Published online Sep 19, 2025. doi: 10.5498/wjp.v15.i9.109478
Revised: June 9, 2025
Accepted: July 8, 2025
Published online: September 19, 2025
Processing time: 106 Days and 1.1 Hours
Older adults with mild cognitive impairment (MCI) often show motor dysfun
To assess gait and handwriting differences and their potential for screening MCI in older adults.
Ninety-five participants, including 34 with MCI and 61 cognitively normal controls, were assessed for gait using the GAITRite® system and handwriting with a dot-matrix pen. Five machine learning models were developed to assess the discriminative power of gait and handwriting data for MCI screening.
Compared to the cognitively normal group, the MCI group had slower gait velocity (Z = -2.911, P = 0.004), shorter stride and step lengths (t = -3.005, P = 0.003; t = 2.863, P = 0.005), and longer cycle, standing, and double support times (t = -2.274, P = 0.025; t = -2.376, P = 0.018; t = -2.717, P = 0.007). They also had reduced cadence (t = 2.060, P = 0.042) and increased double support time variability (Z = -2.614, P = 0.009). In handwriting, the MCI group showed lower average pressure (all tasks: Z = -2.135, P = 0.033) and decreased accuracy (graphic task: Z = -2.447, P = 0.014; Chinese character task: Z = -3.078, P = 0.002). In the graphic task, they demonstrated longer time in air (Z = -2.865, P = 0.004), reduced X-axis maximum velocities (Z = -3.237, P = 0.001), and lower accelerations (X-axis: Z = -2.880, P = 0.004; Y-axis: Z = -1.987, P = 0.047) and maximum accelerations (X-axis: Z = -3.998, P < 0.001; Y-axis: Z = -2.050, P = 0.040). The multimodal analysis achieved the highest accuracy (74.4%) with the Gradient Boosting Classifier.
Integrating gait and handwriting kinematics parameters provides a viable method for distinguishing MCI, po
Core Tip: This study introduces sensitive biomarkers for assisted screening of mild cognitive impairment (MCI) by integrating gait and handwriting kinematic parameters. The findings demonstrate that the combination of gait analysis and handwriting tasks achieves a detection accuracy of 74.44% for MCI, outperforming single-task assessments. These parameters could complement traditional tool like the Montreal cognitive assessment by reducing reliance on subjective assessments and minimizing cultural, language, and educational biases. This research establishes a foundation for translating these precision biomarkers into cost-effective, portable, and scalable wearable technologies, which could enable large-scale, rapid screening of high-risk individuals with MCI in community settings.
- Citation: Ren YX, Wu B, Lou JL, Zhu XR, Zhang C, Lang Q, Wei ZQ, Su LM, Qi HN, Wang LN. Community-based assisted screening for mild cognitive impairment using gait and handwriting kinematic parameters analysis. World J Psychiatry 2025; 15(9): 109478
- URL: https://www.wjgnet.com/2220-3206/full/v15/i9/109478.htm
- DOI: https://dx.doi.org/10.5498/wjp.v15.i9.109478
Mild cognitive impairment (MCI) represents an intermediate stage between normal aging and dementia, without substantial impact on daily activities or meeting the criteria for dementia diagnostic[1]. The global prevalence of MCI over the age of 60 is currently 17.3%, with a higher prevalence of 26.48% reported in China[2,3]. Estimates suggest that 34% of individuals with MCI progress to dementia[4], while 27.57% experience a reversion to normal cognitive function[5]. However, 99.2% of older adults with MCI are unaware of their condition, and 97.2% remain undiagnosed and un
Cortical function decline due to specific brain atrophy affects both cognitive and motor functions[9]. Older adults with MCI exhibit slower gait speeds, shorter stride and step lengths, and longer standing and double support times compared with cognitively normal individuals[9,10]. Previous studies have shown that a decline gait speed often precedes cognitive decline, indicating its potential as a biomarker for cognitive impairment[9,11]. Given its objectivity and practicality, gait testing may be a valuable tool for cognitive impairment screening[12]. Currently, machine learning analysis of gait data alone achieves an accuracy of approximately 70% in distinguishing older adults with MCI from those with normal cognition[13]. Lin et al[14] found that combining gait and eye movement features improved the area under the receiver operating characteristic curve (AUC) to 0.74. These findings indicate that while gait kinematic parameters are valuable for early MCI screening, and a multimodal approach offers greater accuracy than gait data alone.
Additionally, motor dysfunction can also manifest as upper extremity fine motor deficits[15]. Writing, a complex process involving fine motor skills, kinesthetic components, and multiple cognitive domains[16], is often affected in older adults with MCI. Compared with cognitively normal individuals, those with MCI exhibit slower writing speed, smaller characters, longer pauses between strokes, reduced pressure, and decreased writing accuracy[17,18]. However, due to cultural differences and varying writing habits, the English alphabet and Chinese characters differ in writing re
While single-modality analysis of gait and or handwriting kinematic parameters has proven effective in distinguishing older adults with MCI from cognitively normal individuals, the potential enhancement of screening accuracy through the integration of these modalities remains to be fully elucidated. Including both gait and handwriting kinematic parameters provides a more thorough assessment of motor function, ensures that both gross and fine motor skills are considered, and helps capture the full extent of motor impairment or improvement. In addition, machine learning-based methods have shown promise as valuable tools for cognitive impairment screening[21]. Building on the diagnostic relevance of gait and handwriting kinematic parameters, this study is the first time to apply machine learning algorithms for an integrated analysis of these multimodal data in screening older adults with MCI. The aim of this study is to develop an objective, accessible, and accurate screening tool. Two hypotheses are proposed: (1) Gait and handwriting kinematic parameters differ significantly between older adults with MCI and cognitively normal individuals; and (2) A multimodal approach combining gait and handwriting kinematic parameters offers superior accuracy for MCI screening compared to unimodal methods.
A cross-sectional design was used in this study to examine differences in gait and handwriting kinematics and evaluate their potential for MCI screening among older adults in Huzhou city, Zhejiang Province, China, with data collected from August 2024 to October 2024. This study was approved by the Institutional Review Board of the third affiliated hospital of Huzhou university (2024-474) in accordance with the guidelines of the Declaration of Helsinki and registered at Chictr.org.cn (ChiCTR 2400089057). All eligible participants voluntarily signed the written informed consent after being fully informed.
Community-dwelling adults aged 60 years or older were recruited from senior centers and healthcare centers in 2 randomly selected districts of Huzhou, Zhejiang Province, China. Recruitment leaflets were distributed at these centres, and local healthcare providers referred potential participants. Individuals who showed interest were invited for an in-person interview to screen for eligibility by three trained staff, and a trained neurologist-psychiatrist examined the potential participants to provide the final diagnosis of MCI.
The inclusion criteria for the gait and handwriting tasks were: (1) Age ≥ 60 years old; (2) Elementary school level and above; (3) Absence of self-reported visual or auditory impairment; (4) Right-handed dominance[17]; and (5) Able to make an informed consent. The exclusion criteria included: (1) A history of neurological, psychiatric and other severe medical issues that may affect brain function; (2) Taking any medications in the past 6 months which may cause impaired or improved cognitive performance (e.g., memantine and antipsychotic)[22]; (3) Inability to walk independently or walking independently with the aid of auxiliary tools; (4) Unable to perform handwriting tasks due to hand movement disorders; and (5) Recent or upcoming mobility-impairing medical procedures (e.g., hemodialysis and elective surgery). Participants could withdraw if they developed a serious illness or requested to leave the study.
