005 | 0.008 | ||
| Serum 5-HT level | 0.984 | 0.975-0.993 | 0.001 | ||
| Depression | H-Y staging | 2.156 | 1.234-3.767 | 0.007 | 0.823 (0.731-0.915) |
| UPDRS I | 1.387 | 1.178-1.632 | 0.001 | ||
| Disease duration | 1.142 | 1.012-1.289 | 0.031 | ||
| Serum 5-HT level | 0.981 | 0.971-0.991 | 0.001 |
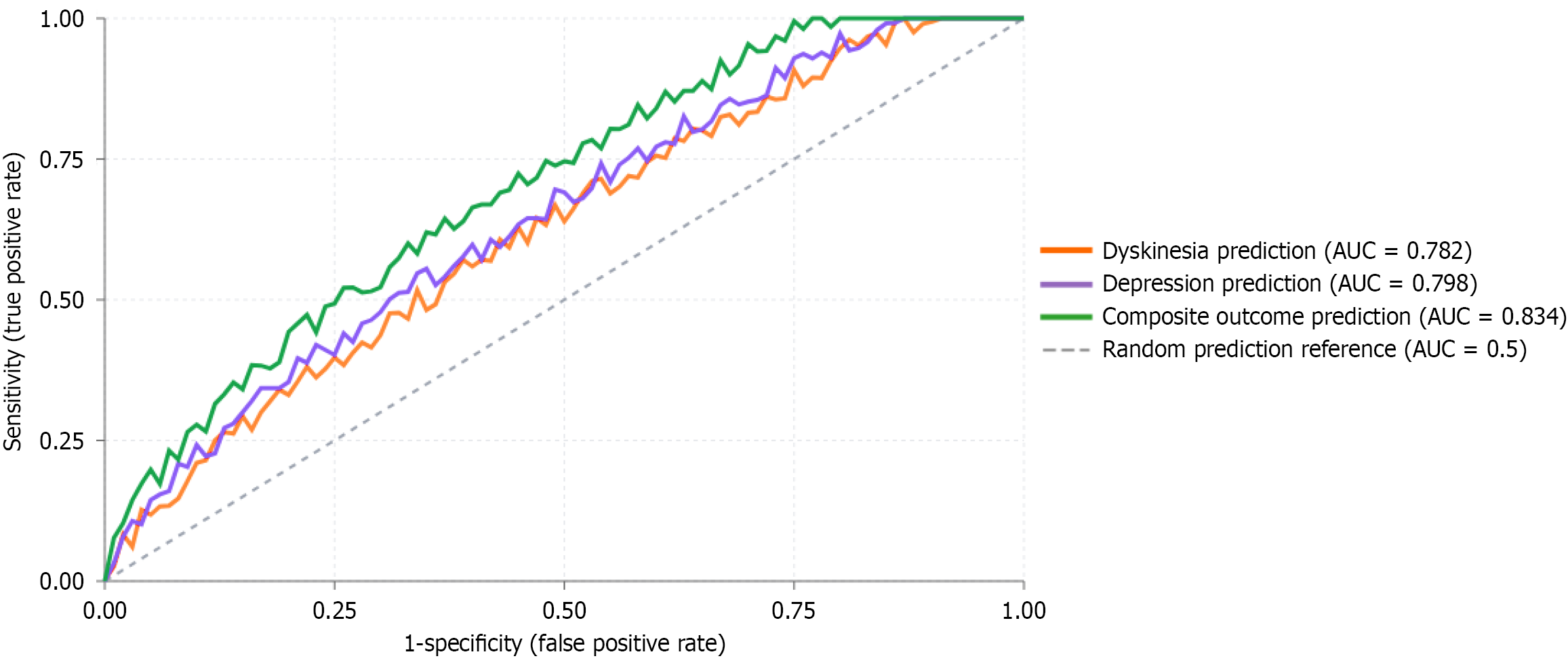
Receiver operating characteristic curve analysis was conducted to evaluate the diagnostic accuracy of serum 5-HT levels for predicting dyskinesia and depression in PD patients. For dyskinesia prediction, the optimal cut-off value was 168.5 ng/mL, yielding a sensitivity of 78.8% (95%CI: 61.1-91.0%) and specificity of 75.7% (95%CI: 58.8-88.2%), with an AUC of 0.782 (95%CI: 0.671-0.894, P < 0.001). For depression prediction, the optimal cut-off value was 158.2 ng/mL, achieving a sensitivity of 80.6% (95%CI: 62.5-92.5%) and specificity of 74.4% (95%CI: 60.4-85.3%), with an AUC of 0.798 (95%CI: 0.693-0.903, P < 0.001). The positive predictive value was 69.4% and negative predictive value was 84.3% for depression prediction. When combining both dyskinesia and depression as a composite outcome (presence of either or both complications), serum 5-HT levels demonstrated enhanced diagnostic performance with an AUC of 0.834 (95%CI: 0.741-0.927, P < 0.001). The optimal cut-off value of 162.4 ng/mL provided a sensitivity of 82.1% and specificity of 78.9%. These results suggest that serum 5-HT levels could serve as a valuable screening tool for identifying PD patients at risk for developing these common complications, potentially enabling early intervention and personalized treatment stra
In this study, we found that 5-HT levels were significantly lower in the dyskinesia group compared to the non-dyskinesia group (Z = -3.047, P = 0.002 < 0.017). In another study used Western blotting to measure 5-HT levels directly in the caudate nucleus and putamen of autopsy brains from 7 dyskinesia patients and 6 non-dyskinesia patients and found a statistically significant difference in the molar ratio of dopamine (77%). In addition, 5-HTP in patients with dyskinesia increased by 2 times compared with non-dyskinesia patients. This indicates that the 5-HT levels of the dyskinesia group were lower than that of the non-dyskinesia group. Some studies have shown that the release of dopamine from serotonergic neurons is the main mechanism of dyskinesia.
Previous neuroimaging studies have provided evidence for serotonergic dysfunction in PD-related depression[21]. Study used PET imaging with [¹¹C]DASB to demonstrate reduced serotonin transporter binding in the raphe nuclei, thalamus, and cingulate cortex of depressed PD patients compared to non-depressed patients[22]. Similarly, study found significantly reduced serotonin levels in the frontal cortex and hippocampus of PD patients with depression in post-mortem brain tissue analysis. Our peripheral measurement of serum 5-HT levels appears to reflect these central nervous system changes, supporting the utility of serum 5-HT as a biomarker for depression in PD.
