Published online Mar 19, 2026. doi: 10.5498/wjp.v16.i3.112022
Revised: July 27, 2025
Accepted: November 12, 2025
Published online: March 19, 2026
Processing time: 227 Days and 23.6 Hours
Per- and polyfluoroalkyl substances (PFAS) are persistent environmental contaminants increasingly associated with adverse health outcomes, including psy
To answer the following two critical questions: What key findings regarding the impacts of PFAS exposure on psychiatric outcomes have emerged in the past five years? What research trends currently prevail in terms of methodologies, targeted populations, and mechanistic investigations?
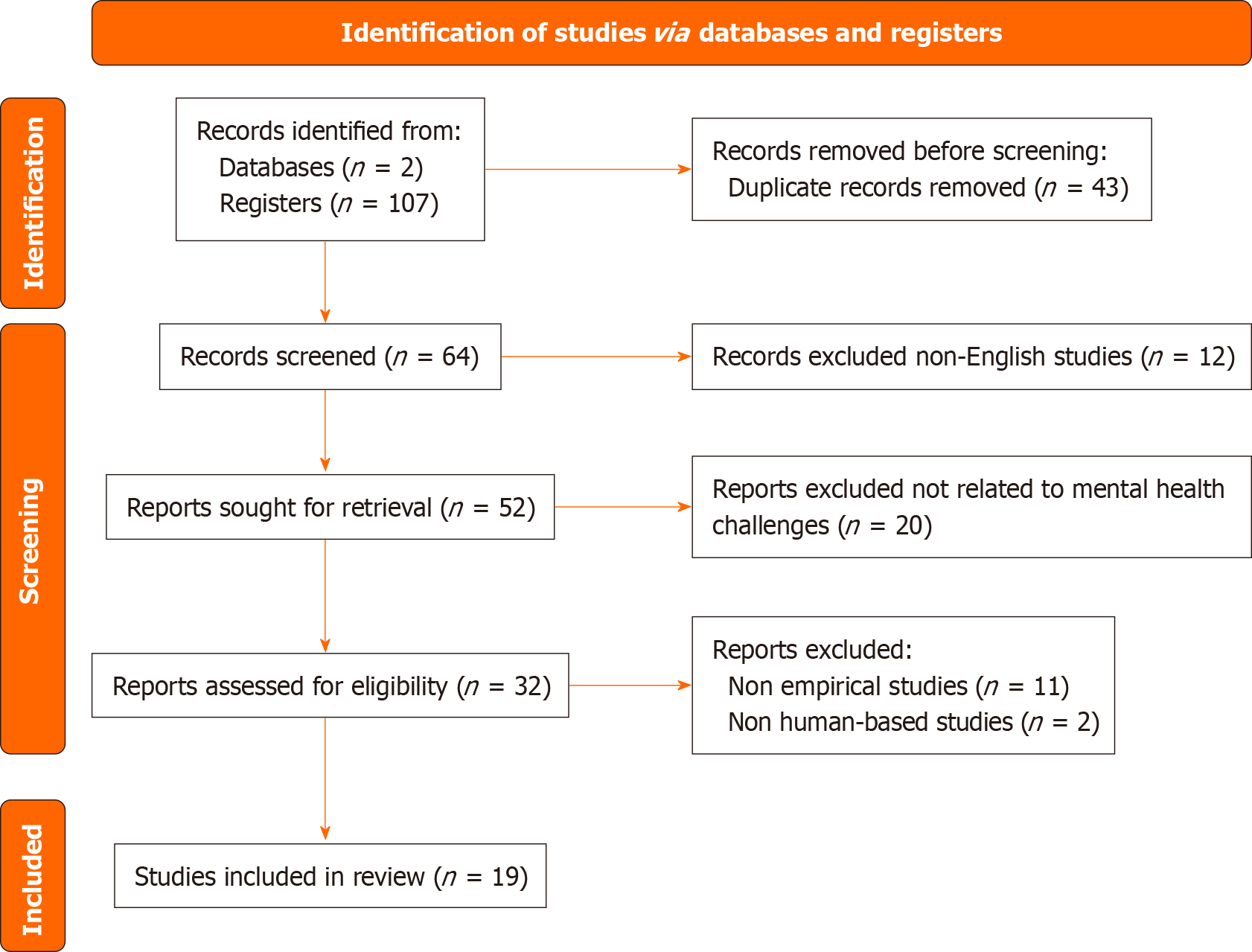
A systematic search guided by the PRISMA framework was conducted using Web of Science and Scopus, covering studies published between July 2021 and July 2025. Nineteen eligible studies were identified and thematically analyzed.
The article found increasing evidence linking PFAS exposure to psychiatric conditions, including depression, attention-deficit/hyperactivity disorder, autism spectrum disorder, and cognitive impairments. Proposed mechanisms include thyroid hormone disruption, neuroinflammation, and increased blood-brain barrier permeability. However, inconsistencies in findings - stemming from vari
Despite growing evidence of PFAS-related neuropsychiatric risks, current res
Core Tip: This article synthesizes recent evidence linking per- and polyfluoroalkyl substances (PFAS) exposure to psychiatric disorders. It highlights the growing rele
- Citation: Jiang YL, Samah NA, Xiao ZS. Forever chemicals and mental health: A systematic review of per- and polyfluoroalkyl substances-related psychiatric outcomes and mechanistic insights. World J Psychiatry 2026; 16(3): 112022
- URL: https://www.wjgnet.com/2220-3206/full/v16/i3/112022.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i3.112022
Per- and polyfluoroalkyl substances (PFAS) comprise a diverse class of synthetic chemicals defined by strong carbon-fluorine bonds. This group includes thousands of compounds, notably perfluorooctanoic acid (PFOA), perfluorooctane sulfonate (PFOS), and shorter-chain alternatives such as perfluorobutanoic acid and perf
As a result, PFAS are now globally ubiquitous found in water, soil, air, and living organisms, even in remote locations such as Antarctica leading to widespread human exposure at various concentrations[3,6]. Although some developed regions, including the European Union and the United States, have implemented regulations limiting specific PFAS, the lack of a coordinated global framework permits continued environmental release and accumulation[7]. Amid rising global concern over environmental health risks, the psychiatric implications of PFAS exposure have gained renewed att
For the past two decades, PFAS research has largely concentrated on their physiological toxicities, particularly their impacts on the liver, kidneys, and immune system[3,5,8]. A growing body of toxicological and epidemiological evidence has linked PFAS exposure to a range of systemic health impairments, including liver dysfunction, kidney damage, immune suppression, endocrine disruption particularly thyroid hormone imbalances and reproductive abnormalities[2,4,9]. Notably, PFAS possess strong bioaccumulative properties, allowing them to magnify through food chains and create chronic exposure risks - now recognized as a serious global public health concern[6,10]. Accordingly, the International Agency for Research on Cancer has classified PFOA as a group 2B carcinogen, meaning it is “possibly carcinogenic to humans”[11]. Importantly, the potential harms of PFAS extend well beyond their physiological impacts, prompting growing concern about their neurological and psychiatric effects.
As scientific understanding deepens, increasing attention has been directed toward the potential neurotoxic effects of PFAS on the central nervous system[12]. Emerging evidences suggest that PFAS exposure is associated with elevated risks of neuropsychiatric and neurodevelopmental disorders, including depression, anxiety, attention-deficit/hyperactivity disorder (ADHD), and autism spectrum disorder (ASD). These associations are particularly pronounced among children and adolescents, who are especially susceptible during key neurodevelopmental windows[13]. Such findings have broadened the scope of PFAS-related health research and underscore the importance of incorporating psychiatric outcomes into future risk assessment frameworks.
Recent investigations into the mechanisms underlying PFAS-related psychiatric outcomes have proposed several key biological pathways: First, PFAS exposure has been shown to disrupt thyroid hormone synthesis and regulation processes that are critical for brain development during fetal and early childhood stages potentially impairing cognitive functions[14]. Second, PFAS-induced systemic inflammation and alterations in neurotransmitter systems particularly in serotonin and dopamine pathways critical for mood regulation may increase the risk of developing depression and anxiety disorders[15]. Third, prenatal PFAS exposure may allow these substances to cross the placental barrier, potentially disrupting central nervous system development and contributing to cognitive and behavioral impairments in early childhood[16]. Furthermore, the timing of PFAS exposure plays a crucial role in shaping psychiatric outcomes. Fetal and early childhood periods represent sensitive windows of brain development, marked by heightened plasticity and vulnerability to environmental insults[17]. Exposure during these critical windows may lead to irreversible alterations in neural structure and function, manifesting as persistent psychological and behavioral disorders extending into adole
Against this backdrop, the psychiatric impacts of PFAS exposure have emerged as a growing focus within psychiatric outcomes research. An increasing volume of empirical research suggests potential associations between PFAS exposure and a range of psychiatric outcomes particularly among vulnerable populations including children, adolescents, and pregnant women[11,13,14,18]. However, as previously noted, this remains a relatively nascent field of study, with many critical questions yet to be explored and validated. Therefore, a comprehensive synthesis of current evidence is essential not only to chart the existing research landscape and identify promising avenues, but also to inform future psychiatric research and support the formulation of robust, evidence-based environmental health policies.
This review addresses two central research questions: (1) What key findings regarding the impacts of PFAS exposure on psychiatric outcomes have emerged in the past five years; and (2) What research trends currently prevail in terms of methodologies, targeted populations, and mechanistic investigations?
By addressing these questions, this review aims to provide a structured synthesis to inform future psychiatric research, enhance understanding of PFAS-related psychiatric outcomes, and support translational efforts linking environmental exposure to psychiatric outcomes.
To establish a clear methodological framework, this review followed the PRISMA guidelines and employed a Boolean search strategy across two major databases: Web of Science and Scopus. The search query was designed to capture literature at the intersection of PFAS exposure and psychiatric outcomes, using the following terms: (“PFAS” OR “Per- and Polyfluoroalkyl Substances” OR “PFOA” OR “Perfluorooctanoic acid” OR “PFOS” OR “Perfluorooctane sulfonate” OR “PFNA” OR “Perfluorononanoic acid” OR “PFHxS” OR “Perfluorohexane sulfonic acid”) AND (“mental health” OR “mental disorder” OR “mental conditions” OR “psychiatry”)[19]. The search covered publications from July 2021 to July 2025.
During the screening process, we strictly adhered to PRISMA procedures, clearly outlining inclusion and exclusion criteria to ensure transparency and reproducibility. Studies were included if they met the following criteria: Peer-reviewed original empirical research; published in English; directly examined associations between PFAS exposure and psychological or neurobehavioral outcomes. Exclusion criteria comprised review articles, commentaries, and studies not involving human participants. The selection process is summarized in Figure 1. While a formal risk-of-bias assessment tool was not employed due to the qualitative approach nature of this review, potential sources of bias were mitigated through independent screening by multiple reviewers and consensus resolution of discrepancies. We acknowledge the possibility of publication bias and language bias, and highlight this as a limitation in the discussion.
A thematic analysis approach was employed to synthesize findings across studies. Following Braun and Clarke’s (2006) framework, the process involved six steps: (1) Familiarization with the data through repeated reading; (2) Generating initial codes by identifying patterns related to PFAS exposure and psychiatric effects; (3) Searching for themes across coded data, such as “depression risk”, “prenatal exposure effects”, “endocrine disruption”, and “neuroinflammation”; (4) Reviewing themes for coherence and consistency; (5) Defining and naming themes to reflect key questions; and (6) Producing the report by integrating themes into a coherent narrative supported by representative study findings[20]. This approach allowed for the identification of recurring patterns, conceptual linkages, and emerging areas of consensus or controversy in the current literature. The analysis prioritized both the epidemiological associations and the underlying mechanistic explanations proposed in the studies.
After rigorous screening, a total of 19 studies were included in the analysis of this study, as shown in Table 1[21-39]. In terms of research design, the majority of studies (n = 13, 68.4%) employed a longitudinal cohort approach. These studies primarily investigated the long-term effects of PFAS exposure on various neurodevelopmental and psychiatric con
| Ref. | Research design | Sample size | Outcomes focus |
| Ahrens et al[21], 2024 | Longitudinal cohort study | 16440 | ASD |
| Carrizosa et al[22], 2021 | Longitudinal cohort study | 1240 | Neuropsychological development |
| Tang et al[23], 2024 | Cross-sectional observational study | 7301 | Depression |
| Dalsager et al[24], 2021 | Longitudinal cohort study | 1138 | ADHD |
| England-Mason et al[25], 2025 | Retrospective machine learning analysis | 406 | Neurodevelopment |
| Hong et al[26], 2024 | Cross-sectional study with paired subjects | 59 | Cognitive impairment |
| Hu et al[27], 2024 | Longitudinal cohort study | 50 | Depression |
| Itoh et al[28], 2022 | Longitudinal cohort study | 770 | ADHD |
| Itoh et al[29], 2025 | Longitudinal cohort study | 15131 | Neuropsychological development |
| Kim et al[30], 2023 | Longitudinal cohort study | 221 | Postpartum depression, Infant neurodevelopment |
| Kim et al[31], 2023 | Longitudinal cohort study | 521 | ADHD |
| Lefèvre-Arbogast et al[32], 2024 | Longitudinal cohort study | 1288 | Dementia |
| Oh et al[33], 2021 | Longitudinal cohort study | 173 | ASD |
| Ogundare and Obeng-Gyasi[34], 2025 | Cross-sectional observational study | 181 | Depression |
| Sun et al[35], 2024 | Cross-sectional observational study | 7700 | Depression |
| Tillaut et al[36], 2023 | Longitudinal cohort study | 444 | ADHD, depression, anxiety |
| Yao et al[37], 2022 | Longitudinal cohort study | 274 | Neuropsychological development |
| Wu et al[38], 2024 | Experimental in vitro study | 3-6 biological replicates | Alzheimer’s disease |
| Zhang et al[39], 2025 | Longitudinal cohort study | 1779 | Neurodevelopment |
Cross-sectional observational studies comprised 4 out of 19 studies (21.1%), generally focusing on depressive sym
Only one study (5.3%) utilized an in vitro experimental design, contributing mechanistic insights by exploring molecular and cellular changes induced by PFAS exposure under controlled laboratory conditions.
Similarly, one study (5.3%) applied a retrospective machine learning approach, using predictive modeling techniques to examine the relationship between PFAS exposure and neurodevelopmental outcomes. This computational method represents an emerging trend in environmental health research, offering new possibilities for analyzing complex exp
Overall, the dominance of longitudinal designs indicates a growing interest in developmental trajectories and delayed effects, while the inclusion of experimental and computational studies highlights the importance of mechanistic exploration and data-driven modeling in this evolving field. The key findings regarding the impacts of PFAS exposure on psychiatric outcomes which have emerged in the past five years are as follows.
Recent studies examining the impacts of PFAS exposure on depression and mood disorders have presented mixed results. A study analyzing the National Health and Nutrition Examination Survey (NHANES) data from 2005 to 2018 suggested that higher exposure to specific PFAS compounds, such asPFOA, perfluorohexane sulfonic acid (PFHxS), perfluoroundecanoic acid was associated with a lower risk of depressive symptoms; however, the cross-sectional design limited causal interpretation[35]. In contrast, another NHANES-based study identified cadmium as a significant factor contributing to depression, finding that PFAS demonstrated antagonistic interactions when co-exposed with heavy metals, reducing their combined depressive effects[23]. Additionally, research on postpartum depression (PPD) in women carrying twins indicated that prenatal exposure to PFAS mixtures significantly increased the risk of PPD, particularly at 1 month and 6 months postpartum[27].
Findings on PFAS exposure and neurodevelopmental outcomes also vary. Exposure to PFAS during early childhood (at around 2 years of age) was linked to increased ADHD symptoms at age 8, particularly at lower to moderate exposure levels[30]. However, several other studies found no significant associations between prenatal or early-childhood PFAS exposure and ADHD symptoms, highlighting inconsistencies in this area[24,28]. Further, prenatal exposure to PFOA and PFNA was associated with heightened ASD risk in specific high-risk cohorts, whereas PFHxS exhibited an inverse relationship[33]. Additionally, prenatal PFAS exposure correlated with reduced gross motor development in infants, notably when mothers experienced elevated psychological distress[39]. Exposure to perfluorobutane sulfonate and PFHxS was similarly linked to decreased neurodevelopmental scores in infants, partly mediated by disruptions in thyroid hormone regulation[37].
Mechanistic studies have identified potential biological pathways through which PFAS may influence psychiatric outcomes. Thyroid hormone disruption emerged as a significant mediator, with prenatal PFAS exposure altering hormone levels and consequently impacting neurodevelopment[28,29,37]. Neurotoxic effects were also observed; for example, PFOA exposure in human-derived cortical neurons triggered pathways analogous to Alzheimer’s disease, including tau protein phosphorylation and related transcriptomic changes[38]. Additionally, increased permeability of the blood-brain barrier to PFOS, leading to cognitive impairments, underscored the direct neurotoxic potential of PFAS[26].
Emerging evidence highlights the complex interactions between PFAS and other environmental or social determinants. Co-exposure to cadmium and PFOS demonstrated antagonistic interactions affecting depression-related biological pathways, suggesting intricate effects dependent on environmental contaminant mixtures[23]. Dietary patterns, speci
Despite accumulating evidence, some studies reported no significant relationships between PFAS exposure and neurodevelopmental outcomes, reflecting ongoing uncertainties. For instance, investigations into prenatal or childhood PFAS exposure sometimes showed no correlation with ADHD or other neurodevelopmental indicators[22,29]. These inconsistencies underscore critical gaps in understanding, emphasizing the need for further research on the timing of exposure, individual susceptibility, and the complex interplay of contaminant mixtures. Research trends currently prevailing in terms of methodologies, targeted populations, and mechanistic investigations are as follows.
Recent studies on PFAS and psychiatric outcomes have embraced increasingly sophisticated methodologies to better understand complex exposure-outcome relationships. One prominent advancement involves the use of mixture analysis and multi-pollutant models. Techniques such as Bayesian Kernel Machine Regression and quantile g-computation are now commonly used to assess the combined effects of PFAS mixtures, often in the context of co-exposure to metals and sociodemographic modifiers[23,35]. Furthermore, machine learning approaches are emerging as powerful tools for modeling the intricate interactions among PFAS, other contaminants, and individual- or population-level risk factors[25].
In terms of study design, there has been a clear trend toward longitudinal and cohort-based methodologies. Large-scale birth cohorts, such as the INfancia y Medio Ambiente Project, Hokkaido Study, and Shanghai Birth Cohort, have allowed researchers to track prenatal PFAS exposure and subsequent neurodevelopmental outcomes across several years[22,28,39]. While cross-sectional studies like NHANES remain widely used, they are increasingly complemented by designs incorporating retrospective exposure assessment or repeat sampling over time[35].
The integration of biomarkers and multi-omics data has also enhanced mechanistic understanding. Analyses of cord blood, serum, and cerebrospinal fluid have facilitated the quantification of PFAS penetration into neurological tissues[26]. In parallel, transcriptomic and metabolomic analyses have elucidated molecular mechanisms - such as tau phosph
Research has increasingly focused on populations that are especially susceptible to the psychiatric outcomes of PFAS exposure. Prenatal and early-life exposure remains a key area of concern, with numerous studies examining pregnant women and infants due to their heightened vulnerability to hormonal and neurodevelopmental disruptions[27,37]. High-risk cohorts such as MARBLES (for autism risk) and the Odense Child Cohort (for ADHD) have been instrumental in identifying specific neurodevelopmental impacts of early PFAS exposure[24,33].
Vulnerable subgroups have also been prioritized in recent research. For instance, stronger associations between PFAS exposure and PPD have been reported in women with twin pregnancies[27]. Populations with low socioeconomic status or high-fat dietary intake appear more susceptible to PFAS-related neurotoxic effects, likely due to both higher exposure levels and reduced physiological or social resilience[32,34].
Adult and aging populations are increasingly represented in epidemiological studies as well. Data from NHANES and other national surveys have helped identify associations between PFAS exposure and depression in adults[23,35]. Research is also extending into geriatric contexts, with some studies examining dietary PFAS exposure and its potential link to dementia risk in older adults[32].
Mechanistic research has provided critical insights into how PFAS exposure may lead to mental health impairments. One prominent pathway involves thyroid hormone disruption: Prenatal exposure to PFAS has been shown to alter levels of free thyroxine, free triiodothyronine, and thyroid-stimulating hormone, which in turn are associated with developmental deficits[28,37].
In addition to endocrine disruption, PFAS compounds have been implicated in neuroinflammatory and neurodegenerative processes. For instance, exposure to PFOA in human pluripotent stem cell-derived cortical neurons has been shown to induce Alzheimer’s-like pathology, including tau aggregation and calcium signaling dysregulation[38]. PFOS has been found in cerebrospinal fluid and is linked to impaired cognitive function, highlighting its potential to cross the blood-brain barrier and affect brain health directly[26].
Emerging research also suggests that PFAS may exert epigenetic and multi-generational effects. Animal and in vitro models have indicated that PFAS exposure may disrupt the hypothalamic-pituitary-adrenal axis and impair synaptic plasticity[38]. Furthermore, maternal stress appears to interact synergistically with PFAS exposure, compounding neuro
This discussion synthesizes current findings on the psychiatric implications of PFAS exposure, critically evaluates methodological and conceptual trends in the literature, and outlines directions for future research and policy deve
Current methodological approaches rely heavily on observational designs, which constrain causal inference and limit temporal resolution. Most studies investigating PFAS and psychiatric outcomes employ cross-sectional surveys or prospective birth cohorts. While these approaches offer initial insights into associations, they are often hampered by residual confounding, misclassification of exposure, and limited capacity to capture long-term effects. Advanced statistical models - such as Bayesian kernel machine regression, quantile g-computation, and machine learning - have been introduced to manage complex mixture exposures[23,25]. However, methodological challenges persist, particularly the reliance on single-timepoint serum measurements and inconsistent biomarker use, which obscure cumulative exp
The current research agenda focuses narrowly on selected psychiatric endpoints and underrepresents both vulnerable populations and dimensional symptomatology. Most studies concentrate on clinically defined outcomes such as ADHD, ASD, and depression, often overlooking intermediate cognitive and affective features such as executive dysfunction, irritability, or social cognition deficits[27,33,35]. This diagnostic approach, while pragmatic, may not fully capture the spectrum of neurobehavioral effects associated with PFAS exposure. Additionally, while children, pregnant women, and adolescents receive appropriate attention, other vulnerable groups - including the elderly, occupationally exposed indi
Future research must adopt integrative, developmentally sensitive, and mechanistically grounded frameworks. Longitudinal cohort designs with multi-wave follow-ups and repeated biomarker assessments are needed to trace the neurobehavioral trajectories associated with PFAS from prenatal stages through adolescence and adulthood. Transitioning from categorical diagnoses to dimensional models, such as the National Institute of Mental Health’s Research Domain Criteria, would enable a more precise mapping of PFAS effects onto fundamental mental processes, such as attention regulation, emotional response, and cognitive flexibility[39]. At the mechanistic level, integrating multi-omics (e.g., epigenomics, metabolomics), neuroimaging, and toxicokinetic modeling will help uncover the molecular pathways and brain network disruptions underlying psychiatric risk. Equally important, future research must address PFAS mixtures in the context of co-exposures (e.g., heavy metals, endocrine disruptors) and psychosocial stressors - factors likely to interact in shaping psychiatric vulnerability.
Environmental justice and translational impact should guide the evolving research agenda. Studies must proactively include diverse global populations and stratify results by social determinants such as income, occupation, and geographic vulnerability. Importantly, research must move beyond exposure characterization toward testing interventions. Clinical and community-based trials evaluating whether reducing PFAS exposure - via dietary interventions, water filtration, or pharmacological modulation of hormonal and inflammatory pathways - can improve psychiatric outcomes are critically needed. These efforts will bridge environmental science with psychiatric care and yield actionable insights for at-risk populations.
The potential psychiatric burden of PFAS exposure calls for a reevaluation of current regulatory and public health strategies. Current safety thresholds predominantly emphasize hepatic, reproductive, and immunological endpoints, frequently overlooking critical neurobehavioral effects. Our findings highlight that PFAS exposure, particularly involving PFHxS and PFOS, is linked to early-life motor delays detectable as early as 14 months, and sex-specific impairments in working memory evident by ages 4-5 both representing essential neurodevelopmental domains during sensitive developmental windows[26,28,35,37,39]. These cognitive and motor domains serve as significant predictors of later psychiatric outcomes, including ADHD, mood disorders, and executive dysfunction. However, major regulatory agencies, such as the United States Environmental Protection Agency and the European Food Safety Authority still largely neglect these neurobehavioral endpoints in their risk assessments[40]. Moreover, vulnerable groups such as pregnant women, young children, and the elderly warrant heightened consideration[1,29,38]. Given the consistent evidence of neurodevelopmental vulnerabilities tied to early-life PFAS exposure, incorporating psychiatric and cognitive outcomes particularly those involving attention, memory, and affect regulation into regulatory frameworks would enhance the comprehensiveness and public-health relevance of PFAS toxicity evaluations. Our analysis thus strongly supports revising current safety thresholds and prioritizing mental health endpoints in environmental health surveillance and regulatory policies.
Policy action must prioritize precautionary regulation, transparency, and equitable exposure reduction. A global phase-out of long-chain PFAS should be accelerated, while stricter evaluation of short-chain alternatives is urgently required. Mandated labeling of PFAS-containing products and public disclosure of industrial emissions would enhance consumer awareness and community oversight. Concurrently, integrating PFAS monitoring into maternal and child health programs and linking this to mental health surveillance systems would support early detection and prevention. Community-centered interventions and environmental justice initiatives are essential to mitigate exposure in high-risk populations[41]. Educational campaigns, behavioral nudges, and provision of PFAS-free alternatives must be tailored for communities with limited resources. Special attention should be given to coastal, industrial, and low-income regions where cumulative exposures are high and mitigation resources scarce. Long-term policy solutions require cross-sector collaboration among governments, industry, academia, and local stakeholders to ensure sustainable reductions in PFAS-related psychiatric risk.
Notably, this systematic review still has several limitations that should be acknowledged. First, although we followed the PRISMA framework to guide search and selection procedures, we restricted our search to two databases (Web of Science and Scopus) and included only peer-reviewed English-language publications. This may have excluded relevant studies from non-English literature or grey literature sources such as governmental reports, dissertations, or conference proceedings potentially introducing publication bias. Second, as this is a qualitative systematic review, formal quality appraisal tools such as the Newcastle-Ottawa Scale or the Risk Of Bias In Non-randomized Studies of Interventions were not employed to assess the methodological rigor or risk of bias of the included studies. Consequently, the synthesis reflects reported findings without a standardized evaluation of study quality, which limits the strength of any causal interpretations. Third, while the thematic analysis allowed us to identify conceptual patterns and mechanistic insights, the interpretation of themes is inherently subjective and may vary with researcher perspective. Fourth, although several included studies reported sex-stratified results, this review did not systematically examine sex-based differences. This is a notable omission given established sex-related variability in both PFAS toxicokinetics and psychiatric susceptibility.
This article underscores the emerging significance of PFAS exposure as a potential environmental risk factor for psychiatric disorders. Over the past five years, a growing body of evidence has linked PFAS exposure to various mental health outcomes, including depression, anxiety, ADHD, ASD, and cognitive impairments. These associations are particularly evident in vulnerable groups such as pregnant women, children, and communities with socioeconomic or environmental disadvantages.
However, inconsistencies across studies stemming from differences in exposure timing, PFAS compound variability, and methodological approaches highlight the complex and multifactorial nature of PFAS-related psychiatric outcomes. While several biological pathways have been proposed to explain PFAS-related neuropsychiatric effects, including dis
To address these gaps, future research must prioritize longitudinal, developmentally sensitive cohort studies, integrate multi-omics and neuroimaging technologies, and enhance exposure assessment protocols. Equally important, studies should reflect environmental justice considerations by including underrepresented populations and exploring the psy
Beyond scientific inquiry, urgent policy and public health actions are needed to mitigate PFAS exposure and its psy
Ultimately, addressing PFAS-related psychiatric risks will require sustained interdisciplinary collaboration across environmental science, psychiatry, and public policy. Integrating rigorous evidence with actionable solutions will be ess
| 1. | Cordner A, De La Rosa VY, Schaider LA, Rudel RA, Richter L, Brown P. Guideline levels for PFOA and PFOS in drinking water: the role of scientific uncertainty, risk assessment decisions, and social factors. J Expo Sci Environ Epidemiol. 2019;29:157-171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 324] [Cited by in RCA: 194] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 2. | Kurwadkar S, Dane J, Kanel SR, Nadagouda MN, Cawdrey RW, Ambade B, Struckhoff GC, Wilkin R. Per- and polyfluoroalkyl substances in water and wastewater: A critical review of their global occurrence and distribution. Sci Total Environ. 2022;809:151003. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 785] [Cited by in RCA: 444] [Article Influence: 111.0] [Reference Citation Analysis (0)] |
| 3. | Beans C. News Feature: How "forever chemicals" might impair the immune system. Proc Natl Acad Sci U S A. 2021;118:e2105018118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 4. | Thapa BS, Pandit S, Mishra RK, Joshi S, Idris AM, Tusher TR. Emergence of per- and poly-fluoroalkyl substances (PFAS) and advances in the remediation strategies. Sci Total Environ. 2024;916:170142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 5. | Rhee J, Chang VC, Cheng I, Calafat AM, Botelho JC, Shearer JJ, Sampson JN, Setiawan VW, Wilkens LR, Silverman DT, Purdue MP, Hofmann JN. Serum concentrations of per- and polyfluoroalkyl substances and risk of renal cell carcinoma in the Multiethnic Cohort Study. Environ Int. 2023;180:108197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 6. | Celis JE, Espejo W, Groffen T, Bervoets L, Padilha J, Mello FV, Sandoval M, Chiang G. Per- and polyfluoroalkylated substances (PFAS) in the feathers and excreta of Gentoo penguins (Pygoscelis papua) from the Antarctic Peninsula. Sci Total Environ. 2025;959:178333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 7. | Brennan NM, Evans AT, Fritz MK, Peak SA, von Holst HE. Trends in the Regulation of Per- and Polyfluoroalkyl Substances (PFAS): A Scoping Review. Int J Environ Res Public Health. 2021;18:10900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 365] [Cited by in RCA: 211] [Article Influence: 42.2] [Reference Citation Analysis (1)] |
| 8. | Solan ME, Koperski CP, Senthilkumar S, Lavado R. Short-chain per- and polyfluoralkyl substances (PFAS) effects on oxidative stress biomarkers in human liver, kidney, muscle, and microglia cell lines. Environ Res. 2023;223:115424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 76] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 9. | Bonato M, Corrà F, Bellio M, Guidolin L, Tallandini L, Irato P, Santovito G. PFAS Environmental Pollution and Antioxidant Responses: An Overview of the Impact on Human Field. Int J Environ Res Public Health. 2020;17:8020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 173] [Cited by in RCA: 129] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 10. | Cheng H, Lv C, Li J, Wu D, Zhan X, Song Y, Zhao N, Jin H. Bioaccumulation and biomagnification of emerging poly- and perfluoroalkyl substances in marine organisms. Sci Total Environ. 2022;851:158117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 11. | Zahm S, Bonde JP, Chiu WA, Hoppin J, Kanno J, Abdallah M, Blystone CR, Calkins MM, Dong GH, Dorman DC, Fry R, Guo H, Haug LS, Hofmann JN, Iwasaki M, Machala M, Mancini FR, Maria-Engler SS, Møller P, Ng JC, Pallardy M, Post GB, Salihovic S, Schlezinger J, Soshilov A, Steenland K, Steffensen IL, Tryndyak V, White A, Woskie S, Fletcher T, Ahmadi A, Ahmadi N, Benbrahim-Tallaa L, Bijoux W, Chittiboyina S, de Conti A, Facchin C, Madia F, Mattock H, Merdas M, Pasqual E, Suonio E, Viegas S, Zupunski L, Wedekind R, Schubauer-Berigan MK. Carcinogenicity of perfluorooctanoic acid and perfluorooctanesulfonic acid. Lancet Oncol. 2024;25:16-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 276] [Cited by in RCA: 214] [Article Influence: 107.0] [Reference Citation Analysis (1)] |
| 12. | Coperchini F, Croce L, Ricci G, Magri F, Rotondi M, Imbriani M, Chiovato L. Thyroid Disrupting Effects of Old and New Generation PFAS. Front Endocrinol (Lausanne). 2020;11:612320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 160] [Article Influence: 32.0] [Reference Citation Analysis (1)] |
| 13. | Roy VC, Bala R, Mehta S. Poly- and per-fluoroalkyl substances toxicity on skeletal and cognitive well-being: a comprehensive review. J Environ Sci Health C Toxicol Carcinog. 2025;43:159-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 14. | Vuong AM, Yolton K, Xie C, Dietrich KN, Braun JM, Webster GM, Calafat AM, Lanphear BP, Chen A. Prenatal and childhood exposure to poly- and perfluoroalkyl substances (PFAS) and cognitive development in children at age 8 years. Environ Res. 2019;172:242-248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 59] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 15. | Brown-Leung JM, Cannon JR. Neurotransmission Targets of Per- and Polyfluoroalkyl Substance Neurotoxicity: Mechanisms and Potential Implications for Adverse Neurological Outcomes. Chem Res Toxicol. 2022;35:1312-1333. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 103] [Article Influence: 25.8] [Reference Citation Analysis (1)] |
| 16. | Szilagyi JT, Avula V, Fry RC. Perfluoroalkyl Substances (PFAS) and Their Effects on the Placenta, Pregnancy, and Child Development: a Potential Mechanistic Role for Placental Peroxisome Proliferator-Activated Receptors (PPARs). Curr Environ Health Rep. 2020;7:222-230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 275] [Cited by in RCA: 226] [Article Influence: 37.7] [Reference Citation Analysis (1)] |
| 17. | Rocabois A, Sanchez M, Philippat C, Crépet A, Wies B, Vrijheid M, Nieuwenhuijsen M, Slama R. Chemical exposome and children health: Identification of dose-response relationships from meta-analyses and epidemiological studies. Environ Res. 2024;262:119811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 18. | Rappazzo KM, Coffman E, Hines EP. Exposure to Perfluorinated Alkyl Substances and Health Outcomes in Children: A Systematic Review of the Epidemiologic Literature. Int J Environ Res Public Health. 2017;14:691. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 355] [Cited by in RCA: 299] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 19. | Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, Shekelle P, Stewart LA; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev. 2015;4:1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20319] [Cited by in RCA: 16752] [Article Influence: 1522.9] [Reference Citation Analysis (7)] |
| 20. | Braun V, Clarke V. Is thematic analysis used well in health psychology? A critical review of published research, with recommendations for quality practice and reporting. Health Psychol Rev. 2023;17:695-718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 439] [Cited by in RCA: 257] [Article Influence: 85.7] [Reference Citation Analysis (0)] |
| 21. | Ahrens AP, Hyötyläinen T, Petrone JR, Igelström K, George CD, Garrett TJ, Orešič M, Triplett EW, Ludvigsson J. Infant microbes and metabolites point to childhood neurodevelopmental disorders. Cell. 2024;187:1853-1873.e15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 84] [Article Influence: 42.0] [Reference Citation Analysis (4)] |
