Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.112270
Revised: August 13, 2025
Accepted: February 2, 2026
Published online: March 20, 2026
Processing time: 235 Days and 10 Hours
Cardiac amyloidosis (CA) is a progressive infiltrative cardiomyopathy resulting from the deposition of misfolded amyloid fibrils in the myocardium and heart valves, with the main forms being amyloid light-chain (AL), transthyretin-related (ATTR) (wild-type or hereditary), and amyloid A amyloidosis. Although historically underdiagnosed, recent advances in non-invasive imaging techniques such as cardiac magnetic resonance imaging and bone scintigraphy have significantly improved its recognition. This review outlines the current diagnostic approaches and therapeutic advancements in CA, emphasizing a multimodal strategy that includes imaging, cardiac biomarkers, and tissue biopsy with precise amyloid subtyping. Diagnosis remains challenging and requires a high degree of clinical suspicion, as delayed recognition can lead to rapid clinical deterioration. Clear diagnostic algorithms and a multidisciplinary care model are essential to im
Core Tip: This review highlights recent advances in cardiac amyloidosis (CA), a progressive infiltrative cardiomyopathy caused by amyloid fibril deposition in cardiac tissues. Improved recognition through advanced non-invasive imaging techniques and significant progress in disease-modifying therapies have transformed diagnosis and treatment. A multimodal diagnostic strategy and early identification are critical to improving patient outcomes. The review emphasizes the need for standardized diagnostic algorithms, increased clinical awareness, and a multidisciplinary approach to optimize care for patients with CA.
- Citation: Bouziane M, Moufid O, Habbal R. Cardiac amyloidosis: From diagnosis to therapeutics breakthroughs. World J Exp Med 2026; 16(1): 112270
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/112270.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.112270
Amyloidosis is a systemic disease characterized by the accumulation of amyloid fibrils and their deposition in extracellular spaces. Those amyloid fibrils are the result of self-aggregated misfolded proteins. It’s a rare and heterogeneous group of disorders that can affect various organs, including the heart, liver, brain, kidneys, and gastrointestinal tract. Thus far, 36 different proteins have been recognized as capable of forming amyloid deposits in humans[1].
Despite originating from different precursor proteins, amyloidogenic proteins follow similar pathogenic pathways. The process of amyloid formation begins when a protein loses its ability to fold properly into its native, functional con
Cardiac amyloidosis (CA) is an infiltrative cardiomyopathy due to the deposition of amyloid fibrils in the myocardium and valvular tissues. The three major forms of CA are amyloid light-chain (AL), transthyretin-related (ATTR) amyloidosis, and serum amyloid A (AA) amyloidosis. AL amyloidosis and ATTR amyloidosis represent more than 95% of CA[2].
Despite recent advances in understanding and diagnosing CA, several challenges remain. These include delayed diagnosis due to non-specific clinical manifestations, limited availability of specific diagnostic tools in routine practice, and therapeutic options that are still evolving. Furthermore, differences in disease progression and response to treatment across amyloid subtypes present additional complexities.
This review aims to synthesize current knowledge on the pathophysiology, clinical features, diagnostic modalities, and therapeutic strategies of CA, while highlighting ongoing challenges and areas in need of further research.
For many years, AL-CA was the most predominant form, affecting equally male and female patients with an estimated incidence of 8 to 15.2 cases per million persons per year. While the overall incidence has remained relatively stable, there has been a notable increase in the recognition of AL-related heart involvement, particularly among individuals aged 18-64 years. Recent data suggest that nearly 12000 patients with AL amyloidosis currently reside in the United States[2].
The rising awareness of this disease from physicians, along with the development of non-invasive tools and algorithm for ATTR-CA has contributed significantly to the rising prevalence of ATTR-CA[3], to the point where it now represents the most frequent form of CA[4,5].
There are two main subtypes of ATTR amyloidosis: The wild-type ATTR (ATTRwt) and the hereditary ATTR (ATTRv). Recent epidemiological data indicate a rising trend in the incidence and prevalence of CA, particularly among older adults. A United States based study involving Medicare beneficiaries aged ≥ 65 years reported a steady increase in both incidence and prevalence of the disease between 2000 and 2012. In hospitalized heart failure patients, the sex distribution was balanced, with the majority (85%) being of white ethnicity[6].
The Transthyretin Amyloidosis Outcomes Survey (THAOS) registry highlights that ATTR amyloidosis predominantly affects elderly males, often presenting with a cardiac-dominant phenotype[7]. The ATTRwt amyloidosis shows increasing prevalence with age, primarily affecting individuals over 60 years. Although historically considered a male-predominant condition, emerging data suggest that women are also significantly affected[8]. In ATTRwt, cardiac involvement is the principal clinical manifestation[6,9].
Further insights into ATTR amyloidosis come from the THAOS registry, a global longitudinal survey established in 2007, which tracks genotype-phenotype relationships, disease progression, and treatment outcomes in both ATTRwt and ATTRv patients, including asymptomatic carriers[10].
The analysis conducted between 2007 and 2022 included 6000 patients worldwide and showed Val30Met as the most common genotype in South America (78.6%), Japan (74.2%), and Europe (62.2%), mainly causing neurological symptoms and cardiac disease mostly limited to conduction system abnormalities[10].
The Ile68 leu mutation is the second most frequent in Europe and typically results in a predominantly cardiac phe
The Val122Ile mutation, associated with hereditary ATTR-CA, likely originated in West Africa, where allele frequencies range from 2.5%-5% in countries like Sierra Leone, Ghana, and Nigeria[13]. Despite the high genetic prevalence, clinical data remain scarce due to underdiagnosis, limited diagnostic access, and low disease awareness. Structural barriers such as a lack of trained personnel, inadequate infrastructure, and weak public health systems further hinder detection and management across the region[14].
Although amyloidogenic proteins originate from distinct precursors, they share common pathogenic mechanisms. Amyloidogenesis typically occurs when a protein fails to attain or maintain its native, functionally active structure, which can lead to protein misfolding and subsequent aggregation. These misfolded proteins may assemble into soluble oligomers, which can ultimately deposit as insoluble fibrils within the extracellular matrix of target tissues[15]. In CA, organ dysfunction is thought to arise through multiple mechanisms. These include mechanical interference with tissue architecture due to extracellular fibril deposition, as well as direct cytotoxic effects of amyloid fibrils and their precursors. Such proteotoxicity may provoke inflammatory responses, oxidative stress, apoptosis, and impaired autophagic processes. Extensive amyloid deposition within the myocardial extracellular matrix disrupts normal tissue architecture and impairs cardiac function. This process results in progressive biventricular wall thickening and increased myocardial stiffness, occurring in the absence of compensatory ventricular dilation and ultimately culminating in both hypertrophic and restrictive cardiomyopathy with reduced cardiac output. In the early stages, cardiac involvement typically presents as isolated diastolic dysfunction with preserved systolic function. As the disease advances, features of restrictive physiology become more pronounced. In addition, atrial amyloid infiltration is common and often leads to impaired atrial contractility[16].
AL amyloidosis results from a clonal proliferation of plasma cells that produces free light chains (FLC), Kappa (κ) and Lambda (λ). Although elevated FLC production is a common feature of plasma cell dyscrasias such as monoclonal gammopathy of undetermined significance, multiple myeloma, and Waldenström macroglobulinemia, only a limited subset of FLC can form amyloid deposits in vivo. In systemic AL amyloidosis, λ light chains are approximately twice as prevalent as κ light chains[17].
In ATTR amyloidosis, the precursor protein is transthyretin. It’s a homotetramer, mainly synthesized in the liver but also in the choroid plexus of the brain and cerebrospinal fluid. It acts as a carrier protein for thyroxine and for retinol.
In ATTRv, various mutations destabilize the transthyretin tetramer, enhancing its tendency to dissociate into monomers that are prone to misfolding and subsequent aggregation. Over 120 amyloidogenic transthyretin mutations have been identified to date, with several variants exhibiting geographic or ethnic clustering. These mutations are associated with variable age at disease onset and differing risks of developing cardiac involvement. Among them, the Val30Met variant is the most extensively documented, while mutations such as Ile68 Leu and Leu111Met are predominantly linked to a cardiac phenotype[2].
In ATTRwt, age-related factors such as oxidative modifications of the transthyretin protein and impairments in proteostasis and cellular repair mechanisms contribute to the destabilization of the native TTR tetramer, promoting its dissociation and subsequent aggregation into amyloid fibrils[16].
AA amyloidosis is commonly associated with autoinflammatory conditions, including rheumatoid arthritis, inflammatory bowel disease, and hidradenitis suppurativa, especially in cases of delayed diagnosis. The disease is characterized by sustained elevation of serum amyloid-associated protein A, an acute-phase reactant which is essential in immune-mediated inflammatory processes. Cardiac involvement in AA amyloidosis is rare and typically occurs after renal involvement. Timely and effective control of the underlying inflammatory disorder can arrest disease progression and, in some cases, lead to partial reversal of organ dysfunction[16].
All types of CA share some particularities, and the most common early clinical manifestation is heart failure with preserved ejection fraction. It mostly starts with dyspnea on exertion that progresses rapidly. Patients can also develop right heart failure signs, such as edema, hepatomegaly, or jugular venous pressure. They can also suffer from atrial arrhythmias, especially atrial fibrillation (AF) and flutter. Furthermore, the intolerance of beta-blockade treatment should raise the suspicion of the diagnosis. Patients can also develop symptoms of peripheral neuropathy or autonomic dysfunction[16,18]. However, there are some clinical clues for each type of CA.
Periorbital purpura and macroglossia (almost pathognomic of AL amyloidosis). Severe hypotension with ACE inhibitors. Amyloid-related nephrotic syndrome and proteinuria, mostly precede the cardiac involvement. Gastrointestinal motility disorder.
Bilateral carpal tunnel. Lumbar spinal stenosis principally with ATTRwt. Biceps tendon rupture. Conduction disease. Paradoxical low flow, low gradient aortic stenosis.
The diagnosis of CA remains a challenge for clinicians. A multimodality approach is essential including electrocardiogram, echocardiography, bone scintigraphy, cardiac magnetic resonance (CMR), endomyocardial or extracardiacbiopsy, biomarkers and genetic.
The most frequent electrocardiographic abnormality in CA is low QRS voltage, and the disproportion between QRS voltages and left ventricular (LV) thickness in imaging techniques. Low QRS voltage range from 25% to 90% of cases. Numerous studies have demonstrated that low QRS voltage is detected more frequently in patients with AL CA than in those with the ATTR subtype. The clinical and echocardiographic correlates of low QRS voltage (LQRSV) have been thoroughly investigated, demonstrating that LQRSV is a marker of more advanced disease and worse prognosis. It’s associated with higher New York Heart Association (NYHA) functional class, elevated natriuretic peptide levels, and impaired right ventricular systolic function[19].
Another frequently observed QRS abnormality in CA is the pseudo-infarction or pseudo-necrosis pattern, characterized by pathological Q waves or QS complexes in at least two contiguous leads, in the absence of prior myocardial infarction or corresponding akinetic segments on echocardiography. This finding, which can be present in up to 70% of CA patients regardless of amyloid subtype, is believed to reflect increased myocardial wall thickness and underlying tissue disruption. Importantly, it often leads to misdiagnosis of coronary artery disease and, in the context of AL CA, has been associated with reduced survival[19].
In AL CA, QRS fragmentation-characterized by notching or an RsR′ pattern without QRS prolongation-is a common finding. In a study of 264 patients excluding those with coronary artery disease, Perlini et al[20] reported a 30% pre
Conduction abnormalities are common in CA, including atrioventricular blocks and His-Purkinje system disease (e.g., bundle branch and fascicular blocks). These result from amyloid infiltration of conduction tissue and cardiac ganglia. In older patients with ATTRwt, age-related degenerative changes of the conduction system may also contribute to these findings[20].