The application of the serotonergic hypothesis to depression in PD is considerably more complex than in primary major depressive disorder due to the profound neuroanatomical overlap and functional interactions between dopaminer
The bidirectional interaction between these systems creates several mechanistic possibilities for depression in PD. First, serotonergic dysfunction may impair compensatory mechanisms that normally buffer against dopaminergic depletion in mood-related circuits. Second, the documented raphe nucleus pathology in PD, including Lewy body deposition and neuronal loss, suggests that serotonergic deficits may represent an independent pathological process that additively or synergistically contributes to depression beyond dopaminergic loss alone. Third, chronic levodopa treatment may dysregulate both systems simultaneously - while providing motor benefit through striatal dopamine restoration, it may exacerbate serotonergic dysfunction through the competitive mechanisms described in our study, potentially explaining why some patients develop depression despite adequate motor control.
Our finding of reduced serum 5-HT levels in depressed PD patients, independent of motor severity (no significant correlation with UPDRS III scores, r = 0.186, P = 0.124), suggests that serotonergic dysfunction may represent a relatively selective pathological pathway for mood disturbances rather than simply a reflection of overall disease burden. However, we acknowledge that disentangling the relative contributions of dopaminergic vs serotonergic dysfunction to depression in PD remains challenging and likely varies across individual patients. Future studies employing multimodal neuro
The co-occurrence of dyskinesia and depression in PD patients has been reported in several studies, with prevalence rates ranging from 20%-40%[23]. Our findings provide a potential biological explanation for this association. The pro
In this study, we found no statistically significant difference in serum 5-HT levels among patients at different H-Y stages of dyskinesia (P = 0.073). Furthermore, our comparison of serum 5-HT levels in patients receiving the highest dose of levodopa across different days revealed no statistically significant difference (P = 0.815). This indicates that the change in 5-HT levels is neither related to the overall disease severity nor the maximum daily dose of levodopa, suggesting that 5-HT alterations may be more specifically related to the development of complications rather than general disease pro
However, this study compared serum 5-HT levels in patients with varying severities of dyskinesia and found diffe
Similarly, for depression severity, we observed that patients with more severe depressive symptoms (higher HAM-D and BDI-II scores) had correspondingly lower serum 5-HT levels. This relationship was maintained across different depression severity categories, from mild to severe depression, suggesting that serum 5-HT levels could potentially be used to monitor treatment response and disease progression in PD-related depression.
