Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.113329
Revised: November 24, 2025
Accepted: December 18, 2025
Published online: April 19, 2026
Processing time: 183 Days and 0.9 Hours
Postpartum depression (PPD) is closely associated with adverse pregnancy out
To clarify the roles played by maternal serum thyroid-stimulating hormone (TSH) and thyroid peroxidase antibody (TPOAb) in PPD risk and APOs.
We enrolled 83 women with PPD and 80 healthy pregnant controls from Sh
Compared with controls, PPD cases had higher TSH and TPOAb levels, both positively correlated with Edinburgh Postnatal Depression Scale and Self-Rating Depression Scale. In the final multivariable model, independent pre
Serum TSH and TPOAb levels were significantly elevated in mothers with PPD and correlated strongly with depressive severity. Multiparity ≥ 3, PPD, TSH ≥ 3 mIU/L, and TPOAb ≥ 7 U/mL each increase the risk of APOs.
Core Tip: This study included 83 patients with postpartum depression (PPD) and 80 healthy controls. By comparing serum-stimulating hormone and thyroid peroxidase antibody levels and assessing their associations with depressive symptoms, we identified both makers as positively correlated with PPD severity and as independent contributors to adverse pregnancy outcomes. Multiparity and PPD further heightened adverse pregnancy outcomes risk.
- Citation: Chen JJ, Chen XJ, She QM, Ouyang S. Maternal thyroid-stimulating hormone and thyroid peroxidase antibody levels: Associations with postpartum depression and impacts on adverse pregnancy outcomes. World J Psychiatry 2026; 16(4): 113329
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/113329.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.113329
Postpartum depression (PPD) is a common psychiatric disorder occurring within a woman’s first postnatal year and is characterized by persistent anxiety, depression, fatigue, and functional impairment[1]. More than 80% of women exp
Abnormally elevated serum thyroid-stimulating hormone (TSH) and the presence of thyroid autoantibodies, including thyroid peroxidase antibody (TPOAb), are known to increase the likelihood of PPD and its related symptoms. Prenatal TPOAb titer may help identify individuals at high risk of PPD through mechanisms involving positive feedback between placental corticotropin-releasing hormone and cortisol[7]. Ciolac et al[8] also reported that elevated TSH contributes to recognizing PPD-associated hypothyroidism. Increased TSH levels may adversely impact childbirth outcomes, particularly when accompanied by persistent severe maternal anxiety, which further elevates the risk of delivering a large for gestational age (LGA) infant[9]. Additionally, Wang et al[10], found that TPOAb positivity was inked to higher rates of postpartum thyroiditis and premature rupture of membranes (PROM), suggesting broader associations with adverse maternal and neonatal outcomes. Based on this evidence, we hypothesized that serum TSH and TPOAb levels are asso
A total of 163 pregnant women from Shenzhen Bao’an District Songgang People’s Hospital (March 2021 to March 2024) were included, comprising 83 subjects with PPD group and 80 healthy pregnancies. Demographic characteristics were comparable between groups (P > 0.05).
Inclusion criteria: (1) Meeting diagnostic requirements for PPD or an Edinburgh Postnatal Depression Scale (EPDS) score ≥ 13[11,12]; (2) Age 22-45 years; (3) 6-7 weeks postpartum; (4) Normal cognitive, communication, and executive ability; and (5) Delivery at our center with complete clinical data.
Exclusion criteria: (1) Current or past personality or psychiatric disorders; (2) Use of psychotropic agents or sedative-hypnotics within ≤ 4 weeks; (3) Antepartum depression or anxiety; (4) Gestational hypertension or diabetes; (5) Organic brain or systemic disease; (6) Thyroid dysfunction; (7) Major physical illness; and (8) Anxiety-disorders or schizophrenia spectrum disorders.
