Published online Mar 19, 2026. doi: 10.5498/wjp.v16.i3.116848
Revised: December 20, 2025
Accepted: December 31, 2025
Published online: March 19, 2026
Processing time: 94 Days and 23.7 Hours
Anxiety and depression during pregnancy are relatively common among pregnant women, with a prevalence rate reaching 30% in some regions of China. Existing studies suggest that maternal psychological states during pregnancy may influence fetal development through neuroendocrine mechanisms and are associated with neonatal outcomes. However, the relationship between these factors and specific neonatal indicators such as Apgar score and birth weight remains incompletely understood.
To investigate the effects of anxiety and depressive symptoms during pregnancy on the Apgar score and birth weight of newborns.
This study enrolled 100 primiparous women who registered and delivered at our hospital between October 2021 and October 2024. Participants were categorized into a normal group (70 cases) and an adverse outcome group (30 cases) based on neonatal outcomes. We collected and compared the general information, sleep status, mode of delivery, fasting blood glucose, and other clinical indicators of the two groups of pregnant women in the third trimester. Using logistic regression analysis, receiver operator characteristic (ROC) curves, and correlation analysis, we examined the relationship between pregnancy anxiety/depression symptoms and neonatal Apgar scores and birth weight.
The adverse outcome group exhibited significantly higher Self-Rating Anxiety Scale (SAS), Self-Rating Depression Scale (SDS), pregnancy stress scores, and fasting blood glucose levels compared to the normal group (all P < 0.05). Additionally, they showed poorer sleep quality, lower natural delivery rates, shorter gestational age, lower newborn birth weight, shorter body length, and lower 5-minute Apgar scores. Logistic regression analysis revealed that SAS, SDS, poor sleep quality, and pregnancy stress scores were independent risk factors for adverse neonatal outcomes (all P < 0.05). ROC analysis demonstrated that SAS and SDS had area under the curve values of 0.958 and 0.979, respectively, indicating strong predictive power for adverse neonatal outcomes (all P < 0.05). Correlation analysis showed negative correlations between anxiety/depression scores and 5-minute Apgar scores (R = -0.60, P < 0.001; R = -0.66, P < 0.001, respectively) and birth weight (R = -0.80, P < 0.001; R = -0.81, P < 0.001, respectively).
Pregnancy-related anxiety, depressive symptoms, poor sleep quality, and high stress levels are independent risk factors for adverse neonatal outcomes. The SAS and SDS scales demonstrate strong predictive value for such outcomes. Clinicians should prioritize maternal mental health, which supports healthy neonatal development.
Core Tip: This study demonstrates that anxiety [Self-Rating Anxiety Scale (SAS)] and depression [Self-Rating Depression Scale (SDS)] symptoms during pregnancy are significant independent risk factors for adverse neonatal outcomes, including lower Apgar scores and reduced birth weight. The SAS and SDS showed high predictive value (area under the curve: 0.958 and 0.979, respectively). The findings underscore the critical need for early screening and intervention for maternal psychological distress to improve neonatal health.
- Citation: Wu SJ, Wang JX, Kang HX, Li P. Effect of anxiety and depression symptoms in pregnancy on Apgar score and birth weight of newborns. World J Psychiatry 2026; 16(3): 116848
- URL: https://www.wjgnet.com/2220-3206/full/v16/i3/116848.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i3.116848
Pregnant women are in a uniquely challenging period both physiologically and psychologically. A global survey on maternal psychology revealed that the prevalence of depressive and anxious symptoms during pregnancy was 19.7% and 24.8%, respectively[1]. Data from China indicate that in some regions, economic pressures and marital stressors may lead to prevalence rates of prenatal anxiety and depression as high as 30%, highlighting the widespread prevalence of pregnancy-related psychological issues among expectant mothers[2]. Growing evidence suggests that maternal psychological states during pregnancy may influence fetal development through neuroendocrine and immune regulatory pathways. For instance, Abrishamcar et al[3] found a significant association between maternal stress/depression and DNA methylation patterns in the first year of life. Smew et al[4] demonstrated that the relaxation group exhibited significantly lower preterm birth rates compared to the anxiety/tension group, with newborns averaging 3.5 cm longer at birth, indicating substantial maternal psychological impact on neonatal outcomes. Apgar score and birth weight remain core indicators for assessing neonatal health[5]. The Apgar score comprehensively reflects cardiopulmonary function and neurological responsiveness at birth, while birth weight serves as a critical measure of fetal nutrition and maturity[6]. However, the specific mechanisms linking maternal anxiety/depression symptoms to neonatal Apgar scores and birth weight remain incompletely understood. Therefore, this study aims to investigate the relationship between maternal anxiety and depressive symptoms during pregnancy and neonatal outcomes (Apgar score and birth weight), in order to reveal the underlying mechanisms and thereby improve pregnancy outcomes.
