Published online Mar 19, 2026. doi: 10.5498/wjp.v16.i3.114301
Revised: October 22, 2025
Accepted: November 7, 2025
Published online: March 19, 2026
Processing time: 165 Days and 19 Hours
Circadian disruption is not merely a common symptom of bipolar disorder (BD) but also serves as a pivotal driver in its pathophysiology. Grounded in the “gene-brain-behavior-environment” multidimensional interaction model, this review established an integrative framework to examine recent advances across mole
Core Tip: Based on the multifaceted role of circadian rhythm disruption in bipolar disorder, this review explored its un
- Citation: Liu Y, Zhu LY, Xiao Q, Zeng HM, Zhan YX, Yang RH, Lin FZ, Liu DL, Zeng XX, Chen BF. Circadian rhythm disruption in bipolar disorder: Mechanisms, clinical significance, and rhythm-oriented interventions. World J Psychiatry 2026; 16(3): 114301
- URL: https://www.wjgnet.com/2220-3206/full/v16/i3/114301.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i3.114301
Bipolar disorder (BD) is a mood disorder with high rates of recurrence and disability and is characterized by alternating episodes of depression and mania or hypomania[1]. Beyond these core symptoms BD leads to broad functional impairments across multiple dimensions, including declines in mental health, cognitive dysfunction, and occupational and social difficulties[2,3]. At the same time BD markedly elevates the risk of premature death. Life expectancy is shortened by 12-14 years compared with the general population, the suicide rate is approximately 64 times higher, and mortality from cardiovascular disease and the prevalence of metabolic disorders are significantly increased[4].
Globally, the disease burden of BD continues to rise. The Global Burden of Disease study reported that globally the number of new cases of BD increased from 3.06 million to 4.53 million between 1990 and 2017, representing a 47.74% growth. Meanwhile, BD accounted for 9.29 million disability-adjusted life years. These figures indicate a continuous rise in the number of incident BD cases and underscore that BD has become a major public health challenge[5]. Current treatments for BD are diverse, encompassing both pharmacological and psychosocial strategies. Nevertheless, its core pathophysiology remains incompletely defined, and treatment responses vary substantially across individuals. Achieving sustained favorable outcomes remains challenging and is reflected in limited efficacy, prominent adverse effects, and complex treatment regimens[6-8].
Growing evidence indicates that circadian rhythm disruption may be a common clinical manifestation of BD as well as one of its core pathophysiological mechanisms[9,10]. We propose that rhythm dysregulation plays a causal role in the development and progression of BD rather than being a mere epiphenomenon. On this basis we propose a multidimensional interactive model that integrates gene, brain, behavior, and environment to explain how the circadian system through dynamic interactions with genetic susceptibility, neural circuitry function, behavioral patterns, and environmental factors drives the disease course of BD.
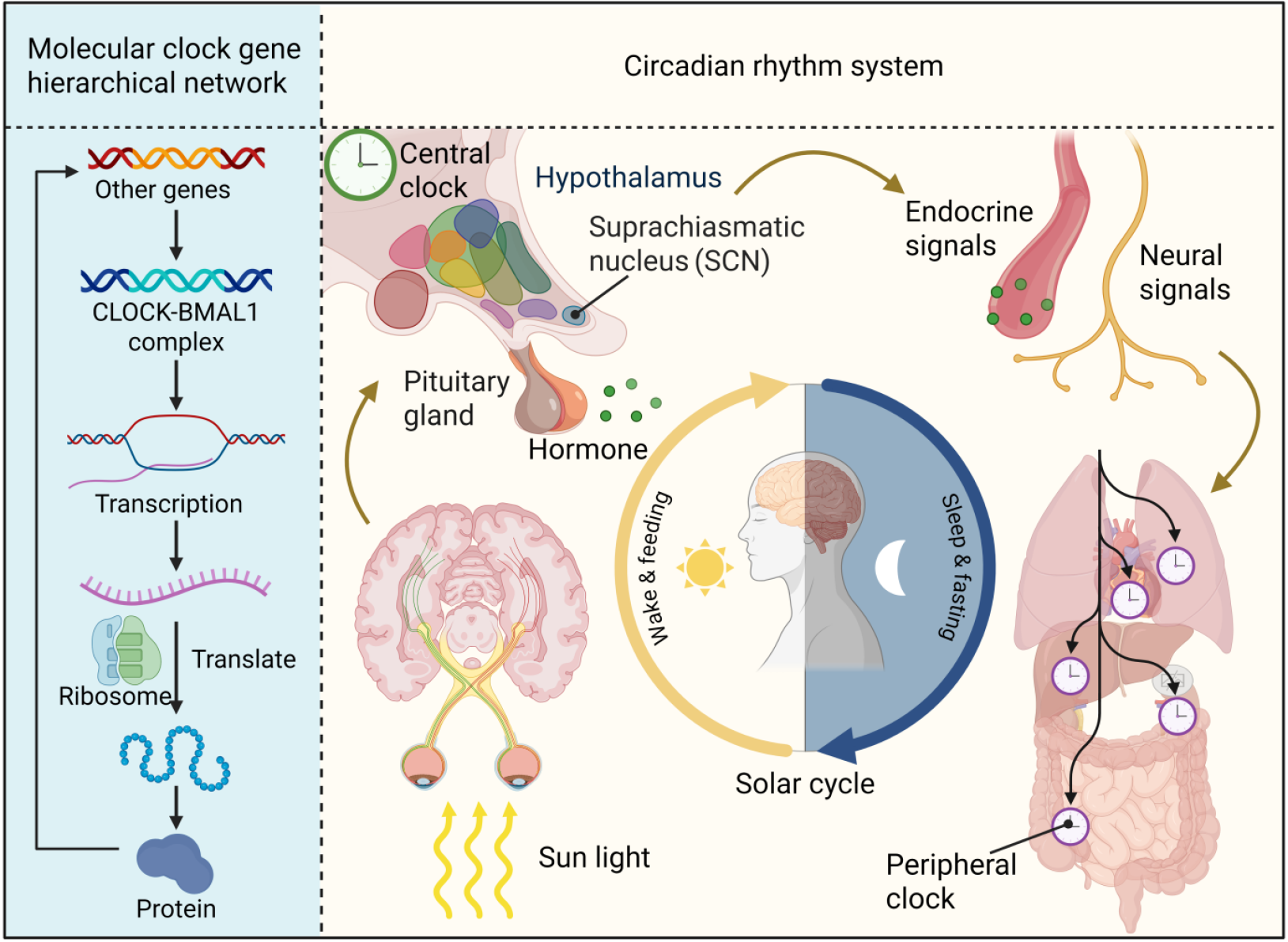
The circadian system is the key physiological mechanism that maintains synchrony between internal rhythms and the external environment. Its central structure is the suprachiasmatic nucleus (SCN) of the hypothalamus, which functions as the master pacemaker. SCN neurons possess intrinsic, cell-autonomous oscillatory capacity, and distinctive coupling mechanisms that sustain strong internal synchrony, thereby coordinating physiological and behavioral rhythms throughout the body[11,12]. At the molecular level the rhythm is driven by transcription and translation feedback loops formed by core clock genes such as CLOCK and BMAL1, creating a complex hierarchical network that underpins endogenous near 24-h oscillations[13-15]. The SCN receives photic input via the retinohypothalamic tract, adjusts the phase and period of the biological clock accordingly[16,17], and relays temporal information to peripheral tissues through endocrine and neural signals to ensure body wide rhythmic synchrony[18,19] (Figure 1). This precise, multilayered regulatory architecture provides essential physiological grounding for understanding the central role of circadian dis
Despite frequent reports linking circadian disruption with BD, a cross-scale, systematic theoretical framework that treats it as a core pathophysiological mechanism is still lacking. A comprehensive synthesis and appraisal of this field is therefore needed to clarify the role of circadian dysregulation in BD and to open new perspectives for mechanistic res
This review identified circadian disruption as a core pathophysiological mechanism of BD from a novel perspective and innovatively proposed a forward-looking framework that advances understanding of mechanisms, clinical signi
Single nucleotide polymorphisms (SNPs) and abnormalities in the expression or function of core clock genes such as CLOCK, BMAL1, PER, and CRY are considered important biological contributors to the pathogenesis of BD. Substantial evidence indicates that variants in core clock genes including CLOCK, BMAL1 (ARNTL), the PER family (PER1, PER2, PER3), the CRY family (CRY1, CRY2), REV-ERBα (NR1D1), retinoid-related orphan receptor (ROR) α, TIMELESS, and CSNK1E are closely associated with BD phenotypes, disease course, and treatment response[20,21]. In humans the circadian cycle begins with dimerization of the positive regulators CLOCK and BMAL1, and their expression depends on the binding of enhancer sequences within the promoter regions of the negative regulators PER (PER1, PER2, PER3) and CRY (CRY1, CRY2). ROR and REV-ERB form an interconnected feedback loop that competes for ROR response elements on BMAL1 to regulate oscillator stability. Once the negative regulators PER and CRY proteins accumulate to sufficient levels, they suppress BMAL1/CLOCK-mediated transcription[22,23]. This section reviewed key variants in these core clock genes and the corresponding evidence in BD.
CLOCK encodes a principal positive regulator of the circadian clock, and its variants are closely linked to BD pathogenesis and clinical features. Animal models provide critical evidence: Mice carrying a deletion in exon 19 of CLOCK, a dominant-negative mutation, exhibit an extended circadian period, sleep disturbances, hyperactivity, mania-like and depression-like behaviors, enhanced cocaine reward, increased excitability of dopaminergic neurons, and metabolic dysregulation, which together model multiple core features of BD[24]. In humans several SNPs have been identified in CLOCK among which rs1801260 (3111T/C) has been most extensively studied. Patients with BD carrying the rs1801260C allele show significantly higher lifetime recurrence, poorer response to treatment for sleep disturbances, more frequent reports of insomnia during depressive episodes, and greater depressive severity[25].
BMAL1 (ARNTL), the essential dimerization partner of CLOCK, also harbors SNPs associated with BD susceptibility and clinical presentation. Multiple studies have linked BMAL1 SNPs to BD susceptibility and to lithium treatment response. For example, Maciukiewicz et al[26] reported that rs11022778 is associated with reduced appetite in patients with BD. Rybakowski et al[27] and Rybakowski et al[28] found that rs11824092 and other BMAL1 SNPs are significantly associated with the prophylactic efficacy of lithium, a relationship that may reflect the role of the gene in BD vulnerability. In addition, epigenetic analyses indicate BMAL1 hypermethylation in BD, suggesting dysregulated expression[29]. Although not all studies consistently show differences in BMAL1 expression between patients and controls, its expression varies markedly across mood states with lower levels during depressive episodes. BMAL1 expression also correlates strongly with monoamine oxidase A, underscoring its role in neurotransmitter regulation and mood stabilization[30]. Collectively, these findings support BMAL1 as a key clock gene involved in the pathophysiology of BD although vali
The G allele at PER2 rs2304672 significantly increases the risk of BD, whereas the 5/5 repeat genotype of the PER3 variable number of tandem repeats shows a protective effect. Carriers display a higher proportion of hypomanic sym
| Gene | Type | Variation | Effect | Strength |
| CLOCK | Core clock gene | ClockΔ19, SNPs (rs1801260C) | Simulated several core characteristics of BD | Highly correlated |
| BMAL1 | SNPs, hypermethylation | Increased the susceptibility of BD and participated in shaping its clinical phenotype | Highly correlated | |
| PER2 | SNPs (rs2304672G) | Significantly increased the risk of BD | Significant correlation | |
| PER3 VNTR | 5/5 repeat genotype | Reduced the risk of BD | Significant correlation | |
| CRY1 | R263Q mutation, risk allele | Caused abnormal circadian rhythm pattern and affected BD | Significant correlation | |
| REV-ERBα | Downregulated expression | Induced extensive apoptosis of NPC in patients with BD | Significant correlation | |
| AKAP11 | Other genes | Heterozygous mutation | Significantly increased the risk of BD | Correlation |
Rhythmic abnormalities in neurotransmitters: As the central pacemaker of circadian rhythms, SCN regulates sleep, wakefulness, body temperature, and cortisol secretion. Its dysfunction is viewed as a core hub for biological rhythm disruption and mood dysregulation in BD. The first manifestation of impaired SCN pacing is abnormal neurotransmitter rhythmicity. Circadian transcription of tyrosine hydroxylase, the rate limiting enzyme for dopamine synthesis, is driven by CLOCK and BMAL1. SCN dysrhythmia shifts the phase of the daytime peak of striatal dopamine, precipitating reward hyperresponsivity during mania, anhedonia during depression, and fragmentation of motor rhythms[35,36]. Notably, dopamine and dopamine receptors, especially D1 like dopamine receptors, can in turn modulate SCN neuronal oscill
The 5-hydroxytryptamine (5-HT) system is also regulated by the SCN. Serotonergic projections from the dorsal raphe nucleus (DRN) to the SCN form an anatomical bridge between circadian timing and mood control. When SCN pacing is disrupted, DRN 5-HT neurons lose circadian drive, leading to excessive nocturnal release and insufficient daytime secretion. This pattern contributes to delayed sleep onset and morning low mood in BD[38]. Irregular light schedules cause phase shifts in the SCN together with phase shifts and lower median levels in DRN 5-HT expression. Mice exhibit delayed sleep onset and morning depression-like behavior, and the severity of depressive behavior correlates positively with the duration of DRN 5-HT rhythm disruption[39]. A study in Science further showed that primary cilia in the SCN regulate oscillations of several core clock genes and neurotransmitters by rhythmically activating the Hedgehog pathway[40]. These findings suggest that abnormalities in primary cilia-mediated signaling may represent a new mechanism linking SCN pacemaker dysfunction to 5-HT dysregulation.
In recent years the focus has expanded from monoamines to inhibitory neurotransmission. Beyond monoamines the role of gamma aminobutyric acid (GABA)-ergic signaling has garnered increasing attention. The impact of SCN pacemaker dysfunction on GABA has become a key topic. GABA is widely expressed in SCN neurons. Although its intracellular actions are debated, the prevailing view is inhibitory[41]. Anatomical and electrophysiological evidence shows a monosynaptic GABA-ergic projection from the SCN to the paraventricular nucleus of the hypothalamus. Optogenetic stimulation of the SCN evokes fast inhibitory postsynaptic currents in the paraventricular nucleus that are mediated by GABA-A receptors, confirming a critical role for this pathway in circadian output control[11,42]. When SCN function is compromised, weakened rhythmic GABA-ergic output produces a glutamate to GABA imbalance, leading to excitation to inhibition imbalance in corticolimbic circuits and amplifying mood polarity in BD[43].
Dysregulation of the hypothalamic-pituitary-adrenal axis rhythm: The hypothalamic-pituitary-adrenal (HPA) axis is a core neuroendocrine stress pathway under SCN control and normally exhibits a cortisol rhythm with higher levels in the morning and lower levels at night[44]. In shift work models chronic nocturnal light exposure misaligns the SCN with the light and dark cycle, delays the cortisol peak, and reduces amplitude. Light therapy or timed melatonin can resyn
Circadian disruption of mitochondrial energy metabolism: Mitochondrial functions, including fusion and fission dynamics and respiration, show marked circadian rhythmicity and depend on an intact molecular clock[47]. Mito
BD shows pronounced seasonality in symptoms and episodes. Systematic reviews indicate that hospitalizations for depressive episodes peak around the winter solstice when the photoperiod is shortest, whereas the risk of manic episodes is greatest near the spring and autumn equinoxes when the photoperiod changes most rapidly[53]. Photoperiodic signals induce SCN to adjust the expression of core clock genes such as CLOCK, BMAL1, and PER2, which are key components of the transcription and translation feedback loop of the circadian clock. These adjustments in turn precipitate metabolic changes and mood fluctuations[9]. Beyond light-dark cycles viral infections like severe acute respiratory syndrome coronavirus 2 represent a significant environmental inducer of circadian disruption through epigenetic mechanisms. Research indicates that long coronavirus disease involves dysregulation of core clock genes (CLOCK, BMAL1, PER2) and mitochondrial dysfunction, which subsequently trigger epigenetic alterations such as DNA methylation. These modifications mediate the adaptation of circadian gene plasticity to environmental stressors, acting as a critical bridge between infection and persistent biorhythm disorder[54]. These processes highlight the crucial plastic role of epigenetic mechanisms in mediating environmental control over clock genes.