Serum indices: All enrolled mothers fasted overnight (> 8 hours). Morning venous blood (3 mL) was collected from re
Negative emotions: All participants completed the EPDS and the Self-rating Depression Scale (SDS)[13]. The EPDS contains 10 items scored 0-3, yielding a total score of 0-30, with ≥ 13 commonly used as the PPD threshold. The SDS com
APOs: The occurrence of APOs, including PROM, preterm birth, postpartum hemorrhage, LGA infants, hypertensive disorder complicating pregnancy (HDP), and gestational diabetes mellitus (GDM), was recorded in both groups, and total incidence rates were calculated.
Statistical analyses were performed using SPSS 22.0. Continuous variables are expressed as mean ± SD and compared using t-tests, whereas categorical variables are shown as n (%) and compared with χ2 tests. Spearman correlations assessed associations of TSH and TPOAb concentrations with PPD severity in PPD cases. Predictors of APOs were identified th
Table 1 summarizes baseline characteristics for the PPD (n = 83) and control (n = 80) groups. Age, gravidity, gestational age at delivery, neonatal birth weight, and feeding practices did not differ significantly (P > 0.05).
| Indicators | PPD group (n = 83) | Control group (n = 80) | χ2/t | P value |
| Age (years) | 28.37 ± 4.41 | 29.16 ± 4.19 | 1.172 | 0.243 |
| Gravidity (times) | 2.10 ± 0.91 | 2.20 ± 0.89 | 0.709 | 0.479 |
| Gestational age at delivery (weeks) | 38.72 ± 1.54 | 38.34 ± 1.55 | 1.570 | 0.118 |
| Neonatal birth weight (g) | 3110.55 ± 279.95 | 3161.71 ± 208.20 | 1.320 | 0.189 |
| Newborn feeding practices | 2.555 | 0.279 | ||
| Breast milk | 42 (50.60) | 43 (53.75) | ||
| Milk powder | 15 (18.07) | 20 (25.00) | ||
| Mixed | 26 (31.33) | 17 (21.25) |
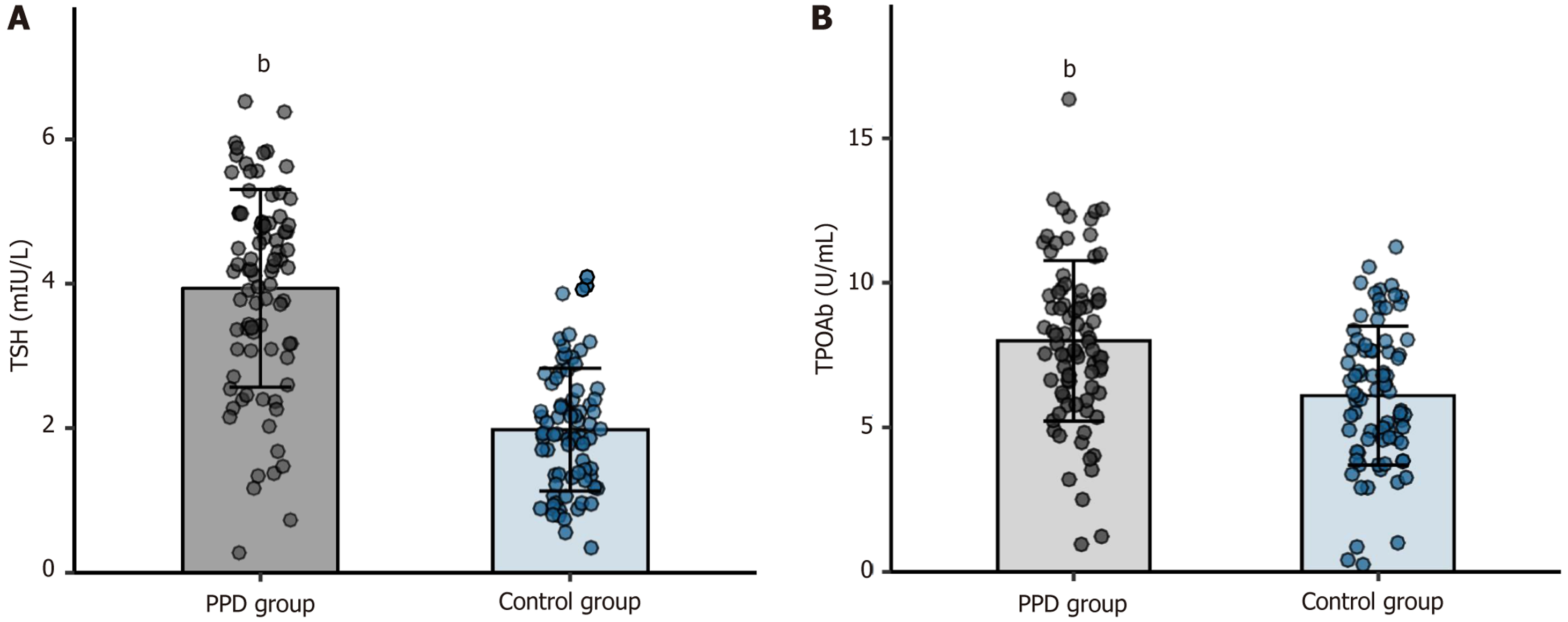
As shown in Figure 1, serum TSH and TPOAb concentrations were significantly higher in PPD cases than in controls (P < 0.01).