This study adopted a case-control study design, selecting 100 primiparous women who established pregnancy records and delivered at our hospital between October 2021 and October 2024 as the study subjects.
Inclusion criteria: (1) Singleton pregnancy and gestational age between 14 weeks and 37 weeks at enrollment; (2) Intact communication and cognitive function; (3) Local permanent residents who agreed to return for postpartum follow-up; (4) Provided written informed consent; and (5) Had a prenatal record at our hospital and planned to deliver here.
Exclusion criteria: (1) Pre-existing diagnosis of depression, anxiety, or other mental disorders before pregnancy; (2) Occu
Sample size calculation: According to Kendall's sample size estimation principle[7], the required sample size should be 5 to 10 times the number of independent variables. This study included 15 independent variables, and the initial estimated sample size range was 75-150 cases. Considering approximately a 15% rate of invalid questionnaires, the final sample size was increased to a range of 88-176 cases. During the actual survey, 115 questionnaires were distributed, 110 were ret
Psychological assessment: During the third trimester, the psychological status of pregnant women was assessed using the Self-Rating Anxiety Scale (SAS) and the Self-Rating Depression Scale (SDS) during routine prenatal examinations. Both scales consist of 20 items each, employing a 1-4-point scoring system. The cut-off scores for SAS and SDS were 50 and 53, respectively, with higher scores indicating more severe anxiety or depressive symptoms. In this study, the Cronbach’s α for SAS and SDS were 0.81 and 0.82, respectively. Based on the scores from these two scales at the time of hospital admission, the corresponding standard scores for each pregnant woman were calculated[8].
Collection of general information: General data including maternal age, gestational age, SAS and SDS scores, as well as disease-related information such as employment status during pregnancy, sleep quality, pregnancy stress status, gra
Assessment of pregnancy stress: Pregnancy stress was evaluated using the Pregnancy Stress Rating Scale (PSRS)[9]. This scale consists of 30 items, with 27 of them distributed across three dimensions such as parental role identification stress, while the remaining 3 are independent items not included in the dimensional scoring. All items are rated on a 4-point scale from 0 to 3, with the total score ranging from 0 to 90. A higher score indicates a greater level of pregnancy stress. In this study sample, the scale demonstrated a Cronbach's α coefficient of 0.87, indicating good reliability.
Based on medical records, the collected pregnancy outcome indicators primarily included gestational age at delivery, mode of delivery, as well as neonatal birth weight, length, and Apgar score. In this study, neonates meeting any of the following criteria were defined as having an "adverse outcome": (1) Birth weight < 2500 g; and (2) 5-minute Apgar score < 7. Accordingly, the cases were divided into a normal group (70 cases) and an adverse outcome group (30 cases).
This study implemented rigorous quality control measures. All research personnel received uniform training to ensure consistent assessment standards. Data were collected using standardized scales widely adopted both domestically and internationally, all of which demonstrated good reliability in this study. Questionnaires were distributed and collected on-site immediately for data acquisition to ensure questionnaire completeness. Data management utilized a dual inde
Data analysis was performed using SPSS 27.0 software. Measurement data conforming to a normal distribution are expressed as mean ± SD, and intergroup comparisons were conducted using the t-test. Count data are expressed as n (%), and intergroup comparisons were conducted using the χ2 test. The influencing factors of adverse outcomes were analyzed by multivariate Logistic regression. The predictive efficacy of influencing factors on pregnancy outcomes was analyzed using the receiver operator characteristic (ROC) curve. Furthermore, Pearson correlation analysis was used to examine the relationships between anxiety and depression levels and neonatal Apgar scores and birth weight. The test level was set at α = 0.05, and a P value < 0.05 was considered statistically significant.
The proportion of pregnant women working during pregnancy (43.33% vs 65.71%) and the rate of natural delivery (23.33% vs 81.43%) in the adverse outcome group were significantly lower than those in the normal group (P < 0.05). Conversely, the proportion of poor sleep status (36.67% vs 22.86%), SAS score (57.23 ± 8.18 vs 40.77 ± 4.06), SDS score (56.23 ± 6.13 vs 41.01 ± 5.06), pregnancy stress scale score (53.13 ± 6.75 vs 47.56 ± 6.41), and fasting blood glucose (6.35 ± 0.77 mmol/L vs 6.03 ± 0.65 mmol/L) were significantly higher than those in the normal group (all P < 0.05). Additionally, the gestational age (37.00 ± 2.38 weeks vs 38.67 ± 2.03 weeks), birth weight (2712.15 ± 532.75 g vs 2986.45 ± 237.13 g), length (47.83 ± 3.49 cm vs 49.63 ± 1.46 cm), and 5-minute Apgar score (9.54 ± 0.45 vs 9.76 ± 0.28) of neonates in the adverse outcome group were significantly lower than those in the normal group (P < 0.05; Table 1).
