Published online Mar 19, 2026. doi: 10.5498/wjp.v16.i3.111523
Revised: August 7, 2025
Accepted: November 27, 2025
Published online: March 19, 2026
Processing time: 241 Days and 20.1 Hours
Crohn’s disease (CD) is a chronic inflammatory bowel disease associated with both physical and psychological stressors. Dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis has been implicated in various autoimmune disea
To evaluate HPA axis responses to acute/chronic stress in patients with CD and assess perceived stress, anxiety, and depression.
This study involved two groups: (1) Surgical group: 22 patients with CD and 28 surgery-controls without inflammatory bowel disease; and (2) Outpatient group: 101 patients with CD and 52 positive-control patients with cancer. In the surgical group, acute stress was measured using six postoperative salivary cortisol samples and chronic stress was assessed by hair cortisol measurements. In the outpatient group, emotional distress was evaluated using the Depression Anxiety Stress Scale 21 questionnaire. Demographic and clinical data were analyzed for associations with stress, anxiety, and depression.
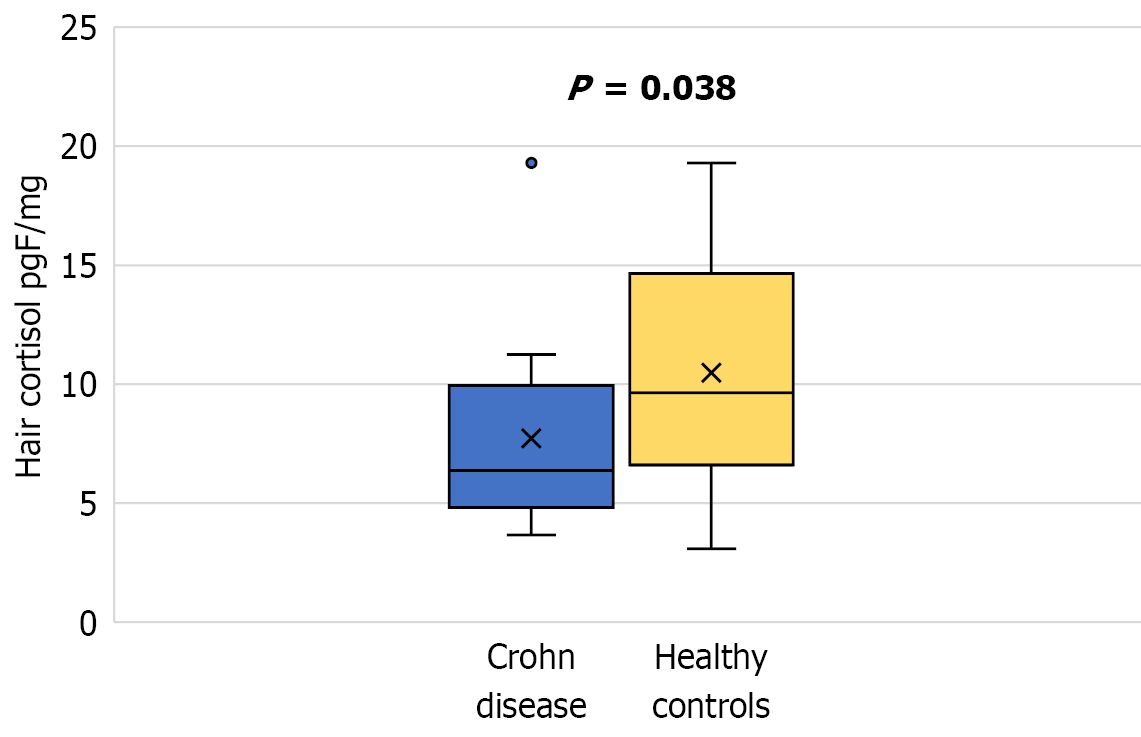
Hair cortisol levels were significantly lower in patients with CD compared to controls (median 6.375 pgF/mg vs 9.643 pgF/mg, P = 0.038), suggesting impaired HPA axis function in response to chronic stress. No significant differences in postoperative salivary cortisol levels were found between patients with CD and controls. According to Depression Anxiety Stress Scale 21 responses, 58% of patients with CD experienced elevated stress levels, while 27% and 24% experienced severe or extremely severe anxiety and depression, respectively. Higher Harvey-Bradshaw Index scores, smoking, and lower hemoglobin levels were independently associated with higher perceived stress and anxiety. Among the controls, 53% exhibited elevated stress levels, while 37% and 13% ex
Patients with CD exhibit a blunted HPA axis response to chronic stress and high psychological distress - com
Core Tip: In this prospective study, we evaluated both acute and chronic stress responses in patients with Crohn’s disease using objective biomarkers - salivary and hair cortisol - in two distinct patient groups. In addition, we assessed perceived psychological stress via validated psychometric tools and compared these findings with those of a control group of patients with cancer. Our findings reveal a significantly decreased cortisol response to chronic stress in patients with Crohn’s disease, along with high levels of perceived stress, anxiety, and depression.
