Published online Sep 19, 2025. doi: 10.5498/wjp.v15.i9.109458
Revised: June 27, 2025
Accepted: July 24, 2025
Published online: September 19, 2025
Processing time: 90 Days and 2.2 Hours
Treatment-resistant depression (TRD) has a poor response to clinical treatment. Patients with TRD do not respond adequately to standard antidepressants. Even after receiving a full dose and sufficient duration of combined antidepressant therapy, significant improvement is still difficult to achieve. At present, electroco
To explore the effect of esketamine and etomidate anesthesia on the clinical efficacy of ECT for TRD.
A total of 120 patients with TRD, treated at the Department of Psychiatry, The Second Affiliated Hospital of Shandong First Medical University, China between April 2020 and April 2024, were selected for the study. The patients were allocated at random into two groups using a random number table: The combination and control groups, with 60 patients in each group. Both groups underwent ECT; the combination group received esketamine and etomidate anesthesia, while the control group received etomidate anesthesia. The following parameters were compared between the two groups: Heart rate (HR); mean arterial pressure (MAP); peripheral capillary oxygen saturation (SpO2); initial and final threshold charges; and serum brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), 5-hydroxytryptamine (5-HT), and interleukin-4 (IL-4) levels. Neurological functions, basic executive function scores, and adverse reactions were compared during the treatment process.
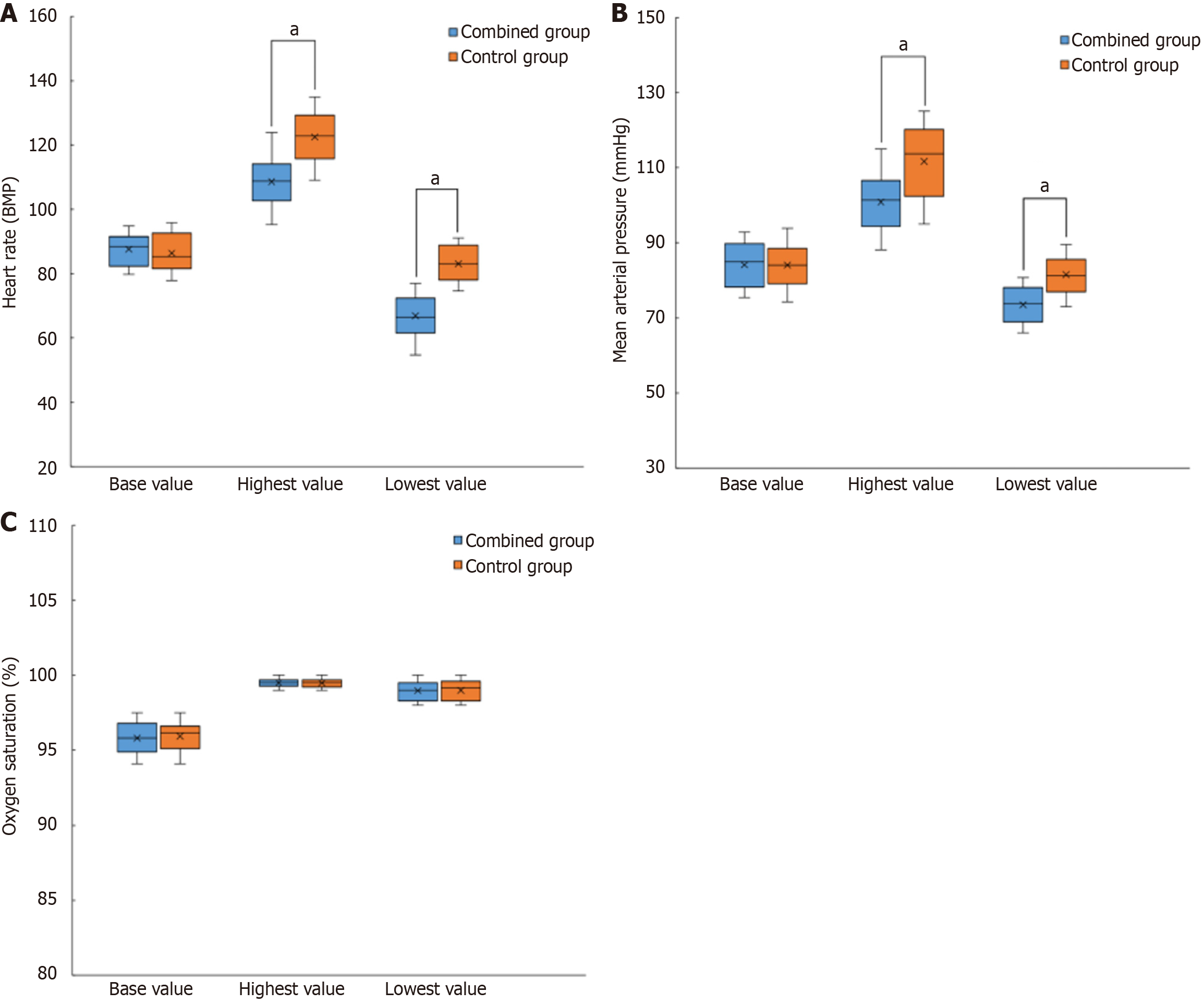
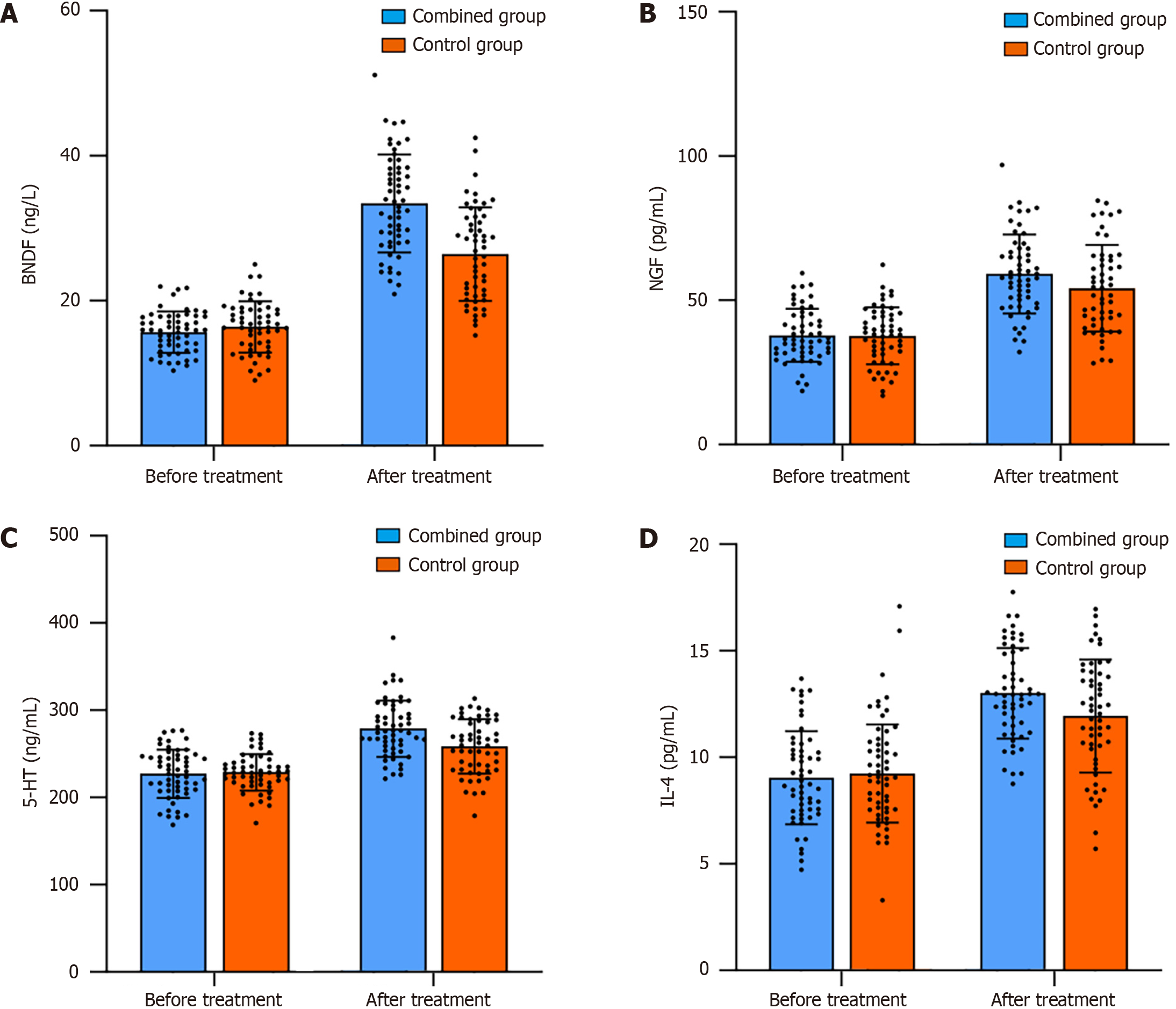
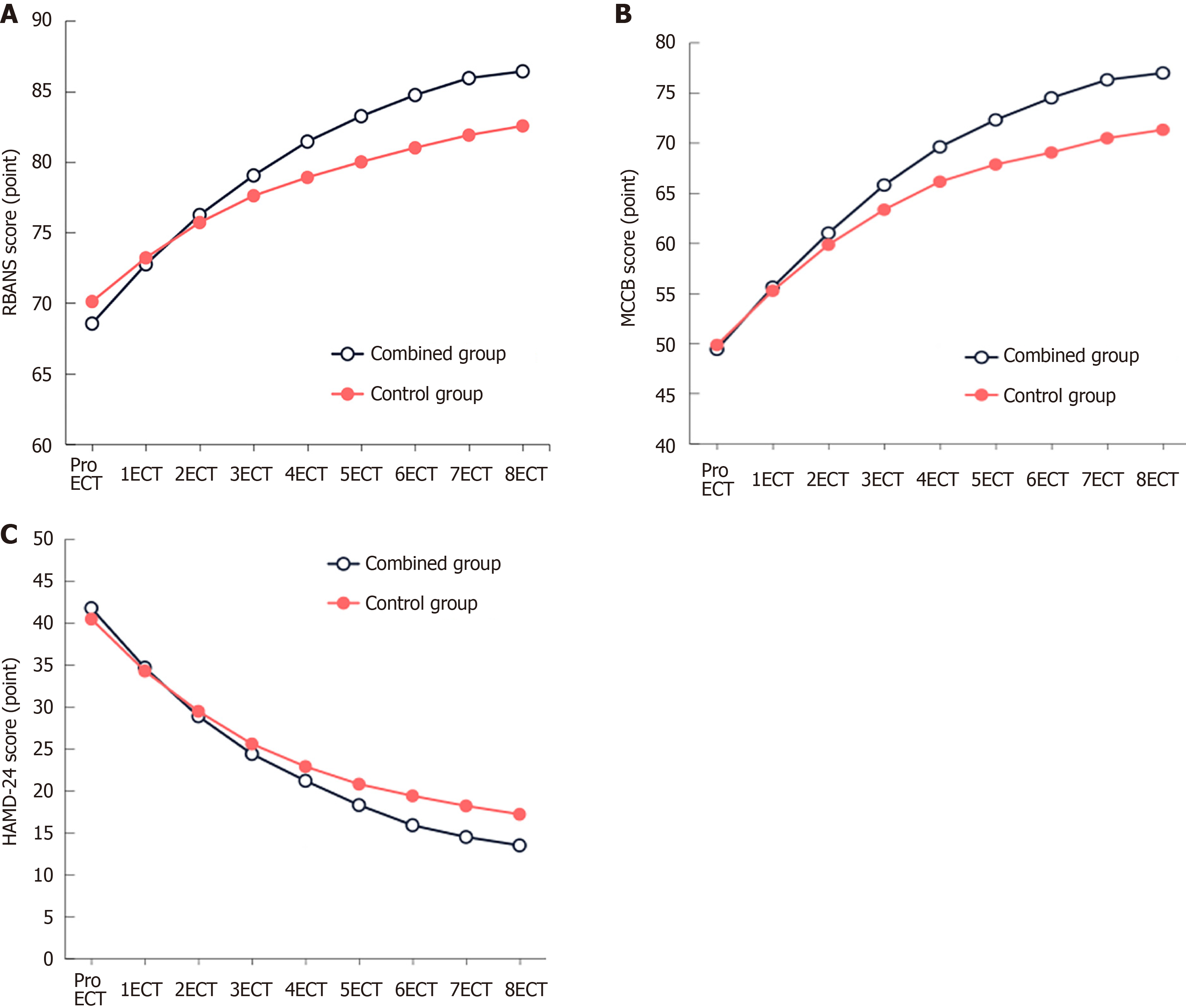
