Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.112061
Revised: August 6, 2025
Accepted: January 5, 2026
Published online: March 20, 2026
Processing time: 241 Days and 22.5 Hours
Febrile seizures (FS) are common pediatric neurological disorders typically triggered by fever, affecting children aged 6 months to 5 years. Although usually benign, early and accurate diagnosis is crucial for effective mana
To investigate the diagnostic potential of interleukin-1 beta (IL-1β) and micro
Pediatric patients were grouped into FS (n = 68), HS (n = 52), FO (n = 52), and SO (n = 32). Blood samples were collected within 30 minutes of seizure onset or fever presentation. Plasma samples were analyzed for IL-1β (enzyme-linked immuno
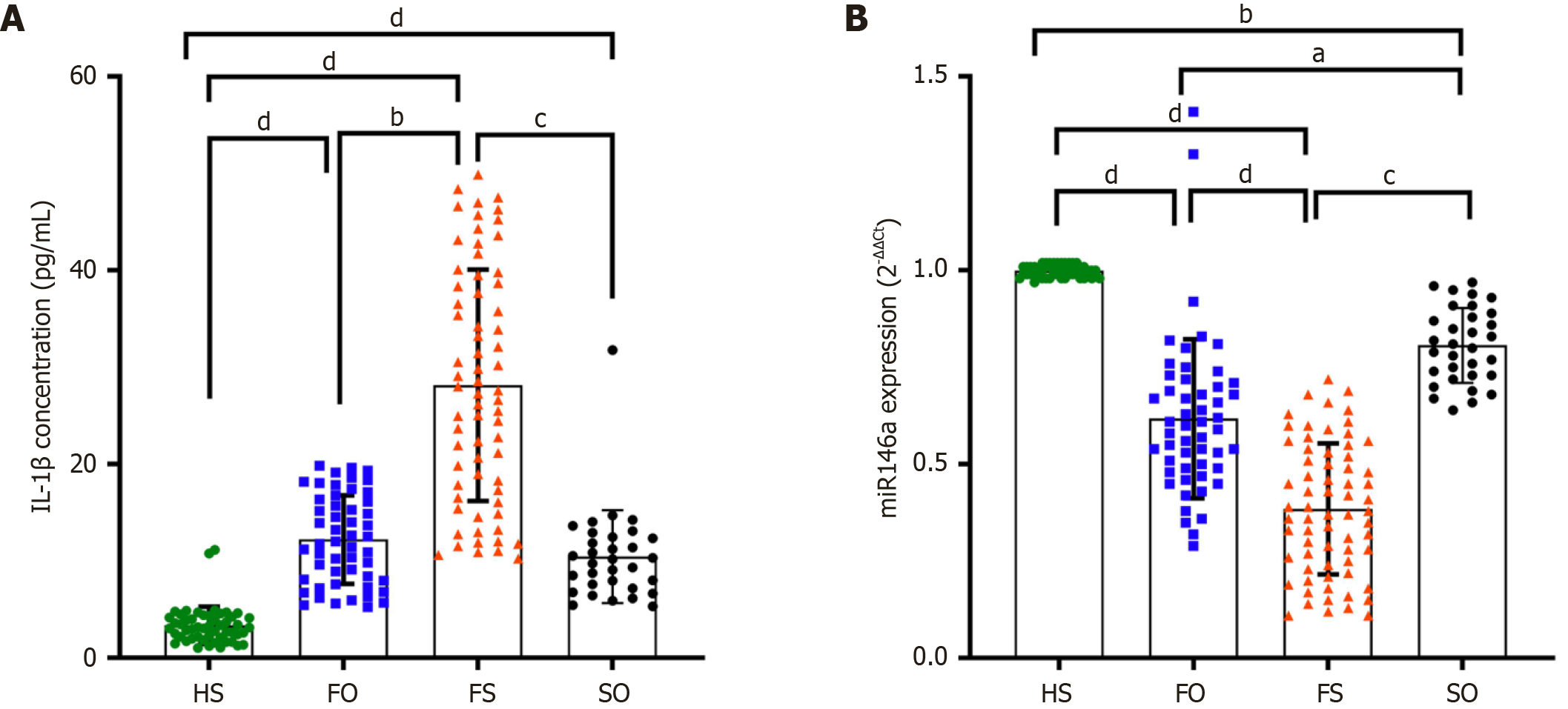
IL-1β levels showed the highest level in FS, followed by FO and SO, with HS showing the lowest levels. IL-1β distinguished FS from HS with an area under the curve (AUC) of 0.9983 (sensitivity: 100%, specificity: 96.15%). miR-146a was lowest in FS and highest in HS, achieving an AUC of 1.000 (sensitivity: 100%, specificity: 98.08%). IL-1β and miR-146a also effectively differentiated FS from FO and SO, with respective AUCs of 0.8862 and 0.8173 for FS vs FO.