| Group | Adverse outcome group | Normal group | t/χ2 | P value | |
| Age (year) | 29.17 ± 2.23 | 28.87 ± 2.12 | 0.629 | 0.531 | |
| Gestational week | 37.83 ± 1.78 | 38.17 ± 2.01 | 0.795 | 0.428 | |
| BMI (kg/m2) | 25.28 ± 1.56 | 25.48 ± 1.45 | 0.619 | 0.538 | |
| Education | Primary school | 7 (23.33) | 15 (21.43) | 0.077 | 0.994 |
| Junior high school | 7 (23.33) | 17 (24.29) | |||
| High school | 8 (26.67) | 18 (25.71) | |||
| College or above | 8 (26.67) | 20 (28.57) | |||
| SAS score | 57.23 ± 8.18 | 40.77 ± 4.06 | 10.481 | 0.000 | |
| SDS score | 56.23 ± 6.13 | 41.01 ± 5.06 | 12.923 | 0.000 | |
| Did you work during pregnancy | Yes | 13 (43.33) | 46 (65.71) | 4.349 | 0.037 |
| No | 17 (56.67) | 24 (34.29) | |||
| Occupation | Administrative organ | 1 (3.33) | 4 (5.71) | 5.267 | 0.261 |
| Public institution | 1 (3.33) | 14 (20.00) | |||
| Enterprise | 10 (33.33) | 21 (30.00) | |||
| Freelancer | 6 (20.00) | 10 (14.29) | |||
| Farming | 0 (0.00) | 0 (0.00) | |||
| Student | 0 (0.00) | 0 (0.00) | |||
| Othe | 12 (40.00) | 21 (30.00) | |||
| Monthly income (yuan) | < 5000 | 21 (70.00) | 40 (57.14) | 1.814 | 0.404 |