During treatment, the maximum and minimum HR and MAP values in the combination group were markedly lower than those in the control group (P < 0.05), whereas there was no significant difference in SpO2 between the two groups (P > 0.05). During the treatment, there were no significant differences in the initial threshold charge and average duration of seizures during ECT between the two groups (P > 0.05). However, the final threshold charge and total charge in the combination group were considerably lower than those in the control group (P < 0.05). After treatment, the BDNF, NGF, 5-HT, and IL-4 levels were evidently higher in the combination group than in the control group (P < 0.05). During treatment, as the number of ECT sessions increased, both BRNAS and Measurement and Treatment Research to Improve Cognition in Schizophrenia Consensus Cognitive Battery (MCCB) scores increased, whereas Hamilton Depression Rating Scale (HAMD)-24 scores decreased in both groups. Starting from the third treatment session, the BRNAS and MCCB scores in the combination group were higher than in the control group, whereas the Hamilton Depression Scale-24 score was lower in the combination group than in the control group (P < 0.05). After treatment, the Wisconsin Card Sorting Test scores and Tower of Hanoi test results in the combination group were significantly better than those in the control group (P < 0.05). The occurrence of adverse reactions was compared between the two groups (P > 0.05).
Esketamine and etomidate anesthesia during ECT for patients with TRD helps maintain stable vital signs during the treatment process, improves depressive symptoms, and enhances neurological and basic executive functions.
Core Tip: This study aims to evaluate the clinical efficacy of the combined application of esketamine and etomidate in electroconvulsive therapy (ECT) anesthesia for patients with refractory depression. The preliminary study shows that during ECT anesthesia for patients with refractory depression, esketamine-etomidate helps maintain the stability of vital signs during the treatment process, improves depressive symptoms, and enhances neurological and basic executive functions.