T-wave inversion (TWI), often seen in hypertrophic cardiomyopathy, typically affects inferior and lateral leads and may be accompanied by ST depression, reflecting ischemia or interstitial expansion. In CA, TWI is more common in AL than in ATTR forms, likely due to differences in myocardial infiltration and toxicity[21].
AF is common in CA, and its early detection is clinically important. Independent predictors of AF in CA include age, heart failure, reduced left ventricular ejection fraction (LVEF), renal involvement, enlarged left atrium, and elevated right atrial pressure. While AF is more strongly linked to heart failure hospitalizations than to overall mortality, it significantly increases the risk of intracardiac thrombi and ischemic stroke. Notably, a prothrombotic state may exist even in sinus rhythm, often with reduced responsiveness to anticoagulation. High rates of intracardiac thrombus have been reported despite adequate anticoagulation. As a result, anticoagulation is recommended in all CA patients with AF, irrespective of the CHA2DS2-VA score[22]. Less commonly, patients with CA may develop other supraventricular arrhythmias, such as atrial flutter, atrial tachycardia, and atrioventricular nodal reentry tachycardia[19].
Although less frequent than supraventricular arrhythmias, ventricular arrhythmias are increasingly recognized in CA. Non-sustained ventricular tachycardia has been reported in up to 74% of patients, with no significant difference between CA subtypes. Contributing mechanisms include autonomic dysfunction, capillary amyloid infiltration with microvascular ischemia, and heterogeneous myocardial involvement. While sudden cardiac death has classically been linked to electromechanical dissociation or bradyarrhythmias, growing evidence suggests a potential role for malignant ventricular arrhythmias (sustained ventricular tachycardia, ventricular fibrillation). However, the role of prophylactic implantable cardioverter-defibrillator implantation in primary prevention remains inconclusive[23].
Echocardiography remains a cornerstone in the diagnostic workup of CA, particularly due to its non-invasive nature, wide availability, and ability to assess structural and functional myocardial alterations.
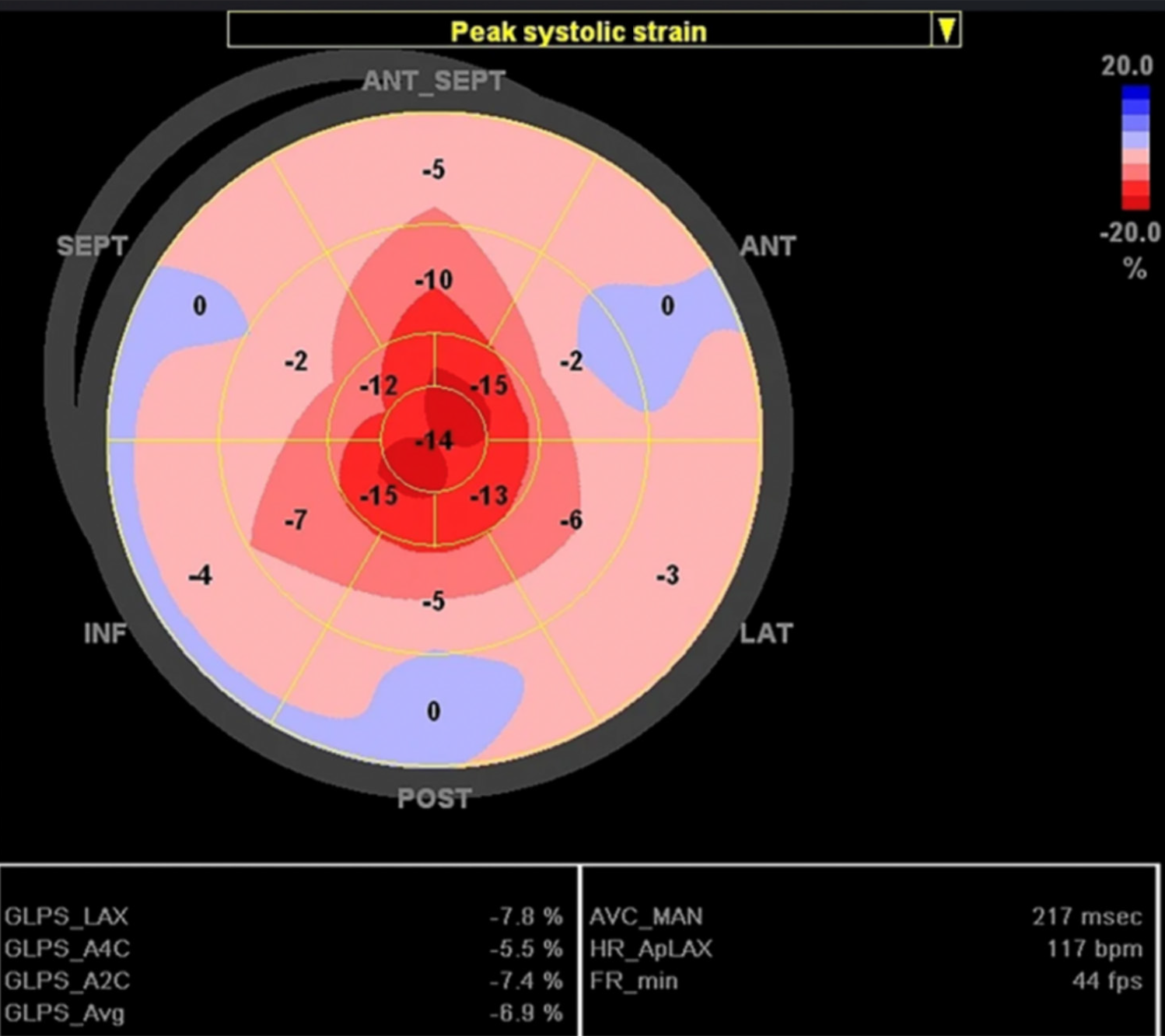
The first tier of evaluation centers around hallmark echocardiographic abnormalities commonly observed in CA. Diastolic dysfunction, particularly grade 2 or higher, is a consistent early manifestation, reflecting the restrictive physiology caused by extracellular amyloid deposition. Similarly, the reduction in tissue Doppler velocities (s’, e’, a’ < 5 cm/s) is indicative of impaired myocardial relaxation and contractility. Perhaps most specific among these is the decrease in global longitudinal strain (GLS), with values < -15% serving as a sensitive marker of subclinical systolic dysfunction. The characteristic “apical sparing” pattern of strain, where basal and mid-wall segments are more severely impaired than apical ones, is often a visual clue pointing toward CA over other hypertrophic conditions (Figure 1)[24].
The European Society of Cardiology working group on myocardial and pericardial diseases proposed in 2020 to use a multiparametric echocardiographic score (Table 1) for the diagnosis of CA, which provides an objective and quantifiable method for risk stratification and diagnostic probability. A score ≥ 8 has shown good sensitivity and specificity in identifying patients with suspected CA. Among its components, a relative wall thickness > 0.6 and an apex-to-base strain ratio > 2.9 are the strongest contributors, highlighting the importance of structural remodeling and regional strain heterogeneity in amyloid cardiomyopathy. Other elements, such as elevated E/e’ ratio, reduced Tricuspid Annular Plane Systolic Excursion (TAPSE), and lower absolute GLS, add complementary information reflecting diastolic burden, right ventricular involvement, and global systolic dysfunction, respectively[22].
| Echocardiographic parameter | Diagnostic threshold | Score |
| Relative LV wall thickness (IVS + PWT)/LVEDD | > 0.6 | 3 points |
| E/e’ ratio (Doppler E wave to e’ velocity) | > 11 | 1 point |
| Tricuspid annular plane systolic excursion | ≤ 19 mm | 2 points |
| LV global longitudinal strain absolute value | ≤ -13% | 1 point |
| Systolic longitudinal strain apex-to-base ratio | > 2.9 | 3 points |
Unexplained LV wall thickening ≥ 12 mm accompanied by either typical echocardiographic features or a multiparametric echocardiographic score ≥ 8, offers a pragmatic framework to raise suspicion for CA, especially in settings where tissue diagnosis or advanced imaging modalities may be limited, but the score needs further studies to validate the performance of these echocardiographic criteria across diverse populations and CA subtypes[22].
Importantly, these echocardiographic criteria also correlate with disease severity and prognosis. Lower GLS and TAPSE values, are associated with worse functional class and higher natriuretic peptide levels. Incorporating these findings into a standardized diagnostic algorithm facilitates early recognition, guides further investigations [e.g., cardiac magnetic resonance imaging (MRI), bone scintigraphy, or biopsy], and expedites timely therapeutic interventions, which is particularly critical in the context of AL amyloidosis where delays in diagnosis adversely impact survival[25].
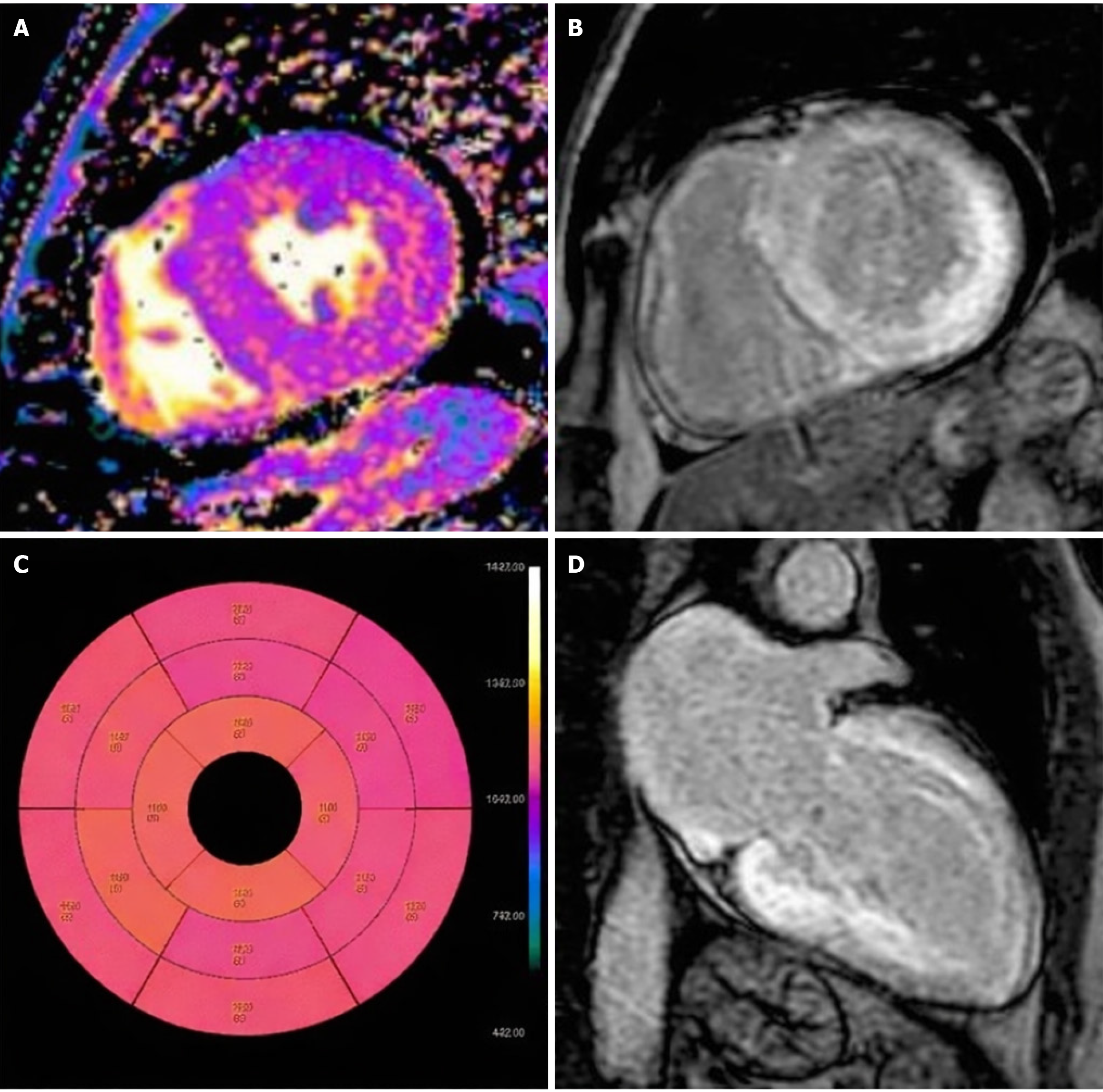
CMR imaging has emerged as a pivotal tool in the non-invasive assessment of CA offering both diagnostic and prognostic value. The hallmark CMR findings characteristic of CA includes diffuse subendocardial or transmural late gadolinium enhancement (LGE) and abnormal gadolinium kinetics, both of which must be present to establish a strong diagnostic suspicion[25].