| 5000-10000 | 7 (23.33) | 26 (37.14) | |||
| > 10000 | 2 (6.67) | 4 (5.71) | |||
| Family history of mental illness | Have | 0 (0.00) | 0 (0.00) | - | 1.000 |
| None | 30 (100.00) | 70 (100.00) | |||
| The impact of the epidemic on pregnancy | Hardly | 14 (46.67) | 44 (62.86) | 6.202 | 0.185 |
| Occasionally | 9 (30.00) | 18 (25.71) | |||
| Sometimes | 5 (16.67) | 8 (11.43) | |||
| Often | 1 (3.33) | 0 (0.00) | |||
| Always | 1 (3.33) | 0 (0.00) | |||
| Payment method for medical expenses | Employee medical insurance | 5 (16.67) | 22 (31.43) | 2.633 | 0.268 |
| Resident medical insurance | 14 (46.67) | 30 (42.86) | |||
| Out-of-pocket | 11 (36.67) | 18 (25.71) | |||
| Pregnancy times | 2.07 ± 0.78 | 1.91 ± 0.74 | 0.929 | 0.355 | |
| Sleep condition | Good | 6 (20.00) | 33 (47.14) | 6.548 | 0.038 |
| Average | 13 (43.33) | 21 (30.00) | |||
| Poor | 11 (36.67) | 16 (22.86) | |||
| Pregnancy stress scale score | 53.13 ± 6.75 | 47.56 ± 6.41 | 3.929 | 0.000 | |
| Fasting blood glucose (mmol/L) | 6.35 ± 0.77 | 6.03 ± 0.65 | 2.054 | 0.043 | |
| Mode of delivery | Spontaneous delivery | 7 (23.33) | 57 (81.43) | 30.789 | 0.000 |