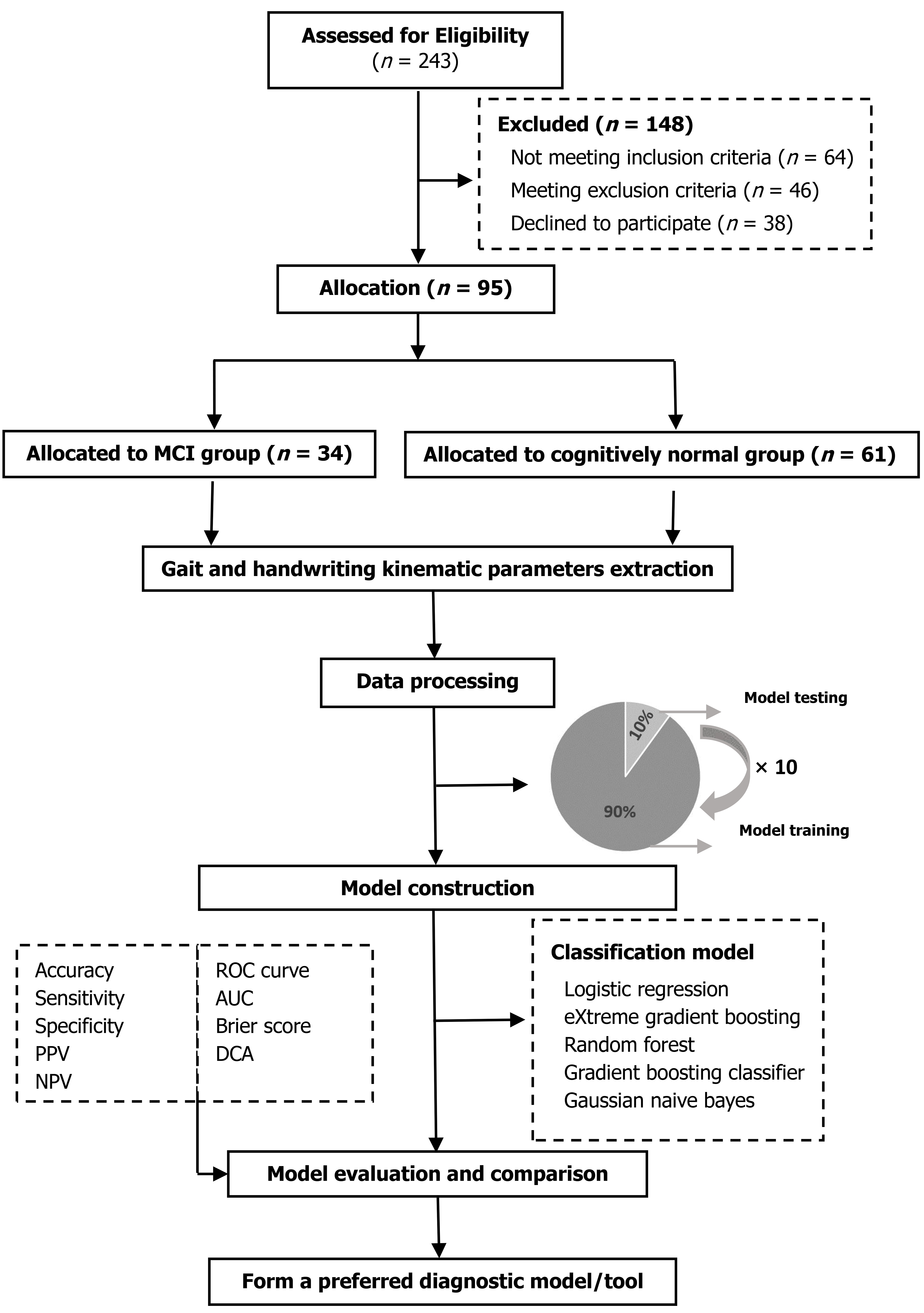
The operational criteria for MCI screening included[23]: (1) A reported decline in cognitive function over the past year by either the participant or an informant; (2) Objective cognitive impairment inconsistent with age and education, assessed by the Chinese MoCA-basic (MoCA-B) with cutoffs of 18 for elementary, 21 for secondary, and 23 for tertiary education[24]; (3) Intact activities of daily living (ADL), evaluated by physical self-maintenance with six items and instrumental activities with eight items, with a total ADL score ≤ 16 indicating intact ADL; and (4) Absence of dementia, determined by community physicians using DSM-IV criteria. This assessment considered family/medical history, chief complaints, and the MMSE scores with thresholds of ≥ 17 for illiterate, ≥ 20 for primary, and ≥ 24 for secondary education and above. Individuals without signs of MCI were classified as cognitively normal (CN group). During the initial eligibility assessment, 243 potential participants with independent walking and writing ability were recruited. The second round included assessments of functional status in ADL, general cognitive function, medical health and medication history. A total of 95 eligible participants were included in this study.
Demographic characteristics: Demographic characteristics included age, sex, education level, occupation, marital status, residence, monthly income, region of residence, smoking and alcohol use, general cognitive function, physical activity, and multimorbidity.
The dataset comprised three unimodal types: Gait kinematic parameters, graphic kinematic, and Chinese character writing kinematic parameters. Multimodal data were subsequently synthesized by integrating gait with graphic and Chinese character handwriting data.
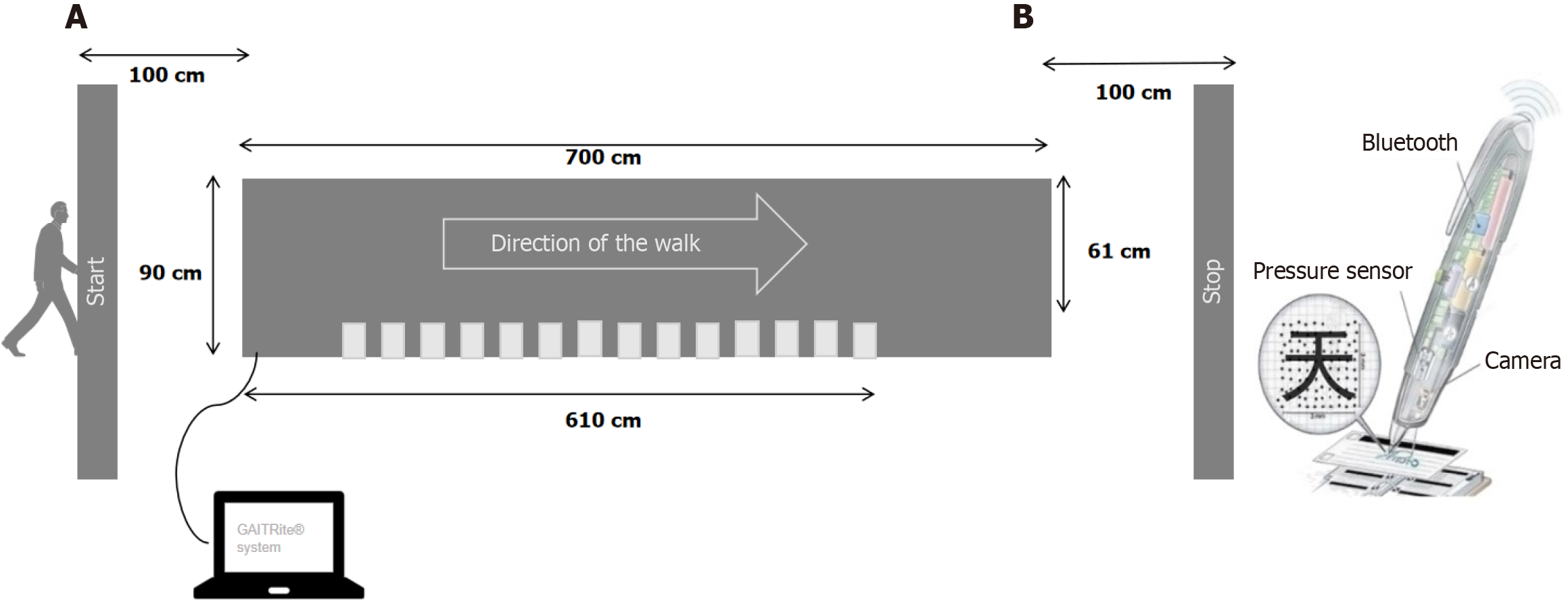
Measurement tools: Gait kinematic parameters were collected using the GAITRite® system (GAITRite platinum plus classic, CIR systems Inc., Havertown, PA, United States), a portable instrumented walkway with built-in pressure sensor (as shown Figure 1A). The walkway measures 7 meters in length and 0.9 meters in width, of which the effective length is 6.1 meters and the width is 0.61 meters, containing over 18000 sensors with a sampling rate of 120 Hz. As participant walks, the system continuously recorded spatiotemporal gait data in real-time, including velocity, cadence, step length, cycle time, stride length, stance time, double support time, swing time, and step time. The GAITRite® system is factory-calibrated and undergoes regular maintenance as recommended by the manufacturer’s guidelines to ensure data accuracy and reliability. Its validity and reliability for measuring spatial and temporal gait parameters have been demonstrated in previous studies[25,26]. Handwriting kinematic parameters were recorded using dot-matrix digital pens and dot-matrix paper (TSTUDY, China), which provide coordinate parameter information for movement trajectory (Figure 1B) and have been validated for reliable handwriting analysis[27,28]. The device was also factory-calibrated and maintained according to the manufacturer’s guidelines. The digital pen is equipped with built-in pressure sensor and camera that is activated upon contact with the dot-matrix paper, enabling the real-time recording of writing pressure and speed. Additionally, the tool mimics traditional pen-and-paper writing, matching participants’ writing habits. Studies have shown that declines in geometric copying and handwriting skills correspond with cognitive decline, making these tasks useful for screening cognitive impairment in older adults; and notably, the pentagon and cube copying tasks demonstrating greater sensitivity to cognitive change than other graphic tasks[29]. Given the participants’ familiarity with Chinese characters, two graphic handwriting tasks and four Chinese character handwriting tasks were set in this study (Supplementary Table 1).