- Citation: Zhao GG, Zhao J, Kong Y, Pang YP, Zheng XN, Zhang YW. Effect of esketamine and etomidate anesthesia on neuroplasticity in electroconvulsive therapy for treatment-resistant depression. World J Psychiatry 2025; 15(9): 109458
- URL: https://www.wjgnet.com/2220-3206/full/v15/i9/109458.htm
- DOI: https://dx.doi.org/10.5498/wjp.v15.i9.109458
Treatment-resistant depression (TRD) responds poorly to clinical treatments. Patients with TRD do not respond adequately to standard antidepressant medications, and significant improvements remain elusive even after receiving full doses and sufficient duration of combined antidepressant therapy. This severely impacts the patient's quality of life and social functioning[1,2]. Electroconvulsive therapy (ECT) remains a clinically validated approach for addressing TRD. ECT works by inducing specific brain electrical activities that help regulate neural network functions, thereby improving mood symptoms[3]. Etomidate is a commonly used anesthetic in ECT and induces rapid sedation and fast recovery[4]. However, etomidate suppresses the adrenal cortex and affects cortisol synthesis[5]. This becomes particularly problematic in prolonged ECT courses as it may interfere with neurological recovery in patients, limiting its clinical use. Esketamine, an NMDA antagonist, modulates the activity of the glutamate system and promotes the release of brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF). This mechanism enhances the recovery of cognitive function in patients after ECT treatment for severe depression[6]. Currently, there are few reports on the use of esketamine and etomidate in ECT treatment for TRD. Therefore, this study aimed to assess the clinical efficacy of the combined use of esketamine and etomidate for ECT anesthesia in patients with TRD.
