Copyright: ©Author(s) 2026.
World J Psychiatry. Apr 19, 2026; 16(4): 115567
Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.115567
Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.115567
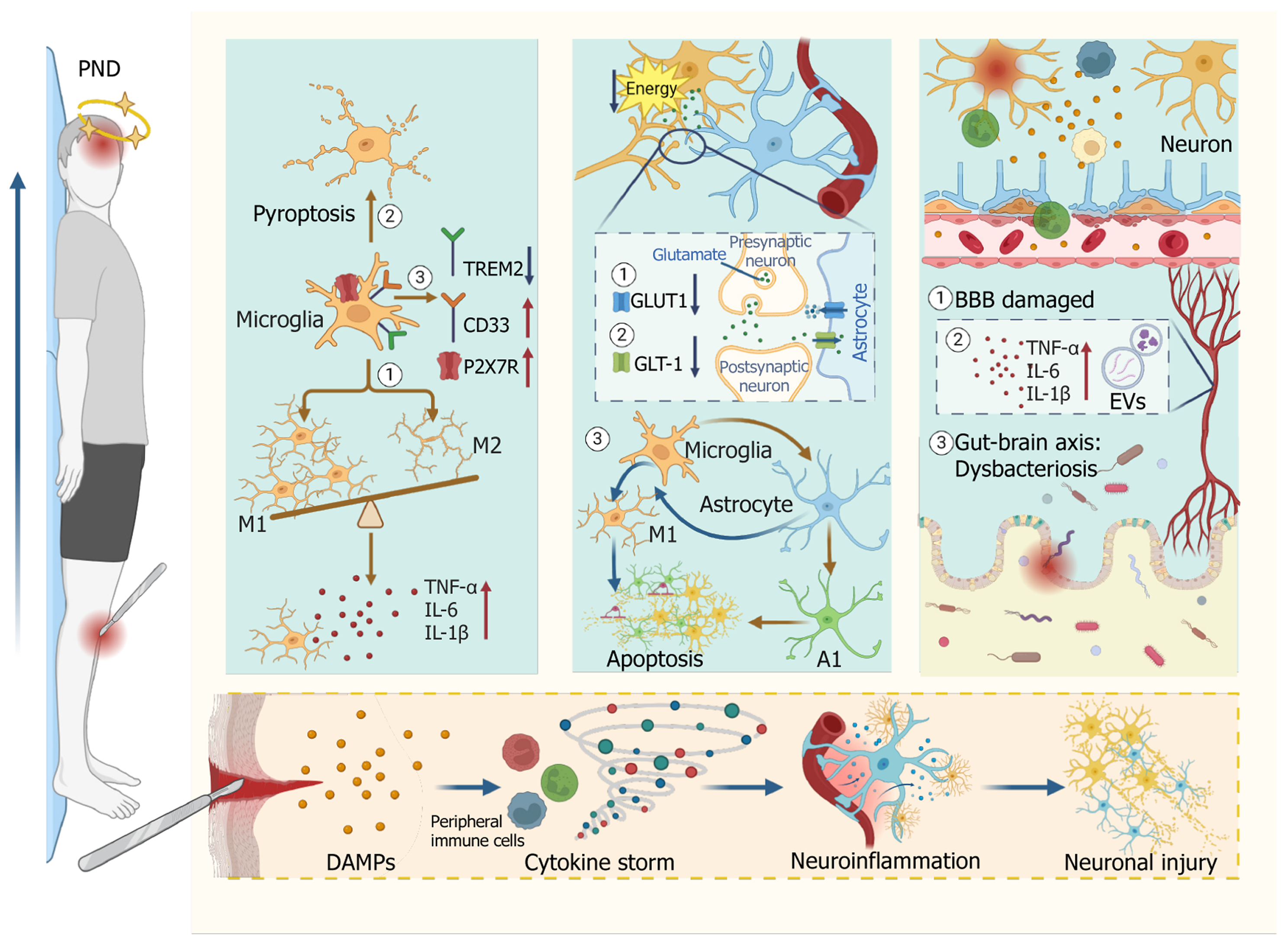
Figure 1 Schematic illustration of the neuroinflammatory mechanisms underlying the pathogenesis of postoperative neurocognitive disorders.
Perioperative factors such as surgery and anesthesia activate the peripheral immune system, triggering the release of damage-associated molecular patterns and a subsequent cytokine surge. These inflammatory signals reach the central nervous system through multiple routes, including disruption of the blood-brain barrier and dysregulation of the gut-brain axis, ultimately resulting in neuronal injury. Within the central nervous system, microglia become activated and polarize toward a pro-inflammatory M1 phenotype, releasing inflammatory mediators and undergoing pyroptosis. Key regulatory molecules such as triggering receptor expressed on myeloid cells 2, CD33, and P2X7 receptor, which modulate microglial activity, display abnormal expression and further amplify neuroinflammation. At the same time, astrocytic dysfunction occurs. This includes reduced expression of the glucose transporter 1 transporter on the cell membrane and abnormal expression of neurotransmitter-regulating transporters such as glutamate transporter 1. These changes disrupt energy metabolism and impair neurotransmitter homeostasis. In addition, harmful interactions between microglia and astrocytes continually intensify the neuroinflammatory cascade. Other mechanisms, including blood-brain barrier impairment, disturbances of the gut-brain axis, extracellular vesicle trafficking, and excessive release of pro-inflammatory cytokines, also contribute to the pathogenesis of postoperative neurocognitive disorders. Together, these processes lead to synaptic dysfunction and neuronal injury, which ultimately result in cognitive decline and the clinical onset of postoperative neurocognitive disorders. PND: Postoperative neurocognitive disorders; M1: Pro-inflammatory microglial phenotype; M2: Anti-inflammatory microglial phenotype; TREM2: Triggering receptor expressed on myeloid cells 2; P2X7R: P2X7 receptor; TNF-α: Tumor necrosis factor-α; IL: Interleukin; GLUT1: Glucose transporter 1; GLT-1: Glutamate transporter 1; A1: Neurotoxic reactive astrocyte phenotype; BBB: Blood-brain barrier; EVs: Extracellular vesicles; DAMPs: Damage-associated molecular patterns.
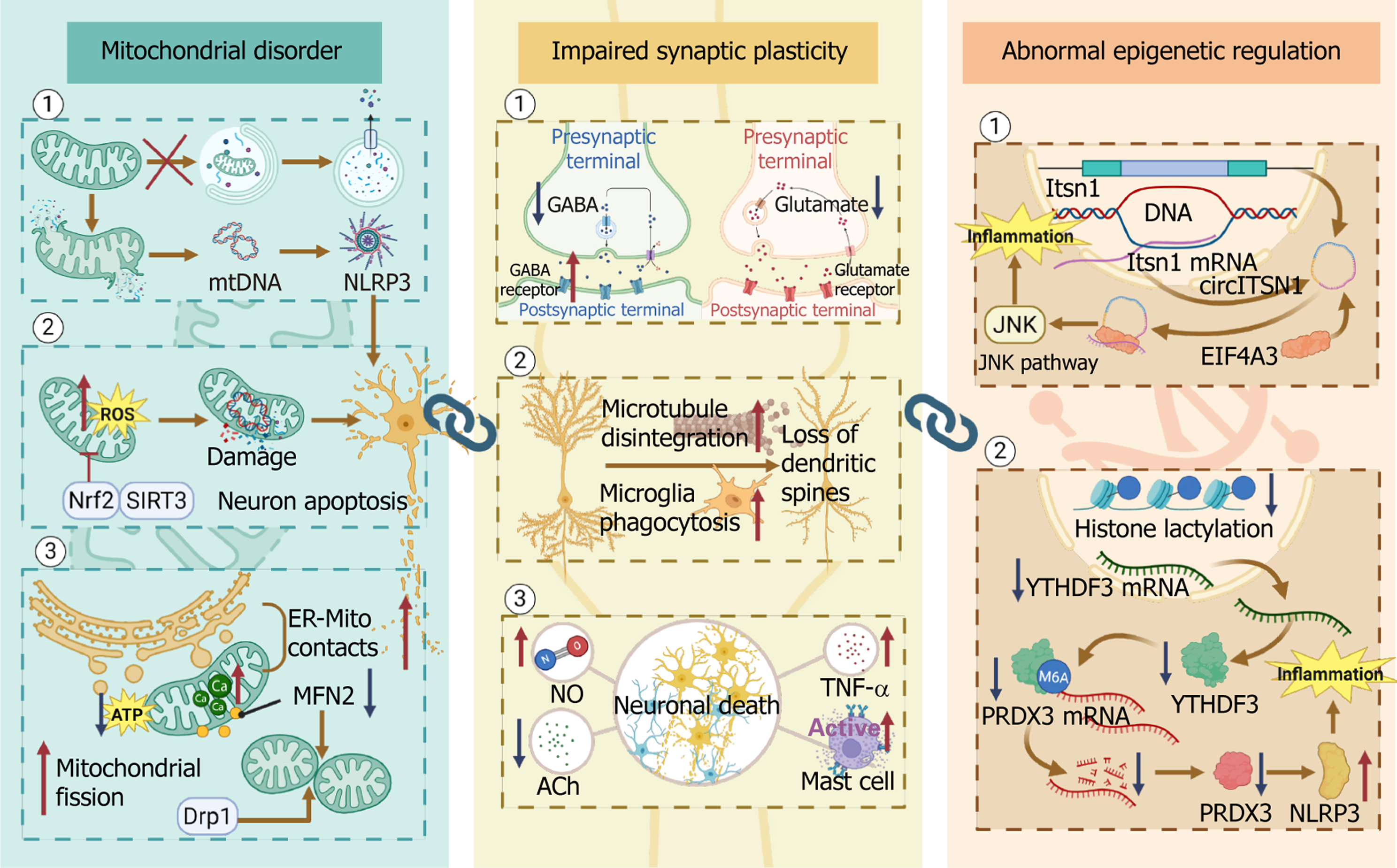
Figure 2 Three core pathological links of postoperative neurocognitive disorders: Mitochondrial dysfunction, impaired synaptic plasticity and abnormal epigenetic regulation.
(1) Mitochondrial dysfunction: Impaired mitophagy leads to the accumulation of dysfunctional mitochondria. Leakage of mitochondrial DNA activates the nucleotide-binding oligomerization domain, leucine-rich repeat and pyrin domain-containing protein 3 inflammasome, while dysregulation of antioxidant pathways, reduced ATP production, disrupted endoplasmic reticulum-mitochondria contacts, and excessive mitochondrial fission collectively promote neuronal apoptosis; (2) Impairment of synaptic plasticity: Imbalances in excitatory and inhibitory neurotransmission, loss of dendritic spines, neuronal death, and related mechanisms ultimately damage synaptic structure and function; and (3) Abnormal epigenetic regulation: The circular RNA circITSN1 modulates inflammatory signaling by sequestering eukaryotic initiation factor 4A-III, whereas histone lactylation regulates nucleotide-binding oligomerization domain, leucine-rich repeat and pyrin domain-containing protein 3 inflammasome activation through the YTH domain family member 3/peroxiredoxin-3 axis. These epigenetic mechanisms contribute to the long-term pathophysiological changes of postoperative neurocognitive disorders. Together, these three interconnected levels form a cascade of damage that extends from subcellular organelles to neural network function, constituting the molecular basis of cognitive impairment in postoperative neurocognitive disorders. MtDNA: Mitochondrial DNA; NLRP3: Nucleotide-binding oligomerization domain, leucine-rich repeat and pyrin domain-containing protein 3; Nrf2: Nuclear factor erythroid 2 related factor 2; SIRT3: Sirtuin 3; ROS: Reactive oxygen species; Mito: Mitochondria; ER: Endoplasmic reticulum; MFN2: Mitofusin 2; Drp1: Dynamin-related protein 1; GABA: γ-aminobutyric acid; NO: Nitric oxide; TNF-α: Tumor necrosis factor-α; Ach: Acetylcholine; JNK: C-Jun N-terminal kinase; YTHDF3: YTH domain family member 3; PRDX3: Peroxiredoxin-3.
- Citation: Jie Y, Lai ZW, Zhou W, Li YC, Zhong BL, Zeng XX, Jiang Q. Neuroimmune synapse and modulation by anesthetics: Inflammatory mechanisms and therapeutic perspectives for postoperative neurocognitive disorders. World J Psychiatry 2026; 16(4): 115567
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/115567.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.115567