IL-1β and miR-146a could be promising diagnostic biomarkers for FS, distinguishing FS from HS, FO, and SO with high sensitivity and specificity. These markers possess the potential to enhance early FS diagnosis.
Core Tip: This study assessed the diagnostic potential of interleukin-1 beta and microRNA-146a in distinguishing febrile seizures (FS) from healthy subjects, fever-only, and seizure-only cases. Interleukin-1 beta levels were significantly highest in the FS group, while microRNA-146a levels were lowest in FS and highest in healthy subjects. Both markers demonstrated high diagnostic accuracy in differentiating FS from the other groups, suggesting they are promising diagnostic biomarkers for FS and support the role of neuroinflammation in FS development.
- Citation: Mohammadi S, Abbood Hameed Ati M, Seyedhosseini FS, Zare Ebrahimabad M, Saeidi M, Hosseini SA, Darweesh M, Al-Harrasi A, Yazdani Y. Diagnostic utility of circulating interleukin-1 beta and microRNA-146a in differentiating febrile seizures from other pediatric conditions. World J Exp Med 2026; 16(1): 112061
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/112061.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.112061
Febrile seizures (FS) represent a common pediatric neurological condition, affecting 2%-5% of children aged six months to five years[1,2]. These seizures are triggered during episodes of fever that are not a result of intracranial infection, electrolyte imbalance, or other systemic disorders[3]. FS can be classified into two types: Simple FS, which are brief, generalized, and occur once during a febrile episode, and complex FS, which are prolonged, recurrent, or focal in nature[4]. Despite their generally benign prognosis, FS pose many diagnostic challenges, as they can mimic other febrile illnesses or seizure-related conditions[5]. Accurate diagnosis is critical to avoid unnecessary diagnostic procedures, alleviate parental anxiety, and guide proper management[6].
The pathophysiology of FS remains incompletely understood but is believed to involve genetic predisposition, immune dysregulation, and febrile-induced neuronal excitability[7]. Among the key players in FS pathogenesis are cytokines, immune signaling molecules that mediate inflammation and fever[8]. Interleukin-1 beta (IL-1β), a pro-inflammatory cytokine, has been previously studied for its role in febrile responses and neuronal excitability[9]. Elevated IL-1β levels during febrile episodes have been demonstrated in blood-brain barrier disruption and hyper-excitability of neuronal circuits, increasing seizure susceptibility[10]. Studies have consistently demonstrated higher IL-1β levels in children with FS compared to febrile controls, implying its potential as a biomarker for FS diagnosis[11]. Mahyar et al[12] highlighted elevated serum IL-1β levels in children with FS, suggesting its direct contribution to seizure generation. Similarly, Soltani et al[13] linked IL-1β gene polymorphisms to increased FS susceptibility, further supporting the role of IL-1β in FS pathophysiology.
MicroRNAs (miRNAs), a class of small non-coding RNA molecules that post-transcriptionally regulate gene expression. It has also emerged as a potential biomarker and therapeutic target in FS[14,15]. MiR-146a, a key regulator of inflammatory pathways, has attracted particular interest due to its ability to suppress IL-1β signaling via targeting interleukin-1 receptor-associated kinase 1 and tumor necrosis factor receptor-associated factor 6, components of the toll-like receptor pathway[16]. Dysregulated miR-146a expression has been linked to several inflammatory and neurological conditions, including epilepsy and FS[17]. Omran et al[18] demonstrated stage-specific alterations in miR-146a expression in children and animal models with epilepsy, with miR-146a levels being inversely correlated with IL-1β expression during acute stages. Similarly, Çarman et al[19] reported elevated miR-146a levels in FS patients compared to controls, further suggesting miR-146a in the inflammatory cascade of FS.
Despite these advancements, the diagnostic utility of IL-1β and miR-146a in FS remains less studied. Previous studies have primarily focused on isolated analyses of either IL-1β or miR-146a, often with small sample sizes and limited comparative groups. While Kwon et al[8] and Omran et al[18] have separately highlighted the significance of IL-1β and miR-146a in febrile and seizure conditions, no study has systematically evaluated their diagnostic value across multiple control groups. Furthermore, the interaction between these biomarkers, particularly their mechanistic relationship in modulating inflammatory responses during FS, has not been studied. The aim of this study was to address these gaps by evaluating the plasma levels of IL-1β and miR-146a in children with FS and comparing them to three distinct control groups: Fever-only (FO), seizure-only (SO), and healthy subjects (HS). We also aimed to determine the diagnostic accuracy and potential utility of these biomarkers in distinguishing FS from other pediatric conditions.