- Citation: Laoudi E, Papalouka D, Kokkotis G, Gkizis M, Mantzou A, Lyrakos G, Markopoulos P, Prapa P, Voulgaris T, Vlachogiannakos J, Papaconstantinou I, Bamias G. Hypothalamic-pituitary-adrenal dysregulation and psychological distress in Crohn’s disease: Insights from acute and chronic stress responses. World J Psychiatry 2026; 16(3): 111523
- URL: https://www.wjgnet.com/2220-3206/full/v16/i3/111523.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i3.111523
In a medical context, stress refers to the body’s physiological response to injury or trauma[1]. In contrast, psychological stress refers to an individual’s response to challenges or pressures perceived as overwhelming or beyond their ability to cope effectively[2]. Both forms of stress responses involve a complex interplay of hormonal, neural, and behavioral changes, which can have both short-term and long-term effects on multiple organ systems[2]. The biochemical response to stress is primarily regulated by the hypothalamic-pituitary-adrenal (HPA) axis. Cortisol, the final effector hormone of the HPA axis, is widely studied as a biomarker of both acute and chronic stress. In several autoimmune diseases, including rheumatoid arthritis, Graves’ disease, and systemic lupus erythematosus, HPA axis dysfunction has been observed in the context of chronic inflammation[3]. This kind of dysfunction in chronic inflammatory diseases arises from prolonged overactivation of the axis and is characterized by lower levels of endogenous cortisol, reflecting an impaired ability to respond to chronic stress[4].
Crohn’s disease (CD) acts as a chronic stressor, encompassing both physical and psychological dimensions. Patients with inflammatory bowel disease (IBD), especially those with CD, face several limitations in their daily lives owing to frequent bowel movements, abdominal pain, anemia, perianal disease, and chronic fatigue[5]. Many patients report experiencing stress and emotional distress, such as anxiety and depression, which significantly compromise their quality of life. Furthermore, psychological stress has been identified as an aggravating factor that may contribute to disease relapse in patients with CD[1,6]. Recent literature emphasizes that bidirectional communication along the gut-brain axis in IBD contributes to the development of mental health disorders such as anxiety and depression[7].
Despite the availability of both traditional and newer medications for the treatment of CD, surgery remains necessary in 20%-30% of patients, particularly those with structuring or penetrating disease[8]. Surgical intervention is widely recognized as a traumatic event and constitutes a substantial source of acute physical and psychological stress[1]. The resultant physiological stress response triggers a cascade of metabolic and hormonal changes, prominently involving the activation of the sympathy-adrenomedullary system and the HPA axis[9]. Activation of the HPA axis increases circulating cortisol levels. Postoperative cortisol secretion rises rapidly as part of the acute stress response and typically peaks within 4-6 hours following surgery, although levels may remain elevated for up to 7 days in cases of major surgical procedures[7].
In the present study, we assessed the ability of patients with CD to respond to both acute and chronic stress through activation of the HPA axis. Salivary cortisol was used as an indicator of acute postoperative stress response, as collecting saliva using salivettes was more convenient for patients than undergoing multiple blood draws. Chronic stress was assessed by measuring hair cortisol concentrations, which reflect cumulative cortisol levels over the preceding 3 months and have been used in recent studies as a stress biomarker[4]. Considering that hair grows approximately 1 cm per month, the proximal 3 cm of hair (closest to the scalp) indicated cortisol exposure over the past 3 months. Additionally, we evaluated perceived stress, anxiety, and depression in patients with CD and explored their correlation with disease parameters and lifestyle factors.
This prospective study consisted of two separate groups of consecutive patients. In the first group, we measured the cortisol levels of patients with CD who underwent surgery and compared them with the levels of surgical patients without IBD, who served as a control group. The HPA axis response to both the acute stress of surgery and the chronic stress of CD was assessed. Furthermore, levels of perceived stress, anxiety, and depression were compared between the two groups using the Depression Anxiety Stress Scales (DASS) 21 questionnaire. In the second group, which consisted of outpatients with CD, who were compared with a positive control group of patients with cancer, we evaluated the levels of stress, anxiety, and depression compared to the general population using the DASS-21 questionnaire. This questionnaire provides severity ratings based on established cut-offs for defining mild, moderate, severe, and extremely severe scores for each scale[10,11].
Samples from the surgical patients included in our study were collected between 2019 and 2023. All patients provided written informed consent before sample collection. Surgical patients from both groups were treated at the Surgical Department of Aretaeio Hospital in Athens (Greece).
