Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.115720
Revised: November 19, 2025
Accepted: December 25, 2025
Published online: April 19, 2026
Processing time: 158 Days and 11.2 Hours
Previously proposed mechanisms of action of antidepressants have involved effects on biogenic amines, and glutamic acid systems to explain their clinically beneficial effects. These models of neurotransmission have also attempted to explain the pathophysiology of depression. These approaches, however, have failed to explain the long latency of clinical actions of classic antidepressants or provide a convincing explanation for the pathophysiology of depression. There is a need for a paradigm shift to understand the mechanism of action of antidepressants and pathophysiology of depression. Translational research on cerebral structures that are involved in depression and short-latency antidepressant actions of N-methyl-D-aspartate receptor antagonists have provided some insights on: (1) Neuroinflammatory processes in depression; (2) The participation of neuroglia and neurotrophic factors; (3) Alterations of the functional activity of corticolimbic structures; and (4) Common antiinflammatory actions among both old and new antidepressants. Our hypothetical model consists of the following: When there is a need to cope with adversity, allostatic processes allow natural functional recovery (resilient individuals), whereas allostatic overload results in a neuroinflammatory process that leads to anxiety and depression (vulnerable individuals). Such a model encourages the development of modulators of antiinflammatory processes that involve microglia, astrocytes, and neurotrophic factors.
Core Tip: Antidepressant drug treatments have been classified based on their actions on neurotransmitters. However, these approaches have not reached solid conclusions about their mechanisms of action, their long latency to achieve clinical efficacy, or the pathophysiological processes that are involved in depression. Recent research suggests a continuum among stress, anxiety, and depression, in which the common denominator is neuroinflammation. Notably, N-methyl-D-aspartate receptor antagonists and classic antidepressants share actions on neurotrophic factors and glia-neuronal interactions. Therefore, one promising approach for the treatment of pathological anxiety and depression may lie in the search for antiinflammatory drugs with specific actions on neural tissue.
- Citation: Contreras CM, Gutiérrez-García AG. Depression and antidepressant drugs: Beyond a purely neurotransmitter approach. World J Psychiatry 2026; 16(4): 115720
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/115720.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.115720
Behavioral, molecular, and electrophysiological techniques suggest that clinical characteristics of depression are a consequence of maladaptive responses to stress that lead to functional changes in the neuroplasticity of neuronal circuits that are related to emotion regulation[1]. There are two main systems that mediate emotional responses: The ventral system and the dorsal system. The ventral system includes the amygdala, the insula, the ventral striatum, and ventral regions of the anterior cingulate gyrus and prefrontal cortex (PFC), which are in charge of identifying the emotional significance of a stimulus and the production of an affective state. The dorsal system includes the hippocampus, dorsal regions of the anterior cingulate gyrus, and the PFC, which are in charge of regulating affective states[2]. Therefore, the dysfunction of connections among the PFC and related areas, including the amygdala, ventral striatum, pallidum, medial thalamus, hypothalamus, and periaqueductal gray and other parts of the brainstem may be considered as the basis of several psychiatric disorders[3-5].
Antidepressants share multiple sites of action. At least three kinds of antidepressants-the tricyclic desipramine, the selective serotonin reuptake inhibitor (SSRI) fluoxetine, and the N-methyl-D-aspartate (NMDA) receptor antagonist ketamine-share the property of increasing c Fos expression in deep layers of the ventromedial PFC in rats[6]. The action of ketamine in the CA3 hippocampus-medial PFC (mPFC) circuit depends on prior exposure to a stressful situation. In a control group, CA3 stimulation produced an inhibitory response in the infralimbic region, and ketamine administration after stress abolished this inhibitory response[7]. Fluoxetine and ketamine produce similar actions on lateral septal nucleus-mPFC responsivity[8]. Therefore, the mPFC and its connections are a good place to explore antidepressant actions, and most of their actions cannot be explained purely by effects on neurotransmitter systems. Ketamine exerts its actions through insulin-like growth factor 1 in the mPFC, in addition to, and in parallel with, its other actions on brain-derived neurotrophic factor (BDNF)[9]. Fluoxetine and ketamine directly bind tyrosine kinase receptor 2, leading to the facilitation of BDNF’s actions[10,11].
The discovery that some cerebral areas are susceptible to the phenomenon of intracranial self-stimulation (ICSS) opened new ways to understand essential aspects of behavior[12]. Initially, regions that were identified as “pleasure centers” were those that connected with the medial forebrain bundle, such as the septal nucleus and PFC, in addition to the entorhinal cortex and pyriform cortex, albeit with less sensitivity[13]. The concept of pleasure centers emerged in parallel with the emerging relationship between these centers and antidepressant drug effects[14-16].
However, the concept of pleasure centers is better understood by considering the ICSS phenomenon as an adaptive process that is directed toward survival[17]. Anhedonia, or the inability to feel or find pleasure, is one of the core sym
Although an exact map of regions that are implicated in ICSS is still incomplete, the concept of pleasure or adaptive survival centers in the brain suggests a relationship between anhedonia and stress, anxiety, and depression. The concept of hotspots may be a good approach because the same structures that are defined as being involved in the anxiety process or pleasure-adaptive centers may produce opposite actions that depend on the subregion studied and the previous state of activity of such centers[20,21].
Hedonic behavior includes the construction of reward valuation, decision-making, anticipation, and motivation. The neural basis of anhedonia and depression may correspond to functional alterations of the cerebral reward circuit[22], involving the ventral striatum, PFC regions, their afferent and efferent connections[23], and the PFC connection to the anterior paraventricular thalamus[24]. In major depressive disorder, the dysfunction of striatal structures, mainly the bilateral putamen, is related to anhedonia as demonstrated by reward anticipation tasks[25]. In humans, high distress is related to ventral striatum-medial orbital frontal cortex connectivity during reward anticipation, and anhedonia is related to greater connectivity from the ventral striatum to amygdala regions during reward anticipation[26]. In a longitudinal study, the presence of anhedonia was shown to predict depression, whereas anxiety-depression comorbidity occurred when the level of anhedonia was high[27-29]. Therefore, when the systems that are responsible for stress management and brain reward systems lose their coordination, anhedonia becomes manifest[30].
The lateral habenula is strongly activated in the absence of reward or expectation of punishment[31], and basal forebrain connections to the lateral habenula display activation in the presence of aversive stimuli and inhibition during reward stimulation[32,33], with the participation of the ventral tegmental area[34]. Recent literature considers that dysfunction of the lateral habenula may explain components of the symptomatology of depression[35]. Ketamine blocks NMDA receptors particularly in the lateral habenula[36] but not in the hippocampus[37] in rats that are subjected to chronic unpredictable mild stress, and ketamine reduces c Fos immunoreactivity in the lateral habenula and increases it in the nucleus accumbens shell[38]. Some SSRIs reduce regional cerebral metabolic rates for glucose in the hippocampus and lateral habenula[39], and imipramine increases the phosphorylation of binding proteins in the nucleus accumbens and lateral habenula, supposedly contributing to long-term actions[40].