Table 2 presents Spearman correlation analyses between serum TSH/TPOAb levels and EPDS/SDS scores in PPD patients (n = 83). TSH levels correlated positively with EPDS (r = 0.500, P < 0.001) and SDS (r = 0.303, P = 0.005). TPOAb levels also showed positive correlations with EPDS (r = 0.347, P = 0.001) and SDS (r = 0.331, P = 0.002).
| Indicators | EPDS (points) | SDS (points) | ||
| r | P value | r | P value | |
| TSH | 0.500 | < 0.001 | 0.303 | 0.005 |
| TPOAb | 0.347 | 0.001 | 0.331 | 0.002 |
Table 3 compares pregnancy complications between cohorts. Women with PPD exhibited higher incidences of PROM, preterm birth, postpartum hemorrhage, LGA infants, HDP, and GDM than controls (P < 0.001).
| Indicators | PPD group (n = 83) | Control group (n = 80) | χ2 | P value |
| PROM | 6 (7.23) | 1 (1.25) | ||
| Preterm birth | 5 (6.02) | 2 (2.50) | ||
| Postpartum hemorrhage | 4 (4.82) | 1 (1.25) | ||
| LGA infants | 3 (3.61) | 0 (0.00) | ||
| HDP | 3 (3.61) | 1 (1.25) | ||
| GDM | 2 (2.41) | 1 (1.25) | ||
| Total | 23 (27.71) | 6 (7.50) | 11.380 | < 0.001 |
Univariable comparisons (Table 4), separating adverse (n = 29) from favorable (n = 134) outcomes, showed no significant associations with maternal age, gestational age at delivery, neonatal birth weight, or infant feeding (P > 0.05). Gravidity, PPD, TSH, and TPOAb were significantly associated with APOs (P < 0.05).
| Indicators | Adverse outcome group (n = 29) | Favorable outcome group (n = 134) | χ2 | P value |
| Age (years) | 0.409 | 0.522 | ||
| < 30 (n = 93) | 15 (51.72) | 78 (58.21) | ||
| ≥ 30 (n = 70) | 14 (48.28) | 56 (41.79) | ||
| Gravidity (times) | 9.766 | 0.002 | ||
| < 3 (n = 108) | 12 (41.38) | 96 (71.64) | ||
| ≥ 3 (n = 55) | 17 (58.62) | 38 (28.36) | ||
| Gestational age at delivery (weeks) | 1.754 | 0.185 | ||
| < 39 (n = 80) | 11 (37.93) | 69 (51.49) | ||
| ≥ 39 (n = 83) | 18 (62.07) | 65 (48.51) | ||
| Neonatal birth weight (g) | 0.891 | 0.345 | ||
| < 3150 (n = 86) | 13 (44.83) | 73 (54.48) | ||
| ≥ 3150 (n = 77) | 16 (55.17) | 61 (45.52) | ||
| Newborn feeding practices | 0.585 | 0.746 | ||
| Breast milk (n = 85) | 15 (51.72) | 70 (52.24) | ||
| Milk powder (n = 35) | 5 (17.24) | 30 (22.39) | ||
| Mixed (n = 43) | 9 (31.03) | 34 (25.37) | ||
| PPD | 11.380 | < 0.001 | ||
| No (n = 80) | 6 (20.69) | 74 (55.22) | ||
| Yes (n = 83) | 23 (79.31) | 60 (44.78) | ||
| TSH (mIU/L) | 13.210 | < 0.001 | ||
| < 3 (n = 89) | 7 (24.14) | 82 (61.19) | ||
| ≥ 3 (n = 74) | 22 (75.86) | 52 (38.81) | ||
| TPOAb (U/mL) | 7.284 | 0.007 | ||
| < 7 (n = 82) | 8 (27.59) | 74 (55.22) | ||
| ≥ 7 (n = 81) | 21 (72.41) | 60 (44.78) |
Table 5 presents the multifactorial modeling results, showing that multiparity ≥ 3 [odds ratio (OR) = 3.782, P = 0.007], PPD (OR = 2.921, P = 0.046), TSH ≥ 3 mIU/L (OR = 3.333, P = 0.019), and TPOAb ≥ 7 U/mL (OR = 3.958, P = 0.008) independently predicted APOs.
| Indicators | B | SE | Wald | P value | OR | 95%CI |
| Gravidity (times) | 1.330 | 0.494 | 7.253 | 0.007 | 3.782 | 1.436-9.960 |
| PPD | 1.072 | 0.538 | 3.964 | 0.046 | 2.921 | 1.017-8.393 |
| TSH (mIU/L) | 1.204 | 0.513 | 5.511 | 0.019 | 3.333 | 1.220-9.109 |
| TPOAb (U/mL) | 1.376 | 0.516 | 7.097 | 0.008 | 3.958 | 1.438-10.889 |
PPD is a common perinatal psychiatric disorder affecting up to 30% of mothers and may contribute to postpartum deaths from suicide, with an estimated risk of approximately 20%[14]. Postpartum stress, reduced quality of life, and low marital satisfaction all heighten PPD risk[15]. PPD is also linked to APOs, such as preterm birth, small for gestational age infants, preeclampsia, HDP, and GDM, which in turn increase long-term maternal mortality[16]. To mitigate these risks, the present study analyzed the association between PPD, thyroid function indicators, and APOs.
Our findings show that PPD cases had elevated serum TSH and TPOAb levels, implicating both makers in PPD pathophysiology. Elevated serum TSH level in PPD may partly reflect dysregulation of the microRNA-624-5p-silent information regulator sirtuin 1 axis in neuronal tissue; inhibition of this pathway reduces serum TSH levels and improves depressive symptoms[17]. Experimental in vivo work in rats indicates that TPOAb elevation may down-regulate brain-derived neurotrophic factor and serotonin (5-hydroxytryptamine) levels in the prefrontal cortex, thereby increasing PPD risk[18]. Consistent with our findings, Zhang et al[19] described elevated TSH level as a strong predictor of PPD and identified associations with reduced fetal weight, prenatal depression, and advanced maternal age.
Both serum TSH and TPOAb levels correlated significantly with EPDS and SDS scores, suggesting their contribution to PPD severity. Ciolac et al[8] similarly reported a significant positive correlation between TSH and EPDS scores. Kowalcze et al[20] also suggested that TSH and TPOAb levels may increase with increasing depression severity, supporting a positive correlation between these biomarkers and depression severity, and aligning with the trends observed in this study.
According to the statistical results, the overall incidence of APOs, including PROM, preterm birth, postpartum hemorrhage, LGA infants, HDP, and GDM, was significantly higher in the PPD group, indicating a strong association between PPD and APO occurrence. Multifactorial analysis further identified multiparity ≥ 3 (OR = 3.782, P = 0.007), PPD (OR = 2.921, P = 0.046), TSH ≥ 3 mIU/L (OR = 3.333, P = 0.019), and TPOAb ≥ 7 U/mL (OR = 3.958, P = 0.008) as independent predictors of APOs. Gravidity ≥ 3 may reflect cumulative reproductive tract stress, including potential endometrial injury and reduced myometrium elasticity, thereby increasing risks of PROM, preterm delivery, and postpartum hemorrhage. PPD may elevate APO likelihood by inducing chronic maternal psychological stress, which can contribute to placental dysfunction and inflammatory immune activation that adversely affect fetal development. Elevated TSH (≥ 3 mIU/L) and TPOAb (≥ 7 U/mL) may signal thyroid insufficiency and autoimmune dysregulation, respectively; together, they may exacerbate placental dysfunction and inflammatory pathways, compounding APO risk. Larsen et al[21] tracked pregnant women across 20 prenatal care facilities in western Nias and observed high APO rates linked to prenatal depression and inadequate social support, consistent with our results. Similarly, Wu et al[22] reported that TPOAb not only increased PPD risks but also contributes to GDM, hypertension, anemia, and postpartum thy
This study has several limitations. First, the single-center sample may introduce selection bias, underscoring the need for broader multicenter study to enhance generalizability. Second, maternal TSH and TPOAb levels were not dynamically monitored throughout pregnancy, future research should include longitudinal measurements to clarify their value in predicting PPD and APOs at different gestational stages. Third, although multivariable modeling was conducted, unmeasured confounders, such as psychosocial stress and family support, may still influence outcomes. Subsequent res
Elevated serum TSH and TPOAb levels were observed in pregnant women with PPD and showed significant positive correlations with depression severity. Both markers independently predict APOs, and multiparity and PPD further amplified these risks. Enhanced clinical surveillance and individualized management are therefore warranted for high-risk populations.
| 1. | Khamidullina Z, Marat A, Muratbekova S, Mustapayeva NM, Chingayeva GN, Shepetov AM, Ibatova SS, Terzic M, Aimagambetova G. Postpartum Depression Epidemiology, Risk Factors, Diagnosis, and Management: An Appraisal of the Current Knowledge and Future Perspectives. J Clin Med. 2025;14:2418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 2. | Dominiak M, Antosik-Wojcinska AZ, Baron M, Mierzejewski P, Swiecicki L. Recommendations for the prevention and treatment of postpartum depression. Ginekol Pol. 2021;92:153-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 3. | Tolossa T, Fetensa G, Yilma MT, Abadiga M, Wakuma B, Besho M, Fekadu G, Etafa W. Postpartum depression and associated factors among postpartum women in Ethiopia: a systematic review and meta-analysis, 2020. Public Health Rev. 2020;41:21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 4. | Domacassé D, de Rooij SR, Vrijkotte T, de Jonge A, Henrichs J. Associations Between Early-Pregnancy Vitamin D Status and Postpartum Depressive and Anxiety Symptoms. Psychosom Med. 2024;86:648-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 5. | Niimura J, Yamasaki S, Nakanishi M, Yamaguchi S, Baba K, Nakajima N, Miyashita M, Stanyon D, Knowles G, DeVylder J, Hiraiwa-Hasegawa M, Ando S, Kasai K, Nishida A. Investigating the association between the number of interpersonal supporters during first-time pregnancy and postpartum depression symptoms. Epidemiol Psychiatr Sci. 2025;34:e34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Roy U, Swain D. A prospective cohort study to assess the prevalence and risk factors of antepartum depression and its effect on maternal and fetal outcome. Asian J Psychiatr. 2024;91:103873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 7. | Backer S, Yancheva J, Garcia C, Khanna D. Thyroid Predictors of Postpartum Mood Disorders. Cureus. 2023;15:e45554. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 8. | Ciolac L, Bernad ES, Tudor A, Nițu DR, Buleu F, Popa DI, Toc T, Haivas C, Craina ML. Postpartum Depression: Interacting Biological Pathways and the Promising Validation of Blood-Based Biomarkers. J Clin Med. 2025;14:4286. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 9. | Wu P, Ouyang J, Cai W, Chen L, Tong J, Gao G, Wu X, Han Y, Yan S, Tao F, Huang K. Interaction between maternal thyroid hormone levels and pregnancy-related anxiety on birth outcomes-A birth cohort study. J Affect Disord. 2025;389:119646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 10. | Wang L, Tang Y, Yuan Y, Yu L, Jin B, Xia J, Yuan Q. Effects of Thyroperoxidase Antibody and Thyroglobulin Antibody on Maternal and Neonatal Outcomes in Pregnant Women. Horm Metab Res. 2022;54:76-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 11. | Moore Simas TA, Whelan A, Byatt N. Screening Recommendations and Treatments for Postpartum Depression-Reply. JAMA. 2024;331:1154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 12. | Fijean AL, Marçais M, Banasiak C, Morel O, Dahlhoff S, Olieric MF, Mottet N, Epstein J, Bertholdt C. Universal screening of postpartum depression with Edinburgh Postpartum Depression Scale: A prospective observational study. Int J Gynaecol Obstet. 2024;167:758-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | Tan J, Xiong Y, Wang X, Wei S, Luo C, Huang S, Yang Y, Chen J, Chen J, Xu M, Wu F. Influencing factors for postpartum depression in women with gestational diabetes mellitus. Front Endocrinol (Lausanne). 2024;15:1423127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Zhao X, Liu L. Mendelian randomization analyses for the causal relationship between early age at first sexual intercourse, early age at first live birth, and postpartum depression in pregnant women. Front Psychiatry. 2024;15:1287934. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 15. | Shim SH, Lee SY, Jung I, Heo SJ, Han YJ, Kwak DW, Kim MH, Park HJ, Chung JH, Lim JH, Kim MY, Cha DH, Shim SS, Cho HY, Ryu HM. Risk Factors of Postpartum Depression Among Korean Women: An Analysis Based on the Korean Pregnancy Outcome Study (KPOS). J Korean Med Sci. 2024;39:e31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 16. | Crump C, Sundquist J, Sundquist K. Adverse Pregnancy Outcomes and Long-Term Mortality in Women. JAMA Intern Med. 2024;184:631-640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 61] [Article Influence: 30.5] [Reference Citation Analysis (0)] |
| 17. | Wu W, Cao X, Wang Y. The role of miRNA-624-5p in congenital hypothyroidism and its molecular mechanism by targeting SIRT1. Genes Genomics. 2022;44:1137-1147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 18. | Zhou Y, Wang X, Zhao Y, Liu A, Zhao T, Zhang Y, Shan Z, Teng W. Elevated Thyroid Peroxidase Antibody Increases Risk of Post-partum Depression by Decreasing Prefrontal Cortex BDNF and 5-HT Levels in Mice. Front Cell Neurosci. 2016;10:307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 19. | Zhang R, Liu Y, Zhang Z, Luo R, Lv B. Interpretable Machine Learning Model for Predicting Postpartum Depression: Retrospective Study. JMIR Med Inform. 2025;13:e58649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 20. | Kowalcze K, Cucinella G, Gullo G, Krysiak R. Sexual Functioning and Depressive Symptoms in Women with Postpartum Thyroiditis. Diagnostics (Basel). 2025;15:1286. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 21. | Larsen A, Pintye J, Abuna F, Bhat A, Dettinger JC, Gomez L, Marwa MM, Ngumbau N, Odhiambo B, Phipps AI, Richardson BA, Watoyi S, Stern J, Kinuthia J, John-Stewart G. Risks of adverse perinatal outcomes in relation to maternal depressive symptoms: A prospective cohort study in Kenya. Paediatr Perinat Epidemiol. 2023;37:489-504. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 22. | Wu P, Yang M, Teng Y, Ouyang J, Cai W, Tong J, Gao G, Wu X, Han Y, Yan S, Tao F, Huang K. Association of maternal thyroid peroxidase antibody exposure with children's emotional and behavioral problems. Eur Thyroid J. 2025;14:e240302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 23. | Qin Z, Qiu Y, Lin J, Yang Y, Hao L, He L. Analysis of the influencing factors for adverse reproductive outcomes in patients with positive TPOAb and the establishment of a nomogram prediction model. Sci Rep. 2025;15:19637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |