Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.117024
Revised: December 23, 2025
Accepted: February 12, 2026
Published online: March 20, 2026
Processing time: 108 Days and 21.2 Hours
Very low-density lipoprotein (VLDL), released in the liver, is the only lipoprotein that includes apolipoprotein B (marker for cardiovascular risk), triglycerides, and cholesterol. VLDL is essential in transporting lipids and cholesterol to organs and cells for utilization. VLDL also contributes significantly to the advancement of atherosclerotic heart disease. We comprehensively summarize VLDL’s physio
Core Tip: The liver secretes very low-density lipoprotein (VLDL), implicated in diseases such as atherosclerosis, metabolic-associated fatty liver disease, cognitive impairment, metabolic syndrome, autoimmune disorders, breast cancer, and head and neck cancer. VLDL contributes to atrial myopathy in the preclinical stage of atrial fibrillation and exhibits cytotoxic effects in myocardial infarction, though the mechanism remains unclear. Endocrine disorders can alter VLDL levels, and VLDL has been shown to increase aldosterone production. Its pathogenic role extends to extrahepatic release, neurological disorders, sleep disturbances, and various malignancies. Understanding VLDL regulation and metabolism, rather than just its levels, may clarify its role in disease and guide targeted therapeutic strategies.
- Citation: Bharadwaj A, Taneja M, Dubey S, Saxena A. Very low-density lipoprotein and the human health. World J Exp Med 2026; 16(1): 117024
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/117024.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.117024
Very low-density lipoprotein (VLDL) is synthesized and secreted by the liver and is the precursor of intermediate-density lipoprotein (IDL), which is further metabolized to low-density lipoprotein (LDL). Lipoproteins such as chylomicrons, VLDL, IDL, LDL, and high-density lipoprotein (HDL) can be separated from blood or plasma using density-gradient ultracentrifugation techniques[1,2]. VLDL is predominantly composed of triglycerides (TGs; 50%-70%), while cholesterol constitutes approximately 10%-25%, and free fatty acids account for less than 10%. The principal structural protein of VLDL is apolipoprotein (apo) B100, along with exchangeable apos including apo E, apo C-I, apo C-II, and apo C-III. These apos act as ligands that regulate lipolysis and mediate interactions with cell-surface receptors[3]. VLDL transports TGs, cholesterol, and associated proteins from the liver to peripheral tissues to support essential metabolic functions. Hepatic availability of TGs and cholesterol, together with apo B100 synthesis, strongly influences the lipid composition and rate of VLDL production[4].
In the circulation, VLDL is hydrolyzed by lipoprotein lipase (LPL) located on the capillary endothelium, resulting in the removal of TGs and the formation of VLDL remnants, also known as IDL. Some VLDL particles may remain partially unmodified. During this process, apo C-II and other surface components are transferred to HDL. Cholesteryl ester transfer protein facilitates the exchange of TGs and cholesteryl esters between VLDL/IDL and HDL. IDL is subsequently taken up by the liver via LDL receptors or further metabolized into LDL following the loss of TGs and apo E[3]. The assembly and metabolism of VLDL are strongly influenced by insulin resistance and chronic nutrient excess, conditions commonly associated with metabolic disorders[5]. Beyond lipid transport, VLDL has been implicated in nitric oxide signaling, which plays a key role in vascular tone regulation and blood pressure homeostasis[6]. Additionally, VLDL has been shown to stimulate phospholipase D activity in the adrenal gland, leading to increased cytosolic calcium levels and enhanced aldosterone secretion[7]. Therefore, VLDL plays a multifaceted role not only in lipid transport but also in metabolic regulation and vascular function.
Hormonal regulation and endocrine disorders play a central role in modulating VLDL secretion and TG metabolism. Insulin is the primary inhibitory hormone regulating hepatic VLDL-TG production by suppressing adipose tissue lipolysis and limiting free fatty acid flux to the liver; insulin resistance therefore leads to VLDL overproduction and hypertriglyceridemia. Counter-regulatory hormones such as glucagon, cortisol, and catecholamines promote lipolysis and hepatic TG synthesis, indirectly enhancing VLDL secretion. Thyroid hormones influence TG clearance by regulating LPL activity and hepatic lipid oxidation, with hypothyroidism commonly associated with elevated VLDL-TG levels. Sex hormones also modulate VLDL metabolism, as estrogens generally reduce hepatic VLDL secretion, whereas androgen excess may exacerbate hypertriglyceridemia. Consequently, endocrine disorders including diabetes mellitus, metabolic syndrome (MetS), Cushing’s syndrome, hypothyroidism, and polycystic ovary syndrome are frequently characterized by dysregulated VLDL-TG metabolism, linking hormonal imbalance to cardiometabolic disease risk.
This review explores the hypothesis that dysregulated VLDL metabolism serves as a central node connecting diverse metabolic stressors to multi-organ pathogenesis, highlighting novel experimental insights into underlying mechanisms and potential therapeutic targets.
Nuclear magnetic resonance spectroscopy measures VLDL particle diameter. VLDL can be classified based on particle size, obtaining different fractions of diameters. High reproducibility is vital for measuring serum lipoproteins sub-fractions. To evaluate the quality control of plasma of lipoprotein samples, reproducibility was complied with, and nuclear magnetic resonance results from 5 laboratories and 11 spectrometers were compared. Subclasses have been defined: 6 for LDL, 4 for HDL, and 6 for VLDL[8].
However, an agreement has not been reached on consensus standard diameter ranges for classifying VLDL subfractions. For example, Phillips and Perry[9], 2015 categories are defined as small VLDL (< 42 nm), medium VLDL (42-60 nm) and large VLDL (including chylomicrons, if present, > 60 nm). Wang et al[10], 2012 explain the 6 classes of VLDL as follows: Small (36.8 nm), very small (31.3 nm), medium (44.5 nm), most significant chylomicrons (mean diameter, 64.0 nm). Garvey et al[11] identify the following three groups: Small VLDL (< 35 nm), medium VLDL (35-60 nm), and large VLDL (> 60 nm). Avogaro et al[12], 1988 identified the LDL for the first time based on surface charge but not on the size of solid exchange chromatography and divided it into LDL (-) and LDL (+) in 1988. In addition, Yang et al[13], 2003 and Chen et al[14], 2003 have classified them into five categories from L1-L5. Similarly, an attempt was made by Chen et al[15], 2012 to classify the VLDL into five subcategories from V1-V5 using anion exchange chromatography (Table 1).
| Classes | Associated impact | Ref. |
| Small VLDL | Metabolically fit individuals with smaller (below medium) VLDL size | [9] |
| Largest VLDL (including chylomicrons) and five different VLDL subclasses | The level of all lipid components in the VLDL subclasses was enhanced as glucose tolerance was reduced | [10] |
| Large and intermediate VLDL | Progressive insulin resistance was associated with enhanced VLDL size and an enhancement in large VLDL particles | [11] |
Although VLDL and the VLDL receptor (VLDLR) share nomenclature, they represent functionally and biologically distinct entities. VLDL particles act as lipid transport vehicles and signaling mediators, whereas VLDLR is a cell-surface endocytic and signaling receptor that governs tissue-specific lipid uptake and intracellular signaling pathways. Conflation of their roles has led to ambiguity in mechanistic interpretations, particularly in cardiometabolic and inflammatory diseases.
Nature: Circulating TG-rich lipoproteins synthesized and secreted by hepatocytes.
Primary function: Transport of endogenous TGs, cholesterol esters, and fat-soluble molecules to peripheral tissues.
Key apos: Apo B100 (structural), apo E, apo C-II, apo C-III.
Pathophysiological effects: Promote atherogenesis, endothelial dysfunction, lipotoxicity, inflammation, insulin resistance, and ectopic lipid deposition.
Mode of action: Systemic - via lipolysis by LPL, remnant formation, oxidative modification, and receptor-independent cellular effects.
Nature: Transmembrane member of the LDL receptor family.
Primary function: Tissue-specific uptake of TG-rich lipoproteins and modulation of intracellular signaling.
Expression profile: High in adipose tissue, heart, skeletal muscle, macrophages, brain, and endothelial cells; absent in adult hepatocytes.
Pathophysiological effects: Regulates lipid accumulation, cellular energy balance, inflammation, angiogenesis, neurodevelopment, and vascular remodeling.
Mode of action: Local - via receptor-mediated endocytosis and signal transduction pathways.
Metabolic-associated fatty liver disease (MAFLD) exhibits a raised VLDL secretion rate attributed to increased intrahepatic TGs hydrolysis. They apparent the absence of immediate VLDL secretion reduction yet maintained a consistent apo B100 secretion rate, as informed by previous studies[16,17]. The power to suppress insulin results on VLDL is compromised in males with MAFLD in terms of particle oxidation, concentration, secretion, and the decrease in particle size due to oxidation[18]. In individuals with insulin resistance and higher body weight, there is an elevation in apo C-III levels within VLDL. This increase enhances hepatocyte VLDL uptake[19-21]. However, in MAFLD patients with a severe TG/VLDL ratio, plasma TG levels, liver fibrosis, and total circulating VLDL were significantly reduced[22]. VLDL showed significant gender differentiation as far as MAFLD is concerned. Men are more likely to develop MAFLD than women[23]. The risk of MAFLD is increased due to decreased estrogen levels in postmenopausal women[24-26]. Recent animal experimentation has reflected that insufficiency of estrogen-related receptor results in the decreased production of VLDL, which causes increased lipid concentration and MAFLD progression. Estrogen-related receptor ERR is a nuclear hormone involved in various metabolic activities[23].
Decreased adiponectin levels (usually seen in patients with MetS and MAFLD) lead to a rise in VLDL size mass (Lucero et al[22], 2017). Activated protein kinase and mammalian target of rapamycin were the two molecules that were concerned with the expression of VLDL and MAFLD[27-29]. It has been found that ceramides (lipid portion among VLDL) are also associated with the MAFLD severity[30,31]. VLDL is also responsible for transporting plasma dihydroceramide (i.e., also connected to MAFLD exposure during type 2 diabetes)[31,32].
Hepatitis C virus (HCV) attaches to the TG-rich lipoproteins and VLDL, synthesizing lipoviral particles. A classic example is lipoviral particles that contain HCV glycoprotein E2, which has the capability to guard HCV against the immune system as well as help in spreading the pathogenicity of lipoprotein receptors inside the liver[33-35]. It has been noticed that HCV viral load negatively impacts LPL activity, while it reflects a positive impact with VLDL (apo C-III portion)[36]. Furthermore, HCV interferes with the host’s lipid metabolism through various mechanisms like reducing fatty acid oxidation, increasing lipogenesis, and reducing the amount of TG in secreted VLDL, thereby affecting lipid homeostasis[37]. Direct-acting antivirals that directly affect HCV have been shown to correct TG abnormalities in cholesterol and lipoprotein metabolism in VLDL and LDL[38].
Insulin-hampered VLDL production, along with insulin resistance, leads to increased and decreased production of VLDL, often associated with hypertriglyceridemia[39-42]. Hepatic VLDL production is decreased by glucagon[43]. Due to insulin resistance, adipose tissue reflects low lipid concentration (i.e., responsible for the accumulation of ectopic lipids and hyperlipidemia)[44,45]. It has been observed that insulin has the capability to reduce VLDL production[46] among individuals suffering from type 2 diabetes. However, postprandial VLDL concentrations are enhanced, but postprandial VLDL clearance is like nondiabetic subjects[47]. A study examining VLDL kinetics during hyperinsulinemia using pre-labeled VLDL1 and VLDL2 particles showed that the size (TG/apo B ratio) and apo B100 level were reduced in a healthy individual. The rate of fatty acid oxidation is reduced in diabetic patients[48]. Emerging evidence indicates that VLDL is cytotoxic, proinflammatory, and atherogenic in MetS and is linked with many infections[49-54]. Enhanced levels of apo C-III, apo E-III, and apo E-IV among VLDL in MetS are responsible for decreased LPL concentration[55,56]. VLDL in MetS can encourage apoptosis via reactive oxygen species, especially in endothelial cells and subendothelial macrophages[55]. Moreover, proinflammatory impacts have been shown to be stimulated by VLDL lipolysis products[57]. VLDL size is thought to be related to the regulation of peroxisome proliferator activated receptor-α during plasma fatty acid clearance after VLDL hydrolysis[58]. The size of VLDL particles has been affected through the LPL activity and relates to MetS, insulin resistance, and VLDL particle size[59].