This study was conducted at Taleghani Medical Center, Golestan University of Medical Sciences, Iran. Ethical approval was granted by the Ethics Committee of Golestan University of Medical Sciences. Written informed consent was obtained from all participants prior to enrollment. The study enrolled 204 children aged six months to six years, categorized into four groups: FS, FO, SO, and healthy control (HS). The FS group included 68 children presenting with seizures associated with fever (≥ 38 °C) and no evidence of central nervous system infection or neurological disorders. The FO group comprised 52 children with fever but no seizures or history of FS. The SO group included 32 afebrile children with seizures unrelated to fever. The FS and SO groups included both patients with their first episode and those with a history of recurrent seizures. The HS group was composed of children who presented to the medical center for routine health checkups or non-systemic health issues. A comprehensive medical history was taken to ensure they had no prior history of fever, seizures, or chronic systemic illnesses, including immune system disorders. Inclusion criteria for all groups required confirmation by a specialist. Children with chronic illnesses, epilepsy, neurodevelopmental disorders, or recent use of immunosuppressive drugs were excluded from the study. Patients who were later diagnosed with central nervous system infections by lumbar puncture examination or other exclusion criteria were not included in the final data analysis.
Blood samples (2 mL) were collected via venipuncture within 30 minutes of seizure onset or fever presentation. To ensure timely sample collection, a dedicated study team was on standby in the emergency department. Upon presentation of a child with an acute seizure or fever, the clinical team immediately alerted the study personnel, who obtained informed consent and collected the blood sample within the 30-minute window. The samples were placed in ethylene diamine tetraacetic acid-coated collection tubes (Pole Ideal Pars, Iran) and transported on ice to the central laboratory. Plasma was separated by centrifugation at 3500 rpm for 10 minutes at 4 °C. Plasma aliquots were stored at -80 °C until further analysis.
Plasma IL-1β levels were measured using an enzyme-linked immunosorbent assay kit (ZellBio GmbH, Germany; Cat. No. RK00058-96) following the manufacturer’s protocol. Standards were reconstituted and serially diluted to prepare a standard curve ranging from 12.5 pg/mL to 800 pg/mL. Plasma samples (50 μL) were added to the pre-coated 96-well microplate along with standards and incubated for two hours at room temperature. After washing with the provided wash buffer, biotin-conjugated anti-IL-1β antibody diluted at 1:100 was added and incubated for one hour. Subsequently, streptavidin-horseradish peroxidase conjugate diluted at 1:100 was added and incubated for 30 minutes, followed by the addition of the TMB substrate solution. After 10 minutes, the reaction was stopped with the stop solution, and absorbance was measured at 450 nm using a StatFax 2200 microplate reader (Awareness Technology, United States). IL-1β concentrations in plasma samples were calculated using a standard curve generated by linear regression in Microsoft Excel, as previously described[20].
The expression levels of miR-146a in plasma were quantified using real-time reverse transcription polymerase chain reaction (qRT-PCR). Total RNA, including miRNAs, was extracted from 500 μL of plasma using the SinaPure ONE RNA Isolation Kit (Sinaclon, Iran; Cat. No. EX6051), with modifications[21]. RNA extraction was performed according to the manufacturer’s instructions. The concentration and purity of RNA were determined using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, United States).
Complementary DNA (cDNA) synthesis was carried out using the SinaClon First Strand cDNA Synthesis Kit (Sinaclon, Iran; Cat. No. RT5201). For miR-146a, a stem-loop primer was used during reverse transcription. Each reaction mixture contained 1 μg of total RNA, 4 μL of 10 × buffer, 1 μL of dNTP mix, 1 μL of stem-loop primer for miR-146a (5′-CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGACCTGTT-3′), 1 μL of U6-specific primer (5′-AACGCTTCACGAATTT GCGT-3′), 1 μL of reverse transcriptase, and nuclease-free water to a final volume of 20 μL. The reverse transcription reaction was performed at 42 °C for 60 minutes, followed by enzyme inactivation at 85 °C for 5 minutes.
qRT-PCR was performed using the SinaSYBR Green HS-qPCR Mix (Sinaclon, Iran; Cat. No. MM2171) on a Step One Plus qPCR system (ABI, United States). Each 20 μL reaction contained 10 μL of 2 × SYBR Green mix, 1 μL of forward primer for miR-146a (5′-TGAGAACTGAATTCCATGGGT-3′), 1 μL of reverse primer (5′-CGACGTGAGTAAAGTAGCCT-3′), 1 μL of cDNA, and 7 μL of nuclease-free water. For U6, the forward primer (5′-GCTTCGGCAGCACATATACTAAA AT-3′) and reverse primer (5′-CGCTTCACGAATTTGCGTGTCAT-3′) were used. The thermal cycling conditions were as follows: Initial denaturation at 95 °C for 5 minutes, followed by 40 cycles of denaturation at 95 °C for 10 seconds, annealing at 60 °C for 15 seconds, and extension at 72 °C for 20 seconds. The relative expression of miR-146a was calculated using the ΔΔCt method, normalized to U6 RNA levels. All qRT-PCR reactions were conducted in triplicate to ensure accuracy.