The PFC in humans corresponds to the mPFC in rodents[41,42], and its connections have been related to depressive-like behavior in rodents[43]. Translational research has yielded similar results as human studies. A reduction of glial fibrillary acidic protein (i.e., a filament protein that is involved in the function of both activated and non-activated ast
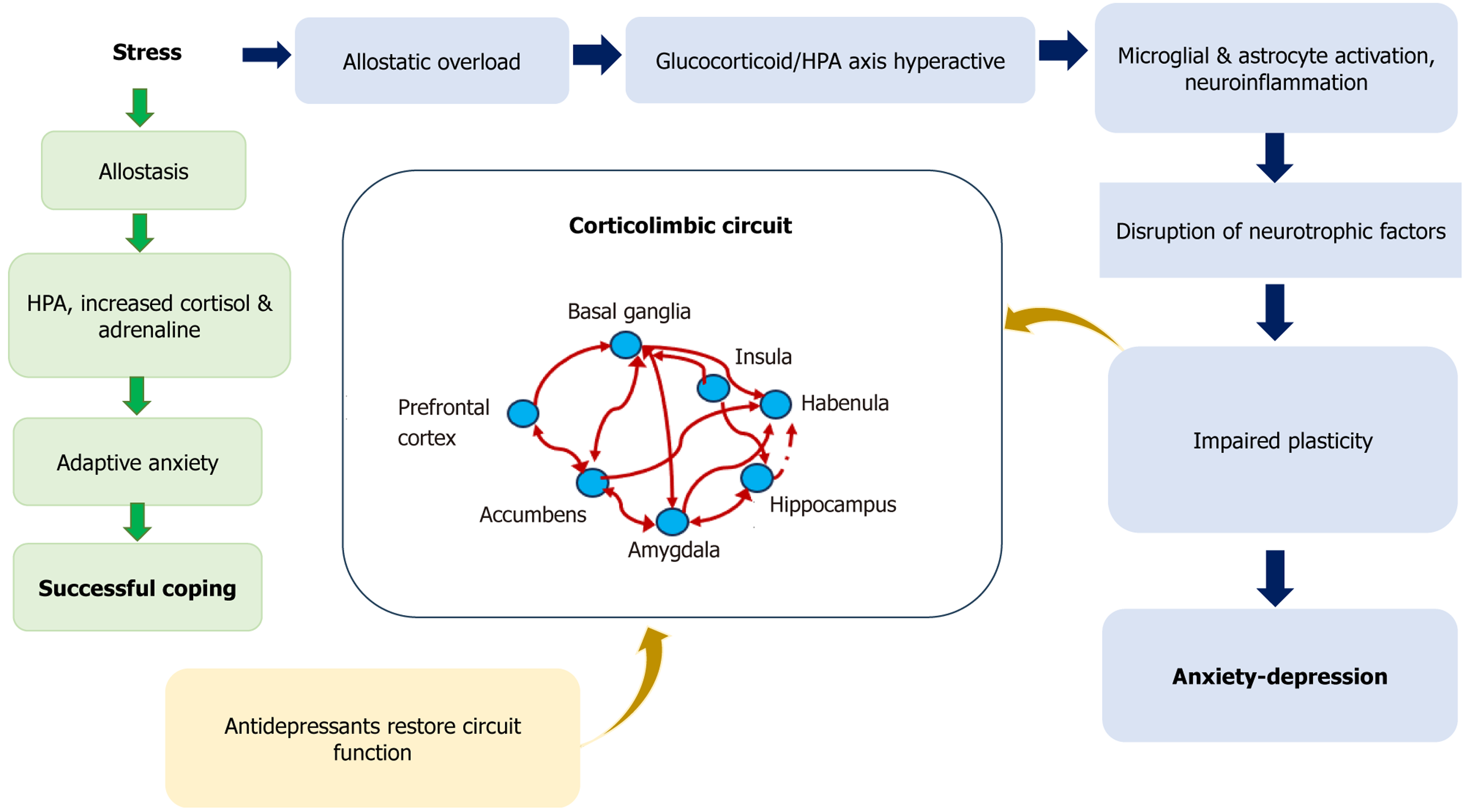
The innate and adaptive immune systems interact with neurotransmitters and neurocircuits to cope with threatening situations. In dysfunctional cases, however, it may constitute a risk for psychiatric diseases[46]. Allostasis is a functional process that maintains homeostasis, but environmental situations in some cases overload the organism’s ability to maintain homeostasis in a process called allostatic load that, in resilient individuals, normally vanishes as soon as the stressful situation resolves. When frequent activation of the allostatic process occurs, when the allostatic process is longer than the stimulus, or when several allostatic processes occur simultaneously, allostatic overload can occur[47], in vulnerable individuals (Figure 1).
The continuous secretion of glucocorticoids and autonomic nervous system hyperactivity that accompanies allostatic overload lead to the delivery of inflammatory cytokines, promoting disease processes[48]. Stress normally activates the hypothalamic-pituitary-adrenal axis, leading to glucocorticoid release. Allostatic load may correspond to anxiety as an adaptive process[49], whereas the pathological process of anxiety may be considered a consequence of allostatic overload. One example of allostatic overload is represented by an inadequate response that leads to the compensatory long-term high secretion of glucocorticoids, which normally exert protective actions when coping with a threatening situation but can result in higher levels of cytokines that are normally counter regulated by glucocorticoids[50]. High allostatic load may predict a higher risk of later anxiety, depression, and suicide[51].
In the long-term when confronting a stressful situation, a vicious cycle may be established through the activation of microglial cells that release proinflammatory factors that stimulate the hypothalamic-pituitary-adrenal axis[52]. The secretion of stress hormones, including glucocorticoids, by the adrenal glands coordinates the body’s response to stress. In the brain, glucocorticoid receptors are expressed by various cell types, including microglia, which are its resident immune cells that regulate stress-induced inflammatory processes. Chronic unpredictable mild stress produces anhedonia- and anxiety-like behavior and increases serum corticosterone and the delivery of proinflammatory factors[53]. The participation of microglia leads to the production of excitotoxic substances, including excess glutamate, and lowers the release of 5-hydroxytryptamine (serotonin) and BDNF[54].
An excess of stress hormones or their dysfunction, mainly corticosteroids, appears to be related to an action that is opposite to their main function, from a protective antiinflammatory role to a dangerous inflammatory process. New-generation antidepressants and ketamine share similar actions on the stress response. Chronic corticosterone administration downregulates glucocorticoid receptors and reduces dendritic branching in the hippocampus, actions that are reversed by fluoxetine[55]. Chronic corticosterone stimulates noradrenergic activity through α2-adrenergic receptors in the mPFC, and fluoxetine, through actions on presynaptic α2-adrenergic receptors, reduces these actions[56]. Long-term corticosterone decreases dendritic spine density in the hippocampal CA1 region, which is also reversed by fluoxetine[57]. Meanwhile, ketamine decreases[58] or is able to antagonize glucocorticoid receptor activity[59], at least in the hipp
There are experimental approaches to uncover the participation of neuroinflammation processes in anxiety. Emotional distress is associated with increases in cortisol and α-amylase in the acute phase, and long-term salivary examination indicates increases in interleukin (IL)-6 and tumor necrosis factor-α (TNF-α) cytokines and C-reactive protein, indicating chronic inflammation[61]. In patients who suffer from psychotic disorders, depressive disorder, or generalized anxiety disorder, there is a high content of plasma soluble urokinase plasminogen activator receptors and IL-6[62].