Surgical patients with CD: Adult patients with confirmed CD who underwent surgery for disease-related complications were included. Most of these patients required segmental resection and subtotal/total colectomy owing to structuring terminal ileitis that was unresponsive to biological treatment. Patients with other immunological comorbidities and or those who had received corticosteroids within the 3 months before surgery were excluded. No additional corticosteroids were administered during the surgery.
Surgical control group: Adult patients without a known diagnosis of IBD who underwent abdominal surgery for other indications, most commonly neoplasm resection, were included as controls. Patients with other immunological comorbi
Outpatients with CD: Adult patients with confirmed CD who were attending the Department of Inflammatory Bowel Disease of Sotiria Hospital in Athens were included. Patients deemed incapable of completing the questionnaires, such as those with cognitive impairment, were excluded.
Outpatient positive control group: Adult patients with neoplastic disease without a known diagnosis of IBD who were attending the Infusion Center of the Oncology Clinic at Sotiria Hospital to receive treatment were included as the positive control group. Patients deemed incapable of completing the questionnaires, such as those with cognitive impairment, were excluded.
Salivary cortisol: Postoperative salivary cortisol secretion was evaluated by six measurements taken on the second postoperative day. Patients were instructed to chew six salivettes according to a predefined schedule, as follows: The first sample upon awakening in the morning; the second sample 30 minutes later; and the remaining samples at 12:00, 15:00, 18:00, and 21:00, corresponding to the circadian pattern of cortisol secretion. Saliva cortisol was extracted after centrifugation, and samples were stored at -80 °C. Cortisol concentrations were quantified using a chemiluminescence assay.
Hair cortisol: Hair samples were collected by a trained clinician or a member of the surgical team. Strands approximately 1 cm in width were cut using scissors from the posterior vertex, as close to the scalp as possible, and stored at room temperature. The cortisol extraction procedure involved pulverization, methanol extraction, and centrifugation. Patients with less than 2 cm of hair length or those with dyed hair were excluded. Cortisol concentrations were measured using a chemiluminescence assay.
The hair and saliva samples were processed at the Choremeio Research Laboratory, First Department of Pediatrics, National and Kapodistrian University of Athens Medical School.
DASS questionnaire: Patients from both groups were asked to complete the validated Greek version of the DASS-21 questionnaire, which assesses emotional discomfort experienced over the preceding week[9,10]. The questionnaire consists of 21 questions, with 7 questions assigned to each of the three measurement subscales: Stress, anxiety, and de
Patient data: Clinical disease activity was assessed using the Harvey-Bradshaw Index (HBI)[14] at the time of each patient’s visit. Demographic and disease-related data were collected retrospectively from patients’ medical records.
All statistical analyses were performed using SPSS version 23.0 (IBM, Armonk, NY, United States). The results are expressed as percentages, means ± SD, or medians (interquartile ranges), as appropriate. The independent samples t-test and the non-parametric Mann-Whitney U test were used to compare normally and non-normally distributed variables, respectively. A sensitivity analysis with linear regression was performed to adjust for possible confounding effects of sex and age on hair cortisol levels. Repeated-measures ANOVA was performed to assess fluctuations in salivary cortisol. The DASS-21 subscale scores were analyzed as continuous variables rather than ordinal variables to increase the sensitivity of the analyses. Univariate linear regression models were used to identify factors associated with each section of the DASS-21 questionnaire. Variables with P ≤ 0.1 in univariate analyses were included in a multivariate logistic regression model. Statistical significance was set at P < 0.05.
In the surgical group, 27 patients with CD (51.9% female) and 32 controls (46.9% female) were included. Among patients with CD (mean age: 41 years), most (52.0%) underwent right hemicolectomy. Patients from the control group (mean age: 63.3 years) underwent various surgical procedures, including operations for colon cancer (51.0%), pancreatic cancer (13.0%), and liver or stomach cancer (13.0%).