This section elaborates on various hormones and endocrine-related disorders concerned with VLDL/TG metabolic pathways (Table 2).
| Disorder/syndrome | Impact due to VLDL | Ref. |
| Cushing syndrome | Enhanced production | [61] |
| Exogenous cortisol | Decrease in degradation and enhanced adipose lipolysis | [62] |
| Aldosterone | Stimulation of aldosterone secretion | [65-67] |
| Growth hormone deficiency | Enhanced secretion and decline in clearance | [61] |
| Growth hormone treatment | Increased adipose lipolysis and increased clearance | [61] |
| Hypothyroidism | Decreased degradation with enhanced secretion | [73,74] |
| Androgen | Androgen-deprivation treatment: Enhanced concentration; transgender males with testosterone treatment: Enhanced concentration | [80-82] |
| Polycystic ovary syndrome | Enhanced concentration | [84] |
| Estrogen/progesterone therapy | Enhanced concentration | [85,86] |
| Prolactinoma | Uncertain | [94-96] |
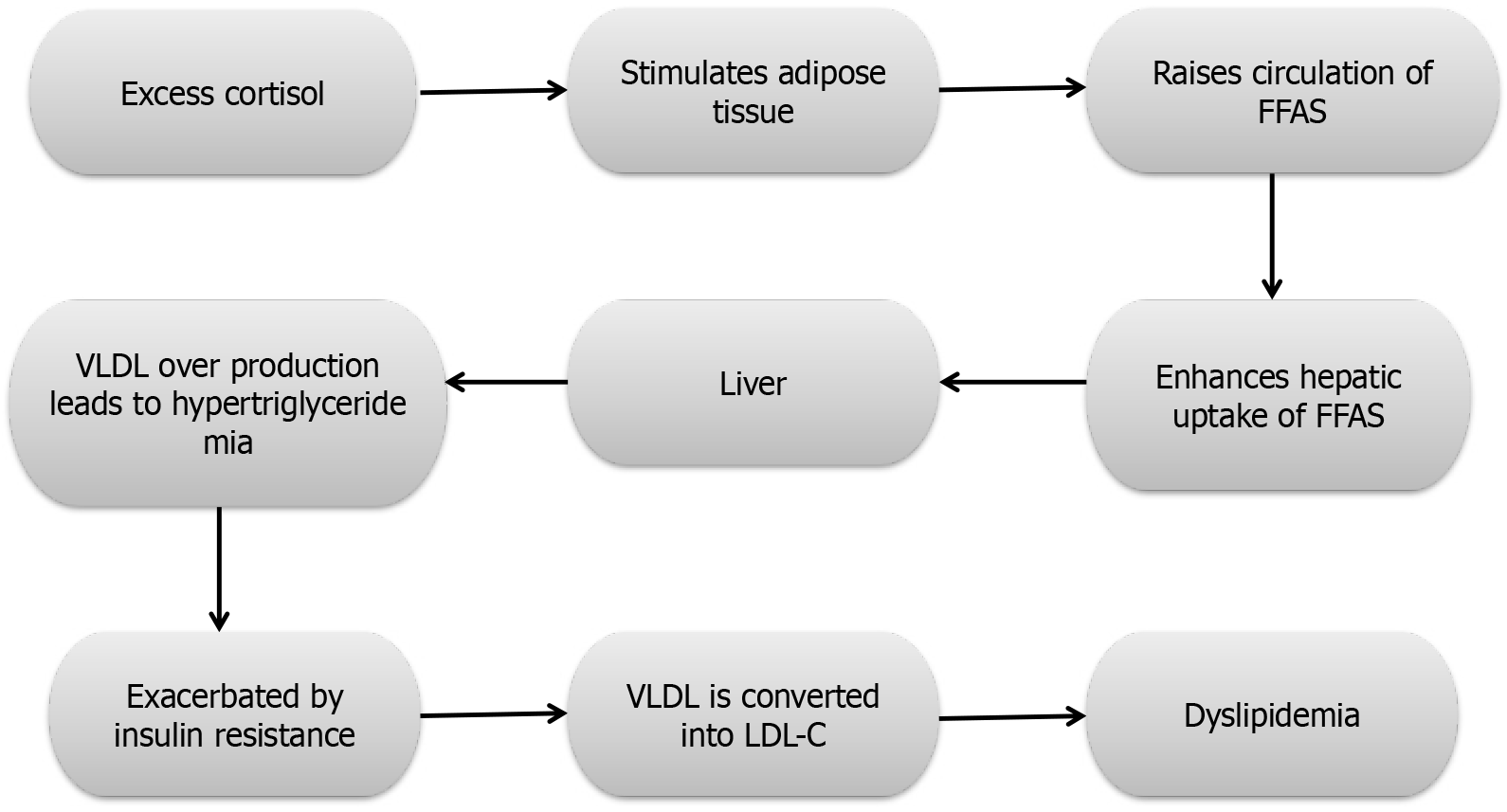
Cushing syndrome: This syndrome occurs when the enhanced concentration of LDL and VLDL manifests as dyslipidemia along with increased levels of plasma cholesteryl ester and TG[60]. Enhanced release of VLDL in comparison to the usual amount with no change in VLDL clearance is responsible for the high VLDL concentration[61]. Figure 1 illustrates how excess cortisol drives dysregulated VLDL-TG metabolism. Elevated cortisol stimulates adipose tissue lipolysis, increasing the circulation of free fatty acids. These free fatty acids are taken up by the liver, where they fuel hepatic TG synthesis and VLDL overproduction, leading to hypertriglyceridemia. This process is further exacerbated by insulin resistance, which impairs normal suppression of VLDL secretion. Circulating VLDL is subsequently converted into LDL-cholesterol, contributing to atherogenic dyslipidemia. Overall, the pathway links cortisol excess to insulin resistance, VLDL overproduction, and adverse lipid profiles.
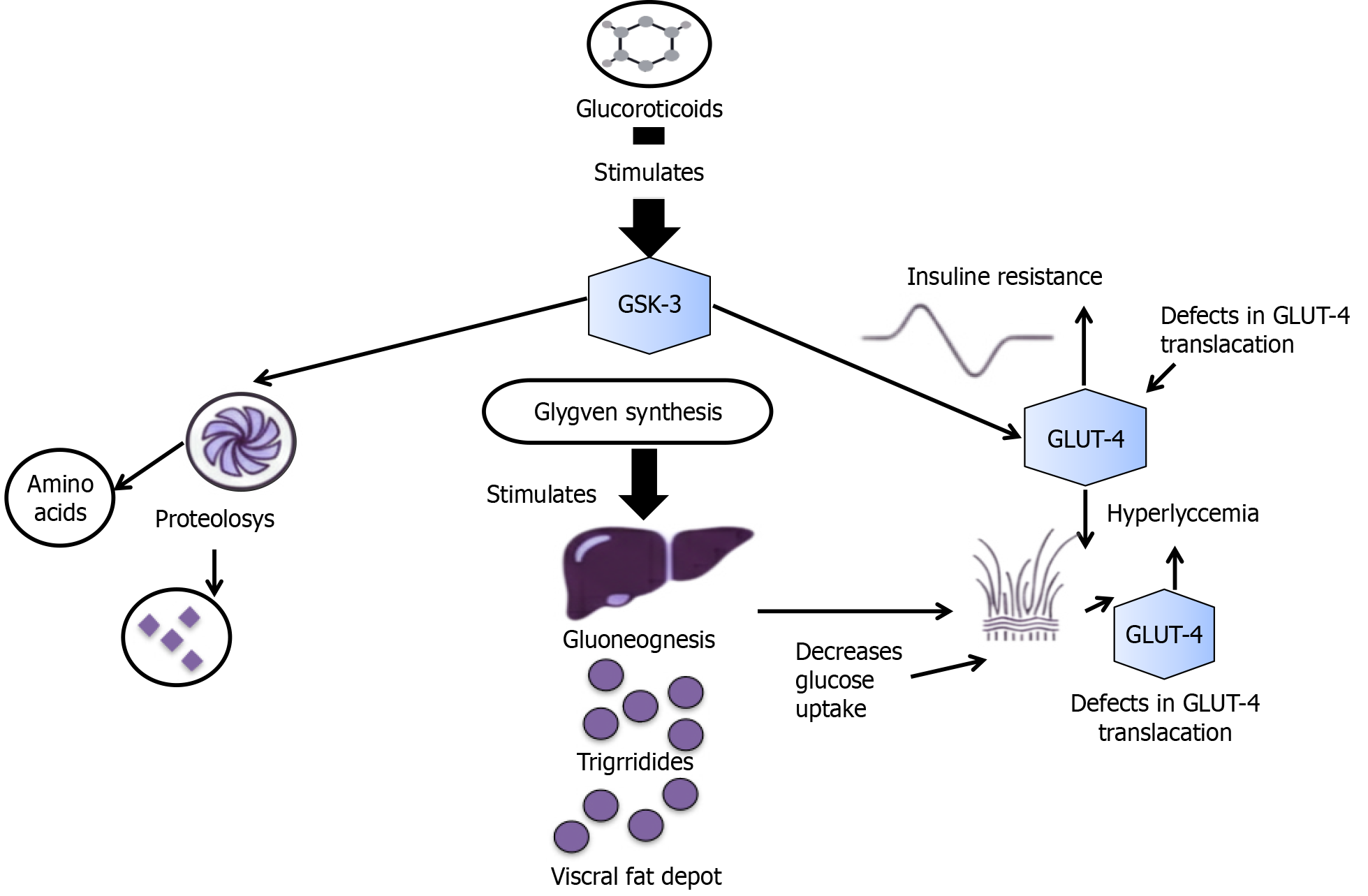
Exogenous cortisol: Stimulation of exogenous cortisol during glucocorticoid therapy decreases the apo B degradation and enhances adipose tissue lipolysis, ensuring increased VLDL[62]. Risk of cardiovascular sickness along with Cushing’s syndrome and dyslipidemia on corticosteroid therapy are associated with increased VLDL/TG levels[63,64]. Figure 2 depicts the metabolic effects of glucocorticoid excess on glucose and lipid homeostasis. Glucocorticoids activate glycogen synthase kinase-3, which inhibits glycogen synthesis and promotes hepatic gluconeogenesis, increasing glucose output. Concurrently, glucocorticoids induce insulin resistance by impairing glucose transporters type 4 translocation in skeletal muscle, reducing peripheral glucose uptake and leading to hyperglycemia. In parallel, glucocorticoid-stimulated proteolysis increases circulating amino acids that further fuel gluconeogenesis. Excess glucose is diverted toward TG synthesis and visceral fat accumulation, linking glucocorticoid signaling to insulin resistance, hyperglycemia, and central adiposity.
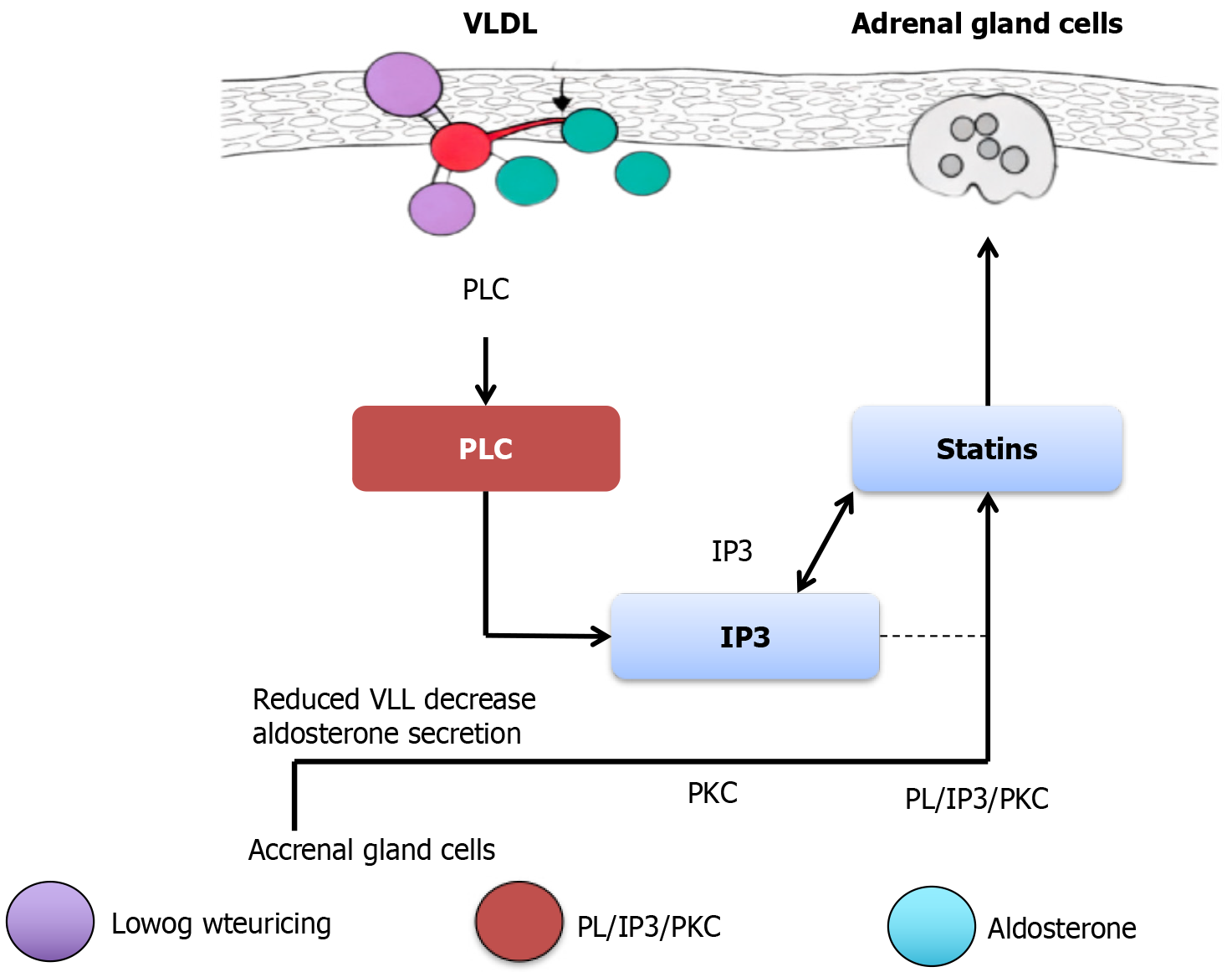
Aldosterone: Aldosterone, secreted from the adrenal gland, is also related to VLDL. Stimulation in VLDL endorses the secretion of aldosterone, which occurs through the phospholipase C (PLC)/inositol 1,4,5-trisphosphate (IP3)/protein kinase C (PKC) signaling pathway[65-67]. Through this mechanism, we can partly clarify how statins, a commonly used lipid-lowering drug, are associated with low aldosterone levels in high blood pressure and diabetes[68]. Figure 3 illustrates a signaling link between VLDL and aldosterone secretion in adrenal gland cells. VLDL interacts with adrenal cell surface receptors, activating the PLC pathway, which generates IP3 and activates PKC, leading to intracellular signaling that promotes aldosterone production. Statins interfere with this pathway by reducing circulating VLDL levels and attenuating PLC-IP3-PKC signaling, thereby decreasing aldosterone secretion. Overall, the diagram highlights a mechanistic connection between lipid metabolism and adrenal hormone regulation, suggesting that VLDL reduction can modulate aldosterone-dependent endocrine effects.