In a sample of psychiatric patients, abnormally high cerebrospinal fluid IL-6 Levels correlated with high trait anxiety scores[63]. IL-10 is elevated in patients who suffer from generalized anxiety disorder[64], and levels of this cytokine may predict anticipatory anhedonia and consummatory anhedonia[65]. In generalized anxiety disorder, high plasma levels of IL17A and IL23A are associated with the pathophysiology[66].
Under normal conditions, γ-aminobutyric acid-A (GABAA) receptors modulate amygdala activity, but low levels of GABA activity are present during anxiety[67]. The metabotropic glutamate family of G protein-coupled receptors in the mPFC, basolateral amygdala, nucleus accumbens, and ventral hippocampus regulate anhedonia, anxiety, and fear[68]. Additionally, in rodents, exposure to prolonged periods of stress causes dendritic atrophy in the hippocampus and increases arborization in the amygdala[69] and mPFC[70]. Likewise, rodents that spend a long time immobile in the forced swim test express anxiety-like behavior, an increase in peroxidase activity, a decrease in catalase activity in the hippocampus, and an increase in plasma levels of IL-17 and interferon-γ (IFN-γ) inflammatory cytokines[71].
In young rodents that were exposed to severe stress conditions and later tested in adulthood, the abnormal function of fear circuits was detected, and anhedonia-like behavior occurred in parallel to high corticotropin-releasing factor-expressing neurons in the central nucleus of the amygdala[72]. The infiltration of monocytes that express IL-1β that derive from monocytes into the brain produces anxiety, and if this proinflammatory cytokine reaches the nucleus accumbens, depression-like behavior occurs[73]. Additionally, IL-17 receptors in the amygdala participate in the anxiogenic process[74], together with the extended amygdala (i.e., central nucleus of the amygdala and bed nucleus of the stria terminalis) cortical-subcortical circuit[75]. In children and adolescents who suffer from anxiety disorder, imaging studies indicate a larger volume of the amygdala[76].
Stress that is produced by long-term sleep deprivation produces anhedonia- and anxiety-like behavior, in parallel with greater serum corticosterone secretion, an upregulation of clock genes, and an increase in plasma proinflammatory cytokine levels[77]. Acute restraint stress produces anxiety-like behavior and high IL-19 and IL-20Rα expression in mPFC pyramidal neurons[78]. When an animal copes with a stressful situation, mPFC connections to the periaqueductal gray participate in anxiety regulation[79].
New-generation antidepressants that are effective in the treatment of generalized anxiety and depression also reduce anxiety scores, improve clinical aspects, and reduce proinflammatory cytokine levels[80,81]. Ketamine administration in a single dose was shown to also exert antiinflammatory effects[82] and produce anxiolytic actions together with a decrease in depression-like behavior, but these anxiolytic actions of ketamine and fluoxetine did not occur after a cholinergic lesion in the diagonal band of Broca[83].
Human imaging studies have shown a decrease in the functional connectivity of prefrontal-limbic circuits, which may explain some symptoms of depression[84], and increases in plasma C-reactive protein levels coincided with lower functional connectivity in the ventral striatum, the parahippocampal gyrus, the amygdala, orbitofrontal and insular cortices, and posterior cingulate cortex circuits, with a main effect on the PFC[85]. Postmortem analyses of depressed patients revealed an abnormal reduction of gray matter at the expense of a lower presence of glial cells in the subgenual PFC and its connections. Additionally, depressed patients exhibit alterations of the PFC, the amygdala, and related parts of the striatum and thalamus[86].
Positron emission tomography allows the observation of changes in glucose metabolism. In non-medicated depressed patients, this metabolism increased in the left and right lateral orbital and ventrolateral portions of the PFC, left amygdala, and posterior cingulate cortex and decreased in the subgenual anterior cingulate cortex and dorsomedial-anterolateral PFC. The antidepressant drugs treatment decreased metabolism in the left amygdala and left subgenual anterior cingulate cortex[87].
Depression in aged humans is related to a reduction of connectivity between the amygdala and dorsal PFC regions[88]. In younger depressed patients, a reduction of gray matter volume in the PFC, hippocampus, and striatum was also obs
These observations have been replicated in animal models. Prelimbic and infralimbic mPFC regions are connected to various brain regions, and the infralimbic region and its midbrain connections appear to be more related to antidepressant actions than the prelimbic region[96]. Retrograde labeling after chronic restraint stress in rodents revealed a decrease in the distribution density of basal, proximal, and distal dendrites, as well as an increase in the loss of dendritic spines of distal dendrites in infralimbic regions of the mPFC, hippocampus, septum/basal forebrain, hypothalamus, and thalamus and an increase in the distribution density of fluorescence-positive neurons in the amygdala[97].
Among other actions, antidepressants produce their effects based on the balance between excitatory glutamatergic pyramidal neurons and GABA inhibitory interneurons [i.e., the functional excitation: Inhibition (E/I) model][98]. For example, dexamethasone administration in the long term produces depression-like behavior in animal models and a reduction of GABAergic interneurons in the mPFC, leading to an imbalance in the E/I process[99].
An initial action on glutamate activity is necessary for a later action of GABA[100]. Chronic stress-induced imp
The difference in the onset of action of classic antidepressants and NMDA receptor antagonists could hypothetically be explained by considering that classic antidepressants establish initial changes in neurotransmitter systems that involve GABAergic receptors, among others, but a long latency may influence the action on cerebral plasticity. In turn, NMDA receptor antagonists exert a direct blocking action on NMDA receptors that can explain their anesthetic properties but not antidepressant actions. Instead, after NMDA receptor blockade, a secondary effect occurs, involving a cascade of events in which the activation of neurotrophic factors restores the natural functions of microglia and astrocytes that reestablish neuronal function through a plastic process. In this case, clinical effects appear within minutes or hours. The persistence of these antiinflammatory factors may explain long-term clinical effects.
Neuroimaging studies show that neuroinflammation is associated with a decrease in activation of the ventral striatum and ventromedial PFC, leading to anhedonia[104]. Chronic stress produces neuroinflammatory responses through the activation of microglia and astrocytes, and then proinflammatory cytokines are released to aggravate symptomatology[105]. Additionally, proinflammatory cytokines may cross the blood-brain barrier and activate microglia[106].
The chronic unpredictable mild stress model produces depression-like behavior and glial cell activation but also inhibits IL-1β, IFN-γ, and TNF-α expression in the hippocampal dentate gyrus. All of these changes are reversed by fluoxetine, which produces reductions of neuronal apoptosis and downregulates apoptotic protein Bax, cleaved caspase 3, and cleaved caspase 9 levels[107]. Under normal conditions, microglia regulate synaptic plasticity and synaptic function. The dysregulation of cytokine activity and presence of neurotoxic elements may be precursors of depression through disruptions of microglial function[108], thereby affecting neuronal plasticity.
The inhibition of astrocyte activation may be a fundamental action of antidepressants[109]. Fluoxetine increases levels of BDNF as an initial effect[110-112] and reduces glycogen levels and increases glucose utilization and lactate release by astrocytes, without any relationship with serotonergic processes[113]. Fluoxetine is also able to promote autophagosome formation and increase the clearance of injured mitochondria in the hippocampus, consequently protecting astrocytes in rodents that are subjected to chronic mild stress[114] and attenuating astrocytic activity[115].
