Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.117125
Revised: December 22, 2025
Accepted: February 3, 2026
Published online: March 20, 2026
Processing time: 106 Days and 17.1 Hours
Periodontitis is a chronic inflammatory disease triggered by a polymicrobial challenge to the periodontium. While scaling and root planing (SRP) is the gold standard for treatment, its efficacy is often limited by incomplete pathogen eradication and rising antimicrobial resistance. Antimicrobial photodynamic therapy (aPDT) has emerged as a potent adjunctive treatment, utilizing light-activated photosensitizers to induce targeted bacterial lysis. Specifically, the delivery of indocyanine green (ICG) via chitosan nanoparticles (CNPs) may enhance stability and penetration, offering a synergistic approach to improving clinical outcomes when combined with SRP.
To evaluate the overall improvements in various clinical and microbiological periodontal parameters in periodontitis patients undergoing aPDT with CNPs-ICG.
Thirty periodontitis patients having interdental clinical attachment loss (CAL) ‘detectable’ at ≥ 2 non-adjacent teeth, buccal or oral CAL ≥ 3 mm with pocketing > 3 mm detectable at ≥ 2 teeth were randomly assigned to aPDT with CNPs-ICG + SRP (test) group and conventional SRP + laser irradiation only (control) group in a split-mouth design. At baseline, 1-month and 3-months plaque index (PI), modified sulcular bleeding index, pocket probing depth (PPD) and relative clinical attachment level (RCAL) and microbiological tests were recorded.
The test group showed more improvement in PPD (7 mm to 3.26 mm in 1-month and 3.13 mm in 3-months) and RCAL (12 mm to 8.26 mm in 1 month and 8.4 mm in 3 months) than control group that showed PPD reduction from 6.7 mm (at baseline) to 3.73 mm at 1-month and 3.76 mm at 3 months and RCAL reduction from 12.23 mm (at baseline) to 8.7 mm at 1-month and 8.9 mm at 3-months with a statistically significant difference (P = 0.001). The test sites showed a statistically significant (P = 0.001) increase in negative microbiological test results (from 10% at baseline to 80% at 1 month and 3 months follow-up) compared to control sites (6.7% at both baseline and 1-month and 3-months follow-up).
aPDT using CNPs-ICG + SRP can be considered as a better procedure in improving periodontal clinical parameters like PPD and RCAL and in reducing periodontal pathogens than conventional SRP alone.
Core Tip: This trial assesses the adjunctive use of indocyanine green-doped chitosan nanoparticles (CNPs-ICG) in antimicrobial photodynamic therapy (aPDT) with scaling and root planing (SRP) for periodontitis management. Conducted at SCB Dental College and Hospital, Cuttack, on 30 patients, the trial demonstrated significantly superior clinical and microbiological outcomes-particularly in improvement in probing depth, gain in clinical attachment level, and bacterial elimination-in comparison to conventional SRP alone. CNPs-ICG enhanced biofilm targeting and treatment precision through improved photosensitizer delivery. The findings highlight aPDT with CNPs-ICG as an effective, non-surgical, and minimally invasive alternative for patients unsuitable for periodontal surgery.
- Citation: Raj SC, Sen S, Baral D, Mohanty D, Tabassum S, Sil S, Sahu M. Indocyanine green doped with chitosan nanoparticles based antimicrobial photodynamic therapy in periodontitis patients: A randomized clinical trial. World J Exp Med 2026; 16(1): 117125
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/117125.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.117125
Periodontitis is a prevalent disease affecting oral health. It typically begins as gingivitis, a condition that can be reversed with adequate oral hygiene measures; but when ignored, it may advance to a severe and permanent form of periodontitis[1]. Chronic periodontitis generally develops as a result of persistent exposure to a diverse community of microbial pathogens[2] this process results in an immune reaction in the host, leading to a sustained state of inflammation[3]. Numerous bacterial species associated with periodontal disease are present in dental plaque; however, certain species show increased prevalence in chronic periodontitis and are critically involved in its onset, progression and resolution[4]. Earlier studies have demonstrated a marked rise in the prevalence of Porphyromonas gingivalis among individuals with chronic form of periodontitis, particularly at sites with active disease characterized by bleeding on probing and ongoing loss of clinical attachment[5].
Mechanical disruption of periodontal biofilms through SRP continues to be the foundational approach in periodontal therapy. Although antibiotics are sometimes employed as adjuncts, their use is limited by issues of antimicrobial re
A highly promising strategy to address these limitations is aPDT. This technique utilizes a photosensitizer (PS) absorbed by microorganisms, which, upon exposure to visible light of a particular wavelength, generates reactive oxygen species such as the cytotoxic singlet oxygen[8]. The primary benefits of aPDT compared to traditional antimicrobial treatments are its rapid therapeutic effect, ability to destroy resistant microbes and their virulence factors, localized PS administration, and dual selectivity, as its cytotoxic action occurs exclusively at locations where both PS and irradiation are simultaneously applied[9]. The adjunctive use of aPDT in treating periodontitis has been proposed as an alternative approach to chemical antimicrobials for targeting and eliminating subgingival microorganisms[10]. A key advantage of aPDT is its dual selectivity. The PS preferentially localizes within diseased tissues, and because the activating light is applied in a precisely targeted manner, the likelihood of unintended damage to surrounding healthy tissues is greatly minimized[11].
A variety of dyes have been employed for the destruction of periodontal pathogens. Among them, the most widely utilized and preferred in photodynamic therapy (PDT) are nonporphyrin phenothiazine derivatives such as toluidine blue O (TBO), methylene blue (MB), and curcumin, applied in different concentration levels[12]. MB and TBO have demonstrated efficacy as photosensitizers against target microorganisms in the past. However, drawbacks such as unintended staining, the requirement for dedicated laser equipment, and residual therapeutic activity even without laser activation highlight the necessity for developing novel photosensitizers that can overcome these limitations[13].
Indocyanine green (ICG), a dye approved by the United States Food and Drug Administration, was first utilized for assessing liver function and subsequently found applications in the field of cardiology[14]. During the 1980s, the application of ICG became well established as a standard technique in ophthalmic fluorescent angiography, although its use in dentistry remains comparatively recent. In vitro investigations have also shown encouraging outcomes, where commercially available 810 nm dental diode lasers employing ICG effectively targeted and eliminated Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans[15].
However, limitations of photosensitizers-such as low water solubility, inadequate control over drug release, limited target specificity, and a low extinction coefficient-have restricted the broader clinical adoption of PDT. Nanotechnology offers potential solutions by enhancing various aspects of PDT, particularly through the development of nanostructured photosensitizers or the use of nanoscale carriers for photosensitizer delivery to address these challenges[16]. The improved efficacy of aPDT with the incorporation of nanoparticles can be attributed to either enhanced delivery of the photosensitizer to the target microorganisms or an amplification of the photosensitizer’s activity.
Chitosan, a naturally occurring polysaccharide, serves as an effective biological vehicle for drug delivery in medical and pharmaceutical applications because of its inherent biocompatibility, bioactivity, biodegradability, and nominal cytotoxicity[17]. According to prior research, chitosan nanoparticles exhibit optimal characteristics for serving as delivery vehicles for photosensitizers in aPDT[18].
In this study, indocyanine green-doped chitosan nanoparticles (CNPs-ICG) were utilized as the photosensitizing agent. The limitations associated with previously used dyes, combined with the superior efficacy and safety profile of CNPs-ICG demonstrated in successful in vitro experiments against periopathogenic bacteria[19,20], provided the rationale for conducting this research.
This single-centered, hospital-based, split-mouth randomized clinical trial with a placebo control was conducted between November 2022 and February 2024 in the Department of Periodontics and Oral Implantology at Sriram Chandra Bhanja Dental College and Hospital, Cuttack, Odisha.
The trial was carried out in accordance with the Helsinki Guidelines, 1964 as revised in 2013 and recommended by the Institutional Ethics Committee (IEC), SCB Dental College and Hospital, Cuttack, Odisha under IEC/SCBDCH/170/2022 dated August 22, 2022. The trial was registered prospectively in the Clinical Trial Registry (ctri.nic.in) with registration number CTRI/2023/02/050002 (Registered on: February 22, 2023). All participants signed an informed consent form before getting included in the study.
The study participants were drawn from individuals attending the outpatient department of the Post Graduate Department of Periodontics, SCBDCH, Cuttack, Odisha. Selection was carried out according to established criteria for inclusion and exclusion, without considering gender or religion.
Systemically healthy patient. Age > 30 years. Interdental clinical attachment loss (CAL) is ‘detectable’ at ≥ 2 non-adjacent teeth[21]. Buccal or oral CAL ≥ 3 mm with pocketing > 3 mm is detectable at ≥ 2 teeth. Presence of bleeding on probing.
With known systemic conditions such as diabetes mellitus, osteoporosis and arthritis. Individuals receiving systemic pharmacological treatments such as corticosteroids, calcium channel blockers, chronic non-steroidal anti-inflammatory drugs, bisphosphonates, or calcium supplements. Habit of using tobacco in any form and alcohol. History of periodontal treatment in the last 6 months. Teeth with grade-III mobility and/or furcation involvement. Immunodeficient individuals. Pregnant or nursing females. Use of antimicrobials within the preceding 6 months that would interfere with bone healing. Tooth with hopeless prognosis. Tooth exhibiting loss of vitality, with or without associated apical pathology. History of radiotherapy therapy in the preceding 1 year in head and neck region.
Preparation of CNPs-ICG as photosensitizer[22]: CNPs-ICG were synthesized in the Molecular Laboratory of Binfosol Private Limited, Belghoria, Kolkata. To prepare the nanoparticles, 50 mg of chitosan powder (75% deacetylated, low molecular weight 1-3 kDa; sourced from shrimp shells, LOBA CHEMIE PVT. LTD.) was held in solution in 50 mL of deionized distilled water (Figure 1). After maintaining the solution at ambient temperature for 1-hour, magnetic stirring was initiated, and acetic acid (1%) was gradually added over a 30-minute period to obtain a uniform chitosan mixture. Subsequently, sodium tripolyphosphate (TPP) (0.5% w/v, 2 mL) was incorporated dropwise for crosslinking purpose, followed by additional stirring for 30 minutes to facilitate nanoparticle formation. Based on the study by Xu and Du[23], TPP acts to reinforce the matrix structure of chitosan nanoparticles (CNPs). The mechanical properties of the CNPs matrix are enhanced through the increased formation of crosslinks between TPP and the chitosan nanoparticles, resulting in nanoparticles with improved strength and hardness.