Epigenetic regulation in BD also involves histone modifications in which dynamic oscillations directly reflect the plasticity of clock genes. Histone methylation levels are tightly coupled to transcriptional rhythms across the circadian cycle and dynamically regulate gene activity through bidirectional control by activating and repressive marks. In the activating arm the methyltransferase MLL1 associates with the CLOCK and BMAL1 complex and rhythmically catalyzes H3K4me3 to increase chromatin accessibility, thereby promoting transcription of genes such as Per1. In contrast, the repressive arm involves SUV39H1 and SUV39H2, which catalyze H3K9me2/3, drive heterochromatin formation, and silence gene expression[55]. Unlike the bidirectional nature of methylation, histone acetylation such as H3K9Ac and H3K27Ac primarily marks transcriptional activation by weakening the interaction between histones and DNA, facilitating transcription factor binding[56].
Alterations in circadian clock genes and their expression compromise the timekeeping function of the SCN as the central pacemaker. This dysfunction manifests at the cellular and tissue levels as a diminished capacity for synchronization among SCN neurons and a weakened entrainment signal to peripheral tissues. Consequently, internal desy
Patients with BD show an increased percentage of rapid eye movement sleep during depressive episodes, whereas manic or mixed episodes are characterized by shorter total sleep time, lower sleep efficiency, and longer sleep latency. Even during euthymia the duration and continuity of sleep display greater variability, suggesting that disruption of the sleep and wake cycle may reflect both pathophysiology and phenotypic features of the disorder[57-59]. Genetic evidence supports its validity as a biomarker. A study in Cell reported that familial delayed sleep phase disorder is associated with a dominant variant in the core clock gene CRY1[60]. Recent work further shows that patients with BD commonly exhibit delayed sleep phase together with altered tryptophan metabolism and circadian dysregulation, indicating potential markers for early identification[61].
Under normal conditions the rhythm of core body temperature peaks about 1-2 h before sleep, reaches a trough about 2 h before waking, and then rises steadily during the daytime. Night shift workers and individuals exposed to nocturnal light show reduced amplitude of core body temperature, suppression of the melatonin peak, and poorer sleep quality. These findings establish core body temperature as a key biomarker for assessing circadian disruption[62]. Animal models provide additional insight. Mice with bilateral lesions of the SCN together with conditional knockout of BMAL1 display disruption of the core body temperature rhythm and anxiety and depression-like behaviors[63]. In a chronic jet lag model, female mice and castrated male mice show reduced robustness of the core body temperature rhythm along with weakened rhythmic expression of clock genes in the liver and adrenal gland[64]. These observations indicate that reduced amplitude of the core body temperature rhythm is a potential biomarker for BD.
Cortisol is a circadian sensitive marker governed by the HPA axis and normally shows a morning peak and a nighttime trough[35,62]. A recent prospective study in patients with Cushing syndrome found markedly flattened circadian oscillations of CLOCK, PER1 to PER3, and TIMELESS in peripheral blood mononuclear cells, suggesting that phase shifts of the cortisol rhythm can mark circadian disruption[65]. Melatonin is a key downstream hormone of the circadian system. Through the melatonin (MT) receptors (i.e. MT1 receptor) it regulates the sleep phase, and through the MT2 receptor it calibrates circadian timing[66,67]. Notably, studies of shift working nurses observed preserved melatonin rhythms together with disorganized cortisol rhythms, indicating differential sensitivity of endocrine axes to circadian perturbation[68]. In summary, phase shifts in endocrine rhythms are important indicators for identifying circadian disruption.
The transcription and translation feedback loop that drives circadian timing oscillates autonomously in cells throughout the body and is synchronized by the SCN through neural and humoral pathways[22]. Metabolic disturbances that arise from misalignment between the environment and the central clock are influenced by peripheral clocks. Even under misalignment matching the periods of peripheral and central clocks can mitigate metabolic dysregulation[69]. Recent evidence has increasingly indicated that the failure of this central coordination function leads to desynchronization between the SCN and peripheral oscillators. In patients with attention deficit hyperactivity disorder, the PER1 rhythm in skin fibroblasts is markedly attenuated under norepinephrine stimulation, suggesting that reduced sensitivity of peripheral clocks may be a shared feature in BD[70]. Moreover, peripheral clock genes such as PER2 and BMAL1 can operate independently of SCN regulation, and their aberrant expression or desynchronization of peripheral rhythms disrupts the circadian blood pressure rhythm[71]. Notably, genes including PER2 and BMAL1 are also associated with BD, further suggesting the role of peripheral tissue rhythm desynchronization in BD. These observed disruptions in physiological rhythms are reflected to some extent in a diminished ability of individuals to maintain regular daily behaviors, manifesting as instability in behavioral and social rhythms (Table 2).
| Biomarker | Rhythmic change | Related variant clock gene | BD performance | Ref. |
| Sleep-wake cycle | Sleep phase delayed | CRY1 | Sleep wake cycle CBT rhythm disordered, accompanied by changes in tryptophan metabolism and circadian rhythm disorder | Marchetti et al[57]; Panchal et al[58]; Scott and McClung[59]; Patke et al[60]; Yavuz Ataşlar and Altınbaş[61] |
| CBT | Decreased rhythm amplitude | BMAL1 | CBT rhythm disordered | National Toxicology Program[62]; Liang et al[63]; Ma et al[64] |
| Cortisol | Rhythmic phase shifted | CLOCK, PER1, PER2, PER3, TIMELESS | Circadian rhythm disordered | Rathor and Ch[35]; National Toxicology Program[62]; Hasenmajer et al[65] |
| Melatonin | Differed in rhythm phase shift and rhythm interference sensitivity | Circadian rhythm disordered | Yang et al[68] | |
| Peripheral clock | Reduced sensitivity and rhythm desynchronization | PER2, BMAL1 | SCN level control is broken, and the whole-body rhythm is not synchronized | Woodie et al[69]; Palm et al[70]; Costello and Gumz[71] |
The etiology of BD is complex and may involve genetic susceptibility[72-74] and social and psychological factors[75]. Prior work indicates that dysfunction of social rhythms may be related to the development of the disorder. In a study of children at familial high risk, Veddum et al[76] found broadly impaired social responsivity, and such deficits were detectable early in life. Historical data converge on similar conclusions. Retrospective and longitudinal studies of adults with BD suggest that social impairment is already present during childhood and adolescence before illness onset[77]. These findings imply that reduced stability of social rhythms may play a role in the course of BD and underscore the importance of preventive interventions tailored to children with poor social competence. In patients with BD Walsh et al[78] observed greater irregularity of social rhythms. Cross sectional studies consistently report a robust association between social rhythm disruption and bipolar spectrum disorder, and longitudinal studies suggest that social rhythm dysregulation predicts mood symptoms[79,80].
The social rhythm metric (SRM) is a valid and reliable self-report tool that quantifies the regularity of daily habitual behaviors and interactions[81]. The SRM index reflects the regularity of an individual’s life, and higher scores indicate more consistent social rhythms[82]. Using the modified SRM, Shen et al[83] examined the predictive value of social rhythm regularity for time to mood episode in late adolescent and young adult participants with bipolar spectrum conditions. Participants with bipolar spectrum conditions reported lower social rhythm regularity than healthy controls, and social rhythm regularity prospectively predicted survival time to a mood episode.
External environmental factors such as artificial light have been shown to cause sleep disturbances and to affect the timing system of circadian rhythms and other intrinsic rhythms that align human behavior with daily changes in light and with seasonal cues[84,85]. Abnormal light exposure patterns, including increased blue light at night and insufficient daytime light, may impair photic entrainment pathways, increase rhythm instability, and may be involved in the occurrence of BD[86-88]. Several lines of evidence support this view. Nighttime blue light exposure can directly influence mood and cognition. In mice LeGates et al[89] demonstrated that abnormal light cycles increased depression-like behavior and impaired learning, and these effects were mediated by intrinsically photosensitive retinal ganglion cells. In humans nighttime blue light exposure correlates with more depressive symptoms, likely through suppression of melatonin secretion[90]. Nighttime light also disrupts physiological rhythms. It elevates cortisol levels and alters the expression of circadian clock genes[91], and it may enhance systemic inflammation by disturbing the circadian rhythmicity of inflammatory markers[92]. These physiological changes can further exacerbate mood instability and the risk of BD.
In patients with BD altered light exposure is closely linked to mood fluctuation. Nighttime light exposure is associated with the occurrence of manic symptoms, whereas insufficient daytime light may contribute to relapse of depressive episodes[93,94]. Circadian disruption is common in BD and may be driven by abnormal light exposure patterns[95]. Adjusting light exposure by increasing daylight and reducing nocturnal light may be an important strategy to improve mood stability in patients.
The bidirectional worsening mechanism that links behavior and biological rhythms is complex. Irregular behavior can weaken the timing function of the SCN, and this process is closely related to the formation of BD. Multiple studies indicate that circadian disruption is involved in onset, progression, outcomes, and relapse and that dysfunction of the circadian system may partly cause the disorder[96]. In mice subjected to repeated inversions of the light dark cycle, simulating irregular schedules during adolescence, Gallego-Landin et al[97] observed disrupted PER2 rhythms in the hypothalamus that contains the SCN, and similar results were reported by Leise et al[98]. Ortiz et al[99] further proposed that changes in daily activity were among the earliest indicators of depressive symptoms in patients with BD. These findings suggest that behavioral and rhythm irregularities can weaken SCN timing. The resulting dysfunction promotes the development of BD. At present mood lability and impaired emotion regulation are recognized features of the disorder[100]. Numerous studies show that, relative to the premorbid state, patients exhibit more unstable mood and more disorganized behavior[101,102]. Thus, irregular behaviors disrupt biological rhythms, which in turn exacerbate beh
Taken together, the interaction between behavioral irregularity and circadian disruption may form a vicious cycle. In the context of BD, circadian disruption is not only a consequence of worsening illness but may also be an etiological factor. This bidirectional worsening highlights the need to prioritize circadian regulation in both treatment and pre
For patients with psychiatric disorders, especially those with BD, relapse represents a challenging treatment failure. It can be defined as becoming ill again after clear recovery followed by clinical deterioration[103]. Many studies show that relapse rates are high, approximately 71%, and the risk relates to the psychosocial context of disease, environment, deve
Life events, particularly stressful ones, are widely recognized as triggers of BD[109]. Recent work by Cusell and Kok[110] confirmed this view. In older patients followed for 5 years, relapse or recurrence was common, and about half of the episodes were preceded by a life event. Koenders et al[111] found that negative life events are strongly associated with subsequent severity of manic and depressive symptoms, whereas positive life events precede manic symptoms only. Although methods for distinguishing whether life events are causes or consequences of mood symptoms have limi
Social jetlag, defined as misalignment between sleep times on workdays and weekends, is a specific form of sleep disruption. Late bedtimes and wake times on weekends with early rising on workdays can create misalignment that affects circadian rhythms[112]. Smith et al[113] reported that individuals with bipolar spectrum disorder may be more vulnerable to the adverse effects of social jetlag, which is associated with greater depressive symptoms in patients and in those at risk. These observations suggest that social jetlag may act as a relapse trigger. Future research should clarify its role in onset, progression, and prognosis. The destabilization of rhythms and their persistent negative interaction with the environment ultimately manifest as clinical features that impact disease progression, prognosis, and treatment.
Circadian rhythm disruption is closely linked to the pathophysiology of BD. It modulates daily feeding behavior[114], regulation of the sleep and wake cycle[115], neuroendocrine hormone release[116], and homeostatic and metabolic processes[117]. Dysregulation of these functions is among the most common clinical features in patients with BD[118]. During manic or depressive episodes, characteristic rhythm disturbances are prominent. Sleep problems typically present as reduced sleep need during mania and near daily insomnia or hypersomnia during depression, which can affect emotion regulation and the course of illness[119]. Circadian disruption is also associated with suicide risk, and this association may vary across bipolar subtypes[120].
Although treatment has advanced, the risk of relapse remains high[104]. The role of circadian disruption in BD has been widely investigated, and many studies suggest it may serve as a core symptom dimension or a predictor of relapse[121]. In a 48-week prospective study, Takaesu et al[122] found that circadian rhythm sleep wake disorders, primarily delayed sleep wake phase disorder, may be important predictors of relapse in patients with BD. Cretu et al[123] further showed that in remitted patients, poor sleep quality was associated with residual mood symptoms and independently predicted recurrence of mood episodes. Taken together, indices of rhythm stability are likely to serve as early warning markers of relapse risk, and circadian disruption may promote mood instability and increased relapse risk in BD[124].
Since approval in the 1970s, lithium has remained the gold standard for treatment, and its regulatory effects on circadian rhythms have received extensive attention[125]. Lithium can influence treatment response by modulating the expression of clock genes[126]. In particular, lithium affects expression of genes such as NR1D1, which may serve as biomarkers of therapeutic response[127]. The impact of lithium on circadian rhythms appears to be closely related to its clinical efficacy, especially in improving mood symptoms[128,129].
In mixed states, rapid cycling, and in the presence of comorbid anxiety or substance use disorders, the efficacy of lithium may be limited. In such cases valproic acid (VPA) can be used alone or in combination with lithium as an effective alternative[130]. The effects of VPA on clock gene expression have been studied extensively. For example, Griggs et al[131] reported that VPA altered expression of genes in both the positive limb (BMAL1) and the negative limb (CRY1 and CRY2) of the clock with effects depending on dosing time at circadian time 0 vs circadian time 12. By altering the oscillatory patterns of core clock transcription factors, VPA modulates circadian function[131]. In line with these findings, Ferraro et al[132] observed abnormal BMAL1 expression patterns in the SCN of both male and female mice exposed to VPA, suggesting changes to core clock mechanisms[132]. Landgraf et al[133] provided in vivo and in vitro cross species evidence that VPA shortens the circadian period in fibroblasts from patients with BD and from controls. These results suggest that VPA improves mood in patients with BD by normalizing a prolonged circadian period. This condition appears to stem from reduced dopamine transporter function and consequent elevated dopamine levels. By correcting this period the treatment helps synchronize the patients’ internal rhythm with the external 24-h light-dark cycle[133]. Thus, VPA related clock effects may serve as biomarkers of treatment response.
Circadian disruption not only shapes symptom expression but also interacts with comorbid insomnia and circadian rhythm sleep disorders (CRSD), thereby influencing prognosis[134,135]. Circadian dysregulation correlates strongly with insomnia severity and may exacerbate symptoms by impairing mood and cognitive function[136]. In a prospective cohort study by Bertrand et al[137], insomnia in patients with BD complemented depressive and anxiety symptoms as a potential signal of suicidal ideation and influenced prognosis.
In mammals sleep is tightly regulated by the circadian system. Misalignment between internal timing and the external environment, such as shift work, jet lag, or nocturnal light exposure, can lead to CRSD[138]. Findings by Liu et al[139] indicate that such disorders are associated with mutations in related genes including PER2, CRY1, CRY2, and PER3. As noted earlier these genes belong to the core clock gene set implicated in BD. These observations indirectly suggest that comorbid CRSD in BD may produce bidirectional amplification that worsens prognosis.