Cortisol levels from hair samples were measured in 18 patients with CD and 21 controls. Patients with CD had signifi
Cortisol levels from saliva samples were measured in 22 patients with CD and 28 controls to assess postoperative cortisol fluctuations (acute stress). No statistically significant change in salivary cortisol levels was observed over time in either group. For patients with CD, cortisol levels reported as median (interquartile range) were as follows: First: 0.26 (0.13-0.82); second: 0.38 (0.15-0.62); third: 0.26 (0.14-0.57); fourth: 0.22 (0.13-0.40); fifth: 0.29 (0.16-0.51); and sixth: 0.22 (0.13-0.44), repeated-measures ANOVA, (P = 0.249). For controls, cortisol levels were as follows: First: 0.20 (0.14-0.50); second: 0.19 (0.15-0.40); third: 0.19 (0.13-0.41); fourth: 0.17 (0.13-0.40); fifth: 0.18 (0.14-0.40); and sixth: 0.17 (0.14-0.36), repeated-measures ANOVA (P = 0.662). There was also no significant difference between the two groups (patients with CD vs controls) at each studied timepoint: First: 0.26 (0.13-0.82) vs 0.20 (0.14-0.50), P = 0.494; second: 0.38 (0.15-0.62) vs 0.19 (0.15-0.40), P = 0.272; third: 0.26 (0.14-0.57) vs 0.19 (0.13-0.41), P = 0.363; fourth: 0.22 (0.13-0.40) vs 0.17 (0.13-0.40), P = 0.789; fifth: 0.29 (0.16-0.51) vs 0.18 (0.14-0.40), P = 0.134; and sixth: 0.22 (0.13-0.44) vs 0.17 (0.14-0.36), P = 0.647 (Supplementary Figure 1).
No significant correlation was observed between hair or salivary cortisol concentrations and DASS-21 questionnaire scores.
We analyzed data from 101 outpatients with CD (61 male; mean age: 38.2 years) who attended our Inflammatory Bowel Disease Unit (Table 1). The positive control group consisted of 52 patients with cancer (mean age: 63 years), who completed the questionnaire while receiving chemotherapy in the short-stay clinic.
| Characteristic | Value |
| Age in years | 38.2 (13.2) |
| Male, % | 57.9 |
| Educational status, % | |
| Illiterate | 1.5 |
| Basic | 46.2 |
| Secondary | 20 |
| Technical/university degree | 32.3 |
| HBI | 1 (1–3) |
| Smoker, % | 40.8 |
| Montreal classification, % | |
| A1/A2/A31 | 8.5/70.7/20.7 |
| B1/B2/B32 | 51.3/38.5/10.3 |
| L1/L2/L3/L43 | 43.6/10.3/46.2 |
| P value | 28.4 |
| Under biologic treatment, % | 72 |
| Hemoglobin in g/dL | 13.4 (1.8) |
| Anemia, % | 10.3 |
| CRP in mg/L | 0.8 (0.3–2.7) |
| History of Crohn’s-related surgery, % | 34.7 |
| Extra-intestinal manifestations, % | 39.4 |
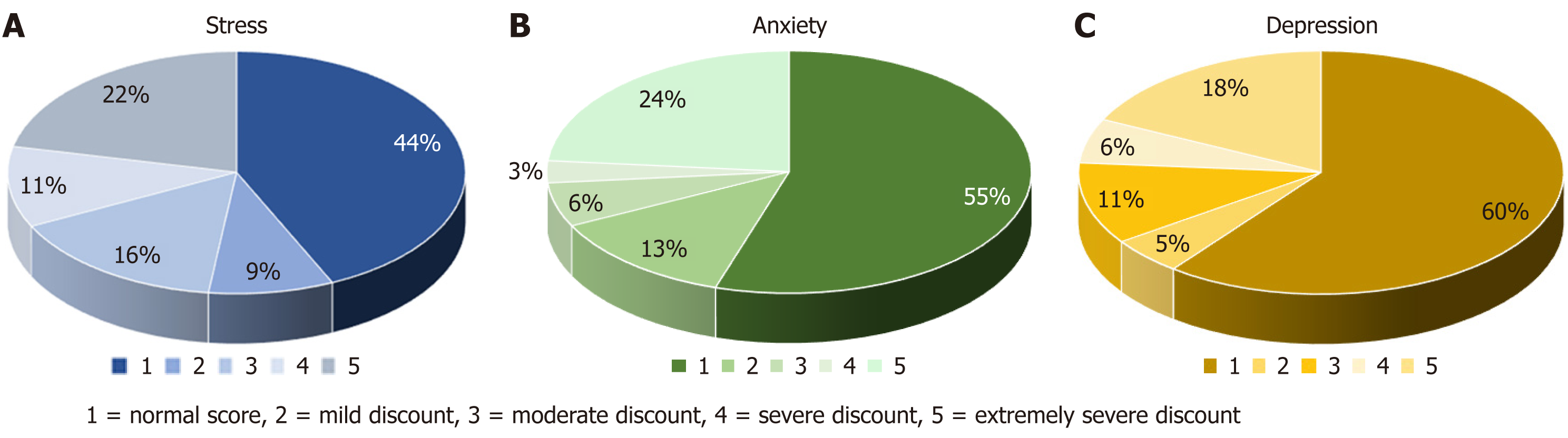
According to the validated DASS-21 cut-off scores for the general population, the distribution of emotional distress (including stress, anxiety, and depression scores) among patients with CD were as follows.