Our correlation analysis revealed strong negative correlations between serum 5-HT levels and quality of life measures, particularly the PDQ-39 total score (r = -0.567, P < 0.001) and the emotional well-being domain (r = -0.612, P < 0.001)[24]. These findings highlight the functional significance of serotonergic dysfunction in PD patients. The emotional well-being domain showed the strongest correlation, which is consistent with the known role of serotonin in mood regulation and emotional processing.
From a therapeutic perspective, these findings suggest that treatments targeting serotonergic function might be beneficial for both motor and non-motor complications in PD. Selective serotonin reuptake inhibitors have shown mixed results in treating PD-related depression[25-27], and our findings might help identify patients most likely to benefit from such interventions. Additionally, novel therapeutic approaches targeting specific serotonin receptor subtypes or serotonin synthesis pathways might offer new treatment options for PD patients with low serum 5-HT levels. Some experimental studies on animals have shown that long-term treatment with 5-HT reuptake inhibitors, such as citalopram, increases the level of serotonin in the synaptic space, thereby promoting serotonergic neurotransmission and leading to downregulation of presynaptic 5-HT1B receptors[28-30]. There is a significant positive correlation between the magnitude of the increase in receptor mRNA expression and the severity of dyskinesia, so the severity of dyskinesia may be related to serum 5-HT levels[31,32]. There was no significant difference in 5-HT levels between the severe group and the non-dyskinesia group, which may be due to the small sample size and large interquartile variation in the severe group.
A fundamental limitation of our study is the inferential nature of linking serum 5-HT concentrations to central nervous system serotonergic tone - a challenge inherent to virtually all peripheral biomarker research in neurology. While several studies have reported correlations between peripheral and central serotonin measurements, this levodopa-induced angiogenesis and blood-brain barrier disruption might enhance permeability not only to amino acids like levodopa but also to monoamines including 5-HT, potentially creating a pathological communication between peripheral and central serotonin pools that does not exist in healthy individuals or early-stage PD.
A significant limitation of our study is the relatively modest sample size of 70 patients, which becomes more pro
Future studies with larger sample sizes, ideally multi-center collaborative efforts enrolling 150-200 patients, would be necessary to adequately power more nuanced subgroup analyses, particularly for examining potential interactions between dyskinesia severity, depression severity, and 5-HT levels.