Growth hormone deficiency and treatment: Growth hormone insufficiency is associated with increased VLDL synthesis, reduced VLDL clearance, and increased TG concentration. VLDL clearance is promoted through growth hormone replacement treatment but concurrently enhances the VLDL release from adipose tissue by promoting lipolysis[69]. As a result, even if growth hormones encourage fatty acid oxidation, there is no decrease in VLDL production. However, it can also enhance plasma VLDL and TG concentration[70]. This incidence may clarify why patients with hypopituitarism may develop cardiovascular and cerebrovascular diseases[71]. Another form of acromegaly is associated with excess growth hormone causing LPL activity, increased plasma non-esterified fatty acids, and excess liver VLDL[72].
Hypothyroidism: It has been found that lipoprotein metabolism, LPL function, and cholesterol activity are affected by thyroid hormones. LPL activity decreases, and hepatic VLDL secretion increases during hypothyroidism[73,74]. Hypothyroidism develops when the level of thyroid stimulating hormone > 10 mIU/L and is responsible for heart and blood-related infections and dyslipidemia, like an increase in total LDL, cholesterol, TG, and low HDL. However, the meta-analysis did not reflect considerable differentiation among VLDL, apo A-I, or apo B levels[75]. Thyromimetic drugs (thyroid regulators) can increase energy disbursement and lipid-lowering by improving the lipid profile[76].
Androgen: As far as sex hormones are concerned, VLDL metabolism is not affected by testosterone[77,78]. On the other hand, a deficiency of androgen results in the rise of VLDL levels[79]. It has been found that patients undergoing testosterone treatment along with androgen deprivation showed partial impact over VLDL levels. However, an increase in VLDL/TG was observed after testosterone treatment in transgenic men, possibly due to the estrogen combination[80-82].
Polycystic ovary syndrome: Enhanced VLDL concentration is typical in patients with polycystic ovary syndrome and may resolve after successful treatment of polycystic ovary syndrome (polycystic ovary syndrome). It reflects no impact on remaining lipid profiles[83,84].
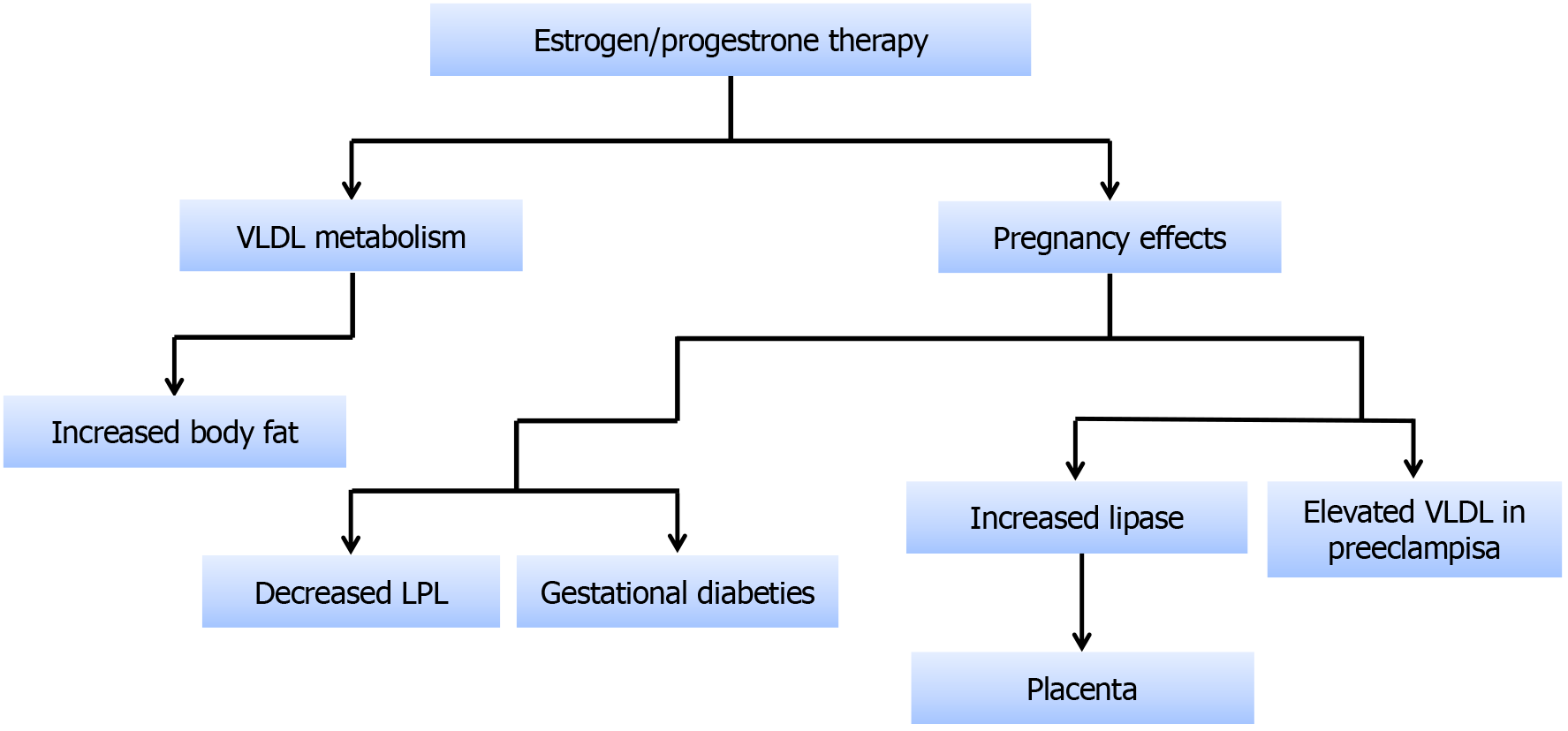
Estrogen/progesterone therapy: VLDL metabolism is affected through hormone replacement therapy, and it is still disputed. Several findings have shown that due to estrogen supplementation, the VLDL and body fat mass have increased[85], and similar results were obtained with progesterone remedy[86]. For adult females of childbearing age, an increased risk of heart-related issues has been observed with increased estrogen in addition to oral contraceptives alone[87]. Increased VLDL concentration in hormone replacement therapy has also been noted and is found to be related to high cardiac hazard (Figure 4)[88].
VLDL concentration is elevated in pregnant women due to decreased LPL and high lipase activity[89,90]. Hormone-sensitive lipase in adipose tissue also promotes VLDL production during pregnancy[91]. Mother and placenta get energy from fatty acids and cholesterol through VLDL[92]. VLDL levels also increase in pregnant women with gestational diabetes and preeclampsia due to increased insulin resistance[91,93].
Prolactinoma: Elevated LDL may cause Prolactinomas, though the impact of prolactin on VLDL/TG is still unclear[94-97]. The usual prolactinoma treatment is dopamine agonist therapy, which improves insulin sensitivity, lowers LDL cholesterol and improves body mass index[75]. Various animal studies have shown that the circadian rhythm affects lipid metabolism, suggesting the underlying mechanism is changes in the gut microbiota[98-100].
Experimental studies collectively indicate that hormonal regulation of VLDL-TG metabolism converges on a limited set of shared mechanistic nodes rather than hormone-specific pathways. A central mechanism is the control of adipose tissue lipolysis and hepatic free fatty acid flux, whereby glucocorticoids, catecholamines, and insulin resistance enhance free fatty acid release, providing substrate for hepatic TG synthesis and VLDL overproduction. At the hepatic level, hormones modulate intracellular signaling pathways (e.g., PLC-IP3-PKC, glycogen synthase kinase-3, and insulin-protein kinase B signaling) that regulate lipogenesis, apo B stability, and VLDL assembly. Concurrently, impaired insulin signaling and glucose transporters type 4 translocation in peripheral tissues reduces glucose uptake, indirectly diverting excess carbon toward TG synthesis and visceral fat accumulation. Hormonal effects also converge on lipoprotein clearance mechanisms, particularly through regulation of LPL activity and VLDL-to-LDL conversion, shaping circulating lipid profiles. Col
The effects of VLDL on cardiovascular infection are reflected through the connection between atherosclerosis and coronary events[101], as it is directly linked with carotid intima-media thickness and arterial stiffness[102]. Current research reflected that plasma VLDL concentration is positively associated with the severity of symptoms in peripheral arterial occlusive infection[103,104]. The addition of TG-rich lipoproteins results in the rupturing of atherosclerotic plaque. A massive study from China reflects a connection between increased VLDL concentrations and heart disease[105]. VLDL concentration is also associated with enhanced mortality among patients with coronary heart disease[106]. Increasing VLDL levels enhance blood viscosity, increasing microvascular events in type 2 diabetes[107]. Other potential mechanisms include proatherogenic effects and hypercoagulable states that promote thromboembolism[108,109].
The apo compounds influence the atherogenic effects of VLDL. It has become evident through the clinical analysis that cardiovascular risk relates to the increased apo B and decreased apo C-III in VLDL[110]. The main component of atherosclerosis development is apo B. In addition, apo C-III loss-of-function mutations are associated with a reduced risk of cardiovascular disease[111]. However, the effect of apo C-III on VLDL is problematic because it inhibits the interaction of VLDL with its receptor. Moreover, apo B, apo C, and apo E can act as ligands for receptors on macrophages to promote cell swelling and inflammatory processes, thereby triggering atherosclerosis[112-114].
The expression of VLDLR is very high in the peripheral nervous system, cerebral cortex, cerebellum, and cerebral cortex. Clinical studies have shown that VLDLR is implicated in Alzheimer’s disease[115-117]. Moreover, the senile plaques of the brain have been the prime site for VLDLR detection[118]. Recent studies have shown that VLDLR can interact with various ligands and molecules associated with Alzheimer’s disease, such as reelin and clusterin. While reelin depletion is considered an early event in Alzheimer’s disease[119,120], clusterin promotes the removal of amyloid-β[121,122]. At present, VLDLR, but not VLDL, is thought to influence the commencement and development of Alzheimer’s disease.
There is some confirmation that VLDL plays a role in mental illness. The data show the association between insulin resistance, increased VLDL concentration[123], and enhanced medium and large VLDL concentrations in patients with schizophrenia[124,125]. In addition, the risk of suicide and cognitive impairment is also connected with the increased TG/VLDL ratio[126,127]. A decrease in VLDL and apo B levels was observed with the increase of fatty acids during autism spectrum disorders[128]. Insufficient sleep can affect VLDL metabolism. Poor daytime activity due to poor sleep, use of sleeping pills, and poor sleep have been associated with increased VLDL levels in clinical studies[129]. This study can link cardiovascular risk and poor sleep[130].
The patients having chronic kidney disease with dyslipidemia were generally affected with hypertriglyceridemia[131], mainly due to impaired VLDL clearance[132]. During chronic kidney disease, the hydrolysis of VLDL gets impaired, and HDL levels decrease. Increased concentration of VLDL results in oxidative stress in chronic kidney disease[133]. In addition, plasma apo C-III in the chronic kidney disease group was higher than in the average population, resulting in increased insulin resistance and hyperglycemia[134]. Decreased concentration of VLDL is responsible for nephrotic syndrome. An animal study showed that the mechanism was the inhibition of VLDLR[135], inhibiting LPL activity due to increased levels of angiopoietin-like protein 4[136,137]. Moreover, VLDL can be absorbed by mesangial cells and exert cytotoxicity, leading to the development of nephrotic syndrome.
In general, MetS is regarded as a chronic disease condition, and VLDL affects the microinflammation of endothelial cells, activation of monocytes in extrahepatic tissue, and cytokines expression[138,139].
During MetS and insulin resistance, ceramide expression in blood plasma is enhanced[140,141], and excessive consumption of VLDL lipolysis products increases the activity of macrophages[142-144]. Upregulation of intracellular ceramides occurs in macrophages and shows a proinflammatory response after incubation with VLDL[57]. On the other hand, the VLDL concentration can be enhanced by specific cytokines like interleukin (IL)-1, IL-2, and IL-6[145]. It has been noticed that when these ILs encounter lipopolysaccharides, the VLDL concentrations are enhanced within 2 hours and remain constant for 24 hours[146].
In several autoimmune diseases, the VLDL concentration is increased. Similar results were also obtained with the patients suffering from systemic lupus erythematosus[147,148]. Moreover, females suffering from systemic lupus erythematosus were more prone to cardiovascular disorders related to dyslipidemia and irregular VLDL expression[149]. By regulating the action of apo C-III, there is a reduction in VLDL clearance and VLDL concentration enhanced among patients suffering from antiphospholipid syndrome[150]. It has been noticed that VLDL concentration among MetS patients is positively linked with rheumatoid arthritis[151]. Steroids are found to be the most potent therapeutic agent for individuals having autoimmune diseases, and they also enhance the plasma VLDL concentration[152]. Because VLDL can cause inflammation, monitoring lipids, including VLDL concentrations, is essential during disease management among autoimmune infections. VLDL was also found to affect skin infections. Due to the oxidative stress in melanocytes, the issue related to hyper-pigmentation occurs and is called vitiligo, usually at increased TG concentration[153]. MetS is found to be a poor interpreter of psoriasis. Moreover, it has been noticed that enhanced VLDL concentration was found among psoriasis individuals along with MetS in comparison to psoriasis individuals without MetS[154].