Statistical analysis was conducted using SPSS version 26.0 (IBM, United States). The normality of the data distribution was assessed using the Shapiro-Wilk test. Data that were not normally distributed were expressed as median (in
The demographic, clinical, and laboratory characteristics of the study participants, including the age distribution, gender distribution, family history of seizures, fever temperature, and hematological parameters, including white blood cell count, red blood cell count, hemoglobin, hematocrit, platelet count, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, and erythrocyte sedimentation rate are summarized in Table 1.
| Groups | HS | FO | FS | SO | P value |
| Age (Months) | 39.23 ± 26.713 | 23.98 ± 17.006 | 21.75 ± 11.962 | 38.03 ± 23.345 | < 0.001 |
| Sex (M/F) | 21/31 | 30/22 | 30/38 | 21/11 | > 0.05 |
| Family history of seizures | - | 3.8% | 26.5% | 0% | < 0.001 |
| Fever temperature (°C) | - | 38.63 ± 0.562 | 38.69 ± 0.817 | - | > 0.05 |
| WBC count | 9796.15 ± 2783.56 | 10750.00 ± 4320.79 | 10275.00 ± 6054.19 | 9662.50 ± 1255.89 | > 0.05 |
| RBC count | 4.5537 ± 0.50459 | 4.4108 ± 0.44087 | 4.5354 ± 0.51568 | 4.6834 ± 0.45657 | > 0.05 |
| Hb (g/dL) | 11.3288 ± 0.91058 | 10.8327 ± 1.37209 | 10.7221 ± 1.53466 | 11.3469 ± 1.54293 | > 0.05 |
| Hct (%) | 33.4404 ± 4.90035 | 32.6788 ± 3.42925 | 32.4603 ± 3.18251 | 33.2844 ± 3.74649 | > 0.05 |
| Platelet count | 318761.54 ± 90056.32 | 316192.31 ± 101805.67 | 308882.35 ± 107568.31 | 336593.75 ± 149717.12 | > 0.05 |
| MCH (pg) | 25.0740 ± 2.82654 | 27.7602 ± 3.2323149 | 26.1144 ± 9.13959 | 24.6069 ± 4.75111 | < 0.01 |
| ESR (mm/hour) | 12.3269 ± 6.07703 | 22.2885 ± 15.78202 | 14.4706 ± 9.12021 | 6.0313 ± 1.44768 | < 0.01 |
In our analysis of IL-1β expression, significant differences were observed across the four study groups. FS patients exhibited the highest mean IL-1β expression, with a value of 28.16 ± 11.95. This was markedly elevated compared to FO individuals (P < 0.01), who had a mean IL-1β expression of 12.24 ± 4.57, and SO subjects (P < 0.001), who showed a mean of 10.48 ± 4.79. The HS group displayed the lowest mean IL-1β expression at 3.38 ± 1.92, compared to the FS group (P < 0.0001). Moreover, both FO and SO groups showed significantly higher IL-1β levels compared to the HS group (P < 0.001) (Figure 1A). In our evaluation of plasma miR-146a expression across the study groups, the HS group displayed the highest mean miR-146a expression at 0.9992 ± 0.01467, while the FS patients showed the lowest levels with a mean expression of 0.3853 ± 0.16847. Intermediate levels were observed in the FO group, with a mean expression of 0.6181 ± 0.20504, and the SO group, which had a mean expression of 0.8072 ± 0.09646, as shown in Figure 1B. The Independent-Samples Kruskal-Wallis test was employed and demonstrated significant differences in miR-146a expression among the groups (P < 0.0001). Post-hoc pairwise comparisons using the Dunn-Bonferroni test further confirmed that miR-146a expression in FS patients was significantly lower than in all other groups (P < 0.0001), and HS subjects exhibited significantly higher miR-146a levels compared to the FO (P < 0.0001) and SO groups (P < 0.001).