ICG 2.0 mg/mL (Tokyo Chemical Industry Co. Ltd.) (Figure 1) was then added to the prepared chitosan solution and mixed with a magnetic mixer for 10 minutes at a speed of 1000 rpm in at a pH 4.8 (Figure 1). After that, this solution was centrifuged at 10000 rpm for 30 minutes and the collected sediment CNPs-ICG was lyophilized for further use in the study (Figure 1). The powder 100 mg CNPs-ICG was then dissolved in 1000 mL of sterile distilled water (Figure 1) and gently shaken for 1 minute (Figure 1), resulting in a final concentration of 1000 μg/mL CNPs-ICG solution (Figure 1).
Characterization of CNPs-ICG[22]: Fourier transform infrared (FTIR) (PerkinElmer Spectrum Two) spectroscopy was utilised to analyse the morphological and chemical composition of CNPS-ICG.
The average particle size was measured using an Anton Paar Nano ZS, India system at 25 °C through dynamic light scattering (DLS).
The crystalline structure and phase purity of the synthesized CNPs-ICG were investigated using X-ray diffraction (XRD) (Rigaku Ultima IV) with Cu-Kα radiation (λ = 1.5406 Å). The samples were scanned in the 2θ range of 50 to 800 at a scanning rate of 20/minute.
Preparation of light source[24]: The light source utilized was the PIOON, Diode Laser Therapy System (Dongguan Yuhan Electronic Technology Co. Ltd., China), operating in pulsed wave mode at a wavelength of 810 nm, with a frequency of pulse repetition of 2 kHz, a power density of 1 W, and an average output power of 300 mW, delivering an applied radiation energy of 1414.7 J/cm² per treatment location[24]. Protective eye wears were worn by the patient, operator, and assistant during the procedure to ensure safety.
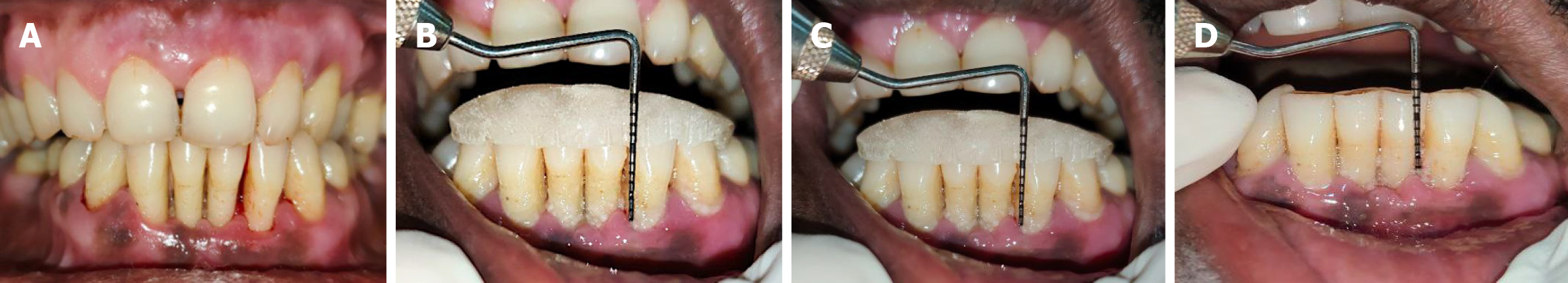
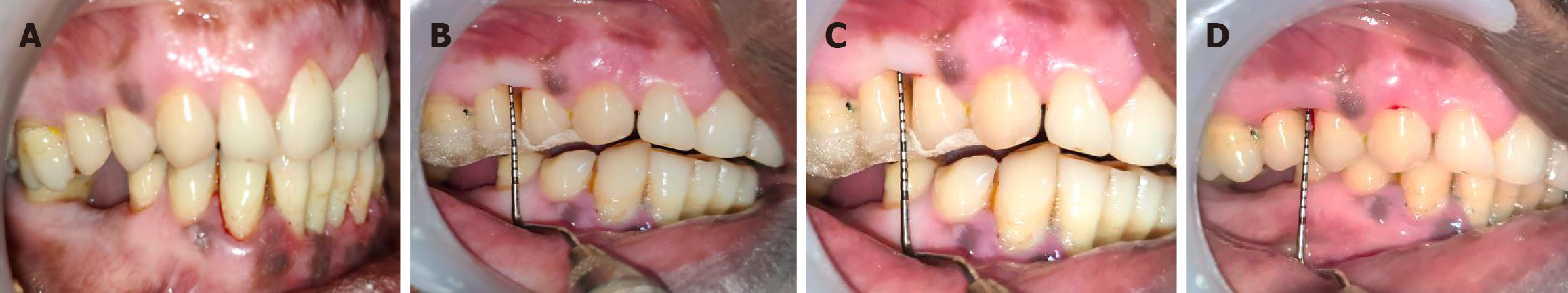
PI was recorded at baseline, and at 1-month and 3-month follow-up visits[25]. Modified sulcus bleeding index (mSBI) was evaluated at baseline, and at 1-month and 3-month follow-up visits[26]. Pocket probing depth (PPD) was measured using a UNC-15 probe as the distance from the gingival margin to the base of the gingival sulcus. Relative clinical attachment level (RCAL) was recorded using a UNC-15 probe at a distance from the apical end of the reference groove created on the acrylic stent to the base of the pocket.
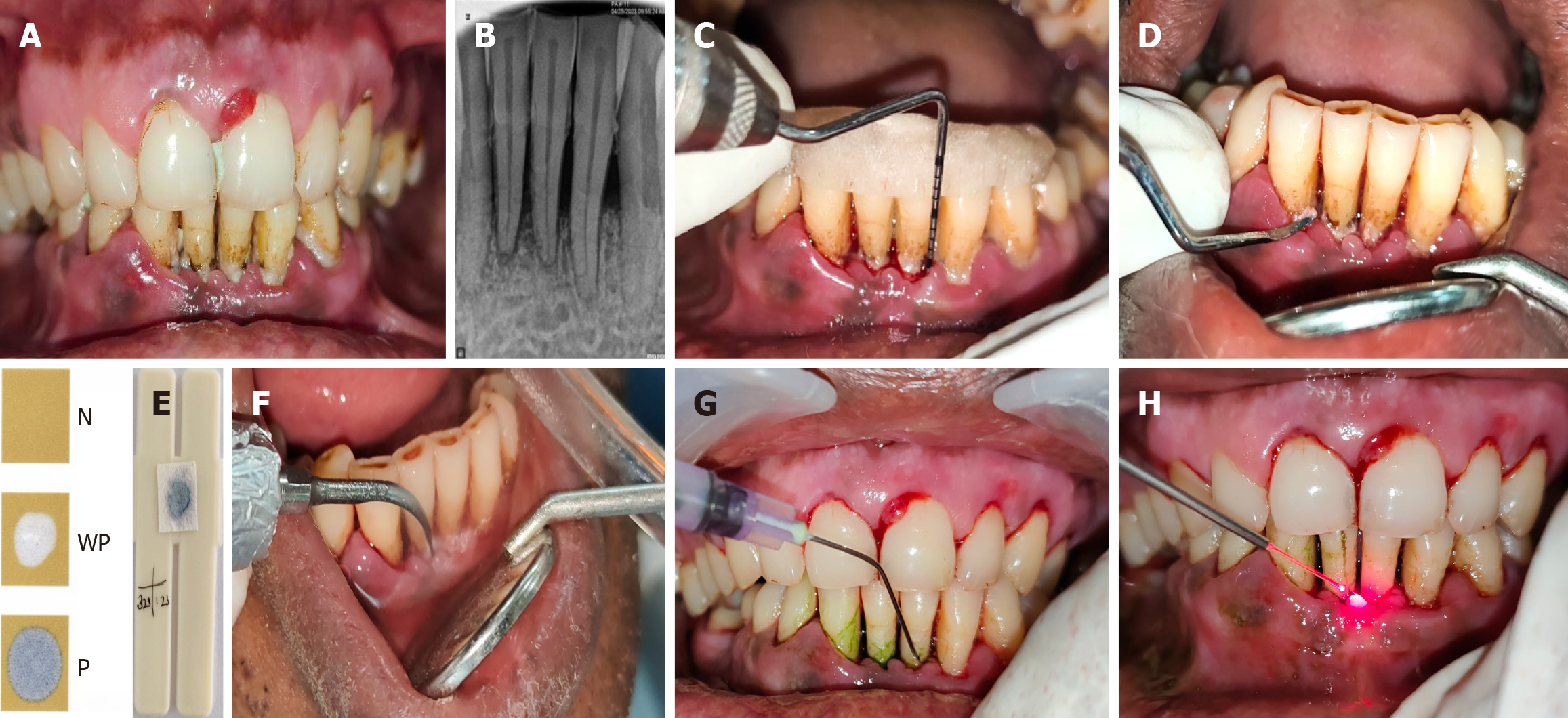
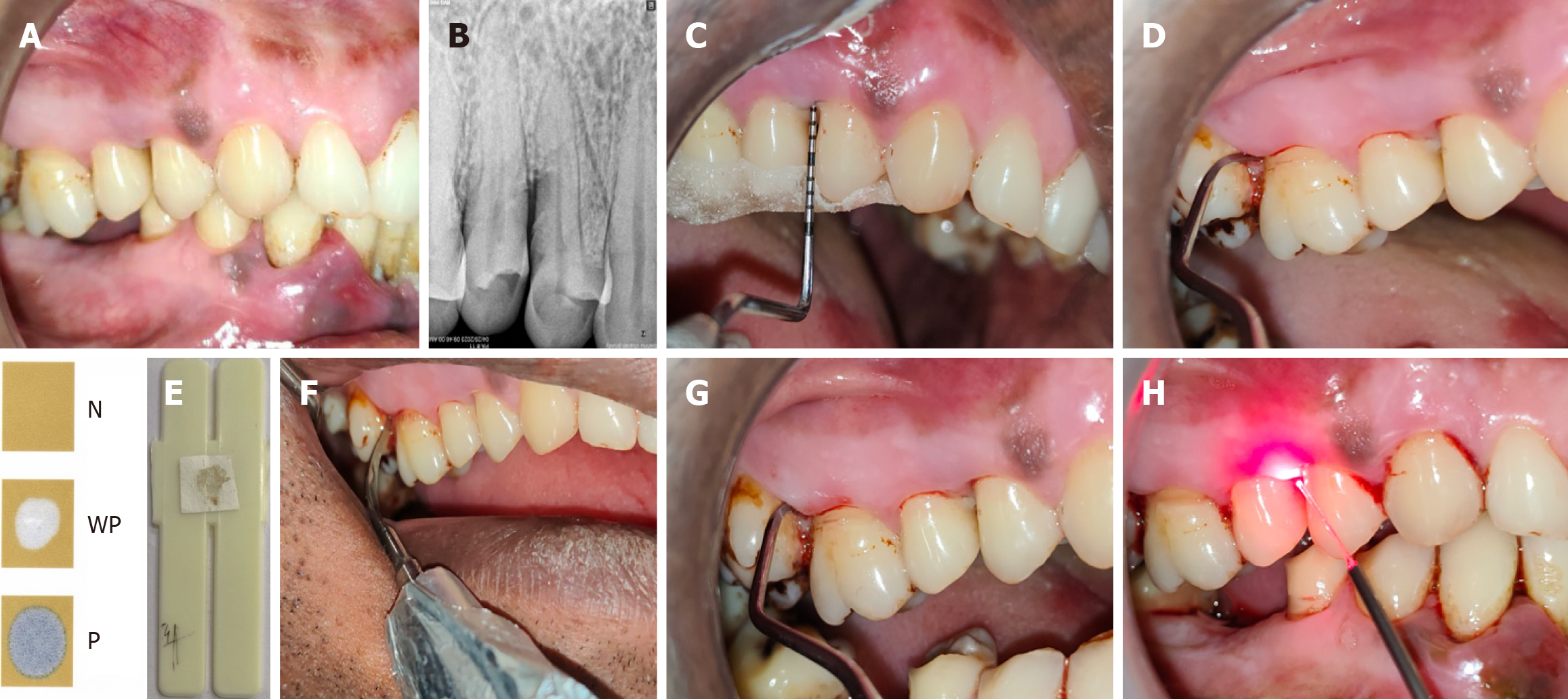
The microbiological parameter assessed was: BANA enzymatic test[27]. Subgingival plaque specimens were obtained at baseline, immediately following treatment, and during the 1-month and 3-month follow-up visits. For each participant, six non-adjacent interproximal sites were selected and distributed randomly across various dental quadrants. Upon recording the clinical parameters, supragingival deposits were carefully removed, and plaque samples were gathered using separate sterile Gracey curettes (Figures 2 and 3). Each specimen was then promptly transferred onto BANA-impregnated filter papers (B4750, Sigma Aldrich; Supertek Co. Ltd., India) positioned along the lower edge of a plastic diagnostic strip (Pride 015, Pvt. Ltd., India). The prepared strips were subsequently incubated at a temperature of 55 °C for 15 minutes[27].
The findings were categorized based on colour intensity as follows: Distinct dark blue coloration indicating a strong positive response (score 2); faint light blue coloration representing a weak positive response (score 1); and absence of any colour change denoting a negative result (score 0) (Figures 2 and 4).
A preliminary investigation was conducted in the Department of Periodontics, SCBDCH. The sample size was calculated using G Power software version 3.1.9.2 (SPSS Software India, Norman H. Nie, 2015). Parameters considered for computation included a statistical power of 85%, a confidence level of 95%, and a significance level of 5%, with an effect size of 0.8 to detect a mean intergroup difference of 1 mm (δ). Based on these criteria, the required sample size was estimated at 30 participants. To accommodate an anticipated dropout rate of about 10%, a total of 33 subjects who satisfied the criteria for inclusion and exclusion were recruited for the study following acquisition of informed consent.
Prior to commencement, the investigator underwent structured training in all measurement and data recording procedures, which included calibration sessions conducted at the Postgraduate Department of Periodontics, SCBDCH. Intra-examiner reliability was assessed by re-examining 40 sites across 20 participants at an interval of 24 hours before study initiation. Calibration was considered satisfactory when measurements differed by no more than 1 mm at the 95% confidence level.