At present pharmacologic approaches are recommended as first line therapies for BD with lithium monotherapy and combination regimens being the mainstays[140-142]. These approaches are not invariably effective[143], and patient acceptance varies[144]. This variability increases the need for multidisciplinary treatment that includes psychosocial and behavioral interventions[145,146]. Interpersonal and social rhythm therapy (IPSRT) is an evidence-based psychotherapy that targets disrupted social rhythms by integrating the core elements of interpersonal psychotherapy for unipolar depression with SRT[147]. IPSRT is applicable to the acute treatment of bipolar depression and to the prevention of mood episodes[148,149].
A randomized controlled trial (RCT) in 2020 suggested that IPSRT may be clinically effective for major depressive disorder (MDD)[150]. Using data from clinical trials, Moot et al[151] found that compared with treatment as usual IPSRT produced positive effects over 18 months on two subscales of the social adjustment scale self-report, indicating beneficial effects of IPSRT. In a study of 38 older outpatients with a mean age of 65.4 ± 10.0 years who had MDD or BD, twice weekly group-based IPSRT was feasible and acceptable[152]. In a clinical effectiveness trial, Crowe et al[153] reported functional improvement although the combination of IPSRT and medication management for more than 18 months did not significantly reduce mood relapse.
Recently, Swartz et al[154] innovated on IPSRT by developing an online IPSRT-based intervention for BD called rhythms and you (RAY). A randomized pilot study showed that remote delivery of RAY was feasible and acceptable, and time spent on RAY correlated positively with improvement in social rhythm regularity[154]. By contrast, an RCT by Douglas et al[155] found that combining IPSRT with cognitive remediation may attenuate the effect of IPSRT when targeting symptoms and cognitive or functional recovery simultaneously. These findings indicate that further comprehensive research is needed to evaluate the short-term and long-term efficacy of IPSRT and the effectiveness of combinations with other therapies.
SRT is a component of IPSRT that focuses on behavioral strategies. It has been tested separately in comprehensive interventions for MDD[156] and for BD[154]. Among patients with BD aged 15-35 years, the SRT component of IPSRT has been shown to be most helpful for maintenance over more than 5 years[157]. Crowe et al[158] found that SRT has multiple features that can be translated into clinical practice across different settings, showing consistency with recent findings by Darwish et al[159]. Sankar et al[160] delivered a modified SRT remotely to 13 adolescents and young adults with BD and showed that telehealth-based SRT was feasible and acceptable and could reduce mood symptoms and suicide risk. These results suggest that SRT stabilizes daily activity anchors and rebuilds zeitgeber signals. It is a promising adjunct for future recommendations in the treatment of BD.
Bright light therapy (BLT) has developed over the past 50 years and has proven to be highly versatile[161]. It is widely used to treat seasonal affective disorder[162] and depressive symptoms in non-seasonal MDD[163]. Bright light stimulates intrinsically photosensitive retinal ganglion cells and induces a cascade of changes that involve melatonergic, neurotrophic, GABAergic, glutamatergic, noradrenergic, and serotonergic systems as well as the HPA axis. Although the exact mechanism remains incompletely understood, bright light is thought to modulate circadian dysfunction and autonomic function and thereby improve mood symptoms[164]. The International Society for BDs task force recommends BLT for acute treatment of bipolar depression[165].
An increasing number of RCTs has examined the efficacy of BLT for depressive symptoms in BD with most showing benefit[166,167]. In contrast, a meta-analysis by Takeshima et al[168] that included six RCTs reported that BLT did not significantly improve depressive symptoms in BD. Given the high heterogeneity and small number of trials, these results are not definitive. Larger studies with appropriate control groups are needed to determine efficacy and safety. A recent dose escalation phase 1 and 2 randomized double blind trial by Geoffroy et al[169] further confirmed that BLT is a feasible antidepressive strategy in BD.
In a cohort of 799 patients with BD receiving antidepressant light therapy, Benedetti[170] observed that the highest reported rate of switch to mania after light therapy approximated the expected 4% switch rate during placebo treatment in BD. This finding suggests no specific concern about manic switch with light therapy, and the switch rate was unrelated to treatment parameters that included light intensity, duration, and circadian timing. This potential risk is theorized to stem from a mechanism shared with many antidepressants: Increased serotonergic activity[171,172] and use of BLT without a mood stabilizer may increase the risk of a manic switch.
Abnormalities in melatonin secretion and regulation have been documented in unipolar and bipolar depression[116]. In BD these abnormalities may reflect reduced pineal secretion or dysregulated secretion due to internal or external causes[173,174]. Studies have begun to examine whether melatonin receptor agonists such as ramelteon and agomelatine can serve as adjunctive therapies for BD[175]. A 2022 systematic review and meta-analysis that included 11 studies, 6 RCTs and 5 feasibility studies, reported only partial evidence that ramelteon prevented recurrent depression in BD[176]. Agomelatine is both a melatonin receptor agonist and a 5-HT2C receptor antagonist. The two mechanisms act synergistically to restore circadian rhythms, improve depressive symptoms, and benefit anxiety, sleep, and sexual function[177].
Agomelatine is an effective antidepressant for MDD. A recent systematic review by Li et al[178] suggested that agomelatine showed significant efficacy and good tolerability in bipolar depression. These studies provide encouraging signals for melatonin receptor agonists as antidepressant therapies while emphasizing the need for rigorously designed trials with large samples to establish definitive efficacy. Reduced sleep need is a core symptom of mania, and insomnia or hypersomnia is a core feature of depression. Sleep disruption is a hallmark of BD[179]. These features are crucial for dia
Sleep deprivation (SD), also called wake therapy, is defined as a period of enforced wakefulness. Since the 1970s it has been considered an effective antidepressant intervention with potential utility for major depressive episodes and can be used alone or combined with medication as part of chronotherapy[182]. The evidence base is largely from noncontrolled clinical trials[183,184] with relatively few RCTs[185], and its true effectiveness remains debated[186]. For example, Sarzetto et al[187] treated 11 inpatients with bipolar depression using three cycles of total SD combined with daily light therapy for 1 week. Polysomnography before and after treatment showed significant improvement in depressive symptoms, supporting a positive effect of the combined chronotherapy for bipolar depression. The composite design, however, makes it difficult to attribute effects to SD alone.
A meta-analysis of rapid antidepressant chronotherapy found benefits that included 5-7 days of SD, but the effect size in randomized trials was smaller than in open label case series with 33% vs 62% of patients classified as responders. This difference suggests that some of the apparent effectiveness of SD may arise from nonspecific factors such as expectancy rather than from true efficacy[188]. In a systematic review and meta-analysis of 29 RCTs, Mitter et al[186] concluded that adding SD to treatment regimens did not enhance antidepressant effects.
More recent work by Gottlieb et al[189] analyzed SD in acute bipolar depression and found a response rate close to 50%, supporting enhanced early efficacy. In line with this conclusion, a systematic review and meta-analysis by Ramirez-Mahaluf et al[190] reported that adding medication to total SD produced positive acute effects with lithium being especially recommended, and higher response rates were maintained at 3 months. Therefore, while SD shows rapid antidepressant effects, its utility as a standalone, sustained treatment is limited, and it may be most effective as part of a combined chronotherapeutic protocol (Table 3).
| Ref. | Year | Type | Medication/therapy | Impact on BD |
| Crowe et al[150] | 2020 | IPSRT | IPSRT | Reduced depressive symptoms |
| Moot et al[151] | 2022 | IPSRT within 18 months | Improved social adaptation | |
| Orhan et al[152] | 2024 | Twice weekly group IPSRT | Feasibility demonstrated | |
| Crowe et al[153] | 2020 | IPSRT exceeding 18 months | No significant effect on relapse | |
| Swartz et al[154] | 2021 | Online intervention of BD based on IPSRT | Improved social rhythm regularity | |
| Douglas et al[155] | 2022 | Combining IPSRT and CR | Reduced the effectiveness of IPSRT | |
| Crowe et al[158] | 2020 | SRT | SRT | Feasibility demonstrated in clinical practice |
| Sankar et al[160] | 2021 | Improved SRT | Reduced emotional symptoms and suicide risk | |
| Takeshima et al[168] | 2020 | BLT | BLT | No significant improvement in depressive symptoms |
| Geoffroy et al[169] | 2025 | BLT | Reduced depressive symptoms | |
| Benedetti[170] | 2018 | BLT | Found the phase change rate of mania was not related to the treatment method | |
| McGowan et al[176] | 2022 | Melatonin receptor agonist | Ramelteon | Prevented recurrent depressive symptoms in patients with BD |
| Li et al[178] | 2024 | Agomelatine | Significant therapeutic effect and good tolerability Excellent treatment and tolerance | |
| Sarzetto et al[187] | 2022 | SD | 3 cycles of TSD + daily one week BLT | Had a positive therapeutic effect on BD depressive episodes |
| Humpston et al[188] | 2020 | SD | Not the actual therapeutic effect | |
| Mitter et al[186] | 2022 | SD | Unable to prove the effect of antidepressant treatment | |
| Gottlieb et al[189] | 2021 | SD | Enhanced early efficacy | |
| Ramirez-Mahaluf et al[190] | 2020 | TSD + drug therapy (such as lithium) | Had a positive impact on acute reactions |
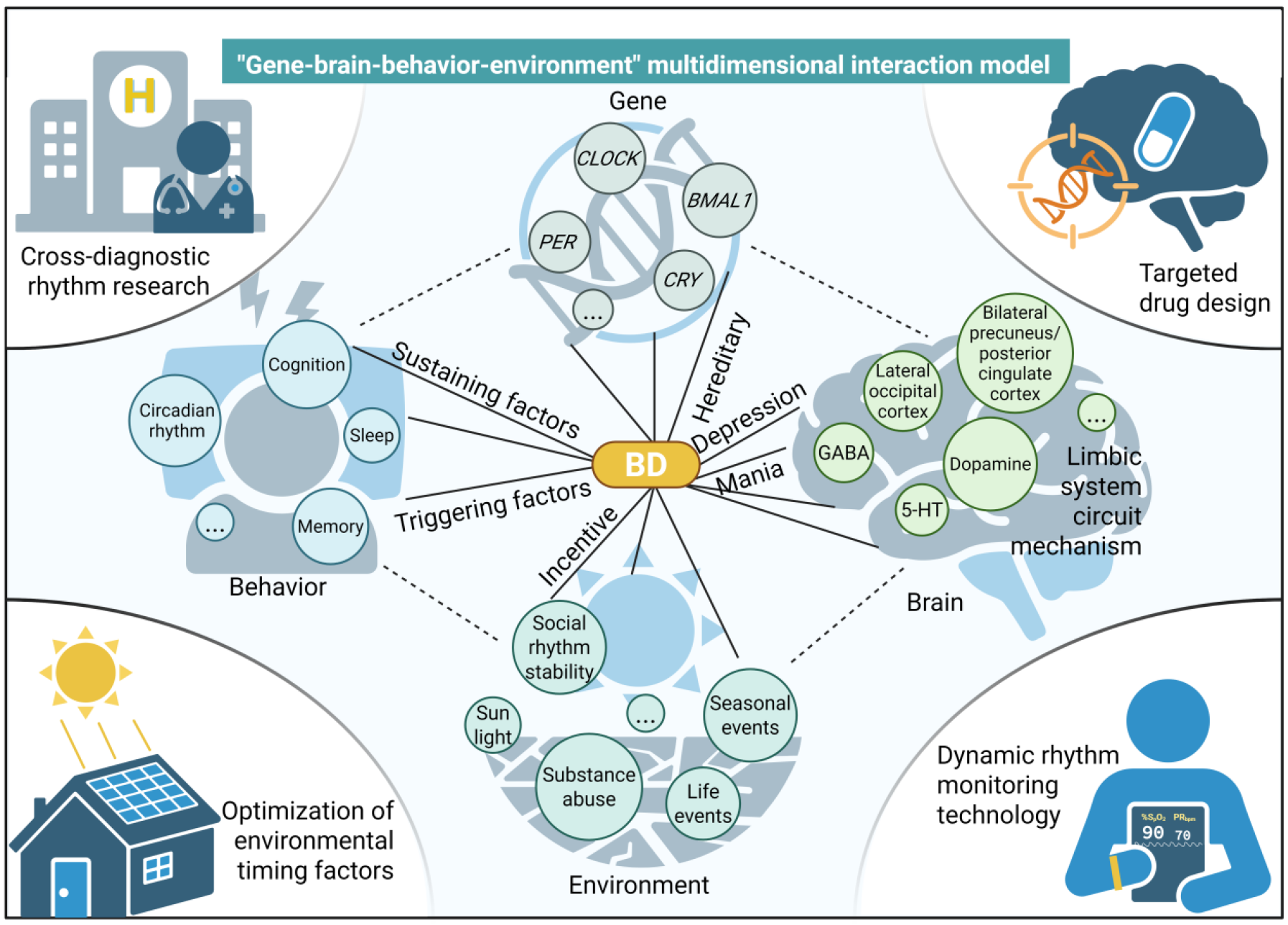
When dissecting pathways involved in the onset and progression of BD, researchers often focus on the complex relationships among gene expression, brain function, and observable behavior. By integrating multiple methods such as functional magnetic resonance imaging and genomic analyses, the pathophysiology of the disorder can be understood more comprehensively. At the level of clock genes, BD shows substantial heritability[191], and multiple studies have identified evidence for variants and expression abnormalities in core clock genes associated with the disorder[192], including SNPs in CLOCK[25], BMAL1[193], PER[194], and CRY[194]. These findings highlight the importance of altered gene expression and provide direction for models that trace disease development in depth.
At the brain level patients show marked functional alterations across clinical stages that are closely related to heterogeneity in gene expression. For example, a functional magnetic resonance imaging study reported significantly increased regional homogeneity in the bilateral precuneus and posterior cingulate cortex as well as the lateral occipital cortex, and the connectivity features of these regions were closely linked to clinical symptoms[195]. The spatial correspondence between these imaging features and gene expression suggests that biological pathways involving limbic circuitry may operate at different stages of the disorder.
Multiple studies also indicate rhythmic abnormalities in neurotransmission during the course of the illness. Disrupted oscillations of dopamine, serotonin, and GABA can perturb limbic circuits, produce manic and depressive symptoms, and profoundly influence cognition, memory, and the regulation of circadian timing and other behaviors[196]. The envir
Manic, depressive, and mixed states are the core observable symptom clusters and represent the final outputs of gene and brain abnormalities under specific environmental conditions. Circadian and sleep disturbances are not only core symptoms but also important triggers and maintaining factors[121], and they interact closely with brain function and environmental cues[138]. Epidemiologic evidence shows that individuals with genetic vulnerability, such as children of mothers with BD, display marked fragility in behavioral phenotypes related to circadian regulation. In the pathway from maternal diagnosis to offspring emotional and behavioral problems, as much as 75% of the effect is mediated by the child’s own circadian disruption[197]. This finding underscores the deep interaction between genetic susceptibility and behavior.
Genes, brain, behavior, and environment all contribute to onset and progression to varying degrees. Their interactions shape relapse, treatment, and prognosis. Different individuals may ultimately develop the disorder through different combinations of gene, brain, behavior, and environment. Building on the preceding discussion, we propose a multidimensional interactive model that integrates gene, brain, behavior, and environment. This model not only reveals potential biological mechanisms but also provides a scientific basis for new therapeutic strategies. Future work should explore these interactions more deeply and comprehensively in order to deliver more effective prevention and intervention for patients (Figure 2).
In applied technology the combination of artificial intelligence and wearable devices offers new possibilities for the critical area of dynamic rhythm monitoring. With artificial intelligence wearables can continuously track physiological parameters and thereby support individualized health management[198]. Several studies show that new real time mobile tools for dynamic rhythm monitoring can derive individualized multilevel dynamical representations of multiple homeostatic systems. These tools can inform clinical research by identifying heterogeneity in presentation and can provide more objective indicators of treatment response in real world settings[199].