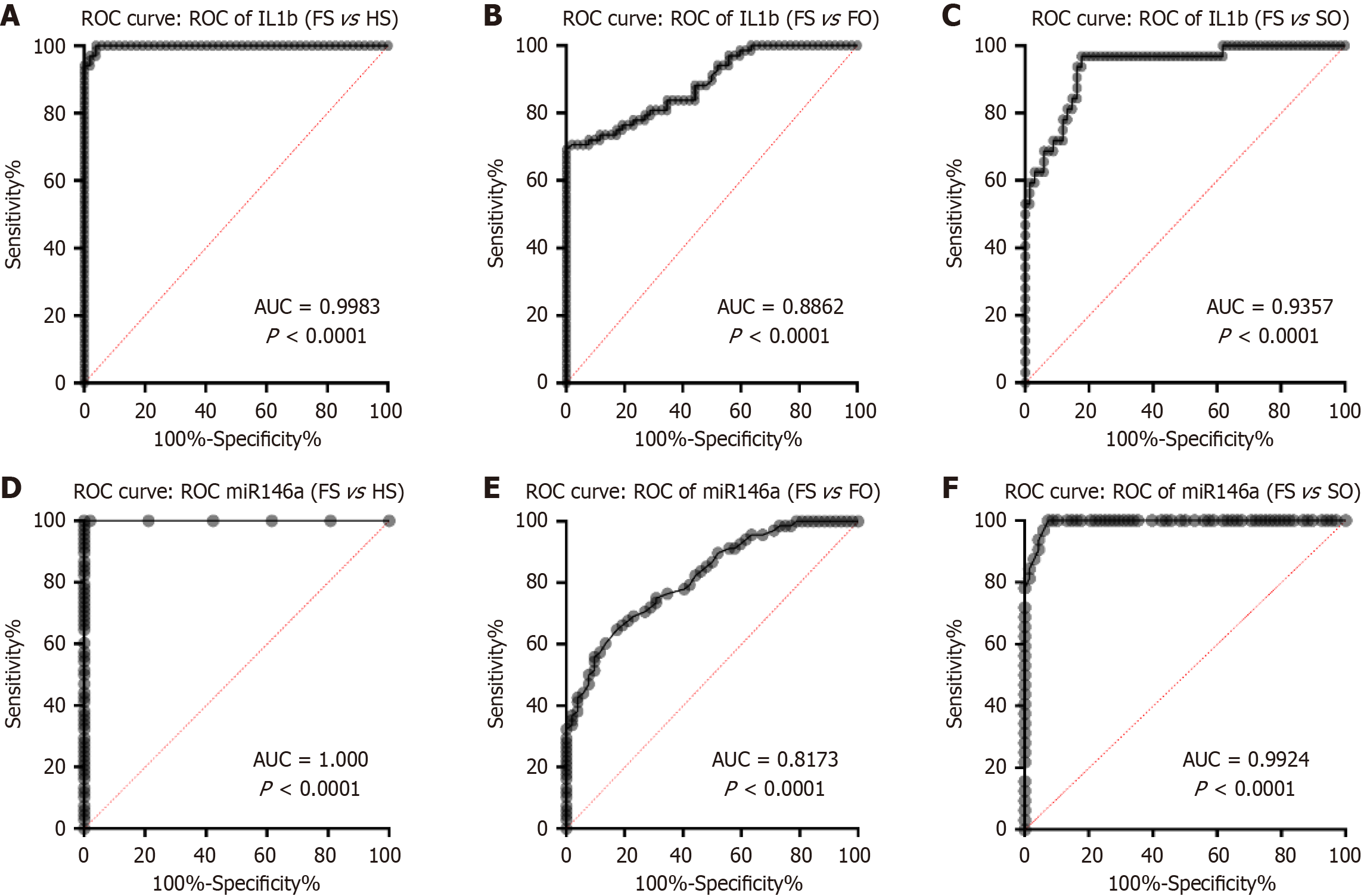
The diagnostic potential of IL-1β in differentiating FS patients from other groups, including HS, FO, and SO subjects, was evaluated using ROC curve analysis. Distinguishing FS patients from HS, the AUC was 0.9983, with a 95% confidence interval (CI): 0.9951-1.000 (P < 0.0001). A cutoff value of 7.595 pg/mL provided a sensitivity of 100.0% (95%CI: 94.65%-100.0%) and specificity of 96.15% (95%CI: 87.02%-99.32%). The likelihood ratio (LR) of 26.00 demonstrated a significant increase in the odds of a positive result when FS was present (Figure 2A). In the FS vs FO comparison, the AUC was 0.8862, with a 95%CI: 0.8298-0.9425 (P < 0.0001). A cutoff value of 19.75 pg/mL achieved a sensitivity of 70.59% (95%CI: 58.89%-80.08%) and a specificity of 98.08% (95%CI: 89.88%-99.90%). The LR was calculated at 36.71, emphasizing the significant diagnostic potential of IL-1β in distinguishing FS cases from FO individuals (Figure 2B). For the FS vs SO comparison, the AUC was 0.9357, with a 95%CI: 0.8862-0.9851 (P < 0.0001). A cutoff value of 14.82 pg/mL provided a sensitivity of 96.88% (95%CI: 84.26%-99.84%) and specificity of 82.35% (95%CI: 71.64%-89.61%). The LR of 5.49 further confirmed IL-1β’s effectiveness in distinguishing FS patients from those with SO conditions (Figure 2C).