Diffuse LGE patterns in CA reflect widespread interstitial amyloid deposition. While subendocardial LGE is more commonly seen in earlier stages or in AL amyloidosis, transmural enhancement often indicates more advanced infiltration and is frequently observed in ATTR amyloidosis (Figure 2)[26]. The presence of abnormal gadolinium kinetics, particularly the difficulty in achieving myocardial nulling during inversion recovery sequences, is a distinct feature of amyloid infiltration and results from rapid gadolinium washout from the blood pool relative to the myocardium. This contrast behavior is attributable to the expanded interstitial space caused by amyloid fibril accumulation.
Although not essential for diagnosis, quantitative assessment of extracellular volume (ECV) fraction, particularly values ≥ 0.40, is strongly supportive of cardiac amyloid involvement. ECV quantification correlates with amyloid burden and is reproducible across institutions, making it a valuable marker for disease monitoring and response to therapy[25,26].
CMR also reveals morphologic and functional features consistent with restrictive cardiomyopathy. Typical findings include increased LV wall thickness exceeding 12 mm, predominantly involving the basal segments. In AL amyloidosis, this hypertrophy tends to be concentric, while asymmetric patterns may be observed in ATTR subtypes. LV systolic function is often preserved or mildly reduced (ejection fraction < 60%), with relative apical sparing detectable in advanced stages. Functional parameters frequently show reduced LV end-diastolic volume (< 90 mL) and a decreased stroke volume index (< 35 mL/m²). Biatrial enlargement is a common feature, defined as a left atrial diameter > 41 mm and a right atrial diameter > 44 mm. Additional CMR findings may include interatrial septal thickening (≥ 6 mm), and the presence of pericardial and/or pleural effusions, reflecting advanced disease and ECV overload.
Native T1 mapping provides a quantitative assessment of myocardial tissue characteristics by measuring T1 relaxation times prior to contrast administration. In CA, native T1 values are elevated due to expanded interstitial space from amyloid infiltration, with higher values observed in AL-CA compared to ATTR-CA (Figure 2) This technique allows for non-invasive tissue characterization and correlates with measures of systolic and diastolic dysfunction. Although native T1 mapping is limited by variability across scanners and magnetic field strengths, it remains particularly valuable in patients for whom contrast agents are contraindicated. Reference ranges vary, but values exceeding 1050-1150 ms are typically suggestive of amyloid involvement[26].
The integration of these CMR features into a standardized diagnostic algorithm enhances the accuracy of CA detection, particularly in cases where echocardiographic findings are equivocal. Moreover, CMR not only facilitates early diagnosis but also provides prognostic insights; for instance, transmural LGE and elevated ECV values have been associated with reduced survival and more advanced functional impairment. As a result, CMR plays a critical role in both the diagnostic workup and risk stratification of patients with suspected CA[26].
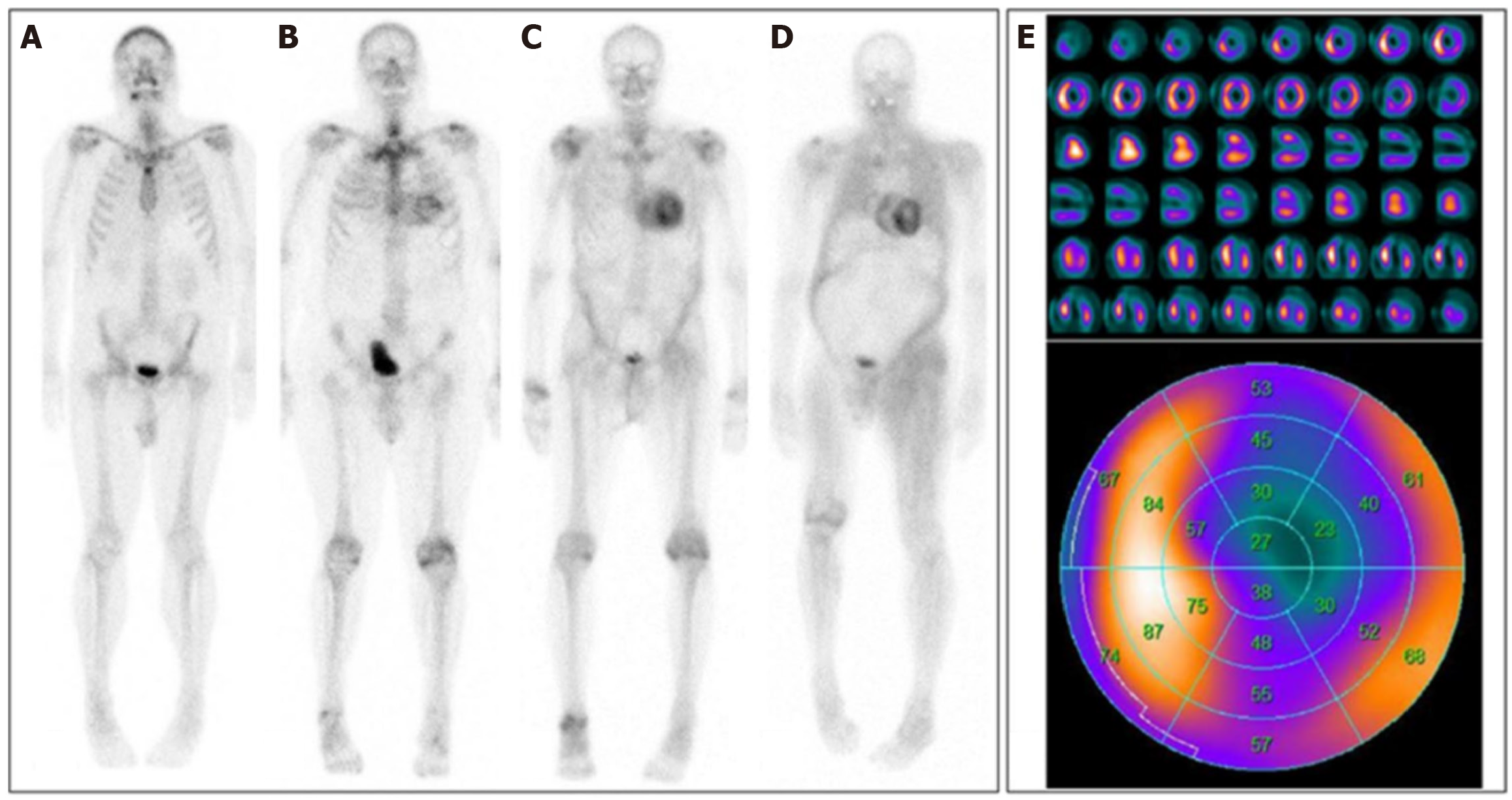
Bone scintigraphy using technetium-labeled radio tracers was discovered in the 1970’s as a CA imaging. The exact mechanism underlying the binding of these tracers to myocardial fibrils is not known but was proposed to be modulated by the calcium P component of the amyloid fibrils which binds to the radio tracers[27]. Other studies have linked the myocardial retention of these tracers to the endomyocardial microcalcifications that are more common in ATTR CA compared to AL CA[28,29].
Only three technetium-labeled radio tracers have been validated for ATTR-CA identification: 99mTc- pyrophosphate, 99mTc-3,3-diphosphono-1,2-propanodicarboxylic acid and 99mTc-hydroxy methylene diphosphonate. Comparison of these three tracers in a 6 studies metanalysis revealed no differences in the diagnosis accuracy with a sensitivity and a specificity above 90%[30].
The cardiac uptake on the planar imaging is assessed visually based on the Perugini grading score[31]. The degree of the tracer uptake is measured comparing the radiotracer activity in the heart and in the bones (Table 2). Note that a grade 2 or above are considered significant[32].
| Perugini score | Cardiac uptake | Bone uptake | Interpretation |
| 0 | Absent | Normal | No evidence of cardiac amyloid deposition |
| 1 | Mild, less than bone uptake | Normal | Low-grade uptake, non-specific |
| 2 | Moderate | Reduced | Suggestive of transthyretin cardiac amyloidosis |
| 3 | Strong | Mild or absent | Highly suggestive of transthyretin cardiac amyloidosis |
In this case bone scintigraphy is considered a sufficient noninvasive tool for ATTR CA, and the extracardiac or endomyocardial biopsy (EMB) are not a must in the setting of typical echocardiography and CMR findings when clonal dyscrasia is excluded by all the serum and urine tests. In this case the specificity has been shown to be near 100%[33].
The results of the cardiac scintigraphy can be influenced by multiple factors, such as rib fractures and valvular or annular calcifications[22], this is why other nuclear imaging techniques can be of good use. Single-photon emission computed tomography (SPECT) techniques for example, and three-dimensional visualization planes in scintigraphy can give more detailed and accurate assessment of radiotracer uptake in the myocardial wall, compared to the 99mTc blood pool imaging[34]. It is then used to assist quantification in scintigraphy by acquiring peak standard uptake values (SUVs) in the myocardium offering three-dimensional assessment[35].
Cardiac SUV and SUV retention index, which reflects how much tracer is retained in the heart relative to a stable reference tissue are well correlated with the Perugini visual score, as it can differentiate between grade 2 or 3 and grades 0 or 1[36] as well as with biomarkers such as troponin and NT-proBNP, as well as strain values in echocardiography[37]. However, bone scintigraphy results can give us false positives when it comes to AL CA[33]. In fact, in some patients with AL CA we can see some cardiac localization of the radiotracers which can make it more difficult to distinguish between AL and ATTR CA subtypes. This is why AL CA should be ruled out first with laboratory tests before interpreting scintigraphy results (Figure 3)[38].
Although still in its early stages, positron emission tomography imaging for the diagnosis of CA holds promise, with further advancements in the technique expected. It uses amyloid-binding radioactive tracers with the 11C-Pib and the 18F-labelled agents being the most used ones[39-42].
Even though high cardiac uptake of these two most used classes of radioactive tracers was observed in both AL and ATTR CA, a meta-analysis demonstrated that higher uptake was observed in AL CA, pointing to the potential in differentiating between AL and ATTR CA[43].
NT-pro-BNP can be elevated in any heart disease, but can be excessively high in CA due to the cellular damage caused by direct myocyte compression and the elevated filling pressure.
Troponin levels are usually elevated in CA which may lead to false diagnosis of myocardial infarction.
A certain level of discordance between biomarkers and clinical symptoms is often found. Heart failure symptoms are usually contradictory with high NT-proBNP levels and persistence in the elevation of cardiac troponin[44]. And due to direct toxicity of amyloid fibril on cardiomyocytes this elevation can precede both clinical symptoms and echocardiography findings in CA[45,46], which makes these biomarkers stand as initial tests when CA is suspected. In fact, NT-proBNP levels under 180 ng/L along with HS-TnT less than 14 ng/L can effectively exclude CA and stop further investigations[47].
Other nonspecific abnormalities such as hypoalbuminemia in the presence of a nephrotic syndrome, or hypercholesterolemia may give a clue to do diagnosis.