Recently, a prospective observational study monitored sleep using wearable devices and assessed emotional symptoms via smartphones in 139 patients with mood disorders (including patients with BD). The study found that 66.7% of patients with MDD and 85.7% of patients with BDI exhibited a causal relationship in which circadian rhythm phase disruption preceded the onset of emotional symptoms. This finding provides evidence for individualized treatments targeting circadian rhythms, such as light intervention[200].
In a randomized sham controlled double blind trial, Yeom et al[201] found that algorithms using daily self-monitoring and feedback through wearables and smartphones predicted mood fluctuations 3 days in advance with accuracy of about 90%-95% when compared with clinician assessments conducted every 3 months. Their earlier positive findings indicated that digital data and circadian parameters can provide useful clinical information, support individualized rhythm phenotyping, and guide subsequent treatment decisions[201]. These conclusions have been supported by additional studies using diverse designs[202]. With these technologies we can achieve precise individualized dynamic monitoring and implement closed-loop interventions (e.g., recommending light exposure based on real-time rhythm data), thereby enabling more accurate medical interventions and providing support for therapies such as precision drug design.
Research on the biological clock has become a key field in modern medicine. The development of novel timing agents such as agonists of REV-ERB and ROR is based on a deep understanding of circadian biology and may offer future treat
Mishra et al[126] compared NPCs from patients and from mice and found that reduced REV-ERBα expression caused more severe cell death in cells from patients than in controls, supporting REV-ERB as a potential therapeutic target for the link between circadian disruption and neuronal loss in the disorder. Elevated risks of premature mortality include nearly doubled cardiovascular mortality and increased prevalence of metabolic disease. The role of REV-ERB in cardiovascular and metabolic disorders has been widely studied[205-208]. For example, Wang et al[209] reported that REV-ERBα protects against atherosclerosis, myocardial infarction and reperfusion injury, and heart failure by regulating myocardial energy metabolism. Raza et al[210] identified REV-ERB as an attractive target that can beneficially affect metabolic dysregulation, inflammation, and fibrosis. Synthetic REV-ERB agonists have been shown to modulate circadian behavior and expression of metabolic genes and to improve sleep disorders, obesity, and metabolic disease[211,212].
Recently, a study conducted by Liu et al[213] has revealed that ROR-β, as a susceptibility gene for BD, exerts an impact on hippocampal neuronal activity and the fluctuations of depressive/mania-like behaviors by regulating the circadian rhythm of pancreatic insulin secretion. Targeting ROR-β (e.g., via the use of its antagonist 4-OA) or modulating its downstream insulin signaling pathways (e.g., phosphatidylinositol 3-kinase-α) can ameliorate circadian rhythm dis
In mammals the cell autonomous molecular clock relies on interconnected transcription and translation feedback loops composed of positive activators CLOCK and BMAL1 and inhibitors PER1 and PER2 and cryptochromes CRY1 and CRY2 that together generate a stable 24-h rhythm. Nuclear receptors of the ROR family regulate transcription of BMAL1 and ensure stability and robustness of rhythms[214]. Recent studies show that the feedback loop formed by ROR and BMAL1 regulates diverse metabolic pathways across organs. He et al[215] demonstrated that the ROR agonist nobiletin, a small molecule that enhances clock amplitude, markedly strengthens circadian rhythms and counters metabolic disease through the circadian gene network[215]. These data suggest that ROR agonists may serve as novel timing agents that target the molecular clock in the disorder. Further studies and drug design efforts are needed.
Environmental factors are widely recognized contributors to the disorder. The condition is closely related to circadian disruption, and pronounced rhythmic behavioral instability can aggravate onset and becomes even more unstable after onset. Growing attention has been paid to optimizing environmental zeitgebers in treatment, especially the design of artificial lighting and community-based rhythm support[216]. In modern society artificial lighting and luminous devices have altered the temporal pattern of human life. Nocturnal light disrupts molecular clock rhythms and produces circadian dysregulation[217,218]. Designed artificial lighting such as BLT can improve mood although long-term effects require further study[169]. Rational lighting that is tailored in real time to the clinical state is therefore essential. These observations indicate that well designed artificial lighting environments may become viable strategies for optimizing environmental zeitgebers as adjuncts to treatment.
Rhythm-oriented interventions discussed earlier, such as SRT and IPSRT, show beneficial effects. By stabilizing daily activities and social rhythms, these therapies help reduce mood fluctuations and relapse risk[158]. Such multilayered strategies have important potential in clinical care. By resynchronizing the biological clock and stabilizing social rhythms, they can improve mood and reduce relapse, offering a comprehensive treatment package. Future studies should evaluate long-term effects and mechanisms to better guide practice.
BD, depression, and schizophrenia are common psychiatric disorders with substantial clinical heterogeneity. These conditions may share transdiagnostic features of rhythm disruption that include disturbances of sleep, biological timing, mood, and cognition. Circadian dysregulation plays an important role in the onset and progression of these disorders and may predict relapse and treatment response[219]. It is also closely associated with mood instability, cognitive impairment, and increased suicide risk[220].
In BD circadian disturbance presents as irregular sleep and wake cycles, an evening chronotype, abnormal melatonin secretion, and susceptibility related to clock genes[221]. These abnormalities are evident during episodes and may persist during euthymia[222]. In mood disorders circadian disruption is tightly linked to reduced functioning and quality of life[136]. In schizophrenia sleep and circadian disturbances are also common and interact in complex ways with alterations in dopamine signaling[223]. Rhythm abnormalities are observed in other psychiatric conditions as well. The link between seasonal affective disorder and BD suggests that the former may serve as a marker of severity of the latter[224].
Among adolescents and college students, circadian disruption is associated with high rates of depression and anxiety, which may relate to pubertal biology, academic stress, and widespread use of electronic devices[225]. Interventions that target circadian disruption, including light therapy, chronotherapy, and cognitive behavioral therapy, may play important roles in improving diagnosis and treatment across these conditions[226]. Future research should continue to clarify the role of circadian rhythms in these disorders in order to develop more personalized and effective therapeutic strategies.
Over the past several decades, circadian disruption has become a research focus because of its broad associations with the course of psychiatric disorders, including BD, depression, and schizophrenia. This review centered on its pivotal regulatory role in BD and analyzed the underlying pathophysiology. Variants in core clock genes such as CLOCK, BMAL1, PER, and CRY produce downstream effects of SCN dysfunction. These effects manifest as biomarkers of circadian disruption, including abnormalities in the sleep and wake cycle, reduced rhythmic amplitude, phase shifts in endocrine rhythms, and desynchronization of rhythms in peripheral tissues, leading to rhythmic disturbances in behavior. At the same time environmental feedback such as abnormal light exposure patterns, life events, and social jetlag intensifies the vicious cycle of behavioral and circadian instability, triggers new mood fluctuations, and contributes to the onset of BD. We reviewed theoretical and clinical studies from roughly the past 15 years on the relationship between circadian disruption and BD to improve understanding of rhythm-oriented interventions and to elucidate its clinical implications for diagnosis, prediction of relapse, and treatment personalization.
In practice, however, these interventions still face many challenges. Evidence for the efficacy of approaches such as BLT and SD relies mainly on noncontrolled and nonrandomized studies or on trials that primarily enrolled participants with unipolar depression. Results may differ when only RCTs are included. In addition, it remains unclear whether combinations of multiple interventions can better stabilize illness and produce synergistic effects that exceed simple additivity. More specifically, research gaps in therapeutic areas are manifested in the need to integrate wearable monitoring into clinical practice and accelerate the development of molecularly targeted drugs in the future. There is little doubt that research on circadian resynchronization will expand rapidly. Such work will help further evaluate and refine multidimensional, integrated, rhythm-oriented interventions for BD.
This review provided a reference for continued study of the multidimensional interactive model that integrates gene, brain, behavior, and environment. It also offers guidance for developing precision, individualized, time-based treatments that target circadian disruption. Ultimately, targeting circadian rhythm disruption represents a paradigm shift from symptomatic management to addressing a core pathophysiological mechanism in BD. We anticipate that circadian resynchronization strategies will inform future applications across diverse diseases and offer new therapeutic directions for psychiatric disorders.
| 1. | Deckersbach T, Nierenberg AA, McInnis MG, Salcedo S, Bernstein EE, Kemp DE, Shelton RC, McElroy SL, Sylvia LG, Kocsis JH, Bobo WV, Friedman ES, Singh V, Tohen M, Bowden CL, Ketter TA, Calabrese JR, Thase ME, Reilly-Harrington NA, Rabideau DJ, Kinrys G, Kamali M. Baseline disability and poor functioning in bipolar disorder predict worse outcomes: results from the Bipolar CHOICE study. J Clin Psychiatry. 2016;77:100-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 2. | Bradley AJ, Anderson KN, Gallagher P, McAllister-Williams RH. The association between sleep and cognitive abnormalities in bipolar disorder. Psychol Med. 2020;50:125-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 3. | Haglili O, Sixsmith A, Star AP, Shmueli M, O'Rourke N. Perceived cognitive loss, symptomology, and psychological well-being with bipolar disorder. Int J Bipolar Disord. 2024;12:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Nierenberg AA, Agustini B, Köhler-Forsberg O, Cusin C, Katz D, Sylvia LG, Peters A, Berk M. Diagnosis and Treatment of Bipolar Disorder: A Review. JAMA. 2023;330:1370-1380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 299] [Cited by in RCA: 241] [Article Influence: 80.3] [Reference Citation Analysis (0)] |
| 5. | He H, Hu C, Ren Z, Bai L, Gao F, Lyu J. Trends in the incidence and DALYs of bipolar disorder at global, regional, and national levels: Results from the global burden of Disease Study 2017. J Psychiatr Res. 2020;125:96-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 92] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 6. | Post RM, Grunze H. The Challenges of Children with Bipolar Disorder. Medicina (Kaunas). 2021;57:601. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 7. | Wozniak J, O'Connor H, Iorini M, Ambrose AJH. Pediatric Bipolar Disorder: Challenges in Diagnosis and Treatment. Paediatr Drugs. 2025;27:125-142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 8. | Goes FS. Diagnosis and management of bipolar disorders. BMJ. 2023;381:e073591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 70] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 9. | Campbell I, Frye M, Campbell H. Metabolic Plasticity. An Evolutionary Perspective on Bipolar Disorder, Insulin Signalling and Ketosis. 2025 Preprint. [DOI] [Full Text] |
| 10. | Scott J, Etain B, Miklowitz D, Crouse JJ, Carpenter J, Marwaha S, Smith D, Merikangas K, Hickie I. A systematic review and meta-analysis of sleep and circadian rhythms disturbances in individuals at high-risk of developing or with early onset of bipolar disorders. Neurosci Biobehav Rev. 2022;135:104585. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 11. | Maejima T, Tsuno Y, Miyazaki S, Tsuneoka Y, Hasegawa E, Islam MT, Enoki R, Nakamura TJ, Mieda M. GABA from vasopressin neurons regulates the time at which suprachiasmatic nucleus molecular clocks enable circadian behavior. Proc Natl Acad Sci U S A. 2021;118:e2010168118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 12. | Harvey JRM, Plante AE, Meredith AL. Ion Channels Controlling Circadian Rhythms in Suprachiasmatic Nucleus Excitability. Physiol Rev. 2020;100:1415-1454. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 76] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 13. | Fagiani F, Di Marino D, Romagnoli A, Travelli C, Voltan D, Di Cesare Mannelli L, Racchi M, Govoni S, Lanni C. Molecular regulations of circadian rhythm and implications for physiology and diseases. Signal Transduct Target Ther. 2022;7:41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 375] [Cited by in RCA: 305] [Article Influence: 76.3] [Reference Citation Analysis (0)] |
| 14. | Patke A, Young MW, Axelrod S. Molecular mechanisms and physiological importance of circadian rhythms. Nat Rev Mol Cell Biol. 2020;21:67-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1222] [Cited by in RCA: 956] [Article Influence: 159.3] [Reference Citation Analysis (0)] |
| 15. | Takahashi JS. Transcriptional architecture of the mammalian circadian clock. Nat Rev Genet. 2017;18:164-179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2377] [Cited by in RCA: 1974] [Article Influence: 219.3] [Reference Citation Analysis (0)] |
| 16. | Lall GS, Atkinson LA, Corlett SA, Broadbridge PJ, Bonsall DR. Circadian entrainment and its role in depression: a mechanistic review. J Neural Transm (Vienna). 2012;119:1085-1096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 17. | von Gall C. The Effects of Light and the Circadian System on Rhythmic Brain Function. Int J Mol Sci. 2022;23:2778. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 18. | Moeller JS, Bever SR, Finn SL, Phumsatitpong C, Browne MF, Kriegsfeld LJ. Circadian Regulation of Hormonal Timing and the Pathophysiology of Circadian Dysregulation. Compr Physiol. 2022;12:4185-4214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 19. | Chung S, Son GH, Kim K. Adrenal peripheral oscillator in generating the circadian glucocorticoid rhythm. Ann N Y Acad Sci. 2011;1220:71-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 20. | Koronowski KB, Sassone-Corsi P. Communicating clocks shape circadian homeostasis. Science. 2021;371:eabd0951. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 243] [Cited by in RCA: 222] [Article Influence: 44.4] [Reference Citation Analysis (0)] |