This study used a randomized controlled trial design with equal sample sizes in both groups, and the neurocognitive function measurement scale [Measurement and Treatment Research to Improve Cognition in Schizophrenia Consensus Cognitive Battery (MCCB)] score[7] was the primary outcome measure. The sample size required for the study was calculated using the formula:
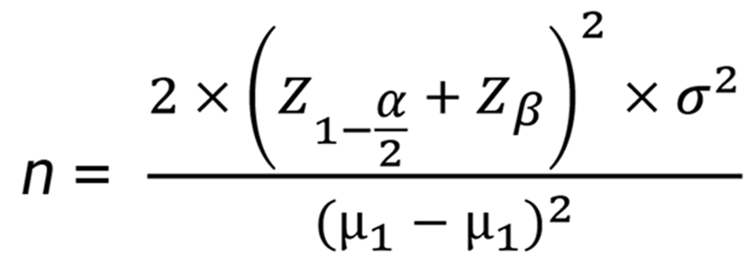
In the formula, n represents the minimum required sample size per group; α is the confidence level, set at two-tailed α = 0.05 in this study, corresponding to a Z value of Z1-α/2 = 1.96; β is the maximum allowable error between the estimated and true values, set at β = 0.1 in this study, corresponding to a Z value of Zβ = 1.28; σ is the standard deviation of the pre-trial efficacy values for the core outcome measure; μ1 and μ2 represent the pre-trial efficacy values for the core indicators in the two groups.
Based on the pre-trial results, the MCCB scores for the esketamine + etomidate and Etomidate anesthesia groups were 27.88 ± 9.21 and 21.47 ± 8.95, respectively. Using these values, the required sample size was calculated to be 43.34, which was 43. Considering the dropout rate, 60 patients were finally included in this study. From April 2020 to April 2024, 120 patients with TRD were selected from the Department of Psychiatry in Second Affiliated Hospital of Shandong First Medical University, China, and randomly divided into a combined and control group using a random number table. Both groups received ECT, with the combined group receiving esketamine + etomidate anesthesia and the control group receiving etomidate anesthesia alone. In the combined group, four patients dropped out owing to poor tolerability of ECT, leaving 56 patients who completed the study. In the control group, six patients dropped out due to poor tolerability of ECT, leaving 54 patients who completed the study. Blinding was maintained for patients and their families, care providers, outcome assessors, and data analysts. This study was approved by the Medical Ethics Committee of our hospital (approval number: 20200122) and informed consent was obtained from all patients and their families.
The diagnostic criteria for TRD were based on the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition[8].
The inclusion criteria were as follows: (1) Age ≥ 19 years; (2) Hamilton Depression Rating Scale (HAMD)-24 score > 35; (3) Exhibits suicidal behavior or tendencies, self-harm tendencies, mutism, self-blame, and has not responded to antidepressant treatment; and (4) Informed consent obtained from the patient's family before treatment.
The exclusion criteria were as follows: (1) Malignant tumors; (2) History of drug abuse or long-term use of sedative-hypnotic drugs; (3) History of cerebrovascular disease; (4) History of cranial trauma; and (5) Bipolar disorder.
All patients were instructed to refrain from drinking for 4 hours and fast for > 12 hours before the procedure. Thirty minutes before the procedure, mice were administered a muscle injection of atropine sulfate (National Drug Approval No. H37020482; Qingdao Jinfeng Pharmaceutical Co. Ltd.; 2 mL, 10 mg). Upon entering the treatment room, an HWM-112W Dynamic Electrocardiogram Monitor (Jizheng Medical Device Approval No. 20212070665, Shijiazhuang Hanwei Medical Equipment Co., Ltd.) and a DMT-101 Electroencephalogram Bispectral Index Monitor (Heilongjiang Medical Device Approval No. 20232070022, Heilongjiang DMT Medical Technology Co., Ltd.) were connected.
In the control group, the etomidate anesthesia protocol was used. After establishing an intravenous (IV) line, the patients were administered 3 L/minute of oxygen via a face mask. Etomidate (National Drug Approval No. H32022379; Jiangsu Hengrui Medicine Co., Ltd.; 10 mL, 20 mg) was slowly injected intravenously at a dose of 0.3 mg/kg.
Combination group underwent the esketamine + etomidate anesthesia protocol. After establishing an IV line, the patients were administered 3 L/minute of oxygen via a face mask. Esketamine (esketamine hydrochloride; National Drug Approval No. H20247040, Jiangsu Hengrui Medicine Co., Ltd., 5 mL: 25 mg) was injected intravenously at a dose of 0.2 mg/kg, followed by etomidate injection at a dose of 0.2 mg/kg.
When the bispectral index value reached 45-50, all patients were administered succinylcholine chloride injections (National Drug Approval No. H20054746; Xi'an Hanfeng Pharmaceutical Co., Ltd.; 2 mL, 100 mg) intravenously at a dose of 1.0 mg/kg. Positive pressure-assisted ventilation was provided using a facemask. After muscle twitching was stopped, laryngeal mask ventilation was used for volume-controlled mechanical ventilation. Eight ECT treatments were performed, with two sessions per week, completing the treatment regimen within 4 weeks.
During treatment, the heart rate (HR), mean arterial pressure (MAP), and oxygen saturation (SpO2) of the patients were monitored using a dynamic electrocardiogram monitor, including the baseline, highest, and lowest values. The basic value refers to the measurement before anesthesia, the highest value refers to the highest value within 5 min after anesthesia to ECT, and the lowest value refers to the lowest value from anesthesia to 5 minutes after ECT.
The Thymatron System IV ECT instrument was used to monitor the initial threshold electric quantity (electric quantity of epileptic seizure for the first time), final threshold electric quantity, total electric quantity of treatment, and average duration of epileptic seizures (total duration of epileptic seizure/number of treatments). Referring to the average of eight treatments for all patients.
At two time points, before and after treatment, 3 mL of peripheral venous blood was collected early in the morning under fasting conditions. The samples were processed using an AmiCORE centrifugal blood component separator (registration No. Guoxie Zhujin 20183100418, Fresenius Kabi Co., Ltd.). Centrifugation was performed at 3000 rpm with a radius of 14 cm at room temperature for 10 minutes. The supernatant was collected and stored until further analysis. Serum levels of BDNF, NGF, 5-hydroxytryptamine (5-HT), and interleukin-4 (IL-4) were measured using an enzyme-linked immunosorbent assay. All reagents were purchased from Shanghai Roche Diagnostics Co., Ltd.