Stress: Exactly 44% of patients had normal scores, while 9% had mild, 16% had moderate, 11% had severe, and 22% had extremely severe stress levels (Figure 2A).
Anxiety: Of the total patients, 55% had normal scores, while 13% had mild, 6% had moderate, 3% had severe, and 24% had extremely severe anxiety levels (Figure 2B).
Depression: Among the patients, 60% had normal scores, whereas 5% had mild, 9% had moderate, 6% had severe, and 18% had extremely severe depression levels (Figure 2C).
No statistically significant differences were observed in DASS-21 scores for stress (P = 0.373), anxiety (P = 0.335), or depression (P = 0.603) between patients with CD and those with cancer (Supplementary Figure 2).
In univariate analyses, stress was significantly associated with sex (P = 0.003), HBI (P = 0.03), hemoglobin levels (P = 0.022), smoking status (P = 0.003), and the A (age at diagnosis) 3 Montreal classification (P = 0.014). In multivariate linear regression analysis, stress remained independently associated with hemoglobin levels (β = -4.1, P = 0.023) and smoking status (β = 9.5, P = 0.008; Table 2).
| Stress | Univariate | Multivariate | ||
| β (95%CI) | P value | β (95%CI) | P value | |
| Female | 4.5 (1.5 to 7.5) | 0.003 | -8.7 (-20.5 to 3.2) | 0.145 |
| HBI | 1.2 (0.1 to 2.2) | 0.03 | 1.7 (-1.5 to 4.9) | 0.281 |
| HGB | -1.6 (-3 to -0.2) | 0.022 | -4.1 (-7.6 to -0.6) | 0.023 |
| Smoking | 6.9 (2.5 to 11.3) | 0.003 | 9.5 (3.1 to 18.4) | 0.008 |
| A Montreal1 | ||||
| A1 | Reference | |||
| A2 | 4.0 (-2.0 to 9.9) | 0.239 | -3.7 (-15.9 to 8.6) | 0.541 |
| A3 | 7.7 (1.2 to 14.2) | 0.014 | 8.5 (-4.2 to 21.3) | 0.181 |
In addition, univariate analyses identified significant associations between anxiety and sex (P = 0.001), HBI (P < 0.001), smoking status (P = 0.008), A3 Montreal classification (P = 0.092), and L (location) 3 Montreal classification (P = 0.024). In multivariate linear regression, anxiety was independently associated with smoking (β = 5.9, P = 0.012), L3 disease location compared to L1 (β = 4.9, P = 0.046), and disease activity (HBI) (β = 2.3, P = 0.002). A trend toward an association with female sex was also observed (β = 4.2, P = 0.065; Table 3).
| Anxiety | Univariate | Multivariate | ||
| β (95%CI) | P value | β (95%CI) | P value | |
| Female | 3.8 (1.5-6.1) | 0.001 | 4.2 (-0.3 to 10.5) | 0.065 |
| HBI | 1.7 (0.8-2.5) | < 0.001 | 2.3 (0.9-3.8) | 0.002 |
| Smoking | 5.0 (1.3-8.7) | 0.008 | 5.9 (1.4-10.5) | 0.012 |
| A Montreal1 | ||||
| A1 | Reference | |||
| A2 | 2.2 (-2.7 to 7.0) | 0.380 | -5.6 (-13.7 to 2.4) | 0.164 |
| A3 | 4.6 (-0.8 to 9.9) | 0.092 | -0.5 (-8.9 to 8.0) | 0.913 |
| L Montreal2 | ||||
| L1 | Reference | |||
| L2 | 1.5 (-3 to 5.9) | 0.515 | -0.3 (-6.8 to 7.4) | 0.933 |
| L3 | 3.4 (0.5-6.4) | 0.024 | 4.9 (0.1-9.7) | 0.046 |
In univariate models, depression was associated with sex (P = 0.016), age (P = 0.011), HBI (P = 0.039), hemoglobin levels (P = 0.030), smoking (P = 0.010), and A3 Montreal classification (P = 0.003). Multivariate linear regression revealed that depression was independently associated with hemoglobin levels (β = -5.1, P = 0.027). A non-significant trend toward an association with smoking was also observed (β = 7.9, P = 0.084; Table 4).