Measure methods: Gait kinematic parameters were collected in a quiet, well-lit environment with participants wearing comfortable footwear. Start and end markers were placed 1 meter from the walkway to allow acceleration and deceleration. The researcher briefed participants to ensure their comprehension before walking tests. To minimize emotional influences, participants walked the walkway 3-5 times at their usual pace, with the mean of the three most consistent trials used for final analysis[30]. Handwriting kinematic parameters were recorded with digital pens, with participants completing two copying graphic handwriting tasks: Pentagons and a cube, as shown in Tasks 1-2 (Supplementary Table 1). Task 1 was scored up to 3 points, Task 2 had a maximum of 4 points, resulting in a total graphic handwriting task score of 7 points. Another handwriting task involved writing Chinese characters, which is designed to identify cognitive deficits and motor dysfunctions in individuals with neuropsychiatric disorders[19]. Based on the task setting of previous studies and taking into account the writing habits and writing features of Chinese older adults[19,31], four Chinese character handwriting tasks were set up, including signature (Task 3), memorizing and writing three words (Task 4), naming three pictures (Task 5), and copying a sentence (Task 6). The content and scoring methods for handwriting tasks were in Supplementary Table 1. Handwriting data collection was also conducted in a quiet, well-lit setting, and participants were briefed on the task to ensure understanding before writing in their usual style.
Parameters extraction: A total of 14 gait kinematic parameters were extracted, including 1 item in velocity (velocity), 8 in the time (ambulation time, step time, cycle time, swing time, stance time, double support time, swing time variability, double support time variability), 4 in the length [step length, stride length, heel-to-heel (HH) base support, stride length variability], and 1 item in other (cadence). Twenty-six handwriting kinematic parameters were extracted: 1 item in pressure (average pressure), 13 in velocity (average velocity, average velocity in X/Y-axis, maximum velocity in X/Y-axis, minimum velocity in X/Y-axis, average acceleration in X/Y-axis, maximum acceleration in X/Y-axis, minimum acceleration in X/Y-axis), 2 in time (time in air, single stroke time), 6 in length (single stroke length, X/Y-axis stroke lengths, total single stroke length, total X/Y-axis stroke lengths), and 4 items in others (tilt angle, number of times velocity slows down, number of times acceleration slows down, task score). Supplementary Tables 2 and 3 for parameter definitions.
Participant demographic differences, along with gait and handwriting kinematic parameters between groups, were analyzed using appropriate statistical tests based on variable type and distribution. Categorical variables were analyzed using χ2 tests. For continuous variables, normality was assessed using the Shapiro-Wilk test. Normally distributed data were analyzed using independent samples t-tests, while non-normally distributed data were analyzed using Wilcoxon rank-sum tests. Statistical significance was set at P < 0.05.
To evaluate the classification accuracy between MCI and CN, five classification models were used, including logistic regression (LR), eXtreme Gradient Boosting (XGB), random forest (RF), gradient boosting classifier (GBC), and Gaussian Naive Bayes (Gaussian NB). Default hyperparameters were applied, with manual adjustments as needed. Variance thresholding and sequential feature selection algorithms were implemented to identify the optimal gait and handwriting parameters subsets. Model performance was assessed using ten-fold cross-validation, with evaluation metrics including accuracy, sensitivity, specificity, positive predictive value, negative predictive value, receiver operating characteristic curve, AUC, brier score, and decision curve analysis (DCA). Statistical analyses were conducted using SPSS 27.0, and PyCharm Community Edition 2022.2 × 64.
Ninety-five participants were recruited, including 34 with MCI and 61 cognitively normal (selection process in Figure 2). Significant differences were found in MMSE and MoCA-B scores, while other characteristics were comparable. Table 1[32,33] summarized the demographics characteristics of all participants.
| Basic characteristics | MCI group (n = 34) | CN group (n = 61) | t/χ2/Z | P value |
| Age | 84.26 ± 7.85 | 80.26 ± 11.02 | -1.868 | 0.065 |
| Sex | 0.623 | 0.430 | ||
| Male | 8 (23.53) | 19 (31.15) | ||
| Female | 26 (76.47) | 42 (68.85) | ||
| Education level | -0.953 | 0.340 | ||
| Primary school | 10 (29.41) | 23 (37.70) | ||
| Junior school | 10 (29.41) | 17 (27.87) | ||
| High School | 9 (26.47) | 16 (26.23) | ||
| College and above | 5 (14.71) | 5 (8.20) | ||
| Occupation1 | 0.411 | 0.522 | ||
| Brain work | 25 (73.53) | 41 (67.21) | ||
| Physical work | 9 (26.47) | 20 (32.79) | ||
| Marital status | 0.012 | 0.914 | ||
| Single (divorced, widowed) | 16 (47.06) | 28 (45.90) | ||
| Married | 18 (52.94) | 33 (54.10) | ||
| Residence | 2.473 | 0.116 | ||
| Living alone | 10 (29.41) | 28 (73.68) | ||
| Living with spouse/children | 24 (70.59) | 33 (54.10) | ||
| Monthly income | -0.625 | 0.532 | ||
| Low (≤ 1000) | 1 (2.94) | 0 (0.00) | ||
| Middle (1000-3000) | 1 (2.94) | 2 (3.28) | ||
| High (≥ 3000) | 32 (94.12) | 59 (96.72) | ||
| Region of residence | 1.212 | 0.271 | ||
| Urban | 32 (94.12) | 53 (86.89) | ||
| Rural | 2 (5.88) | 8 (13.11) | ||
| Physical activity2 | -1.456 | 0.145 | ||
| Low | 19 (55.88) | 25 (40.98) | ||
| Middle | 11 (32.35) | 24 (39.34) | ||
| High | 4 (11.76) | 12 (19.67) | ||
| Family history of Dementia | 2 (5.88) | 3 (4.92) | 0.041 | 0.840 |
| Current smoker | 0 (0.00) | 2 (3.28) | 1.139 | 0.286 |
| Alcohol Consumption | 0 (0.00) | 6 (9.84) | 3.570 | 0.059 |
| Multimorbidity3 | 14 (41.18) | 26 (42.62) | 0.019 | 0.891 |
| MMSE | 25.03 ± 2.04 | 27.07 ± 2.12 | 4.549 | < 0.001 |
| MoCA-B | 17.35 ± 2.89 | 23.23 ± 2.53 | 10.318 | < 0.001 |
Comparisons of kinematic parameters between the MCI and CN groups during gait and handwriting tasks revealed significant differences in gait parameters, including velocity, cadence, step length, cycle time, stride length, stance time, double support time, and double support time variability (all P < 0.05); In handwriting tasks involving graphic tasks, significant differences were found in average pressure, time in air, maximum velocity in the x and Y-axis, average acceleration in the X-axis, maximum acceleration in the x and Y-axis, and task score (all P < 0.05); in Chinese character handwriting tasks, both average pressure and task score were statistically significant between the two groups (all P < 0.05). The above results showed that compared with the CN older adults, individuals with MCI exhibited slower walking speeds, shorter stride lengths, and reduced step lengths, alongside longer standing and cycle times when walking. In handwriting tasks, they demonstrated slower writing speeds, lower pressure, extended thinking times, and an increased number of writing errors. Detailed task-related kinematic parameters were provided in Tables 2, 3, and 4.