| 21. | Gold AK, Kinrys G. Treating Circadian Rhythm Disruption in Bipolar Disorder. Curr Psychiatry Rep. 2019;21:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 54] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 22. | Yalçin M, Grande V, Outeiro TF, Relógio A. Circadian clock dysfunction in Parkinson's disease: mechanisms, consequences, and therapeutic strategy. NPJ Parkinsons Dis. 2025;11:213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 23. | Abreu T, Bragança M. The bipolarity of light and dark: A review on Bipolar Disorder and circadian cycles. J Affect Disord. 2015;185:219-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 98] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 24. | Rahati S, Qorbani M, Naghavi A, Pishva H. The interaction between the Circadian Locomotor Output Cycles Kaput and Melanocortin-4-receptor gene variants on obesity and parameters related to obesity. Clin Nutr. 2025;45:193-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | Yegin Z, Sarisoy G, Erguner Aral A, Koc H. For whom the circadian clock ticks? Investigation of PERIOD and CLOCK gene variants in bipolar disorder. Chronobiol Int. 2021;38:1109-1119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 26. | Maciukiewicz M, Dmitrzak-Weglarz M, Pawlak J, Leszczynska-Rodziewicz A, Zaremba D, Skibinska M, Hauser J. Analysis of genetic association and epistasis interactions between circadian clock genes and symptom dimensions of bipolar affective disorder. Chronobiol Int. 2014;31:770-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 27. | Rybakowski JK, Dmitrzak-Weglar M, Kliwicki S, Hauser J. Polymorphism of circadian clock genes and prophylactic lithium response. Bipolar Disord. 2014;16:151-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 28. | Rybakowski JK, Dmitrzak-Weglarz M, Dembinska-Krajewska D, Hauser J, Akiskal KK, Akiskal HH. Polymorphism of circadian clock genes and temperamental dimensions of the TEMPS-A in bipolar disorder. J Affect Disord. 2014;159:80-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 29. | Bengesser SA, Reininghaus EZ, Lackner N, Birner A, Fellendorf FT, Platzer M, Kainzbauer N, Tropper B, Hörmanseder C, Queissner R, Kapfhammer HP, Wallner-Liebmann SJ, Fuchs R, Petek E, Windpassinger C, Schnalzenberger M, Reininghaus B, Evert B, Waha A. Is the molecular clock ticking differently in bipolar disorder? Methylation analysis of the clock gene ARNTL. World J Biol Psychiatry. 2018;19:S21-S29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 30. | Bengesser SA, Hohenberger H, Tropper B, Dalkner N, Birner A, Fellendorf FT, Platzer M, Rieger A, Maget A, Hamm C, Queissner R, Pilz R, Bauer K, Lenger M, Mörkl S, Wagner-Skacel J, Kapfhammer HP, Meier-Allard N, Stracke A, Holasek SJ, Murphy L, Reininghaus EZ. Gene expression analysis of MAOA and the clock gene ARNTL in individuals with bipolar disorder compared to healthy controls. World J Biol Psychiatry. 2022;23:287-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 31. | Swaminathan A, Kenzior A, Price A, Li H, McCoin C, Hintermann A, Weaver K, Morris N, Keene AC, Rohner N. A repeatedly evolved mutation in Cryptochrome-1 of subterranean animals alters behavioral and molecular circadian rhythms. iScience. 2025;28:112874. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 32. | Toffoli B, Tonon F, Giudici F, Ferretti T, Ghirigato E, Contessa M, Francica M, Candido R, Puato M, Grillo A, Fabris B, Bernardi S. Preliminary Study on the Effect of a Night Shift on Blood Pressure and Clock Gene Expression. Int J Mol Sci. 2023;24:9309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 33. | Mishra HK, Wei H, LeRoux M, Ko D, Rohr K, Nievergelt CM, Maihofer AX, Shilling PD, Alda M, Berrettini WH, Coryell WH, Frye M, Gershon E, McInnis MG, Nurnberger J, Oedegaard KJ, Zandi PP, Kelsoe JR, McCarthy MJ. Circadian clock genes and cell survival in bipolar disorder: Insights into lithium responsiveness and molecular mechanisms in patient-derived neural progenitor cells and mouse neurons. FEBS J. 2025;292:2806-2822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 34. | Farhangdoost N, Liao C, Liu Y, Alda M, Dion PA, Rouleau GA, Khayachi A, Chaumette B. Transcriptomic and epigenomic consequences of heterozygous loss of function mutations in AKAP11, the first large-effect shared risk gene for bipolar disorder and schizophrenia. 2024 Preprint. [DOI] [Full Text] |
| 35. | Rathor P, Ch R. Metabolic Basis of Circadian Dysfunction in Parkinson's Disease. Biology (Basel). 2023;12:1294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 36. | Pradel K, Drwięga G, Chrobok L, Błasiak T. Racing and Pacing in the Reward System: A Multi-Clock Circadian Control Over Dopaminergic Signalling. Front Physiol. 2022;13:932378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 37. | Mesgar S, Eskandari K, Karimian-Sani-Varjovi H, Salemi-Mokri-Boukani P, Haghparast A. The Dopaminergic System Modulates the Electrophysiological Activity of the Suprachiasmatic Nucleus Dependent on the Circadian Cycle. Neurochem Res. 2023;48:3420-3429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 38. | Siemann JK, Grueter BA, McMahon DG. Rhythms, Reward, and Blues: Consequences of Circadian Photoperiod on Affective and Reward Circuit Function. Neuroscience. 2021;457:220-234. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 39. | Liu Q, Meng Q, Qi J, Cheng J, Jiang J, Shang L, Wei X, Hao W. Unfixed light pattern-related circadian disruption impairs circadian-orexinergic-serotoninergic system and induces depression-like behaviors in mice. Toxicol Appl Pharmacol. 2025;503:117464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 40. | Tu HQ, Li S, Xu YL, Zhang YC, Li PY, Liang LY, Song GP, Jian XX, Wu M, Song ZQ, Li TT, Hu HB, Yuan JF, Shen XL, Li JN, Han QY, Wang K, Zhang T, Zhou T, Li AL, Zhang XM, Li HY. Rhythmic cilia changes support SCN neuron coherence in circadian clock. Science. 2023;380:972-979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 57] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 41. | Albers HE, Walton JC, Gamble KL, McNeill JK 4th, Hummer DL. The dynamics of GABA signaling: Revelations from the circadian pacemaker in the suprachiasmatic nucleus. Front Neuroendocrinol. 2017;44:35-82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 82] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 42. | Ono D, Honma KI, Honma S. GABAergic mechanisms in the suprachiasmatic nucleus that influence circadian rhythm. J Neurochem. 2021;157:31-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 43. | Steardo L Jr, D'Angelo M, Monaco F, Di Stefano V, Steardo L. Decoding neural circuit dysregulation in bipolar disorder: Toward an advanced paradigm for multidimensional cognitive, emotional, and psychomotor treatment. Neurosci Biobehav Rev. 2025;169:106030. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 44. | Andreadi A, Andreadi S, Todaro F, Ippoliti L, Bellia A, Magrini A, Chrousos GP, Lauro D. Modified Cortisol Circadian Rhythm: The Hidden Toll of Night-Shift Work. Int J Mol Sci. 2025;26:2090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 36] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 45. | Robertson-Dixon I, Murphy MJ, Crewther SG, Riddell N. The Influence of Light Wavelength on Human HPA Axis Rhythms: A Systematic Review. Life (Basel). 2023;13:1968. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 46. | Yurgel ME, Gao C, O'Malley JJ, Tang Q, Yanay N, Bashford AR, Zhan JJ, Lutas A, Krashes MJ, Zhao H, Penzo MA, Hattar S. A stress-sensing circuit signals to the central pacemaker to reprogram circadian rhythms. Sci Adv. 2025;11:eadr7960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 47. | Dollish HK, Tsyglakova M, McClung CA. Circadian rhythms and mood disorders: Time to see the light. Neuron. 2024;112:25-40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 92] [Article Influence: 46.0] [Reference Citation Analysis (0)] |
| 48. | Madamanchi K, Zhang J, Melkani GC. Linkage of circadian rhythm disruptions with Alzheimer's disease and therapeutic interventions. Acta Pharm Sin B. 2025;15:2945-2965. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 49. | Kato T, Kato N. Mitochondrial dysfunction in bipolar disorder. Bipolar Disord. 2000;2:180-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 312] [Cited by in RCA: 289] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 50. | Berk M, Corrales A, Trisno R, Dodd S, Yatham LN, Vieta E, McIntyre RS, Suppes T, Agustini B. Bipolar II disorder: a state-of-the-art review. World Psychiatry. 2025;24:175-189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 51. | Du T, Liu S, Yu H, Hu T, Huang L, Gao L, Jia L, Hu J, Yu Y, Sun Q. Chronic sleep deprivation disturbs energy balance modulated by suprachiasmatic nucleus efferents in mice. BMC Biol. 2024;22:296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 52. | Stoiljkovic M, Song JE, Hong HK, Endle H, Varela L, Catarino J, Gao XB, Liu ZW, Sotonyi P, Diano S, Cedernaes J, Bass J, Horvath TL. Mitofusin 2 controls mitochondrial and synaptic dynamics of suprachiasmatic VIP neurons and related circadian rhythms. J Clin Invest. 2025;135:e185000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 53. | Della DF, Allison S, Bidargaddi N, Wa SK, Bastiampillai T. An Umbrella Systematic Review of Seasonality in Mood Disorders and Suicide Risk: The Impact on Demand for Primary Behavioral Health Care and Acute Psychiatric Services. Prim Care Companion CNS Disord. 2023;25:22r03395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 54. | Livieratos A, Lockley SW, Tsiodras S. Post infectious fatigue and circadian rhythm disruption in long-COVID and other infections: a need for further research. EClinicalMedicine. 2025;80:103073. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 55. | Mao W, Ge X, Chen Q, Li JD. Epigenetic Mechanisms in the Transcriptional Regulation of Circadian Rhythm in Mammals. Biology (Basel). 2025;14:42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 56. | Kim YH, Lazar MA. Transcriptional Control of Circadian Rhythms and Metabolism: A Matter of Time and Space. Endocr Rev. 2020;41:707-732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 106] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 57. | Marchetti M, Mayeli A, Sanguineti C, Donati FL, Chaichian O, Kim A, Piskun K, D'Agostino A, Meyer N, Wilson JD, Fusar-Poli P, Phillips ML, Ferrarelli F. Sleep abnormalities in bipolar disorders across mood phases: A systematic review and meta-analysis. Sleep Med Rev. 2025;83:102137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 58. | Panchal P, de Queiroz Campos G, Goldman DA, Auerbach RP, Merikangas KR, Swartz HA, Sankar A, Blumberg HP. Toward a Digital Future in Bipolar Disorder Assessment: A Systematic Review of Disruptions in the Rest-Activity Cycle as Measured by Actigraphy. Front Psychiatry. 2022;13:780726. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 59. | Scott MR, McClung CA. Bipolar Disorder. Curr Opin Neurobiol. 2023;83:102801. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 60. | Patke A, Murphy PJ, Onat OE, Krieger AC, Özçelik T, Campbell SS, Young MW. Mutation of the Human Circadian Clock Gene CRY1 in Familial Delayed Sleep Phase Disorder. Cell. 2017;169:203-215.e13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 350] [Cited by in RCA: 285] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 61. | Yavuz Ataşlar E, Altınbaş K. A comprehensive examination of circadian rhythm and tryptophan pathway parameters: Assessing their role in predicting bipolar disorder in patients, siblings, and controls. Chronobiol Int. 2025;42:755-769. [PubMed] [DOI] [Full Text] |
| 62. | National Toxicology Program. NTP Cancer Hazard Assessment Report on Night Shift Work and Light at Night. Research Triangle Park (NC): National Toxicology Program; 2021. [PubMed] [DOI] [Full Text] |
| 63. | Liang X, Ding Y, Zhu X, Qiu J, Shen X, Xiong Y, Zhou J, Liang X, Xie W. Suprachiasmatic nucleus dysfunction induces anxiety- and depression-like behaviors via activating the BDNF-TrkB pathway of the striatum. Transl Psychiatry. 2025;15:92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 64. | Ma T, Matsuo R, Kurogi K, Miyamoto S, Morita T, Shinozuka M, Taniguchi F, Ikegami K, Yasuo S. Sex-dependent effects of chronic jet lag on circadian rhythm and metabolism in mice. Biol Sex Differ. 2024;15:102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 65. | Hasenmajer V, Sbardella E, Sciarra F, Simeoli C, Pivonello C, Ceccato F, Pofi R, Minnetti M, Rizzo F, Ferrari D, Bonaventura I, Barbagallo F, Giannetta E, Alunni Fegatelli D, Conia S, Navigli R, Arnaldi G, Scaroni C, Pivonello R, Gianfrilli D, Venneri MA, Isidori AM. Circadian clock disruption impairs immune oscillation in chronic endogenous hypercortisolism: a multi-level analysis from a multicentre clinical trial. EBioMedicine. 2024;110:105462. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 66. | Ghavamikia N, Mehrnoosh F, Zare F, Ali-Khiavi P, Sinehsepehr A, Boushehri YG, Vahedinezhad M, Abdollahi E, Hjazi A, Aminnezhad S, Saffarfar H, Hamzehzadeh S, Nourizadeh M, KarkonShayan S. Mitochondrial Quality Control and Melatonin: A Strategy Against Myocardial Injury. J Biochem Mol Toxicol. 2025;39:e70194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 67. | Matsui S, Yanai-Inamura H, Kajiro M, Takamatsu H, Tanahashi M, Yokono M. Melatonin induces urethral contraction and increases intraurethral pressure via MT(1) receptor activation in female rats. Eur J Pharmacol. 2025;998:177539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 68. | Yang Z, Black K, Ohman-Strickland P, Graber JM, Kipen HM, Fang M, Zarbl H. Disruption of central and peripheral circadian clocks and circadian controlled estrogen receptor rhythms in night shift nurses in working environments. FASEB J. 2024;38:e23719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 69. | Woodie LN, Alberto AJ, Krusen BM, Melink LC, Lazar MA. Genetic synchronization of the brain and liver molecular clocks defend against chrono-metabolic disease. Proc Natl Acad Sci U S A. 2024;121:e2417678121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 70. | Palm D, Uzoni A, Simon F, Tucha O, Thome J, Faltraco F. Norepinephrine influences the circadian clock in human dermal fibroblasts from study participants with a diagnosis of attention-deficit hyperactivity disorder. J Neural Transm (Vienna). 2021;128:1147-1157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 71. | Costello HM, Gumz ML. Circadian Rhythm, Clock Genes, and Hypertension: Recent Advances in Hypertension. Hypertension. 2021;78:1185-1196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 105] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 72. | Torsvik A, Brattbakk HR, Trentani A, Holdhus R, Stansberg C, Bartz-Johannessen CA, Hughes T, Steen NE, Melle I, Djurovic S, Andreassen OA, Steen VM. Patients with schizophrenia and bipolar disorder display a similar global gene expression signature in whole blood that reflects elevated proportion of immature neutrophil cells with association to lipid changes. Transl Psychiatry. 2023;13:147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 73. | Nakajima K, Miranda A, Craig DW, Shekhtman T, Kmoch S, Bleyer A, Szelinger S, Kato T, Kelsoe JR. Ntrk1 mutation co-segregating with bipolar disorder and inherited kidney disease in a multiplex family causes defects in neuronal growth and depression-like behavior in mice. Transl Psychiatry. 