| Depression | Univariate | Multivariate | ||
| β (95%CI) | P value | β (95%CI) | P value | |
| Female | 3.7 (0.7-6.7) | 0.016 | -5.2 (-19.0 to 8.6) | 0.447 |
| Age | 0.2 (0.0-0.3) | 0.011 | -0.2 (-0.8 to 0.4) | 0.491 |
| HBI | 1.1 (0.1-2.2) | 0.039 | 1.8 (-1.8 to 5.4) | 0.323 |
| HGB | -1.6 (-3.0 to -0.2) | 0.030 | -5.1 (-9.6 to -0.6) | 0.027 |
| Smoking | 6.1 (1.5-10.6) | 0.010 | 7.9 (-1.1 to 16.8) | 0.084 |
| A Montreal1 | ||||
| A1 | Reference | |||
| A2 | 5 (-1.1 to 11.0) | 0.125 | 6.3 (-11.7 to 24.3) | 0.477 |
| A3 | 10.1 (3.5-16.7) | 0.003 | 21.3 (-6.9 to 49.5) | 0.132 |
In this study, we obtained data from assessment of the response of patients with CD to chronic stress, mediated by their immunological disease and treatment courses, as well as to acute stress induced by an acute stressor, such as a surgical procedure. Impairment of the HPA axis has been described in several other immunological diseases, including multiple sclerosis[15], rheumatoid arthritis, and Sjögren’s syndrome[16], suggesting a compromised response to chronic stress in these patients. More specifically, patients with rheumatoid arthritis have demonstrated significantly lower serum cortisol levels during sequential circadian measurements on the first postoperative day compared to patients with osteoarthritis[17]. Moreover, Silverman et al[18] reported that in patients having conditions associated with chronic HPA stimulation - such as several immunological diseases - glucocorticoid receptors may gradually develop resistance to circulating cortisol, subsequently leading to impaired negative feedback regulation of the axis. Our findings suggest that patients with CD may also exhibit impaired HPA axis stimulation in the context of their chronic immunological dysfunction, as evidenced by significantly lower hair cortisol levels compared to controls. Although cortisol levels typically remain elevated in patients experiencing chronic stress owing to sustained HPA axis activation, several circulating mediators in CD may downregulate the axis, leading to reduced cortisol release[17]. Furthermore, according to a review by Ge et al[19], chronic psychological stress affects the neuroendocrine-immune regulatory network, intestinal mucosal immunity, and gut microbiome homeostasis in patients with IBD.
Regarding acute physical stress, our study found no significant differences in salivary cortisol levels between surgical patients with IBD and those without IBD. These findings contrast with those of Chikanza et al[20], who reported significant differences in salivary cortisol levels between patients with rheumatoid arthritis and those with osteoarthritis. However, it is important to note that rheumatoid arthritis and CD are distinct conditions, each characterized by different immunobiological mechanisms. Furthermore, the rheumatoid arthritis group in the referenced study belonged to a different age group than our group, with mean participant ages of 51 years and 40 years, respectively, which may further account for the discrepancy in cortisol responses.
Our analysis did not reveal any correlation between cortisol levels, measured in both hair and saliva, and the ques
Another important finding of our study is that more than half of the outpatients with CD (58%) who completed the DASS-21 questionnaire scored higher for perceived stress compared to the general population, although their scores for anxiety and depression were mainly within normal ranges (55% and 60% of patients, respectively). Importantly, a substantial number of patients fell within the severe or extremely severe range for stress, anxiety, and depression (33%, 27%, and 24%, respectively), reflecting the extent to which the quality of life may be impaired in individuals with persis
Interestingly, the questionnaire results of patients with CD were comparable to those of patients with neoplasms, suggesting that patients with CD may experience emotional disturbances as severe as those faced by patients with cancer[29]. Although differences in age and comorbidities between the two groups may confound the results, the observation that patients with CD exhibit similar levels of stress, anxiety, and depression underscores the need for a holistic treatment approach, which should include not only medical management of the disease but also appropriate psychological support.
Our study has several limitations. First, patients in the CD group were significantly younger (mean age: 40 years) than those in the control group (mean age: 60 years). This age difference may act as a confounding factor[30-32], given that the HPA axis function declines with aging. Second, comparing HPA axis function between patients with CD and those with cancer may introduce bias. However, according to existing data, most studies assessing the HPA axis in patients with cancer report increased baseline cortisol levels or a hyperactive stress response. In approximately half of the studies reporting decreased HPA axis activity, patients were receiving cortisol-suppressing treatments. While this represents an area for further research, the comparison in our study nonetheless suggests distinct patterns of HPA axis dysfunction between the two groups[33]. Lastly, our salivary cortisol measurements were conducted on postoperative day 2 rather than day 1, which may more accurately reflect the physiological response to acute surgical stress. This adjustment was necessary, as most patients were unable to chew the salivettes shortly after surgery owing to the presence of a nasogastric tube.
Despite the limited number of surgical patients included, our study suggests that individuals with CD exhibit an impaired response to chronic physical stress. Dysfunction of the HPA axis may have significant implications for disease progression; however, its precise impact on disease recurrence and treatment failure remains unclear, potentially representing a novel area for future therapeutic intervention. Although psychological treatment has not yet been proven to alter the course of the disease, emotional support remains a crucial component of patient care. Given that treatment adherence and overall patient well-being are primary therapeutic goals, addressing psychological health is essential in the comprehensive management of CD.
| 1. | Desborough JP. The stress response to trauma and surgery. Br J Anaesth. 2000;85:109-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1598] [Cited by in RCA: 1324] [Article Influence: 50.9] [Reference Citation Analysis (1)] |
| 2. | Mawdsley JE, Rampton DS. Psychological stress in IBD: new insights into pathogenic and therapeutic implications. Gut. 2005;54:1481-1491. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 483] [Cited by in RCA: 447] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 3. | da Rosa Franchi Santos LF, Stadtlober NP, Costa Dall'Aqua LG, Scavuzzi BM, Guimarães PM, Flauzino T, Batisti Lozovoy MA, Mayumi Iriyoda TV, Vissoci Reiche EM, Dichi I, Maes M, Colado Simão A. Increased adhesion molecule levels in systemic lupus erythematosus: relationships with severity of illness, autoimmunity, metabolic syndrome and cortisol levels. Lupus. 2018;27:380-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 4. | Sternberg EM, Hill JM, Chrousos GP, Kamilaris T, Listwak SJ, Gold PW, Wilder RL. Inflammatory mediator-induced hypothalamic-pituitary-adrenal axis activation is defective in streptococcal cell wall arthritis-susceptible Lewis rats. Proc Natl Acad Sci U S A. 1989;86:2374-2378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 512] [Cited by in RCA: 453] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 5. | Cohen S, Kamarck T, Mermelstein R. A Global Measure of Perceived Stress. J Health Soc Behav. 1983;24:385-396. [RCA] [DOI] [Full Text] [Cited by in Crossref: 24867] [Cited by in RCA: 18312] [Article Influence: 425.9] [Reference Citation Analysis (15)] |
| 6. | Sternberg EM, Young WS 3rd, Bernardini R, Calogero AE, Chrousos GP, Gold PW, Wilder RL. A central nervous system defect in biosynthesis of corticotropin-releasing hormone is associated with susceptibility to streptococcal cell wall-induced arthritis in Lewis rats. Proc Natl Acad Sci U S A. 1989;86:4771-4775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 383] [Cited by in RCA: 344] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 7. | Barberio B, Zamani M, Black CJ, Savarino EV, Ford AC. Prevalence of symptoms of anxiety and depression in patients with inflammatory bowel disease: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2021;6:359-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 614] [Cited by in RCA: 520] [Article Influence: 104.0] [Reference Citation Analysis (1)] |
| 8. | Shah RS, Click BH. Medical therapies for postoperative Crohn's disease. Therap Adv Gastroenterol. 2021;14:1756284821993581. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 9. | Finnerty CC, Mabvuure NT, Ali A, Kozar RA, Herndon DN. The surgically induced stress response. JPEN J Parenter Enteral Nutr. 2013;37:21S-29S. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 280] [Cited by in RCA: 267] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 10. | Ivascu R, Torsin LI, Hostiuc L, Nitipir C, Corneci D, Dutu M. The Surgical Stress Response and Anesthesia: A Narrative Review. J Clin Med. 2024;13:3017. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 90] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 11. | Lyrakos GN, Arvaniti C, Smyrnioti M, Kostopanagiotou G. Translation and validation study of the depression anxiety stress scale in the greek general population and in a psychiatric patient's sample. Eur Psychiatr. 2011;26:1731-1731. [RCA] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 77] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 12. | Lovibond SH, Lovibond PF. Depression Anxiety Stress Scales (DASS-21, DASS-42). APA PsycTests. 1995. [DOI] [Full Text] |
| 13. | Lovibond PF, Lovibond SH. The structure of negative emotional states: comparison of the Depression Anxiety Stress Scales (DASS) with the Beck Depression and Anxiety Inventories. Behav Res Ther. 1995;33:335-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10682] [Cited by in RCA: 7939] [Article Influence: 256.1] [Reference Citation Analysis (2)] |