Before enrollment and after each ECT session, patients’ neurological function was assessed.
The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS)[9] was used to evaluate cognitive function. This tool is designed for the rapid screening of cognitive status and includes five cognitive domains: Immediate memory, visuospatial/constructional ability, language, attention, and delayed memory. The total score reflects the overall cognitive functioning, with higher scores indicating better cognitive health.
MCCB[7]: This scale is used to evaluate neurocognitive function. It includes 7 cognitive domains and 10 subtests, with a composite total score of 44.3. Higher scores indicate better overall cognitive abilities.
Hamilton Depression Scale-24 (HAMD-24)[10]: This 24-item assessment tool is extensively used to gauge the level of depressive symptoms. The total score is 72, with higher scores indicating more severe depressive symptoms.
Wisconsin Card Sorting Test: The Wisconsin Card Sorting Test (WCST)[11] assesses executive function, particularly cognitive flexibility, abstract thinking, and problem-solving ability. The test included 4 stimulus cards and 128 response cards. The stimulus cards were placed in front of the participants, who were asked to sort the response cards underneath them based on certain criteria. The sorting rule—color, shape, or number—is determined by the examiner and not disclosed to the participant, who must learn the rule through feedback (“correct” or “incorrect”). Each sorting rule required ten consecutive correct responses. The test ended when the participants correctly completed the six categories or used all 128 cards. The following indicators were recorded: Total number of responses, accuracy rate, percentage of conceptual-level responses, number of perseverative responses, number of non-perseverative errors, and number of categories completed.
The Tower of Hanoi test: The Tower of Hanoi (TOH) test[12] is a commonly used neuropsychological tool for assessing executive function, particularly planning ability, problem solving, and working memory. Participants were permitted to move only a single disk at a time, ensuring that no larger disk was positioned atop a smaller one. Disks must be moved onto pegs or held temporarily in the participant’s hand following specific rules. In this study, the test consisted of 12 tasks: The first six tasks involved three disks, the latter six involved four disks, and the task difficulty increased progressively. The participants were required to transfer the disks from a starting configuration to a goal configuration according to predefined rules. Each task was allowed a maximum of six attempts. If participants successfully completed a task twice consecutively, they progressed to the next task; otherwise, the test was terminated. The total score, average planning time, and average execution time of the patients were tested.
The total occurrence of adverse events, including headache, urinary incontinence, postoperative agitation, memory impairment, bradycardia, nausea and vomiting, and muscle soreness, was assessed and recorded across eight treatment sessions.
All patients underwent a 6month followup after treatment completion. We collected the RBANS, MCCB, HAMD24, WCST, and TOH scores at two time points (immediately post-treatment and 6 months later) and calculated the difference as (6-months score-immediate post-treatment score). Recurrence rates and any potential delayed adverse events were also documented.
SPSS software (version 26.0) was used for the statistical analysis. All measurement data were tested by the Shapiro-Wilk test, which were normally distributed and expressed as mean ± SD, and the t-test was used for comparison between groups. Count data were expressed as n (%), and the χ2 test was used for comparison between groups (Fisher's exact test was used if the expected frequency was < 5). Statistical significance was set at P < 0.05.
There were no statistically significant differences between the two groups in terms of age, body mass index, sex composition, smoking history, alcohol consumption, years of education, or HAMD score at baseline (P > 0.05; Table 1).
| Group | n | Age (years) | BMI (kg/m2) | Gender | Smoke | Alcohol | Course of disease (months) | Years of education (years) | Pretherapy HAMD-24 | |
| Male | Female | |||||||||
| Combined group | 56 | 33.8 ± 8.1 | 24.10 ± 2.14 | 38 (67.86) | 18 (32.14) | 31 (55.36) | 35 (62.50) | 15.81 ± 8.80 | 11.7 ± 3.0 | 41.8 ± 4.3 |
| Control group | 54 | 34.5 ± 8.9 | 23.76 ± 2.34 | 34 (62.96) | 20 (37.04) | 28 (51.85) | 31 (57.41) | 16.32 ± 8.73 | 12.4 ± 3.4 | 40.5 ± 4.1 |
| t/χ2 | -1.043 | 0.792 | 0.291 | 0.136 | 0.297 | -1.449 | -1.140 | 1.615 | ||
| P value | 0.299 | 0.430 | 0.589 | 0.712 | 0.586 | 0.150 | 0.257 | 0.109 | ||
The baseline values of HR and MAP were compared between the two groups (P > 0.05). During treatment, the maximum and minimum HR and MAP values in the combination group were significantly lower than those in the control group (P < 0.05). However, there were no significant differences in the baseline SpO2 values or the maximum and minimum SpO2 values during treatment between the two groups (P > 0.05; Figure 1).
The initial threshold charge for ECT was compared between the two groups (P > 0.05). Additionally, the average seizure duration was compared during ECT treatment (P > 0.05). However, the final threshold charge and total treatment charge in the combination group were significantly lower than those in the control group (P < 0.05; Table 2).
| n | Initial threshold charge (mc) | Final threshold charge (mc) | Total treatment electricity (mc) | Mean seizure duration (mc) | |
| Combined group | 56 | 116.02 ± 13.76 | 144.01 ± 13.73 | 1266.07 ± 212.01 | 50.06 ± 8.08 |
| Control group | 54 | 114.18 ± 14.56 | 170.23 ± 24.76 | 1866.02 ± 260.82 | 48.77 ± 8.82 |
| t value | 2.007 | -52.149 | -99.285 | 2.016 | |
| P value | 0.475 | 0.000 | 0.000 | 0.478 |
Before treatment, the BDNF, NGF, 5-HT, and IL-4 Levels were compared between the two groups (P > 0.05). After treatment, the BDNF, NGF, 5-HT, and IL-4 Levels in the combination group were significantly higher than those in the control group (P < 0.05; Figure 2).
During ECT treatment, both the BRNAS and MCCB scores in the combined and control groups showed an increasing trend, while the HAMD-24 scores showed a decreasing trend as the number of treatments increased. Starting from the third ECT session, the BRNAS and MCCB scores in the combined group were higher than those in the control group, and the HAMD-24 scores were lower in the combined group than in the control group (P < 0.05; Figure 3).
Before treatment, the WCST scores were compared between the combined group and the control group (P > 0.05). After treatment, compared to pre-treatment scores, both groups showed a significant decrease in total responses, perseverative responses, and non-perseverative errors, while accuracy, conceptualization percentage, and number of completed categories increased (P < 0.05). After treatment, the combined group had significantly lower total responses, perseverative responses, and non-perseverative errors, as well as higher accuracy, conceptualization percentage, and number of completed categories compared to the control group (P < 0.05; Table 3).
| n | Total number of answers | Accuracy | Percentage of conceptualization level | Continuous response | Non persistent error | Number of categories completed | |
| Combined group | 56 | ||||||
| Before treatment | 129.73 ± 14.97 | 0.58 ± 0.15 | 0.45 ± 0.22 | 13.42 ± 7.34 | 45.68 ± 14.82 | 2.46 ± 1.82 | |
| After treatment | 108.22 ± 15.11a,b | 0.68 ± 0.14a,b | 0.61 ± 0.18a,b | 7.39 ± 6.78a,b | 31.23 ± 17.07a,b | 3.72 ± 2.14a,b | |
| Control group | 54 | ||||||
| Before treatment | 129.12 ± 15.28 | 0.59 ± 0.14 | 0.45 ± 0.23 | 13.97 ± 7.11 | 45.41 ± 15.35 | 2.55 ± 2.04 | |
| After treatment | 114.59 ± 16.84a | 0.65 ± 0.15a | 0.58 ± 0.20a | 8.72 ± 7.14a | 34.65 ± 18.26a | 3.47 ± 2.41a |
Before treatment, there were no statistically significant differences in the TOH test total score, average planning time, and average execution time between the combined group and the control group (P > 0.05). After treatment, compared to pre-treatment values, both groups showed an increase in total score, and a decrease in average planning time and average execution time (P < 0.05). After treatment, the combined group had a significantly higher total score and lower average planning time and average execution time compared to the control group (P < 0.05; Table 4).
| n | Total score | Average planning time (second) | Average execution time (second) | |
| Combined group | 56 | |||
| Before treatment | 42.87 ± 15.71 | 16.42 ± 9.54 | 29.76 ± 10.58 | |
| After treatment | 64.75 ± 10.27a,b | 8.62 ± 7.41a,b | 22.79 ± 9.52a,b | |
| Control group | 54 | |||
| Before treatment | 43.16 ± 16.98 | 15.94 ± 10.08 | 29.41 ± 10.74 | |
| After treatment | 61.23 ± 17.49a | 9.76 ± 8.28a | 24.35 ± 10.66a |
The incidence of postoperative agitation, confusion, bradycardia, headache, muscle soreness, nausea, and vomiting in the combined group was compared to the control group (P > 0.05; Table 5).
| Group | n | Postoperative dysphoria | Confusion of consciousness | Bradycardia | Headache | Muscle soreness | Bauseated | Emesis |
| Combined group | 55 | 3 (5.45) | 4 (7.27) | 1 (1.82) | 4 (7.27) | 3 (5.45) | 5 (9.09) | 2 (3.64) |
| Control group | 54 | 5 (9.26) | 7 (12.96) | 3 (5.56) | 6 (11.11) | 8 (14.81) | 8 (14.81) | 5 (9.26) |
| χ2 | 0.580 | 0.972 | 1.077 | 0.482 | 2.631 | 0.850 | 1.434 | |
| P value | 0.446 | 0.324 | 0.299 | 0.488 | 0.105 | 0.357 | 0.231 |
In both the combined and control groups, there were no statistically significant differences in the RBANS, MCCB, HAMD-24, WCST, or TOH score immediately post-treatment and 6 months later (P < 0.05). During the 6month follow-up, one recurrence occurred in the combined group and two in the control group, with no significant difference between the two groups (χ² = 0.359, P = 0.618). Neither group experienced any potential delayed adverse effects during the follow-up period, including headache, urinary incontinence, memory impairment, bradycardia, nausea or vomiting, and muscle soreness (Table 6).
| n | BRANS | MCCB | HAMD-24 | WCST | TOH | |
| Combined group | 55 | 1.49 ± 0.49 | 1.16 ± 0.44 | -0.54 ± 0.37 | 0.07 ± 0.02 | 1.28 ± 0.65 |
| Control group | 54 | 1.44 ± 0.52 | 1.12 ± 0.47 | -0.52 ± 0.41 | 0.06 ± 0.02 | 1.25 ± 0.59 |
| t value | 0.182 | 0.106 | 0.124 | 0.085 | 0.127 | |
| P value | 0.874 | 0.926 | 0.908 | 0.956 | 0.905 |
TRD is a mental disorder with high disability and recurrence rates. ECT is a fast and effective intervention for this condition. However, patients often experience varying degrees of neurological dysfunction following ECT. Research has shown that the mechanism of neurological decline after ECT is related to the excessive activation of glutamate receptors. This process can interfere with the plasticity of the hippocampal structure, thereby impairing the normal functioning of neural networks[13]. Etomidate, a commonly used anesthetic for ECT, has limited effects on the glutamate system. In contrast, esketamine inhibits NMDA receptors, which block glutamate-mediated calcium ion influx, thereby preventing excessive activation of signaling pathways, such as calcium/calmodulin-dependent protein kinase II. This helps reduce intracellular calcium overload and free radical generation, thereby alleviating hippocampal damage[14].