With regard to fast-acting antidepressant drugs, ketamine increases BDNF immunostaining in the PFC and hipp
In neurodegenerative processes, such as Alzheimer’s disease, targeting astrocyte function has been proposed, based on stem cell therapy and gene editing technology, with a focus on astrocyte transformation[121]. Additional models have been proposed, such as chemogenetic tools that are based on Designer Receptors Exclusively Activated by Designer Drugs in astrocytes and microglia[122]. There is a need to develop novel antidepressant drugs that act on perisynaptic astrocytes[123].
The discovery of areas in the brain that are susceptible to ICSS, dubbed the pleasure zone concept, sparked great enthusiasm for explaining anhedonia, a common symptom in various psychopathological processes, from stress to anxiety and depression and even other psychiatric conditions. However, these centers that are susceptible to ICSS could be better conceived as centers that regulate adaptive processes that are aimed at survival. ICSS centers, sites of antidepressant actions, and nuclei that are related to the pathophysiology of anhedonia have commonalities. Hedonia is necessary for the survival of species because it involves basic behaviors, such as eating and reproductive behavior. Mapping these structures that are related to hedonia and anhedonia has not yielded conclusive results because their neural activity responds differently depending on prior activity and experience (i.e., hotspots). Monoaminergic and catecholaminergic theories have not yielded expected results with regard to satisfactorily explaining either the mech
Notably, classic tricyclic and newer antidepressants and NMDA antagonists share antiinflammatory actions through processes that are beginning to be identified. A promising therapeutic approach consists of finding drugs with specific actions on neuroinflammation processes, particularly with actions on neurotrophic factors, microglia, astrocytes, and proinflammatory cytokines.
The authors thank Michael Arends for proofreading the manuscript.
| 1. | Krishnan V, Nestler EJ. The molecular neurobiology of depression. Nature. 2008;455:894-902. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2380] [Cited by in RCA: 2135] [Article Influence: 118.6] [Reference Citation Analysis (5)] |
| 2. | Phillips ML, Drevets WC, Rauch SL, Lane R. Neurobiology of emotion perception I: The neural basis of normal emotion perception. Biol Psychiatry. 2003;54:504-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1690] [Cited by in RCA: 1491] [Article Influence: 64.8] [Reference Citation Analysis (0)] |
| 3. | Phillips ML, Drevets WC, Rauch SL, Lane R. Neurobiology of emotion perception II: Implications for major psychiatric disorders. Biol Psychiatry. 2003;54:515-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1400] [Cited by in RCA: 1264] [Article Influence: 55.0] [Reference Citation Analysis (4)] |
| 4. | Price JL, Drevets WC. Neurocircuitry of mood disorders. Neuropsychopharmacology. 2010;35:192-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1317] [Cited by in RCA: 1198] [Article Influence: 74.9] [Reference Citation Analysis (4)] |
| 5. | Price JL, Drevets WC. Neural circuits underlying the pathophysiology of mood disorders. Trends Cogn Sci. 2012;16:61-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 706] [Cited by in RCA: 611] [Article Influence: 43.6] [Reference Citation Analysis (3)] |
| 6. | Chang CH, Chen MC, Lu J. Effect of antidepressant drugs on the vmPFC-limbic circuitry. Neuropharmacology. 2015;92:116-124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 7. | Contreras CM, Gutiérrez-García AG. Differential actions of ketamine on CA3-prelimbic and CA3-infralimbic connection responsivity depend on prior exposure to stress. Behav Brain Res. 2025;493:115712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 8. | Contreras CM, Gutiérrez-García AG. Ketamine and fluoxetine exert similar actions on prelimbic and infralimbic responsivity to lateral septal nucleus stimulation in Wistar rats. Neurosci Lett. 2024;834:137848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 9. | Tutuska AM, Nahigian E. Competency based evaluation: one hospital's approach. J Nurs Staff Dev. 1997;13:44-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 33] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 10. | Casarotto PC, Girych M, Fred SM, Kovaleva V, Moliner R, Enkavi G, Biojone C, Cannarozzo C, Sahu MP, Kaurinkoski K, Brunello CA, Steinzeig A, Winkel F, Patil S, Vestring S, Serchov T, Diniz CRAF, Laukkanen L, Cardon I, Antila H, Rog T, Piepponen TP, Bramham CR, Normann C, Lauri SE, Saarma M, Vattulainen I, Castrén E. Antidepressant drugs act by directly binding to TRKB neurotrophin receptors. Cell. 2021;184:1299-1313.e19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 614] [Cited by in RCA: 503] [Article Influence: 100.6] [Reference Citation Analysis (0)] |
| 11. | Diniz CRAF, Crestani AP, Casarotto PC, Biojone C, Cannarozzo C, Winkel F, Prozorov MA, Kot EF, Goncharuk SA, Benette Marques D, Rakauskas Zacharias L, Autio H, Sahu MP, Borges-Assis AB, Leite JP, Mineev KS, Castrén E, Resstel LBM. Fluoxetine and Ketamine Enhance Extinction Memory and Brain Plasticity by Triggering the p75 Neurotrophin Receptor Proteolytic Pathway. Biol Psychiatry. 2025;97:248-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 12. | Olds J, Milner P. Positive reinforcement produced by electrical stimulation of septal area and other regions of rat brain. J Comp Physiol Psychol. 1954;47:419-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2191] [Cited by in RCA: 1412] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 13. | Prado-Alcala R, Streather A, Wise RA. Brain stimulation reward and dopamine terminal fields. II. Septal and cortical projections. Brain Res. 1984;301:209-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 14. | Frank RA, Pommering T, Nitz D. The interactive effects of cocaine and imipramine on self-stimulation train-duration thresholds. Pharmacol Biochem Behav. 1988;30:1-4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 15. | Markou A, Hauger RL, Koob GF. Desmethylimipramine attenuates cocaine withdrawal in rats. Psychopharmacology (Berl). 1992;109:305-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 63] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 16. | Olds ME. Opposite effects of PCA and chlorimipramine on ICSS and on its facilitation by amphetamine. Pharmacol Biochem Behav. 1994;47:803-817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 17. | LeDoux J. Rethinking the emotional brain. Neuron. 2012;73:653-676. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1200] [Cited by in RCA: 853] [Article Influence: 60.9] [Reference Citation Analysis (0)] |
| 18. | Barthel AL, Pinaire MA, Curtiss JE, Baker AW, Brown ML, Hoeppner SS, Bui E, Simon NM, Hofmann SG. Anhedonia is central for the association between quality of life, metacognition, sleep, and affective symptoms in generalized anxiety disorder: A complex network analysis. J Affect Disord. 2020;277:1013-1021. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 19. | Craske MG, Meuret AE, Ritz T, Treanor M, Dour HJ. Treatment for Anhedonia: A Neuroscience Driven Approach. Depress Anxiety. 2016;33:927-938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 326] [Cited by in RCA: 276] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 20. | Berridge KC, Kringelbach ML. Pleasure systems in the brain. Neuron. 2015;86:646-664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1270] [Cited by in RCA: 934] [Article Influence: 84.9] [Reference Citation Analysis (0)] |