In AL amyloidosis, amyloid fibrils result from the misfolding of monoclonal immunoglobulin light chains, typically reflecting an underlying plasma cell dyscrasia. This condition is often associated with suppression of multiple immunoglobulin subclasses resulting in possibly normal serum and urine electrophoresis in patients with AL amyloidosis. On the other hand, Serum free AL and immunofixation of the serum and urine is a much more sensitive test and should always be measured when CA is suspected. Studies showed that the association of serum and urine protein electrophoresis with immunofixation has a 99% sensitivity in identifying AL amyloidosis[48].
FLC κ/λ ratio should always be taken in consideration. An abnormal ratio is found in more than 90% of patient with untreated AL CA. Even though a normal ratio makes the diagnosis less likely, it cannot rule it out or exclude other forms of non-AL CA. The normal serum κ/λ FLC ratio is 0.26-1.65 with the Freelite® assay, and 0.53-1.51 with the N Latex® method. A ratio below 0.26 suggests monoclonal λ light chain production, while a ratio above 1.65 indicates monoclonal κ light chain production. It is important to note that renal dysfunction, especially in patients with an epidermal growth factor receptor (eGFR) ≤ 45 mL/minute/1.73 m2, can raise the κ/λ ratio even in the absence of a clonal disorder. This should be considered when evaluating for monoclonal gammopathy.
Also, the Mayo staging system has shown that cardiac biomarkers such as cTnT, NT-ProBNP, and the serum FLC are powerful prognostic features in patients diagnosed with AL amyloidosis[49-51].
Although the combination of bone scintigraphy with technetium labeled radiotracers and the laboratory tests can be highly suggestive of CA, the definitive diagnosis of CA in any scenario other than negative monoclonal protein assessment and grade 2-3 myocardial uptake, the diagnosis still depends on histopathological confirmation via tissue biopsy demonstrating amyloid deposition[22,52].
This can be obtained from the affected organ or tissue which are the most sensitive sites[22,52] in localized amyloidosis, or from abdominal fat, rectum, kidney, bone marrow, or salivary glands in the case of systemic amyloidosis[2,53,54].
For example, subcutaneous fat is a more accessible tissue and abdominal fat pad aspiration biopsy detects amyloid deposition with a sensitivity of 85.1% and a specificity of 97.1%[55].
Rectal biopsy has a sensitivity of up to 85% combined with fat pad aspiration[56]. But still, complications such as perforation or bleeding are not to be neglected.
When combined, bone marrow biopsy and abdominal fat pad aspiration demonstrate a diagnostic sensitivity approaching 90% for systemic AL amyloidosis[57].
High diagnostic accuracy for AL amyloidosis with sensitivity and specificity > 90% and additional utility in detecting certain hereditary ATTR variants[58,59]. This procedure is also known to have minor complications, such as local swelling or paresthesia in about 10% of cases[58].
Kidney biopsy is especially associated with a major risk of bleeding in patients with a suspicion of CA, and the severity of this complication can go from simple hematuria to major life-threatening bleeding.
EMB in both right or left ventricle maintains its role as the diagnostic gold standard for CA, reserved for definitive diagnosis when non-invasive approaches and extracardiac biopsies yield inconclusive results[22,52].
The number of EMB performed nowadays is very reduced because of the development of non-invasive diagnostic techniques. And so, EMB indications in this matter have been refined and limited to when: Extracardiac biopsies are negative despite high clinical suspicion of CA with monoclonal protein detection and/or abnormal serum FLC. Non-invasive evaluation is inconclusive: Negative or equivocal scintigraphy or unavailable cardiac scintigraphy. Suspected dual pathology: Rare cases of concomitant AL and ATTR amyloidosis[60,61].
Another reason for the reduced use of EMB is the non-negligible risk of major complications, such as thromboembolic risk with a 2% to 4% incidence with the LV EMB, and free wall rupture with an incidence up to 1% to 3% associated with right ventricle (RV) EMB[62], especially in centers with little experience[61,63]. Another major complication in RV EMB is the occurrence of right bundle branch block during sampling in patients with pre-existing left bundle branch block, which may progress to complete heart block necessitating permanent pacing[64].
Thus, this emphasizes the importance of performing these procedures in high-volume centers by experienced operators.
One other reason for this, is that the diagnostic accuracy based on tissue biopsy may be compromised by false-negative results (due to inadequate sampling) or false-positive readings (from staining artifacts)[65,66]. Accurate histopathological staining of biopsy specimens is critical.
Standard immunohistochemistry (IHC) and immunofluorescence for amyloid typing can give misleading results and are now known for their important limitations in staining for amyloid subtyping. Currently, proteomic analysis of amyloid deposits by mass spectrometry represents the diagnostic gold standard. This technique identifies the precise protein composition of amyloid fibrils with high sensitivity and specificity[53,54]. A study reported that it can identify the amyloid type in 80% of amyloid specimens that could not be typed by IHC, but is still limited to very few centers[67].
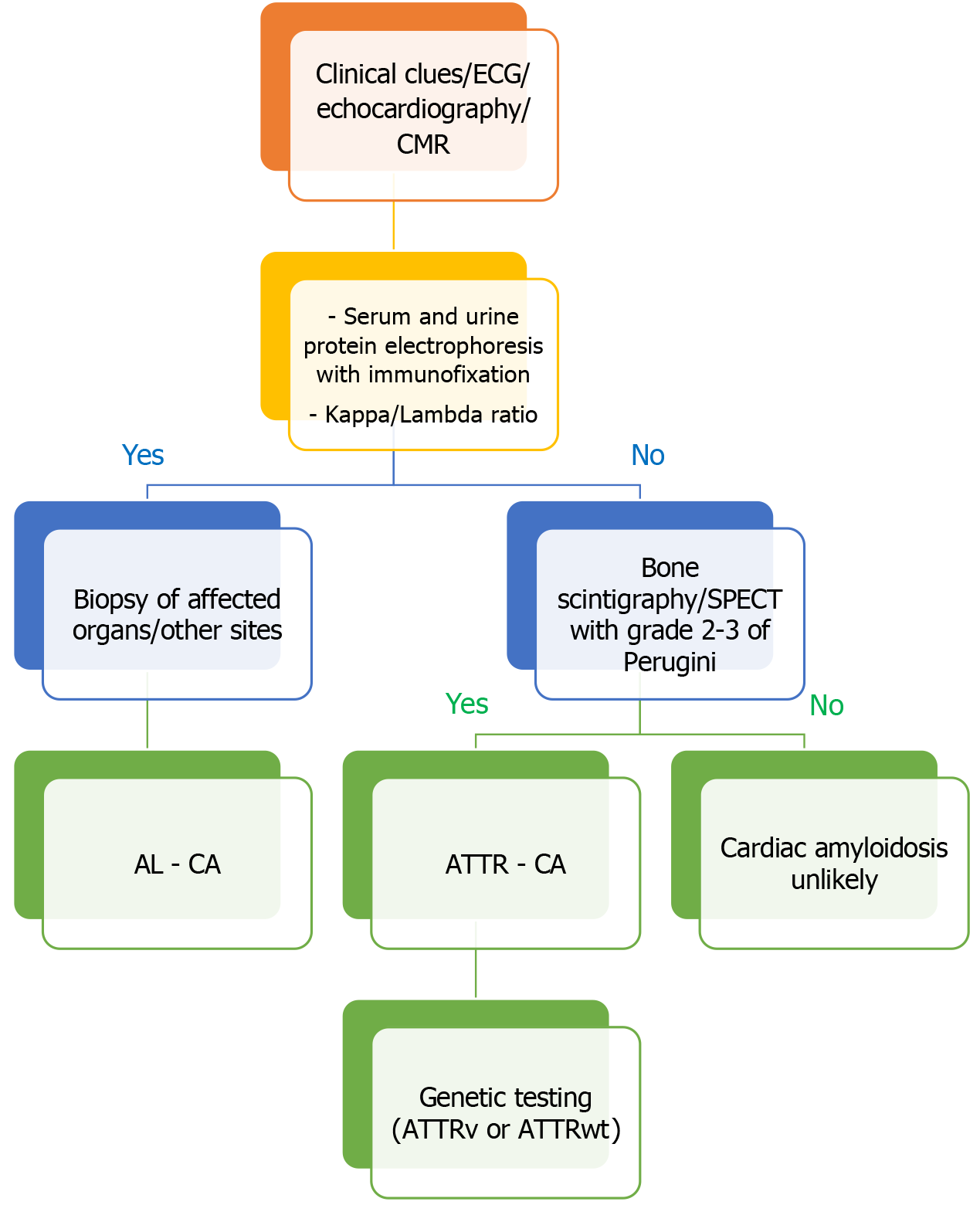
Given the complexity and multiplicity of current diagnostic modalities, we propose a simplified diagnostic algorithm to support a pragmatic and structured approach to the evaluation of suspected CA: The first step when clinical symptoms, electrocardiogram, Echocardiography and CMR is suggestive of CA, is to screen for monoclonal protein using serum and urine electrophoresis with immunofixation along with κ/λ ratio. If this screening is positive, tissue biopsy should be done to confirm AL-CA. (Cardiac scintigraphy cannot differentiate between AL and ATTR CA in this case, since AL-CA can occasionally give positive scintigraphy findings). If negative, 99mTc-labeled bone scintigraphy is performed with SPECT imaging: A grade 0 or 1 uptake makes CA unlikely. Grade 2 or 3 uptake can confirm ATTR-CA, and genetic testing is then required to differentiate between Wild and Hereditary ATTR type. NB: To improve efficiency and reduce time to diagnosis, bone scintigraphy can be performed simultaneously with the hematologic workup. But it’s only interpreted when monoclonal protein screening is negative (Figure 4).
General management of CA basis is the management of the complications and comorbidities to improve the quality of life. It focuses on different clinical aspects, especially heart failure, arrhythmias, conduction disorders, thromboembolism, and even the concomitant presence of severe aortic stenosis.
Cardiac amyloid infiltration stiffens the myocardium, leading to small-to-normal ventricular dimensions, severe diastolic dysfunction, and impaired atrial contraction. This results in reduced stroke volume and cardiac output, often with elevated intracardiac pressures due to restricted filling, which can easily cause volume overload with pulmonary and renal congestion.
The foundation of volume management in this case remains loop diuretics, which must be used with high caution due to the risk of hypoperfusion causing renal insufficiency and fatigue.
Heart failure with reduced ejection fraction therapies, such as angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, angiotensin receptor neprilysin inhibitors, beta-blockers and mineralocorticoid receptor antagonists are often poorly tolerated in patients with CA[68]. Studies has shown that even with low doses, they can cause hypotension, especially in patients with AL CA.
No robust evidence supports the superiority of rhythm control over rate control in CA from a clinical viewpoint[69]. But, when rhythm control strategy fails, rate control becomes necessary, although it is very challenging due to the disease’s unique hemodynamics.
The severe diastolic dysfunction observed in CA due to restrictive physiology is requiring compensatory tachycardia to maintain a good cardiac output, which explains the poor tolerance of antiarrhythmic drugs (beta-blockers, calcium channel blockers and digoxine).
These antiarrhythmics can be harmful through excessive bradycardia on already compromised cardiac output[70,71], hypotension, especially with concomitant autonomic dysfunction observed in CA[72], and the aggravation of cardiac conduction defects[73,74]. In a rate control strategy, low-dose beta-blockers can still be used as a first-line drug[22,75].
Digoxin has high affinity for amyloid fibrils, this binding can potentially increase toxicity risk but can be used in second line with high drug monitoring. Verapamil and diltiazem can be an alternative, but only if LVEF ≥ 40%.
For patients with inadequate rate control despite medical therapy and ineligible for catheter ablation, AV node ablation + cardiac resynchronization therapy/physiological pacing is a viable option[75].
In long-term rhythm control, the absence of randomized control trials studying the safety and efficiency of catheter ablation in atrial arrhythmias, pharmacological strategies are now preferred, with amiodarone being a well-tolerated option in these patients and is considered the antiarrhythmic of choice[75].