The diagnostic potential of microRNA-146a (miR-146a) in differentiating FS patients from HS, FO, and SO subjects was evaluated using ROC curve analysis. In distinguishing FS patients from HS, the AUC was 1.000 (95%CI: 1.000-1.000, P < 0.0001). By setting a cutoff point at a fold change of 0.9750, miR-146a achieved a sensitivity of 100.00% (95%CI: 94.65%-100.0%) and a specificity of 98.08% (95%CI: 89.88%-99.90%). The LR was calculated at 52.00, signifying a substantial increase in the odds of a positive result when FS was present (Figure 2D). In distinguishing FS patients from FO individuals, miR-146a exhibited moderate discriminatory ability, with an AUC of 0.8173 (95%CI: 0.7442-0.8904, P < 0.0001). A cutoff point of 0.5050 provided a sensitivity of 72.06% (95%CI: 60.44%-81.32%) and a specificity of 71.15% (95%CI: 57.73%-81.67%). The LR was calculated at 2.498, reflecting a modest improvement in diagnostic accuracy for FS cases compared to FO cases. While miR-146a displayed moderate performance in this context, the results highlight its potential as a supplementary diagnostic marker for differentiating these two groups (Figure 2E). Moreover, miR-146a demonstrated outstanding discriminatory power in distinguishing FS patients from SO individuals, with an AUC of 0.9924 (95%CI: 0.9821-1.000, P < 0.0001). Using a cutoff point of 0.6650, the sensitivity was calculated at 93.75% (95%CI: 79.85%-98.89%), while the specificity reached 95.59% (95%CI: 87.81%-98.80%). The LR of 21.25 indicated a strong enhancement in diagnostic accuracy for FS cases compared to SO cases (Figure 2F).
In this study, we evaluated the diagnostic potential of IL-1β and miR-146a in distinguishing FS (FS) patients from other conditions, including HS, FO individuals, and SO patients. Our findings demonstrated the remarkable diagnostic performance of both biomarkers, delineating their potential clinical utility in the accurate and timely identification of FS cases. Our results revealed that IL-1β expression was significantly elevated in FS patients compared to all other groups, supporting its role as a potential diagnostic biomarker for FS. The diagnostic performance of IL-1β in distinguishing FS from HS was exceptional, with a sensitivity of 100% and a specificity of 96.15%. The high AUC of 0.9983 indicates near-perfect discriminatory power, consistent with prior research emphasizing the critical role of inflammatory cytokines in seizure susceptibility[8,10].
We also observed that miR-146a expression was significantly lower in FS patients compared to other groups. This reduction was most observed when distinguishing FS from HS, where miR-146a exhibited perfect discriminatory performance with an AUC of 1.000, a sensitivity of 100%, and a specificity of 98.08%. These findings highlight miR-146a as a reliable diagnostic biomarker for FS. The observed exceptionally high sensitivity (100%) and AUC (1.000) for miR-146a in distinguishing FS from HS needs careful interpretation. While these results highlight the strong discriminatory power of this biomarker within our study cohort, such high values in clinical research are rare and may reflect the stark difference between the inflammatory state of FS patients and the healthy state of our control group. We suggest that these findings require validation in larger, prospective cohorts to confirm their generalizability and clinical utility.
When comparing FS to FO and SO cases, both IL-1β and miR-146a demonstrated varying levels of diagnostic per
The reduced expression of miR-146a in FS patients further supports its role in modulating inflammation. MiR-146a is a critical regulator of the nuclear factor-κB (NF-κB) signaling pathway, which governs the expression of pro-inflammatory cytokines such as IL-1β[26]. Its decreased levels may lead to unchecked NF-κB activation, exacerbating inflammatory cascades and increasing seizure susceptibility[27]. These findings are consistent with previous studies, including those by Çarman et al[19] and Perry et al[28], which highlight the regulatory role of miR-146a in inflammation and its potential contribution to FS pathogenesis. Our study confirms and extends previous research on the roles of IL-1β and miR-146a in FS. While prior studies have reported elevated IL-1β levels in FS patients[8,10,23], we observed one of the highest reported diagnostic accuracies for this biomarker, particularly in distinguishing FS from HS. This finding may reflect the specific neuroinflammatory characteristics of FS, which differ from other seizure-related conditions.
Similarly, our findings on miR-146a align with prior research that highlighted its role in regulating inflammatory responses[16,18]. The exceptional diagnostic performance of miR-146a, particularly in distinguishing FS from HS and SO groups, emphasizes its potential as a biomarker for FS diagnosis. The elevated IL-1β expression in FS patients observed in our study may result from fever-induced activation of immune cells, including microglia, which release pro-inflammatory cytokines in response to pyrogenic stimuli[29]. This inflammatory cascade disrupts neuronal homeostasis and promotes excitability, contributing to seizure onset.
The reduced miR-146a expression observed in FS patients likely reflects a failure to suppress inflammation adequately[30-32]. MiR-146a’s role in regulating NF-κB activity is well-documented, and its dysregulation may lead to hypera
We acknowledge several limitations in our study. The cross-sectional design precludes an assessment of temporal changes in biomarker expression, and the relatively small sample size may limit the generalizability of our findings. Larger, longitudinal studies are needed to validate our results and explore the dynamic nature of IL-1β and miR-146a expression in FS. Moreover, while we focused on two biomarkers, the inclusion of other inflammatory and neuroimmune markers could provide a more comprehensive understanding of FS pathogenesis. We did not measure C-reactive protein (CRP) or high-sensitivity CRP, which are other common markers of inflammation. A future study that correlates IL-1β and miR-146a levels with CRP could provide a more comprehensive picture of the inflammatory response in FS. While our study focused on the diagnostic performance of the biomarkers, a comparison of their utility with the clinical judgment of experienced pediatricians was not performed. This is an important area for future research to provide a more comprehensive understanding of the clinical value of these markers. Moreover, the duration of fever prior to seizure onset was not uniformly recorded for all FS patients. Although samples were collected within 30 minutes of either seizure onset or fever presentation, the time elapsed between the initial febrile episode and sample collection could be a factor influencing biomarker expression. Future studies are needed to explore how the duration of fever and the timing of sample collection might affect the levels of these biomarkers.