| Forceps delivery | 10 (33.33) | 6 (8.57) | |||
| Cesarean section | 13 (43.33) | 7 (10.00) | |||
| Gestational age (weeks) | 37.00 ± 2.38 | 38.67 ± 2.03 | 3.585 | 0.001 | |
| Newborn weight (g) | 2712.15 ± 532.75 | 2986.45 ± 237.13 | 2.708 | 0.011 | |
| Head circumference (cm) | 32.67 ± 2.04 | 32.84 ± 10.67 | 0.133 | 0.895 | |
| Body length (cm) | 47.83 ± 3.49 | 49.63 ± 1.46 | 2.722 | 0.010 | |
| 5-minute Apgar score (points) | 9.54 ± 0.45 | 9.76 ± 0.28 | 2.487 | 0.017 | |
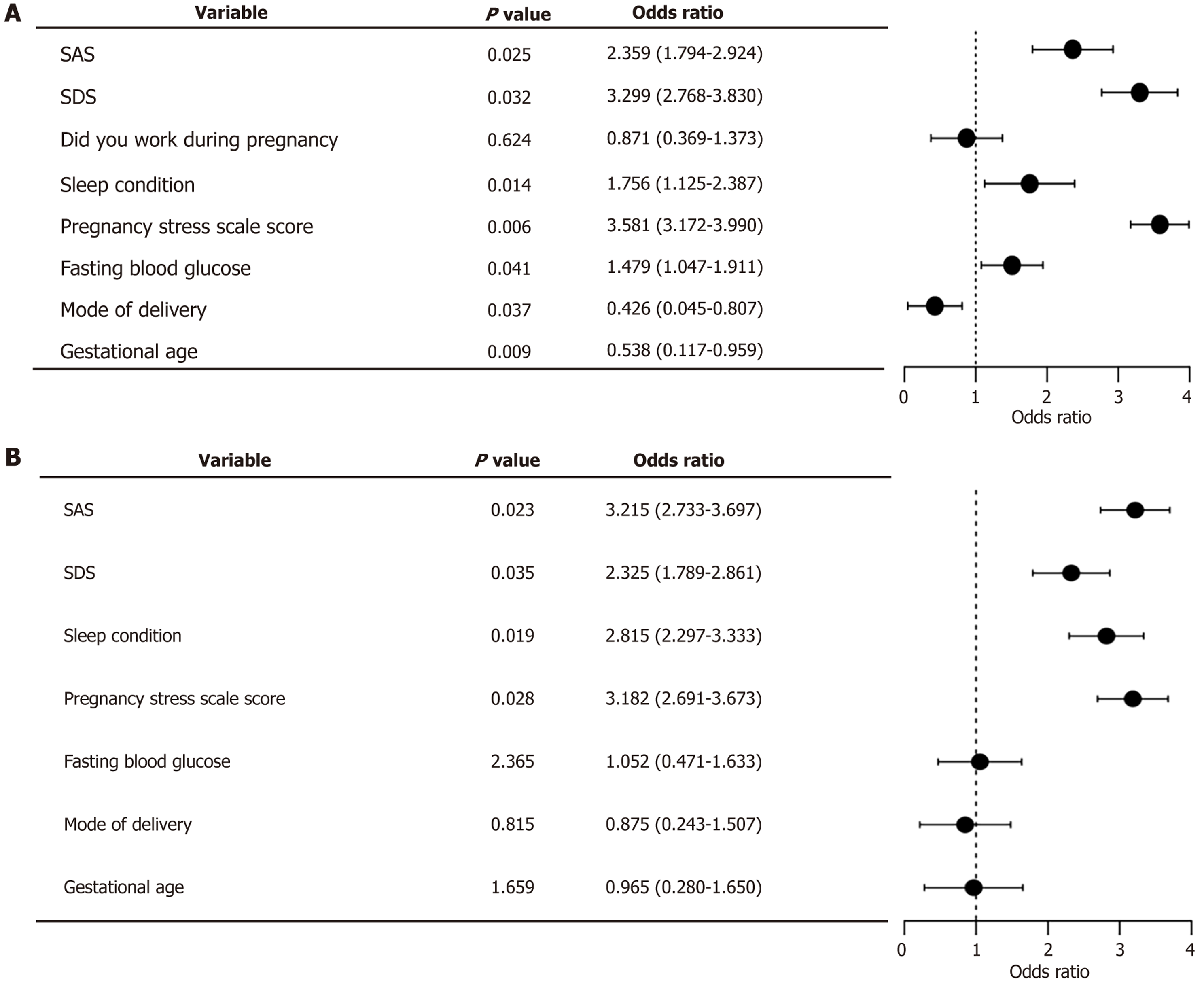
Using the occurrence of adverse neonatal outcomes as the dependent variable (assigned values: Yes = 1, no = 0), and the factors with P < 0.05 from sections 2.1 and 2.2 as independent variables. Measurement data including SAS, SDS, PSRS score, fasting blood glucose, and gestational age were entered as actual values. Assignments were as follows: Emp
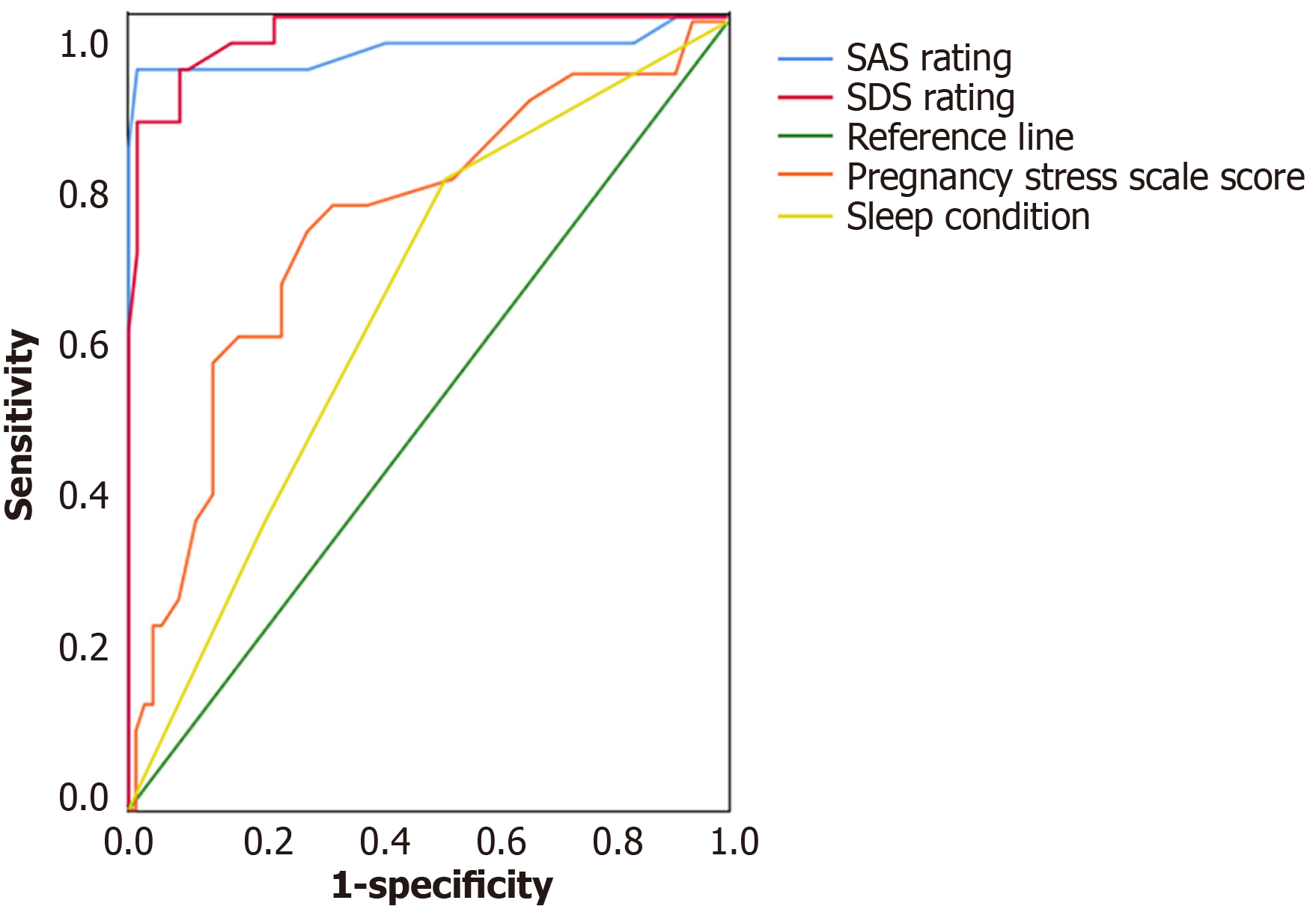
ROC analysis demonstrated that the area under the curve (AUC) values for SAS, SDS, poor sleep quality, and PSRS score were 0.958, 0.979, 0.739, and 0.641, respectively. Among these, SAS and SDS showed superior predictive performance, with P < 0.05 (Figure 2).