The hemodynamic comparison between the two groups during treatment in this study showed that the combination group had lower maximum and minimum HR and MAP values than the control group. This finding suggests that the esketamine + etomidate anesthesia protocol provides better hemodynamic stability during ECT treatment. Etomidate itself has a relatively good cardiovascular stabilizing effect, whereas esketamine, by inhibiting NMDA receptors, reduces excessive glutamate release, alleviates neuronal excitability, and lowers the hyperreactive state of the thalamocortical pathways, which helps suppress the abnormal activation of the sympathetic nervous system. The combined effects of both drugs help maintain HR and blood pressure stability during ECT. Additionally, esketamine promotes the release of norepinephrine from neurons[15], which further supports blood pressure perfusion and autonomic nervous balance. There were no significant differences between the two groups, which may have been due to the effective respiratory support measures provided during ECT, ensuring stable arterial oxygen pressure.
The therapeutic mechanism of ECT relies on inducing widespread synchronized seizure-like discharges in specific brain regions that activate neuroendocrine–immune pathways, thereby triggering antidepressant effects[16]. The stimulation threshold mainly depends on the excitability of the cortical neurons and their ability to synchronize discharges. The results showed that the final threshold charge and total treatment charge in the combined group were both lower than those in the control group, suggesting that esketamine + etomidate anesthesia enhances the sensitivity of neural excitability thresholds, thereby reducing the intensity of stimulation required for effective treatment. This finding is consistent with previous studies[17]. An abnormal increase in excitatory neurotransmitters such as glutamate in the synaptic cleft is one of the key mechanisms underlying the pathophysiology of depression. This leads to overactivation of NMDA receptors, resulting in excessive calcium ion influx into neurons, which damages neuronal membrane structures and mitochondrial function, ultimately triggering excitotoxicity. Esketamine blocks NMDA receptors in postsynaptic neurons, effectively mitigating pathological processes. This helps reduce the excessive depolarization of membrane potentials, thereby stabilizing the neuronal resting state. Under this condition, neuron firing during ECT treatment is more likely to follow a "synchronous yet non-hyperactive" firing pattern, which helps lower the threshold charge required for effective ECT treatment, thereby improving the overall efficacy of the treatment.
The results of this study illustrated that after treatment, BDNF, NGF, 5-HT, and IL-4 Levels were higher in the combination group than in the control group. These findings further highlight that the use of esketamine combined with etomidate anesthesia during ECT for TRD can regulate neurotrophic factors, modulate neurotransmitter metabolism, restore immune balance, and synergistically enhance antidepressant efficacy. Studies have confirmed that esketamine can activate the mTOR-CREB-BDNF signaling pathway, rapidly increasing the synthesis and release of neurotrophic factors and promoting synaptic protein upregulation and dendritic spine sprouting, thus accelerating the reconstruction of neural function[18]. Animal model studies have shown that esketamine significantly upregulates tryptophan hydroxylase 2 (TPH2) mRNA in the dorsal raphe nucleus within 24-72 hours of administration, accompanied by an increase in hippocampal 5-HT levels[19]. This indicates that esketamine enhances central serotonin (5-HT) synthesis by promoting the expression of TPH2 in the hypothalamus, further improving depressive symptoms. Moreover, esketamine can promote the transformation of microglial cells from the M1 to the M2 phenotype, thereby increasing IL-4 secretion, suppressing neuroinflammation, and improving the neuronal microenvironment[20].
In terms of treatment efficacy, this study found that as the number of ECT sessions increased, patients in both anesthesia protocols showed gradual improvements in neurological function and basic cognitive abilities. Notably, from the third ECT session onward, the esketamine + etomidate anesthesia combination demonstrated advantages in terms of the time to clinical antidepressant response and early recovery of neurological function and cognitive levels. On the one hand, esketamine + etomidate anesthesia helps reduce glutamate-mediated excitotoxicity, stabilizes postsynaptic membrane potential, and enhances the synchronization and plasticity of neural network firing. This increases the sensitivity of cortical neurons to electrical stimulation, improves treatment efficiency, and reduces the risk of neuronal damage. In contrast, esketamine + etomidate anesthesia promotes the synergistic regulation of neurotrophic factors, neurotransmitter synthesis, and anti-inflammatory responses, which in turn improves the neuronal microenvironment, strengthens synaptic stability, and offers neuroprotection, facilitating the recovery of neurological function and cognition after treatment. Additionally, the results of this study demonstrated that the combined group exhibited superior cognitive flexibility and executive planning ability after treatment. This suggests that esketamine + etomidate anesthesia may help enhance higher-order cognitive functions in ECT-treated patients with TRD. The underlying mechanism may be that esketamine promotes dendritic spine growth and increases synaptic density, which helps repair the prefrontal cortical structures damaged by depression. This restoration improves the efficiency of information integration between neurons and enhances cooperative efficiency across multiple brain regions, ultimately leading to improved cognitive function in patients.
There were no significant differences in adverse reactions between the two groups, indicating that esketamine + etomidate anesthesia for the ECT treatment of TRD has good clinical safety. In both groups, the 6-month follow-up vs immediate post-treatment RBANS, MCCB, HAMD24, WCST, and TOH score differences as well as the recurrence rates showed no statistically significant differences, indicating that both treatment methods maintained stable effects throughout the observation period. No group experienced any potential delayed adverse reactions during the follow-up period, indicating that neither treatment method poses any risk of late-onset adverse effects. This study has some limitations. First, our findings need validation through large, multicenter, stratified studies, and comparative analyses with other anesthetic adjuncts are needed. Future research should involve multicenter trials with larger sample sizes and evaluate the efficacy of various anesthetic adjuncts in comparison.