Our study provides compelling evidence for the diagnostic potential of IL-1β and miR-146a in FS. Both biomarkers demonstrated exceptional performance in distinguishing FS from HS, and their ability to differentiate FS from FO and SO cases further highlights their clinical relevance. These findings not only enhance our understanding of the patho
We would like to thank all the participants and their families for their cooperation in this study. We also thank the staff at Golestan University of Medical Sciences for their technical assistance. Special thanks to the Research Central Laboratory, Golestan University of Medical Sciences, for providing the necessary facilities and resources.
| 1. | Leung AK, Hon KL, Leung TN. Febrile seizures: an overview. Drugs Context. 2018;7:212536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 112] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 2. | Hesdorffer DC, Benn EK, Bagiella E, Nordli D, Pellock J, Hinton V, Shinnar S; FEBSTAT Study Team. Distribution of febrile seizure duration and associations with development. Ann Neurol. 2011;70:93-100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 73] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 3. | Auvin S, Vallée L. [Febrile seizures: current understanding of pathophysiological mechanisms]. Arch Pediatr. 2009;16:450-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 4. | Dubé CM, Brewster AL, Baram TZ. Febrile seizures: mechanisms and relationship to epilepsy. Brain Dev. 2009;31:366-371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 133] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 5. | Patel AD, Vidaurre J. Complex febrile seizures: a practical guide to evaluation and treatment. J Child Neurol. 2013;28:762-767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 6. | McClelland S, Dubé CM, Yang J, Baram TZ. Epileptogenesis after prolonged febrile seizures: mechanisms, biomarkers and therapeutic opportunities. Neurosci Lett. 2011;497:155-162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 53] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 7. | Kjeldsen MJ, Kyvik KO, Friis ML, Christensen K. Genetic and environmental factors in febrile seizures: a Danish population-based twin study. Epilepsy Res. 2002;51:167-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 56] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 8. | Kwon A, Kwak BO, Kim K, Ha J, Kim SJ, Bae SH, Son JS, Kim SN, Lee R. Cytokine levels in febrile seizure patients: A systematic review and meta-analysis. Seizure. 2018;59:5-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 9. | Yu X, Zhang N, Liu S, Xi Z, Zhang Y. Polymorphisms in the interleukin-1β (IL-1B) and interleukin-1α (IL-1A) genes on risk of febrile seizures: a meta-analysis. Neurol Sci. 2018;39:1529-1536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 10. | Yu HM, Liu WH, He XH, Peng BW. IL-1β: an important cytokine associated with febrile seizures? Neurosci Bull. 2012;28:301-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 11. | Virta M, Hurme M, Helminen M. Increased plasma levels of pro- and anti-inflammatory cytokines in patients with febrile seizures. Epilepsia. 2002;43:920-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 134] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 12. | Mahyar A, Ayazi P, Orangpour R, Daneshi-Kohan MM, Sarokhani MR, Javadi A, Habibi M, Talebi-Bakhshayesh M. Serum interleukin-1beta and tumor necrosis factor-alpha in febrile seizures: is there a link? Korean J Pediatr. 2014;57:440-444. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 13. | Soltani S, Zare-Shahabadi A, Shahrokhi A, Rezaei A, Zoghi S, Zamani GR, Mohammadi M, Ashrafi MR, Rezaei N. Association of Interleukin-1 Gene Cluster and Interleukin-1 Receptor Polymorphisms With Febrile Seizures. J Child Neurol. 2016;31:673-677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 14. | El Abed F, Baraket G, Nyamari MN, Naitore C, Awe OI. Differential Expression Analysis of miRNAs and mRNAs in Epilepsy Uncovers Potential Biomarkers. 2023 Preprint. Available from: bioRxiv: 2023.09.11.557132. [DOI] [Full Text] |
| 15. | Hammond SM. An overview of microRNAs. Adv Drug Deliv Rev. 2015;87:3-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1126] [Cited by in RCA: 1092] [Article Influence: 99.3] [Reference Citation Analysis (1)] |
| 16. | Saba R, Sorensen DL, Booth SA. MicroRNA-146a: A Dominant, Negative Regulator of the Innate Immune Response. Front Immunol. 2014;5:578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 335] [Cited by in RCA: 325] [Article Influence: 27.1] [Reference Citation Analysis (1)] |