In summary, our study demonstrates that serum 5-HT levels are significantly reduced in PD patients with both dyskine
| 1. | Connolly BS, Lang AE. Pharmacological treatment of Parkinson disease: a review. JAMA. 2014;311:1670-1683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1285] [Cited by in RCA: 1028] [Article Influence: 85.7] [Reference Citation Analysis (0)] |
| 2. | Espay AJ, Morgante F, Merola A, Fasano A, Marsili L, Fox SH, Bezard E, Picconi B, Calabresi P, Lang AE. Levodopa-induced dyskinesia in Parkinson disease: Current and evolving concepts. Ann Neurol. 2018;84:797-811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 272] [Article Influence: 34.0] [Reference Citation Analysis (0)] |
| 3. | Kwon DK, Kwatra M, Wang J, Ko HS. Levodopa-Induced Dyskinesia in Parkinson's Disease: Pathogenesis and Emerging Treatment Strategies. Cells. 2022;11:3736. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 113] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 4. | A randomized controlled trial comparing pramipexole with levodopa in early Parkinson's disease: design and methods of the CALM-PD Study. Parkinson Study Group. Clin Neuropharmacol. 2000;23:34-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 80] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 5. | Wu L, Liu Q, Ruan X, Luan X, Zhong Y, Liu J, Yan J, Li X. Multiple Omics Analysis of the Role of RBM10 Gene Instability in Immune Regulation and Drug Sensitivity in Patients with Lung Adenocarcinoma (LUAD). Biomedicines. . 2023;11:1861. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 78] [Article Influence: 26.0] [Reference Citation Analysis (1)] |
| 6. | Aarsland D, Påhlhagen S, Ballard CG, Ehrt U, Svenningsson P. Depression in Parkinson disease--epidemiology, mechanisms and management. Nat Rev Neurol. 2011;8:35-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 479] [Cited by in RCA: 388] [Article Influence: 25.9] [Reference Citation Analysis (1)] |
| 7. | Schrag A. Quality of life and depression in Parkinson's disease. J Neurol Sci. 2006;248:151-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 254] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 8. | Wu L, Zheng Y, Ruan X, Wu D, Xu P, Liu J, Wu D, Li X. Long-chain noncoding ribonucleic acids affect the survival and prognosis of patients with esophageal adenocarcinoma through the autophagy pathway: construction of a prognostic model. Anticancer Drugs. . 2022;33:e590-e603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 91] [Article Influence: 22.8] [Reference Citation Analysis (5)] |
| 9. | Richard IH. Depression in Parkinson's Disease. Curr Treat Options Neurol. 2000;2:263-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (2)] |
| 10. | Zesiewicz TA, Sullivan KL, Hauser RA. Levodopa-induced dyskinesia in Parkinson's disease: epidemiology, etiology, and treatment. Curr Neurol Neurosci Rep. 2007;7:302-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 39] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 11. | Carta M, Carlsson T, Kirik D, Björklund A. Dopamine released from 5-HT terminals is the cause of L-DOPA-induced dyskinesia in parkinsonian rats. Brain. 2007;130:1819-1833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 533] [Cited by in RCA: 485] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 12. | Wu L, Zhong Y, Yu X, Wu D, Xu P, Lv L, Ruan X, Liu Q, Feng Y, Liu J, Li X. Selective poly adenylation predicts the efficacy of immunotherapy in patients with lung adenocarcinoma by multiple omics research. Anticancer Drugs. 2022;33:943-959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 86] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 13. | Nicholson SL, Brotchie JM. 5-hydroxytryptamine (5-HT, serotonin) and Parkinson's disease - opportunities for novel therapeutics to reduce the problems of levodopa therapy. Eur J Neurol. 2002;9 Suppl 3:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 88] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 14. | Politis M, Loane C. Serotonergic dysfunction in Parkinson's disease and its relevance to disability. ScientificWorldJournal. 2011;11:1726-1734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 84] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 15. | Mayeux R, Stern Y, Cote L, Williams JB. Altered serotonin metabolism in depressed patients with parkinson's disease. Neurology. 1984;34:642-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 195] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 16. | Boileau I, Warsh JJ, Guttman M, Saint-Cyr JA, McCluskey T, Rusjan P, Houle S, Wilson AA, Meyer JH, Kish SJ. Elevated serotonin transporter binding in depressed patients with Parkinson's disease: a preliminary PET study with [11C]DASB. Mov Disord. 2008;23:1776-1780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 121] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 17. | Maillet A, Krack P, Lhommée E, Météreau E, Klinger H, Favre E, Le Bars D, Schmitt E, Bichon A, Pelissier P, Fraix V, Castrioto A, Sgambato-Faure V, Broussolle E, Tremblay L, Thobois S. The prominent role of serotonergic degeneration in apathy, anxiety and depression in de novo Parkinson's disease. Brain. 2016;139:2486-2502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 207] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 18. | Wu L, Zhong Y, Wu D, Xu P, Ruan X, Yan J, Liu J, Li X. Immunomodulatory Factor TIM3 of Cytolytic Active Genes Affected the Survival and Prognosis of Lung Adenocarcinoma Patients by Multi-Omics Analysis. Biomedicines. . 2022;10:2248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 85] [Article Influence: 21.3] [Reference Citation Analysis (1)] |