A total of sixty-six periodontal sites from thirty-three individuals aged over 30 years, attending the outpatient Department of Periodontics at SCBDCH, Cuttack, Odisha, were enrolled in accordance with the predefined inclusion criteria. Following recruitment, the 33 participants (66 sites) were allocated to either the test group (33 sites treated with aPDT using CNPs-ICG in addition to SRP) or the control group (33 sites managed with conventional SRP followed by laser irradiation alone) via randomization, which was performed through a computer-generated sequence prepared in Microsoft Excel 2013, version 15.0, for Windows.
The software-generated allocation sequence was revealed exactly prior to the clinical procedure to determine, the side or arch to be treated as either the test or control site. Participants received detailed information regarding the treatment protocol, postoperative care instructions, and potential adverse effects. Randomization of treatment modalities was executed using pre-established randomization tables, with participants numbered sequentially based on their order of enrolment. A minimum interval of two weeks was maintained between interventions performed on opposite sides or arches.
(1) Conventional SRP: All participants underwent full-mouth supragingival scaling utilizing a piezoelectric ultrasonic device [NSK scaler (OEM) unit] in conjunction with manual hand instruments to achieve smooth tooth surfaces. Subsequently, comprehensive subgingival root planing was performed under local anaesthesia employing Gracey curettes (Hu-Friedy, United States). The procedure was prolonged until complete debridement and planing of the root surfaces were confirmed (Figure 2F and Figure 4F); (2) Test site: They received aPDT with CNPs-ICG at a concentration of 1000 μg/mL. The active solution was supplied to the designated test sites using a disposable 3-mL syringe fitted with a fine cannula, which was carefully inserted into the deep periodontal pockets until overflow of the liquid was observed surrounding the tooth margins. The treated sites were then allowed to remain undisturbed for approximately 1 minute to 2 minutes (Figure 2G). The patients were then instructed to rinse and remove any excess. Irradiation of CNPs-ICG delivered sites was performed keeping the previously mentioned laser parameters[24] (Figure 2H); and (3) Control site: The control sites went through the identical irradiation protocol during the baseline visit; however, the procedure was performed without the use of CNPs-ICG (Figure 4).
Post treatment instructions: Following the procedure, all patients were advised to brush and floss gently for 1 week & then resume their regular oral hygiene routines. They were advised to rinse with a warm saline solution, prepared by dissolving approximately half a teaspoon of salt in 8 ounces of water, three times daily for a duration of one week.
Recall appointments: The participants were scheduled for follow-up evaluations at 1 month and 3 months. During each visit, any reported adverse effects were documented, oral hygiene instructions were reiterated, and both clinical and microbiological parameters were re-assessed. All newly formed supragingival deposits were thoroughly removed at each appointment (Figures 3 and 5).
Data were analysed utilizing SPSS software for Windows (version 22.0; IBM Corp., Armonk, NY, United States). Continuous variables between groups were compared using the unpaired t-test. Within group comparisons at various time points were conducted using repeated-measures ANOVA, followed by post hoc pairwise analyses. A P value of less than 0.05 was considered statistically significant. The results were represented through tables and graphical illustrations.
A sample of 33 subjects were initially recruited in this split mouth study; however, 3 subjects were lost to follow-up and the remaining, 30 subjects were assigned into 2 groups, group-I: Test sites (30 sites in which aPDT with CNPs-ICG + SRP was performed) and in group-II: Control sites (30 sites in which conventional SRP with laser irradiation only was performed). All the 30 subjects completed the study and there were no dropouts.
The study participants were of a mean age of 38.07 ± 5.1 years (range: 30-48 years). Out of 30 participants, 17 were males and 13 were females. The test and control groups were statistically identical in terms of age and gender distribution. Among the periodontitis affected sites analysed, 29 were located in the mandibular arch and 31 in the maxillary arch. With respect to tooth position, 17 sites were located in the anterior region and 43 sites in the posterior region.
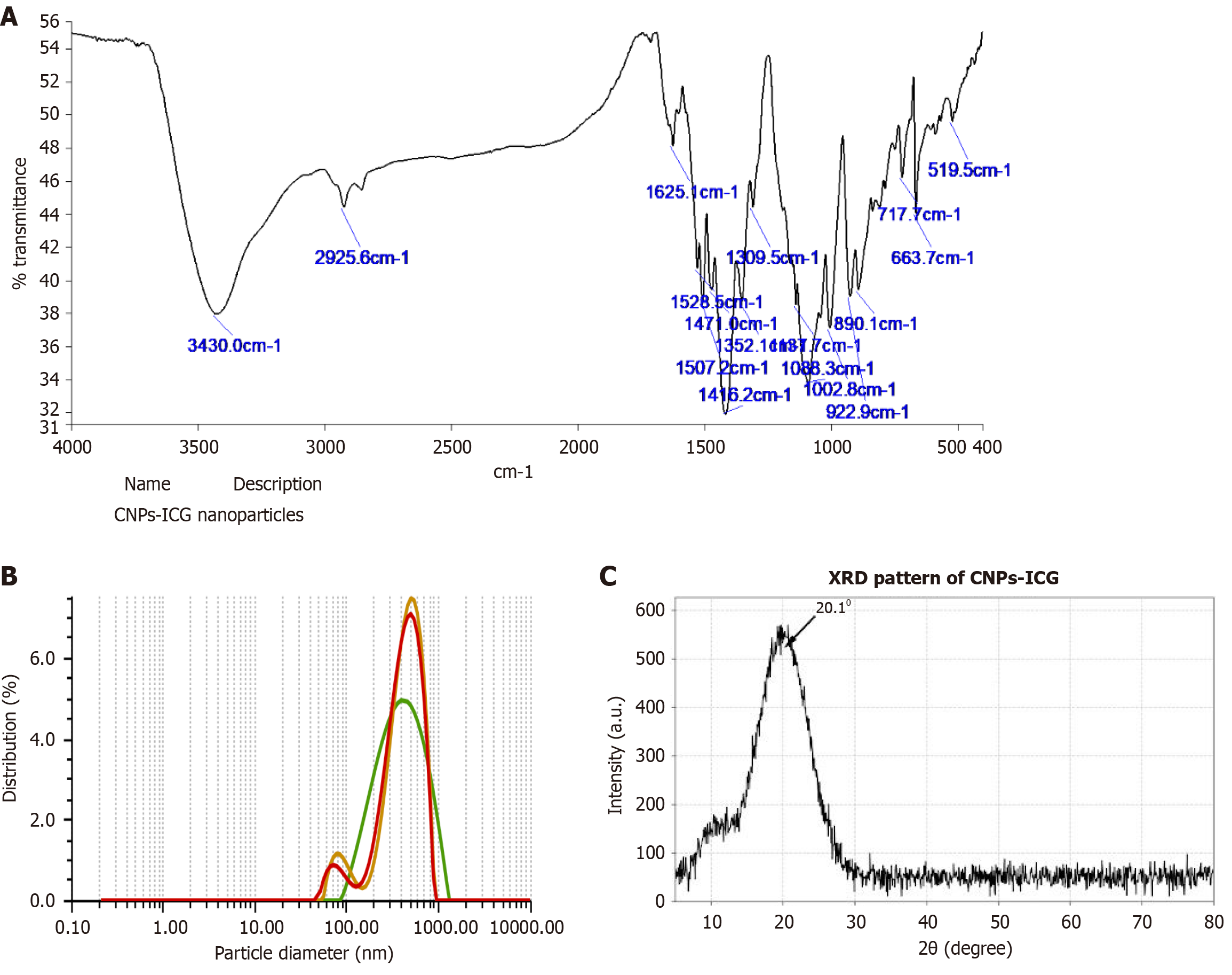
The FT-IR spectrum of CNPs-ICG (Figure 6A) verifies the effective conjugation of ICG molecules onto CNPs, as evidenced by the appearance of characteristic absorption bands near 1002.8 cm-1, corresponding to the S = O, C-S, and S-O stretching vibrations of ICG. Further evidence of ICG incorporation was indicated by the characteristic stretching vibrations of C = C and C = N observed around 1416.2 cm-1 and 1625.1 cm-1, respectively, along with the C-N single-bond stretching near 1309.5 cm-1, all consistent with the spectral features of ICG. These findings collectively confirm the successful synthesis of CNPs-ICG.
The hydrodynamic diameter of CNPs-ICG was measured using an Anton Paar Nano ZS, India system at 250C by DLS (Figure 6B). It was found that the mean hydrodynamic diameter of CNPs-ICG size was 417.4 ± 5.67 nm.
The XRD pattern of CNPs-ICG (Figure 6C) exhibited a broad diffraction peak at approximately 2θ = 20.10, which is characteristic of the semi-crystalline nature of chitosan. The presence of this broad peak, rather than sharp, intense reflections, suggests that the Chitosan nanoparticles exist in a predominantly amorphous state with short-range order.
Upon the incorporation of ICG, no additional crystalline peaks corresponding to pure ICG were observed. This absence of distinct ICG peaks indicates that the ICG molecules were successfully dispersed within the nanoparticle matrix at a molecular level or existed in an amorphous state within the CNPs. Furthermore, a slight decrease in the intensity of the characteristic chitosan peak at 200 was noted compared to pure chitosan, suggesting that the cross-linking process and ICG doping slightly disrupted the intermolecular hydrogen bonding of the chitosan chains, further reducing its crystallinity. These results are consistent with the successful formation of a stable, nano-encapsulated complex.
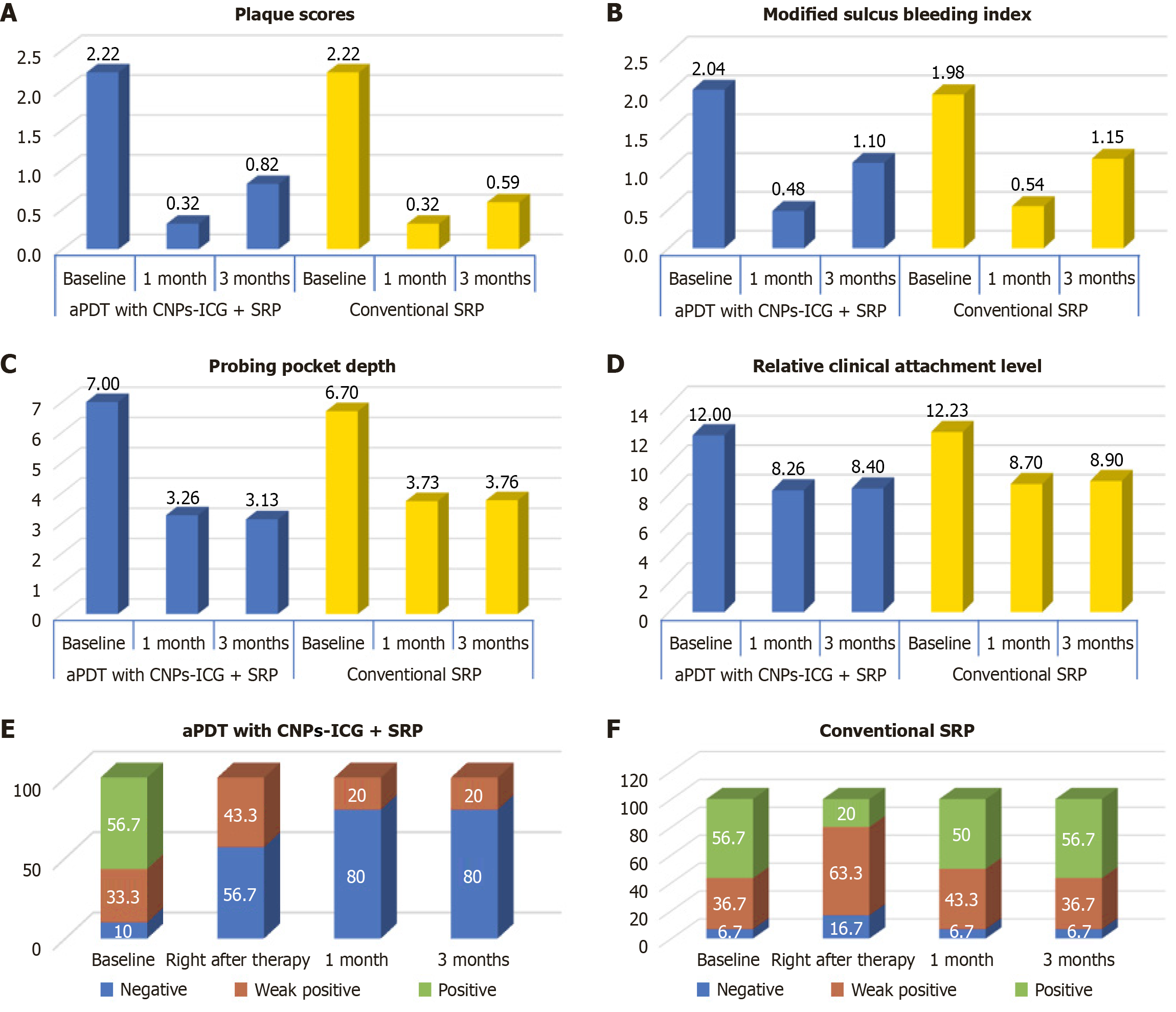
Both test and control groups demonstrated a significant decrease in mean plaque score from baseline to 1 month (2.22 to 0.32 and 2.2 to 0.32, respectively), which then increased at 3 months (0.82 for aPDT, 0.59 for SRP) but the scores remained lower than baseline. All inter-interval changes were statistically significant (P = 0.001), with significant reduction from baseline to 1 month, increase from 1 month to 3 months, and net decrease from baseline to 3 months in both groups (P = 0.001 for each comparison) (Tables 1 and 2, Figure 7A).
| aPDT with CNPs-ICG + SRP | Conventional SRP | ||||||||
| Mean | SD | MD | P value | Mean | SD | MD | P value | ||
| Baseline | 2.22 | 0.64 | 1.907 | 0.0011 | Baseline | 2.2 | 0.52 | 1.883 | 0.0011 |
| 1 month | 0.32 | 0.04 | 1 month | 0.32 | 0.04 | ||||
| 1 month | 0.32 | 0.04 | -0.499 | 0.0011 | 1 month | 0.32 | 0.04 | -2.69 | 0.0011 |
| 3 months | 0.82 | 0.4 | 3 months | 0.59 | 0.05 | ||||
| 3 months | 0.82 | 0.4 | -1.408 | 0.0011 | 3 months | 0.59 | 0.05 | -1.614 | 0.0011 |
| Baseline | 2.22 | 0.64 | Baseline | 2.22 | 0.52 | ||||
Both test and control groups showed statistically significant reductions in mean sulcular bleeding score from baseline to 1 month (2.04 to 0.48 and 1.98 to 0.54, respectively), with a subsequent increase at 3 months (1.1 for aPDT, 1.15 for SRP), although scores remained below baseline. All inter-interval changes were statistically significant (P = 0.001), with reduction in the 1st month after therapy, increase from 1 month to 3 months, and net decrease from baseline to 3 months in both groups (P = 0.001) (Tables 3 and 4, Figure 7B).
| aPDT with CNPs-ICG + SRP | Conventional SRP | ||||||||
| Mean | SD | MD | P value | Mean | SD | MD | P value | ||
| Baseline | 2.04 | 0.44 | 1.56 | 0.0011 | Baseline | 1.98 | 0.53 | 1.435 | 0.0011 |
| 1 month | 0.48 | 0.36 | 1 month | 0.54 | 0.42 | ||||
| 1 month | 0.48 | 0.36 | -0.632 | 0.0011 | 1 month | 0.54 | 0.42 | -0.607 | 0.0011 |
| 3 months | 1.1 | 0.2 | 3 months | 1.15 | 0.27 | ||||
| 3 months | 1.1 | 0.2 | -0.943 | 0.0011 | 3 months | 1.15 | 0.27 | -0.828 | 0.0011 |
| Baseline | 2.04 | 0.44 | Baseline | 1.98 | 0.53 | ||||
aPDT with CNPs-ICG group demonstrated a reduction in mean probing pocket depth from 7 mm at baseline to 3.26 mm at 1 month and 3.13 mm at 3 months, with statistically significant differences at all time points (P = 0.001) except between 1 and 3 months (P = 0.87). The SRP group showed a decrease from 6.7 mm to 3.73 mm at 1 month, with a mi
| aPDT with CNPs-ICG + SRP | Conventional SRP | ||||||||
| Mean | SD | MD | P value | Mean | SD | MD | P value | ||
| Baseline | 7 | 1.08 | 3.733 | 0.0011 | Baseline | 6.7 | 1.104 | 3.03 | 0.0011 |
| 1 month | 3.26 | 0.52 | 1 month | 3.73 | 0.63 | ||||
| 1 month | 3.26 | 0.52 | 0.133 | 0.87 | 1 month | 3.73 | 0.63 | -0.033 | 0.99 |
| 3 months | 3.13 | 0.62 | NS | 3 months | 3.76 | 0.77 | |||
| 3 months | 3.13 | 0.62 | -3.86 | 0.0011 | 3 months | 3.76 | 0.77 | -3 | 0.0011 |
| Baseline | 7 | 1.08 | Baseline | 6.7 | 1.104 | ||||
In the aPDT with CNPs-ICG group the mean RCAL reduced from 12 mm at baseline to 8.26 mm at 1 month and 8.4 mm at 3 months, all changes were statistically significant (P = 0.001) except for the slight increase between 1 month and 3 months (P = 0.31). In the SRP group RCAL decreased from 12.23 mm to 8.7 mm at 1 month, and increased slightly to 8.9 mm at 3 months; these changes were statistically significant (P = 0.001) except for the difference between 1-month and 3-months intervals (P = 0.068) (Tables 7 and 8, Figure 7D).
| aPDT with CNPs-ICG + SRP | Conventional SRP | ||||||||
| Mean | SD | MD | P value | Mean | SD | MD | P value | ||
| Baseline | 12 | 1.01 | 3.73 | 0.0011 | Baseline | 12.2 | 1.16 | 3.5 | 0.0011 |
| 1 month | 8.26 | 0.52 | 1 month | 8.7 | 0.98 | ||||
| 1 month | 8.26 | 0.52 | -0.133 | 0.31 | 1 month | 8.7 | 0.98 | -0.167 | 0.068 |
| 3 months | 8.4 | 0.62 | NS | 3 months | 8.9 | 0.99 | NS | ||
| 3 months | 8.4 | 0.62 | -3.6 | 0.0011 | 3 months | 8.9 | 0.99 | -3.33 | 0.0011 |
| Baseline | 12 | 1.01 | Baseline | 12.2 | 1.16 | ||||
aPDT with CNPs + ICG group showed an elevation in the percentage of sites with negative results of BANA Test from 10% at baseline to 56.7% immediately post-therapy and subsequently to 80% at both 1-month and 3-months follow-up visits, the distribution was statistically significant (P = 0.001) (Table 9, Figure 7E). Conventional SRP group showed an elevation in the percentage of sites with negative results from 6.7% to 16.7% between baseline and immediate post-therapy and again decreasing to 6.7% at 1-month and 3-months follow-up visits, the distribution was statistically non-significant (P = 0.07) (Table 10, Figure 7F).
| Negative | Weak positive | Positive | Total | P value | |
| Baseline | 3 (10) | 10 (33.3) | 17 (56.7) | 30 (100) | 0.0011 |
| Right after therapy | 17 (56.7) | 13 (43.3) | 0 | 30 (100) | |
| 1 month | 24 (80) | 6 (20) | 0 | 30 (100) | |
| 3 months | 24 (80) | 6 (20) | 0 | 30 (100) | |
| Total | 68 (56.7) | 35 (29.2) | 17 (14.2) | 120 (100) |
| Negative | Weak positive | Positive | Total | P value | |
| Baseline | 2 (6.7) | 11 (36.7) | 17 (56.7) | 30 (100) | 0.07 |
| Right after therapy | 5 (16.7) | 19 (63.3) | 6 (20) | 30 (100) | NS |
| 1 month | 2 (6.7) | 13 (43.3) | 15 (50) | 30 (100) | |
| 3 months | 2 (6.7) | 11 (36.7) | 17 (56.7) | 30 (100) | |
| Total | 11 (9.2) | 54 (45) | 55 (45.8) | 120 (100) |
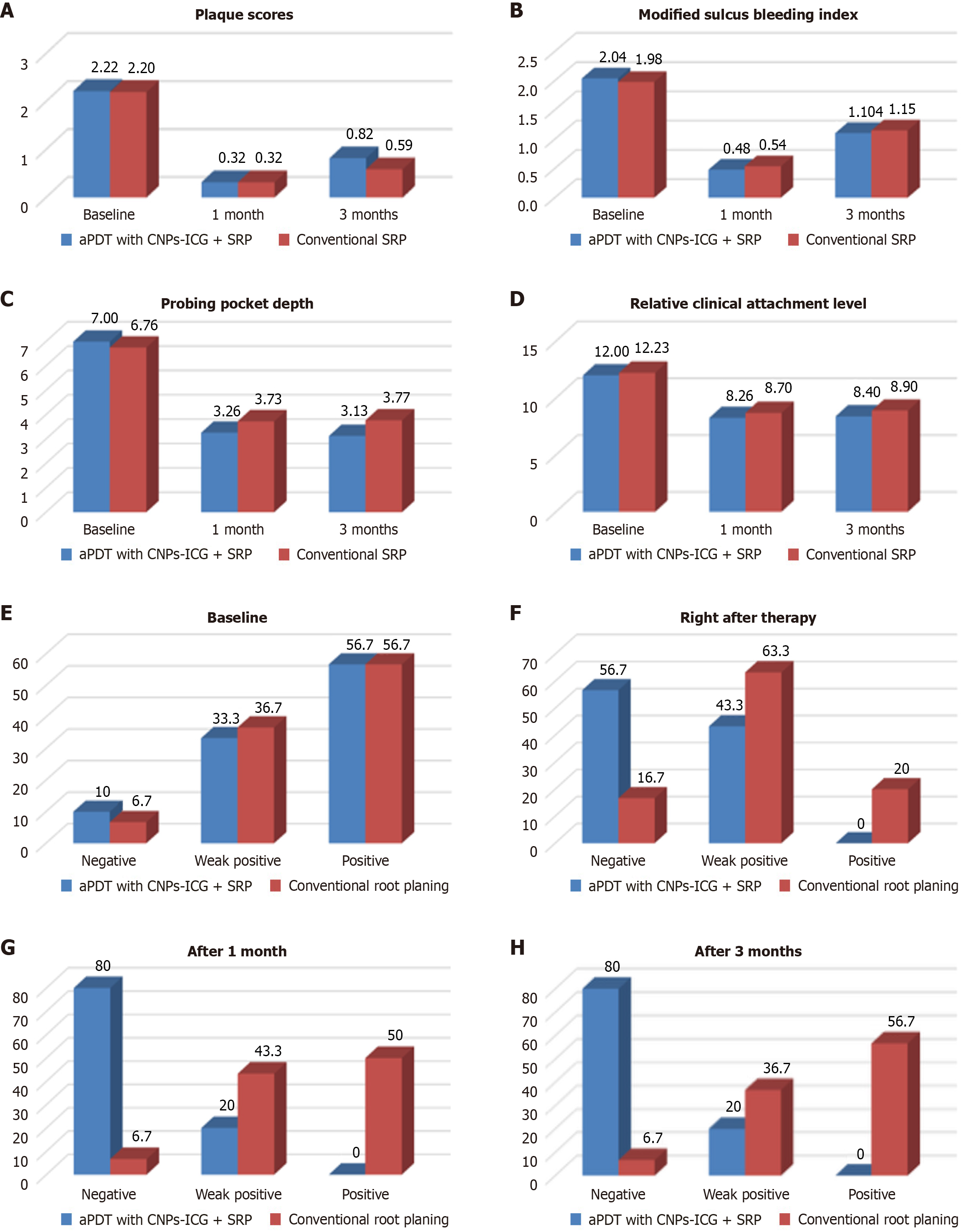
Baseline comparisons showed no significant differences between the aPDT with CNPs-ICG + SRP and conventional SRP groups in plaque score (2.22 and 2.2, P = 0.84) (Table 11, Figure 8A), sulcular bleeding index (2.04 and 1.98, P = 0.6) (Table 12, Figure 8B), probing pocket depth (7 mm and 6.76 mm, P = 0.41) (Table 13, Figure 8C) and relative clinical attachment level (12 mm and 12.23 mm, P = 0.41) (Table 14, Figure 8D). At 1 month, both groups had similar plaque scores (0.32, P = 0.99) and sulcular bleeding scores (0.48 and 0.54, P = 0.52), but aPDT group showed significantly lower probing depth (3.26 mm and 3.73 mm, P = 0.003) and RCAL (8.26 mm and 8.7 mm, P = 0.026) than SRP group. At 3 months, SRP group had a lower plaque score (0.59 and 0.82, P = 0.005), while no statistically significant differences were observed in bleeding scores (1.15 and 1.104, P = 0.43). aPDT group maintained significantly lower probing depth (3.13 mm and 3.77 mm, P = 0.001) and RCAL (8.4 mm and 8.9 mm, P = 0.023) compared to SRP group.
| Plaque index | Number | Mean | SD | t | P value | |
| Baseline | aPDT with CNPs-ICG + SRP | 30 | 2.22 | 0.64 | 0.159 | 0.84 |
| Conventional SRP | 30 | 2.2 | 0.52 | NS | ||
| 1 month | aPDT with CNPs-ICG + SRP | 30 | 0.32 | 0.04 | 0 | 0.99 |
| Conventional SRP | 30 | 0.32 | 0.04 | NS | ||
| 3 months | aPDT with CNPs-ICG + SRP | 30 | 0.82 | 0.4 | 3.07 | 0.0051 |
| Conventional SRP | 30 | 0.59 | 0.05 |
| Modified sulcus bleeding index | Number | Mean | SD | t | P value | |
| Baseline | aPDT with CNPs-ICG + SRP | 30 | 2.04 | 0.44 | 0.52 | 0.6 |
| Conventional SRP | 30 | 1.98 | 0.53 | NS | ||
| 1 month | aPDT with CNPs-ICG + SRP | 30 | 0.48 | 0.36 | -0.63 | 0.52 |
| Conventional SRP | 30 | 0.54 | 0.42 | NS | ||
| 3 months | aPDT with CNPs-ICG + SRP | 30 | 1.104 | 0.2 | -0.787 | 0.43 |
| Conventional SRP | 30 | 1.15 | 0.27 | NS | ||
At baseline, both aPDT with CNPs-ICG + SRP and SRP groups showed 56.7% positive BANA sites, with no significant difference between groups (P = 0.88) (Table 15, Figure 8E). Immediately after therapy, 56.7% of sites were negative in the aPDT group vs 16.7% for SRP; all other BANA results (weak/positive) also favoured aPDT, with significant distribution (P = 0.001) (Table 16, Figure 8F). One month later, 80% of sites in aPDT group were negative compared to 6.7% in SRP, with no positive results for aPDT and significantly fewer weak positives (P = 0.001) (Table 17, Figure 8G). At 3 months, 80% sites remained negative for aPDT vs 6.7% for SRP, none were positive for aPDT, and weak positives were markedly fewer in test group than in the control, all differences being significant statistically (P = 0.001) (Table 18, Figure 8H).
| Baseline | aPDT with CNPs-ICG + SRP | Conventional SRP | Total | P value |
| Negative | 3 (10) | 2 (6.7) | 5 (8.3) | 0.88 |
| Weak positive | 10 (33.3) | 11 (36.7) | 21 (35) | NS |
| Positive | 17 (56.7) | 17 (56.7) | 34 (56.7) | |
| Total | 30 (100) | 30 (100) | 60 (100) |
| Right after therapy | aPDT with CNPs-ICG + SRP | Conventional SRP | Total | P value |
| Negative | 17 (56.7) | 5 (16.7) | 22 (36.7) | 0.0011 |
| Weak positive | 13 (43.3) | 19 (63.3) | 32 (53.3) | |
| Positive | 0 | 6 (20) | 6 (10) | |
| Total | 30 (100) | 30 (100) | 60 (100) |
| After 1 month | aPDT with CNPs-ICG + SRP | Conventional SRP | Total | P value |
| Negative | 24 (80) | 2 (6.7) | 26 (43.3) | 0.0011 |
| Weak positive | 6 (20) | 13 (43.3) | 19 (31.7) | |
| Positive | 0 | 15 (50) | 15 (25) | |
| Total | 30 (100) | 30 (100) | 60 (100) |
This single-centered, split-mouth randomized controlled clinical trial was undertaken to evaluate and compare both clinical and microbiological responses following aPDT employing chitosan nanoparticles incorporated with indocyanine green dye, in comparison to conventional SRP.
According to existing literature, only a single clinical trial has been documented that assessed the in vivo effects of aPDT using MB-loaded poly (lactic-co-glycolic acid) nanoparticles on dental plaque microorganisms in individuals with chronic periodontitis[28].
The split-mouth approach was adopted to minimize inter-individual variability, thereby enhancing the reliability of outcome estimation with a reduced sample size. This design was considered appropriate since the light source by itself does not possess intrinsic antimicrobial activity. A fundamental prerequisite for utilizing this design is the assurance that no cross-contamination or spill-over effect occurs between the treated and control sites[29]. A minimum interval of two weeks was maintained between the test and control interventions, ensuring the absence of any potential spill-over effects between groups. This design enabled an unbiased and independent evaluation of all clinical and microbiological parameters[29].
No participants reported any adverse clinical effects subsequent to the procedure. The sample size employed in this trial was consistent with that commonly adopted in most human periodontal clinical investigations and was further determined through a preliminary pilot study combined with appropriate statistical sample size estimation[24]. Both groups showed statistically significant improvements in clinical findings from baseline to one and three months. A notable decrease in sites exhibiting positive BANA test results was also found in both groups during the same review periods. The test group treated with CNPs-ICG combined with SRP exhibited greater improvement in PPD and RCAL[30] compared with the control group receiving conventional SRP with laser irradiation alone at both follow-up intervals, and these differences were statistically significant. Additionally, the test sites showed a significantly larger decrease in BANA-positive sites relative to the control treatment.
A thorough literature search yielded few reports on clinical and microbiological changes that take place following aPDT using ICG[20,22,24,31,32]. In the current study, the evaluated parameters included PI score[25], mSBI score[26], PPD, RCAL[30] and microbiological evaluation using BANA enzymatic test[27]. Furthermore, our research appears to be the first to undertake comparative analysis of both clinical and microbiological outcomes between aPDT utilizing chitosan nanoparticles incorporated with indocyanine green dye and conventional SRP, based on current knowledge in the field.
The primary objective of integrating indocyanine green with chitosan nanoparticles is to overcome the major limitations associated with ICG, namely its aqueous instability, susceptibility to photo and thermal degradation under both in vitro and in vivo conditions[33]. Physical and chemical alterations in indocyanine green may cause discoloration, diminished light absorption capacity, reduced fluorescence intensity, and a change in its absorption wavelength[34,35].
Polymeric nanoparticles such as CNPs have recently been shown to enhance the aqueous, photostability, and thermal stability of ICG[36]. Chitosan is a common ingredient in many pharmaceutical nano-formulations because it is bio
FTIR analysis of the CNPs-ICG complex revealed morphological and structural characteristics indicative of successful immobilization of indocyanine green within the chitosan nanoparticles, as evidenced by the appearance of specific characteristic bands, identical with the findings reported by Pourhajibagher et al[22]. Similarly the XRD analysis report also confirms the immobilization of indocyanine green within the chitosan nanoparticles.
Moreover, the distribution of hydrodynamic diameter as assessed by DLS analysis showed a mean hydrodynamic diameter of CNPs + ICG to be 417.4 ± 5.67 nm, in contrast study by Pourhajibagher et al[22] demonstrated a size range of CNPs-ICG particle of 10-20 nm.
In the current study, comparison of mean plaque scores between the test group and the control group demonstrated a statistically significant difference (P = 0.005). Both groups demonstrated a declining trend in mean plaque scores at the one-month (mean ± SD = 0.32 ± 0.04) and three-month follow-up periods. It was similar to the results found in studies by Srikanth et al[38] and Monzavi et al[39]. This observation could be attributed to the meticulous periodontal maintenance performed throughout the follow-up period. As supported by existing literature, effective cleansing of dental surfaces to remove plaque and calculus continues to be central to successful periodontal management and disease eradication.
The mean bleeding score was less in the test group compared to the control group; however, the difference was not statistically significant (test group: mean ± SD = 1.104 ± 0.2 and control group: mean ± SD = 1.15 ± 0.27; P = 0.43). In both test and control groups there was statistically significant reduction in mean bleeding score from baseline to 1 month, which again increased statistically on 3 months re-evaluation visit when compared to the 1 month follow up although the values persisted below the baseline readings which was similar to the studies by Polansky et al[40], Ge et al[41], Segarra-Vidal et al[42], Tabenski et al[43] and Annunziata et al[44]. There was reduction of bleeding scores in both the approaches suggesting reduction in inflammation during both the treatment modalities. Bleeding on probing primarily occurs as a result of increased vascularity, epithelial thinning, degeneration, and the presence of dilated blood vessels near the inner surface. Mechanical debridement alone can effectively remove local irritants, promoting restoration and healing of the attachment apparatus. However, the observed increase in bleeding scores during follow-up visits could be due to multiple factors, such as tooth positioning or other local anatomical challenges that restrict adequate access for treatment and effective plaque control.
Both the test and control groups showed statistically significant decrease in mean PPD from baseline to 1 month and 3 months, respectively however the difference between first month and third months were not statistically significant in both the groups. While comparing the CNPs-ICG + SRP group and control group, none of the differences found were statistically significant at baseline, but at 1 month re-evaluation visit, mean PPD was statistically significant (P = 0.003) in case of CNPS-ICG + SRP group (3.26 ± 0.52) than the control group (3.73 ± 0.63), which was even more at 3 months re-evaluation visit (P = 0.001) between the test (3.13 ± 0.62) and control groups (3.77 ± 0.77). This result is in accordance with results of PPD reduction from Alwaeli et al[45], Monzavi et al[39], Sigusch et al[46], Sethi et al[47], Lulic et al[48] and Giannelli et al[49]. Furthermore, a recent systematic review by Feng et al[50] concluded that PDT serves as an effective adjunct for grade B periodontitis by significantly improving clinical attachment levels and reducing both probing depths and bleeding on probing, often yielding outcomes comparable or superior to conventional SRP.
However, studies by Polansky et al[40], Ge et al[41], Bassir et al[51] and Rühling et al[52] failed to show any significant difference in mean probing depth between test and control groups when aPDT was compared to conventional SRP.
A pairwise comparison revealed a statistically significant improvement of relative clinical attachment level from baseline to 1 month and from baseline to 3 months in the groups. However, the deterioration from 1 month to 3 months was not statistically significant. When comparison was done between test and control group, it was found that at baseline there was no significant difference between aPDT with CNPs-ICG group and conventional SRP group. In 1 month, follow-up visit, it was found that there was significantly more gain in attachment in case of aPDT with CNPs-ICG group than control group (P = 0.026) and although there was a non-significant loss of attachment on 3 months re-evaluation visit in both the groups, test group was still better than control group (P = 0.023). This result was in accordance to that of Betsy et al[53], Sigusch et al[46], and Sethi et al[47] However Srikanth et al[38], Monzavi et al[39], Bassir et al[51] and Rühling et al[52] reported findings that differed from the present study, observing no statistically significant variation between the test and control groups with respect to clinical attachment level.
The BANA test is a quick and dependable chairside method of diagnosis that can be completed within approximately 15 minutes. It provides information on the presence of three key putative periodontal pathogens-Porphyromonas gingivalis, Tannerella forsythia, and Treponema denticola-all of which can hydrolyse the trypsin substrate, BANA.
In the present study, when BANA test results were evaluated, at baseline, there was no statistically significant differences between aPDT with CNPs-ICG group and conventional SRP group in terms of sites with positive results (P = 0.88). However right after therapy there was statistically significant improvement in sites with negative result in case of test group than control group (P = 0.001) which was even better 1 month after the therapy (P = 0.001) that maintained for 3 months. Although this is probably the initial clinical study evaluating the microbiological effect of aPDT using CNPs-ICG photosensitizer with the help of BANA microbiological test, there were several studies in the literature evaluating the microbiological effects of aPDT with various other dyes. Corroborating the antimicrobial potential of this specific formulation, Sayar et al[54] demonstrated that aPDT utilizing indocyanine green-loaded chitosan nanoparticles significantly reduces Aggregatibacter actinomycetemcomitans colony counts on dental implants, achieving a decontamination efficacy comparable to that of chlorhexidine. Result of this study is in accordance with that of Sigusch et al[46], Petelin et al[55], and Segarra-Vidal et al[42] which have shown significant reduction in abundance of periodontopathogens following aPDT.
Dhalla et al[27] demonstrated a significant statistical correlation between the results of BANA test and the extent of periodontal damage, as well as a strong association between positive BANA test results and the amount of microbial plaque present.
A follow up at 1-month and 3-months after aPDT was decided on the basis of the methodology as described by AlSarhan et al[24] and Monzavi et al[39]. In the present study, the duration of the follow-up period was relatively short. Moreover, findings from the study by Boehm and Ciancio[15] indicated that the inclusion of indocyanine green markedly enhanced bacterial elimination within a brief exposure period. Moreover Morrison et al[56] suggested to wait for at least 1 month before re-evaluating the clinical findings after any non-surgical periodontal therapy.
Blinding of treatment: In a split-mouth study design, blinding of participants, when possible, helps reduce treatment response bias. Although blinding of both patients and the operator was challenging in the present study, adequate measures were undertaken to minimize bias by administering laser irradiation to the control sites following conventional SRP.
Photosensitizer concentration and laser parameters: The present study examined only a specific concentration of CNPs-ICG in conjunction with defined physical characteristics of the laser system. This limitation may influence the generalizability of the findings, as the therapeutic efficacy of the photosensitizer could vary under different laser parameters.
Follow-up period: The application of CNPs-ICG-mediated aPDT in periodontitis management remains debatable, particularly the concern between long-term and short-term efficacy. Most available studies, including the present one, have utilized relatively short follow-up durations ranging from 8 weeks to 36 weeks, which may not fully capture the long-term therapeutic outcomes. Extended follow-up periods could potentially yield different clinical results.
Microbiological assessment: Although both clinical and microbiological outcomes were assessed in this present trial, it would have been more specific to analyse oral microbiome using 16S rRNA metagenomic sequencing for periodontopathogens than doing BANA test that is an indirect microbiological test to assess the bacterial enzymes.
Based upon the review of previous literatures, it was proposed that aPDT using CNPs-ICG may be used as an effective adjunct to SRP, potentially enhancing treatment outcomes compared to mechanical therapy alone in periodontitis management. Therefore, the objective of this study was to evaluate both the clinical and microbiological effects of CNPs-ICG mediated aPDT as a supplementary approach in the non-surgical therapy of periodontitis.
Based on the findings of this study it can be concluded that aPDT using CNPs-ICG + SRP can be a promising non-surgical treatment option in periodontitis patients having uncontrolled systemic conditions e.g., unstable angina, uncontrolled hypertension etc. in which surgical treatments are contraindicated; patients with residual deep pockets and in patients having resistant strains of bacteria causing repeated periodontal infections without causing any harm to adjacent tissues and bacteraemia in patients having compromised immunity.
Future clinical trials employing a standardized PDT protocol, larger sample sizes, and uniform clinical, biochemical, and microbiological evaluations are necessary to accurately assess the long-term efficacy of aPDT using CNPs-ICG.
Mr. Mayukh Bose, MSc, Biotechnology, his whole team are acknowledged for their contribution to the study with a high level of expertise and commitment.
| 1. | Loe H, Theilade E, Jensen SB. Experimental gingivitis in man. J Periodontol (1930). 1965;36:177-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2266] [Cited by in RCA: 2154] [Article Influence: 35.3] [Reference Citation Analysis (1)] |
| 2. | Armitage GC, Cullinan MP. Comparison of the clinical features of chronic and aggressive periodontitis. Periodontol 2000. 2010;53:12-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 158] [Article Influence: 9.9] [Reference Citation Analysis (1)] |
| 3. | Taubman MA, Valverde P, Han X, Kawai T. Immune response: the key to bone resorption in periodontal disease. J Periodontol. 2005;76:2033-2041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 346] [Cited by in RCA: 321] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 4. | Kumar PS, Griffen AL, Barton JA, Paster BJ, Moeschberger ML, Leys EJ. New bacterial species associated with chronic periodontitis. J Dent Res. 2003;82:338-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 444] [Cited by in RCA: 376] [Article Influence: 16.3] [Reference Citation Analysis (1)] |
| 5. | Dahlén G, Wikström M, Renvert S. Treatment of periodontal disease based on microbiological diagnosis. A 5-year follow-up on individual patterns. J Periodontol. 1996;67:879-887. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 6. | Cieplik F, Tabenski L, Buchalla W, Maisch T. Antimicrobial photodynamic therapy for inactivation of biofilms formed by oral key pathogens. Front Microbiol. 2014;5:405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 195] [Cited by in RCA: 162] [Article Influence: 13.5] [Reference Citation Analysis (1)] |
| 7. | Nguyen S, Hiorth M. Advanced drug delivery systems for local treatment of the oral cavity. Ther Deliv. 2015;6:595-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 91] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 8. | Allison RR, Moghissi K. Photodynamic Therapy (PDT): PDT Mechanisms. Clin Endosc. 2013;46:24-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 609] [Cited by in RCA: 438] [Article Influence: 33.7] [Reference Citation Analysis (0)] |
| 9. | Dai T, Huang YY, Hamblin MR. Photodynamic therapy for localized infections--state of the art. Photodiagnosis Photodyn Ther. 2009;6:170-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 663] [Cited by in RCA: 490] [Article Influence: 28.8] [Reference Citation Analysis (1)] |
| 10. | Haag PA, Steiger-Ronay V, Schmidlin PR. The in Vitro Antimicrobial Efficacy of PDT against Periodontopathogenic Bacteria. Int J Mol Sci. 2015;16:27327-27338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 11. | Castano AP, Demidova TN, Hamblin MR. Mechanisms in photodynamic therapy: part one-photosensitizers, photochemistry and cellular localization. Photodiagnosis Photodyn Ther. 2004;1:279-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1746] [Cited by in RCA: 1340] [Article Influence: 60.9] [Reference Citation Analysis (1)] |
| 12. | Qin YL, Luan XL, Bi LJ, Sheng YQ, Zhou CN, Zhang ZG. Comparison of toluidine blue-mediated photodynamic therapy and conventional scaling treatment for periodontitis in rats. J Periodontal Res. 2008;43:162-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 52] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 13. | George S, Hamblin MR, Kishen A. Uptake pathways of anionic and cationic photosensitizers into bacteria. Photochem Photobiol Sci. 2009;8:788-795. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 225] [Cited by in RCA: 174] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 14. | Boni L, David G, Mangano A, Dionigi G, Rausei S, Spampatti S, Cassinotti E, Fingerhut A. Clinical applications of indocyanine green (ICG) enhanced fluorescence in laparoscopic surgery. Surg Endosc. 2015;29:2046-2055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 455] [Cited by in RCA: 387] [Article Influence: 35.2] [Reference Citation Analysis (1)] |
| 15. | Boehm TK, Ciancio SG. Diode laser activated indocyanine green selectively kills bacteria. J Int Acad Periodontol. 2011;13:58-63. [PubMed] |
| 16. | Huang YY, Sharma SK, Dai T, Chung H, Yaroslavsky A, Garcia-Diaz M, Chang J, Chiang LY, Hamblin MR. Can nanotechnology potentiate photodynamic therapy? Nanotechnol Rev. 2012;1:111-146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 95] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 17. | Mati-Baouche N, Elchinger P, de Baynast H, Pierre G, Delattre C, Michaud P. Chitosan as an adhesive. Eur Polym J. 2014;60:198-212. [RCA] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 118] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 18. | Chen R, Wang X, Yao X, Zheng X, Wang J, Jiang X. Near-IR-triggered photothermal/photodynamic dual-modality therapy system via chitosan hybrid nanospheres. Biomaterials. 2013;34:8314-8322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 144] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 19. | Nagahara A, Mitani A, Fukuda M, Yamamoto H, Tahara K, Morita I, Ting CC, Watanabe T, Fujimura T, Osawa K, Sato S, Takahashi S, Iwamura Y, Kuroyanagi T, Kawashima Y, Noguchi T. Antimicrobial photodynamic therapy using a diode laser with a potential new photosensitizer, indocyanine green-loaded nanospheres, may be effective for the clearance of Porphyromonas gingivalis. J Periodontal Res. 2013;48:591-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 93] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 20. | Rad MR, Pourhajibagher M, Rokn AR, Barikani HR, Bahador A. Effect of Antimicrobial Photodynamic Therapy Using Indocyanine Green Doped with Chitosan Nanoparticles on Biofilm Formation-Related Gene Expression of Aggregatibacter actinomycetemcomitans. Front Dent. 2019;16:187-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 21. | Papapanou PN, Sanz M, Buduneli N, Dietrich T, Feres M, Fine DH, Flemmig TF, Garcia R, Giannobile WV, Graziani F, Greenwell H, Herrera D, Kao RT, Kebschull M, Kinane DF, Kirkwood KL, Kocher T, Kornman KS, Kumar PS, Loos BG, Machtei E, Meng H, Mombelli A, Needleman I, Offenbacher S, Seymour GJ, Teles R, Tonetti MS. Periodontitis: Consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Periodontol. 2018;89 Suppl 1:S173-S182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1599] [Cited by in RCA: 1422] [Article Influence: 177.8] [Reference Citation Analysis (1)] |
| 22. | Pourhajibagher M, Rokn AR, Barikani HR, Bahador A. Photo-sonodynamic antimicrobial chemotherapy via chitosan nanoparticles-indocyanine green against polymicrobial periopathogenic biofilms: Ex vivo study on dental implants. Photodiagnosis Photodyn Ther. 2020;31:101834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 23. | Xu Y, Du Y. Effect of molecular structure of chitosan on protein delivery properties of chitosan nanoparticles. Int J Pharm. 2003;250:215-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 663] [Cited by in RCA: 481] [Article Influence: 20.9] [Reference Citation Analysis (1)] |
| 24. | AlSarhan MA, Altammami MA, Alaqeely RS, AlEbdi A, Jasser RA, Otaibi DA, Oraini SA, Habib SR, Alqahtani L, Alduhaymi IS, Alrabiah DK, Alaradi M, Alyamani EJ. Short-term improvement of clinical parameters and microbial diversity in periodontitis patients following Indocyanine green-based antimicrobial photodynamic therapy: A randomized single-blind split-mouth cohort. Photodiagnosis Photodyn Ther. 2021;35:102349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 25. | Löe H. The Gingival Index, the Plaque Index and the Retention Index Systems. J Periodontol. 1967;38 Suppl:610-Suppl:616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1927] [Cited by in RCA: 1851] [Article Influence: 31.4] [Reference Citation Analysis (1)] |
| 26. | Mombelli A, van Oosten MA, Schurch E Jr, Land NP. The microbiota associated with successful or failing osseointegrated titanium implants. Oral Microbiol Immunol. 1987;2:145-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1451] [Cited by in RCA: 1334] [Article Influence: 34.2] [Reference Citation Analysis (0)] |
| 27. | Dhalla N, Patil S, Chaubey KK, Narula IS. The detection of BANA micro-organisms in adult periodontitis before and after scaling and root planing by BANA-Enzymatic™ test kit: An in vivo study. J Indian Soc Periodontol. 2015;19:401-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 28. | de Freitas LM, Calixto GM, Chorilli M, Giusti JS, Bagnato VS, Soukos NS, Amiji MM, Fontana CR. Polymeric Nanoparticle-Based Photodynamic Therapy for Chronic Periodontitis in Vivo. Int J Mol Sci. 2016;17:769. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 74] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 29. | Antczak-Bouckoms AA, Tulloch JF, Berkey CS. Split-mouth and cross-over designs in dental research. J Clin Periodontol. 1990;17:446-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 74] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 30. | Singh S, Vandana KL. Stent as an accessory tool in periodontal measurements: An insight. J Indian Soc Periodontol. 2019;23:81-84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 31. | Sukumar K, Tadepalli A, Parthasarathy H, Ponnaiyan D. Evaluation of combined efficacy of photodynamic therapy using indocyanine green photosensitizer and non-surgical periodontal therapy on clinical and microbial parameters in the management of chronic periodontitis subjects: A randomized split-mouth design. Photodiagnosis Photodyn Ther. 2020;31:101949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 32. | Braun A, Dehn C, Krause F, Jepsen S. Short-term clinical effects of adjunctive antimicrobial photodynamic therapy in periodontal treatment: a randomized clinical trial. J Clin Periodontol. 2008;35:877-884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 181] [Article Influence: 10.1] [Reference Citation Analysis (1)] |
| 33. | Gomes AJ, Lunardi LO, Marchetti JM, Lunardi CN, Tedesco AC. Indocyanine green nanoparticles useful for photomedicine. Photomed Laser Surg. 2006;24:514-521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 77] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 34. | Saxena V, Sadoqi M, Shao J. Degradation kinetics of indocyanine green in aqueous solution. J Pharm Sci. 2003;92:2090-2097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 258] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 35. | Holzer W, Mauerer M, Penzkofer A, Szeimies RM, Abels C, Landthaler M, Bäumler W. Photostability and thermal stability of indocyanine green. J Photochem Photobiol B. 1998;47:155-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 142] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 36. | Panyam J, Labhasetwar V. Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Adv Drug Deliv Rev. 2003;55:329-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2668] [Cited by in RCA: 1910] [Article Influence: 83.0] [Reference Citation Analysis (1)] |
| 37. | Naqvi S, Mohiyuddin S, Gopinath P. Niclosamide loaded biodegradable chitosan nanocargoes: an in vitro study for potential application in cancer therapy. R Soc Open Sci. 2017;4:170611. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 38. | Srikanth K, Chandra RV, Reddy AA, Reddy BH, Reddy C, Naveen A. Effect of a single session of antimicrobial photodynamic therapy using indocyanine green in the treatment of chronic periodontitis: a randomized controlled pilot trial. Quintessence Int. 2015;46:391-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 39. | Monzavi A, Chinipardaz Z, Mousavi M, Fekrazad R, Moslemi N, Azaripour A, Bagherpasand O, Chiniforush N. Antimicrobial photodynamic therapy using diode laser activated indocyanine green as an adjunct in the treatment of chronic periodontitis: A randomized clinical trial. Photodiagnosis Photodyn Ther. 2016;14:93-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 89] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 40. | Polansky R, Haas M, Heschl A, Wimmer G. Clinical effectiveness of photodynamic therapy in the treatment of periodontitis. J Clin Periodontol. 2009;36:575-580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 92] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 41. | Ge L, Shu R, Li Y, Li C, Luo L, Song Z, Xie Y, Liu D. Adjunctive effect of photodynamic therapy to scaling and root planing in the treatment of chronic periodontitis. Photomed Laser Surg. 2011;29:33-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 42. | Segarra-Vidal M, Guerra-Ojeda S, Vallés LS, López-Roldán A, Mauricio MD, Aldasoro M, Alpiste-Illueca F, Vila JM. Effects of photodynamic therapy in periodontal treatment: A randomized, controlled clinical trial. J Clin Periodontol. 2017;44:915-925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 43. | Tabenski L, Moder D, Cieplik F, Schenke F, Hiller KA, Buchalla W, Schmalz G, Christgau M. Antimicrobial photodynamic therapy vs. local minocycline in addition to non-surgical therapy of deep periodontal pockets: a controlled randomized clinical trial. Clin Oral Investig. 2017;21:2253-2264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 44. | Annunziata M, Donnarumma G, Guida A, Nastri L, Persico G, Fusco A, Sanz-Sánchez I, Guida L. Clinical and microbiological efficacy of indocyanine green-based antimicrobial photodynamic therapy as an adjunct to non-surgical treatment of periodontitis: a randomized controlled clinical trial. Clin Oral Investig. 2023;27:2385-2394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 45. | Alwaeli HA, Al-Khateeb SN, Al-Sadi A. Long-term clinical effect of adjunctive antimicrobial photodynamic therapy in periodontal treatment: a randomized clinical trial. Lasers Med Sci. 2015;30:801-807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 46. | Sigusch B, Beier M, Klinger G, Pfister W, Glockmann E. A 2-step non-surgical procedure and systemic antibiotics in the treatment of rapidly progressive periodontitis. J Periodontol. 2001;72:275-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 86] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 47. | Sethi KS, Raut CP. Antimicrobial photodynamic therapy using indocyanine green as a photosensitizer in treatment of chronic periodontitis: A clinico-microbial study. Indian J Dent Res. 2019;30:870-876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 48. | Lulic M, Leiggener Görög I, Salvi GE, Ramseier CA, Mattheos N, Lang NP. One-year outcomes of repeated adjunctive photodynamic therapy during periodontal maintenance: a proof-of-principle randomized-controlled clinical trial. J Clin Periodontol. 2009;36:661-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 120] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 49. | Giannelli M, Formigli L, Lorenzini L, Bani D. Combined photoablative and photodynamic diode laser therapy as an adjunct to non-surgical periodontal treatment: a randomized split-mouth clinical trial. J Clin Periodontol. 2012;39:962-970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 50. | Feng W, Khattak YR, Majeed AA, Shah Z, Hasan US, Ahmad I. Photodynamic therapy for the treatment of grade B periodontitis: a systematic review of randomized controlled trials. Lasers Med Sci. 2025;40:327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 51. | Bassir SH, Moslemi N, Jamali R, Mashmouly S, Fekrazad R, Chiniforush N, Shamshiri AR, Nowzari H. Photoactivated disinfection using light-emitting diode as an adjunct in the management of chronic periodontitis: a pilot double-blind split-mouth randomized clinical trial. J Clin Periodontol. 2013;40:65-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 54] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 52. | Rühling A, Fanghänel J, Houshmand M, Kuhr A, Meisel P, Schwahn C, Kocher T. Photodynamic therapy of persistent pockets in maintenance patients-a clinical study. Clin Oral Investig. 2010;14:637-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 57] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 53. | Betsy J, Prasanth CS, Baiju KV, Prasanthila J, Subhash N. Efficacy of antimicrobial photodynamic therapy in the management of chronic periodontitis: a randomized controlled clinical trial. J Clin Periodontol. 2014;41:573-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 101] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 54. | Sayar F, Karimi MR, Boroujerdi S. Efficacy of antimicrobial photodynamic therapy with chitosan nanoparticles for decontamination of dental implants contaminated with Aggregatibacter actinomycetemcomitans. Sci Rep. 2025;15:34213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 55. | Petelin M, Perkič K, Seme K, Gašpirc B. Effect of repeated adjunctive antimicrobial photodynamic therapy on subgingival periodontal pathogens in the treatment of chronic periodontitis. Lasers Med Sci. 2015;30:1647-1656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 56. | Morrison EC, Ramfjord SP, Hill RW. Short-term effects of initial, nonsurgical periodontal treatment (hygienic phase). J Clin Periodontol. 1980;7:199-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 158] [Article Influence: 3.4] [Reference Citation Analysis (1)] |