| Parameters | MCI group (n = 34) | CN group (n = 61) | t/Z | P value |
| Ambulation time | 25.08 ± 7.55 | 22.77 ± 7.77 | -1.402 | 0.164 |
| Velocity | 84.55 (23.83) | 93.30 (25.35) | -2.911 | 0.004 |
| Cadence | 106.50 ± 11.51 | 110.91 ± 9.05 | 2.060 | 0.042 |
| Step time | 0.57 (0.08) | 0.54 (0.06) | -1.825 | 0.068 |
| Step length | 47.92 ± 6.42 | 52.56 ± 8.12 | 2.863 | 0.005 |
| Cycle time | 1.14 ± 0.12 | 1.09 ± 0.09 | -2.274 | 0.025 |
| Stride length | 95.48 ± 13.35 | 105.52 ± 16.71 | 3.005 | 0.003 |
| HH base support | 11.24 (4.51) | 10.72 (4.30) | -1.475 | 0.140 |
| Swing time | 0.40 ± 0.04 | 0.40 ± 0.03 | -0.451 | 0.653 |
| Stance time | 0.73 (0.13) | 0.69 (0.07) | -2.376 | 0.018 |
| Double support time | 0.32 (0.09) | 0.28 (0.08) | -2.717 | 0.007 |
| Stride length variability | 3.25 (1.43) | 3.13 (1.10) | -0.679 | 0.497 |
| Swing time variability | 0.02 (0.01) | 0.02 (0.01) | -1.658 | 0.097 |
| Double support time variability | 0.03 (0.01) | 0.02 (0.01) | -2.614 | 0.009 |
| Parameters | Handwriting with graphic tasks | |||
| MCI group (n = 34) | CN group (n = 61) | t/Z | P value | |
| Tilt angle | -42.07 (22.51) | -39.79 (19.18) | -0.823 | 0.411 |
| Average pressure | 593.05 (258.14) | 666.84 (172.65) | -2.135 | 0.033 |
| Time in air | 25920.00 (18039.00) | 17468.00 (11452.00) | -2.865 | 0.004 |
| Single stroke time | 1526.07 (1304.64) | 1605.43 (1058.23) | -0.116 | 0.907 |
| Single stroke length | 13.23 (7.44) | 14.84 (7.15) | -1.623 | 0.105 |
| X-axis stroke length | 8.22 (4.41) | 9.34 (4.69) | -1.669 | 0.095 |
| Y-axis stroke length | 8.32 (4.63) | 9.61 (5.13) | -1.304 | 0.192 |
| Total single stroke length | 255.93 (71.32) | 260.29 (77.99) | -0.155 | 0.877 |
| Total X-axis stroke length, mean (SD) | 167.93 ± 36.92 | 168.48 ± 35.30 | 0.072 | 0.943 |
| Total Y-axis stroke length | 157.47 (50.29) | 157.95 (47.77) | -0.738 | 0.461 |
| Average velocity | 10.40 (4.93) | 11.12 (5.53) | -1.623 | 0.105 |
| Average velocity in X-axis | 6.91 (2.92) | 8.12 (3.46) | -1.933 | 0.053 |
| Average velocity in Y-axis | 7.59 (3.34) | 7.71 (3.57) | -1.102 | 0.270 |
| Maximum velocity in X-axis | 29.07 (9.53) | 36.22 (16.09) | -3.237 | 0.001 |
| Minimum velocity in X-axis | 0.14 (0.32) | 0.08 (0.25) | -0.927 | 0.354 |
| Maximum velocity in Y-axis | 33.27 (16.84) | 38.45 (17.21) | -1.723 | 0.085 |
| Minimum velocity in Y-axis | 0.13 (0.26) | 0.11 (0.25) | -0.617 | 0.537 |
| Number of times velocity slows down | 26.33 (22.87) | 28.95 (23.08) | -0.078 | 0.938 |
| Average acceleration in X-axis | 0.21 (0.07) | 0.25 (0.09) | -2.880 | 0.004 |
| Average acceleration in Y-axis | 0.23 (0.11) | 0.26 (0.08) | -1.987 | 0.047 |
| Maximum acceleration in X-axis | 1.07 (0.32) | 1.38 (0.55) | -3.998 | < 0.001 |
| Minimum acceleration in X-axis | 0.00 (0.01) | 0.00 (0.01) | -0.163 | 0.870 |
| Maximum acceleration in Y-axis | 1.27 (0.64) | 1.45 (0.65) | -2.050 | 0.040 |
| Minimum acceleration in Y-axis | 0.00 (0.01) | 0.00 (0.01) | -0.513 | 0.608 |
| Number of times acceleration slows down | 26.19 (24.49) | 30.21 (24.99) | -0.039 | 0.969 |
| Task score | 5.00 (3.00) | 6.00 (2.00) | -2.447 | 0.014 |
| Parameters | Handwriting with Chinese character tasks | |||
| MCI group (n = 34) | CN group (n = 61) | t/Z | P value | |
| Tilt angle | -57.08 (9.06) | -54.93 (8.81) | -0.668 | 0.504 |
| Average pressure | 499.05 (213.36) | 582.68 (233.95) | -2.135 | 0.033 |
| Time in air | 36336.00 (25185.00) | 29228.00 (21512.00) | -1.394 | 0.163 |
| Single stroke time | 352.1 (152.30) | 356.65 (160.66) | -0.303 | 0.762 |
| Single stroke length | 8.93 (3.87) | 9.09 (5.10) | -0.753 | 0.451 |
| X-axis stroke length | 5.35 (2.53) | 5.20 (3.10) | -0.738 | 0.461 |
| Y-axis stroke length | 6.02 (2.66) | 6.26 (3.69) | -0.706 | 0.480 |
| Total single stroke length | 791.40 ± 151.25 | 829.42 ± 175.50 | 1.062 | 0.291 |
| Total X-axis stroke length | 455.63 ± 99.02 | 478.62 ± 99.95 | 1.078 | 0.284 |
| Total Y-axis stroke length | 521.39 (132.81) | 557.07 (181.17) | -0.831 | 0.406 |
| Average velocity | 25.93 ± 8.70 | 27.69 ± 9.31 | 0.907 | 0.367 |
| Average velocity in X-axis | 17.29 ± 6.17 | 18.55 ± 6.47 | 0.919 | 0.360 |
| Average velocity in Y-axis | 21.28 ± 7.44 | 22.41 ± 7.89 | 0.680 | 0.498 |
| Maximum velocity in X-axis | 51.57 ± 17.45 | 55.05 ± 16.51 | 0.964 | 0.338 |
| Minimum velocity in X-axis | 1.47 (1.63) | 1.58 (1.32) | -0.109 | 0.913 |
| Maximum velocity in Y-axis | 61.86 ± 20.04 | 65.30 ± 20.87 | 0.781 | 0.437 |
| Minimum velocity in Y-axis | 2.02 (1.55) | 1.63 (1.27) | -0.606 | 0.545 |
| Number of times velocity slows down | 6.22 (2.97) | 6.07 (3.19) | -0.008 | 0.994 |
| Average acceleration in X-axis | 0.59 (0.30) | 0.61 (0.34) | -0.660 | 0.509 |
| Average acceleration in Y-axis | 0.67 (0.45) | 0.72 (0.35) | -0.481 | 0.630 |
| Maximum acceleration in X-axis | 1.64 (0.93) | 1.78 (0.99) | -1.196 | 0.232 |
| Minimum acceleration in X-axis | 0.08 (0.11) | 0.07 (0.09) | -0.124 | 0.901 |
| Maximum acceleration in Y-axis | 1.97 (0.80) | 2.08 (1.13) | -1.079 | 0.281 |
| Minimum acceleration in Y-axis | 0.11 (0.14) | 0.10 (0.11) | -0.109 | 0.913 |
| Number of times acceleration slows down | 5.60 (3.11) | 5.37 (3.07) | -0.093 | 0.926 |
| Task score | 20.00 (0.00) | 20.00 (1.00) | -3.078 | 0.002 |
The unimodal classification task consists of two components: Kinematic parameters from the gait task and the handwriting task, which includes both graphics and Chinese character handwriting tasks. Considering that the graphic and Chinese character handwriting tasks have large number and consistent parameter categories, they are prone to overfitting[34], the multimodal classification task incorporates combinations of kinematic parameters from the gait and graphics handwriting tasks, as well as the gait and Chinese character handwriting tasks.