A recent meta-analysis based on studies up to February 2024 looked at how well catheter ablation works for patients with CA who also have atrial arrhythmias. The analysis included data from multiple medical databases and different types of studies. The findings show that while catheter ablation can be used in these patients, it is still associated with significant recurrence rates, and the overall mortality rate remains high. This suggests that treating these patients is particularly challenging[76].
The profound atrial abnormalities in CA create a highly thrombogenic environment, similar to that seen in patients with the highest CHA2DS2-VA scores. Amyloid deposition disrupts the atrial endocardium, promoting clot formation, and severe atrial stiffness impairs both left atrial appendage and body contraction. Additionally, the left atrium loses its reservoir function failing to expand during systole or contract during diastole leading to significant blood stasis and a marked increase in thrombotic risk. Thus, in CA with AF, anticoagulation is essential, and stroke risk should not be assessed using the CHA2DS2-VA score alone[77].
AL amyloidosis: The primary treatment goal is to eliminate the clonal plasma cells responsible for pathogenic light chain overproduction, since the AL CA results from misfolded immunoglobulin AL proteins that infiltrate cardiac muscle and cause heart failure.
The treatment choice in AL CA depends on the risk assessment, which is defined by the cardiac involvement in many patients and the cardiac and hematological response. Two main treatment options are available: Chemotherapy regimens and autologous stem cell transplantation (ASCT). In newly diagnosed AL CA the management strategy is dictated by risk stratification.
Thus, low-risk patients are the ones meeting the ASCT eligibility criteria: Physiological age ≤ 70 years-old; LVEF ≥ 40% (preferred if ≥ 50%); Eastern Cooperative Oncology Group performance status (ECOG PS)/World Health Organization performance score ≤ 2; NYHA Class I-II; normal troponin levels; systolic blood pressure ≥ 90 mmHg (preferred if ≥ 100 mmHg); creatinine clearance ≥ 30 mL/minute (unless on chronic dialysis).
In case of intermediate risk patients that are not eligible for ASCT, chemotherapy is initiated as first-line treatment. The standard chemotherapy protocol was high dose melphalan with dexamethasone. However, the introduction of proteasome inhibitor–based therapy marked a paradigm shift. Adding bortezomib to backbone regimens significantly improves hematologic response rates and overall survival, establishing it as the new standard of care[78].
The two most adopted bortezomib-based regimens are: Cyclophosphamide-bortezomib-dexamethasone; bortezomib-melphalan-dexamethasone.
Patients with high-risk are characterized by poor functional status (ECOG PS = 4) and advanced cardiac disease (NYHA class IIIb/IV), often exhibit limited tolerance to standard chemotherapy. In such cases, treatment strategies should prioritize: Dose reduction or sequential drug introduction to minimize toxicity and frequent reassessment to guide potential escalation if tolerated.
Given emerging evidence supporting daratumumab’s efficacy and tolerability in this population, ongoing research are evaluating daratumumab monotherapy as a potential frontline option[79].
Haematological response criteria: Complete response (CR): Negative serum and urine immunofixation and normal FLC ratio. Very good partial response (VGPR): Reduction in the difference between the involved and uninvolved FLC to < 40 mg/L. PR: Requires any of the following: ≥ 50% reduction in current serum monoclonal protein levels > 0.5 g/dL; ≥ 50% reduction in current urine m-protein levels > 100 mg/day with a visible peak; ≥ 50% reduction in current FLC levels > 10 mg/dL. No response/stable disease: Does not meet the criteria for CR, PR, or progressive disease.
Cardiac response criteria: ≥ 2 mm decrease in mean intraventricular septal wall thickness by echocardiogram; ≥ 20% increase in LV ejection fraction; ≥ 2 grade decrease in NYHA class without an increase in diuretic use and no increase in wall thickness. Reduction (≥ 30% and ≥ 300 ng/L) of NT-proBNP in patients in whom the eGFR is ≥ 45 mL/minute/1.73 m2[22].
Also, GLS and cardiac MRI with T1 mapping and ECV assessment can detect subtle changes in cardiac structure and function, offering a more sensitive measure of treatment response[80,81].
For long, ATTR-CA management focused on symptom treatment and complication control. However emergent therapies are now shifting the paradigm towards disease modification, as it is targeting key steps described earlier in amyloid genesis including.
ATTR stabilizers: Diflunisal and tafamidis. ATTR stabilizers bind to transthyretin and prevent tetramer dissociation and amyloidogenesis.
Diflunisal, a nonsteroidal anti-inflammatory, has shown in vitro[82] TTR-stabilizing effects, but clinical data remain limited to small, nonrandomized, single-arm cohort studies primarily in ATTRv-CA patients[83-86]. While these trials suggest potential stabilization of cardiac biomarkers and echocardiographic measures, robust randomized controlled trials are still needed to confirm efficacy and assess impact on hard outcomes like morbidity and mortality. Though generally well-tolerated, diflunisal carries typical nonsteroidal anti-inflammatory risks: It is contraindicated in patients with significant thrombocytopenia or renal impairment (GFR < 40 mL/minute/1.73 m2) and may exacerbate heart failure, cause gastrointestinal bleeding, or provoke intolerance.
On the other hand, Tafamidis, a Diflunisal analog, is considered as the only Food and Drug Administration (FDA)-approved treatment for ATTR-CA both types, since positive results were demonstrated in the ATTR-ACT study in 2019[87].
The ATTR-ACT trial[88] demonstrated that Tafamidis is more effective than a placebo in improving outcomes for patients with transthyretin amyloid cardiomyopathy. the findings of this study were: Reduced mortality and hospitalizations as Tafamidis significantly lowered the combined risk of death from any cause and cardiovascular-related hospitalizations, with benefits across most patient groups, except for those with advanced heart failure (NYHA Class III), where hospitalizations were not significantly reduced.
Improved functional capacity and quality of life as patients on Tafamidis showed slower decline in walking distance (measured by the 6-minute walk test), and a better quality of life (assessed by the KCCQ-OS questionnaire).
Also, since Tafamidis works by stabilizing transthyretin protein and preventing further amyloid buildup, starting treatment early in the disease is likely to provide the greatest benefit[87].
TTR protein synthesis silencing: Both patisiran and inotersen achieve gene silencing but through distinct molecular pathways. Patisiran is a small interfering RNA that guides the RNA-induced silencing complex to degrade transthyretin mRNA, preventing protein synthesis. Inotersen is an antisense oligonucleotide that binds directly to transthyretin mRNA, enabling its destruction by RNase H1 enzyme.
Both treatments are FDA approved for ATTRv polyneuropathy. In addition, the APOLLO-B trial demonstrated significant benefits in ATTR CA, improving functional capacity (6-minute walk test), reducing cardiac biomarkers, and slowing disease progression leading to its FDA approval in October 2023 for both hereditary and wild-type ATTR-CA[89]. Other ongoing randomized controlled trial, helios-B, evaluating a novel silencer treatment for ATTR-CA: Vutrisiran.
The HELIOS-B phase 3 trial evaluated vutrisiran, a subcutaneous RNAi therapy given every 3 months, in patients with ATTR-CA, including both ATTRv and ATTRwt forms and the key findings were: Vutisiran significantly reduced the composite endpoint of all-cause mortality and recurrent cardiovascular events compared to placebo. Improved functional capacity (6-minute walk distance), quality of life (KCCQ score), and slowed heart failure progression. Consistent effects: Benefits were observed across all subgroups, including patients on background tafamidis therapy[90].
ATTR degraders: While current ATTR-CA therapies primarily prevent new amyloid formation, an innovative approach targets existing deposits through amyloid degradation. The combination of doxycycline with TUDCA/UDCA has demonstrated amyloid-clearing potential in preclinical studies[91]. However, clinical data remain limited, showing modest benefits with significant tolerability issues (10% discontinuation rate primarily due to doxycycline-related esophageal and dermatologic side effects)[92-94]. Despite these challenges, this low-cost combination therapy remains promising. If proven effective, this degradation strategy could offer a much-needed affordable alternative to current high-cost disease-modifying therapies[95].
ATTR immunotherapy: Anti-TTR humanized monoclonal antibody is currently being studied in a phase 1 randomized, double-blind, placebo-controlled trial for ATTR-CA, with a planned open-label extension to further evaluate its safety and efficacy[96].
Despite advances in diagnostic tools, the diagnosis and management of CA remain highly challenging. The disease’s nonspecific clinical presentation frequently mimics other cardiomyopathies or heart failure symptoms, leading to delayed or missed diagnoses. This is further compounded by limited availability of advanced imaging modalities, such as cardiac MRI, bone scintigraphy, and hematologic testing in many centers. Standard heart failure therapies are often ineffective or potentially harmful in CA, necessitating specialized disease modifying treatments. Unfortunately, these therapies are costly and not widely accessible, particularly in developing countries. Moreover, the development of new treatments faces delays due to complex approval processes and the intricate pathophysiology of the disease. Effective management also requires coordinated multidisciplinary care involving cardiology, neurology, and hematology specialists, a collaboration that remains limited in many healthcare settings. Genetic screening is similarly restricted to specialized centers, limiting early diagnosis opportunities for at risk family members in transthyretin amyloidosis[97]. In summary, overcoming challenges related to delayed diagnosis, limited diagnostic and therapeutic resources, high treatment costs, and fragmented care is essential to improve outcomes for patients with CA. Improving access to diagnostics and treatment, fostering multidisciplinary care, and supporting ongoing research are essential steps toward better outcomes in this complex and increasingly recognized condition.
| 1. | Picken MM. The Pathology of Amyloidosis in Classification: A Review. Acta Haematol. 2020;143:322-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 176] [Article Influence: 29.3] [Reference Citation Analysis (2)] |
| 2. | Papingiotis G, Basmpana L, Farmakis D. Diagnosis and therapy of cardiac amyloidosis: epidemiology, diagnosis and therapy. E J Cardiol Pract. 2021;19:19-21. |
| 3. | Çavuşoğlu Y, Özpelit E, Çelik A, İkitimur B, Kayıkçıoğlu M, Tokgözoğlu L, Tüfekçioğlu O, Yılmaz MB. Cardiac amyloidosis: Recent advances in the diagnosis and therapy. Turk Kardiyol Dern Ars. 2019;47:1-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 4. | Zampieri M, Nardi G, Del Monaco G, Allinovi M, Gabriele M, Zocchi C, Casagrande S, Fumagalli C, Di Mario C, Olivotto I, Perfetto F, Cappelli F. Changes in the perceived epidemiology of amyloidosis: 20 year-experience from a Tertiary Referral Centre in Tuscany. Int J Cardiol. 2021;335:123-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 5. | Bajwa F, O'Connor R, Ananthasubramaniam K. Epidemiology and clinical manifestations of cardiac amyloidosis. Heart Fail Rev. 2022;27:1471-1484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 6. | Gilstrap LG, Dominici F, Wang Y, El-Sady MS, Singh A, Di Carli MF, Falk RH, Dorbala S. Epidemiology of Cardiac Amyloidosis-Associated Heart Failure Hospitalizations Among Fee-for-Service Medicare Beneficiaries in the United States. Circ Heart Fail. 2019;12:e005407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 193] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 7. | Maurer MS, Hanna M, Grogan M, Dispenzieri A, Witteles R, Drachman B, Judge DP, Lenihan DJ, Gottlieb SS, Shah SJ, Steidley DE, Ventura H, Murali S, Silver MA, Jacoby D, Fedson S, Hummel SL, Kristen AV, Damy T, Planté-Bordeneuve V, Coelho T, Mundayat R, Suhr OB, Waddington Cruz M, Rapezzi C; THAOS Investigators. Genotype and Phenotype of Transthyretin Cardiac Amyloidosis: THAOS (Transthyretin Amyloid Outcome Survey). J Am Coll Cardiol. 2016;68:161-172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 473] [Cited by in RCA: 426] [Article Influence: 42.6] [Reference Citation Analysis (1)] |
| 8. | González-López E, Gagliardi C, Dominguez F, Quarta CC, de Haro-Del Moral FJ, Milandri A, Salas C, Cinelli M, Cobo-Marcos M, Lorenzini M, Lara-Pezzi E, Foffi S, Alonso-Pulpon L, Rapezzi C, Garcia-Pavia P. Clinical characteristics of wild-type transthyretin cardiac amyloidosis: disproving myths. Eur Heart J. 2017;38:1895-1904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 330] [Cited by in RCA: 285] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 9. | Ruberg FL, Grogan M, Hanna M, Kelly JW, Maurer MS. Transthyretin Amyloid Cardiomyopathy: JACC State-of-the-Art Review. J Am Coll Cardiol. 2019;73:2872-2891. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 909] [Cited by in RCA: 812] [Article Influence: 116.0] [Reference Citation Analysis (1)] |
| 10. | Gentile L, Coelho T, Dispenzieri A, Conceição I, Waddington-Cruz M, Kristen A, Wixner J, Diemberger I, Gonzalez-Moreno J, Cariou E, Maurer MS, Planté-Bordeneuve V, Garcia-Pavia P, Tournev I, Gonzalez-Costello J, Duarte AG, Grogan M, Mazzeo A, Chapman D, Gupta P, Glass O, Amass L; THAOS investigators. A 15-year consolidated overview of data in over 6000 patients from the Transthyretin Amyloidosis Outcomes Survey (THAOS). Orphanet J Rare Dis. 2023;18:350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 51] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 11. | Damy T, Kristen AV, Suhr OB, Maurer MS, Planté-Bordeneuve V, Yu CR, Ong ML, Coelho T, Rapezzi C; THAOS Investigators. Transthyretin cardiac amyloidosis in continental Western Europe: an insight through the Transthyretin Amyloidosis Outcomes Survey (THAOS). Eur Heart J. 2022;43:391-400. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 123] [Article Influence: 30.8] [Reference Citation Analysis (1)] |
| 12. | Falk RH. Senile systemic amyloidosis: are regional differences real or do they reflect different diagnostic suspicion and use of techniques? Amyloid. 2012;19 Suppl 1:68-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 13. | Jacobson DR, Alexander AA, Tagoe C, Garvey WT, Williams SM, Tishkoff S, Modiano D, Sirima SB, Kalidi I, Toure A, Buxbaum JN. The prevalence and distribution of the amyloidogenic transthyretin (TTR) V122I allele in Africa. Mol Genet Genomic Med. 2016;4:548-556. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 14. | Madu EC, Mezue K. Uneven burden of cardiac amyloidosis in people of African descent - global imbalance in resources and access. BMC Glob Public Health. 2023;1:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 15. | Dobson CM. Protein folding and misfolding. Nature. 2003;426:884-890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4249] [Cited by in RCA: 3443] [Article Influence: 149.7] [Reference Citation Analysis (1)] |
| 16. | Griffin JM, Rosenblum H, Maurer MS. Pathophysiology and Therapeutic Approaches to Cardiac Amyloidosis. Circ Res. 2021;128:1554-1575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 137] [Article Influence: 27.4] [Reference Citation Analysis (1)] |
| 17. | Bellotti V, Mangione P, Merlini G. Review: immunoglobulin light chain amyloidosis--the archetype of structural and pathogenic variability. J Struct Biol. 2000;130:280-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 145] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 18. | Falk RH, Alexander KM, Liao R, Dorbala S. AL (Light-Chain) Cardiac Amyloidosis: A Review of Diagnosis and Therapy. J Am Coll Cardiol. 2016;68:1323-1341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 568] [Cited by in RCA: 473] [Article Influence: 47.3] [Reference Citation Analysis (1)] |
| 19. | Martini N, Sinigiani G, De Michieli L, Mussinelli R, Perazzolo Marra M, Iliceto S, Zorzi A, Perlini S, Corrado D, Cipriani A. Electrocardiographic features and rhythm disorders in cardiac amyloidosis. Trends Cardiovasc Med. 2024;34:257-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 35] [Article Influence: 17.5] [Reference Citation Analysis (1)] |
| 20. | Perlini S, Salinaro F, Cappelli F, Perfetto F, Bergesio F, Alogna A, Mussinelli R, Boldrini M, Raimondi A, Musca F, Palladini G, Merlini G. Prognostic value of fragmented QRS in cardiac AL amyloidosis. Int J Cardiol. 2013;167:2156-2161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 21. | Porcari A, Rossi M, Cappelli F, Canepa M, Musumeci B, Cipriani A, Tini G, Barbati G, Varrà GG, Morelli C, Fumagalli C, Zampieri M, Argirò A, Vianello PF, Sessarego E, Russo D, Sinigiani G, De Michieli L, Di Bella G, Autore C, Perfetto F, Rapezzi C, Sinagra G, Merlo M. Incidence and risk factors for pacemaker implantation in light-chain and transthyretin cardiac amyloidosis. Eur J Heart Fail. 2022;24:1227-1236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 22. | Garcia-Pavia P, Rapezzi C, Adler Y, Arad M, Basso C, Brucato A, Burazor I, Caforio ALP, Damy T, Eriksson U, Fontana M, Gillmore JD, Gonzalez-Lopez E, Grogan M, Heymans S, Imazio M, Kindermann I, Kristen AV, Maurer MS, Merlini G, Pantazis A, Pankuweit S, Rigopoulos AG, Linhart A. Diagnosis and treatment of cardiac amyloidosis: a position statement of the ESC Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2021;42:1554-1568. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1022] [Cited by in RCA: 844] [Article Influence: 168.8] [Reference Citation Analysis (1)] |
| 23. | Donnellan E, Wazni OM, Hanna M, Saliba W, Jaber W, Kanj M. Primary prevention implantable cardioverter-defibrillators in transthyretin cardiac amyloidosis. Pacing Clin Electrophysiol. 2020;43:1401-1403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 24. | Gil J, Abreu L, Antunes H, Gonçalves ML, Pires MI, Moreira D, Correia E, Santos LS, Cabral JC. Apical sparing of longitudinal strain in speckle-tracking echocardiography: A sensitive and specific finding in cardiac amyloidosis. Neth Heart J. 2018;26:635. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 25. | Bonderman D, Pölzl G, Ablasser K, Agis H, Aschauer S, Auer-Grumbach M, Binder C, Dörler J, Duca F, Ebner C, Hacker M, Kain R, Kammerlander A, Koschutnik M, Kroiss AS, Mayr A, Nitsche C, Rainer PP, Reiter-Malmqvist S, Schneider M, Schwarz R, Verheyen N, Weber T, Zaruba MM, Badr Eslam R, Hülsmann M, Mascherbauer J. Diagnosis and treatment of cardiac amyloidosis: an interdisciplinary consensus statement. Wien Klin Wochenschr. 2020;132:742-761. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 26. | Maggialetti N, Torrente A, Lorusso G, Villanova I, Ficco M, Gravina M, Ferrari C, Giordano L, Granata V, Rubini D, Lucarelli NM, Stabile Ianora AA, Scardapane A. Role of Cardiovascular Magnetic Resonance in Cardiac Amyloidosis: A Narrative Review. J Pers Med. 2024;14:407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 27. | Pepys MB, Dyck RF, de Beer FC, Skinner M, Cohen AS. Binding of serum amyloid P-component (SAP) by amyloid fibrils. Clin Exp Immunol. 1979;38:284-293. [PubMed] |
| 28. | Pilebro B, Suhr OB, Näslund U, Westermark P, Lindqvist P, Sundström T. (99m)Tc-DPD uptake reflects amyloid fibril composition in hereditary transthyretin amyloidosis. Ups J Med Sci. 2016;121:17-24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 100] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 29. | Stats MA, Stone JR. Varying levels of small microcalcifications and macrophages in ATTR and AL cardiac amyloidosis: implications for utilizing nuclear medicine studies to subtype amyloidosis. Cardiovasc Pathol. 2016;25:413-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 110] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 30. | Treglia G, Glaudemans AWJM, Bertagna F, Hazenberg BPC, Erba PA, Giubbini R, Ceriani L, Prior JO, Giovanella L, Slart RHJA. Diagnostic accuracy of bone scintigraphy in the assessment of cardiac transthyretin-related amyloidosis: a bivariate meta-analysis. Eur J Nucl Med Mol Imaging. 2018;45:1945-1955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 95] [Article Influence: 11.9] [Reference Citation Analysis (1)] |
| 31. | Hutt DF, Fontana M, Burniston M, Quigley AM, Petrie A, Ross JC, Page J, Martinez-Naharro A, Wechalekar AD, Lachmann HJ, Quarta CC, Rezk T, Mahmood S, Sachchithanantham S, Youngstein T, Whelan CJ, Lane T, Gilbertson JA, Rowczenio D, Hawkins PN, Gillmore JD. Prognostic utility of the Perugini grading of 99mTc-DPD scintigraphy in transthyretin (ATTR) amyloidosis and its relationship with skeletal muscle and soft tissue amyloid. Eur Heart J Cardiovasc Imaging. 2017;18:1344-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 147] [Article Influence: 16.3] [Reference Citation Analysis (1)] |
| 32. | Perugini E, Guidalotti PL, Salvi F, Cooke RM, Pettinato C, Riva L, Leone O, Farsad M, Ciliberti P, Bacchi-Reggiani L, Fallani F, Branzi A, Rapezzi C. Noninvasive etiologic diagnosis of cardiac amyloidosis using 99mTc-3,3-diphosphono-1,2-propanodicarboxylic acid scintigraphy. J Am Coll Cardiol. 2005;46:1076-1084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 857] [Cited by in RCA: 715] [Article Influence: 34.0] [Reference Citation Analysis (2)] |
| 33. | Gillmore JD, Maurer MS, Falk RH, Merlini G, Damy T, Dispenzieri A, Wechalekar AD, Berk JL, Quarta CC, Grogan M, Lachmann HJ, Bokhari S, Castano A, Dorbala S, Johnson GB, Glaudemans AW, Rezk T, Fontana M, Palladini G, Milani P, Guidalotti PL, Flatman K, Lane T, Vonberg FW, Whelan CJ, Moon JC, Ruberg FL, Miller EJ, Hutt DF, Hazenberg BP, Rapezzi C, Hawkins PN. Nonbiopsy Diagnosis of Cardiac Transthyretin Amyloidosis. Circulation. 2016;133:2404-2412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1789] [Cited by in RCA: 1552] [Article Influence: 155.2] [Reference Citation Analysis (1)] |
| 34. | Grigoratos C, Aimo A, Rapezzi C, Genovesi D, Barison A, Aquaro GD, Vergaro G, Pucci A, Passino C, Marzullo P, Gimelli A, Emdin M. Diphosphonate single-photon emission computed tomography in cardiac transthyretin amyloidosis. Int J Cardiol. 2020;307:187-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 35. | Manrique A, Dudoignon D, Brun S, N'Ganoa C, Cassol E, Legallois D, Lavie-Badie Y, Agostini D, Lairez O. Quantification of myocardial (99m)Tc-labeled bisphosphonate uptake with cadmium zinc telluride camera in patients with transthyretin-related cardiac amyloidosis. EJNMMI Res. 2019;9:117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 36. | Wollenweber T, Rettl R, Kretschmer-Chott E, Rasul S, Kulterer O, Rainer E, Raidl M, Schaffarich MP, Matschitsch S, Stadler M, Traub-Weidinger T, Beiztke D, Loewe C, Duca F, Mascherbauer J, Bonderman D, Hacker M. In Vivo Quantification of Myocardial Amyloid Deposits in Patients with Suspected Transthyretin-Related Amyloidosis (ATTR). J Clin Med. 2020;9:3446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 37. | Löfbacka V, Axelsson J, Pilebro B, Suhr OB, Lindqvist P, Sundström T. Cardiac transthyretin amyloidosis (99m)Tc-DPD SPECT correlates with strain echocardiography and biomarkers. Eur J Nucl Med Mol Imaging. 2021;48:1822-1832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 38. | Yilmaz A, Bauersachs J, Bengel F, Büchel R, Kindermann I, Klingel K, Knebel F, Meder B, Morbach C, Nagel E, Schulze-Bahr E, Aus dem Siepen F, Frey N. Diagnosis and treatment of cardiac amyloidosis: position statement of the German Cardiac Society (DGK). Clin Res Cardiol. 2021;110:479-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 78] [Article Influence: 15.6] [Reference Citation Analysis (1)] |
| 39. | Bokhari S, Castaño A, Pozniakoff T, Deslisle S, Latif F, Maurer MS. (99m)Tc-pyrophosphate scintigraphy for differentiating light-chain cardiac amyloidosis from the transthyretin-related familial and senile cardiac amyloidoses. Circ Cardiovasc Imaging. 2013;6:195-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 564] [Cited by in RCA: 489] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 40. | Shukla AK, Kumar U. Positron emission tomography: An overview. J Med Phys. 2006;31:13-21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 139] [Cited by in RCA: 87] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 41. | Lee JH, Lee GY, Kim SJ, Kim KH, Jeon ES, Lee KH, Kim BT, Choi JY. Imaging Findings and Literature Review of (18)F-FDG PET/CT in Primary Systemic AL Amyloidosis. Nucl Med Mol Imaging. 2015;49:182-190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 42. | Wolk DA, Zhang Z, Boudhar S, Clark CM, Pontecorvo MJ, Arnold SE. Amyloid imaging in Alzheimer's disease: comparison of florbetapir and Pittsburgh compound-B positron emission tomography. J Neurol Neurosurg Psychiatry. 2012;83:923-926. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 89] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 43. | Kim YJ, Ha S, Kim YI. Cardiac amyloidosis imaging with amyloid positron emission tomography: A systematic review and meta-analysis. J Nucl Cardiol. 2020;27:123-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 62] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 44. | Witteles RM, Bokhari S, Damy T, Elliott PM, Falk RH, Fine NM, Gospodinova M, Obici L, Rapezzi C, Garcia-Pavia P. Screening for Transthyretin Amyloid Cardiomyopathy in Everyday Practice. JACC Heart Fail. 2019;7:709-716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 229] [Article Influence: 32.7] [Reference Citation Analysis (0)] |
| 45. | Hu K, Liu D, Salinger T, Oder D, Knop S, Ertl G, Weidemann F, Frantz S, Störk S, Nordbeck P. Value of cardiac biomarker measurement in the differential diagnosis of infiltrative cardiomyopathy patients with preserved left ventricular systolic function. J Thorac Dis. 2018;10:4966-4975. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 46. | Palladini G, Campana C, Klersy C, Balduini A, Vadacca G, Perfetti V, Perlini S, Obici L, Ascari E, d'Eril GM, Moratti R, Merlini G. Serum N-terminal pro-brain natriuretic peptide is a sensitive marker of myocardial dysfunction in AL amyloidosis. Circulation. 2003;107:2440-2445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 439] [Cited by in RCA: 367] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 47. | Vergaro G, Castiglione V, Aimo A, Prontera C, Masotti S, Musetti V, Nicol M, Cohen Solal A, Logeart D, Georgiopoulos G, Chubuchny V, Giannoni A, Clerico A, Buda G, Patel KN, Razvi Y, Patel R, Wechalekar A, Lachmann H, Hawkins PN, Passino C, Gillmore J, Emdin M, Fontana M. N-terminal pro-B-type natriuretic peptide and high-sensitivity troponin T hold diagnostic value in cardiac amyloidosis. Eur J Heart Fail. 2023;25:335-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 44] [Article Influence: 14.7] [Reference Citation Analysis (1)] |
| 48. | Palladini G, Russo P, Bosoni T, Verga L, Sarais G, Lavatelli F, Nuvolone M, Obici L, Casarini S, Donadei S, Albertini R, Righetti G, Marini M, Graziani MS, Melzi D'Eril GV, Moratti R, Merlini G. Identification of amyloidogenic light chains requires the combination of serum-free light chain assay with immunofixation of serum and urine. Clin Chem. 2009;55:499-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 196] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 49. | Dispenzieri A, Gertz MA, Kyle RA, Lacy MQ, Burritt MF, Therneau TM, Greipp PR, Witzig TE, Lust JA, Rajkumar SV, Fonseca R, Zeldenrust SR, McGregor CG, Jaffe AS. Serum cardiac troponins and N-terminal pro-brain natriuretic peptide: a staging system for primary systemic amyloidosis. J Clin Oncol. 2004;22:3751-3757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 822] [Cited by in RCA: 728] [Article Influence: 33.1] [Reference Citation Analysis (1)] |
| 50. | Kumar SK, Gertz MA, Lacy MQ, Dingli D, Hayman SR, Buadi FK, Short-Detweiler K, Zeldenrust SR, Leung N, Greipp PR, Lust JA, Russell SJ, Kyle RA, Rajkumar SV, Dispenzieri A. Recent improvements in survival in primary systemic amyloidosis and the importance of an early mortality risk score. Mayo Clin Proc. 2011;86:12-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 162] [Cited by in RCA: 138] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 51. | Kumar S, Dispenzieri A, Katzmann JA, Larson DR, Colby CL, Lacy MQ, Hayman SR, Buadi FK, Leung N, Zeldenrust SR, Ramirez-Alvarado M, Clark RJ, Kyle RA, Rajkumar SV, Gertz MA. Serum immunoglobulin free light-chain measurement in primary amyloidosis: prognostic value and correlations with clinical features. Blood. 2010;116:5126-5129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 123] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 52. | Writing Committee; Kittleson MM, Ruberg FL, Ambardekar AV, Brannagan TH, Cheng RK, Clarke JO, Dember LM, Frantz JG, Hershberger RE, Maurer MS, Nativi-Nicolau J, Sanchorawala V, Sheikh FH. 2023 ACC Expert Consensus Decision Pathway on Comprehensive Multidisciplinary Care for the Patient With Cardiac Amyloidosis: A Report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2023;81:1076-1126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 492] [Cited by in RCA: 406] [Article Influence: 135.3] [Reference Citation Analysis (1)] |
| 53. | Manolis AS, Manolis AA, Manolis TA, Melita H. Cardiac amyloidosis: An underdiagnosed/underappreciated disease. Eur J Intern Med. 2019;67:1-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 54. | Varga C, Dorbala S, Lousada I, Polydefkis MJ, Wechalekar A, Maurer MS, Comenzo RL. The diagnostic challenges of cardiac amyloidosis: A practical approach to the two main types. Blood Rev. 2021;45:100720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 55. | Kyriakou P, Mouselimis D, Tsarouchas A, Rigopoulos A, Bakogiannis C, Noutsias M, Vassilikos V. Diagnosis of cardiac amyloidosis: a systematic review on the role of imaging and biomarkers. BMC Cardiovasc Disord. 2018;18:221. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 83] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 56. | Hazenberg BPC, van Rijswijk MH, Meijer S, Marrink J, Limburg PC, Janssen S. Systemic Amyloidosis: A Clinical Survey of 162 Cases. In: Isobe T, Araki S, Uchino F, Kito S, Tsubura E, editors. Amyloid and Amyloidosis. Boston: Springer, 1988. [DOI] [Full Text] |
| 57. | Muchtar E, Dispenzieri A, Lacy MQ, Buadi FK, Kapoor P, Hayman SR, Gonsalves W, Warsame R, Kourelis TV, Chakraborty R, Russell S, Lust JA, Lin Y, Go RS, Zeldenrust S, Rajkumar SV, Dingli D, Leung N, Kyle RA, Kumar SK, Gertz MA. Overuse of organ biopsies in immunoglobulin light chain amyloidosis (AL): the consequence of failure of early recognition. Ann Med. 2017;49:545-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 61] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 58. | Suzuki T, Kusumoto S, Yamashita T, Masuda A, Kinoshita S, Yoshida T, Takami-Mori F, Takino H, Ito A, Ri M, Ishida T, Komatsu H, Ueda M, Ando Y, Inagaki H, Iida S. Labial salivary gland biopsy for diagnosing immunoglobulin light chain amyloidosis: a retrospective analysis. Ann Hematol. 2016;95:279-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 59. | Do Amaral B, Coelho T, Sousa A, Guimarães A. Usefulness of labial salivary gland biopsy in familial amyloid polyneuropathy Portuguese type. Amyloid. 2009;16:232-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 57] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 60. | Porcari A, Baggio C, Fabris E, Merlo M, Bussani R, Perkan A, Sinagra G. Endomyocardial biopsy in the clinical context: current indications and challenging scenarios. Heart Fail Rev. 2023;28:123-135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 52] [Article Influence: 17.3] [Reference Citation Analysis (1)] |
| 61. | Vergaro G, Castiglione V, Poletti R, Buda G, Pucci A, Musetti V, Genovesi D, Aimo A, Passino C, Emdin M. Biopsy Evidence of Sequential Transthyretin and Immunoglobulin Light-Chain Cardiac Amyloidosis in the Same Patient. JACC Case Rep. 2021;3:450-454. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 62. | Bermpeis K, Esposito G, Gallinoro E, Paolisso P, Bertolone DT, Fabbricatore D, Mileva N, Munhoz D, Buckley J, Wyffels E, Sonck J, Collet C, Barbato E, De Bruyne B, Bartunek J, Vanderheyden M. Safety of Right and Left Ventricular Endomyocardial Biopsy in Heart Transplantation and Cardiomyopathy Patients. JACC Heart Fail. 2022;10:963-973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 63. | Leone O, Veinot JP, Angelini A, Baandrup UT, Basso C, Berry G, Bruneval P, Burke M, Butany J, Calabrese F, d'Amati G, Edwards WD, Fallon JT, Fishbein MC, Gallagher PJ, Halushka MK, McManus B, Pucci A, Rodriguez ER, Saffitz JE, Sheppard MN, Steenbergen C, Stone JR, Tan C, Thiene G, van der Wal AC, Winters GL. 2011 consensus statement on endomyocardial biopsy from the Association for European Cardiovascular Pathology and the Society for Cardiovascular Pathology. Cardiovasc Pathol. 2012;21:245-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 466] [Cited by in RCA: 395] [Article Influence: 28.2] [Reference Citation Analysis (1)] |
| 64. | Flodrova P, Flodr P, Pika T, Vymetal J, Holub D, Dzubak P, Hajduch M, Scudla V. Cardiac amyloidosis: from clinical suspicion to morphological diagnosis. Pathology. 2018;50:261-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 65. | Vrana JA, Theis JD, Dasari S, Mereuta OM, Dispenzieri A, Zeldenrust SR, Gertz MA, Kurtin PJ, Grogg KL, Dogan A. Clinical diagnosis and typing of systemic amyloidosis in subcutaneous fat aspirates by mass spectrometry-based proteomics. Haematologica. 2014;99:1239-1247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 152] [Article Influence: 12.7] [Reference Citation Analysis (1)] |
| 66. | Libbey CA, Skinner M, Cohen AS. Use of abdominal fat tissue aspirate in the diagnosis of systemic amyloidosis. Arch Intern Med. 1983;143:1549-1552. [PubMed] |
| 67. | Rezk T, Gilbertson JA, Mangione PP, Rowczenio D, Rendell NB, Canetti D, Lachmann HJ, Wechalekar AD, Bass P, Hawkins PN, Bellotti V, Taylor GW, Gillmore JD. The complementary role of histology and proteomics for diagnosis and typing of systemic amyloidosis. J Pathol Clin Res. 2019;5:145-153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 63] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 68. | Wechalekar AD, Gillmore JD, Hawkins PN. Systemic amyloidosis. Lancet. 2016;387:2641-2654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 875] [Cited by in RCA: 708] [Article Influence: 70.8] [Reference Citation Analysis (1)] |
| 69. | Di Lisi D, Di Caccamo L, Damerino G, Portelli MC, Comparato F, Di Stefano V, Brighina F, Corrado E, Galassi AR, Novo G. Effectiveness and Safety of Oral Anticoagulants in Cardiac Amyloidosis: Lights and Shadows. Curr Probl Cardiol. 2023;48:101188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 70. | Kittleson MM, Maurer MS, Ambardekar AV, Bullock-Palmer RP, Chang PP, Eisen HJ, Nair AP, Nativi-Nicolau J, Ruberg FL; American Heart Association Heart Failure and Transplantation Committee of the Council on Clinical Cardiology. Cardiac Amyloidosis: Evolving Diagnosis and Management: A Scientific Statement From the American Heart Association. Circulation. 2020;142:e7-e22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 617] [Cited by in RCA: 518] [Article Influence: 86.3] [Reference Citation Analysis (1)] |
| 71. | Ruberg FL, Berk JL. Transthyretin (TTR) cardiac amyloidosis. Circulation. 2012;126:1286-1300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 619] [Cited by in RCA: 535] [Article Influence: 38.2] [Reference Citation Analysis (1)] |
| 72. | Yamada S, Yoshihisa A, Hijioka N, Kamioka M, Kaneshiro T, Yokokawa T, Misaka T, Ishida T, Takeishi Y. Autonomic dysfunction in cardiac amyloidosis assessed by heart rate variability and heart rate turbulence. Ann Noninvasive Electrocardiol. 2020;25:e12749. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 73. | Gertz MA, Skinner M, Connors LH, Falk RH, Cohen AS, Kyle RA. Selective binding of nifedipine to amyloid fibrils. Am J Cardiol. 1985;55:1646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 47] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 74. | Assaf A, Mekhael M, Noujaim C, Chouman N, Younes H, Kreidieh O, Marrouche N, Donnellan E. Conduction system disease in cardiac amyloidosis. Trends Cardiovasc Med. 2024;34:250-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 75. | Arbelo E, Protonotarios A, Gimeno JR, Arbustini E, Barriales-Villa R, Basso C, Bezzina CR, Biagini E, Blom NA, de Boer RA, De Winter T, Elliott PM, Flather M, Garcia-Pavia P, Haugaa KH, Ingles J, Jurcut RO, Klaassen S, Limongelli G, Loeys B, Mogensen J, Olivotto I, Pantazis A, Sharma S, Van Tintelen JP, Ware JS, Kaski JP; ESC Scientific Document Group. 2023 ESC Guidelines for the management of cardiomyopathies. Eur Heart J. 2023;44:3503-3626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2099] [Cited by in RCA: 1693] [Article Influence: 564.3] [Reference Citation Analysis (2)] |
| 76. | Mazetto Silva Vieira R, Bulhoes E, Antunes VLJ, Defante MLR, Garcez LPCF, Bastos E, Pinto MV, Guerreiro I, Farias CAG, Ximenes PN, Sousa PAMG, Oliveira VMR, Bueno BVK, Fernandes F, Darrieux FC. Safety and efficacy of catheter ablation in patients with atrial arrhythmias and cardiac amyloidosis: a systematic review and meta-analysis. Europace. 2025;27:euaf085.152. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 77. | Falk RH, Ben Zadok OI. Invited Commentary: Four Questions About Atrial Fibrillation in Cardiac Amyloidosis: Why Is This Arrhythmia Different From All Other Arrhythmias? Can J Cardiol. 2025;41:178-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 78. | Kastritis E, Leleu X, Arnulf B, Zamagni E, Cibeira MT, Kwok F, Mollee P, Hájek R, Moreau P, Jaccard A, Schönland SO, Filshie R, Nicolas-Virelizier E, Augustson B, Mateos MV, Wechalekar A, Hachulla E, Milani P, Dimopoulos MA, Fermand JP, Foli A, Gavriatopoulou M, Klersy C, Palumbo A, Sonneveld P, Johnsen HE, Merlini G, Palladini G. Bortezomib, Melphalan, and Dexamethasone for Light-Chain Amyloidosis. J Clin Oncol. 2020;38:3252-3260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 115] [Article Influence: 19.2] [Reference Citation Analysis (1)] |
| 79. | Stichting European Myeloma Network. A Study of Daratumumab Monotherapy in Previously Untreated Patients With Stage 3B Light Chain (AL) Amyloidosis. [cited 2022 Jan 31]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://ClinicalTrials.gov/show/NCT04131309 ClinicalTrials.gov Identifier: NCT04131309. |
| 80. | Salinaro F, Meier-Ewert HK, Miller EJ, Pandey S, Sanchorawala V, Berk JL, Seldin DC, Ruberg FL. Longitudinal systolic strain, cardiac function improvement, and survival following treatment of light-chain (AL) cardiac amyloidosis. Eur Heart J Cardiovasc Imaging. 2017;18:1057-1064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 63] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 81. | Martinez-Naharro A, Abdel-Gadir A, Treibel TA, Zumbo G, Knight DS, Rosmini S, Lane T, Mahmood S, Sachchithanantham S, Whelan CJ, Lachmann HJ, Wechalekar AD, Kellman P, Gillmore JD, Moon JC, Hawkins PN, Fontana M. Abstract 14407: Regression of Cardiac AL Amyloid by Cardiovascular Magnetic Resonance. Circulation. 2016;134 Suppl 1:A14407. |
| 82. | Sekijima Y, Dendle MA, Kelly JW. Orally administered diflunisal stabilizes transthyretin against dissociation required for amyloidogenesis. Amyloid. 2006;13:236-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 195] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 83. | Castaño A, Helmke S, Alvarez J, Delisle S, Maurer MS. Diflunisal for ATTR cardiac amyloidosis. Congest Heart Fail. 2012;18:315-319. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 140] [Cited by in RCA: 126] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 84. | Rosenblum H, Castano A, Alvarez J, Goldsmith J, Helmke S, Maurer MS. TTR (Transthyretin) Stabilizers Are Associated With Improved Survival in Patients With TTR Cardiac Amyloidosis. Circ Heart Fail. 2018;11:e004769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 82] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 85. | Sekijima Y, Tojo K, Morita H, Koyama J, Ikeda S. Safety and efficacy of long-term diflunisal administration in hereditary transthyretin (ATTR) amyloidosis. Amyloid. 2015;22:79-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 100] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 86. | Wixner J, Westermark P, Ihse E, Pilebro B, Lundgren HE, Anan I. The Swedish open-label diflunisal trial (DFNS01) on hereditary transthyretin amyloidosis and the impact of amyloid fibril composition. Amyloid. 2019;26:39-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 87. | Maurer MS, Schwartz JH, Gundapaneni B, Elliott PM, Merlini G, Waddington-Cruz M, Kristen AV, Grogan M, Witteles R, Damy T, Drachman BM, Shah SJ, Hanna M, Judge DP, Barsdorf AI, Huber P, Patterson TA, Riley S, Schumacher J, Stewart M, Sultan MB, Rapezzi C; ATTR-ACT Study Investigators. Tafamidis Treatment for Patients with Transthyretin Amyloid Cardiomyopathy. N Engl J Med. 2018;379:1007-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2423] [Cited by in RCA: 2080] [Article Influence: 260.0] [Reference Citation Analysis (6)] |
| 88. | Keohane D, Schwartz J, Gundapaneni B, Stewart M, Amass L. Tafamidis delays disease progression in patients with early stage transthyretin familial amyloid polyneuropathy: additional supportive analyses from the pivotal trial. Amyloid. 2017;24:30-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 52] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 89. | Maurer MS, Kale P, Fontana M, Berk JL, Grogan M, Gustafsson F, Hung RR, Gottlieb RL, Damy T, González-Duarte A, Sarswat N, Sekijima Y, Tahara N, Taylor MS, Kubanek M, Donal E, Palecek T, Tsujita K, Tang WHW, Yu WC, Obici L, Simões M, Fernandes F, Poulsen SH, Diemberger I, Perfetto F, Solomon SD, Di Carli M, Badri P, White MT, Chen J, Yureneva E, Sweetser MT, Jay PY, Garg PP, Vest J, Gillmore JD; APOLLO-B Trial Investigators. Patisiran Treatment in Patients with Transthyretin Cardiac Amyloidosis. N Engl J Med. 2023;389:1553-1565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 240] [Article Influence: 80.0] [Reference Citation Analysis (1)] |
| 90. | Fontana M, Berk JL, Gillmore JD, Witteles RM, Grogan M, Drachman B, Damy T, Garcia-Pavia P, Taubel J, Solomon SD, Sheikh FH, Tahara N, González-Costello J, Tsujita K, Morbach C, Pozsonyi Z, Petrie MC, Delgado D, Van der Meer P, Jabbour A, Bondue A, Kim D, Azevedo O, Hvitfeldt Poulsen S, Yilmaz A, Jankowska EA, Algalarrondo V, Slugg A, Garg PP, Boyle KL, Yureneva E, Silliman N, Yang L, Chen J, Eraly SA, Vest J, Maurer MS; HELIOS-B Trial Investigators. Vutrisiran in Patients with Transthyretin Amyloidosis with Cardiomyopathy. N Engl J Med. 2025;392:33-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 263] [Article Influence: 263.0] [Reference Citation Analysis (1)] |
| 91. | Cardoso I, Martins D, Ribeiro T, Merlini G, Saraiva MJ. Synergy of combined doxycycline/TUDCA treatment in lowering Transthyretin deposition and associated biomarkers: studies in FAP mouse models. J Transl Med. 2010;8:74. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 147] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 92. | Obici L, Cortese A, Lozza A, Lucchetti J, Gobbi M, Palladini G, Perlini S, Saraiva MJ, Merlini G. Doxycycline plus tauroursodeoxycholic acid for transthyretin amyloidosis: a phase II study. Amyloid. 2012;19 Suppl 1:34-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 167] [Article Influence: 11.9] [Reference Citation Analysis (1)] |
| 93. | Wixner J, Pilebro B, Lundgren HE, Olsson M, Anan I. Effect of doxycycline and ursodeoxycholic acid on transthyretin amyloidosis. Amyloid. 2017;24:78-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 94. | Karlstedt E, Jimenez-Zepeda V, Howlett JG, White JA, Fine NM. Clinical Experience With the Use of Doxycycline and Ursodeoxycholic Acid for the Treatment of Transthyretin Cardiac Amyloidosis. J Card Fail. 2019;25:147-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (1)] |
| 95. | Fondazione IRCCS Policlinico San Matteo di Pavia. A Study of Doxycycline and Tauroursodeoxycholic Acid (Doxy/TUDCA) Plus Standard Supportive Therapy Versus Standard Supportive Therapy Alone in Cardiac Amyloidosis Caused by Transthyretin. [cited 7 November 2025]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://ClinicalTrials.gov/show/NCT03481972 ClinicalTrials.gov Identifier: NCT03481972. |
| 96. | Neurimmune AG. First-in-Human Study of NI006 in Patients With Amyloid Transthyretin Cardiomyopathy. [cited 7 November 2025]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://ClinicalTrials.gov/show/NCT04360434 ClinicalTrials.gov Identifier: NCT04360434. |
| 97. | Çavuşoğlu Y, Başarıcı İ, Tüfekçioğlu O, Özpelit E, Özdemir E, Sivrikoz İA, Altay H, Değertekin M, Dinçer İ, İkitimur B, Kahveci G, Bozkurt MF, Erkılıç M, Kaya GÇ, Beksaç M, Salihoğlu A, Tokgözoğlu L. Current barriers and recommendations on the diagnosis of transthyretin amyloid cardiomyopathy: a Delphi study. Front Cardiovasc Med. 2024;11:1299261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (1)] |