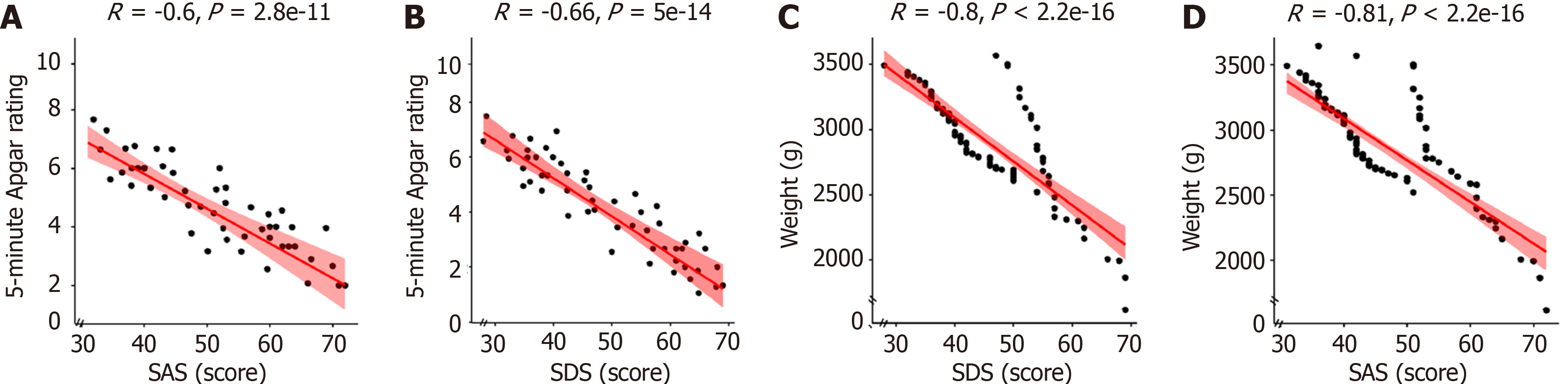
Pearson correlation analysis was used to examine the relationship between maternal SAS and SDS scores during pregnancy and neonatal 5-minute Apgar scores and birth weight. The results showed that both anxiety and depression scores were negatively correlated with the neonatal 5-minute Apgar score (anxiety: R = -0.60, P < 0.001; depression: R =
| Project | 5-minute Apgar rating | Birth weight | ||
| r value | P value | r value | P value | |
| Anxiety | -0.60 | < 0.001 | -0.80 | < 0.001 |
| Depression | -0.66 | < 0.001 | -0.81 | < 0.001 |
Pregnancy is a significant life event for women, exerting profound impacts on their physical health, psychological state, and family dynamics. With evolving lifestyles and increasing workplace pressures, the risk of anxiety and depression during pregnancy is showing an upward trend[10]. Relevant studies indicate that prolonged negative emotional states in pregnant women can easily lead to endocrine disorders and a deterioration in living conditions, thereby adversely affecting childbirth[11]. The results of this study demonstrate that poor maternal psychological status during pregnancy is a risk factor for adverse neonatal outcomes, providing a theoretical foundation for clinically improving pregnancy outcomes.
This study collected maternal clinical data and found through comparison that the adverse outcome group had significantly higher SAS scores, SDS scores, prevalence of work during pregnancy, poor sleep quality, PSRS scores, fasting blood glucose levels, and proportion of non-spontaneous deliveries compared to the normal group. Conversely, gestational age, birth weight, length, and 5-minute Apgar scores were significantly lower. This indicates that mothers of neonates with adverse outcomes had poorer psychological status, suboptimal sleep quality, and were in a prolonged state of physiological stress. The underlying mechanism is hypothesized as follows: Poor psychological status or sleep disturbances in pregnant women may activate the hypothalamic-pituitary-adrenal axis, promoting the release of corticotropin-releasing hormone and cortisol[12]. Studies have shown that high cortisol levels during pregnancy can cross the placental barrier, directly inhibiting the expression of genes related to fetal growth, leading to fetal growth restriction and shortened gestational age[13]. Concurrently, psychological stress can trigger inflammatory responses, increasing levels of pro-inflammatory cytokines such as tumor necrosis factor-α and interleukin-6. This may subsequently cause placental hypoperfusion and oxidative stress damage, further impeding fetal neurodevelopment[14]. Additionally, elevated maternal fasting blood glucose, often associated with insulin resistance. The hyperglycemic environment can activate the advanced glycation end products and their receptor signaling pathway, exacerbating placental vascular endothelial dysfunction and apoptosis, thereby increasing the risks of preterm birth and non-spontaneous delivery[15]. These maternal indicators during pregnancy are interrelated, collectively contributing to placental insufficiency and abnormal fetal development, ultimately elevating the risk of adverse neonatal outcomes. The study by Suda-Całus et al[16]. explicitly stated that mothers with diabetes have a higher risk of adverse maternal and infant outcomes, which aligns with the conclusions of this study.