| 17. | Wang J, Zhao J. MicroRNA Dysregulation in Epilepsy: From Pathogenetic Involvement to Diagnostic Biomarker and Therapeutic Agent Development. Front Mol Neurosci. 2021;14:650372. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 18. | Omran A, Peng J, Zhang C, Xiang QL, Xue J, Gan N, Kong H, Yin F. Interleukin-1β and microRNA-146a in an immature rat model and children with mesial temporal lobe epilepsy. Epilepsia. 2012;53:1215-1224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 116] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 19. | Çarman KB, Karal Y, Gül Mert G, Ekici A, Perk P, Arslantaş D, Yarar C, Dinleyici EÇ. Expression of MicroRNA 146a, 155, 181 and 223 in febrile seizure. Turk J Pediatr. 2021;63:594-601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 20. | Mohammadi S, Seyedhoseini FS, Asadi J, Yazdani Y. Effects of berberine on the secretion of cytokines and expression of genes involved in cell cycle regulation in THP-1 monocytic cell line. Iran J Basic Med Sci. 2017;20:530-537. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 21. | Alsbihawi TQ, Zare Ebrahimabad M, Seyedhosseini FS, Davoodi H, Abdolahi N, Nazari A, Mohammadi S, Yazdani Y. Altered expression of miR-125a and dysregulated cytokines in systemic lupus erythematosus: Unveiling diagnostic and prognostic markers. World J Exp Med. 2023;13:102-114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 22. | Tütüncüoğlu S, Kütükçüler N, Kepe L, Coker C, Berdeli A, Tekgül H. Proinflammatory cytokines, prostaglandins and zinc in febrile convulsions. Pediatr Int. 2001;43:235-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 58] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 23. | Ichiyama T, Nishikawa M, Yoshitomi T, Hayashi T, Furukawa S. Tumor necrosis factor-alpha, interleukin-1 beta, and interleukin-6 in cerebrospinal fluid from children with prolonged febrile seizures. Comparison with acute encephalitis/encephalopathy. Neurology. 1998;50:407-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 134] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 24. | Nemeth DP, Quan N. Modulation of Neural Networks by Interleukin-1. Brain Plast. 2021;7:17-32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 25. | Lopalco G, Cantarini L, Vitale A, Iannone F, Anelli MG, Andreozzi L, Lapadula G, Galeazzi M, Rigante D. Interleukin-1 as a common denominator from autoinflammatory to autoimmune disorders: premises, perils, and perspectives. Mediators Inflamm. 2015;2015:194864. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 75] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 26. | Rusca N, Monticelli S. MiR-146a in Immunity and Disease. Mol Biol Int. 2011;2011:437301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 176] [Article Influence: 11.7] [Reference Citation Analysis (1)] |
| 27. | Taganov KD, Boldin MP, Chang KJ, Baltimore D. NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc Natl Acad Sci U S A. 2006;103:12481-12486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3685] [Cited by in RCA: 3562] [Article Influence: 178.1] [Reference Citation Analysis (1)] |
| 28. | Perry MM, Moschos SA, Williams AE, Shepherd NJ, Larner-Svensson HM, Lindsay MA. Rapid changes in microRNA-146a expression negatively regulate the IL-1beta-induced inflammatory response in human lung alveolar epithelial cells. J Immunol. 2008;180:5689-5698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 368] [Cited by in RCA: 369] [Article Influence: 20.5] [Reference Citation Analysis (1)] |
| 29. | Monif M, Reid CA, Powell KL, Drummond KJ, O'Brien TJ, Williams DA. Interleukin-1β has trophic effects in microglia and its release is mediated by P2X7R pore. J Neuroinflammation. 2016;13:173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 95] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 30. | Fan W, Liang C, Ou M, Zou T, Sun F, Zhou H, Cui L. MicroRNA-146a Is a Wide-Reaching Neuroinflammatory Regulator and Potential Treatment Target in Neurological Diseases. Front Mol Neurosci. 2020;13:90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 83] [Article Influence: 13.8] [Reference Citation Analysis (1)] |
| 31. | Zhou C, Zhao L, Wang K, Qi Q, Wang M, Yang L, Sun P, Mu H. MicroRNA-146a inhibits NF-κB activation and pro-inflammatory cytokine production by regulating IRAK1 expression in THP-1 cells. Exp Ther Med. 2019;18:3078-3084. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 32. | Brennan GP, Henshall DC. microRNAs in the pathophysiology of epilepsy. Neurosci Lett. 2018;667:47-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 54] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 33. | Aurelius E, Andersson B, Forsgren M, Sköldenberg B, Strannegård O. Cytokines and other markers of intrathecal immune response in patients with herpes simplex encephalitis. J Infect Dis. 1994;170:678-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 77] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 34. | Vezzani A, Balosso S, Ravizza T. Neuroinflammatory pathways as treatment targets and biomarkers in epilepsy. Nat Rev Neurol. 2019;15:459-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 641] [Article Influence: 91.6] [Reference Citation Analysis (1)] |