2020;10:407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 74. | Mullins N, Forstner AJ, O'Connell KS, Coombes B, Coleman JRI, Qiao Z, Als TD, Bigdeli TB, Børte S, Bryois J, Charney AW, Drange OK, Gandal MJ, Hagenaars SP, Ikeda M, Kamitaki N, Kim M, Krebs K, Panagiotaropoulou G, Schilder BM, Sloofman LG, Steinberg S, Trubetskoy V, Winsvold BS, Won HH, Abramova L, Adorjan K, Agerbo E, Al Eissa M, Albani D, Alliey-Rodriguez N, Anjorin A, Antilla V, Antoniou A, Awasthi S, Baek JH, Bækvad-Hansen M, Bass N, Bauer M, Beins EC, Bergen SE, Birner A, Bøcker Pedersen C, Bøen E, Boks MP, Bosch R, Brum M, Brumpton BM, Brunkhorst-Kanaan N, Budde M, Bybjerg-Grauholm J, Byerley W, Cairns M, Casas M, Cervantes P, Clarke TK, Cruceanu C, Cuellar-Barboza A, Cunningham J, Curtis D, Czerski PM, Dale AM, Dalkner N, David FS, Degenhardt F, Djurovic S, Dobbyn AL, Douzenis A, Elvsåshagen T, Escott-Price V, Ferrier IN, Fiorentino A, Foroud TM, Forty L, Frank J, Frei O, Freimer NB, Frisén L, Gade K, Garnham J, Gelernter J, Giørtz Pedersen M, Gizer IR, Gordon SD, Gordon-Smith K, Greenwood TA, Grove J, Guzman-Parra J, Ha K, Haraldsson M, Hautzinger M, Heilbronner U, Hellgren D, Herms S, Hoffmann P, Holmans PA, Huckins L, Jamain S, Johnson JS, Kalman JL, Kamatani Y, Kennedy JL, Kittel-Schneider S, Knowles JA, Kogevinas M, Koromina M, Kranz TM, Kranzler HR, Kubo M, Kupka R, Kushner SA, Lavebratt C, Lawrence J, Leber M, Lee HJ, Lee PH, Levy SE, Lewis C, Liao C, Lucae S, Lundberg M, MacIntyre DJ, Magnusson SH, Maier W, Maihofer A, Malaspina D, Maratou E, Martinsson L, Mattheisen M, McCarroll SA, McGregor NW, McGuffin P, McKay JD, Medeiros H, Medland SE, Millischer V, Montgomery GW, Moran JL, Morris DW, Mühleisen TW, O'Brien N, O'Donovan C, Olde Loohuis LM, Oruc L, Papiol S, Pardiñas AF, Perry A, Pfennig A, Porichi E, Potash JB, Quested D, Raj T, Rapaport MH, DePaulo JR, Regeer EJ, Rice JP, Rivas F, Rivera M, Roth J, Roussos P, Ruderfer DM, Sánchez-Mora C, Schulte EC, Senner F, Sharp S, Shilling PD, Sigurdsson E, Sirignano L, Slaney C, Smeland OB, Smith DJ, Sobell JL, Søholm Hansen C, Soler Artigas M, Spijker AT, Stein DJ, Strauss JS, Świątkowska B, Terao C, Thorgeirsson TE, Toma C, Tooney P, Tsermpini EE, Vawter MP, Vedder H, Walters JTR, Witt SH, Xi S, Xu W, Yang JMK, Young AH, Young H, Zandi PP, Zhou H, Zillich L; HUNT All-In Psychiatry, Adolfsson R, Agartz I, Alda M, Alfredsson L, Babadjanova G, Backlund L, Baune BT, Bellivier F, Bengesser S, Berrettini WH, Blackwood DHR, Boehnke M, Børglum AD, Breen G, Carr VJ, Catts S, Corvin A, Craddock N, Dannlowski U, Dikeos D, Esko T, Etain B, Ferentinos P, Frye M, Fullerton JM, Gawlik M, Gershon ES, Goes FS, Green MJ, Grigoroiu-Serbanescu M, Hauser J, Henskens F, Hillert J, Hong KS, Hougaard DM, Hultman CM, Hveem K, Iwata N, Jablensky AV, Jones I, Jones LA, Kahn RS, Kelsoe JR, Kirov G, Landén M, Leboyer M, Lewis CM, Li QS, Lissowska J, Lochner C, Loughland C, Martin NG, Mathews CA, Mayoral F, McElroy SL, McIntosh AM, McMahon FJ, Melle I, Michie P, Milani L, Mitchell PB, Morken G, Mors O, Mortensen PB, Mowry B, Müller-Myhsok B, Myers RM, Neale BM, Nievergelt CM, Nordentoft M, Nöthen MM, O'Donovan MC, Oedegaard KJ, Olsson T, Owen MJ, Paciga SA, Pantelis C, Pato C, Pato MT, Patrinos GP, Perlis RH, Posthuma D, Ramos-Quiroga JA, Reif A, Reininghaus EZ, Ribasés M, Rietschel M, Ripke S, Rouleau GA, Saito T, Schall U, Schalling M, Schofield PR, Schulze TG, Scott LJ, Scott RJ, Serretti A, Shannon Weickert C, Smoller JW, Stefansson H, Stefansson K, Stordal E, Streit F, Sullivan PF, Turecki G, Vaaler AE, Vieta E, Vincent JB, Waldman ID, Weickert TW, Werge T, Wray NR, Zwart JA, Biernacka JM, Nurnberger JI, Cichon S, Edenberg HJ, Stahl EA, McQuillin A, Di Florio A, Ophoff RA, Andreassen OA. Genome-wide association study of more than 40,000 bipolar disorder cases provides new insights into the underlying biology. Nat Genet. 2021;53:817-829. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1397] [Cited by in RCA: 1101] [Article Influence: 220.2] [Reference Citation Analysis (0)] |
| 75. | Alloy LB, Abramson LY, Urosevic S, Walshaw PD, Nusslock R, Neeren AM. The psychosocial context of bipolar disorder: environmental, cognitive, and developmental risk factors. Clin Psychol Rev. 2005;25:1043-1075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 126] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 76. | Veddum L, Greve AN, Andreassen AK, Knudsen CB, Brandt JM, Gregersen M, Krantz MF, Søndergaard A, Ohland J, Burton BK, Jepsen JRM, Hemager N, Thorup AAE, Nordentoft M, Mors O, Bliksted V. Development of social responsiveness and theory of mind in children of parents with schizophrenia or bipolar disorder. Schizophr Res Cogn. 2022;28:100242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 77. | Cannon M, Jones P, Gilvarry C, Rifkin L, McKenzie K, Foerster A, Murray RM. Premorbid social functioning in schizophrenia and bipolar disorder: similarities and differences. Am J Psychiatry. 1997;154:1544-1550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 68] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 78. | Walsh RFL, Klugman J, Moriarity DP, Titone MK, Ng TH, Goel N, Alloy LB. Reward sensitivity and social rhythms during goal-striving: An ecological momentary assessment investigation of bipolar spectrum disorders. J Affect Disord. 2024;344:510-518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 79. | Alloy LB, Ng TH, Titone MK, Boland EM. Circadian Rhythm Dysregulation in Bipolar Spectrum Disorders. Curr Psychiatry Rep. 2017;19:21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 98] [Article Influence: 10.9] [Reference Citation Analysis (1)] |
| 80. | Murray G, Harvey A. Circadian rhythms and sleep in bipolar disorder. Bipolar Disord. 2010;12:459-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 170] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 81. | Monk TH, Frank E, Potts JM, Kupfer DJ. A simple way to measure daily lifestyle regularity. J Sleep Res. 2002;11:183-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 77] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 82. | Monk TH, Kupfer DJ, Frank E, Ritenour AM. The Social Rhythm Metric (SRM): measuring daily social rhythms over 12 weeks. Psychiatry Res. 1991;36:195-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 111] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 83. | Shen GH, Alloy LB, Abramson LY, Sylvia LG. Social rhythm regularity and the onset of affective episodes in bipolar spectrum individuals. Bipolar Disord. 2008;10:520-529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 101] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 84. | Wyse CA, Biello SM, Gill JM. The bright-nights and dim-days of the urban photoperiod: implications for circadian rhythmicity, metabolism and obesity. Ann Med. 2014;46:253-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 85. | Carta MG, Preti A, Akiskal HS. Coping with the New Era: Noise and Light Pollution, Hperactivity and Steroid Hormones. Towards an Evolutionary View of Bipolar Disorders. Clin Pract Epidemiol Ment Health. 2018;14:33-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 86. | Deprato A, Haldar P, Navarro JF, Harding BN, Lacy P, Maidstone R, Moitra S, Palomar-Cros A, Durrington H, Kogevinas M, Moitra S, Adan A. Associations between light at night and mental health: A systematic review and meta-analysis. Sci Total Environ. 2025;974:179188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 87. | Esaki Y, Obayashi K, Saeki K, Fujita K, Iwata N, Kitajima T. Association between light exposure at night and manic symptoms in bipolar disorder: cross-sectional analysis of the APPLE cohort. Chronobiol Int. 2020;37:887-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 88. | Esaki Y, Obayashi K, Saeki K, Fujita K, Iwata N, Kitajima T. Bedroom light exposure at night and obesity in individuals with bipolar disorder: A cross-sectional analysis of the APPLE cohort. Physiol Behav. 2021;230:113281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 89. | LeGates TA, Altimus CM, Wang H, Lee HK, Yang S, Zhao H, Kirkwood A, Weber ET, Hattar S. Aberrant light directly impairs mood and learning through melanopsin-expressing neurons. Nature. 2012;491:594-598. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 484] [Cited by in RCA: 417] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 90. | Li Y, Zou X, Ma Y, Cheng J, Yu X, Shao W, Zheng F, Guo Z, Yu G, Wu S, Li H, Hu H. Lactic acid contributes to the emergence of depression-like behaviors triggered by blue light exposure during sleep. Ecotoxicol Environ Saf. 2025;289:117643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 91. | Ren W, Wang Z, Dong Y, Cao J, Gao T, Guo Q, Chen Y. Dim blue light at night worsens high-fat diet-induced kidney damage via increasing corticosterone levels and modulating the expression of circadian clock genes. Ecotoxicol Environ Saf. 2025;289:117636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 92. | Xu YX, Shen YT, Li J, Ding WQ, Wan YH, Su PY, Tao FB, Sun Y. Real-ambient bedroom light at night increases systemic inflammation and disrupts circadian rhythm of inflammatory markers. Ecotoxicol Environ Saf. 2024;281:116590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 93. | Esaki Y, Obayashi K, Saeki K, Fujita K, Iwata N, Kitajima T. Effect of nighttime bedroom light exposure on mood episode relapses in bipolar disorder. Acta Psychiatr Scand. 2022;146:64-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 94. | Esaki Y, Obayashi K, Saeki K, Fujita K, Iwata N, Kitajima T. Preventive effect of morning light exposure on relapse into depressive episode in bipolar disorder. Acta Psychiatr Scand. 2021;143:328-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 95. | Esaki Y, Obayashi K, Saeki K, Fujita K, Iwata N, Kitajima T. Habitual light exposure and circadian activity rhythms in bipolar disorder: A cross-sectional analysis of the APPLE cohort. J Affect Disord. 2023;323:762-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 96. | Chen Y, Hong W, Fang Y. Role of biological rhythm dysfunction in the development and management of bipolar disorders: a review. Gen Psychiatr. 2020;33:e100127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 97. | Gallego-Landin I, Berbegal-Sáez P, Valverde O. Chronic circadian disruption in adolescent mice impairs hippocampal memory disrupting gene expression oscillations. Sci Rep. 2025;15:26291. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 98. | Leise TL, Goldberg A, Michael J, Montoya G, Solow S, Molyneux P, Vetrivelan R, Harrington ME. Recurring circadian disruption alters circadian clock sensitivity to resetting. Eur J Neurosci. 2020;51:2343-2354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 99. | Ortiz A, Halabi R, Alda M, DeShaw A, Husain MI, Nunes A, O'Donovan C, Patterson R, Mulsant BH, Hintze A. Day-to-day variability in activity levels detects transitions to depressive symptoms in bipolar disorder earlier than changes in sleep and mood. Int J Bipolar Disord. 2025;13:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 100. | McIntyre RS, Berk M, Brietzke E, Goldstein BI, López-Jaramillo C, Kessing LV, Malhi GS, Nierenberg AA, Rosenblat JD, Majeed A, Vieta E, Vinberg M, Young AH, Mansur RB. Bipolar disorders. Lancet. 2020;396:1841-1856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 891] [Cited by in RCA: 693] [Article Influence: 115.5] [Reference Citation Analysis (0)] |
| 101. | Ramírez-Martín A, Sirignano L, Foo JC, Streit F, Frank J, Witt SH, Rietschel M, Mayoral-Cleries F, Moreno-Küstner B, Guzmán-Parra J. The relationships between impulsivity and mood in bipolar disorder: An ecological momentary assessment study. PLoS One. 2025;20:e0314963. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 102. | Hafeman DM, Feldman J, Mak J, Merranko J, Goldstein TR, Gratton C, Phillips ML, Birmaher B. Longitudinal stability of mood-related resting-state networks in youth with symptomatic bipolar-I/II disorder. Transl Psychiatry. 2025;15:187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 103. | Mwaba K, Molamu RB. Perceived causes of relapse among a sample of recovering psychiatric patients at a Mafikeng hospital. Curationis. 1998;21:55-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 104. | Belete H, Ali T, Legas G. Relapse and Clinical Characteristics of Patients with Bipolar Disorders in Central Ethiopia: A Cross-Sectional Study. Psychiatry J. 2020;2020:8986014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 105. | Kripke DF, Mullaney DJ, Atkinson M, Wolf S. Circadian rhythm disorders in manic-depressives. Biol Psychiatry. 1978;13:335-351. [PubMed] |
| 106. | McCarty R, Josephs T, Kovtun O, Rosenthal SJ. Enlightened: addressing circadian and seasonal changes in photoperiod in animal models of bipolar disorder. Transl Psychiatry. 2021;11:373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 107. | Aguglia A, Borsotti A, Maina G. Bipolar disorders: is there an influence of seasonality or photoperiod? Braz J Psychiatry. 2018;40:6-11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 108. | Geoffroy PA, Bellivier F, Scott J, Etain B. Seasonality and bipolar disorder: a systematic review, from admission rates to seasonality of symptoms. J Affect Disord. 2014;168:210-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 178] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 109. | Sam SP, Nisha A, Varghese PJ. Stressful Life Events and Relapse in Bipolar Affective Disorder: A Cross-Sectional Study from a Tertiary Care Center of Southern India. Indian J Psychol Med. 2019;41:61-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 110. | Cusell H, Kok R. A Retrospective Study of the Influence of Life Events and Social Support on Relapses and Recurrences in Older Patients with Bipolar Disorder. Geriatrics (Basel). 2025;10:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 111. | Koenders MA, Giltay EJ, Spijker AT, Hoencamp E, Spinhoven P, Elzinga BM. Stressful life events in bipolar I and II disorder: cause or consequence of mood symptoms? J Affect Disord. 2014;161:55-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 53] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 112. | Grandin LD, Alloy LB, Abramson LY. The social zeitgeber theory, circadian rhythms, and mood disorders: review and evaluation. Clin Psychol Rev. 2006;26:679-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 262] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 113. | Smith LT, Walsh RFL, Goel N, Alloy LB. Social jetlag and trajectories of mood symptoms and reward responsiveness in individuals at low-risk, high-risk, and with bipolar spectrum disorders: An ecological momentary assessment study. Psychiatry Res. 2023;329:115499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 114. | Stokkan KA, Yamazaki S, Tei H, Sakaki Y, Menaker M. Entrainment of the circadian clock in the liver by feeding. Science. 2001;291:490-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1485] [Cited by in RCA: 1306] [Article Influence: 52.2] [Reference Citation Analysis (1)] |
| 115. | Manoogian ENC, Chow LS, Taub PR, Laferrère B, Panda S. Time-restricted Eating for the Prevention and Management of Metabolic Diseases. Endocr Rev. 2022;43:405-436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 286] [Cited by in RCA: 257] [Article Influence: 64.3] [Reference Citation Analysis (1)] |
| 116. | Dmitrzak-Weglarz M, Banach E, Bilska K, Narozna B, Szczepankiewicz A, Reszka E, Jablonska E, Kapelski P, Skibinska M, Pawlak J. Molecular Regulation of the Melatonin Biosynthesis Pathway in Unipolar and Bipolar Depression. Front Pharmacol. 2021;12:666541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 117. | Javeed N, Matveyenko AV. Circadian Etiology of Type 2 Diabetes Mellitus. Physiology (Bethesda). 2018;33:138-150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 112] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 118. | Ahmad A, Anderson KN, Watson S. Sleep and Circadian Rhythm Disorder in Bipolar Affective Disorder. Curr Top Behav Neurosci. 2021;48:133-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 119. | Harvey AG, Talbot LS, Gershon A. Sleep Disturbance in Bipolar Disorder Across the Lifespan. Clin Psychol (New York). 2009;16:256-277. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 95] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 120. | Goldschmied JR, Palermo E, Yocum A, McInnis M, Gehrman P. The relationship between sleep and circadian patterns with risk for suicide in bipolar disorder varies by subtype. J Psychiatr Res. 2025;181:23-28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 121. | Gonzalez R, Tohen M. Circadian Rhythm and the Prediction of Relapse in Bipolar Disorder. J Clin Psychiatry. 2018;79:17com11821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 122. | Takaesu Y, Inoue Y, Ono K, Murakoshi A, Futenma K, Komada Y, Inoue T. Circadian Rhythm Sleep-Wake Disorders Predict Shorter Time to Relapse of Mood Episodes in Euthymic Patients With Bipolar Disorder: A Prospective 48-Week Study. J Clin Psychiatry. 2018;79:17m11565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 123. | Cretu JB, Culver JL, Goffin KC, Shah S, Ketter TA. Sleep, residual mood symptoms, and time to relapse in recovered patients with bipolar disorder. J Affect Disord. 2016;190:162-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 82] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 124. | Esaki Y, Obayashi K, Saeki K, Fujita K, Iwata N, Kitajima T. Circadian variability of objective sleep measures predicts the relapse of a mood episode in bipolar disorder: findings from the APPLE cohort. Psychiatry Clin Neurosci. 2023;77:442-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 125. | Rohr KE, McCarthy MJ. The impact of lithium on circadian rhythms and implications for bipolar disorder pharmacotherapy. Neurosci Lett. 2022;786:136772. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 126. | Mishra HK, Ying NM, Luis A, Wei H, Nguyen M, Nakhla T, Vandenburgh S, Alda M, Berrettini WH, Brennand KJ, Calabrese JR, Coryell WH, Frye MA, Gage FH, Gershon ES, McInnis MG, Nievergelt CM, Nurnberger JI, Shilling PD, Oedegaard KJ, Zandi PP; Pharmacogenomics of Bipolar Disorder Study, Kelsoe JR, Welsh DK, McCarthy MJ. Circadian rhythms in bipolar disorder patient-derived neurons predict lithium response: preliminary studies. Mol Psychiatry. 2021;26:3383-3394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 127. | Uçak EF, Altınbaş K, Koçak N, Güleç A. Circadian rhythm and lithium response in bipolar disorder: Insights from actigraphy and NR1D1 polymorphism. Chronobiol Int. 2025;42:225-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 128. | Federoff M, McCarthy MJ, Anand A, Berrettini WH, Bertram H, Bhattacharjee A, Calkin CV, Conroy C, Coryell WH, D'Arcangelo N, DeModena A, Fisher C, Feeder S, Frazier N, Frye MA, Gao K, Garnham J, Gershon ES, Alliey-Rodriguez N, Glazer K, Goes F, Karberg T, Harrington G, Jakobsen P, Kamali M, Kelly M, Leckband SG, Lohoff F, Maihofer AX, McInnis MG, Mondimore F, Morken G, Nurnberger JI, Oedegaard KJ, Ritchey M, Ryan K, Schinagle M, Schoeyen H, Schwebel C, Shaw M, Shilling PD, Slaney C, Stautland A, Tarwater B, Calabrese JR, Alda M, Nievergelt CM, Zandi PP, Kelsoe JR. Correction of depression-associated circadian rhythm abnormalities is associated with lithium response in bipolar disorder. Bipolar Disord. 2022;24:521-529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 129. | Xu N, Shinohara K, Saunders KEA, Geddes JR, Cipriani A. Effect of lithium on circadian rhythm in bipolar disorder: A systematic review and meta-analysis. Bipolar Disord. 2021;23:445-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 130. | Carli M, Risaliti E, Francomano M, Kolachalam S, Longoni B, Bocci G, Maggio R, Scarselli M. A 5-Year Study of Lithium and Valproic Acid Drug Monitoring in Patients with Bipolar Disorders in an Italian Clinical Center. Pharmaceuticals (Basel). 2022;15:105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 131. | Griggs CA, Malm SW, Jaime-Frias R, Smith CL. Valproic acid disrupts the oscillatory expression of core circadian rhythm transcription factors. Toxicol Appl Pharmacol. 2018;339:110-120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 132. | Ferraro S, de Zavalia N, Belforte N, Amir S. In utero Exposure to Valproic-Acid Alters Circadian Organisation and Clock-Gene Expression: Implications for Autism Spectrum Disorders. Front Behav Neurosci. 2021;15:711549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 133. | Landgraf D, Joiner WJ, McCarthy MJ, Kiessling S, Barandas R, Young JW, Cermakian N, Welsh DK. The mood stabilizer valproic acid opposes the effects of dopamine on circadian rhythms. Neuropharmacology. 2016;107:262-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 134. | Kragh M, Dyrberg H, Kristiansen ST, Speed M, Pedersen P, Martiny K. Efficacy of a Transdiagnostic Sleep and Circadian Intervention for Outpatients With Sleep Problems and Depression, Attention Deficit Disorder, or Bipolar Disorder: A Randomised Controlled Trial. J Sleep Res. 2025;e70088. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (3)] |
| 135. | Palagini L, Miniati M, Caruso D, Massa L, Novi M, Pardini F, Salarpi G, Pini S, Marazziti D, Etain B, Riemann D. Association between affective temperaments and mood features in bipolar disorder II: The role of insomnia and chronobiological rhythms desynchronization. J Affect Disord. 2020;266:263-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 136. | Slyepchenko A, Allega OR, Leng X, Minuzzi L, Eltayebani MM, Skelly M, Sassi RB, Soares CN, Kennedy SH, Frey BN. Association of functioning and quality of life with objective and subjective measures of sleep and biological rhythms in major depressive and bipolar disorder. Aust N Z J Psychiatry. 2019;53:683-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 137. | Bertrand L, Bourguignon C, Beaulieu S, Storch KF, Linnaranta O. Suicidal Ideation and Insomnia in Bipolar Disorders: Idéation suicidaire et insomnie dans les troubles bipolaires. Can J Psychiatry. 2020;65:802-810. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 138. | Khosravipour M, Khanlari P, Khazaie S, Khosravipour H, Khazaie H. A systematic review and meta-analysis of the association between shift work and metabolic syndrome: The roles of sleep, gender, and type of shift work. Sleep Med Rev. 2021;57:101427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 139. | Liu C, Tang X, Gong Z, Zeng W, Hou Q, Lu R. Circadian Rhythm Sleep Disorders: Genetics, Mechanisms, and Adverse Effects on Health. Front Genet. 2022;13:875342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 140. | Sakurai H, Kato M, Yasui-Furukori N, Suzuki T, Baba H, Watanabe K, Inada K, Kishida I, Sugawara Kikuchi Y, Kikuchi T, Katsuki A, Uchida H; Medical Education Panel of the Japanese Society of Clinical Neuropsychopharmacology. Pharmacological management of bipolar disorder: Japanese expert consensus. Bipolar Disord. 2020;22:822-830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 141. | Yatham LN, Kennedy SH, Parikh SV, Schaffer A, Bond DJ, Frey BN, Sharma V, Goldstein BI, Rej S, Beaulieu S, Alda M, MacQueen G, Milev RV, Ravindran A, O'Donovan C, McIntosh D, Lam RW, Vazquez G, Kapczinski F, McIntyre RS, Kozicky J, Kanba S, Lafer B, Suppes T, Calabrese JR, Vieta E, Malhi G, Post RM, Berk M. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018;20:97-170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1573] [Cited by in RCA: 1239] [Article Influence: 154.9] [Reference Citation Analysis (6)] |
| 142. | Baldessarini RJ, Tondo L, Vázquez GH. Commentary: Lithium treatment for bipolar disorder. Bipolar Disord. 2021;23:93-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 143. | Rybakowski JK. Recent advances in the understanding and management of bipolar disorder in adults. F1000Res. 2017;6:2033. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 144. | Le Clerc S, Lombardi L, Baune BT, Amare AT, Schubert KO, Hou L, Clark SR, Papiol S, Cearns M, Heilbronner U, Degenhardt F, Tekola-Ayele F, Hsu YH, Shekhtman T, Adli M, Akula N, Akiyama K, Ardau R, Arias B, Aubry JM, Backlund L, Bhattacharjee AK, Bellivier F, Benabarre A, Bengesser S, Biernacka JM, Birner A, Brichant-Petitjean C, Cervantes P, Chen HC, Chillotti C, Cichon S, Cruceanu C, Czerski PM, Dalkner N, Dayer A, Del Zompo M, DePaulo JR, Étain B, Jamain S, Falkai P, Forstner AJ, Frisen L, Frye MA, Fullerton JM, Gard S, Garnham JS, Goes FS, Grigoroiu-Serbanescu M, Grof P, Hashimoto R, Hauser J, Herms S, Hoffmann P, Jiménez E, Kahn JP, Kassem L, Kuo PH, Kato T, Kelsoe JR, Kittel-Schneider S, Ferensztajn-Rochowiak E, König B, Kusumi I, Laje G, Landén M, Lavebratt C, Leckband SG, Tortorella A, Manchia M, Martinsson L, McCarthy MJ, McElroy SL, Colom F, Millischer V, Mitjans M, Mondimore FM, Monteleone P, Nievergelt CM, Nöthen MM, Novák T, O'Donovan C, Ozaki N, Ösby U, Pfennig A, Potash JB, Reif A, Reininghaus E, Rouleau GA, Rybakowski JK, Schalling M, Schofield PR, Schweizer BW, Severino G, Shilling PD, Shimoda K, Simhandl C, Slaney CM, Pisanu C, Squassina A, Stamm T, Stopkova P, Maj M, Turecki G, Vieta E, Veeh J, Witt SH, Wright A, Zandi PP, Mitchell PB, Bauer M, Alda M, Rietschel M, McMahon FJ, Schulze TG, Spadoni JL, Boukouaci W, Richard JR, Le Corvoisier P, Barrau C, Zagury JF, Leboyer M, Tamouza R. HLA-DRB1 and HLA-DQB1 genetic diversity modulates response to lithium in bipolar affective disorders. Sci Rep. 2021;11:17823. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 145. | Morton E, Murray G. Assessment and treatment of sleep problems in bipolar disorder-A guide for psychologists and clinically focused review. Clin Psychol Psychother. 2020;27:364-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 146. | Kato T. Current understanding of bipolar disorder: Toward integration of biological basis and treatment strategies. Psychiatry Clin Neurosci. 2019;73:526-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 84] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 147. | Bonfils KA, Novick DM. Application of Interpersonal and Social Rhythm Therapy (IPSRT) for Depression Associated With Schizophrenia Spectrum Disorders. Am J Psychother. 2021;74:127-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 148. | Geoffroy PA, Palagini L. Biological rhythms and chronotherapeutics in depression. Prog Neuropsychopharmacol Biol Psychiatry. 2021;106:110158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 149. | Steardo L Jr, Luciano M, Sampogna G, Zinno F, Saviano P, Staltari F, Segura Garcia C, De Fazio P, Fiorillo A. Efficacy of the interpersonal and social rhythm therapy (IPSRT) in patients with bipolar disorder: results from a real-world, controlled trial. Ann Gen Psychiatry. 2020;19:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 150. | Crowe M, Inder M, Douglas K, Carlyle D, Wells H, Jordan J, Lacey C, Mulder R, Beaglehole B, Porter R. Interpersonal and Social Rhythm Therapy for Patients With Major Depressive Disorder. Am J Psychother. 2020;73:29-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 151. | Moot W, Crowe M, Inder M, Eggleston K, Frampton C, Porter RJ. Domain-Based Functional Improvements in Bipolar Disorder After Interpersonal and Social Rhythm Therapy. Front Psychiatry. 2022;13:767629. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 152. | Orhan M, Korten N, Mans N, van Schaik D, Kupka R, Stek M, Steenhuis D, van Dijk M, Swartz HA, van Oppen P, Dols A. Feasibility and Acceptability of Group Interpersonal and Social Rhythm Therapy for Recurrent Mood Disorders: A Pilot Study. Am J Psychother. 2024;77:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 153. | Crowe M, Porter R, Inder M, Carlyle D, Luty S, Lacey C, Frampton C. Clinical Effectiveness Trial of Adjunctive Interpersonal and Social Rhythm Therapy for Patients With Bipolar Disorder. Am J Psychother. 2020;73:107-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 154. | Swartz HA, Rollman BL, Mohr DC, Sadow S, Frank E. A randomized pilot study of Rhythms And You (RAY): An internet-based program for bipolar disorder administered with and without clinical helper support in primary care. J Affect Disord. 2021;295:183-191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 155. | Douglas KM, Groves S, Crowe MT, Inder ML, Jordan J, Carlyle D, Wells H, Beaglehole B, Mulder R, Lacey C, Luty SE, Eggleston K, Frampton CMA, Bowie CR, Porter RJ. A randomised controlled trial of psychotherapy and cognitive remediation to target cognition in mood disorders. Acta Psychiatr Scand. 2022;145:278-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 156. | Corruble E, Swartz HA, Bottai T, Vaiva G, Bayle F, Llorca PM, Courtet P, Frank E, Gorwood P. Telephone-administered psychotherapy in combination with antidepressant medication for the acute treatment of major depressive disorder. J Affect Disord. 2016;190:6-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 157. | Crowe M, Inder M. Staying well with bipolar disorder: A qualitative analysis of five-year follow-up interviews with young people. J Psychiatr Ment Health Nurs. 2018;25:236-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 158. | Crowe M, Inder M, Swartz HA, Murray G, Porter R. Social rhythm therapy-A potentially translatable psychosocial intervention for bipolar disorder. Bipolar Disord. 2020;22:121-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 159. | Darwish E, Mubarak A, Eissa M, Amer R. Effect of combining social rhythm therapy to treatment of bipolar disorder versus treatment as usual: a comparative study on a sample of Egyptian patients. Middle East Curr Psy. 2024;31:6. [DOI] [Full Text] |
| 160. | Sankar A, Panchal P, Goldman DA, Colic L, Villa LM, Kim JA, Lebowitz ER, Carrubba E, Lecza B, Silverman WK, Swartz HA, Blumberg HP. Telehealth Social Rhythm Therapy to Reduce Mood Symptoms and Suicide Risk Among Adolescents and Young Adults With Bipolar Disorder. Am J Psychother. 2021;74:172-177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 161. | Cuomo A, Carmellini P, Garo ML, Barillà G, Libri C, Spiti A, Goracci A, Bolognesi S, Fagiolini A. Effectiveness of light therapy as adjunctive treatment in bipolar depression: A pilot study. J Affect Disord. 2023;321:102-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 162. | Rosenthal NE, Sack DA, Gillin JC, Lewy AJ, Goodwin FK, Davenport Y, Mueller PS, Newsome DA, Wehr TA. Seasonal affective disorder. A description of the syndrome and preliminary findings with light therapy. Arch Gen Psychiatry. 1984;41:72-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1929] [Cited by in RCA: 1393] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 163. | Morton E, Michalak EE, Levitt A, Levitan RD, Cheung A, Morehouse R, Ramasubbu R, Yatham LN, Tam EM, Lam RW. Quality of Life Impacts of Bright Light Treatment, Fluoxetine, and the Combination in Patients with Nonseasonal Major Depressive Disorder: A Randomized Clinical Trial. Can J Psychiatry. 2021;66:289-297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 164. | Kosanovic Rajacic B, Sagud M, Pivac N, Begic D. Illuminating the way: the role of bright light therapy in the treatment of depression. Expert Rev Neurother. 2023;23:1157-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 165. | Gottlieb JF, Benedetti F, Geoffroy PA, Henriksen TEG, Lam RW, Murray G, Phelps J, Sit D, Swartz HA, Crowe M, Etain B, Frank E, Goel N, Haarman BCM, Inder M, Kallestad H, Jae Kim S, Martiny K, Meesters Y, Porter R, Riemersma-van der Lek RF, Ritter PS, Schulte PFJ, Scott J, Wu JC, Yu X, Chen S. The chronotherapeutic treatment of bipolar disorders: A systematic review and practice recommendations from the ISBD task force on chronotherapy and chronobiology. Bipolar Disord. 2019;21:741-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 123] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 166. | Sit DK, McGowan J, Wiltrout C, Diler RS, Dills JJ, Luther J, Yang A, Ciolino JD, Seltman H, Wisniewski SR, Terman M, Wisner KL. Adjunctive Bright Light Therapy for Bipolar Depression: A Randomized Double-Blind Placebo-Controlled Trial. Am J Psychiatry. 2018;175:131-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 122] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 167. | Legenbauer T, Kirschbaum-Lesch I, Jörke C, Kölch M, Reis O, Berger C, Dück A, Schulte-Markwort M, Becker-Hebly I, Bienioschek S, Schroth J, Ruckes C, Deuster O, Holtmann M. Bright Light Therapy as Add-On to Inpatient Treatment in Youth With Moderate to Severe Depression: A Randomized Clinical Trial. JAMA Psychiatry. 2024;81:655-662. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 168. | Takeshima M, Utsumi T, Aoki Y, Wang Z, Suzuki M, Okajima I, Watanabe N, Watanabe K, Takaesu Y. Efficacy and safety of bright light therapy for manic and depressive symptoms in patients with bipolar disorder: A systematic review and meta-analysis. Psychiatry Clin Neurosci. 2020;74:247-256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 169. | Geoffroy PA, Chevret S, Mauries S, Chaffaut C, Amad A, Bellivier F, Benard V, Courtet P, Dubertret C, Gorwood P, Mazer N, Mekaoui L, Olié E, Pataud G, Vaiva G, Lejoyeux M, Sit D, Maruani J. Bright Light Therapy in the Morning or Midday for the Treatment of Nonseasonal Depression in Bipolar Disorder (LuBi): A Dose-Escalation Phase 1/2 Randomized Double-Blind Trial. J Clin Psychiatry. 2025;86:25m15826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 170. | Benedetti F. Rate of switch from bipolar depression into mania after morning light therapy: A historical review. Psychiatry Res. 2018;261:351-356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 171. | Lambert GW, Reid C, Kaye DM, Jennings GL, Esler MD. Effect of sunlight and season on serotonin turnover in the brain. Lancet. 2002;360:1840-1842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 465] [Cited by in RCA: 328] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 172. | Sit D, Haigh S. Use of "Lights" for Bipolar Depression. Curr Psychiatry Rep. 2019;21:45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 173. | Cowen PJ, Browning M. What has serotonin to do with depression? World Psychiatry. 2015;14:158-160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 289] [Cited by in RCA: 218] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 174. | Takahashi T, Sasabayashi D, Yücel M, Whittle S, Lorenzetti V, Walterfang M, Suzuki M, Pantelis C, Malhi GS, Allen NB. Pineal Gland Volume in Major Depressive and Bipolar Disorders. Front Psychiatry. 2020;11:450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 175. | Kaçar A, Karakuş OB, Aydın ZE, Adak İ. Effectiveness of Agomelatine in Generalized Anxiety Disorder Comorbid to Bipolar 1 Disorder in a Male Adolescent Patient. Clin Neuropharmacol. 2024;47:143-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 176. | McGowan NM, Kim DS, de Andres Crespo M, Bisdounis L, Kyle SD, Saunders KEA. Hypnotic and Melatonin/Melatonin-Receptor Agonist Treatment in Bipolar Disorder: A Systematic Review and Meta-Analysis. CNS Drugs. 2022;36:345-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 177. | Stein DJ. Evidence-Based Pharmacotherapy of Anxiety Symptoms in Patients with Major Depressive Disorder: Focus on Agomelatine. Neurol Ther. 2023;12:13-19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 178. | Li J, Luo H, Luo Q. Agomelatine bears promising potential in treating bipolar depression- a systematic review. Int J Psychiatry Clin Pract. 2024;28:189-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 179. | Kaplan KA. Sleep and sleep treatments in bipolar disorder. Curr Opin Psychol. 2020;34:117-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 180. | Chevance A, Ravaud P, Tomlinson A, Le Berre C, Teufer B, Touboul S, Fried EI, Gartlehner G, Cipriani A, Tran VT. Identifying outcomes for depression that matter to patients, informal caregivers, and health-care professionals: qualitative content analysis of a large international online survey. Lancet Psychiatry. 2020;7:692-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 138] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 181. | Gordon-Smith K, Saunders KE, Savage J, Craddock N, Jones I, Jones L. Have I argued with my family this week? J Affect Disord. 2021;281:918-925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 182. | Pflug B, Tölle R. Disturbance of the 24-hour rhythm in endogenous depression and the treatment of endogenous depression by sleep deprivation. Int Pharmacopsychiatry. 1971;6:187-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 96] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 183. | Danilenko KV, Lebedinskaia MY, Gadetskaia EV, Markov AA, Ivanova YA, Aftanas LI. A 6-day combined wake and light therapy trial for unipolar depression. J Affect Disord. 2019;259:355-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 184. | Wirz-Justice A, Benedetti F. Perspectives in affective disorders: Clocks and sleep. Eur J Neurosci. 2020;51:346-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 70] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 185. | Boland EM, Rao H, Dinges DF, Smith RV, Goel N, Detre JA, Basner M, Sheline YI, Thase ME, Gehrman PR. Meta-Analysis of the Antidepressant Effects of Acute Sleep Deprivation. J Clin Psychiatry. 2017;78:e1020-e1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 91] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 186. | Mitter P, De Crescenzo F, Loo Yong Kee K, Xia J, Roberts S, Chi W, Kurtulmus A, Kyle SD, Geddes JR, Cipriani A. Sleep deprivation as a treatment for major depressive episodes: A systematic review and meta-analysis. Sleep Med Rev. 2022;64:101647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 187. | Sarzetto A, Cavallini MC, Fregna L, Pacchioni F, Attanasio F, Barbini B, Franchini L, Colombo C. Sleep architecture modifications after double chronotherapy: A case series of bipolar depressed inpatients. Psychiatry Res. 2022;316:114781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 188. | Humpston C, Benedetti F, Serfaty M, Markham S, Hodsoll J, Young AH, Veale D. Chronotherapy for the rapid treatment of depression: A meta-analysis. J Affect Disord. 2020;261:91-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 189. | Gottlieb JF, Goel N, Chen S, Young MA. Meta-analysis of sleep deprivation in the acute treatment of bipolar depression. Acta Psychiatr Scand. 2021;143:319-327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 190. | Ramirez-Mahaluf JP, Rozas-Serri E, Ivanovic-Zuvic F, Risco L, Vöhringer PA. Effectiveness of Sleep Deprivation in Treating Acute Bipolar Depression as Augmentation Strategy: A Systematic Review and Meta-Analysis. Front Psychiatry. 2020;11:70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 191. | Fabbri C. The Role of Genetics in Bipolar Disorder. Curr Top Behav Neurosci. 2021;48:41-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 192. | Chung J, Kim YC, Jeong JH. Bipolar Disorder, Circadian Rhythm and Clock Genes. Clin Psychopharmacol Neurosci. 2024;22:211-221. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 193. | Daudali H, Anderson J, Bailey MES, Fradera A, Niedzwiedz CL, Lyall D, Lyall LMM, Strawbridge RJ. Genetic variation in circadian regulator gene BMAL1 in psychiatric, psychological and cardiometabolic traits: a trans-ancestry UK Biobank study. BMJ Ment Health. 2024;27:e301267. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 194. | Zhang C, Ni P, Liang S, Li X, Tian Y, Du X, Wei W, Meng Y, Wei J, Ma X, Deng W, Guo W, Li M, Yu H, Zhao L, Wang Q, Pak SC, Li T. Alterations in CRY2 and PER3 gene expression associated with thalamic-limbic community structural abnormalities in patients with bipolar depression or unipolar depression. J Affect Disord. 2022;298:472-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 195. | Li T, Ding Y, Zhang L, Li H, Liu F, Li P, Zhao J, Lv D, Lang B, Guo W. Potential associations between altered brain function, cognitive deficits and gene expressing profiles in bipolar disorder across three clinical stages. J Affect Disord. 2025;374:606-615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 196. | Bi B, Che D, Bai Y. Neural network of bipolar disorder: Toward integration of neuroimaging and neurocircuit-based treatment strategies. Transl Psychiatry. 2022;12:143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 75] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 197. | Jansen K, Grellert M, Nexha A, Oses JP, Silva RAD, Souza LDM, Kapczinski F, Frey BN, de Azevedo Cardoso T. Biological rhythms disruption mediates the association between mother's diagnosis of bipolar disorder and offspring's emotional/behavioral problems. J Affect Disord. 2023;327:230-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 198. | Yadav N, Zhang Q, Qi D, Yadav A, Zheng H. AI-integrated wearable strain sensors: advances in e-skin, robotics, and personalized health monitoring. Nanoscale Adv. 2025;7:4803-4819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 199. | Dunster GP, Swendsen J, Merikangas KR. Real-time mobile monitoring of bipolar disorder: a review of evidence and future directions. Neuropsychopharmacology. 2021;46:197-208. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 200. | Song YM, Jeong J, de Los Reyes AA 5th, Lim D, Cho CH, Yeom JW, Lee T, Lee JB, Lee HJ, Kim JK. Causal dynamics of sleep, circadian rhythm, and mood symptoms in patients with major depression and bipolar disorder: insights from longitudinal wearable device data. EBioMedicine. 2024;103:105094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 34] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 201. | Yeom JW, Yoon Y, Seo JY, Cho CH, Lee T, Lee JB, Jeon S, Kim L, Lee HJ. Daily Self-Monitoring and Feedback of Circadian Rhythm Measures in Major Depression and Bipolar Disorder Using Wearable Devices and Smartphones-The Circadian Rhythm for Mood (CRM®) Trial Protocol: A Randomized Sham Controlled Double-Blind Trial. Psychiatry Investig. 2024;21:918-924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 202. | Lim D, Jeong J, Song YM, Cho CH, Yeom JW, Lee T, Lee JB, Lee HJ, Kim JK. Accurately predicting mood episodes in mood disorder patients using wearable sleep and circadian rhythm features. NPJ Digit Med. 2024;7:324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 39] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 203. | Zhao X, Hirota T, Han X, Cho H, Chong LW, Lamia K, Liu S, Atkins AR, Banayo E, Liddle C, Yu RT, Yates JR 3rd, Kay SA, Downes M, Evans RM. Circadian Amplitude Regulation via FBXW7-Targeted REV-ERBα Degradation. Cell. 2016;165:1644-1657. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 133] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 204. | Chung S, Lee EJ, Yun S, Choe HK, Park SB, Son HJ, Kim KS, Dluzen DE, Lee I, Hwang O, Son GH, Kim K. Impact of circadian nuclear receptor REV-ERBα on midbrain dopamine production and mood regulation. Cell. 2014;157:858-868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 249] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 205. | Gomatou G, Karachaliou A, Veloudiou OZ, Karvela A, Syrigos N, Kotteas E. The Role of REV-ERB Receptors in Cancer Pathogenesis. Int J Mol Sci. 2023;24:8980. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 206. | Luo X, Song S, Qi L, Tien CL, Li H, Xu W, Mathuram TL, Burris T, Zhao Y, Sun Z, Zhang L. REV-ERB is essential in cardiac fibroblasts homeostasis. Front Pharmacol. 2022;13:899628. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 207. | Makhija S, Griffett JD, Veerakanellore GB, Burris TP, Elgendy B, Griffett K. REV-ERB activation as a novel pharmacological approach for treating inflammatory pain. Front Pharmacol. 2023;14:1171931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 208. | Adlanmerini M, Krusen BM, Nguyen HCB, Teng CW, Woodie LN, Tackenberg MC, Geisler CE, Gaisinsky J, Peed LC, Carpenter BJ, Hayes MR, Lazar MA. REV-ERB nuclear receptors in the suprachiasmatic nucleus control circadian period and restrict diet-induced obesity. Sci Adv. 2021;7:eabh2007. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 209. | Wang C, Yang J, Yuan J, Wang X, Li Q, Ren C, Zhi X, Lv X, Liu K, Zhao X, Li Y. Role of circadian transcription factor REV-ERB in cardiovascular diseases: a review. Front Cardiovasc Med. 2025;12:1516279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 210. | Raza GS, Sodum N, Kaya Y, Herzig KH. Role of Circadian Transcription Factor Rev-Erb in Metabolism and Tissue Fibrosis. Int J Mol Sci. 2022;23:12954. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 211. | Solt LA, Wang Y, Banerjee S, Hughes T, Kojetin DJ, Lundasen T, Shin Y, Liu J, Cameron MD, Noel R, Yoo SH, Takahashi JS, Butler AA, Kamenecka TM, Burris TP. Regulation of circadian behaviour and metabolism by synthetic REV-ERB agonists. Nature. 2012;485:62-68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 739] [Cited by in RCA: 691] [Article Influence: 49.4] [Reference Citation Analysis (0)] |
| 212. | Cho H, Zhao X, Hatori M, Yu RT, Barish GD, Lam MT, Chong LW, DiTacchio L, Atkins AR, Glass CK, Liddle C, Auwerx J, Downes M, Panda S, Evans RM. Regulation of circadian behaviour and metabolism by REV-ERB-α and REV-ERB-β. Nature. 2012;485:123-127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 970] [Cited by in RCA: 872] [Article Influence: 62.3] [Reference Citation Analysis (0)] |
| 213. | Liu YN, Wang QW, She XY, Li LJ, Wang B, Yang R, Li Q, Lu SY, Wang YH, Shen W, Fu CL, Li D, Yi L, Wang CX, Shi W, Cheng X, Cao L, Mi S, Yao J. A pancreas-hippocampus feedback mechanism regulates circadian changes in depression-related behaviors. Nat Neurosci. 2025;28:2078-2091. [PubMed] [DOI] [Full Text] |
| 214. | Yi JS, Díaz NM, D'Souza S, Buhr ED. The molecular clockwork of mammalian cells. Semin Cell Dev Biol. 2022;126:87-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 215. | He B, Nohara K, Park N, Park YS, Guillory B, Zhao Z, Garcia JM, Koike N, Lee CC, Takahashi JS, Yoo SH, Chen Z. The Small Molecule Nobiletin Targets the Molecular Oscillator to Enhance Circadian Rhythms and Protect against Metabolic Syndrome. Cell Metab. 2016;23:610-621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 468] [Cited by in RCA: 418] [Article Influence: 41.8] [Reference Citation Analysis (0)] |
| 216. | Porcu A, Gonzalez R, McCarthy MJ. Pharmacological Manipulation of the Circadian Clock: A Possible Approach to the Management of Bipolar Disorder. CNS Drugs. 2019;33:981-999. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 217. | Meléndez-Fernández OH, Liu JA, Nelson RJ. Circadian Rhythms Disrupted by Light at Night and Mistimed Food Intake Alter Hormonal Rhythms and Metabolism. Int J Mol Sci. 2023;24:3392. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 176] [Cited by in RCA: 120] [Article Influence: 40.0] [Reference Citation Analysis (0)] |
| 218. | Walker WH 2nd, Bumgarner JR, Becker-Krail DD, May LE, Liu JA, Nelson RJ. Light at night disrupts biological clocks, calendars, and immune function. Semin Immunopathol. 2022;44:165-173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 219. | Geoffroy PA, Maruani J. Chronobiology of Mood Disorders: The Role of the Biological Clock in Depression and Bipolar Disorder. Biol Psychiatry. 2025;98:830-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 220. | Liu D, Zhang M, Ding L, Huang J, Wang Y, Su Y, Chen Z, Cai Y, He S, Peng D. Relationship between biological rhythm dysregulation and suicidal ideation in patients with major depressive disorder. BMC Psychiatry. 2024;24:87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 221. | Takaesu Y. Circadian rhythm in bipolar disorder: A review of the literature. Psychiatry Clin Neurosci. 2018;72:673-682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 134] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 222. | Hennion V, Scott J, Martinot V, Godin O, Marie-Claire C, Bellivier F, Jamain S, Etain B. Polygenic risk scores for mood disorders and actigraphy estimates of sleep and circadian rhythms: A preliminary study in bipolar disorders. J Sleep Res. 2025;34:e14307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 223. | Ashton A, Jagannath A. Disrupted Sleep and Circadian Rhythms in Schizophrenia and Their Interaction With Dopamine Signaling. Front Neurosci. 2020;14:636. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 69] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 224. | Lesobre M, Bonilla A, Foucher A, Calvet B, Lacroix A, Charles É. Bipolar disorders in the rhythm of day and seasons: Link between bipolarity and seasonal affective disorder. Chronobiol Int. 2025;42:440-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 225. | Yeom JW, Park S, Lee HJ. Managing Circadian Rhythms: A Key to Enhancing Mental Health in College Students. Psychiatry Investig. 2024;21:1309-1317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 226. | Tonon AC, Nexha A, Mendonça da Silva M, Gomes FA, Hidalgo MP, Frey BN. Sleep and circadian disruption in bipolar disorders: From psychopathology to digital phenotyping in clinical practice. Psychiatry Clin Neurosci. 2024;78:654-666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/