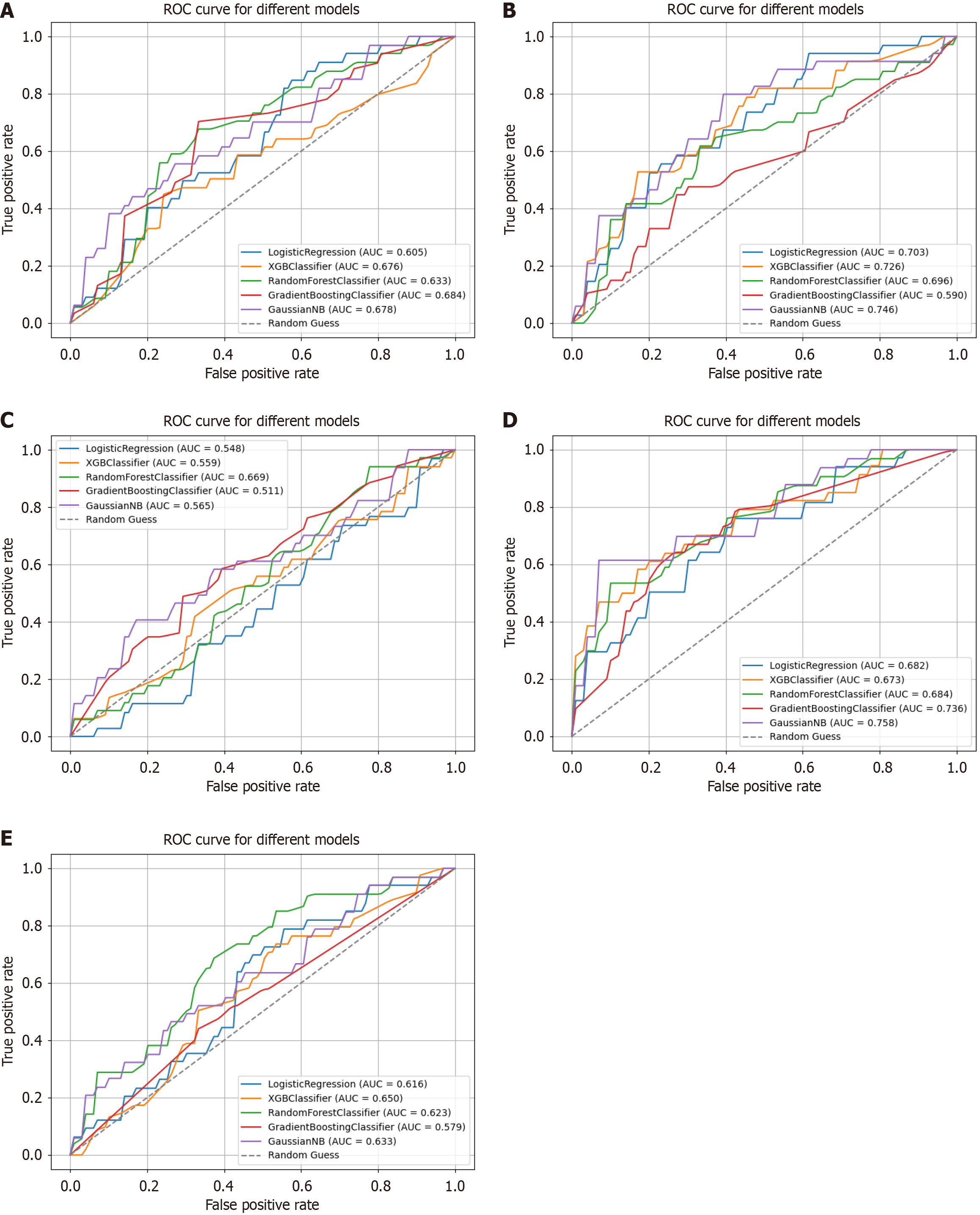
Unimodal analysis of kinematic parameters in gait tasks: All gait kinematic parameters were included in the unimodal classification model. In the gait tasks, the optimal gait parameter subsets differed across models: The LR model included velocity, step length, and cycle time; the XGB model included cadence and cycle time; the RF model included ambulation time, cadence, and cycle time; the GBC model included cadence, step time, and cycle time; and the Gaussian NB model included ambulation time, velocity, cadence, and step length. The GBC and Gaussian NB models demonstrated the highest classification performance, with an accuracy of 69.44% and an AUC of 0.68 (Table 5, Figure 3A). DCA showed that the RF and Gaussian NB models offered a greater net benefit in the testing cohort at a threshold probability range of 0.2 to 0.7. The DCA was shown in Supplementary Figure 1.
| LR (%) | XGB (%) | RF (%) | GBC (%) | Gaussian NB (%) | |
| Gait tasks | |||||
| Accuracy | 66.33 | 69.33 | 63.00 | 69.44 | 69.44 |
| Sensitivity | 29.17 | 49.83 | 44.50 | 54.00 | 50.67 |
| Specificity | 91.87 | 85.44 | 82.06 | 82.88 | 85.02 |
| Positive predictive value | 53.33 | 65.33 | 49.17 | 64.17 | 64.00 |
| Negative predictive value | 68.00 | 73.74 | 71.94 | 73.53 | 72.90 |
| AUC | 0.61 | 0.68 | 0.63 | 0.68 | 0.68 |
| Brier | 0.34 | 0.31 | 0.37 | 0.31 | 0.31 |
| Graphics handwriting tasks | |||||
| Accuracy | 69.56 | 72.44 | 70.44 | 60.78 | 73.44 |
| Sensitivity | 54.17 | 64.00 | 57.33 | 43.67 | 66.00 |
| Specificity | 86.51 | 81.21 | 81.81 | 74.41 | 83.27 |
| Positive predictive value | 66.67 | 72.00 | 69.00 | 43.33 | 71.67 |
| Negative predictive value | 74.82 | 75.82 | 73.19 | 68.51 | 78.76 |
| AUC | 0.70 | 0.73 | 0.70 | 0.59 | 0.75 |
| Brier | 0.30 | 0.28 | 0.30 | 0.39 | 0.27 |
| Chinese character handwriting tasks | |||||
| Accuracy | 63.22 | 63.00 | 64.44 | 55.78 | 68.44 |
| Sensitivity | 18.67 | 26.83 | 59.33 | 25.33 | 20.67 |
| Specificity | 90.98 | 84.88 | 74.45 | 76.95 | 92.40 |
| Positive predictive value | 31.67 | 34.17 | 53.50 | 39.17 | 36.67 |
| Negative predictive value | 66.32 | 67.99 | 74.51 | 65.13 | 68.37 |
| AUC | 0.55 | 0.56 | 0.67 | 0.51 | 0.57 |
| Brier | 0.37 | 0.37 | 0.36 | 0.42 | 0.32 |
| Gait and graphics handwriting tasks | |||||
| Accuracy | 69.67 | 69.44 | 71.67 | 74.44 | 73.67 |
| Sensitivity | 48.67 | 57.67 | 46.67 | 59.00 | 68.50 |
| Specificity | 87.78 | 76.90 | 90.10 | 88.21 | 83.17 |
| Positive predictive value | 66.67 | 57.50 | 55.83 | 77.50 | 68.33 |
| Negative predictive value | 72.66 | 72.74 | 72.63 | 75.73 | 80.29 |
| AUC | 0.68 | 0.67 | 0.68 | 0.74 | 0.76 |
| Brier | 0.30 | 0.31 | 0.28 | 0.26 | 0.26 |
| Gait and Chinese character handwriting tasks | |||||
| Accuracy | 62.44 | 68.44 | 67.44 | 60.33 | 69.78 |
| Sensitivity | 42.50 | 44.33 | 37.00 | 39.50 | 41.50 |
| specificity | 80.66 | 85.71 | 87.52 | 76.39 | 85.04 |
| Positive predictive value | 47.50 | 56.67 | 58.17 | 44.83 | 57.50 |
| Negative predictive value | 69.86 | 71.77 | 71.81 | 67.96 | 71.92 |
| AUC | 0.62 | 0.65 | 0.62 | 0.58 | 0.63 |
| Brier | 0.38 | 0.32 | 0.33 | 0.40 | 0.30 |
Unimodal analysis of kinematic parameters in graphic handwriting tasks: All graphic handwriting kinematic parameters were incorporated into the unimodal classification model. For the graphics handwriting tasks, the optimal handwriting parameter subsets differed by model: The LR model selected seven items (time in air, total X-axis stroke length, maximum velocity in the X and Y-axis, minimum velocity in the X and Y-axis, and average acceleration in the Y-axis); the XGB model selected four items (X-axis stroke length, total single stroke length, minimum velocity in the X-axis, and average acceleration in the Y-axis), the RF model identified six items (average pressure, total single stroke length, minimum velocity in the X-axis, average acceleration in both X and Y-axis, and maximum acceleration in the X-axis), the GBC model included 13 items (tilt angle, time in air, single stroke time, single stroke length, X-axis stroke length, total single stroke length, total X-axis stroke length, average velocity, average velocity in the Y-axis, minimum velocity in the X-axis, number of times velocity slows down, average acceleration in the Y-axis, and minimum acceleration in the X-axis); and the Gaussian NB model incorporated five items (time in air, Y-axis stroke length, total Y-axis stroke length, average velocity, and average acceleration in the Y-axis). The classification results showed that the Gaussian NB model had the highest classification performance (accuracy = 73.44%, AUC = 0.75) (Table 5, Figure 3B). DCA indicated that the Gaussian NB model provided greater net benefit at threshold probabilities of 0.2 to 0.7 in the testing cohort. The DCA was shown in Supplementary Figure 1.