| 19. | Weintraub D, Moberg PJ, Duda JE, Katz IR, Stern MB. Effect of psychiatric and other nonmotor symptoms on disability in Parkinson's disease. J Am Geriatr Soc. 2004;52:784-788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 253] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 20. | Tong Q, Zhang L, Yuan Y, Jiang S, Zhang R, Xu Q, Ding J, Li D, Zhou X, Zhang K. Reduced plasma serotonin and 5-hydroxyindoleacetic acid levels in Parkinson's disease are associated with nonmotor symptoms. Parkinsonism Relat Disord. 2015;21:882-887. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 21. | Ballanger B, Strafella AP, van Eimeren T, Zurowski M, Rusjan PM, Houle S, Fox SH. Serotonin 2A receptors and visual hallucinations in Parkinson disease. Arch Neurol. 2010;67:416-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 175] [Article Influence: 10.9] [Reference Citation Analysis (1)] |
| 22. | Wu L, Li X, Qian X, Wang S, Liu J, Yan J. Lipid Nanoparticle (LNP) Delivery Carrier-Assisted Targeted Controlled Release mRNA Vaccines in Tumor Immunity. Vaccines (Basel). . 2024;12:186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 85] [Article Influence: 42.5] [Reference Citation Analysis (0)] |
| 23. | Pontone G, Williams JR, Bassett SS, Marsh L. Clinical features associated with impulse control disorders in Parkinson disease. Neurology. 2006;67:1258-1261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 164] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 24. | Jenkinson C, Fitzpatrick R, Peto V, Greenhall R, Hyman N. The Parkinson's Disease Questionnaire (PDQ-39): development and validation of a Parkinson's disease summary index score. Age Ageing. 1997;26:353-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1071] [Cited by in RCA: 956] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 25. | Gao R, Zhao P, Yan K. Selective Serotonin Reuptake Inhibitors for the Treatment of Depression in Parkinson's Disease: A Systematic Review and Meta-Analysis. Clin Drug Investig. 2024;44:459-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 26. | Heras-Garvin A, Weckbecker D, Ryazanov S, Leonov A, Griesinger C, Giese A, Wenning GK, Stefanova N. Anle138b modulates α-synuclein oligomerization and prevents motor decline and neurodegeneration in a mouse model of multiple system atrophy. Mov Disord. 2019;34:255-263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 88] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 27. | Starkstein SE, Brockman S. Management of Depression in Parkinson's Disease: A Systematic Review. Mov Disord Clin Pract. 2017;4:470-477. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 46] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 28. | Muñoz A, Li Q, Gardoni F, Marcello E, Qin C, Carlsson T, Kirik D, Di Luca M, Björklund A, Bezard E, Carta M. Combined 5-HT1A and 5-HT1B receptor agonists for the treatment of L-DOPA-induced dyskinesia. Brain. 2008;131:3380-3394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 196] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 29. | Lindenbach D, Palumbo N, Ostock CY, Vilceus N, Conti MM, Bishop C. Side effect profile of 5-HT treatments for Parkinson's disease and L-DOPA-induced dyskinesia in rats. Br J Pharmacol. 2015;172:119-130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 30. | Kuan WL, Zhao JW, Barker RA. The role of anxiety in the development of levodopa-induced dyskinesias in an animal model of Parkinson's disease, and the effect of chronic treatment with the selective serotonin reuptake inhibitor citalopram. Psychopharmacology (Berl). 2008;197:279-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 31. | Pinna A, Parekh P, Morelli M. Serotonin 5-HT(1A) receptors and their interactions with adenosine A(2A) receptors in Parkinson's disease and dyskinesia. Neuropharmacology. 2023;226:109411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 32. | Scholtissen B, Verhey FR, Steinbusch HW, Leentjens AF. Serotonergic mechanisms in Parkinson's disease: opposing results from preclinical and clinical data. J Neural Transm (Vienna). 2006;113:59-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 63] [Article Influence: 3.2] [Reference Citation Analysis (0)] |