| 21. | Kringelbach ML, Berridge KC. Towards a functional neuroanatomy of pleasure and happiness. Trends Cogn Sci. 2009;13:479-487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 374] [Cited by in RCA: 270] [Article Influence: 15.9] [Reference Citation Analysis (4)] |
| 22. | Russo SJ, Nestler EJ. The brain reward circuitry in mood disorders. Nat Rev Neurosci. 2013;14:609-625. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1643] [Cited by in RCA: 1421] [Article Influence: 109.3] [Reference Citation Analysis (0)] |
| 23. | Der-Avakian A, Markou A. The neurobiology of anhedonia and other reward-related deficits. Trends Neurosci. 2012;35:68-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 901] [Cited by in RCA: 798] [Article Influence: 57.0] [Reference Citation Analysis (0)] |
| 24. | Li H, Kawatake-Kuno A, Inaba H, Miyake Y, Itoh Y, Ueki T, Oishi N, Murai T, Suzuki T, Uchida S. Discrete prefrontal neuronal circuits determine repeated stress-induced behavioral phenotypes in male mice. Neuron. 2024;112:786-804.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 25. | Daniels A, Wellan SA, Beck A, Erk S, Wackerhagen C, Romanczuk-Seiferth N, Schwarz K, Schweiger JI, Meyer-Lindenberg A, Heinz A, Walter H. Anhedonia relates to reduced striatal reward anticipation in depression but not in schizophrenia or bipolar disorder: A transdiagnostic study. Cogn Affect Behav Neurosci. 2025;25:501-514. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 26. | Anderson Z, Damme KSF, Carroll AL, Ka-Yi Chat I, Young KS, Craske MG, Bookheimer S, Zinbarg R, Nusslock R. Association between reward-related functional connectivity and tri-level mood and anxiety symptoms. Neuroimage Clin. 2023;37:103335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 27. | Collins AC, Gallagher MR, Calafiore C, Jordan DG, Winer ES. From anxiety to depression: A longitudinal investigation into the role of anhedonia. J Affect Disord. 2025;380:17-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 28. | Liu Q, Ely BA, Schwartz JJ, Alonso CM, Stern ER, Gabbay V. Reward function as an outcome predictor in youth with mood and anxiety symptoms. J Affect Disord. 2021;278:433-442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 29. | Winer ES, Bryant J, Bartoszek G, Rojas E, Nadorff MR, Kilgore J. Mapping the relationship between anxiety, anhedonia, and depression. J Affect Disord. 2017;221:289-296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 89] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 30. | Pizzagalli DA. Depression, stress, and anhedonia: toward a synthesis and integrated model. Annu Rev Clin Psychol. 2014;10:393-423. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 942] [Cited by in RCA: 836] [Article Influence: 69.7] [Reference Citation Analysis (4)] |
| 31. | Proulx CD, Hikosaka O, Malinow R. Reward processing by the lateral habenula in normal and depressive behaviors. Nat Neurosci. 2014;17:1146-1152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 426] [Cited by in RCA: 361] [Article Influence: 30.1] [Reference Citation Analysis (0)] |
| 32. | Cui Y, Huang X, Huang P, Huang L, Feng Z, Xiang X, Chen X, Li A, Ren C, Li H. Reward ameliorates depressive-like behaviors via inhibition of the substantia innominata to the lateral habenula projection. Sci Adv. 2022;8:eabn0193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 33. | Matsumoto M, Hikosaka O. Representation of negative motivational value in the primate lateral habenula. Nat Neurosci. 2009;12:77-84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 463] [Cited by in RCA: 422] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 34. | Lammel S, Lim BK, Ran C, Huang KW, Betley MJ, Tye KM, Deisseroth K, Malenka RC. Input-specific control of reward and aversion in the ventral tegmental area. Nature. 2012;491:212-217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1161] [Cited by in RCA: 1037] [Article Influence: 74.1] [Reference Citation Analysis (0)] |
| 35. | Yang Y, Wang H, Hu J, Hu H. Lateral habenula in the pathophysiology of depression. Curr Opin Neurobiol. 2018;48:90-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 195] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 36. | Ma S, Chen M, Jiang Y, Xiang X, Wang S, Wu Z, Li S, Cui Y, Wang J, Zhu Y, Zhang Y, Ma H, Duan S, Li H, Yang Y, Lingle CJ, Hu H. Sustained antidepressant effect of ketamine through NMDAR trapping in the LHb. Nature. 2023;622:802-809. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 170] [Cited by in RCA: 142] [Article Influence: 47.3] [Reference Citation Analysis (0)] |
| 37. | Chen M, Ma S, Liu H, Dong Y, Tang J, Ni Z, Tan Y, Duan C, Li H, Huang H, Li Y, Cao X, Lingle CJ, Yang Y, Hu H. Brain region-specific action of ketamine as a rapid antidepressant. Science. 2024;385:eado7010. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 58] [Article Influence: 29.0] [Reference Citation Analysis (0)] |
| 38. | Kingir E, Sevinc C, Unal G. Chronic oral ketamine prevents anhedonia and alters neuronal activation in the lateral habenula and nucleus accumbens in rats under chronic unpredictable mild stress. Neuropharmacology. 2023;228:109468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 39. | Freo U, Merico A, Ermani M, Ori C. Cerebral metabolic effects of fluoxetine, fluvoxamine, paroxetine and sertraline in the conscious rat. Neurosci Lett. 2008;436:148-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 40. | Hutchinson AN, Deng JV, Cohen S, West AE. Phosphorylation of MeCP2 at Ser421 contributes to chronic antidepressant action. J Neurosci. 2012;32:14355-14363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 41. | Vertes RP. Differential projections of the infralimbic and prelimbic cortex in the rat. Synapse. 2004;51:32-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1294] [Cited by in RCA: 1180] [Article Influence: 53.6] [Reference Citation Analysis (0)] |
| 42. | Vertes RP. Interactions among the medial prefrontal cortex, hippocampus and midline thalamus in emotional and cognitive processing in the rat. Neuroscience. 2006;142:1-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 651] [Cited by in RCA: 613] [Article Influence: 30.7] [Reference Citation Analysis (0)] |
| 43. | Mayberg HS. Targeted electrode-based modulation of neural circuits for depression. J Clin Invest. 2009;119:717-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 411] [Cited by in RCA: 338] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 44. | Codeluppi SA, Xu M, Bansal Y, Lepack AE, Duric V, Chow M, Muir J, Bagot RC, Licznerski P, Wilber SL, Sanacora G, Sibille E, Duman RS, Pittenger C, Banasr M. Prefrontal cortex astroglia modulate anhedonia-like behavior. Mol Psychiatry. 2023;28:4632-4641. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 27] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 45. | Der-Avakian A, Mazei-Robison MS, Kesby JP, Nestler EJ, Markou A. Enduring deficits in brain reward function after chronic social defeat in rats: susceptibility, resilience, and antidepressant response. Biol Psychiatry. 2014;76:542-549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 122] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 46. | Miller AH, Raison CL. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nat Rev Immunol. 2016;16:22-34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3262] [Cited by in RCA: 2844] [Article Influence: 284.4] [Reference Citation Analysis (4)] |