The development and progression of several malignancies are frequently accompanied by dyslipidemia[155]. Similar to normal cells, lipid-regulated cellular and intracellular signaling in tumor cells can modulate membrane fluidity and lipid raft organization, thereby influencing lipid-derived signaling mediators. These lipid-driven alterations play critical roles in cancer biology, including tumor cell invasion, metastasis, and immune evasion[156,157]. Recent studies have identified CD36, a well-characterized fatty acid translocase, as a key regulator of cancer metastatic potential, with emerging evi
Table 3 summarizes how strong the experimental evidence is for VLDL/VLDLR involvement across different cancers: Hepatocellular carcinoma (HCC): Direct functional experiments show that knocking down VLDLR in hepatoma cells reduces proliferation, clearly demonstrating that VLDLR actively supports tumor cell growth rather than being a passive marker. Clear-cell renal cell carcinoma: SiRNA studies reveal that VLDLR is required for abnormal lipid uptake in RCC cells, linking VLDLR to the lipid-rich phenotype characteristic of this cancer subtype. Breast cancer: Both receptor-level manipulation (VLDLR knockdown/overexpression) and VLDL exposure in animal models show enhanced invasion and metastasis, indicating that VLDLR-mediated lipid uptake and circulating VLDL directly promote tumor aggressiveness. Gastrointestinal cancers: Changes in VLDLR subtype expression are observed in tumor cell lines and tissues, suggesting a role in tumor differentiation; however, functional knockdown or in vivo validation is still limited. Colorectal cancer: Most evidence is correlative, with altered VLDLR expression associated with tumor features, but direct mechanistic and causal studies are still emerging.
| Cancer type | Model evidence | Mechanistic insight |
| HCC | shRNA knockdown reduces proliferation in hepatoma cells | VLDLR supports tumor cell growth |
| ccRCC | siRNA knockdown reduces lipid uptake in RCC cells | VLDLR mediates pathological lipid uptake |
| Breast cancer | VLDLR manipulation alters cancer cell behavior; VLDL increases metastasis in vivo | VLDLR/VLDL enhance tumor aggressiveness |
| Gastrointestinal cancer | Altered VLDLR subtype expression in tumor cell lines/tissues | Suggests involvement in differentiation but not fully functional yet |
| Colorectal cancer | Altered expression correlates with tumor features | Functional causality still emerging |
Breast cancer: The most critical research analysis in breast cancer has paid attention to the effects of VLDL, which has been shown to encourage cancer growth and development by facilitating cell migration, invasion, and angiogenesis. MDA-MB-231 cells, which cause breast cancer in a mouse model, are first loaded with LDL (L1 and L5) and VLDL sub-fractions and then enter the animals’ muscles. The outcomes reflected that incubation of lung tumor cells with VLDL, L5, or L1 support invasion, but VLDL incubation alone was addictive and resulted in more lung metastases. Based on these observations, we can conclude that VLDL encourages lung metastasis under in vivo conditions[160]. Ingestion of VLDL takes up lipids and provides stable energy to cancer cells[161]. Comparison of the impacts of various lipoproteins on human epidermal growth factor receptor 2 and excessive expression of breast tumor cells reflected that VLDL induces tumor cell development and morphological modification and encourages cell viability[162].
HCC: The prime risk factor for HCC is lipid deposition because of the MAFLD and/or genetic predisposition[163]. Like breast tumors, VLDL is associated to influenced the growth and development of HCC. Current animal experimentation has shown that mutations in the transmembrane 6 superfamily member 2 (TM6SF2) genes are responsible for fibrosis and support carcinogenesis in MAFLD. TM6SF2 is contained to the endoplasmic reticulum membrane and is necessary for the lipidation of apo B at the duration of VLDL synthesis. Damage to TM6SF2 leads to reduced VLDL secretion, leading to hepatic steatosis (MAFLD) and severe liver fibrosis[164,165]. In HCC cells, hypoxia-inducible factor-1 (HIF-1) is upre
Table 4 summarizes that in breast cancer; experimental models show that circulating VLDL mainly serves as an external lipid fuel. Breast cancer cells take up VLDL-derived fatty acids through CD36 and VLDLR, which supports energy production, membrane synthesis, and redox balance. This lipid supply directly enhances cell migration, invasion, cancer stem-like properties, and metastatic spread, as demonstrated in cell culture studies and mouse metastasis models. Hypoxia plays only an indirect role, mainly by increasing metabolic stress and lipid dependence rather than directly regulating VLDLRs. In HCC, VLDL functions primarily as an external lipid source that supports tumor cell survival and proliferation. A key mechanistic difference is the direct involvement of hypoxia: Stabilization of HIF-1α under low-oxygen conditions transcriptionally upregulates VLDLR, leading to increased VLDL uptake. This enhanced lipid acquisition promotes cell growth and resistance to apoptosis, as shown in HCC cell culture and hypoxia-based experimental models. Overall, while both cancers exploit VLDL to meet metabolic demands, breast cancer uses VLDL mainly to drive aggressiveness and metastasis, whereas HCC uses hypoxia-induced VLDLR expression to sustain proliferation and survival.
| Feature | Breast cancer | Hepatocellular carcinoma |
| Primary role of VLDL | External lipid fuel for growth and metastasis | External lipid source supporting survival |
| Key receptors | CD36, VLDLR | VLDLR |
| Hypoxia involvement | Indirect (metabolic stress adaptation) | Direct via HIF-1α to VLDLR |
| Demonstrated outcomes | Increased metastasis, invasion, stemness | Increased proliferation, survival |
| Experimental proof | Cell culture + mouse metastasis models | Cell culture + hypoxia models |
Other cancers: The concentration of VLDL increased among cancer patients in comparison to non-cancer patients[167]. On the other hand, the most predictive factor for cancer patients is the HDL level[168]. Yet the exact function of VLDL during lung cancer is still not clear. Two studies conducted in India have shown an association between blood VLDL concentration and leukoplakia, a predecessor to oral cancer[169,170]. The key risk factors include smoking, drinking alcohol, and eating nuts may result in oral cancer. These factors lead to the production of free radicals and further cause harm to cell membranes. Reduced VLDL concentration is thought to result from increased oxidative stress and cell membrane repair. Blood VLDL concentrations have also been shown to be the benchmark of oral cancer and early lesions. While research on VLDL and other cancers is scarce, several studies have shown an association between high TG concentration and cancer risk for ovarian cancer, pancreatic cancer, small cell lung cancer, and breast cancer. However, the fundamental mechanisms are still not precise.
Table 5 summarizes that breast cancer and HCC represent the strongest cases for a causal role of VLDL/VLDLR in cancer biology. In breast cancer, both in vitro and in vivo functional studies demonstrate that VLDL uptake and its receptors directly drive tumor growth, invasion, stemness, and metastasis, establishing a mechanistic link. Similarly, in HCC, cell culture and hypoxia-based mechanistic studies show that HIF-1α-mediated upregulation of VLDLR enhances VLDL uptake, promoting tumor cell proliferation and survival, again supporting direct causality. In contrast, evidence in oral/head and neck squamous cell carcinoma is largely associative, relying mainly on correlations between tumor VLDLR expression or circulating lipid levels and disease features, without functional validation. Ovarian cancer shows a similar pattern, where clinical correlations and expression analyses suggest a possible role for VLDL/VLDLR, but direct experimental manipulation is limited, resulting in only weak to moderate causal support. Finally, in pancreatic cancer, the proposed involvement of VLDL/VLDLR is largely speculative, inferred from general lipid metabolic dependencies rather than direct experimental evidence.
| Cancer type | Evidence type | Strength of causality |
| Breast cancer | In vitro + in vivo functional studies | Strong (mechanistic) |
| Hepatocellular carcinoma | In vitro + hypoxia-driven mechanistic studies | Strong (mechanistic) |
| Oral/HNSCC | Expression + serum lipid correlations | Weak (associative) |
| Ovarian cancer | Clinical correlations + expression data | Weak-moderate (associative) |
| Pancreatic cancer | Indirect metabolic inference | Speculative (hypothesis) |
Overall, the table highlights a clear evidence hierarchy, distinguishing cancers with demonstrated mechanistic involvement of VLDL/VLDLR from those where roles remain correlative or hypothetical and require rigorous functional validation.
Experimental studies using in vitro cell systems, animal models, and translational approaches have revealed that VLDL exerts its effects through multiple, interconnected molecular and cellular mechanisms that extend well beyond its classical role as a lipid transport particle. These effects are mediated through lipolytic products, receptor-dependent signaling, intracellular lipid accumulation, inflammatory activation, oxidative stress, and endocrine modulation.
One of the most consistently demonstrated mechanisms of VLDL action in experimental models is lipolysis-driven cytotoxicity. VLDL particles are hydrolyzed by LPL at the endothelial surface, releasing free fatty acids, monoacylglycerols, and remnant particles. In vitro models of cardiomyocytes, endothelial cells, hepatocytes, and pancreatic β-cells, excess free fatty acids derived from VLDL lipolysis induce lipotoxic stress, characterized by mitochondrial dysfunction, endoplasmic reticulum stress, and apoptosis[57].
Animal studies of insulin resistance and MetS demonstrate that chronic exposure to VLDL-derived free fatty acids promotes intracellular lipid droplet accumulation, activation of stress kinases (e.g., c-Jun N-terminal kinase, p38 mitogen-activated protein kinase), and impaired cellular metabolism. These effects are particularly evident in cardiac tissue, where VLDL lipolysis products contribute to cardiomyocyte hypertrophy, reduced contractility, and atrial structural remo
Beyond lipid delivery, VLDL interacts directly with cell-surface receptors, triggering intracellular signaling cascades. Experimental models have identified several key receptors involved in VLDL-mediated effects, including: (1) VLDLR; (2) LDL receptor; (3) LDL receptor-related protein 1; and (4) Scavenger receptors.
Binding of VLDL or its remnants to these receptors activates signaling pathways independent of lipid uptake. For instance, VLDLR-mediated signaling has been shown to regulate cellular calcium flux, cytoskeletal remodeling, and metabolic gene expression. In endothelial and smooth muscle cells, receptor engagement by VLDL promotes pro-inflammatory gene transcription via nuclear factor kappaB activation and enhances vascular dysfunction[65].
Multiple experimental studies demonstrate that VLDL increases reactive oxygen species (ROS) production in endothelial cells. VLDL exposure impairs nitric oxide bioavailability by uncoupling endothelial nitric oxide synthase, leading to reduced vasodilation and increased vascular tone. This oxidative stress-nitric oxide imbalance contributes to endothelial dysfunction, a key early event in atherosclerosis[92].
Animal models of hypertriglyceridemia further show that elevated VLDL promotes vascular inflammation, increased adhesion molecule expression (vascular cell adhesion molecule 1, intercellular adhesion molecule 1), and monocyte recruitment, thereby accelerating plaque development independently of LDL cholesterol levels.
VLDL has been shown in macrophage and immune cell models to exert direct immunomodulatory effects. VLDL and its remnants activate toll-like receptor-associated pathways and inflammasome components, leading to increased secretion of pro-inflammatory cytokines such as tumor necrosis factor-α, IL-6, and IL-1β. These responses are amplified under conditions of insulin resistance and chronic nutrient excess[57].
In autoimmune and inflammatory disease models, altered VLDL composition (e.g., enriched in saturated fatty acids or apo C-III) enhances macrophage foam cell formation and skews immune responses toward a pro-inflammatory phenotype. Apo C-III-rich VLDL, in particular, has been shown to inhibit lipoprotein clearance and directly activate inflammatory signaling pathways.
Experimental evidence indicates that VLDL plays a role in endocrine signaling, particularly within the adrenal gland. VLDL exposure stimulates phospholipase D activity, leading to increased intracellular calcium concentrations and enhanced aldosterone synthesis. This mechanism provides a link between dyslipidemia and hypertension, as excess aldosterone promotes sodium retention, vascular remodeling, and cardiac fibrosis[61]. Additionally, VLDL-induced hormonal alterations may exacerbate insulin resistance, creating a feed-forward loop that further increases hepatic VLDL production.
Emerging experimental models suggest that VLDL may influence neurological function indirectly through vascular and inflammatory mechanisms. In animal models, elevated VLDL is associated with blood-brain barrier dysfunction, neuroinflammation, and impaired cerebral perfusion, which may contribute to cognitive decline. Altered sleep patterns observed in metabolic disease models have also been linked to dysregulated VLDL metabolism, possibly through hypothalamic inflammation and hormonal imbalance[115].
Although VLDL has traditionally been viewed as a lipid transport particle, emerging evidence suggests that VLDL exerts direct, cell-specific signaling effects independent of its role in TG delivery[65]. Future studies should: Elucidate VLDL-induced intracellular signaling cascades (e.g., mitogen-activated protein kinase, nuclear factor kappaB, phosphoinositide 3 kinase-protein kinase B) in hepatocytes, endothelial cells, cardiomyocytes, adipocytes, immune cells, and atrial myocytes. Distinguish signaling effects mediated by VLDL particles per se vs those mediated through VLDLR, LDLR, and LDL receptor-related protein 1. Define how lipid composition (TG-rich vs cholesterol-enriched VLDL) alters receptor engagement and downstream signaling. Therapeutic implication is that targeting VLDL-triggered signaling pathways may prevent inflammation, fibrosis, or electrical remodeling without necessarily lowering circulating lipid levels.
VLDL particles are highly heterogeneous in size, density, apo content, and lipid composition. Key research priorities include: Characterization of pathogenic VLDL subfractions using lipidomic, proteomics, and cryo-electron microscopy. Understanding how apo C-III, apo E isoforms, and oxidized lipids influence VLDL clearance, receptor binding, and pro-inflammatory activity. Investigating disease-specific VLDL signatures in MAFLD, atrial fibrillation, neurodegeneration, cancer, and autoimmune disorders[23]. Therapeutic implication is that precision therapies targeting specific VLDL subclasses rather than total VLDL may yield better efficacy with fewer metabolic side effects.
The VLDLR is increasingly recognized as a mediator of tissue lipid uptake and pathological remodeling, particularly in the heart, skeletal muscle, adipose tissue, and brain[166]. Future research should: Define context-dependent roles of VLDLR under physiological vs disease states. Explore how VLDLR overexpression or aberrant activation contributes to lipotoxicity, atrial myopathy, insulin resistance, and neuroinflammation. Investigate VLDLR crosstalk with reelin signaling, Wnt pathways, and inflammatory mediators. Therapeutic implication is that tissue-selective modulation of VLDLR (rather than systemic lipid lowering) could prevent local lipid overload and organ-specific pathology.
Excessive hepatic VLDL secretion is a hallmark of insulin resistance and MAFLD. However, the causal mechanisms linking hepatic VLDL output to extrahepatic disease remain poorly defined. Future directions include: Identifying how hepatic metabolic stress alters VLDL composition and bioactivity. Understanding endocrine-like actions of liver-derived VLDL on distant organs. Exploring transcriptional regulators (sterol regulatory element-binding protein-1c, car
VLDL has been shown to interact with immune cells and promote low-grade chronic inflammation. Future research should: Clarify how VLDL influences macrophage polarization, T-cell activation, and inflammasome signaling. Examine the role of modified VLDL in autoimmune and inflammatory diseases. Investigate interactions between VLDL, gut microbiota, and endotoxemia. Therapeutic implication is that anti-inflammatory strategies targeting VLDL-immune cell interactions could complement lipid-lowering therapies.
Mechanistic insights support multiple therapeutic avenues: Apo C-III inhibitors (antisense oligonucleotides, siRNA) to enhance VLDL clearance. Angiopoietin-like 3/4 inhibition to improve LPL activity and reduce VLDL-TG. Small molecules or biologics targeting VLDL-receptor interactions or downstream signaling. Lifestyle and nutraceutical interventions that specifically alter VLDL composition and secretion. Future clinical trials should incorporate VLDL-specific biomarkers, tissue imaging, and functional endpoints rather than relying solely on plasma TG levels.
Integrative approaches combining lipidomic, transcriptomics, metabolomics, and machine learning will be essential to: Identify individuals with VLDL-driven disease phenotypes. Predict therapeutic responsiveness to VLDL-targeted interventions. Develop personalized treatment strategies across metabolic, cardiovascular, and inflammatory disorders.
The liver is the primary site of VLDL synthesis and secretion, and dysregulated VLDL metabolism has been implicated in a wide spectrum of diseases, including atherosclerosis, MAFLD, cognitive impairment, MetS, autoimmune disorders, breast cancer, and head and neck cancers. Emerging evidence suggests that elevated VLDL levels may contribute to atrial myopathy during the preclinical stage of atrial fibrillation. Moreover, VLDL has been reported to exert cytotoxic effects on cardiomyocytes under MetS-associated conditions, although the precise molecular mechanisms underlying these effects remain unclear.
Endocrine disturbances significantly influence circulating VLDL concentrations, and experimental studies indicate that VLDL is associated with the stimulation of aldosterone synthesis, thereby linking lipoprotein metabolism with hormonal regulation. Despite increasing associations between VLDL and diverse pathological states - including extrahepatic VLDL secretion, neurological disorders, sleep disturbances, and various malignancies - the causal and pathogenic roles of VLDL remain incompletely defined.
Nonetheless, accumulating experimental and clinical evidence supports a predominantly deleterious role of elevated VLDL levels in multisystem disorders. Advancing our understanding of VLDL biology will require a shift in focus from simple associations with circulating levels toward a deeper investigation of VLDL regulation, assembly, secretion, and metabolism. Such mechanistic insights are expected to facilitate the identification and development of VLDL-targeted therapeutic strategies for metabolic, cardiovascular, and systemic diseases. Future research will ultimately lead to discovering and implementing potential VLDL-targeted solutions.
The authors are grateful to Dr. Shoor Vir Singh, Department of Biotechnology at GLA University (Mathura) for help and support during the present study.
| 1. | Niimi M, Yan H, Chen Y, Wang Y, Fan J. Isolation and Analysis of Plasma Lipoproteins by Ultracentrifugation. J Vis Exp. 2021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (2)] |
| 2. | Chapman MJ, Goldstein S, Lagrange D, Laplaud PM. A density gradient ultracentrifugal procedure for the isolation of the major lipoprotein classes from human serum. J Lipid Res. 1981;22:339-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 419] [Cited by in RCA: 416] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 3. | Huang JK, Lee HC. Emerging Evidence of Pathological Roles of Very-Low-Density Lipoprotein (VLDL). Int J Mol Sci. 2022;23:4300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 66] [Article Influence: 16.5] [Reference Citation Analysis (1)] |
| 4. | Tiwari S, Siddiqi SA. Intracellular trafficking and secretion of VLDL. Arterioscler Thromb Vasc Biol. 2012;32:1079-1086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 190] [Article Influence: 13.6] [Reference Citation Analysis (1)] |
| 5. | Ginsberg HN, Packard CJ, Chapman MJ, Borén J, Aguilar-Salinas CA, Averna M, Ference BA, Gaudet D, Hegele RA, Kersten S, Lewis GF, Lichtenstein AH, Moulin P, Nordestgaard BG, Remaley AT, Staels B, Stroes ESG, Taskinen MR, Tokgözoğlu LS, Tybjaerg-Hansen A, Stock JK, Catapano AL. Triglyceride-rich lipoproteins and their remnants: metabolic insights, role in atherosclerotic cardiovascular disease, and emerging therapeutic strategies-a consensus statement from the European Atherosclerosis Society. Eur Heart J. 2021;42:4791-4806. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 668] [Cited by in RCA: 670] [Article Influence: 134.0] [Reference Citation Analysis (2)] |
| 6. | Magnifico MC, Oberkersch RE, Mollo A, Giambelli L, Grooten Y, Sarti P, Calabrese GC, Arese M. VLDL Induced Modulation of Nitric Oxide Signalling and Cell Redox Homeostasis in HUVEC. Oxid Med Cell Longev. 2017;2017:2697364. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 7. | Tsai YY, Rainey WE, Bollag WB. Very low-density lipoprotein (VLDL)-induced signals mediating aldosterone production. J Endocrinol. 2017;232:R115-R129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 8. | Jiménez B, Holmes E, Heude C, Tolson RF, Harvey N, Lodge SL, Chetwynd AJ, Cannet C, Fang F, Pearce JTM, Lewis MR, Viant MR, Lindon JC, Spraul M, Schäfer H, Nicholson JK. Quantitative Lipoprotein Subclass and Low Molecular Weight Metabolite Analysis in Human Serum and Plasma by (1)H NMR Spectroscopy in a Multilaboratory Trial. Anal Chem. 2018;90:11962-11971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 198] [Article Influence: 24.8] [Reference Citation Analysis (1)] |
| 9. | Phillips CM, Perry IJ. Lipoprotein particle subclass profiles among metabolically healthy and unhealthy obese and non-obese adults: does size matter? Atherosclerosis. 2015;242:399-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 10. | Wang J, Stančáková A, Soininen P, Kangas AJ, Paananen J, Kuusisto J, Ala-Korpela M, Laakso M. Lipoprotein subclass profiles in individuals with varying degrees of glucose tolerance: a population-based study of 9399 Finnish men. J Intern Med. 2012;272:562-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 11. | Garvey WT, Kwon S, Zheng D, Shaughnessy S, Wallace P, Hutto A, Pugh K, Jenkins AJ, Klein RL, Liao Y. Effects of insulin resistance and type 2 diabetes on lipoprotein subclass particle size and concentration determined by nuclear magnetic resonance. Diabetes. 2003;52:453-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 427] [Cited by in RCA: 459] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 12. | Avogaro P, Bon GB, Cazzolato G. Presence of a modified low density lipoprotein in humans. Arteriosclerosis. 1988;8:79-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 238] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 13. | Yang CY, Raya JL, Chen HH, Chen CH, Abe Y, Pownall HJ, Taylor AA, Smith CV. Isolation, characterization, and functional assessment of oxidatively modified subfractions of circulating low-density lipoproteins. Arterioscler Thromb Vasc Biol. 2003;23:1083-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 87] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 14. | Chen CH, Jiang T, Yang JH, Jiang W, Lu J, Marathe GK, Pownall HJ, Ballantyne CM, McIntyre TM, Henry PD, Yang CY. Low-density lipoprotein in hypercholesterolemic human plasma induces vascular endothelial cell apoptosis by inhibiting fibroblast growth factor 2 transcription. Circulation. 2003;107:2102-2108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 128] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 15. | Chen CH, Lu J, Chen SH, Huang RY, Yilmaz HR, Dong J, Elayda MA, Dixon RA, Yang CY. Effects of electronegative VLDL on endothelium damage in metabolic syndrome. Diabetes Care. 2012;35:648-653. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 16. | Fabbrini E, Mohammed BS, Magkos F, Korenblat KM, Patterson BW, Klein S. Alterations in adipose tissue and hepatic lipid kinetics in obese men and women with nonalcoholic fatty liver disease. Gastroenterology. 2008;134:424-431. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 501] [Cited by in RCA: 451] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 17. | Sparks JD, Sparks CE, Adeli K. Selective hepatic insulin resistance, VLDL overproduction, and hypertriglyceridemia. Arterioscler Thromb Vasc Biol. 2012;32:2104-2112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 175] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 18. | Poulsen MK, Nellemann B, Stødkilde-Jørgensen H, Pedersen SB, Grønbæk H, Nielsen S. Impaired Insulin Suppression of VLDL-Triglyceride Kinetics in Nonalcoholic Fatty Liver Disease. J Clin Endocrinol Metab. 2016;101:1637-1646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 19. | Choi SH, Ginsberg HN. Increased very low density lipoprotein (VLDL) secretion, hepatic steatosis, and insulin resistance. Trends Endocrinol Metab. 2011;22:353-363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 299] [Cited by in RCA: 292] [Article Influence: 19.5] [Reference Citation Analysis (1)] |
| 20. | Cohn JS, Patterson BW, Uffelman KD, Davignon J, Steiner G. Rate of production of plasma and very-low-density lipoprotein (VLDL) apolipoprotein C-III is strongly related to the concentration and level of production of VLDL triglyceride in male subjects with different body weights and levels of insulin sensitivity. J Clin Endocrinol Metab. 2004;89:3949-3955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 130] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 21. | Rodriguez V, Newman JD, Schwartzbard AZ. Towards more specific treatment for diabetic dyslipidemia. Curr Opin Lipidol. 2018;29:307-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 22. | Lucero D, Miksztowicz V, Gualano G, Longo C, Landeira G, Álvarez E, Zago V, Brites F, Berg G, Fassio E, Schreier L. Nonalcoholic fatty liver disease associated with metabolic syndrome: Influence of liver fibrosis stages on characteristics of very low-density lipoproteins. Clin Chim Acta. 2017;473:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 23. | Yang M, Liu Q, Huang T, Tan W, Qu L, Chen T, Pan H, Chen L, Liu J, Wong CW, Lu WW, Guan M. Dysfunction of estrogen-related receptor alpha-dependent hepatic VLDL secretion contributes to sex disparity in NAFLD/NASH development. Theranostics. 2020;10:10874-10891. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 63] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 24. | Yang JD, Abdelmalek MF, Guy CD, Gill RM, Lavine JE, Yates K, Klair J, Terrault NA, Clark JM, Unalp-Arida A, Diehl AM, Suzuki A; Nonalcoholic Steatohepatitis Clinical Research Network. Patient Sex, Reproductive Status, and Synthetic Hormone Use Associate With Histologic Severity of Nonalcoholic Steatohepatitis. Clin Gastroenterol Hepatol. 2017;15:127-131.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 88] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 25. | Garcia-Carretero R, Vigil-Medina L, Barquero-Perez O, Ramos-Lopez J. Relevant Features in Nonalcoholic Steatohepatitis Determined Using Machine Learning for Feature Selection. Metab Syndr Relat Disord. 2019;17:444-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 26. | Ballestri S, Nascimbeni F, Baldelli E, Marrazzo A, Romagnoli D, Lonardo A. NAFLD as a Sexual Dimorphic Disease: Role of Gender and Reproductive Status in the Development and Progression of Nonalcoholic Fatty Liver Disease and Inherent Cardiovascular Risk. Adv Ther. 2017;34:1291-1326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 458] [Cited by in RCA: 431] [Article Influence: 47.9] [Reference Citation Analysis (2)] |
| 27. | Shen B, Zhao C, Wang Y, Peng Y, Cheng J, Li Z, Wu L, Jin M, Feng H. Aucubin inhibited lipid accumulation and oxidative stress via Nrf2/HO-1 and AMPK signalling pathways. J Cell Mol Med. 2019;23:4063-4075. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 130] [Article Influence: 18.6] [Reference Citation Analysis (1)] |
| 28. | Shen B, Feng H, Cheng J, Li Z, Jin M, Zhao L, Wang Q, Qin H, Liu G. Geniposide alleviates non-alcohol fatty liver disease via regulating Nrf2/AMPK/mTOR signalling pathways. J Cell Mol Med. 2020;24:5097-5108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 108] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 29. | Sano A, Kakazu E, Hamada S, Inoue J, Ninomiya M, Iwata T, Tsuruoka M, Sato K, Masamune A. Steatotic Hepatocytes Release Mature VLDL Through Methionine and Tyrosine Metabolism in a Keap1-Nrf2-Dependent Manner. Hepatology. 2021;74:1271-1286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 30. | Nikolova-Karakashian M. Alcoholic and non-alcoholic fatty liver disease: Focus on ceramide. Adv Biol Regul. 2018;70:40-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 31. | Carlier A, Phan F, Szpigel A, Hajduch E, Salem JE, Gautheron J, Le Goff W, Guérin M, Lachkar F, Ratziu V, Hartemann A, Ferré P, Foufelle F, Bourron O. Dihydroceramides in Triglyceride-Enriched VLDL Are Associated with Nonalcoholic Fatty Liver Disease Severity in Type 2 Diabetes. Cell Rep Med. 2020;1:100154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 32. | Al-Mrabeh A, Zhyzhneuskaya SV, Peters C, Barnes AC, Melhem S, Jesuthasan A, Aribisala B, Hollingsworth KG, Lietz G, Mathers JC, Sattar N, Lean MEJ, Taylor R. Hepatic Lipoprotein Export and Remission of Human Type 2 Diabetes after Weight Loss. Cell Metab. 2020;31:233-249.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 126] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 33. | André P, Komurian-Pradel F, Deforges S, Perret M, Berland JL, Sodoyer M, Pol S, Bréchot C, Paranhos-Baccalà G, Lotteau V. Characterization of low- and very-low-density hepatitis C virus RNA-containing particles. J Virol. 2002;76:6919-6928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 541] [Cited by in RCA: 515] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 34. | Vercauteren K, Mesalam AA, Leroux-Roels G, Meuleman P. Impact of lipids and lipoproteins on hepatitis C virus infection and virus neutralization. World J Gastroenterol. 2014;20:15975-15991. [PubMed] [DOI] [Full Text] |
| 35. | Nielsen SU, Bassendine MF, Burt AD, Martin C, Pumeechockchai W, Toms GL. Association between hepatitis C virus and very-low-density lipoprotein (VLDL)/LDL analyzed in iodixanol density gradients. J Virol. 2006;80:2418-2428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 273] [Cited by in RCA: 260] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 36. | Sun HY, Lin CC, Lee JC, Wang SW, Cheng PN, Wu IC, Chang TT, Lai MD, Shieh DB, Young KC. Very low-density lipoprotein/lipo-viro particles reverse lipoprotein lipase-mediated inhibition of hepatitis C virus infection via apolipoprotein C-III. Gut. 2013;62:1193-1203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 37. | Serfaty L. Metabolic Manifestations of Hepatitis C Virus: Diabetes Mellitus, Dyslipidemia. Clin Liver Dis. 2017;21:475-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 38. | Sun HY, Cheng PN, Tseng CY, Tsai WJ, Chiu YC, Young KC. Favouring modulation of circulating lipoproteins and lipid loading capacity by direct antiviral agents grazoprevir/elbasvir or ledipasvir/sofosbuvir treatment against chronic HCV infection. Gut. 2018;67:1342-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 39. | Sørensen LP, Andersen IR, Søndergaard E, Gormsen LC, Schmitz O, Christiansen JS, Nielsen S. Basal and insulin mediated VLDL-triglyceride kinetics in type 2 diabetic men. Diabetes. 2011;60:88-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 40. | Tricò D, Natali A, Mari A, Ferrannini E, Santoro N, Caprio S. Triglyceride-rich very low-density lipoproteins (VLDL) are independently associated with insulin secretion in a multiethnic cohort of adolescents. Diabetes Obes Metab. 2018;20:2905-2910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 41. | Mucinski JM, Manrique-Acevedo C, Kasumov T, Garrett TJ, Gaballah A, Parks EJ. Relationships between Very Low-Density Lipoproteins-Ceramides, -Diacylglycerols, and -Triacylglycerols in Insulin-Resistant Men. Lipids. 2020;55:387-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 42. | Nielsen S, Karpe F. Determinants of VLDL-triglycerides production. Curr Opin Lipidol. 2012;23:321-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 71] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 43. | Xiao C, Pavlic M, Szeto L, Patterson BW, Lewis GF. Effects of acute hyperglucagonemia on hepatic and intestinal lipoprotein production and clearance in healthy humans. Diabetes. 2011;60:383-390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 44. | Lambert JE, Parks EJ. Postprandial metabolism of meal triglyceride in humans. Biochim Biophys Acta. 2012;1821:721-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 130] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 45. | Shulman GI. Ectopic fat in insulin resistance, dyslipidemia, and cardiometabolic disease. N Engl J Med. 2014;371:1131-1141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 894] [Cited by in RCA: 791] [Article Influence: 65.9] [Reference Citation Analysis (1)] |
| 46. | Gormsen LC, Nellemann B, Sørensen LP, Jensen MD, Christiansen JS, Nielsen S. Impact of body composition on very-low-density lipoprotein-triglycerides kinetics. Am J Physiol Endocrinol Metab. 2009;296:E165-E173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 47. | Søndergaard E, Johansen RF, Jensen MD, Nielsen S. Postprandial VLDL-TG metabolism in type 2 diabetes. Metabolism. 2017;75:25-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 48. | Johansen RF, Søndergaard E, Sørensen LP, Jurik AG, Christiansen JS, Nielsen S. Basal and insulin-regulated VLDL1 and VLDL2 kinetics in men with type 2 diabetes. Diabetologia. 2016;59:833-843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 49. | Lee HC, Chen CC, Tsai WC, Lin HT, Shiao YL, Sheu SH, Wu BN, Chen CH, Lai WT. Very-Low-Density Lipoprotein of Metabolic Syndrome Modulates Gap Junctions and Slows Cardiac Conduction. Sci Rep. 2017;7:12050. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 50. | Lee HC, Shin SJ, Huang JK, Lin MY, Lin YH, Ke LY, Jiang HJ, Tsai WC, Chao MF, Lin YH. The role of postprandial very-low-density lipoprotein in the development of atrial remodeling in metabolic syndrome. Lipids Health Dis. 2020;19:210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 51. | Lee HC, Lin HT, Ke LY, Wei C, Hsiao YL, Chu CS, Lai WT, Shin SJ, Chen CH, Sheu SH, Wu BN. VLDL from Metabolic Syndrome Individuals Enhanced Lipid Accumulation in Atria with Association of Susceptibility to Atrial Fibrillation. Int J Mol Sci. 2016;17:134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 52. | Lin YS, Liu CK, Lee HC, Chou MC, Ke LY, Chen CH, Chen SL. Electronegative very-low-density lipoprotein induces brain inflammation and cognitive dysfunction in mice. Sci Rep. 2021;11:6013. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 53. | Li CL, Chu CH, Lee HC, Chou MC, Liu CK, Chen CH, Ke LY, Chen SL. Immunoregulatory effects of very low density lipoprotein from healthy individuals and metabolic syndrome patients on glial cells. Immunobiology. 2019;224:632-637. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 54. | Kornetova EG, Kornetov AN, Mednova IA, Dubrovskaya VV, Boiko AS, Bokhan NA, Loonen AJM, Ivanova SA. Changes in Body Fat and Related Biochemical Parameters Associated With Atypical Antipsychotic Drug Treatment in Schizophrenia Patients With or Without Metabolic Syndrome. Front Psychiatry. 2019;10:803. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 55. | Mendivil CO, Rimm EB, Furtado J, Sacks FM. Apolipoprotein E in VLDL and LDL with apolipoprotein C-III is associated with a lower risk of coronary heart disease. J Am Heart Assoc. 2013;2:e000130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 56. | Whitacre BE, Howles P, Street S, Morris J, Swertfeger D, Davidson WS. Apolipoprotein E content of VLDL limits LPL-mediated triglyceride hydrolysis. J Lipid Res. 2022;63:100157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 57. | Shin KC, Hwang I, Choe SS, Park J, Ji Y, Kim JI, Lee GY, Choi SH, Ching J, Kovalik JP, Kim JB. Macrophage VLDLR mediates obesity-induced insulin resistance with adipose tissue inflammation. Nat Commun. 2017;8:1087. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 75] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 58. | Ruby MA, Goldenson B, Orasanu G, Johnston TP, Plutzky J, Krauss RM. VLDL hydrolysis by LPL activates PPAR-alpha through generation of unbound fatty acids. J Lipid Res. 2010;51:2275-2281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 58] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 59. | Wood AC, Glasser S, Garvey WT, Kabagambe EK, Borecki IB, Tiwari HK, Tsai MY, Hopkins PN, Ordovas JM, Arnett DK. Lipoprotein lipase S447X variant associated with VLDL, LDL and HDL diameter clustering in the MetS. Lipids Health Dis. 2011;10:143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 60. | Giordano R, Picu A, Marinazzo E, D'Angelo V, Berardelli R, Karamouzis I, Forno D, Zinnà D, Maccario M, Ghigo E, Arvat E. Metabolic and cardiovascular outcomes in patients with Cushing's syndrome of different aetiologies during active disease and 1 year after remission. Clin Endocrinol (Oxf). 2011;75:354-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 91] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 61. | Olejarz M, Szczepanek-Parulska E, Ruchala M. Lipoprotein alterations in endocrine disorders - a review of the recent developments in the field. Front Endocrinol (Lausanne). 2024;15:1354098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 62. | de Guia RM, Herzig S. How Do Glucocorticoids Regulate Lipid Metabolism? Adv Exp Med Biol. 2015;872:127-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 63. | Barbot M, Zilio M, Scaroni C. Cushing's syndrome: Overview of clinical presentation, diagnostic tools and complications. Best Pract Res Clin Endocrinol Metab. 2020;34:101380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 75] [Article Influence: 12.5] [Reference Citation Analysis (1)] |
| 64. | Hakami OA, Ahmed S, Karavitaki N. Epidemiology and mortality of Cushing's syndrome. Best Pract Res Clin Endocrinol Metab. 2021;35:101521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 98] [Article Influence: 19.6] [Reference Citation Analysis (1)] |
| 65. | Tsai YY, Rainey WE, Johnson MH, Bollag WB. VLDL-activated cell signaling pathways that stimulate adrenal cell aldosterone production. Mol Cell Endocrinol. 2016;433:138-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 66. | Lee HC, Akhmedov A, Chen CH. Spotlight on very-low-density lipoprotein as a driver of cardiometabolic disorders: Implications for disease progression and mechanistic insights. Front Cardiovasc Med. 2022;9:993633. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 33] [Reference Citation Analysis (1)] |
| 67. | Bollag WB. Role of phospholipases in adrenal steroidogenesis. J Endocrinol. 2016;229:R29-R41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 68. | Baudrand R, Pojoga LH, Vaidya A, Garza AE, Vöhringer PA, Jeunemaitre X, Hopkins PN, Yao TM, Williams J, Adler GK, Williams GH. Statin Use and Adrenal Aldosterone Production in Hypertensive and Diabetic Subjects. Circulation. 2015;132:1825-1833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 47] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 69. | Moller N, Vendelbo MH, Kampmann U, Christensen B, Madsen M, Norrelund H, Jorgensen JO. Growth hormone and protein metabolism. Clin Nutr. 2009;28:597-603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 47] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 70. | Møller N, Jørgensen JO. Effects of growth hormone on glucose, lipid, and protein metabolism in human subjects. Endocr Rev. 2009;30:152-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 612] [Cited by in RCA: 706] [Article Influence: 41.5] [Reference Citation Analysis (1)] |
| 71. | Stochholm K, Gravholt CH, Laursen T, Laurberg P, Andersen M, Kristensen LØ, Feldt-Rasmussen U, Christiansen JS, Frydenberg M, Green A. Mortality and GH deficiency: a nationwide study. Eur J Endocrinol. 2007;157:9-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 83] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 72. | Twickler TB, Dallinga-Thie GM, Zelissen PM, Koppeschaar HP, Erkelens DW. The atherogenic plasma remnant-like particle cholesterol concentration is increased in the fasting and postprandial state in active acromegalic patients. Clin Endocrinol (Oxf). 2001;55:69-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 73. | Fabbrini E, Magkos F, Patterson BW, Mittendorfer B, Klein S. Subclinical hypothyroidism and hyperthyroidism have opposite effects on hepatic very-low-density lipoprotein-triglyceride kinetics. J Clin Endocrinol Metab. 2012;97:E414-E418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 74. | Yang L, Yin R, Wang Z, Wang X, Zhang Y, Zhao D. Circulating Angptl3 and Angptl8 Are Increased in Patients with Hypothyroidism. Biomed Res Int. 2019;2019:3814687. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 75. | Treister-Goltzman Y, Yarza S, Peleg R. Lipid profile in mild subclinical hypothyroidism: systematic review and meta-analysis. Minerva Endocrinol (Torino). 2021;46:428-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 76. | Duntas LH, Brenta G. The effect of thyroid disorders on lipid levels and metabolism. Med Clin North Am. 2012;96:269-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 147] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 77. | Høst C, Gormsen LC, Christensen B, Jessen N, Hougaard DM, Christiansen JS, Pedersen SB, Jensen MD, Nielsen S, Gravholt CH. Independent effects of testosterone on lipid oxidation and VLDL-TG production: a randomized, double-blind, placebo-controlled, crossover study. Diabetes. 2013;62:1409-1416. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 78. | Corona G, Rastrelli G, Di Pasquale G, Sforza A, Mannucci E, Maggi M. Testosterone and Cardiovascular Risk: Meta-Analysis of Interventional Studies. J Sex Med. 2018;15:820-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 97] [Article Influence: 13.9] [Reference Citation Analysis (1)] |
| 79. | Salvador C, Planas J, Agreda F, Placer J, Trilla E, Lopez MA, Morote J. Analysis of the lipid profile and atherogenic risk during androgen deprivation therapy in prostate cancer patients. Urol Int. 2013;90:41-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 80. | Maraka S, Singh Ospina N, Rodriguez-Gutierrez R, Davidge-Pitts CJ, Nippoldt TB, Prokop LJ, Murad MH. Sex Steroids and Cardiovascular Outcomes in Transgender Individuals: A Systematic Review and Meta-Analysis. J Clin Endocrinol Metab. 2017;102:3914-3923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 199] [Article Influence: 22.1] [Reference Citation Analysis (1)] |
| 81. | Gava G, Mancini I, Cerpolini S, Baldassarre M, Seracchioli R, Meriggiola MC. Testosterone undecanoate and testosterone enanthate injections are both effective and safe in transmen over 5 years of administration. Clin Endocrinol (Oxf). 2018;89:878-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 82. | van Velzen DM, Paldino A, Klaver M, Nota NM, Defreyne J, Hovingh GK, Thijs A, Simsek S, T'Sjoen G, den Heijer M. Cardiometabolic Effects of Testosterone in Transmen and Estrogen Plus Cyproterone Acetate in Transwomen. J Clin Endocrinol Metab. 2019;104:1937-1947. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 81] [Article Influence: 11.6] [Reference Citation Analysis (1)] |
| 83. | Couto Alves A, Valcarcel B, Mäkinen VP, Morin-Papunen L, Sebert S, Kangas AJ, Soininen P, Das S, De Iorio M, Coin L, Ala-Korpela M, Järvelin MR, Franks S. Metabolic profiling of polycystic ovary syndrome reveals interactions with abdominal obesity. Int J Obes (Lond). 2017;41:1331-1340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 59] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 84. | Samimi M, Dadkhah A, Haddad Kashani H, Tajabadi-Ebrahimi M, Seyed Hosseini E, Asemi Z. The Effects of Synbiotic Supplementation on Metabolic Status in Women With Polycystic Ovary Syndrome: a Randomized Double-Blind Clinical Trial. Probiotics Antimicrob Proteins. 2019;11:1355-1361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 85. | Marchand GB, Carreau AM, Weisnagel SJ, Bergeron J, Labrie F, Lemieux S, Tchernof A. Increased body fat mass explains the positive association between circulating estradiol and insulin resistance in postmenopausal women. Am J Physiol Endocrinol Metab. 2018;314:E448-E456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 48] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 86. | Lamon-Fava S, Diffenderfer MR, Barrett PHR, Wan WY, Postfai B, Nartsupha C, Dolnikowski GG, Schaefer EJ. Differential Effects of Estrogen and Progestin on Apolipoprotein B100 and B48 Kinetics in Postmenopausal Women. Lipids. 2018;53:167-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 87. | Roach RE, Helmerhorst FM, Lijfering WM, Stijnen T, Algra A, Dekkers OM. Combined oral contraceptives: the risk of myocardial infarction and ischemic stroke. Cochrane Database Syst Rev. 2015;2015:CD011054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 110] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 88. | Kuller LH; Women's Health Initiative. Hormone replacement therapy and risk of cardiovascular disease: implications of the results of the Women's Health Initiative. Arterioscler Thromb Vasc Biol. 2003;23:11-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 65] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 89. | Basaran A. Pregnancy-induced hyperlipoproteinemia: review of the literature. Reprod Sci. 2009;16:431-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 94] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 90. | Alvarez JJ, Montelongo A, Iglesias A, Lasunción MA, Herrera E. Longitudinal study on lipoprotein profile, high density lipoprotein subclass, and postheparin lipases during gestation in women. J Lipid Res. 1996;37:299-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 196] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 91. | Cibickova L, Schovanek J, Karasek D. Changes in serum lipid levels during pregnancy in women with gestational diabetes. A narrative review. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2021;165:8-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 92. | Toescu V, Nuttall SL, Martin U, Nightingale P, Kendall MJ, Brydon P, Dunne F. Changes in plasma lipids and markers of oxidative stress in normal pregnancy and pregnancies complicated by diabetes. Clin Sci (Lond). 2004;106:93-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 82] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 93. | Yadav S, Agrawal M, Hariharan C, Dewani D, Vadera K, Krishna N. A comparative study of serum lipid profile of women with preeclampsia and normotensive pregnancy. J Datta Meghe Inst Med Sci Univ. 2018;13:83-86. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 94. | dos Santos Silva CM, Barbosa FR, Lima GA, Warszawski L, Fontes R, Domingues RC, Gadelha MR. BMI and metabolic profile in patients with prolactinoma before and after treatment with dopamine agonists. Obesity (Silver Spring). 2011;19:800-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 131] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 95. | Inancli SS, Usluogullari A, Ustu Y, Caner S, Tam AA, Ersoy R, Cakir B. Effect of cabergoline on insulin sensitivity, inflammation, and carotid intima media thickness in patients with prolactinoma. Endocrine. 2013;44:193-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 37] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 96. | Schwetz V, Librizzi R, Trummer C, Theiler G, Stiegler C, Pieber TR, Obermayer-Pietsch B, Pilz S. Treatment of hyperprolactinaemia reduces total cholesterol and LDL in patients with prolactinomas. Metab Brain Dis. 2017;32:155-161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 97. | Byberg S, Futtrup J, Andreassen M, Krogh J. Metabolic effects of dopamine agonists in patients with prolactinomas: a systematic review and meta-analysis. Endocr Connect. 2019;8:1395-1404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 98. | Bursać B, Djordjevic A, Veličković N, Milutinović DV, Petrović S, Teofilović A, Gligorovska L, Preitner F, Tappy L, Matić G. Involvement of glucocorticoid prereceptor metabolism and signaling in rat visceral adipose tissue lipid metabolism after chronic stress combined with high-fructose diet. Mol Cell Endocrinol. 2018;476:110-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 99. | Wu T, Yang L, Jiang J, Ni Y, Zhu J, Zheng X, Wang Q, Lu X, Fu Z. Chronic glucocorticoid treatment induced circadian clock disorder leads to lipid metabolism and gut microbiota alterations in rats. Life Sci. 2018;192:173-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 103] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 100. | Li Y, Ma J, Yao K, Su W, Tan B, Wu X, Huang X, Li T, Yin Y, Tosini G, Yin J. Circadian rhythms and obesity: Timekeeping governs lipid metabolism. J Pineal Res. 2020;69:e12682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 109] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 101. | Sacks FM, Alaupovic P, Moye LA, Cole TG, Sussex B, Stampfer MJ, Pfeffer MA, Braunwald E. VLDL, apolipoproteins B, CIII, and E, and risk of recurrent coronary events in the Cholesterol and Recurrent Events (CARE) trial. Circulation. 2000;102:1886-1892. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 339] [Cited by in RCA: 355] [Article Influence: 13.7] [Reference Citation Analysis (1)] |
| 102. | Gentile M, Iannuzzi A, Giallauria F, D'Andrea A, Venturini E, Pacileo M, Covetti G, Panico C, Mattiello A, Vitale G, Sarullo FM, Rubba P, Vigorito C, Panico S, Iannuzzo G. Association between Very Low-Density Lipoprotein Cholesterol (VLDL-C) and Carotid Intima-Media Thickness in Postmenopausal Women Without Overt Cardiovascular Disease and on LDL-C Target Levels. J Clin Med. 2020;9:1422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 103. | Heidemann BE, Koopal C, Bots ML, Asselbergs FW, Westerink J, Visseren FLJ. The relation between VLDL-cholesterol and risk of cardiovascular events in patients with manifest cardiovascular disease. Int J Cardiol. 2021;322:251-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 104. | Duran EK, Aday AW, Cook NR, Buring JE, Ridker PM, Pradhan AD. Triglyceride-Rich Lipoprotein Cholesterol, Small Dense LDL Cholesterol, and Incident Cardiovascular Disease. J Am Coll Cardiol. 2020;75:2122-2135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 287] [Cited by in RCA: 255] [Article Influence: 42.5] [Reference Citation Analysis (2)] |
| 105. | Ren J, Grundy SM, Liu J, Wang W, Wang M, Sun J, Liu J, Li Y, Wu Z, Zhao D. Long-term coronary heart disease risk associated with very-low-density lipoprotein cholesterol in Chinese: the results of a 15-Year Chinese Multi-Provincial Cohort Study (CMCS). Atherosclerosis. 2010;211:327-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 81] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 106. | Varbo A, Freiberg JJ, Nordestgaard BG. Extreme nonfasting remnant cholesterol vs extreme LDL cholesterol as contributors to cardiovascular disease and all-cause mortality in 90000 individuals from the general population. Clin Chem. 2015;61:533-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 155] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 107. | Rosenson RS, Lee ML, Chen Q. Association of total VLDL particle concentrations with elevated blood viscosity in patients with type 2 diabetes. Diabetes Res Clin Pract. 2022;183:109180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 108. | Hollstein T, Vogt A, Grenkowitz T, Stojakovic T, März W, Laufs U, Bölükbasi B, Steinhagen-Thiessen E, Scharnagl H, Kassner U. Treatment with PCSK9 inhibitors reduces atherogenic VLDL remnants in a real-world study. Vascul Pharmacol. 2019;116:8-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 109. | Boulet MM, Cheillan D, Di Filippo M, Buisson C, Michalski MC, Moulin P, Calzada C. Large triglyceride-rich lipoproteins from fasting patients with type 2 diabetes activate platelets. Diabetes Metab. 2020;46:54-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 110. | Kawakami A, Aikawa M, Libby P, Alcaide P, Luscinskas FW, Sacks FM. Apolipoprotein CIII in apolipoprotein B lipoproteins enhances the adhesion of human monocytic cells to endothelial cells. Circulation. 2006;113:691-700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 173] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 111. | Wulff AB, Nordestgaard BG, Tybjærg-Hansen A. APOC3 Loss-of-Function Mutations, Remnant Cholesterol, Low-Density Lipoprotein Cholesterol, and Cardiovascular Risk: Mediation- and Meta-Analyses of 137 895 Individuals. Arterioscler Thromb Vasc Biol. 2018;38:660-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 90] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 112. | Nordestgaard BG. Triglyceride-Rich Lipoproteins and Atherosclerotic Cardiovascular Disease: New Insights From Epidemiology, Genetics, and Biology. Circ Res. 2016;118:547-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 502] [Cited by in RCA: 844] [Article Influence: 84.4] [Reference Citation Analysis (2)] |
| 113. | Pourcet B, Staels B. Alternative macrophages in atherosclerosis: not always protective! J Clin Invest. 2018;128:910-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (2)] |
| 114. | Barrett TJ. Macrophages in Atherosclerosis Regression. Arterioscler Thromb Vasc Biol. 2020;40:20-33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 486] [Article Influence: 69.4] [Reference Citation Analysis (1)] |
| 115. | Okuizumi K, Onodera O, Namba Y, Ikeda K, Yamamoto T, Seki K, Ueki A, Nanko S, Tanaka H, Takahashi H, Oyanagi K, Mizusawa H, Kanazawa I, Tsuji S. Genetic association of the very low density lipoprotein (VLDL) receptor gene with sporadic Alzheimer's disease. Nat Genet. 1995;11:207-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 101] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 116. | Pritchard ML, Saunders AM, Gaskell PC, Small GW, Conneally PM, Rosi B, Yamaoka LH, Roses AD, Haines JL, Pericak-Vance MA. No association between very low density lipoprotein receptor (VLDL-R) and Alzheimer disease in American Caucasians. Neurosci Lett. 1996;209:105-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 117. | Christie RH, Chung H, Rebeck GW, Strickland D, Hyman BT. Expression of the very low-density lipoprotein receptor (VLDL-r), an apolipoprotein-E receptor, in the central nervous system and in Alzheimer's disease. J Neuropathol Exp Neurol. 1996;55:491-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 82] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 118. | Nakamura Y, Yamamoto M, Kumamaru E. Significance of the variant and full-length forms of the very low density lipoprotein receptor in brain. Brain Res. 2001;922:209-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 119. | Herring A, Donath A, Steiner KM, Widera MP, Hamzehian S, Kanakis D, Kölble K, ElAli A, Hermann DM, Paulus W, Keyvani K. Reelin depletion is an early phenomenon of Alzheimer's pathology. J Alzheimers Dis. 2012;30:963-979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 73] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 120. | Rahimi-Balaei M, Jiao X, Dalvand A, Shabanipour S, Chung SH, Amiri S, Kong J, Marzban H. Mutations in the Reelin pathway are associated with abnormal expression of microglial IgG FC receptors in the cerebellar cortex. Mol Biol Rep. 2020;47:5323-5331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 121. | Nelson AR, Sagare AP, Zlokovic BV. Role of clusterin in the brain vascular clearance of amyloid-β. Proc Natl Acad Sci U S A. 2017;114:8681-8682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 68] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 122. | Wojtas AM, Kang SS, Olley BM, Gatherer M, Shinohara M, Lozano PA, Liu CC, Kurti A, Baker KE, Dickson DW, Yue M, Petrucelli L, Bu G, Carare RO, Fryer JD. Loss of clusterin shifts amyloid deposition to the cerebrovasculature via disruption of perivascular drainage pathways. Proc Natl Acad Sci U S A. 2017;114:E6962-E6971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 114] [Article Influence: 12.7] [Reference Citation Analysis (1)] |
| 123. | Anjum S, Bathla M, Panchal S, Singh GP, Singh M. Metabolic syndrome in drug naïve schizophrenic patients. Diabetes Metab Syndr. 2018;12:135-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 124. | Solberg DK, Bentsen H, Refsum H, Andreassen OA. Lipid profiles in schizophrenia associated with clinical traits: a five year follow-up study. BMC Psychiatry. 2016;16:299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 125. | Lin BD, Alkema A, Peters T, Zinkstok J, Libuda L, Hebebrand J, Antel J, Hinney A, Cahn W, Adan R, Luykx JJ. Assessing causal links between metabolic traits, inflammation and schizophrenia: a univariable and multivariable, bidirectional Mendelian-randomization study. Int J Epidemiol. 2019;48:1505-1514. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 126. | Tatar ZB. The relationship between serum lipid levels and lifetime suicide attempts in patients with schizophrenia. Med Science. 2018;7:826-830. [DOI] [Full Text] |
| 127. | Nandeesha H, Keshri N, Rajappa M, Menon V. Association of hyperglycaemia and hyperlipidaemia with cognitive dysfunction in schizophrenia spectrum disorder. Arch Physiol Biochem. 2023;129:497-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 128. | Usui N, Iwata K, Miyachi T, Takagai S, Wakusawa K, Nara T, Tsuchiya KJ, Matsumoto K, Kurita D, Kameno Y, Wakuda T, Takebayashi K, Iwata Y, Fujioka T, Hirai T, Toyoshima M, Ohnishi T, Toyota T, Maekawa M, Yoshikawa T, Maekawa M, Nakamura K, Tsujii M, Sugiyama T, Mori N, Matsuzaki H. VLDL-specific increases of fatty acids in autism spectrum disorder correlate with social interaction. EBioMedicine. 2020;58:102917. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 129. | Geovanini GR, Lorenzi-Filho G, de Paula LK, Oliveira CM, de Oliveira Alvim R, Beijamini F, Negrão AB, von Schantz M, Knutson KL, Krieger JE, Pereira AC. Poor sleep quality and lipid profile in a rural cohort (The Baependi Heart Study). Sleep Med. 2019;57:30-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 130. | Depner CM, Stothard ER, Wright KP Jr. Metabolic consequences of sleep and circadian disorders. Curr Diab Rep. 2014;14:507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 328] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 131. | Navaneethan SD, Schold JD, Arrigain S, Thomas G, Jolly SE, Poggio ED, Schreiber MJ Jr, Sarnak MJ, Nally JV Jr. Serum triglycerides and risk for death in Stage 3 and Stage 4 chronic kidney disease. Nephrol Dial Transplant. 2012;27:3228-3234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 132. | Björnson E, Adiels M, Taskinen MR, Borén J. Kinetics of plasma triglycerides in abdominal obesity. Curr Opin Lipidol. 2017;28:11-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 62] [Article Influence: 6.9] [Reference Citation Analysis (1)] |
| 133. | Miljkovic M, Stefanovic A, Simic-Ogrizovic S, Vekic J, Bogavac-Stanojevic N, Cerne D, Kocbek P, Marc J, Jelic-Ivanovic Z, Spasojevic-Kalimanovska V, Kotur-Stevuljevic J. Association of Dyslipidemia, Oxidative Stress, and Inflammation With Redox Status in VLDL, LDL, and HDL Lipoproteins in Patients With Renal Disease. Angiology. 2018;69:861-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 134. | Chan DT, Dogra GK, Irish AB, Ooi EM, Barrett PH, Chan DC, Watts GF. Chronic kidney disease delays VLDL-apoB-100 particle catabolism: potential role of apolipoprotein C-III. J Lipid Res. 2009;50:2524-2531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 59] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 135. | Liang K, Vaziri ND. Acquired VLDL receptor deficiency in experimental nephrosis. Kidney Int. 1997;51:1761-1765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 44] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 136. | Clement LC, Macé C, Avila-Casado C, Joles JA, Kersten S, Chugh SS. Circulating angiopoietin-like 4 links proteinuria with hypertriglyceridemia in nephrotic syndrome. Nat Med. 2014;20:37-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 128] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 137. | Macé C, Chugh SS. Nephrotic syndrome: components, connections, and angiopoietin-like 4-related therapeutics. J Am Soc Nephrol. 2014;25:2393-2398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 43] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 138. | Hirao Y, Nakajima K, Machida T, Murakami M, Ito Y. Development of a Novel Homogeneous Assay for Remnant Lipoprotein Particle Cholesterol. J Appl Lab Med. 2018;3:26-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 139. | Iqbal J, Al Qarni A, Hawwari A, Alghanem AF, Ahmed G. Metabolic Syndrome, Dyslipidemia and Regulation of Lipoprotein Metabolism. Curr Diabetes Rev. 2018;14:427-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 140. | Chaurasia B, Summers SA. Ceramides - Lipotoxic Inducers of Metabolic Disorders. Trends Endocrinol Metab. 2015;26:538-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 394] [Cited by in RCA: 491] [Article Influence: 44.6] [Reference Citation Analysis (1)] |
| 141. | Xia JY, Holland WL, Kusminski CM, Sun K, Sharma AX, Pearson MJ, Sifuentes AJ, McDonald JG, Gordillo R, Scherer PE. Targeted Induction of Ceramide Degradation Leads to Improved Systemic Metabolism and Reduced Hepatic Steatosis. Cell Metab. 2015;22:266-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 296] [Article Influence: 26.9] [Reference Citation Analysis (1)] |
| 142. | Raichur S, Wang ST, Chan PW, Li Y, Ching J, Chaurasia B, Dogra S, Öhman MK, Takeda K, Sugii S, Pewzner-Jung Y, Futerman AH, Summers SA. CerS2 haploinsufficiency inhibits β-oxidation and confers susceptibility to diet-induced steatohepatitis and insulin resistance. Cell Metab. 2014;20:687-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 399] [Article Influence: 33.3] [Reference Citation Analysis (1)] |
| 143. | Turpin SM, Nicholls HT, Willmes DM, Mourier A, Brodesser S, Wunderlich CM, Mauer J, Xu E, Hammerschmidt P, Brönneke HS, Trifunovic A, LoSasso G, Wunderlich FT, Kornfeld JW, Blüher M, Krönke M, Brüning JC. Obesity-induced CerS6-dependent C16:0 ceramide production promotes weight gain and glucose intolerance. Cell Metab. 2014;20:678-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 441] [Cited by in RCA: 581] [Article Influence: 48.4] [Reference Citation Analysis (1)] |
| 144. | Gosejacob D, Jäger PS, Vom Dorp K, Frejno M, Carstensen AC, Köhnke M, Degen J, Dörmann P, Hoch M. Ceramide Synthase 5 Is Essential to Maintain C16:0-Ceramide Pools and Contributes to the Development of Diet-induced Obesity. J Biol Chem. 2016;291:6989-7003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 106] [Article Influence: 10.6] [Reference Citation Analysis (1)] |
| 145. | Khovidhunkit W, Kim MS, Memon RA, Shigenaga JK, Moser AH, Feingold KR, Grunfeld C. Effects of infection and inflammation on lipid and lipoprotein metabolism: mechanisms and consequences to the host. J Lipid Res. 2004;45:1169-1196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1223] [Cited by in RCA: 1100] [Article Influence: 50.0] [Reference Citation Analysis (0)] |
| 146. | Bale BF, Doneen AL, Leimgruber PP, Vigerust DJ. The critical issue linking lipids and inflammation: Clinical utility of stopping oxidative stress. Front Cardiovasc Med. 2022;9:1042729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 147. | Batún Garrido JA, Radillo Alba HA, Hernández Núñez É, Olán F. [Dyslipidaemia and atherogenic risk in patients with systemic lupus erythematosus]. Med Clin (Barc). 2016;147:63-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 148. | Pocovi-Gerardino G, Correa-Rodríguez M, Rubio JC, Fernández RR, Amada MM, Caparros MC, Rueda-Medina B, Ortego-Centeno N. The Relationships of High-Sensitivity C-Reactive Protein and Homocysteine Levels With Disease Activity, Damage Accrual, and Cardiovascular Risk in Systemic Lupus Erythematosus. Biol Res Nurs. 2020;22:169-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 149. | Zhou B, Xia Y, She J. Dysregulated serum lipid profile and its correlation to disease activity in young female adults diagnosed with systemic lupus erythematosus: a cross-sectional study. Lipids Health Dis. 2020;19:40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 150. | Araújo DM, Rodrigues CEM, Gonçalves NGG, Rabelo-Júnior CN, Lobo MDP, Moreira RA, Monteiro-Moreira ACO. Proteins Involved in the Induction of Procoagulant Activity and Autoimmune Response in Patients With Primary Antiphospholipid Syndrome. Clin Appl Thromb Hemost. 2020;26:1076029620905338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 151. | García-Chagollán M, Hernández-Martínez SE, Rojas-Romero AE, Muñoz-Valle JF, Sigala-Arellano R, Cerpa-Cruz S, Morales-Núñez JJ, Lomelí-Nieto JA, Macedo Ojeda G, Hernández-Bello J. Metabolic syndrome in rheumatoid arthritis patients: Relationship among its clinical components. J Clin Lab Anal. 2021;35:e23666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 152. | Robinson G, Pineda-Torra I, Ciurtin C, Jury EC. Lipid metabolism in autoimmune rheumatic disease: implications for modern and conventional therapies. J Clin Invest. 2022;132:e148552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 76] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 153. | Sinha PK, Nigam P, Swain JP. Association of Metabolic Syndrome with Vitiligo- A Case Control Study. J Evol Med Dent Sci. 2019;8:2783-2786. [DOI] [Full Text] |
| 154. | Teklu M, Zhou W, Kapoor P, Patel N, Dey AK, Sorokin AV, Manyak GA, Teague HL, Erb-Alvarez JA, Sajja A, Abdelrahman KM, Reddy AS, Uceda DE, Lateef SS, Shanbhag SM, Scott C, Prakash N, Svirydava M, Parel P, Rodante JA, Keel A, Siegel EL, Chen MY, Bluemke DA, Playford MP, Gelfand JM, Mehta NN. Metabolic syndrome and its factors are associated with noncalcified coronary burden in psoriasis: An observational cohort study. J Am Acad Dermatol. 2021;84:1329-1338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 155. | Butler LM, Perone Y, Dehairs J, Lupien LE, de Laat V, Talebi A, Loda M, Kinlaw WB, Swinnen JV. Lipids and cancer: Emerging roles in pathogenesis, diagnosis and therapeutic intervention. Adv Drug Deliv Rev. 2020;159:245-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 367] [Cited by in RCA: 458] [Article Influence: 76.3] [Reference Citation Analysis (1)] |
| 156. | Cheng H, Wang M, Su J, Li Y, Long J, Chu J, Wan X, Cao Y, Li Q. Lipid Metabolism and Cancer. Life (Basel). 2022;12:784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 62] [Reference Citation Analysis (1)] |
| 157. | Paoli P, Giannoni E, Chiarugi P. Anoikis molecular pathways and its role in cancer progression. Biochim Biophys Acta. 2013;1833:3481-3498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 614] [Cited by in RCA: 861] [Article Influence: 66.2] [Reference Citation Analysis (7)] |
| 158. | Love-Gregory L, Sherva R, Schappe T, Qi JS, McCrea J, Klein S, Connelly MA, Abumrad NA. Common CD36 SNPs reduce protein expression and may contribute to a protective atherogenic profile. Hum Mol Genet. 2011;20:193-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 125] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 159. | Enciu AM, Radu E, Popescu ID, Hinescu ME, Ceafalan LC. Targeting CD36 as Biomarker for Metastasis Prognostic: How Far from Translation into Clinical Practice? Biomed Res Int. 2018;2018:7801202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 68] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 160. | Lu CW, Lo YH, Chen CH, Lin CY, Tsai CH, Chen PJ, Yang YF, Wang CH, Tan CH, Hou MF, Yuan SF. VLDL and LDL, but not HDL, promote breast cancer cell proliferation, metastasis and angiogenesis. Cancer Lett. 2017;388:130-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 90] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 161. | Lupien LE, Bloch K, Dehairs J, Traphagen NA, Feng WW, Davis WL, Dennis T, Swinnen JV, Wells WA, Smits NC, Kuemmerle NB, Miller TW, Kinlaw WB. Endocytosis of very low-density lipoproteins: an unexpected mechanism for lipid acquisition by breast cancer cells. J Lipid Res. 2020;61:205-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 40] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 162. | Siti ZS, Seoparjoo AMI, Shahrul H. Lipoproteins modulate growth and P-glycoprotein expression in drug-resistant HER2-overexpressed breast cancer cells. Heliyon. 2019;5:e01573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 163. | Bianco C, Jamialahmadi O, Pelusi S, Baselli G, Dongiovanni P, Zanoni I, Santoro L, Maier S, Liguori A, Meroni M, Borroni V, D'Ambrosio R, Spagnuolo R, Alisi A, Federico A, Bugianesi E, Petta S, Miele L, Vespasiani-Gentilucci U, Anstee QM, Stickel F, Hampe J, Fischer J, Berg T, Fracanzani AL, Soardo G, Reeves H, Prati D, Romeo S, Valenti L. Non-invasive stratification of hepatocellular carcinoma risk in non-alcoholic fatty liver using polygenic risk scores. J Hepatol. 2021;74:775-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 322] [Cited by in RCA: 305] [Article Influence: 61.0] [Reference Citation Analysis (1)] |
| 164. | Jiang X, Qian H, Ding WX. New Glance at the Role of TM6SF2 in Lipid Metabolism and Liver Cancer. Hepatology. 2021;74:1141-1144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 165. | Newberry EP, Hall Z, Xie Y, Molitor EA, Bayguinov PO, Strout GW, Fitzpatrick JAJ, Brunt EM, Griffin JL, Davidson NO. Liver-Specific Deletion of Mouse Tm6sf2 Promotes Steatosis, Fibrosis, and Hepatocellular Cancer. Hepatology. 2021;74:1203-1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 90] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 166. | Shen GM, Zhao YZ, Chen MT, Zhang FL, Liu XL, Wang Y, Liu CZ, Yu J, Zhang JW. Hypoxia-inducible factor-1 (HIF-1) promotes LDL and VLDL uptake through inducing VLDLR under hypoxia. Biochem J. 2012;441:675-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 167. | Şahin F, Aslan AF. Relationship between Inflammatory and Biological Markers and Lung Cancer. J Clin Med. 2018;7:160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 168. | Chi PD, Liu W, Chen H, Zhang JP, Lin Y, Zheng X, Liu W, Dai S. High-density lipoprotein cholesterol is a favorable prognostic factor and negatively correlated with C-reactive protein level in non-small cell lung carcinoma. PLoS One. 2014;9:e91080. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 63] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 169. | Ramaswamy P, Kiran CS, Raju BM, Swathi M, Anusha A, Sharanya GE. Serum lipid profile in patients with oral potentially malignant disorders. J Indian Acad Oral Med Radiol. 2019;31:323-327. [RCA] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 170. | Goel P, Garg R, Raghavan V. Lipid profile in oral potentially malignant disorders. J Indian Acad Oral Med Radiol. 2014;26:374-378. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (1)] |