| 22. | Carrizosa C, Murcia M, Ballesteros V, Costa O, Manzano-Salgado CB, Ibarluzea J, Iñiguez C, Casas M, Andiarena A, Llop S, Lertxundi A, Schettgen T, Sunyer J, Ballester F, Vrijheid M, Lopez-Espinosa MJ. Prenatal perfluoroalkyl substance exposure and neuropsychological development throughout childhood: The INMA Project. J Hazard Mater. 2021;416:125185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 23. | Tang C, Wang Y, Hong H. Unraveling the link between heavy metals, perfluoroalkyl substances and depression: Insights from epidemiological and bioinformatics strategies. Ecotoxicol Environ Saf. 2024;279:116482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 24. | Dalsager L, Jensen TK, Nielsen F, Grandjean P, Bilenberg N, Andersen HR. No association between maternal and child PFAS concentrations and repeated measures of ADHD symptoms at age 2½ and 5 years in children from the Odense Child Cohort. Neurotoxicol Teratol. 2021;88:107031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 25. | England-Mason G, MacEachern SJ, Amador K, Soomro MH, Reardon AJF, MacDonald AM, Kinniburgh DW, Letourneau N, Giesbrecht GF, Martin JW, Forkert ND, Dewey D. Using machine learning to investigate the influence of the prenatal chemical exposome on neurodevelopment of young children. Neurotoxicology. 2025;108:218-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 26. | Hong X, Tao L, Guo L, Luo L, Lv J, Li R, Hu J, Gao C, Wang H, Xu DX, Cheng ZZ, Mai BX, Tang Q, Huang Y. PFASs in Cerebrospinal Fluids and Blood-CSF Barrier Permeability in Patients with Cognitive Impairment. Environ Sci Technol. 2024;58:5129-5138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 27. | Hu L, Mei H, Cai X, Song L, Xu Q, Gao W, Zhang D, Zhou J, Sun C, Li Y, Xiang F, Wang Y, Zhou A, Xiao H. Prenatal exposure to poly- and perfluoroalkyl substances and postpartum depression in women with twin pregnancies. Int J Hyg Environ Health. 2024;256:114324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 28. | Itoh S, Yamazaki K, Suyama S, Ikeda-Araki A, Miyashita C, Ait Bamai Y, Kobayashi S, Masuda H, Yamaguchi T, Goudarzi H, Okada E, Kashino I, Saito T, Kishi R. The association between prenatal perfluoroalkyl substance exposure and symptoms of attention-deficit/hyperactivity disorder in 8-year-old children and the mediating role of thyroid hormones in the Hokkaido study. Environ Int. 2022;159:107026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 29. | Itoh M, Kobayashi S, Iwata H, Ait Bamai Y, Ketema RM, Yamaguchi T, Yamazaki K, Tamura N, Tojo M, Suyama S, Itoh S, Miyashita C, Saijo Y, Ito Y, Nakayama SF, Kamijima M, Kishi R; Japan Environment and Children’s Study (JECS) Group. Impact of prenatal exposure to per- and polyfluoroalkyl substances on developmental delays in 4-year-old children: The Japan Environment and Children's study. Environ Int. 2025;198:109434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 30. | Kim JH, Moon N, Ji E, Moon HB. Effects of postnatal exposure to phthalate, bisphenol a, triclosan, parabens, and per- and poly-fluoroalkyl substances on maternal postpartum depression and infant neurodevelopment: a korean mother-infant pair cohort study. Environ Sci Pollut Res Int. 2023;30:96384-96399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 31. | Kim JI, Kim BN, Lee YA, Shin CH, Hong YC, Døssing LD, Hildebrandt G, Lim YH. Association between early-childhood exposure to perfluoroalkyl substances and ADHD symptoms: A prospective cohort study. Sci Total Environ. 2023;879:163081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 27] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 32. | Lefèvre-Arbogast S, Duquenne P, Helmer C, Auriacombe S, Sirot V, Samieri C. Association between dietary exposure to chemical contaminants and risk of dementia in older persons. Environ Int. 2024;192:109033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 33. | Oh J, Bennett DH, Calafat AM, Tancredi D, Roa DL, Schmidt RJ, Hertz-Picciotto I, Shin HM. Prenatal exposure to per- and polyfluoroalkyl substances in association with autism spectrum disorder in the MARBLES study. Environ Int. 2021;147:106328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (1)] |
| 34. | Ogundare O, Obeng-Gyasi E. The Combined Effects of Per- and Polyfluoroalkyl Substances, Metals, and Behavioral and Social Factors on Depressive Symptoms. Med Sci (Basel). 2025;13:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 35. | Sun X, Yang X, Zhang Y, Liu Y, Xiao F, Guo H, Liu X. Correlation analysis between per-fluoroalkyl and poly-fluoroalkyl substances exposure and depressive symptoms in adults: NHANES 2005-2018. Sci Total Environ. 2024;906:167639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 36. | Tillaut H, Monfort C, Rouget F, Pelé F, Lainé F, Gaudreau E, Cordier S, Warembourg C, Saint-Amour D, Chevrier C. Prenatal Exposure to Perfluoroalkyl Substances and Child Behavior at Age 12: A PELAGIE Mother-Child Cohort Study. Environ Health Perspect. 2023;131:117009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 37. | Yao Q, Vinturache A, Lei X, Wang Z, Pan C, Shi R, Yuan T, Gao Y, Tian Y. Prenatal exposure to per- and polyfluoroalkyl substances, fetal thyroid hormones, and infant neurodevelopment. Environ Res. 2022;206:112561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 38. | Wu S, Xie J, Zhao H, Zhao X, Sánchez OF, Rochet JC, Freeman JL, Yuan C. Developmental neurotoxicity of PFOA exposure on hiPSC-derived cortical neurons. Environ Int. 2024;190:108914. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 39. | Zhang S, Ma M, Zhang Y, Luo J, Ouyang F, Tian Y, Gao Y; Shanghai Birth Cohort. Maternal psychological distress modifies the association between prenatal exposure to per- and polyfluoroalkyl substances and infants' neurodevelopment. Sci Total Environ. 2025;977:179351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 40. | S J, Kannaiah KP, Narayanasamy D. Polyfluoroalkyl-Substances Detection in Junk Food Packing Materials Using Various Analytical Methods: A Review. Cureus. 2024;16:e70301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 41. | Kikanme KN, Dennis NM, Orikpete OF, Ewim DRE. PFAS in Nigeria: Identifying data gaps that hinder assessments of ecotoxicological and human health impacts. Heliyon. 2024;10:e29922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/