In summary, esketamine + etomidate anesthesia for the ECT treatment of patients with TRD helps maintain stable vital signs during the treatment process, improves depressive symptoms, and enhances neurological and basic executive functions in patients.
Esketamine + etomidate anesthesia during ECT for patients with TRD helps maintain stable vital signs during the treatment process, improves depressive symptoms, and enhances neurological and basic executive functions.
| 1. | Steffens DC. Treatment-Resistant Depression in Older Adults. N Engl J Med. 2024;390:630-639. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 41] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 2. | Darby I. Treatment-resistant depression. Aust Dent J. 2023;68:221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 3. | Kverno KS, Mangano E. Treatment-Resistant Depression: Approaches to Treatment. J Psychosoc Nurs Ment Health Serv. 2021;59:7-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 70] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 4. | Ninke T, Groene P. Electroconvulsive therapy: recent advances and anesthetic considerations. Curr Opin Anaesthesiol. 2023;36:441-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 5. | Srivilaithon W, Bumrungphanithaworn A, Daorattanachai K, Limjindaporn C, Amnuaypattanapon K, Imsuwan I, Diskumpon N, Dasanadeba I, Siripakarn Y, Ueamsaranworakul T, Pornpanit C, Pornpachara V. Clinical outcomes after a single induction dose of etomidate versus ketamine for emergency department sepsis intubation: a randomized controlled trial. Sci Rep. 2023;13:6362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 6. | Yang Q, Yao Y, Yuan X, Chen C, Wang Y, Liu H, Zhang K. Effects of subanesthetic repeated esketamine infusions on memory function and NGF in patients with depression: An open-label study. J Affect Disord. 2025;369:1183-1189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 7. | Nuechterlein KH, Green MF, Kern RS. The MATRICS Consensus Cognitive Battery: An Update. Curr Top Behav Neurosci. 2023;63:1-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 8. | Battle DE. Diagnostic and Statistical Manual of Mental Disorders (DSM). Codas. 2013;25:191-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 349] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 9. | Høiland K, Raudeberg R, Egeland J. The repeatable battery for the assessment of neuropsychological status (RBANS) and substance use disorders: a systematic review. Subst Abuse Treat Prev Policy. 2025;20:12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 10. | Rosenberg LI. The Ham-D is not Hamilton's Depression Scale. Psychopharmacol Bull. 2022;52:117-153. [PubMed] |
| 11. | Kopp B, Lange F, Steinke A. The Reliability of the Wisconsin Card Sorting Test in Clinical Practice. Assessment. 2021;28:248-263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 12. | Hardy DJ, Wright MJ. Assessing workload in neuropsychology: An illustration with the Tower of Hanoi test. J Clin Exp Neuropsychol. 2018;40:1022-1029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 13. | Zou L, Min S, Chen Q, Li X, Ren L. Subanesthetic dose of ketamine for the antidepressant effects and the associated cognitive impairments of electroconvulsive therapy in elderly patients-A randomized, double-blind, controlled clinical study. Brain Behav. 2021;11:e01775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 14. | Lisek M, Zylinska L, Boczek T. Ketamine and Calcium Signaling-A Crosstalk for Neuronal Physiology and Pathology. Int J Mol Sci. 2020;21:8410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 15. | Reif A, Bitter I, Buyze J, Cebulla K, Frey R, Fu DJ, Ito T, Kambarov Y, Llorca PM, Oliveira-Maia AJ, Messer T, Mulhern-Haughey S, Rive B, von Holt C, Young AH, Godinov Y; ESCAPE-TRD Investigators. Esketamine Nasal Spray versus Quetiapine for Treatment-Resistant Depression. N Engl J Med. 2023;389:1298-1309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 142] [Article Influence: 47.3] [Reference Citation Analysis (0)] |
| 16. | Espinoza RT, Kellner CH. Electroconvulsive Therapy. N Engl J Med. 2022;386:667-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 234] [Article Influence: 58.5] [Reference Citation Analysis (0)] |
| 17. | Zeng QB, Zou DC, Huang XB, Shang DW, Huang X, Yang XH, Ning YP, Balbuena L, Xiang YT, Zheng W. Efficacy and safety of esketamine versus propofol in electroconvulsive therapy for treatment-resistant depression: A randomized, double-blind, controlled, non-inferiority trial. J Affect Disord. 2025;368:320-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 18. | Qin H, Yu M, Han N, Zhu M, Li X, Zhou J. Antidepressant effects of esketamine via the BDNF/AKT/mTOR pathway in mice with postpartum depression and their offspring. Prog Neuropsychopharmacol Biol Psychiatry. 2024;132:110992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 19. | Chen M, Wang C, Lin Y, Chen Y, Xie W, Huang X, Zhang F, Fu C, Zhuang K, Zou T, Can D, Li H, Wu S, Luo C, Zhang J. Dorsal raphe nucleus-hippocampus serotonergic circuit underlies the depressive and cognitive impairments in 5×FAD male mice. Transl Neurodegener. 2024;13:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 20. | Gao Y, Li L, Zhao F, Cheng Y, Jin M, Xue FS. Esketamine at a Clinical Dose Attenuates Cerebral Ischemia/Reperfusion Injury by Inhibiting AKT Signaling Pathway to Facilitate Microglia M2 Polarization and Autophagy. Drug Des Devel Ther. 2025;19:369-387. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/