Unimodal analysis of kinematic parameters in Chinese character handwriting tasks: All Chinese character handwriting kinematic parameters were incorporated into the model. In the Chinese character handwriting tasks, the optimal handwriting parameters subsets varied across models, with 14 items identified for LR (average pressure, time in air, single stroke time, single stroke length, X and Y-axis stroke length, average velocity, average velocity in the X-axis, minimum velocity in the X-axis, number of times velocity slows down, average acceleration in the x and Y-axis, and maximum acceleration in the x and Y-axis), 10 for XGB (tilt angle, time in air, X and Y-axis stroke length, total Y-axis stroke length, average velocity, minimum velocity in the X-axis, average acceleration in the X-axis, and minimum acceleration in the X and Y-axis), two for RF (minimum velocity in the Y-axis and minimum acceleration in the Y-axis), three for GBC (Y-axis stroke length, minimum velocity in the Y-axis, and minimum acceleration in the Y-axis), and five for Gaussian NB (single stroke length, X-axis stroke length, total Y-axis stroke length, minimum velocity in the X-axis, and minimum acceleration in the Y-axis). The Gaussian NB model achieved the highest classification performance (accuracy = 68.44%, AUC = 0.57) (Table 5, Figure 3C). DCA showed that the GBC and Gaussian NB models provided a greater net benefit at threshold probabilities of 0.3 to 0.5 in the testing cohort. The DCA was presented in Supplementary Figure 1.
Multimodal analysis of kinematic parameters in gait and graphic handwriting tasks: In gait and graphic handwriting tasks, the optimal multimodal parameters subsets identified varied by model, with eight items selected for LR (step time, step length, stride length, stance time, single stroke time, total single stroke length, total X-axis stroke length, and maximum velocity in the X-axis), nine for XGB (velocity, stance time, stride length variability, double support time variability, average pressure, total X-axis stroke length, average velocity in the X and Y-axis, and maximum velocity in the X-axis), eight for RF (ambulation time, velocity, stride length variability, average pressure, total X-axis stroke length, average velocity in the Y-axis, and maximum velocity in the X and Y-axis), six for GBC (velocity, cycle time, double support time, total X-axis stroke length, and maximum velocity in the x and Y-axis), and 14 for Gaussian NB (velocity, cadence, step time, step length, stride length, HH base support, double support time, time in air, single stroke time, total X and Y-axis stroke length, average velocity in the Y-axis, maximum velocity in the X-axis, and minimum velocity in the Y-axis). The GBC model attained the highest classification performance (accuracy = 74.44%, AUC = 0.74) (Table 5, Figure 3D). Additionally, the gait and graphics handwriting tasks also had the lowest brier score among all classification tasks at 0.26. DCA showed that the Gaussian NB model yielded a greater net benefit at threshold probabilities between 0.2 and 0.7 in the testing cohort. The DCA was provided in Supplementary Figure 1. In addition, a MCI screener was developed based on gait and handwriting parameters (Supplementary Figure 2).
Multimodal analysis of kinematic parameters in gait and Chinese character handwriting tasks: In gait and Chinese character handwriting tasks, the multimodal parameters subsets included 17 items for LR (velocity, step length, step length, cycle time, stride length, HH base support, stance time, double support time, swing time variability, double support time variability, average pressure, time in air, total Y-axis stroke length, average velocity in the Y-axis, maximum velocity in the X and Y-axis, and minimum velocity in the X-axis), four for XGB (cadence, step length, single stroke time, and number of times velocity slows down), three for RF (cycle time, stance time, and average velocity), four for GBC (cycle time, stride length variability, total Y-axis stroke length, and maximum velocity in the Y-axis), four for Gaussian NB (ambulation time, velocity, swing time, and number of times velocity slows down). The classification results showed that the Gaussian NB model achieved the highest classification performance (accuracy = 69.78%, AUC = 0.63) (Table 5, Figure 3E). DCA showed that the RF and Gaussian NB models offered greater net benefit in the testing cohort at threshold probabilities of 0.2 to 0.6. The DCA was presented in Supplementary Figure 1.
This study is the first to introduce a novel screening tool for MCI by integrating gait and handwriting kinematic parameter analyses. Eight gait parameters, eight graphics parameters and two Chinese character parameters were identified as significantly differentiating older adults with MCI and from cognitively normal controls. Utilizing machine learning classification models, the combined gait and graphic handwriting tasks achieved a 74.44% accuracy, surpassing single tasks and the combination of gait and Chinese character handwriting tasks, underscoring the superior efficacy of the integrated approach.
Notably, the gait measurement equipment in this study was non-invasive, requiring no sensors to be worn by participants, thereby ensuring that the data collected were both objective and unbiased. Moreover, this study expanded upon previously identified gait parameter by incorporating a more extensive analysis of 14 parameters, including cadence, HH base support, and double support time variability, providing a more comprehensive evaluation of gait data. The findings of this study indicated that older adults with MCI exhibit slower gait speeds compared to cognitively normal older adults, aligning with the majority of prior studies[35,36]. Given that gait kinematic parameters are linked to cognitive functions in specific brain regions, slower gait speed may be indicative of declines in executive function, processing speed, and visuospatial abilities[36,37]. Furthermore, the association between gait slowing and cognitive impairment is supported by a shared neural substrate, including a smaller right hippocampus, with cognitive decline being linked to hippocampal volume reduction[38]. Additionally, this study found increased double support time variability in older adults with MCI, consistent with the findings of Jayakody et al[37]. Increased double support time variability may be related to declining memory functions. Moreover, older adults with MCI demonstrate higher cadence, shorter stride and step lengths, and longer cycle, standing and double support times, which may indicate decreased balance[39]. Decreased balance may be attributed to diminished visual function. Previous studies have shown reduced gray matter volume in the right periaqueductal area, indicating the periaqueductal gyrus is vital for the visual network[9]. While cognitively normal older adults maintain balance through visual feedback, cognitive decline in MCI may impair this function[40]. Additionally, balance decline may be associated with hippocampal dysfunction, given the hippocampus’s role in sensorimotor integration during spatial navigation[41,42]. Although stride time variability did not significantly differ between the two groups in this study, older adults with MCI still exhibited higher stride time variability than cognitively normal controls. This could be due to stride time variability being more sensitive under dual-task gait assessments[43]. Future studies should consider dual-task gait kinematics measurements to capture more distinguishing indicators.
The dot-matrix digital pen and paper used in this study preserved the advantages of traditional pen-and-paper writing, providing a more natural environment for participants while also incorporating the advantages of electronic recording, enabling real-time capture of spatial and temporal data during the writing process[16,31]. Our study observed that older adults with MCI had prolonged in-air time during writing, indicating a need for additional thinking time, which may be linked to declines in cognitive function and visuospatial ability[17,18]. Additionally, the slowing of writing speed, especially in the X- and Y-axes, showed varying degrees of reduced speed and acceleration, consistent with findings from previous studies[19,44]. Fine-motor studies suggest that horizontal and vertical movements are primarily executed through wrist and finger movements, making X- and Y-axis stroke speeds may be more reflective of fine-motor impairment[17,19]. Although Kawa et al[44] reported that older adults with MCI tended to write in larger font sizes, both the present study and Yu et al[17] found no significant difference in writing length between the groups, potentially due to differences in the writing tasks employed. In fact, conclusions regarding changes in writing pressure among older adults with MCI remain inconsistent. While Zhào et al[19] found higher writing pressure in MCI, Yu et al[17] observed no significant changes, and the present study found that older adults with MCI exhibited lower writing pressure, consistent with the findings of Werner et al[18]. This variation thought to be a manifestation of fine-motor control deficits[17-19,45]. A possible inference is that writing pressure may be related to cognitive function, with groups with poorer cognitive functioning having lower writing pressure[18]. Furthermore, the present study assessed writing correctness and found that older adults with MCI had significantly lower writing scores than cognitively normal older adults, supporting the conclusion of cognitive decline in this population.
In the unimodal categorization results, the graphic handwriting task demonstrated highest accuracy. This finding is likely because graphic handwriting tasks, particularly replicating pentagons and squares, are more challenging and thus more sensitive to cognitive decline[27]. In contrast, the signature and Chinese character copying tasks were relatively less demanding on cognitive function[16,29,46]. In addition, text writing, being more frequently used in daily life with a higher degree of automation and lower cognitive demands compared to graphic replication, may have lower discriminative power in identifying MCI among older adults with some education[16,46]. Combining gait and handwriting kinematic parameters in this study, the accuracy of the multimodal classification results was improved to 74.4%. MCI-related impairments in multiple cognitive domains often manifest as subtle changes in motor performance. Gait ab
This study introduces a novel approach to MCI-assisted screening by integrating gait and handwriting kinematic parameters, offering important clinical implications. The objective, quantifiable nature of kinematics analysis enhances screening accuracy over traditional cognitive tests, enabling a standardized evaluation of cognitive function that minimizes the influence of educational or linguistic factors[48]. This combined approach may provide a sensitive and comprehensive method for early detection by engaging multiple cognitive domains, thereby improving the precision in identifying individuals at risk of cognitive decline. While the present analysis focused on individuals with confirmed MCI and cognitively normal controls, this approach may also hold promise for identifying elevated MCI risk in broader populations without prior cognitive screening. Validation of this method in diverse settings is essential to confirm its predictive utility and to support its integration into clinical and public health protocols aimed at early intervention. Additionally, portable technologies such as cranio- corpo- graphy (CCG) have been demonstrated to sensitively detect subclinical gait disturbances via balance assessments[49]. The present study utilized the GAITRite® system for comprehensive gait analysis, CCG may offer complementary insights, particularly in settings requiring cost-effective and portable solutions. Integrating these technologies in future study could enhance screening sensitivity and broaden applicability across diverse settings.
This study has several limitations that must be considered: (1) The study’s focus on Huzhou with a relatively small sample size limits the generalizability of the findings. Moreover, potential marginal effects due to differences in age distribution between groups may have impacted the findings. Future research with larger, more diverse populations is warranted to confirm these results; (2) This study only included older adults with MCI without distinguishing specific subtypes (e.g., amnesic MCI and non-memory MCI). Future research should differentiate these subtypes to better understand how they uniquely affect gait and handwriting kinematics, which could enhance the precision and effectiveness of community-based screening efforts; (3) The inclusion of only right-hand dominant participants limits the generalizability of the findings to individuals with left or mixed-hand dominance. Future studies should include participants with varying hand dominances to improve both generalizability and clinical relevance; (4) Gait kinematic parameters was assessed using a single-task paradigm in the present study; however, incorporating dual-task gait measurements in future research may provide a more comprehensive understanding of motor and cognitive dysfunction in older adults with MCI; (5) Although the multimodal approach achieved 74.4% classification accuracy, the absence of explicit memory-related assessments, integral to MCI diagnosis, may have constrained the sensitivity. In addition, the analysis in this study relied solely on behavioral data, which may limit its ability to fully capture the complexity of cognitive impairment. Future studies should consider incorporating memory tasks as well as methods such as neu
This study introduces a promising approach for effectively distinguishing older adults with MCI by combining gait and handwriting kinematic parameters, demonstrating potential for early detection. The integration of motor-cognitive assessments may improve classification accuracy and provide a practical, scalable tool for community-based MCI screening. Further validation in diverse populations is needed to enhance early intervention strategies for MCI.
The authors would like to thank all participants in this research.
| 1. | Petersen RC, Caracciolo B, Brayne C, Gauthier S, Jelic V, Fratiglioni L. Mild cognitive impairment: a concept in evolution. J Intern Med. 2014;275:214-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 846] [Cited by in RCA: 1192] [Article Influence: 99.3] [Reference Citation Analysis (0)] |
| 2. | Pessoa RMP, Bomfim AJL, Ferreira BLC, Chagas MHN. Diagnostic criteria and prevalence of mild cognitive impairment in older adults living in the community: a systematic review and meta-analysis. Arch Clin Psychiatry (São Paulo). 2019;46:72-79. [RCA] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 3. | Cong L, Ren Y, Wang Y, Hou T, Dong Y, Han X, Yin L, Zhang Q, Feng J, Wang L, Tang S, Grande G, Laukka EJ, Du Y, Qiu C. Mild cognitive impairment among rural-dwelling older adults in China: A community-based study. Alzheimers Dement. 2023;19:56-66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 105] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 4. | Hu C, Yu D, Sun X, Zhang M, Wang L, Qin H. The prevalence and progression of mild cognitive impairment among clinic and community populations: a systematic review and meta-analysis. Int Psychogeriatr. 2017;29:1595-1608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 135] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 5. | Xue H, Hou P, Li Y, Mao X, Wu L, Liu Y. Factors for predicting reversion from mild cognitive impairment to normal cognition: A meta-analysis. Int J Geriatr Psychiatry. 2019;34:1361-1368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 6. | Jia L, Du Y, Chu L, Zhang Z, Li F, Lyu D, Li Y, Li Y, Zhu M, Jiao H, Song Y, Shi Y, Zhang H, Gong M, Wei C, Tang Y, Fang B, Guo D, Wang F, Zhou A, Chu C, Zuo X, Yu Y, Yuan Q, Wang W, Li F, Shi S, Yang H, Zhou C, Liao Z, Lv Y, Li Y, Kan M, Zhao H, Wang S, Yang S, Li H, Liu Z, Wang Q, Qin W, Jia J; COAST Group. Prevalence, risk factors, and management of dementia and mild cognitive impairment in adults aged 60 years or older in China: a cross-sectional study. Lancet Public Health. 2020;5:e661-e671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1453] [Cited by in RCA: 1208] [Article Influence: 201.3] [Reference Citation Analysis (0)] |
| 7. | Du S, Ma X, Wang J, Mi Y, Zhang J, Du C, Li X, Tan H, Liang C, Yang T, Shi W, Zhang G, Tian Y. Spatiotemporal gait parameter fluctuations in older adults affected by mild cognitive impairment: comparisons among three cognitive dual-task tests. BMC Geriatr. 2023;23:603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 8. | Haque RU, Pongos AL, Manzanares CM, Lah JJ, Levey AI, Clifford GD. Deep Convolutional Neural Networks and Transfer Learning for Measuring Cognitive Impairment Using Eye-Tracking in a Distributed Tablet-Based Environment. IEEE Trans Biomed Eng. 2021;68:11-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 9. | Kim SY, Park J, Choi H, Loeser M, Ryu H, Seo K. Digital Marker for Early Screening of Mild Cognitive Impairment Through Hand and Eye Movement Analysis in Virtual Reality Using Machine Learning: First Validation Study. J Med Internet Res. 2023;25:e48093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 10. | Saini M, Subramanian MS, Rao AR, Thakral M, Singh V, Chakrawarty A, Chatterjee P, Dey AB. Gait Parameters Change can be an Early Marker of Cognitive Impairment. Neurol India. 2024;72:603-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Kueper JK, Speechley M, Lingum NR, Montero-Odasso M. Motor function and incident dementia: a systematic review and meta-analysis. Age Ageing. 2017;46:729-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 101] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 12. | Wang Y, Yang Q, Tian C, Zeng J, Yang M, Li J, Mao J. A dual-task gait test detects mild cognitive impairment with a specificity of 91.2. Front Neurosci. 2022;16:1100642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 13. | Shahzad A, Dadlani A, Lee H, Kim K. Automated Prescreening of Mild Cognitive Impairment Using Shank-Mounted Inertial Sensors Based Gait Biomarkers. IEEE Access. 2022;10:15835-15844. [DOI] [Full Text] |
| 14. | Lin J, Xu T, Yang X, Yang Q, Zhu Y, Wan M, Xiao X, Zhang S, Ouyang Z, Fan X, Sun W, Yang F, Yuan L, Bei Y, Wang J, Guo J, Tang B, Shen L, Jiao B. A detection model of cognitive impairment via the integrated gait and eye movement analysis from a large Chinese community cohort. Alzheimers Dement. 2024;20:1089-1101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 36] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 15. | Spooner RK, Bahners BH, Schnitzler A, Florin E. Time-resolved quantification of fine hand movements as a proxy for evaluating bradykinesia-induced motor dysfunction. Sci Rep. 2024;14:5340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 16. | Fernandes CP, Montalvo G, Caligiuri M, Pertsinakis M, Guimarães J. Handwriting Changes in Alzheimer's Disease: A Systematic Review. J Alzheimers Dis. 2023;96:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 17. | Yu NY, Chang SH. Characterization of the fine motor problems in patients with cognitive dysfunction - A computerized handwriting analysis. Hum Mov Sci. 2019;65:S0167-9457(17)30841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 18. | Werner P, Rosenblum S, Bar-On G, Heinik J, Korczyn A. Handwriting process variables discriminating mild Alzheimer's disease and mild cognitive impairment. J Gerontol B Psychol Sci Soc Sci. 2006;61:P228-P236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 84] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 19. | Zhào H, Zhang Y, Xia C, Liu Y, Li Z, Huang Y. Digital Handwriting Analysis of Characters in Chinese Patients with Mild Cognitive Impairment. J Vis Exp. 2021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 20. | Kobayashi M, Yamada Y, Shinkawa K, Nemoto M, Nemoto K, Arai T. Automated Early Detection of Alzheimer's Disease by Capturing Impairments in Multiple Cognitive Domains with Multiple Drawing Tasks. J Alzheimers Dis. 2022;88:1075-1089. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 21. | Yim D, Yeo TY, Park MH. Mild cognitive impairment, dementia, and cognitive dysfunction screening using machine learning. J Int Med Res. 2020;48:300060520936881. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 22. | Chandramouleeshwaran S, Khan WU, Inglis F, Rajji TK. Impact of psychotropic medications on cognition among older adults: a systematic review. Int Psychogeriatr. 2024;36:1110-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 23. | Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2004;256:183-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6176] [Cited by in RCA: 5613] [Article Influence: 255.1] [Reference Citation Analysis (0)] |
| 24. | Chen KL, Xu Y, Chu AQ, Ding D, Liang XN, Nasreddine ZS, Dong Q, Hong Z, Zhao QH, Guo QH. Validation of the Chinese Version of Montreal Cognitive Assessment Basic for Screening Mild Cognitive Impairment. J Am Geriatr Soc. 2016;64:e285-e290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 333] [Cited by in RCA: 303] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 25. | Bilney B, Morris M, Webster K. Concurrent related validity of the GAITRite walkway system for quantification of the spatial and temporal parameters of gait. Gait Posture. 2003;17:68-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 528] [Cited by in RCA: 524] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 26. | McDonough AL, Batavia M, Chen FC, Kwon S, Ziai J. The validity and reliability of the GAITRite system's measurements: A preliminary evaluation. Arch Phys Med Rehabil. 2001;82:419-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 354] [Cited by in RCA: 331] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 27. | Qi H, Zhang R, Wei Z, Zhang C, Wang L, Lang Q, Zhang K, Tian X. A study of auxiliary screening for Alzheimer's disease based on handwriting characteristics. Front Aging Neurosci. 2023;15:1117250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 28. | Wei ZQ, Zhang RY, Zhang C, Su LM, Huang C, Zhang JW, Qian MC, Qi HN, Wang LN. [Characteristics and application value of handwriting in elderly patients with mild cognitive impairment]. Zhongguo Quanke Yixue. 2023;26:1224-1233. [DOI] [Full Text] |
| 29. | Ericsson K, Forssell LG, Holmén K, Viitanen M, Winblad B. Copying and handwriting ability in the screening of cognitive dysfunction in old age. Arch Gerontol Geriatr. 1996;22:103-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 30. | Montero-Odasso M, Bergman H, Phillips NA, Wong CH, Sourial N, Chertkow H. Dual-tasking and gait in people with mild cognitive impairment. The effect of working memory. BMC Geriatr. 2009;9:41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 153] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 31. | Cilia ND, Stefano CD, Fontanella F, Di Freca AS. An Experimental Protocol to Support Cognitive Impairment Diagnosis by using Handwriting Analysis. Procedia Computer Sci. 2018;141:466-471. [RCA] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 32. | Kirk MA, Rhodes RE. Occupation correlates of adults' participation in leisure-time physical activity: a systematic review. Am J Prev Med. 2011;40:476-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 194] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 33. | Fan M, Lyu J, He P. [Chinese guidelines for data processing and analysis concerning the International Physical Activity Questionnaire]. Zhonghua Liu Xing Bing Xue Za Zhi. 2014;35:961-964. [PubMed] |
| 34. | Aliferis C, Simon G. Overfitting, Underfitting and General Model Overconfidence and Under-Performance Pitfalls and Best Practices in Machine Learning and AI. 2024 Mar 5. In: Artificial Intelligence and Machine Learning in Health Care and Medical Sciences: Best Practices and Pitfalls [Internet]. Cham (CH): Springer; 2024–. [PubMed] |
| 35. | Kostic E, Kwak K, Kim D. Changes in sensory, postural stability and gait functions depending on cognitive decline, and possible markers for detection of cognitive status. BMC Med Inform Decis Mak. 2022;22:252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 36. | Lindh-Rengifo M, Jonasson SB, Ullén S, Stomrud E, Palmqvist S, Mattsson-Carlgren N, Hansson O, Nilsson MH. Components of gait in people with and without mild cognitive impairment. Gait Posture. 2022;93:83-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 37. | Jayakody O, Breslin M, Srikanth VK, Callisaya ML. Gait Characteristics and Cognitive Decline: A Longitudinal Population-Based Study. J Alzheimers Dis. 2019;71:S5-S14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 38. | Rosso AL, Verghese J, Metti AL, Boudreau RM, Aizenstein HJ, Kritchevsky S, Harris T, Yaffe K, Satterfield S, Studenski S, Rosano C. Slowing gait and risk for cognitive impairment: The hippocampus as a shared neural substrate. Neurology. 2017;89:336-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 128] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 39. | Marchetti GF, Whitney SL, Blatt PJ, Morris LO, Vance JM. Temporal and spatial characteristics of gait during performance of the Dynamic Gait Index in people with and people without balance or vestibular disorders. Phys Ther. 2008;88:640-651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 62] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 40. | Kuan YC, Huang LK, Wang YH, Hu CJ, Tseng IJ, Chen HC, Lin LF. Balance and gait performance in older adults with early-stage cognitive impairment. Eur J Phys Rehabil Med. 2021;57:560-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 41. | Leandri M, Cammisuli S, Cammarata S, Baratto L, Campbell J, Simonini M, Tabaton M. Balance features in Alzheimer's disease and amnestic mild cognitive impairment. J Alzheimers Dis. 2009;16:113-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 73] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 42. | Coughlan G, Laczó J, Hort J, Minihane AM, Hornberger M. Spatial navigation deficits - overlooked cognitive marker for preclinical Alzheimer disease? Nat Rev Neurol. 2018;14:496-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 302] [Article Influence: 43.1] [Reference Citation Analysis (0)] |
| 43. | Bahureksa L, Najafi B, Saleh A, Sabbagh M, Coon D, Mohler MJ, Schwenk M. The Impact of Mild Cognitive Impairment on Gait and Balance: A Systematic Review and Meta-Analysis of Studies Using Instrumented Assessment. Gerontology. 2017;63:67-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 300] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 44. | Kawa J, Bednorz A, Stępień P, Derejczyk J, Bugdol M. Spatial and dynamical handwriting analysis in mild cognitive impairment. Comput Biol Med. 2017;82:21-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 45. | Chernov Y. Handwriting Markers for the Onset of Alzheimer's Disease. Curr Alzheimer Res. 2024;20:791-801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 46. | Fernandes C, Montalvo G, Pertsinakis M, Guimarães J. Signature Execution in Alzheimer’s Disease: An Analysis of Motor Features. In: Carmona-Duarte C, Diaz M, Ferrer MA, Morales A, editors. Intertwining Graphonomics with Human Movements. IGS 2022. Lecture Notes in Computer Science, vol 13424. Cham: Springer, 2022: 349-354. [DOI] [Full Text] |
| 47. | Mc Ardle R, Galna B, Donaghy P, Thomas A, Rochester L. Do Alzheimer's and Lewy body disease have discrete pathological signatures of gait? Alzheimers Dement. 2019;15:1367-1377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 48. | Beauchet O, Launay CP, Sejdić E, Allali G, Annweiler C. Motor imagery of gait: a new way to detect mild cognitive impairment? J Neuroeng Rehabil. 2014;11:66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 49. | Bogdanova D. Gait disorders in unipolar and bipolar depression. Heliyon. 2023;9:e15864. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/