This study further conducted logistic regression analysis, revealing that SAS scores, SDS scores, poor sleep quality, and PSRS scores are independent risk factors for adverse neonatal outcomes. This finding is consistent with that of Arvanitidou et al[17], who reported substantial interactions between depression, anxiety, stress, and childbirth problems. The potential mechanism can be analyzed as follows: A meta-analysis showed that pregnant women with high anxiety levels had significantly increased salivary cortisol concentrations[18]. Elevated maternal cortisol and catecholamine levels can persistently act on the placenta, directly causing functional pathological changes. High cortisol levels can down
The study employed correlation analysis to examine the relationship between SAS and SDS scores and neonatal Apgar scores and birth weight. It was found that as SAS and SDS scores increased, the risk of adverse neonatal outcomes also rose. The underlying mechanism may be explained as follows: Maternal anxiety levels show a significant negative correlation with the degree of DNA methylation in the placental glucocorticoid receptor gene. When hypomethylation occurs in the promoter region of this gene, the placental stress response to cortisol becomes abnormally amplified[22]. More critically, a high-cortisol environment can induce mitochondrial dysfunction. Research indicates that placental tissue from highly anxious mothers shows a significant reduction in mitochondrial DNA copy number and decreased respiratory chain complex activity[23]. Furthermore, severe psychological distress during pregnancy can suppress the mTOR signaling pathway, downregulating the expression of placental amino acid transporters. This leads to a deficiency in the raw materials necessary for the fetus to synthesize its own proteins, directly resulting in decreased neonatal birth weight[24]. Consequently, the fetus experiences not only growth restriction but also inhibited organ development. This directly explains the comprehensive decline in Apgar scores observed in neonates at birth, which is attributed to neu
In summary, SAS and SDS scores, poor sleep quality, and PSRS scores are independent risk factors for adverse neonatal outcomes. Elevated SAS and SDS scores are associated with decreased neonatal Apgar scores and reduced birth weight, demonstrating good predictive efficacy for adverse neonatal outcomes. These findings are expected to provide a the
We sincerely thank all the participants of this study for their valuable contributions.
| 1. | Rondung E, Massoudi P, Nieminen K, Wickberg B, Peira N, Silverstein R, Moberg K, Lundqvist M, Grundberg Å, Hultcrantz M. Identification of depression and anxiety during pregnancy: A systematic review and meta-analysis of test accuracy. Acta Obstet Gynecol Scand. 2024;103:423-436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 2. | Li Y, Zeng Y, Zhu W, Cui Y, Li J. Path model of antenatal stress and depressive symptoms among Chinese primipara in late pregnancy. BMC Pregnancy Childbirth. 2016;16:180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 3. | Abrishamcar S, Zhuang BC, Thomas M, Gladish N, MacIsaac JL, Jones MJ, Simons E, Moraes TJ, Mandhane PJ, Brook JR, Subbarao P, Turvey SE, Chen E, Miller GE, Kobor MS, Hüls A. Association between maternal perinatal stress and depression and infant DNA methylation in the first year of life. Transl Psychiatry. 2024;14:445. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 4. | Smew AI, Lundholm C, Gong T, Sävendahl L, Lichtenstein P, Brew BK, Almqvist C. Maternal depression or anxiety during pregnancy and offspring type 1 diabetes: a population-based family-design cohort study. BMJ Open Diabetes Res Care. 2023;11:e003303. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 5. | Lungameni J, Nghitanwa EM, Uusiku L. Neonatal factors associated with immediate low Apgar score in newborn babies in an intermediate hospital in Namibia: a case control study. Afr Health Sci. 2023;23:141-148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Ehrhardt H, Aubert AM, Ådén U, Draper ES, Gudmundsdottir A, Varendi H, Weber T, Zemlin M, Maier RF, Zeitlin J; EPICE-SHIPS Research Group. Apgar Score and Neurodevelopmental Outcomes at Age 5 Years in Infants Born Extremely Preterm. JAMA Netw Open. 2023;6:e2332413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 7. | Bolarinwa OA. Sample size estimation for health and social science researchers: The principles and considerations for different study designs. Niger Postgrad Med J. 2020;27:67-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 70] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 8. | Lovibond PF, Lovibond SH. The structure of negative emotional states: comparison of the Depression Anxiety Stress Scales (DASS) with the Beck Depression and Anxiety Inventories. Behav Res Ther. 1995;33:335-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10682] [Cited by in RCA: 7940] [Article Influence: 256.1] [Reference Citation Analysis (2)] |
| 9. | Chen CH. Revision and validation of a scale to assess pregnancy stress. J Nurs Res. 2015;23:25-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 10. | Vigod SN, Frey BN, Clark CT, Grigoriadis S, Barker LC, Brown HK, Charlebois J, Dennis CL, Fairbrother N, Green SM, Letourneau NL, Oberlander TF, Sharma V, Singla DR, Stewart DE, Tomasi P, Ellington BD, Fleury C, Tarasoff LA, Tomfohr-Madsen LM, Da Costa D, Beaulieu S, Brietzke E, Kennedy SH, Lam RW, Milev RV, Parikh SV, Ravindran AV, Samaan Z, Schaffer A, Taylor VH, Tourjman SV, Ameringen MV, Yatham LN, Van Lieshout RJ. Canadian Network for Mood and Anxiety Treatments 2024 Clinical Practice Guideline for the Management of Perinatal Mood, Anxiety, and Related Disorders: Guide de pratique 2024 du Canadian Network for Mood and Anxiety Treatments pour le traitement des troubles de l'humeur, des troubles anxieux et des troubles connexes périnatals. Can J Psychiatry. 2025;70:429-489. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 37] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 11. | Wu Y, Niu Y, Guo Q, Liu X, Hu H, Gong L, Xu Y, Hu Y, Li G, Xia X. Severity of depressive and anxious symptoms and its association with birth outcomes among pregnant women during the COVID-19 pandemic: a prospective case-control study. J Psychosom Obstet Gynaecol. 2024;45:2356212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 12. | La Marca-Ghaemmaghami P, Dainese SM, Stalla G, Haller M, Zimmermann R, Ehlert U. Second-trimester amniotic fluid corticotropin-releasing hormone and urocortin in relation to maternal stress and fetal growth in human pregnancy. Stress. 2017;20:231-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 13. | Pignatti E, du Toit T, Flück CE. Development and function of the fetal adrenal. Rev Endocr Metab Disord. 2023;24:5-21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 14. | Ohkuma K, Ono T, Oshima Y, So K, Tsumura K, Yamasaki F, Nakura Y, Yanagihara I, Nomiyama M, Yokoyama M. Diagnostic accuracy of amniotic fluid interleukin-6 for fetal inflammatory response syndrome. J Obstet Gynaecol Res. 2023;49:1498-1505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Zhang Y, Chen L, Ouyang Y, Wang X, Fu T, Yan G, Liang Z, Chen D. A new classification method for gestational diabetes mellitus: a study on the relationship between abnormal blood glucose values at different time points in oral glucose tolerance test and adverse maternal and neonatal outcomes in pregnant women with gestational diabetes mellitus. AJOG Glob Rep. 2024;4:100390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 16. | Suda-Całus M, Dąbrowska K, Gulczyńska E. Infant of a diabetic mother: clinical presentation, diagnosis and treatment. Pediatr Endocrinol Diabetes Metab. 2024;30:36-41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 17. | Arvanitidou O, Kosmas I, Michalopoulos CK, Doumanidou M, Ierodiakonou-Benou I, Athanasiadis A, Daniilidis A. The Impact of Stress and Depression on the Outcome of Human Gestation. Cureus. 2023;15:e48700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 18. | Garay SM, Savory KA, Sumption LA, Penketh RJA, Jones IR, Janssen AB, John RM. Seasonal variation in salivary cortisol but not symptoms of depression and trait anxiety in pregnant women undergoing an elective caesarean section. Psychoneuroendocrinology. 2019;108:14-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 19. | Zhao C, He L, Li L, Deng F, Zhang M, Wang C, Qiu J, Gao Q. Prenatal glucocorticoids exposure and adverse cardiovascular effects in offspring. Front Endocrinol (Lausanne). 2024;15:1430334. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 20. | Duroux M, Stuijfzand S, Sandoz V, Horsch A. Investigating prenatal perceived support as protective factor against adverse birth outcomes: a community cohort study. J Reprod Infant Psychol. 2023;41:289-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 21. | İpek Aytok G, Akkaya Fırat A, Mungan Akın İ, Özel A. How does maternal anemia affect the levels of umbilical cord brain-derived neurotrophic factor? Eur J Obstet Gynecol Reprod Biol. 2024;297:182-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Liu H, Zhu BB, Tao FB, Shao SS. [The role of methylation of placental glucocorticoid response gene in the association between pregnancy-related anxiety in the third trimester and birth outcomes]. Zhonghua Yu Fang Yi Xue Za Zhi. 2023;57:208-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 23. | Scorza P, Duarte CS, Lee S, Wu H, Posner J, Baccarelli A, Monk C. Stage 2 Registered Report: Epigenetic Intergenerational Transmission: Mothers' Adverse Childhood Experiences and DNA Methylation. J Am Acad Child Adolesc Psychiatry. 2023;62:1110-1122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 24. | Jiang N, Zhou M, Le Y, Xiao L, Zhang C, Li S, Guo J, Wu Y, Zhang M, Wang X. The Effect of FPR2 on the Regulation of Trophoblast Autophagy via the PI3K/AKT/mTOR Signaling Pathway in Preeclampsia. FASEB J. 2025;39:e70697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/