| 14. | Khanna R, Son S, Zou G, Roshanov PS. Estimation of the Harvey Bradshaw Index from the Patient-Reported Outcome 2 in Crohn's Disease: Results Based on a Large Scale Randomized Controlled Trial. Inflamm Bowel Dis. 2025;31:2097-2105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 15. | Heesen C, Gold SM, Huitinga I, Reul JM. Stress and hypothalamic-pituitary-adrenal axis function in experimental autoimmune encephalomyelitis and multiple sclerosis - a review. Psychoneuroendocrinology. 2007;32:604-618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 64] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 16. | Crofford LJ. The hypothalamic-pituitary-adrenal axis in the pathogenesis of rheumatic diseases. Endocrinol Metab Clin North Am. 2002;31:1-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 55] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 17. | Sun Y, Li L, Xie R, Wang B, Jiang K, Cao H. Stress Triggers Flare of Inflammatory Bowel Disease in Children and Adults. Front Pediatr. 2019;7:432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 118] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 18. | Silverman MN, Sternberg EM. Glucocorticoid regulation of inflammation and its functional correlates: from HPA axis to glucocorticoid receptor dysfunction. Ann N Y Acad Sci. 2012;1261:55-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 600] [Cited by in RCA: 526] [Article Influence: 37.6] [Reference Citation Analysis (4)] |
| 19. | Ge L, Liu S, Li S, Yang J, Hu G, Xu C, Song W. Psychological stress in inflammatory bowel disease: Psychoneuroimmunological insights into bidirectional gut-brain communications. Front Immunol. 2022;13:1016578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 128] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 20. | Chikanza IC, Petrou P, Kingsley G, Chrousos G, Panayi GS. Defective hypothalamic response to immune and inflammatory stimuli in patients with rheumatoid arthritis. Arthritis Rheum. 1992;35:1281-1288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 225] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 21. | Abdelall ES, Eagle Z, Finseth T, Mumani AA, Wang Z, Dorneich MC, Stone RT. The Interaction Between Physical and Psychosocial Stressors. Front Behav Neurosci. 2020;14:63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 22. | Häuser W, Janke KH, Klump B, Gregor M, Hinz A. Anxiety and depression in adult patients with celiac disease on a gluten-free diet. World J Gastroenterol. 2010;16:2780-2787. [PubMed] [DOI] [Full Text] |
| 23. | Herman JP, Ostrander MM, Mueller NK, Figueiredo H. Limbic system mechanisms of stress regulation: hypothalamo-pituitary-adrenocortical axis. Prog Neuropsychopharmacol Biol Psychiatry. 2005;29:1201-1213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1082] [Cited by in RCA: 981] [Article Influence: 46.7] [Reference Citation Analysis (3)] |
| 24. | Iob E, Kirschbaum C, Steptoe A. Persistent depressive symptoms, HPA-axis hyperactivity, and inflammation: the role of cognitive-affective and somatic symptoms. Mol Psychiatry. 2020;25:1130-1140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 254] [Cited by in RCA: 223] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 25. | Crawford JR, Henry JD. The Depression Anxiety Stress Scales (DASS): normative data and latent structure in a large non-clinical sample. Br J Clin Psychol. 2003;42:111-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1272] [Cited by in RCA: 990] [Article Influence: 43.0] [Reference Citation Analysis (0)] |
| 26. | Weckmann G, Kiel S, Chenot JF, Angelow A. Association of Anemia with Clinical Symptoms Commonly Attributed to Anemia-Analysis of Two Population-Based Cohorts. J Clin Med. 2023;12:921. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 27. | Lawless MH, Harrison KA, Grandits GA, Eberly LE, Allen SS. Perceived stress and smoking-related behaviors and symptomatology in male and female smokers. Addict Behav. 2015;51:80-83. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 98] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 28. | Lakatos PL, Szamosi T, Lakatos L. Smoking in inflammatory bowel diseases: good, bad or ugly? World J Gastroenterol. 2007;13:6134-6139. [PubMed] [DOI] [Full Text] |
| 29. | Singh RP, Singh H, Singh CJ, Kaur KT. Screening of Psychological Distress in Cancer Patients During Chemotherapy: A Cross-sectional Study. Indian J Palliat Care. 2015;21:305-310. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 30. | Ferrari E, Magri F, Dori D, Migliorati G, Nescis T, Molla G, Fioravanti M, Solerte SB. Neuroendocrine correlates of the aging brain in humans. Neuroendocrinology. 1995;61:464-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 57] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 31. | Gaffey AE, Bergeman CS, Clark LA, Wirth MM. Aging and the HPA axis: Stress and resilience in older adults. Neurosci Biobehav Rev. 2016;68:928-945. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 234] [Cited by in RCA: 201] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 32. | Varadhan R, Seplaki CL, Xue QL, Bandeen-Roche K, Fried LP. Stimulus-response paradigm for characterizing the loss of resilience in homeostatic regulation associated with frailty. Mech Ageing Dev. 2008;129:666-670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 112] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 33. | Kanter NG, Cohen-Woods S, Balfour DA, Burt MG, Waterman AL, Koczwara B. Hypothalamic-Pituitary-Adrenal Axis Dysfunction in People With Cancer: A Systematic Review. Cancer Med. 2024;13:e70366. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/