| 47. | McEwen BS. Stress, adaptation, and disease. Allostasis and allostatic load. Ann N Y Acad Sci. 1998;840:33-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3748] [Cited by in RCA: 2947] [Article Influence: 105.3] [Reference Citation Analysis (3)] |
| 48. | McEwen BS, Wingfield JC. The concept of allostasis in biology and biomedicine. Horm Behav. 2003;43:2-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2543] [Cited by in RCA: 1861] [Article Influence: 80.9] [Reference Citation Analysis (0)] |
| 49. | Gutiérrez-García AG, Contreras CM. Anxiety: An Adaptive Emotion. New Insights into Anxiety Disorders. United Kingdom: IntechOpen, 2013. [DOI] [Full Text] |
| 50. | McEwen BS. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev. 2007;87:873-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3640] [Cited by in RCA: 2949] [Article Influence: 155.2] [Reference Citation Analysis (4)] |
| 51. | Gou Y, Cheng S, Kang M, Zhou R, Liu C, Hui J, Liu Y, Wang B, Shi P, Zhang F. Association of Allostatic Load With Depression, Anxiety, and Suicide: A Prospective Cohort Study. Biol Psychiatry. 2025;97:786-793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 24.0] [Reference Citation Analysis (1)] |
| 52. | Cheiran Pereira G, Piton E, Moreira Dos Santos B, Ramanzini LG, Muniz Camargo LF, Menezes da Silva R, Bochi GV. Microglia and HPA axis in depression: An overview of participation and relationship. World J Biol Psychiatry. 2022;23:165-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 53. | Zhang C, Liu B, Pawluski J, Steinbusch HWM, Kirthana Kunikullaya U, Song C. The effect of chronic stress on behaviors, inflammation and lymphocyte subtypes in male and female rats. Behav Brain Res. 2023;439:114220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 54. | Li Q, Xie Y, Lin J, Li M, Gu Z, Xin T, Zhang Y, Lu Q, Guo Y, Xing Y, Wang W. Microglia Sing the Prelude of Neuroinflammation-Associated Depression. Mol Neurobiol. 2025;62:5311-5332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 55. | Fraga DB, Camargo A, Olescowicz G, Padilha DA, Mina F, Budni J, Brocardo PS, Rodrigues ALS. Ketamine, but not fluoxetine, rapidly rescues corticosterone-induced impairments on glucocorticoid receptor and dendritic branching in the hippocampus of mice. Metab Brain Dis. 2021;36:2223-2233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 56. | Horrillo I, Ortega JE, Diez-Alarcia R, Urigüen L, Meana JJ. Chronic fluoxetine reverses the effects of chronic corticosterone treatment on α(2)-adrenoceptors in the rat frontal cortex but not locus coeruleus. Neuropharmacology. 2019;158:107731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 57. | Wang G, Cheng Y, Gong M, Liang B, Zhang M, Chen Y, Zhang C, Yuan X, Xu J. Systematic correlation between spine plasticity and the anxiety/depression-like phenotype induced by corticosterone in mice. Neuroreport. 2013;24:682-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 58. | Wang Z, Hu X, Wang Z, Chen J, Wang L, Li C, Deng J, Yue K, Wang L, Kong Y, Sun L. Ketamine alleviates PTSD-like effect and improves hippocampal synaptic plasticity via regulation of GSK-3β/GR signaling of rats. J Psychiatr Res. 2024;178:259-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 59. | Menke A. The HPA Axis as Target for Depression. Curr Neuropharmacol. 2024;22:904-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 80] [Article Influence: 40.0] [Reference Citation Analysis (1)] |
| 60. | Melanson B, Leri F. Effect of ketamine on the physiological responses to combined hypoglycemic and psychophysical stress. IBRO Neurosci Rep. 2021;11:81-87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Reference Citation Analysis (0)] |
| 61. | Budala DG, Luchian I, Virvescu DI, Tudorici T, Constantin V, Surlari Z, Butnaru O, Bosinceanu DN, Bida C, Hancianu M. Salivary Biomarkers as a Predictive Factor in Anxiety, Depression, and Stress. Curr Issues Mol Biol. 2025;47:488. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 62. | Mongan D, Raj Susai S, Föcking M, Byrne JF, Zammit S, Cannon M, Cotter DR. Associations between plasma inflammatory markers and psychotic disorder, depressive disorder and generalised anxiety disorder in early adulthood: A nested case-control study. Brain Behav Immun. 2023;111:90-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 63. | Enokida T, Hattori K, Okabe K, Noda T, Ota M, Sato N, Ogawa S, Tatsumi M, Hoshino M, Kunugi H, Nakagome K. Possible association of elevated CSF IL-6 levels with anxiety and frustration in psychiatric disorders. Psychiatry Clin Neurosci. 2024;78:792-799. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 64. | Sarmin N, Roknuzzaman ASM, Sarker R, -Or-Rashid M, Qusar MS, Bachar SC, Kabir ER, Islam MR, Al Mahmud Z. Association of interleukin-2 and interleukin-10 with the pathophysiology and development of generalized anxiety disorder: a case-control study. BMC Psychiatry. 2024;24:462. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 65. | Fu L, Ren J, Lei X, Wang Y, Chen X, Zhang R, Li Q, Teng X, Guo C, Wu Z, Yu L, Wang D, Chen Y, Qin J, Yuan A, Zhang C. Association of anhedonia with brain-derived neurotrophic factor and interleukin-10 in major depressive disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2024;133:111023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 66. | Mamun-Or-Rashid, Roknuzzaman ASM, Sarker R, Nayem J, Bhuiyan MA, Islam MR, Al Mahmud Z. Altered serum interleukin-17A and interleukin-23A levels may be associated with the pathophysiology and development of generalized anxiety disorder. Sci Rep. 2024;14:15097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 67. | Arora I, Mal P, Arora P, Paul A, Kumar M. GABAergic implications in anxiety and related disorders. Biochem Biophys Res Commun. 2024;724:150218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 31] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 68. | Modrak CG, Wilkinson CS, Blount HL, Schwendt M, Knackstedt LA. The role of mGlu receptors in susceptibility to stress-induced anhedonia, fear, and anxiety-like behavior. Int Rev Neurobiol. 2023;168:221-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 69. | Patel D, Anilkumar S, Chattarji S, Buwalda B. Repeated social stress leads to contrasting patterns of structural plasticity in the amygdala and hippocampus. Behav Brain Res. 2018;347:314-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 75] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 70. | Shansky RM, Hamo C, Hof PR, McEwen BS, Morrison JH. Stress-induced dendritic remodeling in the prefrontal cortex is circuit specific. Cereb Cortex. 2009;19:2479-2484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 216] [Cited by in RCA: 201] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 71. | Dos Santos BM, Pereira GC, Piton E, Fialho MFP, Becker G, da Silva Carlotto M, Camargo LFM, Ramanzini LG, Oliveira SM, Trevisan G, Zanchet EM, Pillat MM, Bochi GV. Lower antidepressant response to fluoxetine is associated with anxiety-like behavior, hippocampal oxidative imbalance, and increase on peripheral IL-17 and IFN-γ levels. Behav Brain Res. 2022;425:113815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 72. | Bolton JL, Molet J, Regev L, Chen Y, Rismanchi N, Haddad E, Yang DZ, Obenaus A, Baram TZ. Anhedonia Following Early-Life Adversity Involves Aberrant Interaction of Reward and Anxiety Circuits and Is Reversed by Partial Silencing of Amygdala Corticotropin-Releasing Hormone Gene. Biol Psychiatry. 2018;83:137-147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 212] [Cited by in RCA: 187] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 73. | Kitaoka S. Inflammation in the brain and periphery found in animal models of depression and its behavioral relevance. J Pharmacol Sci. 2022;148:262-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 74. | Lee B, Kwon JT, Jeong Y, Caris H, Oh D, Feng M, Davila Mejia I, Zhang X, Ishikawa T, Watson BR, Moffitt JR, Chung K, Huh JR, Choi GB. Inflammatory and anti-inflammatory cytokines bidirectionally modulate amygdala circuits regulating anxiety. Cell. 2025;188:2190-2202.e15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 44] [Article Influence: 44.0] [Reference Citation Analysis (0)] |
| 75. | Pessoa L. How many brain regions are needed to elucidate the neural bases of fear and anxiety? Neurosci Biobehav Rev. 2023;146:105039. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 76. | Liu J, Wang L, Zhang L, Ding Y, Zhang X, Hu Z, Zhao X. Abnormal amygdala volume moderates parenting and anxiety symptoms in children and adolescents with anxiety disorder. J Psychiatr Res. 2024;175:316-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 77. | Cao D, Zhao Y, Wang Y, Wei D, Yan M, Su S, Pan H, Wang Q. Effects of sleep deprivation on anxiety-depressive-like behavior and neuroinflammation. Brain Res. 2024;1836:148916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 78. | Jiang Y, Xu L, Cao Y, Meng F, Jiang S, Yang M, Zheng Z, Zhang Y, Yang L, Wang M, Sun G, Liu J, Li C, Cui M. Effects of Interleukin-19 overexpression in the medial prefrontal cortex on anxiety-related behaviors, BDNF expression and p38/JNK/ERK pathways. Brain Res Bull. 2024;212:110952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 79. | Qian J, Wu W, Qiu L, Liu X, Luo Y, Chen F, Surento W, Liu Y, Lu G, Qi R. Medial prefrontal cortex-periaqueductal gray circuit overcomes anxiety-like behavior in male mice following adversity. J Affect Disord. 2025;372:149-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 80. | Hou R, Ye G, Liu Y, Chen X, Pan M, Zhu F, Fu J, Fu T, Liu Q, Gao Z, Baldwin DS, Tang Z. Effects of SSRIs on peripheral inflammatory cytokines in patients with Generalized Anxiety Disorder. Brain Behav Immun. 2019;81:105-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 67] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 81. | Alboni S, Poggini S, Garofalo S, Milior G, El Hajj H, Lecours C, Girard I, Gagnon S, Boisjoly-Villeneuve S, Brunello N, Wolfer DP, Limatola C, Tremblay MÈ, Maggi L, Branchi I. Fluoxetine treatment affects the inflammatory response and microglial function according to the quality of the living environment. Brain Behav Immun. 2016;58:261-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 91] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 82. | Jiang Y, Wang Y, Sun X, Lian B, Sun H, Wang G, Du Z, Li Q, Sun L. Short- and long-term antidepressant effects of ketamine in a rat chronic unpredictable stress model. Brain Behav. 2017;7:e00749. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 83. | Chen L, Ke Y, Ma H, Gao L, Zhou Y, Zhu H, Liu H, Zhang F, Zhou W. Fluoxetine and Ketamine Reverse the Depressive but Not Anxiety Behavior Induced by Lesion of Cholinergic Neurons in the Horizontal Limb of the Diagonal Band of Broca in Male Rat. Front Behav Neurosci. 2021;15:602708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 84. | Price RB, Duman R. Neuroplasticity in cognitive and psychological mechanisms of depression: an integrative model. Mol Psychiatry. 2020;25:530-543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 595] [Cited by in RCA: 513] [Article Influence: 85.5] [Reference Citation Analysis (0)] |
| 85. | Yin L, Xu X, Chen G, Mehta ND, Haroon E, Miller AH, Luo Y, Li Z, Felger JC. Inflammation and decreased functional connectivity in a widely-distributed network in depression: Centralized effects in the ventral medial prefrontal cortex. Brain Behav Immun. 2019;80:657-666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 87] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 86. | Drevets WC. Neuroimaging studies of mood disorders. Biol Psychiatry. 2000;48:813-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 999] [Cited by in RCA: 866] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 87. | Drevets WC, Bogers W, Raichle ME. Functional anatomical correlates of antidepressant drug treatment assessed using PET measures of regional glucose metabolism. Eur Neuropsychopharmacol. 2002;12:527-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 491] [Cited by in RCA: 412] [Article Influence: 17.2] [Reference Citation Analysis (3)] |
| 88. | Leaver AM, Yang H, Siddarth P, Vlasova RM, Krause B, St Cyr N, Narr KL, Lavretsky H. Resilience and amygdala function in older healthy and depressed adults. J Affect Disord. 2018;237:27-34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 89. | Fossati P, Radtchenko A, Boyer P. Neuroplasticity: from MRI to depressive symptoms. Eur Neuropsychopharmacol. 2004;14 Suppl 5:S503-S510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 111] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 90. | Jacob Y, Morris LS, Verma G, Rutter SB, Balchandani P, Murrough JW. Altered hippocampus and amygdala subregion connectome hierarchy in major depressive disorder. Transl Psychiatry. 2022;12:209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 91. | Drevets WC. Functional anatomical abnormalities in limbic and prefrontal cortical structures in major depression. Prog Brain Res. 2000;126:413-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 502] [Cited by in RCA: 459] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 92. | Drevets WC, Price JL, Furey ML. Brain structural and functional abnormalities in mood disorders: implications for neurocircuitry models of depression. Brain Struct Funct. 2008;213:93-118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1775] [Cited by in RCA: 1619] [Article Influence: 89.9] [Reference Citation Analysis (0)] |
| 93. | Tripp A, Oh H, Guilloux JP, Martinowich K, Lewis DA, Sibille E. Brain-derived neurotrophic factor signaling and subgenual anterior cingulate cortex dysfunction in major depressive disorder. Am J Psychiatry. 2012;169:1194-1202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 247] [Cited by in RCA: 213] [Article Influence: 15.2] [Reference Citation Analysis (1)] |
| 94. | Dunham JS, Deakin JF, Miyajima F, Payton A, Toro CT. Expression of hippocampal brain-derived neurotrophic factor and its receptors in Stanley consortium brains. J Psychiatr Res. 2009;43:1175-1184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 133] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 95. | Qi XR, Zhao J, Liu J, Fang H, Swaab DF, Zhou JN. Abnormal retinoid and TrkB signaling in the prefrontal cortex in mood disorders. Cereb Cortex. 2015;25:75-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 96. | Gasull-Camós J, Soto-Montenegro ML, Casquero-Veiga M, Desco M, Artigas F, Castañé A. Differential Patterns of Subcortical Activity Evoked by Glial GLT-1 Blockade in Prelimbic and Infralimbic Cortex: Relationship to Antidepressant-Like Effects in Rats. Int J Neuropsychopharmacol. 2017;20:988-993. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 97. | Ge MJ, Chen G, Zhang ZQ, Yu ZH, Shen JX, Pan C, Han F, Xu H, Zhu XL, Lu YP. Chronic restraint stress induces depression-like behaviors and alterations in the afferent projections of medial prefrontal cortex from multiple brain regions in mice. Brain Res Bull. 2024;213:110981. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 98. | Yin YY, Wang YH, Liu WG, Yao JQ, Yuan J, Li ZH, Ran YH, Zhang LM, Li YF. The role of the excitation:inhibition functional balance in the mPFC in the onset of antidepressants. Neuropharmacology. 2021;191:108573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 99. | Hu L, Qiu MJ, Fan WJ, Wang WE, Liu SH, Liu XQ, Liu SW, Shen ZJ, Zheng YF, Liu GC, Jia ZY, Wang XQ, Fang N. Characterization of GABAergic marker expression in prefrontal cortex in dexamethasone induced depression/anxiety model. Front Endocrinol (Lausanne). 2024;15:1433026. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 100. | Fogaça MV, Daher F, Picciotto MR. Effects of ketamine on GABAergic and glutamatergic activity in the mPFC: biphasic recruitment of GABA function in antidepressant-like responses. Neuropsychopharmacology. 2025;50:673-684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 101. | Fong TH, Li T, Ma X, Cai X, Zhou Q. Prefrontal contribution to passive coping behaviour in chronic stress and treatment by fast-acting antidepressant. Neuropsychopharmacology. 2026;51:741-752. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 102. | Jetsonen E, Didio G, Suleymanova I, Teino I, Castrén E, Umemori J. Chronic treatment with fluoxetine regulates mitochondrial features and plasticity-associated transcriptomic pathways in parvalbumin-positive interneurons of prefrontal cortex. Neuropsychopharmacology. 2025;50:1864-1874. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 103. | Guirado R, Perez-Rando M, Sanchez-Matarredona D, Castrén E, Nacher J. Chronic fluoxetine treatment alters the structure, connectivity and plasticity of cortical interneurons. Int J Neuropsychopharmacol. 2014;17:1635-1646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 85] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 104. | Lucido MJ, Bekhbat M, Goldsmith DR, Treadway MT, Haroon E, Felger JC, Miller AH. Aiding and Abetting Anhedonia: Impact of Inflammation on the Brain and Pharmacological Implications. Pharmacol Rev. 2021;73:1084-1117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 105. | Maeng SH, Hong H. Inflammation as the Potential Basis in Depression. Int Neurourol J. 2019;23:S63-S71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 106. | Troubat R, Barone P, Leman S, Desmidt T, Cressant A, Atanasova B, Brizard B, El Hage W, Surget A, Belzung C, Camus V. Neuroinflammation and depression: A review. Eur J Neurosci. 2021;53:151-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 989] [Cited by in RCA: 833] [Article Influence: 166.6] [Reference Citation Analysis (0)] |
| 107. | Zhao Y, Shang P, Wang M, Xie M, Liu J. Neuroprotective Effects of Fluoxetine Against Chronic Stress-Induced Neural Inflammation and Apoptosis: Involvement of the p38 Activity. Front Physiol. 2020;11:351. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 108. | Li B, Yang W, Ge T, Wang Y, Cui R. Stress induced microglial activation contributes to depression. Pharmacol Res. 2022;179:106145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 111] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 109. | Mariani N, Everson J, Pariante CM, Borsini A. Modulation of microglial activation by antidepressants. J Psychopharmacol. 2022;36:131-150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 69] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 110. | Mostert JP, Koch MW, Heerings M, Heersema DJ, De Keyser J. Therapeutic potential of fluoxetine in neurological disorders. CNS Neurosci Ther. 2008;14:153-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 72] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 111. | Talaee N, Azadvar S, Khodadadi S, Abbasi N, Asli-Pashaki ZN, Mirabzadeh Y, Kholghi G, Akhondzadeh S, Vaseghi S. Comparing the effect of fluoxetine, escitalopram, and sertraline, on the level of BDNF and depression in preclinical and clinical studies: a systematic review. Eur J Clin Pharmacol. 2024;80:983-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 112. | Wu Z, Chen H, Li L, Huang Y, Lan Q, Zhu H, Luo S. Effects of sertraline on depressive symptoms, serum brain-derived neurotrophic factor (BDNF), 5-HT, and inflammatory cytokine expression in pediatric depression patients. Exp Clin Psychopharmacol. 2025;33:416-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 113. | Allaman I, Fiumelli H, Magistretti PJ, Martin JL. Fluoxetine regulates the expression of neurotrophic/growth factors and glucose metabolism in astrocytes. Psychopharmacology (Berl). 2011;216:75-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 163] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 114. | Shu X, Sun Y, Sun X, Zhou Y, Bian Y, Shu Z, Ding J, Lu M, Hu G. The effect of fluoxetine on astrocyte autophagy flux and injured mitochondria clearance in a mouse model of depression. Cell Death Dis. 2019;10:577. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 187] [Cited by in RCA: 175] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 115. | La Barbera L, Krashia P, Loffredo G, Cauzzi E, De Paolis ML, Montanari M, Saba L, Spoleti E, Ficchì S, Zaccone C, De Bardi M, Palazzo C, Marino R, Latagliata EC, Puglisi-Allegra S, Borsellino G, Keller F, Lo Iacono L, Viscomi MT, Nobili A, D'Amelio M. Midbrain degeneration triggers astrocyte reactivity and tau pathology in experimental Alzheimer's Disease. Mol Neurodegener. 2025;20:105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 116. | Viana GSB, Vale EMD, Araujo ARA, Coelho NC, Andrade SM, Costa ROD, Aquino PEA, Sousa CNS, Medeiros IS, Vasconcelos SMM, Neves KRT. Rapid and long-lasting antidepressant-like effects of ketamine and their relationship with the expression of brain enzymes, BDNF, and astrocytes. Braz J Med Biol Res. 2020;54:e10107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 117. | Medeiros GC, Gould TD, Prueitt WL, Nanavati J, Grunebaum MF, Farber NB, Singh B, Selvaraj S, Machado-Vieira R, Achtyes ED, Parikh SV, Frye MA, Zarate CA Jr, Goes FS. Blood-based biomarkers of antidepressant response to ketamine and esketamine: A systematic review and meta-analysis. Mol Psychiatry. 2022;27:3658-3669. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 118. | Xu R, Zhu W, Xu X, Yao Y, Liu Q, Yang Y, Huang Y, Ma Z. S-Ketamine Alleviates Anxiety-Induced Chronic Postoperative Pain by Affecting Glucose Metabolism of Striatal Microglia in a Rat Model. J Neuroimmune Pharmacol. 2025;20:90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 119. | Yang X, Wang Y, Jiang L, Huang S, Jiao Y, Wu Q, Zhang Y, Huang Y, Gu X, Xu R, Zhang W, Ma Z. S-ketamine relieves neuropathic pain by inhibiting microglia phagocytosis of the perineuronal nets. Sci Rep. 2025;15:33596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 120. | Zhong H, Xue R, Han Y, Liu L, Zhao J, Cai M, Wang S, Wei P, Zhao G, Dong H. S-ketamine exposure in early postnatal period induces social deficit mediated by excessive microglial synaptic pruning. Mol Psychiatry. 2025;30:3615-3631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 121. | Bi W, Lei T, Cai S, Zhang X, Yang Y, Xiao Z, Wang L, Du H. Potential of astrocytes in targeting therapy for Alzheimer's disease. Int Immunopharmacol. 2022;113:109368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 122. | Bossuyt J, Van Den Herrewegen Y, Nestor L, Buckinx A, De Bundel D, Smolders I. Chemogenetic modulation of astrocytes and microglia: State-of-the-art and implications in neuroscience. Glia. 2023;71:2071-2095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 123. | Frizzo ME, Ohno Y. Perisynaptic astrocytes as a potential target for novel antidepressant drugs. J Pharmacol Sci. 2021;145:60-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |